Adipose Tissue Paracrine-, Autocrine-, and Matrix-Dependent Signaling during the Development and Progression of Obesity
Abstract
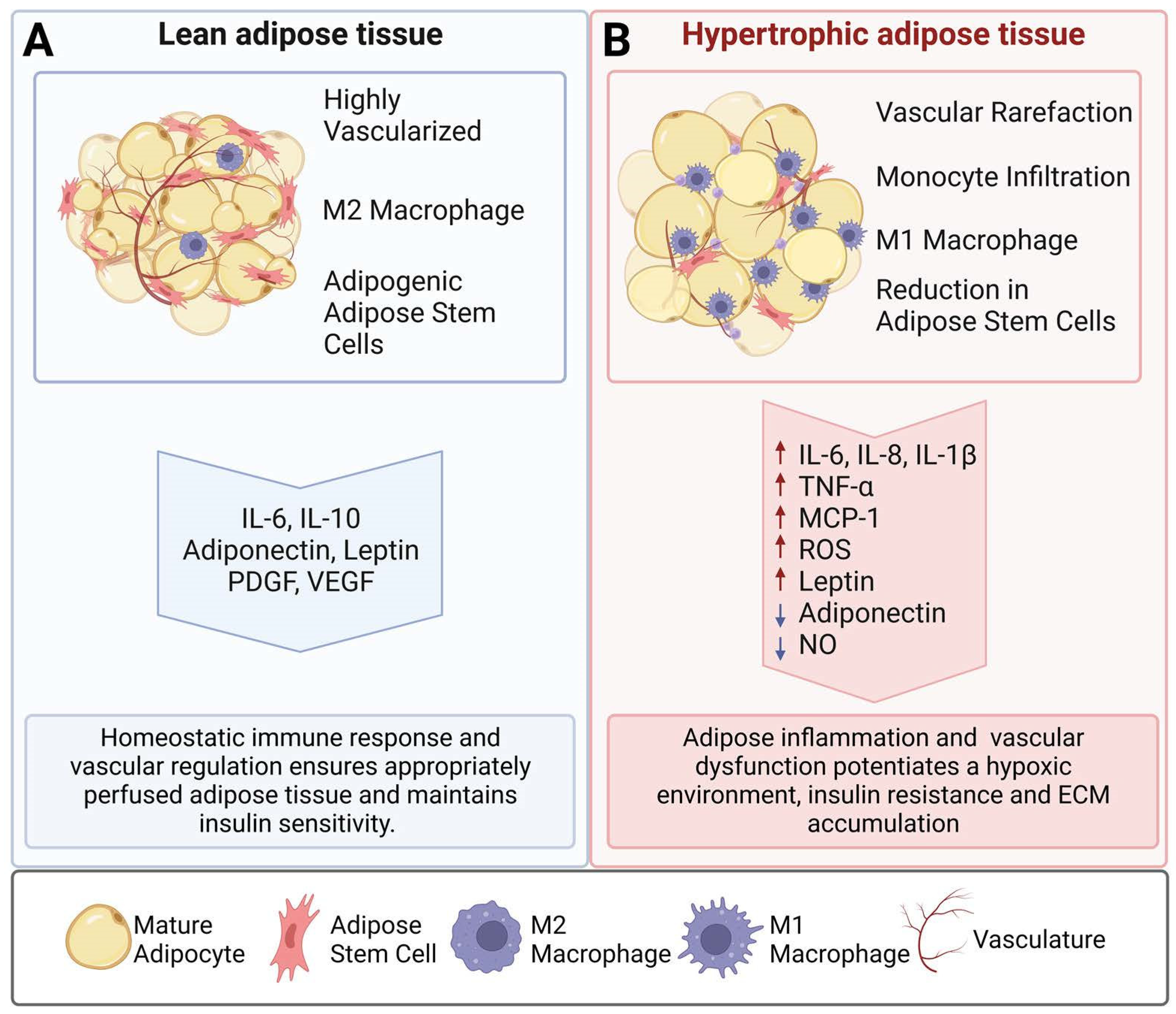
1. Introduction
2. Paracrine Signals Involved in Obesity
2.1. Inflammatory Cytokines
| Signaling Molecule | Physiological Role in Adipose Tissue | Obese State | Regulation | Citation | |
|---|---|---|---|---|---|
| Inflammatory Cytokines | TNF | Involved in immunity Regulates the functions of immune cells, but is found in low levels | Increase in TNF in obesity Stimulates: Adipocyte lipolysis Leptin production IL-6 secretion Plasminogen Activator Inhibitor-1 (PAI-1) biosynthesis ROS production Activates NFκB Inhibits adipocyte differentiation | Triggered through adipocyte cell death | [10,19,25,46,56,57] |
| Interleukins | Classical signaling through the IL-6 canonical pathway is anti-inflammatory and regulates glucose and insulin sensitivity IL-10 is a more prominent interleukin in lean adipose tissue | IL-1β, IL-6, and IL-8 increase adipose inflammation during obesity IL-6 induces lipolytic stimulation and leptin secretion from adipocytes through the trans-signaling pathway while upregulating MCP-1 secretion from endothelial cells that then upregulate their expression of monocyte adhesion molecules | Cellular source of IL-6 production regulates response Endothelial cells release IL-6 in response to TNF, IFN-γ, and IL-1 | [10,46,54,58,59,60] | |
| Interferons | A balance in pro and anti-inflammatory interferons is maintained in order to properly identify and combat invading viruses. This is done through communication between the immune system and the target cells. | Type 1 interferons are upregulated through obesity-derived metabolic endotoxemia. Lipopolysaccharide (LPS) stimulates the production of IFN-β in mouse adipocytes Type II interferon, IFN-γ, is upregulated in obesity due to the influx of IFN-γ-secreting T cells in obese adipose tissue Promotes both a local and systemic inflammatory network between immune cells, adipocytes, and endothelial cells | Adiponectin limits IFN-γ release from CD4+ T cells | [61,62,63,64] | |
| MCP-1 | Chemokine that is secreted by both immune and non-immune cells to attract monocytes/macrophages into the tissue | Increased circulating levels in individuals with obesity Adipocyte progenitors secrete MCP-1 to promote M1 macrophage accumulation in adipose tissue Induces endothelial apoptosis in vitro, thus promoting atherosclerosis | Secreted upon injury or ROS exposure Regulated by TNF and trans-IL-6 signaling Insulin-responsive gene | [25,27,54,65,66,67,68,69,70] | |
| Adipose Hormones | Leptin | Produced by adipose tissue and the stomach but acts on the central nervous system (hypothalamus) in order to regulate satiety. | Increase in leptin production alongside an increase in fat mass Chronic hyperleptinemia can result in leptin resistance and atherogenic consequences Induces SPARC and collagen II and IV expressions and increases profibrotic signaling | Regulated by insulin and steroids | [71,72,73,74] |
| Adiponectin | Encourages angiogenesis through the upregulation of Vascular endothelial growth factor (VEGF)-A, MMP-2 and MMP-9 Regulates glucose and lipid homeostasis and maintains insulin sensitivity | Reduced in obesity | Regulated by peroxisome-proliferator-activated receptor (PPAR)-γ ligands Inhibited by endoplasmic reticulum stress and proinflammatory cytokines | [75,76,77,78,79,80,81,82,83,84] | |
| Vasoconstrictor | Endothelin-1 | Endothelial cells release ET-1 in response to low shear stress and hypoxia to increase blood flow | ET-1 elevation in obesity resulting in vascular vasoconstriction and increased vascular permeability, adipocyte lipolysis, insulin resistance and endothelial inflammation, and perivascular fibrosis Can increase reactive oxidative species (ROS) production | Regulated by blood flow, leptin, and insulin | [85,86,87,88] |
| Vasodilator | Nitric Oxide | Adipose-derived nitric oxide inhibits lipolytic activity and promotes endothelial relaxation and vasodilation Adiponectin stimulates NO production | NO is reduced in obesity through TNF-mediated destabilization and free fatty acid (FFA) inhibition of NOS3 phosphorylation Increased ROS will quench NO | ROS FFA TNF | [89,90] |
| Serpin (serine protease inhibitor) | PAI-1 | Induces fibrinogenesis by suppressing intravascular fibrinolysis | Marked increase in PAI-1 in individuals with obesity and diabetes (positive correlation with insulin resistance) When released into the blood stream, it negatively impacts vascular metabolism Fibrinolytic activities decrease in individuals with obesity individuals. Sustained impairment accelerates atherosclerosis | TNF TGF-β1 and Angiotensin II promote PAI-1 production in adipocytes and stromal cells Insulin responsive | [25,79,91,92,93] |
| Growth Factors | VEGF | Secreted by endothelial cells and adipocytes Acts as a chemotactic factor VEGF-A increases vascularization VEGF-B controls endothelial uptake of fatty acids | Suggestions of a positive VEGF correlation with BMI in humans Dysfunctional VEGF signaling results in impaired vascularization, increased vascular permeability, and endothelial dysfunction in individuals with obesity | SPARC regulates VEGF in individuals with diabetes Leptin modulates VEGF-A expression | [10,79,83,94,95,96,97,98] |
| PDGF | Involved in angiogenesis and developmental adipogenesis ASCs become pro angiogenic in response to Platelet-derived growth factor (PDGF) | M1 macrophages overexpress PDGF-B during obesity PDGF-B induces proliferation and migration of aortic smooth muscle cells in vitro, which results in the thickening of the artery Myofibroblast mitogen that contributes to adipose fibrosis | Stromal-cell-derived factor 1 Positively regulated by IL-1β and TGF-β1 | [99,100,101,102,103,104,105] | |
| TGF-β | Regulates the rate of adipogenesis | TGF-β1 and TGF-β3 increase and can cause mesenchymal transitions and dedifferentiation during obesity These isoforms increase basement membrane production, crosslinking, and inflammatory cytokine production in both adipocytes and endothelial cells | TNF can promote TGF-β secretion in differentiated adipocytes | [24,31,106,107] | |
2.2. Adipokine Secretion
2.3. Vasoregulators in Obesity
3. Extracellular Matrix Remodeling during Obesity
4. Obesity-Related Comorbidities: The Role of Adipose Tissue
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BMI | Body Mass Index |
| PVAT | Perivascular adipose tissue |
| IL | Interleukin |
| PDGF | Platelet-derived growth factor |
| VEGF | Vascular Endothelial Growth Factor |
| TNF | Tumor Necrosis Factor-α |
| MCP-1 | Monocyte Chemoattractant Protein-1 |
| ROS | Reactive oxidative species |
| NO | Nitric Oxide |
| ET-1 | Endothelin-1 |
| MMP | Matrix Metalloproteinase |
| GM-CSF | Granulocyte-Macrophage Colony Stimulating Factor |
| IL-1β | Interleukin-1β |
| MIP-1α | Macrophage Inflammatory Protein-1α |
| HUVECs | Human umbilical vein endothelial cells |
| NFκB | Nuclear factor kappa B |
| IFN-y | Interferon-Gamma |
| ECM | Extracellular Matrix |
| PREP1 | Pre-B-cell leukemia homeobox (Pbx)-regulating protein-1 |
| eNOS | Endothelial nitric oxide synthase |
| HSL | Hormone sensitive lipase |
| iNOS | Inducible NO synthase |
| FFA | Free Fatty Acids |
| f-actin | Filamentous actin |
| SPARC | Secreted Protein, Acidic, Rich in Cysteine |
| LOX | Lysyl Oxidase |
| CTGF | Connective Tissue Growth Factor |
| PAI-1 | Plasminogen Activator Inhibitor-1 |
| AGE | Advanced glycation end products |
| VCAM | Vascular cell adhesion molecule |
| ICAM | Intercellular adhesion molecule-1 |
| ERK ½ | Extracellular signal-regulated kinase ½ |
| ADAM | A disintegrin and metalloprotease |
| STAT3 | Signal transducer and activator of transcription 3 |
| PI3K-Akt | Phosphatidylinositol 3-kinase- protein kinase B |
| LPS | Lipopolysaccharides |
| PPARγ | Peroxisome proliferator-activated receptors |
| TLR4 | Toll-like receptor 4 |
| SVF | Stromal vascular fraction |
| FGF | Fibroblast growth factor |
| HSPG2 | Heparan sulfate proteoglycan 2 |
| TGF-β | Transforming growth factor- β |
References
- Hales, C.M.; Carroll, M.D.; Fryar, C.D.; Ogden, C.L. Prevalence of Obesity and Severe Obesity Among Adults: United States, 2017–2018; CDC National Center for Health Statistics: Hyattsville, MD, USA, 2020; pp. 1–8.
- Hales, C.M.; Carroll, M.D.; Fryar, C.D.; Ogden, C.L. Prevalence of Obesity among Adults and Youth: United States, 2015–2016; National Center for Health Statistics: Hyattsville, MD, USA, 2017.
- Cao, Y. Adipose tissue angiogenesis as a therapeutic target for obesity and metabolic diseases. Nat. Rev. Drug Discov. 2010, 9, 107–115. [Google Scholar] [CrossRef]
- Christiaens, V.; Lijnen, H.R. Angiogenesis and development of adipose tissue. Mol. Cell. Endocrinol. 2010, 318, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, S.; Manabe, I.; Nagasaki, M.; Hosoya, Y.; Yamashita, H.; Fujita, H.; Ohsugi, M.; Tobe, K.; Kadowaki, T.; Nagai, R.; et al. Adipogenesis in Obesity Requires Close Interplay Between Differentiating Adipocytes, Stromal Cells, and Blood Vessels. Diabetes 2007, 56, 1517–1526. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.-K.; Doh, K.-O.; Son, J.E.; Park, J.G.; Bae, Y.; Choi, S.; Nelson, S.M.L.; Cowling, R.; Nagy, K.; Michael, I.P.; et al. Adipose vascular endothelial growth factor regulates metabolic homeostasis through angiogenesis. Cell Metab. 2013, 17, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Rupnick, M.A.; Panigrahy, D.; Zhang, C.-Y.; Dallabrida, S.M.; Lowell, B.B.; Langer, R.; Folkman, M.J. Adipose tissue mass can be regulated through the vasculature. Proc. Natl. Acad. Sci. USA 2002, 99, 10730–10735. [Google Scholar] [CrossRef]
- Hammarstedt, A.; Gogg, S.; Hedjazifar, S.; Nerstedt, A.; Smith, U. Impaired Adipogenesis and Dysfunctional Adipose Tissue in Human Hypertrophic Obesity. Physiol. Rev. 2018, 98, 1911–1941. [Google Scholar] [CrossRef]
- Belligoli, A.; Compagnin, C.; Sanna, M.; Favaretto, F.; Fabris, R.; Busetto, L.; Foletto, M.; Prà, C.D.; Serra, R.; Prevedello, L.; et al. Characterization of subcutaneous and omental adipose tissue in patients with obesity and with different degrees of glucose impairment. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Goossens, G.H.; Bizzarri, A.; Venteclef, N.; Essers, Y.; Cleutjens, J.P.; Konings, E.; Jocken, J.W.E.; Čajlaković, M.; Ribitsch, V.; Clément, K.; et al. Increased adipose tissue oxygen tension in obese compared with lean men is accompanied by insulin resistance, impaired adipose tissue capillarization, and inflammation. Circulation 2011, 124, 67–76. [Google Scholar] [CrossRef]
- Cifarelli, V.; Beeman, S.C.; Smith, G.I.; Yoshino, J.; Morozov, D.; Beals, J.W.; Kayser, B.D.; Watrous, J.D.; Jain, M.; Patterson, B.W.; et al. Decreased adipose tissue oxygenation associates with insulin resistance in individuals with obesity. J. Clin. Investig. 2020, 130, 6688–6699. [Google Scholar] [CrossRef]
- Pasarica, M.; Sereda, O.R.; Redman, L.M.; Albarado, D.C.; Hymel, D.T.; Roan, L.E.; Rood, J.C.; Burk, D.H.; Smith, S.R. Reduced adipose tissue oxygenation in human obesity: Evidence for rarefaction, macrophage chemotaxis, and inflammation without an angiogenic response. Diabetes 2009, 58, 718–725. [Google Scholar] [CrossRef]
- Hansson, B.; Morén, B.; Fryklund, C.; Vliex, L.; Wasserstrom, S.; Albinsson, S.; Berger, K.; Stenkula, K.G. Adipose cell size changes are associated with a drastic actin remodeling. Sci. Rep. 2019, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Eliasson, B.; Smith, U.; Cushman, S.W.; Sherman, A.S. The Size of Large Adipose Cells Is a Predictor of Insulin Resistance in First-Degree Relatives of Type 2 Diabetic Patients. Obesity 2012, 20, 932–938. [Google Scholar] [CrossRef] [PubMed]
- Hammarstedt, A.; E Graham, T.; Kahn, B.B. Adipose tissue dysregulation and reduced insulin sensitivity in non-obese individuals with enlarged abdominal adipose cells. Diabetol. Metab. Syndr. 2012, 4, 42–49. [Google Scholar] [CrossRef]
- Fuster, J.J.; Ouchi, N.; Gokce, N.; Walsh, K. Obesity-Induced Changes in Adipose Tissue Microenvironment and Their Impact on Cardiovascular Disease. Circ. Res. 2016, 118, 1786–1807. [Google Scholar] [CrossRef]
- E Rausch, M.; Weisberg, S.; Vardhana, P.; Tortoriello, D.V. Obesity in C57BL/6J mice is characterized by adipose tissue hypoxia and cytotoxic T-cell infiltration. Int. J. Obes. 2007, 32, 451–463. [Google Scholar] [CrossRef] [PubMed]
- Lumeng, C.N.; Bodzin, J.L.; Saltiel, A.R. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Investig. 2007, 117, 175–184. [Google Scholar] [CrossRef]
- Xu, H.; Uysal, K.T.; Becherer, J.D.; Arner, P.; Hotamisligil, G.k.S. Altered Tumor Necrosis Factor-α (TNF-α) Processing in Adipocytes and Increased Expression of Transmembrane TNF-α in Obesity. Diabetes 2002, 51, 1876–1883. [Google Scholar] [CrossRef]
- Black, R.A. Tumor necrosis factor-alpha converting enzyme. Int. J. Biochem. Cell Biol. 2002, 34, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Dandona, P.; Weinstock, R.; Thusu, K.; Abdel-Rahman, E.; Aljada, A.; Wadden, T. Tumor Necrosis Factor-α in Sera of Obese Patients: Fall with Weight Loss. J. Clin. Endocrinol. Metab. 1998, 83, 2907–2910. [Google Scholar] [CrossRef]
- Feingold, K.R.; Doerrler, W.; A Dinarello, C.; Fiers, W.; Grunfeld, C. Stimulation of lipolysis in cultured fat cells by tumor necrosis factor, interleukin-1, and the interferons is blocked by inhibition of prostaglandin synthesis. Endocrinology 1992, 130, 10–16. [Google Scholar] [CrossRef]
- Aubin, K.; Safoine, M.; Proulx, M.; Audet-Casgrain, M.-A.; Côté, J.-F.; Têtu, F.-A.; Roy, A.; Fradette, J. Characterization of In Vitro Engineered Human Adipose Tissues: Relevant Adipokine Secretion and Impact of TNF-α. PLoS ONE 2015, 10, e0137612. [Google Scholar] [CrossRef]
- Weiner, F.R.; Shah, A.; Smith, P.J.; Rubin, C.S.; Zern, M.A. Regulation of collagen gene expression in 3T3-L1 cells. Effects of adipocyte differentiation and tumor necrosis factor alpha. Biochemistry 1989, 28, 4094–4099. [Google Scholar] [CrossRef]
- Gustafson, B.; Smith, U. Cytokines Promote Wnt Signaling and Inflammation and Impair the Normal Differentiation and Lipid Accumulation in 3T3-L1 Preadipocytes. J. Biol. Chem. 2006, 281, 9507–9516. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, B.; Gogg, S.; Hedjazifar, S.; Jenndahl, L.; Hammarstedt, A.; Smith, U. Inflammation and impaired adipogenesis in hypertrophic obesity in man. Am. J. Physiol.-Endocrinol. Metab. 2009, 297, E999–E1003. [Google Scholar] [CrossRef] [PubMed]
- Gerhardt, C.; Romero, I.; Cancello, R.; Camoin, L.; Strosberg, A. Chemokines control fat accumulation and leptin secretion by cultured human adipocytes. Mol. Cell. Endocrinol. 2001, 175, 81–92. [Google Scholar] [CrossRef]
- Kralisch, S.; Sommer, G.; Stangl, V.; Köhler, U.; Kratzsch, J.; Stepan, H.; Faber, R.; Schubert, A.; Lössner, U.; Vietzke, A.; et al. Secretory products from human adipocytes impair endothelial function via nuclear factor kappaB. Atherosclerosis 2008, 196, 523–531. [Google Scholar] [CrossRef]
- Sommer, G.; Kralisch, S.; Stangl, V.; Vietzke, A.; Köhler, U.; Stepan, H.; Faber, R.; Schubert, A.; Lössner, U.; Bluher, M.; et al. Secretory products from human adipocytes stimulate proinflammatory cytokine secretion from human endothelial cells. J. Cell. Biochem. 2009, 106, 729–737. [Google Scholar] [CrossRef]
- Cousin, B.; Munoz, O.; Andre, M.; Fontanilles, A.M.; Dani, C.; Cousin, J.L.; Laharrague, P.; Casteilla, L.; Énicaud, L.P. A role for preadipocytes as macrophage-like cells. FASEB J. 1999, 13, 305–312. [Google Scholar] [CrossRef]
- Haynes, B.A.; Yang, L.F.; Huyck, R.W.; Lehrer, E.J.; Turner, J.M.; Barabutis, N.; Correll, V.L.; Mathiesen, A.; McPheat, W.; Semmes, O.J.; et al. Endothelial-to-Mesenchymal Transition in Human Adipose Tissue Vasculature Alters the Particulate Secretome and Induces Endothelial Dysfunction. Arterioscler. Thromb. Vasc. Biol. 2019, 39, 2168–2191. [Google Scholar] [CrossRef] [PubMed]
- Nieto-Vazquez, I.; Fernández-Veledo, S.; Krämer, D.K.; Vila, R.; García, L.; Lorenzo, M. Insulin resistance associated to obesity: The link TNF-alpha. Arch. Physiol. Biochem. 2008, 114, 183–194. [Google Scholar] [CrossRef]
- da Costa, R.M.; Neves, K.B.; Mestriner, F.L.; Louzada-Junior, P.; Bruder-Nascimento, T.; Tostes, R.C. TNF-α induces vascular insulin resistance via positive modulation of PTEN and decreased Akt/eNOS/NO signaling in high fat diet-fed mice. Cardiovasc. Diabetol. 2016, 15, 119. [Google Scholar] [CrossRef] [PubMed]
- Mohallem, R.; Aryal, U.K. Regulators of TNFα mediated insulin resistance elucidated by quantitative proteomics. Sci. Rep. 2020, 10, 1–15. [Google Scholar] [CrossRef]
- Abbott, R.D.; Wang, R.Y.; Reagan, M.R.; Chen, Y.; Borowsky, F.E.; Zieba, A.; Marra, K.G.; Rubin, J.P.; Ghobrial, I.M.; Kaplan, D.L. The Use of Silk as a Scaffold for Mature, Sustainable Unilocular Adipose 3D Tissue Engineered Systems. Adv. Heal. Mater. 2016, 5, 1667–1677. [Google Scholar] [CrossRef]
- Abbott, R.D.; Borowsky, F.E.; Alonzo, C.A.; Zieba, A.; Georgakoudi, I.; Kaplan, D.L. Variability in responses observed in human white adipose tissue models. J. Tissue Eng. Regen. Med. 2017, 12, 840–847. [Google Scholar] [CrossRef]
- Miyazaki, Y.; Pipek, R.; Mandarino, L.J.; DeFronzo, R.A. Tumor necrosis factor α and insulin resistance in obese type 2 diabetic patients. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 88–94. [Google Scholar] [CrossRef]
- Wascher, T.C.; Lindeman, J.H.N.; Sourij, H.; Kooistra, T.; Pacini, G.; Roden, M. Chronic TNF-α Neutralization Does Not Improve Insulin Resistance or Endothelial Function in “Healthy” Men with Metabolic Syndrome. Mol. Med. 2010, 17, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, L.E.; Berry, J.; Kim, S.; Canavan, B.; Grinspoon, S.K. Effects of Etanercept in Patients With the Metabolic Syndrome. Arch. Intern. Med. 2006, 166, 902–908. [Google Scholar] [CrossRef]
- Carey, A.L.; Steinberg, G.R.; Macaulay, S.L.; Thomas, W.G.; Holmes, A.G.; Ramm, G.; Prelovsek, O.; Hohnen-Behrens, C.; Watt, M.J.; James, D.E.; et al. Interleukin-6 Increases Insulin-Stimulated Glucose Disposal in Humans and Glucose Uptake and Fatty Acid Oxidation In Vitro via AMP-Activated Protein Kinase. Diabetes 2006, 55, 2688–2697. [Google Scholar] [CrossRef] [PubMed]
- van Hall, G.; Steensberg, A.; Sacchetti, M.; Fischer, C.; Keller, C.; Schjerling, P.; Hiscock, N.; Møller, K.; Saltin, B.; Febbraio, M.A.; et al. Interleukin-6 Stimulates Lipolysis and Fat Oxidation in Humans. J. Clin. Endocrinol. Metab. 2003, 88, 3005–3010. [Google Scholar] [CrossRef]
- Mahlakõiv, T.; Flamar, A.-L.; Johnston, L.K.; Moriyama, S.; Putzel, G.G.; Bryce, P.J.; Artis, D. Stromal cells maintain immune cell homeostasis in adipose tissue via production of interleukin-33. Sci. Immunol. 2019, 4. [Google Scholar] [CrossRef]
- Scheller, J.; Chalaris, A.; Schmidt-Arras, D.; Rose-John, S. The pro- and anti-inflammatory properties of the cytokine interleukin-6. Biochim. Biophys. Acta 2011, 1813, 878–888. [Google Scholar] [CrossRef] [PubMed]
- Antuna-Puente, B.; Fellahi, S.; McAvoy, C.; Fève, B.; Bastard, J.-P. Interleukins in adipose tissue: Keeping the balance. Mol. Cell. Endocrinol. 2022, 542, 111531. [Google Scholar] [CrossRef]
- Mohamed-Ali, V.; Goodrick, S.; Rawesh, A.; Katz, D.R.; Miles, J.M.; Yudkin, J.S.; Klein, S.; Coppack, S.W. Subcutaneous Adipose Tissue Releases Interleukin-6, But Not Tumor Necrosis Factor-α, in Vivo1. J. Clin. Endocrinol. Metab. 1997, 82, 4196–4200. [Google Scholar] [CrossRef] [PubMed]
- Kern, P.A.; Ranganathan, S.; Li, C.; Wood, L.; Ranganathan, G. Adipose tissue tumor necrosis factor and interleukin-6 expression in human obesity and insulin resistance. Am. J. Physiol. Metab. 2001, 280, E745–E751. [Google Scholar] [CrossRef]
- Hajer, G.R.; van Haeften, T.W.; Visseren, F.L.J. Adipose tissue dysfunction in obesity, diabetes, and vascular diseases. Eur. Heart J. 2008, 29, 2959–2971. [Google Scholar] [CrossRef] [PubMed]
- Han, M.S.; White, A.; Perry, R.J.; Camporez, J.-P.; Hidalgo, J.; Shulman, G.I.; Davis, R.J. Regulation of adipose tissue inflammation by interleukin 6. Proc. Natl. Acad. Sci. USA 2020, 117, 2751–2760. [Google Scholar] [CrossRef]
- El-Mikkawy, D.M.E.; El-Sadek, M.A.; El-Badawy, M.A.; Samaha, D. Circulating level of interleukin-6 in relation to body mass indices and lipid profile in Egyptian adults with overweight and obesity. Egypt. Rheumatol. Rehabil. 2020, 47, 1–7. [Google Scholar] [CrossRef]
- Trujillo, M.E.; Sullivan, S.; Harten, I.; Schneider, S.H.; Greenberg, A.S.; Fried, S.K. Interleukin-6 Regulates Human Adipose Tissue Lipid Metabolism and Leptin Production in Vitro. J. Clin. Endocrinol. Metab. 2004, 89, 5577–5582. [Google Scholar] [CrossRef]
- Sindhu, S.; Thomas, R.; Shihab, P.; Sriraman, D.; Behbehani, K.; Ahmad, R. Obesity Is a Positive Modulator of IL-6R and IL-6 Expression in the Subcutaneous Adipose Tissue: Significance for Metabolic Inflammation. PLoS ONE 2015, 10, e0133494. [Google Scholar] [CrossRef]
- Meijer, K.; De Vries, M.; Al-Lahham, S.; Bruinenberg, M.; Weening, D.; Dijkstra, M.; Kloosterhuis, N.; Van Der Leij, R.J.; Van Der Want, H.; Kroesen, B.-J.; et al. Human Primary Adipocytes Exhibit Immune Cell Function: Adipocytes Prime Inflammation Independent of Macrophages. PLoS ONE 2011, 6, e17154. [Google Scholar] [CrossRef] [PubMed]
- Reeh, H.; Rudolph, N.; Billing, U.; Christen, H.; Streif, S.; Bullinger, E.; Schliemann-Bullinger, M.; Findeisen, R.; Schaper, F.; Huber, H.J.; et al. Response to IL-6 trans- and IL-6 classic signalling is determined by the ratio of the IL-6 receptor α to gp130 expression: Fusing experimental insights and dynamic modelling. Cell Commun. Signal. 2019, 17, 1–21. [Google Scholar] [CrossRef]
- Montgomery, A.; Tam, F.; Gursche, C.; Cheneval, C.; Besler, K.; Enns, W.; Manku, S.; Rey, K.; Hanson, P.J.; Rose-John, S.; et al. Overlapping and distinct biological effects of IL-6 classic and trans-signaling in vascular endothelial cells. Am. J. Physiol. Physiol. 2021, 320, C554–C565. [Google Scholar] [CrossRef] [PubMed]
- Podor, T.J.; Jirik, F.R.; Loskutoff, D.J.; Carson, D.A.; Lotz, M. Human endothelial cells produce IL-6. Lack of responses to exogenous IL-6. Ann. N. Y. Acad. Sci. 1989, 557, 374–385, discussion 386–377. [Google Scholar] [CrossRef]
- Cawthorn, W.P.; Sethi, J.K. TNF-α and adipocyte biology. FEBS Lett. 2007, 582, 117–131. [Google Scholar] [CrossRef] [PubMed]
- Sethi, J.K.; Hotamisligil, G.S. Metabolic Messengers: Tumour necrosis factor. Nat. Metab. 2021, 3, 1302–1312. [Google Scholar] [CrossRef] [PubMed]
- Sopasakis, V.R.; Sandqvist, M.; Gustafson, B.; Hammarstedt, A.; Schmelz, M.; Yang, X.; Jansson, P.-A.; Smith, U. High Local Concentrations and Effects on Differentiation Implicate Interleukin-6 as a Paracrine Regulator. Obes. Res. 2004, 12, 454–460. [Google Scholar] [CrossRef]
- Sironi, M.; Breviario, F.; Proserpio, P.; Biondi, A.; Vecchi, A.; Van Damme, J.; Dejana, E.; Mantovani, A. IL-1 stimulates IL-6 production in endothelial cells. J. Immunol. 1989, 142, 549–553. [Google Scholar] [CrossRef]
- Liddle, D.M.; Hutchinson, A.L.; Wellings, H.R.; Power, K.A.; Robinson, L.E.; Monk, J.M. Integrated Immunomodulatory Mechanisms through which Long-Chain n-3 Polyunsaturated Fatty Acids Attenuate Obese Adipose Tissue Dysfunction. Nutrients 2017, 9, 1289. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.C.; Damen, M.S.M.A.; Moreno-Fernandez, M.E.; Stankiewicz, T.E.; Cappelletti, M.; Alarcon, P.C.; Oates, J.R.; Doll, J.R.; Mukherjee, R.; Chen, X.; et al. Type I interferon sensing unlocks dormant adipocyte inflammatory potential. Nat. Commun. 2020, 11, 1–15. [Google Scholar] [CrossRef]
- Rocha, V.Z.; Folco, E.J.; Sukhova, G.; Shimizu, K.; Gotsman, I.; Vernon, A.H.; Libby, P. Interferon-γ, a Th1 Cytokine, Regulates Fat Inflammation. Circ. Res. 2008, 103, 467–476. [Google Scholar] [CrossRef]
- Surendar, J.; Frohberger, S.J.; Karunakaran, I.; Schmitt, V.; Stamminger, W.; Neumann, A.-L.; Wilhelm, C.; Hoerauf, A.; Hübner, M.P. Adiponectin Limits IFN-γ and IL-17 Producing CD4 T Cells in Obesity by Restraining Cell Intrinsic Glycolysis. Front. Immunol. 2019, 10, 2555. [Google Scholar] [CrossRef]
- Zhang, H.; Potter, B.J.; Cao, J.-M.; Zhang, C. Interferon-gamma induced adipose tissue inflammation is linked to endothelial dysfunction in type 2 diabetic mice. Basic Res. Cardiol. 2011, 106, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Anshita, D.; Ravichandiran, V. MCP-1: Function, regulation, and involvement in disease. Int. Immunopharmacol. 2021, 101, 107598. [Google Scholar] [CrossRef]
- Sartipy, P.; Loskutoff, D.J. Monocyte chemoattractant protein 1 in obesity and insulin resistance. Proc. Natl. Acad. Sci. USA 2003, 100, 7265–7270. [Google Scholar] [CrossRef]
- Dahlman, I.; Kaaman, M.; Olsson, T.; Tan, G.D.; Bickerton, A.S.T.; Wåhlén, K.; Andersson, J.; Nordström, E.A.; Blomqvist, L.; Sjögren, A.; et al. A Unique Role of Monocyte Chemoattractant Protein 1 among Chemokines in Adipose Tissue of Obese Subjects. J. Clin. Endocrinol. Metab. 2005, 90, 5834–5840. [Google Scholar] [CrossRef]
- Kanda, H.; Tateya, S.; Tamori, Y.; Kotani, K.; Hiasa, K.-I.; Kitazawa, R.; Kitazawa, S.; Miyachi, H.; Maeda, S.; Egashira, K.; et al. MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J. Clin. Investig. 2006, 116, 1494–1505. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, X.; Shang, H.; Xu, Y.; Qian, M. Monocyte chemoattractant protein-1 induces endothelial cell apoptosis in vitro through a p53-dependent mitochondrial pathway. Acta Biochim. Biophys. Sin. 2011, 43, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, J.L.; Marshall, M.A.; McSkimming, C.C.; Harmon, D.B.; Garmey, J.C.; Oldham, S.N.; Hallowell, P.; McNamara, C.A. Adipocyte progenitor cells initiate monocyte chemoattractant protein-1-mediated macrophage accumulation in visceral adipose tissue. Mol. Metab. 2015, 4, 779–794. [Google Scholar] [CrossRef] [PubMed]
- Wabitsch, M.; Bo Jensen, P.; Blum, W.F.; Christoffersen, C.T.; Englaro, P.; Heinze, E.; Rascher, W.; Teller, W.; Tornqvist, H.; Hauner, H. Insulin and cortisol promote leptin production in cultured human fat cells. Diabetes 1996, 45, 1435–1438. [Google Scholar] [CrossRef]
- Chrysafi, P.; Perakakis, N.; Farr, O.M.; Stefanakis, K.; Peradze, N.; Sala-Vila, A.; Mantzoros, C.S. Leptin alters energy intake and fat mass but not energy expenditure in lean subjects. Nat. Commun. 2020, 11, 1–15. [Google Scholar] [CrossRef]
- Bouloumié, A.; Marumo, T.; Lafontan, M.; Busse, R. Leptin induces oxidative stress in human endothelial cells. FASEB J. 1999, 13, 1231–1238. [Google Scholar] [CrossRef]
- Picó, C.; Oliver, P.; Sánchez, J.; Palou, A. Gastric leptin: A putative role in the short-term regulation of food intake. Br. J. Nutr. 2003, 90, 735–741. [Google Scholar] [CrossRef]
- Ouchi, N.; Kihara, S.; Arita, Y.; Maeda, K.; Kuriyama, H.; Okamoto, Y.; Hotta, K.; Nishida, M.; Takahashi, M.; Nakamura, T.; et al. Novel modulator for endothelial adhesion molecules: Adipocyte-derived plasma protein adiponectin. Circulation 1999, 100, 2473–2476. [Google Scholar] [CrossRef]
- Ouchi, N.; Kihara, S.; Arita, Y.; Okamoto, Y.; Maeda, K.; Kuriyama, H.; Hotta, K.; Nishida, M.; Takahashi, M.; Muraguchi, M.; et al. Adiponectin, an adipocyte-derived plasma protein, inhibits endothelial NF-kappaB signaling through a cAMP-dependent pathway. Circulation 2000, 102, 1296–1301. [Google Scholar] [CrossRef] [PubMed]
- Chandran, M.; Phillips, S.A.; Ciaraldi, T.; Henry, R.R. Adiponectin: More Than Just Another Fat Cell Hormone? Diabetes Care 2003, 26, 2442–2450. [Google Scholar] [CrossRef] [PubMed]
- Kern, P.A.; Di Gregorio, G.B.; Lu, T.; Rassouli, N.; Ranganathan, G. Adiponectin Expression From Human Adipose Tissue. Diabetes 2003, 52, 1779–1785. [Google Scholar] [CrossRef]
- Hauner, H. Secretory factors from human adipose tissue and their functional role. Proc. Nutr. Soc. 2005, 64, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wang, F.; Wang, B.-J.; Chu, G.; Cao, Q.; Sun, B.-G.; Dai, Q.-Y. Inhibition of leptin-induced vascular extracellular matrix remodelling by adiponectin. J. Mol. Endocrinol. 2014, 53, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Adya, R.; Tan, B.K.; Randeva, H.S. Differential Effects of Leptin and Adiponectin in Endothelial Angiogenesis. J. Diabetes Res. 2015, 2015, 1–12. [Google Scholar] [CrossRef]
- Sena, C.M.; Pereira, A.; Fernandes, R.; Letra, L.; Seiça, R.M. Adiponectin improves endothelial function in mesenteric arteries of rats fed a high-fat diet: Role of perivascular adipose tissue. Br. J. Pharmacol. 2017, 174, 3514–3526. [Google Scholar] [CrossRef]
- Nigro, E.; Mallardo, M.; Polito, R.; Scialò, F.; Bianco, A.; Daniele, A. Adiponectin and Leptin Exert Antagonizing Effects on HUVEC Tube Formation and Migration Modulating the Expression of CXCL1, VEGF, MMP-2 and MMP-9. Int. J. Mol. Sci. 2021, 22, 7516. [Google Scholar] [CrossRef]
- Hoffstedt, J.; Arvidsson, E.; Sjölin, E.; Wåhlén, K.; Arner, P. Adipose Tissue Adiponectin Production and Adiponectin Serum Concentration in Human Obesity and Insulin Resistance. J. Clin. Endocrinol. Metab. 2004, 89, 1391–1396. [Google Scholar] [CrossRef]
- Ferri, C.; Bellini, C.; Desideri, G.; Baldoncini, R.; Properzi, G.; Santucci, A.; De Mattia, G. Circulating endothelin-1 levels in obese patients with the metabolic syndrome. Exp. Clin. Endocrinol. Diabetes 1997, 105, 38–40. [Google Scholar] [CrossRef] [PubMed]
- Quehenberger, P.; Exner, M.; Sunder-Plassmann, R.; Ruzicka, K.; Bieglmayer, C.; Endler, G.; Muellner, C.; Speiser, W.; Wagner, O. Leptin Induces Endothelin-1 in Endothelial Cells In Vitro. Circ. Res. 2002, 90, 711–718. [Google Scholar] [CrossRef]
- van Harmelen, V.; Eriksson, A.; Åström, G.; Wåhlén, K.; Näslund, E.; Karpe, F.; Frayn, K.; Olsson, T.; Andersson, J.; Rydén, M.; et al. Vascular Peptide Endothelin-1 Links Fat Accumulation With Alterations of Visceral Adipocyte Lipolysis. Diabetes 2008, 57, 378–386. [Google Scholar] [CrossRef] [PubMed]
- Yoshizumi, M.; Kurihara, H.; Sugiyama, T.; Takaku, F.; Yanagisawa, M.; Masaki, T.; Yazaki, Y. Hemodynamic shear stress stimulates endothelin production by cultured endothelial cells. Biochem. Biophys. Res. Commun. 1989, 161, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Gruber, H.-J.; Mayer, C.; Mangge, H.; Fauler, G.; Grandits, N.; Wilders-Truschnig, M. Obesity reduces the bioavailability of nitric oxide in juveniles. Int. J. Obes. 2008, 32, 826–831. [Google Scholar] [CrossRef] [PubMed]
- Sansbury, B.E.; Hill, B.G. Regulation of obesity and insulin resistance by nitric oxide. Free. Radic. Biol. Med. 2014, 73, 383–399. [Google Scholar] [CrossRef]
- Alessi, M.C.; Bastelica, D.; Morange, P.; Berthet, B.; Leduc, I.; Verdier, M.; Geel, O.; Juhan-Vague, I. Plasminogen activator inhibitor 1, transforming growth factor-beta1, and BMI are closely associated in human adipose tissue during morbid obesity. Diabetes 2000, 49, 1374–1380. [Google Scholar] [CrossRef] [PubMed]
- Kaji, H. Adipose Tissue-Derived Plasminogen Activator Inhibitor-1 Function and Regulation. Compr. Physiol. 2016, 6, 1873–1896. [Google Scholar] [CrossRef]
- Wang, L.; Chen, L.; Liu, Z.; Liu, Y.; Luo, M.; Chen, N.; Deng, X.; Luo, Y.; He, J.; Zhang, L.; et al. PAI-1 Exacerbates White Adipose Tissue Dysfunction and Metabolic Dysregulation in High Fat Diet-Induced Obesity. Front. Pharmacol. 2018, 9, 1087. [Google Scholar] [CrossRef]
- Hagberg, C.E.; Falkevall, A.; Wang, X.; Larsson, E.; Huusko, J.; Nilsson, I.; van Meeteren, L.A.; Samen, E.; Lu, L.; Vanwildemeersch, M.; et al. Vascular endothelial growth factor B controls endothelial fatty acid uptake. Nature 2010, 464, 917–921. [Google Scholar] [CrossRef] [PubMed]
- Silha, J.V.; Kršek, M.; Sucharda, P.; Murphy, L.J. Angiogenic factors are elevated in overweight and obese individuals. Int. J. Obes. 2005, 29, 1308–1314. [Google Scholar] [CrossRef] [PubMed]
- Loebig, M.; Klement, J.; Schmoller, A.; Betz, S.; Heuck, N.; Schweiger, U.; Peters, A.; Schultes, B.; Oltmanns, K.M. Evidence for a Relationship between VEGF and BMI Independent of Insulin Sensitivity by Glucose Clamp Procedure in a Homogenous Group Healthy Young Men. PLoS ONE 2010, 5, e12610. [Google Scholar] [CrossRef]
- Lamers, D.; Schlich, R.; Greulich, S.; Sasson, S.; Sell, H.; Eckel, J. Oleic acid and adipokines synergize in inducing proliferation and inflammatory signalling in human vascular smooth muscle cells. J. Cell. Mol. Med. 2010, 15, 1177–1188. [Google Scholar] [CrossRef] [PubMed]
- Schlich, R.; Willems, M.; Greulich, S.; Ruppe, F.; Knoefel, W.T.; Ouwens, D.M.; Maxhera, B.; Lichtenberg, A.; Eckel, J.; Sell, H. VEGF in the Crosstalk between Human Adipocytes and Smooth Muscle Cells: Depot-Specific Release from Visceral and Perivascular Adipose Tissue. Mediat. Inflamm. 2013, 2013, 1–10. [Google Scholar] [CrossRef]
- Onogi, Y.; Wada, T.; Kamiya, C.; Inata, K.; Matsuzawa, T.; Inaba, Y.; Kimura, K.; Inoue, H.; Yamamoto, S.; Ishii, Y.; et al. PDGFRβ Regulates Adipose Tissue Expansion and Glucose Metabolism via Vascular Remodeling in Diet-Induced Obesity. Diabetes 2017, 66, 1008–1021. [Google Scholar] [CrossRef]
- Watanabe, E.; Wada, T.; Okekawa, A.; Kitamura, F.; Komatsu, G.; Onogi, Y.; Yamamoto, S.; Sasahara, M.; Kitada, M.; Koya, D.; et al. Stromal cell-derived factor 1 (SDF1) attenuates platelet-derived growth factor-B (PDGF-B)-induced vascular remodeling for adipose tissue expansion in obesity. Angiogenesis 2020, 23, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Onogi, Y.; Wada, T.; Okekawa, A.; Matsuzawa, T.; Watanabe, E.; Ikeda, K.; Nakano, M.; Kitada, M.; Koya, D.; Tsuneki, H.; et al. Pro-inflammatory macrophages coupled with glycolysis remodel adipose vasculature by producing platelet-derived growth factor-B in obesity. Sci. Rep. 2020, 10, 1–13. [Google Scholar] [CrossRef]
- Lopatina, T.; Bruno, S.; Tetta, C.; Kalinina, N.; Porta, M.; Camussi, G. Platelet-derived growth factor regulates the secretion of extracellular vesicles by adipose mesenchymal stem cells and enhances their angiogenic potential. Cell Commun. Signal. 2014, 12, 26. [Google Scholar] [CrossRef]
- Shin, S.; Pang, Y.; Park, J.; Liu, L.; Lukas, B.E.; Kim, S.H.; Kim, K.-W.; Xu, P.; Berry, D.C.; Jiang, Y. Dynamic control of adipose tissue development and adult tissue homeostasis by platelet-derived growth factor receptor alpha. eLife 2020, 9, e56189. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Murray, D.L.; Choy, L.N.; Spiegelman, B.M. Tumor necrosis factor alpha inhibits signaling from the insulin receptor. Proc. Natl. Acad. Sci. USA 1994, 91, 4854–4858. [Google Scholar] [CrossRef]
- Bonner, J.C. Regulation of PDGF and its receptors in fibrotic diseases. Cytokine Growth Factor Rev. 2004, 15, 255–273. [Google Scholar] [CrossRef] [PubMed]
- Reggio, S.; Rouault, C.; Poitou, C.; Bichet, J.-C.; Prifti, E.; Bouillot, J.-L.; Rizkalla, S.; Lacasa, D.; Tordjman, J.; Clément, K. Increased Basement Membrane Components in Adipose Tissue During Obesity: Links With TGFβ and Metabolic Phenotypes. J. Clin. Endocrinol. Metab. 2016, 101, 2578–2587. [Google Scholar] [CrossRef] [PubMed]
- Choy, L.; Skillington, J.; Derynck, R. Roles of Autocrine TGF-β Receptor and Smad Signaling in Adipocyte Differentiation. J. Cell Biol. 2000, 149, 667–682. [Google Scholar] [CrossRef]
- Elmquist, J.K.; Maratos-Flier, E.; Saper, C.B.; Flier, J.S. Unraveling the central nervous system pathways underlying responses to leptin. Nat. Neurosci. 1998, 1, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Korbonits, M.; Trainer, P.J.; Little, J.A.; Edwards, R.; Kopelman, P.G.; Besser, G.M.; Svec, F.; Grossman, A.B. Leptin levels do not change acutely with food administration in normal or obese subjects, but are negatively correlated with pituitary-adrenal activity. Clin. Endocrinol. 1997, 46, 751–757. [Google Scholar] [CrossRef]
- Zhang, Y.; Proenca, R.; Maffei, M.; Barone, M.; Leopold, L.; Friedman, J.M. Positional cloning of the mouse obese gene and its human homologue. Nature 1994, 372, 425–432. [Google Scholar] [CrossRef]
- Myers, M.G., Jr.; Heymsfield, S.B.; Haft, C.; Kahn, B.B.; Laughlin, M.; Leibel, R.L.; Tschöp, M.H.; Yanovski, J.A. Challenges and Opportunities of Defining Clinical Leptin Resistance. Cell Metab. 2012, 15, 150–156. [Google Scholar] [CrossRef]
- Saladin, R.; De Vos, P.; Guerre-Millot, M.; Leturque, A.; Girard, J.; Staels, B.; Auwerx, J. Transient increase in obese gene expression after food intake or insulin administration. Nature 1995, 377, 527–528. [Google Scholar] [CrossRef]
- Park, H.-Y.; Kwon, H.M.; Lim, H.J.; Hong, B.K.; Lee, J.Y.; Park, B.E.; Jang, Y.S.; Cho, S.Y.; Kim, H.-S. Potential role of leptin in angiogenesis: Leptin induces endothelial cell proliferation and expression of matrix metalloproteinases in vivo and in vitro. Exp. Mol. Med. 2001, 33, 95–102. [Google Scholar] [CrossRef]
- Artwohl, M.; Roden, M.; Hölzenbein, T.; Freudenthaler, A.; Waldhäusl, W.; Baumgartner-Parzer, S. Modulation by leptin of proliferation and apoptosis in vascular endothelial cells. Int. J. Obes. 2002, 26, 577–580. [Google Scholar] [CrossRef]
- Kiernan, K.; MacIver, N.J. The Role of the Adipokine Leptin in Immune Cell Function in Health and Disease. Front. Immunol. 2021, 11. [Google Scholar] [CrossRef]
- Bruno, A.; Di Sano, C.; Simon, H.-U.; Chanez, P.; Patti, A.M.; Di Vincenzo, S.; Dino, P.; D’Esposito, V.; Formisano, P.; Beguinot, F.; et al. Leptin and TGF-β1 Downregulate PREP1 Expression in Human Adipose-Derived Mesenchymal Stem Cells and Mature Adipocytes. Front. Cell Dev. Biol. 2021, 9, 1756. [Google Scholar] [CrossRef]
- Wagoner, B.; Hausman, D.B.; Harris, R.B.S. Direct and indirect effects of leptin on preadipocyte proliferation and differentiation. Am. J. Physiol. Integr. Comp. Physiol. 2006, 290, R1557–R1564. [Google Scholar] [CrossRef]
- Mick, G.J.; Wang, X.; Fu, C.L.; McCormick, K.L. Inhibition of leptin secretion by insulin and metformin in cultured rat adipose tissue. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2000, 1502, 426–432. [Google Scholar] [CrossRef]
- Nolan, J.J.; Olefsky, J.M.; Nyce, M.R.; Considine, R.V.; Caro, J.F. Effect of Troglitazone on Leptin Production: Studies In Vitro and in Human Subjects. Diabetes 1996, 45, 1276–1278. [Google Scholar] [CrossRef]
- Kolaczynski, J.W.; Ohannesian, J.P.; Considine, R.V.; Marco, C.C.; Caro, J.F. Response of leptin to short-term and prolonged overfeeding in humans. J. Clin. Endocrinol. Metab. 1996, 81, 4162–4165. [Google Scholar] [CrossRef]
- Kolaczynski, J.W.; Nyce, M.R.; Considine, R.V.; Boden, G.; Nolan, J.J.; Henry, R.; Mudaliar, S.R.; Olefsky, J.; Caro, J.F. Acute and Chronic Effect of Insulin on Leptin Production in Humans: Studies In Vivo and In Vitro. Diabetes 1996, 45, 699–701. [Google Scholar] [CrossRef]
- Morad, V.; Abrahamsson, A.; Kjölhede, P.; Dabrosin, C. Adipokines and Vascular Endothelial Growth Factor in Normal Human Breast Tissue in Vivo – Correlations and Attenuation by Dietary Flaxseed. J. Mammary Gland. Biol. Neoplasia 2016, 21, 69–76. [Google Scholar] [CrossRef]
- Cao, R.; Brakenhielm, E.; Wahlestedt, C.; Thyberg, J.; Cao, Y. Leptin induces vascular permeability and synergistically stimulates angiogenesis with FGF-2 and VEGF. Proc. Natl. Acad. Sci. USA 2001, 98, 6390–6395. [Google Scholar] [CrossRef]
- Arita, Y.; Kihara, S.; Ouchi, N.; Takahashi, M.; Maeda, K.; Miyagawa, J.-I.; Hotta, K.; Shimomura, I.; Nakamura, T.; Miyaoka, K.; et al. Paradoxical Decrease of an Adipose-Specific Protein, Adiponectin, in Obesity. Biochem. Biophys. Res. Commun. 1999, 257, 79–83. [Google Scholar] [CrossRef]
- Fu, Y.; Luo, N.; Klein, R.L.; Garvey, W.T. Adiponectin promotes adipocyte differentiation, insulin sensitivity, and lipid accumulation. J. Lipid Res. 2005, 46, 1369–1379. [Google Scholar] [CrossRef]
- Kobashi, C.; Urakaze, M.; Kishida, M.; Kibayashi, E.; Kobayashi, H.; Kihara, S.; Funahashi, T.; Takata, M.; Temaru, R.; Sato, A.; et al. Adiponectin Inhibits Endothelial Synthesis of Interleukin-8. Circ. Res. 2005, 97, 1245–1252. [Google Scholar] [CrossRef]
- Okamoto, Y.; Folco, E.J.; Minami, M.; Wara, A.K.; Feinberg, M.W.; Sukhova, G.K.; Colvin, R.A.; Kihara, S.; Funahashi, T.; Luster, A.D.; et al. Adiponectin Inhibits the Production of CXC Receptor 3 Chemokine Ligands in Macrophages and Reduces T-Lymphocyte Recruitment in Atherogenesis. Circ. Res. 2008, 102, 218–225. [Google Scholar] [CrossRef]
- Chen, H.; Montagnani, M.; Funahashi, T.; Shimomura, I.; Quon, M.J. Adiponectin Stimulates Production of Nitric Oxide in Vascular Endothelial Cells. J. Biol. Chem. 2003, 278, 45021–45026. [Google Scholar] [CrossRef]
- Orlando, A.; Viazzi, F.; Giussani, M.; Nava, E.; Cazzaniga, E.; Bonino, B.; Palestini, P.; Parati, G.; Genovesi, S. Endothelin-1/nitric oxide balance and HOMA index in children with excess weight and hypertension: A pathophysiological model of hypertension. Hypertens. Res. 2019, 42, 1192–1199. [Google Scholar] [CrossRef]
- Padilla, J.; Carpenter, A.J.; Das, N.A.; Kandikattu, H.K.; López-Ongil, S.; Martinez-Lemus, L.A.; Siebenlist, U.; DeMarco, V.G.; Chandrasekar, B. TRAF3IP2 mediates high glucose-induced endothelin-1 production as well as endothelin-1-induced inflammation in endothelial cells. Am. J. Physiol.-Heart Circ. Physiol. 2017, 314, H52–H64. [Google Scholar] [CrossRef]
- Jurrissen, T.J.; Grunewald, Z.I.; Woodford, M.L.; Winn, N.C.; Ball, J.R.; Smith, T.N.; Wheeler, A.; Rawlings, A.L.; Staveley-O’Carroll, K.F.; Ji, Y.; et al. Overproduction of endothelin-1 impairs glucose tolerance but does not promote visceral adipose tissue inflammation or limit metabolic adaptations to exercise. Am. J. Physiol. Metab. 2019, 317, E548–E558. [Google Scholar] [CrossRef]
- Briançon-Marjollet, A.; Monneret, D.; Henri, M.; Puch, F.; Pépin, J.L.; Faure, P.; Godin-Ribuot, D. Endothelin regulates intermittent hypoxia-induced lipolytic remodelling of adipose tissue and phosphorylation of hormone-sensitive lipase. J. Physiol. 2016, 594, 1727–1740. [Google Scholar] [CrossRef]
- Schieber, M.; Chandel, N.S. ROS Function in Redox Signaling and Oxidative Stress. Curr. Biol. 2014, 24, R453–R462. [Google Scholar] [CrossRef] [PubMed]
- Greenstein, A.S.; Khavandi, K.; Withers, S.B.; Sonoyama, K.; Clancy, O.; Jeziorska, M.; Laing, I.; Yates, A.P.; Pemberton, P.W.; Malik, R.A.; et al. Local Inflammation and Hypoxia Abolish the Protective Anticontractile Properties of Perivascular Fat in Obese Patients. Circulation 2009, 119, 1661–1670. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, R.; González, J.; Paoletto, F. The role of oxidative stress in the pathophysiology of hypertension. Hypertens. Res. 2011, 34, 431–440. [Google Scholar] [CrossRef]
- Williams, C.R.; Lu, X.; Sutliff, R.L.; Hart, C.M. Rosiglitazone attenuates NF-κB-mediated Nox4 upregulation in hyperglycemia-activated endothelial cells. Am. J. Physiol. Cell Physiol. 2012, 303, C213–C223. [Google Scholar] [CrossRef] [PubMed]
- Shiraki, A.; Oyama, J.; Komoda, H.; Asaka, M.; Komatsu, A.; Sakuma, M.; Kodama, K.; Sakamoto, Y.; Kotooka, N.; Hirase, T.; et al. The glucagon-like peptide 1 analog liraglutide reduces TNF-α-induced oxidative stress and inflammation in endothelial cells. Atherosclerosis 2012, 221, 375–382. [Google Scholar] [CrossRef]
- Xia, F.; Wang, C.; Jin, Y.; Liu, Q.; Meng, Q.; Liu, K.; Sun, H. Luteolin Protects HUVECs from TNF-α-induced Oxidative Stress and Inflammation via its Effects on the Nox4/ROS-NF-κB and MAPK Pathways. J. Atheroscler. Thromb. 2014, 21, 768–783. [Google Scholar] [CrossRef]
- Elizalde, M.; Rydén, M.; van Harmelen, V.; Eneroth, P.; Gyllenhammar, H.; Holm, C.; Ramel, S.; Olund, A.; Arner, P.; Andersson, K. Expression of nitric oxide synthases in subcutaneous adipose tissue of nonobese and obese humans. J. Lipid Res. 2000, 41, 1244–1251. [Google Scholar] [CrossRef]
- Yan, H.; Aziz, E.; Shillabeer, G.; Wong, A.; Shanghavi, D.; Kermouni, A.; Abdel-Hafez, M.; Lau, D.C. Nitric oxide promotes differentiation of rat white preadipocytes in culture. J. Lipid Res. 2002, 43, 2123–2129. [Google Scholar] [CrossRef]
- Gaudiot, N.; Jaubert, A.-M.; Charbonnier, E.; Sabourault, D.; Lacasa, D.; Giudicelli, Y.; Ribiére, C. Modulation of White Adipose Tissue Lipolysis by Nitric Oxide. J. Biol. Chem. 1998, 273, 13475–13481. [Google Scholar] [CrossRef]
- Engeli, S.; Janke, J.; Gorzelniak, K.; Böhnke, J.; Ghose, N.; Lindschau, C.; Luft, F.C.; Sharma, A.M. Regulation of the nitric oxide system in human adipose tissue. J. Lipid Res. 2004, 45, 1640–1648. [Google Scholar] [CrossRef]
- Rajapakse, N.; Head, G.; Kaye, D.M. Say NO to Obesity-Related Hypertension. Hypertension 2016, 67, 813–819. [Google Scholar] [CrossRef]
- Tan, K.C.B.; Xu, A.; Chow, W.S.; Lam, M.C.W.; Ai, V.H.G.; Tam, S.C.F.; Lam, K.S.L. Hypoadiponectinemia Is Associated with Impaired Endothelium-Dependent Vasodilation. J. Clin. Endocrinol. Metab. 2004, 89, 765–769. [Google Scholar] [CrossRef]
- Hara, Y.; Wakino, S.; Tanabe, Y.; Saito, M.; Tokuyama, H.; Washida, N.; Tatematsu, S.; Yoshioka, K.; Homma, K.; Hasegawa, K.; et al. Rho and Rho-Kinase Activity in Adipocytes Contributes to a Vicious Cycle in Obesity That May Involve Mechanical Stretch. Sci. Signal. 2011, 4, ra3. [Google Scholar] [CrossRef] [PubMed]
- Soták, M.; Rajan, M.R.; Clark, M.; Biörserud, C.; Wallenius, V.; Hagberg, C.E.; Börgeson, E. Healthy Subcutaneous and Omental Adipose Tissue Is Associated with High Expression of Extracellular Matrix Components. Int. J. Mol. Sci. 2022, 23, 520. [Google Scholar] [CrossRef]
- Henegar, C.; Tordjman, J.; Achard, V.; Lacasa, D.; Cremer, I.; Guerre-Millo, M.; Poitou, C.; Basdevant, A.; Stich, V.; Viguerie, N.; et al. Adipose tissue transcriptomic signature highlights the pathological relevance of extracellular matrix in human obesity. Genome Biol. 2008, 9, R14. [Google Scholar] [CrossRef]
- Bradshaw, A.D. The role of SPARC in extracellular matrix assembly. J. Cell Commun. Signal. 2009, 3, 239–246. [Google Scholar] [CrossRef]
- Johnston, E.K.; Abbott, R.D. Adipose Tissue Development Relies on Coordinated Extracellular Matrix Remodeling, Angiogenesis, and Adipogenesis. Biomedicines 2022, 10, 2227. [Google Scholar] [CrossRef]
- Kos, K.; Wong, S.; Tan, B.; Gummesson, A.; Jernas, M.; Franck, N.; Kerrigan, D.; Nystrom, F.H.; Carlsson, L.M.; Randeva, H.S.; et al. Regulation of the Fibrosis and Angiogenesis Promoter SPARC/Osteonectin in Human Adipose Tissue by Weight Change, Leptin, Insulin, and Glucose. Diabetes 2009, 58, 1780–1788. [Google Scholar] [CrossRef]
- Luo, L.; Sun, X.; Tang, M.; Wu, J.; Qian, T.; Chen, S.; Guan, Z.; Jiang, Y.; Fu, Y.; Zheng, Z. Secreted Protein Acidic and Rich in Cysteine Mediates the Development and Progression of Diabetic Retinopathy. Front. Endocrinol. 2022, 13. [Google Scholar] [CrossRef]
- Spencer, M.; Unal, R.; Zhu, B.; Rasouli, N.; McGehee, R.E.; Peterson, C.A.; Kern, P.A. Adipose Tissue Extracellular Matrix and Vascular Abnormalities in Obesity and Insulin Resistance. J. Clin. Endocrinol. Metab. 2011, 96, E1990–E1998. [Google Scholar] [CrossRef]
- Sasso, M.; Abdennour, M.; Liu, Y.; Hazrak, H.; Aron-Wisnewsky, J.; Bouillot, J.-L.; Le Naour, G.; Bedossa, P.; Torjman, J.; Clément, K.; et al. Relevance of Adipose Tissue Stiffness Evaluated by Transient Elastography (AdipoScan™) in Morbidly Obese Patients before Bariatric Surgery. Phys. Procedia 2015, 70, 1264–1268. [Google Scholar] [CrossRef]
- Pi-Sunyer, F.X. Comorbidities of overweight and obesity: Current evidence and research issues. Med. Sci. Sports Exerc. 1999, 31, S602–S608. [Google Scholar] [CrossRef] [PubMed]
- Arroyo-Jousse, V.; Jaramillo, A.; Castaño-Moreno, E.; Lépez, M.; Carrasco-Negüe, K.; Casanello, P. Adipokines underlie the early origins of obesity and associated metabolic comorbidities in the offspring of women with pregestational obesity. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2019, 1866, 165558. [Google Scholar] [CrossRef] [PubMed]
- Pellegrinelli, V.; Rouault, C.; Veyrie, N.; Clément, K.; Lacasa, D. Endothelial Cells From Visceral Adipose Tissue Disrupt Adipocyte Functions in a Three-Dimensional Setting: Partial Rescue by Angiopoietin-1. Diabetes 2014, 63, 535–549. [Google Scholar] [CrossRef] [PubMed]
- Tornavaca, O.; Chia, M.; Dufton, N.; Almagro, L.O.; Conway, D.E.; Randi, A.M.; Schwartz, M.A.; Matter, K.; Balda, M.S. ZO-1 controls endothelial adherens junctions, cell–cell tension, angiogenesis, and barrier formation. J. Cell Biol. 2015, 208, 821–838. [Google Scholar] [CrossRef]
- Virdis, A.; Colucci, R.; Bernardini, N.; Blandizzi, C.; Taddei, S.; Masi, S. Microvascular Endothelial Dysfunction in Human Obesity: Role of TNF-α. J. Clin. Endocrinol. Metab. 2018, 104, 341–348. [Google Scholar]
- Gogg, S.; Smith, U.; Jansson, P.-A. Increased MAPK Activation and Impaired Insulin Signaling in Subcutaneous Microvascular Endothelial Cells in Type 2 Dia-betes: The Role of Endothelin-1. Diabetes 2019, 58, 2238–2245. [Google Scholar] [CrossRef]
- Steppan, C.M.; Bailey, S.T.; Bhat, S.; Brown, E.J.; Banerjee, R.R.; Wright, C.M.; Patel, H.R.; Ahima, R.S.; Lazar, M.A. The hormone resistin links obesity to diabetes. Nature 2001, 409, 307–312. [Google Scholar] [CrossRef]
- Verma, S.; Li, S.-H.; Bhat, S.; Wang, C.-H.; Fedak, P.W.; Li, R.-K.; Weisel, R.D.; Mickle, D.A. Resistin promotes endothelial cell activation: Further evidence of adipokine-endothelial interaction. Circulation 2003, 108, 736–740. [Google Scholar]
- Yudkin, J.S.; Eringa, E.; Stehouwer, C.D.A. “Vasocrine” signalling from perivascular fat: A mechanism linking insulin resistance to vascular disease. Circulation 2005, 365, 1817–1820. [Google Scholar] [CrossRef]
- Calles-Escandon, J.; Cipolla, M. Diabetes and Endothelial Dysfunction: A Clinical Perspective. Endocr. Rev. 2001, 22, 36–52. [Google Scholar]
- Wang, Z.; Wang, D.; Wang, Y. Cigarette Smoking and Adipose Tissue: The Emerging Role in Progression of Atherosclerosis. Mediat. Inflamm. 2017, 2017, 1–11. [Google Scholar] [CrossRef]
- Antoniades, C.; Kotanidis, C.; Berman, D.S. State-of-the-art review article. Atherosclerosis affecting fat: What can we learn by imaging perivascular adipose tissue? J. Cardiovasc. Comput. Tomogr. 2019, 13, 288–296. [Google Scholar] [CrossRef]
- Cai, Z.; Gong, Z.; Li, Z.; Li, L.; Kong, W. Vascular Extracellular Matrix Remodeling and Hypertension. Antioxidants Redox Signal. 2021, 34, 765–783. [Google Scholar] [CrossRef]
- Savage, D.B.; Petersen, K.F.; Shulman, G.I. Disordered Lipid Metabolism and the Pathogenesis of Insulin Resistance. Physiol. Rev. 2007, 87, 507–520. [Google Scholar] [CrossRef]
- Sun, K.; Tordjman, J.; Clément, K.; Scherer, P.E. Fibrosis and Adipose Tissue Dysfunction. Cell Metab. 2013, 18, 470–477. [Google Scholar] [CrossRef]
- Debari, M.K.; Abbott, R.D. Adipose Tissue Fibrosis: Mechanisms, Models, and Importance. Int. J. Mol. Sci. 2020, 21, 6030. [Google Scholar] [CrossRef]
- Chait, A.; den Hartigh, L.J. Adipose Tissue Distribution, Inflammation and Its Metabolic Consequences, Including Diabetes and Cardiovascular Disease. Front. Cardiovasc. Med. 2020, 7, 22. [Google Scholar] [CrossRef]
- Crewe, C.; An, Y.A.; Scherer, P.E. The ominous triad of adipose tissue dysfunction: Inflammation, fibrosis, and impaired angiogenesis. J. Clin. Investig. 2017, 127, 74–85. [Google Scholar] [CrossRef]
- Baynes, J.W. Role of Oxidative Stress in Development of Complications in Diabetes. Diabetes 1991, 40, 405–412. [Google Scholar]
- Busik, J.V.; Mohr, S.; Grant, M.B. Hyperglycemia-Induced Reactive Oxygen Species Toxicity to Endothelial Cells Is Dependent on Paracrine Mediators. Diabetes 2008, 57, 1952–1965. [Google Scholar] [CrossRef]
- Bouloumié, A.; Sengenès, C.; Portolan, G.; Galitzky, J.; Lafontan, M. Adipocyte produces matrix metalloproteinases 2 and 9: Involvement in adipose differentiation. Diabetes 2001, 50, 2080–2086. [Google Scholar]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnston, E.K.; Abbott, R.D. Adipose Tissue Paracrine-, Autocrine-, and Matrix-Dependent Signaling during the Development and Progression of Obesity. Cells 2023, 12, 407. https://doi.org/10.3390/cells12030407
Johnston EK, Abbott RD. Adipose Tissue Paracrine-, Autocrine-, and Matrix-Dependent Signaling during the Development and Progression of Obesity. Cells. 2023; 12(3):407. https://doi.org/10.3390/cells12030407
Chicago/Turabian StyleJohnston, Elizabeth K., and Rosalyn D. Abbott. 2023. "Adipose Tissue Paracrine-, Autocrine-, and Matrix-Dependent Signaling during the Development and Progression of Obesity" Cells 12, no. 3: 407. https://doi.org/10.3390/cells12030407
APA StyleJohnston, E. K., & Abbott, R. D. (2023). Adipose Tissue Paracrine-, Autocrine-, and Matrix-Dependent Signaling during the Development and Progression of Obesity. Cells, 12(3), 407. https://doi.org/10.3390/cells12030407







