Abstract
Transient receptor potential classical or canonical 4 (TRPC4) and TRPC5 channels are members of the classical or canonical transient receptor potential (TRPC) channel family of non-selective cation channels. TRPC4 and TRPC5 channels are widely accepted as receptor-operated cation channels that are activated in a phospholipase C-dependent manner, following the Gq/11 protein-coupled receptor activation. However, their precise activation mechanism has remained largely elusive for a long time, as the TRPC4 and TRPC5 channels were considered as being insensitive to the second messenger diacylglycerol (DAG) in contrast to the other TRPC channels. Recent findings indicate that the C-terminal interactions with the scaffolding proteins Na+/H+ exchanger regulatory factor 1 and 2 (NHERF1 and NHERF2) dynamically regulate the DAG sensitivity of the TRPC4 and TRPC5 channels. Interestingly, the C-terminal NHERF binding suppresses, while the dissociation of NHERF enables, the DAG sensitivity of the TRPC4 and TRPC5 channels. This leads to the assumption that all of the TRPC channels are DAG sensitive. The identification of the regulatory function of the NHERF proteins in the TRPC4/5-NHERF protein complex offers a new starting point to get deeper insights into the molecular basis of TRPC channel activation. Future studies will have to unravel the physiological and pathophysiological functions of this multi-protein channel complex.
1. Introduction
Transient receptor potential classical or canonical 4 (TRPC4) and TRPC5 channels belong to the transient receptor potential classical or canonical (TRPC) cation channel subfamily, which comprises seven members. TRPC channels are regarded as non-selective, receptor-operated cation channels that are important for calcium homeostasis. They are activated via the Gq/11-signaling cascade as a function of phospholipase C (PLC) [1]. Moreover, the TRPC4 and TRPC5 channels can also be activated following the Gi/o-protein coupled receptor activation [2,3,4]. However, the mechanism of the Gi/o-mediated TRPC4/5 channel activation is still not completely understood and might involve either a direct Gi/o-protein interaction [3], or the activation of the PLC isoform, PLCδ1 [2]. Even a Gs-protein mediated activation mechanism was proposed for the TRPC5 channels [5]. Therefore, elevating the intracellular cyclic adenosine monophosphate (cAMP) levels might potentiate the TRPC5 currents by increasing the channel trafficking to the plasma membrane. Additionally, a store-operated activation mechanism for the TRPC4 [6,7,8,9] and TRPC5 channels [10,11,12] was proposed, and is still under discussion [13,14]. This article will only focus on the Gq/11-protein mediated signaling pathway leading to the TRPC4 and TRPC5 channel activation.
All of the TRPC channels are tetramers formed by four TRPC protein subunits, which was confirmed recently by structural analysis [15,16,17,18]. TRPC proteins can form not only homotetrameric but also heterotetrameric cation channels [19,20,21,22] with distinct channel properties, such as altered calcium permeability [21,23,24,25]. Because of their sequence homology, the TRPC channel family can be divided into the following subgroups: TRPC1, TRPC4/5, and TRPC3/6/7. TRPC2 has a special role as it represents a pseudogene in humans that is not functionally expressed because of several stop codons in the open reading frame [26,27]. However, in rodents, the TRPC2 channels are functionally expressed (e.g., in the olfactory cells of the vomeronasal organ, where they are important for pheromone sensing [28]).
Although the Gq/11-protein mediated TRPC channel activation is widely accepted, the precise signaling pathway resulting in the channel opening has remained elusive for quite some time. The activation of the Gq/11-protein coupled receptors leads to the activation of PLC, which cleaves phosphatidylinositol-4,5-bisphosphate (PIP2) into the second messengers, inositol-1,4,5-trisphopshate (IP3) and 1,2-diacyl-sn-glycerol (DAG), and to an oxonium ion [29,30]. There is broad agreement that the TRPC3/6/7 subfamily is directly activated by DAG, the cleavage product of PIP2 [31]. However, IP3 might also contribute to the activation of endogenously expressed TRPC7 channels [32]. Notably, a DAG binding site has not been identified until now, and it is not clear whether DAG directly activates the channel or whether it first interacts with an additional protein, which in turn causes the channel activation. Unfortunately, the recent structural analysis of the TRPC3, TRPC4, and TRPC6 channels only displays a closed channel conformation [15,16,17,18]. However, the structural model of TRPC3 reveals two lipid-binding sites, one being sandwiched between the pre-S1 elbow and the S4–S5 linker, and the other being close to the pore-forming domain, where the conserved “LWF” motif of the TRPC family is located [16]. Perhaps these lipid binding sites reflect potential DAG binding sites. Interestingly, it was recently reported that the exchange of a highly conserved amino acid located close to the pore-forming domain, affects the DAG recognition of the TRPC3 channels [33]. Thus, the second lipid binding site might indeed reflect a potential DAG binding site. TRPC2 channels are also regarded as being DAG sensitive [34,35]. In contrast to the TRPC2, TRPC3, TRPC6, and TRPC7 channels, the TRPC4 and TRPC5 channels were commonly considered as DAG insensitive [31], as DAG or the membrane permeable DAG analogue 1-oleoyl-2-acetyl-sn-glycerol (OAG) even inhibited the basal TRPC5 currents [36]. Interestingly, Venkatachalam and colleagues showed that the DAG-induced TRPC5 channel inhibition is related to the protein kinase C (PKC) activation [36]. Moreover, it was demonstrated that the homotetrameric TRPC4 and TRPC5 channels are activated by PIP2 depletion [37,38]. In contrast, the heterotetrameric TRPC1/4 and TRPC1/5 [39] and homotetrameric TRPC6 and TRPC7 channels [40] are instead inhibited by the PIP2 depletion. These contradictory findings suggest that the TRPC channel-lipid interaction is rather complex. Another unique structural feature of the TRPC4 and TRPC5 channels is their capability to interact with the PDZ I domain of the scaffolding proteins Na+/H+ exchanger regulatory factor 1 and 2 (NHERF1 and NHERF2) [41,42,43]. The NHERF1 and NHERF2 proteins are structurally related; can form homodimers [44]; and possess two PDZ binding domains as well as a C-terminal binding domain, which enables crosslinking with the actin cytoskeleton via ezrin, radixin, and moesin proteins [41,45]. Thus, NHERF1 and NHERF2 proteins are commonly regarded as adapter proteins that crosslink integral membrane proteins with the cytoskeleton, thereby increasing their membrane localization [46,47,48].
2. DAG-Mediated Activation Mechanism of TRPC4 and TRPC5 Channels
The first evidence that the TRPC5 channels might be DAG sensitive was presented by Lee and colleagues, who performed electrophysiological whole-cell measurements on murine gastric smooth muscle cells endogenously expressing TRPC5 channels, and found that the channels are activated by OAG [49]. However, the mechanistic insights into the DAG mediated TRPC5 channel activation were missing. A remarkable structural difference between TRPC4 and TRPC5, and the well characterized DAG sensitive TRPC3/6/7 channels, is the PDZ binding motif with the amino acid sequence “VTTRL” at the very end of the C-terminus [41,42,50]. This PDZ binding motif includes a potential PKC phosphorylation site, which was identified as being important for the TRPC5 current inactivation following the receptor activation [51]. The amino acid exchange from threonine to alanine at position 972 (T972A) in the murine TRPC5 channels resulted in a loss of current desensitization during the receptor activation with carbachol [51]. Thus, this was the first evidence that the PKC phosphorylation might regulate the TRPC5 channel function [36,51].
The C-terminal PDZ binding motif allows for interactions with the adapter proteins NHERF1 and NHERF2. This C-terminal TRPC4/5 interaction was demonstrated several times by performing co-immunoprecipitations and functional studies using the patch-clamp technique [41,42,43]. However, only a slight enhancement of the membrane expression was found to be similar to what was observed when analyzing the chloride channel cystic fibrosis transmembrane conductance regulator (CFTR) [52] and other integral membrane proteins. Thus, the functional implications of the TRPC4/5-NHERF interaction were missing. Interestingly, our recent findings indicate that the TRPC4 and TRPC5 channels are DAG-sensitive similar to other TRPC channels [53]. However, in contrast to TRPC3/6/7, their DAG-sensitivity is tightly regulated by the C-terminal NHERF1 and NHERF2 interaction. The Gq/11-protein coupled receptor activation causes a cleavage of PIP2, resulting in a conformational change of the C-terminus, which in turn causes the dissociation of the NHERF proteins from the C-terminus of the channel. This dynamic dissociation was monitored employing the method of dynamic intermolecular fluorescence resonance energy transfer (FRET) between fluorescence tagged TRPC5 and NHERF proteins. The separation of the NHERF proteins from the C-terminus was a prerequisite for DAG sensitivity. Moreover, the C-terminal NHERF interaction strongly depended on the PKC phosphorylation status of the C-terminal PDZ binding motif “VTTRL” [53]. The PKC inhibition resulted in the DAG sensitivity, and the PKC phosphorylation mutant T972A of murine TRPC5 was sensitive to DAG, suggesting that PKC phosphorylation is a prerequisite for NHERF binding, thereby suppressing the DAG sensitivity. Thus, the NHERF proteins are dynamic regulators of the TRPC4 and TRPC5 channel activity. This signaling pathway was also observed in the primary cell lines (e.g., in proximal tubule cells and in hippocampal neurons, which endogenously express TRPC4 and TRPC5 channels, respectively) [53]. This activation model has the potential to integrate the contradictory findings of different research groups concerning the TRPC4 and TRPC5 channel activation. Therefore, the pieces of the puzzle like the PLC dependent receptor-mediated TRPC4/5 channel activation [1], the inhibitory effect of DAG or DAG analogues [31,36] on the native TRPC4/5-NHERF-channel complex [41,42,43], the activation by PIP2 depletion [37,38], the inhibitory effect of PKC phosphorylation on DAG sensitivity [36], and the DAG sensitivity of the endogenous gastric TRPC5 channels [49] coalesce into a consistent picture. These findings lead to a new concept, that all of the TRPC channels are DAG-sensitive, and that classifying the TRPC channels as DAG-sensitive and -insensitive channels should be avoided. Consequently, a common DAG binding site for TRPC channels can be proposed. Perhaps the highly conserved amino acid near the TRPC pore domain that affects the DAG activation [33] participates in DAG binding.
Altogether, a new model of Gq/11-protein mediated TRPC4 and TRPC5 channel activation can be proposed. The agonist-induced Gq/11-protein coupled receptor activation causes the activation of PLC, which in turn cleaves PIP2 into the two second messengers IP3 and DAG. The PIP2 cleavage causes a conformational change at the C-terminus of TRPC4 and TRPC5, which results in a dissociation of the NHERF proteins from the C-terminus, thereby evoking a DAG-sensitive channel conformation. Then, the PIP2 cleavage product DAG can activate the channel. This model is illustrated in Figure 1.
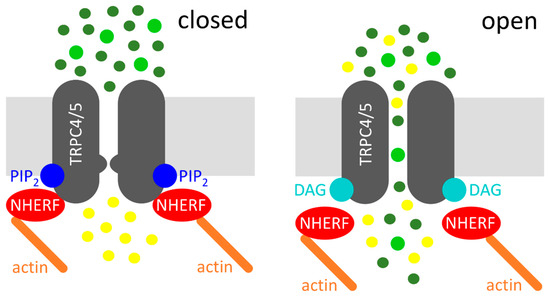
Figure 1.
Diacylglycerol (DAG)-mediated activation of transient receptor potential classical or canonical 4/5 (TRPC4/5) channels. Left: Na+/H+ exchanger regulatory factor (NHERF) proteins and phosphatidylinositol-4,5-bisphosphate (PIP2) interact with the C-termini of TRPC4/5, which depicts the closed state of the channel. Right: receptor activation (not displayed) leads to the cleavage of PIP2, resulting in the dissociation of NHERF and in DAG binding, which represents the open state of the channel. Sodium cations (dark green circles), calcium cations (light green circles), and potassium cations (yellow circles) are displayed. The potassium efflux is mainly relevant in the excitable cells.
3. Physiological and Pathophysiological Roles of NHERF Proteins
NHERF1 and NHERF2 proteins belong to a family of scaffolding proteins that crosslink the integral membrane proteins with the cytoskeleton. It is commonly accepted that NHERF proteins increase the membrane localization of several membrane proteins, like transporters, receptors, and ion channels [46,47,48]. However, besides their anchoring function, the NHERF proteins are of the utmost importance for the maintenance of essential cellular functions (e.g., in the kidney or in the small intestine, where they interact with transporters, ion channels, signaling proteins, transcription factors, enzymes, G-protein coupled receptors, and tyrosine kinase receptors) [47,48,54,55,56]. Thus, NHERF proteins are involved in numerous physiological processes. For example, in proximal tubule cells, NHERF proteins regulate phosphate transport [57]. A mutation in the PDZ I domain of human NHERF1 was found in patients with impaired renal phosphate reabsorption due to a reduced expression of the renal phosphate transporter NPT2a [58]. In astrocytes, NHERF proteins regulate the activity of the glutamate transporter (GLAST) and of the metabotropic glutamate receptor (mGlu5) [59,60], and in the small intestine, they control ion transport via interactions with the Na+/H+ exchanger (NHE3) [61]. Moreover, the mice lacking NHERF1 and adult humans harboring NHERF1 mutations suffer from osteopenia [62,63], which might be due to abnormal osteoblast differentiation [64]. Furthermore, NHERF proteins influence proliferation [65,66], and they may be involved in carcinogenesis and in the progression of liver, breast, and colon cancer; small-cell lung carcinoma; and glioblastoma [67,68,69,70]. A mutation in the PDZ I domain of NHERF1 was identified in the patients with medullar breast carcinoma [71]. This mutation resulted in a reduced interaction of NHERF1 with the epidermal growth factor receptor, thereby promoting the progression of breast cancer. Another mutation in the PDZ II domain of human NHERF1 resulted in a nuclear translocation of NHERF1, thereby inducing carcinogenesis [72]. Recently, in the tumors from ovarian cancer patients, a mutation in the PDZ II domain of NHERF1 was identified, which might contribute to the disease progression [73]. These data suggest that wild-type NHERF1 may act as a tumor suppressor. The subcellular NHERF expression also plays a critical role in cancer cells. In breast cancer cells, the subcellular NHERF expression might even serve as a prognostic marker, as a high cytoplasmic expression of NHERF was associated with a high aggressiveness and poor prognosis [74]. Furthermore, aberrant nuclear NHERF1 expression might be important for the carcinogenesis and progression of colon [74] and breast cancer [72].
The specific role of NHERF proteins for channel function is poorly understood. Beside the regulatory role of NHERF proteins on TRPC4 and TRPC5 channel function [53], the interaction with NHERF was identified as playing an important role in the proper function of the CFTR chloride channel [52]. Mutations in the CFTR channel are known to cause cystic fibrosis [75], which is characterized by the accumulation of viscous mucus, because of impaired fluid transport. Furthermore, CFTR mutations can cause congenital absence of the vas deferens and male infertility [76]. Interestingly, the NHERF2 interaction with the lysophosphatidic acid receptor 2 (LPAR2) promotes the assembly of CFTR–NHERF2–LPAR2 protein complexes, which results in an impaired CFTR function [77,78]. The disruption of this protein complex might enhance the CFTR function of patients suffering from cystic fibrosis [52]. Altogether, the NHERF proteins or their protein complexes might be interesting novel targets for the treatment of diseases
4. Physiological and Pathophysiological Roles of TRPC4 and TRPC5 Channels
TRPC4 and TRPC5 channels are expressed in several cells and tissues (e.g., in the brain [79,80,81,82,83], kidney [80,84,85], and vascular system [7,8,86]). In particular, the TRPC4 and TRPC5 channel expression is very high in the central nervous system [7,79,82]. Here, the TRPC4 and TRPC5 channels are involved in the amygdala function and account for fear-related behavior against aversive stimuli [87,88]. In addition, the TRPC4 and TRPC5 channels are important for peripherally induced neuropathic pain behavior. Microinjections of the TRPC4 and TRPC5 channel blocker ML-204 into the amygdala of rats reduced the sensory and the affective pain sensitivity [89]. Thus, in the future, TRPC4/5 blockers that are able to cross the blood–brain barrier might be used as novel anxiolytics or even as innovative analgesics against peripheral neuropathies. Furthermore, the TRPC4 and TRPC5 channels are expressed in the dorsal root ganglia, where they contribute to axonal regeneration after nerve injury [90], to itching [91], and to cold detection [92].
Moreover, in the hippocampal CA1 pyramidal cells from rats, the calcium and sodium influx through the TRPC5 channels generated a plateau potential [93] that is also observed during epileptic seizures [94,95]. In accordance with this neurophysiological evidence, the TRPC5 gene-deficient mice exhibited significantly reduced seizures. Thus, future studies will have to show whether TRPC5 channels represent interesting novel target structures for the treatment of epileptic disorders.
Furthermore, the TRPC5 channels reduce the hippocampal neurite length and growth cone morphology by reducing the filopodia length growth, which leads to impaired axon guidance [96]. The TRPC5 channels also play a role in podocytes and in fibroblasts. Interestingly, in these cells, the TRPC5 and TRPC6 channels have opposite effects on the actin cytoskeleton [97]. The receptor-operated TRPC5 channel activation by angiotensin II decreased the number of parallel stress fibers via the activation of the small guanosine-5′-triphosphate (GTP)ase protein Rac1, leading to a motile and non-contractile phenotype in vitro, while the TRPC6 activation by angiotensin II increased the formation of parallel stress fibers via the activation of the small GTPase protein Rho A, resulting in a contractile but non-motile phenotype [97]. These differential channel functions might be due to a specific subcellular localization of TRPC5 and TRPC6 channels in podocytes, or be caused by a distinct signaling elicited by the podocyte-specific TRPC6 and slit membrane protein channel complex [98,99]. The reorganization of the actin cytoskeleton characterized by a reduction of the parallel stress fibers results in podocyte injury, leading to the loss of podocyte foot processes, which in the end results in the disruption of the slit diaphragm and in massive proteinuria [100,101,102]. Thus, the TRPC5 channel blockers might be useful for the treatment of podocyte diseases by preventing end-stage renal disease [103,104].
In addition, the TRPC4 and TRPC5 channels might play a pathophysiological role in cancer cells. The increased TRPC5 channel activity in breast cancer [105] and in colorectal cancer cells [106] caused an increased expression of the ABC transporter P-glycoprotein (MDR1). MDR1 is an important molecular correlate for drug-resistance against chemotherapeutic agents. For example, MDR1 eliminates the well-known and commonly used DNA intercalating drug doxorubicin, the tubulin-targeting drug paclitaxel, and the antimetabolite 5-fluorouracil. In breast cancer cells, the transcription factor NFATc3, and in colorectal cancer cells, the structural protein and transcription factor β-catenin, are thought to enhance the MDR1 expression [105,106]. In contrast, the potent TRPC4 and TRPC5 channel activator (-)-englerin A showed pronounced cytotoxic effects on diverse cancer cell lines, with an EC50 value of ~20 nM [107,108]. (-)-englerin A was even effective on triple-negative breast cancer cells [109], which do not express the drug targets of estrogen, progesterone, and human epithelial growth factor (HER2) receptor. Of all breast cancer patients, 15% have triple-negative breast cancer [110], which is regarded as very aggressive [111]. The main treatment is surgery, but specific targets for target-orientated chemotherapeutic agents are missing, and patients suffer from frequent relapses within the first three years [112]. A more targeted medical treatment would be of great benefit for these patients. Notably, although (-)-englerin A was selectively cytotoxic to cancer cell lines, adverse reactions were observed in mice and rats after (-)-englerin A injections [108], which were mediated by the TRPC4/5 channels [113]. Thus, the therapeutic application of (-)-englerin A might be limited.
There is evidence that the TRPC5 channel activity increases angiogenesis in cases of breast cancer by the activation of the transcription factor hypoxia-inducible factor 1 (HIF-1), leading to vascular endothelial growth factor (VEGF) formation [114], thereby promoting cancer growth. In contrast, it was reported that the TRPC4 channel activation reduces angiogenesis in cases of renal cell carcinoma cells by the secretion of the inhibitor of angiogenesis thrombospondin-1 [115]. Moreover, the TRPC4 and TRPC5 channel activation in the endothelial cells increases vasculogenesis [116,117], indicative of a pro-angiogenetic effect of these channels. Furthermore, the increased expression of TRPC1, TRPC3, TRPC4, and TRPC6 in ovarian cancer cells increased the migration and proliferation, and therefore had a tumorigenic effect [118]. Thus, other TRPC channels, like TRPC1, TRPC3, and TRPC6, might also function as targets for chemotherapeutic agents. However, at present, the majority of publications point to the TRPC4 and TRPC5 channels as potential new drug targets [119].
The novel role of the NHERF adapter proteins as dynamic regulators of the TRPC4 and TRPC5 channel activity [53,120] might also be important for several other physiological or pathophysiological processes. Interestingly, the NHERF1 protein/channel interaction is of the utmost importance for the proper function of CFTR channels. The NHERF1 proteins stabilized and enhanced the membrane expression of the misfolded CFTR mutant channels [121], which partially restored the channel activity after the NHERF binding.
The TRPC4 and TRPC5 channels, and the NHERF proteins are co-expressed in various excitable and non-excitable cells and tissues. Hence, it can be speculated that the inhibitory effect of the NHERF interaction on the TRPC4/5 channel function may contribute to several physiological or pathophysiological conditions. For example, in tumor cells, the TRPC4/5 channels and the NHERF proteins and NHERF mutations account for cancer progression. However, the effect of the TRPC4/5-NHERF protein complex on tumor growth has largely remained elusive until now. The NHERF interaction also inhibited the DAG mediated TRPC4/5 channel activation in murine hippocampal neurons and in proximal tubule cells [53], suggesting a regulatory role. Altogether, further studies will be needed to show whether the TRPC4/5-NHERF protein complexes have the potential to serve as novel target structures for therapeutics.
5. Conclusions
The new concept that all TRPC channels are DAG sensitive is not only a new starting point for deeper insights into the activation mechanism of TRPC channels on a molecular level, but it might also help to unravel the physiological and pathophysiological roles of these channels, which have not been fully understood until now. On the molecular level, the conformational changes and the kinetics of the conformational changes leading to the TRPC4 and TRPC5 channel activation are largely elusive, as a structure analysis only revealed the inactive channel states. Moreover, the role of the NHERF proteins as dynamic regulators of the TRPC4 and TRC5 channel activation sheds new light on the function of ion channels and adapter proteins in multi-protein complexes. In the TRPC4/5-NHERF protein complexes, the NHERF proteins suppress the DAG sensitivity. Thus, for screening purposes, wildtype TRPC5 channels as well as DAG sensitive PKC phosphorylation site mutant T972A, which cannot interact with NHERF, might be used. Without the inhibitory NHERF binding, it can be speculated that other hits will be identified. As the expression pattern of NHERF proteins and TRPC4/5 channels is altered in several cancer cell lines and might be linked to cancer progression, the TRPC4/5–NHERF channel complexes should also be reconsidered as potential novel targets for cancer therapeutics. Future studies will have to unravel the physiological and pathophysiological roles of these channel complexes. However, a thorough analysis of the TRPC channel functions on a molecular level is of the utmost importance to lay the foundation for a better understanding of the role of TRPC channels in health and disease.
Author Contributions
U.S. and M.M.y.S. planned the review; U.S. performed the literature searches; U.S., M.M.y.S., and T.G. wrote the review; M.M.y.S. created Figure 1.
Funding
This work was supported by the German Research Foundation (Deutsche Forschungsgemeinschaft) (project no. 406028471 and TRR-152).
Acknowledgments
We thank Anna-Lena Forst for her excellent work, and we thank Laura Danner for her brilliant technical support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Rohacs, T. Regulation of transient receptor potential channels by the phospholipase c pathway. Adv. Biol. Reg. 2013, 53, 341–355. [Google Scholar] [CrossRef] [PubMed]
- Thakur, D.P.; Tian, J.B.; Jeon, J.; Xiong, J.; Huang, Y.; Flockerzi, V.; Zhu, M.X. Critical roles of gi/o proteins and phospholipase c-delta1 in the activation of receptor-operated TRPC4 channels. Proc. Natl. Acad. Sci. USA 2016, 113, 1092–1097. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.P.; Hong, C.; Park, E.J.; Jeon, J.H.; Cho, N.H.; Kim, I.G.; Choe, H.; Muallem, S.; Kim, H.J.; So, I. Selective Gαi subunits as novel direct activators of transient receptor potential canonical (TRPC)4 and TRPC5 channels. J. Biol. Chem. 2012, 287, 17029–17039. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.P.; Lee, K.P.; Park, E.J.; Sung, T.S.; Kim, B.J.; Jeon, J.H.; So, I. The specific activation of TRPC4 by Gi protein subtype. Biochem. Biophys. Res. Commun. 2008, 377, 538–543. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.; Kim, J.; Jeon, J.P.; Wie, J.; Kwak, M.; Ha, K.; Kim, H.; Myeong, J.; Kim, S.Y.; Jeon, J.H.; et al. Gs cascade regulates canonical transient receptor potential 5 (TRPC5) through camp mediated intracellular Ca2+ release and ion channel trafficking. Biochem. Biophys. Res. Commun. 2012, 421, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Philipp, S.; Trost, C.; Warnat, J.; Rautmann, J.; Himmerkus, N.; Schroth, G.; Kretz, O.; Nastainczyk, W.; Cavalie, A.; Hoth, M.; et al. TRP4 (CCE1) protein is part of native calcium release-activated Ca2+-like channels in adrenal cells. J. Biol. Chem. 2000, 275, 23965–23972. [Google Scholar] [CrossRef] [PubMed]
- Freichel, M.; Suh, S.H.; Pfeifer, A.; Schweig, U.; Trost, C.; Weissgerber, P.; Biel, M.; Philipp, S.; Freise, D.; Droogmans, G.; et al. Lack of an endothelial store-operated Ca2+ current impairs agonist-dependent vasorelaxation in TRP4-/- mice. Nat. Cell Biol. 2001, 3, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Tiruppathi, C.; Freichel, M.; Vogel, S.M.; Paria, B.C.; Mehta, D.; Flockerzi, V.; Malik, A.B. Impairment of store-operated Ca2+ entry in TRPC4(-/-) mice interferes with increase in lung microvascular permeability. Circ. Res. 2002, 91, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Pluznick, J.L.; Wei, P.; Padanilam, B.J.; Sansom, S.C. TRPC4 forms store-operated Ca2+ channels in mouse mesangial cells. Am. J. Physiol. Cell Physiol. 2004, 287, C357–C364. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.Z.; Boulay, G.; Flemming, R.; Beech, D.J. E3-targeted anti-TRPC5 antibody inhibits store-operated calcium entry in freshly isolated pial arterioles. Am. J. Physiol. Heart Circ. Physiol. 2006, 291, H2653–H2659. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.Y.; Thilo, F.; Scholze, A.; Wittstock, A.; Zhao, Z.G.; Harteneck, C.; Zidek, W.; Zhu, Z.M.; Tepel, M. Increased store-operated and 1-oleoyl-2-acetyl-sn-glycerol-induced calcium influx in monocytes is mediated by transient receptor potential canonical channels in human essential hypertension. J. Hypertens. 2007, 25, 799–808. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.; Xu, S.Z.; Jackson, P.K.; McHugh, D.; Kumar, B.; Fountain, S.J.; Beech, D.J. Human TRPC5 channel activated by a multiplicity of signals in a single cell. J. Physiol. 2004, 559, 739–750. [Google Scholar] [CrossRef] [PubMed]
- DeHaven, W.I.; Jones, B.F.; Petranka, J.G.; Smyth, J.T.; Tomita, T.; Bird, G.S.; Putney, J.W., Jr. TRPC channels function independently of STIM1 and Orai1. J. Physiol. 2009, 587, 2275–2298. [Google Scholar] [CrossRef] [PubMed]
- Putney, J.W.; Steinckwich-Besancon, N.; Numaga-Tomita, T.; Davis, F.M.; Desai, P.N.; D’Agostin, D.M.; Wu, S.; Bird, G.S. The functions of store-operated calcium channels. Biochim. Biophys. Acta Mol. Cell Res. 2017, 1864, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Li, J.; Zeng, B.; Chen, G.L.; Peng, X.; Zhang, Y.; Wang, J.; Clapham, D.E.; Li, Z.; Zhang, J. Structure of the mouse TRPC4 ion channel. Nat. Commun. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.; Choi, W.; Sun, W.; Du, J.; Lu, W. Structure of the human lipid-gated cation channel TRPC3. eLife 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q.; Guo, W.; Zheng, L.; Wu, J.X.; Liu, M.; Zhou, X.; Zhang, X.; Chen, L. Structure of the receptor-activated human TRPC6 and TRPC3 ion channels. Cell Res. 2018, 28, 746–755. [Google Scholar] [CrossRef] [PubMed]
- Vinayagam, D.; Mager, T.; Apelbaum, A.; Bothe, A.; Merino, F.; Hofnagel, O.; Gatsogiannis, C.; Raunser, S. Electron cryo-microscopy structure of the canonical TRPC4 ion channel. eLife 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Strübing, C.; Krapivinsky, G.; Krapivinsky, L.; Clapham, D.E. TRPC1 and TRPC5 form a novel cation channel in mammalian brain. Neuron 2001, 29, 645–655. [Google Scholar] [CrossRef]
- Strübing, C.; Krapivinsky, G.; Krapivinsky, L.; Clapham, D.E. Formation of novel TRPC channels by complex subunit interactions in embryonic brain. J. Biol. Chem. 2003, 278, 39014–39019. [Google Scholar] [CrossRef] [PubMed]
- Storch, U.; Forst, A.L.; Philipp, M.; Gudermann, T.; Mederos y Schnitzler, M. Transient receptor potential channel 1 (TRPC1) reduces calcium permeability in heteromeric channel complexes. J. Biol. Chem. 2012, 287, 3530–3540. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, T.; Schaefer, M.; Schultz, G.; Gudermann, T. Subunit composition of mammalian transient receptor potential channels in living cells. Proc. Natl. Acad. Sci. USA 2002, 99, 7461–7466. [Google Scholar] [CrossRef] [PubMed]
- Medic, N.; Desai, A.; Olivera, A.; Abramowitz, J.; Birnbaumer, L.; Beaven, M.A.; Gilfillan, A.M.; Metcalfe, D.D. Knockout of the Trpc1 gene reveals that TRPC1 can promote recovery from anaphylaxis by negatively regulating mast cell TNF-α production. Cell Calcium 2013, 53, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kwak, M.; Jeon, J.P.; Myeong, J.; Wie, J.; Hong, C.; Kim, S.Y.; Jeon, J.H.; Kim, H.J.; So, I. Isoform- and receptor-specific channel property of canonical transient receptor potential (TRPC)1/4 channels. Pflugers Arch. 2014, 466, 491–504. [Google Scholar] [CrossRef] [PubMed]
- Erac, Y.; Selli, C.; Tosun, M. TRPC1 ion channel gene regulates store-operated calcium entry and proliferation in human aortic smooth muscle cells. Turk. J. Biol. 2016, 40, 1336–1344. [Google Scholar] [CrossRef]
- Vannier, B.; Peyton, M.; Boulay, G.; Brown, D.; Qin, N.; Jiang, M.; Zhu, X.; Birnbaumer, L. Mouse trp2, the homologue of the human trpc2 pseudogene, encodes mtrp2, a store depletion-activated capacitative Ca2+ entry channel. Proc. Natl. Acad. Sci. USA 1999, 96, 2060–2064. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, E.; Dietrich, A.; Birnbaumer, L. The mouse c-type transient receptor potential 2 (TRPC2) channel: Alternative splicing and calmodulin binding to its n terminus. Proc. Natl. Acad. Sci. USA 2003, 100, 2220–2225. [Google Scholar] [CrossRef] [PubMed]
- Liman, E.R.; Dulac, C. TRPC2 and the molecular biology of pheromone detection in mammals. In TRP Ion Channel Function in Sensory Transduction and Cellular Signaling Cascades; Liedtke, W.B., Heller, S., Eds.; Taylor and Francis: Boca Raton, FL, USA, 2007; ISBN 9781420005844. [Google Scholar]
- Gudermann, T.; Mederos y Schnitzler, M. Phototransduction: Keep an eye out for acid-labile TRPs. Curr. Biol. 2010, 20, R149–R152. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Liu, C.H.; Hughes, S.A.; Postma, M.; Schwiening, C.J.; Hardie, R.C. Activation of TRP channels by protons and phosphoinositide depletion in drosophila photoreceptors. Curr. Biol. 2010, 20, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, T.; Obukhov, A.G.; Schaefer, M.; Harteneck, C.; Gudermann, T.; Schultz, G. Direct activation of human TRPC6 and TRPC3 channels by diacylglycerol. Nature 1999, 397, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Vazquez, G.; Bird, G.S.; Mori, Y.; Putney, J.W., Jr. Native TRPC7 channel activation by an inositol trisphosphate receptor-dependent mechanism. J. Biol. Chem. 2006, 281, 25250–25258. [Google Scholar] [CrossRef] [PubMed]
- Lichtenegger, M.; Tiapko, O.; Svobodova, B.; Stockner, T.; Glasnov, T.N.; Schreibmayer, W.; Platzer, D.; de la Cruz, G.G.; Krenn, S.; Schober, R.; et al. An optically controlled probe identifies lipid-gating fenestrations within the TRPC3 channel. Nat. Chem. Biol. 2018, 14, 396–404. [Google Scholar] [CrossRef] [PubMed]
- Lucas, P.; Ukhanov, K.; Leinders-Zufall, T.; Zufall, F. A diacylglycerol-gated cation channel in vomeronasal neuron dendrites is impaired in TRPC2 mutant mice: Mechanism of pheromone transduction. Neuron 2003, 40, 551–561. [Google Scholar] [CrossRef]
- Leinders-Zufall, T.; Storch, U.; Bleymehl, K.; Mederos, Y.S.M.; Frank, J.A.; Konrad, D.B.; Trauner, D.; Gudermann, T.; Zufall, F. PhoDAGs enable optical control of diacylglycerol-sensitive transient receptor potential channels. Cell Chem. Biol. 2018, 25, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Venkatachalam, K.; Zheng, F.; Gill, D.L. Regulation of canonical transient receptor potential (TRPC) channel function by diacylglycerol and protein kinase c. J. Biol. Chem. 2003, 278, 29031–29040. [Google Scholar] [CrossRef] [PubMed]
- Otsuguro, K.; Tang, J.; Tang, Y.; Xiao, R.; Freichel, M.; Tsvilovskyy, V.; Ito, S.; Flockerzi, V.; Zhu, M.X.; Zholos, A.V. Isoform-specific inhibition of TRPC4 channel by phosphatidylinositol 4,5-bisphosphate. J. Biol. Chem. 2008, 283, 10026–10036. [Google Scholar] [CrossRef] [PubMed]
- Trebak, M.; Lemonnier, L.; DeHaven, W.I.; Wedel, B.J.; Bird, G.S.; Putney, J.W., Jr. Complex functions of phosphatidylinositol 4,5-bisphosphate in regulation of TRPC5 cation channels. Pflugers Arch. 2009, 457, 757–769. [Google Scholar] [CrossRef] [PubMed]
- Myeong, J.; Ko, J.; Kwak, M.; Kim, J.; Woo, J.; Ha, K.; Hong, C.; Yang, D.; Kim, H.J.; Jeon, J.H.; et al. Dual action of the Gαq-PLCβ-PI(4,5)P2 pathway on TRPC1/4 and TRPC1/5 heterotetramers. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Itsuki, K.; Imai, Y.; Hase, H.; Okamura, Y.; Inoue, R.; Mori, M.X. Plc-mediated PI(4,5)P2 hydrolysis regulates activation and inactivation of TRPC6/7 channels. J. Gen. Physiol. 2014, 143, 183–201. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Tang, J.; Chen, Z.; Trost, C.; Flockerzi, V.; Li, M.; Ramesh, V.; Zhu, M.X. Association of mammalian trp4 and phospholipase c isozymes with a PDZ domain-containing protein, NHERF. J. Biol. Chem. 2000, 275, 37559–37564. [Google Scholar] [CrossRef] [PubMed]
- Obukhov, A.G.; Nowycky, M.C. TRPC5 activation kinetics are modulated by the scaffolding protein ezrin/radixin/moesin-binding phosphoprotein-50 (ebp50). J. Cell. Physiol. 2004, 201, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Lee-Kwon, W.; Wade, J.B.; Zhang, Z.; Pallone, T.L.; Weinman, E.J. Expression of TRPC4 channel protein that interacts with NHERF-2 in rat descending vasa recta. Am. J. Physiol. Cell Physiol. 2005, 288, C942–C949. [Google Scholar] [CrossRef] [PubMed]
- Shenolikar, S.; Minkoff, C.M.; Steplock, D.A.; Evangelista, C.; Liu, M.; Weinman, E.J. N-terminal pdz domain is required for NHERF dimerization. FEBS Lett. 2001, 489, 233–236. [Google Scholar] [CrossRef]
- Mamonova, T.; Kurnikova, M.; Friedman, P.A. Structural basis for NHERF1 PDZ domain binding. Biochemistry 2012, 51, 3110–3120. [Google Scholar] [CrossRef] [PubMed]
- Shenolikar, S.; Weinman, E.J. NHERF: Targeting and trafficking membrane proteins. Am. J. Physiol. Renal. Physiol. 2001, 280, F389–F395. [Google Scholar] [CrossRef] [PubMed]
- Hall, R.A.; Ostedgaard, L.S.; Premont, R.T.; Blitzer, J.T.; Rahman, N.; Welsh, M.J.; Lefkowitz, R.J. A C-terminal motif found in the beta2-adrenergic receptor, P2Y1 receptor and cystic fibrosis transmembrane conductance regulator determines binding to the Na+/H+ exchanger regulatory factor family of PDZ proteins. Proc. Natl. Acad. Sci. USA 1998, 95, 8496–8501. [Google Scholar] [CrossRef] [PubMed]
- Hall, R.A.; Premont, R.T.; Chow, C.W.; Blitzer, J.T.; Pitcher, J.A.; Claing, A.; Stoffel, R.H.; Barak, L.S.; Shenolikar, S.; Weinman, E.J.; et al. The beta2-adrenergic receptor interacts with the Na+/H+-exchanger regulatory factor to control Na+/H+ exchange. Nature 1998, 392, 626–630. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.M.; Kim, B.J.; Kim, H.J.; Yang, D.K.; Zhu, M.H.; Lee, K.P.; So, I.; Kim, K.W. TRPC5 as a candidate for the nonselective cation channel activated by muscarinic stimulation in murine stomach. Am. J. Physiol. Gastrointest. Liver Physiol. 2003, 284, G604–G616. [Google Scholar] [CrossRef] [PubMed]
- Mery, L.; Strauss, B.; Dufour, J.F.; Krause, K.H.; Hoth, M. The PDZ-interacting domain of TRPC4 controls its localization and surface expression in HEK293 cells. J. Cell Sci. 2002, 115, 3497–3508. [Google Scholar] [PubMed]
- Zhu, M.H.; Chae, M.; Kim, H.J.; Lee, Y.M.; Kim, M.J.; Jin, N.G.; Yang, D.K.; So, I.; Kim, K.W. Desensitization of canonical transient receptor potential channel 5 by protein kinase c. Am. J. Physiol. Cell Physiol. 2005, 289, C591–C600. [Google Scholar] [CrossRef] [PubMed]
- Holcomb, J.; Spellmon, N.; Trescott, L.; Sun, F.; Li, C.; Yang, Z. PDZ structure and implication in selective drug design against cystic fibrosis. Curr. Drug Targets 2015, 16, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Storch, U.; Forst, A.L.; Pardatscher, F.; Erdogmus, S.; Philipp, M.; Gregoritza, M.; Mederos y Schnitzler, M.; Gudermann, T. Dynamic NHERF interaction with TRPC4/5 proteins is required for channel gating by diacylglycerol. Proc. Natl. Acad. Sci. USA 2017, 114, E37–E46. [Google Scholar] [CrossRef] [PubMed]
- Hall, R.A.; Spurney, R.F.; Premont, R.T.; Rahman, N.; Blitzer, J.T.; Pitcher, J.A.; Lefkowitz, R.J. G protein-coupled receptor kinase 6a phosphorylates the na(+)/h(+) exchanger regulatory factor via a pdz domain-mediated interaction. J. Biol. Chem. 1999, 274, 24328–24334. [Google Scholar] [CrossRef] [PubMed]
- Maudsley, S.; Zamah, A.M.; Rahman, N.; Blitzer, J.T.; Luttrell, L.M.; Lefkowitz, R.J.; Hall, R.A. Platelet-derived growth factor receptor association with Na+/H+-exchanger regulatory factor potentiates receptor activity. Mol. Cell Biol. 2000, 20, 8352–8363. [Google Scholar] [CrossRef] [PubMed]
- Weinman, E.J.; Hall, R.A.; Friedman, P.A.; Liu-Chen, L.Y.; Shenolikar, S. The association of NHERF adaptor proteins with g protein-coupled receptors and receptor tyrosine kinases. Annu. Rev. Physiol. 2006, 68, 491–505. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, R.; Biswas, R.; Steplock, D.; Shenolikar, S.; Weinman, E. Role of NHERF and scaffolding proteins in proximal tubule transport. Urol. Res. 2010, 38, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Courbebaisse, M.; Leroy, C.; Bakouh, N.; Salaun, C.; Beck, L.; Grandchamp, B.; Planelles, G.; Hall, R.A.; Friedlander, G.; Prie, D. A New Human NHERF1 Mutation Decreases Renal Phosphate Transporter NPT2a Expression by a PTH-Independent Mechanism. PloS ONE 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Paquet, M.; Asay, M.J.; Fam, S.R.; Inuzuka, H.; Castleberry, A.M.; Oller, H.; Smith, Y.; Yun, C.C.; Traynelis, S.F.; Hall, R.A. The PDZ scaffold NHERF-2 interacts with mGluR5 and regulates receptor activity. J. Biol. Chem. 2006, 281, 29949–29961. [Google Scholar] [CrossRef] [PubMed]
- Ritter, S.L.; Asay, M.J.; Paquet, M.; Paavola, K.J.; Reiff, R.E.; Yun, C.C.; Hall, R.A. GLAST stability and activity are enhanced by interaction with the PDZ scaffold NHERF-2. Neurosci. Lett. 2011, 487, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Ghishan, F.K.; Kiela, P.R. Small intestinal ion transport. Curr. Opin. Gastroenterol. 2012, 28, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Weinman, E.J.; Mohanlal, V.; Stoycheff, N.; Wang, F.; Steplock, D.; Shenolikar, S.; Cunningham, R. Longitudinal study of urinary excretion of phosphate, calcium, and uric acid in mutant NHERF-1 null mice. Am. J. Physiol. Renal. Physiol. 2006, 290, F838–F843. [Google Scholar] [CrossRef] [PubMed]
- Karim, Z.; Gerard, B.; Bakouh, N.; Alili, R.; Leroy, C.; Beck, L.; Silve, C.; Planelles, G.; Urena-Torres, P.; Grandchamp, B.; et al. NHERF1 mutations and responsiveness of renal parathyroid hormone. N. Engl. J. Med. 2008, 359, 1128–1135. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Alonso, V.; Guo, L.; Tourkova, I.; Henderson, S.E.; Almarza, A.J.; Friedman, P.A.; Blair, H.C. Na+/H+ exchanger regulatory factor 1 (NHERF1) directly regulates osteogenesis. J. Biol. Chem. 2012, 287, 43312–43321. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, R.; Wang, E.; Dutta, S.K.; Vohra, P.K.; E, G.; Prakash, Y.S.; Mukhopadhyay, D. NHERF-2 maintains endothelial homeostasis. Blood 2012, 119, 4798–4806. [Google Scholar] [CrossRef] [PubMed]
- Kruger, W.A.; Monteith, G.R.; Poronnik, P. NHERF-1 regulation of EGF and neurotensin signalling in HT-29 epithelial cells. Biochem. Biophys. Res. Commun. 2013, 432, 568–573. [Google Scholar] [CrossRef] [PubMed]
- Voltz, J.W.; Weinman, E.J.; Shenolikar, S. Expanding the role of NHERF, a PDZ-domain containing protein adapter, to growth regulation. Oncogene 2001, 20, 6309–6314. [Google Scholar] [CrossRef] [PubMed]
- Georgescu, M.M.; Morales, F.C.; Molina, J.R.; Hayashi, Y. Roles of NHERF1/EBP50 in cancer. Curr. Mol. Med. 2008, 8, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Ritter, S.L.; Zhang, H.; Shim, H.; Hall, R.A.; Yun, C.C. MAGI-3 competes with NHERF-2 to negatively regulate LPA2 receptor signaling in colon cancer cells. Gastroenterology 2011, 140, 924–934. [Google Scholar] [CrossRef] [PubMed]
- Mangia, A.; Partipilo, G.; Schirosi, L.; Saponaro, C.; Galetta, D.; Catino, A.; Scattone, A.; Simone, G. Fine needle aspiration cytology: A tool to study NHERF1 expression as a potential marker of aggressiveness in lung cancer. Mol. Biotechnol. 2015, 57, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Du, G.; Hao, C.; Gu, Y.; Wang, Z.; Jiang, W.G.; He, J.; Cheng, S. A novel NHERF1 mutation in human breast cancer inactivates inhibition by NHERF1 protein in EGFR signaling. Anticancer Res. 2016, 36, 1165–1173. [Google Scholar] [PubMed]
- Yang, X.; Du, G.; Yu, Z.; Si, Y.; Martin, T.A.; He, J.; Cheng, S.; Jiang, W.G. A novel NHERF1 mutation in human breast cancer and effects on malignant progression. Anticancer Res. 2017, 37, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Kreimann, E.L.; Ratajska, M.; Kuzniacka, A.; Demacopulo, B.; Stukan, M.; Limon, J. A novel splicing mutation in the SLC9A3R1 gene in tumors from ovarian cancer patients. Oncology Lett. 2015, 10, 3722–3726. [Google Scholar] [CrossRef] [PubMed]
- Saponaro, C.; Malfettone, A.; Dell’Endice, T.S.; Brunetti, A.E.; Achimas-Cadariu, P.; Paradiso, A.; Mangia, A. The prognostic value of the Na+/H+ exchanger regulatory factor 1 (NHERF1) protein in cancer. Cancer Biomark. 2014, 14, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Guggino, W.B.; Stanton, B.A. New insights into cystic fibrosis: Molecular switches that regulate cftr. Nat. Rev. Mol. Cell Biol. 2006, 7, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Cuppens, H.; Cassiman, J.J. CFTR mutations and polymorphisms in male infertility. Int. J. Androl. 2004, 27, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Holcomb, J.; Jiang, Y.; Lu, G.; Trescott, L.; Brunzelle, J.; Sirinupong, N.; Li, C.; Naren, A.P.; Yang, Z. Structural insights into PDZ-mediated interaction of NHERF2 and LPA(2), a cellular event implicated in CFTR channel regulation. Biochem. Biophys. Res. Commun. 2014, 446, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Penmatsa, H.; Ren, A.; Punchihewa, C.; Lemoff, A.; Yan, B.; Fujii, N.; Naren, A.P. Functional regulation of cystic fibrosis transmembrane conductance regulator-containing macromolecular complexes: A small-molecule inhibitor approach. Biochem. J. 2011, 435, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Sossey-Alaoui, K.; Lyon, J.A.; Jones, L.; Abidi, F.E.; Hartung, A.J.; Hane, B.; Schwartz, C.E.; Stevenson, R.E.; Srivastava, A.K. Molecular cloning and characterization of TRPC5 (HTRP5), the human homologue of a mouse brain receptor-activated capacitative Ca2+ entry channel. Genomics 1999, 60, 330–340. [Google Scholar] [CrossRef] [PubMed]
- Philipp, S.; Hambrecht, J.; Braslavski, L.; Schroth, G.; Freichel, M.; Murakami, M.; Cavalie, A.; Flockerzi, V. A novel capacitative calcium entry channel expressed in excitable cells. EMBO J. 1998, 17, 4274–4282. [Google Scholar] [CrossRef] [PubMed]
- Philipp, S.; Cavalie, A.; Freichel, M.; Wissenbach, U.; Zimmer, S.; Trost, C.; Marquart, A.; Murakami, M.; Flockerzi, V. A mammalian capacitative calcium entry channel homologous to drosophila TRP and TRPL. EMBO J. 1996, 15, 6166–6171. [Google Scholar] [CrossRef] [PubMed]
- Mori, Y.; Takada, N.; Okada, T.; Wakamori, M.; Imoto, K.; Wanifuchi, H.; Oka, H.; Oba, A.; Ikenaka, K.; Kurosaki, T. Differential distribution of TRP Ca2+ channel isoforms in mouse brain. Neuroreport 1998, 9, 507–515. [Google Scholar] [PubMed]
- Munsch, T.; Freichel, M.; Flockerzi, V.; Pape, H.C. Contribution of transient receptor potential channels to the control of GABA release from dendrites. Proc. Natl. Acad. Sci. USA 2003, 100, 16065–16070. [Google Scholar] [CrossRef] [PubMed]
- Okada, T.; Shimizu, S.; Wakamori, M.; Maeda, A.; Kurosaki, T.; Takada, N.; Imoto, K.; Mori, Y. Molecular cloning and functional characterization of a novel receptor-activated TRP Ca2+ channel from mouse brain. J. Biol. Chem. 1998, 273, 10279–10287. [Google Scholar] [CrossRef] [PubMed]
- Turvey, M.R.; Wang, Y.; Gu, Y. The effects of extracellular nucleotides on [Ca2+]i signalling in a human-derived renal proximal tubular cell line (HKC-8). J. Cell. Biochem. 2010, 109, 132–139. [Google Scholar] [PubMed]
- Yip, H.; Chan, W.Y.; Leung, P.C.; Kwan, H.Y.; Liu, C.; Huang, Y.; Michel, V.; Yew, D.T.; Yao, X. Expression of TRPC homologs in endothelial cells and smooth muscle layers of human arteries. Histochem. Cell Biol. 2004, 122, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Riccio, A.; Li, Y.; Moon, J.; Kim, K.S.; Smith, K.S.; Rudolph, U.; Gapon, S.; Yao, G.L.; Tsvetkov, E.; Rodig, S.J.; et al. Essential role for TRPC5 in amygdala function and fear-related behavior. Cell 2009, 137, 761–772. [Google Scholar] [CrossRef] [PubMed]
- Riccio, A.; Li, Y.; Tsvetkov, E.; Gapon, S.; Yao, G.L.; Smith, K.S.; Engin, E.; Rudolph, U.; Bolshakov, V.Y.; Clapham, D.E. Decreased anxiety-like behavior and Gαq/11-dependent responses in the amygdala of mice lacking TRPC4 channels. J. Neurosci. 2014, 34, 3653–3667. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Sagalajev, B.; Yuzer, M.A.; Koivisto, A.; Pertovaara, A. Regulation of neuropathic pain behavior by amygdaloid TRPC4/C5 channels. Neurosci. Lett. 2015, 608, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Huang, W.; Richardson, P.M.; Priestley, J.V.; Liu, M. TRPC4 in rat dorsal root ganglion neurons is increased after nerve injury and is necessary for neurite outgrowth. J. Biol. Chem. 2008, 283, 416–426. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Cho, P.S.; Tonello, R.; Lee, H.K.; Jang, J.H.; Park, G.Y.; Hwang, S.W.; Park, C.K.; Jung, S.J.; Berta, T. Peripheral serotonin receptor 2b and transient receptor potential channel 4 mediate pruritus to serotonergic antidepressants in mice. J. Allergy Clin. Immunol. 2018, 142, 1349–1352. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, K.; Lennerz, J.K.; Hein, A.; Link, A.S.; Kaczmarek, J.S.; Delling, M.; Uysal, S.; Pfeifer, J.D.; Riccio, A.; Clapham, D.E. Transient receptor potential cation channel, subfamily c, member 5 (TRPC5) is a cold-transducer in the peripheral nervous system. Proc. Natl. Acad. Sci. USA 2011, 108, 18114–18119. [Google Scholar] [CrossRef] [PubMed]
- Tai, C.; Hines, D.J.; Choi, H.B.; MacVicar, B.A. Plasma membrane insertion of TRPC5 channels contributes to the cholinergic plateau potential in hippocampal CA1 pyramidal neurons. Hippocampus 2011, 21, 958–967. [Google Scholar] [CrossRef] [PubMed]
- Dichter, M.A.; Ayala, G.F. Cellular mechanisms of epilepsy: A status report. Science 1987, 237, 157–164. [Google Scholar] [CrossRef]
- Fraser, D.D.; MacVicar, B.A. Cholinergic-dependent plateau potential in hippocampal CA1 pyramidal neurons. J. Neurosci. 1996, 16, 4113–4128. [Google Scholar] [CrossRef] [PubMed]
- Greka, A.; Navarro, B.; Oancea, E.; Duggan, A.; Clapham, D.E. TRPC5 is a regulator of hippocampal neurite length and growth cone morphology. Nat. Neurosci. 2003, 6, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Tian, D.; Jacobo, S.M.; Billing, D.; Rozkalne, A.; Gage, S.D.; Anagnostou, T.; Pavenstadt, H.; Hsu, H.H.; Schlondorff, J.; Ramos, A.; et al. Antagonistic regulation of actin dynamics and cell motility by TRPC5 and TRPC6 channels. Sci. Signal. 2010, 3. [Google Scholar] [CrossRef] [PubMed]
- Huber, T.B.; Schermer, B.; Benzing, T. Podocin organizes ion channel-lipid supercomplexes: Implications for mechanosensation at the slit diaphragm. Nephron Exp. Nephrol. 2007, 106, e27–e31. [Google Scholar] [CrossRef] [PubMed]
- Huber, T.B.; Schermer, B.; Muller, R.U.; Hohne, M.; Bartram, M.; Calixto, A.; Hagmann, H.; Reinhardt, C.; Koos, F.; Kunzelmann, K.; et al. Podocin and MEC-2 bind cholesterol to regulate the activity of associated ion channels. Proc. Natl. Acad. Sci. USA 2006, 103, 17079–17086. [Google Scholar] [CrossRef] [PubMed]
- Takeda, T.; McQuistan, T.; Orlando, R.A.; Farquhar, M.G. Loss of glomerular foot processes is associated with uncoupling of podocalyxin from the actin cytoskeleton. J. Clin. Invest. 2001, 108, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Asanuma, K.; Yanagida-Asanuma, E.; Faul, C.; Tomino, Y.; Kim, K.; Mundel, P. Synaptopodin orchestrates actin organization and cell motility via regulation of RhoA signalling. Nat. Cell Biol. 2006, 8, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Faul, C.; Asanuma, K.; Yanagida-Asanuma, E.; Kim, K.; Mundel, P. Actin up: Regulation of podocyte structure and function by components of the actin cytoskeleton. Trends Cell Biol. 2007, 17, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Castonguay, P.; Sidhom, E.H.; Clark, A.R.; Dvela-Levitt, M.; Kim, S.; Sieber, J.; Wieder, N.; Jung, J.Y.; Andreeva, S.; et al. A small-molecule inhibitor of TRPC5 ion channels suppresses progressive kidney disease in animal models. Science 2017, 358, 1332–1336. [Google Scholar] [CrossRef] [PubMed]
- Schaldecker, T.; Kim, S.; Tarabanis, C.; Tian, D.; Hakroush, S.; Castonguay, P.; Ahn, W.; Wallentin, H.; Heid, H.; Hopkins, C.R.; et al. Inhibition of the TRPC5 ion channel protects the kidney filter. J. Clin. Invest. 2013, 123, 5298–5309. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Cai, Y.; He, D.; Zou, C.; Zhang, P.; Lo, C.Y.; Xu, Z.; Chan, F.L.; Yu, S.; Chen, Y.; et al. Transient receptor potential channel TRPC5 is essential for p-glycoprotein induction in drug-resistant cancer cells. Proc. Natl. Acad. Sci. USA 2012, 109, 16282–16287. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Chen, Z.; Zhu, Y.; Pan, Q.; Liu, Y.; Qi, X.; Jin, L.; Jin, J.; Ma, X.; Hua, D. Inhibition of transient receptor potential channel 5 reverses 5-fluorouracil resistance in human colorectal cancer cells. J. Biol. Chem. 2015, 290, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Akbulut, Y.; Gaunt, H.J.; Muraki, K.; Ludlow, M.J.; Amer, M.S.; Bruns, A.; Vasudev, N.S.; Radtke, L.; Willot, M.; Hahn, S.; et al. (-)-Englerin a is a potent and selective activator of TRPC4 and TRPC5 calcium channels. Angew Chem. Int. Ed. Engl. 2015, 54, 3787–3791. [Google Scholar] [CrossRef] [PubMed]
- Carson, C.; Raman, P.; Tullai, J.; Xu, L.; Henault, M.; Thomas, E.; Yeola, S.; Lao, J.M.; McPate, M.; Verkuyl, J.M.; et al. Englerin A agonizes the TRPC4/C5 cation channels to inhibit tumor cell line proliferation. PloS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Ratnayake, R.; Covell, D.; Ransom, T.T.; Gustafson, K.R.; Beutler, J.A. Englerin A, a selective inhibitor of renal cancer cell growth, from Phyllanthus engleri. Org. Lett. 2009, 11, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Gluz, O.; Liedtke, C.; Gottschalk, N.; Pusztai, L.; Nitz, U.; Harbeck, N. Triple-negative breast cancer--current status and future directions. Ann. Oncol. 2009, 20, 1913–1927. [Google Scholar] [CrossRef] [PubMed]
- Dent, R.; Trudeau, M.; Pritchard, K.I.; Hanna, W.M.; Kahn, H.K.; Sawka, C.A.; Lickley, L.A.; Rawlinson, E.; Sun, P.; Narod, S.A. Triple-negative breast cancer: Clinical features and patterns of recurrence. Clin. Cancer Res. 2007, 13, 4429–4434. [Google Scholar] [CrossRef] [PubMed]
- Liedtke, C.; Mazouni, C.; Hess, K.R.; Andre, F.; Tordai, A.; Mejia, J.A.; Symmans, W.F.; Gonzalez-Angulo, A.M.; Hennessy, B.; Green, M.; et al. Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J. Clin. Oncol. 2008, 26, 1275–1281. [Google Scholar] [CrossRef] [PubMed]
- Cheung, S.Y.; Henrot, M.; Al-Saad, M.; Baumann, M.; Muller, H.; Unger, A.; Rubaiy, H.N.; Mathar, I.; Dinkel, K.; Nussbaumer, P.; et al. TRPC4/TRPC5 channels mediate adverse reaction to the cancer cell cytotoxic agent (-)-Englerin A. Oncotarget 2018, 9, 29634–29643. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Pan, Q.; Meng, H.; Jiang, Y.; Mao, A.; Wang, T.; Hua, D.; Yao, X.; Jin, J.; Ma, X. Enhancement of vascular endothelial growth factor release in long-term drug-treated breast cancer via transient receptor potential channel 5-Ca(2+)-hypoxia-inducible factor 1α pathway. Pharmacol. Res. 2015, 93, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Veliceasa, D.; Ivanovic, M.; Hoepfner, F.T.; Thumbikat, P.; Volpert, O.V.; Smith, N.D. Transient potential receptor channel 4 controls thrombospondin-1 secretion and angiogenesis in renal cell carcinoma. FEBS J. 2007, 274, 6365–6377. [Google Scholar] [CrossRef] [PubMed]
- Antigny, F.; Girardin, N.; Frieden, M. Transient receptor potential canonical channels are required for in vitro endothelial tube formation. J. Biol. Chem. 2012, 287, 5917–5927. [Google Scholar] [CrossRef] [PubMed]
- Song, H.B.; Jun, H.O.; Kim, J.H.; Fruttiger, M.; Kim, J.H. Suppression of transient receptor potential canonical channel 4 inhibits vascular endothelial growth factor-induced retinal neovascularization. Cell Calcium 2015, 57, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Zeng, B.; Yuan, C.; Yang, X.; Atkin, S.L.; Xu, S.Z. TRPC channels and their splice variants are essential for promoting human ovarian cancer cell proliferation and tumorigenesis. Curr. Cancer Drug Targets 2013, 13, 103–116. [Google Scholar] [CrossRef] [PubMed]
- Gaunt, H.J.; Vasudev, N.S.; Beech, D.J. Transient receptor potential canonical 4 and 5 proteins as targets in cancer therapeutics. Eur. Biophys. J. 2016, 45, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Gough, N.R. New connections: NHERF gates activity. Sci. Signal. 2017, 10. [Google Scholar] [CrossRef] [PubMed]
- Loureiro, C.A.; Matos, A.M.; Dias-Alves, A.; Pereira, J.F.; Uliyakina, I.; Barros, P.; Amaral, M.D.; Matos, P. A molecular switch in the scaffold NHERF1 enables misfolded CFTR to evade the peripheral quality control checkpoint. Sci. Signal. 2015, 8. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).