Spatiotemporal Control of Intracellular Membrane Trafficking by Rho GTPases
Abstract
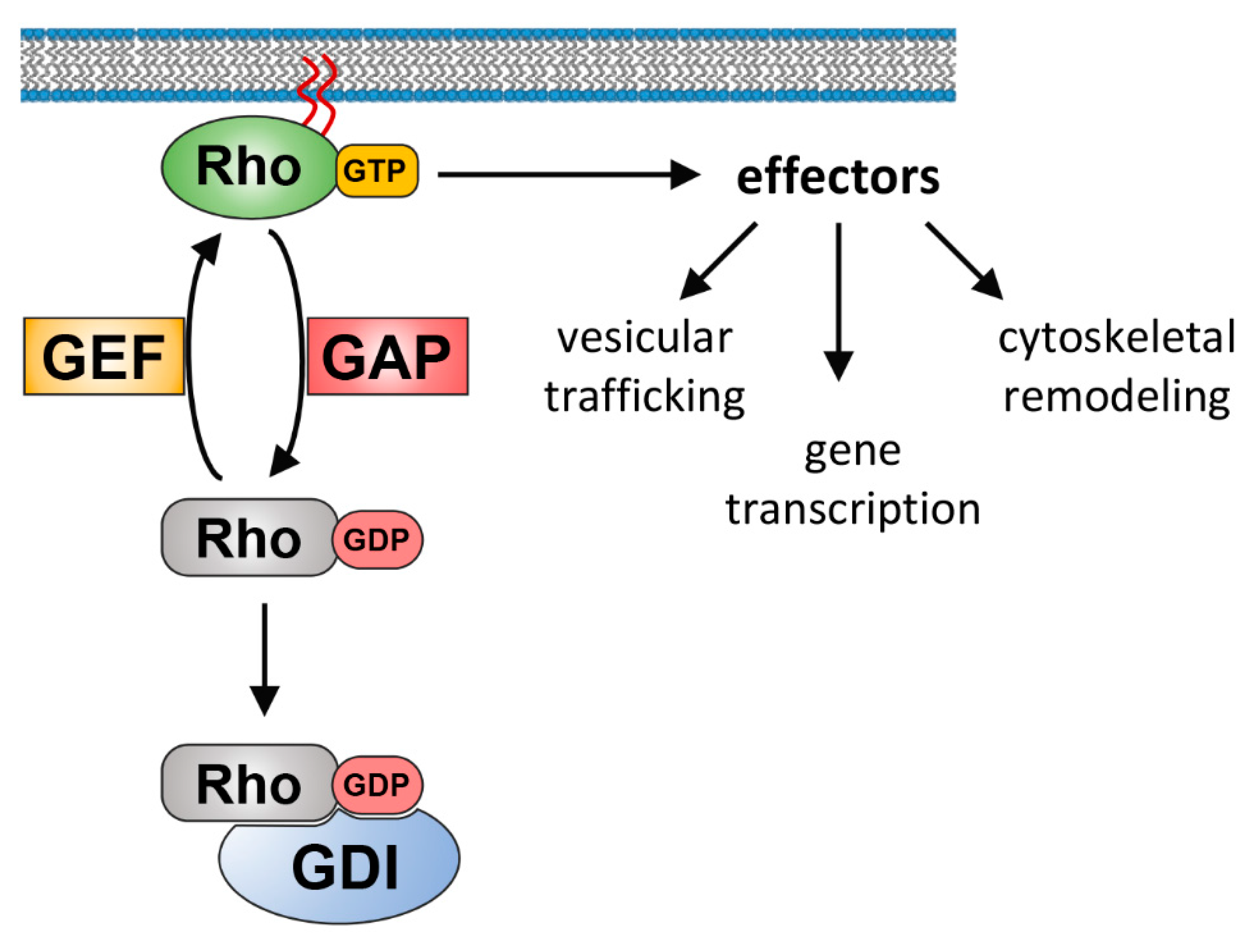
1. Introduction
2. Spatial Organization of Rho GTPase Signaling Complexes
3. Spatiotemporal Rho Regulation by GEFs and GAPs
4. Rho GTPases Acting along the Secretory Pathway
5. Rho GTPases Acting along the Endocytic Pathway
6. Rho GTPases and Membrane Trafficking—Implications for Cancer
7. Conclusions and Outlook
Funding
Acknowledgments
Conflicts of Interest
References
- Chavrier, P.; Goud, B. The role of ARF and Rab GTPases in membrane transport. Curr. Opin. Cell Biol. 1999, 11, 466–475. [Google Scholar] [CrossRef]
- Ridley, A.J. Rho GTPases and actin dynamics in membrane protrusions and vesicle trafficking. Trends Cell Biol. 2006, 16, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Hall, A. Rho family GTPases. Biochem. Soc. Trans. 2012, 40, 1378–1382. [Google Scholar] [CrossRef] [PubMed]
- Vega, F.M.; Ridley, A.J. SnapShot: Rho family GTPases. Cell 2007, 129, 1430. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Mata, R.; Boulter, E.; Burridge, K. The ‘invisible hand’: Regulation of RHO GTPases by RHOGDIs. Nat. Rev. Mol. Cell Biol. 2011, 12, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Bos, J.L.; Rehmann, H.; Wittinghofer, A. GEFs and GAPs: Critical elements in the control of small G proteins. Cell 2007, 129, 865–877. [Google Scholar] [CrossRef]
- Hodge, R.G.; Ridley, A.J. Regulating Rho GTPases and their regulators. Nat. Rev. Mol. Cell Biol. 2016, 17, 496–510. [Google Scholar] [CrossRef]
- Mitin, N.; Roberts, P.J.; Chenette, E.J.; Der, C.J. Posttranslational lipid modification of Rho family small GTPases. Methods Mol. Biol. 2012, 827, 87–95. [Google Scholar] [CrossRef]
- Aicart-Ramos, C.; Valero, R.A.; Rodriguez-Crespo, I. Protein palmitoylation and subcellular trafficking. Biochim. Et Biophys. Acta 2011, 1808, 2981–2994. [Google Scholar] [CrossRef]
- Williams, C.L. The polybasic region of Ras and Rho family small GTPases: A regulator of protein interactions and membrane association and a site of nuclear localization signal sequences. Cell. Signal. 2003, 15, 1071–1080. [Google Scholar] [CrossRef]
- Peurois, F.; Peyroche, G.; Cherfils, J. Small GTPase peripheral binding to membranes: Molecular determinants and supramolecular organization. Biochem. Soc. Trans. 2019, 47, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, M.A.; Glogauer, M. Pivotal Advance: Phospholipids determine net membrane surface charge resulting in differential localization of active Rac1 and Rac2. J. Leukoc. Biol. 2010, 87, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, K.N.; Zhou, Y.; Hancock, J.F. Rac1 Nanoscale Organization on the Plasma Membrane Is Driven by Lipid Binding Specificity Encoded in the Membrane Anchor. Mol. Cell. Biol. 2018, 38. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Yin, T.; Yang, Q.; Zhang, J.; Wu, Y.I.; Yu, J. Single-molecule tracking of small GTPase Rac1 uncovers spatial regulation of membrane translocation and mechanism for polarized signaling. Proc. Natl. Acad. Sci. USA 2015, 112, E267–E276. [Google Scholar] [CrossRef] [PubMed]
- Olson, M.F. Rho GTPases, their post-translational modifications, disease-associated mutations and pharmacological inhibitors. Small Gtpases 2018, 9, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Nethe, M.; Hordijk, P.L. The role of ubiquitylation and degradation in RhoGTPase signalling. J. Cell Sci. 2010, 123, 4011–4018. [Google Scholar] [CrossRef]
- Remorino, A.; De Beco, S.; Cayrac, F.; Di Federico, F.; Cornilleau, G.; Gautreau, A.; Parrini, M.C.; Masson, J.B.; Dahan, M.; Coppey, M. Gradients of Rac1 Nanoclusters Support Spatial Patterns of Rac1 Signaling. Cell Rep. 2017, 21, 1922–1935. [Google Scholar] [CrossRef]
- Erickson, J.W.; Zhang, C.; Kahn, R.A.; Evans, T.; Cerione, R.A. Mammalian Cdc42 is a brefeldin A-sensitive component of the Golgi apparatus. J. Biol. Chem. 1996, 271, 26850–26854. [Google Scholar] [CrossRef]
- Wu, W.J.; Erickson, J.W.; Lin, R.; Cerione, R.A. The gamma-subunit of the coatomer complex binds Cdc42 to mediate transformation. Nature 2000, 405, 800–804. [Google Scholar] [CrossRef]
- Fucini, R.V.; Chen, J.L.; Sharma, C.; Kessels, M.M.; Stamnes, M. Golgi vesicle proteins are linked to the assembly of an actin complex defined by mAbp1. Mol. Biol. Cell 2002, 13, 621–631. [Google Scholar] [CrossRef]
- Lam, B.D.; Hordijk, P.L. The Rac1 hypervariable region in targeting and signaling: A tail of many stories. Small Gtpases 2013, 4, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Golding, A.E.; Visco, I.; Bieling, P.; Bement, W.M. Extraction of active RhoGTPases by RhoGDI regulates spatiotemporal patterning of RhoGTPases. ELife 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Bishop, A.L.; Hall, A. Rho GTPases and their effector proteins. Biochem. J. 2000, 348, 241–255. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.; Paterson, H.F.; Adamson, P.; Ridley, A.J. Cellular responses regulated by rho-related small GTP-binding proteins. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 1993, 340, 267–271. [Google Scholar] [CrossRef]
- Takesono, A.; Heasman, S.J.; Wojciak-Stothard, B.; Garg, R.; Ridley, A.J. Microtubules regulate migratory polarity through Rho/ROCK signaling in T cells. PLoS ONE 2010, 5, e8774. [Google Scholar] [CrossRef]
- Meriane, M.; Mary, S.; Comunale, F.; Vignal, E.; Fort, P.; Gauthier-Rouviere, C. Cdc42Hs and Rac1 GTPases induce the collapse of the vimentin intermediate filament network. J. Biol. Chem. 2000, 275, 33046–33052. [Google Scholar] [CrossRef]
- Jiu, Y.; Peranen, J.; Schaible, N.; Cheng, F.; Eriksson, J.E.; Krishnan, R.; Lappalainen, P. Vimentin intermediate filaments control actin stress fiber assembly through GEF-H1 and RhoA. J. Cell Sci. 2017, 130, 892–902. [Google Scholar] [CrossRef]
- Wing, M.R.; Snyder, J.T.; Sondek, J.; Harden, T.K. Direct activation of phospholipase C-epsilon by Rho. J. Biol. Chem. 2003, 278, 41253–41258. [Google Scholar] [CrossRef]
- Watanabe, G.; Saito, Y.; Madaule, P.; Ishizaki, T.; Fujisawa, K.; Morii, N.; Mukai, H.; Ono, Y.; Kakizuka, A.; Narumiya, S. Protein kinase N (PKN) and PKN-related protein rhophilin as targets of small GTPase Rho. Science 1996, 271, 645–648. [Google Scholar] [CrossRef]
- Mellor, H.; Flynn, P.; Nobes, C.D.; Hall, A.; Parker, P.J. PRK1 is targeted to endosomes by the small GTPase, RhoB. J. Biol. Chem. 1998, 273, 4811–4814. [Google Scholar] [CrossRef]
- Gampel, A.; Parker, P.J.; Mellor, H. Regulation of epidermal growth factor receptor traffic by the small GTPase rhoB. Curr. Biol. 1999, 9, 955–958. [Google Scholar] [CrossRef]
- Gong, X.; Didan, Y.; Lock, J.G.; Stromblad, S. KIF13A-regulated RhoB plasma membrane localization governs membrane blebbing and blebby amoeboid cell migration. EMBO J. 2018, 37. [Google Scholar] [CrossRef]
- Baschieri, F.; Confalonieri, S.; Bertalot, G.; Di Fiore, P.P.; Dietmaier, W.; Leist, M.; Crespo, P.; Macara, I.G.; Farhan, H. Spatial control of Cdc42 signalling by a GM130-RasGRF complex regulates polarity and tumorigenesis. Nat. Commun. 2014, 5, 4839. [Google Scholar] [CrossRef] [PubMed]
- Iden, S.; Collard, J.G. Crosstalk between small GTPases and polarity proteins in cell polarization. Nat. Rev. Mol. Cell Biol. 2008, 9, 846–859. [Google Scholar] [CrossRef]
- Vigil, D.; Cherfils, J.; Rossman, K.L.; Der, C.J. Ras superfamily GEFs and GAPs: Validated and tractable targets for cancer therapy? Nat. Rev. Cancer 2010, 10, 842–857. [Google Scholar] [CrossRef]
- Viaud, J.; Gaits-Iacovoni, F.; Payrastre, B. Regulation of the DH-PH tandem of guanine nucleotide exchange factor for Rho GTPases by phosphoinositides. Adv. Biol. Regul. 2012, 52, 303–314. [Google Scholar] [CrossRef]
- Lemmon, M.A. Membrane recognition by phospholipid-binding domains. Nat. Rev. Mol. Cell Biol. 2008, 9, 99–111. [Google Scholar] [CrossRef]
- Hendrick, J.; Franz-Wachtel, M.; Moeller, Y.; Schmid, S.; Macek, B.; Olayioye, M.A. The polarity protein Scribble positions DLC3 at adherens junctions to regulate Rho signaling. J. Cell Sci. 2016, 129, 3583–3596. [Google Scholar] [CrossRef]
- Braun, A.C.; Olayioye, M.A. Rho regulation: DLC proteins in space and time. Cell. Signal. 2015, 27, 1643–1651. [Google Scholar] [CrossRef]
- Frank, S.R.; Bell, J.H.; Frodin, M.; Hansen, S.H. A betaPIX-PAK2 complex confers protection against Scrib-dependent and cadherin-mediated apoptosis. Curr. Biol. 2012, 22, 1747–1754. [Google Scholar] [CrossRef]
- Braun, A.C.; Hendrick, J.; Eisler, S.A.; Schmid, S.; Hausser, A.; Olayioye, M.A. The Rho-specific GAP protein DLC3 coordinates endocytic membrane trafficking. J. Cell Sci. 2015, 128, 1386–1399. [Google Scholar] [CrossRef] [PubMed]
- Noll, B.; Benz, D.; Frey, Y.; Meyer, F.; Lauinger, M.; Eisler, S.A.; Schmid, S.; Hordijk, P.L.; Olayioye, M.A. DLC3 suppresses MT1-MMP-dependent matrix degradation by controlling RhoB and actin remodeling at endosomal membranes. J. Cell Sci. 2019, 132. [Google Scholar] [CrossRef] [PubMed]
- Vega, F.M.; Colomba, A.; Reymond, N.; Thomas, M.; Ridley, A.J. RhoB regulates cell migration through altered focal adhesion dynamics. Open Biol. 2012, 2, 120076. [Google Scholar] [CrossRef] [PubMed]
- Arnette, C.; Frye, K.; Kaverina, I. Microtubule and Actin Interplay Drive Intracellular c-Src Trafficking. PLoS ONE 2016, 11, e0148996. [Google Scholar] [CrossRef]
- Pathak, R.; Delorme-Walker, V.D.; Howell, M.C.; Anselmo, A.N.; White, M.A.; Bokoch, G.M.; Dermardirossian, C. The microtubule-associated Rho activating factor GEF-H1 interacts with exocyst complex to regulate vesicle traffic. Dev. Cell 2012, 23, 397–411. [Google Scholar] [CrossRef]
- Eisler, S.A.; Curado, F.; Link, G.; Schulz, S.; Noack, M.; Steinke, M.; Olayioye, M.A.; Hausser, A. A Rho signaling network links microtubules to PKD controlled carrier transport to focal adhesions. ELife 2018, 7. [Google Scholar] [CrossRef]
- Azoitei, M.L.; Noh, J.; Marston, D.J.; Roudot, P.; Marshall, C.B.; Daugird, T.A.; Lisanza, S.L.; Sandi, M.J.; Ikura, M.; Sondek, J.; et al. Spatiotemporal dynamics of GEF-H1 activation controlled by microtubule- and Src-mediated pathways. J. Cell Biol. 2019, 218, 3077–3097. [Google Scholar] [CrossRef]
- Jacquemet, G.; Humphries, M.J. IQGAP1 is a key node within the small GTPase network. Small Gtpases 2013, 4, 199–207. [Google Scholar] [CrossRef][Green Version]
- Wilkinson, B.; Li, J.; Coba, M.P. Synaptic GAP and GEF Complexes Cluster Proteins Essential for GTP Signaling. Sci. Rep. 2017, 7, 5272. [Google Scholar] [CrossRef]
- Um, K.; Niu, S.; Duman, J.G.; Cheng, J.X.; Tu, Y.K.; Schwechter, B.; Liu, F.; Hiles, L.; Narayanan, A.S.; Ash, R.T.; et al. Dynamic control of excitatory synapse development by a Rac1 GEF/GAP regulatory complex. Dev. Cell 2014, 29, 701–715. [Google Scholar] [CrossRef]
- Vega, F.M.; Ridley, A.J. Rho GTPases in cancer cell biology. FEBS Lett. 2008, 582, 2093–2101. [Google Scholar] [CrossRef] [PubMed]
- Farhan, H.; Rabouille, C. Signalling to and from the secretory pathway. J. Cell Sci. 2011, 124, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Luna, A.; Matas, O.B.; Martinez-Menarguez, J.A.; Mato, E.; Duran, J.M.; Ballesta, J.; Way, M.; Egea, G. Regulation of protein transport from the Golgi complex to the endoplasmic reticulum by CDC42 and N-WASP. Mol. Biol. Cell 2002, 13, 866–879. [Google Scholar] [CrossRef] [PubMed]
- Matas, O.B.; Martinez-Menarguez, J.A.; Egea, G. Association of Cdc42/N-WASP/Arp2/3 signaling pathway with Golgi membranes. Traffic 2004, 5, 838–846. [Google Scholar] [CrossRef]
- Osmani, N.; Peglion, F.; Chavrier, P.; Etienne-Manneville, S. Cdc42 localization and cell polarity depend on membrane traffic. J. Cell Biol. 2010, 191, 1261–1269. [Google Scholar] [CrossRef]
- Egorov, M.V.; Capestrano, M.; Vorontsova, O.A.; Di Pentima, A.; Egorova, A.V.; Mariggio, S.; Ayala, M.I.; Tete, S.; Gorski, J.L.; Luini, A.; et al. Faciogenital dysplasia protein (FGD1) regulates export of cargo proteins from the golgi complex via Cdc42 activation. Mol. Biol. Cell 2009, 20, 2413–2427. [Google Scholar] [CrossRef]
- Hehnly, H.; Longhini, K.M.; Chen, J.L.; Stamnes, M. Retrograde Shiga toxin trafficking is regulated by ARHGAP21 and Cdc42. Mol. Biol. Cell 2009, 20, 4303–4312. [Google Scholar] [CrossRef]
- Park, S.Y.; Yang, J.S.; Schmider, A.B.; Soberman, R.J.; Hsu, V.W. Coordinated regulation of bidirectional COPI transport at the Golgi by CDC42. Nature 2015, 521, 529–532. [Google Scholar] [CrossRef]
- Musch, A.; Cohen, D.; Kreitzer, G.; Rodriguez-Boulan, E. cdc42 regulates the exit of apical and basolateral proteins from the trans-Golgi network. EMBO J. 2001, 20, 2171–2179. [Google Scholar] [CrossRef]
- Dubois, T.; Paleotti, O.; Mironov, A.A.; Fraisier, V.; Stradal, T.E.; De Matteis, M.A.; Franco, M.; Chavrier, P. Golgi-localized GAP for Cdc42 functions downstream of ARF1 to control Arp2/3 complex and F-actin dynamics. Nat. Cell Biol. 2005, 7, 353–364. [Google Scholar] [CrossRef]
- Nalbant, P.; Hodgson, L.; Kraynov, V.; Toutchkine, A.; Hahn, K.M. Activation of endogenous Cdc42 visualized in living cells. Science 2004, 305, 1615–1619. [Google Scholar] [CrossRef] [PubMed]
- Herrington, K.A.; Trinh, A.L.; Dang, C.; O’Shaughnessy, E.; Hahn, K.M.; Gratton, E.; Digman, M.A.; Sutterlin, C. Spatial analysis of Cdc42 activity reveals a role for plasma membrane-associated Cdc42 in centrosome regulation. Mol. Biol. Cell 2017, 28, 2135–2145. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, L.; Shen, F.; Hahn, K. Biosensors for characterizing the dynamics of rho family GTPases in living cells. Curr. Protoc. Cell Biol. 2010, 46, 14.11.11–14.11.26. [Google Scholar] [CrossRef] [PubMed]
- Hodgson, L.; Spiering, D.; Sabouri-Ghomi, M.; Dagliyan, O.; DerMardirossian, C.; Danuser, G.; Hahn, K.M. FRET binding antenna reports spatiotemporal dynamics of GDI-Cdc42 GTPase interactions. Nat. Chem. Biol. 2016, 12, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Kage, F.; Steffen, A.; Ellinger, A.; Ranftler, C.; Gehre, C.; Brakebusch, C.; Pavelka, M.; Stradal, T.; Rottner, K. FMNL2 and -3 regulate Golgi architecture and anterograde transport downstream of Cdc42. Sci. Rep. 2017, 7, 9791. [Google Scholar] [CrossRef]
- Kodani, A.; Kristensen, I.; Huang, L.; Sutterlin, C. GM130-dependent control of Cdc42 activity at the Golgi regulates centrosome organization. Mol. Biol. Cell 2009, 20, 1192–1200. [Google Scholar] [CrossRef]
- Salazar, M.A.; Kwiatkowski, A.V.; Pellegrini, L.; Cestra, G.; Butler, M.H.; Rossman, K.L.; Serna, D.M.; Sondek, J.; Gertler, F.B.; De Camilli, P. Tuba, a novel protein containing bin/amphiphysin/Rvs and Dbl homology domains, links dynamin to regulation of the actin cytoskeleton. J. Biol. Chem. 2003, 278, 49031–49043. [Google Scholar] [CrossRef]
- Estrada, L.; Caron, E.; Gorski, J.L. Fgd1, the Cdc42 guanine nucleotide exchange factor responsible for faciogenital dysplasia, is localized to the subcortical actin cytoskeleton and Golgi membrane. Hum. Mol. Genet. 2001, 10, 485–495. [Google Scholar] [CrossRef]
- Egorov, M.V.; Polishchuk, R.S. Emerging role of Cdc42-specific guanine nucleotide exchange factors as regulators of membrane trafficking in health and disease. Tissue Cell 2017, 49, 157–162. [Google Scholar] [CrossRef]
- Egorov, M.; Polishchuk, R. Identification of CDC42 Effectors Operating in FGD1-Dependent Trafficking at the Golgi. Front. Cell Dev. Biol. 2019, 7, 7. [Google Scholar] [CrossRef]
- Fitzpatrick, E.R.; Hu, T.; Ciccarelli, B.T.; Whitehead, I.P. Regulation of vesicle transport and cell motility by Golgi-localized Dbs. Small Gtpases 2014, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Makhoul, C.; Gosavi, P.; Duffield, R.; Delbridge, B.; Williamson, N.A.; Gleeson, P.A. Intersectin-1 interacts with the golgin GCC88 to couple the actin network and Golgi architecture. Mol. Biol. Cell 2019, 30, 370–386. [Google Scholar] [CrossRef] [PubMed]
- Friesland, A.; Zhao, Y.; Chen, Y.H.; Wang, L.; Zhou, H.; Lu, Q. Small molecule targeting Cdc42-intersectin interaction disrupts Golgi organization and suppresses cell motility. Proc. Natl. Acad. Sci. USA 2013, 110, 1261–1266. [Google Scholar] [CrossRef] [PubMed]
- Bruurs, L.J.M.; van der Net, M.C.; Zwakenberg, S.; Zwartkruis, F.J.T.; Bos, J.L. A Tuba/Cdc42/Par6A complex is required to ensure singularity in apical domain formation during enterocyte polarization. PLoS ONE 2018, 13, e0207159. [Google Scholar] [CrossRef]
- Kovacs, E.M.; Makar, R.S.; Gertler, F.B. Tuba stimulates intracellular N-WASP-dependent actin assembly. J. Cell Sci. 2006, 119, 2715–2726. [Google Scholar] [CrossRef]
- Reinhard, N.R.; Van Der Niet, S.; Chertkova, A.; Postma, M.; Hordijk, P.L.; Gadella, T.W.J., Jr.; Goedhart, J. Identification of guanine nucleotide exchange factors that increase Cdc42 activity in primary human endothelial cells. Small Gtpases 2019, 1–15. [Google Scholar] [CrossRef]
- Müller, P.M.; Rademacher, J.; Bagshaw, R.D.; Alp, K.M.; Giudice, G.; Heinrich, L.E.; Barth, C.; Eccles, R.L.; Sanchez-Castro, M.; Brandenburg, L.; et al. Spatial Organization of Rho GTPase signaling by RhoGEF/RhoGAP proteins. Biorxiv 2018. [Google Scholar] [CrossRef]
- Bhattacharya, K.; Swaminathan, K.; Peche, V.S.; Clemen, C.S.; Knyphausen, P.; Lammers, M.; Noegel, A.A.; Rastetter, R.H. Novel Coronin7 interactions with Cdc42 and N-WASP regulate actin organization and Golgi morphology. Sci. Rep. 2016, 6, 25411. [Google Scholar] [CrossRef]
- Rybakin, V. Role of Mammalian coronin 7 in the biosynthetic pathway. Sub-Cell. Biochem. 2008, 48, 110–115. [Google Scholar] [CrossRef]
- Dighe, S.A.; Kozminski, K.G. Secretory vesicles deliver Cdc42p to sites of polarized growth in S. cerevisiae. PLoS ONE 2014, 9, e99494. [Google Scholar] [CrossRef]
- Michaelson, D.; Silletti, J.; Murphy, G.; D’Eustachio, P.; Rush, M.; Philips, M.R. Differential localization of Rho GTPases in live cells: Regulation by hypervariable regions and RhoGDI binding. J. Cell Biol. 2001, 152, 111–126. [Google Scholar] [CrossRef] [PubMed]
- Pertz, O.; Hodgson, L.; Klemke, R.L.; Hahn, K.M. Spatiotemporal dynamics of RhoA activity in migrating cells. Nature 2006, 440, 1069–1072. [Google Scholar] [CrossRef] [PubMed]
- Zilberman, Y.; Alieva, N.O.; Miserey-Lenkei, S.; Lichtenstein, A.; Kam, Z.; Sabanay, H.; Bershadsky, A. Involvement of the Rho-mDia1 pathway in the regulation of Golgi complex architecture and dynamics. Mol. Biol. Cell 2011, 22, 2900–2911. [Google Scholar] [CrossRef] [PubMed]
- Cole, N.B.; Sciaky, N.; Marotta, A.; Song, J.; Lippincott-Schwartz, J. Golgi dispersal during microtubule disruption: Regeneration of Golgi stacks at peripheral endoplasmic reticulum exit sites. Mol. Biol. Cell 1996, 7, 631–650. [Google Scholar] [CrossRef]
- Quassollo, G.; Wojnacki, J.; Salas, D.A.; Gastaldi, L.; Marzolo, M.P.; Conde, C.; Bisbal, M.; Couve, A.; Caceres, A. A RhoA Signaling Pathway Regulates Dendritic Golgi Outpost Formation. Curr. Biol. 2015, 25, 971–982. [Google Scholar] [CrossRef]
- Ren, Y.; Li, R.; Zheng, Y.; Busch, H. Cloning and characterization of GEF-H1, a microtubule-associated guanine nucleotide exchange factor for Rac and Rho GTPases. J. Biol. Chem. 1998, 273, 34954–34960. [Google Scholar] [CrossRef]
- Tao, T.; Sun, J.; Peng, Y.; Li, Y.; Wang, P.; Chen, X.; Zhao, W.; Zheng, Y.Y.; Wei, L.; Wang, W.; et al. Golgi-resident TRIO regulates membrane trafficking during neurite outgrowth. J. Biol. Chem. 2019, 294, 10954–10968. [Google Scholar] [CrossRef]
- Tang, B.L. Emerging aspects of membrane traffic in neuronal dendrite growth. Biochim. Et Biophys. Acta 2008, 1783, 169–176. [Google Scholar] [CrossRef][Green Version]
- Camera, P.; da Silva, J.S.; Griffiths, G.; Giuffrida, M.G.; Ferrara, L.; Schubert, V.; Imarisio, S.; Silengo, L.; Dotti, C.G.; Di Cunto, F. Citron-N is a neuronal Rho-associated protein involved in Golgi organization through actin cytoskeleton regulation. Nat. Cell Biol. 2003, 5, 1071–1078. [Google Scholar] [CrossRef]
- Camera, P.; Schubert, V.; Pellegrino, M.; Berto, G.; Vercelli, A.; Muzzi, P.; Hirsch, E.; Altruda, F.; Dotti, C.G.; Di Cunto, F. The RhoA-associated protein Citron-N controls dendritic spine maintenance by interacting with spine-associated Golgi compartments. EMBO Rep. 2008, 9, 384–392. [Google Scholar] [CrossRef]
- Espinosa, E.J.; Calero, M.; Sridevi, K.; Pfeffer, S.R. RhoBTB3: A Rho GTPase-family ATPase required for endosome to Golgi transport. Cell 2009, 137, 938–948. [Google Scholar] [CrossRef] [PubMed]
- Lu, A.; Pfeffer, S.R. Golgi-associated RhoBTB3 targets cyclin E for ubiquitylation and promotes cell cycle progression. J. Cell Biol. 2013, 203, 233–250. [Google Scholar] [CrossRef] [PubMed]
- Riento, K.; Totty, N.; Villalonga, P.; Garg, R.; Guasch, R.; Ridley, A.J. RhoE function is regulated by ROCK I-mediated phosphorylation. EMBO, J. 2005, 24, 1170–1180. [Google Scholar] [CrossRef] [PubMed]
- Riento, K.; Guasch, R.M.; Garg, R.; Jin, B.; Ridley, A.J. RhoE binds to ROCK I and inhibits downstream signaling. Mol. Cell. Biol. 2003, 23, 4219–4229. [Google Scholar] [CrossRef]
- Blom, M.; Reis, K.; Nehru, V.; Blom, H.; Gad, A.K.; Aspenstrom, P. RhoD is a Golgi component with a role in anterograde protein transport from the ER to the plasma membrane. Exp. Cell Res. 2015, 333, 208–219. [Google Scholar] [CrossRef]
- Gad, A.K.; Nehru, V.; Ruusala, A.; Aspenstrom, P. RhoD regulates cytoskeletal dynamics via the actin nucleation-promoting factor WASp homologue associated with actin Golgi membranes and microtubules. Mol. Biol. Cell 2012, 23, 4807–4819. [Google Scholar] [CrossRef]
- Russo, A.J.; Mathiowetz, A.J.; Hong, S.; Welch, M.D.; Campellone, K.G. Rab1 recruits WHAMM during membrane remodeling but limits actin nucleation. Mol. Biol. Cell 2016, 27, 967–978. [Google Scholar] [CrossRef]
- Chiang, S.H.; Baumann, C.A.; Kanzaki, M.; Thurmond, D.C.; Watson, R.T.; Neudauer, C.L.; Macara, I.G.; Pessin, J.E.; Saltiel, A.R. Insulin-stimulated GLUT4 translocation requires the CAP-dependent activation of TC10. Nature 2001, 410, 944–948. [Google Scholar] [CrossRef]
- Cheng, J.; Wang, H.; Guggino, W.B. Regulation of cystic fibrosis transmembrane regulator trafficking and protein expression by a Rho family small GTPase TC10. J. Biol. Chem. 2005, 280, 3731–3739. [Google Scholar] [CrossRef]
- Cuadra, A.E.; Kuo, S.H.; Kawasaki, Y.; Bredt, D.S.; Chetkovich, D.M. AMPA receptor synaptic targeting regulated by stargazin interactions with the Golgi-resident PDZ protein Npist. J. Neurosci. 2004, 24, 7491–7502. [Google Scholar] [CrossRef]
- Inoue, M.; Chang, L.; Hwang, J.; Chiang, S.H.; Saltiel, A.R. The exocyst complex is required for targeting of Glut4 to the plasma membrane by insulin. Nature 2003, 422, 629–633. [Google Scholar] [CrossRef] [PubMed]
- Kawase, K.; Nakamura, T.; Takaya, A.; Aoki, K.; Namikawa, K.; Kiyama, H.; Inagaki, S.; Takemoto, H.; Saltiel, A.R.; Matsuda, M. GTP hydrolysis by the Rho family GTPase TC10 promotes exocytic vesicle fusion. Dev. Cell 2006, 11, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Watson, R.T.; Furukawa, M.; Chiang, S.H.; Boeglin, D.; Kanzaki, M.; Saltiel, A.R.; Pessin, J.E. The exocytotic trafficking of TC10 occurs through both classical and nonclassical secretory transport pathways in 3T3L1 adipocytes. Mol. Cell. Biol. 2003, 23, 961–974. [Google Scholar] [CrossRef] [PubMed]
- Miserey-Lenkei, S.; Bousquet, H.; Pylypenko, O.; Bardin, S.; Dimitrov, A.; Bressanelli, G.; Bonifay, R.; Fraisier, V.; Guillou, C.; Bougeret, C.; et al. Coupling fission and exit of RAB6 vesicles at Golgi hotspots through kinesin-myosin interactions. Nat. Commun. 2017, 8, 1254. [Google Scholar] [CrossRef]
- Shibata, S.; Kawanai, T.; Hara, T.; Yamamoto, A.; Chaya, T.; Tokuhara, Y.; Tsuji, C.; Sakai, M.; Tachibana, T.; Inagaki, S. ARHGEF10 directs the localization of Rab8 to Rab6-positive executive vesicles. J. Cell Sci. 2016, 129, 3620–3634. [Google Scholar] [CrossRef]
- Shibata, S.; Teshima, Y.; Niimi, K.; Inagaki, S. Involvement of ARHGEF10, GEF for RhoA, in Rab6/Rab8-mediating membrane traffic. Small Gtpases 2019, 10, 169–177. [Google Scholar] [CrossRef]
- Chi, X.; Wang, S.; Huang, Y.; Stamnes, M.; Chen, J.L. Roles of rho GTPases in intracellular transport and cellular transformation. Int. J. Mol. Sci. 2013, 14, 7089–7108. [Google Scholar] [CrossRef]
- Croise, P.; Estay-Ahumada, C.; Gasman, S.; Ory, S. Rho GTPases, phosphoinositides, and actin: A tripartite framework for efficient vesicular trafficking. Small Gtpases 2014, 5, e29469. [Google Scholar] [CrossRef]
- Symons, M.; Rusk, N. Control of vesicular trafficking by Rho GTPases. Curr. Biol. 2003, 13, R409–R418. [Google Scholar] [CrossRef]
- Adamson, P.; Paterson, H.F.; Hall, A. Intracellular localization of the P21rho proteins. J. Cell Biol. 1992, 119, 617–627. [Google Scholar] [CrossRef]
- Gampel, A.; Mellor, H. Small interfering RNAs as a tool to assign Rho GTPase exchange-factor function in vivo. Biochem. J. 2002, 366, 393–398. [Google Scholar] [CrossRef]
- Neel, N.F.; Lapierre, L.A.; Goldenring, J.R.; Richmond, A. RhoB plays an essential role in CXCR2 sorting decisions. J. Cell Sci. 2007, 120, 1559–1571. [Google Scholar] [CrossRef]
- Fernandez-Borja, M.; Janssen, L.; Verwoerd, D.; Hordijk, P.; Neefjes, J. RhoB regulates endosome transport by promoting actin assembly on endosomal membranes through Dia1. J. Cell Sci. 2005, 118, 2661–2670. [Google Scholar] [CrossRef]
- Wallar, B.J.; Deward, A.D.; Resau, J.H.; Alberts, A.S. RhoB and the mammalian Diaphanous-related formin mDia2 in endosome trafficking. Exp. Cell Res. 2007, 313, 560–571. [Google Scholar] [CrossRef]
- Sandilands, E.; Cans, C.; Fincham, V.J.; Brunton, V.G.; Mellor, H.; Prendergast, G.C.; Norman, J.C.; Superti-Furga, G.; Frame, M.C. RhoB and actin polymerization coordinate Src activation with endosome-mediated delivery to the membrane. Dev. Cell 2004, 7, 855–869. [Google Scholar] [CrossRef]
- Tominaga, T.; Sahai, E.; Chardin, P.; McCormick, F.; Courtneidge, S.A.; Alberts, A.S. Diaphanous-related formins bridge Rho GTPase and Src tyrosine kinase signaling. Mol. Cell 2000, 5, 13–25. [Google Scholar] [CrossRef]
- Wherlock, M.; Gampel, A.; Futter, C.; Mellor, H. Farnesyltransferase inhibitors disrupt EGF receptor traffic through modulation of the RhoB GTPase. J. Cell Sci. 2004, 117, 3221–3231. [Google Scholar] [CrossRef]
- Murphy, C.; Saffrich, R.; Grummt, M.; Gournier, H.; Rybin, V.; Rubino, M.; Auvinen, P.; Lutcke, A.; Parton, R.G.; Zerial, M. Endosome dynamics regulated by a Rho protein. Nature 1996, 384, 427–432. [Google Scholar] [CrossRef]
- Gasman, S.; Kalaidzidis, Y.; Zerial, M. RhoD regulates endosome dynamics through Diaphanous-related Formin and Src tyrosine kinase. Nat. Cell Biol. 2003, 5, 195–204. [Google Scholar] [CrossRef]
- Sandilands, E.; Brunton, V.G.; Frame, M.C. The membrane targeting and spatial activation of Src, Yes and Fyn is influenced by palmitoylation and distinct RhoB/RhoD endosome requirements. J. Cell Sci. 2007, 120, 2555–2564. [Google Scholar] [CrossRef]
- Murphy, C.; Saffrich, R.; Olivo-Marin, J.C.; Giner, A.; Ansorge, W.; Fotsis, T.; Zerial, M. Dual function of rhoD in vesicular movement and cell motility. Eur. J. Cell Biol. 2001, 80, 391–398. [Google Scholar] [CrossRef]
- Nehru, V.; Voytyuk, O.; Lennartsson, J.; Aspenstrom, P. RhoD binds the Rab5 effector Rabankyrin-5 and has a role in trafficking of the platelet-derived growth factor receptor. Traffic 2013, 14, 1242–1254. [Google Scholar] [CrossRef]
- Marcos-Ramiro, B.; Garcia-Weber, D.; Barroso, S.; Feito, J.; Ortega, M.C.; Cernuda-Morollon, E.; Reglero-Real, N.; Fernandez-Martin, L.; Duran, M.C.; Alonso, M.A.; et al. RhoB controls endothelial barrier recovery by inhibiting Rac1 trafficking to the cell border. J. Cell Biol. 2016, 213, 385–402. [Google Scholar] [CrossRef]
- Huang, M.; Satchell, L.; Duhadaway, J.B.; Prendergast, G.C.; Laury-Kleintop, L.D. RhoB links PDGF signaling to cell migration by coordinating activation and localization of Cdc42 and Rac. J. Cell. Biochem. 2011, 112, 1572–1584. [Google Scholar] [CrossRef]
- Menard, L.; Parker, P.J.; Kermorgant, S. Receptor tyrosine kinase c-Met controls the cytoskeleton from different endosomes via different pathways. Nat. Commun. 2014, 5, 3907. [Google Scholar] [CrossRef]
- Strehle, A.; Schleicher, M.; Faix, J. Trix, a novel Rac guanine-nucleotide exchange factor from Dictyostelium discoideum is an actin-binding protein and accumulates at endosomes. Eur. J. Cell Biol. 2006, 85, 1035–1045. [Google Scholar] [CrossRef]
- Palamidessi, A.; Frittoli, E.; Garre, M.; Faretta, M.; Mione, M.; Testa, I.; Diaspro, A.; Lanzetti, L.; Scita, G.; Di Fiore, P.P. Endocytic trafficking of Rac is required for the spatial restriction of signaling in cell migration. Cell 2008, 134, 135–147. [Google Scholar] [CrossRef]
- Donaldson, J.G.; Porat-Shliom, N.; Cohen, L.A. Clathrin-independent endocytosis: A unique platform for cell signaling and PM remodeling. Cell. Signal. 2009, 21, 1–6. [Google Scholar] [CrossRef]
- Margiotta, A.; Progida, C.; Bakke, O.; Bucci, C. Rab7a regulates cell migration through Rac1 and vimentin. Biochim. Et Biophys. Acta. Mol. Cell Res. 2017, 1864, 367–381. [Google Scholar] [CrossRef]
- Garcia-Weber, D.; Millan, J. Parallels between single cell migration and barrier formation: The case of RhoB and Rac1 trafficking. Small Gtpases 2018, 9, 332–338. [Google Scholar] [CrossRef][Green Version]
- Bouchet, J.; Del Rio-Iniguez, I.; Lasserre, R.; Aguera-Gonzalez, S.; Cuche, C.; Danckaert, A.; McCaffrey, M.W.; Di Bartolo, V.; Alcover, A. Rac1-Rab11-FIP3 regulatory hub coordinates vesicle traffic with actin remodeling and T-cell activation. EMBO, J. 2016, 35, 1160–1174. [Google Scholar] [CrossRef]
- Bravo-Cordero, J.J.; Cordani, M.; Soriano, S.F.; Diez, B.; Munoz-Agudo, C.; Casanova-Acebes, M.; Boullosa, C.; Guadamillas, M.C.; Ezkurdia, I.; Gonzalez-Pisano, D.; et al. A novel high-content analysis tool reveals Rab8-driven cytoskeletal reorganization through Rho GTPases, calpain and MT1-MMP. J. Cell Sci. 2016, 129, 1734–1749. [Google Scholar] [CrossRef]
- Pelish, H.E.; Peterson, J.R.; Salvarezza, S.B.; Rodriguez-Boulan, E.; Chen, J.L.; Stamnes, M.; Macia, E.; Feng, Y.; Shair, M.D.; Kirchhausen, T. Secramine inhibits Cdc42-dependent functions in cells and Cdc42 activation in vitro. Nat. Chem. Biol. 2006, 2, 39–46. [Google Scholar] [CrossRef]
- de Toledo, M.; Senic-Matuglia, F.; Salamero, J.; Uze, G.; Comunale, F.; Fort, P.; Blangy, A. The GTP/GDP cycling of rho GTPase TCL is an essential regulator of the early endocytic pathway. Mol. Biol. Cell 2003, 14, 4846–4856. [Google Scholar] [CrossRef]
- Vignal, E.; Blangy, A.; Martin, M.; Gauthier-Rouviere, C.; Fort, P. Kinectin is a key effector of RhoG microtubule-dependent cellular activity. Mol. Cell. Biol. 2001, 21, 8022–8034. [Google Scholar] [CrossRef]
- Aspenstrom, P.; Fransson, A.; Saras, J. Rho GTPases have diverse effects on the organization of the actin filament system. Biochem. J. 2004, 377, 327–337. [Google Scholar] [CrossRef]
- Berthold, J.; Schenkova, K.; Ramos, S.; Miura, Y.; Furukawa, M.; Aspenstrom, P.; Rivero, F. Characterization of RhoBTB-dependent Cul3 ubiquitin ligase complexes--evidence for an autoregulatory mechanism. Exp. Cell Res. 2008, 314, 3453–3465. [Google Scholar] [CrossRef]
- Wennerberg, K.; Der, C.J. Rho-family GTPases: it’s not only Rac and Rho (and I like it). J. Cell Sci. 2004, 117, 1301–1312. [Google Scholar] [CrossRef]
- Pridgeon, J.W.; Webber, E.A.; Sha, D.; Li, L.; Chin, L.S. Proteomic analysis reveals Hrs ubiquitin-interacting motif-mediated ubiquitin signaling in multiple cellular processes. FEBS J. 2009, 276, 118–131. [Google Scholar] [CrossRef]
- Hehnly, H.; Xu, W.; Chen, J.L.; Stamnes, M. Cdc42 regulates microtubule-dependent Golgi positioning. Traffic 2010, 11, 1067–1078. [Google Scholar] [CrossRef]
- Wu, W.J.; Tu, S.; Cerione, R.A. Activated Cdc42 sequesters c-Cbl and prevents EGF receptor degradation. Cell 2003, 114, 715–725. [Google Scholar] [CrossRef]
- Randazzo, P.A. RhoD, Src, and hDia2C in endosome motility. Dev. Cell 2003, 4, 287–288. [Google Scholar] [CrossRef][Green Version]
- Murphy, G.A.; Jillian, S.A.; Michaelson, D.; Philips, M.R.; D’Eustachio, P.; Rush, M.G. Signaling mediated by the closely related mammalian Rho family GTPases TC10 and Cdc42 suggests distinct functional pathways. Cell Growth Differ. Mol. Biol. J. Am. Assoc. Cancer Res. 2001, 12, 157–167. [Google Scholar]
- Hobbs, G.A.; Der, C.J.; Rossman, K.L. RAS isoforms and mutations in cancer at a glance. J. Cell Sci. 2016, 129, 1287–1292. [Google Scholar] [CrossRef]
- Cook, D.R.; Rossman, K.L.; Der, C.J. Rho guanine nucleotide exchange factors: Regulators of Rho GTPase activity in development and disease. Oncogene 2014, 33, 4021–4035. [Google Scholar] [CrossRef]
- Yoo, H.Y.; Sung, M.K.; Lee, S.H.; Kim, S.; Lee, H.; Park, S.; Kim, S.C.; Lee, B.; Rho, K.; Lee, J.E.; et al. A recurrent inactivating mutation in RHOA GTPase in angioimmunoblastic T cell lymphoma. Nat. Genet. 2014, 46, 371–375. [Google Scholar] [CrossRef]
- Cools, J. RHOA mutations in peripheral T cell lymphoma. Nat. Genet. 2014, 46, 320–321. [Google Scholar] [CrossRef]
- Sakata-Yanagimoto, M.; Enami, T.; Yoshida, K.; Shiraishi, Y.; Ishii, R.; Miyake, Y.; Muto, H.; Tsuyama, N.; Sato-Otsubo, A.; Okuno, Y.; et al. Somatic RHOA mutation in angioimmunoblastic T cell lymphoma. Nat. Genet. 2014, 46, 171–175. [Google Scholar] [CrossRef]
- Palomero, T.; Couronne, L.; Khiabanian, H.; Kim, M.Y.; Ambesi-Impiombato, A.; Perez-Garcia, A.; Carpenter, Z.; Abate, F.; Allegretta, M.; Haydu, J.E.; et al. Recurrent mutations in epigenetic regulators, RHOA and FYN kinase in peripheral T cell lymphomas. Nat. Genet. 2014, 46, 166–170. [Google Scholar] [CrossRef]
- Zhou, J.; Hayakawa, Y.; Wang, T.C.; Bass, A.J. RhoA mutations identified in diffuse gastric cancer. Cancer Cell 2014, 26, 9–11. [Google Scholar] [CrossRef]
- Kakiuchi, M.; Nishizawa, T.; Ueda, H.; Gotoh, K.; Tanaka, A.; Hayashi, A.; Yamamoto, S.; Tatsuno, K.; Katoh, H.; Watanabe, Y.; et al. Recurrent gain-of-function mutations of RHOA in diffuse-type gastric carcinoma. Nat. Genet. 2014, 46, 583–587. [Google Scholar] [CrossRef] [PubMed]
- Aspenstrom, P. Activated Rho GTPases in Cancer-The Beginning of a New Paradigm. Int. J. Mol. Sci. 2018, 19, 3949. [Google Scholar] [CrossRef] [PubMed]
- Joffre, C.; Barrow, R.; Menard, L.; Calleja, V.; Hart, I.R.; Kermorgant, S. A direct role for Met endocytosis in tumorigenesis. Nat. Cell Biol. 2011, 13, 827–837. [Google Scholar] [CrossRef]
- Barrow-McGee, R.; Kermorgant, S. Met endosomal signalling: In the right place, at the right time. Int. J. Biochem. Cell Biol. 2014, 49, 69–74. [Google Scholar] [CrossRef]
- Goldenring, J.R. A central role for vesicle trafficking in epithelial neoplasia: Intracellular highways to carcinogenesis. Nat. Rev. Cancer 2013, 13, 813–820. [Google Scholar] [CrossRef]
- Liu, A.; Du, W.; Liu, J.P.; Jessell, T.M.; Prendergast, G.C. RhoB alteration is necessary for apoptotic and antineoplastic responses to farnesyltransferase inhibitors. Mol. Cell. Biol. 2000, 20, 6105–6113. [Google Scholar] [CrossRef]
- Du, W.; Prendergast, G.C. Geranylgeranylated RhoB mediates suppression of human tumor cell growth by farnesyltransferase inhibitors. Cancer Res. 1999, 59, 5492–5496. [Google Scholar]
- Calvayrac, O.; Pradines, A.; Favre, G. RHOB expression controls the activity of serine/threonine protein phosphatase PP2A to modulate mesenchymal phenotype and invasion in non-small cell lung cancers. Small Gtpases 2018, 9, 339–344. [Google Scholar] [CrossRef]
- Bousquet, E.; Calvayrac, O.; Mazieres, J.; Lajoie-Mazenc, I.; Boubekeur, N.; Favre, G.; Pradines, A. RhoB loss induces Rac1-dependent mesenchymal cell invasion in lung cells through PP2A inhibition. Oncogene 2016, 35, 1760–1769. [Google Scholar] [CrossRef]
- Canguilhem, B.; Pradines, A.; Baudouin, C.; Boby, C.; Lajoie-Mazenc, I.; Charveron, M.; Favre, G. RhoB protects human keratinocytes from UVB-induced apoptosis through epidermal growth factor receptor signaling. J. Biol. Chem. 2005, 280, 43257–43263. [Google Scholar] [CrossRef]
- Huang, M.; Duhadaway, J.B.; Prendergast, G.C.; Laury-Kleintop, L.D. RhoB regulates PDGFR-beta trafficking and signaling in vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 2597–2605. [Google Scholar] [CrossRef] [PubMed]
- Calvayrac, O.; Mazieres, J.; Figarol, S.; Marty-Detraves, C.; Raymond-Letron, I.; Bousquet, E.; Farella, M.; Clermont-Taranchon, E.; Milia, J.; Rouquette, I.; et al. The RAS-related GTPase RHOB confers resistance to EGFR-tyrosine kinase inhibitors in non-small-cell lung cancer via an AKT-dependent mechanism. EMBO Mol. Med. 2017, 9, 238–250. [Google Scholar] [CrossRef] [PubMed]
- Adini, I.; Rabinovitz, I.; Sun, J.F.; Prendergast, G.C.; Benjamin, L.E. RhoB controls Akt trafficking and stage-specific survival of endothelial cells during vascular development. Genes Dev. 2003, 17, 2721–2732. [Google Scholar] [CrossRef] [PubMed]
- Zaoui, K.; Rajadurai, C.V.; Duhamel, S.; Park, M. Arf6 regulates RhoB subcellular localization to control cancer cell invasion. J. Cell Biol. 2019, 218, 3812–3826. [Google Scholar] [CrossRef]
- Durkin, M.E.; Ullmannova, V.; Guan, M.; Popescu, N.C. Deleted in liver cancer 3 (DLC-3), a novel Rho GTPase-activating protein, is downregulated in cancer and inhibits tumor cell growth. Oncogene 2007, 26, 4580–4589. [Google Scholar] [CrossRef]
- Holeiter, G.; Bischoff, A.; Braun, A.C.; Huck, B.; Erlmann, P.; Schmid, S.; Herr, R.; Brummer, T.; Olayioye, M.A. The RhoGAP protein Deleted in Liver Cancer 3 (DLC3) is essential for adherens junctions integrity. Oncogenesis 2012, 1, e13. [Google Scholar] [CrossRef]
- Stengel, K.; Zheng, Y. Cdc42 in oncogenic transformation, invasion, and tumorigenesis. Cell. Signal. 2011, 23, 1415–1423. [Google Scholar] [CrossRef]
- Cheng, C.M.; Li, H.; Gasman, S.; Huang, J.; Schiff, R.; Chang, E.C. Compartmentalized Ras proteins transform NIH 3T3 cells with different efficiencies. Mol. Cell. Biol. 2011, 31, 983–997. [Google Scholar] [CrossRef]
- Long, M.; Simpson, J.C. Rho GTPases operating at the Golgi complex: Implications for membrane traffic and cancer biology. Tissue Cell 2017, 49, 163–169. [Google Scholar] [CrossRef]
- Ayala, I.; Giacchetti, G.; Caldieri, G.; Attanasio, F.; Mariggio, S.; Tete, S.; Polishchuk, R.; Castronovo, V.; Buccione, R. Faciogenital dysplasia protein Fgd1 regulates invadopodia biogenesis and extracellular matrix degradation and is up-regulated in prostate and breast cancer. Cancer Res. 2009, 69, 747–752. [Google Scholar] [CrossRef]
- Luo, N.; Guo, J.; Chen, L.; Yang, W.; Qu, X.; Cheng, Z. ARHGAP10, downregulated in ovarian cancer, suppresses tumorigenicity of ovarian cancer cells. Cell Death Dis. 2016, 7, e2157. [Google Scholar] [CrossRef] [PubMed]
- Sakurai-Yageta, M.; Recchi, C.; Le Dez, G.; Sibarita, J.B.; Daviet, L.; Camonis, J.; D’Souza-Schorey, C.; Chavrier, P. The interaction of IQGAP1 with the exocyst complex is required for tumor cell invasion downstream of Cdc42 and RhoA. J. Cell Biol. 2008, 181, 985–998. [Google Scholar] [CrossRef] [PubMed]
- Gillingham, A.K.; Bertram, J.; Begum, F.; Munro, S. In vivo identification of GTPase interactors by mitochondrial relocalization and proximity biotinylation. ELife 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Lambrus, B.G.; Moyer, T.C.; Holland, A.J. Applying the auxin-inducible degradation system for rapid protein depletion in mammalian cells. Methods Cell Biol. 2018, 144, 107–135. [Google Scholar] [CrossRef]
- Schapira, M.; Calabrese, M.F.; Bullock, A.N.; Crews, C.M. Targeted protein degradation: Expanding the toolbox. Nat. Rev. Drug Discov. 2019. [Google Scholar] [CrossRef]
- Beyer, H.M.; Naumann, S.; Weber, W.; Radziwill, G. Optogenetic control of signaling in mammalian cells. Biotechnol. J. 2015, 10, 273–283. [Google Scholar] [CrossRef]
- Valon, L.; Etoc, F.; Remorino, A.; di Pietro, F.; Morin, X.; Dahan, M.; Coppey, M. Predictive Spatiotemporal Manipulation of Signaling Perturbations Using Optogenetics. Biophys. J. 2015, 109, 1785–1797. [Google Scholar] [CrossRef]
- De Beco, S.; Vaidziulyte, K.; Manzi, J.; Dalier, F.; di Federico, F.; Cornilleau, G.; Dahan, M.; Coppey, M. Optogenetic dissection of Rac1 and Cdc42 gradient shaping. Nat. Commun. 2018, 9, 4816. [Google Scholar] [CrossRef]
- Valon, L.; Marin-Llaurado, A.; Wyatt, T.; Charras, G.; Trepat, X. Optogenetic control of cellular forces and mechanotransduction. Nat. Commun. 2017, 8, 14396. [Google Scholar] [CrossRef]
- Yin, T.; Wu, Y.I. Optogenetics: Optical control of a photoactivatable Rac in living cells. Methods Mol. Biol. 2015, 1251, 277–289. [Google Scholar] [CrossRef]

| Rho GTPase | Location | GEF | GAP | Effectors | Function | Reference |
|---|---|---|---|---|---|---|
| RhoA | TGN | GEF-H1 Trio (?) | DLC3 | PLCε ROCK | Promotes cargo transport to the PM; Formation of Golgi outposts in neuronal cells | [41,46,82,83,84,85,87,89] |
| RhoB | Endosomes | Vav2 (?) | DLC3 | mDia1 mDia2 WAVE1 | Transport from endosomes to the lysosomes; Src movement and activation | [31,41,42,110,111,113,114,115] |
| Golgi complex | ? | ? | ? | ? | [46,81] | |
| Cdc42 | Golgi complex; Exocytic vesicles | Intersectin-1, Tuba, FGD1, Dbs-130 | ARHGAP21 | ARP2/3, N-WASP; FMNL2/3 | Anterograde trafficking through the Golgi; Golgi-to-PM transport; Retrograde trafficking from the Golgi to the ER | [33,53,54,55,56,57,58,59,60,61,62,65,66,68,70,71,72,76,78,81,140] |
| Early endosomes | β-Pix | ? | ? | Ccd42 delivery to the leading edge during directed cell migration | [55,141] | |
| RhoD | Trans-Golgi membranes | - | - | WHAMM | Golgi-to-PM transport | [95,96] |
| Endosomes | - | - | hDia2C | Inhibition of endosomal motility; Regulation of RTK trafficking | [119,120,121,122,142] | |
| RhoE | Trans-Golgi membranes | - | - | ROCK1 | Inhibits RhoA signaling through competing in binding to ROCK1 | [93,94] |
| RhoBTB3 | Golgi complex; Vesicular structures | - | - | Cyclin E | Promotes retrograde trafficking from the late endosomes to the Golgi; Required for S-phase progression | [91,92,137] |
| TC10 | Golgi complex; Exocytic vesicles | ARHGEF10 (?) | p190RhoGAP | PIST Exo70 | Cargo loading at the TGN; Promotes exocytic vesicle fusion with the PM | [81,98,100,101,102,103,143] |
| Rac1 | Endosomes | TIAM1 Vav2 | ? | ? | Important for Rac1 activation and delivery to the leading edge; Formation of actin-based migratory protrusions; Promotion of cell migration and invasion | [124,127] |
| RhoJ/TCL | Early endosomes | ? | ? | ? | TfR recycling | [134] |
| RhoG | Lysosomes | ? | ? | ? | Lysosomal dynamics | [135] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olayioye, M.A.; Noll, B.; Hausser, A. Spatiotemporal Control of Intracellular Membrane Trafficking by Rho GTPases. Cells 2019, 8, 1478. https://doi.org/10.3390/cells8121478
Olayioye MA, Noll B, Hausser A. Spatiotemporal Control of Intracellular Membrane Trafficking by Rho GTPases. Cells. 2019; 8(12):1478. https://doi.org/10.3390/cells8121478
Chicago/Turabian StyleOlayioye, Monilola A., Bettina Noll, and Angelika Hausser. 2019. "Spatiotemporal Control of Intracellular Membrane Trafficking by Rho GTPases" Cells 8, no. 12: 1478. https://doi.org/10.3390/cells8121478
APA StyleOlayioye, M. A., Noll, B., & Hausser, A. (2019). Spatiotemporal Control of Intracellular Membrane Trafficking by Rho GTPases. Cells, 8(12), 1478. https://doi.org/10.3390/cells8121478





