MicroRNAs in Uteroplacental Vascular Dysfunction
Abstract
1. Introduction
2. MiRNA Expression Patterns in Uteroplacental Tissues
2.1. Biogenesis of miRNAs
2.2. MiRNA on Gene Expression
2.3. Placental miRNAs in Normal Pregnancy
2.4. Placental miRNAs in Compromised Pregnancies
3. MiRNAs and Uteroplacental Circulation Adaptation under Physiological and Pathophysiological Conditions
3.1. Structural Adaptation of Uterine Arteries in Normal Pregnancy
3.2. Functional Adaptation of Uterine Arteries in Normal Pregnancy
3.3. Spiral Artery Maltransformation and Uterine Vascular Maladaptation in Compromised Pregnancies
3.4. Impaired Trophoblast Invasion by miRNAs in Preeclampsia and IUGR
3.4.1. MAPK Signaling Pathway
3.4.2. PI3K/AKT Signaling Pathway
3.4.3. TGF-β Signaling Pathway
3.4.4. Wnt Signaling Pathway
3.4.5. Notch Signaling Pathway
3.4.6. EphrinB2-EphB4 Signaling Pathway
3.4.7. Insulin-like Growth Factor 1 (IGF-1) Signaling Pathway
3.4.8. Matrix Metalloproteinases (MMPs)
3.4.9. VEGF
3.4.10. eNOS
3.4.11. Other Signaling Elements
3.5. miRNAs in Maladaptation of Uterine Vascular Function in Preeclampsia and IUGR
4. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Alles, J.; Fehlmann, T.; Fischer, U.; Backes, C.; Galata, V.; Minet, M.; Hart, M.; Abu-Halima, M.; Grasser, F.A.; Lenhof, H.P.; et al. An estimate of the total number of true human miRNAs. Nucleic Acids Res. 2019, 47, 3353–3364. [Google Scholar] [CrossRef] [PubMed]
- Sayed, D.; Abdellatif, M. MicroRNAs in development and disease. Physiol. Rev. 2011, 91, 827–887. [Google Scholar] [CrossRef] [PubMed]
- American College of Obstetricians and Gynecologists. Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Obstet. Gynecol. 2013, 122, 1122–1131. [Google Scholar] [CrossRef]
- Tranquilli, A.L.; Dekker, G.; Magee, L.; Roberts, J.; Sibai, B.M.; Steyn, W.; Zeeman, G.G.; Brown, M.A. The classification, diagnosis and management of the hypertensive disorders of pregnancy: A revised statement from the ISSHP. Pregnancy Hypertens. 2014, 4, 97–104. [Google Scholar] [CrossRef]
- Vayssiere, C.; Sentilhes, L.; Ego, A.; Bernard, C.; Cambourieu, D.; Flamant, C.; Gascoin, G.; Gaudineau, A.; Grange, G.; Houfflin-Debarge, V.; et al. Fetal growth restriction and intra-uterine growth restriction: Guidelines for clinical practice from the French College of Gynaecologists and Obstetricians. Eur. J. Obstet. Gynecol. Reprod. Biol. 2015, 193, 10–18. [Google Scholar] [CrossRef]
- Phipps, E.A.; Thadhani, R.; Benzing, T.; Karumanchi, S.A. Pre-eclampsia: Pathogenesis, novel diagnostics and therapies. Nat. Rev. Nephrol. 2019, 15, 275–289. [Google Scholar] [CrossRef]
- Goulopoulou, S. Maternal Vascular Physiology in Preeclampsia. Hypertension 2017, 70, 1066–1073. [Google Scholar] [CrossRef]
- Lane-Cordova, A.D.; Khan, S.S.; Grobman, W.A.; Greenland, P.; Shah, S.J. Long-Term Cardiovascular Risks Associated with Adverse Pregnancy Outcomes: JACC Review Topic of the Week. J. Am. Coll. Cardiol. 2019, 73, 2106–2116. [Google Scholar] [CrossRef]
- Huppertz, B. Placental origins of preeclampsia: Challenging the current hypothesis. Hypertension 2008, 51, 970–975. [Google Scholar] [CrossRef]
- Kalafat, E.; Thilaganathan, B. Cardiovascular origins of preeclampsia. Curr. Opin. Obstet. Gynecol. 2017, 29, 383–389. [Google Scholar] [CrossRef]
- Menendez-Castro, C.; Rascher, W.; Hartner, A. Intrauterine growth restriction—Impact on cardiovascular diseases later in life. Mol. Cell. Pediatr. 2018, 5, 4. [Google Scholar] [CrossRef] [PubMed]
- Rashid, C.S.; Bansal, A.; Simmons, R.A. Oxidative Stress, Intrauterine Growth Restriction, and Developmental Programming of Type 2 Diabetes. Physiology 2018, 33, 348–359. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.M.; Escudero, C. The placenta in preeclampsia. Pregnancy Hypertens. 2012, 2, 72–83. [Google Scholar] [CrossRef] [PubMed]
- Burton, G.J.; Jauniaux, E. Pathophysiology of placental-derived fetal growth restriction. Am. J. Obstet. Gynecol. 2018, 218, S745–S761. [Google Scholar] [CrossRef]
- Mouillet, J.F.; Ouyang, Y.; Coyne, C.B.; Sadovsky, Y. MicroRNAs in placental health and disease. Am. J. Obstet. Gynecol. 2015, 213, S163–S172. [Google Scholar] [CrossRef]
- Hayder, H.; O’Brien, J.; Nadeem, U.; Peng, C. MicroRNAs: Crucial regulators of placental development. Reproduction 2018, 155, R259–R271. [Google Scholar] [CrossRef]
- Malnou, E.C.; Umlauf, D.; Mouysset, M.; Cavaille, J. Imprinted MicroRNA Gene Clusters in the Evolution, Development, and Functions of Mammalian Placenta. Front. Genet. 2018, 9, 706. [Google Scholar] [CrossRef]
- Chiofalo, B.; Lagana, A.S.; Vaiarelli, A.; La Rosa, V.L.; Rossetti, D.; Palmara, V.; Valenti, G.; Rapisarda, A.M.C.; Granese, R.; Sapia, F.; et al. Do miRNAs Play a Role in Fetal Growth Restriction? A Fresh Look to a Busy Corner. Biomed. Res. Int. 2017, 2017, 6073167. [Google Scholar] [CrossRef]
- Bounds, K.R.; Chiasson, V.L.; Pan, L.J.; Gupta, S.; Chatterjee, P. MicroRNAs: New Players in the Pathobiology of Preeclampsia. Front. Cardiovasc. Med. 2017, 4, 60. [Google Scholar] [CrossRef]
- Yi, R.; Qin, Y.; Macara, I.G.; Cullen, B.R. Exportin-5 mediates the nuclear export of pre-microRNAs and short hairpin RNAs. Genes Dev. 2003, 17, 3011–3016. [Google Scholar] [CrossRef]
- Desvignes, T.; Batzel, P.; Berezikov, E.; Eilbeck, K.; Eppig, J.T.; McAndrews, M.S.; Singer, A.; Postlethwait, J.H. miRNA Nomenclature: A View Incorporating Genetic Origins, Biosynthetic Pathways, and Sequence Variants. Trends Genet. 2015, 31, 613–626. [Google Scholar] [CrossRef] [PubMed]
- Huntzinger, E.; Izaurralde, E. Gene silencing by microRNAs: Contributions of translational repression and mRNA decay. Nat. Rev. Genet. 2011, 12, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Treiber, T.; Treiber, N.; Meister, G. Regulation of microRNA biogenesis and its crosstalk with other cellular pathways. Nat. Rev. Mol. Cell. Biol. 2019, 20, 5–20. [Google Scholar] [CrossRef] [PubMed]
- Vasudevan, S.; Tong, Y.; Steitz, J.A. Switching from repression to activation: microRNAs can up-regulate translation. Science 2007, 318, 1931–1934. [Google Scholar] [CrossRef]
- Fabian, M.R.; Sonenberg, N.; Filipowicz, W. Regulation of mRNA translation and stability by microRNAs. Annu. Rev. Biochem. 2010, 79, 351–379. [Google Scholar] [CrossRef]
- Donker, R.B.; Mouillet, J.F.; Chu, T.; Hubel, C.A.; Stolz, D.B.; Morelli, A.E.; Sadovsky, Y. The expression profile of C19MC microRNAs in primary human trophoblast cells and exosomes. Mol. Hum. Reprod. 2012, 18, 417–424. [Google Scholar] [CrossRef]
- Morales-Prieto, D.M.; Chaiwangyen, W.; Ospina-Prieto, S.; Schneider, U.; Herrmann, J.; Gruhn, B.; Markert, U.R. MicroRNA expression profiles of trophoblastic cells. Placenta 2012, 33, 725–734. [Google Scholar] [CrossRef]
- Morales-Prieto, D.M.; Ospina-Prieto, S.; Schmidt, A.; Chaiwangyen, W.; Markert, U.R. Elsevier Trophoblast Research Award Lecture: Origin, evolution and future of placenta miRNAs. Placenta 2014, 35, S39–S45. [Google Scholar] [CrossRef]
- Ishibashi, O.; Ohkuchi, A.; Ali, M.M.; Kurashina, R.; Luo, S.S.; Ishikawa, T.; Takizawa, T.; Hirashima, C.; Takahashi, K.; Migita, M.; et al. Hydroxysteroid (17-beta) dehydrogenase 1 is dysregulated by miR-210 and miR-518c that are aberrantly expressed in preeclamptic placentas: A novel marker for predicting preeclampsia. Hypertension 2012, 59, 265–273. [Google Scholar] [CrossRef]
- Wang, W.; Feng, L.; Zhang, H.; Hachy, S.; Satohisa, S.; Laurent, L.C.; Parast, M.; Zheng, J.; Chen, D.B. Preeclampsia up-regulates angiogenesis-associated microRNA (i.e., miR-17, -20a, and -20b) that target ephrin-B2 and EPHB4 in human placenta. J. Clin. Endocrinol. Metab. 2012, 97, E1051–E1059. [Google Scholar] [CrossRef]
- Gu, Y.; Sun, J.; Groome, L.J.; Wang, Y. Differential miRNA expression profiles between the first and third trimester human placentas. Am. J. Physiol. Endocrinol. Metab. 2013, 304, E836–E843. [Google Scholar] [CrossRef] [PubMed]
- Farrokhnia, F.; Aplin, J.D.; Westwood, M.; Forbes, K. MicroRNA regulation of mitogenic signaling networks in the human placenta. J. Biol. Chem. 2014, 289, 30404–30416. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Ye, G.; Nadeem, L.; Fu, G.; Yang, B.B.; Honarparvar, E.; Dunk, C.; Lye, S.; Peng, C. MicroRNA-378a-5p promotes trophoblast cell survival, migration and invasion by targeting Nodal. J. Cell Sci. 2012, 125, 3124–3132. [Google Scholar] [CrossRef] [PubMed]
- Fu, G.; Ye, G.; Nadeem, L.; Ji, L.; Manchanda, T.; Wang, Y.; Zhao, Y.; Qiao, J.; Wang, Y.L.; Lye, S.; et al. MicroRNA-376c impairs transforming growth factor-beta and nodal signaling to promote trophoblast cell proliferation and invasion. Hypertension 2013, 61, 864–872. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.L.; Zhang, H.Z.; Meng, F.R.; Han, S.Y.; Zhang, M. Differential expression of microRNA-411 and 376c is associated with hypertension in pregnancy. Braz. J. Med. Biol. Res. 2019, 52, e7546. [Google Scholar] [CrossRef]
- Pineles, B.L.; Romero, R.; Montenegro, D.; Tarca, A.L.; Han, Y.M.; Kim, Y.M.; Draghici, S.; Espinoza, J.; Kusanovic, J.P.; Mittal, P.; et al. Distinct subsets of microRNAs are expressed differentially in the human placentas of patients with preeclampsia. Am. J. Obstet. Gynecol. 2007, 196, e261–e266. [Google Scholar] [CrossRef]
- Hu, Y.; Li, P.; Hao, S.; Liu, L.; Zhao, J.; Hou, Y. Differential expression of microRNAs in the placentae of Chinese patients with severe pre-eclampsia. Clin. Chem. Lab. Med. 2009, 47, 923–929. [Google Scholar] [CrossRef]
- Zhu, X.M.; Han, T.; Sargent, I.L.; Yin, G.W.; Yao, Y.Q. Differential expression profile of microRNAs in human placentas from preeclamptic pregnancies vs. normal pregnancies. Am. J. Obstet. Gynecol. 2009, 200, e661–e667. [Google Scholar] [CrossRef]
- Enquobahrie, D.A.; Abetew, D.F.; Sorensen, T.K.; Willoughby, D.; Chidambaram, K.; Williams, M.A. Placental microRNA expression in pregnancies complicated by preeclampsia. Am. J. Obstet. Gynecol. 2011, 204, e112–e121. [Google Scholar] [CrossRef]
- Mayor-Lynn, K.; Toloubeydokhti, T.; Cruz, A.C.; Chegini, N. Expression profile of microRNAs and mRNAs in human placentas from pregnancies complicated by preeclampsia and preterm labor. Reprod. Sci. 2011, 18, 46–56. [Google Scholar] [CrossRef]
- Betoni, J.S.; Derr, K.; Pahl, M.C.; Rogers, L.; Muller, C.L.; Packard, R.E.; Carey, D.J.; Kuivaniemi, H.; Tromp, G. MicroRNA analysis in placentas from patients with preeclampsia: Comparison of new and published results. Hypertens. Pregnancy 2013, 32, 321–339. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.Y.; Yun, J.; Lee, O.J.; Han, H.S.; Yeo, M.K.; Lee, M.A.; Suh, K.S. MicroRNA expression profiles in placenta with severe preeclampsia using a PNA-based microarray. Placenta 2013, 34, 799–804. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Zhao, Y.; Liu, M.; Wang, Y.; Wang, H.; Li, Y.X.; Zhu, X.; Yao, Y.; Wang, H.; Qiao, J.; et al. Variations of microRNAs in human placentas and plasma from preeclamptic pregnancy. Hypertension 2014, 63, 1276–1284. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Li, J.; Wu, G.; Miao, Z.; Lu, L.; Ren, G.; Wang, X. Upregulation of microRNA335 and microRNA584 contributes to the pathogenesis of severe preeclampsia through downregulation of endothelial nitric oxide synthase. Mol. Med. Rep. 2015, 12, 5383–5390. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Li, Q.; Ren, N.; Li, C.; Wang, X.; Xie, M.; Gao, Z.; Pan, Z.; Zhao, C.; Ren, C.; et al. Placental miR-106a approximately 363 cluster is dysregulated in preeclamptic placenta. Placenta 2015, 36, 250–252. [Google Scholar] [CrossRef]
- Lykoudi, A.; Kolialexi, A.; Lambrou, G.I.; Braoudaki, M.; Siristatidis, C.; Papaioanou, G.K.; Tzetis, M.; Mavrou, A.; Papantoniou, N. Dysregulated placental microRNAs in Early and Late onset Preeclampsia. Placenta 2018, 61, 24–32. [Google Scholar] [CrossRef]
- Awamleh, Z.; Gloor, G.B.; Han, V.K.M. Placental microRNAs in pregnancies with early onset intrauterine growth restriction and preeclampsia: Potential impact on gene expression and pathophysiology. BMC Med. Genom. 2019, 12, 91. [Google Scholar] [CrossRef]
- Higashijima, A.; Miura, K.; Mishima, H.; Kinoshita, A.; Jo, O.; Abe, S.; Hasegawa, Y.; Miura, S.; Yamasaki, K.; Yoshida, A.; et al. Characterization of placenta-specific microRNAs in fetal growth restriction pregnancy. Prenat. Diagn. 2013, 33, 214–222. [Google Scholar] [CrossRef]
- Thamotharan, S.; Chu, A.; Kempf, K.; Janzen, C.; Grogan, T.; Elashoff, D.A.; Devaskar, S.U. Differential microRNA expression in human placentas of term intra-uterine growth restriction that regulates target genes mediating angiogenesis and amino acid transport. PLoS ONE 2017, 12, e0176493. [Google Scholar] [CrossRef]
- Ostling, H.; Kruse, R.; Helenius, G.; Lodefalk, M. Placental expression of microRNAs in infants born small for gestational age. Placenta 2019, 81, 46–53. [Google Scholar] [CrossRef]
- Hromadnikova, I.; Kotlabova, K.; Ondrackova, M.; Pirkova, P.; Kestlerova, A.; Novotna, V.; Hympanova, L.; Krofta, L. Expression profile of C19MC microRNAs in placental tissue in pregnancy-related complications. DNA Cell Biol. 2015, 34, 437–457. [Google Scholar] [CrossRef] [PubMed]
- Hromadnikova, I.; Kotlabova, K.; Hympanova, L.; Krofta, L. Cardiovascular and Cerebrovascular Disease Associated microRNAs Are Dysregulated in Placental Tissues Affected with Gestational Hypertension, Preeclampsia and Intrauterine Growth Restriction. PLoS ONE 2015, 10, e0138383. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, C.C.; Cheng, H.H.; Tewari, M. MicroRNA profiling: Approaches and considerations. Nat. Rev. Genet. 2012, 13, 358–369. [Google Scholar] [CrossRef] [PubMed]
- Hunt, E.A.; Broyles, D.; Head, T.; Deo, S.K. MicroRNA Detection: Current Technology and Research Strategies. Annu. Rev. Anal. Chem. 2015, 8, 217–237. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.Q.; Dasgupta, C.; Xiao, J.; Yang, S.; Zhang, L. Long-term high altitude hypoxia during gestation suppresses large conductance Ca2+ -activated K+ channel function in uterine arteries: A causal role for microRNA-210. J. Physiol. 2018, 596, 5891–5906. [Google Scholar] [CrossRef]
- Noack, F.; Ribbat-Idel, J.; Thorns, C.; Chiriac, A.; Axt-Fliedner, R.; Diedrich, K.; Feller, A.C. miRNA expression profiling in formalin-fixed and paraffin-embedded placental tissue samples from pregnancies with severe preeclampsia. J. Perinat. Med. 2011, 39, 267–271. [Google Scholar] [CrossRef]
- Timofeeva, A.V.; Gusar, V.A.; Kan, N.E.; Prozorovskaya, K.N.; Karapetyan, A.O.; Bayev, O.R.; Chagovets, V.V.; Kliver, S.F.; Iakovishina, D.Y.; Frankevich, V.E.; et al. Identification of potential early biomarkers of preeclampsia. Placenta 2018, 61, 61–71. [Google Scholar] [CrossRef]
- Dai, X.; Cai, Y. Down-regulation of microRNA let-7d inhibits the proliferation and invasion of trophoblast cells in preeclampsia. J. Cell. Biochem. 2018, 119, 1141–1151. [Google Scholar] [CrossRef]
- Wang, N.; Li, R.; Xue, M. Potential regulatory network in the PSG10P/miR-19a-3p/IL1RAP pathway is possibly involved in preeclampsia pathogenesis. J. Cell. Mol. Med. 2019, 23, 852–864. [Google Scholar] [CrossRef]
- Kumar, P.; Luo, Y.; Tudela, C.; Alexander, J.M.; Mendelson, C.R. The c-Myc-regulated microRNA-17~92 (miR-17~92) and miR-106a~363 clusters target hCYP19A1 and hGCM1 to inhibit human trophoblast differentiation. Mol. Cell. Biol. 2013, 33, 1782–1796. [Google Scholar] [CrossRef]
- Lou, C.X.; Zhou, X.T.; Tian, Q.C.; Xie, H.Q.; Zhang, J.Y. Low expression of microRNA-21 inhibits trophoblast cell infiltration through targeting PTEN. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 6181–6189. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, Y.; Wang, H.; Wang, J.; Zhang, Y.; Wang, Y.; Pan, Z.; Luo, S. Aberrantly up-regulated miR-20a in pre-eclampsic placenta compromised the proliferative and invasive behaviors of trophoblast cells by targeting forkhead box protein A1. Int. J. Biol. Sci. 2014, 10, 973–982. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Li, Q.; Xu, J.; Zhang, X.; Zhang, H.; Xiang, Y.; Fang, C.; Wang, T.; Xia, S.; Zhang, Q.; et al. The aberrantly expressed miR-193b-3p contributes to preeclampsia through regulating transforming growth factor-beta signaling. Sci. Rep. 2016, 6, 19910. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Jiang, Z.; Yu, X.; Zhang, Y.; Sun, M.; Wang, W.; Ge, Z.; De, W.; Sun, L. MiR-101 regulates apoptosis of trophoblast HTR-8/SVneo cells by targeting endoplasmic reticulum (ER) protein 44 during preeclampsia. J. Hum. Hypertens. 2014, 28, 610–616. [Google Scholar] [CrossRef]
- Yan, T.; Liu, Y.; Cui, K.; Hu, B.; Wang, F.; Zou, L. MicroRNA-126 regulates EPCs function: Implications for a role of miR-126 in preeclampsia. J. Cell. Biochem. 2013, 114, 2148–2159. [Google Scholar] [CrossRef]
- Hong, F.; Li, Y.; Xu, Y. Decreased placental miR-126 expression and vascular endothelial growth factor levels in patients with pre-eclampsia. J. Int. Med. Res. 2014, 42, 1243–1251. [Google Scholar] [CrossRef]
- Li, P.; Guo, W.; Du, L.; Zhao, J.; Wang, Y.; Liu, L.; Hu, Y.; Hou, Y. microRNA-29b contributes to pre-eclampsia through its effects on apoptosis, invasion and angiogenesis of trophoblast cells. Clin. Sci. 2013, 124, 27–40. [Google Scholar] [CrossRef]
- Niu, Z.R.; Han, T.; Sun, X.L.; Luan, L.X.; Gou, W.L.; Zhu, X.M. MicroRNA-30a-3p is overexpressed in the placentas of patients with preeclampsia and affects trophoblast invasion and apoptosis by its effects on IGF-1. Am. J. Obstet. Gynecol. 2018, 218, 249. [Google Scholar] [CrossRef]
- Sun, M.; Chen, H.; Liu, J.; Tong, C.; Meng, T. MicroRNA-34a inhibits human trophoblast cell invasion by targeting MYC. BMC Cell Biol. 2015, 16, 21. [Google Scholar] [CrossRef]
- Guo, M.; Zhao, X.; Yuan, X.; Li, P. Elevated microRNA-34a contributes to trophoblast cell apoptosis in preeclampsia by targeting BCL-2. J. Hum. Hypertens. 2017, 31, 815–820. [Google Scholar] [CrossRef]
- Xue, F.; Yang, J.; Li, Q.; Zhou, H. Down-regulation of microRNA-34a-5p promotes trophoblast cell migration and invasion via targetting Smad4. Biosci. Rep. 2019, 39. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Tao, T.; Yin, Y.; Zhao, L.; Yang, L.; Hu, L. miR-144 may regulate the proliferation, migration and invasion of trophoblastic cells through targeting PTEN in preeclampsia. Biomed. Pharmacother. 2017, 94, 341–353. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Li, J.; Tong, M.; Li, Q.; Chen, Y.; Lu, H.; Wang, Y.; Min, L. MicroRNA1443p may participate in the pathogenesis of preeclampsia by targeting Cox2. Mol. Med. Rep. 2019, 19, 4655–4662. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Tsai, S.Q.; Hardison, N.E.; James, A.H.; Motsinger-Reif, A.A.; Thames, B.; Stone, E.A.; Deng, C.; Piedrahita, J.A. Differentially expressed microRNAs and affected biological pathways revealed by modulated modularity clustering (MMC) analysis of human preeclamptic and IUGR placentas. Placenta 2013, 34, 599–605. [Google Scholar] [CrossRef]
- Xiaobo, Z.; Qizhi, H.; Zhiping, W.; Tao, D. Down-regulated miR-149-5p contributes to preeclampsia via modulating endoglin expression. Pregnancy Hypertens. 2019, 15, 201–208. [Google Scholar] [CrossRef]
- Li, Q.; Pan, Z.; Wang, X.; Gao, Z.; Ren, C.; Yang, W. miR-125b-1-3p inhibits trophoblast cell invasion by targeting sphingosine-1-phosphate receptor 1 in preeclampsia. Biochem. Biophys. Res. Commun. 2014, 453, 57–63. [Google Scholar] [CrossRef]
- Lu, T.M.; Lu, W.; Zhao, L.J. MicroRNA-137 Affects Proliferation and Migration of Placenta Trophoblast Cells in Preeclampsia by Targeting ERRalpha. Reprod. Sci. 2017, 24, 85–96. [Google Scholar] [CrossRef]
- Ospina-Prieto, S.; Chaiwangyen, W.; Herrmann, J.; Groten, T.; Schleussner, E.; Markert, U.R.; Morales-Prieto, D.M. MicroRNA-141 is upregulated in preeclamptic placentae and regulates trophoblast invasion and intercellular communication. Transl. Res. 2016, 172, 61–72. [Google Scholar] [CrossRef]
- Brkic, J.; Dunk, C.; O’Brien, J.; Fu, G.; Nadeem, L.; Wang, Y.L.; Rosman, D.; Salem, M.; Shynlova, O.; Yougbare, I.; et al. MicroRNA-218-5p Promotes Endovascular Trophoblast Differentiation and Spiral Artery Remodeling. Mol. Ther. 2018, 26, 2189–2205. [Google Scholar] [CrossRef]
- Liu, E.; Liu, Z.; Zhou, Y.; Chen, M.; Wang, L.; Li, J. MicroRNA1423p inhibits trophoblast cell migration and invasion by disrupting the TGFbeta1/Smad3 signaling pathway. Mol. Med. Rep. 2019, 19, 3775–3782. [Google Scholar] [CrossRef]
- Yang, Y.; Li, H.; Ma, Y.; Zhu, X.; Zhang, S.; Li, J. MiR-221-3p is down-regulated in preeclampsia and affects trophoblast growth, invasion and migration partly via targeting thrombospondin 2. Biomed. Pharmacother. 2019, 109, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Weedon-Fekjaer, M.S.; Sheng, Y.; Sugulle, M.; Johnsen, G.M.; Herse, F.; Redman, C.W.; Lyle, R.; Dechend, R.; Staff, A.C. Placental miR-1301 is dysregulated in early-onset preeclampsia and inversely correlated with maternal circulating leptin. Placenta 2014, 35, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Lazar, L.; Nagy, B.; Molvarec, A.; Szarka, A.; Rigo, J., Jr. Role of hsa-miR-325 in the etiopathology of preeclampsia. Mol. Med. Rep. 2012, 6, 597–600. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, X.; Li, C.; Dong, X.; Gou, W. MicroRNA-155 inhibits migration of trophoblast cells and contributes to the pathogenesis of severe preeclampsia by regulating endothelial nitric oxide synthase. Mol. Med. Rep. 2014, 10, 550–554. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.Y.; Li, S.H.; Chen, W.N.; Tsai, H.L.; Su, M.T. Differential miR-346 and miR-582-3p Expression in Association with Selected Maternal and Fetal Complications. Int. J. Mol. Sci. 2017, 18, 1570. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Wu, L.; Zhang, G.; Tang, R.; Zhou, X. Elevated MicroRNA-181a-5p Contributes to Trophoblast Dysfunction and Preeclampsia. Reprod. Sci. 2019, 26, 1121–1129. [Google Scholar] [CrossRef]
- Fang, Y.N.; Huang, Z.L.; Li, H.; Tan, W.B.; Zhang, Q.G.; Wang, L.; Wu, J.L. Highly expressed miR-182-5p can promote preeclampsia progression by degrading RND3 and inhibiting HTR-8/SVneo cell invasion. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 6583–6590. [Google Scholar] [CrossRef]
- Bai, Y.; Yang, W.; Yang, H.X.; Liao, Q.; Ye, G.; Fu, G.; Ji, L.; Xu, P.; Wang, H.; Li, Y.X.; et al. Downregulated miR-195 detected in preeclamptic placenta affects trophoblast cell invasion via modulating ActRIIA expression. PLoS ONE 2012, 7, e38875. [Google Scholar] [CrossRef]
- Wang, F.; Yan, J. MicroRNA-454 is involved in regulating trophoblast cell proliferation, apoptosis, and invasion in preeclampsia by modulating the expression of ephrin receptor B4. Biomed. Pharmacother. 2018, 107, 746–753. [Google Scholar] [CrossRef]
- Shi, Z.; She, K.; Li, H.; Yuan, X.; Han, X.; Wang, Y. MicroRNA-454 contributes to sustaining the proliferation and invasion of trophoblast cells through inhibiting Nodal/ALK7 signaling in pre-eclampsia. Chem. Biol. Interact. 2019, 298, 8–14. [Google Scholar] [CrossRef]
- Lalevee, S.; Lapaire, O.; Buhler, M. miR455 is linked to hypoxia signaling and is deregulated in preeclampsia. Cell Death Dis. 2014, 5, e1408. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Williams, J., III; Brown, J.; Wang, E.T.; Lee, B.; Gonzalez, T.L.; Cui, J.; Goodarzi, M.O.; Pisarska, M.D. Up-regulation of microRNA-202-3p in first trimester placenta of pregnancies destined to develop severe preeclampsia, a pilot study. Pregnancy Hypertens. 2017, 10, 7–9. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Muralimanoharan, S.; Wortman, A.C.; Mendelson, C.R. Primate-specific miR-515 family members inhibit key genes in human trophoblast differentiation and are upregulated in preeclampsia. Proc. Natl. Acad. Sci. USA 2016, 113, E7069–E7076. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Wu, K.; Wu, W.; Chen, Y.; Wu, H.; Wang, H.; Zhang, W. miR203 contributes to preeclampsia via inhibition of VEGFA expression. Mol. Med. Rep. 2018, 17, 5627–5634. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.C.; Romero, R.; Kim, J.S.; Tarca, A.L.; Montenegro, D.; Pineles, B.L.; Kim, E.; Lee, J.; Kim, S.Y.; Draghici, S.; et al. miR-210 targets iron-sulfur cluster scaffold homologue in human trophoblast cell lines: Siderosis of interstitial trophoblasts as a novel pathology of preterm preeclampsia and small-for-gestational-age pregnancies. Am. J. Pathol. 2011, 179, 590–602. [Google Scholar] [CrossRef]
- Luo, R.; Shao, X.; Xu, P.; Liu, Y.; Wang, Y.; Zhao, Y.; Liu, M.; Ji, L.; Li, Y.X.; Chang, C.; et al. MicroRNA-210 contributes to preeclampsia by downregulating potassium channel modulatory factor 1. Hypertension 2014, 64, 839–845. [Google Scholar] [CrossRef]
- Wang, R.; Liu, W.; Liu, X.; Liu, X.; Tao, H.; Wu, D.; Zhao, Y.; Zou, L. MicroRNA-210 regulates human trophoblast cell line HTR-8/SVneo function by attenuating Notch1 expression: Implications for the role of microRNA-210 in pre-eclampsia. Mol. Reprod. Dev. 2019, 86, 896–907. [Google Scholar] [CrossRef]
- Luo, R.; Wang, Y.; Xu, P.; Cao, G.; Zhao, Y.; Shao, X.; Li, Y.X.; Chang, C.; Peng, C.; Wang, Y.L. Hypoxia-inducible miR-210 contributes to preeclampsia via targeting thrombospondin type I domain containing 7A. Sci. Rep. 2016, 6, 19588. [Google Scholar] [CrossRef]
- Adel, S.; Mansour, A.; Louka, M.; Matboli, M.; Elmekkawi, S.F.; Swelam, N. Evaluation of MicroRNA-210 and Protein tyrosine phosphatase, non-receptor type 2 in Pre-eclampsia. Gene 2017, 596, 105–109. [Google Scholar] [CrossRef]
- Muralimanoharan, S.; Maloyan, A.; Mele, J.; Guo, C.; Myatt, L.G.; Myatt, L. MIR-210 modulates mitochondrial respiration in placenta with preeclampsia. Placenta 2012, 33, 816–823. [Google Scholar] [CrossRef]
- Fang, M.; Du, H.; Han, B.; Xia, G.; Shi, X.; Zhang, F.; Fu, Q.; Zhang, T. Hypoxia-inducible microRNA-218 inhibits trophoblast invasion by targeting LASP1: Implications for preeclampsia development. Int. J. Biochem. Cell Biol. 2017, 87, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; She, R.; Wang, Q.; Li, Y.; Zhang, H. Up-regulation of miR-299 suppressed the invasion and migration of HTR-8/SVneo trophoblast cells partly via targeting HDAC2 in pre-eclampsia. Biomed. Pharmacother. 2018, 97, 1222–1228. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Deng, M.; Wang, Q. MiRNA-320a inhibits trophoblast cell invasion by targeting estrogen-related receptor-gamma. J. Obstet. Gynaecol. Res. 2018, 44, 756–763. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Feng, Y.; Xu, J.; Zou, J.; Chen, M.; He, Y.; Liu, H.; Xue, M.; Gu, Y. miR-362-3p regulates cell proliferation, migration and invasion of trophoblastic cells under hypoxia through targeting Pax3. Biomed. Pharmacother. 2018, 99, 462–468. [Google Scholar] [CrossRef]
- Yang, X.; Meng, T. MicroRNA-431 affects trophoblast migration and invasion by targeting ZEB1 in preeclampsia. Gene 2019, 683, 225–232. [Google Scholar] [CrossRef]
- Fu, J.Y.; Xiao, Y.P.; Ren, C.L.; Guo, Y.W.; Qu, D.H.; Zhang, J.H.; Zhu, Y.J. Up-regulation of miR-517-5p inhibits ERK/MMP-2 pathway: Potential role in preeclampsia. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 6599–6608. [Google Scholar] [CrossRef]
- Anton, L.; Olarerin-George, A.O.; Hogenesch, J.B.; Elovitz, M.A. Placental expression of miR-517a/b and miR-517c contributes to trophoblast dysfunction and preeclampsia. PLoS ONE 2015, 10, e0122707. [Google Scholar] [CrossRef]
- Shi, Z.; Liu, B.; Li, Y.; Liu, F.; Yuan, X.; Wang, Y. MicroRNA-652-3p promotes the proliferation and invasion of the trophoblast HTR-8/SVneo cell line by targeting homeobox A9 to modulate the expression of ephrin receptor B4. Clin. Exp. Pharmacol. Physiol. 2019, 46, 587–596. [Google Scholar] [CrossRef]
- Gao, W.L.; Liu, M.; Yang, Y.; Yang, H.; Liao, Q.; Bai, Y.; Li, Y.X.; Li, D.; Peng, C.; Wang, Y.L. The imprinted H19 gene regulates human placental trophoblast cell proliferation via encoding miR-675 that targets Nodal Modulator 1 (NOMO1). RNA Biol. 2012, 9, 1002–1010. [Google Scholar] [CrossRef]
- Ding, J.; Huang, F.; Wu, G.; Han, T.; Xu, F.; Weng, D.; Wu, C.; Zhang, X.; Yao, Y.; Zhu, X. MiR-519d-3p suppresses invasion and migration of trophoblast cells via targeting MMP-2. PLoS ONE 2015, 10, e0120321. [Google Scholar] [CrossRef]
- Jiang, L.; Long, A.; Tan, L.; Hong, M.; Wu, J.; Cai, L.; Li, Q. Elevated microRNA-520g in pre-eclampsia inhibits migration and invasion of trophoblasts. Placenta 2017, 51, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q.; Wu, W.; Xu, X.; Huang, L.; Gao, Q.; Chen, H.; Sun, H.; Xia, Y.; Sha, J.; Wang, X.; et al. miR-141 contributes to fetal growth restriction by regulating PLAG1 expression. PLoS ONE 2013, 8, e58737. [Google Scholar] [CrossRef] [PubMed]
- Maccani, M.A.; Padbury, J.F.; Marsit, C.J. miR-16 and miR-21 expression in the placenta is associated with fetal growth. PLoS ONE 2011, 6, e21210. [Google Scholar] [CrossRef] [PubMed]
- Cindrova-Davies, T.; Herrera, E.A.; Niu, Y.; Kingdom, J.; Giussani, D.A.; Burton, G.J. Reduced cystathionine gamma-lyase and increased miR-21 expression are associated with increased vascular resistance in growth-restricted pregnancies: Hydrogen sulfide as a placental vasodilator. Am. J. Pathol. 2013, 182, 1448–1458. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Huang, X.; He, Z.; Xiong, Y.; Fang, Q. miRNA-210-3p regulates trophoblast proliferation and invasiveness through fibroblast growth factor 1 in selective intrauterine growth restriction. J. Cell. Mol. Med. 2019, 23, 4422–4433. [Google Scholar] [CrossRef]
- Huang, L.; Shen, Z.; Xu, Q.; Huang, X.; Chen, Q.; Li, D. Increased levels of microRNA-424 are associated with the pathogenesis of fetal growth restriction. Placenta 2013, 34, 624–627. [Google Scholar] [CrossRef]
- Wang, D.; Na, Q.; Song, W.W.; Song, G.Y. Altered Expression of miR-518b and miR-519a in the placenta is associated with low fetal birth weight. Am. J. Perinatol. 2014, 31, 729–734. [Google Scholar] [CrossRef]
- Ducsay, C.A.; Goyal, R.; Pearce, W.J.; Wilson, S.; Hu, X.Q.; Zhang, L. Gestational Hypoxia and Developmental Plasticity. Physiol. Rev. 2018, 98, 1241–1334. [Google Scholar] [CrossRef]
- Annibale, D.J.; Rosenfeld, C.R.; Stull, J.T.; Kamm, K.E. Protein content and myosin light chain phosphorylation in uterine arteries during pregnancy. Am. J. Physiol. 1990, 259, C484–C489. [Google Scholar] [CrossRef]
- Griendling, K.K.; Fuller, E.O.; Cox, R.H. Pregnancy-induced changes in sheep uterine and carotid arteries. Am. J. Physiol. 1985, 248, H658–H665. [Google Scholar] [CrossRef]
- Guenther, A.E.; Conley, A.J.; Van Orden, D.E.; Farley, D.B.; Ford, S.P. Structural and mechanical changes of uterine arteries during pregnancy in the pig. J. Anim. Sci. 1988, 66, 3144–3152. [Google Scholar] [CrossRef] [PubMed]
- Osol, G.; Cipolla, M. Pregnancy-induced changes in the three-dimensional mechanical properties of pressurized rat uteroplacental (radial) arteries. Am. J. Obstet. Gynecol. 1993, 168, 268–274. [Google Scholar] [CrossRef]
- Konje, J.C.; Kaufmann, P.; Bell, S.C.; Taylor, D.J. A longitudinal study of quantitative uterine blood flow with the use of color power angiography in appropriate for gestational age pregnancies. Am. J. Obstet. Gynecol. 2001, 185, 608–613. [Google Scholar] [CrossRef] [PubMed]
- St-Louis, J.; Sicotte, B.; Bedard, S.; Brochu, M. Blockade of angiotensin receptor subtypes in arcuate uterine artery of pregnant and postpartum rats. Hypertension 2001, 38, 1017–1023. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.K.; Zamudio, S.; Coffin, C.; Parker, S.; Stamm, E.; Moore, L.G. Quantitative estimation of human uterine artery blood flow and pelvic blood flow redistribution in pregnancy. Obstet. Gynecol. 1992, 80, 1000–1006. [Google Scholar] [PubMed]
- Rennie, M.Y.; Whiteley, K.J.; Adamson, S.L.; Sled, J.G. Quantification of Gestational Changes in the Uteroplacental Vascular Tree Reveals Vessel Specific Hemodynamic Roles During Pregnancy in Mice. Biol. Reprod. 2016, 95, 43. [Google Scholar] [CrossRef]
- Osol, G.; Mandala, M. Maternal uterine vascular remodeling during pregnancy. Physiology 2009, 24, 58–71. [Google Scholar] [CrossRef]
- Kaufmann, P.; Black, S.; Huppertz, B. Endovascular trophoblast invasion: Implications for the pathogenesis of intrauterine growth retardation and preeclampsia. Biol. Reprod. 2003, 69, 1–7. [Google Scholar] [CrossRef]
- Lyall, F. Priming and remodelling of human placental bed spiral arteries during pregnancy—A review. Placenta 2005, 26, S31–S36. [Google Scholar] [CrossRef]
- Pijnenborg, R.; Bland, J.M.; Robertson, W.B.; Brosens, I. Uteroplacental arterial changes related to interstitial trophoblast migration in early human pregnancy. Placenta 1983, 4, 397–413. [Google Scholar] [CrossRef]
- Kam, E.P.; Gardner, L.; Loke, Y.W.; King, A. The role of trophoblast in the physiological change in decidual spiral arteries. Hum. Reprod. 1999, 14, 2131–2138. [Google Scholar] [CrossRef] [PubMed]
- Ashton, S.V.; Whitley, G.S.; Dash, P.R.; Wareing, M.; Crocker, I.P.; Baker, P.N.; Cartwright, J.E. Uterine spiral artery remodeling involves endothelial apoptosis induced by extravillous trophoblasts through Fas/FasL interactions. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Keogh, R.J.; Harris, L.K.; Freeman, A.; Baker, P.N.; Aplin, J.D.; Whitley, G.S.; Cartwright, J.E. Fetal-derived trophoblast use the apoptotic cytokine tumor necrosis factor-alpha-related apoptosis-inducing ligand to induce smooth muscle cell death. Circ. Res. 2007, 100, 834–841. [Google Scholar] [CrossRef] [PubMed]
- Whitley, G.S.; Cartwright, J.E. Trophoblast-mediated spiral artery remodelling: A role for apoptosis. J. Anat. 2009, 215, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Damsky, C.H.; Fisher, S.J. Preeclampsia is associated with failure of human cytotrophoblasts to mimic a vascular adhesion phenotype. One cause of defective endovascular invasion in this syndrome? J. Clin. Investig. 1997, 99, 2152–2164. [Google Scholar] [CrossRef]
- Bulmer, J.N.; Innes, B.A.; Levey, J.; Robson, S.C.; Lash, G.E. The role of vascular smooth muscle cell apoptosis and migration during uterine spiral artery remodeling in normal human pregnancy. FASEB J. 2012, 26, 2975–2985. [Google Scholar] [CrossRef]
- Burton, G.J.; Woods, A.W.; Jauniaux, E.; Kingdom, J.C. Rheological and physiological consequences of conversion of the maternal spiral arteries for uteroplacental blood flow during human pregnancy. Placenta 2009, 30, 473–482. [Google Scholar] [CrossRef]
- Chang, K.; Lubo, Z. Review article: Steroid hormones and uterine vascular adaptation to pregnancy. Reprod. Sci. 2008, 15, 336–348. [Google Scholar] [CrossRef]
- Pastore, M.B.; Jobe, S.O.; Ramadoss, J.; Magness, R.R. Estrogen receptor-alpha and estrogen receptor-beta in the uterine vascular endothelium during pregnancy: Functional implications for regulating uterine blood flow. Semin. Reprod. Med. 2012, 30, 46–61. [Google Scholar] [CrossRef]
- Chatuphonprasert, W.; Jarukamjorn, K.; Ellinger, I. Physiology and Pathophysiology of Steroid Biosynthesis, Transport and Metabolism in the Human Placenta. Front. Pharmacol. 2018, 9, 1027. [Google Scholar] [CrossRef]
- Berkane, N.; Liere, P.; Oudinet, J.P.; Hertig, A.; Lefevre, G.; Pluchino, N.; Schumacher, M.; Chabbert-Buffet, N. From Pregnancy to Preeclampsia: A Key Role for Estrogens. Endocr. Rev. 2017, 38, 123–144. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, C.R.; Morriss, F.H., Jr.; Makowski, E.L.; Meschia, G.; Battaglia, F.C. Circulatory changes in the reproductive tissues of ewes during pregnancy. Gynecol. Investig. 1974, 5, 252–268. [Google Scholar] [CrossRef]
- Magness, R.R.; Phernetton, T.M.; Gibson, T.C.; Chen, D.B. Uterine blood flow responses to ICI 182 780 in ovariectomized oestradiol-17beta-treated, intact follicular and pregnant sheep. J. Physiol. 2005, 565, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Magness, R.R.; Phernetton, T.M.; Zheng, J. Systemic and uterine blood flow distribution during prolonged infusion of 17beta-estradiol. Am. J. Physiol. 1998, 275, H731–H743. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Huang, X.; Yang, S.; Zhang, L. Direct chronic effect of steroid hormones in attenuating uterine arterial myogenic tone: Role of protein kinase c/extracellular signal-regulated kinase 1/2. Hypertension 2009, 54, 352–358. [Google Scholar] [CrossRef]
- Magness, R.R.; Rosenfeld, C.R. Local and systemic estradiol-17 beta: Effects on uterine and systemic vasodilation. Am. J. Physiol. 1989, 256, E536–E542. [Google Scholar] [CrossRef]
- Corcoran, J.J.; Nicholson, C.; Sweeney, M.; Charnock, J.C.; Robson, S.C.; Westwood, M.; Taggart, M.J. Human uterine and placental arteries exhibit tissue-specific acute responses to 17beta-estradiol and estrogen-receptor-specific agonists. Mol. Hum. Reprod. 2014, 20, 433–441. [Google Scholar] [CrossRef]
- Rosenfeld, C.R.; White, R.E.; Roy, T.; Cox, B.E. Calcium-activated potassium channels and nitric oxide coregulate estrogen-induced vasodilation. Am. J. Physiol. Heart Circ. Physiol. 2000, 279, H319–H328. [Google Scholar] [CrossRef]
- Tropea, T.; De Francesco, E.M.; Rigiracciolo, D.; Maggiolini, M.; Wareing, M.; Osol, G.; Mandala, M. Pregnancy Augments G Protein Estrogen Receptor (GPER) Induced Vasodilation in Rat Uterine Arteries via the Nitric Oxide-cGMP Signaling Pathway. PLoS ONE 2015, 10, e0141997. [Google Scholar] [CrossRef]
- Rupnow, H.L.; Phernetton, T.M.; Shaw, C.E.; Modrick, M.L.; Bird, I.M.; Magness, R.R. Endothelial vasodilator production by uterine and systemic arteries. VII. Estrogen and progesterone effects on eNOS. Am. J. Physiol. Heart Circ. Physiol. 2001, 280, H1699–H1705. [Google Scholar] [CrossRef]
- Rosenfeld, C.R.; Chen, C.; Roy, T.; Liu, X. Estrogen selectively up-regulates eNOS and nNOS in reproductive arteries by transcriptional mechanisms. J. Soc. Gynecol. Investig. 2003, 10, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Nagar, D.; Liu, X.T.; Rosenfeld, C.R. Estrogen regulates {beta}1-subunit expression in Ca2+-activated K+ channels in arteries from reproductive tissues. Am. J. Physiol. Heart Circ. Physiol. 2005, 289, H1417–H1427. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.Q.; Xiao, D.; Zhu, R.; Huang, X.; Yang, S.; Wilson, S.; Zhang, L. Pregnancy upregulates large-conductance Ca2+-activated K+ channel activity and attenuates myogenic tone in uterine arteries. Hypertension 2011, 58, 1132–1139. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Dasgupta, C.; Xiong, F.; Zhang, L. Epigenetic upregulation of large-conductance Ca2+-activated K+ channel expression in uterine vascular adaptation to pregnancy. Hypertension 2014, 64, 610–618. [Google Scholar] [CrossRef]
- Lechuga, T.J.; Bilg, A.K.; Patel, B.A.; Nguyen, N.A.; Qi, Q.R.; Chen, D.B. Estradiol-17beta stimulates H2 S biosynthesis by ER-dependent CBS and CSE transcription in uterine artery smooth muscle cells in vitro. J. Cell. Physiol. 2019, 234, 9264–9273. [Google Scholar] [CrossRef]
- Rosenfeld, C.R.; Cox, B.E.; Roy, T.; Magness, R.R. Nitric oxide contributes to estrogen-induced vasodilation of the ovine uterine circulation. J. Clin. Investig. 1996, 98, 2158–2166. [Google Scholar] [CrossRef]
- Kublickiene, K.R.; Kublickas, M.; Lindblom, B.; Lunell, N.O.; Nisell, H. A comparison of myogenic and endothelial properties of myometrial and omental resistance vessels in late pregnancy. Am. J. Obstet. Gynecol. 1997, 176, 560–566. [Google Scholar] [CrossRef]
- Gokina, N.I.; Kuzina, O.Y.; Fuller, R.; Osol, G. Local uteroplacental influences are responsible for the induction of uterine artery myogenic tone during rat pregnancy. Reprod. Sci. 2009, 16, 1072–1081. [Google Scholar] [CrossRef]
- Rosenfeld, C.R. Mechanisms regulating angiotensin II responsiveness by the uteroplacental circulation. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2001, 281, R1025–R1040. [Google Scholar] [CrossRef]
- Weiner, C.P.; Thompson, L.P.; Liu, K.Z.; Herrig, J.E. Pregnancy reduces serotonin-induced contraction of guinea pig uterine and carotid arteries. Am. J. Physiol. 1992, 263, H1764–H1769. [Google Scholar] [CrossRef]
- Weiner, C.; Liu, K.Z.; Thompson, L.; Herrig, J.; Chestnut, D. Effect of pregnancy on endothelium and smooth muscle: Their role in reduced adrenergic sensitivity. Am. J. Physiol. 1991, 261, H1275–H1283. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.; Meyer, M.; Osol, G. Gestation increases nitric oxide-mediated vasodilation in rat uterine arteries. Am. J. Obstet. Gynecol. 1997, 176, 856–864. [Google Scholar] [CrossRef]
- Ni, Y.; May, V.; Braas, K.; Osol, G. Pregnancy augments uteroplacental vascular endothelial growth factor gene expression and vasodilator effects. Am. J. Physiol. 1997, 273, H938–H944. [Google Scholar] [CrossRef] [PubMed]
- Nelson, S.H.; Steinsland, O.S.; Suresh, M.S.; Lee, N.M. Pregnancy augments nitric oxide-dependent dilator response to acetylcholine in the human uterine artery. Hum. Reprod. 1998, 13, 1361–1367. [Google Scholar] [CrossRef]
- Xiao, D.; Pearce, W.J.; Zhang, L. Pregnancy enhances endothelium-dependent relaxation of ovine uterine artery: Role of NO and intracellular Ca2+. Am. J. Physiol. Heart Circ. Physiol. 2001, 281, H183–H190. [Google Scholar] [CrossRef]
- Cooke, C.L.; Davidge, S.T. Pregnancy-induced alterations of vascular function in mouse mesenteric and uterine arteries. Biol. Reprod. 2003, 68, 1072–1077. [Google Scholar] [CrossRef]
- Gokina, N.I.; Kuzina, O.Y.; Vance, A.M. Augmented EDHF signaling in rat uteroplacental vasculature during late pregnancy. Am. J. Physiol. Heart Circ. Physiol. 2010, 299, H1642–H1652. [Google Scholar] [CrossRef]
- Lechuga, T.J.; Qi, Q.R.; Magness, R.R.; Chen, D.B. Ovine uterine artery hydrogen sulfide biosynthesis in vivo: Effects of ovarian cycle and pregnancydagger. Biol. Reprod. 2019, 100, 1630–1636. [Google Scholar] [CrossRef]
- Jaggar, J.H.; Wellman, G.C.; Heppner, T.J.; Porter, V.A.; Perez, G.J.; Gollasch, M.; Kleppisch, T.; Rubart, M.; Stevenson, A.S.; Lederer, W.J.; et al. Ca2+ channels, ryanodine receptors and Ca2+-activated K+ channels: A functional unit for regulating arterial tone. Acta Physiol. Scand. 1998, 164, 577–587. [Google Scholar] [CrossRef]
- Veerareddy, S.; Cooke, C.L.; Baker, P.N.; Davidge, S.T. Vascular adaptations to pregnancy in mice: Effects on myogenic tone. Am. J. Physiol. Heart Circ. Physiol. 2002, 283, H2226–H2233. [Google Scholar] [CrossRef]
- Xiao, D.; Buchholz, J.N.; Zhang, L. Pregnancy attenuates uterine artery pressure-dependent vascular tone: Role of PKC/ERK pathway. Am. J. Physiol. Heart Circ. Physiol. 2006, 290, H2337–H2343. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.Q.; Xiao, D.; Zhu, R.; Huang, X.; Yang, S.; Wilson, S.M.; Zhang, L. Chronic hypoxia suppresses pregnancy-induced upregulation of large-conductance Ca2+-activated K+ channel activity in uterine arteries. Hypertension 2012, 60, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.Q.; Song, R.; Romero, M.; Dasgupta, C.; Huang, X.; Holguin, M.A.; Williams, V.; Xiao, D.; Wilson, S.M.; Zhang, L. Pregnancy Increases Ca2+ Sparks/Spontaneous Transient Outward Currents and Reduces Uterine Arterial Myogenic Tone. Hypertension 2019, 73, 691–702. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.Q.; Dasgupta, C.; Xiao, D.; Huang, X.; Yang, S.; Zhang, L. MicroRNA-210 Targets Ten-Eleven Translocation Methylcytosine Dioxygenase 1 and Suppresses Pregnancy-Mediated Adaptation of Large Conductance Ca2+-Activated K+ Channel Expression and Function in Ovine Uterine Arteries. Hypertension 2017, 70, 601–612. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, C.R.; Cornfield, D.N.; Roy, T. Ca2+-activated K+ channels modulate basal and E(2)beta-induced rises in uterine blood flow in ovine pregnancy. Am. J. Physiol. Heart Circ. Physiol. 2001, 281, H422–H431. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, C.R.; Roy, T.; DeSpain, K.; Cox, B.E. Large-conductance Ca2+-dependent K+ channels regulate basal uteroplacental blood flow in ovine pregnancy. J. Soc. Gynecol. Investig. 2005, 12, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Osol, G.; Cipolla, M. Interaction of myogenic and adrenergic mechanisms in isolated, pressurized uterine radial arteries from late-pregnant and nonpregnant rats. Am. J. Obstet. Gynecol. 1993, 168, 697–705. [Google Scholar] [CrossRef]
- Eckman, D.M.; Gupta, R.; Rosenfeld, C.R.; Morgan, T.M.; Charles, S.M.; Mertz, H.; Moore, L.G. Pregnancy increases myometrial artery myogenic tone via NOS- or COX-independent mechanisms. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2012, 303, R368–R375. [Google Scholar] [CrossRef]
- Brosens, I.A.; Robertson, W.B.; Dixon, H.G. The role of the spiral arteries in the pathogenesis of pre-eclampsia. J. Pathol. 1970, 101, Pvi. [Google Scholar]
- Gerretsen, G.; Huisjes, H.J.; Elema, J.D. Morphological changes of the spiral arteries in the placental bed in relation to pre-eclampsia and fetal growth retardation. Br. J. Obstet. Gynaecol. 1981, 88, 876–881. [Google Scholar] [CrossRef]
- Meekins, J.W.; Pijnenborg, R.; Hanssens, M.; McFadyen, I.R.; van Asshe, A. A study of placental bed spiral arteries and trophoblast invasion in normal and severe pre-eclamptic pregnancies. Br. J. Obstet. Gynaecol. 1994, 101, 669–674. [Google Scholar] [CrossRef] [PubMed]
- Kadyrov, M.; Kingdom, J.C.; Huppertz, B. Divergent trophoblast invasion and apoptosis in placental bed spiral arteries from pregnancies complicated by maternal anemia and early-onset preeclampsia/intrauterine growth restriction. Am. J. Obstet. Gynecol. 2006, 194, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Pijnenborg, R.; Anthony, J.; Davey, D.A.; Rees, A.; Tiltman, A.; Vercruysse, L.; van Assche, A. Placental bed spiral arteries in the hypertensive disorders of pregnancy. Br. J. Obstet. Gynaecol. 1991, 98, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Lyall, F.; Robson, S.C.; Bulmer, J.N. Spiral artery remodeling and trophoblast invasion in preeclampsia and fetal growth restriction: Relationship to clinical outcome. Hypertension 2013, 62, 1046–1054. [Google Scholar] [CrossRef]
- Ong, S.S.; Baker, P.N.; Mayhew, T.M.; Dunn, W.R. Remodeling of myometrial radial arteries in preeclampsia. Am. J. Obstet. Gynecol. 2005, 192, 572–579. [Google Scholar] [CrossRef]
- Van der Heijden, O.W.; Essers, Y.P.; Fazzi, G.; Peeters, L.L.; De Mey, J.G.; van Eys, G.J. Uterine artery remodeling and reproductive performance are impaired in endothelial nitric oxide synthase-deficient mice. Biol. Reprod. 2005, 72, 1161–1168. [Google Scholar] [CrossRef]
- Rennie, M.Y.; Rahman, A.; Whiteley, K.J.; Sled, J.G.; Adamson, S.L. Site-specific increases in utero- and fetoplacental arterial vascular resistance in eNOS-deficient mice due to impaired arterial enlargement. Biol. Reprod. 2015, 92, 48. [Google Scholar] [CrossRef]
- Leiberman, J.R.; Meizner, I.; Mazor, M.; Insler, V. Blood supply to the uterus in preeclampsia. Eur. J. Obstet. Gynecol. Reprod. Biol. 1988, 28, 23–27. [Google Scholar] [CrossRef]
- Lunell, N.O.; Sarby, B.; Lewander, R.; Nylund, L. Comparison of uteroplacental blood flow in normal and in intrauterine growth-retarded pregnancy. Measurements with Indium-113m and a computer-linked gammacamera. Gynecol. Obstet. Investig. 1979, 10, 106–118. [Google Scholar] [CrossRef]
- Lunell, N.O.; Nylund, L.E.; Lewander, R.; Sarby, B. Uteroplacental blood flow in pre-eclampsia measurements with indium-113m and a computer-linked gamma camera. Clin. Exp. Hypertens. Part B 1982, 1, 105–117. [Google Scholar] [CrossRef]
- Takata, M.; Nakatsuka, M.; Kudo, T. Differential blood flow in uterine, ophthalmic, and brachial arteries of preeclamptic women. Obstet. Gynecol. 2002, 100, 931–939. [Google Scholar] [CrossRef] [PubMed]
- Konje, J.C.; Howarth, E.S.; Kaufmann, P.; Taylor, D.J. Longitudinal quantification of uterine artery blood volume flow changes during gestation in pregnancies complicated by intrauterine growth restriction. BJOG 2003, 110, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Napolitano, R.; Rajakulasingam, R.; Memmo, A.; Bhide, A.; Thilaganathan, B. Uterine artery Doppler screening for pre-eclampsia: Comparison of the lower, mean and higher first-trimester pulsatility indices. Ultrasound Obstet. Gynecol. 2011, 37, 534–537. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishnan, K.; Mishra, J.S.; Chinnathambi, V.; Vincent, K.L.; Patrikeev, I.; Motamedi, M.; Saade, G.R.; Hankins, G.D.; Sathishkumar, K. Elevated Testosterone Reduces Uterine Blood Flow, Spiral Artery Elongation, and Placental Oxygenation in Pregnant Rats. Hypertension 2016, 67, 630–639. [Google Scholar] [CrossRef] [PubMed]
- Kublickiene, K.R.; Lindblom, B.; Kruger, K.; Nisell, H. Preeclampsia: Evidence for impaired shear stress-mediated nitric oxide release in uterine circulation. Am. J. Obstet. Gynecol. 2000, 183, 160–166. [Google Scholar] [CrossRef]
- Chang, K.; Xiao, D.; Huang, X.; Xue, Z.; Yang, S.; Longo, L.D.; Zhang, L. Chronic hypoxia inhibits sex steroid hormone-mediated attenuation of ovine uterine arterial myogenic tone in pregnancy. Hypertension 2010, 56, 750–757. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Reho, J.J.; Toot, J.D.; Peck, J.; Novak, J.; Yun, Y.H.; Ramirez, R.J. Increased Myogenic Reactivity of Uterine Arteries from Pregnant Rats with Reduced Uterine Perfusion Pressure. Pregnancy Hypertens. 2012, 2, 106–114. [Google Scholar] [CrossRef]
- Ashworth, J.R.; Warren, A.Y.; Baker, P.N.; Johnson, I.R. Loss of endothelium-dependent relaxation in myometrial resistance arteries in pre-eclampsia. Br. J. Obstet. Gynaecol. 1997, 104, 1152–1158. [Google Scholar] [CrossRef]
- Ong, S.S.; Moore, R.J.; Warren, A.Y.; Crocker, I.P.; Fulford, J.; Tyler, D.J.; Gowland, P.A.; Baker, P.N. Myometrial and placental artery reactivity alone cannot explain reduced placental perfusion in pre-eclampsia and intrauterine growth restriction. BJOG 2003, 110, 909–915. [Google Scholar] [CrossRef]
- Wimalasundera, R.C.; Thom, S.A.; Regan, L.; Hughes, A.D. Effects of vasoactive agents on intracellular calcium and force in myometrial and subcutaneous resistance arteries isolated from preeclamptic, pregnant, and nonpregnant woman. Am. J. Obstet. Gynecol. 2005, 192, 625–632. [Google Scholar] [CrossRef]
- Luksha, L.; Luksha, N.; Kublickas, M.; Nisell, H.; Kublickiene, K. Diverse mechanisms of endothelium-derived hyperpolarizing factor-mediated dilatation in small myometrial arteries in normal human pregnancy and preeclampsia. Biol. Reprod. 2010, 83, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Zamudio, S. High-altitude hypoxia and preeclampsia. Front. Biosci. 2007, 12, 2967–2977. [Google Scholar] [CrossRef] [PubMed]
- Moore, L.G.; Charles, S.M.; Julian, C.G. Humans at high altitude: Hypoxia and fetal growth. Respir. Physiol. Neurobiol. 2011, 178, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Tissot van Patot, M.; Grilli, A.; Chapman, P.; Broad, E.; Tyson, W.; Heller, D.S.; Zwerdlinger, L.; Zamudio, S. Remodelling of uteroplacental arteries is decreased in high altitude placentae. Placenta 2003, 24, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Lorca, R.A.; Lane, S.L.; Bales, E.S.; Nsier, H.; Yi, H.; Donnelly, M.A.; Euser, A.G.; Julian, C.G.; Moore, L.G. High Altitude Reduces NO-Dependent Myometrial Artery Vasodilator Response During Pregnancy. Hypertension 2019, 73, 1319–1326. [Google Scholar] [CrossRef]
- Zamudio, S.; Palmer, S.K.; Droma, T.; Stamm, E.; Coffin, C.; Moore, L.G. Effect of altitude on uterine artery blood flow during normal pregnancy. J. Appl. Physiol. 1995, 79, 7–14. [Google Scholar] [CrossRef]
- Julian, C.G.; Galan, H.L.; Wilson, M.J.; Desilva, W.; Cioffi-Ragan, D.; Schwartz, J.; Moore, L.G. Lower uterine artery blood flow and higher endothelin relative to nitric oxide metabolite levels are associated with reductions in birth weight at high altitude. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 295, R906–R915. [Google Scholar] [CrossRef]
- Anderson, C.M.; Lopez, F.; Zhang, H.Y.; Pavlish, K.; Benoit, J.N. Reduced uteroplacental perfusion alters uterine arcuate artery function in the pregnant Sprague-Dawley rat. Biol. Reprod. 2005, 72, 762–766. [Google Scholar] [CrossRef]
- Fitzgerald, J.S.; Busch, S.; Wengenmayer, T.; Foerster, K.; de la Motte, T.; Poehlmann, T.G.; Markert, U.R. Signal transduction in trophoblast invasion. Chem. Immunol. Allergy 2005, 88, 181–199. [Google Scholar] [CrossRef]
- Gupta, S.K.; Malhotra, S.S.; Malik, A.; Verma, S.; Chaudhary, P. Cell Signaling Pathways Involved During Invasion and Syncytialization of Trophoblast Cells. Am. J. Reprod. Immunol. 2016, 75, 361–371. [Google Scholar] [CrossRef]
- Morrison, D.K. MAP kinase pathways. Cold Spring Harb. Perspect. Biol. 2012, 4. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Q.; Yang, M.; Tsang, B.K.; Gruslin, A. Both mitogen-activated protein kinase and phosphatidylinositol 3-kinase signalling are required in epidermal growth factor-induced human trophoblast migration. Mol. Hum. Reprod. 2004, 10, 677–684. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Malik, A.; Pal, R.; Gupta, S.K. Interdependence of JAK-STAT and MAPK signaling pathways during EGF-mediated HTR-8/SVneo cell invasion. PLoS ONE 2017, 12, e0178269. [Google Scholar] [CrossRef] [PubMed]
- Suman, P.; Gupta, S.K. STAT3 and ERK1/2 cross-talk in leukaemia inhibitory factor mediated trophoblastic JEG-3 cell invasion and expression of mucin 1 and Fos. Am. J. Reprod. Immunol. 2014, 72, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Appel, S.; Ankerne, J.; Appel, J.; Oberthuer, A.; Mallmann, P.; Dotsch, J. CNN3 regulates trophoblast invasion and is upregulated by hypoxia in BeWo cells. PLoS ONE 2014, 9, e103216. [Google Scholar] [CrossRef] [PubMed]
- LaMarca, H.L.; Dash, P.R.; Vishnuthevan, K.; Harvey, E.; Sullivan, D.E.; Morris, C.A.; Whitley, G.S. Epidermal growth factor-stimulated extravillous cytotrophoblast motility is mediated by the activation of PI3-K, Akt and both p38 and p42/44 mitogen-activated protein kinases. Hum. Reprod. 2008, 23, 1733–1741. [Google Scholar] [CrossRef]
- Cartwright, J.E.; Tse, W.K.; Whitley, G.S. Hepatocyte growth factor induced human trophoblast motility involves phosphatidylinositol-3-kinase, mitogen-activated protein kinase, and inducible nitric oxide synthase. Exp. Cell Res. 2002, 279, 219–226. [Google Scholar] [CrossRef]
- Moon, K.C.; Park, J.S.; Norwitz, E.R.; Kim, D.I.; Oh, K.J.; Park, C.W.; Jun, J.K.; Syn, H.C. Expression of extracellular signal-regulated kinase1/2 and p38 mitogen-activated protein kinase in the invasive trophoblasts at the human placental bed. Placenta 2008, 29, 391–395. [Google Scholar] [CrossRef]
- Wang, Y.; Cheng, K.; Zhou, W.; Liu, H.; Yang, T.; Hou, P.; Li, X. miR-141-5p regulate ATF2 via effecting MAPK1/ERK2 signaling to promote preeclampsia. Biomed. Pharmacother. 2019, 115, 108953. [Google Scholar] [CrossRef]
- Webster, R.P.; Brockman, D.; Myatt, L. Nitration of p38 MAPK in the placenta: Association of nitration with reduced catalytic activity of p38 MAPK in pre-eclampsia. Mol. Hum. Reprod. 2006, 12, 677–685. [Google Scholar] [CrossRef]
- Yang, Q.E.; Giassetti, M.I.; Ealy, A.D. Fibroblast growth factors activate mitogen-activated protein kinase pathways to promote migration in ovine trophoblast cells. Reproduction 2011, 141, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Mendelson, K.; Evans, T.; Hla, T. Sphingosine 1-phosphate signalling. Development 2014, 141, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Li, Q.; Pan, Z. Sphingosine-1-phosphate promotes extravillous trophoblast cell invasion by activating MEK/ERK/MMP-2 signaling pathways via S1P/S1PR1 axis activation. PLoS ONE 2014, 9, e106725. [Google Scholar] [CrossRef] [PubMed]
- Anton, L.; Olarerin-George, A.O.; Schwartz, N.; Srinivas, S.; Bastek, J.; Hogenesch, J.B.; Elovitz, M.A. miR-210 inhibits trophoblast invasion and is a serum biomarker for preeclampsia. Am. J. Pathol. 2013, 183, 1437–1445. [Google Scholar] [CrossRef]
- Hemmings, B.A.; Restuccia, D.F. PI3K-PKB/Akt pathway. Cold Spring Harb. Perspect. Biol. 2012, 4, a011189. [Google Scholar] [CrossRef]
- Tamguney, T.; Stokoe, D. New insights into PTEN. J. Cell Sci. 2007, 120, 4071–4079. [Google Scholar] [CrossRef]
- Kent, L.N.; Rumi, M.A.; Kubota, K.; Lee, D.S.; Soares, M.J. FOSL1 is integral to establishing the maternal-fetal interface. Mol. Cell. Biol. 2011, 31, 4801–4813. [Google Scholar] [CrossRef]
- Xu, Y.; Sui, L.; Qiu, B.; Yin, X.; Liu, J.; Zhang, X. ANXA4 promotes trophoblast invasion via the PI3K/Akt/eNOS pathway in preeclampsia. Am. J. Physiol. Cell Physiol. 2019, 316, C481–C491. [Google Scholar] [CrossRef]
- Budi, E.H.; Duan, D.; Derynck, R. Transforming Growth Factor-beta Receptors and Smads: Regulatory Complexity and Functional Versatility. Trends Cell Biol. 2017, 27, 658–672. [Google Scholar] [CrossRef]
- Feng, X.H.; Derynck, R. Specificity and versatility in tgf-beta signaling through Smads. Annu. Rev. Cell Dev. Biol. 2005, 21, 659–693. [Google Scholar] [CrossRef]
- Lash, G.E.; Otun, H.A.; Innes, B.A.; Bulmer, J.N.; Searle, R.F.; Robson, S.C. Inhibition of trophoblast cell invasion by TGFB1, 2, and 3 is associated with a decrease in active proteases. Biol. Reprod. 2005, 73, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.R.; Qiu, W.; Li, Y.X.; Zhang, Z.B.; Li, D.; Wang, Y.L. Dual effect of transforming growth factor beta1 on cell adhesion and invasion in human placenta trophoblast cells. Reproduction 2006, 132, 333–341. [Google Scholar] [CrossRef] [PubMed]
- Nadeem, L.; Munir, S.; Fu, G.; Dunk, C.; Baczyk, D.; Caniggia, I.; Lye, S.; Peng, C. Nodal signals through activin receptor-like kinase 7 to inhibit trophoblast migration and invasion: Implication in the pathogenesis of preeclampsia. Am. J. Pathol. 2011, 178, 1177–1189. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.C.; Chang, H.M.; Leung, P.C. Transforming growth factor-beta1 inhibits trophoblast cell invasion by inducing Snail-mediated down-regulation of vascular endothelial-cadherin protein. J. Biol. Chem. 2013, 288, 33181–33192. [Google Scholar] [CrossRef]
- Chauvin, S.; Yinon, Y.; Xu, J.; Ermini, L.; Sallais, J.; Tagliaferro, A.; Todros, T.; Post, M.; Caniggia, I. Aberrant TGFbeta Signalling Contributes to Dysregulation of Sphingolipid Metabolism in Intrauterine Growth Restriction. J. Clin. Endocrinol. Metab. 2015, 100, E986–E996. [Google Scholar] [CrossRef]
- Wu, H.; Wang, H.; Liu, M.; Bai, Y.; Li, Y.X.; Ji, L.; Peng, C.; Yu, Y.; Wang, Y.L. MiR-195 participates in the placental disorder of preeclampsia via targeting activin receptor type-2B in trophoblastic cells. J. Hypertens. 2016, 34, 1371–1379. [Google Scholar] [CrossRef]
- Mano, Y.; Kotani, T.; Shibata, K.; Matsumura, H.; Tsuda, H.; Sumigama, S.; Yamamoto, E.; Iwase, A.; Senga, T.; Kikkawa, F. The loss of endoglin promotes the invasion of extravillous trophoblasts. Endocrinology 2011, 152, 4386–4394. [Google Scholar] [CrossRef]
- Caniggia, I.; Taylor, C.V.; Ritchie, J.W.; Lye, S.J.; Letarte, M. Endoglin regulates trophoblast differentiation along the invasive pathway in human placental villous explants. Endocrinology 1997, 138, 4977–4988. [Google Scholar] [CrossRef]
- Gu, Y.; Lewis, D.F.; Wang, Y. Placental productions and expressions of soluble endoglin, soluble fms-like tyrosine kinase receptor-1, and placental growth factor in normal and preeclamptic pregnancies. J. Clin. Endocrinol. Metab. 2008, 93, 260–266. [Google Scholar] [CrossRef]
- Angers, S.; Moon, R.T. Proximal events in Wnt signal transduction. Nat. Rev. Mol. Cell Biol. 2009, 10, 468–477. [Google Scholar] [CrossRef]
- Pollheimer, J.; Loregger, T.; Sonderegger, S.; Saleh, L.; Bauer, S.; Bilban, M.; Czerwenka, K.; Husslein, P.; Knofler, M. Activation of the canonical wingless/T-cell factor signaling pathway promotes invasive differentiation of human trophoblast. Am. J. Pathol. 2006, 168, 1134–1147. [Google Scholar] [CrossRef] [PubMed]
- Knofler, M.; Pollheimer, J. Human placental trophoblast invasion and differentiation: A particular focus on Wnt signaling. Front. Genet. 2013, 4, 190. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, H.; Zhang, L.; Jia, L.; Wang, P. Differential expression of beta-catenin and Dickkopf-1 in the third trimester placentas from normal and preeclamptic pregnancies: A comparative study. Reprod. Biol. Endocrinol. 2013, 11, 17. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhang, Z.; Zeng, X.; Wang, J.; Zhang, L.; Song, W.; Shi, Y. Wnt/beta-catenin signaling pathway in severe preeclampsia. J. Mol. Histol. 2018, 49, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Zhang, Y.; Yang, P.; Gao, X.; Wang, Y. Downregulated low-density lipoprotein receptor-related protein 6 induces the maldevelopment of extravillous trophoblast via Wnt/beta-catenin signaling pathway. Mol. Cell. Probes 2019, 44, 21–28. [Google Scholar] [CrossRef]
- Zhang, Y.; Pan, X.; Yu, X.; Li, L.; Qu, H.; Li, S. MicroRNA-590-3p inhibits trophoblast-dependent maternal spiral artery remodeling by repressing low-density lipoprotein receptor-related protein 6. Mol. Genet. Genom. Med. 2018, 6, 1124–1133. [Google Scholar] [CrossRef]
- Kopan, R. Notch signaling. Cold Spring Harb. Perspect. Biol. 2012, 4. [Google Scholar] [CrossRef]
- Zhao, W.X.; Lin, J.H. Notch signaling pathway and human placenta. Int. J. Med. Sci. 2012, 9, 447–452. [Google Scholar] [CrossRef]
- Haider, S.; Meinhardt, G.; Saleh, L.; Fiala, C.; Pollheimer, J.; Knofler, M. Notch1 controls development of the extravillous trophoblast lineage in the human placenta. Proc. Natl. Acad. Sci. USA 2016, 113, E7710–E7719. [Google Scholar] [CrossRef]
- Cobellis, L.; Mastrogiacomo, A.; Federico, E.; Schettino, M.T.; De Falco, M.; Manente, L.; Coppola, G.; Torella, M.; Colacurci, N.; De Luca, A. Distribution of Notch protein members in normal and preeclampsia-complicated placentas. Cell Tissue Res. 2007, 330, 527–534. [Google Scholar] [CrossRef]
- Zhao, W.X.; Huang, T.T.; Jiang, M.; Feng, R.; Lin, J.H. Expression of notch family proteins in placentas from patients with early-onset severe preeclampsia. Reprod. Sci. 2014, 21, 716–723. [Google Scholar] [CrossRef] [PubMed]
- Fragkiadaki, P.; Soulitzis, N.; Sifakis, S.; Koutroulakis, D.; Gourvas, V.; Vrachnis, N.; Spandidos, D.A. Downregulation of notch signaling pathway in late preterm and term placentas from pregnancies complicated by preeclampsia. PLoS ONE 2015, 10, e0126163. [Google Scholar] [CrossRef] [PubMed]
- Shimanuki, Y.; Mitomi, H.; Fukumura, Y.; Makino, S.; Itakura, A.; Yao, T.; Takeda, S. Alteration of Delta-like ligand 1 and Notch 1 receptor in various placental disorders with special reference to early onset preeclampsia. Hum. Pathol. 2015, 46, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.J.; Zhang, L.; Zhang, F.F.; Luan, T.; Yin, Z.M.; Rui, C.; Ding, H.J. Influence of miR-34a on preeclampsia through the Notch signaling pathway. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 923–931. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.X.; Wu, Z.M.; Liu, W.; Lin, J.H. Notch2 and Notch3 suppress the proliferation and mediate invasion of trophoblast cell lines. Biol. Open 2017, 6, 1123–1129. [Google Scholar] [CrossRef] [PubMed]
- Doridot, L.; Houry, D.; Gaillard, H.; Chelbi, S.T.; Barbaux, S.; Vaiman, D. miR-34a expression, epigenetic regulation, and function in human placental diseases. Epigenetics 2014, 9, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Pang, R.T.; Leung, C.O.; Ye, T.M.; Liu, W.; Chiu, P.C.; Lam, K.K.; Lee, K.F.; Yeung, W.S. MicroRNA-34a suppresses invasion through downregulation of Notch1 and Jagged1 in cervical carcinoma and choriocarcinoma cells. Carcinogenesis 2010, 31, 1037–1044. [Google Scholar] [CrossRef]
- Barquilla, A.; Pasquale, E.B. Eph receptors and ephrins: Therapeutic opportunities. Annu. Rev. Pharmacol. Toxicol. 2015, 55, 465–487. [Google Scholar] [CrossRef]
- Zeng, X.; Hunt, A.; Jin, S.C.; Duran, D.; Gaillard, J.; Kahle, K.T. EphrinB2-EphB4-RASA1 Signaling in Human Cerebrovascular Development and Disease. Trends Mol. Med. 2019, 25, 265–286. [Google Scholar] [CrossRef]
- Dong, H.; Yu, C.; Mu, J.; Zhang, J.; Lin, W. Role of EFNB2/EPHB4 signaling in spiral artery development during pregnancy: An appraisal. Mol. Reprod. Dev. 2016, 83, 12–18. [Google Scholar] [CrossRef]
- Liu, X.; Hu, Y.; Zheng, Y.; Liu, X.; Luo, M.; Liu, W.; Zhao, Y.; Zou, L. EPHB4 Regulates Human Trophoblast Cell Line HTR-8/SVneo Function: Implications for the Role of EPHB4 in Preeclampsia. Biol. Reprod. 2016, 95, 65. [Google Scholar] [CrossRef] [PubMed]
- Red-Horse, K.; Kapidzic, M.; Zhou, Y.; Feng, K.T.; Singh, H.; Fisher, S.J. EPHB4 regulates chemokine-evoked trophoblast responses: A mechanism for incorporating the human placenta into the maternal circulation. Development 2005, 132, 4097–4106. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liu, X.; Liu, W.; Luo, M.; Tao, H.; Wu, D.; Zhao, Y.; Zou, L. HOXA9 transcriptionally regulates the EPHB4 receptor to modulate trophoblast migration and invasion. Placenta 2017, 51, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Fei, M.; Xue, G.; Zhou, Q.; Jia, Y.; Li, L.; Xin, H.; Sun, S. Elevated levels of hypoxia-inducible microRNA-210 in pre-eclampsia: New insights into molecular mechanisms for the disease. J. Cell. Mol. Med. 2012, 16, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Siddle, K. Signalling by insulin and IGF receptors: Supporting acts and new players. J. Mol. Endocrinol. 2011, 47, R1–R10. [Google Scholar] [CrossRef] [PubMed]
- Forbes, K.; Westwood, M.; Baker, P.N.; Aplin, J.D. Insulin-like growth factor I and II regulate the life cycle of trophoblast in the developing human placenta. Am. J. Physiol. Cell Physiol. 2008, 294, C1313–C1322. [Google Scholar] [CrossRef] [PubMed]
- Hiden, U.; Glitzner, E.; Hartmann, M.; Desoye, G. Insulin and the IGF system in the human placenta of normal and diabetic pregnancies. J. Anat. 2009, 215, 60–68. [Google Scholar] [CrossRef]
- Dubova, E.A.; Pavlov, K.A.; Lyapin, V.M.; Kulikova, G.V.; Shchyogolev, A.I.; Sukhikh, G.T. Expression of insulin-like growth factors in the placenta in preeclampsia. Bull. Exp. Biol. Med. 2014, 157, 103–107. [Google Scholar] [CrossRef]
- Nawathe, A.R.; Christian, M.; Kim, S.H.; Johnson, M.; Savvidou, M.D.; Terzidou, V. Insulin-like growth factor axis in pregnancies affected by fetal growth disorders. Clin. Epigenet. 2016, 8, 11. [Google Scholar] [CrossRef]
- Cohen, M.; Meisser, A.; Bischof, P. Metalloproteinases and human placental invasiveness. Placenta 2006, 27, 783–793. [Google Scholar] [CrossRef]
- Isaka, K.; Usuda, S.; Ito, H.; Sagawa, Y.; Nakamura, H.; Nishi, H.; Suzuki, Y.; Li, Y.F.; Takayama, M. Expression and activity of matrix metalloproteinase 2 and 9 in human trophoblasts. Placenta 2003, 24, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Staun-Ram, E.; Goldman, S.; Gabarin, D.; Shalev, E. Expression and importance of matrix metalloproteinase 2 and 9 (MMP-2 and -9) in human trophoblast invasion. Reprod. Biol. Endocrinol. 2004, 2, 59. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Zhong, M.; Pang, Z.; Yu, Y. Dysregulated expression of matrix metalloproteinases and their inhibitors may participate in the pathogenesis of pre-eclampsia and fetal growth restriction. Early Hum. Dev. 2014, 90, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Plaks, V.; Rinkenberger, J.; Dai, J.; Flannery, M.; Sund, M.; Kanasaki, K.; Ni, W.; Kalluri, R.; Werb, Z. Matrix metalloproteinase-9 deficiency phenocopies features of preeclampsia and intrauterine growth restriction. Proc. Natl. Acad. Sci. USA 2013, 110, 11109–11114. [Google Scholar] [CrossRef]
- Jin, M.; Li, H.; Xu, H.; Huo, G.; Yao, Y. MicroRNA-20b inhibits trophoblast cell migration and invasion by targeting MMP-2. Int. J. Clin. Exp. Pathol. 2017, 10, 10901–10909. [Google Scholar]
- Yu, Y.; Wang, L.; Liu, T.; Guan, H. MicroRNA-204 suppresses trophoblast-like cell invasion by targeting matrix metalloproteinase-9. Biochem. Biophys. Res. Commun. 2015, 463, 285–291. [Google Scholar] [CrossRef]
- Su, M.T.; Tsai, P.Y.; Tsai, H.L.; Chen, Y.C.; Kuo, P.L. miR-346 and miR-582-3p-regulated EG-VEGF expression and trophoblast invasion via matrix metalloproteinases 2 and 9. Biofactors 2017, 43, 210–219. [Google Scholar] [CrossRef]
- Zhang, H.; Hou, L.; Li, C.M.; Zhang, W.Y. The chemokine CXCL6 restricts human trophoblast cell migration and invasion by suppressing MMP-2 activity in the first trimester. Hum. Reprod. 2013, 28, 2350–2362. [Google Scholar] [CrossRef]
- Xie, L.; Mouillet, J.F.; Chu, T.; Parks, W.T.; Sadovsky, E.; Knofler, M.; Sadovsky, Y. C19MC microRNAs regulate the migration of human trophoblasts. Endocrinology 2014, 155, 4975–4985. [Google Scholar] [CrossRef]
- Wu, D.; Chen, X.; Wang, L.; Chen, F.; Cen, H.; Shi, L. Hypoxia-induced microRNA-141 regulates trophoblast apoptosis, invasion, and vascularization by blocking CXCL12beta/CXCR2/4 signal transduction. Biomed. Pharmacother. 2019, 116, 108836. [Google Scholar] [CrossRef]
- Qiu, Q.; Yang, M.; Tsang, B.K.; Gruslin, A. EGF-induced trophoblast secretion of MMP-9 and TIMP-1 involves activation of both PI3K and MAPK signalling pathways. Reproduction 2004, 128, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Klausen, C.; Zhu, H.; Leung, P.C. Activin A Increases Human Trophoblast Invasion by Inducing SNAIL-Mediated MMP2 Up-Regulation Through ALK4. J. Clin. Endocrinol. Metab. 2015, 100, E1415–E1427. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.B.; Zheng, J. Regulation of placental angiogenesis. Microcirculation 2014, 21, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Lash, G.E.; Cartwright, J.E.; Whitley, G.S.; Trew, A.J.; Baker, P.N. The effects of angiogenic growth factors on extravillous trophoblast invasion and motility. Placenta 1999, 20, 661–667. [Google Scholar] [CrossRef] [PubMed]
- Lash, G.E.; Warren, A.Y.; Underwood, S.; Baker, P.N. Vascular endothelial growth factor is a chemoattractant for trophoblast cells. Placenta 2003, 24, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Yang, J. Expression and significance of miR155 and vascular endothelial growth factor in placenta of rats with preeclampsia. Int. J. Clin. Exp. Med. 2015, 8, 15731–15737. [Google Scholar] [PubMed]
- Lyall, F.; Young, A.; Boswell, F.; Kingdom, J.C.; Greer, I.A. Placental expression of vascular endothelial growth factor in placentae from pregnancies complicated by pre-eclampsia and intrauterine growth restriction does not support placental hypoxia at delivery. Placenta 1997, 18, 269–276. [Google Scholar] [CrossRef]
- Zhou, Y.; McMaster, M.; Woo, K.; Janatpour, M.; Perry, J.; Karpanen, T.; Alitalo, K.; Damsky, C.; Fisher, S.J. Vascular endothelial growth factor ligands and receptors that regulate human cytotrophoblast survival are dysregulated in severe preeclampsia and hemolysis, elevated liver enzymes, and low platelets syndrome. Am. J. Pathol. 2002, 160, 1405–1423. [Google Scholar] [CrossRef]
- Nanaev, A.; Chwalisz, K.; Frank, H.G.; Kohnen, G.; Hegele-Hartung, C.; Kaufmann, P. Physiological dilation of uteroplacental arteries in the guinea pig depends on nitric oxide synthase activity of extravillous trophoblast. Cell Tissue Res. 1995, 282, 407–421. [Google Scholar] [CrossRef]
- Martin, D.; Conrad, K.P. Expression of endothelial nitric oxide synthase by extravillous trophoblast cells in the human placenta. Placenta 2000, 21, 23–31. [Google Scholar] [CrossRef]
- Cartwright, J.E.; Holden, D.P.; Whitley, G.S. Hepatocyte growth factor regulates human trophoblast motility and invasion: A role for nitric oxide. Br. J. Pharmacol. 1999, 128, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Kulandavelu, S.; Whiteley, K.J.; Qu, D.; Mu, J.; Bainbridge, S.A.; Adamson, S.L. Endothelial nitric oxide synthase deficiency reduces uterine blood flow, spiral artery elongation, and placental oxygenation in pregnant mice. Hypertension 2012, 60, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Poehlmann, T.G.; Fitzgerald, J.S.; Meissner, A.; Wengenmayer, T.; Schleussner, E.; Friedrich, K.; Markert, U.R. Trophoblast invasion: Tuning through LIF, signalling via Stat3. Placenta 2005, 26, S37–S41. [Google Scholar] [CrossRef] [PubMed]
- Meng, H.X.; Xu, L.N.; Jing, G.; Qian, L.; Qi, M.G. MiR-223 promotes trophoblast cell survival and invasion by targeting STAT3 in preeclampsia. Int. J. Clin. Exp. Med. 2017, 10, 6577–6585. [Google Scholar]
- Zhu, X.; Yang, Y.; Han, T.; Yin, G.; Gao, P.; Ni, Y.; Su, X.; Liu, Y.; Yao, Y. Suppression of microRNA-18a expression inhibits invasion and promotes apoptosis of human trophoblast cells by targeting the estrogen receptor alpha gene. Mol. Med. Rep. 2015, 12, 2701–2706. [Google Scholar] [CrossRef]
- Bonagura, T.W.; Pepe, G.J.; Enders, A.C.; Albrecht, E.D. Suppression of extravillous trophoblast vascular endothelial growth factor expression and uterine spiral artery invasion by estrogen during early baboon pregnancy. Endocrinology 2008, 149, 5078–5087. [Google Scholar] [CrossRef]
- Rahl, P.B.; Young, R.A. MYC and transcription elongation. Cold Spring Harb. Perspect. Med. 2014, 4, a020990. [Google Scholar] [CrossRef]
- Butt, E.; Raman, D. New Frontiers for the Cytoskeletal Protein LASP1. Front. Oncol. 2018, 8, 391. [Google Scholar] [CrossRef]
- Zou, A.X.; Chen, B.; Li, Q.X.; Liang, Y.C. MiR-134 inhibits infiltration of trophoblast cells in placenta of patients with preeclampsia by decreasing ITGB1 expression. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 2199–2206. [Google Scholar] [CrossRef]
- Poidatz, D.; Dos Santos, E.; Brule, A.; De Mazancourt, P.; Dieudonne, M.N. Estrogen-related receptor gamma modulates energy metabolism target genes in human trophoblast. Placenta 2012, 33, 688–695. [Google Scholar] [CrossRef]
- Dai, Y.; Qiu, Z.; Diao, Z.; Shen, L.; Xue, P.; Sun, H.; Hu, Y. MicroRNA-155 inhibits proliferation and migration of human extravillous trophoblast derived HTR-8/SVneo cells via down-regulating cyclin D1. Placenta 2012, 33, 824–829. [Google Scholar] [CrossRef] [PubMed]
- Streit, M.; Riccardi, L.; Velasco, P.; Brown, L.F.; Hawighorst, T.; Bornstein, P.; Detmar, M. Thrombospondin-2: A potent endogenous inhibitor of tumor growth and angiogenesis. Proc. Natl. Acad. Sci. USA 1999, 96, 14888–14893. [Google Scholar] [CrossRef] [PubMed]
- Naicker, T.; Dorsamy, E.; Ramsuran, D.; Burton, G.J.; Moodley, J. The role of apoptosis on trophoblast cell invasion in the placental bed of normotensive and preeclamptic pregnancies. Hypertens. Pregnancy 2013, 32, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Hertig, A.; Liere, P.; Chabbert-Buffet, N.; Fort, J.; Pianos, A.; Eychenne, B.; Cambourg, A.; Schumacher, M.; Berkane, N.; Lefevre, G.; et al. Steroid profiling in preeclamptic women: Evidence for aromatase deficiency. Am. J. Obstet. Gynecol. 2010, 203, e471–e479. [Google Scholar] [CrossRef] [PubMed]
- Perez-Sepulveda, A.; Monteiro, L.J.; Dobierzewska, A.; Espana-Perrot, P.P.; Venegas-Araneda, P.; Guzman-Rojas, A.M.; Gonzalez, M.I.; Palominos-Rivera, M.; Irarrazabal, C.E.; Figueroa-Diesel, H.; et al. Placental Aromatase Is Deficient in Placental Ischemia and Preeclampsia. PLoS ONE 2015, 10, e0139682. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Huang, L.; He, Z.; Zou, Z.; Luo, Y. Estrogen-related receptor gamma regulates expression of 17beta-hydroxysteroid dehydrogenase type 1 in fetal growth restriction. Placenta 2018, 67, 38–44. [Google Scholar] [CrossRef]
- He, M.; Li, F.; Yang, M.; Fan, Y.; Beejadhursing, R.; Xie, Y.; Zhou, Y.; Deng, D. Impairment of BKca channels in human placental chorionic plate arteries is potentially relevant to the development of preeclampsia. Hypertens. Res. 2018, 41, 126–134. [Google Scholar] [CrossRef]
- Chang, K.; Xiao, D.; Huang, X.; Longo, L.D.; Zhang, L. Chronic hypoxia increases pressure-dependent myogenic tone of the uterine artery in pregnant sheep: Role of ERK/PKC pathway. Am. J. Physiol. Heart Circ. Physiol. 2009, 296, H1840–H1849. [Google Scholar] [CrossRef]
- Wang, K.; Ahmad, S.; Cai, M.; Rennie, J.; Fujisawa, T.; Crispi, F.; Baily, J.; Miller, M.R.; Cudmore, M.; Hadoke, P.W.; et al. Dysregulation of hydrogen sulfide producing enzyme cystathionine gamma-lyase contributes to maternal hypertension and placental abnormalities in preeclampsia. Circulation 2013, 127, 2514–2522. [Google Scholar] [CrossRef]
- Hu, X.Q.; Song, R.; Zhang, L. Effect of Oxidative Stress on the Estrogen-NOS-NO-KCa Channel Pathway in Uteroplacental Dysfunction: Its Implication in Pregnancy Complications. Oxid. Med. Cell. Longev. 2019, 2019, 9194269. [Google Scholar] [CrossRef]
- Li, F.F.; He, M.Z.; Xie, Y.; Wu, Y.Y.; Yang, M.T.; Fan, Y.; Qiao, F.Y.; Deng, D.R. Involvement of dysregulated IKCa and SKCa channels in preeclampsia. Placenta 2017, 58, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Bird, I.M.; Magness, R.R.; Longo, L.D.; Zhang, L. Upregulation of eNOS in pregnant ovine uterine arteries by chronic hypoxia. Am. J. Physiol. Heart Circ. Physiol. 2001, 280, H812–H820. [Google Scholar] [CrossRef] [PubMed]
- Schiessl, B.; Strasburger, C.; Bidlingmaier, M.; Mylonas, I.; Jeschke, U.; Kainer, F.; Friese, K. Plasma- and urine concentrations of nitrite/nitrate and cyclic Guanosinemonophosphate in intrauterine growth restricted and preeclamptic pregnancies. Arch. Gynecol. Obstet. 2006, 274, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Krause, B.J.; Carrasco-Wong, I.; Caniuguir, A.; Carvajal, J.; Farias, M.; Casanello, P. Endothelial eNOS/arginase imbalance contributes to vascular dysfunction in IUGR umbilical and placental vessels. Placenta 2013, 34, 20–28. [Google Scholar] [CrossRef]
- Jobe, S.O.; Tyler, C.T.; Magness, R.R. Aberrant synthesis, metabolism, and plasma accumulation of circulating estrogens and estrogen metabolites in preeclampsia implications for vascular dysfunction. Hypertension 2013, 61, 480–487. [Google Scholar] [CrossRef]
- Pecks, U.; Rath, W.; Kleine-Eggebrecht, N.; Maass, N.; Voigt, F.; Goecke, T.W.; Mohaupt, M.G.; Escher, G. Maternal Serum Lipid, Estradiol, and Progesterone Levels in Pregnancy, and the Impact of Placental and Hepatic Pathologies. Geburtshilfe Frauenheilkd. 2016, 76, 799–808. [Google Scholar] [CrossRef]
- Ouyang, Y.; Mouillet, J.F.; Coyne, C.B.; Sadovsky, Y. Review: Placenta-specific microRNAs in exosomes—Good things come in nano-packages. Placenta 2014, 35, S69–S73. [Google Scholar] [CrossRef]
- Cronqvist, T.; Tannetta, D.; Morgelin, M.; Belting, M.; Sargent, I.; Familari, M.; Hansson, S.R. Syncytiotrophoblast derived extracellular vesicles transfer functional placental miRNAs to primary human endothelial cells. Sci. Rep. 2017, 7, 4558. [Google Scholar] [CrossRef]
- Shao, X.; Liu, Y.; Liu, M.; Wang, Y.; Yan, L.; Wang, H.; Ma, L.; Li, Y.X.; Zhao, Y.; Wang, Y.L. Testosterone Represses Estrogen Signaling by Upregulating miR-22: A Mechanism for Imbalanced Steroid Hormone Production in Preeclampsia. Hypertension 2017, 69, 721–730. [Google Scholar] [CrossRef]
- Berkane, N.; Liere, P.; Lefevre, G.; Alfaidy, N.; Nahed, R.A.; Vincent, J.; Oudinet, J.P.; Pianos, A.; Cambourg, A.; Rozenberg, P.; et al. Abnormal steroidogenesis and aromatase activity in preeclampsia. Placenta 2018, 69, 40–49. [Google Scholar] [CrossRef]
- Hu, X.Q.; Dasgupta, C.; Chen, M.; Xiao, D.; Huang, X.; Han, L.; Yang, S.; Xu, Z.; Zhang, L. Pregnancy Reprograms Large-Conductance Ca2+-Activated K+ Channel in Uterine Arteries: Roles of Ten-Eleven Translocation Methylcytosine Dioxygenase 1-Mediated Active Demethylation. Hypertension 2017, 69, 1181–1191. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, C.; Chen, M.; Zhang, H.; Yang, S.; Zhang, L. Chronic hypoxia during gestation causes epigenetic repression of the estrogen receptor-alpha gene in ovine uterine arteries via heightened promoter methylation. Hypertension 2012, 60, 697–704. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Park, M.N.; Park, K.H.; Lee, J.E.; Shin, Y.Y.; An, S.M.; Kang, S.S.; Cho, W.S.; An, B.S.; Kim, S.C. The expression and activation of sex steroid receptors in the preeclamptic placenta. Int. J. Mol. Med. 2018, 41, 2943–2951. [Google Scholar] [CrossRef]
- Babicheva, A.; Ayon, R.J.; Makino, A.; Yuan, J.X.J. Increased expression of microRNA-29b attenuates function of Ca2+-activated K+ channels in human PASMC from idiopathic PAH patients. FASEB J. 2018, 32, lb1–lb925. [Google Scholar]
- Tu, J.; Ng, S.H.; Luk, A.C.; Liao, J.; Jiang, X.; Feng, B.; Lun Mak, K.K.; Rennert, O.M.; Chan, W.Y.; Lee, T.L. MicroRNA-29b/Tet1 regulatory axis epigenetically modulates mesendoderm differentiation in mouse embryonic stem cells. Nucleic Acids Res. 2015, 43, 7805–7822. [Google Scholar] [CrossRef]
- Yang, G.; Pei, Y.; Cao, Q.; Wang, R. MicroRNA-21 represses human cystathionine gamma-lyase expression by targeting at specificity protein-1 in smooth muscle cells. J. Cell. Physiol. 2012, 227, 3192–3200. [Google Scholar] [CrossRef]
- Lasabova, Z.; Vazan, M.; Zibolenova, J.; Svecova, I. Overexpression of miR-21 and miR-122 in preeclamptic placentas. Neuroendocrinol. Lett. 2015, 36, 695–699. [Google Scholar]

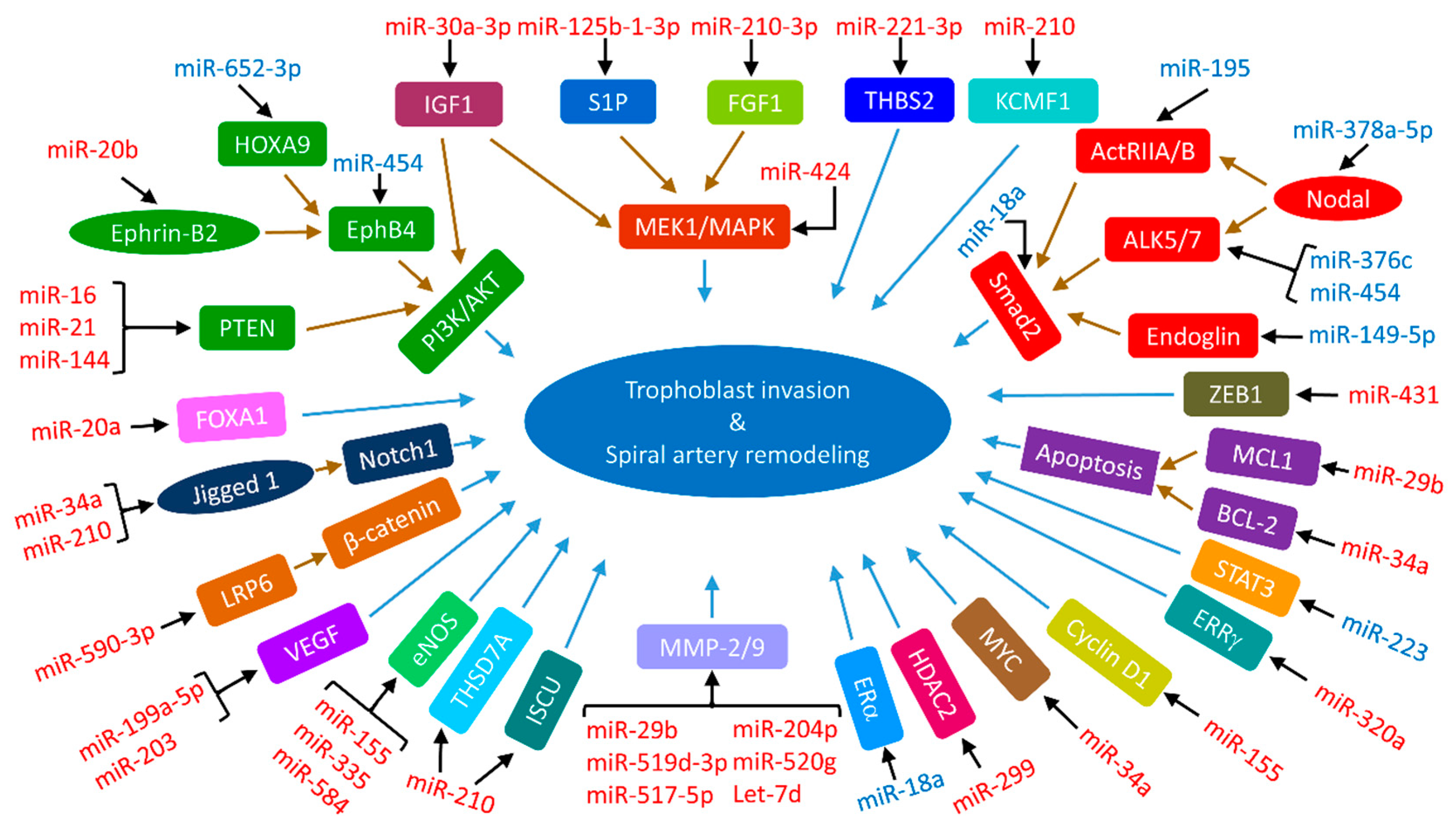
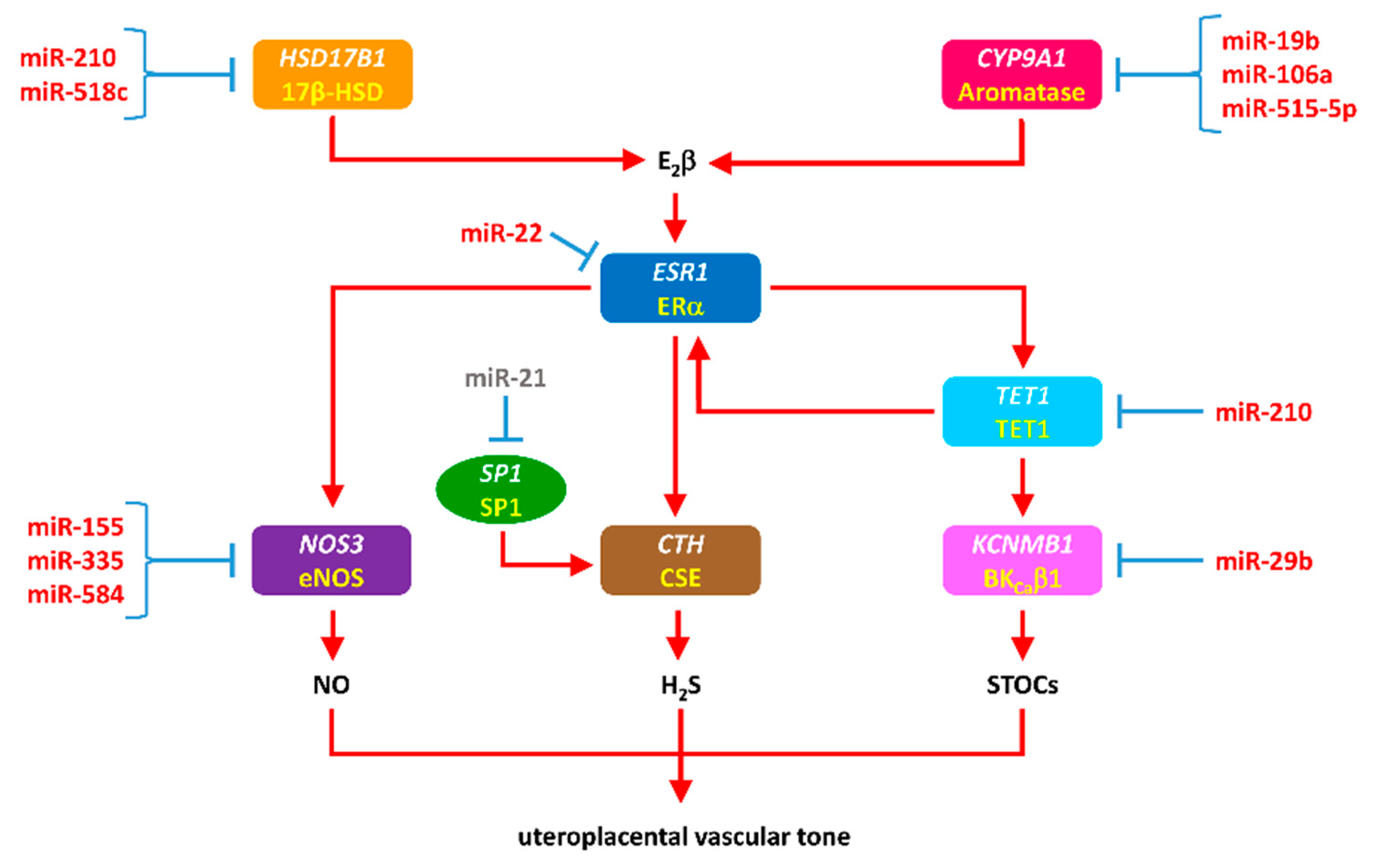
| Preeclampsia | ||||
| Upregulation | Downregulation | |||
| miRNA | Reference(s) | miRNA | Reference(s) | |
| let-7b | [56] | let-7c-5p | [57] | |
| let-7d | [58] | miR-1 | [39] | |
| miR-16 | [37] | miR-18a | [38,43] | |
| miR-17 | [30] | miR-19a-3p | [59] | |
| miR-17-3p | [43] | miR-19b | [43] | |
| miR-19b | [60] | miR-21 | [61] | |
| miR-20a | [30,62] | miR-27a-5p | [63] | |
| miR-20b | [30] | miR-34c-5p | [39] | |
| miR-25 | [42] | miR-101 | [64] | |
| miR-26b | [37,42] | miR-126 | [65,66] | |
| miR-29b | [37,67] | miR-127-3p | [57] | |
| miR-31-5p | [63] | miR-133b | [56] | |
| miR-30a-3p | [43,68] | miR-135b-5p | [63] | |
| miR-34a | [69,70] | miR-136-3p | [63] | |
| miR-34a-5p | [71] | miR-139-5p | [39] | |
| miR-92b | [42] | miR-144 | [72] | |
| miR-104 | [56] | miR-144-3p | [73] | |
| miR-106a | [60] | miR-149 | [74] | |
| miR-124-5p | [46] | miR-149-5p | [75] | |
| miR-125b-1-3p | [76] | miR-194 | [74] | |
| miR-128a | [56] | miR-195 | [43] | |
| miR-137 | [77] | miR-218 | [43] | |
| miR-141 | [78] | miR-218-5p | [79] | |
| miR-142-3p | [77,80] | miR-221-3p | [81] | |
| miR-148a-3p | [63] | miR-223 | [43,82] | |
| miR-151 | [43] | miR-224 | [82] | |
| miR-152 | [38] | miR-325 | [83] | |
| miR-154-3p | [36] | miR-328 | [39] | |
| miR-155 | [36,84] | miR-335 | [45] | |
| miR-181a | [37,45] | miR-346 | [85] | |
| miR-181a-5p | [84,86] | miR-363 | [38,45] | |
| miR-182 | [36,45] | miR-365a-3p | [63] | |
| miR-182-3p | [36,56] | miR-376c | [34,35] | |
| miR-182-5p | [87] | miR-377 | [38] | |
| miR-183 | [36] | miR-378a-5p | [33] | |
| miR-193b | [43,63] | miR-379 | [43] | |
| miR-193b-3p | [63] | miR-411 | [35,38,43] | |
| miR-195 | [37,88] | miR-454 | [89,90] | |
| miR-197 | [42] | miR455-3p | [91] | |
| miR-200b | [36] | miR455-5p | [91] | |
| miR-202-3p | [92] | miR-515-5p | [91,93] | |
| miR-203 | [94] | miR-517-5p | [51] | |
| miR-210 | [36,38,39,43,63,95,96,97,98,99,100] | miR-518f-5p | [51] | |
| miR-218 | [101] | miR-519a | [51] | |
| miR-222 | [37] | miR-519a-3p | [57] | |
| miR-296-3p | [42] | miR-519d | [51] | |
| miR-296-5p | [42] | miR-520a-5p | [51,63] | |
| miR-299 | [102] | miR-520h | [51] | |
| miR-320a | [103] | miR-524-5p | [51] | |
| miR-335 | [37,44] | miR-525 | [51] | |
| miR-342-3p | [42] | miR-526a | [51] | |
| miR-362-3p | [104] | miR-532-5p | [57] | |
| miR-423-5p | [57] | miR-539-5p | [57] | |
| miR-424 | [45] | miR-542-3p | [38] | |
| miR-431 | [46,105] | miR-544 | [46] | |
| miR-517-5p | [106] | miR-584 | [39] | |
| miR-517a/b | [107] | miR-629-5p | [57] | |
| miR-517c | [107] | miR-652-3p | [108] | |
| miR-518a-5p | [46] | miR-675 | [109] | |
| miR-519d-3p | [110] | miR-1301 | [82] | |
| miR-520g | [111] | |||
| miR-524 | [43] | |||
| miR-584 | [44] | |||
| IUGA | ||||
| Upregulation | Downregulation | |||
| miRNA | Reference(s) | miRNA | Reference(s) | |
| miR-10b | [49] | miR-16-5p | [52] | |
| miR-141 | [112] | miR-21 | [113,114] | |
| miR-193b-3p | [47] | miR-26a-5p | [52] | |
| miR-193b-5p | [47] | miR-100-5p | [52] | |
| miR-210 | [95] | miR-103a-3p | [52] | |
| miR-210-3p | [47,115] | miR-122-5p | [52] | |
| miR-221-3p | [47] | miR-125b-5p | [52] | |
| miR-342-3p | [47] | miR-126-3p | [52] | |
| miR-363 | [49] | miR-143-3p | [52] | |
| miR-365a/b-3p | [47] | miR-145-5p | [52] | |
| miR-424 | [116] | miR-149 | [74] | |
| miR-499a-5p | [52] | miR-199a-5p | [52] | |
| miR-519a | [51,117] | miR-346 | [85] | |
| miR-574-3p | [47] | miR-515-5p | [48] | |
| miR-516b | [48] | |||
| miR-517-5p | [51] | |||
| miR-518b | [48,117] | |||
| miR-518f-5p | [51] | |||
| miR-519d | [48,51] | |||
| miR-520a-5p | [51] | |||
| miR-520h | [48] | |||
| miR-525 | [51] | |||
| miR-526b | [48] | |||
| miR-1323 | [48] | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, X.-Q.; Zhang, L. MicroRNAs in Uteroplacental Vascular Dysfunction. Cells 2019, 8, 1344. https://doi.org/10.3390/cells8111344
Hu X-Q, Zhang L. MicroRNAs in Uteroplacental Vascular Dysfunction. Cells. 2019; 8(11):1344. https://doi.org/10.3390/cells8111344
Chicago/Turabian StyleHu, Xiang-Qun, and Lubo Zhang. 2019. "MicroRNAs in Uteroplacental Vascular Dysfunction" Cells 8, no. 11: 1344. https://doi.org/10.3390/cells8111344
APA StyleHu, X.-Q., & Zhang, L. (2019). MicroRNAs in Uteroplacental Vascular Dysfunction. Cells, 8(11), 1344. https://doi.org/10.3390/cells8111344






