Ocrelizumab Depletes CD20+ T Cells in Multiple Sclerosis Patients
Abstract
1. Introduction
2. Patients and Methods
2.1. Patients and Samples
2.2. Multicolor Flow Cytometry
2.3. Statistical Analysis
3. Results
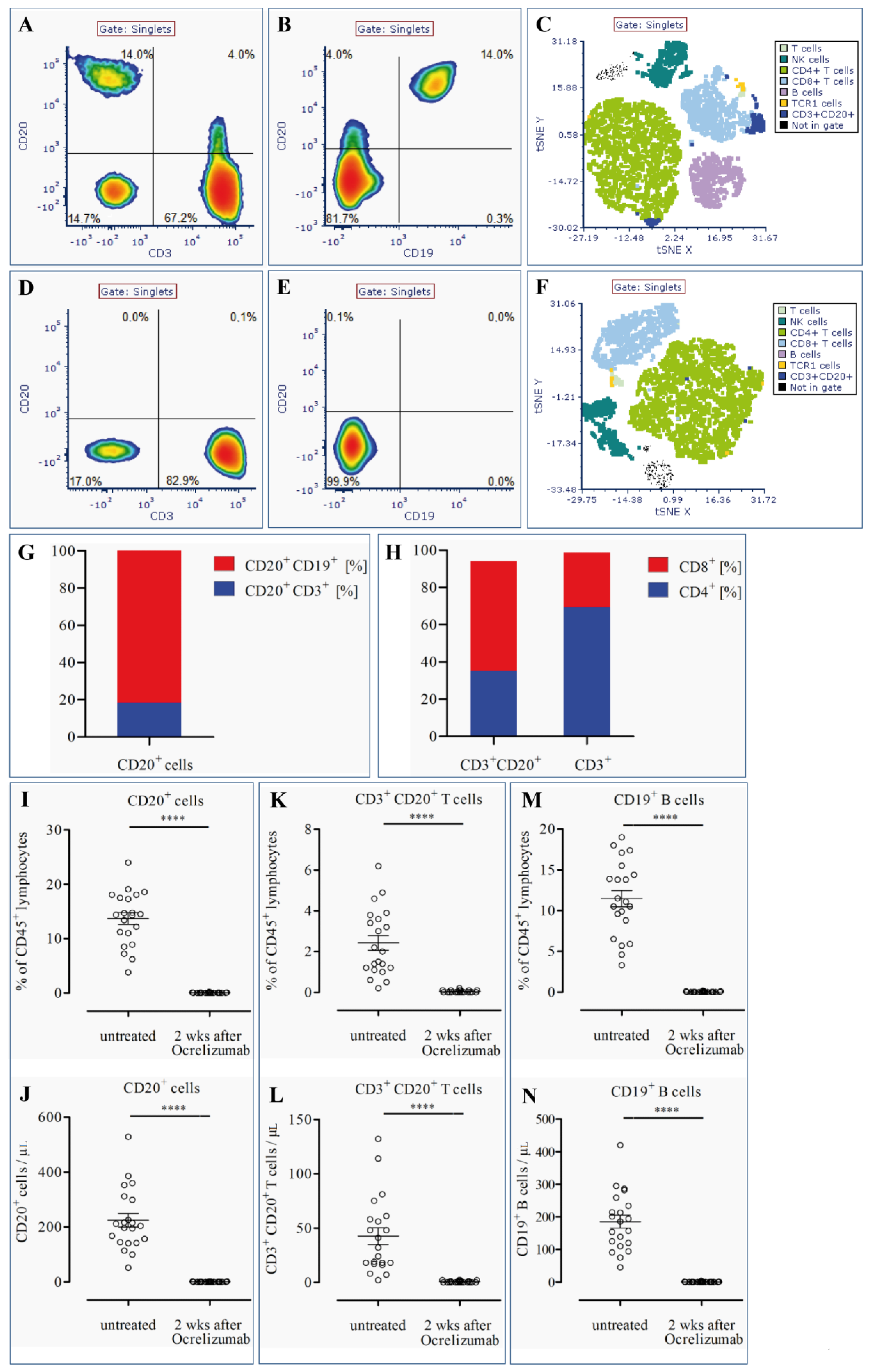
3.1. CD20+ T Cells Constitute a Significant Proportion of CD20+ Cells in the Blood of MS Patients
3.2. CD20+ T Cells Are Effectively Depleted by Ocrelizumab
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sospedra, M.; Martin, R. Immunology of multiple sclerosis. Annu. Rev. Immunol. 2005, 23, 683–747. [Google Scholar] [CrossRef] [PubMed]
- Bittner, S.; Ruck, T.; Wiendl, H.; Grauer, O.M.; Meuth, S.G. Targeting B cells in relapsing-remitting multiple sclerosis: From pathophysiology to optimal clinical management. Ther. Adv. Neurol. Disord. 2017, 10, 51–66. [Google Scholar] [CrossRef] [PubMed]
- Bar-Or, A.; Calabresi, P.A.; Arnold, D.; Markowitz, C.; Shafer, S.; Kasper, L.H.; Waubant, E.; Gazda, S.; Fox, R.J.; Panzara, M.; et al. Rituximab in relapsing-remitting multiple sclerosis: A 72-week, open-label, phase I trial. Ann. Neurol. 2008, 63, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Hauser, S.L.; Waubant, E.; Arnold, D.L.; Vollmer, T.; Antel, J.; Fox, R.J.; Bar-Or, A.; Panzara, M.; Sarkar, N.; Agarwal, S.; et al. B-cell depletion with rituximab in relapsing-remitting multiple sclerosis. N. Engl. J. Med. 2008, 358, 676–688. [Google Scholar] [CrossRef] [PubMed]
- Hauser, S.L.; Bar-Or, A.; Comi, G.; Giovannoni, G.; Hartung, H.P.; Hemmer, B.; Lublin, F.; Montalban, X.; Rammohan, K.W.; Selmaj, K.; et al. Ocrelizumab versus interferon β-1a in relapsing multiple sclerosis. N. Engl. J. Med. 2017, 376, 221–234. [Google Scholar] [CrossRef] [PubMed]
- Montalban, X.; Hauser, S.L.; Kappos, L.; Arnold, D.L.; Bar-Or, A.; Comi, G.; de Seze, J.; Giovannoni, G.; Hartung, H.P.; Hemmer, B.; et al. Ocrelizumab versus placebo in primary progressive multiple sclerosis. N. Engl. J. Med. 2017, 376, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Loken, M.R.; Shah, V.O.; Dattilio, K.L.; Civin, C.I. Flow cytometric analysis of human bone marrow. II. Normal B lymphocyte development. Blood 1987, 70, 1316–1324. [Google Scholar] [PubMed]
- Tedder, T.F.; Engel, P. CD20: A regulator of cell-cycle progression of B lymphocytes. Immunol. Today 1994, 15, 450–454. [Google Scholar] [CrossRef]
- Hultin, L.E.; Hausner, M.A.; Hultin, P.M.; Giorgi, J.V. CD20 (pan-B cell) antigen is expressed at a low level on a subpopulation of human T lymphocytes. Cytometry 1993, 14, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Wilk, E.; Witte, T.; Marquardt, N.; Horvath, T.; Kalippke, K.; Scholz, K.; Wilke, N.; Schmidt, R.E.; Jacobs, R. Depletion of functionally active CD20+ T cells by rituximab treatment. Arthritis Rheumatol. 2009, 60, 3563–3571. [Google Scholar] [CrossRef] [PubMed]
- Forster, F.; Singla, A.; Arora, S.K.; Schmidt, R.E.; Jacobs, R. CD20+ T cell numbers are decreased in untreated HIV-1 patients and recover after HAART. Immunol. Lett. 2012, 146, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Schuh, E.; Berer, K.; Mulazzani, M.; Feil, K.; Meinl, I.; Lahm, H.; Krane, M.; Lange, R.; Pfannes, K.; Subklewe, M.; et al. Features of Human CD3+CD20+ T Cells. J. Immunol. 2016, 197, 1111–1117. [Google Scholar] [CrossRef] [PubMed]
- Holley, J.E.; Bremer, E.; Kendall, A.C.; de Bruyn, M.; Helfrich, W.; Tarr, J.M.; Newcombe, J.; Gutowski, N.J.; Eggleton, P. CD20+ inflammatory T-cells are present in blood and brain of multiple sclerosis patients and can be selectively targeted for apoptotic elimination. Mult. Scler. Relat. Disord. 2014, 3, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Palanichamy, A.; Jahn, S.; Nickles, D.; Derstine, M.; Abounasr, A.; Hauser, S.L.; Baranzini, S.E.; Leppert, D.; von Budingen, H.C. Rituximab efficiently depletes increased CD20-expressing T cells in multiple sclerosis patients. J. Immunol. 2014, 193, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Klein, C.; Lammens, A.; Schafer, W.; Georges, G.; Schwaiger, M.; Mossner, E.; Hopfner, K.P.; Umana, P.; Niederfellner, G. Epitope interactions of monoclonal antibodies targeting CD20 and their relationship to functional properties. MAbs 2013, 5, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Kappos, L.; Li, D.; Calabresi, P.A.; O’Connor, P.; Bar-Or, A.; Barkhof, F.; Yin, M.; Leppert, D.; Glanzman, R.; Tinbergen, J.; et al. Ocrelizumab in relapsing-remitting multiple sclerosis: A phase 2, randomised, placebo-controlled, multicentre trial. Lancet 2011, 378, 1779–1787. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gingele, S.; Jacobus, T.L.; Konen, F.F.; Hümmert, M.W.; Sühs, K.-W.; Schwenkenbecher, P.; Ahlbrecht, J.; Möhn, N.; Müschen, L.H.; Bönig, L.; et al. Ocrelizumab Depletes CD20+ T Cells in Multiple Sclerosis Patients. Cells 2019, 8, 12. https://doi.org/10.3390/cells8010012
Gingele S, Jacobus TL, Konen FF, Hümmert MW, Sühs K-W, Schwenkenbecher P, Ahlbrecht J, Möhn N, Müschen LH, Bönig L, et al. Ocrelizumab Depletes CD20+ T Cells in Multiple Sclerosis Patients. Cells. 2019; 8(1):12. https://doi.org/10.3390/cells8010012
Chicago/Turabian StyleGingele, Stefan, Thais Langer Jacobus, Franz Felix Konen, Martin W. Hümmert, Kurt-Wolfram Sühs, Philipp Schwenkenbecher, Jonas Ahlbrecht, Nora Möhn, Lars H. Müschen, Lena Bönig, and et al. 2019. "Ocrelizumab Depletes CD20+ T Cells in Multiple Sclerosis Patients" Cells 8, no. 1: 12. https://doi.org/10.3390/cells8010012
APA StyleGingele, S., Jacobus, T. L., Konen, F. F., Hümmert, M. W., Sühs, K.-W., Schwenkenbecher, P., Ahlbrecht, J., Möhn, N., Müschen, L. H., Bönig, L., Alvermann, S., Schmidt, R. E., Stangel, M., Jacobs, R., & Skripuletz, T. (2019). Ocrelizumab Depletes CD20+ T Cells in Multiple Sclerosis Patients. Cells, 8(1), 12. https://doi.org/10.3390/cells8010012