Perinatal Mesenchymal Stromal Cells and Their Possible Contribution to Fetal-Maternal Tolerance
Abstract
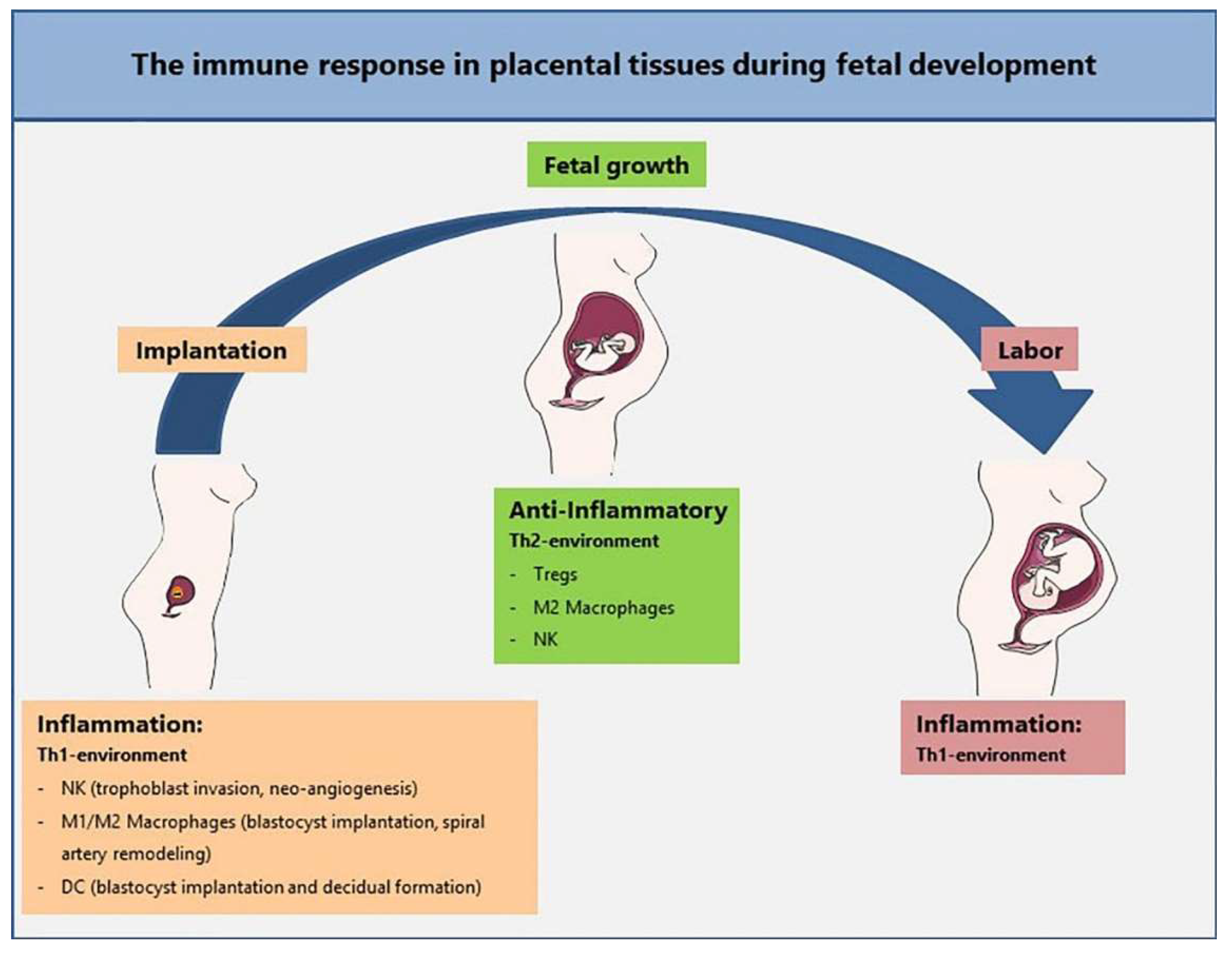
1. Immunology of Pregnancy
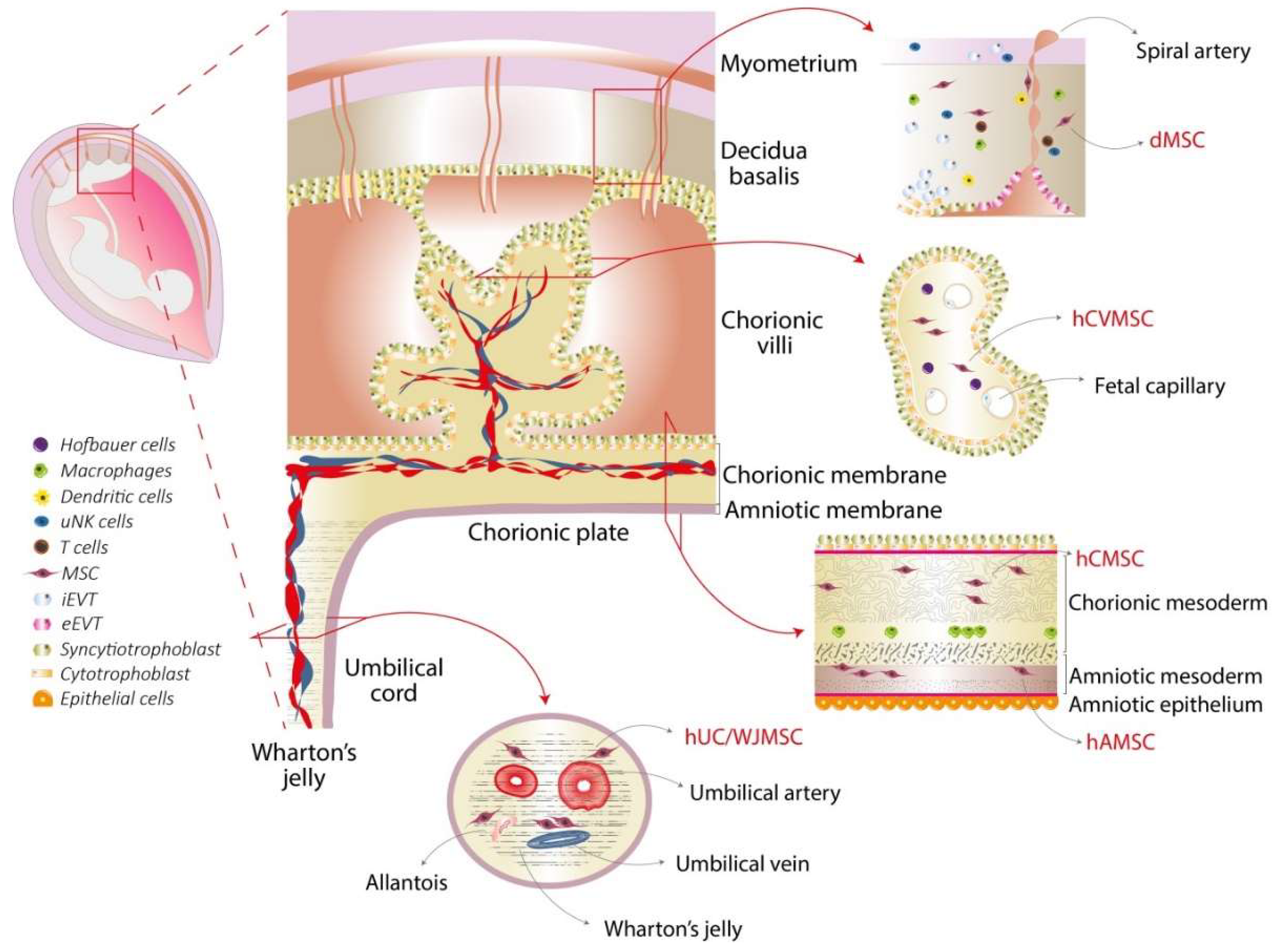
2. Placental Structure and Perinatal MSC
3. Role of MSC in Fetal-Maternal Tolerance
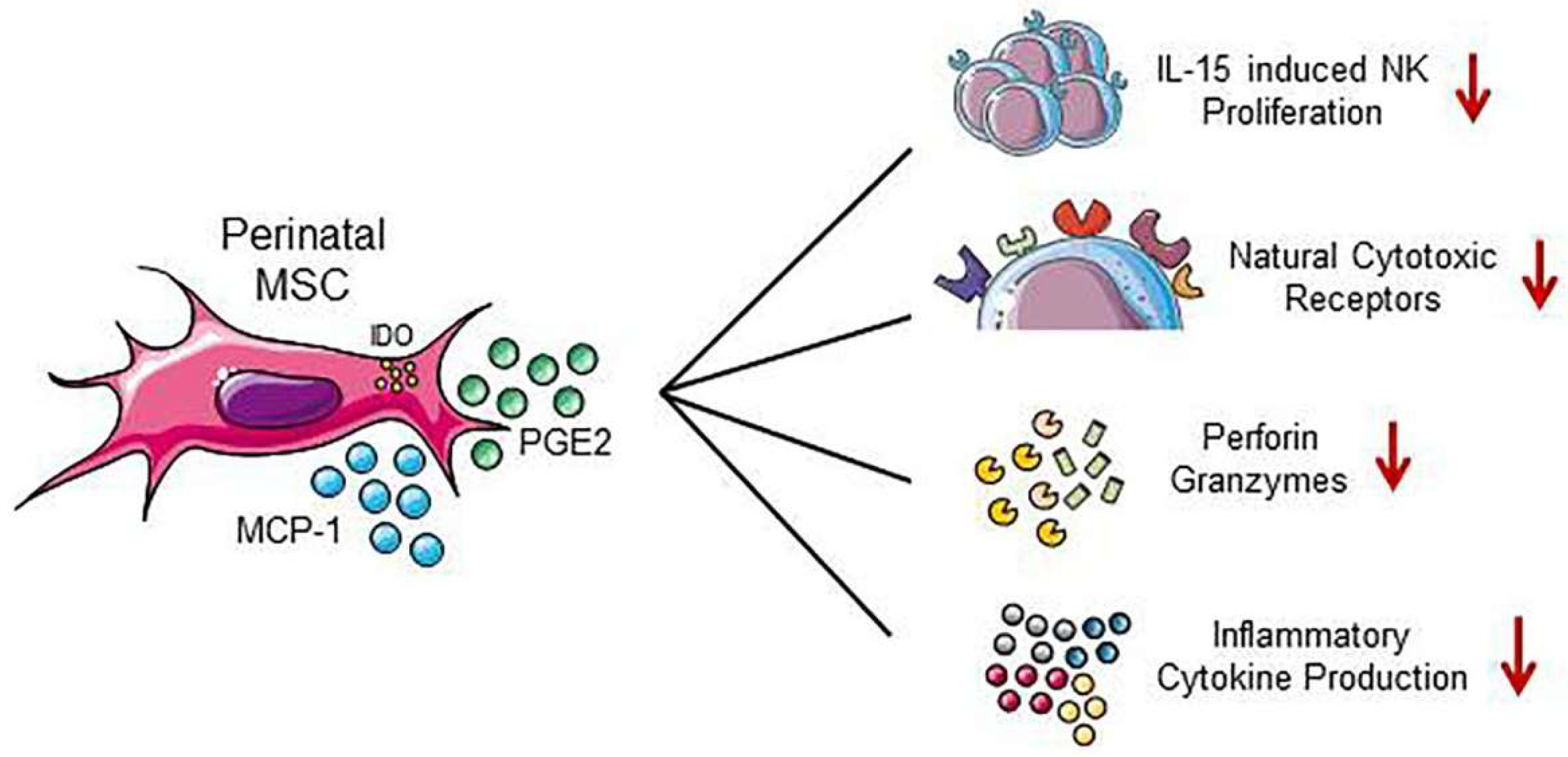
3.1. Effect of Perinatal MSC on NK Cells
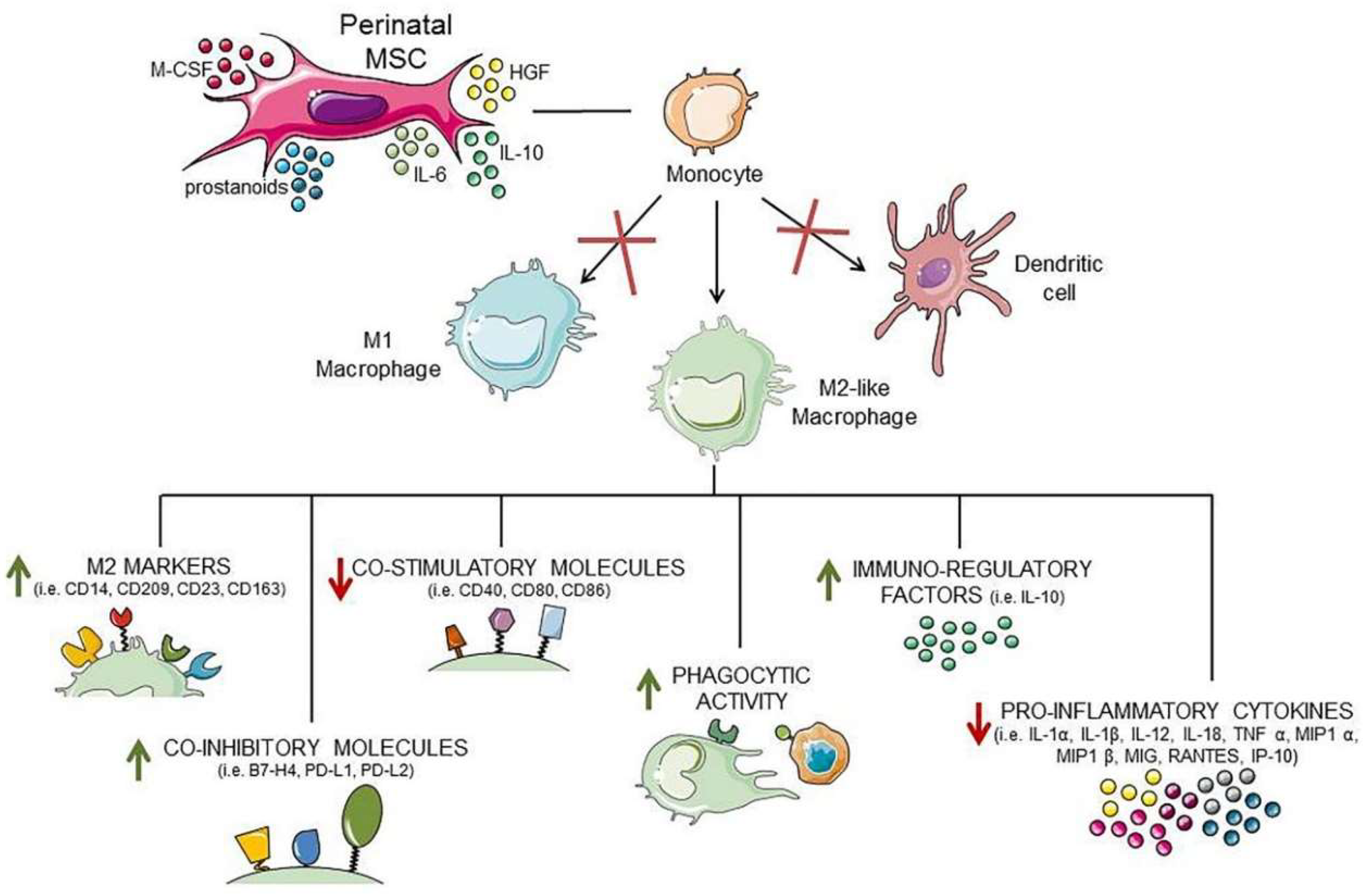
3.2. Effect of Perinatal MSC on Antigen-Presenting Cells (APC): Macrophages
3.3. Effect of Perinatal MSC on APC: Dendritic Cells
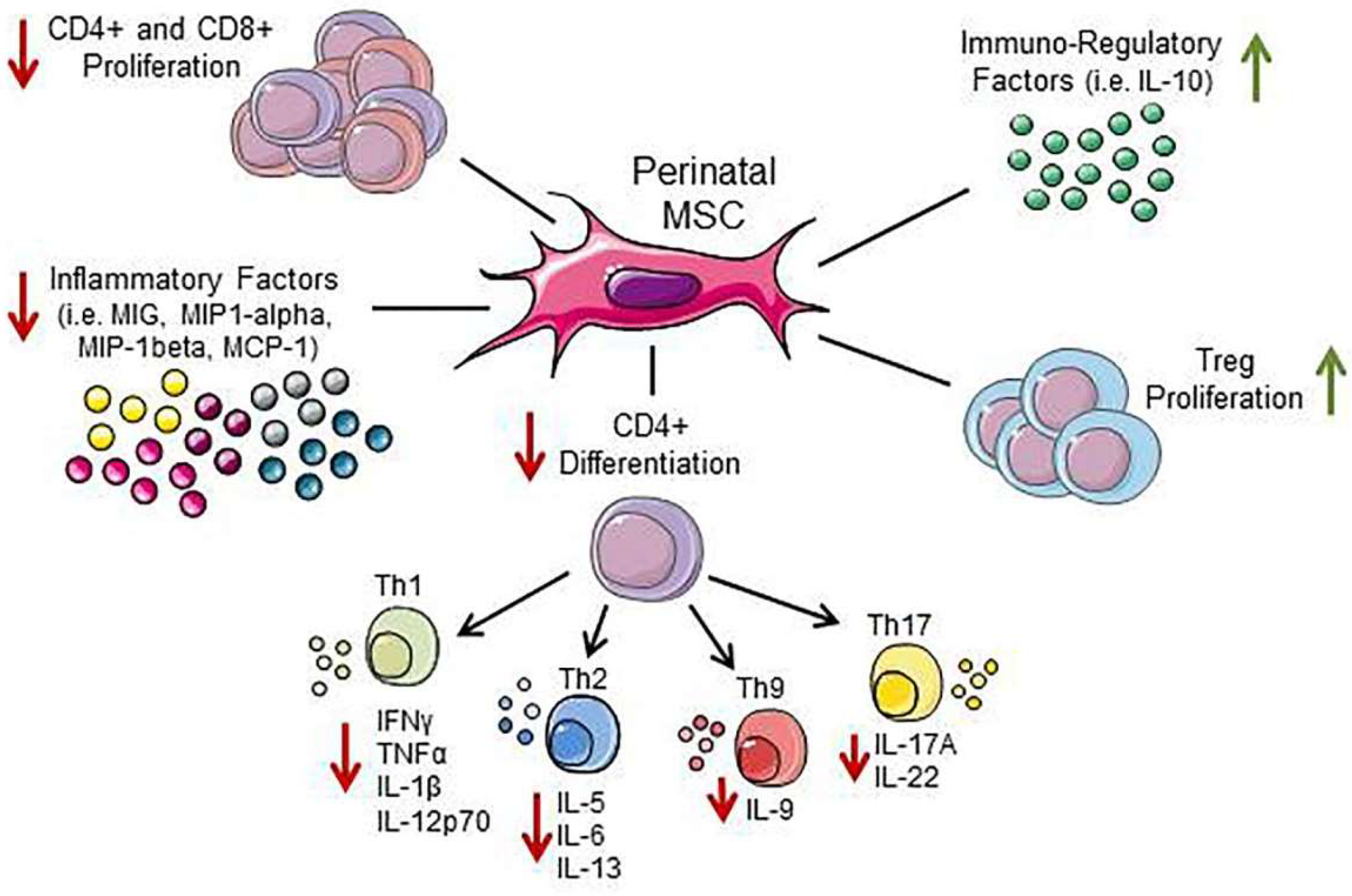
3.4. Effect of Perinatal MSC on T Cells
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Medawar, P.B. Some immunological and endocrinological problems raised by the evolution of viviparity in vertebrates. Symp. Soc. Exp. Biol. 1953, 7, 320–338. [Google Scholar]
- Billingham, R.E.; Brent, L.; Medawar, P.B. Actively acquired tolerance of foreign cells. Nature 1953, 172, 603–606. [Google Scholar] [CrossRef] [PubMed]
- Hoskin, D.W.; Murgita, R.A. Specific maternal anti-fetal lymphoproliferative responses and their regulation by natural immunosuppressive factors. Clin. Exp. Immunol. 1989, 76, 262–267. [Google Scholar] [PubMed]
- Sacks, G.; Sargent, I.; Redman, C. An innate view of human pregnancy. Immunol. Today 1999, 20, 114–118. [Google Scholar] [CrossRef]
- PrabhuDas, M.; Bonney, E.; Caron, K.; Dey, S.; Erlebacher, A.; Fazleabas, A.; Fisher, S.; Golos, T.; Matzuk, M.; McCune, J.M.; et al. Immune mechanisms at the maternal-fetal interface: Perspectives and challenges. Nat. Immunol. 2015, 16, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, D.W.; Zickwolf, G.K.; Weil, G.J.; Sylvester, S.; DeMaria, M.A. Male fetal progenitor cells persist in maternal blood for as long as 27 years postpartum. Proc. Natl. Acad. Sci. USA 1996, 93, 705–708. [Google Scholar] [CrossRef] [PubMed]
- Mor, G.; Aldo, P.; Alvero, A.B. The unique immunological and microbial aspects of pregnancy. Nat. Rev. 2017, 17, 469–482. [Google Scholar] [CrossRef]
- Mor, G.; Cardenas, I.; Abrahams, V.; Guller, S. Inflammation and pregnancy: The role of the immune system at the implantation site. Ann. N.Y. Acad. Sci. 2011, 1221, 80–87. [Google Scholar] [CrossRef]
- Bulmer, J.N.; Pace, D.; Ritson, A. Immunoregulatory cells in human decidua: Morphology, immunohistochemistry and function. Reprod. Nutr. Dev. 1988, 28, 1599–1613. [Google Scholar] [CrossRef]
- Ozenci, C.C.; Korgun, E.T.; Demir, R. Immunohistochemical detection of CD45+, CD56+, and CD14+ cells in human decidua during early pregnancy. Early Pregnancy (Online) 2001, 5, 164–175. [Google Scholar]
- Muzzio, D.O.; Soldati, R.; Ehrhardt, J.; Utpatel, K.; Evert, M.; Zenclussen, A.C.; Zygmunt, M.; Jensen, F. B cell development undergoes profound modifications and adaptations during pregnancy in mice. Biol. Reprod. 2014, 91, 115. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Nakashima, A.; Shima, T.; Ito, M. Th1/Th2/Th17 and regulatory T-cell paradigm in pregnancy. Am. J. Reprod. Immunol. 2010, 63, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Mjosberg, J.; Berg, G.; Jenmalm, M.C.; Ernerudh, J. FOXP3+ regulatory T cells and T helper 1, T helper 2, and T helper 17 cells in human early pregnancy decidua. Biol. Reprod. 2010, 82, 698–705. [Google Scholar] [CrossRef] [PubMed]
- La Rocca, C.; Carbone, F.; Longobardi, S.; Matarese, G. The immunology of pregnancy: Regulatory T cells control maternal immune tolerance toward the fetus. Immunol. Lett. 2014, 162, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Vacca, P.; Montaldo, E.; Croxatto, D.; Loiacono, F.; Canegallo, F.; Venturini, P.L.; Moretta, L.; Mingari, M.C. Identification of diverse innate lymphoid cells in human decidua. Mucosal. Immunol. 2015, 8, 254–264. [Google Scholar] [CrossRef]
- Benirschke, K.; Kaufmann, P. Pathology of the Human Placenta, 4th ed.; Springer-Verlag: New York, NY, USA, 2000; p. 947. [Google Scholar]
- Cunningham, F.G.; Mac Donald, P.C.; Gant, N.F.; Leveno, K.J.; Gilstrap, L.C.; Hankins, G.D.V.; Clark, S.L. Williams Obstetrics; Appleton & Lange: Stamford, CT, USA, 1997; pp. 107–123. [Google Scholar]
- Parolini, O.; Alviano, F.; Bagnara, G.P.; Bilic, G.; Buhring, H.J.; Evangelista, M.; Hennerbichler, S.; Liu, B.; Magatti, M.; Mao, N.; et al. Concise review: Isolation and characterization of cells from human term placenta: Outcome of the first international Workshop on Placenta Derived Stem Cells. Stem Cells 2008, 26, 300–311. [Google Scholar] [CrossRef]
- Ventura Ferreira, M.S.; Bienert, M.; Muller, K.; Rath, B.; Goecke, T.; Oplander, C.; Braunschweig, T.; Mela, P.; Brummendorf, T.H.; Beier, F.; et al. Comprehensive characterization of chorionic villi-derived mesenchymal stromal cells from human placenta. Stem Cell Res. Ther. 2018, 9, 28. [Google Scholar] [CrossRef]
- Anzalone, R.; Lo Iacono, M.; Corrao, S.; Magno, F.; Loria, T.; Cappello, F.; Zummo, G.; Farina, F.; La Rocca, G. New emerging potentials for human Wharton’s jelly mesenchymal stem cells: Immunological features and hepatocyte-like differentiative capacity. Stem Cells Dev. 2010, 19, 423–438. [Google Scholar] [CrossRef] [PubMed]
- Bongso, A.; Fong, C.Y. The therapeutic potential, challenges and future clinical directions of stem cells from the Wharton’s jelly of the human umbilical cord. Stem Cell Rev. Rep. 2013, 9, 226–240. [Google Scholar] [CrossRef]
- Abomaray, F.M.; Al Jumah, M.A.; Alsaad, K.O.; Jawdat, D.; Al Khaldi, A.; AlAskar, A.S.; Al Harthy, S.; Al Subayyil, A.M.; Khatlani, T.; Alawad, A.O.; et al. Phenotypic and Functional Characterization of Mesenchymal Stem/Multipotent Stromal Cells from Decidua Basalis of Human Term Placenta. Stem Cells Int. 2016, 2016, 18. [Google Scholar] [CrossRef]
- Abumaree, M.H.; Abomaray, F.M.; Alshehri, N.A.; Almutairi, A.; AlAskar, A.S.; Kalionis, B.; Al Jumah, M.A. Phenotypic and Functional Characterization of Mesenchymal Stem/Multipotent Stromal Cells From Decidua Parietalis of Human Term Placenta. Reprod. Sci. 2016, 23, 1193–1207. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Araujo, A.B.; Salton, G.D.; Furlan, J.M.; Schneider, N.; Angeli, M.H.; Laureano, A.M.; Silla, L.; Passos, E.P.; Paz, A.H. Comparison of human mesenchymal stromal cells from four neonatal tissues: Amniotic membrane, chorionic membrane, placental decidua and umbilical cord. Cytotherapy 2017, 19, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Wegmeyer, H.; Broske, A.M.; Leddin, M.; Kuentzer, K.; Nisslbeck, A.K.; Hupfeld, J.; Wiechmann, K.; Kuhlen, J.; von Schwerin, C.; Stein, C.; et al. Mesenchymal stromal cell characteristics vary depending on their origin. Stem Cells Dev. 2013, 22, 2606–2618. [Google Scholar] [CrossRef]
- Kmiecik, G.; Spoldi, V.; Silini, A.; Parolini, O. Current View on Osteogenic Differentiation Potential of Mesenchymal Stromal Cells Derived from Placental Tissues. Stem Cell Rev. Rep. 2015, 11, 570–585. [Google Scholar] [CrossRef]
- Magatti, M.; Abumaree, M.H.; Silini, A.R.; Anzalone, R.; Saieva, S.; Russo, E.; Trapani, M.E.; La Rocca, G.; Parolini, O. The Immunomodulatory Features of Mesenchymal Stromal Cells Derived from Wharton’s Jelly, Amniotic Membrane and Chorionic Villi: In Vitro and In Vivo Data. In Placenta: The Tree of Life; Parolini, O., Ed.; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2016; pp. 91–128. [Google Scholar]
- Magatti, M.; Vertua, E.; Cargnoni, A.; Silini, A.; Parolini, O. The Immunomodulatory Properties of Amniotic Cells: The Two Sides of the Coin. Cell Transpl. 2018, 27, 31–44. [Google Scholar] [CrossRef]
- Silini, A.R.; Magatti, M.; Cargnoni, A.; Parolini, O. Is Immune Modulation the Mechanism Underlying the Beneficial Effects of Amniotic Cells and Their Derivatives in Regenerative Medicine? Cell Transpl. 2017, 26, 531–539. [Google Scholar] [CrossRef]
- Roelen, D.L.; van der Mast, B.J.; in’t Anker, P.S.; Kleijburg, C.; Eikmans, M.; van Beelen, E.; de Groot-Swings, G.M.; Fibbe, W.E.; Kanhai, H.H.; Scherjon, S.A.; et al. Differential immunomodulatory effects of fetal versus maternal multipotent stromal cells. Hum. Immunol. 2009, 70, 16–23. [Google Scholar] [CrossRef]
- Vacca, P.; Vitale, C.; Montaldo, E.; Conte, R.; Cantoni, C.; Fulcheri, E.; Darretta, V.; Moretta, L.; Mingari, M.C. CD34+ hematopoietic precursors are present in human decidua and differentiate into natural killer cells upon interaction with stromal cells. Proc. Natl. Acad. Sci. USA 2011, 108, 2402–2407. [Google Scholar] [CrossRef]
- Croxatto, D.; Vacca, P.; Canegallo, F.; Conte, R.; Venturini, P.L.; Moretta, L.; Mingari, M.C. Stromal cells from human decidua exert a strong inhibitory effect on NK cell function and dendritic cell differentiation. PLoS ONE 2014, 9, e89006. [Google Scholar] [CrossRef]
- Chen, Y.; Zhuang, Y.; Chen, X.; Huang, L. Effect of human endometrial stromal cell-derived conditioned medium on uterine natural killer (uNK) cells’ proliferation and cytotoxicity. Am. J. Reprod. Immunol. 2011, 65, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Wang, Q.; Deng, B.; Wang, H.; Dong, Z.; Qu, X.; Kong, B. Monocyte chemoattractant protein-1 secreted by decidual stromal cells inhibits NK cells cytotoxicity by up-regulating expression of SOCS3. PLoS ONE 2012, 7, e41869. [Google Scholar] [CrossRef] [PubMed]
- Manochantr, S.; U-pratya, Y.; Kheolamai, P.; Rojphisan, S.; Chayosumrit, M.; Tantrawatpan, C.; Supokawej, A.; Issaragrisil, S. Immunosuppressive properties of mesenchymal stromal cells derived from amnion, placenta, Wharton’s jelly and umbilical cord. Intern. Med. J. 2013, 43, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Lindau, R.; Mehta, R.B.; Lash, G.E.; Papapavlou, G.; Boij, R.; Berg, G.; Jenmalm, M.C.; Ernerudh, J.; Svensson-Arvelund, J. Interleukin-34 is present at the fetal-maternal interface and induces immunoregulatory macrophages of a decidual phenotype in vitro. Hum. Reprod. 2018, 33, 588–599. [Google Scholar] [CrossRef] [PubMed]
- Tipnis, S.; Viswanathan, C.; Majumdar, A.S. Immunosuppressive properties of human umbilical cord-derived mesenchymal stem cells: Role of B7-H1 and IDO. Immunol. Cell Biol. 2010, 88, 795–806. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, D.; Marquardt, N.; Tufa, D.M.; Beauclair, G.; Low, H.Z.; Hatlapatka, T.; Hass, R.; Kasper, C.; von Kaisenberg, C.; Schmidt, R.E.; et al. Role of gamma-secretase in human umbilical-cord derived mesenchymal stem cell mediated suppression of NK cell cytotoxicity. Cell Commun. Signal. CCS 2014, 12, 63. [Google Scholar] [CrossRef]
- Chatterjee, D.; Marquardt, N.; Tufa, D.M.; Hatlapatka, T.; Hass, R.; Kasper, C.; von Kaisenberg, C.; Schmidt, R.E.; Jacobs, R. Human Umbilical Cord-Derived Mesenchymal Stem Cells Utilize Activin-A to Suppress Interferon-gamma Production by Natural Killer Cells. Front. Immunol. 2014, 5, 662. [Google Scholar] [CrossRef]
- Noone, C.; Kihm, A.; English, K.; O’Dea, S.; Mahon, B.P. IFN-gamma stimulated human umbilical-tissue-derived cells potently suppress NK activation and resist NK-mediated cytotoxicity in vitro. Stem Cells Dev. 2013, 22, 3003–3014. [Google Scholar] [CrossRef]
- Ribeiro, A.; Laranjeira, P.; Mendes, S.; Velada, I.; Leite, C.; Andrade, P.; Santos, F.; Henriques, A.; Graos, M.; Cardoso, C.M.; et al. Mesenchymal stem cells from umbilical cord matrix, adipose tissue and bone marrow exhibit different capability to suppress peripheral blood B, natural killer and T cells. Stem cell Res. Ther. 2013, 4, 125. [Google Scholar] [CrossRef]
- Li, W.; Zhang, Q.; Wang, M.; Wu, H.; Mao, F.; Zhang, B.; Ji, R.; Gao, S.; Sun, Z.; Zhu, W.; et al. Macrophages are involved in the protective role of human umbilical cord-derived stromal cells in renal ischemia-reperfusion injury. Stem Cell Res. 2013, 10, 405–416. [Google Scholar] [CrossRef]
- Donders, R.; Vanheusden, M.; Bogie, J.F.; Ravanidis, S.; Thewissen, K.; Stinissen, P.; Gyselaers, W.; Hendriks, J.J.; Hellings, N. Human Wharton’s jelly-derived stem cells display immunomodulatory properties and transiently improve rat experimental autoimmune encephalomyelitis. Cell Transpl. 2015, 24, 2077–2098. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, H.; Erkers, T.; Nava, S.; Ruhm, S.; Westgren, M.; Ringden, O. Stromal cells from term fetal membrane are highly suppressive in allogeneic settings in vitro. Clin. Exp. Immunol. 2012, 167, 543–555. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Qiu, X.; Ni, P.; Qiu, X.; Lin, X.; Wu, W.; Xie, L.; Lin, L.; Min, J.; Lai, X.; et al. Immunological characteristics of human umbilical cord mesenchymal stem cells and the therapeutic effects of their transplantion on hyperglycemia in diabetic rats. Int. J. Mol. Med. 2014, 33, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Saeidi, M.; Masoud, A.; Shakiba, Y.; Hadjati, J.; Mohyeddin Bonab, M.; Nicknam, M.H.; Latifpour, M.; Nikbin, B. Immunomodulatory effects of human umbilical cord Wharton’s jelly-derived mesenchymal stem cells on differentiation, maturation and endocytosis of monocyte-derived dendritic cells. Iran. J. Allergy Asthma Immunol. 2013, 12, 37–49. [Google Scholar]
- Tsai, P.J.; Wang, H.S.; Lin, G.J.; Chou, S.C.; Chu, T.H.; Chuan, W.T.; Lu, Y.J.; Weng, Z.C.; Su, C.H.; Hsieh, P.S.; et al. Undifferentiated Wharton’s Jelly Mesenchymal Stem Cell Transplantation Induces Insulin-Producing Cell Differentiation and Suppression of T-Cell-Mediated Autoimmunity in Nonobese Diabetic Mice. Cell Transpl. 2015, 24, 1555–1570. [Google Scholar] [CrossRef]
- Pianta, S.; Magatti, M.; Vertua, E.; Bonassi Signoroni, P.; Muradore, I.; Nuzzo, A.M.; Rolfo, A.; Silini, A.; Quaglia, F.; Todros, T.; et al. Amniotic mesenchymal cells from pre-eclamptic placentae maintain immunomodulatory features as healthy controls. J. Cell. Mol. Med. 2016, 20, 157–169. [Google Scholar] [CrossRef]
- Magatti, M.; Caruso, M.; De Munari, S.; Vertua, E.; De, D.; Manuelpillai, U.; Parolini, O. Human Amniotic Membrane-Derived Mesenchymal and Epithelial Cells Exert Different Effects on Monocyte-Derived Dendritic Cell Differentiation and Function. Cell Transpl. 2015, 24, 1733–1752. [Google Scholar] [CrossRef]
- Magatti, M.; Vertua, E.; De Munari, S.; Caro, M.; Caruso, M.; Silini, A.; Delgado, M.; Parolini, O. Human amnion favours tissue repair by inducing the M1-to-M2 switch and enhancing M2 macrophage features. J. Tissue Eng. Regen. Med. 2017, 11, 2895–2911. [Google Scholar] [CrossRef]
- Kang, J.W.; Koo, H.C.; Hwang, S.Y.; Kang, S.K.; Ra, J.C.; Lee, M.H.; Park, Y.H. Immunomodulatory effects of human amniotic membrane-derived mesenchymal stem cells. J. Vet. Sci. 2012, 13, 23–31. [Google Scholar] [CrossRef]
- Yamahara, K.; Harada, K.; Ohshima, M.; Ishikane, S.; Ohnishi, S.; Tsuda, H.; Otani, K.; Taguchi, A.; Soma, T.; Ogawa, H.; et al. Comparison of angiogenic, cytoprotective, and immunosuppressive properties of human amnion- and chorion-derived mesenchymal stem cells. PLoS ONE 2014, 9, e88319. [Google Scholar] [CrossRef]
- Wolbank, S.; Stadler, G.; Peterbauer, A.; Gillich, A.; Karbiener, M.; Streubel, B.; Wieser, M.; Katinger, H.; van Griensven, M.; Redl, H.; et al. Telomerase immortalized human amnion- and adipose-derived mesenchymal stem cells: Maintenance of differentiation and immunomodulatory characteristics. Tissue Eng. 2009, 15, 1843–1854. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, M.; Hosseini, E.; Pourfathollah, A.A.; Ghasemzadeh, M.; Karimi, G. The Role of Amnion Membrane-Derived Mesenchymal Stem Cells on Differentiation and Expansion of Natural Killer Cell Progenitors Originated From Umbilical Cord Blood Mononuclear Cells. Biotech. Health Sci. 2015, 2, e33684. [Google Scholar] [CrossRef]
- Li, J.; Koike-Soko, C.; Sugimoto, J.; Yoshida, T.; Okabe, M.; Nikaido, T. Human Amnion-Derived Stem Cells Have Immunosuppressive Properties on NK Cells and Monocytes. Cell Transpl. 2015, 24, 2065–2076. [Google Scholar] [CrossRef] [PubMed]
- Magatti, M.; De Munari, S.; Vertua, E.; Gibelli, L.; Wengler, G.S.; Parolini, O. Human amnion mesenchyme harbors cells with allogeneic T-cell suppression and stimulation capabilities. Stem Cells 2008, 26, 182–192. [Google Scholar] [CrossRef]
- Rossi, D.; Pianta, S.; Magatti, M.; Sedlmayr, P.; Parolini, O. Characterization of the conditioned medium from amniotic membrane cells: Prostaglandins as key effectors of its immunomodulatory activity. PLoS ONE 2012, 7, e46956. [Google Scholar] [CrossRef]
- Wolbank, S.; Peterbauer, A.; Fahrner, M.; Hennerbichler, S.; van Griensven, M.; Stadler, G.; Redl, H.; Gabriel, C. Dose-Dependent Immunomodulatory Effect of Human Stem Cells from Amniotic Membrane: A Comparison with Human Mesenchymal Stem Cells from Adipose Tissue. Tissue Eng. 2007, 13, 1173–1183. [Google Scholar] [CrossRef]
- Kronsteiner, B.; Wolbank, S.; Peterbauer, A.; Hackl, C.; Redl, H.; van Griensven, M.; Gabriel, C. Human mesenchymal stem cells from adipose tissue and amnion influence T-cells depending on stimulation method and presence of other immune cells. Stem Cells Dev. 2011, 20, 2115–2126. [Google Scholar] [CrossRef]
- Parolini, O.; Souza-Moreira, L.; O’Valle, F.; Magatti, M.; Hernandez-Cortes, P.; Gonzalez-Rey, E.; Delgado, M. Therapeutic effect of human amniotic membrane-derived cells on experimental arthritis and other inflammatory disorders. Arthr. Rheumatol. 2014, 66, 327–339. [Google Scholar] [CrossRef]
- Pianta, S.; Bonassi Signoroni, P.; Muradore, I.; Rodrigues, M.F.; Rossi, D.; Silini, A.; Parolini, O. Amniotic membrane mesenchymal cells-derived factors skew T cell polarization toward Treg and downregulate Th1 and Th17 cells subsets. Stem Cell Rev. 2015, 11, 394–407. [Google Scholar] [CrossRef]
- Kronsteiner, B.; Peterbauer-Scherb, A.; Grillari-Voglauer, R.; Redl, H.; Gabriel, C.; van Griensven, M.; Wolbank, S. Human mesenchymal stem cells and renal tubular epithelial cells differentially influence monocyte-derived dendritic cell differentiation and maturation. Cell. Immunol. 2011, 267, 30–38. [Google Scholar] [CrossRef]
- Magatti, M.; De Munari, S.; Vertua, E.; Nassauto, C.; Albertini, A.; Wengler, G.S.; Parolini, O. Amniotic mesenchymal tissue cells inhibit dendritic cell differentiation of peripheral blood and amnion resident monocytes. Cell Transpl. 2009, 18, 899–914. [Google Scholar] [CrossRef] [PubMed]
- Onishi, R.; Ohnishi, S.; Higashi, R.; Watari, M.; Yamahara, K.; Okubo, N.; Nakagawa, K.; Katsurada, T.; Suda, G.; Natsuizaka, M.; et al. Human Amnion-Derived Mesenchymal Stem Cell Transplantation Ameliorates Dextran Sulfate Sodium-Induced Severe Colitis in Rats. Cell Transpl. 2015, 24, 2601–2614. [Google Scholar] [CrossRef] [PubMed]
- Bailo, M.; Soncini, M.; Vertua, E.; Signoroni, P.B.; Sanzone, S.; Lombardi, G.; Arienti, D.; Calamani, F.; Zatti, D.; Paul, P.; et al. Engraftment potential of human amnion and chorion cells derived from term placenta. Transplantation 2004, 78, 1439–1448. [Google Scholar] [CrossRef] [PubMed]
- Abumaree, M.H.; Al Jumah, M.A.; Kalionis, B.; Jawdat, D.; Al Khaldi, A.; Abomaray, F.M.; Fatani, A.S.; Chamley, L.W.; Knawy, B.A. Human placental mesenchymal stem cells (pMSCs) play a role as immune suppressive cells by shifting macrophage differentiation from inflammatory M1 to anti-inflammatory M2 macrophages. Stem Cell Rev. 2013, 9, 620–641. [Google Scholar] [CrossRef] [PubMed]
- Abomaray, F.M.; Al Jumah, M.A.; Kalionis, B.; AlAskar, A.S.; Al Harthy, S.; Jawdat, D.; Al Khaldi, A.; Alkushi, A.; Knawy, B.A.; Abumaree, M.H. Human Chorionic Villous Mesenchymal Stem Cells Modify the Functions of Human Dendritic Cells, and Induce an Anti-Inflammatory Phenotype in CD1+ Dendritic Cells. Stem Cell Rev. 2014. [Google Scholar] [CrossRef] [PubMed]
- Trundley, A.; Moffett, A. Human uterine leukocytes and pregnancy. Tissue Antigens 2004, 63, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kwan, M.; Hazan, A.; Zhang, J.; Jones, R.L.; Harris, L.K.; Whittle, W.; Keating, S.; Dunk, C.E.; Lye, S.J. Dynamic changes in maternal decidual leukocyte populations from first to second trimester gestation. Placenta 2014, 35, 1027–1034. [Google Scholar] [CrossRef]
- Hanna, J.; Goldman-Wohl, D.; Hamani, Y.; Avraham, I.; Greenfield, C.; Natanson-Yaron, S.; Prus, D.; Cohen-Daniel, L.; Arnon, T.I.; Manaster, I.; et al. Decidual NK cells regulate key developmental processes at the human fetal-maternal interface. Nat. Med. 2006, 12, 1065–1074. [Google Scholar] [CrossRef]
- Montaldo, E.; Vacca, P.; Moretta, L.; Mingari, M.C. Development of human natural killer cells and other innate lymphoid cells. Semin. Immunol. 2014, 26, 107–113. [Google Scholar] [CrossRef]
- Jabrane-Ferrat, N. Features of Human Decidual NK Cells in Healthy Pregnancy and During Viral Infection. Front. Immunol. 2019, 10, 1397. [Google Scholar] [CrossRef]
- Croy, B.A.; Ashkar, A.A.; Minhas, K.; Greenwood, J.D. Can murine uterine natural killer cells give insights into the pathogenesis of preeclampsia? J. Soc. Gynecol. Investig. 2000, 7, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Sivori, S.; Carlomagno, S.; Pesce, S.; Moretta, A.; Vitale, M.; Marcenaro, E. TLR/NCR/KIR: Which One to Use and When? Front. Immunol. 2014, 5, 105. [Google Scholar] [CrossRef] [PubMed]
- Long, E.O.; Kim, H.S.; Liu, D.; Peterson, M.E.; Rajagopalan, S. Controlling natural killer cell responses: Integration of signals for activation and inhibition. Annu. Rev. Immunol. 2013, 31, 227–258. [Google Scholar] [CrossRef] [PubMed]
- Vivier, E.; Artis, D.; Colonna, M.; Diefenbach, A.; Di Santo, J.P.; Eberl, G.; Koyasu, S.; Locksley, R.M.; McKenzie, A.N.J.; Mebius, R.E.; et al. Innate Lymphoid Cells: 10 Years On. Cell 2018, 174, 1054–1066. [Google Scholar] [CrossRef] [PubMed]
- Vacca, P.; Cantoni, C.; Prato, C.; Fulcheri, E.; Moretta, A.; Moretta, L.; Mingari, M.C. Regulatory role of NKp44, NKp46, DNAM-1 and NKG2D receptors in the interaction between NK cells and trophoblast cells. Evidence for divergent functional profiles of decidual versus peripheral NK cells. Int. Immunol. 2008, 20, 1395–1405. [Google Scholar] [CrossRef] [PubMed]
- Vento-Tormo, R.; Efremova, M.; Botting, R.A.; Turco, M.Y.; Vento-Tormo, M.; Meyer, K.B.; Park, J.E.; Stephenson, E.; Polanski, K.; Goncalves, A.; et al. Single-cell reconstruction of the early maternal-fetal interface in humans. Nature 2018, 563, 347–353. [Google Scholar] [CrossRef]
- Vacca, P.; Vitale, C.; Munari, E.; Cassatella, M.A.; Mingari, M.C.; Moretta, L. Human Innate Lymphoid Cells: Their Functional and Cellular Interactions in Decidua. Front. Immunol. 2018, 9, 1897. [Google Scholar] [CrossRef]
- Vacca, P.; Chiossone, L.; Mingari, M.C.; Moretta, L. Heterogeneity of NK Cells and Other Innate Lymphoid Cells in Human and Murine Decidua. Front. Immunol. 2019, 10, 170. [Google Scholar] [CrossRef]
- Vacca, P.; Pesce, S.; Greppi, M.; Fulcheri, E.; Munari, E.; Olive, D.; Mingari, M.C.; Moretta, A.; Moretta, L.; Marcenaro, E. PD-1 is expressed by and regulates human group 3 innate lymphoid cells in human decidua. Mucosal Immunol. 2019, 12, 624–631. [Google Scholar] [CrossRef]
- Gamliel, M.; Goldman-Wohl, D.; Isaacson, B.; Gur, C.; Stein, N.; Yamin, R.; Berger, M.; Grunewald, M.; Keshet, E.; Rais, Y.; et al. Trained Memory of Human Uterine NK Cells Enhances Their Function in Subsequent Pregnancies. Immunity 2018, 48, 951–962. [Google Scholar] [CrossRef]
- Keskin, D.B.; Allan, D.S.; Rybalov, B.; Andzelm, M.M.; Stern, J.N.; Kopcow, H.D.; Koopman, L.A.; Strominger, J.L. TGFbeta promotes conversion of CD16+ peripheral blood NK cells into CD16- NK cells with similarities to decidual NK cells. Proc. Natl. Acad. Sci. USA 2007, 104, 3378–3383. [Google Scholar] [CrossRef] [PubMed]
- Erlebacher, A. Immunology of the maternal-fetal interface. Annu. Rev. Immunol. 2013, 31, 387–411. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.J.; Bulmer, J.N.; Searle, R.F.; Innes, B.A.; Robson, S.C. Altered decidual leucocyte populations in the placental bed in pre-eclampsia and foetal growth restriction: A comparison with late normal pregnancy. Reproduction 2009, 138, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Ning, F.; Liu, H.; Lash, G.E. The Role of Decidual Macrophages During Normal and Pathological Pregnancy. Am. J. Reprod. Immunol. 2016, 75, 298–309. [Google Scholar] [CrossRef]
- Houser, B.L.; Tilburgs, T.; Hill, J.; Nicotra, M.L.; Strominger, J.L. Two unique human decidual macrophage populations. J. Immunol. 2011, 186, 2633–2642. [Google Scholar] [CrossRef]
- Tang, Z.; Abrahams, V.M.; Mor, G.; Guller, S. Placental Hofbauer cells and complications of pregnancy. Ann. N.Y. Acad. Sci. 2011, 1221, 103–108. [Google Scholar] [CrossRef]
- Young, O.M.; Tang, Z.; Niven-Fairchild, T.; Tadesse, S.; Krikun, G.; Norwitz, E.R.; Mor, G.; Abrahams, V.M.; Guller, S. Toll-like receptor-mediated responses by placental Hofbauer cells (HBCs): A potential pro-inflammatory role for fetal M2 macrophages. Am. J. Reprod. Immunol. 2015, 73, 22–35. [Google Scholar] [CrossRef]
- Nagamatsu, T.; Schust, D.J. The contribution of macrophages to normal and pathological pregnancies. Am. J. Reprod. Immunol. 2010, 63, 460–471. [Google Scholar] [CrossRef]
- Renaud, S.J.; Graham, C.H. The role of macrophages in utero-placental interactions during normal and pathological pregnancy. Immunol. Investig. 2008, 37, 535–564. [Google Scholar] [CrossRef]
- Mills, C.D. M1 and M2 Macrophages: Oracles of Health and Disease. Crit. Rev. Immunol. 2012, 32, 463–488. [Google Scholar] [CrossRef]
- Mantovani, A.; Sica, A.; Sozzani, S.; Allavena, P.; Vecchi, A.; Locati, M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 2004, 25, 677–686. [Google Scholar] [CrossRef] [PubMed]
- Ferrante, C.J.; Leibovich, S.J. Regulation of Macrophage Polarization and Wound Healing. Adv. Wound Care 2012, 1, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Bulmer, J.N.; Williams, P.J.; Lash, G.E. Immune cells in the placental bed. Int. J. Dev. Biol. 2010, 54, 281–294. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, M.K.; Mallers, T.M.; Larsen, B.; Kwak-Kim, J.; Chaouat, G.; Gilman-Sachs, A.; Beaman, K.D. V-ATPase upregulation during early pregnancy: A possible link to establishment of an inflammatory response during preimplantation period of pregnancy. Reproduction 2012, 143, 713–725. [Google Scholar] [CrossRef] [PubMed]
- Aldo, P.B.; Racicot, K.; Craviero, V.; Guller, S.; Romero, R.; Mor, G. Trophoblast induces monocyte differentiation into CD14+/CD16+ macrophages. Am. J. Reprod. Immunol. 2014, 72, 270–284. [Google Scholar] [CrossRef]
- Cupurdija, K.; Azzola, D.; Hainz, U.; Gratchev, A.; Heitger, A.; Takikawa, O.; Goerdt, S.; Wintersteiger, R.; Dohr, G.; Sedlmayr, P. Macrophages of human first trimester decidua express markers associated to alternative activation. Am. J. Reprod. Immunol. 2004, 51, 117–122. [Google Scholar] [CrossRef]
- Gustafsson, C.; Mjosberg, J.; Matussek, A.; Geffers, R.; Matthiesen, L.; Berg, G.; Sharma, S.; Buer, J.; Ernerudh, J. Gene expression profiling of human decidual macrophages: Evidence for immunosuppressive phenotype. PLoS ONE 2008, 3, e2078. [Google Scholar] [CrossRef]
- Lidstrom, C.; Matthiesen, L.; Berg, G.; Sharma, S.; Ernerudh, J.; Ekerfelt, C. Cytokine secretion patterns of NK cells and macrophages in early human pregnancy decidua and blood: Implications for suppressor macrophages in decidua. Am. J. Reprod. Immunol. 2003, 50, 444–452. [Google Scholar] [CrossRef]
- Svensson, J.; Jenmalm, M.C.; Matussek, A.; Geffers, R.; Berg, G.; Ernerudh, J. Macrophages at the fetal-maternal interface express markers of alternative activation and are induced by M-CSF and IL-10. J. Immunol. 2011, 187, 3671–3682. [Google Scholar] [CrossRef]
- Seval, Y.; Korgun, E.T.; Demir, R. Hofbauer cells in early human placenta: Possible implications in vasculogenesis and angiogenesis. Placenta 2007, 28, 841–845. [Google Scholar] [CrossRef]
- Loegl, J.; Hiden, U.; Nussbaumer, E.; Schliefsteiner, C.; Cvitic, S.; Lang, I.; Wadsack, C.; Huppertz, B.; Desoye, G. Hofbauer cells of M2a, M2b and M2c polarization may regulate feto-placental angiogenesis. Reproduction 2016, 152, 447–455. [Google Scholar] [CrossRef]
- Bockle, B.C.; Solder, E.; Kind, S.; Romani, N.; Sepp, N.T. DC-sign+ CD163+ macrophages expressing hyaluronan receptor LYVE-1 are located within chorion villi of the placenta. Placenta 2008, 29, 187–192. [Google Scholar] [CrossRef]
- Wetzka, B.; Clark, D.E.; Charnock-Jones, D.S.; Zahradnik, H.P.; Smith, S.K. Isolation of macrophages (Hofbauer cells) from human term placenta and their prostaglandin E2 and thromboxane production. Hum. Reprod. 1997, 12, 847–852. [Google Scholar] [CrossRef]
- Kim, S.S.; Romero, R.; Kim, J.S.; Abbas, A.; Espinoza, J.; Kusanovic, J.P.; Hassan, S.; Yoon, B.H.; Kim, C.J. Coexpression of myofibroblast and macrophage markers: Novel evidence for an in vivo plasticity of chorioamniotic mesodermal cells of the human placenta. Lab. Investig. 2008, 88, 365–374. [Google Scholar] [CrossRef]
- Sutton, L.; Gadd, M.; Mason, D.Y.; Redman, C.W. Cells bearing class II MHC antigens in the human placenta and amniochorion. Immunology 1986, 58, 23–29. [Google Scholar]
- Madsen-Bouterse, S.A.; Romero, R.; Tarca, A.L.; Kusanovic, J.P.; Espinoza, J.; Kim, C.J.; Kim, J.S.; Edwin, S.S.; Gomez, R.; Draghici, S. The transcriptome of the fetal inflammatory response syndrome. Am. J. Reprod. Immunol. 2010, 63, 73–92. [Google Scholar] [CrossRef]
- Galimi, F.; Cottone, E.; Vigna, E.; Arena, N.; Boccaccio, C.; Giordano, S.; Naldini, L.; Comoglio, P.M. Hepatocyte growth factor is a regulator of monocyte-macrophage function. J. Immunol. 2001, 166, 1241–1247. [Google Scholar] [CrossRef]
- Chen, P.M.; Liu, K.J.; Hsu, P.J.; Wei, C.F.; Bai, C.H.; Ho, L.J.; Sytwu, H.K.; Yen, B.L. Induction of immunomodulatory monocytes by human mesenchymal stem cell-derived hepatocyte growth factor through ERK1/2. J. Leukoc. Biol. 2014, 96, 295–303. [Google Scholar] [CrossRef]
- Buechler, C.; Ritter, M.; Orso, E.; Langmann, T.; Klucken, J.; Schmitz, G. Regulation of scavenger receptor CD163 expression in human monocytes and macrophages by pro- and antiinflammatory stimuli. J. Leukoc. Biol. 2000, 67, 97–103. [Google Scholar] [CrossRef]
- Heusinkveld, M.; de Vos van Steenwijk, P.J.; Goedemans, R.; Ramwadhdoebe, T.H.; Gorter, A.; Welters, M.J.; van Hall, T.; van der Burg, S.H. M2 macrophages induced by prostaglandin E2 and IL-6 from cervical carcinoma are switched to activated M1 macrophages by CD4+ Th1 cells. J. Immunol. 2011, 187, 1157–1165. [Google Scholar] [CrossRef]
- Kalinski, P. Regulation of immune responses by prostaglandin E2. J. Immunol. 2012, 188, 21–28. [Google Scholar] [CrossRef]
- Nemeth, K.; Leelahavanichkul, A.; Yuen, P.S.; Mayer, B.; Parmelee, A.; Doi, K.; Robey, P.G.; Leelahavanichkul, K.; Koller, B.H.; Brown, J.M.; et al. Bone marrow stromal cells attenuate sepsis via prostaglandin E(2)-dependent reprogramming of host macrophages to increase their interleukin-10 production. Nat. Med. 2009, 15, 42–49. [Google Scholar] [CrossRef]
- Maggini, J.; Mirkin, G.; Bognanni, I.; Holmberg, J.; Piazzon, I.M.; Nepomnaschy, I.; Costa, H.; Canones, C.; Raiden, S.; Vermeulen, M.; et al. Mouse bone marrow-derived mesenchymal stromal cells turn activated macrophages into a regulatory-like profile. PLoS ONE 2010, 5, e9252. [Google Scholar] [CrossRef]
- Anderson, P.; Souza-Moreira, L.; Morell, M.; Caro, M.; O’Valle, F.; Gonzalez-Rey, E.; Delgado, M. Adipose-derived mesenchymal stromal cells induce immunomodulatory macrophages which protect from experimental colitis and sepsis. Gut 2013, 62, 1131–1141. [Google Scholar] [CrossRef]
- He, S.; Gleason, J.; Fik-Rymarkiewicz, E.; DiFiglia, A.; Bharathan, M.; Morschauser, A.; Djuretic, I.; Xu, Y.; Krakovsky, M.; Jankovic, V.; et al. Human Placenta-Derived Mesenchymal Stromal-Like Cells Enhance Angiogenesis via T Cell-Dependent Reprogramming of Macrophage Differentiation. Stem Cells 2017, 35, 1603–1613. [Google Scholar] [CrossRef]
- Gardner, L.; Moffett, A. Dendritic cells in the human decidua. Biol. Reprod. 2003, 69, 1438–1446. [Google Scholar] [CrossRef]
- Steinman, R.M. Decisions about dendritic cells: Past, present, and future. Annu. Rev. Immunol. 2012, 30, 1–22. [Google Scholar] [CrossRef]
- Plaks, V.; Birnberg, T.; Berkutzki, T.; Sela, S.; BenYashar, A.; Kalchenko, V.; Mor, G.; Keshet, E.; Dekel, N.; Neeman, M.; et al. Uterine DCs are crucial for decidua formation during embryo implantation in mice. J. Clin. Investig. 2008, 118, 3954–3965. [Google Scholar] [CrossRef]
- Collins, M.K.; Tay, C.S.; Erlebacher, A. Dendritic cell entrapment within the pregnant uterus inhibits immune surveillance of the maternal/fetal interface in mice. J. Clin. Investig. 2009, 119, 2062–2073. [Google Scholar] [CrossRef]
- Morelli, A.E.; Thomson, A.W. Tolerogenic dendritic cells and the quest for transplant tolerance. Nat. Rev. 2007, 7, 610–621. [Google Scholar] [CrossRef]
- Dunk, C.; Kwan, M.; Hazan, A.; Walker, S.; Wright, J.K.; Harris, L.K.; Jones, R.L.; Keating, S.; Kingdom, J.C.P.; Whittle, W.; et al. Failure of Decidualization and Maternal Immune Tolerance Underlies Uterovascular Resistance in Intra Uterine Growth Restriction. Front. Endocrinol. 2019, 10, 160. [Google Scholar] [CrossRef]
- Kammerer, U.; Eggert, A.O.; Kapp, M.; McLellan, A.D.; Geijtenbeek, T.B.; Dietl, J.; van Kooyk, Y.; Kampgen, E. Unique appearance of proliferating antigen-presenting cells expressing DC-SIGN (CD209) in the decidua of early human pregnancy. Am. J. Pathol. 2003, 162, 887–896. [Google Scholar] [CrossRef]
- Blois, S.M.; Alba Soto, C.D.; Tometten, M.; Klapp, B.F.; Margni, R.A.; Arck, P.C. Lineage, maturity, and phenotype of uterine murine dendritic cells throughout gestation indicate a protective role in maintaining pregnancy. Biol. Reprod. 2004, 70, 1018–1023. [Google Scholar] [CrossRef]
- Liu, W.; Morschauser, A.; Zhang, X.; Lu, X.; Gleason, J.; He, S.; Chen, H.J.; Jankovic, V.; Ye, Q.; Labazzo, K.; et al. Human placenta-derived adherent cells induce tolerogenic immune responses. Clin. Transl. Immunol. 2014, 3, e14. [Google Scholar] [CrossRef]
- Ou, D.; Wang, X.; Metzger, D.L.; Ao, Z.; Pozzilli, P.; James, R.F.; Chen, L.; Warnock, G.L. Suppression of human T-cell responses to beta-cells by activation of B7-H4 pathway. Cell Transpl. 2006, 15, 399–410. [Google Scholar] [CrossRef]
- Ueno, T.; Yeung, M.Y.; McGrath, M.; Yang, S.; Zaman, N.; Snawder, B.; Padera, R.F.; Magee, C.N.; Gorbatov, R.; Hashiguchi, M.; et al. Intact B7-H3 signaling promotes allograft prolongation through preferential suppression of Th1 effector responses. Eur. J. Immunol. 2012, 42, 2343–2353. [Google Scholar] [CrossRef]
- Carter, L.; Fouser, L.A.; Jussif, J.; Fitz, L.; Deng, B.; Wood, C.R.; Collins, M.; Honjo, T.; Freeman, G.J.; Carreno, B.M. PD-1:PD-L inhibitory pathway affects both CD4(+) and CD8(+) T cells and is overcome by IL-2. Eur. J. Immunol. 2002, 32, 634–643. [Google Scholar] [CrossRef]
- Latchman, Y.; Wood, C.R.; Chernova, T.; Chaudhary, D.; Borde, M.; Chernova, I.; Iwai, Y.; Long, A.J.; Brown, J.A.; Nunes, R.; et al. PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat. Immunol. 2001, 2, 261–268. [Google Scholar] [CrossRef]
- Segerer, S.E.; Muller, N.; Brandt, J.; Kapp, M.; Dietl, J.; Reichardt, H.M.; Rieger, L.; Kammerer, U. The glycoprotein-hormones activin A and inhibin A interfere with dendritic cell maturation. Reprod. Biol. Endocrinol. 2008, 6, 17. [Google Scholar] [CrossRef]
- Segerer, S.E.; Rieger, L.; Kapp, M.; Dombrowski, Y.; Muller, N.; Dietl, J.; Kammerer, U. MIC-1 (a multifunctional modulator of dendritic cell phenotype and function) is produced by decidual stromal cells and trophoblasts. Hum. Reprod. 2012, 27, 200–209. [Google Scholar] [CrossRef]
- Deng, Y.; Yi, S.; Wang, G.; Cheng, J.; Zhang, Y.; Chen, W.; Tai, Y.; Chen, S.; Chen, G.; Liu, W.; et al. Umbilical cord-derived mesenchymal stem cells instruct dendritic cells to acquire tolerogenic phenotypes through the IL-6-mediated upregulation of SOCS1. Stem Cells Dev. 2014, 23, 2080–2092. [Google Scholar] [CrossRef]
- Du, M.R.; Guo, P.F.; Piao, H.L.; Wang, S.C.; Sun, C.; Jin, L.P.; Tao, Y.; Li, Y.H.; Zhang, D.; Zhu, R.; et al. Embryonic trophoblasts induce decidual regulatory T cell differentiation and maternal-fetal tolerance through thymic stromal lymphopoietin instructing dendritic cells. J. Immunol. 2014, 192, 1502–1511. [Google Scholar] [CrossRef]
- Vacca, P.; Cantoni, C.; Vitale, M.; Prato, C.; Canegallo, F.; Fenoglio, D.; Ragni, N.; Moretta, L.; Mingari, M.C. Crosstalk between decidual NK and CD14+ myelomonocytic cells results in induction of Tregs and immunosuppression. Proc. Natl. Acad. Sci. USA 2010, 107, 11918–11923. [Google Scholar] [CrossRef]
- Chomarat, P.; Banchereau, J.; Davoust, J.; Palucka, A.K. IL-6 switches the differentiation of monocytes from dendritic cells to macrophages. Nat. Immunol. 2000, 1, 510–514. [Google Scholar] [CrossRef]
- Palucka, K.A.; Taquet, N.; Sanchez-Chapuis, F.; Gluckman, J.C. Dendritic cells as the terminal stage of monocyte differentiation. J. Immunol. 1998, 160, 4587–4595. [Google Scholar]
- Tilburgs, T.; Claas, F.H.; Scherjon, S.A. Elsevier Trophoblast Research Award Lecture: Unique properties of decidual T cells and their role in immune regulation during human pregnancy. Placenta 2010, 31, S82–S86. [Google Scholar] [CrossRef]
- Tilburgs, T.; Strominger, J.L. CD8+ effector T cells at the fetal-maternal interface, balancing fetal tolerance and antiviral immunity. Am. J. Reprod. Immunol. 2013, 69, 395–407. [Google Scholar] [CrossRef]
- Tripathi, S.; Guleria, I. Role of PD1/PDL1 pathway, and TH17 and treg cells in maternal tolerance to the fetus. Biomed. J. 2015, 38, 25–31. [Google Scholar] [CrossRef]
- Inada, K.; Shima, T.; Ito, M.; Ushijima, A.; Saito, S. Helios-positive functional regulatory T cells are decreased in decidua of miscarriage cases with normal fetal chromosomal content. J. Reprod. Immunol. 2015, 107, 10–19. [Google Scholar] [CrossRef]
- Sasaki, Y.; Darmochwal-Kolarz, D.; Suzuki, D.; Sakai, M.; Ito, M.; Shima, T.; Shiozaki, A.; Rolinski, J.; Saito, S. Proportion of peripheral blood and decidual CD4(+) CD25(bright) regulatory T cells in pre-eclampsia. Clin. Exp. Immunol. 2007, 149, 139–145. [Google Scholar] [CrossRef]
- Nancy, P.; Tagliani, E.; Tay, C.S.; Asp, P.; Levy, D.E.; Erlebacher, A. Chemokine gene silencing in decidual stromal cells limits T cell access to the maternal-fetal interface. Science 2012, 336, 1317–1321. [Google Scholar] [CrossRef] [PubMed]
- Anam, K.; Lazdun, Y.; Davis, P.M.; Banas, R.A.; Elster, E.A.; Davis, T.A. Amnion-derived multipotent progenitor cells support allograft tolerance induction. Am. J. Transpl. 2013, 13, 1416–1428. [Google Scholar] [CrossRef] [PubMed]
- Redman, C.W.; Sargent, I.L. Immunology of pre-eclampsia. Am. J. Reprod. Immunol. 2010, 63, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Hiby, S.E.; Walker, J.J.; O’Shaughnessy, K.M.; Redman, C.W.; Carrington, M.; Trowsdale, J.; Moffett, A. Combinations of maternal KIR and fetal HLA-C genes influence the risk of preeclampsia and reproductive success. J. Exp. Med. 2004, 200, 957–965. [Google Scholar] [CrossRef] [PubMed]
- Obata, Y.; Furusawa, Y.; Hase, K. Epigenetic modifications of the immune system in health and disease. Immunol. Cell Biol. 2015, 93, 226–232. [Google Scholar] [CrossRef]
- Bidarimath, M.; Khalaj, K.; Wessels, J.M.; Tayade, C. MicroRNAs, immune cells and pregnancy. Cell. Mol. Immunol. 2014, 11, 538–547. [Google Scholar] [CrossRef] [PubMed]
- Parham, P.; Moffett, A. Variable NK cell receptors and their MHC class I ligands in immunity, reproduction and human evolution. Nat. Rev. 2013, 13, 133–144. [Google Scholar] [CrossRef]
- Slaats, G.G.; Reinius, L.E.; Alm, J.; Kere, J.; Scheynius, A.; Joerink, M. DNA methylation levels within the CD14 promoter region are lower in placentas of mothers living on a farm. Allergy 2012, 67, 895–903. [Google Scholar] [CrossRef]
- Acevedo, N.; Frumento, P.; Harb, H.; Alashkar Alhamwe, B.; Johansson, C.; Eick, L.; Alm, J.; Renz, H.; Scheynius, A.; Potaczek, D.P. Histone Acetylation of Immune Regulatory Genes in Human Placenta in Association with Maternal Intake of Olive Oil and Fish Consumption. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef]
- Harb, H.; Irvine, J.; Amarasekera, M.; Hii, C.S.; Kesper, D.A.; Ma, Y.; D’Vaz, N.; Renz, H.; Potaczek, D.P.; Prescott, S.L.; et al. The role of PKCzeta in cord blood T-cell maturation towards Th1 cytokine profile and its epigenetic regulation by fish oil. Biosci. Rep. 2017, 37. [Google Scholar] [CrossRef]
- Harb, H.; Alashkar Alhamwe, B.; Acevedo, N.; Frumento, P.; Johansson, C.; Eick, L.; Papadogiannakis, N.; Alm, J.; Renz, H.; Potaczek, D.P.; et al. Epigenetic Modifications in Placenta are Associated with the Child’s Sensitization to Allergens. BioMed Res. Int. 2019, 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Gomez de Aguero, M.; Ganal-Vonarburg, S.C.; Fuhrer, T.; Rupp, S.; Uchimura, Y.; Li, H.; Steinert, A.; Heikenwalder, M.; Hapfelmeier, S.; Sauer, U.; et al. The maternal microbiota drives early postnatal innate immune development. Science 2016, 351, 1296–1302. [Google Scholar] [CrossRef] [PubMed]
- Nardelli, C.; Granata, I.; Iaffaldano, L.; D’Argenio, V.; Del Monaco, V.; Maruotti, G.M.; Omodei, D.; Del Vecchio, L.; Martinelli, P.; Salvatore, F.; et al. miR-138/miR-222 Overexpression Characterizes the miRNome of Amniotic Mesenchymal Stem Cells in Obesity. Stem Cells Dev. 2017, 26, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Capobianco, V.; Caterino, M.; Iaffaldano, L.; Nardelli, C.; Sirico, A.; Del Vecchio, L.; Martinelli, P.; Pastore, L.; Pucci, P.; Sacchetti, L. Proteome analysis of human amniotic mesenchymal stem cells (hA-MSCs) reveals impaired antioxidant ability, cytoskeleton and metabolic functionality in maternal obesity. Sci. Rep. 2016, 6, 25270. [Google Scholar] [CrossRef] [PubMed]
- Iaffaldano, L.; Nardelli, C.; D’Alessio, F.; D’Argenio, V.; Nunziato, M.; Mauriello, L.; Procaccini, C.; Maruotti, G.M.; Martinelli, P.; Matarese, G.; et al. Altered Bioenergetic Profile in Umbilical Cord and Amniotic Mesenchymal Stem Cells from Newborns of Obese Women. Stem Cells Dev. 2018, 27, 199–206. [Google Scholar] [CrossRef]
- Lehnen, H.; Zechner, U.; Haaf, T. Epigenetics of gestational diabetes mellitus and offspring health: The time for action is in early stages of life. Mol. Hum. Reprod. 2013, 19, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Merkhan, M.M.; Forsyth, N.R.; Wu, P. Chorionic and amniotic membrane-derived stem cells have distinct, and gestational diabetes mellitus independent, proliferative, differentiation, and immunomodulatory capacities. Stem Cell Res. 2019, 40, 101537. [Google Scholar] [CrossRef]
- Hwang, J.H.; Lee, M.J.; Seok, O.S.; Paek, Y.C.; Cho, G.J.; Seol, H.J.; Lee, J.K.; Oh, M.J. Cytokine expression in placenta-derived mesenchymal stem cells in patients with pre-eclampsia and normal pregnancies. Cytokine 2010, 49, 95–101. [Google Scholar] [CrossRef]
- Mor, G.; Romero, R.; Aldo, P.B.; Abrahams, V.M. Is the trophoblast an immune regulator? The role of Toll-like receptors during pregnancy. Crit. Rev. Immunol. 2005, 25, 375–388. [Google Scholar] [CrossRef]
- Oreshkova, T.; Dimitrov, R.; Mourdjeva, M. A cross-talk of decidual stromal cells, trophoblast, and immune cells: A prerequisite for the success of pregnancy. Am. J. Reprod. Immunol. 2012, 68, 366–373. [Google Scholar] [CrossRef]
| Origin | Tissue | Acronym | Phenotype | Function |
|---|---|---|---|---|
| Maternal | Decidua | dMSC | Positive for: CD44 [22,23,25,31], CD29 [25,31], CD54 [31], CD73 [25,31], CD90 [22,23,25,31], CD105 [22,23,25,31], CD146 [22,23], CD166 [22,23,31], HLA-A,B,C [22,23,31] Negative for: CD3 [25], CD11b [25], CD14 [22,23,25,31], CD19 [22,23,25], CD25 [31], CD31 [22,23,31], CD34 [25,31], CD40 [22,23,31], CD45 [22,23,25,31], CD56 [25], CD80 [22,23,31], CD83 [22,23], CD86 [22,23,31], CD117 [31], CD123 [31], CD133 [31], HLA-DR [22,23,25,31], HLA-G [25,31] | Differentiation of CD34+ precursor cells into functional natural killer (NK) cells [32] |
| Inhibition of NK cell proliferation and cytotoxicity [33,34,35] | ||||
| Inhibition of T cell proliferation [31,33,36] | ||||
| Inhibition of dendritic cell (DC) differentiation [31,33] | ||||
| Induction of M2 macrophage phenotype [37] | ||||
| Fetal | Umbilical cord | hUCMSC | Positive for: CD73 [25,26], CD90 [25,26], CD105 [25,26], CD29 [25], CD44 [25], CD49d [26], CD56low [25], CD133 [21], CD142 [26], CD146 [26], CD166 [26], CD200 [26], CD235a [21], CD349 [26], B7-H1/PD-L1 [38], HER1 [26], HER2 [26], MCSP [26], cMet [26] Negative for: CD3 [25], CD11b [25], CD14 [25,26], CD19 [25,26], CD31 [26], CD34 [25,26], CD45 [25,26], CD80 [38], CD86 [38], CD136 [26], CD143 [26], CD271 [26], HLA-DR [25,26,38], HLA-G [25], B7-DC/PD-L2 [38] | Inhibition of NK cell cytotoxicity and activation [39,40,41,42] |
| Expansion of T regulatory (Treg) cells [38] | ||||
| Inhibition of monocyte-derived DC differentiation [38] | ||||
| In a murine model of renal damage, decrease of macrophage infiltration [43] | ||||
| Induction of M2 macrophages [43] | ||||
| Wharton’s jelly | hWJMSC | Positive for: CD10 [20,21], CD13 [20,21], CD29 [20,21], CD44 [20,21], CD49e [20], CD51 [20,21], CD68 [20,21], CD73 [20,21], CD80 [20], CD90 [20,21], CD105 [20,21], CD117 [20], CD166 [20,21], CD200 [44], ALP21, CK8 [20], CK18 [20], CK19 [20], Connexin-43 [20], COX-2 [44], DNMT3B [21], GABRB3 [21], GATA-4 [20], GATA-5 [20], GATA-6 [20], GFAP [20], HLA-A,B,C [20,21], HLA-E [44], HLA-G [20,21,44], HNF-4α [20], IDO-1 [44], Nanog [20,21], Nestin [20], NSE [20], OCT3/4A [20], CD274/PD-L1 [44], REX2 [21], α-SMA [20], SOX2 [21], SSEA-4 [21], Tra-1-60 [21], Tra-1-81 [21], TSG-6 [44], Vimentin [20] Negative for: CD14 [20,21], CD31 [20,21], CD33 [20], CD34 [20,21], CD45 [20,21], CD56 [20,21], CD86 [20], CD163 [20], CK-7 [20] Discordant markers: HLA-DR (Negative [20] or positive [21]) | Inhibition of T cell proliferation [36,44,45,46] | |
| Inhibition of monocyte-derived DC differentiation [44,47] | ||||
| In diabetic NOD mice, reduction of systemic and pancreatic levels of T helper 1 (Th1) and Th17, shift toward Th2, increment of Treg cell levels, decrease of DC [48] | ||||
| Amnion | hAMSC | Positive for: CD10 [18,26,49,50], CD13 [18,27,49,50,51], CD26 [49], CD29 [18,25,27,31,52], CD44 [18,25,27,31,49,50,52], CD49a [49,50], CD49b [49,50], CD49c [18,49,50], CD49d [18,26,49,50], CD49e [18,50], CD49f [50], CD54 [18,31], CD59 [49], CD73 [18,25,26,27,31,36,49,50,51,52,53,54], CD85a [49], CD90 [18,25,26,27,31,36,49,50,51,52,53,54], CD105 [18,25,26,27,31,36,49,50,52,53,54], CD140b [18,49], CD142 [26], CD146 [26,49], CD166 [18,26,27], CD200 [26,49], CD252 [49], CD272 [49], CD273/PD-L2 [49], CD274/PD-L1 [49], CD284 [49], CD349 [18,26], HER1 [26], HER2 [26], HLA-A,B,C [18,27,49,50,52,54], MCSP [26], cMet [26], Oct-4 [18] Negative for: B7-H4 [49], CD3 [18,25], CD11b [25], CD14 [18,25,26,31,50,53,54], CD19 [25,26,53], CD25 [31], CD31 [18,26,31,49,52], CD33 [49], CD34 [18,25,26,31,36,49,52,53,54], CD39 [49], CD40 [31], CD45 [18,25,26,31,36,49,50,52,53,54], CD52 [49], CD70 [49], CD80 [27,31], CD86 [27,31], CD106 [49], CD109 [49], CD114 [49], CD116 [49], CD117 [31,49,50], CD120b [49], CD123 [31], CD124 [49], CD133 [18,31], CD136 [26], CD143 [26], CD146 [50], CD152 [49], CD154 [49], CD244 [49], CD271 [49], CD275 [49], CD282 [49], CD324 [18], CD326 [50], HLA-DM [49], HLA-DQ [49], HLA-DR [18,25,26,27,31,50,52,53,54], HLA-G [25,31,49] Discordant markers: CD271 (low [18] or negative [26]), CD166 (weakly positive [31,50] or positive [49]) | Expansion of cord-blood NK cells in the presence of specific cytokines [55] | |
| Inhibition of NK cell cytotoxicity and activation [56] | ||||
| Inhibition of T cell proliferation [31,36,49,52,53,54,57,58,59,60] | ||||
| Expansion of Treg cells [49,61] | ||||
| Inhibition of Th1, Th17 formation [49,61,62] | ||||
| Inhibition of inflammatory cytokines [45,49,52,60,61,62] | ||||
| Inhibition of monocyte-derived DC differentiation [49,50,63,64] | ||||
| Inhibition of monocyte-derived M1 differentiation [49,51] | ||||
| Induction of M2 macrophage phenotype [49,50,51,65] | ||||
| Chorion | hCMSC | Positive for: CD10 [18], CD13 [18], CD29 [18,25,66], CD44 [18,25,66], CD49e [18], CD54 [66], CD73 [18,25,53,66], CD90 [18,25,53,66], CD105 [18,25,53,66], CD140b [18], CD166 [18], CD349 [18], CD271low [18], HLA-A,B,C [18] Negative for: CD3 [25,66], CD11b [25], CD14 [18,25,53,66], CD19 [25,53], CD34 [18,25,53,66], CD45 [18,25,53,66], CD56, [25], CD117 [18], CD133 [18], CD324 [18], HLA-DR [18,25,53], HLA-G [18,25] | Inhibition of T cell proliferation [53,66] | |
| Chorionic villi | hCVMSC | Positive for: CD29 [45], CD44 [45,67,68], CD73 [45], CD90 [45,67,68], CD146 [67,68], CD166 [67,68], CD105 [45,67,68], HLA-class I [45] Negative for: CD14 [45], CD19 [67,68], CD31 [45], CD34 [45], CD40 [67,68], CD45 [45,67,68], CD80 [67,68], CD86 [67,68], CXCR4 [45], HLA-DR [45,67,68], SSEA-3 [45], SSEA-4 [45] | Inhibition of monocyte-derived DC differentiation [68] | |
| Inhibition of monocyte-derived M1 differentiation [67] | ||||
| Induction of M2 macrophage phenotype [67] | ||||
| Decreased secretion of IL-12 and IFN-gamma when co-cultured with T cell and DC [68] | ||||
| Promotion of IL-17/Th17 [45] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magatti, M.; Stefani, F.R.; Papait, A.; Cargnoni, A.; Masserdotti, A.; Silini, A.R.; Parolini, O. Perinatal Mesenchymal Stromal Cells and Their Possible Contribution to Fetal-Maternal Tolerance. Cells 2019, 8, 1401. https://doi.org/10.3390/cells8111401
Magatti M, Stefani FR, Papait A, Cargnoni A, Masserdotti A, Silini AR, Parolini O. Perinatal Mesenchymal Stromal Cells and Their Possible Contribution to Fetal-Maternal Tolerance. Cells. 2019; 8(11):1401. https://doi.org/10.3390/cells8111401
Chicago/Turabian StyleMagatti, Marta, Francesca Romana Stefani, Andrea Papait, Anna Cargnoni, Alice Masserdotti, Antonietta Rosa Silini, and Ornella Parolini. 2019. "Perinatal Mesenchymal Stromal Cells and Their Possible Contribution to Fetal-Maternal Tolerance" Cells 8, no. 11: 1401. https://doi.org/10.3390/cells8111401
APA StyleMagatti, M., Stefani, F. R., Papait, A., Cargnoni, A., Masserdotti, A., Silini, A. R., & Parolini, O. (2019). Perinatal Mesenchymal Stromal Cells and Their Possible Contribution to Fetal-Maternal Tolerance. Cells, 8(11), 1401. https://doi.org/10.3390/cells8111401







