Mechanisms Underlying Cell Therapy in Liver Fibrosis: An Overview
Abstract
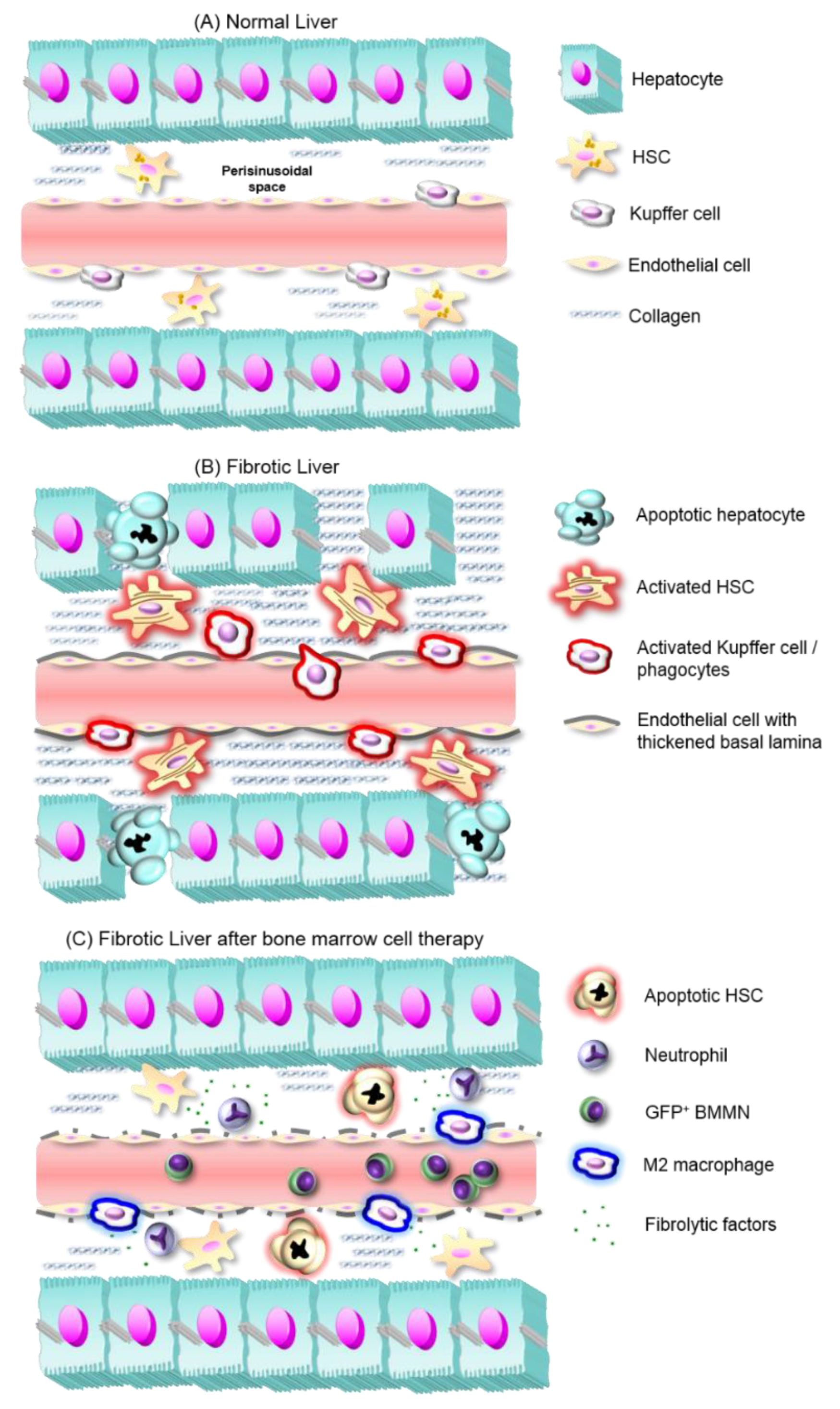
1. Introduction
2. Cell Types Used for the Inhibition of Liver Fibrosis
2.1. Bone Marrow Mononuclear Fraction
2.2. Endothelial Progenitor Cells
2.3. Mesenchymal Stem Cells
2.4. Bone Marrow Mesenchymal Stem Cells
2.5. Adipose-Derived Mesenchymal Stem Cells
2.6. Primary and Cell Line Hepatocytes
2.7. Oval or Hepatic Progenitor Cells
2.8. Pluripotent Cells
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chen, K.Y.; Shen, X.; Diehl, A.M. Prometheus revisited. J. Clin. Investig. 2018, 128, 2192–2193. [Google Scholar] [CrossRef] [PubMed]
- Bizzaro, D.; Russo, F.P.; Burra, P. New Perspectives in Liver Transplantation: From Regeneration to Bioengineering. Bioengineering (Basel) 2019, 6, 81. [Google Scholar] [CrossRef] [PubMed]
- Chamuleau, R.A.F.M.; Hoekstra, R. End-stage liver failure: Filling the treatment gap at the intensive care unit. J. Artif. Organs 2019. [Google Scholar] [CrossRef] [PubMed]
- Borkham-Kamphorst, E.; Weiskirchen, R. The PDGF system and its antagonists in liver fibrosis. Cytokine Growth Factor Rev. 2016, 28, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Krenkel, O.; Hundertmark, J.; Ritz, T.P.; Weiskirchen, R.; Tacke, F. Single Cell RNA Sequencing Identifies Subsets of Hepatic Stellate Cells and Myofibroblasts in Liver Fibrosis. Cells 2019, 8, 503. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Qi, X.; Guo, X. Child-Pugh Versus MELD Score for the Assessment of Prognosis in Liver Cirrhosis: A Systematic Review and Meta-Analysis of Observational Studies. Medicine (Baltimore) 2016, 95, e2877. [Google Scholar] [CrossRef]
- Massarweh, N.N.; El-Serag, H.B. Epidemiology of Hepatocellular Carcinoma and Intrahepatic Cholangiocarcinoma. Cancer Control 2017, 24, 1073274817729245. [Google Scholar] [CrossRef]
- Clements, O.; Eliahoo, J.; Un Kim, J.; Taylor-Robinson, S.D.; Khan, S.A. Risk Factors for Intrahepatic and Extrahepatic Cholangiocarcinoma: A systematic review and meta-analysis. J. Hepatol. 2019. [Google Scholar] [CrossRef]
- Tag, C.G.; Sauer-Lehnen, S.; Weiskirchen, S.; Borkham-Kamphorst, E.; Tolba, R.H.; Tacke, F.; Weiskirchen, R. Bile duct ligation in mice: Induction of inflammatory liver injury and fibrosis by obstructive cholestasis. J. Vis. Exp. 2015, 96, 52438. [Google Scholar] [CrossRef]
- Addante, A.; Roncero, C.; Almalé, L.; Lazcanoiturburu, N.; García-Álvaro, M.; Fernández, M.; Sanz, J.; Hammad, S.; Nwosu, Z.C.; Lee, S.J.; et al. Bone morphogenetic protein 9 as a key regulator of liver progenitor cells in DDC-induced cholestatic liver injury. Liver Int. 2018, 38, 1664–1675. [Google Scholar] [CrossRef]
- Finck, B.N. Targeting Metabolism, Insulin Resistance, and Diabetes to Treat Nonalcoholic Steatohepatitis. Diabetes 2018, 67, 2485–2493. [Google Scholar] [CrossRef] [PubMed]
- Bessone, F.; Razori, M.V.; Roma, M.G. Molecular pathways of nonalcoholic fatty liver disease development and progression. Cell. Mol. Life Sci. 2019, 76, 99–128. [Google Scholar] [CrossRef] [PubMed]
- Denk, H.; Abuja, P.M.; Zatloukal, K. Animal models of NAFLD from the pathologist’s point of view. Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1865, 929–942. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef]
- Tomizawa, M.; Shinozaki, F.; Motoyoshi, Y.; Sugiyama, T.; Yamamoto, S.; Ishige, N. Oncostatin M in William’s E medium is suitable for initiation of hepatocyte differentiation in human induced pluripotent stem cells. Mol. Med. Rep. 2017, 15, 3088–3092. [Google Scholar] [CrossRef]
- Weiskirchen, R.; Weiskirchen, S.; Tacke, F. Organ and tissue fibrosis: Molecular signals, cellular mechanisms and translational implications. Mol. Aspects Med. 2019, 65, 2–15. [Google Scholar] [CrossRef]
- Tacke, F.; Weiskirchen, R. An update on the recent advances in antifibrotic therapy. Expert Rev. Gastroenterol. Hepatol. 2018, 12, 1143–1152. [Google Scholar] [CrossRef]
- Qi, X.; Guo, X.; Su, C. Clinical Outcomes of the Transplantation of Stem Cells from Various Human Tissue Sources in the Management of Liver Cirrhosis: A Systematic Review and Meta-Analysis. Curr. Stem Cell Res. Ther. 2015, 10, 166–180. [Google Scholar] [CrossRef]
- Boyd, A.; Newsome, P.; Lu, W.Y. The role of stem cells in liver injury and repair. Expert Rev. Gastroenterol. Hepatol. 2019, 13, 623–631. [Google Scholar] [CrossRef]
- Morrison, S.J.; Scadden, D.T. The bone marrow niche for haematopoietic stem cells. Nature 2014, 505, 327–334. [Google Scholar] [CrossRef]
- Thomas, E.; Storb, R.; Clift, R.A.; Fefer, A.; Johnson, F.L.; Neiman, P.E.; Lerner, K.G.; Glucksberg, H.; Buckner, C.D. Bone-marrow transplantation (first of two parts). N. Engl. J. Med. 1975, 292, 832–843. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, S.N.; Lira, D.C.; Oliveira, G.P.; Thole, A.A.; Stumbo, A.C.; Caetano, C.E.; Marques, R.G.; Carvalho, L. Decreased collagen types I and IV, laminin, CK-19 and α-SMA expression after bone marrow cell transplantation in rats with liver fibrosis. Histochem. Cell Biol. 2010, 134, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Nunes de Carvalho, S.; Helal-Neto, E.; de Andrade, D.C.; Costa Cortez, E.A.; Thole, A.A.; Barja-Fidalgo, C.; de Carvalho, L. Bone marrow mononuclear cell transplantation increases metalloproteinase-9 and 13 and decreases tissue inhibitors of metalloproteinase-1 and 2 expression in the liver of cholestatic rats. Cells Tissues Organs 2013, 198, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Nunes de Carvalho, S.; da Cunha Lira, D.; Costa Cortez, E.A.; de Andrade, D.C.; Thole, A.A.; Stumbo, A.C.; de Carvalho, L. Bone marrow cell transplantation is associated with fibrogenic cells apoptosis during hepatic regeneration in cholestatic rats. Biochem. Cell Biol. 2013, 91, 88–94. [Google Scholar] [CrossRef]
- De Andrade, D.C.; de Carvalho, S.N.; Pinheiro, D.; Thole, A.A.; Moura, A.S.; de Carvalho, L.; Cortez, E.A. Bone marrow mononuclear cell transplantation improves mitochondrial bioenergetics in the liver of cholestatic rats. Exp. Cell Res. 2015, 336, 15–22. [Google Scholar] [CrossRef]
- Pinheiro, D.; Leirós, L.; Dáu, J.B.T.; Stumbo, A.C.; Thole, A.A.; Cortez, E.A.C.; Mandarim-de-Lacerda, C.A.; Carvalho, L.; Carvalho, S.N. Cytokines, hepatic cell profiling and cell interactions during bone marrow cell therapy for liver fibrosis in cholestatic mice. PLoS ONE 2017, 12, e0187970. [Google Scholar] [CrossRef]
- De Souza, V.C.A.; Pereira, T.A.; Teixeira, V.W.; Carvalho, H.; de Castro, M.C.A.B.; D’assunção, C.G.; de Barros, A.F.; Carvalho, C.L.; de Lorena, V.M.B.; Costa, V.M.A.; et al. Bone marrow-derived monocyte infusion improves hepatic fibrosis by decreasing osteopontin, TGF-β1, IL-13 and oxidative stress. World J. Gastroenterol. 2017, 23, 5146–5157. [Google Scholar] [CrossRef]
- Suh, Y.G.; Kim, J.K.; Byun, J.S.; Yi, H.S.; Lee, Y.S.; Eun, H.S.; Kim, S.Y.; Han, K.H.; Lee, K.S.; Duester, G.; et al. CD11b(+) Gr1(+) bone marrow cells ameliorate liver fibrosis by producing interleukin-10 in mice. Hepatology 2012, 56, 1902–1912. [Google Scholar] [CrossRef]
- Karlmark, K.R.; Weiskirchen, R.; Zimmermann, H.W.; Gassler, N.; Ginhoux, F.; Weber, C.; Merad, M.; Luedde, T.; Trautwein, C.; Tacke, F. Hepatic recruitment of the inflammatory Gr1+ monocyte subset upon liver injury promotes hepatic fibrosis. Hepatology 2009, 50, 261–274. [Google Scholar] [CrossRef]
- Carvalho, S.; Cortez, E.; Stumbo, A.C.; Thole, A.; Caetano, C.; Marques, R.; Pelajo-Machado, M.; Porto, L.C.; Carvalho, L. Laminin expression during bone marrow mononuclear cell transplantation in hepatectomized rats. Cell Biol. Int. 2008, 32, 1014–1018. [Google Scholar] [CrossRef]
- Asri, A.; Sabour, J.; Atashi, A.; Soleimani, M. Homing in hematopoietic stem cells: Focus on regulatory role of CXCR7 on SDF1a/CXCR4 axis. EXCLI J. 2016, 15, 134–143. [Google Scholar] [PubMed]
- Eggenhofer, E.; Luk, F.; Dahlke, M.H.; Hoogduijn, M.J. The life and fate of mesenchymal stem cells. Front. Immunol. 2014, 5, 148. [Google Scholar] [CrossRef] [PubMed]
- Yannaki, E.; Athanasiou, E.; Xagorari, A.; Constantinou, V.; Batsis, I.; Kaloyannidis, P.; Proya, E.; Anagnostopoulos, A.; Fassas, A. G-CSF-primed hematopoietic stem cells or G-CSF per se accelerate recovery and improve survival after liver injury, predominantly by promoting endogenous repair programs. Exp. Hematol. 2005, 33, 108–119. [Google Scholar] [CrossRef] [PubMed]
- Tsolaki, E.; Athanasiou, E.; Gounari, E.; Zogas, N.; Siotou, E.; Yiangou, M.; Anagnostopoulos, A.; Yannaki, E. Hematopoietic stem cells and liver regeneration: Differentially acting hematopoietic stem cell mobilization agents reverse induced chronic liver injury. Blood Cells Mol. Dis. 2014, 53, 124–132. [Google Scholar] [CrossRef] [PubMed]
- El-Akabawy, G.; El-Mehi, A. Mobilization of endogenous bone marrow-derived stem cells in a thioacetamide-induced mouse model of liver fibrosis. Tissue Cell 2015, 47, 257–265. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov. The U.S. National Institutes of Health, Department of Health and Human Services, National Library of Medicine. Available online: www.clinicaltrials.gov (accessed on 16 September 2019).
- Sakai, Y.; Takamura, M.; Seki, A.; Sunagozaka, H.; Terashima, T.; Komura, T.; Yamato, M.; Miyazawa, M.; Kawaguchi, K.; Nasti, A.; et al. Phase I clinical study of liver regenerative therapy for cirrhosis by intrahepatic arterial infusion of freshly isolated autologous adipose tissue-derived stromal/stem (regenerative) cell. Regen. Ther. 2017, 6, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Kharaziha, P.; Hellström, P.M.; Noorinayer, B.; Farzaneh, F.; Aghajani, K.; Jafari, F.; Telkabadi, M.; Atashi, A.; Honardoost, M.; Zali, M.R.; et al. Improvement of liver function in liver cirrhosis patients after autologous mesenchymal stem cell injection: A phase I-II clinical trial. Eur. J. Gastroenterol. Hepatol. 2009, 21, 1199–1205. [Google Scholar] [CrossRef]
- D’Avola, D.; Fernández-Ruiz, V.; Carmona-Torre, F.; Méndez, M.; Pérez-Calvo, J.; Prósper, F.; Andreu, E.; Herrero, J.I.; Iñarrairaegui, M.; Fuertes, C.; et al. Phase 1-2 pilot clinical trial in patients with decompensated liver cirrhosis treated with bone marrow-derived endothelial progenitor cells. Transl. Res. 2017, 188, 80–91.e2. [Google Scholar] [CrossRef]
- Algire, G.H.; Chalkley, H.W.; Legallais, F.Y.; Park, H.D. Vasculae Reactions of Normal and Malignant Tissues in Vivo. I. Vascular Reactions of Mice to Wounds and to Normal and Neoplastic Transplants. J. Natl. Cancer Inst. 1945, 6, 73–85. [Google Scholar] [CrossRef]
- Asahara, T.; Murohara, T.; Sullivan, A.; Silver, M.; van der Zee, R.; Li, T.; Witzenbichler, B.; Schatteman, G.; Isner, J.M. Isolation of putative progenitor endothelial cells for angiogenesis. Science 1997, 275, 964–967. [Google Scholar] [CrossRef]
- Asahara, T.; Masuda, H.; Takahashi, T.; Kalka, C.; Pastore, C.; Silver, M.; Kearne, M.; Magner, M.; Isner, J.M. Bone marrow origin of endothelial progenitor cells responsible for postnatal vasculogenesis in physiological and pathological neovascularization. Circ. Res. 1999, 85, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Aicher, A.; Heeschen, C.; Mildner-Rihm, C.; Urbich, C.; Ihling, C.; Technau-Ihling, K.; Zeiher, A.M.; Dimmeler, S. Essential role of endothelial nitric oxide synthase for mobilization of stem and progenitor cells. Nat. Med. 2003, 9, 1370–1376. [Google Scholar] [CrossRef] [PubMed]
- De Falco, E.; Porcelli, D.; Torella, A.R.; Straino, S.; Iachininoto, M.G.; Orlandi, A.; Truffa, S.; Biglioli, P.; Napolitano, M.; Capogrossi, M.C.; et al. SDF-1 involvement in endothelial phenotype and ischemia-induced recruitment of bone marrow progenitor cells. Blood 2004, 104, 3472–3482. [Google Scholar] [CrossRef] [PubMed]
- Ishida, Y.; Kimura, A.; Kuninaka, Y.; Inui, M.; Matsushima, K.; Mukaida, N.; Kondo, T. Pivotal role of the CCL5/CCR5 interaction for recruitment of endothelial progenitor cells in mouse wound healing. J. Clin. Investig. 2012, 122, 711–721. [Google Scholar] [CrossRef]
- Kaur, S.; Tripathi, D.; Dongre, K.; Garg, V.; Rooge, S.; Mukopadhyay, A.; Sakhuja, P.; Sarin, S.K. Increased number and function of endothelial progenitor cells stimulate angiogenesis by resident liver sinusoidal endothelial cells (SECs) in cirrhosis through paracrine factors. J. Hepatol. 2012, 57, 1193–1198. [Google Scholar] [CrossRef]
- Sieghart, W.; Fellner, S.; Reiberger, T.; Ulbrich, G.; Ferlitsch, A.; Wacheck, V.; Peck-Radosavljevic, M. Differential role of circulating endothelial progenitor cells in cirrhotic patients with or without hepatocellular carcinoma. Dig. Liver Dis. 2009, 41, 902–906. [Google Scholar] [CrossRef]
- Beaudry, P.; Hida, Y.; Udagawa, T.; Alwayn, I.P.; Greene, A.K.; Arsenault, D.; Folkman, J.; Heymach, J.V.; Ryeom, S.; Puder, M. Endothelial progenitor cells contribute to accelerated liver regeneration. J. Pediatr. Surg. 2007, 42, 1190–1198. [Google Scholar] [CrossRef]
- Asahara, T.; Kawamoto, A.; Masuda, H. Concise review: Circulating endothelial progenitor cells for vascular medicine. Stem Cells 2011, 29, 1650–1655. [Google Scholar] [CrossRef]
- Rehman, J.; Li, J.; Orschell, C.M.; March, K.L. Peripheral blood “endothelial progenitor cells” are derived from monocyte/macrophages and secrete angiogenic growth factors. Circulation 2003, 107, 1164–1169. [Google Scholar] [CrossRef]
- Miyamoto, Y.; Suyama, T.; Yashita, T.; Akimaru, H.; Kurata, H. Bone marrow subpopulations contain distinct types of endothelial progenitor cells and angiogenic cytokine-producing cells. J. Mol. Cell. Cardiol. 2007, 43, 627–635. [Google Scholar] [CrossRef]
- Yang, Z.; von Ballmoos, M.W.; Faessler, D.; Voelzmann, J.; Ortmann, J.; Diehm, N.; Kalka-Moll, W.; Baumgartner, I.; Di Santo, S.; Kalka, C. Paracrine factors secreted by endothelial progenitor cells prevent oxidative stress-induced apoptosis of mature endothelial cells. Atherosclerosis 2010, 211, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Di Santo, S.; Yang, Z.; Wyler von Ballmoos, M.; Voelzmann, J.; Diehm, N.; Baumgartner, I.; Kalka, C. Novel cell-free strategy for therapeutic angiogenesis: In vitro generated conditioned medium can replace progenitor cell transplantation. PLoS ONE 2009, 4, e5643. [Google Scholar] [CrossRef] [PubMed]
- Greene, A.K.; Wiener, S.; Puder, M.; Yoshida, A.; Shi, B.; Perez-Atayde, A.R.; Efstathiou, J.A.; Holmgren, L.; Adamis, A.P.; Rupnick, M.; et al. Endothelial-directed hepatic regeneration after partial hepatectomy. Ann. Surg. 2003, 237, 530–535. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chen, C.; Wei, L.; Li, Q.; Niu, X.; Xu, Y.; Wang, Y.; Zhao, J. Exosomes derived from endothelial progenitor cells attenuate vascular repair and accelerate reendothelialization by enhancing endothelial function. Cytotherapy 2016, 18, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, C.; Hu, B.; Niu, X.; Liu, X.; Zhang, G.; Zhang, C.; Li, Q.; Wang, Y. Exosomes Derived from Human Endothelial Progenitor Cells Accelerate Cutaneous Wound Healing by Promoting Angiogenesis Through Erk1/2 Signaling. Int. J. Biol. Sci. 2016, 12, 1472–1487. [Google Scholar] [CrossRef]
- Hu, H.; Wang, B.; Jiang, C.; Li, R.; Zhao, J. Endothelial progenitor cell-derived exosomes facilitate vascular endothelial cell repair through shuttling miR-21-5p to modulate Thrombospondin-1 expression. Clin. Sci. 2019, 33, 1629–1644. [Google Scholar] [CrossRef]
- Nakamura, T.; Torimura, T.; Sakamoto, M.; Hashimoto, O.; Taniguchi, E.; Inoue, K.; Sakata, R.; Kumashiro, R.; Murohara, T.; Ueno, T.; et al. Significance and therapeutic potential of endothelial progenitor cell transplantation in a cirrhotic liver rat model. Gastroenterology 2007, 133, 91–107.e1. [Google Scholar] [CrossRef]
- Liu, F.; Liu, Z.D.; Wu, N.; Cong, X.; Fei, R.; Chen, H.S.; Wei, L. Transplanted endothelial progenitor cells ameliorate carbon tetrachloride-induced liver cirrhosis in rats. Liver Transpl. 2009, 15, 1092–1100. [Google Scholar] [CrossRef]
- Nakamura, T.; Torimura, T.; Iwamoto, H.; Masuda, H.; Naitou, M.; Koga, H.; Abe, M.; Hashimoto, O.; Tsutsumi, V.; Ueno, T.; et al. Prevention of liver fibrosis and liver reconstitution of DMN-treated rat liver by transplanted EPCs. Eur. J. Clin. Investig. 2012, 42, 717–728. [Google Scholar] [CrossRef]
- Sakamoto, M.; Nakamura, T.; Torimura, T.; Iwamoto, H.; Masuda, H.; Koga, H.; Abe, M.; Hashimoto, O.; Ueno, T.; Sata, M. Transplantation of endothelial progenitor cells ameliorates vascular dysfunction and portal hypertension in carbon tetrachloride-induced rat liver cirrhotic model. J. Gastroenterol. Hepatol. 2013, 28, 168–178. [Google Scholar] [CrossRef]
- Lan, L.; Liu, R.; Qin, L.Y.; Cheng, P.; Liu, B.W.; Zhang, B.Y.; Ding, S.Z.; Li, X.L. Transplantation of bone marrow-derived endothelial progenitor cells and hepatocyte stem cells from liver fibrosis rats ameliorates liver fibrosis. World J. Gastroenterol. 2018, 24, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Saile, B.; Matthes, N.; Knittel, T.; Ramadori, G. Transforming growth factor beta and tumor necrosis factor alpha inhibit both apoptosis and proliferation of activated rat hepatic stellate cells. Hepatology 1999, 30, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, Y.; Matsumoto, K.; Yamada, A.; Ichida, T.; Asakura, H.; Komoriya, Y.; Nishiyama, E.; Nakamura, T. Preventive and therapeutic effects in rats of hepatocyte growth factor infusion on liver fibrosis/cirrhosis. Hepatology 1997, 26, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Liu, Z.D.; Wu, N.; Wang, J.H.; Zhang, H.H.; Fei, R.; Cong, X.; Chen, H.S.; Wei, L. In vitro interactions between rat bone marrow-derived endothelial progenitor cells and hepatic stellate cells: Interaction between EPCs and HSCs. In Vitro Cell. Dev. Biol. Anim. 2013, 49, 537–547. [Google Scholar] [CrossRef] [PubMed]
- Souidi, N.; Stolk, M.; Rudeck, J.; Strunk, D.; Schallmoser, K.; Volk, H.D.; Seifert, M. Stromal Cells Act as Guardians for Endothelial Progenitors by Reducing Their Immunogenicity After Co-Transplantation. Stem Cells 2017, 35, 1233–1245. [Google Scholar] [CrossRef] [PubMed]
- Tepper, O.M.; Galiano, R.D.; Capla, J.M.; Kalka, C.; Gagne, P.J.; Jacobowitz, G.R.; Levine, J.P.; Gurtner, G.C. Human endothelial progenitor cells from type II diabetics exhibit impaired proliferation, adhesion, and incorporation into vascular structures. Circulation 2002, 106, 2781–2786. [Google Scholar] [CrossRef]
- Kawamoto, A.; Asahara, T. Role of progenitor endothelial cells in cardiovascular disease and upcoming therapies. Catheter. Cardiovasc. Interv. 2007, 70, 477–484. [Google Scholar] [CrossRef]
- Masuda, J.; Mitsuyama, K.; Yamasaki, H.; Takedatsu, H.; Okamura, T.; Andoh, A.; Murohara, T.; Asahara, T.; Sata, M. Depletion of endothelial progenitor cells in the peripheral blood of patients with ulcerative colitis. Int. J. Mol. Med. 2007, 19, 221–228. [Google Scholar] [CrossRef][Green Version]
- Capla, J.M.; Grogan, R.H.; Callaghan, M.J.; Galiano, R.D.; Tepper, O.M.; Ceradini, D.J.; Gurtner, G.C. Diabetes impairs endothelial progenitor cell-mediated blood vessel formation in response to hypoxia. Plast. Reconstr. Surg. 2007, 119, 59–70. [Google Scholar] [CrossRef]
- Hill, J.M.; Zalos, G.; Halcox, J.P.; Schenke, W.H.; Waclawiw, M.A.; Quyyumi, A.A.; Finkel, T. Circulating endothelial progenitor cells, vascular function, and cardiovascular risk. N. Engl. J. Med. 2003, 348, 593–600. [Google Scholar] [CrossRef]
- Shirakura, K.; Masuda, H.; Kwon, S.M.; Obi, S.; Ito, R.; Shizuno, T.; Kurihara, Y.; Mine, T.; Asahara, T. Impaired function of bone marrow-derived endothelial progenitor cells in murine liver fibrosis. Biosci. Trends 2011, 5, 77–82. [Google Scholar] [CrossRef][Green Version]
- Rautou, P.-E. Endothelial progenitor cells in cirrhosis: The more, the merrier? J. Hepatol. 2012, 57, 1163–1165. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kaur, S.; Anita, K. Angiogenesis in liver regeneration and fibrosis: “a double-edged sword”. Hepatol. Int. 2013, 7, 959–968. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Sehgal, R.; Shastry, S.M.; McCaughan, G.; McGuire, H.M.; Fazekas St de Groth, B.; Sarin, S.; Trehanpati, N.; Seth, D. Circulating Endothelial Progenitor Cells Present an Inflammatory Phenotype and Function in Patients with Alcoholic Liver Cirrhosis. Front. Physiol. 2018, 9, 556. [Google Scholar] [CrossRef]
- Fadini, G.P.; Losordo, D.; Dimmeler, S. Critical reevaluation of endothelial progenitor cell phenotypes for therapeutic and diagnostic use. Circ. Res. 2012, 110, 624–637. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, N.A.; Reinisch, A.; Strunk, D. Endothelial colony-forming progenitor cell isolation and expansion. Methods Mol. Biol. 2012, 879, 381–387. [Google Scholar] [PubMed]
- Tasev, D.; van Wijhe, M.H.; Weijers, E.M.; van Hinsbergh, V.W.; Koolwijk, P. Long-Term Expansion in Platelet Lysate Increases Growth of Peripheral Blood-Derived Endothelial-Colony Forming Cells and Their Growth Factor-Induced Sprouting Capacity. PLoS ONE 2015, 10, e0129935. [Google Scholar] [CrossRef]
- Takizawa, S.; Nagata, E.; Nakayama, T.; Masuda, H.; Asahara, T. Recent Progress in Endothelial Progenitor Cell Culture Systems: Potential for Stroke Therapy. Neurol. Med. Chir. (Tokyo) 2016, 56, 302–309. [Google Scholar] [CrossRef]
- Medina, R.J.; Barber, C.L.; Sabatier, F.; Dignat-George, F.; Melero-Martin, J.M.; Khosrotehrani, K.; Ohneda, O.; Randi, A.M.; Chan, J.K.Y.; Yamaguchi, T.; et al. Endothelial Progenitors: A Consensus Statement on Nomenclature. Stem Cells Transl. Med. 2017, 6, 1316–1320. [Google Scholar] [CrossRef]
- Akita, T.; Murohara, T.; Ikeda, H.; Sasaki, K.; Shimada, T.; Egami, K.; Imaizumi, T. Hypoxic preconditioning augments efficacy of human endothelial progenitor cells for therapeutic neovascularization. Lab. Investig. 2003, 83, 65–73. [Google Scholar] [CrossRef]
- Choi, J.H.; Hur, J.; Yoon, C.H.; Kim, J.H.; Lee, C.S.; Youn, S.W.; Oh, I.Y.; Skurk, C.; Murohara, T.; Park, Y.B.; et al. Augmentation of therapeutic angiogenesis using genetically modified human endothelial progenitor cells with altered glycogen synthase kinase-3beta activity. J. Biol. Chem. 2004, 279, 49430–49438. [Google Scholar] [CrossRef] [PubMed]
- Roncalli, J.; Tongers, J.; Renault, M.A.; Losordo, D.W. Biological approaches to ischemic tissue repair: Gene- and cell-based strategies. Expert Rev. Cardiovasc. Ther. 2008, 6, 653–668. [Google Scholar] [CrossRef] [PubMed]
- Goto, K.; Takemura, G.; Takahashi, T.; Okada, H.; Kanamori, H.; Kawamura, I.; Watanabe, T.; Morishita, K.; Tsujimoto, A.; Miyazaki, N.; et al. Intravenous Administration of Endothelial Colony-Forming Cells Overexpressing Integrin β1 Augments Angiogenesis in Ischemic Legs. Stem Cells Transl. Med. 2016, 5, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Owen, M.; Friedenstein, A.J. Stromal stem cells: Marrow-derived osteogenic precursors. Ciba Found. Symp. 1988, 136, 42–60. [Google Scholar] [PubMed]
- Pereira, R.F.; O’Hara, M.D.; Laptev, A.V.; Halford, K.W.; Pollard, M.D.; Class, R.; Simon, D.; Livezey, K.; Prockop, D.J. Marrow stromal cells as a source of progenitor cells for nonhematopoietic tissues in transgenic mice with a phenotype of osteogenesis imperfecta. Proc. Natl. Acad. Sci. USA 1998, 95, 1142–1147. [Google Scholar] [CrossRef]
- Spees, J.L.; Olson, S.D.; Ylostalo, J.; Lynch, P.J.; Smith, J.; Perry, A.; Peister, A.; Wang, M.Y.; Prockop, D.J. Differentiation, cell fusion, and nuclear fusion during ex vivo repair of epithelium by human adult stem cells from bone marrow stroma. Proc. Natl. Acad. Sci. USA 2003, 100, 2397–2402. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef]
- Mushahary, D.; Spittler, A.; Kasper, C.; Weber, V.; Charwat, V. Isolation, cultivation, and characterization of human mesenchymal stem cells. Cytometry A 2018, 93, 19–31. [Google Scholar] [CrossRef]
- Driscoll, J.; Patel, T. The mesenchymal stem cell secretome as an acellular regenerative therapy for liver disease. J. Gastroenterol. 2019, 54, 763–773. [Google Scholar] [CrossRef]
- Choi, J.S.; Ryu, H.A.; Cheon, S.H.; Kim, S.W. Human Adipose Derived Stem Cells Exhibit Enhanced Liver Regeneration in Acute Liver Injury by Controlled Releasing Hepatocyte Growth Factor. Cell. Physiol. Biochem. 2019, 52, 935–950. [Google Scholar]
- Lange, C.; Bassler, P.; Lioznov, M.V.; Bruns, H.; Kluth, D.; Zander, A.R.; Fiegel, H.C. Liver-specific gene expression in mesenchymal stem cells is induced by liver cells. World J. Gastroenterol. 2005, 11, 4497–4504. [Google Scholar] [CrossRef] [PubMed]
- An, S.Y.; Han, J.; Lim, H.J.; Park, S.Y.; Kim, J.H.; Do, B.R. Valproic acid promotes differentiation of hepatocyte-like cells from whole human umbilical cord-derived mesenchymal stem cells. Tissue Cell 2014, 46, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Ong, S.Y.; Dai, H.; Leong, K.W. Inducing hepatic differentiation of human mesenchymal stem cells in pellet culture. Biomaterials 2006, 27, 4087–4097. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Xu, L.; Lin, N.; Pan, W.; Hu, K.; Xu, R. Activation of Notch1 signaling by marrow-derived mesenchymal stem cells through cell-cell contact inhibits proliferation of hepatic stellate cells. Life Sci 2011, 89, 975–981. [Google Scholar] [CrossRef]
- Qiao, H.; Zhou, Y.; Qin, X.; Cheng, J.; He, Y.; Jiang, Y. NADPH Oxidase Signaling Pathway Mediates Mesenchymal Stem Cell-Induced Inhibition of Hepatic Stellate Cell Activation. Stem Cells Int. 2018, 2018, 1239143. [Google Scholar] [CrossRef]
- Eom, Y.W.; Shim, K.Y.; Baik, S.K. Mesenchymal stem cell therapy for liver fibrosis. Korean J. Intern. Med. 2015, 30, 580–589. [Google Scholar] [CrossRef]
- Zhang, X.; Hu, M.G.; Pan, K.; Li, C.H.; Liu, R. 3D Spheroid Culture Enhances the Expression of Antifibrotic Factors in Human Adipose-Derived MSCs and Improves Their Therapeutic Effects on Hepatic Fibrosis. Stem Cells Int. 2016, 2016, 4626073. [Google Scholar] [CrossRef]
- Harting, M.T.; Srivastava, A.K.; Zhaorigetu, S.; Bair, H.; Prabhakara, K.S.; Toledano Furman, N.E.; Vykoukal, J.V.; Ruppert, K.A.; Cox, C.S., Jr.; Olson, S.D. Inflammation-Stimulated Mesenchymal Stromal Cell-Derived Extracellular Vesicles Attenuate Inflammation. Stem Cells 2018, 36, 79–90. [Google Scholar] [CrossRef]
- Lee, S.C.; Jeong, H.J.; Lee, S.K.; Kim, S.J. Hypoxic Conditioned Medium from Human Adipose-Derived Stem Cells Promotes Mouse Liver Regeneration Through JAK/STAT3 Signaling. Stem Cells Transl. Med. 2016, 5, 816–825. [Google Scholar] [CrossRef]
- Liu, L.; Gao, J.; Yuan, Y.; Chang, Q.; Liao, Y.; Lu, F. Hypoxia preconditioned human adipose derived mesenchymal stem cells enhance angiogenic potential via secretion of increased VEGF and bFGF. Cell Biol. Int. 2013, 37, 551–560. [Google Scholar] [CrossRef]
- Baba, S.; Fujii, H.; Hirose, T.; Yasuchika, K.; Azuma, H.; Hoppo, T.; Naito, M.; Machimoto, T.; Ikai, I. Commitment of bone marrow cells to hepatic stellate cells in mouse. J. Hepatol. 2004, 40, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Russo, F.P.; Alison, M.R.; Bigger, B.W.; Amofah, E.; Florou, A.; Amin, F.; Bou-Gharios, G.; Jeffery, R.; Iredale, J.P.; Forbes, S.J. The bone marrow functionally contributes to liver fibrosis. Gastroenterology 2006, 130, 1807–1821. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, A.B.; Quintanilha, L.F.; Dias, J.V.; Paredes, B.D.; Mannheimer, E.G.; Carvalho, F.G.; Asensi, K.D.; Gutfilen, B.; Fonseca, L.M.; Resende, C.M.; et al. Bone marrow multipotent mesenchymal stromal cells do not reduce fibrosis or improve function in a rat model of severe chronic liver injury. Stem Cells 2008, 26, 1307–1314. [Google Scholar] [CrossRef] [PubMed]
- Mannheimer, E.G.; Quintanilha, L.F.; Carvalho, A.B.; Paredes, B.D.; Gonçalves de Carvalho, F.; Takyia, C.M.; Resende, C.M.; Ferreira da Motta Rezende, G.; Campos de Carvalho, A.C.; Schanaider, A.; et al. Bone marrow cells obtained from cirrhotic rats do not improve function or reduce fibrosis in a chronic liver disease model. Clin. Transplant. 2011, 25, 54–60. [Google Scholar] [CrossRef]
- Zhao, D.C.; Lei, J.X.; Chen, R.; Yu, W.H.; Zhang, X.M.; Li, S.N.; Xiang, P. Bone marrow-derived mesenchymal stem cells protect against experimental liver fibrosis in rats. World J. Gastroenterol. 2005, 11, 3431–3440. [Google Scholar] [CrossRef]
- Hong, J.; Jin, H.; Han, J.; Hu, H.; Liu, J.; Li, L.; Huang, Y.; Wang, D.; Wu, M.; Qiu, L.; et al. Infusion of human umbilical cord-derived mesenchymal stem cells effectively relieves liver cirrhosis in DEN-induced rats. Mol. Med. Rep. 2014, 9, 1103–1111. [Google Scholar] [CrossRef][Green Version]
- Fiorina, P.; Jurewicz, M.; Augello, A.; Vergani, A.; Dada, S.; La Rosa, S.; Selig, M.; Godwin, J.; Law, K.; Placidi, C.; et al. Immunomodulatory function of bone marrow-derived mesenchymal stem cells in experimental autoimmune type 1 diabetes. J. Immunol. 2009, 183, 993–1004. [Google Scholar] [CrossRef]
- Gazdic, M.; Volarevic, V.; Arsenijevic, N.; Stojkovic, M. Mesenchymal stem cells: A friend or foe in immune-mediated diseases. Stem Cell Rev. Rep. 2015, 11, 280–287. [Google Scholar] [CrossRef]
- Lou, G.; Chen, Z.; Zheng, M.; Liu, Y. Mesenchymal stem cell-derived exosomes as a new therapeutic strategy for liver diseases. Exp. Mol. Med. 2017, 49, e346. [Google Scholar] [CrossRef]
- Gronthos, S.; Zannettino, A.C.; Hay, S.J.; Shi, S.; Graves, S.E.; Kortesidis, A.; Simmons, P.J. Molecular and cellular characterisation of highly purified stromal stem cells derived from human bone marrow. J. Cell. Sci. 2003, 116, 1827–1835. [Google Scholar] [CrossRef]
- Meng, F.; Wang, K.; Aoyama, T.; Grivennikov, S.I.; Paik, Y.; Scholten, D.; Cong, M.; Iwaisako, K.; Liu, X.; Zhang, M.; et al. Interleukin-17 signaling in inflammatory, Kupffer cells, and hepatic stellate cells exacerbates liver fibrosis in mice. Gastroenterology 2012, 143, 765–776.e3. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Huang, D.; Weng, J.; Huang, Y.; Liu, S.; Zhang, Q.; Li, N.; Wen, M.; Zhu, G.; Lin, F.; et al. Neutralization of Interleukin-17 Attenuates Cholestatic Liver Fibrosis in Mice. Scand. J. Immunol. 2016, 83, 102–108. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Farouk, S.; Sabet, S.; Abu Zahra, F.A.; El-Ghor, A.A. Bone marrow derived-mesenchymal stem cells downregulate IL17A dependent IL6/STAT3 signaling pathway in CCl4-induced rat liver fibrosis. PLoS ONE 2018, 13, e0206130. [Google Scholar] [CrossRef] [PubMed]
- Milosavljevic, N.; Gazdic, M.; Simovic Markovic, B.; Arsenijevic, A.; Nurkovic, J.; Dolicanin, Z.; Jovicic, N.; Jeftic, I.; Djonov, V.; Arsenijevic, N.; et al. Mesenchymal stem cells attenuate liver fibrosis by suppressing Th17 cells—An experimental study. Transpl. Int. 2018, 31, 102–115. [Google Scholar] [CrossRef] [PubMed]
- Duman, D.G.; Zibandeh, N.; Ugurlu, M.U.; Celikel, C.; Akkoc, T.; Banzragch, M.; Genc, D.; Ozdogan, O. Mesenchymal stem cells suppress hepatic fibrosis accompanied by expanded intrahepatic natural killer cells in rat fibrosis model. Mol. Biol. Rep. 2019, 46, 2997–3008. [Google Scholar] [CrossRef]
- Luo, X.Y.; Meng, X.J.; Cao, D.C.; Wang, W.; Zhou, K.; Li, L.; Guo, M.; Wang, P. Transplantation of bone marrow mesenchymal stromal cells attenuates liver fibrosis in mice by regulating macrophage subtypes. Stem Cell Res. Ther. 2019, 10, 16. [Google Scholar] [CrossRef]
- Rong, X.; Liu, J.; Yao, X.; Jiang, T.; Wang, Y.; Xie, F. Human bone marrow mesenchymal stem cells-derived exosomes alleviate liver fibrosis through the Wnt/β-catenin pathway. Stem Cell Res. Ther. 2019, 10, 98. [Google Scholar] [CrossRef]
- Ye, Z.; Lu, W.; Liang, L.; Tang, M.; Wang, Y.; Li, Z.; Zeng, H.; Wang, A.; Lin, M.; Huang, L.; et al. Mesenchymal stem cells overexpressing hepatocyte nuclear factor-4 alpha alleviate liver injury by modulating anti-inflammatory functions in mice. Stem Cell Res. Ther. 2019, 10, 149. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, R.; Rong, W.; Han, M.; Cui, C.; Feng, Z.; Sun, X.; Jin, S. Therapeutic effect of hepatocyte growth factor-overexpressing bone marrow-derived mesenchymal stem cells on CCl. Cell Death Dis. 2018, 9, 1186. [Google Scholar] [CrossRef]
- Mohamed, H.E.; Elswefy, S.E.; Rashed, L.A.; Younis, N.N.; Shaheen, M.A.; Ghanim, A.M. Bone marrow-derived mesenchymal stem cells effectively regenerate fibrotic liver in bile duct ligation rat model. Exp. Biol. Med. (Maywood) 2016, 241, 581–591. [Google Scholar] [CrossRef]
- Lai, L.; Chen, J.; Wei, X.; Huang, M.; Hu, X.; Yang, R.; Jiang, X.; Shan, H. Transplantation of MSCs Overexpressing HGF into a Rat Model of Liver Fibrosis. Mol. Imaging Biol. 2016, 18, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.D.; Kim, S.S.; Cha, H.Y.; Jang, S.H.; Chang, D.Y.; Kim, W.; Suh-Kim, H.; Lee, J.H. Therapeutic effect of hepatocyte growth factor-secreting mesenchymal stem cells in a rat model of liver fibrosis. Exp. Mol. Med. 2014, 46, e110. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Cheng, X.; Wang, H.; Huang, W.; la Ga Hu, Z.; Wang, D.; Zhang, K.; Zhang, H.; Xue, Z.; Da, Y.; et al. Mesenchymal stem cells and their secreted molecules predominantly ameliorate fulminant hepatic failure and chronic liver fibrosis in mice respectively. J. Transl. Med. 2016, 14, 45. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.O.; Jun, B.G.; Baik, S.K.; Kim, M.Y.; Kwon, S.O. Inhibition of hepatic stellate cells by bone marrow-derived mesenchymal stem cells in hepatic fibrosis. Clin. Mol. Hepatol. 2015, 21, 141–149. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zuk, P.A.; Zhu, M.; Mizuno, H.; Huang, J.; Futrell, J.W.; Katz, A.J.; Benhaim, P.; Lorenz, H.P.; Hedrick, M.H. Multilineage cells from human adipose tissue: Implications for cell-based therapies. Tissue Eng. 2001, 7, 211–228. [Google Scholar] [CrossRef] [PubMed]
- Haldar, D.; Henderson, N.C.; Hirschfield, G.; Newsome, P.N. Mesenchymal stromal cells and liver fibrosis: A complicated relationship. FASEB J. 2016, 30, 3905–3928. [Google Scholar] [CrossRef]
- Di Bonzo, L.V.; Ferrero, I.; Cravanzola, C.; Mareschi, K.; Rustichell, D.; Novo, E.; Sanavio, F.; Cannito, S.; Zamara, E.; Bertero, M.; et al. Human mesenchymal stem cells as a two-edged sword in hepatic regenerative medicine: Engraftment and hepatocyte differentiation versus profibrogenic potential. Gut 2008, 57, 223–231. [Google Scholar] [CrossRef]
- Li, C.; Kong, Y.; Wang, H.; Wang, S.; Yu, H.; Liu, X.; Yang, L.; Jiang, X.; Li, L. Homing of bone marrow mesenchymal stem cells mediated by sphingosine 1-phosphate contributes to liver fibrosis. J. Hepatol. 2009, 50, 1174–1183. [Google Scholar] [CrossRef]
- Mahmoudifar, N.; Doran, P.M. Mesenchymal Stem Cells Derived from Human Adipose Tissue. Methods Mol. Biol. 2015, 1340, 53–64. [Google Scholar]
- Lee, S.C.; Kim, J.O.; Kim, S.J. Secretome from human adipose-derived stem cells protects mouse liver from hepatic ischemia-reperfusion injury. Surgery 2015, 157, 934–943. [Google Scholar] [CrossRef]
- Burchfield, J.S.; Paul, A.L.; Lanka, V.; Tan, W.; Kong, Y.; McCallister, C.; Rothermel, B.A.; Schneider, J.W.; Gillette, T.G.; Hill, J.A. Pharmacological priming of adipose-derived stem cells promotes myocardial repair. J. Investig. Med. 2016, 64, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Maria, A.T.; Maumus, M.; Le Quellec, A.; Jorgensen, C.; Noël, D.; Guilpain, P. Adipose-Derived Mesenchymal Stem Cells in Autoimmune Disorders: State of the Art and Perspectives for Systemic Sclerosis. Clin. Rev. Allergy Immunol. 2017, 52, 234–259. [Google Scholar] [CrossRef] [PubMed]
- Hao, T.; Chen, J.; Zhi, S.; Zhang, Q.; Chen, G.; Yu, F. Comparison of bone marrow-vs. adipose tissue-derived mesenchymal stem cells for attenuating liver fibrosis. Exp. Ther. Med. 2017, 14, 5956–5964. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.; Le, T.; Do, H.; Ngo, D.; Le, H.; Truong, N. Comparative treatment efficiency of adipose and bone marrow derived allogenic mesenchymal stem cell transplantation in mouse models of liver fibrosis. Biomed. Res. Ther. 2017, 4, 1374–1387. [Google Scholar] [CrossRef]
- Yu, F.; Ji, S.; Su, L.; Wan, L.; Zhang, S.; Dai, C.; Wang, Y.; Fu, J.; Zhang, Q. Adipose-derived mesenchymal stem cells inhibit activation of hepatic stellate cells in vitro and ameliorate rat liver fibrosis in vivo. J. Formos. Med. Assoc. 2015, 114, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Nahar, S.; Nakashima, Y.; Miyagi-Shiohira, C.; Kinjo, T.; Toyoda, Z.; Kobayashi, N.; Saitoh, I.; Watanabe, M.; Noguchi, H.; Fujita, J. Cytokines in adipose-derived mesenchymal stem cells promote the healing of liver disease. World J. Stem Cells 2018, 10, 146–159. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.S.; Ju, D.T.; Day, C.H.; Yeh, Y.L.; Chen, R.J.; Viswanadha, V.P.; Chang, R.L.; Lin, Y.C.; Yao, C.H.; Huang, C.Y. Protective effect of autologous transplantation of resveratrol preconditioned adipose-derived stem cells in the treatment of diabetic liver dysfunction in rat model. J. Tissue Eng. Regen. Med. 2019, 13, 1629–1640. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.P.; Akahoshi, T.; Piao, J.S.; Narahara, S.; Murata, M.; Kawano, T.; Hamano, N.; Ikeda, T.; Hashizume, M. Basic fibroblast growth factor-treated adipose tissue-derived mesenchymal stem cell infusion to ameliorate liver cirrhosis via paracrine hepatocyte growth factor. J. Gastroenterol. Hepatol. 2015, 30, 1065–1074. [Google Scholar] [CrossRef]
- De Luna-Saldivar, M.M.; Marino-Martinez, I.A.; Franco-Molina, M.A.; Rivera-Morales, L.G.; Alarcón-Galván, G.; Cordero-Pérez, P.; Rojas-Martínez, A.; Rodríguez-Padilla, C.; Muñoz-Espinosa, L.E. Advantages of adipose tissue stem cells over CD34. Ann. Hepatol. 2019, 18, 620–626. [Google Scholar] [CrossRef]
- Baig, M.T.; Ali, G.; Awan, S.J.; Shehzad, U.; Mehmood, A.; Mohsin, S.; Khan, S.N.; Riazuddin, S. Serum from CCl. Growth Factors 2017, 35, 144–160. [Google Scholar] [CrossRef]
- Zhang, S.; Zhu, Z.; Wang, Y.; Liu, S.; Zhao, C.; Guan, W.; Zhao, Y. Therapeutic potential of Bama miniature pig adipose stem cells induced hepatocytes in a mouse model with acute liver failure. Cytotechnology 2018, 70, 1131–1141. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.L.; Wang, Z.G.; Xiong, L.K.; Pan, L.Y.; Zhu, Q.; Yuan, Y.F.; Liu, Z.S. Hepatogenic differentiation from human adipose-derived stem cells and application for mouse acute liver injury. Artif. Cells Nanomed. Biotechnol. 2017, 45, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Lv, Y.; Chen, F.; Wang, X.; Zhu, J.; Li, H.; Xiao, J. Co-stimulation of LPAR. Stem Cell Res. Ther. 2018, 9, 161. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Seo, E.; Park, J.M.; Han, N.Y.; Lee, H.; Jun, H.S. Effects of FGF21-secreting adipose-derived stem cells in thioacetamide-induced hepatic fibrosis. J. Cell. Mol. Med. 2018, 22, 5165–5169. [Google Scholar] [CrossRef] [PubMed]
- McGill, M.R.; Yan, H.M.; Ramachandran, A.; Murray, G.J.; Rollins, D.E.; Jaeschke, H. HepaRG cells: A human model to study mechanisms of acetaminophen hepatotoxicity. Hepatology 2011, 53, 974–982. [Google Scholar] [CrossRef] [PubMed]
- Mandon, M.; Huet, S.; Dubreil, E.; Fessard, V.; Le Hégarat, L. Three-dimensional HepaRG spheroids as a liver model to study human genotoxicity in vitro with the single cell gel electrophoresis assay. Sci. Rep. 2019, 9, 10548. [Google Scholar] [CrossRef] [PubMed]
- Kreutz, C.; MacNelly, S.; Follo, M.; Wäldin, A.; Binninger-Lacour, P.; Timmer, J.; Bartolomé-Rodríguez, M.M. Hepatocyte Ploidy Is a Diversity Factor for Liver Homeostasis. Front. Physiol. 2017, 8, 862. [Google Scholar] [CrossRef]
- Kostadinova, R.; Boess, F.; Applegate, D.; Suter, L.; Weiser, T.; Singer, T.; Naughton, B.; Roth, A. A long-term three dimensional liver co-culture system for improved prediction of clinically relevant drug-induced hepatotoxicity. Toxicol. Appl. Pharmacol. 2013, 268, 1–16. [Google Scholar] [CrossRef]
- Godoy, P.; Hewitt, N.J.; Albrecht, U.; Andersen, M.E.; Ansari, N.; Bhattacharya, S.; Bode, J.G.; Bolleyn, J.; Borner, C.; Böttger, J.; et al. Recent advances in 2D and 3D in vitro systems using primary hepatocytes, alternative hepatocyte sources and non-parenchymal liver cells and their use in investigating mechanisms of hepatotoxicity, cell signaling and ADME. Arch. Toxicol. 2013, 87, 1315–1530. [Google Scholar] [CrossRef]
- Tostões, R.M.; Leite, S.B.; Serra, M.; Jensen, J.; Björquist, P.; Carrondo, M.J.; Brito, C.; Alves, P.M. Human liver cell spheroids in extended perfusion bioreactor culture for repeated-dose drug testing. Hepatology 2012, 55, 1227–1236. [Google Scholar] [CrossRef]
- Bell, C.C.; Hendriks, D.F.; Moro, S.M.; Ellis, E.; Walsh, J.; Renblom, A.; Fredriksson Puigvert, L.; Dankers, A.C.; Jacobs, F.; Snoeys, J.; et al. Characterization of primary human hepatocyte spheroids as a model system for drug-induced liver injury, liver function and disease. Sci. Rep. 2016, 6, 25187. [Google Scholar] [CrossRef] [PubMed]
- Ito, M.; Nagata, H.; Yamamoto, T.; Yoshihara, D.; Fox, I.J.; Miyakawa, S. Intrasplenic hepatocyte transplantation prolonged the survival in Nagase analbuminemic rats with liver failure induced by common bile duct ligation. Cell. Transplant. 2007, 16, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Ito, M.; Nagata, H.; Westerman, K.A.; Lafleur, D.; Chowdhury, J.R.; Leboulch, P.; Fox, I.J. Treatment of liver failure in rats with end-stage cirrhosis by transplantation of immortalized hepatocytes. Hepatology 2002, 36, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Dusabineza, A.C.; Najimi, M.; van Hul, N.; Legry, V.; Khuu, D.N.; van Grunsven, L.A.; Sokal, E.; Leclercq, I.A. Hepatic Stellate Cells Improve Engraftment of Human Primary Hepatocytes: A Preclinical Transplantation Study in an Animal Model. Cell. Transplant. 2015, 24, 2557–2571. [Google Scholar] [CrossRef] [PubMed]
- Dhawan, A.; Mitry, R.R.; Hughes, R.D. Hepatocyte transplantation for liver-based metabolic disorders. J. Inherit. Metab. Dis. 2006, 29, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.A.; Sinha, S.; Fitzpatrick, E.; Dhawan, A. Hepatocyte transplantation and advancements in alternative cell sources for liver-based regenerative medicine. J. Mol. Med. (Berl.) 2018, 96, 469–481. [Google Scholar] [CrossRef]
- Sato, K.; Marzioni, M.; Meng, F.; Francis, H.; Glaser, S.; Alpini, G. Ductular Reaction in Liver Diseases: Pathological Mechanisms and Translational Significances. Hepatology 2019, 69, 420–430. [Google Scholar] [CrossRef]
- Zhou, H.; Rogler, L.E.; Teperman, L.; Morgan, G.; Rogler, C.E. Identification of hepatocytic and bile ductular cell lineages and candidate stem cells in bipolar ductular reactions in cirrhotic human liver. Hepatology 2007, 45, 716–724. [Google Scholar] [CrossRef]
- Liu, X.B.; Lo, C.M.; Cheng, Q.; Ng, K.T.; Shao, Y.; Li, C.X.; Chung, S.K.; Ng, I.O.L.; Yu, J.; Man, K. Oval Cells Contribute to Fibrogenesis of Marginal Liver Grafts under Stepwise Regulation of Aldose Reductase and Notch Signaling. Theranostics 2017, 7, 4879–4893. [Google Scholar] [CrossRef]
- Lotowska, J.M.; Sobaniec-Lotowska, M.E.; Lebensztejn, D.M.; Daniluk, U.; Sobaniec, P.; Sendrowski, K.; Daniluk, J.; Reszec, J.; Debek, W. Ultrastructural Characteristics of Rat Hepatic Oval Cells and Their Intercellular Contacts in the Model of Biliary Fibrosis: New Insights into Experimental Liver Fibrogenesis. Gastroenterol. Res. Pract. 2017, 2017, 2721547. [Google Scholar] [CrossRef]
- Awan, S.J.; Baig, M.T.; Yaqub, F.; Tayyeb, A.; Ali, G. In vitro differentiated hepatic oval-like cells enhance hepatic regeneration in CCl. Cell Biol. Int. 2017, 41, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Bria, A.; Marda, J.; Zhou, J.; Sun, X.; Cao, Q.; Petersen, B.E.; Pi, L. Hepatic progenitor cell activation in liver repair. Liver Res. 2017, 1, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Ghanekar, A.; Kamath, B.M. Cholangiocytes derived from induced pluripotent stem cells for disease modeling. Curr. Opin. Gastroenterol. 2016, 32, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Dianat, N.; Dubois-Pot-Schneider, H.; Steichen, C.; Desterke, C.; Leclerc, P.; Raveux, A.; Combettes, L.; Weber, A.; Corlu, A.; Dubart-Kupperschmitt, A. Generation of functional cholangiocyte-like cells from human pluripotent stem cells and HepaRG cells. Hepatology 2014, 60, 700–714. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhao, P.; Wan, Z.; Jin, X.; Cheng, Y.; Yan, T.; Qing, S.; Ding, N.; Xin, S. Differentiation of human foreskin fibroblast-derived induced pluripotent stem cells into hepatocyte-like cells. Cell Biochem. Funct. 2016, 34, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Kim, Y.; Sharkis, S.; Marchionni, L.; Jang, Y.Y. In vivo liver regeneration potential of human induced pluripotent stem cells from diverse origins. Sci. Transl. Med. 2011, 3, 82ra39. [Google Scholar] [CrossRef]
- Povero, D.; Pinatel, E.M.; Leszczynska, A.; Goyal, N.P.; Nishio, T.; Kim, J.; Kneiber, D.; de Araujo Horcel, L.; Eguchi, A.; Ordonez, P.M.; et al. Human induced pluripotent stem cell-derived extracellular vesicles reduce hepatic stellate cell activation and liver fibrosis. JCI Insight 2019, 5, 125652. [Google Scholar] [CrossRef]
- Chien, Y.; Huang, C.S.; Lin, H.C.; Lu, K.H.; Tsai, P.H.; Lai, Y.H.; Chen, K.H.; Lee, S.D.; Huang, Y.H.; Wang, C.Y. Improvement of non-alcoholic steatohepatitis by hepatocyte-like cells generated from iPSCs with Oct4/Sox2/Klf4/Parp1. Oncotarget 2018, 9, 18594–18606. [Google Scholar] [CrossRef]
- Park, S.; In Hwang, S.; Kim, J.; Hwang, S.; Kang, S.; Yang, S.; Kang, W.; Kim, K.H.; Han, D.W.; Paik, Y.H. The therapeutic potential of induced hepatocyte-like cells generated by direct reprogramming on hepatic fibrosis. Stem Cell Res. Ther. 2019, 10, 21. [Google Scholar] [CrossRef]
- Takayama, K.; Akita, N.; Mimura, N.; Akahira, R.; Taniguchi, Y.; Ikeda, M.; Sakurai, F.; Ohara, O.; Morio, T.; Sekiguchi, K.; et al. Generation of safe and therapeutically effective human induced pluripotent stem cell-derived hepatocyte-like cells for regenerative medicine. Hepatol. Commun. 2017, 1, 1058–1069. [Google Scholar] [CrossRef]
- Tian, L.; Deshmukh, A.; Ye, Z.; Jang, Y.Y. Efficient and Controlled Generation of 2D and 3D Bile Duct Tissue from Human Pluripotent Stem Cell-Derived Spheroids. Stem Cell Rev. Rep. 2016, 12, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Sampaziotis, F.; de Brito, M.C.; Madrigal, P.; Bertero, A.; Saeb-Parsy, K.; Soares, F.A.C.; Schrumpf, E.; Melum, E.; Karlsen, T.H.; Bradley, J.A.; et al. Cholangiocytes derived from human induced pluripotent stem cells for disease modeling and drug validation. Nat. Biotechnol. 2015, 33, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Tasnim, F.; Xing, J.; Huang, X.; Mo, S.; Wei, X.; Tan, M.H.; Yu, H. Generation of mature Kupffer cells from human induced pluripotent stem cells. Biomaterials 2019, 192, 377–391. [Google Scholar] [CrossRef] [PubMed]
- Hoeffel, G.; Chen, J.; Lavin, Y.; Low, D.; Almeida, F.F.; See, P.; Beaudin, A.E.; Lum, J.; Low, I.; Forsberg, E.C.; et al. C-Myb(+) erythro-myeloid progenitor-derived fetal monocytes give rise to adult tissue-resident macrophages. Immunity 2015, 42, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Acun, A.; Oganesyan, R.; Uygun, B.E. Liver Bioengineering: Promise, Pitfalls, and Hurdles to Overcome. Curr. Transplant. Rep. 2019, 6, 119–126. [Google Scholar] [CrossRef] [PubMed]
| Cells/Dose/Route | Fibrosis Model | Main Results and Mechanisms of Action | Reference |
|---|---|---|---|
| Rat BMMNs, 107 cells, jugular vein | BDL (Wistar rats) | Collagen types I and IV, laminin, CK-19 and α-SMA reduction | [22] |
| Rat BMMNs, 107 cells, jugular vein | BDL (Wistar rats) | MMP-9 and MMP-13 expression were increased by macrophages, TIMP-1 and TIMP-2 reduction | [23] |
| Rat BMMNs, 107 cells, jugular vein | BDL (Wistar rats) | Fibrogenic cell apoptosis | [24] |
| Rat BMMNs, 107 cells, jugular vein | BDL (Wistar rats) | Oxidative stress reduction (4HNE), mitochondrial coupling (UCP2 levels) and biogenesis (PGC1-α) regulation | [25] |
| Mouse BMMNs, 107 cells, jugular vein | BDL (C57BL/6) | BMMNs originated populations of CD144, CD11b and Ly6G cells in the fibrotic liver, anti-fibrotic cytokines augmentation (IL-10, IL-13, IFN-γ, HGF) and pro-inflammatory cytokines reduction (IL-17A, IL-6) | [26] |
| Mouse BMMNs or BMMNs-derived monocytes, 106 cells per 3 weeks | CCl4 (orogastric), 200 μL-20%, 12 weeks, C57BL/6 | BMMN derived-monocyte had a better therapeutic effect, pro-inflammatory/fibrotic cytokines (TNF-α, IL-6, IL-1β, TGF-β1) reduction, IL-10 and MMP-9 were increased | [27] |
| Mouse BMMNs, 106 cells, tail vein | CCl4 (intraperitoneal) 0.4 mL/kg, 3 x per week, 2 weeks, C57BL/6 | CD4+CD25+Foxp3+ Treg cells produced IL-10 and promoted IL-6 and MCP-1 reduction, CD11b+F4/80+ cells were reduced in fibrotic liver | [28] |
| Trial Number (Status) | Cohort | Intervention | Study Phase Type | Follow-Up (Months) | Main Analysis Criteria | Outcomes/Published Results |
|---|---|---|---|---|---|---|
| NCT02297867 (Completed) | Liver cirrhosis (n = 6) | Autologous ADSC by intrahepatic injection | Phase I | 1–6 | MELD | NR |
| NCT02705742 (unknown) | Liver cirrhosis | Autologous ADSC by intravenous injection | Phase I/II | 12 | All-cause mortality | NR |
| NCT04088058 (not yet recruiting) | Liver cirrhosis (n = 20) | Autologous ADSC by intrahepatic injection | Phase II open-label single-arm | 1–12 | MELD | NR |
| NCT03629015 (not yet recruiting) | Acute liver failure (n = 20) | Allogeneic ADSC by intravenous infusion of low (0.5 × 106 cells/kg) or high (2 × 106 cells/kg) dose | Phase I | 12 | Incidence of adverse events and suspected unexpected serious adverse reaction | NR |
| NCT00913289 (terminated) | Liver cirrhosis (n = 6) | Autologous adipose tissue-derived stromal cells | Phase I | 6 | All cause harmful events | NR |
| NCT01062750 (Completed) | Liver cirrhosis (n = 4) | Autologous adipose tissue-derived stromal cells via intrahepatic arterial catheterization | NA | 1 | All cause harmful events | No serious adverse events, albumin serum levels were improved in three patients [37] |
| NCT03254758 (Recruiting) | Decompensated liver cirrhosis | ADSC by intravenous infusion | Phase I/II | 6 | Incidence of adverse events and Child Pugh score | NR |
| NCT01854125 (unknown) | Liver cirrhosis (n = 30) | Autologous BMMSC transplantation via hepatic artery catheterization | Phase III | 3 | LF, MELD, adverse effects | Improvement of LF in cirrhotic patients after autologous mesenchymal stem cell injection in phase I–II [38] |
| NCT00993941 (unknown) | Liver cirrhosis | Autologous BMMSC transplantation via portal vein catheterization or drug therapy (oral or intravenous) | Phase II | 12 | ALT, total bilirubin, prothrombin time, albumin, laminin, prealbumin, procollagen III, collagen IV, hyaluronidase and histology | NR |
| NCT03838250 (Recruiting) | Alcoholic liver cirrhosis (n = 10) | Autologous BMMSC transplantation via hepatic artery | Phase I | 12 | Incidence of serious adverse events | NR |
| NCT03468699 (Recruiting) | Biliary liver cirrhosis (n = 20) | Autologous BMMSC transplantation via hepatic artery | Single group assignment, Phase II | 6 | Cholestasis changes, LF, PELD | NR |
| NCT00713934 (Completed) | Liver cirrhosis (n = 7) | Autologous BM-derived CD133+ and BM mononuclear stem cells transplantation via portal vein | Randomized Phase I/II | 6 | LF, MELD | NR |
| NCT01120925 (Completed) | Liver cirrhosis (n = 30) | Autologous BM-derived CD133+ and BM mononuclear stem cells transplantation via portal vein | Randomized Phase I/II | 6 | LF, MELD and Child Pugh scores | NR |
| NCT01333228 (Completed) | Liver cirrhosis (n = 12), age 18–75 years | Autologous BM-derived EPCs, single group assignment, 8.45 × 106 to 450 × 106 cells administered through the hepatic artery | Single arm non-randomized Phase I/II | 12 | Primary: Number of Participants with adverse events; Secondary: LF, MELD, and Child-Pugh scores, HVPG, complications of liver cirrhosis | Treatment was confirmed safe and feasible, transient (but significant) beneficial effects in LF [39] |
| NCT03109236 (Recruiting) | Decompensated liver cirrhosis (n = 66) | Autologous BM-derived EPCs administrated via the portal vein system | Two arm randomized Phase III | 3, 6, or 12 | Primary: Fibrosis (Ishak, MRE, MELD, quantitative fibrosis), Secondary: overall survival, LF, HVPG, clinical decompensation, patient reported outcome | NR |
| Cells/Dose/Route | Fibrosis Model | Main Results and Mechanisms of Action | Reference |
|---|---|---|---|
| Rat BM-EPCs,3 × 106 cells—single or four-repeated doses once a week for 4 weeks, tail vein | CCl4 or thioacetamide (intraperitoneal) twice a week for 10 weeks (Wistar) | Increased survival rates, liver fibrosis and fibrogenesis reduction (HSC suppression and enhanced MMP activity), increased hepatocyte proliferation and HGF, TGF-α, EGF, and VEGF expression in liver | [58] |
| Rat BM-EPCs, 5 × 105 cells, portal vein | CCl4 by gavage twice a week for 16 weeks (Sprague-Dawley) | Increased survival rates, reduced levels of AST, ALT, and TBIL, albumin levels restoration, liver fibrosis and fibrogenesis reduction (HSC suppression), increased liver cell proliferation | [59] |
| Rat BM-EPCs, 3 × 106 cells—once weekly for four weeks, tail vein | Dimethylnitrosamine (intraperitoneal) three times a week for eight weeks (Sprague–Dawley) | Liver fibrosis and fibrogenesis reduction (HSC suppression), increased hepatocyte proliferation, vascular density and HGF, TGF-α and EGF expression in liver | [60] |
| Rat BM-EPCs, 3 × 106 cells—once a week for four weeks, tail vein | CCl4 (intraperitoneal) twice weekly for 10 weeks (Wistar) | Liver fibrosis and fibrogenesis reduction (HSC suppression), reduced portal venous pressure, increased vascular density and hepatic blood flow | [61] |
| Fibrotic rat BM-EPCs, 2 × 105 and 2 × 106 cells—once a week for three weeks, tail vein and portal vein | CCl4 (subcutaneous) twice a week for six weeks (Wistar) | Liver fibrosis suppression, improved liver function (lower ALT, AST, APTT), increased liver mRNA levels of HGF and VEGF, increased liver cells proliferation | [62] |
| Cells/Dose/Route | Fibrosis Model | Main Results and Mechanisms of Action | Reference |
|---|---|---|---|
| Mouse BMMSC and CM-BMMSC, 106 cells | CCl4 (intraperitoneal) in C57BL/6 mice, 1 µL/g, 2x per week, 1 month | IDO promoted Th17 suppression (IL-17 reduction) and IL-10 production and activation of CD4 T cells. | [115] |
| Human BMMSC and EX-BMMSC, 106 cells | CCl4 (intraperitoneal) in Sprague Dawley rats | EX-BMMSC had better therapeutic effect; IL-1, IL-2, IL-6, IL-8, and TNF-α reduction; PPARγ, Wnt3a, Wnt10b, β-catenin, WISP1, Cyclin D1 were decreased; inhibition of hepatic stellate cell activation | [118] |
| Mouse BMMSC and HNF-4α-overexpressing BMMSC, 106 cells | CCl4 (intragastric gavage) in C57BL/6 mice, 5.0 mL/kg, 2x per week, 3 weeks | HNF-4α-overexpressing BMMSC had better therapeutic effect; reduction in TNF-α, IFN-γ, IL-6; enhanced iNOS expression that depends on NF-κB signalling; Kupffer cell inhibition | [119] |
| Rat BMMSC, 3 × 106 cells | CCl4 (intraperitoneal) in Sprague Dawley rats, 1 mL/g, 2x per week, 6 weeks | Reduction in IL-17, IL-2 and IL-6; downregulation of IL-17a, IL-17ra, IL-17f, Stat3, p-STAT3, Stat5a, p-SMAD3, and TGFβR2; elevation of p-STAT5 protein | [114] |
| Rat BMMSC, HGF overexpressing BMMSC and HGF alone | CCl4 (intraperitoneal) in Sprague Dawley rats, 5.0 mL/kg, 30 consecutive days | AST, ALT, total bilirubin levels reduction; hepatocyte nuclear factor 4α, albumin, and cytokeratin 18 expression were increased | [120] |
| Mouse BMMSC, 5 × 105 cells | CCl4 (intraperitoneal) in C57BL/6 mice, 1 mL/g, 70 days | IL-10 increase and IL-12b, IFN-γ, TNF-α, and IL-6 gene expression were decreased; M2 macrophages activation; MMP13 production and M1 macrophages inhibition | [117] |
| Rat BMMSC, 106 cells | Bile duct ligation in Sprague Dawley rats | T cell proliferation was decreased; inflammatory cytokines were reduced; expansion of intrahepatic NK cells | [116] |
| Rat BMMSC, 106 cells or Silymarin (100 mg/kg) | Bile duct ligation in Wistar rats | BMMSC had a better therapeutic effect; MMP-2 mRNA upregulation and CK-19 mRNA downregulation; HGF augmentation regulate MMP-2 and CK-19 gene expression | [121] |
| Human BMMSC, HGF-overexpressing BMMSC, 4 × 106 cells | CCl4 (intraperitoneal) in Sprague Dawley rats, 0.2 mL/100 mg, 3x per week, 8 weeks | HGF overexpressing BMMSC had better therapeutic effect; HGF overexpressing increased homing of BMMSC in the liver | [122] |
| Human BMMSC, HGF-overexpressing BMMSC, 107 cells | Dimethylnitrosamine (intraperitoneal) in Sprague Dawley rats, 1 mL/g, three consecutive days per week, 4 weeks | HGF overexpressing BMMSC had better therapeutic effect; reduction in PDGF-bb, TGF-β1, and TIMP2; inhibition of α-SMA cells | [123] |
| Cells/Dose/Route | Fibrosis Model | Main Results and Mechanisms of Action | Reference |
|---|---|---|---|
| Human ADSC induced with cocktail of cytokines,1 × 106 cells by intravenous route | thioacetamide (intraperitoneal) in BALB/c nude mice (150 mg/kg) twice a week for 1 month | Degradation of MEC (increased MMP-13), decreased collagen and α-SMA content, activation of p-38 MAPK signaling, angiogenesis and hepatocyte proliferation, inhibition of HSC | [91] |
| Rat ADSC preconditioned with resveratrol, 1 × 106 cells by intravenous route | Streptozotocin (intraperitoneal) in Wistar rats (50 mg/kg) | Decreased collagen I, improved hepatocyte survival, downregulation of apoptotic pathway (caspase-3, cytochrome-c, FAD), upregulation of antioxidant pathway (Sirt1, SOD2), degradation of MEC (increased MMP-2) | [138] |
| Rat ADSC preconditioned with serum of liver- injured rat, 1.5 × 106 cells by intrahepatic route | CCl4 (intraperitoneal) in Sprague Dawley rats, 1 µL/g twice a week for one month | Decreased fibrosis, increased albumin, AFP, CK-18 and HNF4 levels in liver, degradation of MEC (increased MMP-2), increased expression of CK-8, CK-19, albumin, and AFP in ADSC | [141] |
| Rat ADSC, 3.0 x106 cells | CCl4 (intraperitoneal) in Wistar rats, three times a week for two months | Increased HGF and IL-10 serum level, decreased TGF-β and TNF-α, decreased collagen deposition, increased PCNA, hepatocyte proliferation | [140] |
| Pig ADSC induced to hepatocytes, 1.5 × 107 cells by intravenous route | CCl4 (intraperitoneal) in mice, twice a week for five to seven weeks | Decreased collagen deposition, decreased ALT and AST, increased albumin serum levels, decreased TGF-β, IL-6 and IL-10 | [142] |
| Human ADSC induced to hepatocytes, 2–3 × 106 cells by intravenous route | CCl4 (intraperitoneal) in mice, 5 mL/kg single dose and Cyclosporin A (10 mg/kg) daily for three weeks | Decreased ALT and AST | [143] |
| Rat ADSC preconditioned with bFGF, 5.0 × 106 cells by intravenous route | CCl4 (intraperitoneal) in Fischer 344 rats, 1 mg/kg twice a week for eight weeks | Apoptosis of HSC, activation of JNK-p53 signaling, increased expression of HGF | [139] |
| Human ADSC pretreated with Lysophosphatidic acid and sphingosine-1-phosphate, 2 × 106 cells by intravenous route | Intraperitoneal D-galactosamine (600 mg/kg) and LPS (8 μg/kg) in NOD/SCID mice | Decreased serum levels of ALT, AST, MDA, TNF and caspase-3/7 | [144] |
| Rat ADSC, 5.0 × 106 cells injected through portal vein | CCl4 (subcutaneous) in Sprague Dawley rats, 1.5 mL/kg twice a week for 10–12 weeks | Decreased expression of collagen type I and α-SMA, increased serum level of HGF, decreased serum level of TGF-β and NGF | [136] |
| Mouse ADSC transfected with miR-122, 1 × 106 cells injected through tail vein | CCl4 (intraperitoneal), 1 mL/kg twice a week for four weeks | Decreased mature hepatic collagen type I α1, inhibition of cell proliferation and collagen maturation by HSC | [110] |
| Cells/Dose/Route | Fibrosis Model | Main Results and Mechanisms of Action | Reference |
|---|---|---|---|
| Extracellular vesicles derived from human iPSC (iPSC-EVs) for in vitro experiments, animal models: 1.5 × 106 murine iPSC-EVs by tail vein | Activated HSC in vitro, CCl4 and BDL models of fibrosis in mice | Downregulation of collagens, growth factors and TIMPs in HSCs, reduced proliferation and chemotaxis, reduced fibrosis in both models, downregulation of α-SMA, collagen-I α1, and TIMP-1 | [168] |
| Hepatocyte-like cells derived from reprogramming of senescent mouse fibroblasts with Oct4, Sox2, Klf4, and the novel factor poly(ADPribose) polymerase 1 (Parp1), intra-splenic route | Non-alcoholic steatohepatitis (NASH) mouse model using a methionine/choline-deficient diet for four weeks | iPSC-derived hepatocyte-like cells were resistant to oxidative stress induced by hydrogen peroxide and lipid overload with fatty acids, increased IL-10 secretion, cell therapy attenuated macrovacuolar steatosis and restored liver function | [169] |
| Intra-splenic transplantation of 1 × 106 hepatocyte-like cells derived from reprogrammed mouse embryonic fibroblasts | Acute and chronic liver injury was induced using different treatment protocols with CCl4 in BALB/c nude mice | Normalization of ALT and ALS levels in both models, reduced mRNA expression of fibrotic markers COL1α1, α-SMA, TGF-β1, and pro-inflammatory cytokines (TNF-α, IL-6, IL-1β), increased expression of antioxidant molecules NQO-1, HO-1, SOD-1, catalase and GST | [170] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pinheiro, D.; Dias, I.; Ribeiro Silva, K.; Stumbo, A.C.; Thole, A.; Cortez, E.; de Carvalho, L.; Weiskirchen, R.; Carvalho, S. Mechanisms Underlying Cell Therapy in Liver Fibrosis: An Overview. Cells 2019, 8, 1339. https://doi.org/10.3390/cells8111339
Pinheiro D, Dias I, Ribeiro Silva K, Stumbo AC, Thole A, Cortez E, de Carvalho L, Weiskirchen R, Carvalho S. Mechanisms Underlying Cell Therapy in Liver Fibrosis: An Overview. Cells. 2019; 8(11):1339. https://doi.org/10.3390/cells8111339
Chicago/Turabian StylePinheiro, Daphne, Isabelle Dias, Karina Ribeiro Silva, Ana Carolina Stumbo, Alessandra Thole, Erika Cortez, Lais de Carvalho, Ralf Weiskirchen, and Simone Carvalho. 2019. "Mechanisms Underlying Cell Therapy in Liver Fibrosis: An Overview" Cells 8, no. 11: 1339. https://doi.org/10.3390/cells8111339
APA StylePinheiro, D., Dias, I., Ribeiro Silva, K., Stumbo, A. C., Thole, A., Cortez, E., de Carvalho, L., Weiskirchen, R., & Carvalho, S. (2019). Mechanisms Underlying Cell Therapy in Liver Fibrosis: An Overview. Cells, 8(11), 1339. https://doi.org/10.3390/cells8111339






