Machine Learning Allows for Distinguishing Precancerous and Cancerous Human Epithelial Cervical Cells Using High-Resolution AFM Imaging of Adhesion Maps
Abstract
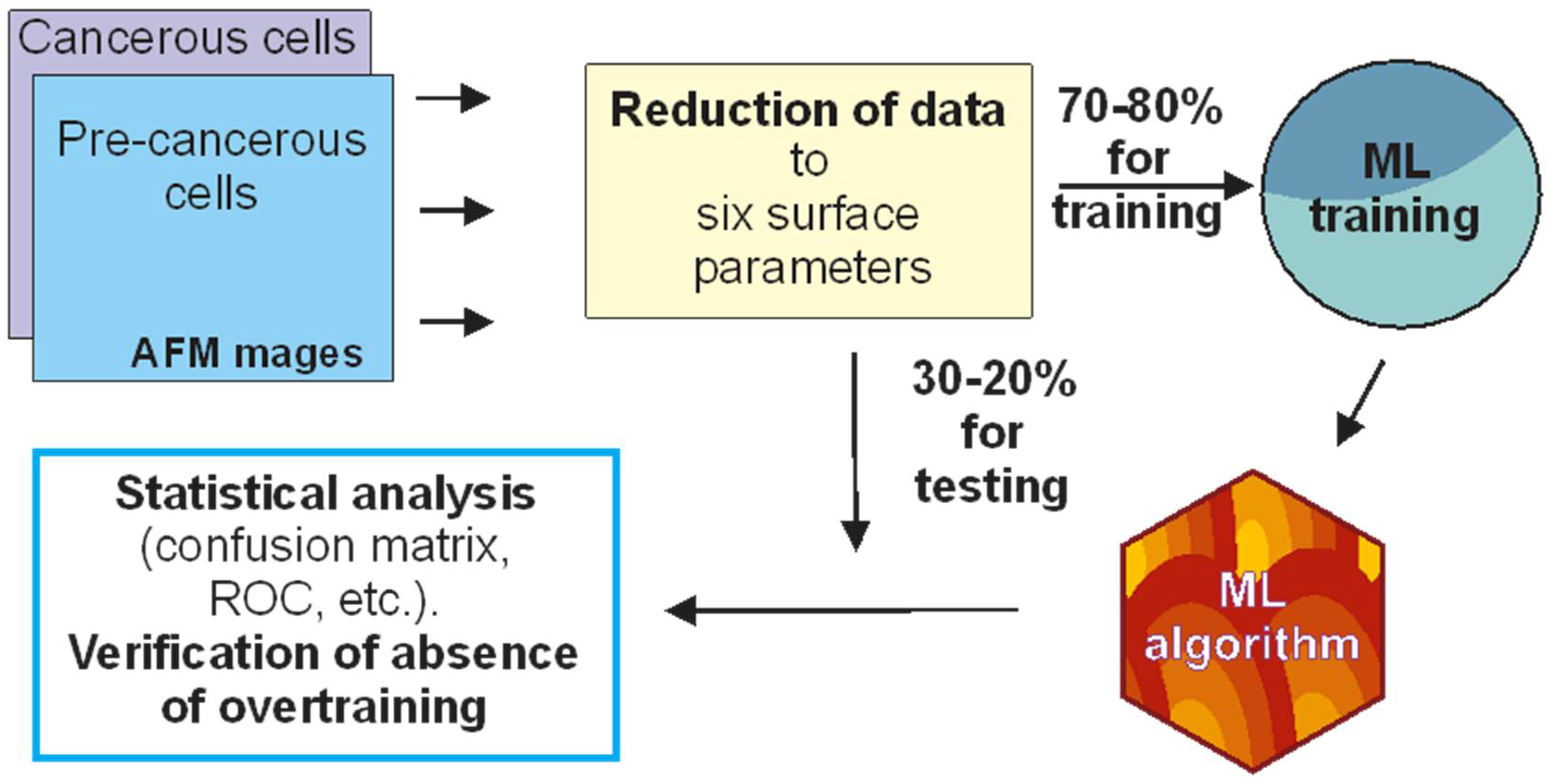
:1. Introduction
2. Results and Discussion
3. Methods
3.1. Cells and AFM imaging
3.2. Surface Parameters Used in This Study
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Costa, S.; Negri, G.; Sideri, M.; Santini, D.; Martinelli, G.; Venturoli, S.; Pelusi, C.; Syrjanen, S.; Syrjanen, K.; Pelusi, G. Human papillomavirus (HPV) test and PAP smear as predictors of outcome in conservatively treated adenocarcinoma in situ (AIS) of the uterine cervix. Gynecol. Oncol. 2007, 106, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Hanley, K.Z.; Tadros, T.S.; Briones, A.J.; Birdsong, G.G.; Mosunjac, M.B. Hematologic malignancies of the female genital tract diagnosed on liquid-based Pap test: Cytomorphologic features and review of differential diagnoses. Diagn. Cytopathol. 2009, 37, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Hoda, R.S.; Colello, C.; Roddy, M.; Houser, P.M. “Fruiting body” of Aspergillus species in a routine cervico-vaginal smear (Pap test). Diagn. Cytopathol. 2005, 33, 244–245. [Google Scholar] [CrossRef] [PubMed]
- Grubisic, G.; Klaric, P.; Jokanovic, L.; Soljacic Vranes, H.; Grbavac, I.; Bolanca, I. Diagnostic approach for precancerous and early invasive cancerous lesions of the uterine cervix. Coll. Antropol. 2009, 33, 1431–1436. [Google Scholar] [PubMed]
- Franco, E.L.; Schlecht, N.F.; Saslow, D. The epidemiology of cervical cancer. Cancer J. 2003, 9, 348–359. [Google Scholar] [CrossRef] [PubMed]
- Saslow, D.; Runowicz, C.D.; Solomon, D.; Moscicki, A.B.; Smith, R.A.; Eyre, H.J.; Cohen, C.; American Cancer, S. American Cancer Society guideline for the early detection of cervical neoplasia and cancer. CA Cancer J. Clin. 2002, 52, 342–362. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.mass.gov/doc/cervical-cancer-in-massachusetts-2004-2014/download (accessed on 8 August 2023).
- Fontham, E.T.H.; Wolf, A.M.D.; Church, T.R.; Etzioni, R.; Flowers, C.R.; Herzig, A.; Guerra, C.E.; Oeffinger, K.C.; Shih, Y.C.T.; Walter, L.C.; et al. Cervical cancer screening for individuals at average risk: 2020 guideline update from the American Cancer Society. Ca-Cancer J. Clin. 2020, 70, 321–346. [Google Scholar] [CrossRef] [PubMed]
- Katki, H.A.; Wentzensen, N. How might HPV testing be integrated into cervical screening? Lancet Oncol. 2012, 13, 8–10. [Google Scholar] [CrossRef]
- Doornewaard, H.; van der Schouw, Y.T.; van der Graaf, Y.; Bos, A.B.; van den Tweel, J.G. Observer variation in cytologic grading for cervical dysplasia of Papanicolaou smears with the PAPNET testing system. Cancer 1999, 87, 178–183. [Google Scholar] [CrossRef]
- Losa, G.A. Fractals in pathology: Are they really useful? Pathologica 1995, 87, 310–317. [Google Scholar]
- Pelling, A.E.; Li, Y.; Shi, W.; Gimzewski, J.K. Nanoscale visualization and characterization of Myxococcus xanthus cells with atomic force microscopy. Proc. Natl. Acad. Sci. USA 2005, 102, 6484–6489. [Google Scholar] [CrossRef] [PubMed]
- Matzke, R.; Jacobson, K.; Radmacher, M. Direct, high-resolution measurement of furrow stiffening during division of adherent cells. Nat. Cell Biol. 2001, 3, 607–610. [Google Scholar] [CrossRef] [PubMed]
- Suresh, S. Biomechanics and biophysics of cancer cells. Acta Biomater. 2007, 3, 413–438. [Google Scholar] [CrossRef] [PubMed]
- Lekka, M.; Laidler, P.; Ignacak, J.; Labedz, M.; Lekki, J.; Struszczyk, H.; Stachura, Z.; Hrynkiewicz, A.Z. The effect of chitosan on stiffness and glycolytic activity of human bladder cells. Biochim. Biophys. Acta 2001, 1540, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Iyer, S.; Woodworth, C.D.; Gaikwad, R.M.; Kievsky, Y.Y.; Sokolov, I. Towards nonspecific detection of malignant cervical cells with fluorescent silica beads. Small 2009, 5, 2277–2284. [Google Scholar] [CrossRef] [PubMed]
- Iyer, S.; Gaikwad, R.M.; Subba-Rao, V.; Woodworth, C.D.; Sokolov, I. AFM Detects Differences in the Surface Brush on Normal and Cancerous Cervical Cells. Nat. Nanotechnol. 2009, 4, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Dokukin, M.E.; Guz, N.V.; Woodworth, C.D.; Sokolov, I. Emergence of fractal geometry on the surface of human cervical epithelial cells during progression towards cancer. N. J. Phys. 2015, 17, 033019. [Google Scholar] [CrossRef]
- Dokukin, M.E.; Guz, N.V.; Gaikwad, R.M.; Woodworth, C.D.; Sokolov, I. Cell Surface as a Fractal: Normal and Cancerous Cervical Cells Demonstrate Different Fractal Behavior of Surface Adhesion Maps at the Nanoscale. Phys. Rev. Lett. 2011, 107, 028101. [Google Scholar] [CrossRef]
- Iyer, K.S.; Gaikwad, R.M.; Woodworth, C.D.; Volkov, D.O.; Sokolov, I. Physical Labeling of Papillomavirus-Infected, Immortal, and Cancerous Cervical Epithelial Cells Reveal Surface Changes at Immortal Stage. Cell Biochem. Biophys. 2012, 63, 109–116. [Google Scholar] [CrossRef]
- Origoni, M.; Cantatore, F.; Sopracordevole, F.; Clemente, N.; Spinillo, A.; Gardella, B.; De Vincenzo, R.; Ricci, C.; Landoni, F.; Di Meo, M.L.; et al. Colposcopy Accuracy and Diagnostic Performance: A Quality Control and Quality Assurance Survey in Italian Tertiary-Level Teaching and Academic Institutions-The Italian Society of Colposcopy and Cervico-Vaginal Pathology (SICPCV). Diagnostics 2023, 13, 1906. [Google Scholar] [CrossRef]
- Zhou, B.; Khosla, A.; Lapedriza, A.; Oliva, A.; Torralba, A. Learning Deep Features for Discriminative Localization. In Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, Las Vegas, NV, USA, 27–30 June 2016; pp. 2921–2929. [Google Scholar] [CrossRef]
- Arandjelovic, R.; Gronat, P.; Torii, A.; Pajdla, T.; Sivic, J. NetVLAD: CNN Architecture for Weakly Supervised Place Recognition. IEEE Trans. Pattern Anal. Mach. Intell. 2018, 40, 1437–1451. [Google Scholar] [CrossRef] [PubMed]
- Azuri, I.; Rosenhek-Goldian, I.; Regev-Rudzki, N.; Fantner, G.; Cohen, S.R. The role of convolutional neural networks in scanning probe microscopy: A review. Beilstein J. Nanotechnol. 2021, 12, 878–901. [Google Scholar] [CrossRef] [PubMed]
- Akbar, S.; Peikari, M.; Salama, S.; Nofech-Mozes, S.; Martel, A.L. The transition module: A method for preventing overfitting in convolutional neural networks. Comput. Methods Biomech. Biomed. Eng. Imaging Vis. 2019, 7, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Fausett, L.V. Fundamentals of Neural Networks: Architectures, Algorithms, and Applications; Prentice-Hall: Englewood Cliffs, NJ, USA, 1994; p. 461. [Google Scholar]
- Khabsa, M.; Elmagarmid, A.; Ilyas, I.; Hammady, H.; Ouzzani, M. Learning to identify relevant studies for systematic reviews using random forest and external information. Mach. Learn. 2016, 102, 465–482. [Google Scholar] [CrossRef]
- Breiman, L. Random forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Ishwaran, H. The effect of splitting on random forests. Mach. Learn. 2015, 99, 75–118. [Google Scholar] [CrossRef] [PubMed]
- Surface Texture (Surface Roughness, Waviness, and Lay) B46.1, 2019th ed.; ASME: New York, NY, USA, 2019.
- Sokolov, I.; Dokukin, M.E.; Kalaparthi, V.; Miljkovic, M.; Wang, A.; Seigne, J.D.; Grivas, P.; Demidenko, E. Noninvasive diagnostic imaging using machine-learning analysis of nanoresolution images of cell surfaces: Detection of bladder cancer. Proc. Natl. Acad. Sci. USA 2018, 115, 12920–12925. [Google Scholar] [CrossRef]
- Makarova, N.; Lekka, M.; Gnanachandran, K.; Sokolov, I. Mechanical Way To Study Molecular Structure of Pericellular Layer. Acs Appl. Mater. Inter. 2023, 15, 35962–35972. [Google Scholar] [CrossRef]
- Meakin, P. Fractals, Scaling, and Growth Far from Equilibrium; Cambridge University Press: Cambridge, UK; New York, NY, USA, 1998; p. 674. [Google Scholar]
- McCauley, J.L. Chaos, Dynamics, and Fractals: An Algorithmic Approach to Deterministic Chaos; Cambridge University Press: Cambridge, UK; New York, NY, USA, 1993; p. 323. [Google Scholar]
- Wu, K.K.S.; Lahav, O.; Rees, M.J. The large-scale smoothness of the Universe. Nature 1999, 397, 225–230. [Google Scholar] [CrossRef]
- Burrough, P.A. Fractal dimensions of landscapes and other environmental data. Nature 1981, 294, 240–242. [Google Scholar] [CrossRef]
- Morse, D.R.; Lawton, J.H.; Dodson, M.M.; Williamson, M.H. Fractal dimension of vegetation and the distribution of arthropod body lengths. Nature 1985, 314, 731–733. [Google Scholar] [CrossRef]
- Chopard, B.; Herrmann, H.J.; Vicsek, T. Structure and growth mechanism of mineral dendrites. Nature 1991, 353, 409–412. [Google Scholar] [CrossRef]
- Lovejoy, S. Area-Perimeter Relation for Rain and Cloud Areas. Science 1982, 216, 185–187. [Google Scholar] [CrossRef] [PubMed]
- Jones-Smith, K.; Mathur, H. Fractal Analysis: Revisiting Pollock’s drip paintings. Nature 2006, 444, E9–E10; discussion E10–E11. [Google Scholar] [CrossRef] [PubMed]
- Falconer, K.J. Fractal Geometry: Mathematical Foundations and Applications; Wiley: Chichester, UK; New York, NY, USA, 1990; p. 288. [Google Scholar]
- Prasad, S.; Rankine, A.; Prasad, T.; Song, P.; Dokukin, M.E.; Makarova, N.; Backman, V.; Sokolov, I. Atomic Force Microscopy Detects the Difference in Cancer Cells of Different Neoplastic Aggressiveness via Machine Learning. Adv. NanoBiomed Res. 2021, 1, 2000116. [Google Scholar] [CrossRef]
- Woodworth, C.D.; Bowden, P.E.; Doniger, J.; Pirisi, L.; Barnes, W.; Lancaster, W.D.; DiPaolo, J.A. Characterization of normal human exocervical epithelial cells immortalized in vitro by papillomavirus types 16 and 18 DNA. Cancer Res. 1988, 48, 4620–4628. [Google Scholar] [PubMed]
- Available online: https://figshare.com/articles/dataset/Data_for_the_surface_parameters_for_paper_M_E_Dokukin_i_et_al_i_2015_i_New_J_Phys_i_b_17_b_033019_b_DOI_b_10_1088_1367-2630_17_3_033019/23810616 (accessed on 8 September 2023).
- Sahin, O.; Magonov, S.; Su, C.; Quate, C.F.; Solgaard, O. An atomic force microscope tip designed to measure time-varying nanomechanical forces. Nat. Nanotechnol. 2007, 2, 507–514. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrov, M.; Sokolov, I. Machine Learning Allows for Distinguishing Precancerous and Cancerous Human Epithelial Cervical Cells Using High-Resolution AFM Imaging of Adhesion Maps. Cells 2023, 12, 2536. https://doi.org/10.3390/cells12212536
Petrov M, Sokolov I. Machine Learning Allows for Distinguishing Precancerous and Cancerous Human Epithelial Cervical Cells Using High-Resolution AFM Imaging of Adhesion Maps. Cells. 2023; 12(21):2536. https://doi.org/10.3390/cells12212536
Chicago/Turabian StylePetrov, Mikhail, and Igor Sokolov. 2023. "Machine Learning Allows for Distinguishing Precancerous and Cancerous Human Epithelial Cervical Cells Using High-Resolution AFM Imaging of Adhesion Maps" Cells 12, no. 21: 2536. https://doi.org/10.3390/cells12212536
APA StylePetrov, M., & Sokolov, I. (2023). Machine Learning Allows for Distinguishing Precancerous and Cancerous Human Epithelial Cervical Cells Using High-Resolution AFM Imaging of Adhesion Maps. Cells, 12(21), 2536. https://doi.org/10.3390/cells12212536




