Tracing IgE-Producing Cells in Allergic Patients
Abstract
1. Introduction
2. IgE Production
2.1. Murine Models to Investigate IgE-Production and Allergic Disease
2.2. Human IgE Production
3. Tracing IgE Producing Cells
3.1. Identification of IgE Producing Cells by IgE Staining Techniques
3.1.1. ELISpot
3.1.2. Flow Cytometry
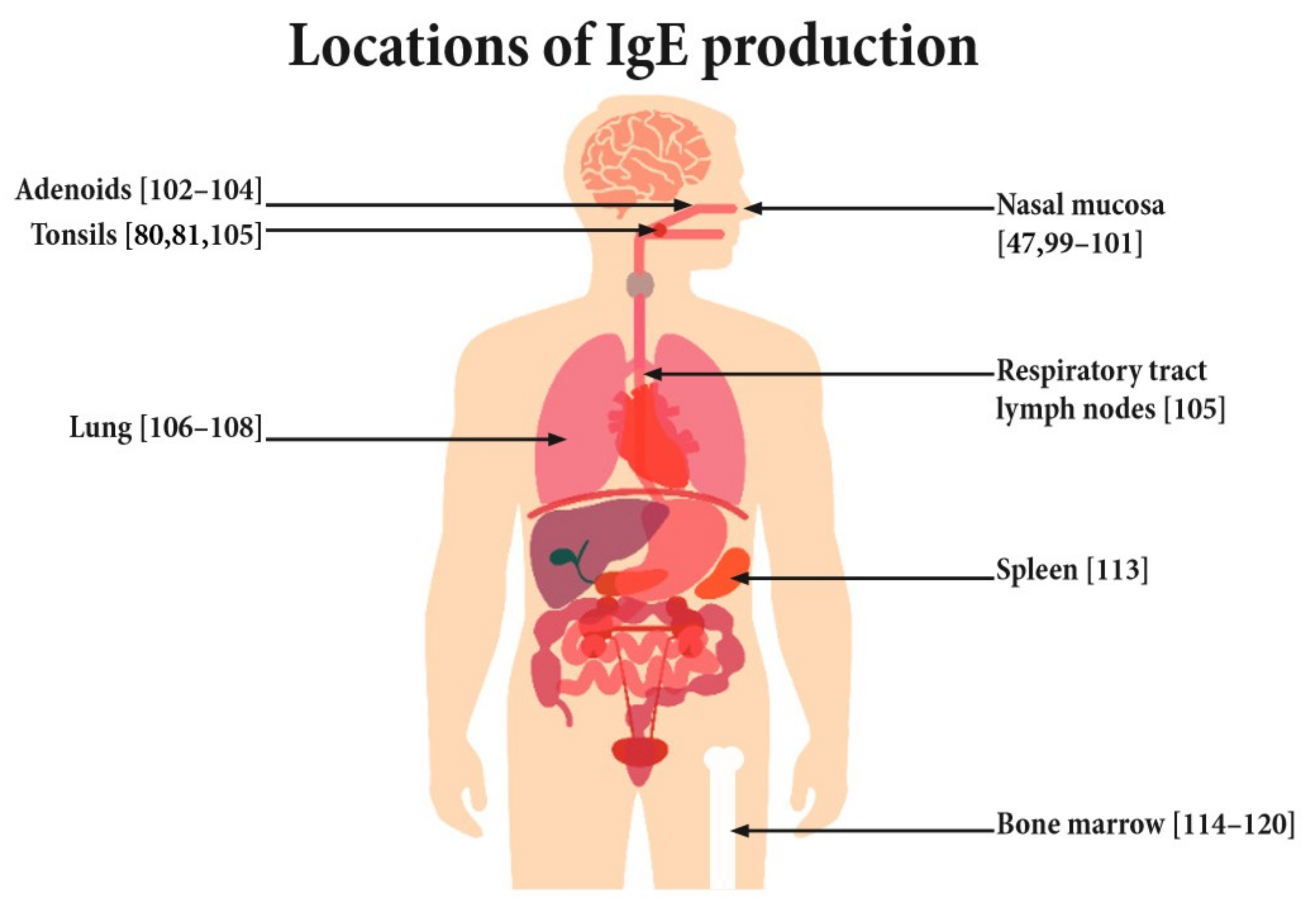
3.2. Sites of IgE Production
4. Role of IgE in Mediating Immediate Allergic Symptoms and T Cell-Mediated Allergic Inflammation
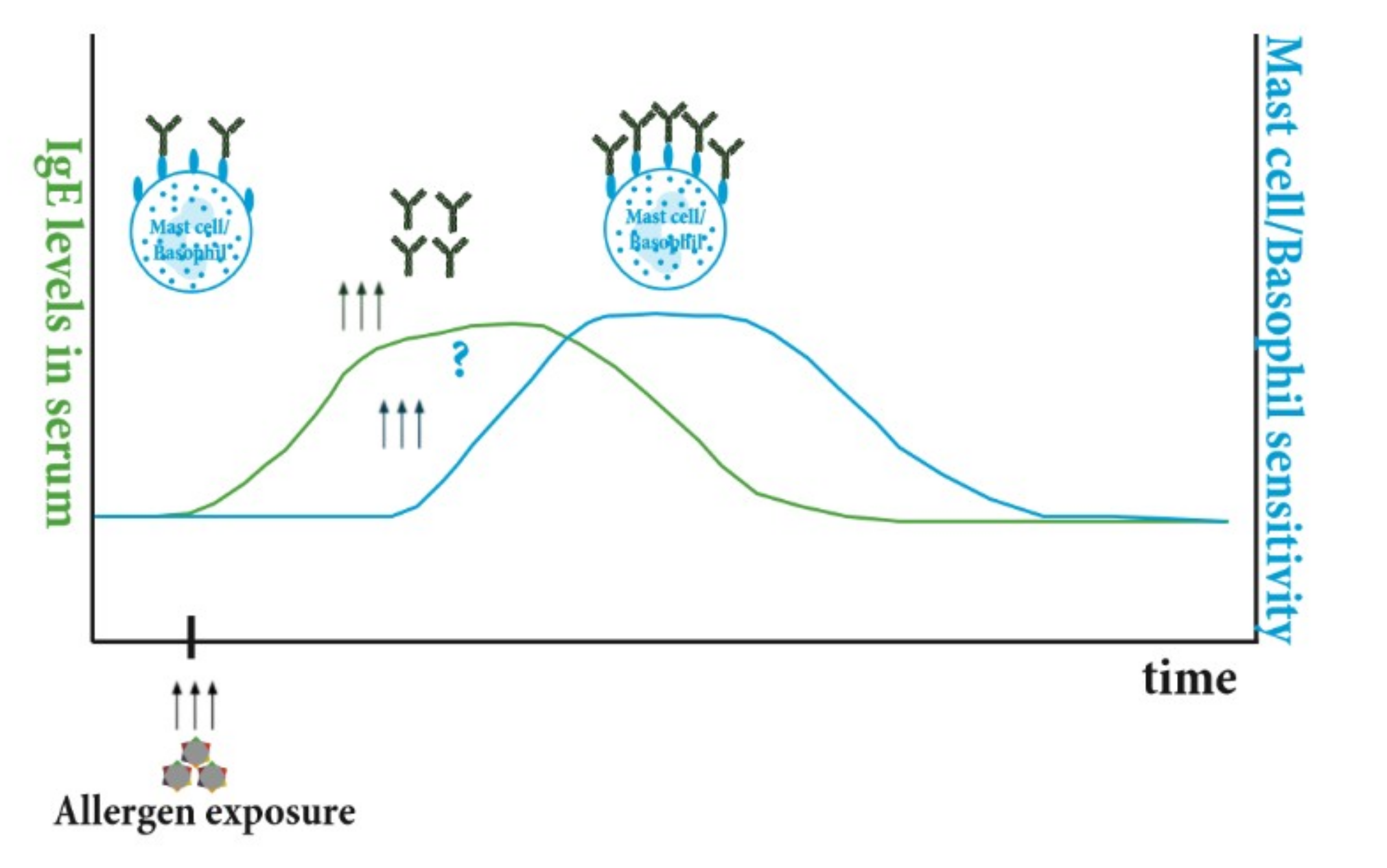
4.1. Connecting IgE Production to Clinical Effect—How Circulating IgE Influences Mast Cell and Basophil Sensitivity
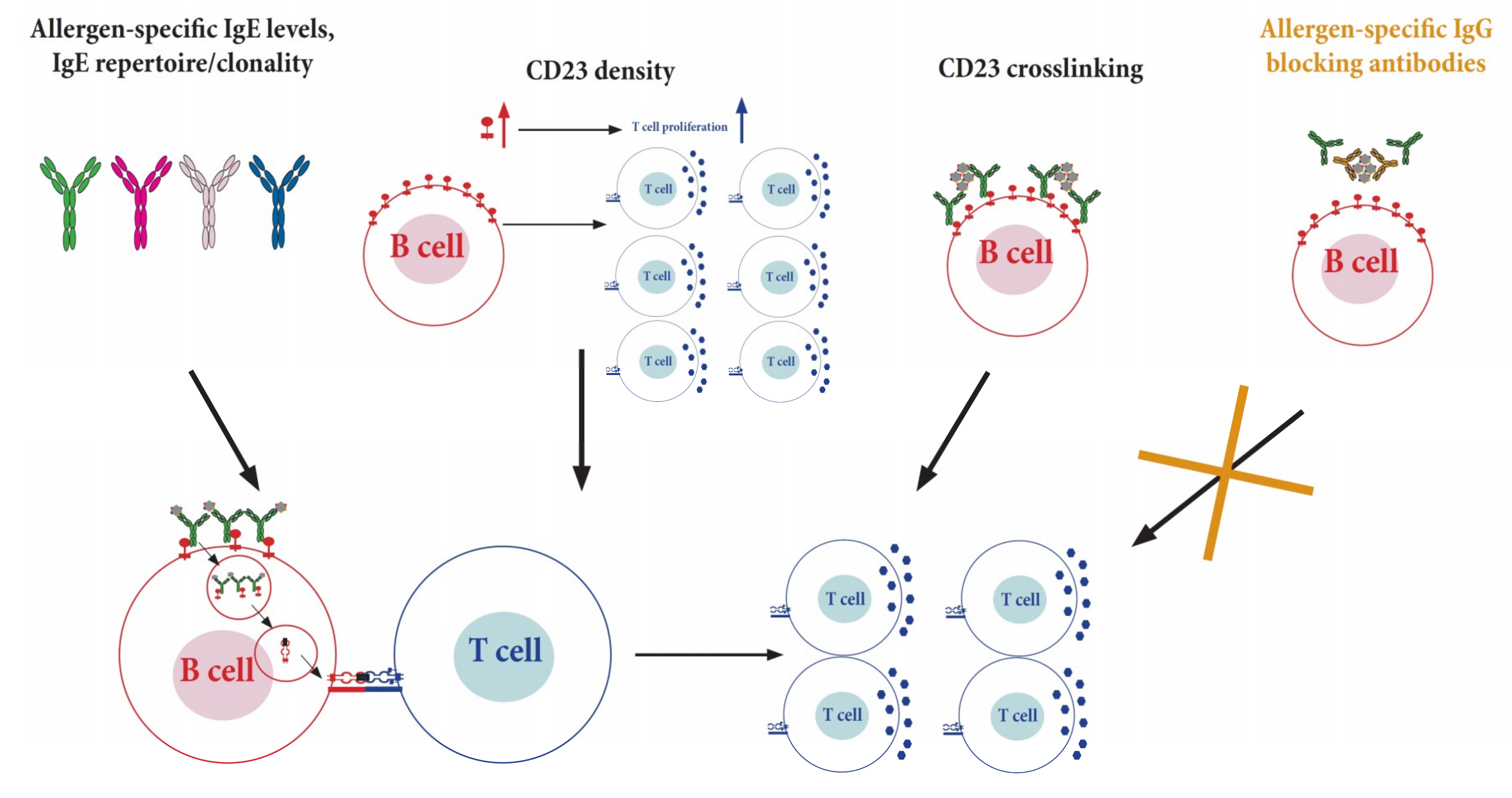
4.2. Importance of IgE-Facilitatated Allergen Presentation Mediated by CD23 and Rules Guiding this Process
5. Targeting of IgE and IgE+ Cells
5.1. Targeting IgE and Interference with Activation of Effector Cells
5.2. Therapeutic Targeting of IgE-Producing Cells
6. Conclusions
Appendix A
| A | Approach | Biological Effect | Study Design | Disease Target | Subjects | Length of Observation | Outcome | Current Phase | References |
| Strategies targeting IgE | Immuno adsorption | Removal of circulating IgE or total Ig through Plasmapheresis | RCT | Allergic Asthma | N = 15 | 16 weeks | ↓Total IgE ↓ basophil activation | Pre-Clinical | [13] |
| RCT | Atopic Dermatitis | N = 50 | 8 weeks | Less AE in IgE group vs pan Ig | Pre-Clinical | [20] | |||
| Omalizumab | Monoclonal antibody against Fc portion of IgE—prevents receptor binding | RCT | Allergic Asthma | N = 317 | 20 weeks | ↓IgE ↓Steroid use | Marketed | [17] | |
| CMAB007 | Biosimilar to Omalizumab developed by China | RCT | Allergic Asthma | N = 400 | 24 weeks | Not yet completed | Phase III NCT03468790 | [163] | |
| DARPins | Ankyrin repeat domains that affect stability and function of target protein | In vitro study | Allergy | Isolated basophils | N/A | Removal of IgE from basophils + ↓ basophil activation | Pre-Clinical | [168] | |
| MEDI4212 | Monoclonal antibody against Fc portion of IgE—prevents receptor binding | RCT | Allergy/Atopy | N = 86 | 12 weeks | Greater ↓total IgE vs Omalizumab worse half life | Phase I NCT01544348 | [166] | |
| MEDI4212 Variant | Monoclonal antibody against Fc portion of IgE and Fc potion of monoclonal antibody binds to inhibitory receptor FcγRIIIa on B-cells | In vitro study | Allergy | Cell lines and human B cells | N/A | Elimination of IgE expressing B cells | Pre-Clinical | [178] | |
| IgE Peptide Vaccine | Induction of autoantibodies against Fc region of IgE | In vitro study | Allergy | FcεRI–ELISA | N/A | Autoantibodies block IgE binding to FcεRI | Pre-Clinical | [171] | |
| QGE031 (Ligelizumab) | Monoclonal antibody against Fc portion of IgE—prevents receptor binding | RCT | Allergic Asthma | N = 37 | 10 weeks | QGE031 > Omalizumab | Phase II NCT01703312 | [164] | |
| B | Approaches | Biological Effect | Study Design | Disease Target | Subjects | Length of Observation | Outcome | Current Trail Phase | References |
| Strategies targeting IgE production or effector cells | Quilizumab | Monoclonal antibody targeting M1-prime segment of membrane bound IgE expressed on IgE switched B cells leading to cell depletion | RCT | Allergic Asthma | N = 578 | 36 weeks with 48 week safety follow-up | Acceptable safety and reduced serum IgE but no clinically meaningful benefit in clinical outcome parameters | Phase II NCT01582503 | [23] |
| DARPins | Ankyrin repeat domains that affect stability and function of target protein | In vitro study | Allergy | Human basophils | N/A | Targets FcγIIB and inhibits basophil degranulation | Pre-Clinical | [169] | |
| Bsc-IgE/CD3 Construct | Monoclonal antibody binding to cells with membrane bound IgE and targets T-cell cytotoxic activity towards them | In vitro study | Allergy | Cells isolated from allergic human donors | N/A | Bsc-IgE/CD3 effective at eliminating IgE+ B cells without inducing degranulation of mast cells | Pre-Clinical | [176] | |
| Anti-FcεRI Fab-conjugated celastrol-loaded micelles | Fusion with cell membrane and induction of apoptosis of FcεRI expressing cells | In vitro study | Allergy | Mast cell line | N/A | Efficient induction of apoptosis of mast cells and reduction of allergic inflammation in mouse model | Pre-Clinical | [21] | |
| CTLA4Fcε Fusion Protein | Binds FcεRI and CD23, prevents CD23 cleavage, and blocks CD80/CD86 costimulation | In vitro study | Allergy | Cell line and human PBMC samples | N/A | Reduces sCD23 and lymphocyte proliferation | Pre-Clinical | [22] | |
| Maternal Anti-IgE Vaccination | IgG anti IgE antibodies transferred from mother to fetus and prevent onset of allergy by targeting IgE memory B cells | In vivo mouse study | Allergy | N/A | 9 weeks after birth | Reduced IgE levels in mouse offspring | Pre-Clinical | [180] |
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| APC | Antigen Presenting Cell |
| BCR | B Cell Receptor |
| CD23 | Low Affinity Receptor for IgE |
| FcεRI | High Affinity Receptor for IgE |
| IgE | Immunoglobulin E |
| MHC | Major Histocompatibility Complex |
| PBMC | Peripheral Blood Mononuclear Cell |
References
- Valenta, R.; Karaulov, A.; Niederberger, V.; Gattinger, P.; van Hage, M.; Flicker, S.; Linhart, B.; Campana, R.; Focke-Tejkl, M.; Curin, M.; et al. Molecular aspects of allergens and allergy. Adv. Immunol. 2018, 138, 195–256. [Google Scholar] [PubMed]
- Bønnelykke, K.; Sparks, R.; Waage, J.; Milner, J.D. Genetics of allergy and allergic sensitization: Common variants, rare mutations. Curr. Opin. Immunol. 2015, 36, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Neunkirchner, A.; Kratzer, B.; Köhler, C.; Smole, U.; Mager, L.F.; Schmetterer, K.G.; Trapin, D.; Leb-Reichl, V.; Rosloniec, E.; Naumann, R.; et al. Genetic restriction of antigen-presentation dictates allergic sensitization and disease in humanized mice. EBioMedicine 2018, 31, 66–78. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, S.C. Role of mast cells in allergic and non-allergic immune responses: Comparison of human and murine data. Nat. Rev. Immunol. 2007, 7, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Bruhns, P.; Jönsson, F. Mouse and human FcR effector functions. Immunol. Rev. 2015, 268, 25–51. [Google Scholar] [CrossRef] [PubMed]
- Van Der Heijden, F.L.; Van Neerven, R.J.J.; Van Katwijk, M.; Bos, J.D.; Kapsenberg, M.L. Serum-IgE-facilitated allergen presentation in atopic disease. J. Immunol. 1993, 150, 3643–3650. [Google Scholar]
- Mudde, G.C.; Bheekha, R.; Bruijnzeel-Koomen, C.A. IgE-mediated antigen presentation. Allergy 1995, 50, 193–199. [Google Scholar] [CrossRef]
- Maurer, D.; Ebner, C.; Reininger, B.; Fiebiger, E.; Kraft, D.; Kinet, J.P.; Stingl, G. The high affinity IgE receptor (FcεRI) mediates IgE-dependent allergen presentation. J. Immunol. 1995, 154, 6285–6290. [Google Scholar]
- Eckl-Dorna, J.; Villazala-Merino, S.; Linhart, B.; Karaulov, A.V.; Zhernov, Y.; Khaitov, M.; Niederberger-Leppin, V.; Valenta, R. Allergen-Specific Antibodies Regulate Secondary Allergen-Specific Immune Responses. Front. Immunol. 2019, 9, 3131. [Google Scholar] [CrossRef]
- Platzer, B.; Ruiter, F.; Van Der Mee, J.; Fiebiger, E. Soluble IgE receptors—Elements of the IgE network. Immunol. Lett. 2011, 141, 36–44. [Google Scholar] [CrossRef]
- Dullaers, M.; De Bruyne, R.; Ramadani, F.; Gould, H.J.; Gevaert, P.; Lambrecht, B.N. The who, where, and when of IgE in allergic airway disease. J. Allergy Clin. Immunol. 2012, 129, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, R.G.; Adkinson, N.F., Jr. 23. Clinical laboratory assessment of IgE-dependent hypersensitivity. J. Allergy Clin. Immunol. 2003, 111, S687–S701. [Google Scholar] [CrossRef] [PubMed]
- Lupinek, C.; Derfler, K.; Lee, S.; Prikoszovich, T.; Movadat, O.; Wollmann, E.; Cornelius, C.; Weber, M.; Fröschl, R.; Selb, R.; et al. Extracorporeal IgE Immunoadsorption in Allergic Asthma: Safety and Efficacy. EBioMedicine 2017, 17, 119–133. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Chen, J.; Ye, L.; Cai, Z.; Sun, J.; Ji, K. Anti-IgE therapy for IgE-mediated allergic diseases: From neutralizing IgE antibodies to eliminating IgE+ B cells. Clin. Transl. Allergy 2018, 8, 27. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.C.; Scheerens, H. Targeting IgE production in mice and humans. Curr. Opin. Immunol. 2014, 31, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Balbino, B.; Conde, E.; Marichal, T.; Starkl, P.; Reber, L.L. Aproaches to target IgE antibodies in allergic disease. Pharmacol. Ther. 2018, 191, 50–64. [Google Scholar] [CrossRef] [PubMed]
- Milgrom, H.; Fick, R.B., Jr.; Su, J.Q.; Reimann, J.D.; Bush, R.K.; Watrous, M.L.; Metzger, W.J. Treatment of allergic asthma with monoclonal anti-IgE antibody. Rhumab-e25 study group. N. Engl. J. Med. 1999, 341, 1966–1973. [Google Scholar] [CrossRef] [PubMed]
- Busse, W.; Corren, J.; Lanier, B.Q.; McAlary, M.; Fowler-Taylor, A.; Della Cioppa, G.; Van As, A.; Gupta, N. Omalizumab, anti-IgE recombinant humanized monoclonal antibody, for the treatment of severe allergic asthma. J. Allergy Clin. Immunol. 2001, 108, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Humbert, M.; Busse, W.; Hanania, N.A.; Lowe, P.J.; Canvin, J.; Erpenbeck, V.J.; Holgate, S. Omalizumab in Asthma: An Update on Recent Developments. J. Allergy Clin. Immunol. Pract. 2014, 2, 525–536. [Google Scholar] [CrossRef] [PubMed]
- Reich, K.; Deinzer, J.; Fiege, A.-K.; Von Gruben, V.; Sack, A.-L.; Thraen, A.; Weisenseel, P.; Breuer, K.; Jäckle, S.; Meier, M. Panimmunoglobulin and IgE-selective extracorporeal immunoadsorption in patients with severe atopic dermatitis. J. Allergy Clin. Immunol. 2016, 137, 1882–1884. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Peng, X.; Wang, J.; Li, X.; Lin, L.; Xie, G.; Cui, Z.; Li, J.; Wang, Y.; Li, L. Targeting Mast Cells and Basophils with Anti-FcεRIα Fab-Conjugated Celastrol-Loaded Micelles Suppresses Allergic Inflammation. J. Biomed. Nanotechnol. 2015, 11, 2286–2299. [Google Scholar] [CrossRef] [PubMed]
- Perez-Witzke, D.; Miranda-García, M.A.; Suárez, N.; Becerra, R.; Duque, K.; Porras, V.; Fuenmayor, J.; Montano, R.F. CTLA4Fcε, a novel soluble fusion protein that binds B7 molecules and the IgE receptors, and reduces human in vitro soluble CD23 production and lymphocyte proliferation. Immunology 2016, 148, 40–55. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.M.; Maciuca, R.; Bradley, M.S.; Cabanski, C.R.; Scheerens, H.; Lim, J.; Cai, F.; Kishnani, M.; Liao, X.C.; Samineni, D.; et al. A randomized trial of the efficacy and safety of quilizumab in adults with inadequately controlled allergic asthma. Respir. Res. 2016, 17, 29. [Google Scholar] [CrossRef] [PubMed]
- Horst, A.; Hunzelmann, N.; Arce, S.; Herber, M.; Manz, R.A.; Radbruch, A.; Nischt, R.; Schmitz, J.; Assenmacher, M. Detection and characterization of plasma cells in peripheral blood: Correlation of IgE+ plasma cell frequency with IgE serum titre. Clin. Exp. Immunol. 2002, 130, 370–378. [Google Scholar] [CrossRef] [PubMed]
- Pree, I.; Reisinger, J.; Marth, K.; Chen, K.-W.; Vrtala, S.; Spitzauer, S.; Valenta, R.; Niederberger, V.; Eckl-Dorna, J.; Eckl-Dorna, J. The majority of allergen-specific IgE in the blood of allergic patients does not originate from blood-derived B cells or plasma cells. Clin. Exp. Allergy 2012, 42, 1347–1355. [Google Scholar]
- Østergaard, P.A. Tonsillar IgE plasma cells predict atopic disease. Clin. Exp. Immunol. 1982, 49, 163–166. [Google Scholar] [PubMed]
- Smurthwaite, L.; Walker, S.N.; Wilson, D.R.; Birch, D.S.; Merrett, T.G.; Durham, S.R.; Gould, H.J. Persistent IgE synthesis in the nasal mucosa of hay fever patients. Eur. J. Immunol. 2001, 31, 3422–3431. [Google Scholar] [CrossRef]
- Wu, L.C.; Zarrin, A.A. The production and regulation of IgE by the immune system. Nat. Rev. Immunol. 2014, 14, 247–259. [Google Scholar] [CrossRef] [PubMed]
- He, J.S.; Meyer-Hermann, M.; Xiangying, D.; Zuan, L.Y.; Jones, L.A.; Ramakrishna, L.; de Vries, V.C.; Dolpady, J.; Aina, H.; Joseph, S.; et al. The distinctive germinal center phase of IgE+ B lymphocytes limits their contribution to the classical memory response. J. Exp. Med. 2013, 210, 2755–2771. [Google Scholar] [CrossRef]
- Erazo, A.; Kutchukhidze, N.; Leung, M.; Christ, A.P.G.; Urban, J.F.; De Lafaille, M.A.C.; Lafaille, J.J. Unique maturation program of the IgE response in vivo. Immunity 2007, 26, 191–203. [Google Scholar] [CrossRef]
- Kühn, R.; Rajewsky, K.; Müller, W. Generation and analysis of interleukin-4 deficient mice. Science 1991, 254, 707–710. [Google Scholar] [CrossRef] [PubMed]
- Punnonen, J.; Yssel, H.; de Vries, J.E. The relative contribution of IL-4 and IL-13 to human IgE synthesis induced by activated CD4+ or CD8+ T cells. J. Allergy Clin. Immunol. 1997, 100, 792–801. [Google Scholar] [CrossRef]
- Vrtala, S.; Ball, T.; Spitzauer, S.; Pandjaitan, B.; Suphioglu, C.; Knox, B.; Sperr, W.R.; Valent, P.; Kraft, D.; Valenta, R. Immunization with purified natural and recombinant allergens induces mouse IgG1 antibodies that recognize similar epitopes as human IgE and inhibit the human IgE-allergen interaction and allergen-induced basophil degranulation. J. Immunol. 1998, 160, 6137–6144. [Google Scholar] [PubMed]
- Xiong, H.; Dolpady, J.; Wabl, M.; De Lafaille, M.A.C.; Lafaille, J.J. Sequential class switching is required for the generation of high affinity IgE antibodies. J. Exp. Med. 2012, 209, 353–364. [Google Scholar] [CrossRef] [PubMed]
- Pree, I.; Reisinger, J.; Focke, M.; Vrtala, S.; Pauli, G.; Van Hage, M.; Cromwell, O.; Gadermaier, E.; Egger, C.; Reider, N.; et al. Analysis of Epitope-Specific Immune Responses Induced by Vaccination with Structurally Folded and Unfolded Recombinant Bet v 1 Allergen Derivatives in Man. J. Immunol. 2007, 179, 5309–5316. [Google Scholar] [CrossRef] [PubMed]
- Curin, M.; Swoboda, I.; Wollmann, E.; Lupinek, C.; Spitzauer, S.; Van Hage, M.; Valenta, R. Microarrayed dog, cat, and horse allergens show weak correlation between allergen-specific IgE and IgG responses. J. Allergy Clin. Immunol. 2014, 133, 918–921. [Google Scholar] [CrossRef]
- Niederberger, V.; Ring, J.; Rakoski, J.; Jager, S.; Spitzauer, S.; Valent, P.; Horak, F.; Kundi, M.; Valenta, R. Antigens drive memory IgE responses in human allergy via the nasal mucosa. Int. Arch. Allergy Immunol. 2007, 142, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Egger, C.; Lupinek, C.; Ristl, R.; Lemell, P.; Horak, F.; Zieglmayer, P.; Spitzauer, S.; Valenta, R.; Niederberger, V. Effects of Nasal Corticosteroids on Boosts of Systemic Allergen-Specific IgE Production Induced by Nasal Allergen Exposure. PLoS ONE 2015, 10, e0114991. [Google Scholar] [CrossRef]
- Doeing, D.C.; Borowicz, J.L.; Crockett, E.T. Gender dimorphism in differential peripheral blood leukocyte counts in mice using cardiac, tail, foot, and saphenous vein puncture methods. BMC Clin. Pathol. 2003, 3, 3. [Google Scholar] [CrossRef] [PubMed]
- Mestas, J.; Hughes, C.C.W. Of Mice and Not Men: Differences between Mouse and Human Immunology. J. Immunol. 2004, 172, 2731–2738. [Google Scholar] [CrossRef]
- Kita, H.; Gleich, G.J. Eosinophils and IgE receptors: A continuing controversy. Blood 1997, 89, 3497–3501. [Google Scholar] [PubMed]
- Gounni, A.S.; Lamkhioued, B.; Ochiai, K.; Tanaka, Y.; Delaporte, E.; Capron, A.; Kinet, J.-P.; Capron, M. High-affinity IgE receptor on eosinophils is involved in defence against parasites. Nature 1994, 367, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Noti, M.; Kim, B.S.; Siracusa, M.C.; Rak, G.D.; Kubo, M.; Moghaddam, A.E.; Sattentau, Q.A.; Comeau, M.R.; Spergel, J.M.; Artis, D. Exposure to food allergens through inflamed skin promotes intestinal food allergy through the thymic stromal lymphopoietin–basophil axis. J. Allergy Clin. Immunol. 2014, 133, 1390–1399. [Google Scholar] [CrossRef] [PubMed]
- Takeda, K.; Gelfand, E.W. Mouse Models of allergic diseases. Curr Opin Immunol 2009, 21, 660–665. [Google Scholar] [CrossRef]
- Spergel, J. Atopic dermatitis and the atopic march. J. Allergy Clin. Immunol. 2003, 112, S118–S127. [Google Scholar] [CrossRef] [PubMed]
- Berkowska, M.A.; Heeringa, J.J.; Hajdarbegovic, E.; van der Burg, M.; Thio, H.B.; van Hagen, P.M.; Boon, L.; Orfao, A.; van Dongen, J.J.; van Zelm, M.C. Human IgE+ B cells are derived from T cell-dependent and T cell-independent pathways. J. Allergy Clin. Immunol. 2014, 134, 688–697. [Google Scholar] [CrossRef] [PubMed]
- Cameron, L.; Gounni, A.S.; Frenkiel, S.; Lavigne, F.; Vercelli, D.; Hamid, Q. Sε, sµ and sγ switch circles in human nasal mucosa following ex vivo allergen challenge: Evidence for direct as well as sequential class switch recombination. J. Immunol. 2003, 171, 3816–3822. [Google Scholar] [CrossRef]
- Ramadani, F.; Bowen, H.; Upton, N.; Hobson, P.S.; Chan, Y.C.; Chen, J.B.; Chang, T.W.; McDonell, J.M.; Sutton, B.J.; Fear, D.J.; et al. Ontogeny of human IgE-expressing B cells and plasma cells. Allergy 2017, 72, 66–76. [Google Scholar] [CrossRef]
- Takhar, P.; Smurthwaite, L.; Coker, H.A.; Fear, D.J.; Banfield, G.K.; Carr, V.A.; Durham, S.R.; Gould, H.J. Allergen Drives Class Switching to IgE in the Nasal Mucosa in Allergic Rhinitis. J. Immunol. 2005, 174, 5024–5032. [Google Scholar] [CrossRef]
- Mills, F.C.; Mitchell, M.P.; Harindranath, N.; Max, E.E. Human Ig S gamma regions and their participation in sequential switching to IgE. J. Immunol. 1995, 155, 3021–3026. [Google Scholar]
- Looney, T.J.; Lee, J.Y.; Roskin, K.M.; Hoh, R.A.; King, J.; Glanville, J.; Liu, Y.; Pham, T.D.; Dekker, C.L.; Davis, M.M.; et al. Human B-cell isotype switching origins of IgE. J. Allergy Clin. Immunol. 2016, 137, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Saunders, S.P.; Ma, E.G.M.; Aranda, C.J.; De Lafaille, M.A.C. Non-classical B Cell Memory of Allergic IgE Responses. Front. Immunol. 2019, 10, 710–715. [Google Scholar] [CrossRef] [PubMed]
- Niederberger, V.; Niggemann, B.; Kraft, D.; Spitzauer, S.; Valenta, R. Evolution of IgM, IgE and IgG1–4 antibody responses in early childhood monitored with recombinant allergen components: Implications for class switch mechanisms. Eur. J. Immunol. 2002, 32, 576–584. [Google Scholar] [CrossRef]
- Wagner, J.G.; Harkema, J.R. Rodent models of allergic rhinitis: Relevance to human pathophysiology. Curr. Allergy Asthma Rep. 2007, 7, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Harkema, J.R.; Carey, S.A.; Wagner, J.G. The Nose Revisited: A Brief Review of the Comparative Structure, Function, and Toxicologic Pathology of the Nasal Epithelium. Toxicol. Pathol. 2006, 34, 252–269. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, S.; Holgate, S.T. The mouse trap: It still yields few answers in asthma. Am. J. Respir. Crit. Care Med. 2006, 174, 1173–1176. [Google Scholar] [CrossRef] [PubMed]
- Gordon, C.J.; Grafton, G.; Wood, P.M.; Larché, M.; Armitage, R.J. Modelling the human immune response: Can mice be trusted? Commentary. Curr. Opin. Pharmacol. 2001, 1, 431–435. [Google Scholar] [CrossRef]
- Hessel, E.M.; Van Oosterhout, A.J.; Van Ark, I.; Van Esch, B.; Hofman, G.; Van Loveren, H.; Savelkoul, H.F.; Nijkamp, F.P. Development of airway hyperresponsiveness is dependent on interferon-gamma and independent of eosinophil infiltration. Am. J. Respir. Cell Mol. Boil. 1997, 16, 325–334. [Google Scholar] [CrossRef]
- Spergel, J.M.; Mizoguchi, E.; Brewer, J.P.; Martin, T.R.; Bhan, A.K.; Geha, R.S. Epicutaneous sensitization with protein antigen induces localized allergic dermatitis and hyperresponsiveness to methacholine after single exposure to aerosolized antigen in mice. J. Clin. Investig. 1998, 101, 1614–1622. [Google Scholar] [CrossRef]
- Mitre, E.; Nutman, T.B. IgE memory: Persistence of antigen-specific IgE responses years after treatment of human filarial infections. J. Allergy Clin. Immunol. 2006, 117, 939–945. [Google Scholar] [CrossRef]
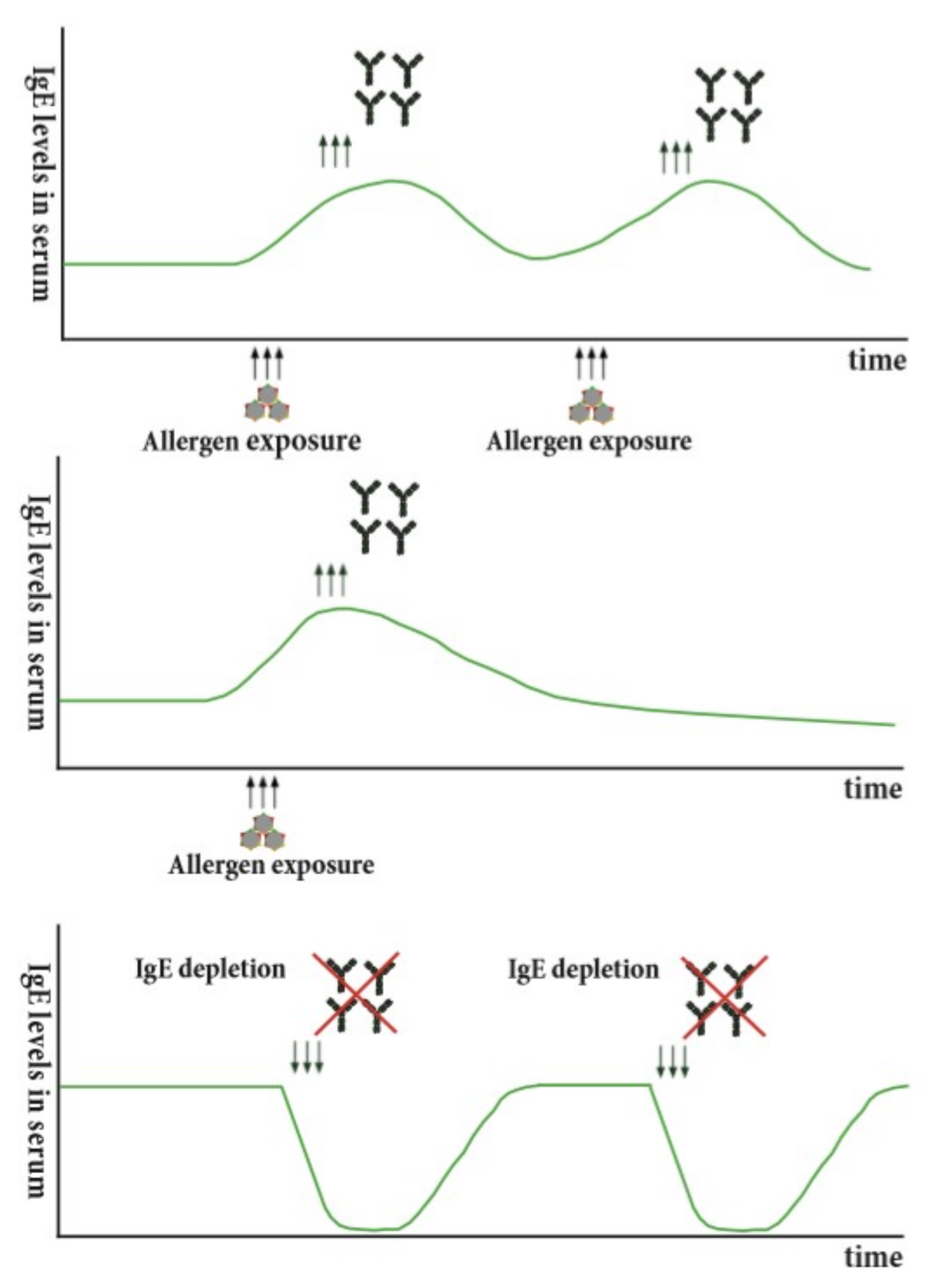
- Henderson, L.; Larson, J.; Gleich, G. Maximal rise in IgE antibody following ragweed pollination season. J. Allergy Clin. Immunol. 1975, 55, 10–15. [Google Scholar] [CrossRef]
- Manz, R.A.; Thiel, A.; Radbruch, A. Lifetime of plasma cells in the bone marrow. Nature 1997, 388, 133–134. [Google Scholar] [CrossRef] [PubMed]
- Manz, R.A.; Arce, S.; Cassese, G.; Hauser, A.E.; Hiepe, F.; Radbruch, A. Humoral immunity and long-lived plasma cells. Curr. Opin. Immunol. 2002, 14, 517–521. [Google Scholar] [CrossRef]
- Traggiai, E.; Bernasconi, N.L.; Lanzavecchia, A. Maintenance of Serological Memory by Polyclonal Activation of Human Memory B Cells. Science 2002, 298, 2199–2202. [Google Scholar]
- Hochwallner, H.; Schulmeister, U.; Swoboda, I.; Balic, N.; Geller, B.; Nystrand, M.; Harlin, A.; Thalhamer, J.; Scheiblhofer, S.; Niggemann, B.; et al. Microarray and allergenic activity assessment of milk allergens. Clin. Exp. Allergy 2010, 40, 1809–1818. [Google Scholar] [CrossRef] [PubMed]
- Hochwallner, H.; Schulmeister, U.; Swoboda, I.; Focke-Tejkl, M.; Civaj, V.; Balic, N.; Nystrand, M.; Harlin, A.; Thalhamer, J.; Scheiblhofer, S.; et al. Visualization of clustered IgE epitopes on alpha-lactalbumin. J. Allergy Clin. Immunol. 2010, 125, 1279–1285. [Google Scholar] [CrossRef] [PubMed]
- Reininger, R.; Exner, H.; Kuderna, C.; Rumpold, H.; Balic, N.; Valenta, R.; Spitzauer, S. Possible Modes of Allergen-Specific Sensitization and Boosting in an Atopic Child. Int. Arch. Allergy Immunol. 2003, 130, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Bradleyeisenbrey, A.; Agarwal, M.; Offord, K.; Adolphson, C.; Yunginger, J.; Gleich, G. Seasonal variation of in vitro lymphocyte proliferative response to ragweed antigen E. J. Allergy Clin. Immunol. 1985, 75, 84–90. [Google Scholar] [CrossRef]
- Eckl-Dorna, J.; Campana, R.; Valenta, R.; Niederberger, V. Poor association of allergen-specific antibody, T- and B-cell responses revealed with recombinant allergens and a CFSE dilution-based assay. Allergy 2015, 70, 1222–1229. [Google Scholar] [CrossRef] [PubMed]
- Lupinek, C.; Marth, K.; Niederberger, V.; Valenta, R. Analysis of serum IgE reactivity profiles with microarrayed allergens indicates absence of de novo IgE sensitizations in adults. J. Allergy Clin. Immunol. 2012, 130, 1418–1420. [Google Scholar] [CrossRef] [PubMed]
- Blauvelt, A.; Simpson, E.L.; Tyring, S.K.; Purcell, L.A.; Shumel, B.; Petro, C.D.; Akinlade, B.; Gadkari, A.; Eckert, L.; Graham, N.M.; et al. Dupilumab does not affect correlates of vaccine-induced immunity: A randomized, placebo-controlled trial in adults with moderate-to-severe atopic dermatitis. J. Am. Acad. Dermatol. 2019, 80, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Bachert, C.; Mannent, L.; Naclerio, R.M.; Mullol, J.; Ferguson, B.J.; Gevaert, P.; Hellings, P.; Jiao, L.; Wang, L.; Evans, P.R.; et al. Effect of subcutaneous Dupilumab on nasal polyp burden in patients with chronic sinusitis and nasal polyposis: A randomized clinical trial. JAMA 2016, 315, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Beck, L.A.; Thaci, D.; Hamilton, J.D.; Graham, N.M.; Bieber, T.; Rocklin, R.; Ming, J.E.; Ren, H.; Kao, R.; Simpson, E.; et al. Dupilumab Treatment in Adults with Moderate-to-Severe Atopic Dermatitis. N. Engl. J. Med. 2014, 371, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, S.; Ford, L.; Pearlman, D.; Spector, S.; Sher, L.; Skobieranda, F.; Wang, L.; Kirkesseli, S.; Rocklin, R.; Bock, B.; et al. Dupilumab in Persistent Asthma with Elevated Eosinophil Levels. N. Engl. J. Med. 2013, 368, 2455–2466. [Google Scholar] [CrossRef] [PubMed]
- Fiser, P.M.; Buckley, R.H. Human IgE biosynthesis in vitro: Studies with atopic and normal blood mononuclear cells and subpopulations. J. Immunol. 1979, 123, 1788–1794. [Google Scholar] [PubMed]
- Romagnani, S.; Maggi, E.; Del Prete, G.F.; Troncone, R.; Ricci, M. In vitro production of IgE by human peripheral blood mononuclear cells. I. Rate of IgE biosynthesis. Clin. Exp. Immunol. 1980, 42, 167–174. [Google Scholar] [PubMed]
- Steinberger, P.; Bohle, B.; DiPadova, F.; Wrann, M.; Liehl, E.; Scheiner, O.; Kraft, D.; Valenta, R. Allergen-specific IgE production of committed B cells from allergic patients in vitro. J. Allergy Clin. Immunol. 1995, 96, 209–218. [Google Scholar] [CrossRef]
- Ohta, K.; Manzara, T.; Harbeck, R.J.; Kirkpatrick, C.H. Human tonsillar IgE biosynthesis in vitro. I. Enhancement of IgE and IgG synthesis in the presence of pokeweed mitogen by T-cell irradiation. J. Allergy Clin. Immunol. 1983, 71, 212–223. [Google Scholar] [CrossRef]
- Takenaka, H.; Kusumi, T.; Mizukoshi, O. In vitro synthesis of IgE antibody by human tonsil mononuclear cells. Preliminary report. Acta Oto-Laryngol. 1988, 105, 133–137. [Google Scholar] [CrossRef]
- Jabara, H.H. CD40 and IgE: Synergism between anti-CD40 monoclonal antibody and interleukin 4 in the induction of IgE synthesis by highly purified human B cells. J. Exp. Med. 1990, 172, 1861–1864. [Google Scholar] [CrossRef]
- Maggi, E.; Del Prete, G.F.; Parronchi, P.; Tiri, A.; Macchia, D.; Biswas, P.; Simonelli, C.; Ricci, M.; Romagnani, S. Role for T cells, IL-2 and IL-6 in the IL-4-dependent in vitro human IgE synthesis. Immunology 1989, 68, 300–306. [Google Scholar] [PubMed]
- Pene, J.; Rousset, F.; Briere, F.; Chretien, I.; Bonnefoy, J.Y.; Spits, H.; Yokota, T.; Arai, N.; Arai, K.; Banchereau, J.; et al. IgE production by normal human lymphocytes is induced by interleukin 4 and suppressed by interferons gamma and alpha and prostaglandin e2. Proc. Natl. Acad. Sci. USA 1988, 85, 6880–6884. [Google Scholar] [CrossRef] [PubMed]
- Gascan, H.; Gauchat, J.F.; Aversa, G.; Van Vlasselaer, P.; de Vries, J.E. Anti-CD40 monoclonal antibodies or CD4+ T cell clones and IL-4 induce IgG4 and IgE switching in purified human B cells via different signaling pathways. J. Immunol. 1991, 147, 8–13. [Google Scholar] [PubMed]
- Steinberger, P.; Kraft, D.; Valenta, R. Construction of a combinatorial IgE library from an allergic patient. Isolation and characterization of human IgE Fabs with specificity for the major timothy grass pollen allergen, Phl p 5. J. Biol. Chem. 1996, 271, 10967–10972. [Google Scholar] [CrossRef]
- Eckl-Dorna, J.; Niederberger, V. What Is the Source of Serum Allergen-Specific IgE? Curr. Allergy Asthma Rep. 2013, 13, 281–287. [Google Scholar] [CrossRef]
- Jiménez-Saiz, R.; Ellenbogen, Y.; Bruton, K.; Spill, P.; Sommer, D.D.; Lima, H.; Waserman, S.; Patil, S.U.; Shreffler, W.G.; Jordana, M. Human BCR analysis of single-sorted, putative IgE. J. Allergy Clin. Immunol. 2019, 144, 336–339. [Google Scholar] [CrossRef]
- Wong, K.J.; Timbrell, V.; Xi, Y.; Upham, J.W.; Collins, A.M.; Davies, J.M. IgE+ B cells are scarce, but allergen-specific B cells with a memory phenotype circulate in patients with allergic rhinitis. Allergy 2015, 70, 420–428. [Google Scholar] [CrossRef]
- Selb, R.; Eckl-Dorna, J.; Neunkirchner, A.; Schmetterer, K.; Marth, K.; Gamper, J.; Jahn-Schmid, B.; Pickl, W.F.; Valenta, R.; Niederberger, V. CD23 surface density on B cells is associated with IgE levels and determines IgE-facilitated allergen uptake, as well as activation of allergen-specific T cells. J. Allergy Clin. Immunol. 2017, 139, 290–299. [Google Scholar] [CrossRef]
- Heeringa, J.J.; Rijvers, L.; Arends, N.J.; Driessen, G.J.; Pasmans, S.G.; Van Dongen, J.J.M.; De Jongste, J.C.; Van Zelm, M.C. IgE-expressing memory B cells and plasmablasts are increased in blood of children with asthma, food allergy, and atopic dermatitis. Allergy 2018, 73, 1331–1336. [Google Scholar] [CrossRef]
- Croote, D.; Darmanis, S.; Nadeau, K.C.; Quake, S.R. High-affinity allergen-specific human antibodies cloned from single IgE B cell transcriptomes. Science 2018, 362, 1306–1309. [Google Scholar] [CrossRef]
- Giesecke, C.; Frölich, D.; Reiter, K.; Mei, H.E.; Wirries, I.; Kuhly, R.; Killig, M.; Glatzer, T.; Stölzel, K.; Perka, C.; et al. Tissue Distribution and Dependence of Responsiveness of Human Antigen-Specific Memory B Cells. J. Immunol. 2014, 192, 3091–3100. [Google Scholar] [CrossRef] [PubMed]
- Franz, B.; May, K.F.; Dranoff, G.; Wucherpfennig, K. Ex vivo characterization and isolation of rare memory B cells with antigen tetramers. Blood 2011, 118, 348–357. [Google Scholar] [CrossRef] [PubMed]
- Blanco, E.; Pérez-Andrés, M.; Arriba-Mendez, S.; Contreras-SanFeliciano, T.; Criado, I.; Pelák, O.; Serra-Caetano, A.; Romero, A.; Puig, N.; Remesal, A.; et al. Age-associated distribution of normal B-cell and plasma cell subsets in peripheral blood. J. Allergy Clin. Immunol. 2018, 141, 2208–2219. [Google Scholar] [CrossRef] [PubMed]
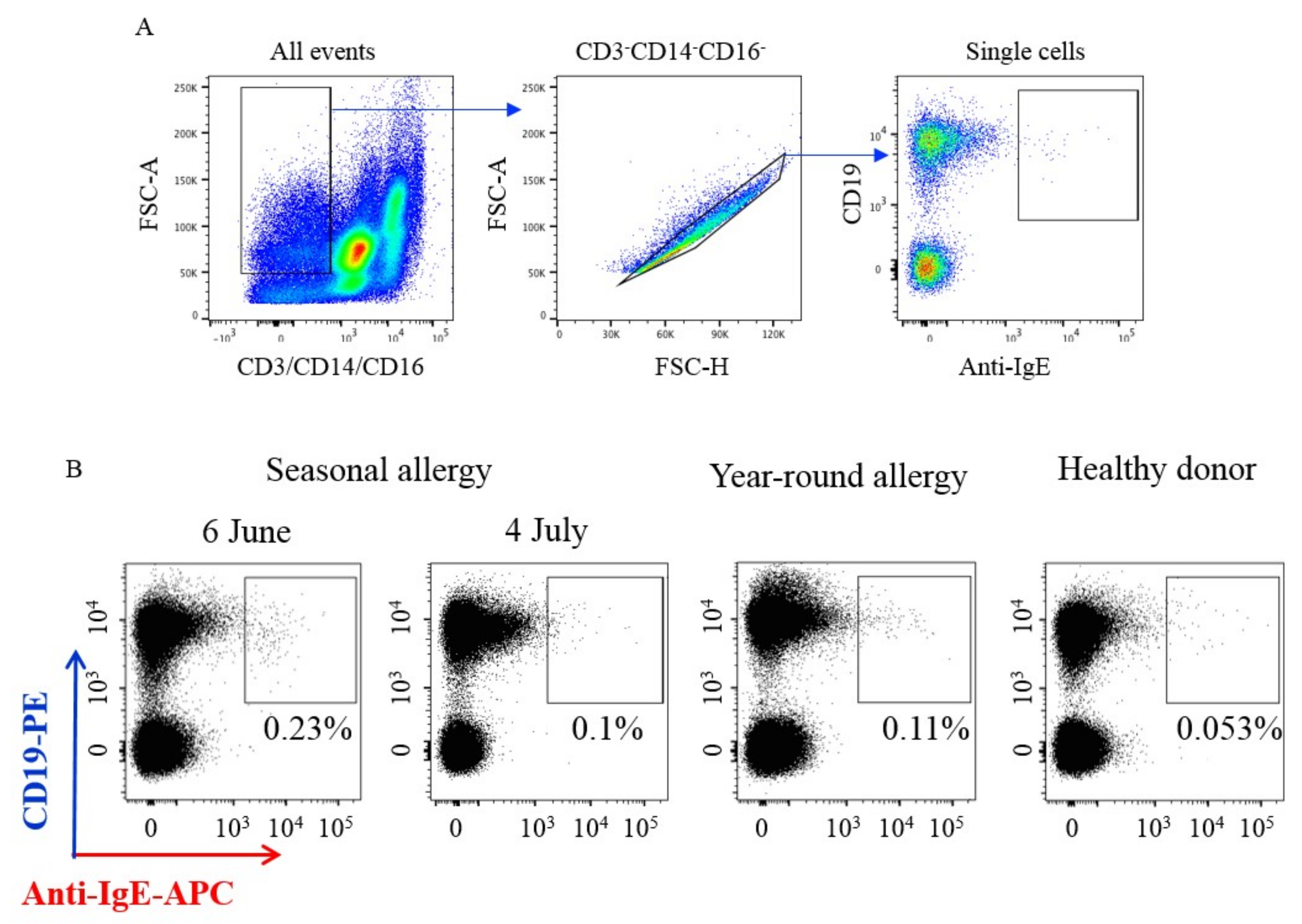
- Oliveria, J.P.; Salter, B.M.; MacLean, J.; Kotwal, S.; Smith, A.; Harris, J.M.; Scheerens, H.; Sehmi, R.; Gauvreau, G.M. Increased IgE+ B cells in sputum, but not blood, bone marrow, or tonsils, after inhaled allergen challenge in subjects with asthma. Am. J. Respir. Crit. Care Med. 2017, 196, 107–109. [Google Scholar] [CrossRef] [PubMed]
- Hedley, B.D.; Keeney, M. Technical issues: Flow cytometry and rare event analysis. Int. J. Lab. Hematol. 2013, 35, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Tangye, S.G.; Liu, Y.-J.; Aversa, G.; Phillips, J.H.; De Vries, J.E. Identification of Functional Human Splenic Memory B Cells by Expression of CD148 and CD27. J. Exp. Med. 1998, 188, 1691–1703. [Google Scholar] [CrossRef]
- Ellyard, J.I.; Avery, D.T.; Phan, T.G.; Hare, N.J.; Hodgkin, P.D.; Tangye, S.G. Antigen-selected, immunoglobulin-secreting cells persist in human spleen and bone marrow. Blood 2004, 103, 3805–3812. [Google Scholar] [CrossRef]
- Martínez-Gamboa, L.; Mei, H.; Loddenkemper, C.; Ballmer, B.; Hansen, A.; Lipsky, P.E.; Emmerich, F.; Radbruch, A.; Salama, A.; Dorner, T. Role of the spleen in peripheral memory B-cell homeostasis in patients with autoimmune thrombocytopenia purpura. Clin. Immunol. 2009, 130, 199–212. [Google Scholar] [CrossRef]
- Kleinjan, A.; Vinke, J.; Severijnen, L.; Fokkens, W. Local production and detection of (specific) IgE in nasal B-cells and plasma cells of allergic rhinitis patients. Eur. Respir. J. 2000, 15, 491–497. [Google Scholar] [CrossRef]
- Cameron, L.; Hamid, Q.; Wright, E.; Nakamura, Y.; Christodoulopoulos, P.; Muro, S.; Frenkiel, S.; Lavigne, F.; Durham, S.; Gould, H. Local synthesis of epsilon germline gene transcripts, IL-4, and IL-13 in allergic nasal mucosa after ex vivo allergen exposure. J. Allergy Clin. Immunol. 2000, 106, 46–52. [Google Scholar] [CrossRef]
- Durham, S.R.; Gould, H.J.; Thienes, C.P.; Jacobson, M.R.; Masuyama, K.; Rak, S.; Lowhagen, O.; Schotman, E.; Cameron, L.; Hamid, Q.A. Expression of ε germ-line gene transcripts and mrna for the ε heavy chain of IgE in nasal B cells and the effects of topical corticosteroid. Eur. J. Immunol. 1997, 27, 2899–2906. [Google Scholar] [CrossRef] [PubMed]
- Papatziamos, G.; van der Ploeg, I.; Hemlin, C.; Patwardhan, A.; Scheynius, A. Increased occurrence of IgE+ and FcεRI+ cells in adenoids from atopic children. Allergy 1999, 54, 916–925. [Google Scholar] [CrossRef] [PubMed]
- Papatziamos, G.; Van Hage-Hamsten, M.; Lundahl, J.; Hemlin, C. IgE-positive plasma cells are present in adenoids of atopic children. Acta Oto-Laryngol. 2006, 126, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.-Y.; Choi, S.-J.; Hur, G.-Y.; Lee, K.-H.; Kim, S.-W.; Cho, J.-S.; Park, H.-S. Local production of total IgE and specific antibodies to the house dust mite in adenoid tissue. Pediatr. Allergy Immunol. 2009, 20, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Ganzer, U.; Bachert, C. Localization of IgE synthesis in immediate-type allergy of the upper respiratory tract. ORL 1988, 50, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Snow, R.E.; Djukanovic, R.; Stevenson, F.K. Analysis of immunoglobulin E VH transcripts in a bronchial biopsy of an asthmatic patient confirms bias towards VH5, and indicates local clonal expansion, somatic mutation and isotype switch events. Immunology 1999, 98, 646–651. [Google Scholar] [CrossRef] [PubMed]
- Nahm, D.H.; Park, H.-S. Analysis of induced sputum for studying allergen-specific IgE antibodies in airway secretion from asthmatic patients. Clin. Exp. Allergy 1998, 28, 686–693. [Google Scholar] [CrossRef] [PubMed]
- Takhar, P.; Corrigan, C.J.; Smurthwaite, L.; O’Connor, B.J.; Durham, S.R.; Lee, T.H.; Gould, H.J. Class switch recombination to IgE in the bronchial mucosa of atopic and nonatopic patients with asthma. J. Allergy Clin. Immunol. 2007, 119, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.-D.; Tokoyoda, K.; Radbruch, A. Immunological memories of the bone marrow. Immunol. Rev. 2018, 283, 86–98. [Google Scholar] [CrossRef]
- Paramithiotis, E.; Cooper, M.D. Memory B lymphocytes migrate to bone marrow in humans. Proc. Natl. Acad. Sci. USA 1997, 94, 208–212. [Google Scholar] [CrossRef]
- Slifka, M.K.; Antia, R.; Whitmire, J.K.; Ahmed, R. Humoral Immunity Due to Long-Lived Plasma Cells. Immunity 1998, 8, 363–372. [Google Scholar] [CrossRef]
- Mamani-Matsuda, M.; Cosma, A.; Weller, S.; Faili, A.; Staib, C.; Garçon, L.; Hermine, O.; Beyne-Rauzy, O.; Fieschi, C.; Pers, J.-O.; et al. The human spleen is a major reservoir for long-lived vaccinia virus-specific memory B cells. Blood 2008, 111, 4653–4659. [Google Scholar] [CrossRef] [PubMed]
- Snow, R.E.; Chapman, C.J.; Frew, A.J.; Holgate, S.T.; Stevenson, F.K. Analysis of Ig VH region genes encoding IgE antibodies in splenic B lymphocytes of a patient with asthma. J. Immunol. 1995, 154, 5576–5581. [Google Scholar] [PubMed]
- Bellou, A.; Kanny, G.; Fremont, S.; Moneret-Vautrin, D. Transfer of Atopy Following Bone Marrow Transplantation. Ann. Allergy Asthma Immunol. 1997, 78, 513–516. [Google Scholar] [CrossRef]
- Walker, S.A.; Riches, P.G.; Wild, G.; Ward, A.M.; Shaw, P.J.; Desai, S.; Hobbs, J.R. Total and allergen-specific IgE in relation to allergic response pattern following bone marrow transplantation. Clin. Exp. Immunol. 1986, 66, 633–639. [Google Scholar] [PubMed]
- Desai, S.; Walker, S.; Shaw, P.; Riches, P.; Hobbs, J.; Wild, G.; Harper, J. Expression of Donor Allergic Response Patterns by Bone Marrow Transplant Recipients. Lancet 1984, 324, 1148. [Google Scholar] [CrossRef]
- Saarinen, U.M. Transfer of latent atopy by bone marrow transplantation? A case report. J. Allergy Clin. Immunol. 1984, 74, 196–200. [Google Scholar] [CrossRef]
- Agosti, J.M.; Sprenger, J.D.; Lum, L.G.; Witherspoon, R.P.; Fisher, L.D.; Storb, R.; Henderson, W.R. Transfer of Allergen-Specific IgE-Mediated Hypersensitivity with Allogeneic Bone Marrow Transplantation. N. Engl. J. Med. 1988, 319, 1623–1628. [Google Scholar] [CrossRef]
- Hallstrand, T.S.; Sprenger, J.D.; Agosti, J.M.; Longton, G.M.; Witherspoon, R.P.; Henderson, W.R. Long-term acquisition of allergen-specific IgE and asthma following allogeneic bone marrow transplantation from allergic donors. Blood 2004, 104, 3086–3090. [Google Scholar] [CrossRef]
- Storek, J.; Vliagoftis, H.; Grizel, A.; Lyon, A.W.; Daly, A.; Khan, F.; Bowen, T.; Game, M.; Larratt, L.; Turner, R.; et al. Allergy transfer with hematopoietic cell transplantation from an unrelated donor. Bone Marrow Transpl. 2011, 46, 605–606. [Google Scholar] [CrossRef]
- Kubo, M. Mast cells and basophils in allergic inflammation. Curr. Opin. Immunol. 2018, 54, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Varricchi, G.; Raap, U.; Rivellese, F.; Marone, G.; Gibbs, B.F. Human mast cells and basophils-How are they similar how are they different? Immunol. Rev. 2018, 282, 8–34. [Google Scholar] [CrossRef] [PubMed]
- Geha, R.S.; Helm, B.; Gould, H. Inhibition of the Prausnitz–Küstner reaction by an immunoglobulin ε-chain fragment synthesized in E. coli. Nature 1985, 315, 577–578. [Google Scholar] [CrossRef] [PubMed]
- Borkowski, T.A.; Jouvin, M.-H.; Lin, S.-Y.; Kinet, J.-P. Minimal Requirements for IgE-Mediated Regulation of Surface FcεRI. J. Immunol. 2001, 167, 1290–1296. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Hong, S.-C.; Lee, H.-M.; Cho, J.H.; Suh, J.D.; Park, I.-H. Correlation between Skin-prick Testing, Individual Specific IgE Tests, and a Multiallergen IgE Assay for Allergy Detection in Patients with Chronic Rhinitis. Am. J. Rhinol. Allergy 2014, 28, 388–391. [Google Scholar]
- Finnerty, J.; Summerell, S.; Holgate, S. Relationship between skin-prick tests, the multiple allergosorbent test and symptoms of allergic disease. Clin. Exp. Allergy 1989, 19, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Naclerio, R.M.; Adkinson, N.F.; Moylan, B.; Baroody, F.M.; Proud, D.; Kagey-Sobotka, A.; Lichtenstein, L.M.; Hamilton, R. Nasal provocation with allergen induces a secondary serum IgE antibody response. J. Allergy Clin. Immunol. 1997, 100, 505–510. [Google Scholar] [CrossRef]
- Eckl-Dorna, J.; Froschl, R.; Lupinek, C.; Kiss, R.; Gattinger, P.; Marth, K.; Campana, R.; Mittermann, I.; Blatt, K.; Valent, P.; et al. Intranasal administration of allergen increases specific IgE whereas intranasal omalizumab does not increase serum IgE levels-a pilot study. Allergy 2018, 73, 1003–1012. [Google Scholar] [CrossRef]
- Lin, H.; Boesel, K.M.; Griffith, D.T.; Prussin, C.; Foster, B.; Romero, F.; Townley, R.; Casale, T.B. Omalizumab rapidly decreases nasal allergic response and FcεRI on basophils. J. Allergy Clin. Immunol. 2004, 113, 297–302. [Google Scholar] [CrossRef]
- Holgate, S.; Casale, T.; Wenzel, S.; Bousquet, J.; Deniz, Y.; Reisner, C. The anti-inflammatory effects of omalizumab confirm the central role of IgE in allergic inflammation. J. Allergy Clin. Immunol. 2005, 115, 459–465. [Google Scholar] [CrossRef]
- MacGlashan, D.W., Jr.; Bochner, B.S.; Adelman, D.C.; Jardieu, P.M.; Togias, A.; McKenzie-White, J.; Sterbinsky, S.A.; Hamilton, R.G.; Lichtenstein, L.M. Down-regulation of FcεRI expression on human basophils during in vivo treatment of atopic patients with anti-IgE antibody. J. Immunol. 1997, 158, 1438–1445. [Google Scholar] [PubMed]
- Noga, O.; Hanf, G.; Kunkel, G.; Kleine-Tebbe, J. Basophil histamine release decreases during omalizumab therapy in allergic asthmatics. Int. Arch. Allergy Immunol. 2008, 146, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Saini, S.S.; MacGlashan, D.W., Jr.; Sterbinsky, S.A.; Togias, A.; Adelman, D.C.; Lichtenstein, L.M.; Bochner, B.S. Down-regulation of human basophil IgE and FcεRIα surface densities and mediator release by anti-IgE-infusions is reversible in vitro and in vivo. J. Immunol. 1999, 162, 5624–5630. [Google Scholar] [PubMed]
- Noga, O.; Hanf, G.; Kunkel, G. Immunological and Clinical Changes in Allergic Asthmatics following Treatment with Omalizumab. Int. Arch. Allergy Immunol. 2003, 131, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Campana, R.; Moritz, K.; Marth, K.; Neubauer, A.; Huber, H.; Henning, R.; Blatt, K.; Hoermann, G.; Brodie, T.M.; Kaider, A.; et al. Frequent occurrence of T cell-mediated late reactions revealed by atopy patch testing with hypoallergenic rBet v 1 fragments. J. Allergy Clin. Immunol. 2016, 137, 601–609. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pirron, U.; Schlunck, T.; Prinz, J.C.; Rieber, E.P. IgE-dependent antigen focusing by human B lymphocytes is mediated by the low-affinity receptor for IgE. Eur. J. Immunol. 1990, 20, 1547–1551. [Google Scholar] [CrossRef] [PubMed]
- Wilcock, L.K.; Francis, J.N.; Durham, S.R. IgE-facilitated antigen presentation: Role in allergy and the influence of allergen immunotherapy. Immunol. Allergy Clin. N. Am. 2006, 26, 333–347. [Google Scholar] [CrossRef]
- Ding, Z.; Dahlin, J.S.; Xu, H.; Heyman, B. IgE-mediated enhancement of CD4+ T cell responses requires antigen presentation by CD8α− conventional dendritic cells. Sci. Rep. 2016, 6, 28290. [Google Scholar] [CrossRef]
- Engeroff, P.; Fellmann, M.; Yerly, D.; Bachmann, M.F.; Vogel, M. A novel recycling mechanism of native IgE-antigen complexes in human B cells facilitates the transfer of antigen to dendritic cells for antigen presentation. J. Allergy Clin. Immunol. 2018, 142, 557–568. [Google Scholar] [CrossRef]
- Tu, Y.; Salim, S.; Bourgeois, J.; Di Leo, V.; Irvine, E.J.; Marshall, J.K.; Perdue, M.H. CD23-Mediated IgE Transport Across Human Intestinal Epithelium: Inhibition by Blocking Sites of Translation or Binding. Gastroenterology 2005, 129, 928–940. [Google Scholar] [CrossRef]
- Palaniyandi, S.; Tomei, E.; Li, Z.; Conrad, D.H.; Zhu, X.; Wang, S.; Zhang, K.; Stepkowski, S.M.; Li, J.; Chen, G.; et al. CD23-Dependent Transcytosis of IgE and Immune Complex across the Polarized Human Respiratory Epithelial Cells. J. Immunol. 2011, 186, 3484–3496. [Google Scholar] [CrossRef] [PubMed]
- Hjelm, F.; Karlsson, M.C.I.; Heyman, B. A novel B cell-mediated transport of IgE-immune complexes to the follicle of the spleen. J. Immunol. 2008, 180, 6604–6610. [Google Scholar] [CrossRef] [PubMed]
- Van Der Heijden, F.L.; Van Neerven, R.J.; Kapsenberg, M.L. Relationship between facilitated allergen presentation and the presence of allergen-specific IgE in serum of atopic patients. Clin. Exp. Immunol. 1995, 99, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Poole, J.A.; Meng, J.; Reff, M.; Spellman, M.C.; Rosenwasser, L.J. Anti-CD23 monoclonal antibody, lumiliximab, inhibited allergen-induced responses in antigen-presenting cells and T cells from atopic subjects. J. Allergy Clin. Immunol. 2005, 116, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Van Neerven, R.J.; Wikborg, T.; Lund, G.; Jacobsen, B.; Brinch-Nielsen, A.; Arnved, J.; Ipsen, H. Blocking antibodies induced by specific allergy vaccination prevent the activation of CD4+ T cells by inhibiting serum-IgE-facilitated allergen presentation. J. Immunol. 1999, 163, 2944–2952. [Google Scholar] [PubMed]
- Wachholz, P.A.; Soni, N.K.; Till, S.J.; Durham, S.R. Inhibition of allergen-IgE binding to B cells by IgG antibodies after grass pollen immunotherapy. J. Allergy Clin. Immunol. 2003, 112, 915–922. [Google Scholar] [CrossRef]
- Van Neerven, R.J.J.; Arvidsson, M.; Ipsen, H.; Sparholt, S.H.; Rak, S.; Würtzen, P. A double-blind, placebo-controlled birch allergy vaccination study: Inhibition of CD23-mediated serum-immunoglobulin E-facilitated allergen presentation. Clin. Exp. Allergy 2004, 34, 420–428. [Google Scholar] [CrossRef]
- Shamji, M.H.; Wilcock, L.K.; Wachholz, P.A.; Dearman, R.J.; Kimber, I.; Würtzen, P.A.; Larché, M.; Durham, S.R.; Francis, J.N. The IgE-facilitated allergen binding (FAB) assay: Validation of a novel flow-cytometric based method for the detection of inhibitory antibody responses. J. Immunol. Methods 2006, 317, 71–79. [Google Scholar] [CrossRef]
- Würtzen, P.A.; Lund, G.; Lund, K.; Arvidsson, M.; Rak, S.; Ipsen, H. A double-blind placebo-controlled birch allergy vaccination study II: Correlation between inhibition of IgE binding, histamine release and facilitated allergen presentation. Clin. Exp. Allergy 2008, 38, 1290–1301. [Google Scholar] [CrossRef]
- Pree, I.; Shamji, M.H.; Kimber, I.; Valenta, R.; Durham, S.R.; Niederberger, V. Inhibition of CD23-dependent facilitated allergen binding to B cells following vaccination with genetically modified hypoallergenic Bet v 1 molecules. Clin. Exp. Allergy 2010, 40, 1346–1352. [Google Scholar] [CrossRef]
- Yu, P.; Kosco-Vilbois, M.; Richards, M.; Köhler, G.; Lamers, M.C. Negative feedback regulation of IgE synthesis by murine CD23. Nature 1994, 369, 753–756. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.E.; Wang, Z.; Locksley, R.M. Murine B cells regulate serum IgE levels in a CD23-dependent manner1. J. Immunol. 2010, 185, 5040–5047. [Google Scholar] [CrossRef] [PubMed]
- Reginald, K.; Eckl-Dorna, J.; Zafred, D.; Focke-Tejkl, M.; Lupinek, C.; Niederberger, V.; Keller, W.; Valenta, R. Different modes of IgE binding to CD23 revealed with major birch allergen, Bet v 1-specific monoclonal IgE. Immunol. Cell Biol. 2013, 91, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Holm, J.; Willumsen, N.; Würtzen, P.A.; Christensen, L.H.; Lund, K. Facilitated antigen presentation and its inhibition by blocking IgG antibodies depends on IgE repertoire complexity. J. Allergy Clin. Immunol. 2011, 127, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Vangelista, L.; Laffer, S.; Turek, R.; Gronlund, H.; Sperr, W.R.; Valent, P.; Pastore, A.; Valenta, R. The immunoglobulin-like modules Cε3 and α2 are the minimal units necessary for human IgE-FcεRI interaction. J. Clin. Investig. 1999, 103, 1571–1578. [Google Scholar] [CrossRef]
- Garman, S.C.; Wurzburg, B.A.; Tarchevskaya, S.S.; Kinet, J.-P.; Jardetzky, T.S. Structure of the Fc fragment of human IgE bound to its high-affinity receptor FcεRIα. Nature 2000, 406, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Jardieu, P. Anti-IgE therapy. Curr. Opin. Immunol. 1995, 7, 779–782. [Google Scholar] [CrossRef]
- Heusser, C. Therapeutic potential of anti-IgE antibodies. Curr. Opin. Immunol. 1997, 9, 805–813. [Google Scholar] [CrossRef]
- Stadler, B.M.; Rudolf, M.P.; Zurcher, A.W.; Miescher, S.; Vogel, M. Anti-IgE in allergic sensitization. Immunol. Cell Biol. 1996, 74, 195–200. [Google Scholar] [CrossRef]
- Segal, M.; Stokes, J.R.; Casale, T.B. Anti-immunoglobulin E therapy. World Allergy Organ. J. 2008, 1, 174–183. [Google Scholar] [CrossRef]
- Chan, M.A.; Gigliotti, N.M.; Dotson, A.L.; Rosenwasser, L.J. Omalizumab may decrease IgE synthesis by targeting membrane IgE+ human B cells. Clin. Transl. Allergy 2013, 3, 29. [Google Scholar] [CrossRef] [PubMed]
- Güzelbey, O.; Ardelean, E.; Magerl, M.; Zuberbier, T.; Maurer, M.; Metz, M. Successful treatment of solar urticaria with anti-immunoglobulin E therapy. Allergy 2008, 63, 1563–1565. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Lin, B.; Li, J.; Qian, W.; Hou, S.; Zhang, D.; Kou, G.; Li, B.; Wang, H.; Chen, Y.; et al. Tolerability, pharmacokinetics and pharmacodynamics of CMAB007, a humanized anti-immunoglobulin E monoclonal antibody, in healthy chinese subjects. mAbs 2012, 4, 110–119. [Google Scholar] [CrossRef]
- Gauvreau, G.M.; Arm, J.P.; Boulet, L.-P.; Leigh, R.; Cockcroft, D.W.; Davis, B.E.; Mayers, I.; Fitzgerald, J.M.; Dahlen, B.; Killian, K.J.; et al. Efficacy and safety of multiple doses of QGE031 (ligelizumab) versus omalizumab and placebo in inhibiting allergen-induced early asthmatic responses. J. Allergy Clin. Immunol. 2016, 138, 1051–1059. [Google Scholar] [CrossRef] [PubMed]
- Cohen, E.S.; Dobson, C.L.; Käck, H.; Wang, B.; Sims, D.A.; Lloyd, C.O.; England, E.; Rees, D.G.; Guo, H.; Karagiannis, S.N.; et al. A novel IgE-neutralizing antibody for the treatment of severe uncontrolled asthma. mAbs 2014, 6, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, E.; Schwickart, M.; Li, J.; Kim, K.; Crouch, S.; Parveen, S.; Kell, C.; Birrell, C. Pharmacokinetics, Pharmacodynamics, and Safety of MEDI4212, an Anti-IgE Monoclonal Antibody, in Subjects with Atopy: A Phase I Study. Adv. Ther. 2016, 33, 225–251. [Google Scholar] [CrossRef]
- Eggel, A.; Baumann, M.J.; Amstutz, P.; Stadler, B.M.; Vogel, M. DARPins as Bispecific Receptor Antagonists Analyzed for Immunoglobulin E Receptor Blockage. J. Mol. Boil. 2009, 393, 598–607. [Google Scholar] [CrossRef]
- Eggel, A.; Baravalle, G.; Hobi, G.; Kim, B.; Buschor, P.; Forrer, P.; Shin, J.-S.; Vogel, M.; Stadler, B.M.; Dahinden, C.A.; et al. Accelerated dissociation of IgE-FcεRI complexes by disruptive inhibitors actively desensitizes allergic effector cells. J. Allergy Clin. Immunol. 2014, 133, 1709–1719. [Google Scholar] [CrossRef]
- Zellweger, F.; Gasser, P.; Brigger, D.; Buschor, P.; Vogel, M.; Eggel, A. A novel bispecific darpin targeting FcγRIIB and FcεRI-bound IgE inhibits allergic responses. Allergy 2017, 72, 1174–1183. [Google Scholar] [CrossRef]
- Hellman, L. Profound reduction in allergen sensitivity following treatment with a novel allergy vaccine. Eur. J. Immunol. 1994, 24, 415–420. [Google Scholar] [CrossRef]
- Peng, Z.; Liu, Q.; Wang, Q.; Rector, E.; Ma, Y.; Warrington, R. Novel IgE peptide-based vaccine prevents the increase of IgE and down-regulates elevated IgE in rodents. Clin. Exp. Allergy 2007, 37, 1040–1048. [Google Scholar] [CrossRef] [PubMed]
- Sethi, G.; Ahn, K.S.; Pandey, M.K.; Aggarwal, B.B. Celastrol, a novel triterpene, potentiates TNF-induced apoptosis and suppresses invasion of tumor cells by inhibiting NF-κB regulated gene products and tak1-mediated NF-κB activation. Blood 2007, 109, 2727–2735. [Google Scholar] [PubMed]
- Brightbill, H.D.; Jeet, S.; Lin, Z.; Yan, D.; Zhou, M.; Tan, M.; Nguyen, A.; Yeh, S.; Delarosa, D.; Leong, S.R.; et al. Antibodies specific for a segment of human membrane IgE deplete IgE-producing B cells in humanized mice. J. Clin. Investig. 2010, 120, 2218–2229. [Google Scholar] [CrossRef] [PubMed]
- Gauvreau, G.M.; Harris, J.M.; Boulet, L.P.; Scheerens, H.; Fitzgerald, J.M.; Putnam, W.S.; Cockcroft, D.W.; Davis, B.E.; Leigh, R.; Zheng, Y.; et al. Targeting membrane-expressed IgE B cell receptor with an antibody to the M1 prime epitope reduces IgE production. Sci. Transl. Med. 2014, 6, 243ra85. [Google Scholar] [CrossRef] [PubMed]
- Scheerens, H.; Samineni, D.; Cochran, C.; Staubach, P.; Metz, M.; Sussman, G.; Maurer, M.; Harris, J.M.; Cabanski, C.R.; Bradley, M.S. A randomized trial of quilizumab in adults with refractory chronic spontaneous urticaria. J. Allergy Clin. Immunol. 2016, 138, 1730–1732. [Google Scholar]
- Kirak, O.; Riethmuller, G. A novel, nonanaphylactogenic, bispecific IgE-CD3 antibody eliminates IgE+ B cells. J. Allergy Clin. Immunol. 2015, 136, 800–802. [Google Scholar] [CrossRef] [PubMed]
- Rudolf, M.P.; Zuercher, A.W.; Nechansky, A.; Ruf, C.; Vogel, M.; Miescher, S.M.; Stadler, B.M.; Kricek, F. Molecular Basis for Nonanaphylactogenicity of a Monoclonal Anti-IgE Antibody. J. Immunol. 2000, 165, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Nyborg, A.C.; Zacco, A.; Ettinger, R.; Jack Borrok, M.; Zhu, J.; Martin, T.; Woods, R.; Kiefer, C.; Bowen, M.A.; Suzanne Cohen, E.; et al. Development of an antibody that neutralizes soluble IgE and eliminates IgE expressing B cells. Cell. Mol. Immunol. 2016, 13, 391–400. [Google Scholar] [CrossRef]
- Chu, S.Y.; Horton, H.M.; Pong, E.; Leung, I.W.; Chen, H.; Nguyen, D.-H.; Bautista, C.; Muchhal, U.S.; Bernett, M.J.; Moore, G.L.; et al. Reduction of total IgE by targeted coengagement of IgE B-cell receptor and FcγRIIb with Fc-engineered antibody. J. Allergy Clin. Immunol. 2012, 129, 1102–1115. [Google Scholar] [CrossRef]
- Morita, H.; Tamari, M.; Motomura, K.; Koezuka, Y.; Ichien, G.; Matsumoto, K.; Ishizaka, K.; Saito, H. IgE-class-specific immunosuppression in offspring by administraton of anti-IgE to pregnant mice. J. Allergy Clin. Immunol. 2019, 143, 1261–1264. [Google Scholar] [CrossRef]
| Mice | Humans | |
|---|---|---|
| Genetic background | Inbred | Outbred |
| Percentage lymphocytes of total leukocytes | 75–90% [39] | 30–50% [40] |
| IgG subclasses | IgG1, IgG2, and IgG3 | IgG1, IgG2, IgG3, and IgG4 |
| IgE receptors on eosinophils | No FcεRI [41] | FcεRI [42] |
| Access to tissue for analysis | All tissues available | Limited access—mainly blood |
| Asthma development | Induced by sensitization, sometimes Th1-like | Induced by natural allergen exposure, mostly Th2-like |
| Allergy | Induced by sensitization [43,44,45] | Spontaneous by natural allergen exposure |
| IgE epitopes of respiratory allergens | Mainly sequential | Mainly conformational |
| T cell epitopes of respiratory allergens | Dominating T cell epitopes | High diversity |
| Cytokines required for IgE class switch | IL-4 [31] | IL-4, IL-13 [32] |
| Mechanisms of class-switch to IgE | Mainly sequential [33] | Evidence for sequential [46,47,48,49,50,51,52] and non-sequential [53] |
| Rise in Ig subtype in response to allergen challenge | IgE, IgG1 | IgE, IgG4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eckl-Dorna, J.; Villazala-Merino, S.; Campion, N.J.; Byazrova, M.; Filatov, A.; Kudlay, D.; Karsonova, A.; Riabova, K.; Khaitov, M.; Karaulov, A.; et al. Tracing IgE-Producing Cells in Allergic Patients. Cells 2019, 8, 994. https://doi.org/10.3390/cells8090994
Eckl-Dorna J, Villazala-Merino S, Campion NJ, Byazrova M, Filatov A, Kudlay D, Karsonova A, Riabova K, Khaitov M, Karaulov A, et al. Tracing IgE-Producing Cells in Allergic Patients. Cells. 2019; 8(9):994. https://doi.org/10.3390/cells8090994
Chicago/Turabian StyleEckl-Dorna, Julia, Sergio Villazala-Merino, Nicholas James Campion, Maria Byazrova, Alexander Filatov, Dmitry Kudlay, Antonina Karsonova, Ksenja Riabova, Musa Khaitov, Alexander Karaulov, and et al. 2019. "Tracing IgE-Producing Cells in Allergic Patients" Cells 8, no. 9: 994. https://doi.org/10.3390/cells8090994
APA StyleEckl-Dorna, J., Villazala-Merino, S., Campion, N. J., Byazrova, M., Filatov, A., Kudlay, D., Karsonova, A., Riabova, K., Khaitov, M., Karaulov, A., Niederberger-Leppin, V., & Valenta, R. (2019). Tracing IgE-Producing Cells in Allergic Patients. Cells, 8(9), 994. https://doi.org/10.3390/cells8090994





