Recent Research Advances in Mitosis during Mammalian Gametogenesis
Abstract
1. Introduction
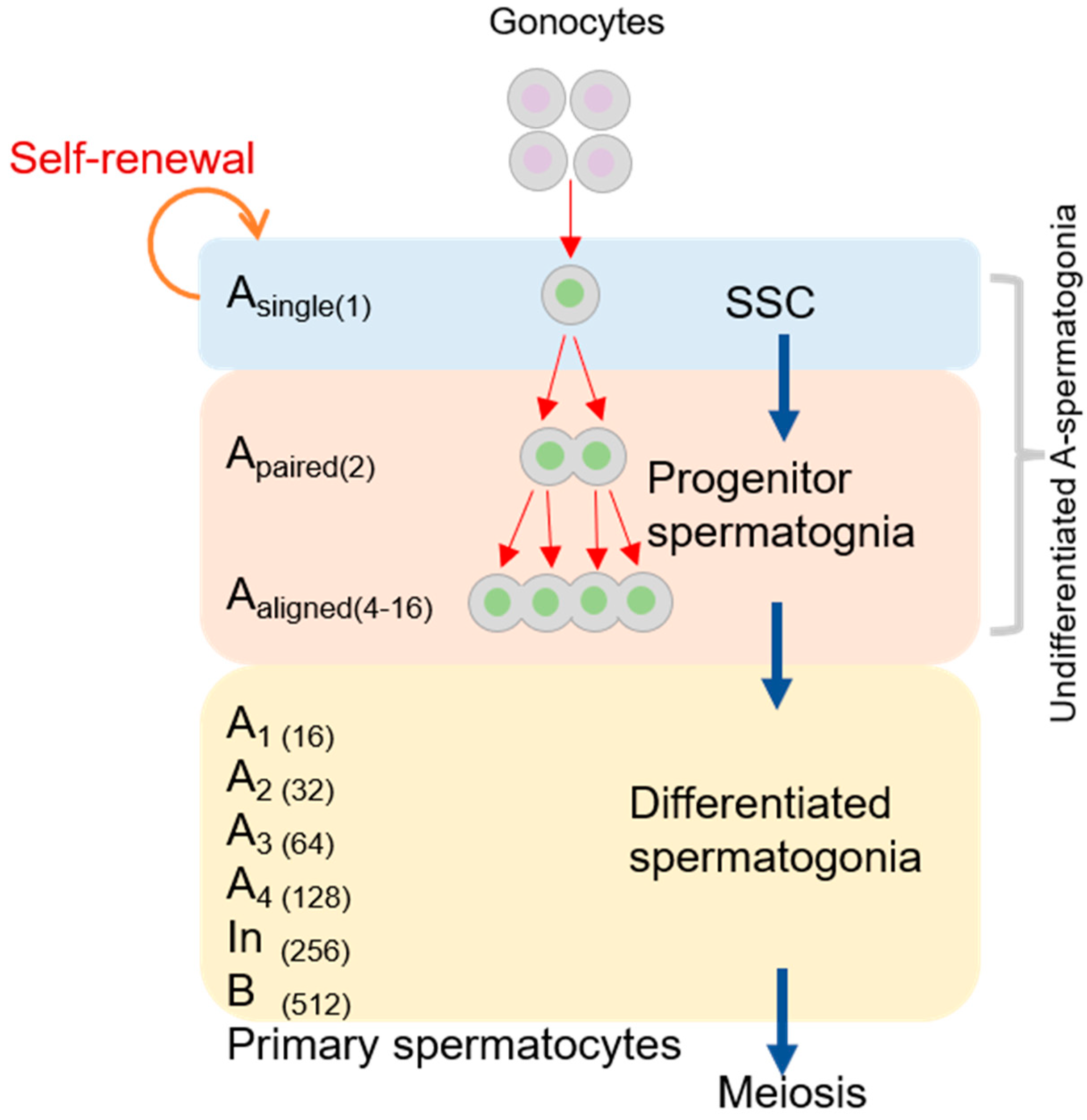
2. Mitosis of Male Gametogenesis
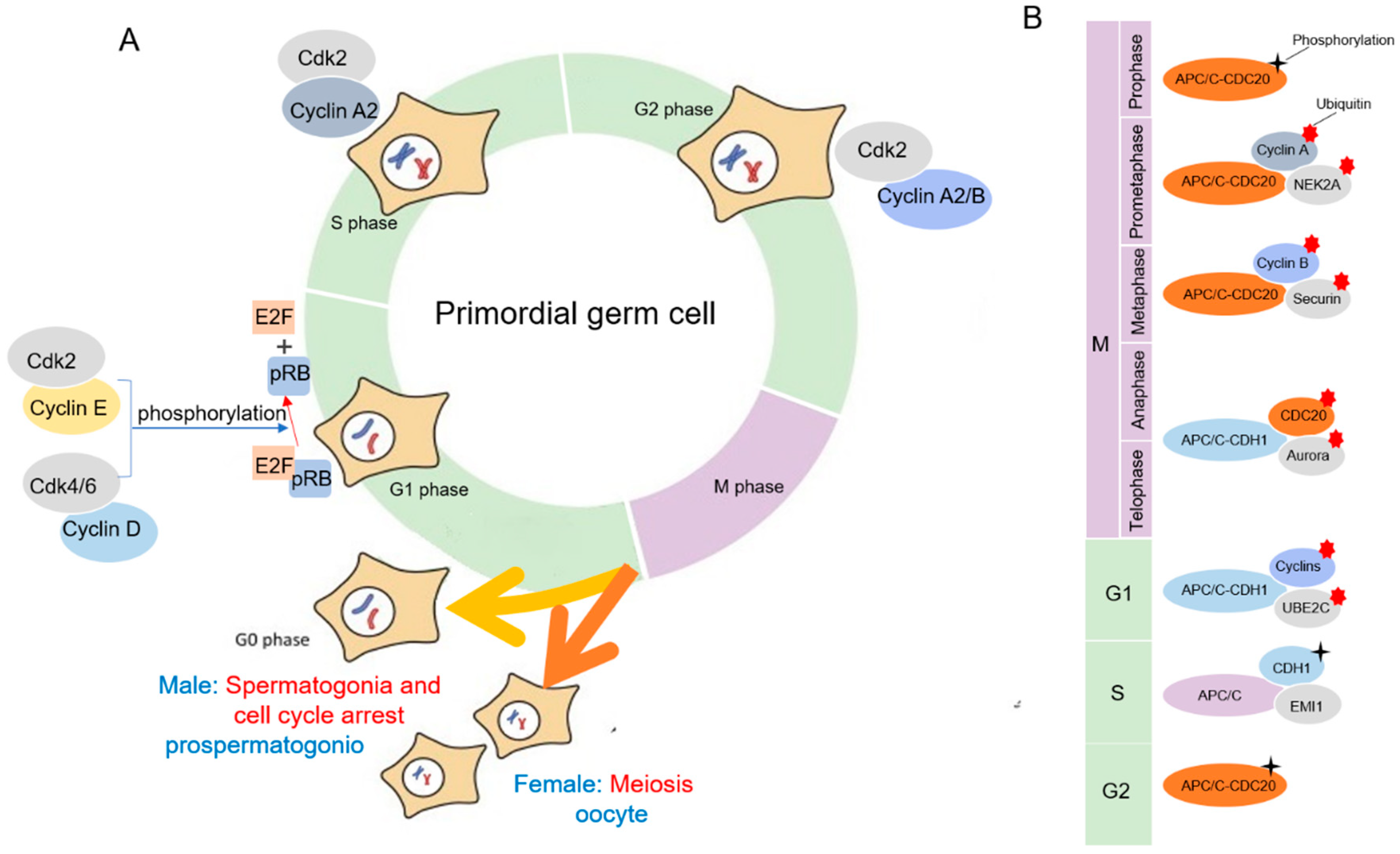
3. Mitosis of Female Gametogenesis
4. Gene Regulation of Mitosis during Mammalian Gametogenesis
4.1. Cyclin/CDK Regulation of Mitosis during Mammalian Gametogenesis
4.2. APC/C Regulation of Mitosis during Mammalian Gametogenesis
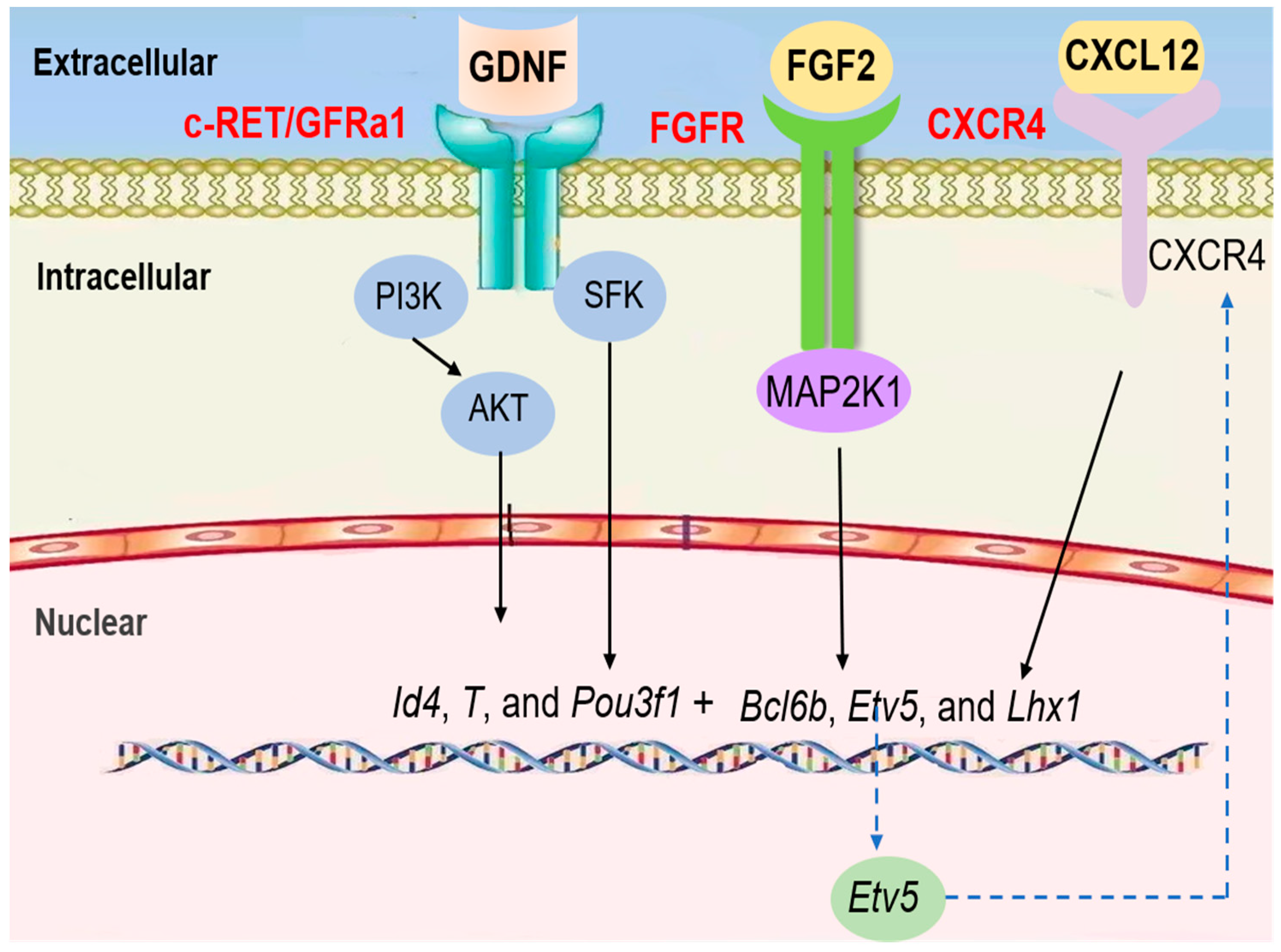
5. Signaling Pathways/Molecules Regulating Mitosis during Mammalian Gametogenesis
6. mRNA Regulation in Mitosis during Mammalian Gametogenesis
7. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| APC/C | anaphase-promoting complex or cyclosome |
| Akt | thymoma viral proto-oncogene 1 |
| ATF | activating transcription factor |
| Bcl6b | B-cell CLL/lymphoma 6, member B |
| Ccnd | cyclin D |
| CDC20 | cell division cycle 20 |
| CDH1 | CDC20 homologue 1 |
| Cdk | cyclin-dependent kinase |
| CXCL12 | chemokine (C-X-C motif) ligand 12 |
| CXCR4 | C-X-C chemokine receptor type 4 |
| EMI1 | early mitotic inhibitor 1 |
| Etv5 | ets variant gene 5 |
| Fgf2 | fibroblast growth factor 2 |
| Gdnf | glial cell line-derived neurotrophic factor |
| Gfrα-1 | glial cell line-derived neurotrophic factor family receptor alpha 1 |
| Kitl | KIT ligand or stem cell factor |
| NEK2A | NIMA-related expressed kinase 2A |
| PI3K | phosphatidylinositol 3-kinase |
| POZ | poxvirus and zinc finger |
| pRB | retinoblastoma protein |
| SCs | Sertoli cells |
| SFK | src family kinase |
| SKAP | Small kinetochore-associated protein |
| SSC | spermatogonial stem cell |
| Taf4B | TATA box-binding protein (TBP)-associated factor, subunit 4B |
| UBE2C | ubiquitin-conjugating enzyme E2C |
References
- Carreau, S.; Hess, R.A. Oestrogens and spermatogenesis. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2010, 365, 1517–1535. [Google Scholar] [CrossRef] [PubMed]
- Lesch, B.J.; Page, D.C. Genetics of germ cell development. Nat. Rev. Genet. 2012, 13, 781–794. [Google Scholar] [CrossRef] [PubMed]
- Sahin, Z.; Szczepny, A.; McLaughlin, E.A.; Meistrich, M.L.; Zhou, W.; Ustunel, I.; Loveland, K.L. Dynamic Hedgehog signalling pathway activity in germline stem cells. Andrology 2014, 2, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Seki, Y.; Yamaji, M.; Yabuta, Y.; Sano, M.; Shigeta, M.; Matsui, Y.; Saga, Y.; Tachibana, M.; Shinkai, Y.; Saitou, M. Cellular dynamics associated with the genome-wide epigenetic reprogramming in migrating primordial germ cells in mice. Development 2007, 134, 2627–2638. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Shirayoshi, Y.; Koshimizu, U.; Hashimoto, S.; Yonehara, S.; Eguchi, Y.; Tsujimoto, Y.; Nakatsuji, N. Gene transfection of mouse primordial germ cells in vitro and analysis of their survival and growth control. Exp. Cell Res. 1997, 230, 76–83. [Google Scholar] [CrossRef]
- Oatley, J.A.; Brinster, R.L. Regulation of Spermatogonial Stem Cell Self-Renewal in Mammals. Annu. Rev. Cell Dev. Bi. 2008, 24, 263–286. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, E.; Tumbar, T.; Guasch, G. Socializing with the neighbors: Stem cells and their niche. Cell 2004, 116, 769–778. [Google Scholar] [CrossRef]
- Cooke, H.J.; Saunders, P.T. Mouse models of male infertility. Nat. Rev. Genet. 2002, 3, 790–801. [Google Scholar] [CrossRef]
- Edson, M.A.; Nagaraja, A.K.; Matzuk, M.M. The mammalian ovary from genesis to revelation. Endocr. Rev. 2009, 30, 624–712. [Google Scholar] [CrossRef]
- De Rooij, D.G. Proliferation and differentiation of spermatogonial stem cells. Reproduction 2001, 121, 347–354. [Google Scholar] [CrossRef]
- Pepling, M.E. From primordial germ cell to primordial follicle: Mammalian female germ cell development. Genesis 2006, 44, 622–632. [Google Scholar] [CrossRef] [PubMed]
- Hunter, N. Meiotic Recombination: The Essence of Heredity. Cold Spring Harb. Perspect. Biol. 2015, 7, a016618. [Google Scholar] [CrossRef] [PubMed]
- Baudat, F.; Imai, Y.; de Massy, B. Meiotic recombination in mammals: Localization and regulation. Nat. Rev. Genet. 2013, 14, 794–806. [Google Scholar] [CrossRef] [PubMed]
- Yanagida, M. The Role of Model Organisms in the History of Mitosis Research. Csh. Perspect. Biol. 2014, 6, a15768. [Google Scholar] [CrossRef] [PubMed]
- Vallier, L. Cell Cycle Rules Pluripotency. Cell Stem Cell 2015, 17, 131–132. [Google Scholar] [CrossRef]
- Takeda, D.Y.; Dutta, A. DNA replication and progression through S phase. Oncogene 2005, 24, 2827–2843. [Google Scholar] [CrossRef]
- DeRan, M.; Pulvino, M.; Greene, E.; Su, C.; Zhao, J. Transcriptional activation of histone genes requires NPAT-dependent recruitment of TRRAP-Tip60 complex to histone promoters during the G1/S phase transition. Mol. Cell. Biol. 2008, 28, 435–447. [Google Scholar] [CrossRef]
- Ramachandran, S.; Henikoff, S. Replicating Nucleosomes. Sci. Adv. 2015, 1, e1500587. [Google Scholar] [CrossRef]
- De Souza, C.P.; Osmani, S.A. Mitosis, not just open or closed. Eukaryot. Cell 2007, 6, 1521–1527. [Google Scholar] [CrossRef]
- Guttinger, S.; Laurell, E.; Kutay, U. Orchestrating nuclear envelope disassembly and reassembly during mitosis. Nat. Rev. Mol. Cell Biol. 2009, 10, 178–191. [Google Scholar] [CrossRef]
- Jongsma, M.L.; Berlin, I.; Neefjes, J. On the move: Organelle dynamics during mitosis. Trends Cell Biol. 2015, 25, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Vicente, J.J.; Wordeman, L. Mitosis, microtubule dynamics and the evolution of kinesins. Exp. Cell Res. 2015, 334, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Hanukoglu, A.; Hanukoglu, I. Localization of epithelial sodium channel (ENaC) and CFTR in the germinal epithelium of the testis, Sertoli cells, and spermatozoa. J. Mol. Histol. 2018, 49, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Rato, L.; Alves, M.G.; Socorro, S.; Duarte, A.I.; Cavaco, J.E.; Oliveira, P.F. Metabolic regulation is important for spermatogenesis. Nat. Rev. Urol. 2012, 9, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Chojnacka, K.; Zarzycka, M.; Mruk, D.D. Biology of the Sertoli Cell in the Fetal, Pubertal, and Adult Mammalian Testis. Results Probl Cell Differ. 2016, 58, 225–251. [Google Scholar] [PubMed]
- De Rooij, D.G.; Russell, L.D. All you wanted to know about spermatogonia but were afraid to ask. J. Androl. 2000, 21, 776–798. [Google Scholar] [PubMed]
- Licatalosi, D.D. Roles of RNA-binding Proteins and Post-transcriptional Regulation in Driving Male Germ Cell Development in the Mouse. Adv. Exp. Med. Biol. 2016, 907, 123–151. [Google Scholar] [PubMed]
- Griswold, M.D. Spermatogenesis: The Commitment to Meiosis. Physiol. Rev. 2016, 96, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Ren, H.P.; Russell, L.D. Clonal development of interconnected germ cells in the rat and its relationship to the segmental and subsegmental organization of spermatogenesis. Am. J. Anat. 1991, 192, 121–128. [Google Scholar] [CrossRef]
- Haglund, K.; Nezis, I.P.; Stenmark, H. Structure and functions of stable intercellular bridges formed by incomplete cytokinesis during development. Commun. Integr. Biol. 2011, 4, 1–9. [Google Scholar] [CrossRef]
- Nishimura, H.; L’Hernault, S.W. Spermatogenesis. Curr. Biol. 2017, 27, R988–R994. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Thompson, L.A.; Dufour, J.M. Sertoli cells—Immunological sentinels of spermatogenesis. Semin. Cell Dev. Biol. 2014, 30, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Johnson, L.; Thompson, D.J.; Varner, D.D. Role of Sertoli cell number and function on regulation of spermatogenesis. Anim. Reprod. Sci. 2008, 105, 23–51. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.C.; Kiyomitsu, T.; Hori, T.; Backer, C.B.; Fukagawa, T.; Cheeseman, I.M. Aurora B kinase controls the targeting of the Astrin-SKAP complex to bioriented kinetochores. J. Cell Biol. 2010, 191, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Grey, C.; Espeut, J.; Ametsitsi, R.; Kumar, R.; Luksza, M.; Brun, C.; Verlhac, M.H.; Suja, J.A.; de Massy, B. SKAP, an outer kinetochore protein, is required for mouse germ cell development. Reproduction 2016, 151, 239–251. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Porras-Gomez, T.J.; Moreno-Mendoza, N. Neo-oogenesis in mammals. Zygote 2017, 25, 404–422. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, F.; Smitz, J. Molecular control of oogenesis. Biochim. Biophys. Acta 2012, 1822, 1896–1912. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; Liu, H.; Gu, X.; Boots, C.; Moley, K.H.; Wang, Q. Metabolic control of oocyte development: Linking maternal nutrition and reproductive outcomes. Cell. Mol. Life Sci. 2015, 72, 251–271. [Google Scholar] [CrossRef] [PubMed]
- McLaren, A. Primordial germ cells in the mouse. Dev. Biol. 2003, 262, 1–15. [Google Scholar] [CrossRef]
- Kurimoto, K.; Saitou, M. Mechanism and Reconstitution In Vitro of Germ Cell Development in Mammals. Cold Spring Harb. Symp. Quant. Biol. 2015, 80, 147–154. [Google Scholar] [CrossRef]
- Western, P. Foetal germ cells: Striking the balance between pluripotency and differentiation. Int. J. Dev. Biol. 2009, 53, 393–409. [Google Scholar] [CrossRef] [PubMed]
- Risal, S.; Zhang, J.; Adhikari, D.; Liu, X.; Shao, J.; Hu, M.; Busayavalasa, K.; Tu, Z.; Chen, Z.; Kaldis, P.; et al. MASTL is essential for anaphase entry of proliferating primordial germ cells and establishment of female germ cells in mice. Cell Discov. 2017, 3, 16052. [Google Scholar] [CrossRef] [PubMed]
- Atchison, F.W.; Capel, B.; Means, A.R. Pin1 regulates the timing of mammalian primordial germ cell proliferation. Development 2003, 130, 3579–3586. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, S.; Nakamura, N.; Vazquez, F.; Batt, D.B.; Perera, S.; Roberts, T.M.; Sellers, W.R. Regulation of G1 progression by the PTEN tumor suppressor protein is linked to inhibition of the phosphatidylinositol 3-kinase/Akt pathway. Proc. Natl. Acad. Sci. USA 1999, 96, 2110–2115. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Lesche, R.; Li, D.M.; Liliental, J.; Zhang, H.; Gao, J.; Gavrilova, N.; Mueller, B.; Liu, X.; Wu, H. PTEN modulates cell cycle progression and cell survival by regulating phosphatidylinositol 3,4,5,-trisphosphate and Akt/protein kinase B signaling pathway. Proc. Natl. Acad. Sci. USA 1999, 96, 6199–6204. [Google Scholar] [CrossRef] [PubMed]
- Neirijnck, Y.; Kuhne, F.; Mayere, C.; Pavlova, E.; Sararols, P.; Foti, M.; Atanassova, N.; Nef, S. Tumor Suppressor PTEN Regulates Negatively Sertoli Cell Proliferation, Testis Size, and Sperm Production In Vivo. Endocrinology 2019, 160, 387–398. [Google Scholar] [CrossRef] [PubMed]
- Ohta, H.; Tohda, A.; Nishimune, Y. Proliferation and differentiation of spermatogonial stem cells in the w/wv mutant mouse testis. Biol. Reprod. 2003, 69, 1815–1821. [Google Scholar] [CrossRef]
- Driancourt, M.A.; Reynaud, K.; Cortvrindt, R.; Smitz, J. Roles of KIT and KIT LIGAND in ovarian function. Rev. Reprod. 2000, 5, 143–152. [Google Scholar] [CrossRef]
- Lutzmann, M.; Grey, C.; Traver, S.; Ganier, O.; Maya-Mendoza, A.; Ranisavljevic, N.; Bernex, F.; Nishiyama, A.; Montel, N.; Gavois, E.; et al. MCM8- and MCM9-deficient mice reveal gametogenesis defects and genome instability due to impaired homologous recombination. Mol. Cell 2012, 47, 523–534. [Google Scholar] [CrossRef]
- Park, J.; Long, D.T.; Lee, K.Y.; Abbas, T.; Shibata, E.; Negishi, M.; Luo, Y.; Schimenti, J.C.; Gambus, A.; Walter, J.C.; et al. The MCM8-MCM9 complex promotes RAD51 recruitment at DNA damage sites to facilitate homologous recombination. Mol. Cell. Biol. 2013, 33, 1632–1644. [Google Scholar] [CrossRef]
- Murakumo, Y.; Roth, T.; Ishii, H.; Rasio, D.; Numata, S.; Croce, C.M.; Fishel, R. A human REV7 homolog that interacts with the polymerase zeta catalytic subunit hREV3 and the spindle assembly checkpoint protein hMAD2. J. Biol. Chem. 2000, 275, 4391–4397. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Schimenti, J.C. MCM9 deficiency delays primordial germ cell proliferation independent of the ATM pathway. Genesis 2015, 53, 678–684. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, N.; Mii, S.; Asai, N.; Asai, M.; Niimi, K.; Ushida, K.; Kato, T.; Enomoto, A.; Ishii, H.; Takahashi, M.; et al. The REV7 subunit of DNA polymerase zeta is essential for primordial germ cell maintenance in the mouse. J. Biol. Chem. 2013, 288, 10459–10471. [Google Scholar] [CrossRef] [PubMed]
- Pandey, V.; Tripathi, A.; Dubey, P.K. Expression and intracellular localization of Nanos2-homologue protein in primordial germ cells and spermatogonial stem cells. Zygote 2019, 27, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Tsuda, M.; Sasaoka, Y.; Kiso, M.; Abe, K.; Haraguchi, S.; Kobayashi, S.; Saga, Y. Conserved role of nanos proteins in germ cell development. Science 2003, 301, 1239–1241. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, A.; Saga, Y. Nanos2 suppresses meiosis and promotes male germ cell differentiation. Genes Dev. 2008, 22, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Arsenault, M.; Ng, E.T.; Longmuss, E.; Chau, T.C.; Hartwig, S.; Koopman, P. SOX4 regulates gonad morphogenesis and promotes male germ cell differentiation in mice. Dev. Biol. 2017, 423, 46–56. [Google Scholar] [CrossRef]
- Cook, M.S.; Munger, S.C.; Nadeau, J.H.; Capel, B. Regulation of male germ cell cycle arrest and differentiation by DND1 is modulated by genetic background. Development 2011, 138, 23–32. [Google Scholar] [CrossRef]
- Lim, S.; Kaldis, P. Cdks, cyclins and CKIs: Roles beyond cell cycle regulation. Development 2013, 140, 3079–3093. [Google Scholar] [CrossRef]
- Malumbres, M.; Barbacid, M. Mammalian cyclin-dependent kinases. Trends Biochem. Sci. 2005, 30, 630–641. [Google Scholar] [CrossRef] [PubMed]
- Sisakhtnezhad, S.; Heshmati, P. Comparative analysis of single-cell RNA sequencing data from mouse spermatogonial and mesenchymal stem cells to identify differentially expressed genes and transcriptional regulators of germline cells. J. Cell. Physiol. 2018, 233, 5231–5242. [Google Scholar] [CrossRef] [PubMed]
- Sorrentino, E.; Nazzicone, V.; Farini, D.; Campagnolo, L.; De Felici, M. Comparative transcript profiles of cell cycle-related genes in mouse primordial germ cells, embryonic stem cells and embryonic germ cells. Gene Expr. Patterns 2007, 7, 714–721. [Google Scholar] [CrossRef] [PubMed]
- Western, P.S.; Miles, D.C.; van den Bergen, J.A.; Burton, M.; Sinclair, A.H. Dynamic regulation of mitotic arrest in fetal male germ cells. Stem Cells 2008, 26, 339–347. [Google Scholar] [CrossRef] [PubMed]
- De Felici, M.; Farini, D. The control of cell cycle in mouse primordial germ cells: Old and new players. Curr. Pharm. Des. 2012, 18, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Ravnik, S.E.; Wolgemuth, D.J. The developmentally restricted pattern of expression in the male germ line of a murine cyclin A, cyclin A2, suggests roles in both mitotic and meiotic cell cycles. Dev. Biol. 1996, 173, 69–78. [Google Scholar] [CrossRef][Green Version]
- Satyanarayana, A.; Kaldis, P. Mammalian cell-cycle regulation: Several Cdks, numerous cyclins and diverse compensatory mechanisms. Oncogene 2009, 28, 2925–2939. [Google Scholar] [CrossRef]
- Wolgemuth, D.J.; Manterola, M.; Vasileva, A. Role of cyclins in controlling progression of mammalian spermatogenesis. Int. J. Dev. Biol. 2013, 57, 159–168. [Google Scholar] [CrossRef]
- Lanza, D.G.; Dawson, E.P.; Rao, P.; Heaney, J.D. Misexpression of cyclin D1 in embryonic germ cells promotes testicular teratoma initiation. Cell Cycle 2016, 15, 919–930. [Google Scholar] [CrossRef]
- Beumer, T.L.; Roepers-Gajadien, H.L.; Gademan, I.S.; Kal, H.B.; de Rooij, D.G. Involvement of the D-type cyclins in germ cell proliferation and differentiation in the mouse. Biol. Reprod. 2000, 63, 1893–1898. [Google Scholar] [CrossRef]
- Tanaka, T.; Kanatsu-Shinohara, M.; Shinohara, T. The CDKN1B-RB1-E2F1 pathway protects mouse spermatogonial stem cells from genomic damage. J. Reprod. Dev. 2015, 61, 305–316. [Google Scholar] [CrossRef]
- Spiller, C.M.; Wilhelm, D.; Koopman, P. Retinoblastoma 1 protein modulates XY germ cell entry into G1/G0 arrest during fetal development in mice. Biol. Reprod. 2010, 82, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, S.; Gorbsky, G.J. Spatiotemporal regulation of the anaphase-promoting complex in mitosis. Nat. Rev. Mol. Cell Biol. 2015, 16, 82–94. [Google Scholar] [CrossRef] [PubMed]
- Alfieri, C.; Chang, L.; Zhang, Z.; Yang, J.; Maslen, S.; Skehel, M.; Barford, D. Molecular basis of APC/C regulation by the spindle assembly checkpoint. Nature 2016, 536, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Jeganathan, K.B.; van Deursen, J.M. Differential mitotic checkpoint protein requirements in somatic and germ cells. Biochem. Soc. Trans. 2006, 34, 583–586. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Hsiao, J.Y.; Davey, N.E.; Van Voorhis, V.A.; Foster, S.A.; Tang, C.; Morgan, D.O. Multiple mechanisms determine the order of APC/C substrate degradation in mitosis. J. Cell Biol. 2014, 207, 23–39. [Google Scholar] [CrossRef]
- Kanatsu-Shinohara, M.; Onoyama, I.; Nakayama, K.I.; Shinohara, T. Skp1-Cullin-F-box (SCF)-type ubiquitin ligase FBXW7 negatively regulates spermatogonial stem cell self-renewal. Proc. Natl. Acad. Sci. USA 2014, 111, 8826–8831. [Google Scholar] [CrossRef] [PubMed]
- Song, H.W.; Wilkinson, M.F. Transcriptional control of spermatogonial maintenance and differentiation. Semin. Cell Dev. Biol. 2014, 30, 14–26. [Google Scholar] [CrossRef]
- Absalan, F.; Movahedin, M.; Mowla, S.J. Spermatogonial stem cell transplantation and subsequent orchidopexy in the bilateral cryptorchid mouse model. Cell J. 2011, 13, 143–148. [Google Scholar]
- Hermann, B.P.; Cheng, K.; Singh, A.; Roa-De, L.C.L.; Mutoji, K.N.; Chen, I.C.; Gildersleeve, H.; Lehle, J.D.; Mayo, M.; Westernstroer, B.; et al. The Mammalian Spermatogenesis Single-Cell Transcriptome, from Spermatogonial Stem Cells to Spermatids. Cell Rep. 2018, 25, 1650–1667. [Google Scholar] [CrossRef]
- Naughton, C.K.; Jain, S.; Strickland, A.M.; Gupta, A.; Milbrandt, J. Glial cell-line derived neurotrophic factor-mediated RET signaling regulates spermatogonial stem cell fate. Biol. Reprod. 2006, 74, 314–321. [Google Scholar] [CrossRef]
- Jijiwa, M.; Kawai, K.; Fukihara, J.; Nakamura, A.; Hasegawa, M.; Suzuki, C.; Sato, T.; Enomoto, A.; Asai, N.; Murakumo, Y.; et al. GDNF-mediated signaling via RET tyrosine 1062 is essential for maintenance of spermatogonial stem cells. Genes Cells 2008, 13, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Kanatsu-Shinohara, M.; Shinohara, T. Spermatogonial stem cell self-renewal and development. Annu. Rev. Cell Dev. Biol. 2013, 29, 163–187. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.; Braun, R.E. Cyclical expression of GDNF is required for spermatogonial stem cell homeostasis. Development 2018, 145, dev151555. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Grow, E.J.; Mlcochova, H.; Maher, G.J.; Lindskog, C.; Nie, X.; Guo, Y.; Takei, Y.; Yun, J.; Cai, L.; et al. The adult human testis transcriptional cell atlas. Cell Res. 2018, 28, 1141–1157. [Google Scholar] [CrossRef] [PubMed]
- Green, C.D.; Ma, Q.; Manske, G.L.; Shami, A.N.; Zheng, X.; Marini, S.; Moritz, L.; Sultan, C.; Gurczynski, S.J.; Moore, B.B.; et al. A Comprehensive Roadmap of Murine Spermatogenesis Defined by Single-Cell RNA-Seq. Dev. Cell 2018, 46, 651–667. [Google Scholar] [CrossRef] [PubMed]
- Eo, J.; Song, H.; Lim, H.J. Etv5, a transcription factor with versatile functions in male reproduction. Clin. Exp. Reprod. Med. 2012, 39, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Oatley, J.M.; Oatley, M.J.; Kaucher, A.V.; Avarbock, M.R.; Brinster, R.L. The POU domain transcription factor POU3F1 is an important intrinsic regulator of GDNF-induced survival and self-renewal of mouse spermatogonial stem cells. Biol. Reprod. 2010, 82, 1103–1111. [Google Scholar] [CrossRef]
- Oatley, J.M.; Avarbock, M.R.; Telaranta, A.I.; Fearon, D.T.; Brinster, R.L. Identifying genes important for spermatogonial stem cell self-renewal and survival. Proc. Natl. Acad. Sci. USA 2006, 103, 9524–9529. [Google Scholar] [CrossRef]
- Lee, J.; Kanatsu-Shinohara, M.; Inoue, K.; Ogonuki, N.; Miki, H.; Toyokuni, S.; Kimura, T.; Nakano, T.; Ogura, A.; Shinohara, T. Akt mediates self-renewal division of mouse spermatogonial stem cells. Development 2007, 134, 1853–1859. [Google Scholar] [CrossRef]
- Ocon-Grove, O.M.; Oatley, J.M. Molecular Mechanisms Regulating Spermatogonial Stem Cell Fate Decisions. In Stem Cell Biology and Regenerative Medicine; Orwig, K.E., Hermann, B.P., Eds.; Humana Press Inc.: Totowa, NJ, USA, 2011; pp. 135–157. [Google Scholar]
- Oatley, J.M.; Brinster, R.L. The germline stem cell niche unit in mammalian testes. Physiol. Rev. 2012, 92, 577–595. [Google Scholar] [CrossRef]
- Oatley, J.M.; Avarbock, M.R.; Brinster, R.L. Glial cell line-derived neurotrophic factor regulation of genes essential for self-renewal of mouse spermatogonial stem cells is dependent on Src family kinase signaling. J. Biol. Chem. 2007, 282, 25842–25851. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Goodyear, S.M.; Tobias, J.W.; Avarbock, M.R.; Brinster, R.L. Spermatogonial stem cell self-renewal requires ETV5-mediated downstream activation of Brachyury in mice. Biol. Reprod. 2011, 85, 1114–1123. [Google Scholar] [CrossRef] [PubMed]
- Parekh, P.; Garcia, T.X.; Hofmann, M.C. Regulation of GDNF expression in Sertoli cells. Reproduction 2019, 157, R95–R107. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, M.C. Gdnf signaling pathways within the mammalian spermatogonial stem cell niche. Mol. Cell. Endocrinol. 2008, 288, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Kanatsu-Shinohara, M.; Ogonuki, N.; Matoba, S.; Morimoto, H.; Ogura, A.; Shinohara, T. Improved serum- and feeder-free culture of mouse germline stem cells. Biol. Reprod. 2014, 91, 88. [Google Scholar] [CrossRef] [PubMed]
- Takashima, S.; Kanatsu-Shinohara, M.; Tanaka, T.; Morimoto, H.; Inoue, K.; Ogonuki, N.; Jijiwa, M.; Takahashi, M.; Ogura, A.; Shinohara, T. Functional differences between GDNF-dependent and FGF2-dependent mouse spermatogonial stem cell self-renewal. Stem Cell Rep. 2015, 4, 489–502. [Google Scholar] [CrossRef] [PubMed]
- Ishii, K.; Kanatsu-Shinohara, M.; Toyokuni, S.; Shinohara, T. FGF2 mediates mouse spermatogonial stem cell self-renewal via upregulation of Etv5 and Bcl6b through MAP2K1 activation. Development 2012, 139, 1734–1743. [Google Scholar] [CrossRef] [PubMed]
- Kitadate, Y.; Jorg, D.J.; Tokue, M.; Maruyama, A.; Ichikawa, R.; Tsuchiya, S.; Segi-Nishida, E.; Nakagawa, T.; Uchida, A.; Kimura-Yoshida, C.; et al. Competition for Mitogens Regulates Spermatogenic Stem Cell Homeostasis in an Open Niche. Cell Stem Cell 2019, 24, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Tian, R.; Yao, C.; Yang, C.; Zhu, Z.; Li, C.; Zhi, E.; Wang, J.; Li, P.; Chen, H.; Yuan, Q.; et al. Fibroblast growth factor-5 promotes spermatogonial stem cell proliferation via ERK and AKT activation. Stem Cell Res. Ther. 2019, 10, 40. [Google Scholar] [CrossRef] [PubMed]
- Chan, F.; Oatley, M.J.; Kaucher, A.V.; Yang, Q.E.; Bieberich, C.J.; Shashikant, C.S.; Oatley, J.M. Functional and molecular features of the Id4+ germline stem cell population in mouse testes. Genes Dev. 2014, 28, 1351–1362. [Google Scholar] [CrossRef]
- Garbuzov, A.; Pech, M.F.; Hasegawa, K.; Sukhwani, M.; Zhang, R.J.; Orwig, K.E.; Artandi, S.E. Purification of GFRalpha1+ and GFRalpha1-Spermatogonial Stem Cells Reveals a Niche-Dependent Mechanism for Fate Determination. Stem Cell Rep. 2018, 10, 553–567. [Google Scholar] [CrossRef]
- Tokue, M.; Ikami, K.; Mizuno, S.; Takagi, C.; Miyagi, A.; Takada, R.; Noda, C.; Kitadate, Y.; Hara, K.; Mizuguchi, H.; et al. SHISA6 Confers Resistance to Differentiation-Promoting Wnt/beta-Catenin Signaling in Mouse Spermatogenic Stem Cells. Stem Cell Rep. 2017, 8, 561–575. [Google Scholar] [CrossRef] [PubMed]
- Kanatsu-Shinohara, M.; Inoue, K.; Takashima, S.; Takehashi, M.; Ogonuki, N.; Morimoto, H.; Nagasawa, T.; Ogura, A.; Shinohara, T. Reconstitution of mouse spermatogonial stem cell niches in culture. Cell Stem Cell 2012, 11, 567–578. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.E.; Kim, D.; Kaucher, A.; Oatley, M.J.; Oatley, J.M. CXCL12-CXCR4 signaling is required for the maintenance of mouse spermatogonial stem cells. J. Cell Sci. 2013, 126, 1009–1020. [Google Scholar] [CrossRef] [PubMed]
- Wienholds, E.; Plasterk, R.H. MicroRNA function in animal development. FEBS Lett. 2005, 579, 5911–5922. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, S.; Li, L.; Li, M.; Guo, C.; Yao, J.; Mi, S. Exosome and exosomal microRNA: Trafficking, sorting, and function. Genom. Proteom. Bioinform. 2015, 13, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Kanellopoulou, C.; Muljo, S.A.; Kung, A.L.; Ganesan, S.; Drapkin, R.; Jenuwein, T.; Livingston, D.M.; Rajewsky, K. Dicer-deficient mouse embryonic stem cells are defective in differentiation and centromeric silencing. Genes Dev. 2005, 19, 489–501. [Google Scholar] [CrossRef]
- Liu, D.; Li, L.; Fu, H.; Li, S.; Li, J. Inactivation of Dicer1 has a severe cumulative impact on the formation of mature germ cells in mouse testes. Biochem. Biophys. Res. Commun. 2012, 422, 114–120. [Google Scholar] [CrossRef]
- Hayashi, K.; Chuva, D.S.L.S.; Kaneda, M.; Tang, F.; Hajkova, P.; Lao, K.; O’Carroll, D.; Das, P.P.; Tarakhovsky, A.; Miska, E.A.; et al. MicroRNA biogenesis is required for mouse primordial germ cell development and spermatogenesis. PLoS ONE 2008, 3, e1738. [Google Scholar] [CrossRef]
- Kimura, T.; Suzuki, A.; Fujita, Y.; Yomogida, K.; Lomeli, H.; Asada, N.; Ikeuchi, M.; Nagy, A.; Mak, T.W.; Nakano, T. Conditional loss of PTEN leads to testicular teratoma and enhances embryonic germ cell production. Development 2003, 130, 1691–1700. [Google Scholar] [CrossRef]
- Fu, H.; Zhou, F.; Yuan, Q.; Zhang, W.; Qiu, Q.; Yu, X.; He, Z. miRNA-31-5p Mediates the Proliferation and Apoptosis of Human Spermatogonial Stem Cells via Targeting JAZF1 and Cyclin A2. Mol. Ther.-Nucleic Acids 2019, 14, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Lichner, Z.; Pall, E.; Kerekes, A.; Pallinger, E.; Maraghechi, P.; Bosze, Z.; Gocza, E. The miR-290-295 cluster promotes pluripotency maintenance by regulating cell cycle phase distribution in mouse embryonic stem cells. Differentiation 2011, 81, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Subramanyam, D.; Lamouille, S.; Judson, R.L.; Liu, J.Y.; Bucay, N.; Derynck, R.; Blelloch, R. Multiple targets of miR-302 and miR-372 promote reprogramming of human fibroblasts to induced pluripotent stem cells. Nat. Biotechnol. 2011, 29, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Cai, T.; Zheng, C.; Lin, X.; Wang, G.; Liao, S.; Wang, X.; Gan, H.; Zhang, D.; Hu, X.; et al. MicroRNA-202 maintains spermatogonial stem cells by inhibiting cell cycle regulators and RNA binding proteins. Nucleic Acids Res. 2017, 45, 4142–4157. [Google Scholar] [CrossRef] [PubMed]
- Matson, C.K.; Murphy, M.W.; Griswold, M.D.; Yoshida, S.; Bardwell, V.J.; Zarkower, D. The mammalian doublesex homolog DMRT1 is a transcriptional gatekeeper that controls the mitosis versus meiosis decision in male germ cells. Dev. Cell 2010, 19, 612–624. [Google Scholar] [CrossRef] [PubMed]
- Cui, N.; Hao, G.; Zhao, Z.; Wang, F.; Cao, J.; Yang, A. MicroRNA-224 regulates self-renewal of mouse spermatogonial stem cells via targeting DMRT1. J. Cell. Mol. Med. 2016, 20, 1503–1512. [Google Scholar] [CrossRef] [PubMed]
- Xie, R.; Lin, X.; Du, T.; Xu, K.; Shen, H.; Wei, F.; Hao, W.; Lin, T.; Lin, X.; Qin, Y.; et al. Targeted Disruption of miR-17-92 Impairs Mouse Spermatogenesis by Activating mTOR Signaling Pathway. Medicine (Baltimore) 2016, 95, e2713. [Google Scholar] [CrossRef] [PubMed]
- Dyce, P.W.; Toms, D.; Li, J. Stem cells and germ cells: MicroRNA and gene expression signatures. Histol. Histopathol. 2010, 25, 505–513. [Google Scholar] [PubMed]
- Qi, J.; Yu, J.Y.; Shcherbata, H.R.; Mathieu, J.; Wang, A.J.; Seal, S.; Zhou, W.; Stadler, B.M.; Bourgin, D.; Wang, L.; et al. microRNAs regulate human embryonic stem cell division. Cell Cycle 2009, 8, 3729–3741. [Google Scholar] [CrossRef]
- Radisky, D.C. miR-200c at the nexus of epithelial-mesenchymal transition, resistance to apoptosis, and the breast cancer stem cell phenotype. Breast Cancer Res. 2011, 13, 110. [Google Scholar] [CrossRef]
- Niu, Z.; Goodyear, S.M.; Rao, S.; Wu, X.; Tobias, J.W.; Avarbock, M.R.; Brinster, R.L. MicroRNA-21 regulates the self-renewal of mouse spermatogonial stem cells. Proc. Natl. Acad. Sci. USA 2011, 108, 12740–12745. [Google Scholar] [CrossRef] [PubMed]
- Kedde, M.; Strasser, M.J.; Boldajipour, B.; Oude, V.J.; Slanchev, K.; le Sage, C.; Nagel, R.; Voorhoeve, P.M.; van Duijse, J.; Orom, U.A.; et al. RNA-binding protein Dnd1 inhibits microRNA access to target mRNA. Cell 2007, 131, 1273–1286. [Google Scholar] [CrossRef] [PubMed]
- Yan, N.; Lu, Y.; Sun, H.; Tao, D.; Zhang, S.; Liu, W.; Ma, Y. A microarray for microRNA profiling in mouse testis tissues. Reproduction 2007, 134, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Yan, N.; Lu, Y.; Sun, H.; Qiu, W.; Tao, D.; Liu, Y.; Chen, H.; Yang, Y.; Zhang, S.; Li, X.; et al. Microarray profiling of microRNAs expressed in testis tissues of developing primates. J. Assist. Reprod. Genet. 2009, 26, 179–186. [Google Scholar] [CrossRef] [PubMed]
| Name | Expression | Proposed Function | Targets Involved in Mammalian Gametogenesis | Reference |
|---|---|---|---|---|
| miR-17-92 cluster | PGCs, ES cells | Regulator of differentiation, proliferation and apoptosis | STAT3, E2F1, PTEN | [110,118,119] |
| miRNA-31-5p | SSCs | Regulator of SSCs proliferation | JAZF1 and Cyclin A2 | [112] |
| miR-290-295 cluster | PGCs | G1 to S phase cell cycle control | WEE1, FBXL5 | [110,113] |
| miR-202 | SSCs | Regulator of cell cycle and apoptosis of mitosis | Rbfox, Cpeb1 | [115] |
| miR-224 | SSCs | Control SSCs self-renewal and cyclical gene expression | DMRT1 | [117] |
| MiR-302-67 cluster | PGCs | targeting inhibitors of the G1/S transition | Cdkn1a | [114,120] |
| miR-125a | Later male PGCs | Control of differentiation | LIN28 | [110] |
| miR-200c | Early PGCs | Control of apoptosis | ZEB1, TRKB | [110,121] |
| miR-21 | SSCs | SSCs self-renewal, anti-apoptosis | ZEB1, TRKB | [122] |
| miR-221 | PGCs | Regulate mitotic arrest in male germ cells | DND1 | [123] |
| miR-34c | PGCs | Cell cycle regulator | CCND3, CCNG1, CCNB1 NOTCH2 | [124,125] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.-H.; Li, Y.; Deng, S.-L.; Liu, Y.-X.; Lian, Z.-X.; Yu, K. Recent Research Advances in Mitosis during Mammalian Gametogenesis. Cells 2019, 8, 567. https://doi.org/10.3390/cells8060567
Wang J-H, Li Y, Deng S-L, Liu Y-X, Lian Z-X, Yu K. Recent Research Advances in Mitosis during Mammalian Gametogenesis. Cells. 2019; 8(6):567. https://doi.org/10.3390/cells8060567
Chicago/Turabian StyleWang, Jia-Hao, Yan Li, Shou-Long Deng, Yi-Xun Liu, Zheng-Xing Lian, and Kun Yu. 2019. "Recent Research Advances in Mitosis during Mammalian Gametogenesis" Cells 8, no. 6: 567. https://doi.org/10.3390/cells8060567
APA StyleWang, J.-H., Li, Y., Deng, S.-L., Liu, Y.-X., Lian, Z.-X., & Yu, K. (2019). Recent Research Advances in Mitosis during Mammalian Gametogenesis. Cells, 8(6), 567. https://doi.org/10.3390/cells8060567







