The Mediator Complex: A Regulatory Hub for Transcriptional Activity of Nuclear Receptors
Abstract
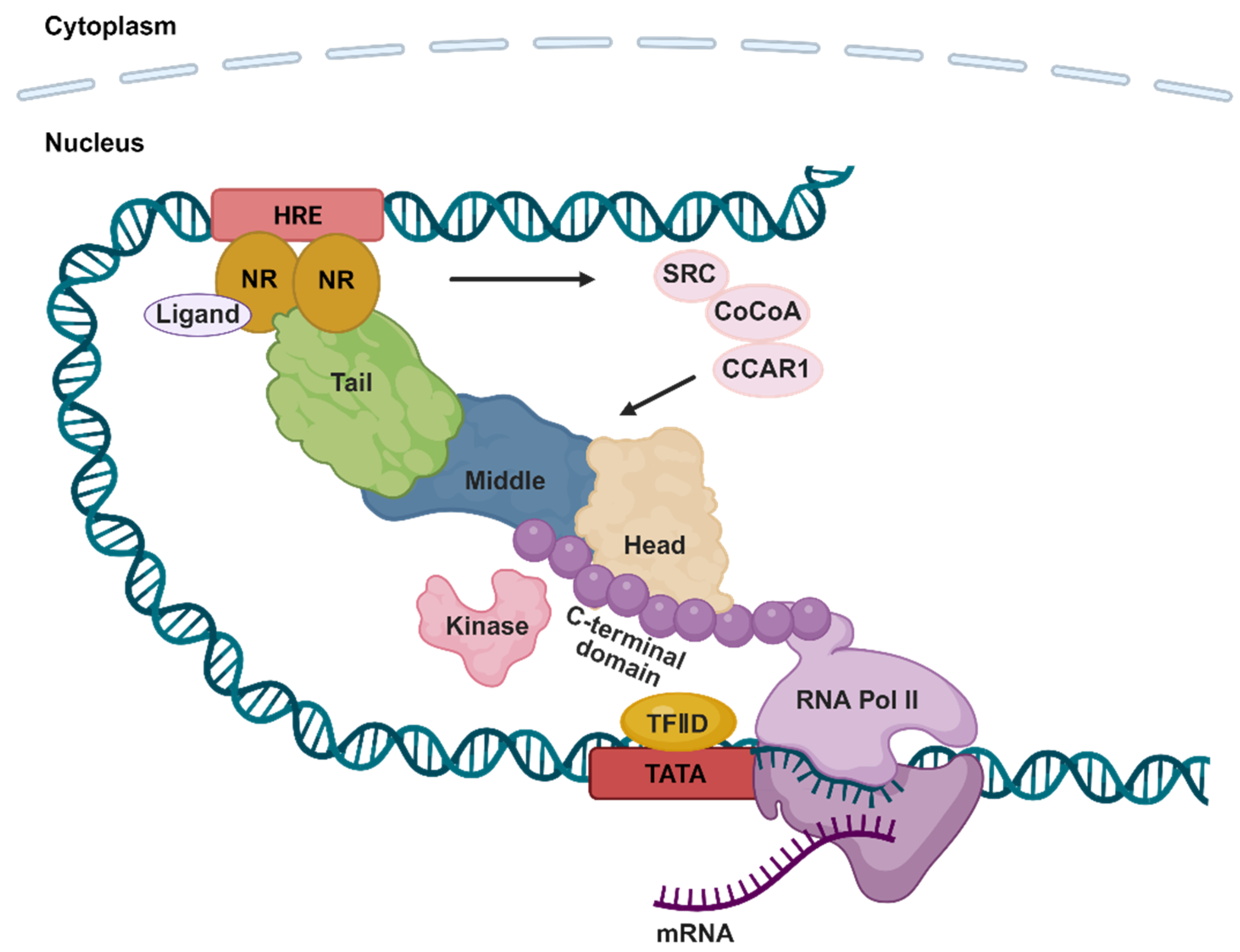
1. Overview of the Mediator Complex
1.1. Composition and Structure of the Mediator Complex
1.2. The Function of the Mediator Complex
2. The Mediator Complex and Nuclear Receptors
3. Interaction and Regulation of Nuclear Receptors by the Mediator Complex
3.1. Head Module
3.2. Middle Module
3.3. Tail Module
3.3.1. Interaction of MED15 with Sterol Regulatory Element-Binding Protein 1α (SREBP1α) in the Nuclear Receptor-Regulated Lipid Homeostasis
3.3.2. The Role of MED23 in Hepatic Fibrosis by Interaction with the Nuclear Receptor Retinoic Acid Receptor-Associated Orphan Receptor α (RORα)
3.3.3. The Role of MED25 in the Orphan Nuclear Receptor Hepatocyte Nuclear Factor 4α (HNF4α)-Driven Insulin Secretion
3.4. CDK8 Kinase Module
3.4.1. Multifaceted Regulatory Roles of MED12 in Nuclear Receptor Signaling
3.4.2. MED13
3.4.3. CDK8
4. Diseases Associated with Interaction of the Mediator Complex with Nuclear Receptors
4.1. Neoplastic Diseases
4.1.1. Prostate Cancer
4.1.2. Breast Cancer
4.1.3. Uterine Fibroids
4.2. Metabolic Diseases
4.2.1. Diabetes
4.2.2. Lipid Metabolism Disorders and Obesity
5. Potential Therapeutic Strategies by Targeting the Interaction Between the Mediator Complex and Nuclear Receptors
5.1. Disrupting the Interaction Interface Between the Mediator Complex and Nuclear Receptors
5.2. Targeting the Cofactors That Mediate Interaction Between the Mediator Complex and Nuclear Receptors
5.3. Targeting the CDK8 Kinase Module
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bourbon, H.M. Comparative genomics supports a deep evolutionary origin for the large, four-module transcriptional mediator complex. Nucleic Acids Res. 2008, 36, 3993–4008. [Google Scholar] [CrossRef]
- Flanagan, P.M.; Kelleher, R.J., 3rd; Sayre, M.H.; Tschochner, H.; Kornberg, R.D. A mediator required for activation of RNA polymerase II transcription in vitro. Nature 1991, 350, 436–438. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Björklund, S.; Li, Y.; Sayre, M.H.; Kornberg, R.D. A multiprotein mediator of transcriptional activation and its interaction with the C-terminal repeat domain of RNA polymerase II. Cell 1994, 77, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Malik, S.; Roeder, R.G. The metazoan Mediator co-activator complex as an integrative hub for transcriptional regulation. Nat. Rev. Genet. 2010, 11, 761–772. [Google Scholar] [CrossRef] [PubMed]
- Richter, W.F.; Nayak, S.; Iwasa, J.; Taatjes, D.J. The Mediator complex as a master regulator of transcription by RNA polymerase II. Nat. Rev. Mol. Cell Biol. 2022, 23, 732–749. [Google Scholar] [CrossRef]
- Chen, X.; Yin, X.; Li, J.; Wu, Z.; Qi, Y.; Wang, X.; Liu, W.; Xu, Y. Structures of the human Mediator and Mediator-bound preinitiation complex. Science 2021, 372, eabg0635. [Google Scholar] [CrossRef]
- Tsai, K.L.; Tomomori-Sato, C.; Sato, S.; Conaway, R.C.; Conaway, J.W.; Asturias, F.J. Subunit Architecture and Functional Modular Rearrangements of the Transcriptional Mediator Complex. Cell 2014, 157, 1430–1444. [Google Scholar] [CrossRef]
- Cevher, M.A.; Shi, Y.; Li, D.; Chait, B.T.; Malik, S.; Roeder, R.G. Reconstitution of active human core Mediator complex reveals a critical role of the MED14 subunit. Nat. Struct. Mol. Biol. 2014, 21, 1028–1034. [Google Scholar] [CrossRef]
- Abdella, R.; Talyzina, A.; Chen, S.; Inouye, C.J.; Tjian, R.; He, Y. Structure of the human Mediator-bound transcription preinitiation complex. Science 2021, 372, 52–56. [Google Scholar] [CrossRef]
- Zhao, H.; Young, N.; Kalchschmidt, J.; Lieberman, J.; El Khattabi, L.; Casellas, R.; Asturias, F.J. Structure of mammalian Mediator complex reveals Tail module architecture and interaction with a conserved core. Nat. Commun. 2021, 12, 1355. [Google Scholar] [CrossRef]
- Luyties, O.; Taatjes, D.J. The Mediator kinase module: An interface between cell signaling and transcription. Trends Biochem. Sci. 2022, 47, 314–327. [Google Scholar] [CrossRef] [PubMed]
- Galbraith, M.D.; Allen, M.A.; Bensard, C.L.; Wang, X.; Schwinn, M.K.; Qin, B.; Long, H.W.; Daniels, D.L.; Hahn, W.C.; Dowell, R.D.; et al. HIF1A employs CDK8-mediator to stimulate RNAPII elongation in response to hypoxia. Cell 2013, 153, 1327–1339. [Google Scholar] [CrossRef] [PubMed]
- Schilbach, S.; Hantsche, M.; Tegunov, D.; Dienemann, C.; Wigge, C.; Urlaub, H.; Cramer, P. Structures of transcription pre-initiation complex with TFIIH and Mediator. Nature 2017, 551, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Parmely, T.J.; Sato, S.; Tomomori-Sato, C.; Banks, C.A.; Kong, S.E.; Szutorisz, H.; Swanson, S.K.; Martin-Brown, S.; Washburn, M.P.; et al. Human mediator subunit MED26 functions as a docking site for transcription elongation factors. Cell 2011, 146, 92–104. [Google Scholar] [CrossRef]
- Takahashi, H.; Ranjan, A.; Chen, S.; Suzuki, H.; Shibata, M.; Hirose, T.; Hirose, H.; Sasaki, K.; Abe, R.; Chen, K.; et al. The role of Mediator and Little Elongation Complex in transcription termination. Nat. Commun. 2020, 11, 1063. [Google Scholar] [CrossRef]
- Elmlund, H.; Baraznenok, V.; Lindahl, M.; Samuelsen, C.O.; Koeck, P.J.; Holmberg, S.; Hebert, H.; Gustafsson, C.M. The cyclin-dependent kinase 8 module sterically blocks Mediator interactions with RNA polymerase II. Proc. Natl. Acad. Sci. USA 2006, 103, 15788–15793. [Google Scholar] [CrossRef]
- Wong, M.M.; Guo, C.; Zhang, J. Nuclear receptor corepressor complexes in cancer: Mechanism, function and regulation. Am. J. Clin. Exp. Urol. 2014, 2, 169–187. [Google Scholar]
- Aoyagi, S.; Archer, T.K. Dynamics of coactivator recruitment and chromatin modifications during nuclear receptor mediated transcription. Mol. Cell. Endocrinol. 2008, 280, 1–5. [Google Scholar] [CrossRef]
- Jin, L.; Li, Y. Structural and functional insights into nuclear receptor signaling. Adv. Drug Deliv. Rev. 2010, 62, 1218–1226. [Google Scholar] [CrossRef]
- Echeverria, P.C.; Picard, D. Molecular chaperones, essential partners of steroid hormone receptors for activity and mobility. Biochim. Biophys. Acta 2010, 1803, 641–649. [Google Scholar] [CrossRef]
- Belakavadi, M.; Fondell, J.D. Role of the mediator complex in nuclear hormone receptor signaling. Rev. Physiol. Biochem. Pharmacol. 2006, 156, 23–43. [Google Scholar] [CrossRef]
- George, A.A.; Schiltz, R.L.; Hager, G.L. Dynamic access of the glucocorticoid receptor to response elements in chromatin. Int. J. Biochem. Cell Biol. 2009, 41, 214–224. [Google Scholar] [CrossRef]
- Fondell, J.D.; Ge, H.; Roeder, R.G. Ligand induction of a transcriptionally active thyroid hormone receptor coactivator complex. Proc. Natl. Acad. Sci. USA 1996, 93, 8329–8333. [Google Scholar] [CrossRef]
- Youn, D.Y.; Xiaoli, A.M.; Pessin, J.E.; Yang, F. Regulation of metabolism by the Mediator complex. Biophys. Rep. 2016, 2, 69–77. [Google Scholar] [CrossRef]
- Poss, Z.C.; Ebmeier, C.C.; Taatjes, D.J. The Mediator complex and transcription regulation. Crit. Rev. Biochem. Mol. Biol. 2013, 48, 575–608. [Google Scholar] [CrossRef]
- Jiang, P.; Hu, Q.; Ito, M.; Meyer, S.; Waltz, S.; Khan, S.; Roeder, R.G.; Zhang, X. Key roles for MED1 LxxLL motifs in pubertal mammary gland development and luminal-cell differentiation. Proc. Natl. Acad. Sci. USA 2010, 107, 6765–6770. [Google Scholar] [CrossRef]
- Chen, W.; Roeder, R.G. Mediator-dependent nuclear receptor function. Semin. Cell Dev. Biol. 2011, 22, 749–758. [Google Scholar] [CrossRef]
- Spaeth, J.M.; Kim, N.H.; Boyer, T.G. Mediator and human disease. Semin. Cell Dev. Biol. 2011, 22, 776–787. [Google Scholar] [CrossRef]
- Hittelman, A.B.; Burakov, D.; Iñiguez-Lluhí, J.A.; Freedman, L.P.; Garabedian, M.J. Differential regulation of glucocorticoid receptor transcriptional activation via AF-1-associated proteins. EMBO J. 1999, 18, 5380–5388. [Google Scholar] [CrossRef]
- Grøntved, L.; Madsen, M.S.; Boergesen, M.; Roeder, R.G.; Mandrup, S. MED14 tethers mediator to the N-terminal domain of peroxisome proliferator-activated receptor gamma and is required for full transcriptional activity and adipogenesis. Mol. Cell. Biol. 2010, 30, 2155–2169. [Google Scholar] [CrossRef]
- Malik, S.; Wallberg, A.E.; Kang, Y.K.; Roeder, R.G. TRAP/SMCC/mediator-dependent transcriptional activation from DNA and chromatin templates by orphan nuclear receptor hepatocyte nuclear factor 4. Mol. Cell. Biol. 2002, 22, 5626–5637. [Google Scholar] [CrossRef]
- Lee, J.; Safe, S. Coactivation of estrogen receptor alpha (ER alpha)/Sp1 by vitamin D receptor interacting protein 150 (DRIP150). Arch. Biochem. Biophys. 2007, 461, 200–210. [Google Scholar] [CrossRef]
- Jin, F.; Claessens, F.; Fondell, J.D. Regulation of androgen receptor-dependent transcription by coactivator MED1 is mediated through a newly discovered noncanonical binding motif. J. Biol. Chem. 2012, 287, 858–870. [Google Scholar] [CrossRef]
- Kang, Y.K.; Guermah, M.; Yuan, C.X.; Roeder, R.G. The TRAP/Mediator coactivator complex interacts directly with estrogen receptors alpha and beta through the TRAP220 subunit and directly enhances estrogen receptor function in vitro. Proc. Natl. Acad. Sci. USA 2002, 99, 2642–2647. [Google Scholar] [CrossRef]
- Ge, K.; Guermah, M.; Yuan, C.X.; Ito, M.; Wallberg, A.E.; Spiegelman, B.M.; Roeder, R.G. Transcription coactivator TRAP220 is required for PPAR gamma 2-stimulated adipogenesis. Nature 2002, 417, 563–567. [Google Scholar] [CrossRef]
- Chen, W.; Roeder, R.G. The Mediator subunit MED1/TRAP220 is required for optimal glucocorticoid receptor-mediated transcription activation. Nucleic Acids Res. 2007, 35, 6161–6169. [Google Scholar] [CrossRef]
- Rachez, C.; Lemon, B.D.; Suldan, Z.; Bromleigh, V.; Gamble, M.; Näär, A.M.; Erdjument-Bromage, H.; Tempst, P.; Freedman, L.P. Ligand-dependent transcription activation by nuclear receptors requires the DRIP complex. Nature 1999, 398, 824–828. [Google Scholar] [CrossRef]
- Yuan, C.X.; Ito, M.; Fondell, J.D.; Fu, Z.Y.; Roeder, R.G. The TRAP220 component of a thyroid hormone receptor- associated protein (TRAP) coactivator complex interacts directly with nuclear receptors in a ligand-dependent fashion. Proc. Natl. Acad. Sci. USA 1998, 95, 7939–7944. [Google Scholar] [CrossRef]
- Taubert, S.; Van Gilst, M.R.; Hansen, M.; Yamamoto, K.R. A Mediator subunit, MDT-15, integrates regulation of fatty acid metabolism by NHR-49-dependent and -independent pathways in C. elegans. Genes Dev. 2006, 20, 1137–1149. [Google Scholar] [CrossRef]
- Lee, H.K.; Park, U.H.; Kim, E.J.; Um, S.J. MED25 is distinct from TRAP220/MED1 in cooperating with CBP for retinoid receptor activation. EMBO J. 2007, 26, 3545–3557. [Google Scholar] [CrossRef]
- Rana, R.; Surapureddi, S.; Kam, W.; Ferguson, S.; Goldstein, J.A. Med25 is required for RNA polymerase II recruitment to specific promoters, thus regulating xenobiotic and lipid metabolism in human liver. Mol. Cell. Biol. 2011, 31, 466–481. [Google Scholar] [CrossRef]
- Amoasii, L.; Holland, W.; Sanchez-Ortiz, E.; Baskin, K.K.; Pearson, M.; Burgess, S.C.; Nelson, B.R.; Bassel-Duby, R.; Olson, E.N. A MED13-dependent skeletal muscle gene program controls systemic glucose homeostasis and hepatic metabolism. Genes Dev. 2016, 30, 434–446. [Google Scholar] [CrossRef]
- Plaschka, C.; Larivière, L.; Wenzeck, L.; Seizl, M.; Hemann, M.; Tegunov, D.; Petrotchenko, E.V.; Borchers, C.H.; Baumeister, W.; Herzog, F.; et al. Architecture of the RNA polymerase II-Mediator core initiation complex. Nature 2015, 518, 376–380. [Google Scholar] [CrossRef]
- Belorusova, A.Y.; Bourguet, M.; Hessmann, S.; Chalhoub, S.; Kieffer, B.; Cianférani, S.; Rochel, N. Molecular determinants of MED1 interaction with the DNA bound VDR-RXR heterodimer. Nucleic Acids Res. 2020, 48, 11199–11213. [Google Scholar] [CrossRef] [PubMed]
- McInerney, E.M.; Rose, D.W.; Flynn, S.E.; Westin, S.; Mullen, T.M.; Krones, A.; Inostroza, J.; Torchia, J.; Nolte, R.T.; Assa-Munt, N.; et al. Determinants of coactivator LXXLL motif specificity in nuclear receptor transcriptional activation. Genes Dev. 1998, 12, 3357–3368. [Google Scholar] [CrossRef] [PubMed]
- Matsui, K.; Oda, K.; Mizuta, S.; Ishino, R.; Urahama, N.; Hasegawa, N.; Roeder, R.G.; Ito, M. Mediator subunit MED1 is a T3-dependent and T3-independent coactivator on the thyrotropin β gene promoter. Biochem. Biophys. Res. Commun. 2013, 440, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Viswakarma, N.; Jia, Y.; Bai, L.; Vluggens, A.; Borensztajn, J.; Xu, J.; Reddy, J.K. Coactivators in PPAR-Regulated Gene Expression. PPAR Res. 2010, 2010, 250126. [Google Scholar] [CrossRef]
- Ren, Y.; Behre, E.; Ren, Z.; Zhang, J.; Wang, Q.; Fondell, J.D. Specific structural motifs determine TRAP220 interactions with nuclear hormone receptors. Mol. Cell. Biol. 2000, 20, 5433–5446. [Google Scholar] [CrossRef]
- Burakov, D.; Wong, C.W.; Rachez, C.; Cheskis, B.J.; Freedman, L.P. Functional interactions between the estrogen receptor and DRIP205, a subunit of the heteromeric DRIP coactivator complex. J. Biol. Chem. 2000, 275, 20928–20934. [Google Scholar] [CrossRef]
- Rachez, C.; Gamble, M.; Chang, C.P.; Atkins, G.B.; Lazar, M.A.; Freedman, L.P. The DRIP complex and SRC-1/p160 coactivators share similar nuclear receptor binding determinants but constitute functionally distinct complexes. Mol. Cell. Biol. 2000, 20, 2718–2726. [Google Scholar] [CrossRef]
- Claessens, F.; Denayer, S.; Van Tilborgh, N.; Kerkhofs, S.; Helsen, C.; Haelens, A. Diverse roles of androgen receptor (AR) domains in AR-mediated signaling. Nucl. Recept. Signal. 2008, 6, e008. [Google Scholar] [CrossRef]
- Yang, F.; Vought, B.W.; Satterlee, J.S.; Walker, A.K.; Jim Sun, Z.Y.; Watts, J.L.; DeBeaumont, R.; Saito, R.M.; Hyberts, S.G.; Yang, S.; et al. An ARC/Mediator subunit required for SREBP control of cholesterol and lipid homeostasis. Nature 2006, 442, 700–704. [Google Scholar] [CrossRef]
- Xiaoli; Yang, F. Mediating lipid biosynthesis: Implications for cardiovascular disease. Trends Cardiovasc. Med. 2013, 23, 269–273. [Google Scholar] [CrossRef]
- Fajas, L.; Schoonjans, K.; Gelman, L.; Kim, J.B.; Najib, J.; Martin, G.; Fruchart, J.C.; Briggs, M.; Spiegelman, B.M.; Auwerx, J. Regulation of peroxisome proliferator-activated receptor gamma expression by adipocyte differentiation and determination factor 1/sterol regulatory element binding protein 1: Implications for adipocyte differentiation and metabolism. Mol. Cell. Biol. 1999, 19, 5495–5503. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Sun, Q.; Zhong, W.; Sun, X.; Zhou, Z. Hepatic Peroxisome Proliferator-Activated Receptor Gamma Signaling Contributes to Alcohol-Induced Hepatic Steatosis and Inflammation in Mice. Alcohol. Clin. Exp. Res. 2016, 40, 988–999. [Google Scholar] [CrossRef] [PubMed]
- Su, F.; Koeberle, A. Regulation and targeting of SREBP-1 in hepatocellular carcinoma. Cancer Metastasis Rev. 2024, 43, 673–708. [Google Scholar] [CrossRef] [PubMed]
- Van Gilst, M.R.; Hadjivassiliou, H.; Jolly, A.; Yamamoto, K.R. Nuclear hormone receptor NHR-49 controls fat consumption and fatty acid composition in C. elegans. PLoS Biol. 2005, 3, e53. [Google Scholar] [CrossRef]
- Sala, A.J.; Grant, R.A.; Imran, G.; Morton, C.; Brielmann, R.M.; Gorgoń, S.; Watts, J.; Bott, L.C.; Morimoto, R.I. Nuclear receptor signaling via NHR-49/MDT-15 regulates stress resilience and proteostasis in response to reproductive and metabolic cues. Genes Dev. 2024, 38, 380–392. [Google Scholar] [CrossRef]
- Shomer, N.; Kadhim, A.Z.; Grants, J.M.; Cheng, X.; Alhusari, D.; Bhanshali, F.; Poon, A.F.; Lee, M.Y.Y.; Muhuri, A.; Park, J.I.; et al. Mediator subunit MDT-15/MED15 and Nuclear Receptor HIZR-1/HNF4 cooperate to regulate toxic metal stress responses in Caenorhabditis elegans. PLoS Genet. 2019, 15, e1008508. [Google Scholar] [CrossRef]
- Chu, Y.; Gómez Rosso, L.; Huang, P.; Wang, Z.; Xu, Y.; Yao, X.; Bao, M.; Yan, J.; Song, H.; Wang, G. Liver Med23 ablation improves glucose and lipid metabolism through modulating FOXO1 activity. Cell Res. 2014, 24, 1250–1265. [Google Scholar] [CrossRef]
- Hintermann, E.; Bayer, M.; Pfeilschifter, J.M.; Luster, A.D.; Christen, U. CXCL10 promotes liver fibrosis by prevention of NK cell mediated hepatic stellate cell inactivation. J. Autoimmun. 2010, 35, 424–435. [Google Scholar] [CrossRef]
- Braunersreuther, V.; Viviani, G.L.; Mach, F.; Montecucco, F. Role of cytokines and chemokines in non-alcoholic fatty liver disease. World J. Gastroenterol. 2012, 18, 727–735. [Google Scholar] [CrossRef]
- Wang, Z.; Cao, D.; Li, C.; Min, L.; Wang, G. Mediator MED23 regulates inflammatory responses and liver fibrosis. PLoS Biol. 2019, 17, e3000563. [Google Scholar] [CrossRef] [PubMed]
- Han, E.H.; Rha, G.B.; Chi, Y.I. MED25 is a mediator component of HNF4α-driven transcription leading to insulin secretion in pancreatic beta-cells. PLoS ONE 2012, 7, e44007. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chiang, J.Y. Hepatocyte nuclear factor 4alpha regulation of bile acid and drug metabolism. Expert Opin. Drug Metab. Toxicol. 2009, 5, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Maechler, P.; Antinozzi, P.A.; Hagenfeldt, K.A.; Wollheim, C.B. Hepatocyte nuclear factor 4alpha regulates the expression of pancreatic beta -cell genes implicated in glucose metabolism and nutrient-induced insulin secretion. J. Biol. Chem. 2000, 275, 35953–35959. [Google Scholar] [CrossRef]
- Gupta, R.K.; Vatamaniuk, M.Z.; Lee, C.S.; Flaschen, R.C.; Fulmer, J.T.; Matschinsky, F.M.; Duncan, S.A.; Kaestner, K.H. The MODY1 gene HNF-4alpha regulates selected genes involved in insulin secretion. J. Clin. Investig. 2005, 115, 1006–1015. [Google Scholar] [CrossRef]
- Conaway, R.C.; Conaway, J.W. Function and regulation of the Mediator complex. Curr. Opin. Genet. Dev. 2011, 21, 225–230. [Google Scholar] [CrossRef]
- Knuesel, M.T.; Meyer, K.D.; Bernecky, C.; Taatjes, D.J. The human CDK8 subcomplex is a molecular switch that controls Mediator coactivator function. Genes Dev. 2009, 23, 439–451. [Google Scholar] [CrossRef]
- Park, M.J.; Shen, H.; Spaeth, J.M.; Tolvanen, J.H.; Failor, C.; Knudtson, J.F.; McLaughlin, J.; Halder, S.K.; Yang, Q.; Bulun, S.E.; et al. Oncogenic exon 2 mutations in Mediator subunit MED12 disrupt allosteric activation of cyclin C-CDK8/19. J. Biol. Chem. 2018, 293, 4870–4882. [Google Scholar] [CrossRef]
- Turunen, M.; Spaeth, J.M.; Keskitalo, S.; Park, M.J.; Kivioja, T.; Clark, A.D.; Mäkinen, N.; Gao, F.; Palin, K.; Nurkkala, H.; et al. Uterine leiomyoma-linked MED12 mutations disrupt mediator-associated CDK activity. Cell Rep. 2014, 7, 654–660. [Google Scholar] [CrossRef] [PubMed]
- Adler, D.; Offermann, A.; Braun, M.; Menon, R.; Syring, I.; Nowak, M.; Halbach, R.; Vogel, W.; Ruiz, C.; Zellweger, T.; et al. MED12 overexpression is a frequent event in castration-resistant prostate cancer. Endocr. Relat. Cancer 2014, 21, 663–675. [Google Scholar] [CrossRef] [PubMed]
- Crona, D.J.; Milowsky, M.I.; Whang, Y.E. Androgen receptor targeting drugs in castration-resistant prostate cancer and mechanisms of resistance. Clin. Pharmacol. Ther. 2015, 98, 582–589. [Google Scholar] [CrossRef] [PubMed]
- Andolfi, C.; Bartolini, C.; Morales, E.; Gündoğdu, B.; Puhr, M.; Guzman, J.; Wach, S.; Taubert, H.; Aigner, A.; Eder, I.E.; et al. MED12 and CDK8/19 Modulate Androgen Receptor Activity and Enzalutamide Response in Prostate Cancer. Endocrinology 2024, 165, bqae114. [Google Scholar] [CrossRef]
- Chen, M.; Carkner, R.; Buttyan, R. The hedgehog/Gli signaling paradigm in prostate cancer. Expert Rev. Endocrinol. Metab. 2011, 6, 453–467. [Google Scholar] [CrossRef]
- Duong, T.M.; Araujo Rincon, M.; Myneni, N.; Burleson, M. Genetic alterations in MED12 promote castration-resistant prostate cancer through modulation of GLI3 signaling. Mol. Biol. Res. Commun. 2023, 12, 63–70. [Google Scholar] [CrossRef]
- Zhou, H.; Kim, S.; Ishii, S.; Boyer, T.G. Mediator modulates Gli3-dependent Sonic hedgehog signaling. Mol. Cell. Biol. 2006, 26, 8667–8682. [Google Scholar] [CrossRef]
- Al-Hendy, A.; Laknaur, A.; Diamond, M.P.; Ismail, N.; Boyer, T.G.; Halder, S.K. Silencing Med12 Gene Reduces Proliferation of Human Leiomyoma Cells Mediated via Wnt/β-Catenin Signaling Pathway. Endocrinology 2017, 158, 592–603. [Google Scholar] [CrossRef]
- Tanos, T.; Sflomos, G.; Echeverria, P.C.; Ayyanan, A.; Gutierrez, M.; Delaloye, J.F.; Raffoul, W.; Fiche, M.; Dougall, W.; Schneider, P.; et al. Progesterone/RANKL is a major regulatory axis in the human breast. Sci. Transl. Med. 2013, 5, 182ra155. [Google Scholar] [CrossRef]
- Ishikawa, H.; Ishi, K.; Serna, V.A.; Kakazu, R.; Bulun, S.E.; Kurita, T. Progesterone is essential for maintenance and growth of uterine leiomyoma. Endocrinology 2010, 151, 2433–2442. [Google Scholar] [CrossRef]
- Liu, S.; Yin, P.; Kujawa, S.A.; Coon, J.S.t.; Okeigwe, I.; Bulun, S.E. Progesterone receptor integrates the effects of mutated MED12 and altered DNA methylation to stimulate RANKL expression and stem cell proliferation in uterine leiomyoma. Oncogene 2019, 38, 2722–2735. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Makala, H.; Sharma, V.; Suri, V.; Sarkar, C.; Kulshreshtha, R. MED12 is overexpressed in glioblastoma patients and serves as an oncogene by targeting the VDR/BCL6/p53 axis. Cell. Mol. Life Sci. CMLS 2022, 79, 104. [Google Scholar] [CrossRef]
- Li, Y.C.; Chao, T.C.; Kim, H.J.; Cholko, T.; Chen, S.F.; Li, G.; Snyder, L.; Nakanishi, K.; Chang, C.E.; Murakami, K.; et al. Structure and noncanonical Cdk8 activation mechanism within an Argonaute-containing Mediator kinase module. Sci. Adv. 2021, 7, eabd4484. [Google Scholar] [CrossRef]
- Chhun, T.; Chong, S.Y.; Park, B.S.; Wong, E.C.; Yin, J.L.; Kim, M.; Chua, N.H. HSI2 Repressor Recruits MED13 and HDA6 to Down-Regulate Seed Maturation Gene Expression Directly During Arabidopsis Early Seedling Growth. Plant Cell Physiol. 2016, 57, 1689–1706. [Google Scholar] [CrossRef]
- Amoasii, L.; Olson, E.N.; Bassel-Duby, R. Control of Muscle Metabolism by the Mediator Complex. Cold Spring Harb. Perspect. Med. 2018, 8, a029843. [Google Scholar] [CrossRef]
- Ziada, S.; Diharce, J.; Serillon, D.; Bonnet, P.; Aci-Sèche, S. Highlighting the Major Role of Cyclin C in Cyclin-Dependent Kinase 8 Activity through Molecular Dynamics Simulations. Int. J. Mol. Sci. 2024, 25, 5411. [Google Scholar] [CrossRef]
- Nemet, J.; Jelicic, B.; Rubelj, I.; Sopta, M. The two faces of Cdk8, a positive/negative regulator of transcription. Biochimie 2014, 97, 22–27. [Google Scholar] [CrossRef]
- McDermott, M.S.; Chumanevich, A.A.; Lim, C.U.; Liang, J.; Chen, M.; Altilia, S.; Oliver, D.; Rae, J.M.; Shtutman, M.; Kiaris, H.; et al. Inhibition of CDK8 mediator kinase suppresses estrogen dependent transcription and the growth of estrogen receptor positive breast cancer. Oncotarget 2017, 8, 12558–12575. [Google Scholar] [CrossRef]
- Lipsey, C.C.; Harbuzariu, A.; Robey, R.W.; Huff, L.M.; Gottesman, M.M.; Gonzalez-Perez, R.R. Leptin Signaling Affects Survival and Chemoresistance of Estrogen Receptor Negative Breast Cancer. Int. J. Mol. Sci. 2020, 21, 3794. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhang, C.; Wu, D.; Chen, H.; Rorick, A.; Zhang, X.; Wang, Q. Phospho-MED1-enhanced UBE2C locus looping drives castration-resistant prostate cancer growth. EMBO J. 2011, 30, 2405–2419. [Google Scholar] [CrossRef] [PubMed]
- Constantin, T.A.; Greenland, K.K.; Varela-Carver, A.; Bevan, C.L. Transcription associated cyclin-dependent kinases as therapeutic targets for prostate cancer. Oncogene 2022, 41, 3303–3315. [Google Scholar] [CrossRef] [PubMed]
- Rasool, R.U.; Natesan, R.; Deng, Q.; Aras, S.; Lal, P.; Sander Effron, S.; Mitchell-Velasquez, E.; Posimo, J.M.; Carskadon, S.; Baca, S.C.; et al. CDK7 Inhibition Suppresses Castration-Resistant Prostate Cancer through MED1 Inactivation. Cancer Discov. 2019, 9, 1538–1555. [Google Scholar] [CrossRef] [PubMed]
- Leonard, M.; Zhang, X. Estrogen receptor coactivator Mediator Subunit 1 (MED1) as a tissue-specific therapeutic target in breast cancer. J. Zhejiang Univ. Sci. B 2019, 20, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Qi, C.; Jain, S.; Le Beau, M.M.; Espinosa, R., 3rd; Atkins, G.B.; Lazar, M.A.; Yeldandi, A.V.; Rao, M.S.; Reddy, J.K. Amplification and overexpression of peroxisome proliferator-activated receptor binding protein (PBP/PPARBP) gene in breast cancer. Proc. Natl. Acad. Sci. USA 1999, 96, 10848–10853. [Google Scholar] [CrossRef]
- Bick, G.; Zhang, J.; Lower, E.E.; Zhang, X. Transcriptional coactivator MED1 in the interface of anti-estrogen and anti-HER2 therapeutic resistance. Cancer Drug Resist. 2022, 5, 498–510. [Google Scholar] [CrossRef]
- Zhang, Y.; Leonard, M.; Shu, Y.; Yang, Y.; Shu, D.; Guo, P.; Zhang, X. Overcoming Tamoxifen Resistance of Human Breast Cancer by Targeted Gene Silencing Using Multifunctional pRNA Nanoparticles. ACS Nano 2017, 11, 335–346. [Google Scholar] [CrossRef]
- Wu, J.; Zou, Y.; Luo, Y.; Guo, J.B.; Liu, F.Y.; Zhou, J.Y.; Zhang, Z.Y.; Wan, L.; Huang, O.P. Prevalence and clinical significance of mediator complex subunit 12 mutations in 362 Han Chinese samples with uterine leiomyoma. Oncol. Lett. 2017, 14, 47–54. [Google Scholar] [CrossRef]
- Mäkinen, N.; Kämpjärvi, K.; Frizzell, N.; Bützow, R.; Vahteristo, P. Characterization of MED12, HMGA2, and FH alterations reveals molecular variability in uterine smooth muscle tumors. Mol. Cancer 2017, 16, 101. [Google Scholar] [CrossRef]
- Zhao, X.; Xiaoli; Zong, H.; Abdulla, A.; Yang, E.S.; Wang, Q.; Ji, J.Y.; Pessin, J.E.; Das, B.C.; Yang, F. Inhibition of SREBP transcriptional activity by a boron-containing compound improves lipid homeostasis in diet-induced obesity. Diabetes 2014, 63, 2464–2473. [Google Scholar] [CrossRef]
- Paulsen, F.O.; Kang, D.; Becker, F.; Roth, D.; Joerg, V.; Dreyer, E.; Roesch, M.C.; Seidel, C.; Merseburger, A.S.; Kirfel, J.; et al. Targeting cyclin-dependent kinase 7-association between CDK7 and pMED1 expression in prostate cancer tissue. Carcinogenesis 2022, 43, 779–786. [Google Scholar] [CrossRef]
- Russo, J.W.; Nouri, M.; Balk, S.P. Androgen Receptor Interaction with Mediator Complex Is Enhanced in Castration-Resistant Prostate Cancer by CDK7 Phosphorylation of MED1. Cancer Discov. 2019, 9, 1490–1492. [Google Scholar] [CrossRef]
- Constantin, T.A.; Varela-Carver, A.; Greenland, K.K.; de Almeida, G.S.; Olden, E.; Penfold, L.; Ang, S.; Ormrod, A.; Leach, D.A.; Lai, C.F.; et al. The CDK7 inhibitor CT7001 (Samuraciclib) targets proliferation pathways to inhibit advanced prostate cancer. Br. J. Cancer 2023, 128, 2326–2337. [Google Scholar] [CrossRef]
- Yang, Y.; Leonard, M.; Zhang, Y.; Zhao, D.; Mahmoud, C.; Khan, S.; Wang, J.; Lower, E.E.; Zhang, X. HER2-Driven Breast Tumorigenesis Relies upon Interactions of the Estrogen Receptor with Coactivator MED1. Cancer Res. 2018, 78, 422–435. [Google Scholar] [CrossRef]
- Broude, E.V.; Győrffy, B.; Chumanevich, A.A.; Chen, M.; McDermott, M.S.; Shtutman, M.; Catroppo, J.F.; Roninson, I.B. Expression of CDK8 and CDK8-interacting Genes as Potential Biomarkers in Breast Cancer. Curr. Cancer Drug Targets 2015, 15, 739–749. [Google Scholar] [CrossRef] [PubMed]
- Crown, J. CDK8: A new breast cancer target. Oncotarget 2017, 8, 14269–14270. [Google Scholar] [CrossRef] [PubMed]
- McGuire, M.M.; Yatsenko, A.; Hoffner, L.; Jones, M.; Surti, U.; Rajkovic, A. Whole exome sequencing in a random sample of North American women with leiomyomas identifies MED12 mutations in majority of uterine leiomyomas. PLoS ONE 2012, 7, e33251. [Google Scholar] [CrossRef]
- Locke, J.M.; Dusatkova, P.; Colclough, K.; Hughes, A.E.; Dennis, J.M.; Shields, B.; Flanagan, S.E.; Shepherd, M.H.; Dempster, E.L.; Hattersley, A.T.; et al. Association of birthweight and penetrance of diabetes in individuals with HNF4A-MODY: A cohort study. Diabetologia 2022, 65, 246–249. [Google Scholar] [CrossRef]
- Lehmann, J.M.; Moore, L.B.; Smith-Oliver, T.A.; Wilkison, W.O.; Willson, T.M.; Kliewer, S.A. An antidiabetic thiazolidinedione is a high affinity ligand for peroxisome proliferator-activated receptor gamma (PPAR gamma). J. Biol. Chem. 1995, 270, 12953–12956. [Google Scholar] [CrossRef]
- Rieusset, J.; Chambrier, C.; Bouzakri, K.; Dusserre, E.; Auwerx, J.; Riou, J.P.; Laville, M.; Vidal, H. The expression of the p85alpha subunit of phosphatidylinositol 3-kinase is induced by activation of the peroxisome proliferator-activated receptor gamma in human adipocytes. Diabetologia 2001, 44, 544–554. [Google Scholar] [CrossRef]
- Bai, L.; Jia, Y.; Viswakarma, N.; Huang, J.; Vluggens, A.; Wolins, N.E.; Jafari, N.; Rao, M.S.; Borensztajn, J.; Yang, G.; et al. Transcription coactivator mediator subunit MED1 is required for the development of fatty liver in the mouse. Hepatolology 2011, 53, 1164–1174. [Google Scholar] [CrossRef]
- Qin, W.; Xie, M.; Qin, X.; Fang, Q.; Yin, F.; Li, Z. Recent advances in peptidomimetics antagonists targeting estrogen receptor α-coactivator interaction in cancer therapy. Bioorganic Med. Chem. Lett. 2018, 28, 2827–2836. [Google Scholar] [CrossRef]
- Gunther, J.R.; Moore, T.W.; Collins, M.L.; Katzenellenbogen, J.A. Amphipathic benzenes are designed inhibitors of the estrogen receptor alpha/steroid receptor coactivator interaction. ACS Chem. Biol. 2008, 3, 282–286. [Google Scholar] [CrossRef]
- Kim, J.H.; Yang, C.K.; Heo, K.; Roeder, R.G.; An, W.; Stallcup, M.R. CCAR1, a key regulator of mediator complex recruitment to nuclear receptor transcription complexes. Mol. Cell 2008, 31, 510–519. [Google Scholar] [CrossRef]
- Sharma, D.; Fondell, J.D. Ordered recruitment of histone acetyltransferases and the TRAP/Mediator complex to thyroid hormone-responsive promoters in vivo. Proc. Natl. Acad. Sci. USA 2002, 99, 7934–7939. [Google Scholar] [CrossRef] [PubMed]
- Burakov, D.; Crofts, L.A.; Chang, C.P.; Freedman, L.P. Reciprocal recruitment of DRIP/mediator and p160 coactivator complexes in vivo by estrogen receptor. J. Biol. Chem. 2002, 277, 14359–14362. [Google Scholar] [CrossRef] [PubMed]
- Redmond, A.M.; Bane, F.T.; Stafford, A.T.; McIlroy, M.; Dillon, M.F.; Crotty, T.B.; Hill, A.D.; Young, L.S. Coassociation of estrogen receptor and p160 proteins predicts resistance to endocrine treatment; SRC-1 is an independent predictor of breast cancer recurrence. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2009, 15, 2098–2106. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Wu, R.C.; O’Malley, B.W. Normal and cancer-related functions of the p160 steroid receptor co-activator (SRC) family. Nat. Rev. Cancer 2009, 9, 615–630. [Google Scholar] [CrossRef]
- Roninson, I.B.; Győrffy, B.; Mack, Z.T.; Shtil, A.A.; Shtutman, M.S.; Chen, M.; Broude, E.V. Identifying Cancers Impacted by CDK8/19. Cells 2019, 8, 821. [Google Scholar] [CrossRef]
- Fant, C.B.; Taatjes, D.J. Regulatory functions of the Mediator kinases CDK8 and CDK19. Transcription 2019, 10, 76–90. [Google Scholar] [CrossRef]

| Module | Subunit |
|---|---|
| Head | MED6 |
| MED8 | |
| MED11 | |
| MED14 | |
| MED17 | |
| MED18 | |
| MED20 | |
| MED22 | |
| MED27 | |
| MED28 | |
| MED29 | |
| MED30 | |
| Middle | MED1 |
| MED4 | |
| MED7 | |
| MED9 | |
| MED10 | |
| MED19 | |
| MED21 | |
| MED26 | |
| MED31 | |
| Tail | MED15 |
| MED16 | |
| MED23 | |
| MED24 | |
| MED25 | |
| CDK8-Kinase module | MED12/12L |
| MED13/13L | |
| CDK8/CDK19 | |
| CycC |
| Subunit | NR | Mediator Subunit Domains Involved | NRs Domains Involved | Reference |
|---|---|---|---|---|
| MED14 | GR | AF1 | [29] | |
| PPARγ | [30] | |||
| HNF4 | [31] | |||
| ERα | [32] | |||
| MED1 | AR | LXXLL | AF1/AF2 | [33] |
| ER | [26,34] | |||
| PPARγ | [35] | |||
| GR | [36] | |||
| TR | [34] | |||
| VDR | [37] | |||
| RAR | [38] | |||
| RXR | [38] | |||
| HNF4 | [31] | |||
| MED15 | NHR-49 | KIX | LBD | [39] |
| MED25 | RAR | LXXLL | AF2 | [40] |
| HNF4α | [41] | |||
| MED13 | NURR1 | [42] |
| Disease | Subunit | Molecular Mechanism | Preclinical Features | Reference |
|---|---|---|---|---|
| Prostate cancer | MED1 | Phosphorylation modification | Cancer progression propensity | [90,91,92] |
| MED12 | Overexpression | Drug resistance | [74,78] | |
| Breast cancer | MED1 | Overexpression | Promotes cancer progression | [93,94] |
| Drug resistance | [95,96] | |||
| CDK8 | Promotes cancer progression | [88] | ||
| Uterine Fibroids | MED12 | Gene mutations | Promotes tumor growth | [97,98] |
| Diabetes | MED25 | Gene mutations | Insulin secretion defect | [64] |
| Lipid Metabolism disorders and Obesity | MED15 | Overexpression | Lipid metabolism disorders | [99] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, L.; Zhao, M.; Zhai, Y.; Lin, Q. The Mediator Complex: A Regulatory Hub for Transcriptional Activity of Nuclear Receptors. Cells 2025, 14, 1335. https://doi.org/10.3390/cells14171335
Zhou L, Zhao M, Zhai Y, Lin Q. The Mediator Complex: A Regulatory Hub for Transcriptional Activity of Nuclear Receptors. Cells. 2025; 14(17):1335. https://doi.org/10.3390/cells14171335
Chicago/Turabian StyleZhou, Liming, Manhan Zhao, Yifei Zhai, and Qiong Lin. 2025. "The Mediator Complex: A Regulatory Hub for Transcriptional Activity of Nuclear Receptors" Cells 14, no. 17: 1335. https://doi.org/10.3390/cells14171335
APA StyleZhou, L., Zhao, M., Zhai, Y., & Lin, Q. (2025). The Mediator Complex: A Regulatory Hub for Transcriptional Activity of Nuclear Receptors. Cells, 14(17), 1335. https://doi.org/10.3390/cells14171335





