Mechanistic Insights into the Pathogenesis of Polycystic Kidney Disease
Abstract
1. Introduction
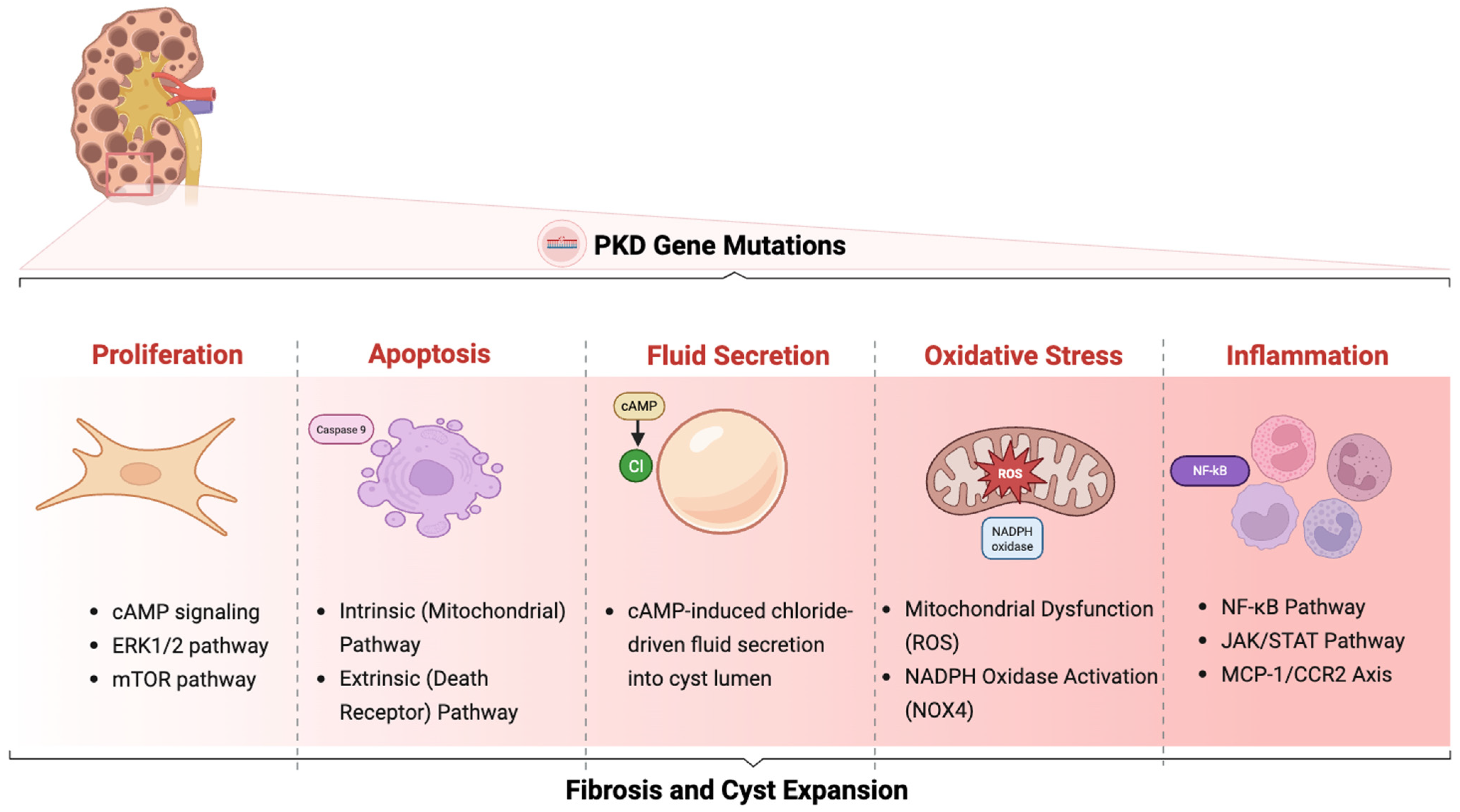
2. Cellular Mechanisms of Cystogenesis
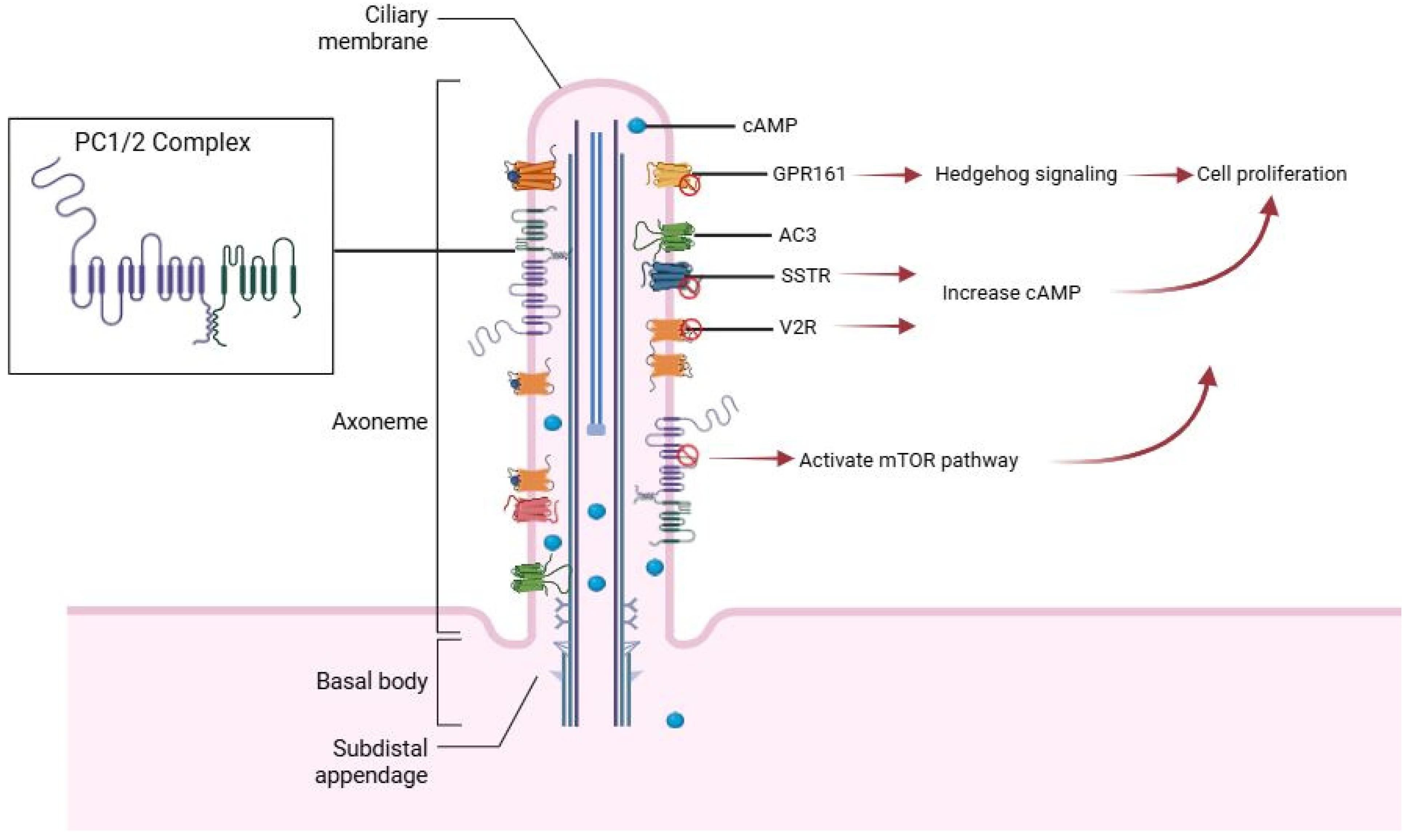
2.1. Role of Cilia in PKD
2.2. Cell Proliferation
2.3. Fluid Secretion
2.4. Fibrosis
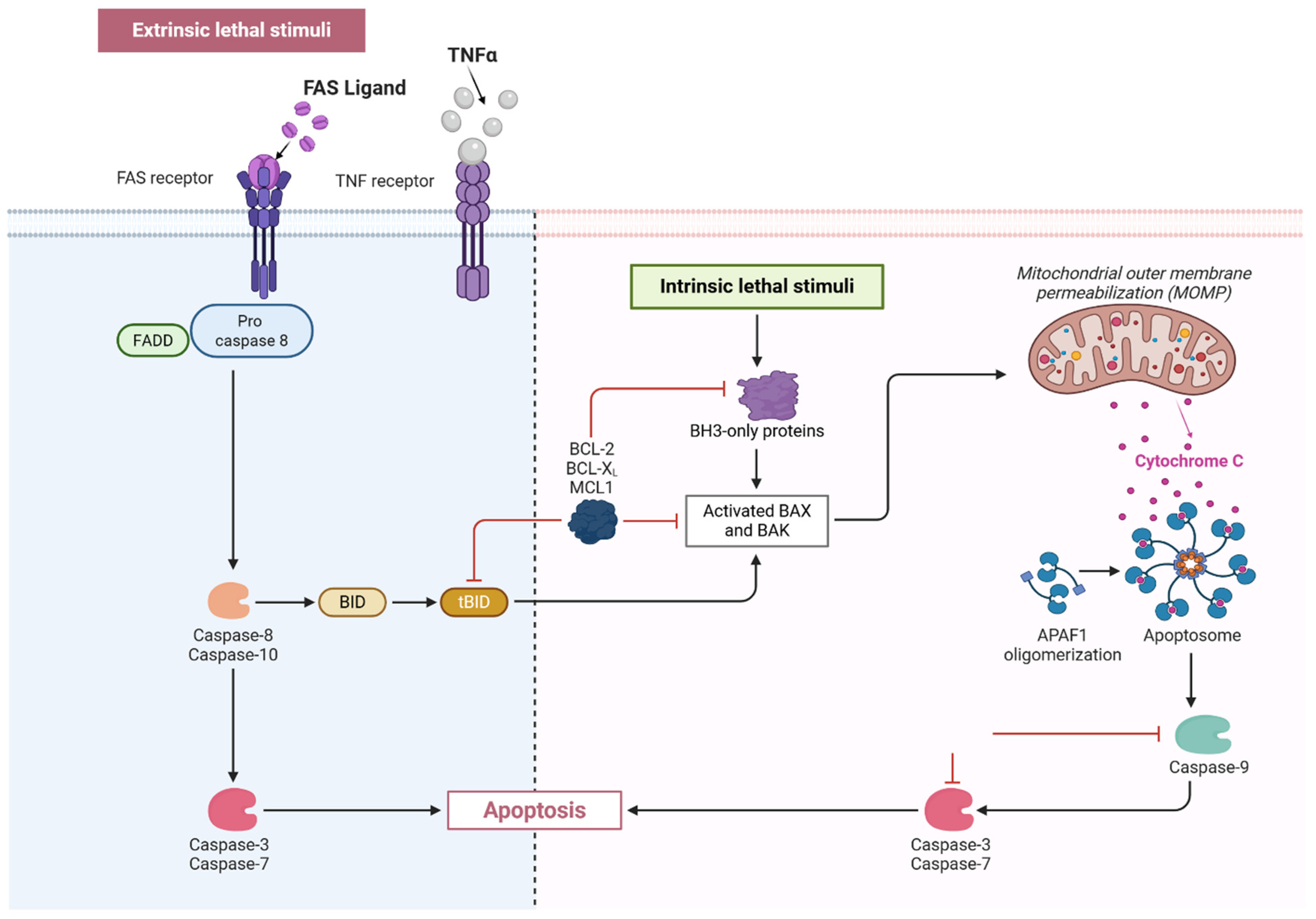
2.5. Apoptosis
2.6. Oxidative Stress and Mitochondrial Dysfunction
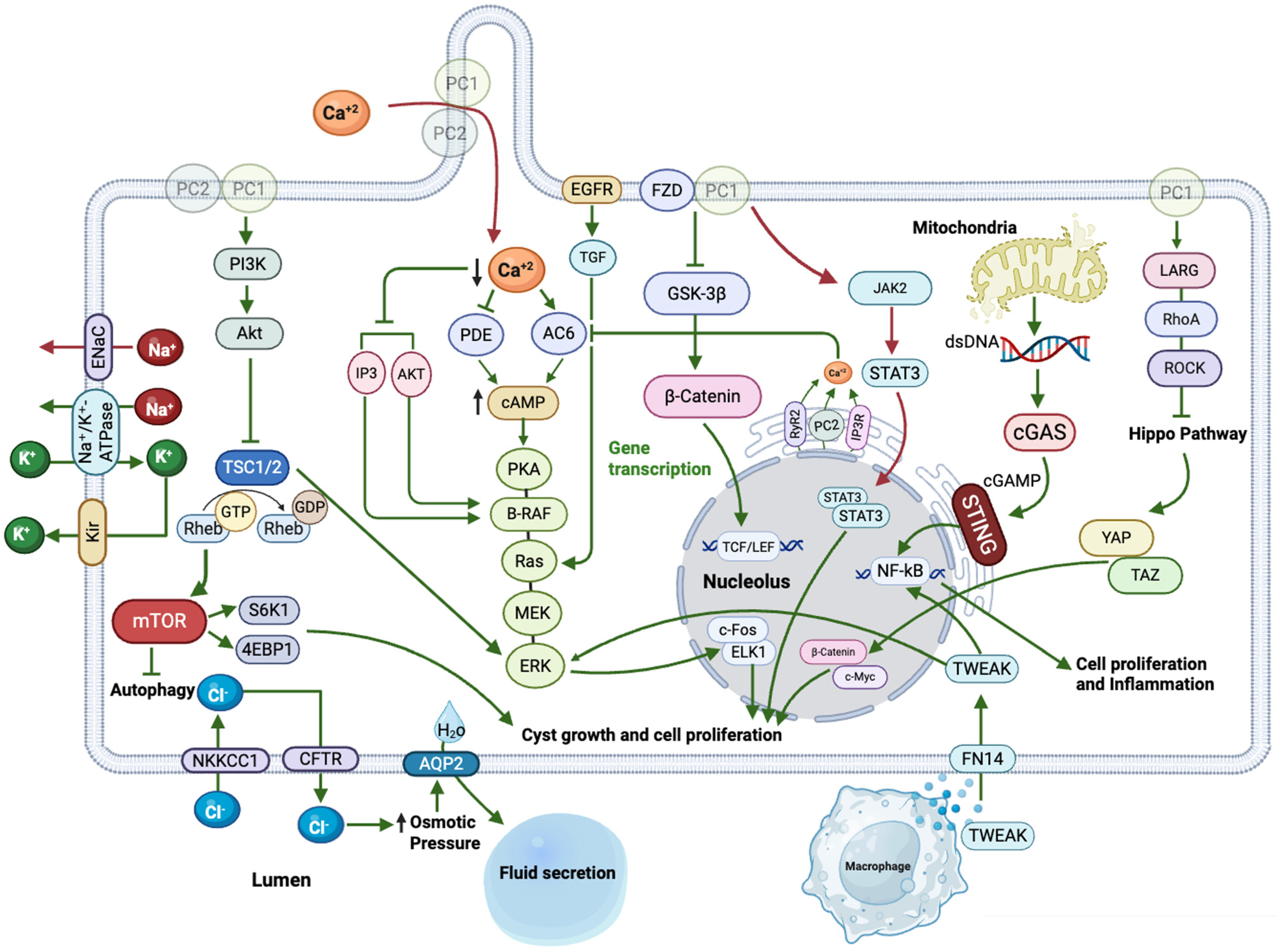
3. Molecular Mechanisms’ Contributions to PKD Pathogenesis
3.1. cAMP-Driven Mechanisms in ADPKD Pathogenesis
3.2. mTOR-Driven Mechanisms in ADPKD Pathogenesis
3.3. Wnt-Driven Mechanisms in ADPKD Pathogenesis
3.4. STING-Driven Mechanisms in ADPKD Pathogenesis
3.5. TWEAK/Fn14-Driven Mechanisms in ADPKD Pathogenesis
3.6. Hippo Signaling-Driven Mechanisms in ADPKD Pathogenesis
4. Epigenetic Mechanisms in ADPKD
5. Role of Emerging Biomarkers in the Pathogenesis of ADPKD
6. Animal Models of Polycystic Kidney Disease
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACs | Adenylate Cyclases |
| ADPKD | Autosomal Dominant Polycystic Kidney Disease |
| ANG II | Angiotensin II |
| ARPKD | Autosomal Recessive Polycystic Kidney Disease |
| Bak | Bcl-2 Antagonist/killer |
| Bax | Bcl-2-associated X Protein |
| BCL-2 | B-cell Lymphoma 2 |
| BCL-XL | B-cell Lymphoma-extra-large |
| B-Raf | B-Rapidly Accelerated Fibrosarcoma |
| Ca+2 | Calcium |
| cAMP | Cyclic Adenosine Monophosphate |
| CD | Collecting Duct |
| c-Myc | Cellular Myelocytomatosis Oncogene |
| cGAS | Cyclic GMP–AMP Synthase |
| CFTR | Cystic Fibrosis Transmembrane Conductance Regulator |
| ECM | Extracellular Matrix |
| EMT | Epithelial-to-Mesenchymal Transition |
| ENaC | Epithelial Sodium Channel |
| eNOS | endothelial nitric oxide synthase |
| ER | Endoplasmic Reticulum |
| ERK | Extracellular Signal-Regulated Kinase |
| ESRD | End Stage Renal Disease |
| FPC | Fibrocystin |
| HGF | Hepatocyte Growth Factor |
| IGF-1 | Insulin-like Growth Factor 1 |
| IP3R | Inositol 1,4,5-Trisphosphate Receptor |
| JAK-STAT | Janus Kinase and Signal Transducer and Activator of Transcription |
| MCL-1 | Myeloid Cell Leukemia-1 |
| MDCK | Madin-Darby Canine Kidney |
| MEK | Mitogen-Activated Protein Kinase |
| MMP | Matrix Metalloproteinase |
| mTOR | Mammalian Target of Rapamycin |
| Na+ | Sodium Ion |
| NADPH | Nicotinamide Adenine Dinucleotide Phosphate |
| NHKS | Normal Human Kidneys |
| NCCE | Non-Capacitative Ca+2 Entry |
| NO | Nitric Oxide |
| NOX4 | NADPH Oxidase 4 |
| PC1 | Polycystin-1 |
| PC2 | Polycystin-2 |
| PDE | Phosphodiesterases |
| PDGF | Platelet-Derived Growth Factor |
| PKA | Protein Kinase A |
| PKD | Polycystic Kidney Disease |
| ROS | Reactive Oxygen Species |
| RyR | Ryanodine Receptor |
| STIM1 | Stromal Interaction Molecule 1 |
| TGF-α/β | Transforming Growth Factor Alpha/Beta |
| TIMPs | Tissue Inhibitors of Metalloproteinases |
| TMEM16A | Transmembrane Member 16A Anoctamin 1 |
| TRP | Transient Receptor Potential |
| TRPP | Transient Receptor Potential Polycystin |
| TSP | Thrombospondin |
| WGS | Whole-Genome Sequencing |
References
- Aung, T.T.; Bhandari, S.K.; Chen, Q.; Malik, F.T.; Willey, C.J.; Reynolds, K.; Jacobsen, S.J.; Sim, J.J. Autosomal Dominant Polycystic Kidney Disease Prevalence among a Racially Diverse United States Population, 2002 through 2018. Kidney360 2021, 2, 2010–2015. [Google Scholar] [CrossRef]
- Nobakht, N.; Hanna, R.M.; Al-Baghdadi, M.; Ameen, K.M.; Arman, F.; Nobahkt, E.; Kamgar, M.; Rastogi, A. Advances in Autosomal Dominant Polycystic Kidney Disease: A Clinical Review. Kidney Med. 2020, 2, 196–208. [Google Scholar] [CrossRef]
- Righini, M.; Mancini, R.; Busutti, M.; Buscaroli, A. Autosomal dominant polycystic kidney disease: Extrarenal involvement. Int. J. Mol. Sci. 2024, 25, 2554. [Google Scholar] [CrossRef]
- Luciano, R.L.; Dahl, N.K. Extra-renal manifestations of autosomal dominant polycystic kidney disease (ADPKD): Considerations for routine screening and management. Nephrol. Dial. Transplant. 2014, 29, 247–254. [Google Scholar] [CrossRef]
- Sagar, P.S.; Rangan, G.K. Cardiovascular manifestations and management in ADPKD. Kidney Int. Rep. 2023, 8, 1924–1940. [Google Scholar] [CrossRef] [PubMed]
- Mahboob, M.; Rout, P.; Leslie, S.W.; Bokhari, S.R.A. Autosomal Dominant Polycystic Kidney Disease. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2025. [Google Scholar]
- Ferreira, F.M.; Watanabe, E.H.; Onuchic, L.F. Polycystins and molecular basis of autosomal dominant polycystic kidney disease. In Polycystic Kidney Disease; Li, X., Ed.; Exon Publications: Brisbane, Australia, 2015; pp. 139–167. [Google Scholar]
- Saternos, H.; Ley, S.; AbouAlaiwi, W. Primary cilia and calcium signaling interactions. Int. J. Mol. Sci. 2020, 21, 7109. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.J.; Seo, E.; Kim, J.W.; Nam, S.A.; Lee, J.Y.; Jun, J.; Oh, S.; Park, M.; Jho, E.-h.; Yoo, K.H. TAZ/Wnt-β-catenin/c-MYC axis regulates cystogenesis in polycystic kidney disease. Proc. Natl. Acad. Sci. USA 2020, 117, 29001–29012. [Google Scholar] [CrossRef]
- Margaria, J.P.; Campa, C.C.; De Santis, M.C.; Hirsch, E.; Franco, I. The PI3K/Akt/mTOR pathway in polycystic kidney disease: A complex interaction with polycystins and primary cilium. Cell. Signal. 2020, 66, 109468. [Google Scholar] [CrossRef]
- Malekshahabi, T.; Khoshdel Rad, N.; Serra, A.L.; Moghadasali, R. Autosomal dominant polycystic kidney disease: Disrupted pathways and potential therapeutic interventions. J. Cell. Physiol. 2019, 234, 12451–12470. [Google Scholar] [CrossRef]
- Cornec-Le Gall, E.; Audrézet, M.-P.; Rousseau, A.; Hourmant, M.; Renaudineau, E.; Charasse, C.; Morin, M.-P.; Moal, M.C.; Dantal, J.; Wehbe, B.; et al. The PROPKD Score: A New Algorithm to Predict Renal Survival in Autosomal Dominant Polycystic Kidney Disease. J. Am. Soc. Nephrol. 2016, 27, 942–951. [Google Scholar] [CrossRef] [PubMed]
- Vasileva, V.Y.; Sultanova, R.F.; Sudarikova, A.V.; Ilatovskaya, D.V. Insights into the molecular mechanisms of polycystic kidney diseases. Front. Physiol. 2021, 12, 693130. [Google Scholar] [CrossRef]
- Boletta, A.; Caplan, M.J. Physiologic mechanisms underlying polycystic kidney disease. Physiol. Rev. 2025, 105, 1001–1045. [Google Scholar] [CrossRef] [PubMed]
- Lapin, B. Renal Cysts Formation: Study on the Organization and Dynamics of 3D Biomimetic Cellular Models for Polycystic Disease. Ph.D. Thesis, Université Paris Sciences et Lettres, Paris, France, 2023. [Google Scholar]
- Koslowski, S.; Latapy, C.; Auvray, P.; Blondel, M.; Meijer, L. An overview of in vivo and in vitro models for autosomal dominant polycystic kidney disease: A journey from 3D-cysts to mini-pigs. Int. J. Mol. Sci. 2020, 21, 4537. [Google Scholar] [CrossRef]
- Masyuk, A.I.; Gradilone, S.A.; LaRusso, N.F. Calcium signaling in cilia and ciliary-mediated intracellular calcium signaling: Are they independent or coordinated molecular events? Hepatology 2014, 60, 1783–1785. [Google Scholar] [CrossRef]
- Kolb, R.J.; Nauli, S.M. Ciliary dysfunction in polycystic kidney disease: An emerging model with polarizing potential. Front. Biosci. A J. Virtual Libr. 2008, 13, 4451. [Google Scholar] [CrossRef]
- Ta, C.M.; Vien, T.N.; Ng, L.C.; DeCaen, P.G. Structure and function of polycystin channels in primary cilia. Cell. Signal. 2020, 72, 109626. [Google Scholar] [CrossRef]
- Reiterová, J.; Tesař, V. Autosomal dominant polycystic kidney disease: From pathophysiology of cystogenesis to advances in the treatment. Int. J. Mol. Sci. 2022, 23, 3317. [Google Scholar] [CrossRef]
- Hoey, D.A.; Downs, M.E.; Jacobs, C.R. The mechanics of the primary cilium: An intricate structure with complex function. J. Biomech. 2012, 45, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Bell, P.D.; Fitzgibbon, W.; Sas, K.; Stenbit, A.E.; Amria, M.; Houston, A.; Reichert, R.; Gilley, S.; Siegal, G.P.; Bissler, J.; et al. Loss of primary cilia upregulates renal hypertrophic signaling and promotes cystogenesis. J. Am. Soc. Nephrol. 2011, 22, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Tian, X.; Igarashi, P.; Pazour, G.J.; Somlo, S. Loss of cilia suppresses cyst growth in genetic models of autosomal dominant polycystic kidney disease. Nat. Genet. 2013, 45, 1004–1012. [Google Scholar] [CrossRef]
- Shao, L.; El-Jouni, W.; Kong, F.; Ramesh, J.; Kumar, R.S.; Shen, X.; Ren, J.; Devendra, S.; Dorschel, A.; Wu, M.; et al. Genetic reduction of cilium length by targeting intraflagellar transport 88 protein impedes kidney and liver cyst formation in mouse models of autosomal polycystic kidney disease. Kidney Int. 2020, 98, 1225–1241. [Google Scholar] [CrossRef]
- Wang, X.; Yin, G.; Yang, Y.; Tian, X. Ciliary and Non-Ciliary Roles of IFT88 in Development and Diseases. Int. J. Mol. Sci. 2025, 26, 2110. [Google Scholar] [CrossRef] [PubMed]
- Lehman, J.M.; Michaud, E.J.; Schoeb, T.R.; Aydin-Son, Y.; Miller, M.; Yoder, B.K. The Oak Ridge Polycystic Kidney mouse: Modeling ciliopathies of mice and men. Dev. Dyn. 2008, 237, 1960–1971. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Beebe, K.; Hernandez, E.J.; Lazaro-Guevara, J.M.; Revelo, M.P.; Huang, Y.; Maschek, J.A.; Cox, J.E.; Kohan, D.E. Multiomic identification of factors associated with progression to cystic kidney disease in mice with nephron Ift88 disruption. Am. J. Physiol. Ren. Physiol. 2022, 322, F175–F192. [Google Scholar] [CrossRef]
- Buqaileh, R.; Alshriem, L.A.; AbouAlaiwi, W. Ciliary G-Protein Coupled Receptor Signaling in Polycystic Kidney Disease. Int. J. Mol. Sci. 2025, 26, 4971. [Google Scholar] [CrossRef]
- Maser, R.L.; Calvet, J.P.; Parnell, S.C. The GPCR properties of polycystin-1- A new paradigm. Front. Mol. Biosci. 2022, 9, 1035507. [Google Scholar] [CrossRef] [PubMed]
- Wingfield, J.L.; Lechtreck, K.F.; Lorentzen, E. Trafficking of ciliary membrane proteins by the intraflagellar transport/BBSome machinery. Essays Biochem. 2018, 62, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Mukhopdhyay, S.; Wen, X.; Ratti, N.; Loktev, A.; Rangell, L.; Scales, S.; Jackson, P. The Ciliary G-Protein-Coupled Receptor Gpr161 Negatively Regulates the Sonic Hedgehog Pathway via cAMP Signaling. Cell 2013, 152, 210–223. [Google Scholar] [CrossRef]
- Balster, D.A.; O’Dorisio, M.S.; Summers, M.A.; Turman, M.A. Segmental expression of somatostatin receptor subtypes sst1 and sst2 in tubules and glomeruli of human kidney. Am. J. Physiol. -Ren. Physiol. 2001, 280, F457–F465. [Google Scholar] [CrossRef]
- Messchendorp, A.L.; Casteleijn, N.F.; Meijer, E.; Gansevoort, R.T. Somatostatin in renal physiology and autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 2020, 35, 1306–1316. [Google Scholar] [CrossRef]
- Bhandari, S.; Watson, N.; Long, E.; Sharpe, S.; Zhong, W.; Xu, S.-Z.; Atkin, S.L. Expression of somatostatin and somatostatin receptor subtypes 1–5 in human normal and diseased kidney. J. Histochem. Cytochem. 2008, 56, 733–743. [Google Scholar] [CrossRef]
- Winkler, S.; Torikai, S.; Levine, B.; Kurokawa, K. Effect of somatostatin on vasopressin-induced antidiuresis and renal cyclic AMP of rats. Miner. Electrolyte Metab. 1982, 7, 8–14. [Google Scholar]
- Boone, M.; Deen, P.M. Physiology and pathophysiology of the vasopressin-regulated renal water reabsorption. Pflügers Arch. Eur. J. Physiol. 2008, 456, 1005–1024. [Google Scholar] [CrossRef] [PubMed]
- Bankir, L.; Bichet, D.G.; Morgenthaler, N.G. Vasopressin: Physiology, assessment and osmosensation. J. Intern. Med. 2017, 282, 284–297. [Google Scholar] [CrossRef]
- Juul, K.V.; Bichet, D.G.; Nielsen, S.; Nørgaard, J.P. The physiological and pathophysiological functions of renal and extrarenal vasopressin V2 receptors. Am. J. Physiol. -Ren. Physiol. 2014, 306, F931–F940. [Google Scholar] [CrossRef] [PubMed]
- Nauli, S.M.; Alenghat, F.J.; Luo, Y.; Williams, E.; Vassilev, P.; Li, X.; Elia, A.E.; Lu, W.; Brown, E.M.; Quinn, S.J. Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat. Genet. 2003, 33, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, M.; Fong, P.; Cheng, J.; Boletta, A.; Qian, F.; Zhang, X.-M.; Cai, H.; Germino, G.G.; Guggino, W.B. A regulatory role of polycystin-1 on cystic fibrosis transmembrane conductance regulator plasma membrane expression. Cell. Physiol. Biochem. 2006, 18, 9–20. [Google Scholar] [CrossRef]
- Araç, D.; Boucard, A.A.; Bolliger, M.F.; Nguyen, J.; Soltis, S.M.; Südhof, T.C.; Brunger, A.T. A novel evolutionarily conserved domain of cell-adhesion GPCRs mediates autoproteolysis. EMBO J. 2012, 31, 1364–1378. [Google Scholar] [CrossRef]
- Boehlke, C.; Kotsis, F.; Patel, V.; Braeg, S.; Voelker, H.; Bredt, S.; Beyer, T.; Janusch, H.; Hamann, C.; Gödel, M. Primary cilia regulate mTORC1 activity and cell size through Lkb1. Nat. Cell Biol. 2010, 12, 1115–1122. [Google Scholar] [CrossRef]
- Matson, J.P.; Cook, J.G. Cell cycle proliferation decisions: The impact of single cell analyses. Febs J. 2017, 284, 362–375. [Google Scholar] [CrossRef]
- Pellarin, I.; Dall’Acqua, A.; Favero, A.; Segatto, I.; Rossi, V.; Crestan, N.; Karimbayli, J.; Belletti, B.; Baldassarre, G. Cyclin-dependent protein kinases and cell cycle regulation in biology and disease. Signal Transduct. Target. Ther. 2025, 10, 11. [Google Scholar] [CrossRef]
- Zhou, X.; Torres, V.E. Emerging therapies for autosomal dominant polycystic kidney disease with a focus on cAMP signaling. Front. Mol. Biosci. 2022, 9, 981963. [Google Scholar] [CrossRef] [PubMed]
- Sanz, A.B.; Sanchez-Niño, M.D.; Ramos, A.M.; Ortiz, A. Regulated cell death pathways in kidney disease. Nat. Rev. Nephrol. 2023, 19, 281–299. [Google Scholar] [CrossRef]
- Zhou, J.X.; Li, X. Apoptosis in polycystic kidney disease: From pathogenesis to treatment. In Polycystic Kidney Disease; Li, X., Ed.; Codon Publications: Brisbane, Australia, 2015; pp. 197–230. [Google Scholar]
- Parys, J.B.; Bultynck, G.; Vervliet, T. IP3 receptor biology and endoplasmic reticulum calcium dynamics in cancer. Cell. Biol. Endoplasmic Reticulum 2021, 59, 215–237. [Google Scholar]
- Santoso, N.G.; Cebotaru, L.; Guggino, W.B. Polycystin-1, 2, and STIM1 interact with IP3R to modulate ER Ca2+ release through the PI3K/Akt pathway. Cell. Physiol. Biochem. 2011, 27, 715–726. [Google Scholar] [CrossRef] [PubMed]
- Sudarikova, A.V.; Vasileva, V.Y.; Sultanova, R.F.; Ilatovskaya, D.V. Recent advances in understanding ion transport mechanisms in polycystic kidney disease. Clin. Sci. 2021, 135, 2521–2540. [Google Scholar] [CrossRef]
- Alshriem, L.A.; Buqaileh, R.; Alorjani, Q.; AbouAlaiwi, W. Ciliary Ion Channels in Polycystic Kidney Disease. Cells 2025, 14, 459. [Google Scholar] [CrossRef]
- Mangolini, A.; de Stephanis, L.; Aguiari, G. Role of calcium in polycystic kidney disease: From signaling to pathology. World J. Nephrol. 2016, 5, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Cabrita, I.; Buchholz, B.; Schreiber, R.; Kunzelmann, K. TMEM16A drives renal cyst growth by augmenting Ca(2+) signaling in M1 cells. J. Mol. Med. 2020, 98, 659–671. [Google Scholar] [CrossRef]
- Patera, F.; Cudzich-Madry, A.; Huang, Z.; Fragiadaki, M. Renal expression of JAK2 is high in polycystic kidney disease and its inhibition reduces cystogenesis. Sci. Rep. 2019, 9, 4491. [Google Scholar] [CrossRef]
- Weimbs, T.; Olsan, E.E.; Talbot, J.J. Regulation of STATs by polycystin-1 and their role in polycystic kidney disease. JAK-STAT 2013, 2, e23650. [Google Scholar] [CrossRef]
- Harskamp, L.R.; Perez-Gomez, M.V.; Heida, J.E.; Engels, G.E.; van Goor, H.; van den Heuvel, M.C.; Streets, A.J.; Ong, A.C.; Ortiz, A.; Gansevoort, R.T. The association of urinary epidermal growth factors with ADPKD disease severity and progression. Nephrol. Dial. Transplant. 2023, 38, 2266–2275. [Google Scholar] [CrossRef]
- Coaxum, S.D.; Blanton, M.G.; Joyner, A.; Akter, T.; Bell, P.D.; Luttrell, L.M.; Raymond, J.R.; Lee, M.-H.; Blichmann, P.A.; Garnovskaya, M.N.; et al. Epidermal growth factor-induced proliferation of collecting duct cells from Oak Ridge polycystic kidney mice involves activation of Na+/H+ exchanger. Am. J. Physiol. Cell Physiol. 2014, 307, C554–C560. [Google Scholar] [CrossRef]
- Nemo, R.; Murcia, N.; Dell, K.M. Transforming growth factor alpha (TGF-α) and other targets of tumor necrosis factor-alpha converting enzyme (TACE) in murine polycystic kidney disease. Pediatr. Res. 2005, 57, 732–737. [Google Scholar] [CrossRef]
- Zhang, Y.; Dai, Y.; Raman, A.; Daniel, E.; Metcalf, J.; Reif, G.; Pierucci-Alves, F.; Wallace, D.P. Overexpression of TGF-β1 induces renal fibrosis and accelerates the decline in kidney function in polycystic kidney disease. Am. J. Physiol. Ren. Physiol. 2020, 319, F1135–F1148. [Google Scholar] [CrossRef]
- Elberg, D.; Jayaraman, S.; Turman, M.A.; Elberg, G. Transforming growth factor-β inhibits cystogenesis in human autosomal dominant polycystic kidney epithelial cells. Exp. Cell Res. 2012, 318, 1508–1516. [Google Scholar] [CrossRef]
- Qin, S.; Taglienti, M.; Cai, L.; Zhou, J.; Kreidberg, J.A. c-Met and NF-κB–dependent overexpression of Wnt7a and-7b and Pax2 promotes cystogenesis in polycystic kidney disease. J. Am. Soc. Nephrol. 2012, 23, 1309–1318. [Google Scholar] [CrossRef]
- Harris, P.C.; Torres, V.E. Genetic mechanisms and signaling pathways in autosomal dominant polycystic kidney disease. J. Clin. Investig. 2014, 124, 2315–2324. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zhang, Y.; Yuan, L.; Fu, L.; Mei, C. Rosiglitazone inhibits insulin-like growth factor-1-induced polycystic kidney disease cell growth and p70S6 kinase activation. Mol. Med. Rep. 2013, 8, 861–864. [Google Scholar] [CrossRef] [PubMed]
- Haffner, D.; Grund, A.; Leifheit-Nestler, M. Renal effects of growth hormone in health and in kidney disease. Pediatr. Nephrol. 2021, 36, 2511–2530. [Google Scholar] [CrossRef] [PubMed]
- Vanichapol, T.; Wangford, R.; Takemoto, M.; Uesugi, M.; Davidson, A.J.; Sander, V. High Efficiency Organoid-Derived Cyst Cultures as a Drug Discovery Platform for Polycystic Kidney Disease. bioRxiv 2025. preprint. [Google Scholar] [CrossRef]
- Montesano, R.; Ghzili, H.; Carrozzino, F.; Rossier, B.C.; Féraille, E. cAMP-dependent chloride secretion mediates tubule enlargement and cyst formation by cultured mammalian collecting duct cells. Am. J. Physiol. Ren. Physiol. 2009, 296, F446–F457. [Google Scholar] [CrossRef]
- Liu, B.; Li, C.; Liu, Z.; Dai, Z.; Tao, Y. Increasing extracellular matrix collagen level and MMP activity induces cyst development in polycystic kidney disease. BMC Nephrol. 2012, 13, 109. [Google Scholar] [CrossRef]
- Hewitson, T.D.; Holt, S.G.; Smith, E.R. Progression of tubulointerstitial fibrosis and the chronic kidney disease phenotype–role of risk factors and epigenetics. Front. Pharmacol. 2017, 8, 520. [Google Scholar] [CrossRef]
- Norman, J. Fibrosis and progression of autosomal dominant polycystic kidney disease (ADPKD). Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2011, 1812, 1327–1336. [Google Scholar] [CrossRef] [PubMed]
- Song, C.J.; Zimmerman, K.A.; Henke, S.J.; Yoder, B.K. Inflammation and fibrosis in polycystic kidney disease. In Kidney Development and Disease; Yoder, B.K., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 323–344. [Google Scholar]
- Dwivedi, N.; Jamadar, A.; Mathew, S.; Fields, T.A.; Rao, R. Myofibroblast depletion reduces kidney cyst growth and fibrosis in autosomal dominant polycystic kidney disease. Kidney Int. 2023, 103, 144–155. [Google Scholar] [CrossRef]
- Zhang, Y.; Reif, G.; Wallace, D.P. Extracellular matrix, integrins, and focal adhesion signaling in polycystic kidney disease. Cell Signal 2020, 72, 109646. [Google Scholar] [CrossRef]
- Yuan, Q.; Tan, R.J.; Liu, Y. Myofibroblast in kidney fibrosis: Origin, activation, and regulation. Ren. Fibros. Mech. Ther. 2019, 1165, 253–283. [Google Scholar]
- Kim, K.P.; Williams, C.E.; Lemmon, C.A. Cell–matrix interactions in renal fibrosis. Kidney Dial. 2022, 2, 607–624. [Google Scholar] [CrossRef] [PubMed]
- Hassane, S.; Leonhard, W.N.; van der Wal, A.; Hawinkels, L.J.; Lantinga-van Leeuwen, I.S.; Dijke, P.T.; Breuning, M.H.; Heer, E.d.; Peters, D.J. Elevated TGFβ–Smad signalling in experimental Pkd1 models and human patients with polycystic kidney disease. J. Pathol. 2010, 222, 21–31. [Google Scholar] [CrossRef]
- Song, X.; Di Giovanni, V.; He, N.; Wang, K.; Ingram, A.; Rosenblum, N.D.; Pei, Y. Systems biology of autosomal dominant polycystic kidney disease (ADPKD): Computational identification of gene expression pathways and integrated regulatory networks. Hum. Mol. Genet. 2009, 18, 2328–2343. [Google Scholar] [CrossRef] [PubMed]
- Kramann, R.; Machado, F.; Wu, H.; Kusaba, T.; Hoeft, K.; Schneider, R.K.; Humphreys, B.D. Parabiosis and single-cell RNA sequencing reveal a limited contribution of monocytes to myofibroblasts in kidney fibrosis. JCI Insight 2018, 3, e99561. [Google Scholar] [CrossRef]
- Li, L.; Okusa, M.D. Macrophages, dendritic cells, and kidney ischemia-reperfusion injury. Semin. Nephrol. 2010, 30, 268–277. [Google Scholar] [CrossRef]
- Chadban, S.J.; Wu, H.; Hughes, J. Macrophages and kidney transplantation. Semin. Nephrol. 2010, 30, 278–289. [Google Scholar] [CrossRef]
- Williams, T.M.; Little, M.H.; Ricardo, S.D. Macrophages in renal development, injury, and repair. Semin. Nephrol. 2010, 30, 255–267. [Google Scholar] [CrossRef]
- Goerdt, S.; Politz, O.; Schledzewski, K.; Birk, R.; Gratchev, A.; Guillot, P.; Hakiy, N.; Klemke, C.-D.; Dippel, E.; Kodelja, V. Alternative versus classical activation of macrophages. Pathobiology 2000, 67, 222–226. [Google Scholar] [CrossRef]
- Vernon, M.A.; Mylonas, K.J.; Hughes, J. Macrophages and Renal Fibrosis. Semin. Nephrol. 2010, 30, 302–317. [Google Scholar] [CrossRef]
- Wynes, M.W.; Frankel, S.K.; Riches, D.W. IL-4-induced macrophage-derived IGF-I protects myofibroblasts from apoptosis following growth factor withdrawal. J. Leukoc. Biol. 2004, 76, 1019–1027. [Google Scholar] [CrossRef] [PubMed]
- MacKinnon, A.C.; Farnworth, S.L.; Hodkinson, P.S.; Henderson, N.C.; Atkinson, K.M.; Leffler, H.; Nilsson, U.J.; Haslett, C.; Forbes, S.J.; Sethi, T. Regulation of alternative macrophage activation by galectin-3. J. Immunol. 2008, 180, 2650–2658. [Google Scholar] [CrossRef]
- Tan, T.K.; Zheng, G.; Hsu, T.-T.; Wang, Y.; Lee, V.W.; Tian, X.; Wang, Y.; Cao, Q.; Wang, Y.; Harris, D.C. Macrophage matrix metalloproteinase-9 mediates epithelial-mesenchymal transition in vitro in murine renal tubular cells. Am. J. Pathol. 2010, 176, 1256–1270. [Google Scholar] [CrossRef] [PubMed]
- Smigiel, K.S.; Parks, W.C. Macrophages, wound healing, and fibrosis: Recent insights. Curr. Rheumatol. Rep. 2018, 20, 1–8. [Google Scholar] [CrossRef]
- Duffield, J.S.; Ware, C.F.; Ryffel, B.; Savill, J. Suppression by apoptotic cells defines tumor necrosis factor-mediated induction of glomerular mesangial cell apoptosis by activated macrophages. Am. J. Pathol. 2001, 159, 1397–1404. [Google Scholar] [CrossRef]
- Duffield, J.S.; Erwig, L.-P.; Wei, X.-q.; Liew, F.Y.; Rees, A.J.; Savill, J.S. Activated macrophages direct apoptosis and suppress mitosis of mesangial cells. J. Immunol. 2000, 164, 2110–2119. [Google Scholar] [CrossRef]
- Liu, Y. Cellular and molecular mechanisms of renal fibrosis. Nat. Rev. Nephrol. 2011, 7, 684–696. [Google Scholar] [CrossRef] [PubMed]
- Leonhard, W.N.; Kunnen, S.J.; Plugge, A.J.; Pasternack, A.; Jianu, S.B.; Veraar, K.; El Bouazzaoui, F.; Hoogaars, W.M.; Ten Dijke, P.; Breuning, M.H. Inhibition of activin signaling slows progression of polycystic kidney disease. J. Am. Soc. Nephrol. 2016, 27, 3589–3599. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, S.; Maeshima, A.; Kojima, I.; Nojima, Y. Activin A is a potent activator of renal interstitial fibroblasts. J. Am. Soc. Nephrol. 2004, 15, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Nowak, K.L.; Edelstein, C.L. Apoptosis and autophagy in polycystic kidney disease (PKD). Cell Signal 2020, 68, 109518. [Google Scholar] [CrossRef]
- Peintner, L.; Borner, C. Role of apoptosis in the development of autosomal dominant polycystic kidney disease (ADPKD). Cell Tissue Res. 2017, 369, 27–39. [Google Scholar] [CrossRef]
- Lossi, L. The concept of intrinsic versus extrinsic apoptosis. Biochem. J. 2022, 479, 357–384. [Google Scholar] [CrossRef]
- Kari, S.; Subramanian, K.; Altomonte, I.A.; Murugesan, A.; Yli-Harja, O.; Kandhavelu, M. Programmed cell death detection methods: A systematic review and a categorical comparison. Apoptosis 2022, 27, 482–508. [Google Scholar] [CrossRef]
- Venugopal, J.; Blanco, G. Ouabain enhances ADPKD cell apoptosis via the intrinsic pathway. Front. Physiol. 2016, 7, 107. [Google Scholar] [CrossRef]
- Lee, O.Y.A.; Wong, A.N.N.; Ho, C.Y.; Tse, K.W.; Chan, A.Z.; Leung, G.P.-H.; Kwan, Y.W.; Yeung, M.H.Y. Potentials of natural antioxidants in reducing inflammation and oxidative stress in chronic kidney disease. Antioxidants 2024, 13, 751. [Google Scholar] [CrossRef] [PubMed]
- Elmore, S. Apoptosis: A Review of Programmed Cell Death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Mallamaci, F.; Zoccali, C.; Parlongo, S.; Cutrupi, S.; Tripepi, G.; Postorino, M. Plasma adrenomedullin during acute changes in intravascular volume in hemodialysis patients. Kidney Int. 1998, 54, 1697–1703. [Google Scholar] [CrossRef]
- Podrini, C.; Cassina, L.; Boletta, A. Metabolic reprogramming and the role of mitochondria in polycystic kidney disease. Cell. Signal. 2020, 67, 109495. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhu, H.; Xu, C.J.; Yuan, J. Cleavage of BID by Caspase 8 Mediates the Mitochondrial Damage in the Fas Pathway of Apoptosis. Cell 1998, 94, 491–501. [Google Scholar] [CrossRef]
- Torres, V.E.; Harris, P.C. Autosomal dominant polycystic kidney disease: The last 3 years. Kidney Int. 2009, 76, 149–168. [Google Scholar] [CrossRef]
- Stel, V.S.; van Dijk, P.C.W.; van Manen, J.G.; Dekker, F.W.; Ansell, D.; Conte, F.; Kramar, R.; Leivestad, T.; Vela, E.; Briggs, J.D.; et al. Prevalence of co-morbidity in different European RRT populations and its effect on access to renal transplantation. Nephrol. Dial. Transplant. 2005, 20, 2803–2811. [Google Scholar] [CrossRef]
- Subhash, S.; Vijayvargiya, S.; Parmar, A.; Sandhu, J.; Simmons, J.; Raina, R. Reactive Oxygen Species in Cystic Kidney Disease. Antioxidants 2024, 13, 1186. [Google Scholar] [CrossRef]
- Gonzalez-Vicente, A.; Hong, N.; Garvin, J.L. Effects of reactive oxygen species on renal tubular transport. Am. J. Physiol. Ren. Physiol. 2019, 317, F444–F455. [Google Scholar] [CrossRef]
- Su, H.; Wan, C.; Song, A.; Qiu, Y.; Xiong, W.; Zhang, C. Oxidative stress and renal fibrosis: Mechanisms and therapies. In Renal Fibrosis: Mechanisms and Therapy; Lan, H.Y., Liu, Y., Eds.; Springer: Singapore, 2019; pp. 585–604. [Google Scholar]
- Kahveci, A.S.; Barnatan, T.T.; Kahveci, A.; Adrian, A.E.; Arroyo, J.; Eirin, A.; Harris, P.C.; Lerman, A.; Lerman, L.O.; Torres, V.E. Oxidative stress and mitochondrial abnormalities contribute to decreased endothelial nitric oxide synthase expression and renal disease progression in early experimental polycystic kidney disease. Int. J. Mol. Sci. 2020, 21, 1994. [Google Scholar] [CrossRef] [PubMed]
- Fedeles, B.I.; Bhardwaj, R.; Ishikawa, Y.; Khumsubdee, S.; Krappitz, M.; Gubina, N.; Volpe, I.; Andrade, D.C.; Westergerling, P.; Staudner, T. A synthetic agent ameliorates polycystic kidney disease by promoting apoptosis of cystic cells through increased oxidative stress. Proc. Natl. Acad. Sci. USA 2024, 121, e2317344121. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, N.; Gambhir, K.; Kumar, S.; Gangenahalli, G.; Verma, Y.K. Interplay of reactive oxygen species (ROS) and tissue engineering: A review on clinical aspects of ROS-responsive biomaterials. J. Mater. Sci. 2021, 56, 16790–16823. [Google Scholar] [CrossRef]
- Menezes, L.F.; Germino, G.G. The pathobiology of polycystic kidney disease from a metabolic viewpoint. Nat. Rev. Nephrol. 2019, 15, 735–749. [Google Scholar] [CrossRef]
- Klemens, C.A.; Fedoriuk, M.; Semenikhina, M.; Stefanenko, M.; Zietara, A.; Levchenko, V.; Dissanayake, L.V.; Palygin, O.; Staruschenko, A. Electrolyte and metabolite composition of cystic fluid from a rat model of ARPKD. Commun. Biol. 2025, 8, 230. [Google Scholar] [CrossRef]
- Irazabal, M.V.; Torres, V.E. Reactive oxygen species and redox signaling in chronic kidney disease. Cells 2020, 9, 1342. [Google Scholar] [CrossRef]
- Haque, M.Z.; Ortiz, P.A. Superoxide increases surface NKCC2 in the rat thick ascending limbs via PKC. Am. J. Physiol. Ren. Physiol. 2019, 317, F99–F106. [Google Scholar] [CrossRef]
- Maskey, D.; Granados Pineda, J.; Ortiz, P.A. Update on NKCC2 regulation in the thick ascending limb (TAL) by membrane trafficking, phosphorylation, and protein-protein interactions. Front. Physiol. 2024, 15, 1508806. [Google Scholar] [CrossRef]
- Gonzalez-Vicente, A.; Saez, F.; Monzon, C.M.; Asirwatham, J.; Garvin, J.L. Thick ascending limb sodium transport in the pathogenesis of hypertension. Physiol. Rev. 2018, 99, 235–309. [Google Scholar] [CrossRef]
- Sun, P.; Yue, P.; Wang, W.-H. Angiotensin II stimulates epithelial sodium channels in the cortical collecting duct of the rat kidney. Am. J. Physiol. Ren. Physiol. 2012, 302, F679–F687. [Google Scholar] [CrossRef] [PubMed]
- Ramkumar, N.; Stuart, D.; Mironova, E.; Abraham, N.; Gao, Y.; Wang, S.; Lakshmipathi, J.; Stockand, J.D.; Kohan, D.E. Collecting duct principal, but not intercalated, cell prorenin receptor regulates renal sodium and water excretion. Am. J. Physiol. Ren. Physiol. 2018, 315, F607–F617. [Google Scholar] [CrossRef]
- Trac, D.; Liu, B.; Pao, A.C.; Thomas, S.V.; Park, M.; Downs, C.A.; Ma, H.-P.; Helms, M.N. Fulvene-5 inhibition of Nadph oxidases attenuates activation of epithelial sodium channels in A6 distal nephron cells. Am. J. Physiol. Ren. Physiol. 2013, 305, F995–F1005. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Bao, H.-F.; Self, J.L.; Eaton, D.C.; Helms, M.N. Aldosterone-induced increases in superoxide production counters nitric oxide inhibition of epithelial Na channel activity in A6 distal nephron cells. Am. J. Physiol. Ren. Physiol. 2007, 293, F1666–F1677. [Google Scholar] [CrossRef] [PubMed]
- Martinou, J.-C.; Féraille, E.; de Seigneux, D.S. ROLE OF NADPH Oxidase 4 (NOX4) in the Pathophysiology of Kidney Diseases. Ph.D. Thesis, Université de Genève, Geneva, Switzerland, 2014. [Google Scholar]
- Fu, Y.; Zhang, R.; Lu, D.; Liu, H.; Chandrashekar, K.; Juncos, L.A.; Liu, R. NOX2 is the primary source of angiotensin II-induced superoxide in the macula densa. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R707–R712. [Google Scholar] [CrossRef]
- Xu, N.; Jiang, S.; Persson, P.B.; Persson, E.A.; Lai, E.Y.; Patzak, A. Reactive oxygen species in renal vascular function. Acta Physiol. 2020, 229, e13477. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, H.; Yi, X.; Dou, Q.; Yang, X.; He, Y.; Chen, J.; Chen, K. Cellular senescence of renal tubular epithelial cells in acute kidney injury. Cell Death Discov. 2024, 10, 62. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, D.; Choi, M.E. Autophagy and mitophagy: Physiological implications in kidney inflammation and diseases. Am. J. Physiol. Ren. Physiol. 2023, 325, F1–F21. [Google Scholar] [CrossRef]
- Cybulsky, A.V. Endoplasmic reticulum stress, the unfolded protein response and autophagy in kidney diseases. Nat. Rev. Nephrol. 2017, 13, 681–696. [Google Scholar] [CrossRef]
- Gallazzini, M.; Pallet, N. Endoplasmic reticulum stress and kidney dysfunction. Biol. Cell 2018, 110, 205–216. [Google Scholar] [CrossRef]
- Richards, T.; Modarage, K.; Malik, S.A.; Goggolidou, P. The cellular pathways and potential therapeutics of Polycystic Kidney Disease. Biochem. Soc. Trans. 2021, 49, 1171–1188. [Google Scholar] [CrossRef]
- Blazer-Yost, B.L.; Wallace, D.P. The Polycystins and Polycystic Kidney Disease. In Studies of Epithelial Transporters and Ion Channels: Ion Channels and Transporters of Epithelia in Health and Disease-Vol. 3; Springer: Berlin/Heidelberg, Germany, 2021; pp. 1149–1186. [Google Scholar]
- Tomilin, V.N.; Pochynyuk, O. A peek into Epac physiology in the kidney. Am. J. Physiol. Ren. Physiol. 2019, 317, F1094–F1097. [Google Scholar] [CrossRef] [PubMed]
- Pochynyuk, O.; Pyrshev, K.; Cheng, X. Multifaceted roles of Epac signaling in renal functions. Biochem. J. 2025, 482, 553–568. [Google Scholar] [CrossRef]
- Torres, V.E.; Harris, P.C. Strategies targeting cAMP signaling in the treatment of polycystic kidney disease. J. Am. Soc. Nephrol. 2014, 25, 18–32. [Google Scholar] [CrossRef]
- Zhang, C.; Balbo, B.; Ma, M.; Zhao, J.; Tian, X.; Kluger, Y.; Somlo, S. Cyclin-dependent kinase 1 activity is a driver of cyst growth in polycystic kidney disease. J. Am. Soc. Nephrol. 2021, 32, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.; Wallace, D.P.; Magenheimer, B.S.; Hempson, S.J.; Grantham, J.J.; Calvet, J.P. Calcium restriction allows cAMP activation of the B-Raf/ERK pathway, switching cells to a cAMP-dependent growth-stimulated phenotype. J. Biol. Chem. 2004, 279, 40419–40430. [Google Scholar] [CrossRef]
- Parnell, S.C.; Raman, A.; Zhang, Y.; Daniel, E.A.; Dai, Y.; Khanna, A.; Reif, G.A.; Vivian, J.L.; Fields, T.A.; Wallace, D.P. Expression of active B-Raf proto-oncogene in kidney collecting ducts induces cyst formation in normal mice and accelerates cyst growth in mice with polycystic kidney disease. Kidney Int. 2022, 102, 1103–1114. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Reif, G.A.; Calvet, J.P.; Wallace, D.P. Sorafenib inhibits cAMP-dependent ERK activation, cell proliferation, and in vitro cyst growth of human ADPKD cyst epithelial cells. Am. J. Physiol. Ren. Physiol. 2010, 299, F944–F951. [Google Scholar] [CrossRef]
- Wang, X.; Jiang, L.; Thao, K.; Sussman, C.R.; LaBranche, T.; Palmer, M.; Harris, P.C.; McKnight, G.S.; Hoeflich, K.P.; Schalm, S.; et al. Protein Kinase A Downregulation Delays the Development and Progression of Polycystic Kidney Disease. J. Am. Soc. Nephrol. 2022, 33, 1087–1104. [Google Scholar] [CrossRef]
- Prosseda, P.P.; Dannewitz Prosseda, S.; Tran, M.; Liton, P.B.; Sun, Y. Crosstalk between the mTOR pathway and primary cilia in human diseases. Curr. Top. Dev. Biol. 2023, 155, 1–37. [Google Scholar] [CrossRef]
- Lai, Y.; Jiang, Y. Reciprocal Regulation between Primary Cilia and mTORC1. Genes 2020, 11, 711. [Google Scholar] [CrossRef] [PubMed]
- Shillingford, J.M.; Murcia, N.S.; Larson, C.H.; Low, S.H.; Hedgepeth, R.; Brown, N.; Flask, C.A.; Novick, A.C.; Goldfarb, D.A.; Kramer-Zucker, A. The mTOR pathway is regulated by polycystin-1, and its inhibition reverses renal cystogenesis in polycystic kidney disease. Proc. Natl. Acad. Sci. USA 2006, 103, 5466–5471. [Google Scholar] [CrossRef]
- Zhu, J.; Liu, F.; Mao, J. Clinical findings, underlying pathogenetic processes and treatment of vascular dysfunction in autosomal dominant polycystic kidney disease. Ren. Fail. 2023, 45, 2282027. [Google Scholar] [CrossRef]
- Laplante, M.; Sabatini, D.M. mTOR signaling in growth control and disease. Cell 2012, 149, 274–293. [Google Scholar] [CrossRef]
- Ponticelli, C.; Moroni, G.; Reggiani, F. Autosomal dominant polycystic kidney disease: Is there a role for autophagy? Int. J. Mol. Sci. 2023, 24, 14666. [Google Scholar] [CrossRef] [PubMed]
- Holditch, S.J.; Brown, C.N.; Atwood, D.J.; Lombardi, A.M.; Nguyen, K.N.; Toll, H.W.; Hopp, K.; Edelstein, C.L. A study of sirolimus and mTOR kinase inhibitor in a hypomorphic Pkd1 mouse model of autosomal dominant polycystic kidney disease. Am. J. Physiol. Ren. Physiol. 2019, 317, F187–F196. [Google Scholar] [CrossRef] [PubMed]
- Tran Nguyen Truc, L.; Matsuda, S.; Miyazaki, T.; Yoshizawa, K.; Tsukaguchi, H. WCN23-0154 Cross-talk of Cilia Sensors and mTOR Signaling Pathway in Cystogenesis of Mouse Polycystic Kidney Disease Model. Kidney Int. Rep. 2023, 8, S208–S209. [Google Scholar] [CrossRef]
- Iliuta, I.-A.; Song, X.; Pickel, L.; Haghighi, A.; Retnakaran, R.; Scholey, J.; Sung, H.-K.; Steinberg, G.R.; Pei, Y. Shared pathobiology identifies AMPK as a therapeutic target for obesity and autosomal dominant polycystic kidney disease. Front. Mol. Biosci. 2022, 9, 962933. [Google Scholar] [CrossRef] [PubMed]
- Saxena, S.; Dagar, N.; Shelke, V.; Puri, B.; Gaikwad, A.B. Wnt/beta-catenin modulation: A promising frontier in chronic kidney disease management. Fundam. Clin. Pharmacol. 2024, 38, 1020–1030. [Google Scholar] [CrossRef]
- Agborbesong, E. Characterizing the Role of the Lysine Methyltransferase Smyd3 and Antioxidant Enzyme Prdx5 in Cysto-genesis and Ciliogenesis in Autosomal Dominant Polycystic Kidney Disease. Ph.D. Thesis, University of Kansas, Lawrence, KS, USA, 2022. [Google Scholar]
- Papavassiliou, K. Molecular Pathways of Polycystin-1 (PC1) Implicated in the Initiation and Progression of Tumors. Ph.D. Thesis, Laboratory of Physiology, Department of Medicine, School of Health Sciences, National and Kapodistrian University of Athens, Athens, Greece, 2019. [Google Scholar]
- Miller, M.M.; Iglesias, D.M.; Zhang, Z.; Corsini, R.; Chu, L.; Murawski, I.; Gupta, I.; Somlo, S.; Germino, G.G.; Goodyer, P.R. T-cell factor/β-catenin activity is suppressed in two different models of autosomal dominant polycystic kidney disease. Kidney Int. 2011, 80, 146–153. [Google Scholar] [CrossRef]
- Paul, B.M.; Vanden Heuvel, G.B. Kidney: Polycystic kidney disease. Wiley Interdiscip. Rev. Dev. Biol. 2014, 3, 465–487. [Google Scholar] [CrossRef]
- Lemos, F.O.; Ehrlich, B.E. Polycystin and calcium signaling in cell death and survival. Cell Calcium 2018, 69, 37–45. [Google Scholar] [CrossRef]
- Kim, S.; Nie, H.; Nesin, V.; Tran, U.; Outeda, P.; Bai, C.-X.; Keeling, J.; Maskey, D.; Watnick, T.; Wessely, O. The polycystin complex mediates Wnt/Ca2+ signalling. Nat. Cell Biol. 2016, 18, 752–764. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.; Hiesberger, T.; Cordes, K.; Sinclair, A.M.; Goldstein, L.S.; Somlo, S.; Igarashi, P. Kidney-specific inactivation of the KIF3A subunit of kinesin-II inhibits renal ciliogenesis and produces polycystic kidney disease. Proc. Natl. Acad. Sci. USA 2003, 100, 5286–5291. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Xu, Y.; Fan, S.; Meng, J.; Shen, X.; Xiao, Q.; Li, Y.; Zhang, L.; Zhang, X.; Wu, G. Canonical Wnt inhibitors ameliorate cystogenesis in a mouse ortholog of human ADPKD. JCI Insight 2018, 3, e95874. [Google Scholar] [CrossRef] [PubMed]
- Konno, H.; Barber, G.N. The STING controlled cytosolic-DNA activated innate immune pathway and microbial disease. Microbes Infect. 2014, 16, 998–1001. [Google Scholar] [CrossRef]
- Chen, P.-L.; Chen, C.-F.; Lin, H.Y.-H.; Riley, D.J.; Chen, Y. The Link between Autosomal Dominant Polycystic Kidney Disease and Chromosomal Instability: Exploring the Relationship. Int. J. Mol. Sci. 2024, 25, 2936. [Google Scholar] [CrossRef]
- Ma, J.; Setton, J.; Lee, N.Y.; Riaz, N.; Powell, S.N. The therapeutic significance of mutational signatures from DNA repair deficiency in cancer. Nat. Commun. 2018, 9, 3292. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Z.; Zhang, H.; Zhou, M.; Huang, C.; Xia, W.; Li, J.; You, H. The effects of cGAS-STING inhibition in liver disease, kidney disease, and cellular senescence. Front. Immunol. 2024, 15, 1346446. [Google Scholar] [CrossRef]
- Yoo, M.; Haydak, J.C.; Azeloglu, E.U.; Lee, K.; Gusella, G.L. cGAS Activation Accelerates the Progression of Autosomal Dominant Polycystic Kidney Disease. J. Am. Soc. Nephrol. 2024, 35, 466–482. [Google Scholar] [CrossRef]
- Wu, J.; Cheng, S.; Lee, G.; Agborbesong, E.; Li, X.; Zhou, X.; Li, X. STING Promotes the Progression of ADPKD by Regulating Mitochondrial Function, Inflammation, Fibrosis, and Apoptosis. Biomolecules 2024, 14, 1215. [Google Scholar] [CrossRef]
- Ruiz-Ortega, M.; Ortiz, A.; Ramos, A.M. Tumor necrosis factor-like weak inducer of apoptosis (TWEAK) and kidney disease. Curr. Opin. Nephrol. Hypertens. 2014, 23, 93–100. [Google Scholar] [CrossRef]
- Cordido, A.; Nuñez-Gonzalez, L.; Martinez-Moreno, J.M.; Lamas-Gonzalez, O.; Rodriguez-Osorio, L.; Perez-Gomez, M.V.; Martin-Sanchez, D.; Outeda, P.; Chiaravalli, M.; Watnick, T.; et al. TWEAK Signaling Pathway Blockade Slows Cyst Growth and Disease Progression in Autosomal Dominant Polycystic Kidney Disease. J. Am. Soc. Nephrol. 2021, 32, 1913–1932. [Google Scholar] [CrossRef]
- Müller, R.U.; Schermer, B. Hippo signaling-a central player in cystic kidney disease? Pediatr. Nephrol. 2020, 35, 1143–1152. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Zhang, Z.; Liu, T.M.; Ge, W. Novel zebrafish polycystic kidney disease models reveal functions of the Hippo pathway in renal cystogenesis. Dis. Model. Mech. 2021, 14, dmm049027. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Guan, K.L. Polycystic kidney disease: A Hippo connection. Genes Dev. 2018, 32, 737–739. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Song, X.; Wang, W.; Watnick, T.; Pei, Y.; Qian, F.; Pan, D. A RhoA-YAP-c-Myc signaling axis promotes the development of polycystic kidney disease. Genes Dev. 2018, 32, 781–793. [Google Scholar] [CrossRef]
- Liu, H.; Ma, H.; Li, Y.; Zhao, H. Advances in epigenetic modifications and cervical cancer research. Biochim. Et. Biophys. Acta (BBA) Rev. Cancer 2023, 1878, 188894. [Google Scholar] [CrossRef]
- Hamilton, J.P. Epigenetics: Principles and practice. Dig. Dis. 2011, 29, 130–135. [Google Scholar] [CrossRef]
- Yan, Z.; Cao, F.; Shao, T.; Liao, B.; Wang, G.; Tang, X.; Luo, H.; Zhu, F.; Liao, Y.; Zhang, F.; et al. Epigenetics in autosomal dominant polycystic kidney disease. Biochim. Biophys. Acta Mol. Basis Dis. 2025, 1871, 167652. [Google Scholar] [CrossRef]
- Agborbesong, E.; Li, L.X.; Li, L.; Li, X. Molecular Mechanisms of Epigenetic Regulation, Inflammation, and Cell Death in ADPKD. Front. Mol. Biosci. 2022, 9, 922428. [Google Scholar] [CrossRef]
- Li, X. Epigenetics in ADPKD: Understanding Mechanisms and Discovering Treatment. In Polycystic Kidney Disease; Li, X., Ed.; Codon Publications: Brisbane, Australia, 2015. [Google Scholar]
- Woo, Y.M.; Bae, J.B.; Oh, Y.H.; Lee, Y.G.; Lee, M.J.; Park, E.Y.; Choi, J.K.; Lee, S.; Shin, Y.; Lyu, J.; et al. Genome-wide methylation profiling of ADPKD identified epigenetically regulated genes associated with renal cyst development. Hum. Genet. 2014, 133, 281–297. [Google Scholar] [CrossRef]
- Hajirezaei, F.; Ghaderian, S.M.H.; Hasanzad, M.; Nafar, M.; Ghadiani, M.H.; Biglari, S.; Sohrabifar, N.; Jafari, H. Methylation of the PKD1 Promoter Inversely Correlates with its Expression in Autosomal Dominant Polycystic Kidney Disease. Rep. Biochem. Mol. Biol. 2020, 9, 193–198. [Google Scholar] [CrossRef]
- Li, X. Epigenetics and autosomal dominant polycystic kidney disease. Biochim. Biophys. Acta 2011, 1812, 1213–1218. [Google Scholar] [CrossRef]
- Gui, Y.; Dai, C. mTOR signaling in kidney diseases. Kidney360 2020, 1, 1319–1327. [Google Scholar] [CrossRef]
- Alessi, D.R.; Pearce, L.R.; Garcia-Martinez, J.M. New insights into mTOR signaling: mTORC2 and beyond. Sci. Signal. 2009, 2, pe27. [Google Scholar] [CrossRef] [PubMed]
- Boletta, A. Emerging evidence of a link between the polycystins and the mTOR pathways. Pathogenetics 2009, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Messing, M.; Torres, J.A.; Holznecht, N.; Weimbs, T. Trigger Warning: How Modern Diet, Lifestyle, and Environment Pull the Trigger on Autosomal Dominant Polycystic Kidney Disease Progression. Nutrients 2024, 16, 3281. [Google Scholar] [CrossRef] [PubMed]
- Trinh, A.; Huang, Y.; Shao, H.; Ram, A.; Morival, J.; Wang, J.; Chung, E.J.; Downing, T.L. Targeting the ADPKD methylome using nanoparticle-mediated combination therapy. APL Bioeng. 2023, 7, 026111. [Google Scholar] [CrossRef]
- Fontecha-Barriuso, M.; Martin-Sanchez, D.; Ruiz-Andres, O.; Poveda, J.; Sanchez-Nino, M.D.; Valino-Rivas, L.; Ruiz-Ortega, M.; Ortiz, A.; Sanz, A.B. Targeting epigenetic DNA and histone modifications to treat kidney disease. Nephrol. Dial. Transpl. 2018, 33, 1875–1886. [Google Scholar] [CrossRef]
- Sun, L.; Hu, C.; Zhang, X. Histone Deacetylase Inhibitors Reduce Cysts by Activating Autophagy in Polycystic Kidney Disease. Kidney Dis. 2019, 5, 163–172. [Google Scholar] [CrossRef]
- Xiong, C.; Masucci, M.V.; Zhou, X.; Liu, N.; Zang, X.; Tolbert, E.; Zhao, T.C.; Zhuang, S. Pharmacological targeting of BET proteins inhibits renal fibroblast activation and alleviates renal fibrosis. Oncotarget 2016, 7, 69291–69308. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Park, S.J.; Lee, M.H.; Kronbichler, A.; Shin, J.I. CRISPR-Cas9 system in autosomal dominant polycystic kidney disease: A comprehensive review. Child. Kidney Dis. 2025, 29, 4–11. [Google Scholar] [CrossRef]
- Xu, J.; Xue, C.; Wang, X.; Zhang, L.; Mei, C.; Mao, Z. Chromatin Methylation Abnormalities in Autosomal Dominant Polycystic Kidney Disease. Front Med. 2022, 9, 921631. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Zhu, Q.; Ma, S.; Peng, X.; Huang, G.; Liu, G.; Zhang, W.; Zhang, Y.; Jiang, C.; Qiu, A.; et al. Comprehensive single-cell transcriptomic analysis reveals cellular heterogeneity and immune landscape in autosomal dominant polycystic kidney disease. bioRxiv 2025. preprint. [Google Scholar] [CrossRef]
- Grampp, S.; Kraus, A.; Skoczynski, K.; Schiffer, M.; Kruger, R.; Naas, S.; Schodel, J.; Buchholz, B. Hypoxia induces polycystin-1 expression in the renal epithelium. R. Soc. Open Sci. 2023, 10, 220992. [Google Scholar] [CrossRef]
- Aboudehen, K.; Farahani, S.; Kanchwala, M.; Chan, S.C.; Avdulov, S.; Mickelson, A.; Lee, D.; Gearhart, M.D.; Patel, V.; Xing, C.; et al. Long noncoding RNA Hoxb3os is dysregulated in autosomal dominant polycystic kidney disease and regulates mTOR signaling. J. Biol. Chem. 2018, 293, 9388–9398. [Google Scholar] [CrossRef]
- Hajarnis, S.; Lakhia, R.; Patel, V. MicroRNAs and polycystic kidney disease. In Polycystic Kidney Disease; Li, X., Ed.; Codon Publications: Brisbane, Australia, 2015; pp. 313–334. [Google Scholar]
- Fragiadaki, M. Lessons from microRNA biology: Top key cellular drivers of Autosomal Dominant Polycystic Kidney Disease. Biochim. Biophys. Acta Mol. Basis Dis. 2022, 1868, 166358. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, M.A.; Gleeson, J.G. Cystic kidney disease: The role of Wnt signaling. Trends Mol. Med. 2010, 16, 349–360. [Google Scholar] [CrossRef]
- Tran, U.; Streets, A.J.; Smith, D.; Decker, E.; Kirschfink, A.; Izem, L.; Hassey, J.M.; Rutland, B.; Valluru, M.K.; Bräsen, J.H. BICC1 Interacts with PKD1 and PKD2 to Drive Cystogenesis in ADPKD. bioRxiv 2024. [Google Scholar] [CrossRef]
- Ghanem, A.; Borghol, A.H.; Munairdjy Debeh, F.G.; Paul, S.; AlKhatib, B.; Harris, P.C.; Garimella, P.S.; Hanna, C.; Kline, T.L.; Dahl, N.K.; et al. Biomarkers of Kidney Disease Progression in ADPKD. Kidney Int. Rep. 2024, 9, 2860–2882. [Google Scholar] [CrossRef] [PubMed]
- Soric Hosman, I.; Cvitkovic Roic, A.; Fistrek Prlic, M.; Vukovic Brinar, I.; Lamot, L. Predicting autosomal dominant polycystic kidney disease progression: Review of promising Serum and urine biomarkers. Front. Pediatr. 2023, 11, 1274435. [Google Scholar] [CrossRef]
- Muto, Y.; Yoshimura, Y.; Wu, H.; Chang-Panesso, M.; Ledru, N.; Woodward, O.M.; Outeda, P.; Cheng, T.; Mahjoub, M.R.; Watnick, T.J.; et al. Multi-omics profiling of mouse polycystic kidney disease progression at a single cell resolution. bioRxiv 2024. [Google Scholar] [CrossRef]
- Jiang, X.; Xu, X.; Wu, M.; Guan, Z.; Su, X.; Chen, S.; Wang, H.; Teng, L. GPRC5A: An Emerging Biomarker in Human Cancer. Biomed. Res. Int. 2018, 2018, 1823726. [Google Scholar] [CrossRef] [PubMed]
- Daniel, E.A.; Sommer, N.A.; Sharma, M. Polycystic kidneys: Interaction of notch and renin. Clin. Sci. 2023, 137, 1145–1150. [Google Scholar] [CrossRef]
- Ren, H.; Mu, C.; Wang, Y.; Cheng, Y.; Hou, Y.; Li, Y.; Liu, N.; Yin, Z.; Xiong, H.; Chen, Y.; et al. Notch2 Inhibition and Kidney Cyst Growth in Autosomal Dominant Polycystic Kidney Disease. J. Am. Soc. Nephrol. 2025, 36, 781–797. [Google Scholar] [CrossRef] [PubMed]
- Yoo, K.H.; Hennighausen, L. EZH2 methyltransferase and H3K27 methylation in breast cancer. Int. J. Biol. Sci. 2012, 8, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Lv, J.; Lan, B.; Fu, L.; He, C.; Zhou, W.; Wang, X.; Zhou, C.; Mao, Z.; Chen, Y.; Mei, C.; et al. EZH2 inhibition or genetic ablation suppresses cyst growth in autosomal dominant polycystic kidney disease. J. Transl. Med. 2024, 22, 979. [Google Scholar] [CrossRef]
- Sieben, C.J.; Harris, P.C. Experimental Models of Polycystic Kidney Disease: Applications and Therapeutic Testing. Kidney360 2023, 4, 1155–1173. [Google Scholar] [CrossRef]
- Shimizu, T.; Mae, S.-I.; Araoka, T.; Okita, K.; Hotta, A.; Yamagata, K.; Osafune, K. A novel ADPKD model using kidney organoids derived from disease-specific human iPSCs. Biochem. Biophys. Res. Commun. 2020, 529, 1186–1194. [Google Scholar] [CrossRef]
- Freedman, B.S.; Brooks, C.R.; Lam, A.Q.; Fu, H.; Morizane, R.; Agrawal, V.; Saad, A.F.; Li, M.K.; Hughes, M.R.; Werff, R.V.; et al. Modelling kidney disease with CRISPR-mutant kidney organoids derived from human pluripotent epiblast spheroids. Nat. Commun. 2015, 6, 8715. [Google Scholar] [CrossRef]
- Mae, S.I.; Hattanda, F.; Morita, H.; Nozaki, A.; Katagiri, N.; Ogawa, H.; Teranaka, K.; Nishimura, Y.; Kudoh, A.; Yamanaka, S.; et al. Human iPSC-derived renal collecting duct organoid model cystogenesis in ADPKD. Cell Rep. 2023, 42, 113431. [Google Scholar] [CrossRef]
- Tran, T.; Song, C.J.; Nguyen, T.; Cheng, S.-Y.; McMahon, J.A.; Yang, R.; Guo, Q.; Der, B.; Lindström, N.O.; Lin, D.C.-H. A scalable organoid model of human autosomal dominant polycystic kidney disease for disease mechanism and drug discovery. Cell Stem Cell 2022, 29, 1083–1101.e1087. [Google Scholar] [CrossRef] [PubMed]
- Czerniecki, S.M.; Cruz, N.M.; Harder, J.L.; Menon, R.; Annis, J.; Otto, E.A.; Gulieva, R.E.; Islas, L.V.; Kim, Y.K.; Tran, L.M.; et al. High-Throughput Screening Enhances Kidney Organoid Differentiation from Human Pluripotent Stem Cells and Enables Automated Multidimensional Phenotyping. Cell Stem Cell 2018, 22, 929–940.e924. [Google Scholar] [CrossRef]
- Subramanian, B.; Rudym, D.; Cannizzaro, C.; Perrone, R.; Zhou, J.; Kaplan, D.L. Tissue-Engineered Three-Dimensional In Vitro Models for Normal and Diseased Kidney. Tissue Eng. Part. A 2010, 16, 2821–2831. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Nagao, S.; Wallace, D.P.; Belibi, F.A.; Cowley, B.D.; Pelling, J.C.; Grantham, J.J. Cyclic AMP activates B-Raf and ERK in cyst epithelial cells from autosomal-dominant polycystic kidneys. Kidney Int. 2003, 63, 1983–1994. [Google Scholar] [CrossRef]
- Booij, T.H.; Bange, H.; Leonhard, W.N.; Yan, K.; Fokkelman, M.; Kunnen, S.J.; Dauwerse, J.G.; Qin, Y.; van de Water, B.; van Westen, G.J.P.; et al. High-Throughput Phenotypic Screening of Kinase Inhibitors to Identify Drug Targets for Polycystic Kidney Disease. SLAS Discov. Adv. Sci. Drug Discov. 2017, 22, 974–984. [Google Scholar] [CrossRef]
- Musah, S.; Mammoto, A.; Ferrante, T.C.; Jeanty, S.S.F.; Hirano-Kobayashi, M.; Mammoto, T.; Roberts, K.; Chung, S.; Novak, R.; Ingram, M.; et al. Mature induced-pluripotent-stem-cell-derived human podocytes reconstitute kidney glomerular-capillary-wall function on a chip. Nat. Biomed. Eng. 2017, 1, 0069. [Google Scholar] [CrossRef]
- Dewhurst, R.M.; Molinari, E.; Sayer, J.A. Spheroids, organoids and kidneys-on-chips: How complex human cellular models have assisted in the study of kidney disease and renal ciliopathies. Microfluid. Nanofluidics 2023, 27, 21. [Google Scholar] [CrossRef]
- Takasato, M.; Er, P.X.; Chiu, H.S.; Maier, B.; Baillie, G.J.; Ferguson, C.; Parton, R.G.; Wolvetang, E.J.; Roost, M.S.; Chuva de Sousa Lopes, S.M.; et al. Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis. Nature 2015, 526, 564–568. [Google Scholar] [CrossRef] [PubMed]
- Hosszu, A.; Kaucsar, T.; Seeliger, E.; Fekete, A. Animal Models of Renal Pathophysiology and Disease. Methods Mol. Biol. 2021, 2216, 27–44. [Google Scholar] [CrossRef]
- Shibazaki, S.; Yu, Z.; Nishio, S.; Tian, X.; Thomson, R.B.; Mitobe, M.; Louvi, A.; Velazquez, H.; Ishibe, S.; Cantley, L.G.; et al. Cyst formation and activation of the extracellular regulated kinase pathway after kidney specific inactivation of Pkd1. Hum. Mol. Genet. 2008, 17, 1505–1516. [Google Scholar] [CrossRef] [PubMed]
- Traykova-Brauch, M.; Schönig, K.; Greiner, O.; Miloud, T.; Jauch, A.; Bode, M.; Felsher, D.W.; Glick, A.B.; Kwiatkowski, D.J.; Bujard, H.; et al. An efficient and versatile system for acute and chronic modulation of renal tubular function in transgenic mice. Nat. Med. 2008, 14, 979–984. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.X.; Torres, V.E. Drug repurposing in autosomal dominant polycystic kidney disease. Kidney Int. 2023, 103, 859–871. [Google Scholar] [CrossRef] [PubMed]
- Sanzen, T.; Harada, K.; Yasoshima, M.; Kawamura, Y.; Ishibashi, M.; Nakanuma, Y. Polycystic kidney rat is a novel animal model of Caroli’s disease associated with congenital hepatic fibrosis. Am. J. Pathol. 2001, 158, 1605–1612. [Google Scholar] [CrossRef]
- Kofotolios, I.; Bonios, M.J.; Adamopoulos, M.; Mourouzis, I.; Filippatos, G.; Boletis, J.N.; Marinaki, S.; Mavroidis, M. The Han:SPRD Rat: A Preclinical Model of Polycystic Kidney Disease. Biomedicines 2024, 12, 1234. [Google Scholar] [CrossRef]
- Otto, E.A.; Trapp, M.L.; Schultheiss, U.T.; Helou, J.; Quarmby, L.M.; Hildebrandt, F. NEK8 mutations affect ciliary and centrosomal localization and may cause nephronophthisis. J. Am. Soc. Nephrol. 2008, 19, 587–592. [Google Scholar] [CrossRef]
- Tran, U.; Zakin, L.; Schweickert, A.; Agrawal, R.; Döger, R.; Blum, M.; De Robertis, E.M.; Wessely, O. The RNA-binding protein bicaudal C regulates polycystin 2 in the kidney by antagonizing miR-17 activity. Development 2010, 137, 1107–1116. [Google Scholar] [CrossRef]
- Gehrig, J.; Pandey, G.; Westhoff, J.H. Zebrafish as a Model for Drug Screening in Genetic Kidney Diseases. Front. Pediatr. 2018, 6, 183. [Google Scholar] [CrossRef]
- He, J.; Wang, Q.; Ye, J.; Hu, X.; Li, N. Identification of porcine polycystic kidney disease 1 (PKD1) gene: Molecular cloning, expression profile, and implication in disease model. Gene 2011, 490, 37–46. [Google Scholar] [CrossRef]
- Gu, J.; Liu, F.; Li, L.; Mao, J. Advances and Challenges in Modeling Autosomal Dominant Polycystic Kidney Disease: A Focus on Kidney Organoids. Biomedicines 2025, 13, 523. [Google Scholar] [CrossRef]
- Marshall, L.J.; Bailey, J.; Cassotta, M.; Herrmann, K.; Pistollato, F. Poor Translatability of Biomedical Research Using Animals—A Narrative Review. Altern. Lab. Anim. 2023, 51, 102–135. [Google Scholar] [CrossRef] [PubMed]
| Pathway | Disease Stage Involved | Functional Role in ADPKD | Evidence Strength | Ref(s) |
|---|---|---|---|---|
| JAK/STAT3 | Cyst Growth | Promotes proliferation, survival, inflammation | Moderate | [54,55] |
| ROS/NOX4 | All Stages | Drives fibrosis, apoptosis, senescence via oxidative damage | Strong | [101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124,125,126,127,128,129,130,131] |
| cAMP/PKA/ERK | Initiation, Cyst Growth | Promotes epithelial proliferation and fluid secretion | Strong (preclinical and clinical) | [132,133,134,135,136,137,138,139] |
| mTOR | Cyst Growth | Drives cell proliferation, inhibits autophagy | Strong (preclinical and clinical trials) | [140,141,142,143,144,145,146,147,148,149,150] |
| Wnt/β-catenin | Initiation, Cyst Growth | Enhances proliferation, disrupts polarity | Moderate (animal and organoid models) | [151,152,153,154,155,156,157,158,159,160,161,162] |
| STING | Cyst Growth, Fibrosis | Promotes inflammation and fibrosis via NF-κB | Emerging (preclinical) | [163,164,165,166,167,168] |
| TWEAK/Fn14 | Cyst Growth, Fibrosis | Activates NF-κB and MAPK, increases proliferation and inflammation | Strong (mouse and human data) | [169,170] |
| Hippo/YAP/TAZ | Cyst Growth | Enhances proliferation via c-Myc with β-catenin cooperation | Emerging (mechanistic models) | [162,170,171,172,173,174] |
| Pathway | Activation in ADPKD | Main Downstream Effects | Therapeutic Potential and Modulators | Ref(s) |
|---|---|---|---|---|
| JAK/STAT | ↑ Activated | Proliferation, inflammation | JAK2 inhibitors (Preclinical) | [54,55] |
| ROS/NOX4 | ↑ Elevated | Apoptosis, fibrosis, mitochondrial dysfunction | MitoQ, antioxidants (In trials) | [101,102,103,104,105,106,107,108,109,110,111,112,113,114,115,116,117,118,119,120,121,122,123,124,125,126,127,128,129,130,131] |
| cAMP/PKA/ERK | ↑ Upregulated | Cell proliferation, CFTR-mediated fluid secretion | Tolvaptan (Approved) | [132,133,134,135,136,137,138,139,175,176,177] |
| mTORC1/2 | ↑ Activated | Protein synthesis, cell growth, suppressed autophagy | Sirolimus, Torin2 (Trials/Preclinical) | [140,141,142,143,144,145,146,147,148,149,150] |
| Wnt/β-catenin | ↑ Context-dependent | Proliferation, polarity loss, EMT | Wnt/β-catenin inhibitors (Preclinical) | [151,152,153,154,155,156,157,158,159,160,161,162] |
| STING | ↑ Upregulated | NF-κB–driven inflammation, fibrosis | C-176, cGAS inhibitors (Preclinical) | [163,164,165,166,167,168] |
| Hippo/YAP/TAZ | ↑ Nuclear accumulation | Drives c-Myc expression, proliferation | ROCK inhibitors (Y-27632) (Preclinical) | [162,170,171,172,173,174] |
| TWEAK/Fn14 | ↑ Overexpressed | MAPK/NF-κB activation, inflammation | Anti-TWEAK antibody (Preclinical) | [169,170] |
| Author(s) | Year | Species/System | PKD Gene/Pathway Targeted | Model Type/Intervention | Study Type | Key Observations/Comments |
|---|---|---|---|---|---|---|
| Lee et al. [9] | 2020 | Mouse | Wnt-β-catenin/TAZ | Endo-IWR1 | in vivo | β-catenin inhibition reduces cyst growth and proliferation |
| Shao et al. [24] | 2020 | Mouse | Ift88 + Pkd1 | Tg737orpk/orpk model | in vivo | Cilia loss impairs PC1/PC2 trafficking; double KO attenuates cystogenesis |
| Cabrita et al. [53] | 2020 | M1 cells | TMEM16A | PC1 loss/TMEM16A activation | in vitro | TMEM16A overactivity (due to PC1 loss) increases Ca2+ signaling and Cl− secretion, promoting cystogenesis |
| Patera et al. [54] | 2019 | Mouse/human renal tubules | JAK2/STAT3 | JAK2 expression and inhibition | in vivo/ex vivo | JAK2 is highly expressed in cystic tubules early in ADPKD; JAK2 inhibition reduces STAT3 activation and cyst growth |
| Zhang et al. [59] | 2020 | Mouse | TGF-β | TGF-β1 overexpression | in vivo | TGF-β1 promotes fibrosis and accelerates functional kidney decline in ADPKD |
| Qin et al. [61] | 2012 | Mouse | Pkd1 | Pkd1 KO | in vivo | β-catenin activation induces cysts; supports Wnt signaling |
| Liu et al. [63] | 2013 | Mouse | HGF, IGF-1 | Transgenic overexpression | in vivo | IGF-1/HGF promote cyst-lining epithelial proliferation |
| Dwivedi et al. [71] | 2023 | Mouse | ECM/myofibroblasts | Myofibroblast depletion | in vivo | Depleting myofibroblasts reduces ECM deposition and cyst expansion, alleviating fibrosis |
| Kim et al. [74] | 2022 | Mouse | ECM/αSMA+ cells | ECM accumulation/cell–matrix interaction | in vivo | αSMA+ myofibroblasts near cysts drive fibrosis and nephron loss, progressing ADPKD |
| Lee et al. [97] | 2024 | Mouse/renal epithelial cells | Cytochrome C/Apoptosome | Oxidative stress/antioxidant modulation | in vivo/in vitro | Oxidative stress promotes apoptosome formation via cytochrome C/APAF1/caspase-9 pathway; antioxidants show protective potential |
| Gonzalez-Vicente et al. [105] | 2019 | Rat nephron segments | ROS | ROS modulation | in vitro | ROS impairs transcellular and paracellular ion transport across nephron segments, contributing to dysfunction |
| Fedeles et al. [108] | 2024 | Mouse | NOX4/ROS | Synthetic agent/ROS modulation | in vivo | NOX4 overexpression in cyst-lining cells enhances ROS; pro-oxidant agent promotes apoptosis of cystic cells and reduces cyst burden |
| Klemens et al. [111] | 2025 | Rat model of ARPKD | Glycolysis/metabolism | Cystic fluid metabolomics | in vivo | PKD cystic cells shift to glycolysis (Warburg effect), altering electrolyte and metabolite profile |
| Haque & Ortiz [113] | 2019 | Rat thick ascending limb (TAL) | Superoxide/NKCC2 | PKC-mediated NKCC2 trafficking | in vitro | Superoxide upregulates NKCC2 activity via PKC, promoting sodium retention in PKD |
| Zhang et al. [132] | 2021 | Mouse | ERK/Cyclin-CDK | CDK1 activity assay | in vivo | ERK activation leads to CDK1 upregulation, driving cyst epithelial cell proliferation |
| Parnell et al. [134] | 2022 | Mouse (Pkd1-mutant) | B-Raf | Active B-Raf overexpression | in vivo | Active B-Raf expression in collecting ducts accelerates cyst growth in Pkd1-mutant mice |
| Wang et al. [136] | 2022 | Mouse/PKD1-mutant cells | PKA-RIα | BLU2864 inhibition | in vivo/in vitro | PKA-RIα promotes cystogenesis; BLU2864 suppresses effects |
| Shillingford et al. [139] | 2006 | Mouse | Pkd1 | Pkd1 KO | in vivo | mTORC1 upregulation; rapamycin reduces cyst expansion |
| Holditch et al. [143] | 2019 | Mouse/Pkd1−/− cells | mTOR | Torin2 vs. sirolimus | in vivo/in vitro | Both drugs reduce mTOR targets, proliferation, and metabolic activity |
| Kim et al. [152] | 2016 | Mouse | Pkd2/Wnt-Ca+2 | Pkd2 KO | in vivo | Pkd2 loss impairs Wnt-Ca+2 signaling and cell migration; non-canonical Wnt is protective |
| Lin et al. [153] | 2003 | Mouse | Kif3a + Pkd1 | Pax8-Cre conditional KO | in vivo | Loss of cilia promotes cysts; double KO surprisingly reduced cyst severity |
| Li et al. [154] | 2018 | Mouse | Wnt/β-catenin | Canonical Wnt inhibition | in vivo | Wnt/β-catenin inhibitors reduce cystogenesis |
| Yoo et al. [159] | 2024 | IMCD cells/mouse | cGAS/Pkd1 | cGAS KO/MitoQ/PC1 tail | in vivo/in vitro | cGAS upregulated with Pkd1 loss; promotes inflammation via mitochondrial/genotoxic stress. MitoQ or PC1-CTT reverses effects. cGAS inhibition reduces cyst growth and preserves renal function |
| Wu et al. [160] | 2024 | Mouse/renal epithelial cells | STING | Pkd1 KO + STING inhibition (C-176) | in vivo/in vitro | Pharmacologic STING inhibition delays cystogenesis in both early and advanced ADPKD models; restores mitochondrial structure, reduces micronuclei formation, promotes p53-mediated apoptosis, and attenuates renal fibrosis |
| Cordido et al. [162] | 2021 | Mouse/Human | TWEAK/Fn14 | TWEAK stimulation/anti-TWEAK antibody | in vivo/Human | TWEAK/Fn14 upregulated in ADPKD; anti-TWEAK Ab reduces cysts, inflammation, and preserves kidney function |
| Cai et al. [166] | 2018 | Mouse/renal cells | Hippo/YAP | ROCK inhibitor (Y-27632) | in vivo/in vitro | ROCK inhibition reduces YAP/TAZ activity and cysts |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-orjani, Q.; Alshriem, L.A.; Gallagher, G.; Buqaileh, R.; Azizi, N.; AbouAlaiwi, W. Mechanistic Insights into the Pathogenesis of Polycystic Kidney Disease. Cells 2025, 14, 1203. https://doi.org/10.3390/cells14151203
Al-orjani Q, Alshriem LA, Gallagher G, Buqaileh R, Azizi N, AbouAlaiwi W. Mechanistic Insights into the Pathogenesis of Polycystic Kidney Disease. Cells. 2025; 14(15):1203. https://doi.org/10.3390/cells14151203
Chicago/Turabian StyleAl-orjani, Qasim, Lubna A. Alshriem, Gillian Gallagher, Raghad Buqaileh, Neela Azizi, and Wissam AbouAlaiwi. 2025. "Mechanistic Insights into the Pathogenesis of Polycystic Kidney Disease" Cells 14, no. 15: 1203. https://doi.org/10.3390/cells14151203
APA StyleAl-orjani, Q., Alshriem, L. A., Gallagher, G., Buqaileh, R., Azizi, N., & AbouAlaiwi, W. (2025). Mechanistic Insights into the Pathogenesis of Polycystic Kidney Disease. Cells, 14(15), 1203. https://doi.org/10.3390/cells14151203






