Gynecological Cancers Translational, Research Implementation, and Harmonization: Gynecologic Cancer InterGroup Consensus and Still Open Questions
Abstract
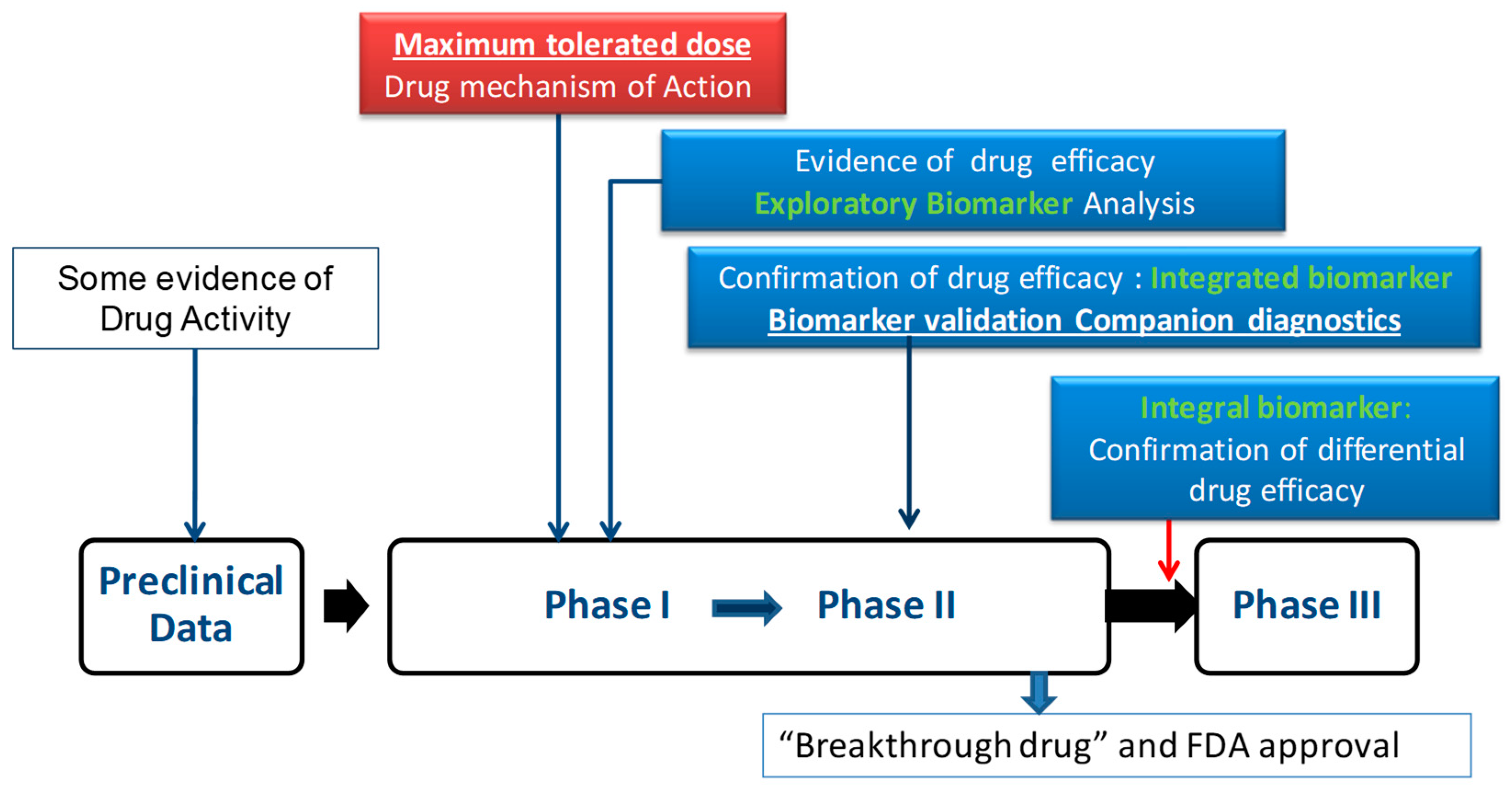
1. Introduction
2. Precision Medicine in Gynecological Cancer
- Translational studies can add complexity and may impact on recruitment to the clinical trial.
- Clear definition of the hypotheses driving correlative studies is essential.
- The trial must be adequately designed and sufficiently powered to address the translational hypotheses.
2.1. Biomarkers: Definitions and Applications
2.2. Biospecimen, Quality Control, and Validation
2.3. Improvement of Biomarker Development
- Assess longitudinally, cancer cell clonal evolution and dynamics in cohorts or individual cancer patients and to capture heterogeneity in space and time [9].
- Guide the use of molecular-targeted therapies and provide critical insight into mechanisms of treatment failure and resistance in cancer patients.
3. Immunoprofiling of Gynecological Cancers: Towards a New Class of Biomarkers?
3.1. Tumor-Infiltrating Immune Cells
3.2. Tumor-Specific Factors
3.3. Tumor Antigenicity
3.4. Reactivating the Immune System
3.5. Microenvironment and Immune Responses
4. Overcoming Roadblocks to Completing Translational Goals
- Pathology and biomarker research should be integral to clinical trial design to reduce the likelihood that a trial produces a negative result without mechanistic information provided.
- Mandatory upfront central collection of pathologically reviewed patients’ tissues is to be preferred to ensure the generation of optimal trial-associated tumor biobanks with complete clinical annotation. Efforts should be made to create a harmonization program of Standard Operating Procedures (SOPs) to be shared across trial centers and across GCIG members as a set of documented guidelines taking into account issues related to sample collection, curation, and clinical data annotation.
- When planning a collaborative translational study, two different scenarios could be hypothesized, a virtual vs. physical biobanking, each of them requiring different costs, management, and governance. In the process of selection of the most appropriate type, it should be considered that while the virtual biobanking is easy from a regulatory perspective with tissues remaining in the original sites, it needs the development of robust protocols for testing the biomarkers, as the assays will be all carried out locally. In addition, while it is useful for validation studies, the virtual biobanking is not practical for rarer tumor types or small trials.
- Expert pathology review is recommended to ensure comprehensive assessment of gynecological cancers [35]. When considering a trial design which includes molecular tests it is recommended that translational/molecular/pathologic expertise should be consulted, to optimize sample collection and translational study design. In any case, the development of a project-specific consortium with a defined data-sharing mechanism, authorship policy and regular meetings, may substantially improve the output of the translational study.
- A process of assay validation is required with regard to reproducibility and reliability between laboratories (use of different/multiple platforms across different labs), to validate fit-for-purpose assay platforms, analysis tools, shared controls, and cut points that should be used in the final setting assay.
- As new analyses are developing over time, studies with molecular end-points should include as part of the trial, pathology-reviewed collection of additional biospecimen(s) for future use, for assay revalidation, and assessment of new or improved biomarkers.
- Biomarker validation and development are related processes that should be made public and transparent at all points by all stakeholders (including industry) to ensure rapid progress with positive outcomes for patients. The early phase studies should include all-comer, non-randomized patients, assessing intra and inter patient heterogeneity (temporal and geographic heterogeneity). In early validation studies, evaluation of the full spectrum is preferable to avoid loss of information about the possible treatment effect in the biomarker-negative population, these studies can be single arm and relatively small. Progression toward validation should be based on randomization, focused on the population of interest, with adequate power for appropriate treatment/outcome interaction analyses.
- Patient consent has to be specific and explicit, referring to future relevant research. However, to improve its acceptance, it should be a one-time consent, remaining broad but yet not unlimited in its terms. The opportunity to introduce a tiered consent has been considered. Potential exemption of consent should be considered (Declaration of Helsinki, art 32) with the development of an ethically defensible plan.
- Engagement of multiple trial groups with the aim of streamlined tissue donation and curation should be advocated, to increase the value/impact of trials through translational research. This is particularly aimed at ending the waste of funding and effort associated with trials which fail to reach their primary or secondary endpoints
- Standardization of the process of selection and optimization of the cutoff point for continuous biomarkers.
- In addition to scientific validity, the cost-benefit impact of biomarkers should also be considered. Whenever possible, based on anatomic feasibility, collection of post-progression biopsies should always be included in patient consent forms. Since this procedure is often restricted due to lack of funding, parallel institutional or trial group protocols and funding should be made available to ensure this collection.
- Issues related to the heterogeneity of the tumor and of the host (ethnic/racial variation) need to be considered.
- The use of an agreed governance document for Material Transfer is important to simplify institutional intellectual property agreements and to reduce delay.
- The creation of a big data consortium for clinical trial data to overcome the limitations related to the poor diffusion of negative results and to the different regulatory issues existing among the different entities involved should be considered.
5. Conclusions, Overall Goals and Key Future Tasks
- Development of a harmonization program of SOPs in a set of documented trial-specific guidelines taking into account issues related to sample collection and curation (pathological definitions and evaluation, clinical data annotation), and biomarker validation (analysis tools, choice of cut-points, impact of geographical and temporal heterogeneity, platforms, laboratory variation, etc.)
- Improve data sharing, transparency (e.g., components of biomarker signature/algorithms) publications (including negative findings) and ensure funding for sample collection. Facilitate development of consortia with a constitution, a data sharing mechanism and authorship policy that might also improve translational research funding success.
- GCIG guidelines/templates for patients’ informed consent including incorporation of global GCIG Material Transfer Agreement for widespread use to ensure streamlined access to patient specimens for the purpose of translational research.
- Increase involvement of advocates and improve reporting of translational data in a forum accessible to patients.
Funding
Acknowledgments
Conflicts of Interest
References
- Dancey, J.E.; Dobbin, K.K.; Groshen, S.; Jessup, J.M.; Hruszkewycz, A.H.; Koehler, M.; Parchment, R.; Ratain, M.J.; Shankar, L.K.; Stadler, W.M.; et al. Guidelines for the development and incorporation of biomarker studies in early clinical trials of novel agents. Clin. Cancer Res. 2010, 16, 1745–1755. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, S.; Panteghini, M. Making new biomarkers a reality: The case of serum human epididymis protein 4. Clin. Chem. Lab. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Lesko, L.J.; Atkinson, A.J., Jr. Use of biomarkers and surrogate endpoints in drug development and regulatory decision making: Criteria, validation, strategies. Annu. Rev. Pharmacol. Toxicol. 2001, 41, 347–366. [Google Scholar] [CrossRef] [PubMed]
- Mirza, M.R.; Monk, B.J.; Herrstedt, J.; Oza, A.M.; Mahner, S.; Redondo, A.; Fabbro, M.; Ledermann, J.A.; Lorusso, D.; Vergote, I.; et al. Niraparib Maintenance Therapy in Platinum-Sensitive, Recurrent Ovarian Cancer. N. Engl. J. Med. 2016, 375, 2154–2164. [Google Scholar] [CrossRef] [PubMed]
- Hyman, D.M.; Puzanov, I.; Subbiah, V.; Faris, J.E.; Chau, I.; Blay, J.Y.; Wolf, J.; Raje, N.S.; Diamond, E.L.; Hollebecque, A.; et al. Vemurafenib in Multiple Nonmelanoma Cancers with BRAF V600 Mutations. N. Engl. J. Med. 2015, 373, 726–736. [Google Scholar] [CrossRef] [PubMed]
- FOCUS4: A Molecularly Stratified Trial Programme in Colorectal Cancer. Available online: http://www.focus4trial.org/ (accessed on 5 November 2018).
- Scholl, S.; De Koning, L.; Popovic, M.; Anne, D.L.R.; Floquet, A.; Berns, E.; Kenter, G.; Kereszt, A.; Girard, E.; von der Leyen, H.; et al. Molecular profiles as a function of treatment response/PFS in a prospective cervical cancer study (RAIDs). Ann. Oncol. 2018, 29. [Google Scholar] [CrossRef]
- Goranova, T.; Ennis, D.; Piskorz, A.M.; Macintyre, G.; Lewsley, L.A.; Stobo, J.; Wilson, C.; Kay, D.; Glasspool, R.M.; Lockley, M.; et al. Safety and utility of image-guided research biopsies in relapsed high-grade serous ovarian carcinoma-experience of the BriTROC consortium. Br. J. Cancer 2017, 116, 1294–1301. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, R.F.; Ng, C.K.; Cooke, S.L.; Newman, S.; Temple, J.; Piskorz, A.M.; Gale, D.; Sayal, K.; Murtaza, M.; Baldwin, P.J.; et al. Spatial and temporal heterogeneity in high-grade serous ovarian cancer: A phylogenetic analysis. PLoS Med. 2015, 12, e1001789. [Google Scholar] [CrossRef] [PubMed]
- Parkinson, C.A.; Gale, D.; Piskorz, A.M.; Biggs, H.; Hodgkin, C.; Addley, H.; Freeman, S.; Moyle, P.; Sala, E.; Sayal, K.; et al. Exploratory Analysis of TP53 Mutations in Circulating Tumour DNA as Biomarkers of Treatment Response for Patients with Relapsed High-Grade Serous Ovarian Carcinoma: A Retrospective Study. PLoS Med. 2016, 13, e1002198. [Google Scholar] [CrossRef] [PubMed]
- Stelloo, E.; Nout, R.A.; Osse, E.M.; Jurgenliemk-Schulz, I.J.; Jobsen, J.J.; Lutgens, L.C.; van der Steen-Banasik, E.M.; Nijman, H.W.; Putter, H.; Bosse, T.; et al. Improved Risk Assessment by Integrating Molecular and Clinicopathological Factors in Early-stage Endometrial Cancer-Combined Analysis of the PORTEC Cohorts. Clin. Cancer Res. 2016, 22, 4215–4224. [Google Scholar] [CrossRef] [PubMed]
- de Boer, S.M.; Wortman, B.G.; Bosse, T.; Powell, M.E.; Singh, N.; Hollema, H.; Wilson, G.; Chowdhury, M.N.; Mileshkin, L.; Pyman, J.; et al. Clinical consequences of upfront pathology review in the randomised PORTEC-3 trial for high-risk endometrial cancer. Ann. Oncol. 2018, 29, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.S.; Mellman, I. Oncology meets immunology: The cancer-immunity cycle. Immunity 2013, 39, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Deniger, D.C.; Pasetto, A.; Robbins, P.F.; Gartner, J.J.; Prickett, T.D.; Paria, B.C.; Malekzadeh, P.; Jia, L.; Yossef, R.; Langhan, M.M.; et al. T-cell Responses to TP53 “Hotspot” Mutations and Unique Neoantigens Expressed by Human Ovarian Cancers. Clin. Cancer Res. 2018, 24, 5562–5573. [Google Scholar] [CrossRef] [PubMed]
- Ribas, A. Adaptive Immune Resistance: How Cancer Protects from Immune Attack. Cancer Discov. 2015, 5, 915–919. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Conejo-Garcia, J.R.; Katsaros, D.; Gimotty, P.A.; Massobrio, M.; Regnani, G.; Makrigiannakis, A.; Gray, H.; Schlienger, K.; Liebman, M.N.; et al. Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N. Engl. J. Med. 2003, 348, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Hwang, W.T.; Adams, S.F.; Tahirovic, E.; Hagemann, I.S.; Coukos, G. Prognostic significance of tumor-infiltrating T cells in ovarian cancer: A meta-analysis. Gynecol. Oncol. 2012, 124, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Sato, E.; Olson, S.H.; Ahn, J.; Bundy, B.; Nishikawa, H.; Qian, F.; Jungbluth, A.A.; Frosina, D.; Gnjatic, S.; Ambrosone, C.; et al. Intraepithelial CD8+ tumor-infiltrating lymphocytes and a high CD8+/regulatory T cell ratio are associated with favorable prognosis in ovarian cancer. Proc. Natl. Acad. Sci. USA 2005, 102, 18538–18543. [Google Scholar] [CrossRef] [PubMed]
- Curiel, T.J.; Coukos, G.; Zou, L.; Alvarez, X.; Cheng, P.; Mottram, P.; Evdemon-Hogan, M.; Conejo-Garcia, J.R.; Zhang, L.; Burow, M.; et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat. Med. 2004, 10, 942–949. [Google Scholar] [CrossRef] [PubMed]
- Jang, M.; Yew, P.Y.; Hasegawa, K.; Ikeda, Y.; Fujiwara, K.; Fleming, G.F.; Nakamura, Y.; Park, J.H. Characterization of T cell repertoire of blood, tumor, and ascites in ovarian cancer patients using next generation sequencing. Oncoimmunology 2015, 4, e1030561. [Google Scholar] [CrossRef] [PubMed]
- Perez-Gracia, J.L.; Labiano, S.; Rodriguez-Ruiz, M.E.; Sanmamed, M.F.; Melero, I. Orchestrating immune check-point blockade for cancer immunotherapy in combinations. Curr. Opin. Immunol. 2014, 27, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Hamanishi, J.; Mandai, M.; Iwasaki, M.; Okazaki, T.; Tanaka, Y.; Yamaguchi, K.; Higuchi, T.; Yagi, H.; Takakura, K.; Minato, N.; et al. Programmed cell death 1 ligand 1 and tumor-infiltrating CD8+ T lymphocytes are prognostic factors of human ovarian cancer. Proc. Natl. Acad. Sci. USA 2007, 104, 3360–3365. [Google Scholar] [CrossRef] [PubMed]
- Darb-Esfahani, S.; Kunze, C.A.; Kulbe, H.; Sehouli, J.; Wienert, S.; Lindner, J.; Budczies, J.; Bockmayr, M.; Dietel, M.; Denkert, C.; et al. Prognostic impact of programmed cell death-1 (PD-1) and PD-ligand 1 (PD-L1) expression in cancer cells and tumor-infiltrating lymphocytes in ovarian high grade serous carcinoma. Oncotarget 2016, 7, 1486–1499. [Google Scholar] [CrossRef] [PubMed]
- Mandai, M.; Hamanishi, J.; Abiko, K.; Matsumura, N.; Baba, T.; Konishi, I. Anti-PD-L1/PD-1 immune therapies in ovarian cancer: Basic mechanism and future clinical application. Int. J. Clin. Oncol. 2016, 21, 456–461. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.D.; Brown, S.D.; Wick, D.A.; Nielsen, J.S.; Kroeger, D.R.; Twumasi-Boateng, K.; Holt, R.A.; Nelson, B.H. Low Mutation Burden in Ovarian Cancer May Limit the Utility of Neoantigen-Targeted Vaccines. PLoS ONE 2016, 11, e0155189. [Google Scholar] [CrossRef] [PubMed]
- Strickland, K.C.; Howitt, B.E.; Shukla, S.A.; Rodig, S.; Ritterhouse, L.L.; Liu, J.F.; Garber, J.E.; Chowdhury, D.; Wu, C.J.; D’Andrea, A.D.; et al. Association and prognostic significance of BRCA1/2-mutation status with neoantigen load, number of tumor-infiltrating lymphocytes and expression of PD-1/PD-L1 in high grade serous ovarian cancer. Oncotarget 2016, 7, 13587–13598. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Uram, J.N.; Wang, H.; Bartlett, B.R.; Kemberling, H.; Eyring, A.D.; Skora, A.D.; Luber, B.S.; Azad, N.S.; Laheru, D.; et al. PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N. Engl. J. Med. 2015, 372, 2509–2520. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Durham, J.N.; Smith, K.N.; Wang, H.; Bartlett, B.R.; Aulakh, L.K.; Lu, S.; Kemberling, H.; Wilt, C.; Luber, B.S.; et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 2017, 357, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Kandoth, C.; Schultz, N.; Cherniack, A.D.; Akbani, R.; Liu, Y.; Shen, H.; Robertson, A.G.; Pashtan, I.; Shen, R.; Benz, C.C.; et al. Integrated genomic characterization of endometrial carcinoma. Nature 2013, 497, 67–73. [Google Scholar] [PubMed]
- van Gool, I.C.; Eggink, F.A.; Freeman-Mills, L.; Stelloo, E.; Marchi, E.; de Bruyn, M.; Palles, C.; Nout, R.A.; de Kroon, C.D.; Osse, E.M.; et al. POLE Proofreading Mutations Elicit an Antitumor Immune Response in Endometrial Cancer. Clin. Cancer Res. 2015, 21, 3347–3355. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, T.N.; Schreiber, R.D. Neoantigens in cancer immunotherapy. Science 2015, 348, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Trimble, C.L.; Morrow, M.P.; Kraynyak, K.A.; Shen, X.; Dallas, M.; Yan, J.; Edwards, L.; Parker, R.L.; Denny, L.; Giffear, M.; et al. Safety, efficacy, and immunogenicity of VGX-3100, a therapeutic synthetic DNA vaccine targeting human papillomavirus 16 and 18 E6 and E7 proteins for cervical intraepithelial neoplasia 2/3: A randomised, double-blind, placebo-controlled phase 2b trial. Lancet 2015, 386, 2078–2088. [Google Scholar] [CrossRef]
- Moughon, D.L.; He, H.; Schokrpur, S.; Jiang, Z.K.; Yaqoob, M.; David, J.; Lin, C.; Iruela-Arispe, M.L.; Dorigo, O.; Wu, L. Macrophage Blockade Using CSF1R Inhibitors Reverses the Vascular Leakage Underlying Malignant Ascites in Late-Stage Epithelial Ovarian Cancer. Cancer Res. 2015, 75, 4742–4752. [Google Scholar] [CrossRef] [PubMed]
- McCracken, M.N.; Cha, A.C.; Weissman, I.L. Molecular Pathways: Activating T Cells after Cancer Cell Phagocytosis from Blockade of CD47 “Don’t Eat Me” Signals. Clin. Cancer Res. 2015, 21, 3597–3601. [Google Scholar] [CrossRef] [PubMed]
- Kommoss, S.; Pfisterer, J.; Reuss, A.; Diebold, J.; Hauptmann, S.; Schmidt, C.; du, B.A.; Schmidt, D.; Kommoss, F. Specialized pathology review in patients with ovarian cancer: Results from a prospective study. Int. J. Gynecol. Cancer 2013, 23, 1376–1382. [Google Scholar] [CrossRef] [PubMed]

| Type of Biomarker | Question Addressed |
|---|---|
| Diagnostic | Cancer type/subtype identification |
| Prognostic | Cancer outcome definition |
| Predictive | Probability of response to a given drug |
| Pharmacodynamic | Definition of the optimal dose for efficient target engagement |
| Aims | Requirements | |
|---|---|---|
| Integral | Used for patient selection within a particular trial; its detection determines patient treatment. | Assessment has to be performed in a CLIA environment; it may require IDE and additional FDA approval. |
| Integrated | Used for patient/tumor characterization; it should provide evidence of function/pathway alteration. | CLIA environment recommended; IDE is not required. |
| Exploratory | Descriptive biomarkers; used to explore other patient characteristics useful for alternative treatment(s). | No particular requirements to be performed. |
| Biomarker-Enrichment Design | Biomarker-Stratified Designs | Adaptive Designs (Randomized Phase II/III Trial Design) | |
|---|---|---|---|
| Design description | • Biomarker positive subgroup is tested. • Control/standard therapy arm controls for marker prognostic effects. | • Randomize for treatment both positive and negative patients. • Several different testing strategies are possible. | • Combine phase II and phase III trials in a phase II/III trial. • It can be used without enrichment or be stratified by biomarker |
| Suitable choice when | • Solid knowledge of biomarker biology. • Convincing evidence that the benefits of treatment are limited to the biomarker-positive subgroup. | • A clinically significant effect in biomarker-negative patients cannot be ruled out. | • The treatment benefit should be definitively assessed; • It allows a decrease of the time and number of patients required from early to later phase development. |
| Major issues: | • It does not provide information on the biomarker-negative population (off-target effects, multiple pathway targeting) • Advantages coming from biomarker refinement within trial are limited to biomarker-positive patients. | • They maintain a good statistical power even in the case of a homogeneous treatment effect across subgroups. • They provide evidence of treatment benefit in both positive and negative population. Interim analysis can guide the accrual of biomarker-negative subgroup. | Critical are • the choice of the intermediate endpoint • the criteria (error rates, timing of analyses) defining a promising activity for phase II • accrual suspension while awaiting phase II data to mature. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bagnoli, M.; Shi, T.Y.; Gourley, C.; Speiser, P.; Reuss, A.; Nijman, H.W.; Creutzberg, C.L.; Scholl, S.; Negrouk, A.; Brady, M.F.; et al. Gynecological Cancers Translational, Research Implementation, and Harmonization: Gynecologic Cancer InterGroup Consensus and Still Open Questions. Cells 2019, 8, 200. https://doi.org/10.3390/cells8030200
Bagnoli M, Shi TY, Gourley C, Speiser P, Reuss A, Nijman HW, Creutzberg CL, Scholl S, Negrouk A, Brady MF, et al. Gynecological Cancers Translational, Research Implementation, and Harmonization: Gynecologic Cancer InterGroup Consensus and Still Open Questions. Cells. 2019; 8(3):200. https://doi.org/10.3390/cells8030200
Chicago/Turabian StyleBagnoli, Marina, Ting Yan Shi, Charlie Gourley, Paul Speiser, Alexander Reuss, Hans W. Nijman, Carien L. Creutzberg, Suzy Scholl, Anastassia Negrouk, Mark F. Brady, and et al. 2019. "Gynecological Cancers Translational, Research Implementation, and Harmonization: Gynecologic Cancer InterGroup Consensus and Still Open Questions" Cells 8, no. 3: 200. https://doi.org/10.3390/cells8030200
APA StyleBagnoli, M., Shi, T. Y., Gourley, C., Speiser, P., Reuss, A., Nijman, H. W., Creutzberg, C. L., Scholl, S., Negrouk, A., Brady, M. F., Hasegawa, K., Oda, K., McNeish, I. A., Kohn, E. C., Oza, A. M., MacKay, H., Millan, D., Bennett, K., Scott, C., & Mezzanzanica, D. (2019). Gynecological Cancers Translational, Research Implementation, and Harmonization: Gynecologic Cancer InterGroup Consensus and Still Open Questions. Cells, 8(3), 200. https://doi.org/10.3390/cells8030200








