Effect of Hyperbaric Oxygen and Inflammation on Human Gingival Mesenchymal Stem/Progenitor Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culturing
2.2. Experimental Groups
2.3. Control Group: Basic Medium
2.4. Flow Cytometric Analysis
2.5. Real-Time-Polymerase-Chain-Reaction
2.6. Next Generation Sequencing
2.7. ELISA pS45/Total β-Catenin
2.8. Cellular Proliferation and Colony-Forming Units (CFUs)
2.9. Multilinear Differentiation
2.10. Statistical Analysis
2.11. Data Availability
3. Results
3.1. G-MSCs’ Characterization
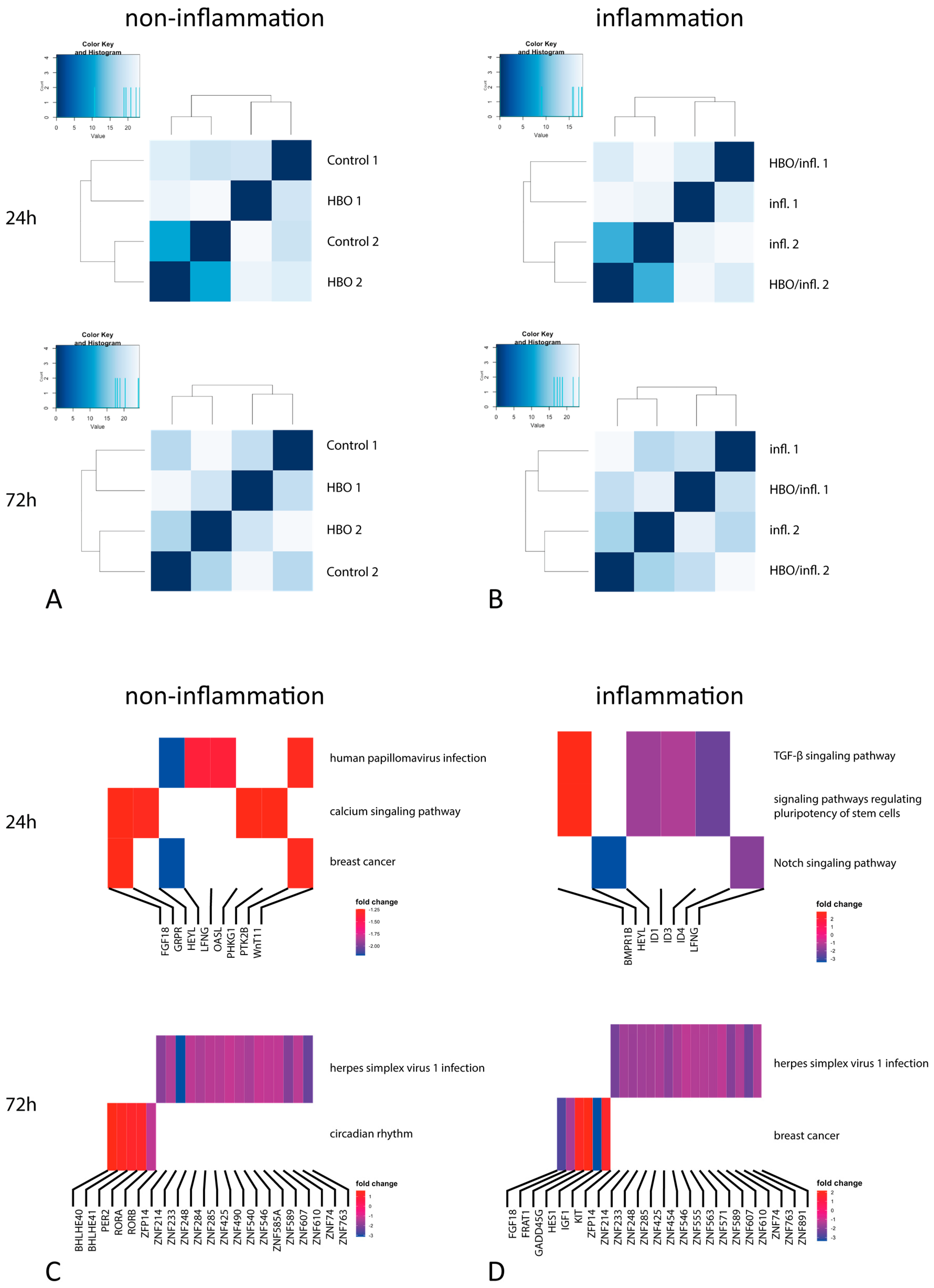
3.2. Next Generation Sequencing
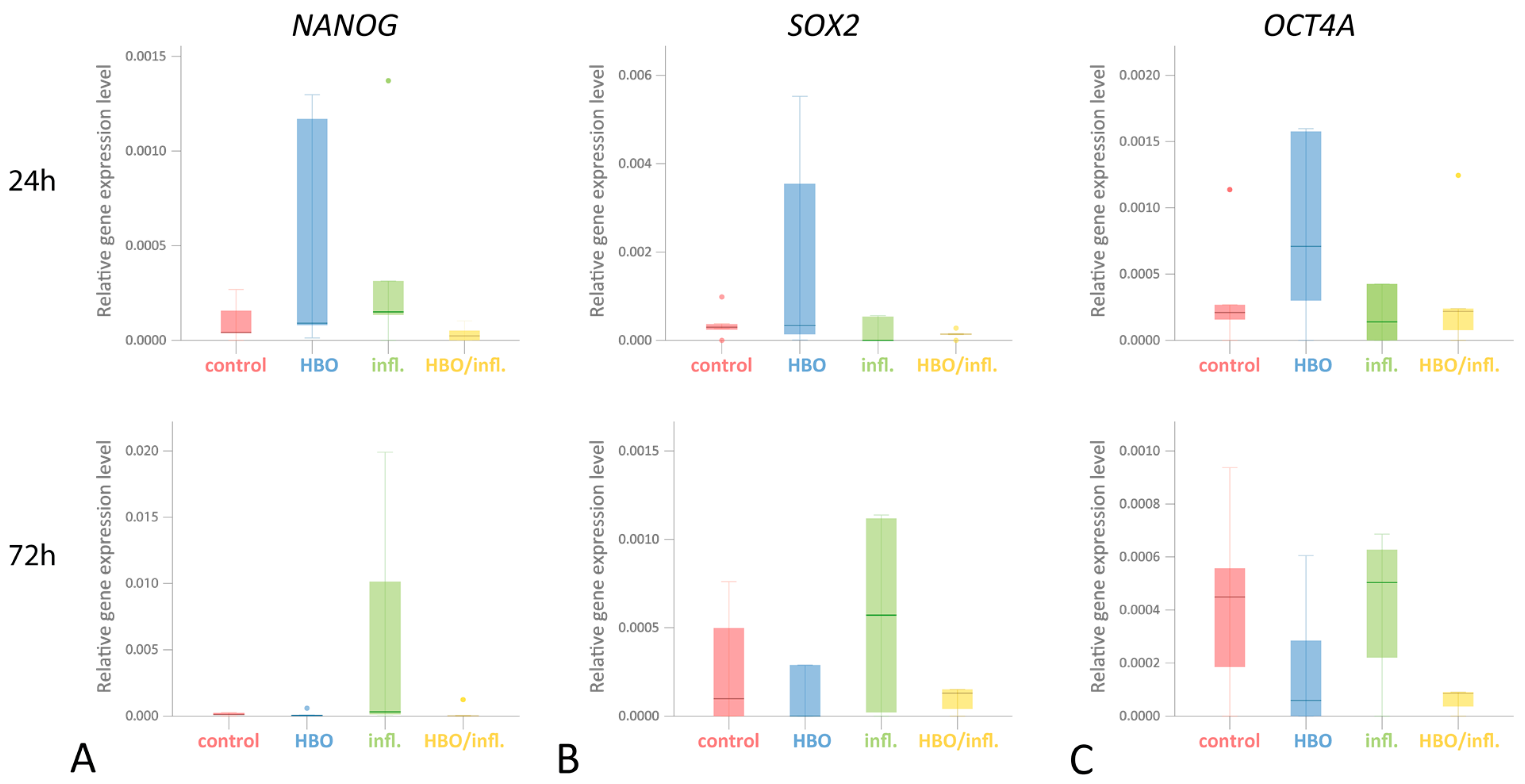
3.3. Pluripotency Genes
3.4. ELISA for Total and pS45-β-Catenin
3.5. Cellular Proliferation and CFUs
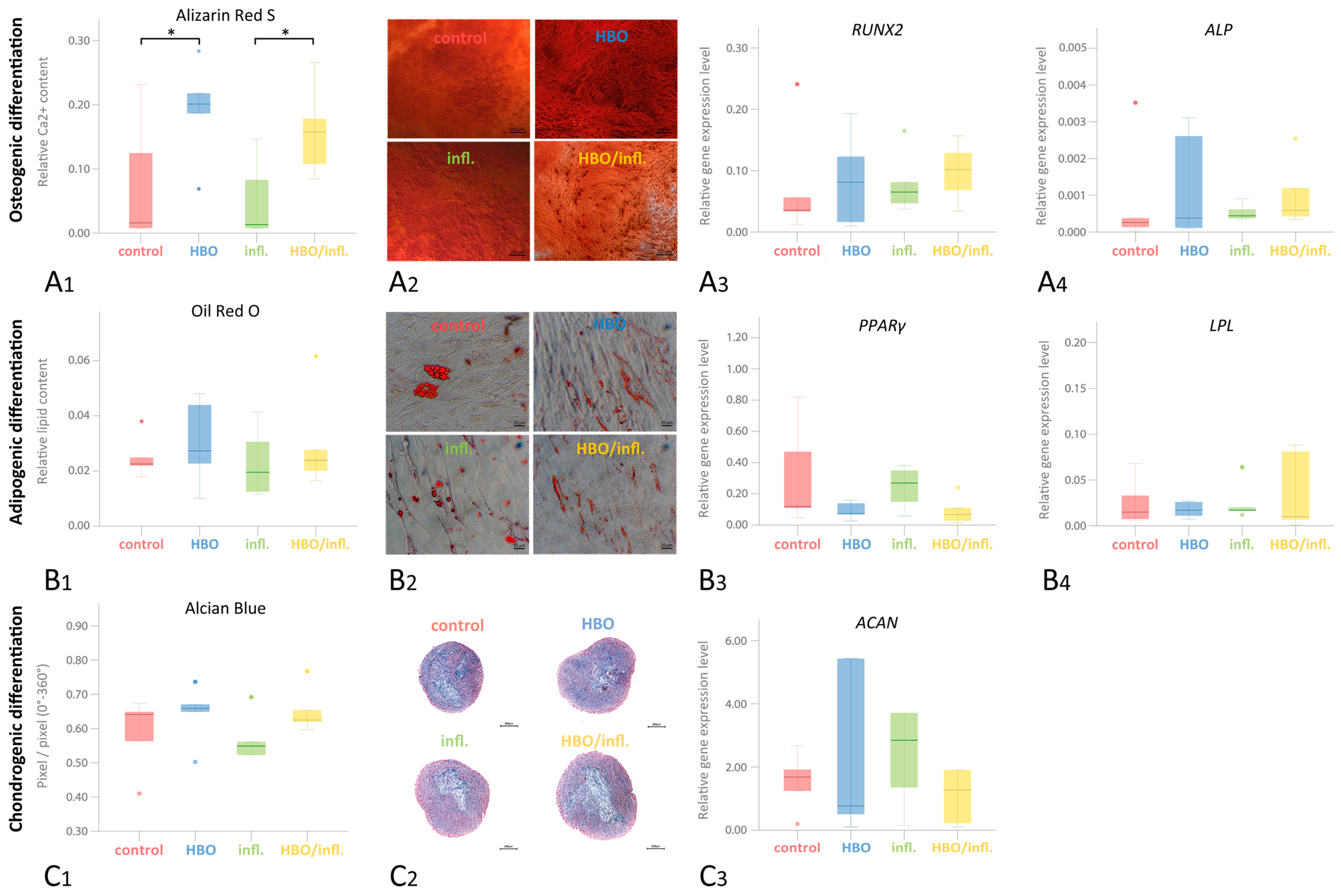
3.6. Multilinear Differentiation Potential
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fawzy El-Sayed, K.M.; Cosgarea, R.; Sculean, A.; Doerfer, C. Can vitamins improve periodontal wound healing/regeneration? Periodontol. 2000 2023. Online ahead of print. [Google Scholar] [CrossRef]
- Shapira, L.; Soskolne, W.A.; Sela, M.N.; Offenbacher, S.; Barak, V. The secretion of PGE2, IL-1 beta, IL-6, and TNF alpha by adherent mononuclear cells from early onset periodontitis patients. J. Periodontol. 1994, 65, 139–146. [Google Scholar] [CrossRef]
- Tan, J.; Dai, A.; Pan, L.; Zhang, L.; Wang, Z.; Ke, T.; Sun, W.; Wu, Y.; Ding, P.-H.; Chen, L. Inflamm-Aging-Related Cytokines of IL-17 and IFN-gamma Accelerate Osteoclastogenesis and Periodontal Destruction. J. Immunol. Res. 2021, 2021, 9919024. [Google Scholar] [CrossRef]
- Bosshardt, D.D. The periodontal pocket: Pathogenesis, histopathology and consequences. Periodontol. 2000 2018, 76, 43–50. [Google Scholar] [CrossRef]
- Sadek, K.M.; El Moshy, S.; Radwan, I.A.; Rady, D.; Abbass, M.M.S.; El-Rashidy, A.A.; Dörfer, C.E.; El-Sayed, K.M.F. Molecular Basis beyond Interrelated Bone Resorption/Regeneration in Periodontal Diseases: A Concise Review. Int. J. Mol. Sci. 2023, 24, 4599. [Google Scholar] [CrossRef]
- Fawzy El-Sayed, K.M.; Elahmady, M.; Adawi, Z.; Aboushadi, N.; Elnaggar, A.; Eid, M.; Hamdy, N.; Sanaa, D.; Dorfer, C.E. The periodontal stem/progenitor cell inflammatory-regenerative cross talk: A new perspective. J. Periodontal Res. 2019, 54, 81–94. [Google Scholar] [CrossRef]
- Zhou, L.; Dorfer, C.E.; Chen, L.; Fawzy El-Sayed, K.M. Porphyromonas gingivalis lipopolysaccharides affect gingival stem/progenitor cells attributes through NF-kappaB, but not Wnt/beta-catenin, pathway. J. Clin. Periodontol. 2017, 44, 1112–1122. [Google Scholar] [CrossRef]
- Zhou, L.L.; Liu, W.; Wu, Y.M.; Sun, W.L.; Dorfer, C.E.; Fawzy El-Sayed, K.M. Oral Mesenchymal Stem/Progenitor Cells: The Immunomodulatory Masters. Stem Cells Int. 2020, 2020, 1327405. [Google Scholar] [CrossRef]
- Li, X.; Hu, L.; Ma, L.; Chang, S.; Wang, W.; Feng, Y.; Xu, Y.; Hu, J.; Zhang, C.; Wang, S. Severe periodontitis may influence cementum and dental pulp through inflammation, oxidative stress, and apoptosis. J. Periodontol. 2019, 90, 1297–1306. [Google Scholar] [CrossRef]
- Sczepanik, F.S.C.; Grossi, M.L.; Casati, M.; Goldberg, M.; Glogauer, M.; Fine, N.; Tenenbaum, H.C. Periodontitis is an inflammatory disease of oxidative stress: We should treat it that way. Periodontol. 2000 2020, 84, 45–68. [Google Scholar] [CrossRef]
- El-Mouelhy, A.T.M.; Nasry, S.A.; Abou El-Dahab, O.; Sabry, D.; Fawzy El-Sayed, K. In vitro evaluation of the effect of the electronic cigarette aerosol, Cannabis smoke, and conventional cigarette smoke on the properties of gingival fibroblasts/gingival mesenchymal stem cells. J. Periodontal Res. 2022, 57, 104–114. [Google Scholar] [CrossRef]
- Mewes, L.; Knappe, C.; Graetz, C.; Wagner, J.; Demetrowitsch, T.J.; Jensen-Kroll, J.; El-Sayed, K.M.F.; Schwarz, K.; Dorfer, C.E.; Schreiber, S.; et al. Vitamin C and Omega-3 Fatty Acid Intake Is Associated with Human Periodontitis-A Nested Case-Control Study. Nutrients 2022, 14, 1936. [Google Scholar] [CrossRef]
- Burcea, A.; Mihai, L.L.; Bechir, A.; Suciu, M.; Bechir, E.S. Clinical Assessment of the Hyperbaric Oxygen Therapy Efficacy in Mild to Moderate Periodontal Affections: A Simple Randomised Trial. Medicina 2022, 58, 234. [Google Scholar] [CrossRef]
- Choudhury, R. Hypoxia and hyperbaric oxygen therapy: A review. Int. J. Gen. Med. 2018, 11, 431–442. [Google Scholar] [CrossRef]
- Parabucki, A.B.; Bozic, I.D.; Bjelobaba, I.M.; Lavrnja, I.C.; Brkic, P.D.; Jovanovic, T.S.; Savic, D.Z.; Stojiljkovic, M.B.; Pekovic, S.M. Hyperbaric oxygenation alters temporal expression pattern of superoxide dismutase 2 after cortical stab injury in rats. Croat. Med. J. 2012, 53, 586–597. [Google Scholar] [CrossRef]
- Re, K.; Patel, S.; Gandhi, J.; Suh, Y.; Reid, I.; Joshi, G.; Smith, N.L.; Khan, S.A. Clinical utility of hyperbaric oxygen therapy in dentistry. Med. Gas Res. 2019, 9, 93–100. [Google Scholar] [CrossRef]
- Mathieu, D.; Marroni, A.; Kot, J. Tenth European Consensus Conference on Hyperbaric Medicine: Recommendations for accepted and non-accepted clinical indications and practice of hyperbaric oxygen treatment. Diving Hyperb. Med. 2017, 47, 24–32. [Google Scholar] [CrossRef]
- Halbach, J.L.; Prieto, J.M.; Wang, A.W.; Hawisher, D.; Cauvi, D.M.; Reyes, T.; Okerblom, J.; Ramirez-Sanchez, I.; Villarreal, F.; Patel, H.H.; et al. Early hyperbaric oxygen therapy improves survival in a model of severe sepsis. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2019, 317, R160–R168. [Google Scholar] [CrossRef]
- Pena-Villalobos, I.; Casanova-Maldonado, I.; Lois, P.; Prieto, C.; Pizarro, C.; Lattus, J.; Osorio, G.; Palma, V. Hyperbaric Oxygen Increases Stem Cell Proliferation, Angiogenesis and Wound-Healing Ability of WJ-MSCs in Diabetic Mice. Front. Physiol. 2018, 9, 995. [Google Scholar] [CrossRef]
- Yoshinoya, Y.; Bocker, A.H.; Ruhl, T.; Siekmann, U.; Pallua, N.; Beier, J.P.; Kim, B.S. The Effect of Hyperbaric Oxygen Therapy on Human Adipose-Derived Stem Cells. Plast. Reconstr. Surg. 2020, 146, 309–320. [Google Scholar] [CrossRef]
- Chen, C.; Yang, Y.; Yao, Y. HBO Promotes the Differentiation of Neural Stem Cells via Interactions Between the Wnt3/beta-Catenin and BMP2 Signaling Pathways. Cell Transplant. 2019, 28, 1686–1699. [Google Scholar] [CrossRef]
- Gardin, C.; Bosco, G.; Ferroni, L.; Quartesan, S.; Rizzato, A.; Tatullo, M.; Zavan, B. Hyperbaric Oxygen Therapy Improves the Osteogenic and Vasculogenic Properties of Mesenchymal Stem Cells in the Presence of Inflammation In Vitro. Int. J. Mol. Sci. 2020, 21, 1452. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.X.; Liu, Z.G.; Liu, X.J.; Chen, Q.X. Umbilical cord-derived mesenchymal stem cell transplantation combined with hyperbaric oxygen treatment for repair of traumatic brain injury. Neural Regen. Res. 2016, 11, 107–113. [Google Scholar] [CrossRef]
- Lin, S.S.; Ueng, S.W.; Niu, C.C.; Yuan, L.J.; Yang, C.Y.; Chen, W.J.; Lee, M.S.; Chen, J.K. Hyperbaric oxygen promotes osteogenic differentiation of bone marrow stromal cells by regulating Wnt3a/beta-catenin signaling--an in vitro and in vivo study. Stem Cell Res. 2014, 12, 260–274. [Google Scholar] [CrossRef]
- Nogueira-Filho, G.R.; Rosa, B.T.; David-Neto, J.R. Effects of hyperbaric oxygen therapy on the treatment of severe cases of periodontitis. Undersea Hyperb. Med. 2010, 37, 107–114. [Google Scholar]
- Chen, T.L.; Xu, B.; Liu, J.C.; Li, S.G.; Li, D.Y.; Gong, G.C.; Wu, Z.F.; Lin, S.L.; Zhou, Y.J. Effects of hyperbaric oxygen on aggressive periodontitis and subgingival anaerobes in Chinese patients. J. Indian. Soc. Periodontol. 2012, 16, 492–497. [Google Scholar] [CrossRef]
- El-Sayed, K.M.; Paris, S.; Graetz, C.; Kassem, N.; Mekhemar, M.; Ungefroren, H.; Fandrich, F.; Dorfer, C. Isolation and characterisation of human gingival margin-derived STRO-1/MACS(+) and MACS(-) cell populations. Int. J. Oral Sci. 2015, 7, 80–88. [Google Scholar] [CrossRef]
- Clarke, D. USN Treatment Table 9. Diving Hyperb. Med. 2017, 47, 65. [Google Scholar]
- Sidney, L.E.; Kirkham, G.R.; Buttery, L.D. Comparison of osteogenic differentiation of embryonic stem cells and primary osteoblasts revealed by responses to IL-1beta, TNF-alpha, and IFN-gamma. Stem Cells Dev. 2014, 23, 605–617. [Google Scholar] [CrossRef]
- Fawzy-El-Sayed, K.; Mekhemar, M.; Adam-Klages, S.; Kabelitz, D.; Dorfer, C. TlR expression profile of human gingival margin-derived stem progenitor cells. Med. Oral Patol. Oral Cir. Bucal 2016, 21, e30–e38. [Google Scholar] [CrossRef]
- Fawzy El-Sayed, K.M.; Hein, D.; Dorfer, C.E. Retinol/inflammation affect stemness and differentiation potential of gingival stem/progenitor cells via Wnt/beta-catenin. J. Periodontal Res. 2019, 54, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Mekhemar, M.K.; Adam-Klages, S.; Kabelitz, D.; Dorfer, C.E.; Fawzy El-Sayed, K.M. TLR-induced immunomodulatory cytokine expression by human gingival stem/progenitor cells. Cell Immunol. 2018, 326, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Si, M.; Wang, H.; Mekhemar, M.K.; Dorfer, C.E.; Fawzy El-Sayed, K.M. IL-1/TNF-alpha Inflammatory and Anti-Inflammatory Synchronization Affects Gingival Stem/Progenitor Cells’ Regenerative Attributes. Stem Cells Int. 2017, 2017, 1349481. [Google Scholar] [CrossRef] [PubMed]
- Fawzy El-Sayed, K.M.; Klingebiel, P.; Dorfer, C.E. Toll-like Receptor Expression Profile of Human Dental Pulp Stem/Progenitor Cells. J. Endod. 2016, 42, 413–417. [Google Scholar] [CrossRef] [PubMed]
- Lv, F.J.; Tuan, R.S.; Cheung, K.M.; Leung, V.Y. Concise review: The surface markers and identity of human mesenchymal stem cells. Stem Cells 2014, 32, 1408–1419. [Google Scholar] [CrossRef]
- Mushahary, D.; Spittler, A.; Kasper, C.; Weber, V.; Charwat, V. Isolation, cultivation, and characterization of human mesenchymal stem cells. Cytom. A 2018, 93, 19–31. [Google Scholar] [CrossRef]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef]
- Mekhemar, M.; Tolle, J.; Dorfer, C.; Fawzy El-Sayed, K. TLR3 ligation affects differentiation and stemness properties of gingival mesenchymal stem/progenitor cells. J. Clin. Periodontol. 2020, 47, 991–1005. [Google Scholar] [CrossRef]
- Santamaria, S.; Sanchez, N.; Sanz, M.; Garcia-Sanz, J.A. Comparison of periodontal ligament and gingiva-derived mesenchymal stem cells for regenerative therapies. Clin. Oral. Investig. 2017, 21, 1095–1102. [Google Scholar] [CrossRef]
- Fawzy El-Sayed, K.M.; Dorfer, C.E. Gingival Mesenchymal Stem/Progenitor Cells: A Unique Tissue Engineering Gem. Stem Cells Int. 2016, 2016, 7154327. [Google Scholar] [CrossRef]
- Jin, S.H.; Lee, J.E.; Yun, J.H.; Kim, I.; Ko, Y.; Park, J.B. Isolation and characterization of human mesenchymal stem cells from gingival connective tissue. J. Periodontal Res. 2015, 50, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Mekhemar, M.; Tolle, J.; Hassan, Y.; Dorfer, C.; El-Sayed, K.F. Thymoquinone-Mediated Modulation of Toll-like Receptors and Pluripotency Factors in Gingival Mesenchymal Stem/Progenitor Cells. Cells 2022, 11, 1452. [Google Scholar] [CrossRef] [PubMed]
- Simmons, P.J.; Torok-Storb, B. Identification of stromal cell precursors in human bone marrow by a novel monoclonal antibody, STRO-1. Blood 1991, 78, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Cooper, J.S.; Phuyal, P.; Shah, N. Oxygen Toxicity. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Allardet-Servent, J.; Sicard, G.; Metz, V.; Chiche, L. Benefits and risks of oxygen therapy during acute medical illness: Just a matter of dose! Rev. Med. Interne 2019, 40, 670–676. [Google Scholar] [CrossRef]
- Kendall, A.C.; Whatmore, J.L.; Harries, L.W.; Winyard, P.G.; Smerdon, G.R.; Eggleton, P. Changes in inflammatory gene expression induced by hyperbaric oxygen treatment in human endothelial cells under chronic wound conditions. Exp. Cell Res. 2012, 318, 207–216. [Google Scholar] [CrossRef]
- Consonni, A.; Cipolla, L.; Guidetti, G.; Canobbio, I.; Ciraolo, E.; Hirsch, E.; Falasca, M.; Okigaki, M.; Balduini, C.; Torti, M. Role and regulation of phosphatidylinositol 3-kinase beta in platelet integrin alpha2beta1 signaling. Blood 2012, 119, 847–856. [Google Scholar] [CrossRef]
- Park, K.Y.; Jung, J.Y.; Park, J.; Hwang, J.U.; Kim, Y.W.; Hwang, I.; Lee, Y. A role for phosphatidylinositol 3-phosphate in abscisic acid-induced reactive oxygen species generation in guard cells. Plant Physiol. 2003, 132, 92–98. [Google Scholar] [CrossRef][Green Version]
- Beyfuss, K.; Hood, D.A. A systematic review of p53 regulation of oxidative stress in skeletal muscle. Redox Rep. 2018, 23, 100–117. [Google Scholar] [CrossRef]
- Song, H.; Ding, Z.; Chen, J.; Chen, T.; Wang, T.; Huang, J. The AMPK-SIRT1-FoxO1-NF-kappaB signaling pathway participates in hesperetin-mediated neuroprotective effects against traumatic brain injury via the NLRP3 inflammasome. Immunopharmacol. Immunotoxicol. 2022, 44, 970–983. [Google Scholar] [CrossRef]
- Mang, T.; Kleinschmidt-Doerr, K.; Ploeger, F.; Schoenemann, A.; Lindemann, S.; Gigout, A. BMPR1A is necessary for chondrogenesis and osteogenesis, whereas BMPR1B prevents hypertrophic differentiation. J. Cell Sci. 2020, 133, jcs246934. [Google Scholar] [CrossRef]
- Sebastian, V.P.; Salazar, G.A.; Coronado-Arrazola, I.; Schultz, B.M.; Vallejos, O.P.; Berkowitz, L.; Alvarez-Lobos, M.M.; Riedel, C.A.; Kalergis, A.M.; Bueno, S.M. Heme Oxygenase-1 as a Modulator of Intestinal Inflammation Development and Progression. Front. Immunol. 2018, 9, 1956. [Google Scholar] [CrossRef]
- Song, N.; Zhong, J.; Hu, Q.; Gu, T.; Yang, B.; Zhang, J.; Yu, J.; Ma, X.; Chen, Q.; Qi, J.; et al. FGF18 Enhances Migration and the Epithelial-Mesenchymal Transition in Breast Cancer by Regulating Akt/GSK3beta/Beta-Catenin Signaling. Cell Physiol. Biochem. 2018, 49, 1019–1032. [Google Scholar] [CrossRef]
- Salvador, J.M.; Brown-Clay, J.D.; Fornace, A.J., Jr. Gadd45 in stress signaling, cell cycle control, and apoptosis. Adv. Exp. Med. Biol. 2013, 793, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Levine, D.C.; Hong, H.; Weidemann, B.J.; Ramsey, K.M.; Affinati, A.H.; Schmidt, M.S.; Cedernaes, J.; Omura, C.; Braun, R.; Lee, C.; et al. NAD(+) Controls Circadian Reprogramming through PER2 Nuclear Translocation to Counter Aging. Mol. Cell 2020, 78, 835–849 e837. [Google Scholar] [CrossRef] [PubMed]
- Ostareck, D.H.; Ostareck-Lederer, A. RNA-Binding Proteins in the Control of LPS-Induced Macrophage Response. Front. Genet. 2019, 10, 31. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Liu, W.; Wong, C.W.; Zhu, W.; Lin, Y.; Hu, J.; Xu, W.; Zhang, J.; Sander, M.; Wang, Z.; et al. Zinc-finger antiviral protein acts as a tumor suppressor in colorectal cancer. Oncogene 2020, 39, 5995–6008. [Google Scholar] [CrossRef]
- Fawzy El-Sayed, K.M.; Nguyen, N.; Dorfer, C.E. Ascorbic Acid, Inflammatory Cytokines (IL-1beta/TNF-alpha/IFN-gamma), or Their Combination’s Effect on Stemness, Proliferation, and Differentiation of Gingival Mesenchymal Stem/Progenitor Cells. Stem Cells Int. 2020, 2020, 8897138. [Google Scholar] [CrossRef]
- Fawzy El-Sayed, K.M.; Bittner, A.; Schlicht, K.; Mekhemar, M.; Enthammer, K.; Hoppner, M.; Es-Souni, M.; Schulz, J.; Laudes, M.; Graetz, C.; et al. Ascorbic Acid/Retinol and/or Inflammatory Stimuli’s Effect on Proliferation/Differentiation Properties and Transcriptomics of Gingival Stem/Progenitor Cells. Cells 2021, 10, 3310. [Google Scholar] [CrossRef]
- Lin, S.S.; Ueng, S.W.; Niu, C.C.; Yuan, L.J.; Yang, C.Y.; Chen, W.J.; Lee, M.S.; Chen, J.K. Effects of hyperbaric oxygen on the osteogenic differentiation of mesenchymal stem cells. BMC Musculoskelet. Disord. 2014, 15, 56. [Google Scholar] [CrossRef]
- Grespi, F.; Soratroi, C.; Krumschnabel, G.; Sohm, B.; Ploner, C.; Geley, S.; Hengst, L.; Hacker, G.; Villunger, A. BH3-only protein Bmf mediates apoptosis upon inhibition of CAP-dependent protein synthesis. Cell Death Differ. 2010, 17, 1672–1683. [Google Scholar] [CrossRef]
- Kang, Y.; Nian, H.; Rajendran, P.; Kim, E.; Dashwood, W.M.; Pinto, J.T.; Boardman, L.A.; Thibodeau, S.N.; Limburg, P.J.; Lohr, C.V.; et al. HDAC8 and STAT3 repress BMF gene activity in colon cancer cells. Cell Death Dis. 2014, 5, e1476. [Google Scholar] [CrossRef] [PubMed]
- Kurppa, K.J.; Liu, Y.; To, C.; Zhang, T.; Fan, M.; Vajdi, A.; Knelson, E.H.; Xie, Y.; Lim, K.; Cejas, P.; et al. Treatment-Induced Tumor Dormancy through YAP-Mediated Transcriptional Reprogramming of the Apoptotic Pathway. Cancer Cell 2020, 37, 104–122 e112. [Google Scholar] [CrossRef] [PubMed]
- Kabakov, A.E.; Kudryavtsev, V.A.; Gabai, V.L. Determination of cell survival or death. Methods Mol. Biol. 2011, 787, 231–244. [Google Scholar] [CrossRef] [PubMed]
- Osnato, A.; Brown, S.; Krueger, C.; Andrews, S.; Collier, A.J.; Nakanoh, S.; Quiroga Londono, M.; Wesley, B.T.; Muraro, D.; Brumm, A.S.; et al. TGFbeta signalling is required to maintain pluripotency of human naive pluripotent stem cells. Elife 2021, 10, e67259. [Google Scholar] [CrossRef] [PubMed]
- Gajendrareddy, P.K.; Junges, R.; Cygan, G.; Zhao, Y.; Marucha, P.T.; Engeland, C.G. Increased oxygen exposure alters collagen expression and tissue architecture during ligature-induced periodontitis. J. Periodontal. Res. 2017, 52, 644–649. [Google Scholar] [CrossRef]
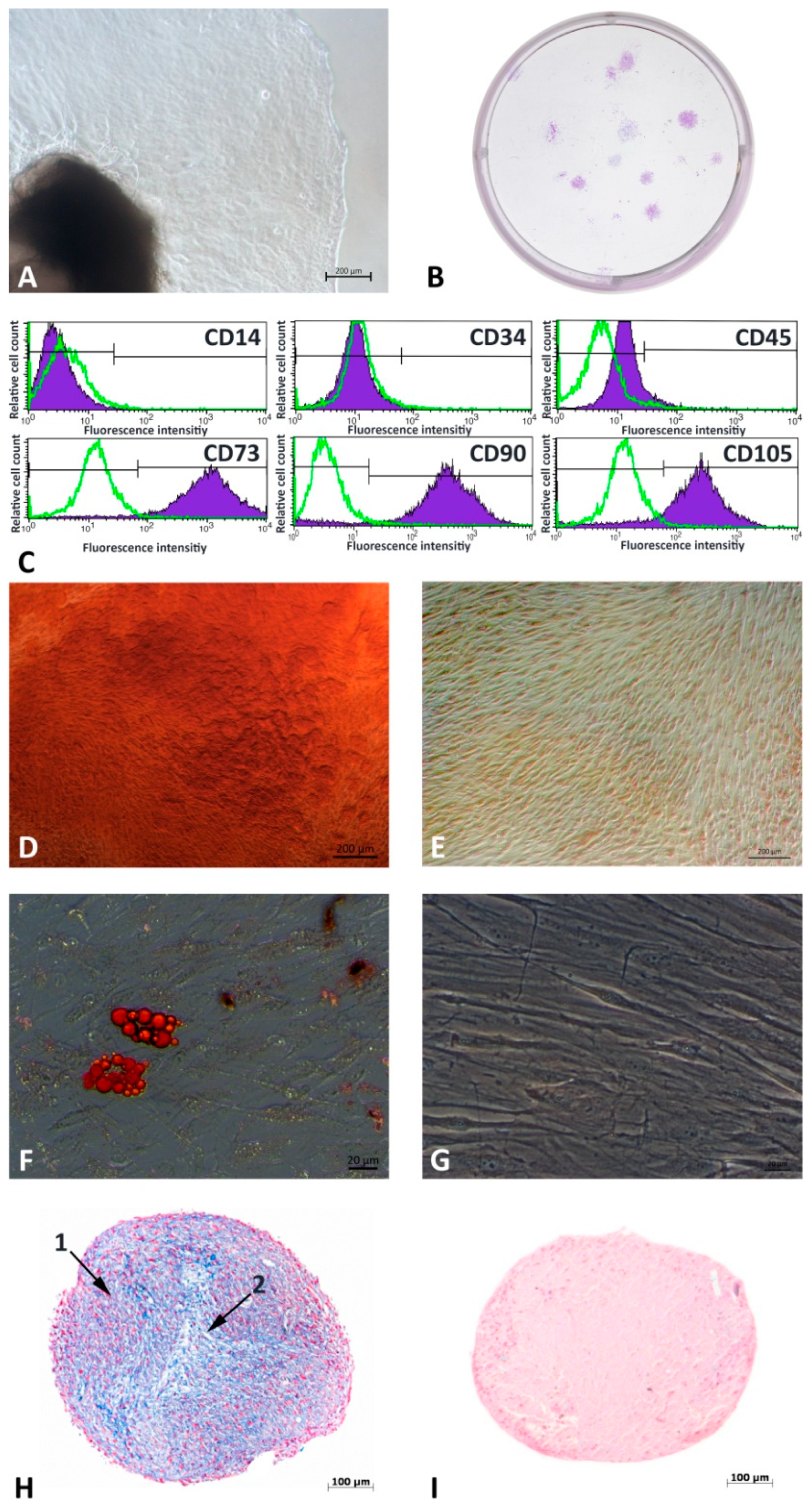

| Antibody | Clone | Color | Distributor | Product/Cat. No. |
|---|---|---|---|---|
| anti-human CD14 | REA599 | FITC | Miltenyi Biotec (Bergisch Gladbach, Germany) | 130-110-576 |
| anti-human CD34 | AC136 | PE | Miltenyi Biotec | 130-113-741 |
| anti-human CD45 | REA747 | APC | Miltenyi Biotec | 130-110-771 |
| anti-human CD73 | REA804 | PE | Miltenyi Biotec | 130-112-060 |
| anti-human CD90 | REA897 | FITC | Miltenyi Biotec | 130-114-901 |
| anti-human CD105 | REA794 | FITC | Miltenyi Biotec | 130-112-327 |
| REA Control (S) | REA293 | FITC | Miltenyi Biotec | 130-113-437 |
| REA Control (S) | REA293 | PE | Miltenyi Biotec | 130-113-438 |
| anti-human STRO-1 | STRO-1 | Alexa Fluor 647 | BioLegend (San Diego, CA, USA) | 340103 |
| anti-Cy5/Alexa Fluor 647 MicroBeads | - | - | Miltenyi Biotec | 130-091-395 |
| Contrast | Time | Gene Name | Name | LogFC | p-Value | p-ValueBH Adjust |
|---|---|---|---|---|---|---|
| non-inflammation | 24 h | PTK2B | Protein Tyrosine Kinase 2 Beta | −1.0 | 2.28 × 10−17 | 3.24 × 10−13 |
| BMF | Bcl2 Modifying Factor | −1.0 | 2.34 × 10−15 | 1.66 × 10−11 | ||
| TLL1 | Tolloid Like 1 | −1.0 | 6.72 × 10−13 | 3.19 × 10−9 | ||
| 72 h | TXNIP | Thioredoxin Interacting Protein | −2.4 | 5.57 × 10−27 | 7.90 × 10−23 | |
| PDK4 | Pyruvate Dehydrogenase Kinase 4 | −2.5 | 2.21 × 10−26 | 1.57 × 10−22 | ||
| ABL2 | ABL Proto-Oncogene 2, Non-Receptor Tyrosine Kinase | 1.7 | 2.24 × 10−24 | 1.06 × 10−20 | ||
| inflammation | 24 h | PI16 | Peptidase Inhibitor 16 | −6.0 | 8.90 × 10−22 | 1.24 × 10−17 |
| HMOX1 | Heme Oxygenase 1 | −1.0 | 7.60 × 10−15 | 5.28 × 10−11 | ||
| ID3 | Inhibitor Of DNA Binding 3, HLH Protein | −1.0 | 1.20 × 10−14 | 5.56 × 10−11 | ||
| 72 h | BHLHE40 | Basic Helix-Loop-Helix Family Member E40 | 3.0 | 1.06 × 10−8 | 0.0001 | |
| ARL4C | ADP Ribosylation Factor Like GTPase 4C | 1.6 | 5.16 × 10−7 | 0.003 | ||
| HMOX1 | Heme Oxygenase 1 | 1.7 | 6.10 × 10−7 | 0.003 |
| Gene | Assay ID | Gene Symbol | Accession ID | Primer Sequence |
|---|---|---|---|---|
| PGK-1 | 102083 | PGK1 H. sapiens | ENST00000373316 | FWD: GGAGAACCTCCGCTTTCAT REV: GCTGGCTCGGCTTTAACC |
| NANOG | 148147 | NANOG H. sapiens | ENST00000229307 | FWD: GAGATGCCTCACACGGAGAC REV: AGGGCTGTCCTGAATAAGCA |
| OCT4A | 113034 | OCT4A H. sapiens | ENST00000259915 | FWD: GCAAAACCCGGAGGAGTC REV: TCCCAGGGTGATCCTCTTCT |
| SOX2 | 111867 | SOX2 H. sapiens | ENST00000325404 | FWD: ATGGGTTCGGTGGTCAAGT REV: GGAGGAAGAGGTAACCACAGG |
| RUNX2 | 113380 | RUNX2 H. sapiens | ENST00000359524 | FWD: GCCTAGGCGCATTTCAGAT REV: CTGAGAGTGGAAGGCCAGAG |
| ALP | 103448 | ALP H. sapiens | ENST00000374840 | FWD: AGAACCCCAAAGGCTTCTTC REV: CTTGGCTTTTCCTTCATGGT |
| PPARγ | 110607 | PPARγ H. sapiens | ENST00000287820 | FWD: GACAGGAAAGACAACAGACAAATC REV: GGGGTGATGTGTTTGAACTTG |
| LPL | 113230 | LPL H. sapiens | ENST00000311322 | FWD: TCGTTCTCAGATGCCCTACA REV: GCCTGATTGGTATGGGTTTC |
| ACAN | 138057 | ACAN H. sapiens | ENST00000439576 | FWD: GAACGACAGGACCATCGAA REV: AAAGTTGTCAGGCTGGTTGG |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tölle, J.; Koch, A.; Schlicht, K.; Finger, D.; Kaehler, W.; Höppner, M.; Graetz, C.; Dörfer, C.; Schulte, D.M.; Fawzy El-Sayed, K. Effect of Hyperbaric Oxygen and Inflammation on Human Gingival Mesenchymal Stem/Progenitor Cells. Cells 2023, 12, 2479. https://doi.org/10.3390/cells12202479
Tölle J, Koch A, Schlicht K, Finger D, Kaehler W, Höppner M, Graetz C, Dörfer C, Schulte DM, Fawzy El-Sayed K. Effect of Hyperbaric Oxygen and Inflammation on Human Gingival Mesenchymal Stem/Progenitor Cells. Cells. 2023; 12(20):2479. https://doi.org/10.3390/cells12202479
Chicago/Turabian StyleTölle, Johannes, Andreas Koch, Kristina Schlicht, Dirk Finger, Wataru Kaehler, Marc Höppner, Christian Graetz, Christof Dörfer, Dominik M. Schulte, and Karim Fawzy El-Sayed. 2023. "Effect of Hyperbaric Oxygen and Inflammation on Human Gingival Mesenchymal Stem/Progenitor Cells" Cells 12, no. 20: 2479. https://doi.org/10.3390/cells12202479
APA StyleTölle, J., Koch, A., Schlicht, K., Finger, D., Kaehler, W., Höppner, M., Graetz, C., Dörfer, C., Schulte, D. M., & Fawzy El-Sayed, K. (2023). Effect of Hyperbaric Oxygen and Inflammation on Human Gingival Mesenchymal Stem/Progenitor Cells. Cells, 12(20), 2479. https://doi.org/10.3390/cells12202479








