Systematic Review of the Current Status of Human Sarcoma Cell Lines
Abstract
:1. Introduction
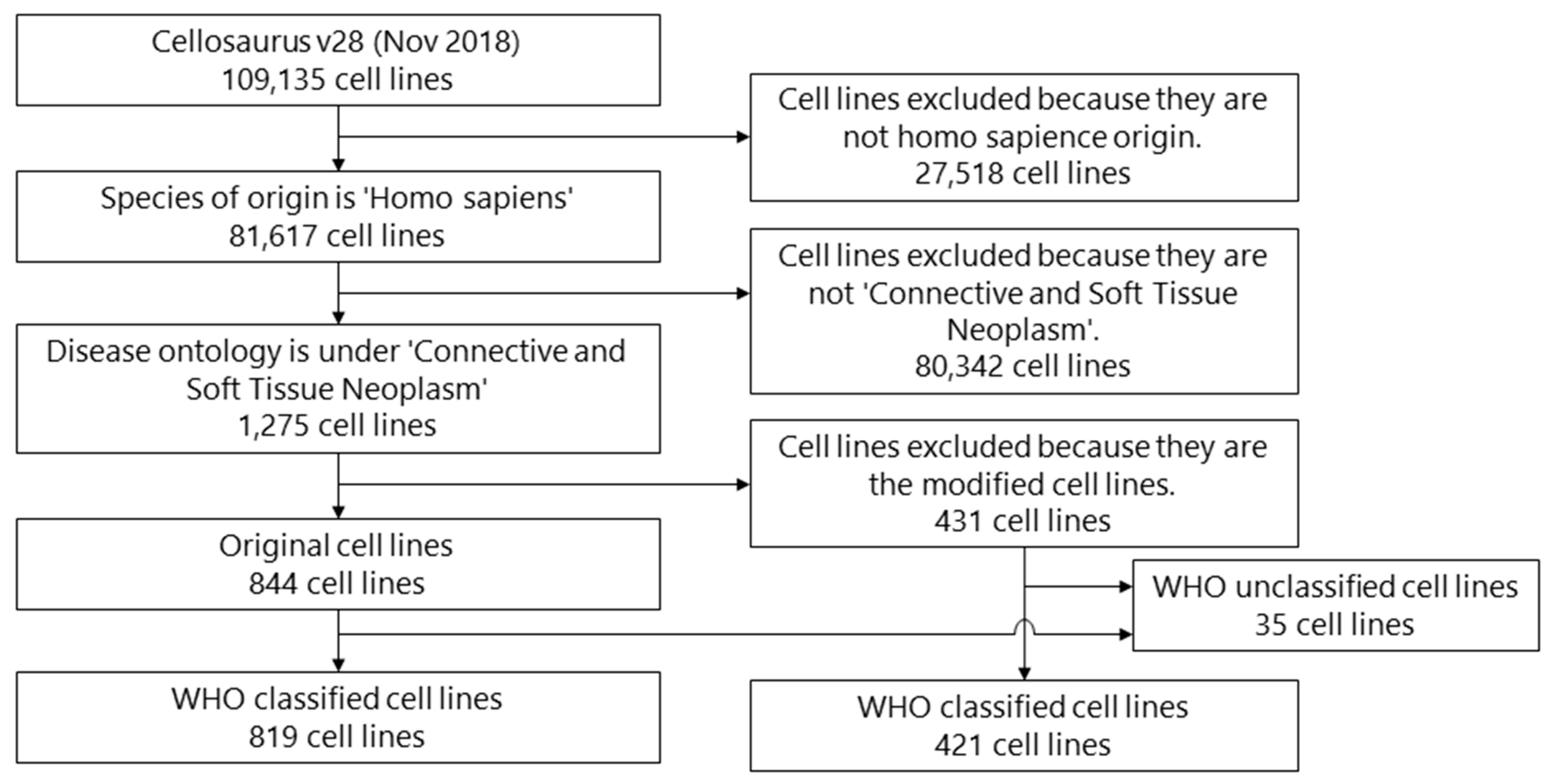
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility Criteria and Selection
2.3. Data Collection Process
2.4. Data Items
2.5. Information Sources
3. Results
3.1. Histology
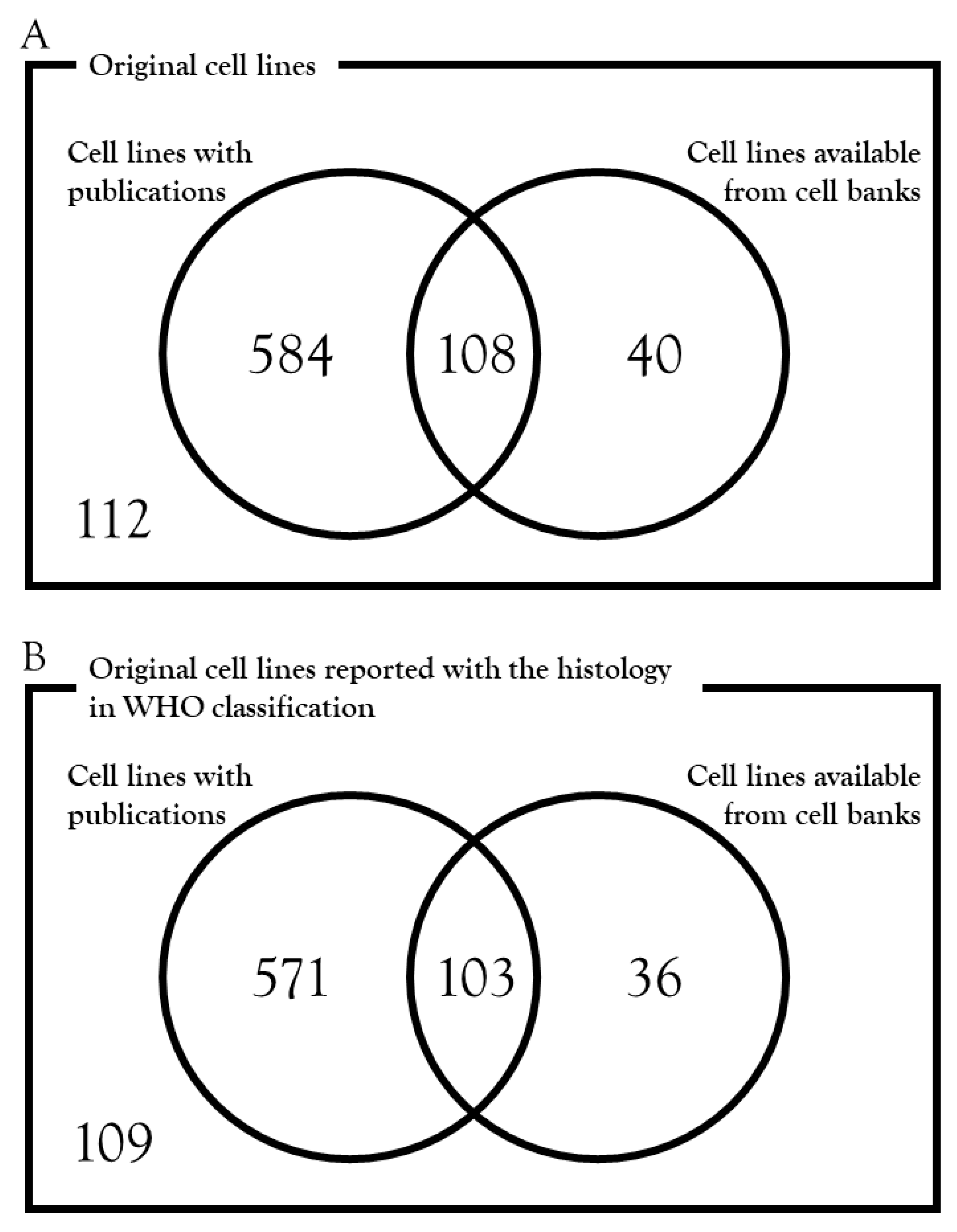
3.2. Availability from Cell Banks
3.3. Publication of Cell Lines
3.4. Availability and Publication of Cell Lines
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Fletcher, C.D.M.; Bridge, J.A.; Hogendoorn, P.; Mertens, F. WHO Classification of Tumours of Soft Tissue and Bone, 4th ed.; WHO Press: Geneva, Switzerland, 2013. [Google Scholar]
- Bovee, J.V.; Hogendoorn, P.C. Molecular pathology of sarcomas: Concepts and clinical implications. Virchows Arch. 2010, 456, 193–199. [Google Scholar] [CrossRef] [PubMed]
- Hameed, M. Molecular diagnosis of soft tissue neoplasia: Clinical applications and recent advances. Expert Rev. Mol. Diagn. 2014, 14, 961–977. [Google Scholar] [CrossRef] [PubMed]
- The Cancer Genome Atlas Research Network. Comprehensive and Integrated Genomic Characterization of Adult Soft Tissue Sarcomas. Cell 2017, 171, 950–965. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2017. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Farid, M.; Ngeow, J. Sarcomas Associated with Genetic Cancer Predisposition Syndromes: A Review. Oncologist 2016, 21, 1002–1013. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, Z. Systematic meta-analysis of genetic variants associated with osteosarcoma susceptibility. Medicine 2018, 97, e12525. [Google Scholar] [CrossRef] [PubMed]
- Benna, C.; Simioni, A.; Pasquali, S.; De Boni, D.; Rajendran, S.; Spiro, G.; Colombo, C.; Virgone, C.; DuBois, S.G.; Gronchi, A.; et al. Genetic susceptibility to bone and soft tissue sarcomas: A field synopsis and meta-analysis. Oncotarget 2018, 9, 18607–18626. [Google Scholar] [CrossRef] [PubMed]
- Ballinger, M.L.; Goode, D.L.; Ray-Coquard, I.; James, P.A.; Mitchell, G.; Niedermayr, E.; Puri, A.; Schiffman, J.D.; Dite, G.S.; Cipponi, A.; et al. Monogenic and polygenic determinants of sarcoma risk: An international genetic study. Lancet Oncol. 2016, 17, 1261–1271. [Google Scholar] [CrossRef]
- Jiang, Y.; Janku, F.; Subbiah, V.; Angelo, L.S.; Naing, A.; Anderson, P.A.; Herzog, C.E.; Fu, S.; Benjamin, R.S.; Kurzrock, R. Germline PTPRD mutations in Ewing sarcoma: Biologic and clinical implications. Oncotarget 2013, 4, 884–889. [Google Scholar] [CrossRef]
- Zhang, J.; Walsh, M.F.; Wu, G.; Edmonson, M.N.; Gruber, T.A.; Easton, J.; Hedges, D.; Ma, X.; Zhou, X.; Yergeau, D.A.; et al. Germline Mutations in Predisposition Genes in Pediatric Cancer. N. Engl. J. Med. 2015, 373, 2336–2346. [Google Scholar] [CrossRef]
- Eaton, K.W.; Tooke, L.S.; Wainwright, L.M.; Judkins, A.R.; Biegel, J.A. Spectrum of SMARCB1/INI1 mutations in familial and sporadic rhabdoid tumors. Pediatr. Blood Cancer 2011, 56, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.H.; Lim, W.K.; Ishak, N.D.B.; Li, S.T.; Goh, W.L.; Tan, G.S.; Lim, K.H.; Teo, M.; Young, C.N.C.; Malik, S.; et al. Germline Mutations in Cancer Predisposition Genes are Frequent in Sporadic Sarcomas. Sci. Rep. 2017, 7, 10660. [Google Scholar] [CrossRef] [PubMed]
- Joensuu, H.; Roberts, P.J.; Sarlomo-Rikala, M.; Andersson., L.C.; Tervahartiala, P.; Tuveson, D.; Silberman, S.L.; Capdeville, R.; Dimitrijevic, S.; Druker, B.; et al. Effect of the tyrosine kinase inhibitor STI571 in a patient with a metastatic gastrointestinal stromal tumor. N. Engl. J. Med. 2001, 344, 1052–1056. [Google Scholar] [CrossRef] [PubMed]
- Van Oosterom, A.T.; Judson, I.; Verweij, J.; Stroobants, S.; di Paola, E.D.; Dimitrijevic, S.; Martens, M.; Webb, A.; Sciot, R.; Van Glabbeke, M.; et al. Safety and efficacy of imatinib (STI571) in metastatic gastrointestinal stromal tumours: A phase I study. Lancet 2001, 358, 1421–1423. [Google Scholar] [CrossRef]
- Demetri, G.D.; von Mehren, M.; Blanke, C.D.; Van den Abbeele, A.D.; Eisenberg, B.; Roberts, P.J.; Heinrich, M.C.; Tuveson, D.A.; Singer, S.; Janicek, M.; et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N. Engl. J. Med. 2002, 347, 472–480. [Google Scholar] [CrossRef]
- Blanke, C.D.; Rankin, C.; Demetri, G.D.; Ryan, C.W.; von Mehren, M.; Benjamin, R.S.; Raymond, A.K.; Bramwell, V.H.; Baker, L.H.; Maki, R.G.; et al. Phase III randomized, intergroup trial assessing imatinib mesylate at two dose levels in patients with unresectable or metastatic gastrointestinal stromal tumors expressing the kit receptor tyrosine kinase: S0033. J. Clin. Oncol. 2008, 26, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Dematteo, R.P.; Ballman, K.V.; Antonescu, C.R.; Maki, R.G.; Pisters, P.W.; Demetri, G.D.; Blackstein, M.E.; Blanke, C.D.; von Mehren, M.; Brennan, M.F.; et al. Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: A randomised, double-blind, placebo-controlled trial. Lancet 2009, 373, 1097–1104. [Google Scholar] [CrossRef]
- Stacchiotti, S.; Pantaleo, M.A.; Negri, T.; Astolfi, A.; Tazzari, M.; Dagrada, G.P.; Urbini, M.; Indio, V.; Maestro, R.; Gronchi, A.; et al. Efficacy and Biological Activity of Imatinib in Metastatic Dermatofibrosarcoma Protuberans (DFSP). Clin. Cancer Res. 2016, 22, 837–846. [Google Scholar] [CrossRef]
- Rutkowski, P.; Klimczak, A.; Ługowska, I.; Jagielska, B.; Wągrodzki, M.; Dębiec-Rychter, M.; Pieńkowska-Grela, B.; Świtaj, T. Long-term results of treatment of advanced dermatofibrosarcoma protuberans (DFSP) with imatinib mesylate—The impact of fibrosarcomatous transformation. Eur. J. Surgical Oncol. 2017, 43, 1134–1141. [Google Scholar] [CrossRef]
- Demetri, G.D.; van Oosterom, A.T.; Garrett, C.R.; Blackstein, M.E.; Shah, M.H.; Verweij, J.; McArthur, G.; Judson, I.R.; Heinrich, M.C.; Morgan, J.A.; et al. Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: A randomised controlled trial. Lancet 2006, 368, 1329–1338. [Google Scholar] [CrossRef]
- Demetri, G.D.; Reichardt, P.; Kang, Y.K.; Blay, J.Y.; Rutkowski, P.; Gelderblom, H.; Hohenberger, P.; Leahy, M.; von Mehren, M.; Joensuu, H.; et al. Efficacy and safety of regorafenib for advanced gastrointestinal stromal tumours after failure of imatinib and sunitinib (GRID): An international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 2013, 381, 295–302. [Google Scholar] [CrossRef]
- Yovine, A.; Riofrio, M.; Blay, J.Y.; Brain, E.; Alexandre, J.; Kahatt, C.; Taamma, A.; Jimeno, J.; Martin, C.; Salhi, Y.; et al. Phase II study of ecteinascidin-743 in advanced pretreated soft tissue sarcoma patients. J. Clin. Oncol. 2004, 22, 890–899. [Google Scholar] [CrossRef] [PubMed]
- Le Cesne, A.; Blay, J.Y.; Judson, I.; Van Oosterom, A.; Verweij, J.; Radford, J.; Lorigan, P.; Rodenhuis, S.; Ray-Coquard, I.; Bonvalot, S.; et al. Phase II study of ET-743 in advanced soft tissue sarcomas: A European Organisation for the Research and Treatment of Cancer (EORTC) soft tissue and bone sarcoma group trial. J. Clin. Oncol. 2005, 23, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Blay, J.Y.; Italiano, A.; Ray-Coquard, I.; Le Cesne, A.; Duffaud, F.; Rios, M.; Collard, O.; Bertucci, F.; Bompas, E.; Isambert, N.; et al. Long-term outcome and effect of maintenance therapy in patients with advanced sarcoma treated with trabectedin: An analysis of 181 patients of the French ATU compassionate use program. BMC Cancer 2013, 13, 64. [Google Scholar] [CrossRef] [PubMed]
- Le Cesne, A.; Ray-Coquard, I.; Duffaud, F.; Chevreau, C.; Penel, N.; Bui Nguyen, B.; Piperno-Neumann, S.; Delcambre, C.; Rios, M.; Chaigneau, L.; et al. Trabectedin in patients with advanced soft tissue sarcoma: A retrospective national analysis of the French Sarcoma Group. Eur. J. Cancer 2015, 51, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Buonadonna, A.; Benson, C.; Casanova, J.; Kasper, B.; López Pousa, A.; Mazzeo, F.; Brodowicz, T.; Penel, N. A noninterventional, multicenter, prospective phase IV study of trabectedin in patients with advanced soft tissue sarcoma. Anticancer Drugs 2017, 28, 1157–1165. [Google Scholar] [CrossRef] [PubMed]
- Hamberg, P.; Verweij, J.; Sleijfer, S. (Pre-)clinical pharmacology and activity of pazopanib, a novel multikinase angiogenesis inhibitor. Oncologist 2010, 15, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Knick, V.B.; Rudolph, S.K.; Johnson, J.H.; Crosby, R.M.; Crouthamel, M.C.; Hopper, T.M.; Miller, C.G.; Harrington, L.E.; Onori, J.A.; et al. Pharmacokinetic-pharmacodynamic correlation from mouse to human with pazopanib, a multikinase angiogenesis inhibitor with potent antitumor and antiangiogenic activity. Mol. Cancer Ther. 2007, 6, 2012–2021. [Google Scholar] [CrossRef] [PubMed]
- Hurwitz, H.I.; Dowlati, A.; Saini, S.; Savage, S.; Suttle, A.B.; Gibson, D.M.; Hodge, J.P.; Merkle, E.M.; Pandite, L. Phase I trial of pazopanib in patients with advanced cancer. Clin. Cancer Res. 2009, 15, 4220–4227. [Google Scholar] [CrossRef]
- Sleijfer, S.; Ray-Coquard, I.; Papai, Z.; Le Cesne, A.; Scurr, M.; Schöffski, P.; Collin, F.; Pandite, L.; Marreaud, S.; De Brauwer, A.; et al. Pazopanib, a multikinase angiogenesis inhibitor, in patients with relapsed or refractory advanced soft tissue sarcoma: A phase II study from the European organisation for research and treatment of cancer-soft tissue and bone sarcoma group (EORTC study 62043). J. Clin. Oncol. 2009, 27, 3126–3132. [Google Scholar]
- Van der Graaf, W.T.; Blay, J.Y.; Chawla, S.P.; Kim, D.W.; Bui-Nguyen, B.; Casali, P.G.; Schöffski, P.; Aglietta, M.; Staddon, A.P.; Beppu, Y.; et al. Pazopanib for metastatic soft-tissue sarcoma (PALETTE): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2012, 379, 1879–1886. [Google Scholar] [CrossRef]
- Bai, R.L.; Paull, K.D.; Herald, C.L.; Malspeis, L.; Pettit, G.R.; Hamel, E. Halichondrin B and homohalichondrin B, marine natural products binding in the vinca domain of tubulin. Discovery of tubulin-based mechanism of action by analysis of differential cytotoxicity data. J. Biol. Chem. 1991, 266, 15882–15889. [Google Scholar]
- Jordan, M.A.; Kamath, K.; Manna, T.; Okouneva, T.; Miller, H.P.; Davis, C.; Littlefield, B.A.; Wilson, L. The primary antimitotic mechanism of action of the synthetic halichondrin E7389 is suppression of microtubule growth. Mol. Cancer Ther. 2005, 4, 1086–1095. [Google Scholar] [CrossRef] [PubMed]
- Funahashi, Y.; Okamoto, K.; Adachi, Y.; Semba, T.; Uesugi, M.; Ozawa, Y.; Tohyama, O.; Uehara, T.; Kimura, T.; Watanabe, H.; et al. Eribulin mesylate reduces tumor microenvironment abnormality by vascular remodeling in preclinical human breast cancer models. Cancer Sci. 2014, 105, 1334–1342. [Google Scholar] [CrossRef]
- Yoshida, T.; Ozawa, Y.; Kimura, T.; Sato, Y.; Kuznetsov, G.; Xu, S.; Uesugi, M.; Agoulnik, S.; Taylor, N.; Funahashi, Y.; Matsui, J. Eribulin mesilate suppresses experimental metastasis of breast cancer cells by reversing phenotype from epithelial-mesenchymal transition (EMT) to mesenchymal-epithelial transition (MET) states. Br. J. Cancer 2014, 110, 1497–1505. [Google Scholar] [CrossRef] [PubMed]
- Schoffski, P.; Chawla, S.; Maki, R.G.; Italiano, A.; Gelderblom, H.; Choy, E.; Grignani, G.; Camargo, V.; Bauer, S.; Rha, S.Y.; et al. Eribulin versus dacarbazine in previously treated patients with advanced liposarcoma or leiomyosarcoma: A randomised, open-label, multicentre, phase 3 trial. Lancet 2016, 387, 1629–1637. [Google Scholar] [CrossRef]
- Wagner, A.J.; Kindler, H.; Gelderblom, H.; Schöffski, P.; Bauer, S.; Hohenberger, P.; Kopp, H.G.; Lopez-Martin, J.A.; Peeters, M.; Reichardt, P.; et al. A phase II study of a human anti-PDGFRalpha monoclonal antibody (olaratumab, IMC-3G3) in previously treated patients with metastatic gastrointestinal stromal tumors. Ann. Oncol. 2017, 28, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Tap, W.D.; Jones, R.L.; Van Tine, B.A.; Chmielowski, B.; Elias, A.D.; Adkins, D.; Agulnik, M.; Cooney, M.M.; Livingston, M.B.; Pennock, G.; et al. Olaratumab and doxorubicin versus doxorubicin alone for treatment of soft-tissue sarcoma: An open-label phase 1b and randomised phase 2 trial. Lancet 2016, 388, 488–497. [Google Scholar] [CrossRef]
- Bekker, P.J.; Holloway, D.L.; Rasmussen, A.S.; Murphy, R.; Martin, S.W.; Leese, P.T.; Holmes, G.B.; Dunstan, C.R.; DePaoli, A.M. A single-dose placebo-controlled study of AMG 162, a fully human monoclonal antibody to RANKL, in postmenopausal women. J. bone Mineral Res. 2004, 19, 1059–1066. [Google Scholar] [CrossRef]
- Yamagishi, T.; Kawashima, H.; Ogose, A.; Ariizumi, T.; Sasaki, T.; Hatano, H.; Hotta, T.; Endo, N. Receptor-Activator of Nuclear KappaB Ligand Expression as a New Therapeutic Target in Primary Bone Tumors. PLoS ONE 2016, 11, e0154680. [Google Scholar] [CrossRef]
- Branstetter, D.; Rohrbach, K.; Huang, L.Y.; Soriano, R.; Tometsko, M.; Blake, M.; Jacob, A.P.; Dougall, W.C. RANK and RANK ligand expression in primary human osteosarcoma. J. Bone Oncol. 2015, 4, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Di Grappa, M.A.; Molyneux, S.D.; McKee, T.D.; Waterhouse, P.; Penninger, J.M.; Khokha, R. RANKL blockade prevents and treats aggressive osteosarcomas. Sci. Transl. Med. 2015, 7, 317ra197. [Google Scholar] [CrossRef] [PubMed]
- Traub, F.; Singh, J.; Dickson, B.C.; Leung, S.; Mohankumar, R.; Blackstein, M.E.; Razak, A.R.; Griffin, A.M.; Ferguson, P.C.; Wunder, J.S. Efficacy of denosumab in joint preservation for patients with giant cell tumour of the bone. Eur. J. Cancer 2016, 59, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Piperno-Neumann, S.; Le Deley, M.C.; Rédini, F.; Pacquement, H.; Marec-Bérard, P.; Petit, P.; Brisse, H.; Lervat, C.; Gentet, J.C.; Entz-Werlé, N.; et al. Zoledronate in combination with chemotherapy and surgery to treat osteosarcoma (OS2006): A randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2016, 17, 1070–1080. [Google Scholar] [CrossRef]
- Casali, P.G.; Abecassis, N.; Bauer, S.; Casali, P.G.; Abecassis, N.; Bauer, S.; Biagini, R.; Bielack, S.; Bonvalot, S.; Boukovinas, I.; et al. Soft tissue and visceral sarcomas: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2018, 29, iv51–iv67. [Google Scholar] [CrossRef]
- Casali, P.G.; Abecassis, N.; Aro, H.T.; Casali, P.G.; Abecassis, N.; Aro, H.T.; Bauer, S.; Biagini, R.; Bielack, S.; Bonvalot, S.; et al. Soft tissue and visceral sarcomas: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2018, 29, iv268–iv269. [Google Scholar] [CrossRef]
- Kawai, A.; Yonemori, K.; Takahashi, S.; Araki, N.; Ueda, T. Systemic Therapy for Soft Tissue Sarcoma: Proposals for the Optimal Use of Pazopanib, Trabectedin, and Eribulin. Adv. Ther. 2017, 34, 1556–1571. [Google Scholar] [CrossRef]
- Hoffman, R.M. Patient-derived orthotopic xenografts: Better mimic of metastasis than subcutaneous xenografts. Nat. Rev. Cancer 2015, 15, 451–452. [Google Scholar] [CrossRef] [PubMed]
- Drost, J.; Clevers, H. Organoids in cancer research. Nat. Rev. Cancer 2018, 18, 407–418. [Google Scholar] [CrossRef]
- Domcke, S.; Sinha, R.; Levine, D.A.; Sander, C.; Schultz, N. Evaluating cell lines as tumour models by comparison of genomic profiles. Nat. Commun. 2013, 4, 2126. [Google Scholar] [CrossRef]
- Rockwell, S. In vivo-in vitro tumour cell lines: Characteristics and limitations as models for human cancer. Br. J. Cancer 1980, 4, 118–122. [Google Scholar]
- Teicher, B.A.; Polley, E.; Kunkel, M.; Evans, D.; Silvers, T.; Delosh, R.; Laudeman, J.; Ogle, C.; Reinhart, R.; Selby, M.; et al. Sarcoma Cell Line Screen of Oncology Drugs and Investigational Agents Identifies Patterns Associated with Gene and microRNA Expression. Mol. Cancer Ther. 2015, 14, 2452–2462. [Google Scholar] [CrossRef] [PubMed]
- Barretina, J.; Caponigro, G.; Stransky, N.; Venkatesan, K.; Margolin, A.A.; Kim, S.; Wilson, C.J.; Lehár, J.; Kryukov, G.V.; Sonkin, D.; et al. The Cancer Cell Line Encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature 2012, 483, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Crystal, A.S.; Shaw, A.T.; Sequist, L.V.; Friboulet, L.; Niederst, M.J.; Lockerman, E.L.; Frias, R.L.; Gainor, J.F.; Amzallag, A.; Greninger, P.; et al. Patient-derived models of acquired resistance can identify effective drug combinations for cancer. Science 2014, 346, 1480–1486. [Google Scholar] [CrossRef] [PubMed]
- The Cellosaurus: A cell line knowledge resource. [updated 2018]. Available online: http://web.expasy.org/cellosaurus/ (accessed on 13 February 2019).
- Bairoch, A. The Cellosaurus, a Cell-Line Knowledge Resource. J. Biomol. Tech. 2018, 29, 25–38. [Google Scholar] [CrossRef] [PubMed]
- SEER Cancer Statistics Review, 1975–2015, National Cancer Institute. Bethesda, MD. [updated 2017]. Available online: https://seer.cancer.gov/csr/1975_2015/ (accessed on 13 February 2019).
- Jo, V.Y.; Doyle, L.A. Refinements in Sarcoma Classification in the Current 2013 World Health Organization Classification of Tumours of Soft Tissue and Bone. Surgical Oncol. Clin. North Am. 2016, 25, 621–643. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, C.; Costa, C.; Sousa, N.; Amado, F.; Santos, L. Patient-derived bladder cancer xenografts: A systematic review. Transl. Res. 2015, 166, 324–331. [Google Scholar] [CrossRef]
- Wan, L.; Tu, C.; Chen, Q.; Su, J.; Li, Z. Patient-derived xenograft models in musculoskeletal malignancies. J. Transl. Med. 2018, 16, 107. [Google Scholar]
| Group | Disease | In Public Cell Banks | Not in Public Cell Banks | Total |
|---|---|---|---|---|
| Adipocytic tumors | Lipoma | 0 | 6 | 6 |
| Dedifferentiated liposarcoma | 2 | 40 | 42 | |
| Myxoid liposarcoma | 0 | 9 | 9 | |
| Pleomorphic liposarcoma | 0 | 1 | 1 | |
| Fibroblastic/myofibroblastic tumors | Desmoids-type fibromatosis | 1 | 0 | 1 |
| Dermatofibrosarcoma protuberans | 2 | 2 | 4 | |
| Myxofibrosarcoma | 2 | 5 | 7 | |
| Fibrosarcoma | 9 | 21 | 30 | |
| So-called fibrohistiocytic tumors | Tenosynovial giant cell tumor | 0 | 1 | 1 |
| Smooth-muscle tumors | Leiomyoma of deep soft tissue | 0 | 10 | 10 |
| Leiomyosarcoma | 6 | 10 | 16 | |
| Pericytic (perivascular) tumors | Glomus tumors | 1 | 2 | 3 |
| Skeletal-muscle tumors | Embryonal rhabdomyosarcoma | 6 | 31 | 37 |
| Alveolar rhabdomyosarcoma | 6 | 40 | 46 | |
| Pleomorphic rhabdomyosarcoma | 0 | 2 | 2 | |
| Rhabdomyosarcoma | 5 | 14 | 19 | |
| Vascular tumors | Lymphangioma | 2 | 0 | 2 |
| Kaposi sarcoma | 0 | 8 | 8 | |
| Gastrointestinal stromal tumors | Gastrointestinal stromal tumors | 2 | 8 | 10 |
| Nerve sheath tumors | Malignant peripheral nerve sheath tumor | 5 | 19 | 24 |
| Tumors of uncertain differentiation | Myoepithelioma/myoepithelial carcinoma/mixed tumor | 0 | 2 | 2 |
| Synovial sarcoma | 5 | 29 | 34 | |
| Epithelioid sarcoma | 4 | 12 | 16 | |
| Alveolar soft-part sarcoma | 1 | 1 | 2 | |
| Clear cell sarcoma of soft tissue | 1 | 12 | 13 | |
| Desmoplastic small round cell tumor | 0 | 1 | 1 | |
| Extrarenal rhabdoid tumor | 0 | 12 | 12 | |
| PEComa | 0 | 1 | 1 | |
| Intimal sarcoma | 1 | 3 | 4 | |
| Chondrogenic tumors | Chondrosarcoma | 5 | 34 | 39 |
| Dedifferentiated chondrosarcoma | 0 | 5 | 5 | |
| Osteogenic tumors | Osteoid osteoma | 2 | 1 | 3 |
| Osteoblastoma | 0 | 1 | 1 | |
| Small cell osteosarcoma | 0 | 1 | 1 | |
| Osteosarcoma | 37 | 111 | 148 | |
| Fibrogenic tumors | Fibrosarcoma of bone | 0 | 1 | 1 |
| Fibrohistiocytic tumors | Benign fibrous histiocytoma/non-ossifying fibroma | 0 | 1 | 1 |
| Ewing sarcoma | Ewing sarcoma | 14 | 142 | 156 |
| Osteoclastic giant cell rich tumors | Giant cell tumor of bone | 4 | 4 | 8 |
| Notochordal tumors | Chordoma | 7 | 16 | 23 |
| Vascular tumors | Angiosarcoma | 2 | 2 | 4 |
| Myogenic, lipogenic and epithelial tumors | Bone leiomyosarcoma | 0 | 2 | 2 |
| Liposarcoma | 2 | 16 | 18 | |
| Undifferentiated high-grade pleomorphic sarcoma | Undifferentiated high-grade pleomorphic sarcoma | 4 | 39 | 43 |
| Tumor syndromes | Multiple osteochondromas | 1 | 2 | 3 |
| Total | 139 | 680 | 819 |
| Group | Disease | In Public Cell Banks | Not in Public Cell Banks | Total |
|---|---|---|---|---|
| Adipocytic tumors | Dedifferentiated liposarcoma | 0 | 1 | 1 |
| Myxoid liposarcoma | 0 | 1 | 1 | |
| Fibroblastic/myofibroblastic tumors | Fibrosarcoma | 26 | 17 | 43 |
| Smooth-muscle tumors | Leiomyosarcoma | 1 | 10 | 11 |
| Skeletal-muscle tumors | Embryonal rhabdomyosarcoma | 2 | 17 | 19 |
| Alveolar rhabdomyosarcoma | 0 | 23 | 23 | |
| Gastrointestinal stromal tumors | Gastrointestinal stromal tumors | 0 | 7 | 7 |
| Nerve sheath tumors | Malignant peripheral nerve sheath tumor | 0 | 1 | 1 |
| Tumors of uncertain differentiation | Epithelioid sarcoma | 0 | 5 | 5 |
| Intimal sarcoma | 2 | 5 | 7 | |
| Chondrogenic tumors | Chondrosarcoma | 0 | 6 | 6 |
| Osteogenic tumors | Osteosarcoma | 228 | 47 | 275 |
| Ewing sarcoma | Ewing sarcoma | 1 | 11 | 12 |
| Notochordal tumors | Chordoma | 0 | 1 | 1 |
| Vascular tumors | Angiosarcoma | 2 | 2 | 4 |
| Undifferentiated high-grade pleomorphic sarcoma | Undifferentiated high-grade pleomorphic sarcoma | 0 | 5 | 5 |
| Total | 262 | 159 | 421 |
| Group | Disease | In Public Cell Banks | Not in Public Cell Banks | Total |
|---|---|---|---|---|
| Not_classified | Ovarian mixed germ cell tumor | 2 | 5 | 7 |
| Thyroid gland sarcoma | 2 | 0 | 2 | |
| Endometrioid stromal sarcoma | 2 | 3 | 5 | |
| Soft tissue sarcoma | 0 | 2 | 2 | |
| Sarcoma | 0 | 8 | 8 | |
| Histiocytoma | 1 | 0 | 1 | |
| Skin sarcoma | 2 | 0 | 2 | |
| Uterine corpus sarcoma | 3 | 3 | 6 | |
| Meningeal sarcoma | 0 | 1 | 1 | |
| Benign synovial neoplasm | 0 | 1 | 1 | |
| Total | 12 | 23 | 35 |
| Group | Disease | PubMed Cited | Not Cited | Total |
|---|---|---|---|---|
| Adipocytic tumors | Lipoma | 5 | 1 | 6 |
| Dedifferentiated liposarcoma | 41 | 1 | 42 | |
| Myxoid liposarcoma | 9 | 0 | 9 | |
| Pleomorphic liposarcoma | 1 | 0 | 1 | |
| Fibroblastic/myofibroblastic tumors | Desmoids-type fibromatosis | 0 | 1 | 1 |
| Dermatofibrosarcoma protuberans | 3 | 1 | 4 | |
| Myxofibrosarcoma | 6 | 1 | 7 | |
| Fibrosarcoma | 12 | 18 | 30 | |
| So-called fibrohistiocytic tumors | Tenosynovial giant cell tumor | 1 | 0 | 1 |
| Smooth-muscle tumors | Leiomyoma of deep soft tissue | 9 | 1 | 10 |
| Leiomyosarcoma | 11 | 5 | 16 | |
| Pericytic(perivascular) tumors | Glomus tumors | 0 | 3 | 3 |
| Skeletal-muscle tumors | Embryonal rhabdomyosarcoma | 36 | 1 | 37 |
| Alveolar rhabdomyosarcoma | 45 | 1 | 46 | |
| Pleomorphic rhabdomyosarcoma | 2 | 0 | 2 | |
| Rhabdomyosarcoma | 13 | 6 | 19 | |
| Vascular tumors | Lymphangioma | 0 | 2 | 2 |
| Kaposi sarcoma | 7 | 1 | 8 | |
| Gastrointestinal stromal tumors | Gastrointestinal stromal tumors | 10 | 0 | 10 |
| Nerve sheath tumors | Malignant peripheral nerve sheath tumor | 22 | 2 | 24 |
| Tumors of uncertain differentiation | Myoepithelioma/myoepithelial carcinoma/mixed tumor | 2 | 0 | 2 |
| Synovial sarcoma | 31 | 3 | 34 | |
| Epithelioid sarcoma | 15 | 1 | 16 | |
| Alveolar soft-part sarcoma | 2 | 0 | 2 | |
| Clear cell sarcoma of soft tissue | 13 | 0 | 13 | |
| Desmoplastic small round cell tumor | 1 | 0 | 1 | |
| Extrarenal rhabdoid tumor | 11 | 1 | 12 | |
| PEComa | 0 | 1 | 1 | |
| Intimal sarcoma | 4 | 0 | 4 | |
| Chondrogenic tumors | Chondrosarcoma | 34 | 5 | 39 |
| Dedifferentiated chondrosarcoma | 5 | 0 | 5 | |
| Osteogenic tumors | Osteoid osteoma | 0 | 3 | 3 |
| Osteoblastoma | 0 | 1 | 1 | |
| Small cell osteosarcoma | 1 | 0 | 1 | |
| Osteosarcoma | 100 | 48 | 148 | |
| Fibrogenic tumors | Fibrosarcoma of bone | 1 | 0 | 1 |
| Fibrohistiocytic tumors | Benign fibrous histiocytoma/non-ossifying fibroma | 1 | 0 | 1 |
| Ewing sarcoma | Ewing sarcoma | 136 | 20 | 156 |
| Osteoclastic giant cell rich tumors | Giant cell tumor of bone | 5 | 3 | 8 |
| Notochordal tumors | Chordoma | 20 | 3 | 23 |
| Vascular tumors | Angiosarcoma | 4 | 0 | 4 |
| Myogenic, lipogenic and epithelial tumors | Bone leiomyosarcoma | 2 | 0 | 2 |
| Liposarcoma | 10 | 8 | 18 | |
| Undifferentiated high-grade pleomorphic sarcoma | Undifferentiated high-grade pleomorphic sarcoma | 42 | 1 | 43 |
| Tumor syndromes | Multiple osteochondromas | 1 | 2 | 3 |
| Total | 674 | 145 | 819 |
| Group | Disease | PubMed Cited | Not Cited | Total |
|---|---|---|---|---|
| Adipocytic tumors | Dedifferentiated liposarcoma | 1 | 0 | 1 |
| Myxoid liposarcoma | 1 | 0 | 1 | |
| Fibroblastic/myofibroblastic tumors | Fibrosarcoma | 18 | 25 | 43 |
| Smooth-muscle tumors | Leiomyosarcoma | 11 | 0 | 11 |
| Skeletal-muscle tumors | Embryonal rhabdomyosarcoma | 18 | 1 | 19 |
| Alveolar rhabdomyosarcoma | 20 | 3 | 23 | |
| Gastrointestinal stromal tumors | Gastrointestinal stromal tumors | 7 | 0 | 7 |
| Nerve sheath tumors | Malignant peripheral nerve sheath tumor | 1 | 0 | 1 |
| Tumors of uncertain differentiation | Epithelioid sarcoma | 5 | 0 | 5 |
| Intimal sarcoma | 3 | 4 | 7 | |
| Chondrogenic tumors | Chondrosarcoma | 6 | 0 | 6 |
| Osteogenic tumors | Osteosarcoma | 51 | 224 | 275 |
| Ewing sarcoma | Ewing sarcoma | 9 | 3 | 12 |
| Notochordal tumors | Chordoma | 1 | 0 | 1 |
| Vascular tumors | Angiosarcoma | 2 | 2 | 4 |
| Undifferentiated high-grade pleomorphic sarcoma | Undifferentiated high-grade pleomorphic sarcoma | 5 | 0 | 5 |
| Total | 159 | 262 | 421 |
| Group | Disease | PubMed Cited | Not Cited | Total |
|---|---|---|---|---|
| Not_classified | Ovarian mixed germ cell tumor | 6 | 1 | 7 |
| Thyroid gland sarcoma | 1 | 1 | 2 | |
| Endometrioid stromal sarcoma | 5 | 0 | 5 | |
| Soft tissue sarcoma | 2 | 0 | 2 | |
| Sarcoma | 6 | 2 | 8 | |
| Histiocytoma | 0 | 1 | 1 | |
| Skin sarcoma | 0 | 2 | 2 | |
| Uterine corpus sarcoma | 5 | 1 | 6 | |
| Meningeal sarcoma | 1 | 0 | 1 | |
| Benign synovial neoplasm | 1 | 0 | 1 | |
| Total | 27 | 8 | 35 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hattori, E.; Oyama, R.; Kondo, T. Systematic Review of the Current Status of Human Sarcoma Cell Lines. Cells 2019, 8, 157. https://doi.org/10.3390/cells8020157
Hattori E, Oyama R, Kondo T. Systematic Review of the Current Status of Human Sarcoma Cell Lines. Cells. 2019; 8(2):157. https://doi.org/10.3390/cells8020157
Chicago/Turabian StyleHattori, Emi, Rieko Oyama, and Tadashi Kondo. 2019. "Systematic Review of the Current Status of Human Sarcoma Cell Lines" Cells 8, no. 2: 157. https://doi.org/10.3390/cells8020157
APA StyleHattori, E., Oyama, R., & Kondo, T. (2019). Systematic Review of the Current Status of Human Sarcoma Cell Lines. Cells, 8(2), 157. https://doi.org/10.3390/cells8020157