Hepatitis C Virus Infection: Host–Virus Interaction and Mechanisms of Viral Persistence
Abstract
1. Introduction
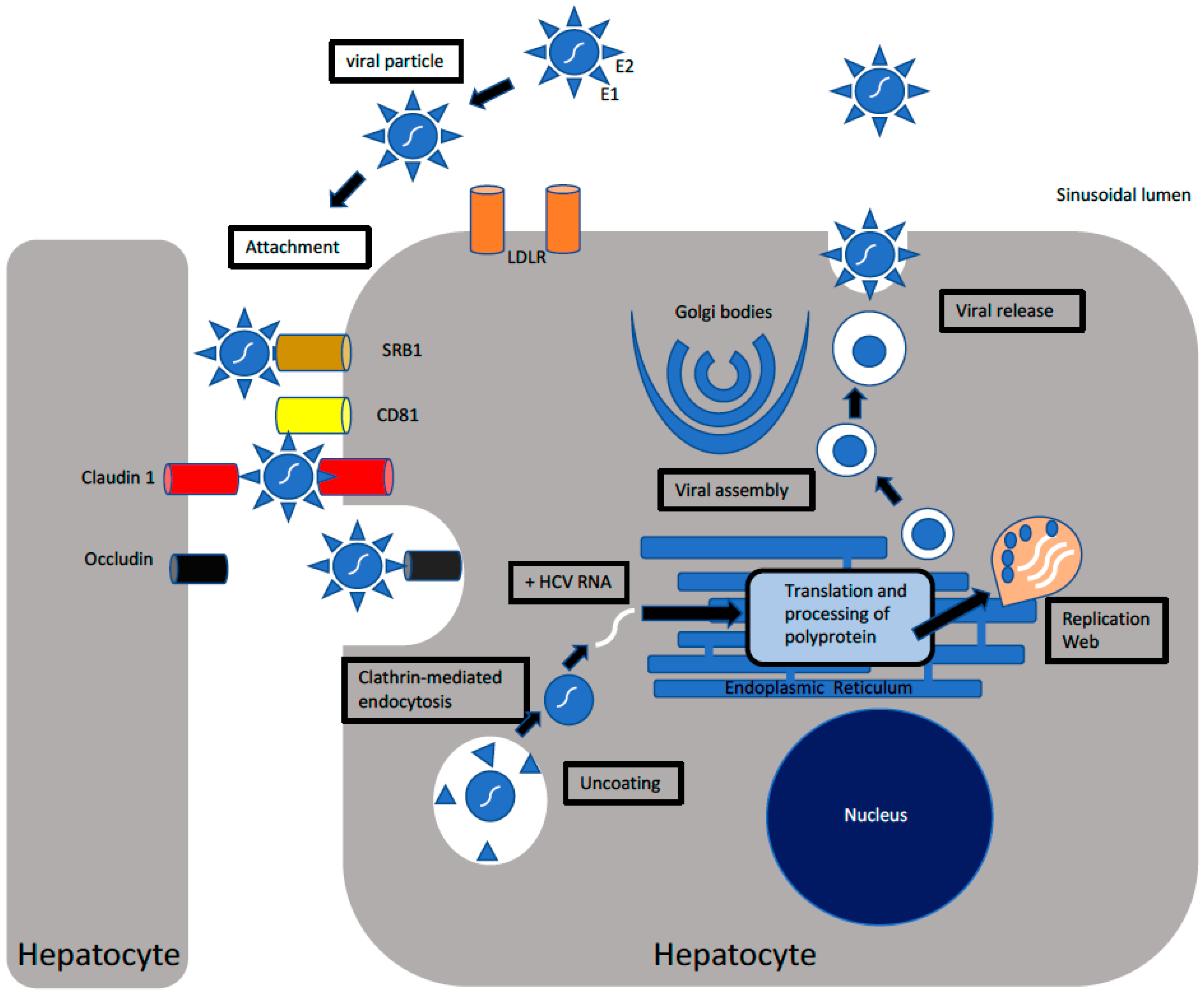
2. Characteristics, Structure, and Pathogenesis
3. Clinical Manifestations
4. HCV and Host Interaction
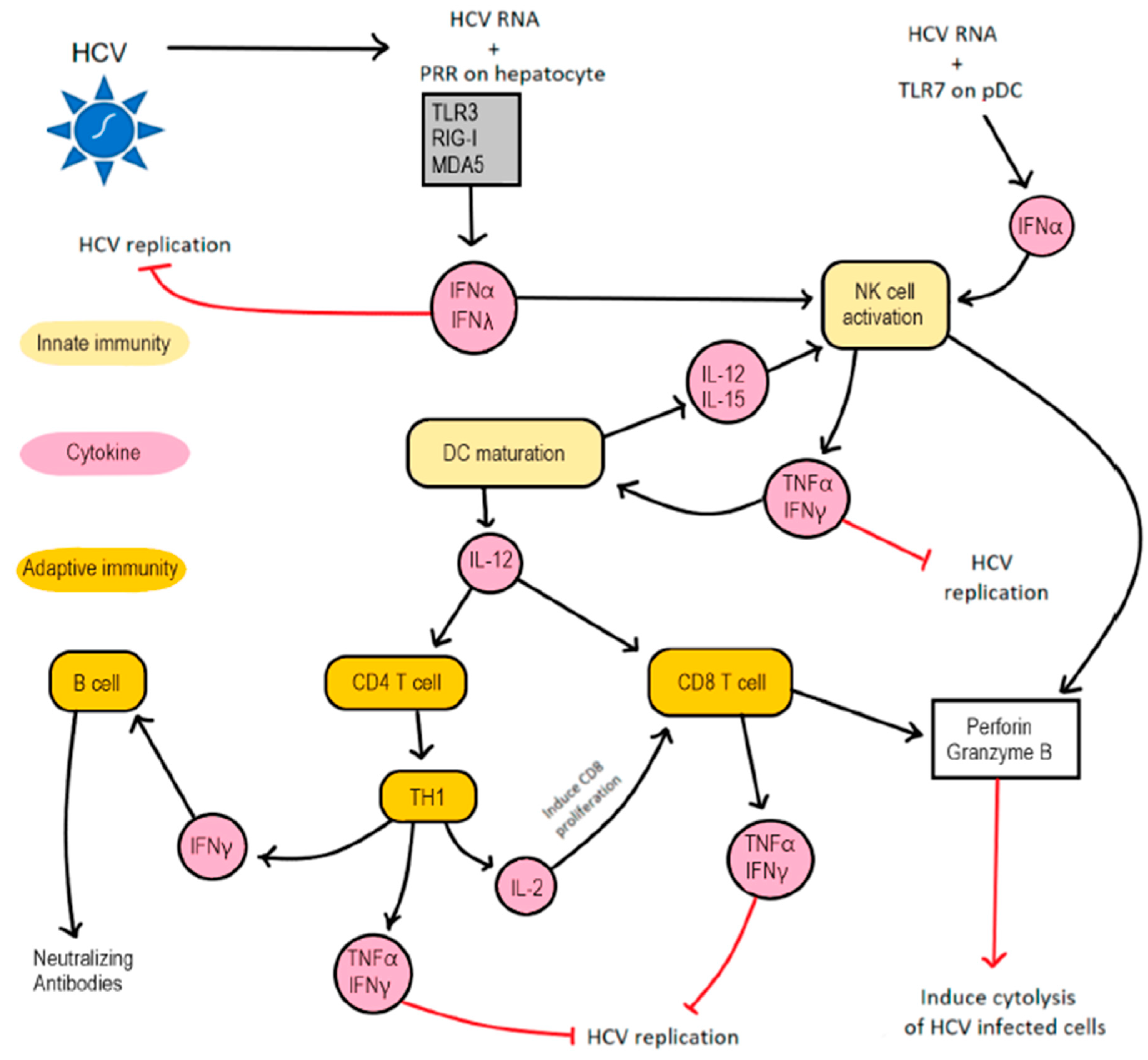
4.1. Innate Immune Response in HCV Infection
4.2. Adaptive Immune Response in HCV Infection
4.3. Effect of HCV on Myeloid Cells
4.4. Effect of HCV on Lymphoid Cells
4.5. Effect of HCV on Nonimmune Cells
5. Mechanisms Responsible for the Development of Chronic HCV Infection
6. Impact of Host–HCV Interactions on HCV Therapy
7. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Bouchard, M.J.; Navas-Martin, S. Hepatitis B and C virus hepatocarcinogenesis: Lessons learned and future challenges. Cancer Lett. 2011, 305, 123–143. [Google Scholar] [CrossRef]
- Robinson, W.S. Hepadnaviruses and hepatocellular carcinoma (HCC). Cancer Detect. Prev. 1989, 14, 245–252. [Google Scholar] [PubMed]
- Hepatitis B. Available online: http://www.who.int/news-room/fact-sheets/detail/hepatitis-b (accessed on 17 April 2019).
- Hepatitis C. Available online: http://www.who.int/news-room/fact-sheets/detail/hepatitis-c (accessed on 17 April 2019).
- Hepatitis B Progression. Available online: https://www.hepmag.com/basics/hepatitis-b-basics/hepatitis-b-progression (accessed on 4 March 2019).
- Jamma, S.; Hussain, G.; Lau, D.T.-Y. Current Concepts of HBV/HCV Coinfection: Coexistence, but Not Necessarily in Harmony. Hepatol. Rep. 2010, 9, 260–269. [Google Scholar] [CrossRef]
- Yang, R.-R.; Gui, X.; Chen, X.-Y.; Zhu, Y. Interference of replication between hepatitis B and C viruses in patients infected with HIV. J. Med Virol. 2011, 83, 1159–1164. [Google Scholar] [CrossRef] [PubMed]
- Hepatitis. Available online: https://www.afro.who.int/health-topics/hepatitis (accessed on 17 April 2019).
- Chen, S.L. The Natural History of Hepatitis C Virus (HCV) Infection. Int. J. Med Sci. 2006, 3, 47. [Google Scholar] [CrossRef]
- Hepatitis C An Epidemic for Anyone. UShealthCareCost73: The epidemic, 2018.
- King, A.M.; Brown, F.; Christian, P.; Hovi, T.; Hyypiä, T.; Knowles, N.J.; Lemon, S.M.; Minor, P.D.; Palmenberg, A.C.; Skern, T.; et al. Virus taxonomy. Seventh report of the International Committee on Taxonomy of Viruses; Academic Press: San Diego, CA, USA, 2000. [Google Scholar]
- Lindenbach, B.D.; Rice, C.M. Unravelling hepatitis C virus replication from genome to function. Nature 2005, 436, 933–938. [Google Scholar] [CrossRef]
- Bode, J.G.; Brenndorfer, E.D.; Haussinger, D. Hepatitis C virus (HCV) employs multiple strategies to subvert the host innate antiviral response. Biol. Chem. 2008, 389, 1283–1298. [Google Scholar] [CrossRef] [PubMed]
- Manns, M.P.; Buti, M.; Gane, E.; Pawlotsky, J.-M.; Razavi, H.; Terrault, N.; Younossi, Z. Hepatitis C virus infection. Nat. Rev. Dis. Primers 2017, 3, 17006. [Google Scholar] [CrossRef]
- Penin, F.; Dubuisson, J.; Rey, F.A.; Moradpour, D.; Pawlotsky, J.M. Structural biology of hepatitis C virus. Hepatology 2004, 39, 5–19. [Google Scholar] [CrossRef]
- Pachiadakis, I.; Pollara, G.; Chain, B.M.; Naoumov, N.V. Is hepatitis C virus infection of dendritic cells a mechanism facilitating viral persistence? Lancet Infect. Dis. 2005, 5, 296–304. [Google Scholar] [CrossRef]
- Sehgal, M.; Khan, Z.K.; Talal, A.H.; Jain, P. Dendritic Cells in HIV-1 and HCV Infection: Can They Help Win the Battle? Virol. Res. Treat. 2013, 4, 1–25. [Google Scholar] [CrossRef]
- Sung, V.M.-H.; Shimodaira, S.; Doughty, A.L.; Picchio, G.R.; Can, H.; Yen, T.S.B.; Lindsay, K.L.; Levine, A.M.; Lai, M.M.C.; Yen, T.S.B. Establishment of B-Cell Lymphoma Cell Lines Persistently Infected with Hepatitis C Virus In Vivo and In Vitro: The Apoptotic Effects of Virus Infection. J. Virol. 2003, 77, 2134–2146. [Google Scholar] [CrossRef]
- Inokuchi, M.; Ito, T.; Uchikoshi, M.; Shimozuma, Y.; Morikawa, K.; Nozawa, H.; Shimazaki, T.; Hiroishi, K.; Miyakawa, Y.; Imawari, M. Infection of B cells with hepatitis C virus for the development of lymphoproliferative disorders in patients with chronic hepatitis C. J. Med Virol. 2009, 81, 619–627. [Google Scholar] [CrossRef]
- García, F.; Roldán, C.; López, I.; Martínez, N.M.; Alvarez, M.; Bernal, M.C.; Hernández, J.; Maroto, M.C. Detection of HCV and GBV-CHGV RNA in peripheral blood mononuclear cells of patients with chronic type C hepatitis. Microbios 2000, 103, 7–15. [Google Scholar]
- Muratori, L.; Gibellini, D.; Lenzi, M.; Cataleta, M.; Muratori, P.; Morelli, M.C.; Bianchi, F.B. Quantification of hepatitis C virus-infected peripheral blood mononuclear cells by in situ reverse transcriptase-polymerase chain reaction. Blood 1996, 88, 2768–2774. [Google Scholar]
- Paul, D.; Hoppe, S.; Saher, G.; Krijnse-Locker, J.; Bartenschlager, R. Morphological and Biochemical Characterization of the Membranous Hepatitis C Virus Replication Compartment. J. Virol. 2013, 87, 10612–10627. [Google Scholar] [CrossRef]
- Breaking bad mitochondria: How hepatitis C survives for so long. Available online: www.sciencedaily.com/releases/2014/04/140415143959.htm (accessed on 17 April 2019).
- Ripoli, M.; Pazienza, V. Impact of HCV genetic differences on pathobiology of disease. Expert Rev. Anti Infect. Ther. 2011, 9, 747–759. [Google Scholar] [CrossRef]
- Wojcik, G.; Latanich, R.; Mosbruger, T.; Astemborski, J.; Kirk, G.D.; Mehta, S.H.; Goedert, J.J.; Kim, A.Y.; Seaberg, E.C.; Busch, M.; et al. Variants in HAVCR1 gene region contribute to hepatitis C persistence in African Americans. J. Infect. Dis. 2014, 209, 355–359. [Google Scholar] [CrossRef]
- Neukam, K.; Nattermann, J.; Rallón, N.; Rivero, A.; Caruz, A.; Macías, J.; Vogel, M.; Benito, J.; Camacho, A.; Mira, J.; et al. Different distributions of hepatitis C virus genotypes among HIV-infected patients with acute and chronic hepatitis C according to interleukin-28B genotype. HIV Med. 2011, 12, 487–493. [Google Scholar] [CrossRef]
- Pár, A.; Pár, G.; Tornai, I.; Szalay, F.; Várszegi, D.; Fráter, E.; Papp, M.; Lengyel, G.; Fehér, J.; Varga, M.; et al. IL28B and IL10R −1087 polymorphisms are protective for chronic genotype 1 HCV infection and predictors of response to interferon-based therapy in an East-Central European cohort. BMC Notes 2014, 7, 12. [Google Scholar] [CrossRef]
- Thomas, D.L.; Thio, C.L.; Martin, M.P.; Qi, Y.; Ge, D.; O’Huigin, C.; Kidd, J.; Kidd, K.; Khakoo, S.I.; Alexander, G.; et al. Genetic variation in IL28B and spontaneous clearance of hepatitis C virus. Nat. Cell Boil. 2009, 461, 798–801. [Google Scholar] [CrossRef]
- Rauch, A.; Kutalik, Z.; Descombes, P.; Cai, T.; Di Iulio, J.; Mueller, T.; Bochud, M.; Battegay, M.; Bernasconi, E.; Borovička, J.; et al. Genetic Variation in IL28B Is Associated With Chronic Hepatitis C and Treatment Failure: A Genome-Wide Association Study. Gastroenterology 2010, 138, 1338–1345. [Google Scholar] [CrossRef]
- Randall, G.; Chen, L.; Panis, M.; Fischer, A.K.; Lindenbach, B.D.; Sun, J.; Heathcote, J.; Rice, C.M.; Edwards, A.M.; McGilvray, I.D. WITHDRAWN: Silencing of USP18 potentiates the antiviral activity of interferon against hepatitis C virus infection. Gastroenterology 2006, 131, 1584–1591. [Google Scholar] [CrossRef]
- Kaplan, D.E. Immunopathogenesis of Hepatitis C Virus Infection. Gastroenterol. Clin. 2015, 44, 735–760. [Google Scholar] [CrossRef]
- McCaughan, G.W.; McGuinness, P.H.; Bishop, G.; Painter, D.M.; Lien, A.S.; Tulloch, R.; Wylie, B.R.; Archer, G.T. Clinical assessment and incidence of hepatitis C RNA in 50 consecutive RIBA-positive volunteer blood donors. Med. J. Aust. 1992, 157, 231–233. [Google Scholar]
- National Institutes of Health. National Institutes of Health Consensus Development Conference Statement: Management of hepatitis C: 2002. Hepatology 2002, 36, S3–20. [Google Scholar] [CrossRef]
- Lingala, S.; Ghany, M.G. Natural History of Hepatitis C. Gastroenterol. Clin. N. Am. 2015, 44, 717–734. [Google Scholar] [CrossRef]
- Sherman, A.C.; Sherman, K.E. Extrahepatic Manifestations of Hepatitis C Infection: Navigating CHASM. HIV/AIDS Rep. 2015, 12, 353–361. [Google Scholar] [CrossRef]
- Potthoff, A.; Manns, M.P.; Wedemeyer, H. Treatment of HBV/HCV coinfection. Expert Opin. Pharmacother. 2010, 11, 919–928. [Google Scholar] [CrossRef]
- Lamontagne, R.J.; Bagga, S.; Bouchard, M.J. Hepatitis B virus molecular biology and pathogenesis. Hepatoma Res. 2016, 2, 163–186. [Google Scholar] [CrossRef]
- Zoulim, F.; Saputelli, J.; Seeger, C. Woodchuck hepatitis virus X protein is required for viral infection in vivo. J. Virol. 1994, 68, 2026–2030. [Google Scholar] [PubMed]
- Zeisel, M.B.; Felmlee, D.J.; Baumert, T.F. Hepatitis C virus entry. Curr. Top Microbiol. Immunol. 2013, 369, 87–112. [Google Scholar] [PubMed]
- Moradpour, D.; Penin, F. Hepatitis C Virus Proteins: From Structure to Function. Curr. Top Microbiol. Immunol. 2013, 369, 113–142. [Google Scholar] [PubMed]
- Lohmann, V. Hepatitis C virus RNA replication. Curr. Top Microbiol. Immunol. 2013, 369, 167–198. [Google Scholar]
- Scheel, T.K.H.; Rice, C.M. Understanding the hepatitis C virus life cycle paves the way for highly effective therapies. Nat. Med. 2013, 19, 837–849. [Google Scholar] [CrossRef] [PubMed]
- Lindenbach, B.D. Virion assembly and release. Curr. Top Microbiol. Immunol. 2013, 369, 199–218. [Google Scholar]
- Thimme, R.; Oldach, D.; Chang, K.-M.; Steiger, C.; Ray, S.C.; Chisari, F.V. Determinants of Viral Clearance and Persistence during Acute Hepatitis C Virus Infection. J. Exp. Med. 2001, 194, 1395–1406. [Google Scholar] [CrossRef]
- Hiroishi, K.; Ito, T.; Imawari, M. Immune responses in hepatitis C virus infection and mechanisms of hepatitis C virus persistence. J. Gastroenterol. Hepatol. 2008, 23, 1473–1482. [Google Scholar] [CrossRef]
- Bigger, C.B.; Brasky, K.M.; Lanford, R.E. DNA Microarray Analysis of Chimpanzee Liver during Acute Resolving Hepatitis C Virus Infection. J. Virol. 2001, 75, 7059–7066. [Google Scholar] [CrossRef]
- Foy, E.; Li, K.; Sumpter, R.; Wang, C.; Yoneyama, M.; Fujita, T.; Gale, M.; Loo, Y.-M.; Johnson, C.L.; Fish, P.M.; et al. Control of antiviral defenses through hepatitis C virus disruption of retinoic acid-inducible gene-I signaling. Proc. Natl. Acad. Sci. USA 2005, 102, 2986–2991. [Google Scholar] [CrossRef]
- Saito, T.; Owen, D.M.; Jiang, F.; Marcotrigiano, J.; Gale, M., Jr. Innate immunity induced by composition-dependent RIG-I recognition of Hepatitis C virus RNA. Nat. Cell Boil. 2008, 454, 523–527. [Google Scholar] [CrossRef]
- Stetson, D.B.; Medzhitov, R. Type I interferons in host defense. Immunity 2006, 25, 373–381. [Google Scholar] [CrossRef]
- Israelow, B.; Narbus, C.M.; Sourisseau, M.; Evans, M.J. HepG2 cells mount an effective antiviral interferon-lambda based innate immune response to hepatitis C virus infection. Hepatology 2014, 60, 1170–1179. [Google Scholar] [CrossRef]
- Jiao, B.; Chen, L. The role of USP18 in interferon signaling and inflammation. Cell Death Dis. 2012, 3, e419. [Google Scholar]
- Zhang, X.; Bogunovic, D.; Payelle-Brogard, B.; Francois-Newton, V.; Speer, S.D.; Yuan, C.; Volpi, S.; Li, Z.; Sanal, O.; Mansouri, D.; et al. Human intracellular ISG15 prevents interferon-alpha/beta over-amplification and auto-inflammation. Nature 2015, 517, 89–93. [Google Scholar] [CrossRef]
- Corado, J.; Toro, F.; Rivera, H.; Bianco, N.E.; Deibis, L.; De Sanctis, J.B. Impairment of natural killer (NK) cytotoxic activity in hepatitis C virus (HCV) infection. Clin. Exp. Immunol. 1997, 109, 451–457. [Google Scholar] [CrossRef]
- Khakoo, S.I. HLA and NK Cell Inhibitory Receptor Genes in Resolving Hepatitis C Virus Infection. Science 2004, 305, 872–874. [Google Scholar] [CrossRef]
- Brown, M.G. Vital Involvement of a Natural Killer Cell Activation Receptor in Resistance to Viral Infection. Science 2001, 292, 934–937. [Google Scholar] [CrossRef]
- Golden-Mason, L.; Rosen, H.R.; Golden-Mason, L. Natural killer cells: Primary target for hepatitis C virus immune evasion strategies? Liver Transplant. 2006, 12, 363–372. [Google Scholar] [CrossRef]
- Guidotti, L.G.; Chisari, F.V. Noncytolytic control of viral infections by the innate and adaptive immune response. Annu. Rev. Immunol. 2001, 19, 65–91. [Google Scholar] [CrossRef]
- Lunemann, S.; Schlaphoff, V.; Cornberg, M.; Wedemeyer, H. NK Cells in Hepatitis C: Role in Disease Susceptibility and Therapy. Dig. Dis. 2012, 30, 48–54. [Google Scholar] [CrossRef]
- Doherty, D.G.; O’Farrelly, C. Innate and adaptive lymphoid cells in the human liver. Immunol. Rev. 2000, 174, 5–20. [Google Scholar] [CrossRef]
- Ahmad, A.; Alvarez, F. Role of NK and NKT cells in the immunopathogenesis of HCV-induced hepatitis. J. Leukoc. Boil. 2004, 76, 743–759. [Google Scholar] [CrossRef]
- Swain, M.G. Hepatic NKT cells: Friend or foe? Clin. Sci. (Lond.) 2008, 114, 457–466. [Google Scholar] [CrossRef]
- Lucas, M.; Gadola, S.; Meier, U.; Young, N.T.; Harcourt, G.; Karadimitris, A.; Coumi, N.; Brown, D.; Dusheiko, G.; Cerundolo, V.; et al. Frequency and phenotype of circulating Valpha24/Vbeta11 double-positive natural killer T cells during hepatitis C virus infection. J. Virol. 2003, 77, 2251–2257. [Google Scholar] [CrossRef]
- Deignan, T.; Curry, M.P.; Doherty, D.G.; Golden-Mason, L.; Volkov, Y.; Norris, S.; Nolan, N.; Traynor, O.; McEntee, G.; Hegarty, J.E.; et al. Decrease in hepatic CD56(+) T cells and V alpha 24(+) natural killer T cells in chronic hepatitis C viral infection. J. Hepatol. 2002, 37, 101–108. [Google Scholar] [CrossRef]
- Inoue, M.; Kanto, T.; Miyatake, H.; Itose, I.; Miyazaki, M.; Yakushijin, T.; Sakakibara, M.; Kuzushita, N.; Hiramatsu, N.; Takehara, T.; et al. Enhanced ability of peripheral invariant natural killer T cells to produce IL-13 in chronic hepatitis C virus infection. J. Hepatol. 2006, 45, 190–196. [Google Scholar] [CrossRef]
- Wynn, T.A. IL-13 effector functions. Annu. Rev. Immunol. 2003, 21, 425–456. [Google Scholar] [CrossRef]
- Liu, Y.-J. Dendritic Cell Subsets and Lineages, and Their Functions in Innate and Adaptive Immunity. Cell 2001, 106, 259–262. [Google Scholar] [CrossRef]
- Lechner, F.; Wong, D.K.; Dunbar, P.R.; Chapman, R.; Chung, R.T.; Dohrenwend, P.; Robbins, G.; Phillips, R.; Klenerman, P.; Walker, B.D. Analysis of Successful Immune Responses in Persons Infected with Hepatitis C Virus. J. Exp. Med. 2000, 191, 1499–1512. [Google Scholar] [CrossRef]
- Kinchen, V.J.; Bailey, J.R. Defining Breadth of Hepatitis C Virus Neutralization. Front. Immunol. 2018, 9, 1703. [Google Scholar] [CrossRef]
- Zeisel, M.B.; Fafi-Kremer, S.; Robinet, E.; Habersetzer, F.; Baumert, T.F.; Stoll-Keller, F. Adaptive Immunity to Hepatitis C Virus. Viruses 2009, 1, 276. [Google Scholar] [CrossRef]
- Timpe, J.M.; Stamataki, Z.; Jennings, A.; Hu, K.; Farquhar, M.J.; Harris, H.J.; Schwarz, A.; Desombere, I.; Roels, G.L.; Balfe, P.; et al. Hepatitis C virus cell-cell transmission in hepatoma cells in the presence of neutralizing antibodies. Hepatology 2008, 47, 17–24. [Google Scholar] [CrossRef]
- Araujo, A.C.; Astrakhantseva, I.V.; Fields, H.A.; Kamili, S. Distinguishing acute from chronic hepatitis C virus (HCV) infection based on antibody reactivities to specific HCV structural and nonstructural proteins. J. Clin. Microbiol. 2011, 49, 54–57. [Google Scholar] [CrossRef]
- Filomena, A.; Göpfert, J.C.; Duffy, D.; Pol, S.; Abdel-Hamid, M.; Esmat, G.; Fontanet, A.; Albert, M.L.; Joos, T.O.; Schneiderhan-Marra, N.; et al. Study of the Humoral Immune Response towards HCV Genotype 4 Using a Bead-Based Multiplex Serological Assay. High-Throughput 2017, 6, 15. [Google Scholar] [CrossRef]
- Keck, Z.-Y.; Li, S.H.; Xia, J.; Von Hahn, T.; Balfe, P.; McKeating, J.A.; Witteveldt, J.; Patel, A.H.; Alter, H.; Rice, C.M.; et al. Mutations in Hepatitis C Virus E2 Located outside the CD81 Binding Sites Lead to Escape from Broadly Neutralizing Antibodies but Compromise Virus Infectivity. J. Virol. 2009, 83, 6149–6160. [Google Scholar] [CrossRef]
- Logvinoff, C.; Major, M.E.; Oldach, D.; Heyward, S.; Talal, A.; Balfe, P.; Feinstone, S.M.; Alter, H.; Rice, C.M.; McKeating, J.A. Neutralizing antibody response during acute and chronic hepatitis C virus infection. Proc. Natl. Acad. Sci. USA 2004, 101, 10149–10154. [Google Scholar] [CrossRef]
- Gerlach, J.T.; Diepolder, H.M.; Jung, M.C.; Gruener, N.H.; Schraut, W.W.; Zachoval, R.; Hoffmann, R.; Schirren, C.A.; Santantonio, T.; Pape, G.R. Recurrence of hepatitis C virus after loss of virus-specific CD4(+) T-cell response in acute hepatitis C. Gastroenterology 1999, 117, 933–941. [Google Scholar] [CrossRef]
- Fitzmaurice, K. Klenerman Cellular immunity and acute hepatitis C infection. Curr. Pharmac. Des. 2008, 14, 1666–1677. [Google Scholar] [CrossRef]
- Semmo, N.; Krashias, G.; Willberg, C.; Klenerman, P. Analysis of the relationship between cytokine secretion and proliferative capacity in hepatitis C virus infection. J. Vrial Hepat. 2007, 14, 492–502. [Google Scholar] [CrossRef]
- Jani, A.B.; Hellman, S. Early Prostate Cancer: Hedonic Prices Model of Provider–Patient Interactions and Decisions. Int. J. Radiat. Oncol. 2008, 70, 1158–1168. [Google Scholar] [CrossRef]
- Dustin, L.B. Innate and Adaptive Immune Responses in Chronic HCV infection. Drug Targets 2017, 18, 826–843. [Google Scholar] [CrossRef]
- Thimme, R.; Bukh, J.; Spangenberg, H.C.; Wieland, S.; Pemberton, J.; Steiger, C.; Govindarajan, S.; Purcell, R.H.; Chisari, F.V. Viral and immunological determinants of hepatitis C virus clearance, persistence, and disease. Proc. Natl. Acad. Sci. USA 2002, 99, 15661–15668. [Google Scholar] [CrossRef]
- Lauer, G.M.; Lucas, M.; Timm, J.; Ouchi, K.; Kim, A.Y.; Day, C.L.; Schulze Zur Wiesch, J.; Paranhos-Baccala, G.; Sheridan, I.; Casson, D.R.; et al. Full-breadth analysis of CD8+ T-cell responses in acute hepatitis C virus infection and early therapy. J. Virol. 2005, 79, 12979–12988. [Google Scholar] [CrossRef]
- Lechner, F.; Gruener, N.H.; Urbani, S.; Uggeri, J.; Santantonio, T.; Kammer, A.R.; Cerny, A.; Phillips, R.; Ferrari, C.; Pape, G.R.; et al. CD8+ T lymphocyte responses are induced during acute hepatitis C virus infection but are not sustained. Eur. J. Immunol. 2000, 30, 2479–2487. [Google Scholar] [CrossRef]
- Kaplan, D.E.; Sugimoto, K.; Newton, K.; Valiga, M.E.; Ikeda, F.; Aytaman, A.; Nunes, F.A.; Lucey, M.R.; Vance, B.A.; Vonderheide, R.H.; et al. Discordant Role of CD4 T-Cell Response Relative to Neutralizing Antibody and CD8 T-Cell Responses in Acute Hepatitis C. Gastroenterology 2007, 132, 654–666. [Google Scholar] [CrossRef]
- Grakoui, A.; Shoukry, N.H.; Woollard, D.J.; Han, J.-H.; Hanson, H.L.; Murthy, K.K.; Ghrayeb, J.; Rice, C.M.; Walker, C.M. HCV Persistence and Immune Evasion in the Absence of Memory T Cell Help. Science 2003, 302, 659–662. [Google Scholar] [CrossRef]
- Shoukry, N.H.; Grakoui, A.; Houghton, M.; Chien, D.Y.; Ghrayeb, J.; Reimann, K.A.; Walker, C.M. Memory CD8+ T cells are required for protection from persistent hepatitis C virus infection. J. Exp. Med. 2003, 197, 1645–1655. [Google Scholar] [CrossRef]
- Kanto, T.; Hayashi, N. Immunopathogenesis of Hepatitis C Virus Infection: Multifaceted Strategies Subverting Innate and Adaptive Immunity. Intern. Med. 2006, 45, 183–191. [Google Scholar] [CrossRef]
- Kondo, Y.; Machida, K.; Liu, H.M.; Ueno, Y.; Kobayashi, K.; Wakita, T.; Shimosegawa, T.; Lai, M.M.C. Hepatitis C Virus Infection of T Cells Inhibits Proliferation and Enhances Fas-Mediated Apoptosis by Down-Regulating the Expression of CD44 Splicing Variant 6. J. Infect. Dis. 2009, 199, 726–736. [Google Scholar] [CrossRef]
- Ando, K.; Hiroishi, K.; Kaneko, T.; Moriyama, T.; Muto, Y.; Kayagaki, N.; Yagita, H.; Okumura, K.; Imawari, M. Perforin, Fas/Fas ligand, and TNF-alpha pathways as specific and bystander killing mechanisms of hepatitis C virus-specific human CTL. J. Immunol. 1997, 158, 5283–5291. [Google Scholar]
- Alric, L.; Fort, M.; Izopet, J.; Vinel, J.; Charlet, J.; Selves, J.; Puel, J.; Pascal, J.; Duffaut, M.; Abbal, M. Genes of the major histocompatibility complex class II influence the outcome of hepatitis C virus infection. Gastroenterology 1997, 113, 1675–1681. [Google Scholar] [CrossRef]
- Thursz, M.; Yallop, R.; Goldin, R.; Trepo, C.; Thomas, H.C. Influence of MHC class II genotype on outcome of infection with hepatitis C virus. The Lancet 1999, 354, 2119–2124. [Google Scholar] [CrossRef]
- Zavaglia, C.; Martinetti, M.; Silini, E.; Bottelli, R.; Daielli, C.; Asti, M.; Airoldi, A.; Salvaneschi, L.; Mondelli, M.U.; Ideo, G. Association between HLA class II alleles and protection from or susceptibility to chronic hepatitis C. J. Hepatol. 1998, 28, 1–7. [Google Scholar] [CrossRef]
- Cramp, M.E.; Carucci, P.; Underhill, J.; Naoumov, N.V.; Williams, R.; Donaldson, P.T. Association between HLA class II genotype and spontaneous clearance of hepatitis C viraemia. J. Hepatol. 1998, 29, 207–213. [Google Scholar] [CrossRef]
- Mangia, A.; Gentile, R.; Cascavilla, I.; Margaglione, M.; Villani, M.R.; Stella, F.; Modola, G.; Agostiano, V.; Gaudiano, C.; Andriulli, A. HLA class II favors clearance of HCV infection and progression of the chronic liver damage. J. Hepatol. 1999, 30, 984–989. [Google Scholar] [CrossRef]
- Thio, C.L.; Thomas, D.L.; Goedert, J.J.; Vlahov, D.; Nelson, K.E.; Hilgartner, M.W.; O’Brien, S.J.; Karacki, P.; Marti, D.; Astemborski, J.; et al. Racial Differences in HLA Class II Associations with Hepatitis C Virus Outcomes. J. Infect. Dis. 2001, 184, 16–21. [Google Scholar] [CrossRef]
- McKiernan, S.M.; Hagan, R.; Curry, M.; McDonald, G.S.A.; Kelly, A.; Nolan, N.; Walsh, A.; Hegarty, J.; Lawlor, E.; Kelleher, D. Distinct MHC class I and II alleles are associated with hepatitis C viral clearance, originating from a single source. Hepatology 2004, 40, 108–114. [Google Scholar] [CrossRef]
- Thio, C.L.; Gao, X.; Goedert, J.J.; Vlahov, D.; Nelson, K.E.; Hilgartner, M.W.; O’Brien, S.J.; Karacki, P.; Astemborski, J.; Carrington, M.; et al. HLA-Cw*04 and Hepatitis C Virus Persistence. J. Virol. 2002, 76, 4792–4797. [Google Scholar] [CrossRef]
- Chuang, W.C.-M.; Sarkodie, F.; Brown, C.J.; Brown, J.; Li, C.; Navarrete, C.; Klenerman, P.; Allain, J.-P.; Owusu-Ofori, S.; Owusu-Ofori, S. Protective effect of HLA-B57 on HCV genotype 2 infection in a West African population. J. Med Virol. 2007, 79, 724–733. [Google Scholar] [CrossRef]
- Metchnikoff, E. Der Kampf der Phagocyten gegen Krankeitserreger. Virchows Arch. 1884, 96, 177–195. [Google Scholar]
- Davies, L.C.; Jenkins, S.J.; Allen, J.E.; Taylor, P.R. Tissue-resident macrophages. Nat. Immunol. 2013, 14, 986–995. [Google Scholar] [CrossRef]
- Murray, P.J.; Wynn, T.A. Protective and pathogenic functions of macrophage subsets. Nat. Rev. Immunol. 2011, 11, 723–737. [Google Scholar] [CrossRef]
- Merad, M.; Sathe, P.; Helft, J.; Miller, J.; Mortha, A. The dendritic cell lineage: Ontogeny and function of dendritic cells and their subsets in the steady state and the inflamed setting. Annu. Immunol. 2013, 31, 563–604. [Google Scholar] [CrossRef]
- Steinman, R.M. Decisions About Dendritic Cells: Past, Present, and Future. Annu. Immunol. 2012, 30, 1–22. [Google Scholar] [CrossRef]
- Swiecki, M.; Colonna, M. The multifaceted biology of plasmacytoid dendritic cells. Nat. Rev. Immunol. 2015, 15, 471–485. [Google Scholar] [CrossRef]
- Veglia, F.; Perego, M.; Gabrilovich, D. Myeloid-derived suppressor cells coming of age. Nat. Immunol. 2018, 19, 108–119. [Google Scholar] [CrossRef]
- Noy, R.; Pollard, J.W. Tumor-associated macrophages: From mechanisms to therapy. Immunity 2014, 41, 49–61. [Google Scholar] [CrossRef]
- Biswas, S.K.; Mantovani, A. Macrophage plasticity and interaction with lymphocyte subsets: Cancer as a paradigm. Nat. Immunol. 2010, 11, 889–896. [Google Scholar] [CrossRef]
- Gabrilovich, D.I. Myeloid-Derived Suppressor Cells. Cancer Immunol. Res. 2017, 5, 3–8. [Google Scholar] [CrossRef]
- Gabrilovich, D.I.; Nagaraj, S. Myeloid-derived-suppressor cells as regulators of the immune system. Nat. Rev. Immunol. 2009, 9, 162–174. [Google Scholar] [CrossRef]
- Tacke, R.S.; Lee, H.-C.; Goh, C.; Courtney, J.; Polyak, S.J.; Rosen, H.R.; Hahn, Y.S. Myeloid Suppressor Cells Induced by Hepatitis C Virus Suppress T-Cell Responses Through the Production of Reactive Oxygen Species. Hepatology 2012, 55, 343–353. [Google Scholar] [CrossRef]
- Ren, J.P.; Zhao, J.; Dai, J.; Griffin, J.W.D.; Wang, L.; Wu, X.Y.; Morrison, Z.D.; Li, G.Y.; El Gazzar, M.; Ning, S.B.; et al. Hepatitis C virus-induced myeloid-derived suppressor cells regulate T-cell differentiation and function via the signal transducer and activator of transcription 3 pathway. Immunology 2016, 148, 377–386. [Google Scholar] [CrossRef]
- Zeng, Q.L.; Yang, B.; Sun, H.-Q.; Feng, G.-H.; Jin, L.; Zou, Z.-S.; Zhang, Z.; Zhang, J.-Y.; Wang, F.-S. Myeloid-Derived Suppressor Cells Are Associated with Viral Persistence and Downregulation of TCR xi Chain Expression on CD8(+) T Cells in Chronic Hepatitis C Patients. Mol. Cells 2014, 37, 66–73. [Google Scholar] [CrossRef]
- Wang, L.; Cao, D.; Wang, L.; Zhao, J.; Nguyen, L.N.; Dang, X.; Ji, Y.; Wu, X.Y.; Morrison, Z.D.; Xie, Q.; et al. HCV-associated exosomes promote myeloid-derived suppressor cell expansion via inhibiting miR-124 to regulate T follicular cell differentiation and function. Cell Discov. 2018, 4, 51. [Google Scholar] [CrossRef]
- Goutagny, N.; Fatmi, A.; De Ledinghen, V.; Penin, F.; Couzigou, P.; Inchauspé, G.; Bain, C. Evidence of Viral Replication in Circulating Dendritic Cells during Hepatitis C Virus Infection. J. Infect. Dis. 2003, 187, 1951–1958. [Google Scholar] [CrossRef]
- Navas, M.-C.; Fuchs, A.; Schvoerer, E.; Bohbot, A.; Aubertin, A.-M. Dendritic cell susceptibility to hepatitis C virus genotype 1 infection. J. Med Virol. 2002, 67, 152–161. [Google Scholar] [CrossRef]
- Marukian, S.; Jones, C.T.; Andrus, L.; Evans, M.J.; Ritola, K.D.; Charles, E.D.; Rice, C.M.; Dustin, L.B. Cell culture-produced hepatitis C virus does not infect peripheral blood mononuclear cells. Hepatology 2008, 48, 1843–1850. [Google Scholar] [CrossRef]
- Lozach, P.-Y.; Lortat-Jacob, H.; Lavalette, A.D.L.D.; Staropoli, I.; Foung, S.; Amara, A.; Houles, C.; Fieschi, F.; Schwartz, O.; Virelizier, J.-L.; et al. DC-SIGN and L-SIGN Are High Affinity Binding Receptors for Hepatitis C Virus Glycoprotein E2. J. Boil. Chem. 2003, 278, 20358–20366. [Google Scholar] [CrossRef]
- Cormier, E.G.; Durso, R.J.; Tsamis, F.; Boussemart, L.; Manix, C.; Olson, W.C.; Gardner, J.P.; Dragic, T. L-SIGN (CD209L) and DC-SIGN (0209) mediate transinfection of liver cells by hepatitis C virus. Proc. Natl. Acad. Sci. USA 2004, 101, 14067–14072. [Google Scholar] [CrossRef]
- Kanto, T.; Inoue, M.; Miyatake, H.; Sato, A.; Sakakibara, M.; Yakushijin, T.; Oki, C.; Itose, I.; Hiramatsu, N.; Takehara, T.; et al. Reduced Numbers and Impaired Ability of Myeloid and Plasmacytoid Dendritic Cells to Polarize T Helper Cells in Chronic Hepatitis C Virus Infection. J. Infect. Dis. 2004, 190, 1919–1926. [Google Scholar] [CrossRef]
- Ulsenheimer, A.; Gerlach, J.T.; Jung, M.-C.; Gruener, N.; Wächtler, M.; Backmund, M.; Santantonio, T.; Schraut, W.; Heeg, M.H.J.; Schirren, C.A.; et al. Plasmacytoid dendritic cells in acute and chronic hepatitis C virus infection. Hepatology 2005, 41, 643–651. [Google Scholar] [CrossRef]
- Wertheimer, A.M.; Bakke, A.; Rosen, H.R. Direct enumeration and functional assessment of circulating dendritic cells in patients with liver disease. Hepatology 2004, 40, 335–345. [Google Scholar] [CrossRef]
- Nattermann, J.; Zimmermann, H.; Iwan, A.; Von Lilienfeld-Toal, M.; Leifeld, L.; Nischalke, H.D.; Langhans, B.; Sauerbruch, T.; Spengler, U. Hepatitis C virus E2 and CD81 interaction may be associated with altered trafficking of dendritic cells in chronic hepatitis C. Hepatology 2006, 44, 945–954. [Google Scholar] [CrossRef]
- Kondo, Y.; Ueno, Y.; Kakazu, E.; Kobayashi, K.; Shiina, M.; Tamai, K.; Machida, K.; Inoue, J.; Wakui, Y. Lymphotropic HCV strain can infect human primary naive CD4+ cells and affect their proliferation and IFN-gamma secretion activity. J. Gastroenterol. 2011, 46, 232–241. [Google Scholar] [CrossRef]
- Kondo, Y.; Sung, V.M.; Machida, K.; Liu, M.; Lai, M.M. Hepatitis C virus infects T cells and affects interferon-gamma signaling in T cell lines. Virology 2007, 361, 161–173. [Google Scholar] [CrossRef][Green Version]
- Wang, R.Y.; Bare, P.; De Giorgi, V.; Matsuura, K.; Salam, K.A.; Grandinetti, T.; Schechterly, C.; Alte, H.J. Preferential association of hepatitis C virus with CD19(+) B cells is mediated by complement system. Hepatology 2016, 64, 1900–1910. [Google Scholar] [CrossRef]
- Ito, M.; Masumi, A.; Mochida, K.; Kukihara, H.; Moriishi, K.; Matsuura, Y.; Yamaguchi, K.; Mizuochi, T. Peripheral B Cells May Serve as a Reservoir for Persistent Hepatitis C Virus Infection. J. Innate Immun. 2010, 2, 607–617. [Google Scholar] [CrossRef]
- Sarhan, M.A.; Chen, A.Y.; Michalak, T.I. Differential Expression of Candidate Virus Receptors in Human T Lymphocytes Prone or Resistant to Infection with Patient-Derived Hepatitis C Virus. PLoS ONE 2013, 8, e62159. [Google Scholar] [CrossRef]
- Kondo, Y.; Shimosegawa, T. Direct effects of hepatitis C virus on the lymphoid cells. World J. Gastroenterol. 2013, 19, 7889–7895. [Google Scholar] [CrossRef]
- Wang, Y.; Li, J.; Wang, X.; Ye, L.; Zhou, Y.; Ho, W. Induction of interferon-lambda contributes to Toll-like receptor-3-activated hepatic stellate cell-mediated hepatitis C virus inhibition in hepatocytes. J. Viral. Hepat. 2013, 20, 385–394. [Google Scholar] [CrossRef]
- Giugliano, S.; Kriss, M.; Golden-Mason, L.; Dobrinskikh, E.; Stone, A.E.; Soto-Gutierrez, A.; Mitchell, A.; Khetani, S.R.; Yamane, D.; Stoddard, M.; et al. Hepatitis C virus infection induces autocrine interferon signaling by human liver endothelial cells and release of exosomes, which inhibits viral replication. Gastroenterology 2015, 148, 392–402. [Google Scholar] [CrossRef]
- Wu, J.; Meng, Z.; Jiang, M.; Zhang, E.; Trippler, M.; Broering, R.; Bucchi, A.; Krux, F.; Dittmer, U.; Yang, D.; et al. Toll-like receptor-induced innate immune responses in non-parenchymal liver cells are cell type-specific. Immunology 2010, 129, 363–374. [Google Scholar] [CrossRef]
- Lau, A.H.; Thomson, A.W. Dendritic cells and immune regulation in the liver. Gut 2003, 52, 307–314. [Google Scholar] [CrossRef]
- Neumann, A.U.; Lam, N.P.; Dahari, H.; Gretch, D.R.; Wiley, E.T.; Layden, T.J.; Perelson, A.S. Hepatitis C viral dynamics in vivo and the antiviral efficacy of interferon-alpha therapy. Science 1998, 282, 103–107. [Google Scholar] [CrossRef]
- Bowen, D.G.; Walker, C.M. Mutational escape from CD8+ T cell immunity: HCV evolution, from chimpanzees to man. J. Exp. Med. 2005, 201, 1709–1714. [Google Scholar] [CrossRef]
- Bukh, J.; Miller, R.; Purcell, R. Genetic Heterogeneity of Hepatitis C Virus: Quasispecies and Genotypes. Semin. Liver 1995, 15, 41–63. [Google Scholar] [CrossRef]
- Kato, N.; Ootsuyama, Y.; Sekiya, H.; Ohkoshi, S.; Nakazawa, T.; Hijikata, M.; Shimotohno, K. Genetic drift in hypervariable region 1 of the viral genome in persistent hepatitis C virus infection. J. Virol. 1994, 68, 4776–4784. [Google Scholar]
- Weiner, A.J.; Geysen, H.M.; Christopherson, C.; Hall, J.E.; Mason, T.J.; Saracco, G.; Bonino, F.; Crawford, K.A.; Marion, C.D.; Crawford, K.A. Evidence for immune selection of hepatitis C virus (HCV) putative envelope glycoprotein variants: Potential role in chronic HCV infections. Proc. Natl. Acad. Sci. USA 1992, 89, 3468–3472. [Google Scholar] [CrossRef]
- Puig, M.; Mihalik, K.; Tilton, J.C.; Williams, O.; Merchlinsky, M.; Connors, M.; Feinstone, S.M.; Major, M.E. CD4+ immune escape and subsequent T-cell failure following chimpanzee immunization against hepatitis C virus. Hepatology 2006, 44, 736–745. [Google Scholar] [CrossRef]
- Tester, I.; Smyk-Pearson, S.; Wang, P.; Wertheimer, A.; Yao, E.; Lewinsohn, D.M.; Tavis, J.E.; Rosen, H.R. Immune evasion versus recovery after acute hepatitis C virus infection from a shared source. J. Exp. Med. 2005, 201, 1725–1731. [Google Scholar] [CrossRef]
- Meyer-Olson, D.; Shoukry, N.H.; Brady, K.W.; Kim, H.; Olson, D.P.; Hartman, K.; Shintani, A.K.; Walker, C.M.; Kalams, S.A. Limited T Cell Receptor Diversity of HCV-specific T Cell Responses Is Associated with CTL Escape. J. Exp. Med. 2004, 200, 307–319. [Google Scholar] [CrossRef]
- Seifert, U.; Liermann, H.; Racanelli, V.; Halenius, A.; Wiese, M.; Wedemeyer, H.; Ruppert, T.; Rispeter, K.; Henklein, P.; Sijts, A.; et al. Hepatitis C virus mutation affects proteasomal epitope processing. J. Clin. Investig. 2004, 114, 250–259. [Google Scholar] [CrossRef] [PubMed]
- Gaudieri, S.; Rauch, A.; Park, L.P.; Freitas, E.; Herrmann, S.; Jeffrey, G.; Cheng, W.; Pfafferott, K.; Naidoo, K.; Chapman, R.; et al. Evidence of Viral Adaptation to HLA Class I-Restricted Immune Pressure in Chronic Hepatitis C Virus Infection. J. Virol. 2006, 80, 11094–11104. [Google Scholar] [CrossRef]
- Timm, J.; Li, B.; Daniels, M.G.; Bhattacharya, T.; Reyor, L.L.; Allgaier, R.; Kuntzen, T.; Fischer, W.; Nolan, B.E.; Duncan, J.; et al. Human leukocyte antigen-associated sequence polymorphisms in hepatitis C virus reveal reproducible immune responses and constraints on viral evolution. Hepatology 2007, 46, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Meylan, E.; Curran, J.; Hofmann, K.; Moradpour, D.; Binder, M.; Bartenschlager, R.; Tschopp, J. Cardif is an adaptor protein in the RIG-I antiviral pathway and is targeted by hepatitis C virus. Nat. Cell Boil. 2005, 437, 1167–1172. [Google Scholar] [CrossRef]
- Li, K.; Foy, E.; Ferreon, J.C.; Nakamura, M.; Ferreon, A.C.M.; Ikeda, M.; Ray, S.C.; Gale, M.; Lemon, S.M. Immune evasion by hepatitis C virus NS3/4A protease-mediated cleavage of the Toll-like receptor 3 adaptor protein TRIF. Proc. Natl. Acad. Sci. USA 2005, 102, 2992–2997. [Google Scholar] [CrossRef]
- Li, X.-D.; Sun, L.; Seth, R.B.; Pineda, G.; Chen, Z.J. Hepatitis C virus protease NS3/4A cleaves mitochondrial antiviral signaling protein off the mitochondria to evade innate immunity. Proc. Natl. Acad. Sci. USA 2005, 102, 17717–17722. [Google Scholar] [CrossRef]
- Lin, W.; Choe, W.H.; Hiasa, Y.; Kamegaya, Y.; Blackard, J.T.; Schmidt, E.V.; Chung, R.T. Hepatitis C virus expression suppresses interferon signaling by degrading STAT1. Gastroenterology 2005, 128, 1034–1041. [Google Scholar] [CrossRef]
- Lin, W.; Kim, S.S.; Yeung, E.; Kamegaya, Y.; Blackard, J.T.; Kim, K.A.; Holtzman, M.J.; Chung, R.T. Hepatitis C Virus Core Protein Blocks Interferon Signaling by Interaction with the STAT1 SH2 Domain. J. Virol. 2006, 80, 9226–9235. [Google Scholar] [CrossRef]
- Bode, J.G.; Brenndörfer, E.D.; Häussinger, D. Subversion of innate host antiviral strategies by the hepatitis C virus. Arch. Biochem. Biophys. 2007, 462, 254–265. [Google Scholar] [CrossRef] [PubMed]
- Bode, J.G.; Ludwig, S.; Ehrhardt, C.; Albrecht, U.; Erhardt, A.; Schaper, F.; Heinrich, P.C.; Häussinger, D. IFN-alpha antagonistic activity of HCV core protein involves induction of suppressor of cytokine signaling-3. FASEB J. 2003, 17, 488–490. [Google Scholar] [CrossRef]
- Anthony, D.D.; Yonkers, N.L.; Post, A.B.; Asaad, R.; Heinzel, F.P.; Lederman, M.M.; Lehmann, P.V.; Valdez, H. Selective Impairments in Dendritic Cell-Associated Function Distinguish Hepatitis C Virus and HIV Infection. J. Immunol. 2004, 172, 4907–4916. [Google Scholar] [CrossRef]
- Amjad, M.; Abdel-Haq, N.; Faisal, M.; Kamal, M.; Moudgal, V. Decreased interferon-alpha production and impaired regulatory function of plasmacytoid dendritic cells induced by the hepatitis C virus NS 5 protein. Microbiol. Immunol. 2008, 52, 499–507. [Google Scholar] [CrossRef]
- Lasarte, J.J.; Casares, N.; López-Díaz de Cerio, A.; Baixeras, E.; Labarga, P.; García, N.; Borrás-Cuesta, F.; Prieto, J. Abnormal priming of CD4(+) T cells by dendritic cells expressing hepatitis C virus core and E1 proteins. J. Virol. 2002, 76, 5062–5070. [Google Scholar]
- Waggoner, S.N.; Hall, C.H.; Hahn, Y.S. HCV core protein interaction with gC1q receptor inhibits Th1 differentiation of CD4+ T cells via suppression of dendritic cell IL-12 production. J. Leukoc. Biol. 2007, 82, 1407–1419. [Google Scholar] [CrossRef]
- Kittlesen, D.J.; Chianese-Bullock, K.A.; Yao, Z.Q.; Braciale, T.J.; Hahn, Y.S. Interaction between complement receptor gC1qR and hepatitis C virus core protein inhibits T-lymphocyte proliferation. J. Clin. Investig. 2000, 106, 1239–1249. [Google Scholar] [CrossRef]
- Accapezzato, D.; Accapezzato, D.; De Salvo, M.; Rawson, P.; Cosimi, O.; Lipp, M.; Cerino, A.; Cividini, A.; Mondelli, M.U.; Barnaba, V. Subversion of effector CD8+ T cell differentiation in acute hepatitis C virus infection: The role of the virus. Eur. J. Immunol. 2004, 34, 438–446. [Google Scholar] [CrossRef]
- Zimmermann, M.; Flechsig, C.; La Monica, N.; Tripodi, M.; Adler, G.; Dikopoulos, N. Hepatitis C virus core protein impairs in vitro priming of specific T cell responses by dendritic cells and hepatocytes. J. Hepatol. 2008, 48, 51–60. [Google Scholar] [CrossRef]
- Song, X.; Yao, Z.; Yang, J.; Zhang, Z.; Deng, Y.; Li, M.; Ma, C.; Yang, L.; Gao, X.; Li, W.; et al. HCV core protein binds to gC1qR to induce A20 expression and inhibit cytokine production through MAPKs and NF-kappaB signaling pathways. Oncotarget 2016, 7, 33796–33808. [Google Scholar]
- Tseng, C.-T.K.; Klimpel, G.R. Binding of the hepatitis C virus envelope protein E2 to CD81 inhibits natural killer cell functions. J. Exp. Med. 2002, 195, 43–49. [Google Scholar] [CrossRef]
- Yoon, J.C.; Shiina, M.; Ahlenstiel, G.; Rehermann, B. Natural killer cell function is intact after direct exposure to infectious hepatitis C virions. Hepatology 2009, 49, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Nattermann, J.; Nischalke, H.D.; Hofmeister, V.; Ahlenstiel, G.; Zimmermann, H.; Leifeld, L.; Weiss, E.H.; Sauerbruch, T.; Spengler, U. The HLA-A2 Restricted T Cell Epitope HCV Core35–44 Stabilizes HLA-E Expression and Inhibits Cytolysis Mediated by Natural Killer Cells. Am. J. Pathol. 2005, 166, 443–453. [Google Scholar] [CrossRef]
- Herzer, K.; Falk, C.S.; Encke, J.; Eichhorst, S.T.; Ulsenheimer, A.; Seliger, B.; Krammer, P.H. Upregulation of Major Histocompatibility Complex Class I on Liver Cells by Hepatitis C Virus Core Protein via p53 and TAP1 Impairs Natural Killer Cell Cytotoxicity†. J. Virol. 2003, 77, 8299–8309. [Google Scholar] [CrossRef] [PubMed]
- Konan, K.V.; Giddings, J.T.H.; Ikeda, M.; Li, K.; Lemon, S.M.; Kirkegaard, K.; Giddings, T.H. Nonstructural Protein Precursor NS4A/B from Hepatitis C Virus Alters Function and Ultrastructure of Host Secretory Apparatus. J. Virol. 2003, 77, 7843–7855. [Google Scholar] [CrossRef] [PubMed]
- Siavoshian, S.; Abraham, J.D.; Thumann, C.; Kieny, M.P.; Schuster, C. Hepatitis C virus core, NS3, NS5A, NS5B proteins induce apoptosis in mature dendritic cells. J. Med Virol. 2005, 75, 402–411. [Google Scholar] [CrossRef]
- Moorman, J.P.; Prayther, D.; McVay, D.; Hahn, Y.S.; Hahn, C.S. The C-terminal region of hepatitis C core protein is required for Fas-ligand independent apoptosis in Jurkat cells by facilitating Fas oligomerization. Virology 2003, 312, 320–329. [Google Scholar] [CrossRef]
- Soguero, C.; Joo, M.; Chianese-Bullock, K.A.; Nguyen, D.T.; Tung, K.; Hahn, Y.S.; Malaspina, A.; Moir, S.; Nickle, D.C.; Donoghue, E.T.; et al. Hepatitis C Virus Core Protein Leads to Immune Suppression and Liver Damage in a Transgenic Murine Model. J. Virol. 2002, 76, 9345–9354. [Google Scholar] [CrossRef] [PubMed]
- Saito, K.; Meyer, K.; Warner, R.; Basu, A.; Ray, R.B.; Ray, R. Hepatitis C Virus Core Protein Inhibits Tumor Necrosis Factor Alpha-Mediated Apoptosis by a Protective Effect Involving Cellular FLICE Inhibitory Protein. J. Virol. 2006, 80, 4372–4379. [Google Scholar] [CrossRef]
- Lee, S.H.; Kim, Y.K.; Kim, C.S.; Seol, S.K.; Kim, J.; Cho, S.; Song, Y.L.; Bartenschlager, R.; Jang, S.K. E2 of Hepatitis C Virus Inhibits Apoptosis. J. Immunol. 2005, 175, 8226–8235. [Google Scholar] [CrossRef]
- Harizi, H. Reciprocal crosstalk between dendritic cells and natural killer cells under the effects of PGE2 in immunity and immunopathology. Cell. Mol. Immunol. 2013, 10, 213–221. [Google Scholar] [CrossRef]
- Jinushi, M.; Takehara, T.; Kanto, T.; Tatsumi, T.; Groh, V.; Spies, T.; Miyagi, T.; Suzuki, T.; Sasaki, Y.; Hayashi, N. Critical role of MHC class I-related chain A and B expression on IFN-alpha-stimulated dendritic cells in NK cell activation: Impairment in chronic hepatitis C virus infection. J. Immunol. 2003, 170, 1249–1256. [Google Scholar] [CrossRef]
- Jinushi, M.; Takehara, T.; Tatsumi, T.; Kanto, T.; Miyagi, T.; Suzuki, T.; Kanazawa, Y.; Hiramatsu, N.; Hayashi, N. Negative Regulation of NK Cell Activities by Inhibitory Receptor CD94/NKG2A Leads to Altered NK Cell-Induced Modulation of Dendritic Cell Functions in Chronic Hepatitis C Virus Infection. J. Immunol. 2004, 173, 6072–6081. [Google Scholar] [CrossRef]
- Nattermann, J.; Feldmann, G.; Ahlenstiel, G.; Langhans, B.; Sauerbruch, T.; Spengler, U. Surface expression and cytolytic function of natural killer cell receptors is altered in chronic hepatitis C. Gut 2006, 55, 869–877. [Google Scholar] [CrossRef][Green Version]
- De Maria, A.; Fogli, M.; Mazza, S.; Basso, M.; Picciotto, A.; Costa, P.; Congia, S.; Mingari, M.C.; Moretta, L. Increased natural cytotoxicity receptor expression and relevant IL-10 production in NK cells from chronically infected viremic HCV patients. Eur. J. Immunol. 2007, 37, 445–455. [Google Scholar] [CrossRef]
- Bonavita, M.S.; Franco, A.; Paroli, M.; Santilio, I.; Benvenuto, R.; De Petrillo, G.; Levrero, M.; Perrone, A.; Balsano, C.; Barnaba, V. Normalization of depressed natural killer activity after interferon-alpha therapy is associated with a low frequency of relapse in patients with chronic hepatitis C. Int. J. Tissue. React. 1993, 15, 11–16. [Google Scholar]
- Goh, C.C.; Roggerson, K.M.; Lee, H.C.; Golden-Mason, L.; Rosen, H.R.; Hahn, Y.S. Hepatitis C Virus-Induced Myeloid-Derived Suppressor Cells Suppress NK Cell IFN-gamma Production by Altering Cellular Metabolism via Arginase-1. J. Immunol. 2016, 196, 2283–2292. [Google Scholar] [CrossRef]
- Murakami, H.; Akbar, S.M.F.; Matsui, H.; Horiike, N.; Onji, M. Decreased interferon-alpha production and impaired T helper 1 polarization by dendritic cells from patients with chronic hepatitis C. Clin. Exp. Immunol. 2004, 137, 559–565. [Google Scholar] [CrossRef]
- Della Bella, S.; Crosignani, A.; Riva, A.; Presicce, P.; Benetti, A.; Longhi, R.; Podda, M.; Villa, M.L. Decrease and dysfunction of dendritic cells correlate with impaired hepatitis C virus-specific CD4+ T-cell proliferation in patients with hepatitis C virus infection. Immunology 2007, 121, 283–292. [Google Scholar] [CrossRef]
- Auffermann-Gretzinger, S. Impaired dendritic cell maturation in patients with chronic, but not resolved, hepatitis C virus infection. Blood 2001, 97, 3171–3176. [Google Scholar] [CrossRef]
- Henderson, E.A.; Louie, T.J.; Ramotar, K.; Ledgerwood, D.; Hope, K.M.; Kennedy, A. Comparison of Higher-Dose Intradermal Hepatitis B Vaccination to Standard Intramuscular Vaccination of Healthcare Workers. Infect. Hosp. Epidemiol. 2000, 21, 264–269. [Google Scholar] [CrossRef]
- Ebihara, T.; Shingai, M.; Matsumoto, M.; Wakita, T.; Seya, T. Hepatitis C virus–infected hepatocytes extrinsically modulate dendritic cell maturation to activate T cells and natural killer cells. Hepatology 2008, 48, 48–58. [Google Scholar] [CrossRef]
- Kanto, T.; Inoue, M.; Miyazaki, M.; Itose, I.; Miyatake, H.; Sakakibara, M.; Yakushijin, T.; Kaimori, A.; Oki, C.; Hiramatsu, N.; et al. Impaired function of dendritic cells circulating in patients infected with hepatitis C virus who have persistently normal alanine aminotransferase levels. Intervirology 2006, 49, 58–63. [Google Scholar] [CrossRef]
- Harari, A.; Dutoit, V.; Cellerai, C.; Bart, P.-A.; Du Pasquier, R.A.; Pantaleo, G. Functional signatures of protective antiviral T-cell immunity in human virus infections. Immunol. Rev. 2006, 211, 236–254. [Google Scholar] [CrossRef]
- Ha, S.-J.; West, E.E.; Araki, K.; Smith, K.A.; Ahmed, R. Manipulating both the inhibitory and stimulatory immune system towards the success of therapeutic vaccination against chronic viral infections. Immunol. Rev. 2008, 223, 317–333. [Google Scholar] [CrossRef]
- Blattman, J.N.; Murali-Krishna, K.; Sourdive, D.J.; Zajac, A.J.; Suresh, M.; Altman, J.D.; Ahmed, R. Viral Immune Evasion Due to Persistence of Activated T Cells Without Effector Function. J. Exp. Med. 1998, 188, 2205–2213. [Google Scholar]
- Wherry, E.J.; Blattman, J.N.; Murali-Krishna, K.; Van Der Most, R.; Ahmed, R. Viral Persistence Alters CD8 T-Cell Immunodominance and Tissue Distribution and Results in Distinct Stages of Functional Impairment. J. Virol. 2003, 77, 4911–4927. [Google Scholar] [CrossRef]
- Wherry, E.J.; Ha, S.J.; Kaech, S.M.; Haining, W.N.; Sarkar, S.; Kalia, V.; Subramaniam, S.; Blattman, J.N.; Barber, D.L.; Ahmed, R. Molecular signature of CD8+ T cell exhaustion during chronic viral infection. Immunity 2007, 27, 670–684. [Google Scholar] [CrossRef]
- Gruener, N.H.; Lechner, F.; Jung, M.-C.; Diepolder, H.; Gerlach, T.; Lauer, G.; Walker, B.; Sullivan, J.; Phillips, R.; Pape, G.R.; et al. Sustained dysfunction of antiviral CD8+ T lymphocytes after infection with hepatitis C virus. J. Virol. 2001, 75, 5550–5558. [Google Scholar] [CrossRef]
- Wedemeyer, H.; He, X.S.; Nascimbeni, M.; Davis, A.R.; Greenberg, H.B.; Hoofnagle, J.H.; Liang, T.J.; Alter, H.; Rehermann, B. Impaired effector function of hepatitis C virus-specific CD8+ T cells in chronic hepatitis C virus infection. J. Immunol. 2002, 169, 3447–3458. [Google Scholar] [CrossRef]
- Missale, G.; Cariani, E.; Ferrari, C. Role of viral and host factors in HCV persistence: Which lesson for therapeutic and preventive strategies? Dig. Liver 2004, 36, 703–711. [Google Scholar] [CrossRef]
- Radziewicz, H.; Ibegbu, C.C.; Hon, H.; Osborn, M.K.; Obideen, K.; Wehbi, M.; Freeman, G.J.; Lennox, J.L.; Workowski, K.A.; Hanson, H.L.; et al. Impaired hepatitis C virus (HCV)-specific effector CD8+ T cells undergo massive apoptosis in the peripheral blood during acute HCV infection and in the liver during the chronic phase of infection. J. Virol. 2008, 82, 9808–9822. [Google Scholar] [CrossRef]
- Bengsch, B.; Seigel, B.; Ruhl, M.; Timm, J.; Kuntz, M.; Blum, H.E.; Pircher, H.; Thimme, R. Coexpression of PD-1, 2B4, CD160 and KLRG1 on exhausted HCV-specific CD8+ T cells is linked to antigen recognition and T cell differentiation. PLoS Pathog. 2010, 6, e1000947. [Google Scholar] [CrossRef]
- Barber, D.L.; Wherry, E.J.; Masopust, D.; Zhu, B.; Allison, J.P.; Sharpe, A.H.; Freeman, G.J.; Ahmed, R. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 2006, 439, 682–687. [Google Scholar] [CrossRef]
- Blackburn, S.D.; Shin, H.; Haining, W.N.; Zou, T.; Workman, C.J.; Polley, A.; Betts, M.R.; Freeman, G.J.; Vignali, D.A.; Wherry, E.J. Coregulation of CD8(+) T cell exhaustion by multiple inhibitory receptors during chronic viral infection. Nat. Immunol. 2009, 10, 29–37. [Google Scholar] [CrossRef]
- Okazaki, T.; Honjo, T. The PD-1–PD-L pathway in immunological tolerance. Nat. Immunol. 2006, 27, 195–201. [Google Scholar] [CrossRef]
- Urbani, S.; Amadei, B.; Tola, D.; Massari, M.; Schivazappa, S.; Missale, G.; Ferrari, C. PD-1 Expression in Acute Hepatitis C Virus (HCV) Infection Is Associated with HCV-Specific CD8 Exhaustion. J. Virol. 2006, 80, 11398–11403. [Google Scholar] [CrossRef] [PubMed]
- Penna, A.; Pilli, M.; Zerbini, A.; Orlandini, A.; Mezzadri, S.; Sacchelli, L.; Missale, G.; Ferrari, C. Dysfunction and functional restoration of HCV-specific CD8 responses in chronic hepatitis C virus infection. Hepatology 2007, 45, 588–601. [Google Scholar] [CrossRef]
- Radziewicz, H.; Ibegbu, C.C.; Fernandez, M.L.; Workowski, K.A.; Obideen, K.; Wehbi, M.; Hanson, H.L.; Steinberg, J.P.; Masopust, D.; Wherry, E.J.; et al. Liver-infiltrating lymphocytes in chronic human hepatitis C virus infection display an exhausted phenotype with high levels of PD-1 and low levels of CD127 expression. J. Virol. 2007, 81, 2545–2553. [Google Scholar] [CrossRef]
- Rutebemberwa, A.; Ray, S.; Astemborski, J.; Levine, J.; Liu, L.; Dowd, K.A.; Clute, S.; Wang, C.; Korman, A.; Sette, A.; et al. High-Programmed Death-1 Levels on Hepatitis C Virus-Specific T Cells during Acute Infection Are Associated with Viral Persistence and Require Preservation of Cognate Antigen during Chronic Infection. J. Immunol. 2008, 181, 8215–8225. [Google Scholar] [CrossRef]
- Wherry, E.J.; Barber, D.L.; Kaech, S.M.; Blattman, J.N.; Ahmed, R. Antigen-independent memory CD8 T cells do not develop during chronic viral infection. Proc. Natl. Acad. Sci. USA 2004, 101, 16004–16009. [Google Scholar] [CrossRef] [PubMed]
- Lang, K.S.; Recher, M.; Navarini, A.A.; Harris, N.L.; Löhning, M.; Junt, T.; Probst, H.C.; Hengartner, H.; Zinkernagel, R.M. Inverse correlation between IL-7 receptor expression and CD8 T cell exhaustion during persistent antigen stimulation. Eur. J. Immunol. 2005, 35, 738–745. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.W.; de Waal Malefyt, R.; Coffman, R.L.; O’Garra, A. Interleukin-10 and the interleukin-10 receptor. Annu. Rev. Immunol. 2001, 19, 683–765. [Google Scholar] [CrossRef] [PubMed]
- O’Garra, A.; Vieira, P. T(H)1 cells control themselves by producing interleukin-10. Nat. Rev. Immunol. 2007, 7, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Accapezzato, D.; Francavilla, V.; Paroli, M.; Casciaro, M.; Chircu, L.V.; Cividini, A.; Abrignani, S.; Mondelli, M.U.; Barnaba, V. Hepatic expansion of a virus-specific regulatory CD8(+) T cell population in chronic hepatitis C virus infection. J. Clin. Invest. 2004, 113, 963–972. [Google Scholar] [CrossRef] [PubMed]
- Rigopoulou, E.I.; Abbott, W.G.; Haigh, P.; Naoumov, N.V. Blocking of interleukin-10 receptor—a novel approach to stimulate T-helper cell type 1 responses to hepatitis C virus. Clin. Immunol. 2005, 117, 57–64. [Google Scholar] [CrossRef]
- Brooks, D.G.; Trifilo, M.J.; Edelmann, K.H.; Teyton, L.; McGAVERN, D.B.; Oldstone, M.B. Interleukin-10 determines viral clearance or persistence in vivo. Nat. Med. 2006, 12, 1301–1309. [Google Scholar] [CrossRef]
- Ejrnaes, M.; Filippi, C.M.; Martinic, M.M.; Ling, E.M.; Togher, L.M.; Crotty, S.; Von Herrath, M.G. Resolution of a chronic viral infection after interleukin-10 receptor blockade. J. Exp. Med. 2006, 203, 2461–2472. [Google Scholar] [CrossRef]
- Kaplan, D.E.; Ikeda, F.; Li, Y.; Nakamoto, N.; Ganesan, S.; Valiga, M.E.; Nunes, F.A.; Reddy, K.R.; Chang, K.-M. Peripheral virus-specific T-cell Interleukin-10 responses develop early in acute hepatitis C infection and become dominant in chronic hepatitis. J. Hepatol. 2008, 48, 903–913. [Google Scholar] [CrossRef]
- Cabrera, R.; Tu, Z.; Xu, Y.; Firpi, R.J.; Rosen, H.R.; Liu, C.; Nelson, D.R. An immunomodulatory role for CD4(+)CD25(+) regulatory T lymphocytes in hepatitis C virus infection. Hepatology 2004, 40, 1062–1071. [Google Scholar] [CrossRef]
- Sugimoto, K. Suppression of HCV-specific T cells without differential hierarchy demonstrated ex vivo in persistent HCV infection. Hepatology 2003, 38, 1437–1448. [Google Scholar] [CrossRef]
- Ward, S.M.; Fox, B.C.; Brown, P.J.; Worthington, J.; Fox, S.B.; Chapman, R.W.; Fleming, K.A.; Banham, A.H.; Klenerman, P. Quantification and localisation of FOXP3+ T lymphocytes and relation to hepatic inflammation during chronic HCV infection. J. Hepatol. 2007, 47, 316–324. [Google Scholar] [CrossRef]
- Sakaki, M.; Hiroishi, K.; Baba, T.; Ito, T.; Hirayama, Y.; Saito, K.; Tonoike, T.; Kushima, M.; Imawari, M. Intrahepatic status of regulatory T cells in autoimmune liver diseases and chronic viral hepatitis. Hepatol. Res. 2008, 38, 354–361. [Google Scholar] [CrossRef]
- Keynan, Y.; Card, C.M.; McLaren, P.J.; Dawood, M.R.; Kasper, K.; Fowke, K.R. The Role of Regulatory T Cells in Chronic and Acute Viral Infections. Clin. Infect. Dis. 2008, 46, 1046–1052. [Google Scholar] [CrossRef]
- Romano, K.P.; Ali, A.; Aydin, C.; Soumana, D.; Özen, A.; Deveau, L.M.; Silver, C.; Cao, H.; Newton, A.; Petropoulos, C.J.; et al. The Molecular Basis of Drug Resistance against Hepatitis C Virus NS3/4A Protease Inhibitors. PLOS Pathog. 2012, 8, e1002832. [Google Scholar] [CrossRef]
- Fried, M.W. Side effects of therapy of hepatitis C and their management. Hepatology 2002, 36, s237–s244. [Google Scholar]
- DrugBank. Available online: https://www.drugbank.ca/ (accessed on 8 November 2017).
- Gieling, R.G.; Wallace, K.; Han, Y.-P. Interleukin-1 participates in the progression from liver injury to fibrosis. Am. J. Physiol. Liver Physiol. 2009, 296, G1324–G1331. [Google Scholar] [CrossRef]
- Negash, A.A.; Ramos, H.J.; Crochet, N.; Lau, D.T.Y.; Doehle, B.; Papic, N.; Delker, D.A.; Jo, J.; Bertoletti, A.; Hagedorn, C.H.; et al. IL-1beta production through the NLRP3 inflammasome by hepatic macrophages links hepatitis C virus infection with liver inflammation and disease. PLoS Pathog. 2013, 9, e1003330. [Google Scholar] [CrossRef]
- Serti, E.; Chepa-Lotrea, X.; Kim, Y.J.; Keane, M.; Fryzek, N.; Liang, T.J.; Ghany, M.; Rehermann, B. Successful Interferon-Free Therapy of Chronic Hepatitis C Virus Infection Normalizes Natural Killer Cell Function. Gastroenterology 2015, 149, 190–200. [Google Scholar] [CrossRef]
- Burchill, M.A.; Roby, J.A.; Crochet, N.; Wind-Rotolo, M.; Stone, A.E.; Edwards, M.G.; Dran, R.J.; Kriss, M.S.; Gale, M.; Rosen, H.R. Rapid reversal of innate immune dysregulation in blood of patients and livers of humanized mice with HCV following DAA therapy. PLoS ONE 2017, 12, e0186213. [Google Scholar] [CrossRef]
- Alao, H.; Cam, M.; Keembiyehetty, C.; Zhang, F.; Serti, E.; Suarez, D.; Park, H.; Fourie, N.H.; Wright, E.C.; Henderson, W.A.; et al. Baseline Intrahepatic and Peripheral Innate Immunity are Associated with Hepatitis C Virus Clearance During Direct-Acting Antiviral Therapy. Hepatology 2018, 68, 2078–2088. [Google Scholar] [CrossRef]
- Martin, B.; Hennecke, N.; Lohmann, V.; Kayser, A.; Neumann-Haefelin, C.; Kukolj, G.; Böcher, W.O.; Thimme, R. Restoration of HCV-specific CD8+ T cell function by interferon-free therapy. J. Hepatol. 2014, 61, 538–543. [Google Scholar] [CrossRef]
- Van der Ree, M.H.; Stelma, F.; Willemse, S.B.; Brown, A.; Swadling, L.; van der Valk, M.; Sinnige, M.J.; van Nuenen, A.C.; de Vree, J.M.L.; Klenerman, P.; et al. Immune responses in DAA treated chronic hepatitis C patients with and without prior RG-101 dosing. Antiviral Res. 2017, 146, 139–145. [Google Scholar] [CrossRef]
- Guo, X.; Zhong, J.-Y.; Li, J.-W. Hepatitis C Virus Infection and Vaccine Development. J. Clin. Exp. Hepatol. 2018, 8, 195–204. [Google Scholar] [CrossRef]
- Callendret, B.; Eccleston, H.B.; Satterfield, W.; Capone, S.; Folgori, A.; Cortese, R.; Nicosia, A.; Walker, C.M. Persistent hepatitis C viral replication despite priming of functional CD8+ T cells by combined therapy with a vaccine and a direct-acting antiviral. Hepatology 2016, 63, 1442–1454. [Google Scholar] [CrossRef]
- Osburn, W.O.; Snider, A.E.; Wells, B.L.; Latanich, R.; Bailey, J.R.; Thomas, D.L.; Cox, A.L.; Ray, S.C. Clearance of hepatitis C infection is associated with the early appearance of broad neutralizing antibody responses. Hepatology 2014, 59, 2140–2151. [Google Scholar] [CrossRef]
- Raghuraman, S.; Park, H.; Osburn, W.O.; Winkelstein, E.; Edlin, B.R.; Rehermann, B. Spontaneous Clearance of Chronic Hepatitis C Virus Infection Is Associated with Appearance of Neutralizing Antibodies and Reversal of T-Cell Exhaustion. J. Infect. Dis. 2012, 205, 763–771. [Google Scholar] [CrossRef]
- Fauvelle, C.; Colpitts, C.C.; Keck, Z.-Y.; Pierce, B.G.; Foung, S.K.H.; Baumert, T.F. Hepatitis C virus vaccine candidates inducing protective neutralizing antibodies. Expert Rev. Vaccines 2016, 15, 1535–1544. [Google Scholar] [CrossRef]
- Shoukry, N.H. Hepatitis C Vaccines, Antibodies, and T Cells. Front. Immunol. 2018, 9, 9. [Google Scholar] [CrossRef]
- Takaki, A.; Wiese, M.; Maertens, G.; Depla, E.; Seifert, U.; Liebetrau, A.; Miller, J.L.; Manns, M.P.; Rehermann, B. Cellular immune responses persist and humoral responses decrease two decades after recovery from a single-source outbreak of hepatitis C. Nat. Med. 2000, 6, 578–582. [Google Scholar] [CrossRef]
- Abdel-Hakeem, M.S.; Bédard, N.; Murphy, D.; Bruneau, J.; Shoukry, N.H. Signatures of protective memory immune responses during hepatitis C virus reinfection. Gastroenterology 2014, 147, 870–881. [Google Scholar] [CrossRef]
- Law, J.L.M.; Chen, C.; Wong, J.; Hockman, D.; Santer, D.M.; Frey, S.E.; Belshe, R.B.; Wakita, T.; Bukh, J.; Jones, C.T.; et al. A Hepatitis C Virus (HCV) Vaccine Comprising Envelope Glycoproteins gpE1/gpE2 Derived from a Single Isolate Elicits Broad Cross-Genotype Neutralizing Antibodies in Humans. PLoS ONE 2013, 8, e59776. [Google Scholar] [CrossRef]
- Swadling, L.; Capone, S.; Antrobus, R.D.; Brown, A.; Richardson, R.; Newell, E.W.; Halliday, J.; Kelly, C.; Bowen, D.; Fergusson, J.; et al. A human vaccine strategy based on chimpanzee adenoviral and MVA vectors that primes, boosts, and sustains functional HCV-specific T cell memory. Sci. Transl. Med. 2014, 6, 261ra153. [Google Scholar] [CrossRef]
- Walker, C.M.; Grakoui, A. Hepatitis C virus: Why do we need a vaccine to prevent a curable persistent infection? Curr. Opin. Immunol. 2015, 35, 137–143. [Google Scholar] [CrossRef]
- Keck, Z.-Y.; Girard-Blanc, C.; Wang, W.; Lau, P.; Zuiani, A.; Rey, F.A.; Krey, T.; Diamond, M.S.; Foung, S.K.H. Antibody Response to Hypervariable Region 1 Interferes with Broadly Neutralizing Antibodies to Hepatitis C Virus. J. Virol. 2016, 90, 3112–3122. [Google Scholar] [CrossRef]
- Brimacombe, C.L.; Grove, J.; Meredith, L.W.; Hu, K.; Syder, A.J.; Flores, M.V.; Timpe, J.M.; Krieger, S.E.; Baumert, T.F.; Tellinghuisen, T.L.; et al. Neutralizing antibody-resistant hepatitis C virus cell-to-cell transmission. J. Virol. 2011, 85, 596–605. [Google Scholar] [CrossRef]
- Andre, P.; Komurian-Pradel, F.; Deforges, S.; Perret, M.; Berland, J.L.; Sodoyer, M.; Pol, S.; Bréchot, C.; Paranhos-Baccalà, G.; Lotteau, V.; et al. Characterization of Low- and Very-Low-Density Hepatitis C Virus RNA-Containing Particles. J. Virol. 2002, 76, 6919–6928. [Google Scholar] [CrossRef]
- Sheridan, D.A.; Hajarizadeh, B.; I Fenwick, F.; Matthews, G.V.; Applegate, T.; Douglas, M.; Neely, D.; Askew, B.; Dore, G.J.; Lloyd, A.R.; et al. Maximum levels of hepatitis C virus lipoviral particles are associated with early and persistent infection. Liver Int. 2016, 36, 1774–1782. [Google Scholar] [CrossRef]
- Keck, Z.-Y.; Xia, J.; Wang, Y.; Wang, W.; Krey, T.; Prentoe, J.; Carlsen, T.; Li, A.Y.-J.; Patel, A.H.; Lemon, S.M.; et al. Human Monoclonal Antibodies to a Novel Cluster of Conformational Epitopes on HCV E2 with Resistance to Neutralization Escape in a Genotype 2a Isolate. PLOS Pathog. 2012, 8, e1002653. [Google Scholar] [CrossRef]
| Class of DAA | DAA (Targeted Genotypes in Brackets) |
|---|---|
| NS3/4A Protease Inhibitors (PIs) | Glecaprevir (1–6) Voxilaprevir (1–6) Galexos (1) Grazoprevir (1, 3, 4) Sunvepra (1, 4) |
| Nucleoside and Nucleotide NS5B Polymerase Inhibitors | Sofosbuvir (1–4) |
| NS5A Inhibitors | Ombitasvir (1, 4) Pibrentasvir (1–6) Daclatasvir (3) Elbasvir (1, 4) Ombitasvir (1) Velpatasvir (1–6) |
| Non-Nucleoside NS5B Polymerase Inhibitors | Dasabuvir (1) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chigbu, D.I.; Loonawat, R.; Sehgal, M.; Patel, D.; Jain, P. Hepatitis C Virus Infection: Host–Virus Interaction and Mechanisms of Viral Persistence. Cells 2019, 8, 376. https://doi.org/10.3390/cells8040376
Chigbu DI, Loonawat R, Sehgal M, Patel D, Jain P. Hepatitis C Virus Infection: Host–Virus Interaction and Mechanisms of Viral Persistence. Cells. 2019; 8(4):376. https://doi.org/10.3390/cells8040376
Chicago/Turabian StyleChigbu, DeGaulle I., Ronak Loonawat, Mohit Sehgal, Dip Patel, and Pooja Jain. 2019. "Hepatitis C Virus Infection: Host–Virus Interaction and Mechanisms of Viral Persistence" Cells 8, no. 4: 376. https://doi.org/10.3390/cells8040376
APA StyleChigbu, D. I., Loonawat, R., Sehgal, M., Patel, D., & Jain, P. (2019). Hepatitis C Virus Infection: Host–Virus Interaction and Mechanisms of Viral Persistence. Cells, 8(4), 376. https://doi.org/10.3390/cells8040376





