Poc1B and Sas-6 Function Together during the Atypical Centriole Formation in Drosophila melanogaster
Abstract
:1. Introduction
2. Methods
2.1. Transgenic Flies
| Reference | Name in Text | Name in Ref | Description |
| [37] | Ana2GFP | pUbq-Ana2-GFP | Ana2 cDNA tagged at the C-terminus with GFP. Expressed from an ubiquitin promoter. |
| [37] | Sas-6GFP | pUbq-GFP-Sas6 | Sas-6 cDNA tagged at the N-terminus with GFP. Expressed from an ubiquitin promoter. |
| [6] | gPoc1BGFP | Poc1A/B-GFP | Poc1 gene tagged at the end of the last poc1B exon with GFP. Expressed from a poc1 promoter. |
| [6] | Poc1BGFP | UAS-Poc1B-GFP | Poc1B cDNA tagged at the C-terminus with GFP. Expressed from a UAS promoter. |
| [37] | Sas-6RFP | Ubq-RFP-Sas-6 | Sas-6 cDNA tagged at the N-terminus with RFP. Expressed from an ubiquitin promoter. |
| VDRC ID-108219 | Poc1RNAi | Poc1-RNAi | RNAi knockdown of Poc1RNAi |
| [6] | Poc1W87X | Poc1W87X | Poc1 truncation mutant |
| [25] | Sas-4GFP | Sas-4-GFP | Sas4 gene tagged at the end of the last exon with GFP. Expressed from a sas-4 promoter. |
| [38] | Bam-Gal4 | P{bam-GAL4:VP16} | GAL4 line expressing GAL4 from a Bam promoter. |
| Bloomington stock #8092 | Poc1 def | Df(3R)ED5066 | A chromosomal deletion that is also missing the poc1 gene |
| [39] | ana2 719 | ana2 | Ana2 truncation at R175 |
| Bloomington stock #12119 | sas-4s2214 | sas-4 | P-element insertion in the Sas-4 codding leading to a strong loss-of-function |
| [13] | Ana1GFP | Ana1-GFP | Ana1 gene tagged at the end of the last exon with GFP. Expressed from an Ana1 promoter |
| [13] | gSas-6GFP | gSas-6-GFP | Sas-6 gene tagged at the end of the last exon with GFP. Expressed from a Sas-6 promoter |
| [13] | Ana1tdtomato | Ana1-tdtomato | Ana1 gene tagged at the end of the last exon with Tdtomato. Expressed from an Ana1 promoter |
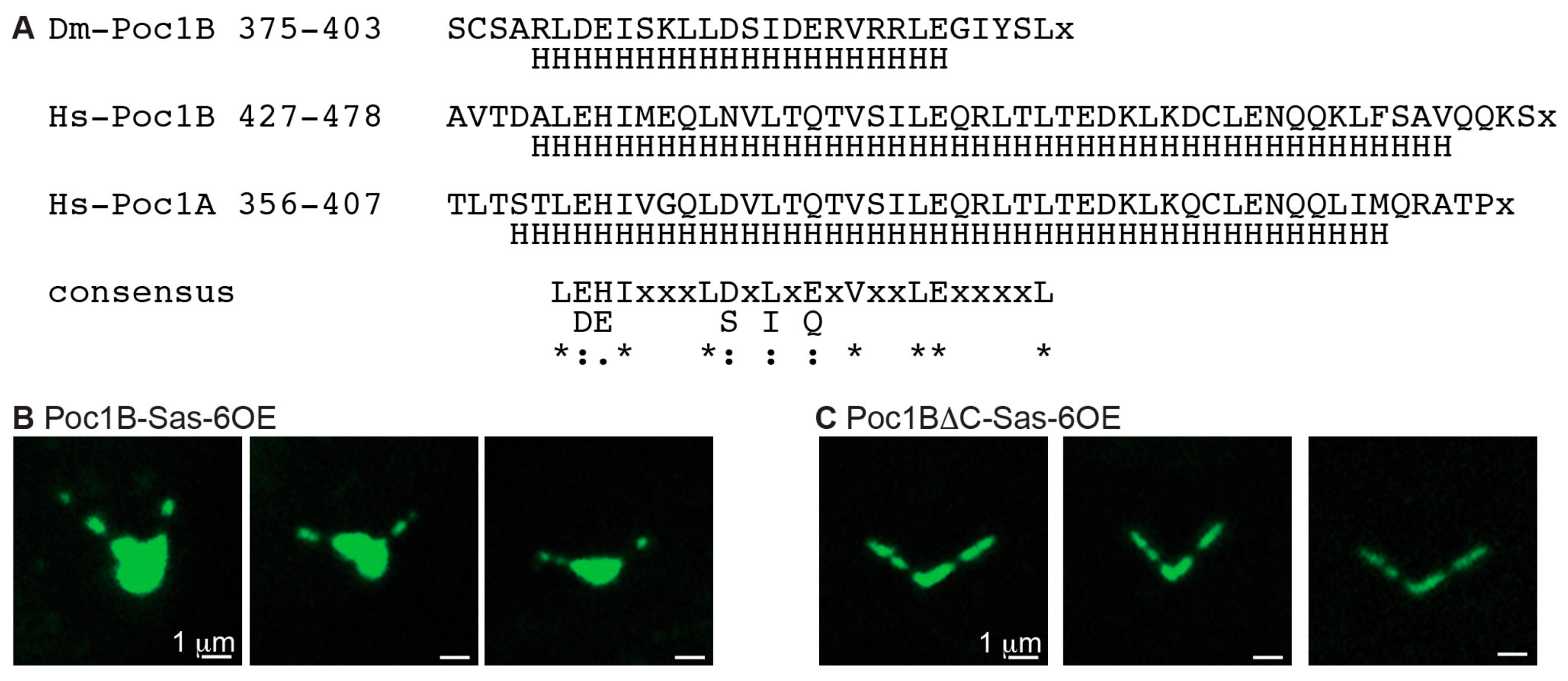
| This study | Poc1BΔCGFP | Poc1B cDNA lacking VLDVSIESGSDLCSCSARL DEISKLLDSIDERVRRLEGIYSL amino acid tagged at the C-terminus with GFP. Expressed from a UAS promoter. | |
| All fly stocks were cultured on standard media at 25 °C. | |||
2.2. Transgenic Flies Functional Test References
| Ubq-Ana2-GFP and Ubq-GFP-Sas-6 “The homo-oligomerisation of both Sas-6 and Ana2 is required for efficient centriole assembly in flies” [40] |
| gPoc1B-GFP and UAS-Poc1B-GFP “Centriole Remodeling during Spermiogenesis in Drosophila” [6] |
| gSas-6-GFP “Self-assembling SAS-6 Multimer Is a Core Centriole Building Block” [41] |
| Ana1-tdtomato “Drosophila Asterless the ortholog of vertebrate Cep152 is essential for centriole duplication” [13] |
2.3. Testes Fluorescence Microscopy
2.4. Antibodies
2.5. Tissue Processing and Transmission Electron Microscopy
2.6. Super-Resolution Microscopy
2.7. Statistical Methods
2.8. Reagent and Data Availability
3. Results
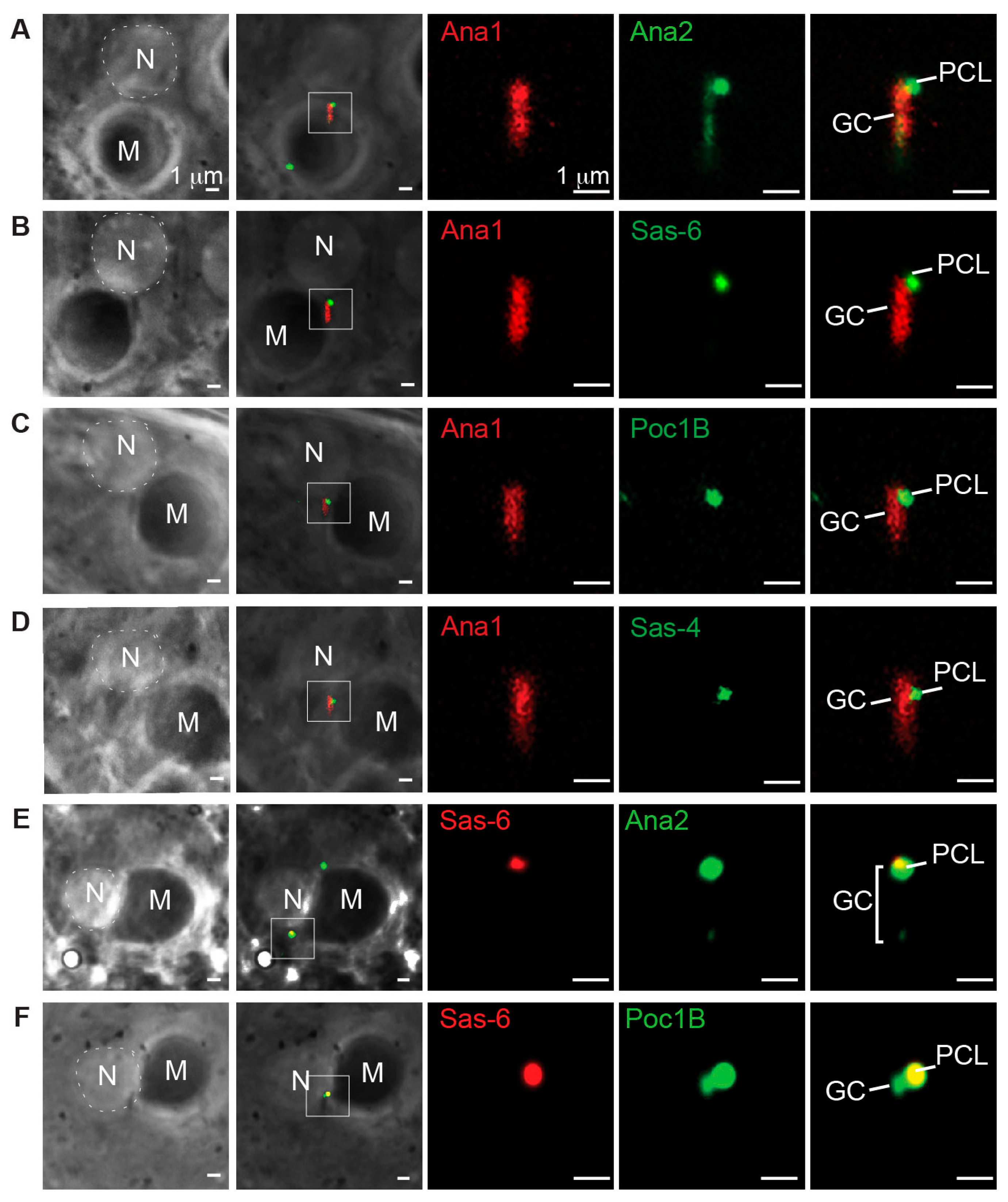
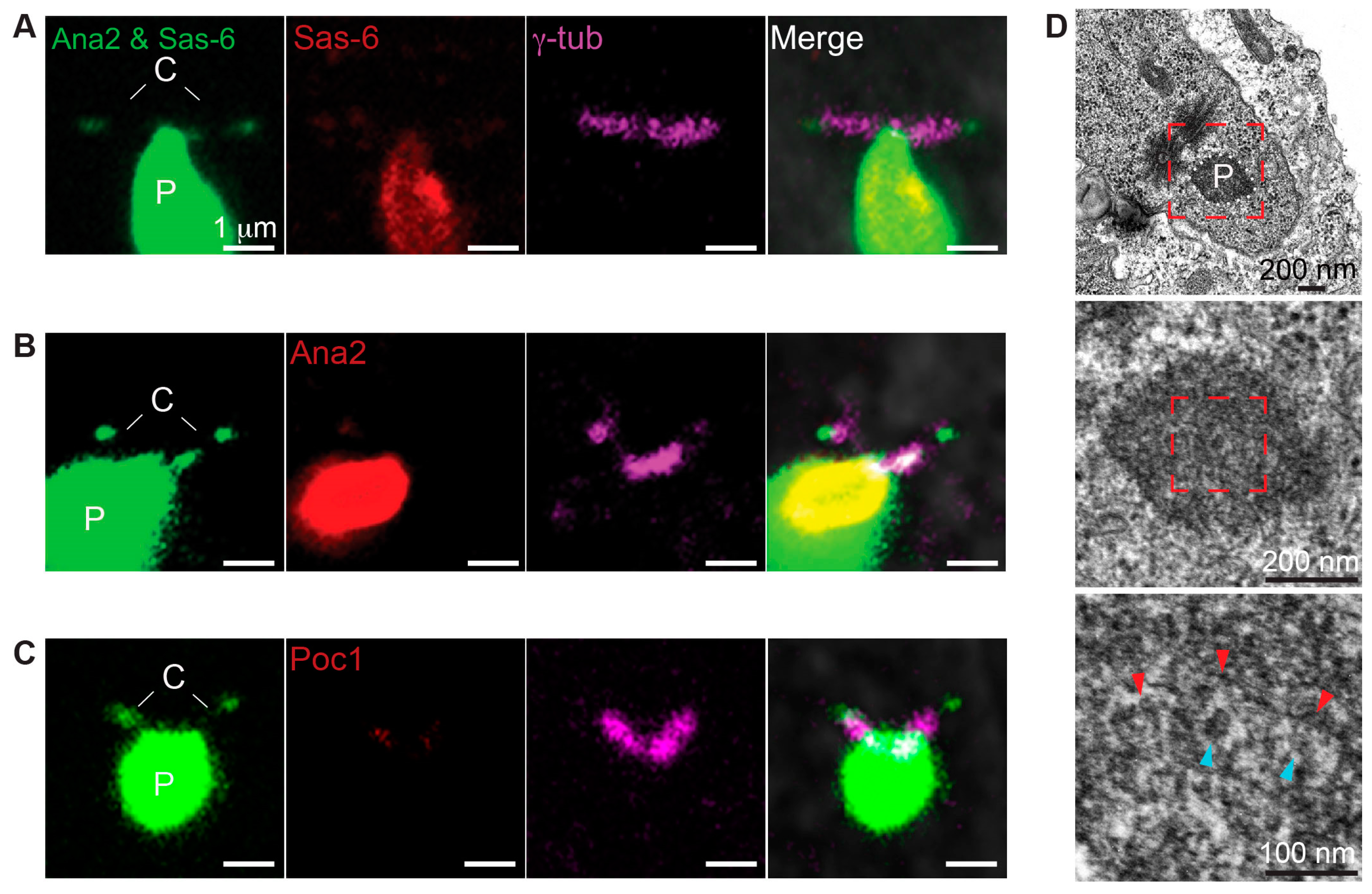
3.1. Poc1B Is Recruited during the Initial Stage of the PCL Formation
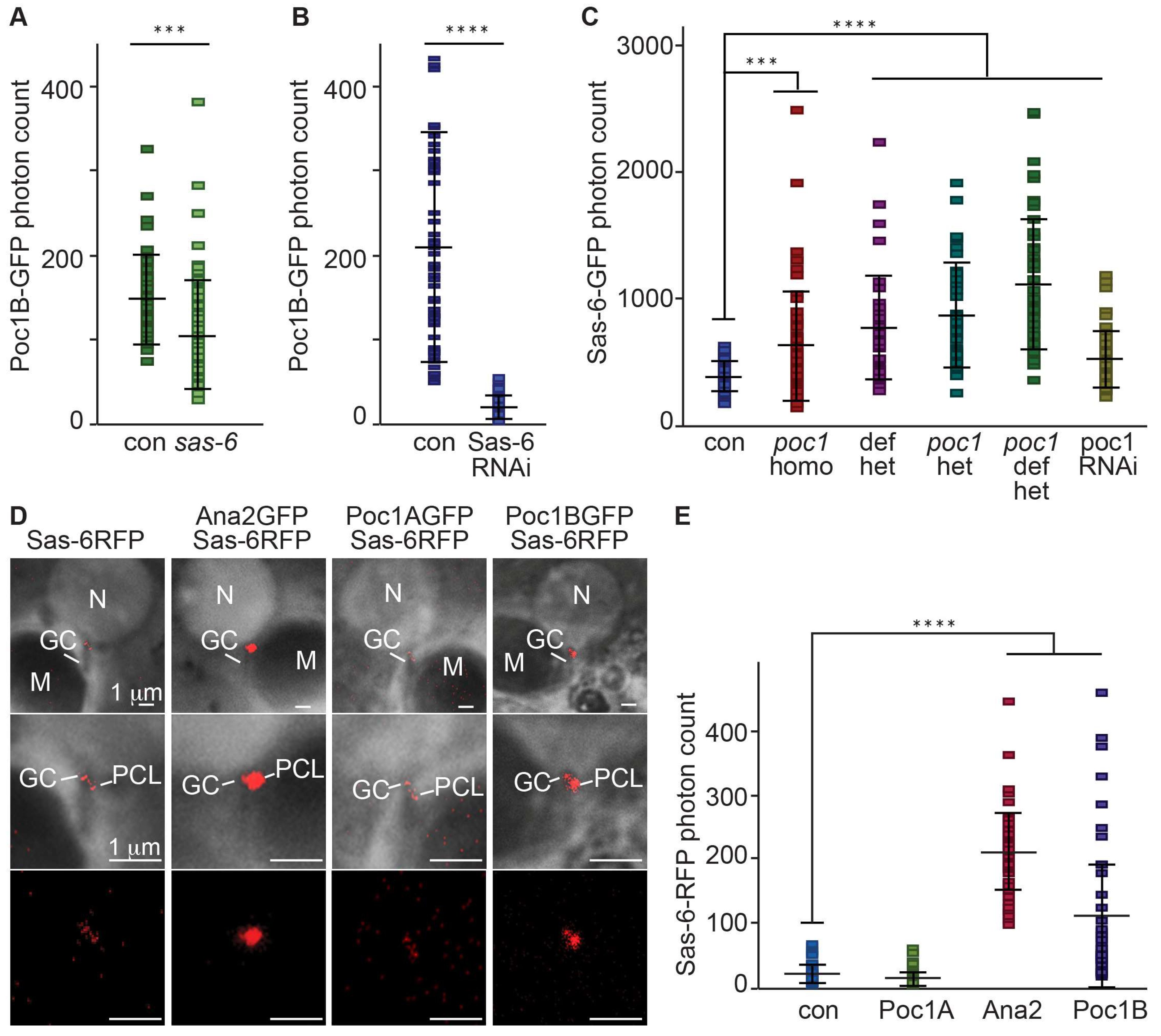
3.2. Poc1B Is Downstream to Ana-2 or Sas-6 during PCL Initiation
3.3. Poc1B Overexpression Induces Increase Cellular and PCL Sas-6 Level
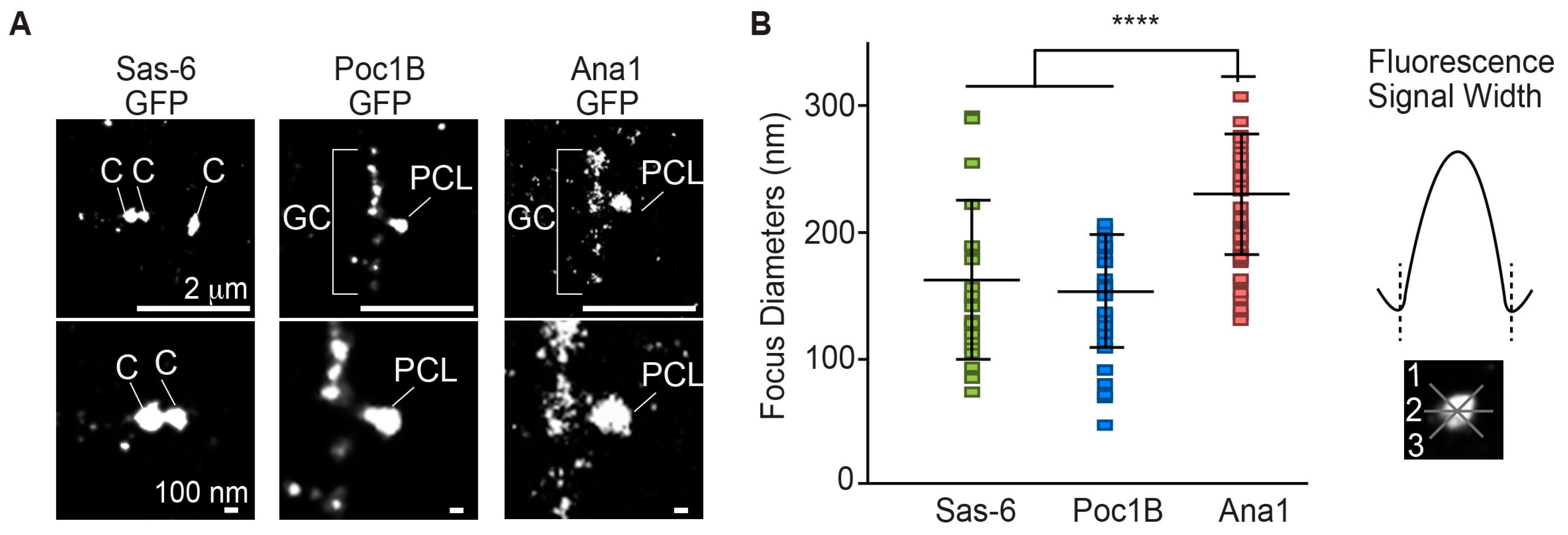
3.4. Poc1B and Sas-6 Are Localized in the PCL Core
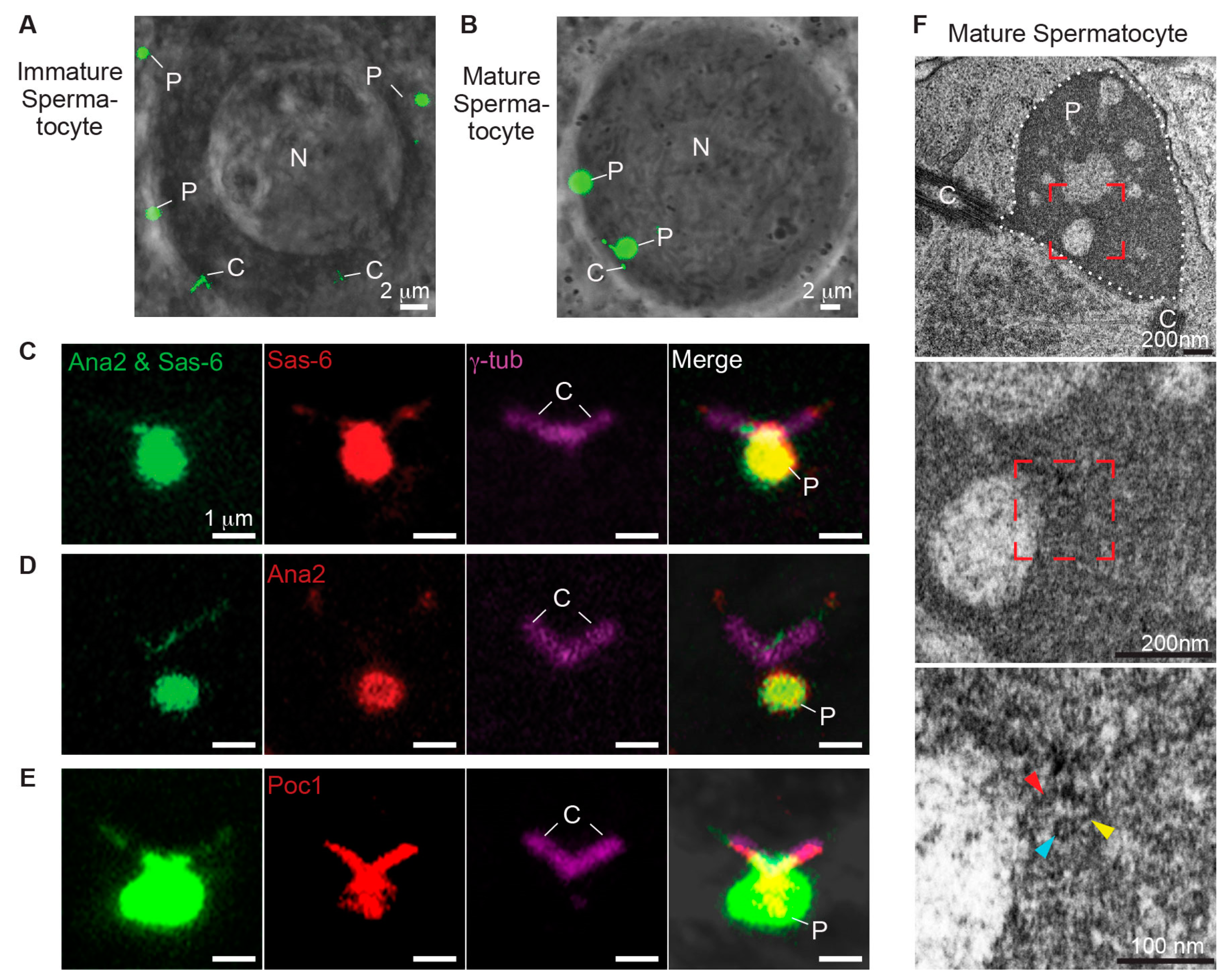
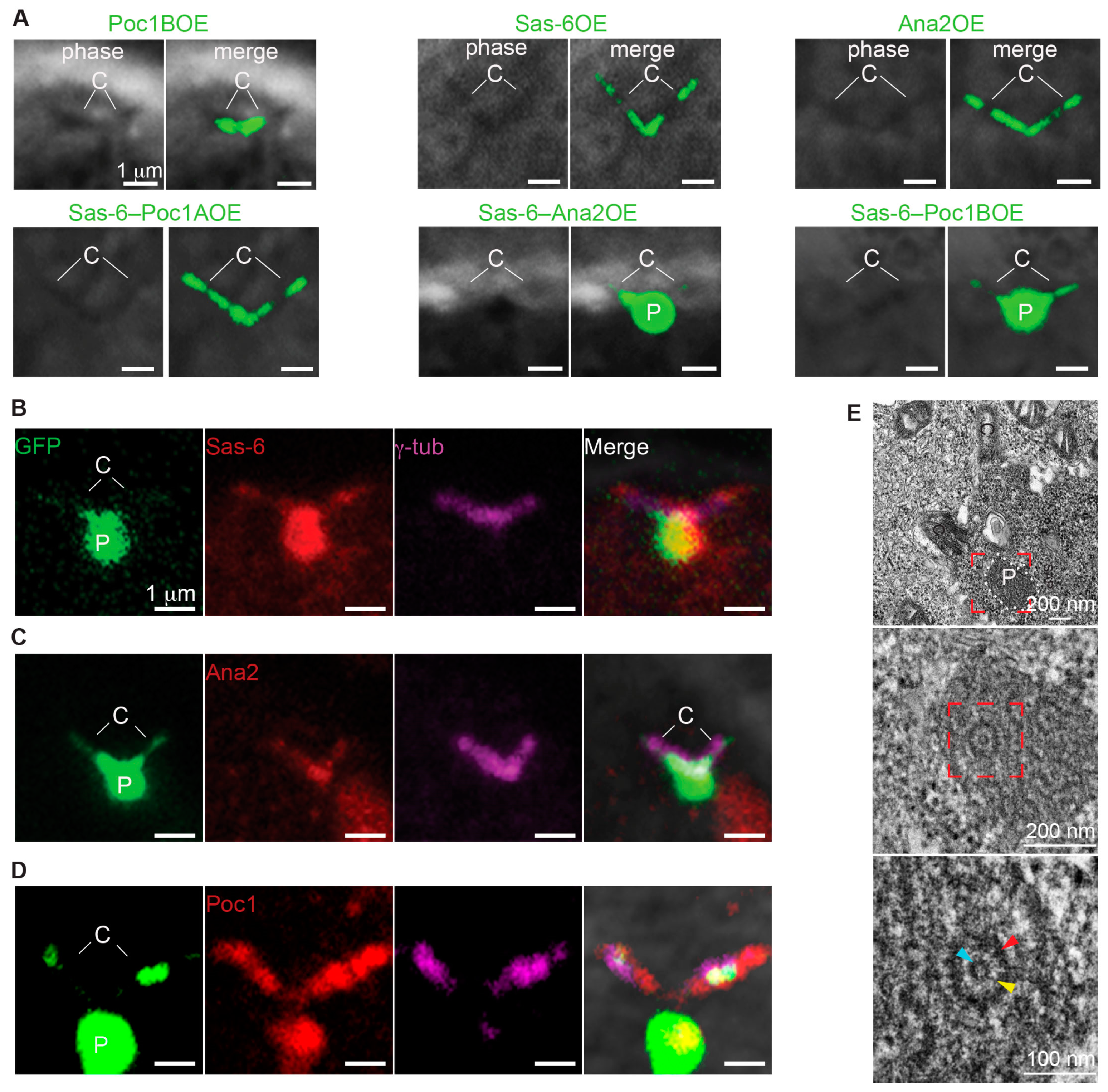
3.5. Ana2 and Sas-6 Overexpression Can Induce PCL-Like Structure That Contains Poc1
3.6. Poc1 Proteins Are Essential for Proper Formation of PCL-Like Structure
3.7. Poc1B and Sas-6 Co-Overexpression Is Sufficient to Induce PCL-Like Structures
3.8. The Coiled Coil Domain of Poc1B Is Essential to Induce Ectopic Particles
4. Discussion
- Poc1B and Sas-6 were recruited during the initial stage of PCL formation.
- Poc1B and Sas-6 were localized in the PCL core.
- Poc1B recruitment was dependent on Sas-6.
- Poc1 mutation increased Sas-6 localization to the PCL.
- Poc1B overexpression increased Sas-6 in the testes and PCL.
- Overexpression of Ana2 and Sas-6 induced the formation of atypical particles in PCL-like structures that include Poc1 proteins.
- Poc1 proteins were essential for forming stable PCL-like structures.
- Overexpression of Poc1B and Sas-6 induced the formation of PCL-like structures.
- The C-terminal coiled coil of Poc1B was essential for formation of ectopic practical.
4.1. PCL Could Be Homologous to the Intermediate Structure during Pro-Centriole Formation
4.2. Poc1B May Be a Component of the Cartwheel Inner Density
4.3. Poc1B May Have Multiple Functions in Centriole Assembly
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bornens, M. The centrosome in cells and organisms. Science 2012, 335, 422–426. [Google Scholar] [CrossRef] [PubMed]
- Avidor-Reiss, T.; Gopalakrishnan, J. Building a centriole. Curr. Opin. Cell Biol. 2013, 25, 72–77. [Google Scholar] [CrossRef]
- Avidor-Reiss, T.; Khire, A.; Fishman, E.L.; Jo, K.H. Atypical centrioles during sexual reproduction. Front. Cell Dev. Biol 2015. [Google Scholar] [CrossRef] [PubMed]
- Avidor-Reiss, T. Rapid Evolution of Sperm Produces Diverse Centriole Structures that Reveal the Most Rudimentary Structure Needed for Function. Cells 2018. [Google Scholar] [CrossRef] [PubMed]
- Fishman, E.L.; Jo, K.; Ha, A.; Royfman, R.; Zinn, A.; Krishnamurthy, M.; Avidor-Reiss, T. Atypical centrioles are present in Tribolium sperm. Open Biol. 2017, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khire, A.; Jo, K.H.; Kong, D.; Akhshi, T.; Blachon, S.; Cekic, A.R.; Hynek, S.; Ha, A.; Loncarek, J.; Mennella, V.; et al. Centriole Remodeling during Spermiogenesis in Drosophila. Curr. Biol. 2016, 26, 3183–3189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fishman, E.L.; Jo, K.; Nguyen, Q.P.H.; Kong, D.; Royfman, R.; Cekic, A.R.; Khanal, S.; Miller, A.L.; Simerly, C.; Schatten, G.; et al. A novel atypical sperm centriole is functional during human fertilization. Nat. Commun. 2018. [Google Scholar] [CrossRef]
- Gottardo, M.; Callaini, G.; Riparbelli, M.G. Structural characterization of procentrioles in Drosophila spermatids. Cytoskeleton 2015, 72, 576–584. [Google Scholar] [CrossRef]
- Blachon, S.; Khire, A.; Avidor-Reiss, T. The origin of the second centriole in the zygote of Drosophila melanogaster. Genetics 2014, 197, 199–205. [Google Scholar] [CrossRef]
- Avidor-Reiss, T.; Fishman, E.L. It Takes Two (Centrioles) to Tango. Reproduction 2018. [Google Scholar] [CrossRef]
- Fu, J.; Lipinszki, Z.; Rangone, H.; Min, M.; Mykura, C.; Chao-Chu, J.; Schneider, S.; Dzhindzhev, N.S.; Gottardo, M.; Riparbelli, M.G.; et al. Conserved molecular interactions in centriole-to-centrosome conversion. Nat. Cell Biol. 2016, 18, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Bettencourt-Dias, M.; Rodrigues-Martins, A.; Carpenter, L.; Riparbelli, M.; Lehmann, L.; Gatt, M.K.; Carmo, N.; Balloux, F.; Callaini, G.; Glover, D.M. SAK/PLK4 is required for centriole duplication and flagella development. Curr. Biol. 2005, 15, 2199–2207. [Google Scholar] [CrossRef] [PubMed]
- Blachon, S.; Gopalakrishnan, J.; Omori, Y.; Polyanovsky, A.; Church, A.; Nicastro, D.; Malicki, J.; Avidor-Reiss, T. Drosophila asterless and vertebrate Cep152 Are orthologs essential for centriole duplication. Genetics 2008, 180, 2081–2094. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, K.F.; Maxwell, K.N.; White, J.G. The spd-2 gene is required for polarization of the anteroposterior axis and formation of the sperm asters in the Caenorhabditis elegans zygote. Dev. Biol. 2000, 222, 55–70. [Google Scholar] [CrossRef] [PubMed]
- Delattre, M.; Leidel, S.; Wani, K.; Baumer, K.; Bamat, J.; Schnabel, H.; Feichtinger, R.; Schnabel, R.; Gonczy, P. Centriolar SAS-5 is required for centrosome duplication in C. elegans. Nat. Cell Biol. 2004, 6, 656–664. [Google Scholar] [CrossRef] [PubMed]
- Leidel, S.; Delattre, M.; Cerutti, L.; Baumer, K.; Gonczy, P. SAS-6 defines a protein family required for centrosome duplication in C. elegans and in human cells. Nat. Cell Biol. 2005, 7, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, K.; Lefebvre, P.A.; Kamiya, R.; Hirono, M. Bld10p, a novel protein essential for basal body assembly in Chlamydomonas: Localization to the cartwheel, the first ninefold symmetrical structure appearing during assembly. J. Cell Biol. 2004, 165, 663–671. [Google Scholar] [CrossRef] [PubMed]
- Hung, L.Y.; Tang, C.J.; Tang, T.K. Protein 4.1 R-135 interacts with a novel centrosomal protein (CPAP) which is associated with the gamma-tubulin complex. Mol. Cell Biol. 2000, 20, 7813–7825. [Google Scholar] [CrossRef] [PubMed]
- Goshima, G.; Wollman, R.; Goodwin, S.S.; Zhang, N.; Scholey, J.M.; Vale, R.D.; Stuurman, N. Genes required for mitotic spindle assembly in Drosophila S2 cells. Science 2007, 316, 417–421. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo, D.; Wang, W.J.; Uryu, K.; Tsou, M.F. Stabilization of Cartwheel-less Centrioles for Duplication Requires CEP295-Mediated Centriole-to-Centrosome Conversion. Cell Reports 2014, 8, 957–965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, C.W.; Hsu, W.B.; Tsai, J.J.; Tang, C.J.; Tang, T.K. CEP295 interacts with microtubules and is required for centriole elongation. J. Cell Sci. 2016, 129, 2501–2513. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Megraw, T.L.; Li, K.; Kao, L.R.; Kaufman, T.C. The centrosomin protein is required for centrosome assembly and function during cleavage in Drosophila. Development 1999, 126, 2829–2839. [Google Scholar] [PubMed]
- Doxsey, S.J.; Stein, P.; Evans, L.; Calarco, P.D.; Kirschner, M. Pericentrin, a highly conserved centrosome protein involved in microtubule organization. Cell 1994, 76, 639–650. [Google Scholar] [CrossRef]
- Bonaccorsi, S.; Giansanti, M.G.; Gatti, M. Spindle self-organization and cytokinesis during male meiosis in asterless mutants of Drosophila melanogaster. J. Cell Biol. 1998, 142, 751–761. [Google Scholar] [CrossRef] [PubMed]
- Blachon, S.; Cai, X.; Roberts, K.A.; Yang, K.; Polyanovsky, A.; Church, A.; Avidor-Reiss, T. A proximal centriole-like structure is present in Drosophila spermatids and can serve as a model to study centriole duplication. Genetics 2009, 182, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Avidor-Reiss, T.; Gopalakrishnan, J.; Blachon, S.; Polyanovsky, A. Centriole duplication and inheritance in Drosophila melanogaster. In The Centrosome: Cell and Molecular Mechanisms of Functions and Dysfunctions in Disease; Schatten, H., Ed.; Humana Press: New York, NY, USA, 2012; pp. 3–31. [Google Scholar]
- Mottier-Pavie, V.; Megraw, T.L. Drosophila bld10 is a centriolar protein that regulates centriole, basal body, and motile cilium assembly. Mol. Biol. Cell 2009, 20, 2605–2614. [Google Scholar] [CrossRef] [PubMed]
- Pearson, C.G.; Osborn, D.P.; Giddings, T.H., Jr.; Beales, P.L.; Winey, M. Basal body stability and ciliogenesis requires the conserved component Poc1. J. Cell Biol. 2009, 187, 905–920. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keller, L.C.; Geimer, S.; Romijn, E.; Yates, J., 3rd; Zamora, I.; Marshall, W.F. Molecular architecture of the centriole proteome: The conserved WD40 domain protein POC1 is required for centriole duplication and length control. Mol. Biol Cell 2009, 20, 1150–1166. [Google Scholar] [CrossRef]
- Venoux, M.; Tait, X.; Hames, R.S.; Straatman, K.R.; Woodland, H.R.; Fry, A.M. Poc1A and Poc1B act together in human cells to ensure centriole integrity. J. Cell Sci. 2013, 126, 163–175. [Google Scholar] [CrossRef] [Green Version]
- Hames, R.S.; Hames, R.; Prosser, S.L.; Euteneuer, U.; Lopes, C.A.; Moore, W.; Woodland, H.R.; Fry, A.M. Pix1 and Pix2 are novel WD40 microtubule-associated proteins that colocalize with mitochondria in Xenopus germ plasm and centrosomes in human cells. Exp. Cell Res. 2008, 314, 574–589. [Google Scholar] [CrossRef]
- Sarig, O.; Nahum, S.; Rapaport, D.; Ishida-Yamamoto, A.; Fuchs-Telem, D.; Qiaoli, L.; Cohen-Katsenelson, K.; Spiegel, R.; Nousbeck, J.; Israeli, S.; et al. Short stature, onychodysplasia, facial dysmorphism, and hypotrichosis syndrome is caused by a POC1A mutation. Am. J. Hum. Genet. 2012, 91, 337–342. [Google Scholar] [CrossRef]
- Shaheen, R.; Faqeih, E.; Shamseldin, H.E.; Noche, R.R.; Sunker, A.; Alshammari, M.J.; Al-Sheddi, T.; Adly, N.; Al-Dosari, M.S.; Megason, S.G.; et al. POC1A truncation mutation causes a ciliopathy in humans characterized by primordial dwarfism. Am. J. Hum. Genet. 2012, 91, 330–336. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, Q.; Wang, F.; Liu, Q. Knockdown of poc1b causes abnormal photoreceptor sensory cilium and vision impairment in zebrafish. Biochem. Biophys. Res. Commun. 2015, 465, 651–657. [Google Scholar] [CrossRef] [Green Version]
- Roosing, S.; Lamers, I.J.; de Vrieze, E.; van den Born, L.I.; Lambertus, S.; Arts, H.H.; Group, P.B.S.; Peters, T.A.; Hoyng, C.B.; Kremer, H.; et al. Disruption of the basal body protein POC1B results in autosomal-recessive cone-rod dystrophy. Am. J. Hum. Genet. 2014, 95, 131–142. [Google Scholar] [CrossRef]
- Sydor, A.M.; Coyaud, E.; Rovelli, C.; Laurent, E.; Liu, H.; Raught, B.; Mennella, V. PPP1R35 is a novel centrosomal protein that regulates centriole length in concert with the microcephaly protein RTTN. eLife 2018. [Google Scholar] [CrossRef]
- Stevens, N.R.; Roque, H.; Raff, J.W. DSas-6 and Ana2 coassemble into tubules to promote centriole duplication and engagement. Dev. Cell 2010, 19, 913–919. [Google Scholar] [CrossRef]
- Shields, A.R.; Spence, A.C.; Yamashita, Y.M.; Davies, E.L.; Fuller, M.T. The actin-binding protein profilin is required for germline stem cell maintenance and germ cell enclosure by somatic cyst cells. Development 2014, 141, 73–82. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Li, S.; Januschke, J.; Rossi, F.; Izumi, Y.; Garcia-Alvarez, G.; Gwee, S.S.; Soon, S.B.; Sidhu, H.K.; Yu, F.; et al. An ana2/ctp/mud complex regulates spindle orientation in Drosophila neuroblasts. Dev. Cell 2011, 21, 520–533. [Google Scholar] [CrossRef]
- Cottee, M.A.; Muschalik, N.; Johnson, S.; Leveson, J.; Raff, J.W.; Lea, S.M. The homo-oligomerisation of both Sas-6 and Ana2 is required for efficient centriole assembly in flies. eLife 2015. [Google Scholar] [CrossRef]
- Gopalakrishnan, J.; Guichard, P.; Smith, A.H.; Schwarz, H.; Agard, D.A.; Marco, S.; Avidor-Reiss, T. Self-assembling SAS-6 multimer is a core centriole building block. J. Biol. Chem. 2010, 285, 8759–8770. [Google Scholar] [CrossRef]
- Basiri, M.L.; Blachon, S.; Chim, Y.C.; Avidor-Reiss, T. Imaging centrosomes in fly testes. J. Vis. Exp. 2013. [Google Scholar] [CrossRef]
- Tates, A.D. Cytodifferentiation during Spermatogenesis in Drosophila melanogaster: An. Electron. Microscope Study; Rijksuniversiteit de Leiden: Leiden, The Netherlands, 1971. [Google Scholar]
- Demarco, R.S.; Eikenes, A.H.; Haglund, K.; Jones, D.L. Investigating spermatogenesis in Drosophila melanogaster. Methods 2014, 68, 218–227. [Google Scholar] [CrossRef]
- Chen, D.; McKearin, D.M. A discrete transcriptional silencer in the bam gene determines asymmetric division of the Drosophila germline stem cell. Development 2003, 130, 1159–1170. [Google Scholar] [CrossRef]
- Stevens, N.R.; Dobbelaere, J.; Brunk, K.; Franz, A.; Raff, J.W. Drosophila Ana2 is a conserved centriole duplication factor. J. Cell Biol 2010, 188, 313–323. [Google Scholar] [CrossRef] [Green Version]
- Cizmecioglu, O.; Arnold, M.; Bahtz, R.; Settele, F.; Ehret, L.; Haselmann-Weiss, U.; Antony, C.; Hoffmann, I. Cep152 acts as a scaffold for recruitment of Plk4 and CPAP to the centrosome. J. Cell Biol 2010, 191, 731–739. [Google Scholar] [CrossRef] [Green Version]
- Guderian, G.; Westendorf, J.; Uldschmid, A.; Nigg, E.A. Plk4 trans-autophosphorylation regulates centriole number by controlling betaTrCP-mediated degradation. J. Cell Sci 2010, 123, 2163–2169. [Google Scholar] [CrossRef]
- Hatch, E.M.; Kulukian, A.; Holland, A.J.; Cleveland, D.W.; Stearns, T. Cep152 interacts with Plk4 and is required for centriole duplication. J. Cell Biol. 2010, 191, 721–729. [Google Scholar] [CrossRef] [Green Version]
- Habedanck, R.; Stierhof, Y.D.; Wilkinson, C.J.; Nigg, E.A. The Polo kinase Plk4 functions in centriole duplication. Nat. Cell Biol. 2005, 7, 1140–1146. [Google Scholar] [CrossRef]
- Gottardo, M.; Callaini, G.; Riparbelli, M.G. Procentriole assembly without centriole disengagement-a paradox of male gametogenesis. J. Cell Sci. 2014, 127, 3434–3439. [Google Scholar] [CrossRef]
- Dzhindzhev, N.S.; Tzolovsky, G.; Lipinszki, Z.; Schneider, S.; Lattao, R.; Fu, J.; Debski, J.; Dadlez, M.; Glover, D.M. Plk4 phosphorylates Ana2 to trigger Sas6 recruitment and procentriole formation. Curr. Biol. 2014, 24, 2526–2532. [Google Scholar] [CrossRef]
- Saurya, S.; Roque, H.; Novak, Z.A.; Wainman, A.; Aydogan, M.G.; Volanakis, A.; Sieber, B.; Pinto, D.M.; Raff, J.W. Drosophila Ana1 is required for centrosome assembly and centriole elongation. J. Cell Sci. 2016, 129, 2514–2525. [Google Scholar] [CrossRef]
- Avidor-Reiss, T.; Turner, K. The Evolution of Centriole Structure: Heterochrony, Neoteny, and Hypermorphosis. In Centriole and Golgi; Kloc, M., Ed.; Springer: New York, NY, USA, In press.
- Nakazawa, Y.; Hiraki, M.; Kamiya, R.; Hirono, M. SAS-6 is a cartwheel protein that establishes the 9-fold symmetry of the centriole. Curr. Biol. 2007, 17, 2169–2174. [Google Scholar] [CrossRef]
- Guichard, P.; Hamel, V.; Gonczy, P. The Rise of the Cartwheel: Seeding the Centriole Organelle. Bioessays 2018. [Google Scholar] [CrossRef]
- Li, S.; Fernandez, J.J.; Marshall, W.F.; Agard, D.A. Electron cryo-tomography provides insight into procentriole architecture and assembly mechanism. eLife 2019. [Google Scholar] [CrossRef]
- Bayless, B.A.; Galati, D.F.; Junker, A.D.; Backer, C.B.; Gaertig, J.; Pearson, C.G. Asymmetrically localized proteins stabilize basal bodies against ciliary beating forces. J. Cell Biol 2016, 215, 457–466. [Google Scholar] [CrossRef]
- Bayless, B.A.; Giddings, T.H., Jr.; Winey, M.; Pearson, C.G. Bld10/Cep135 stabilizes basal bodies to resist cilia-generated forces. Mol. Biol Cell 2012, 23, 4820–4832. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jo, K.H.; Jaiswal, A.; Khanal, S.; Fishman, E.L.; Curry, A.N.; Avidor-Reiss, T. Poc1B and Sas-6 Function Together during the Atypical Centriole Formation in Drosophila melanogaster. Cells 2019, 8, 841. https://doi.org/10.3390/cells8080841
Jo KH, Jaiswal A, Khanal S, Fishman EL, Curry AN, Avidor-Reiss T. Poc1B and Sas-6 Function Together during the Atypical Centriole Formation in Drosophila melanogaster. Cells. 2019; 8(8):841. https://doi.org/10.3390/cells8080841
Chicago/Turabian StyleJo, Kyoung H., Ankit Jaiswal, Sushil Khanal, Emily L. Fishman, Alaina N. Curry, and Tomer Avidor-Reiss. 2019. "Poc1B and Sas-6 Function Together during the Atypical Centriole Formation in Drosophila melanogaster" Cells 8, no. 8: 841. https://doi.org/10.3390/cells8080841





