Let-7g Upregulation Attenuated the KRAS–PI3K–Rac1–Akt Axis-Mediated Bioenergetic Functions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Chemicals, Reagents, and Plasmids
2.3. Antibodies
2.4. Measurement of DNA Fragmentation
2.5. Rac1 Activation Assay
2.6. Measurement of the Oxygen Consumption Rate (OCR) and Extracellular Acidification Rate (ECAR)
2.7. Glucose Uptake Assay
2.8. Measurement of Cellular ATP
2.9. Measurement of Cellular Pyruvate and Lactate
2.10. Measurement of Mitochondrial ROS Production
2.11. Measurement of Intracellular Glucose
2.12. Measurement of Cytosolic Calcium (Ca++)
2.13. Determination of Let-7 Expression by Quantitative Real-Time PCR
2.14. In Vitro RNA Cleavage Assay
2.15. Pyruvate Kinase Type M2 (PKM2) Activity Assay
2.16. Pyruvate Dehydrogenase Kinase 1 (PDK1) Activity Assay
2.17. Hexokinase II (HK-II) Activity Assay
2.18. Lactate Dehydrogenase (LDH) Activity Assay
2.19. Succinate Dehydrogenase (SDH) Activity Assay
2.20. Determination of Succinate
2.21. Mitochondrial DNA (mtDNA) Copy Number
2.22. Statistical Analysis
3. Results
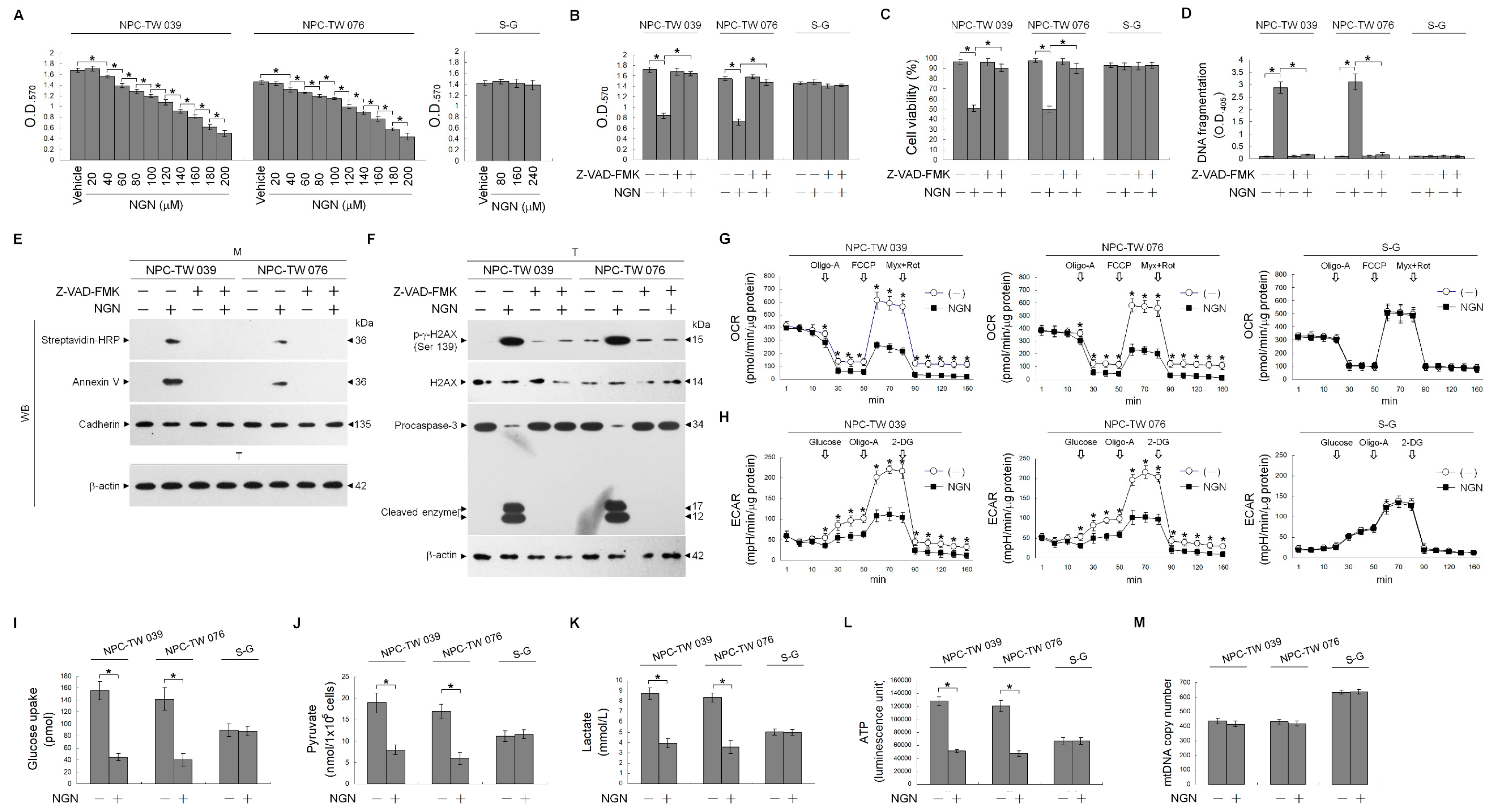
3.1. The Impairment of the Regulation of Glycolysis and Mitochondrial Oxidative Phosphorylation (OXPHOS) Confers the Apoptotic Death of NPC Cells
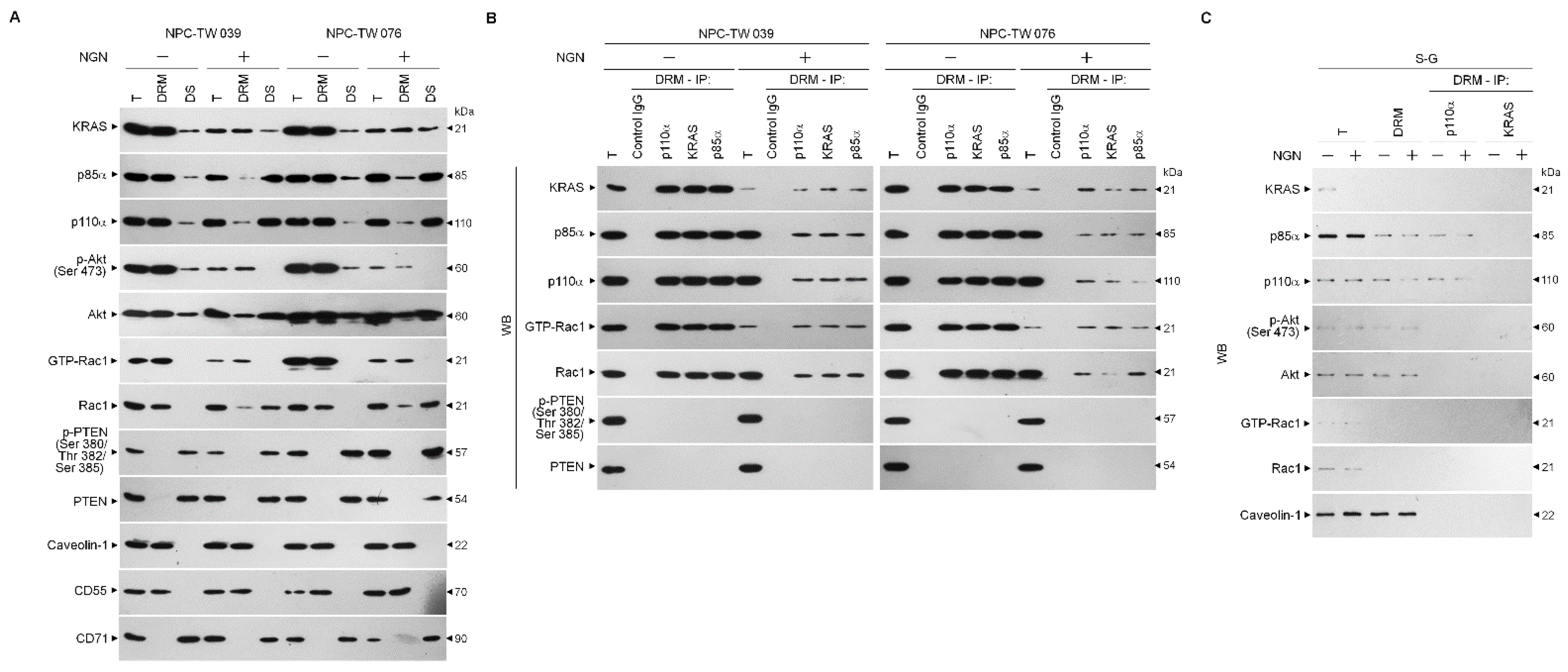
3.2. Decreased Interaction of KRAS with p110α by NGN Confers the Suppression of Lipid Raft-Associated KRAS–PI3K–Rac1–Akt-Mediated Bioenergetic Generation
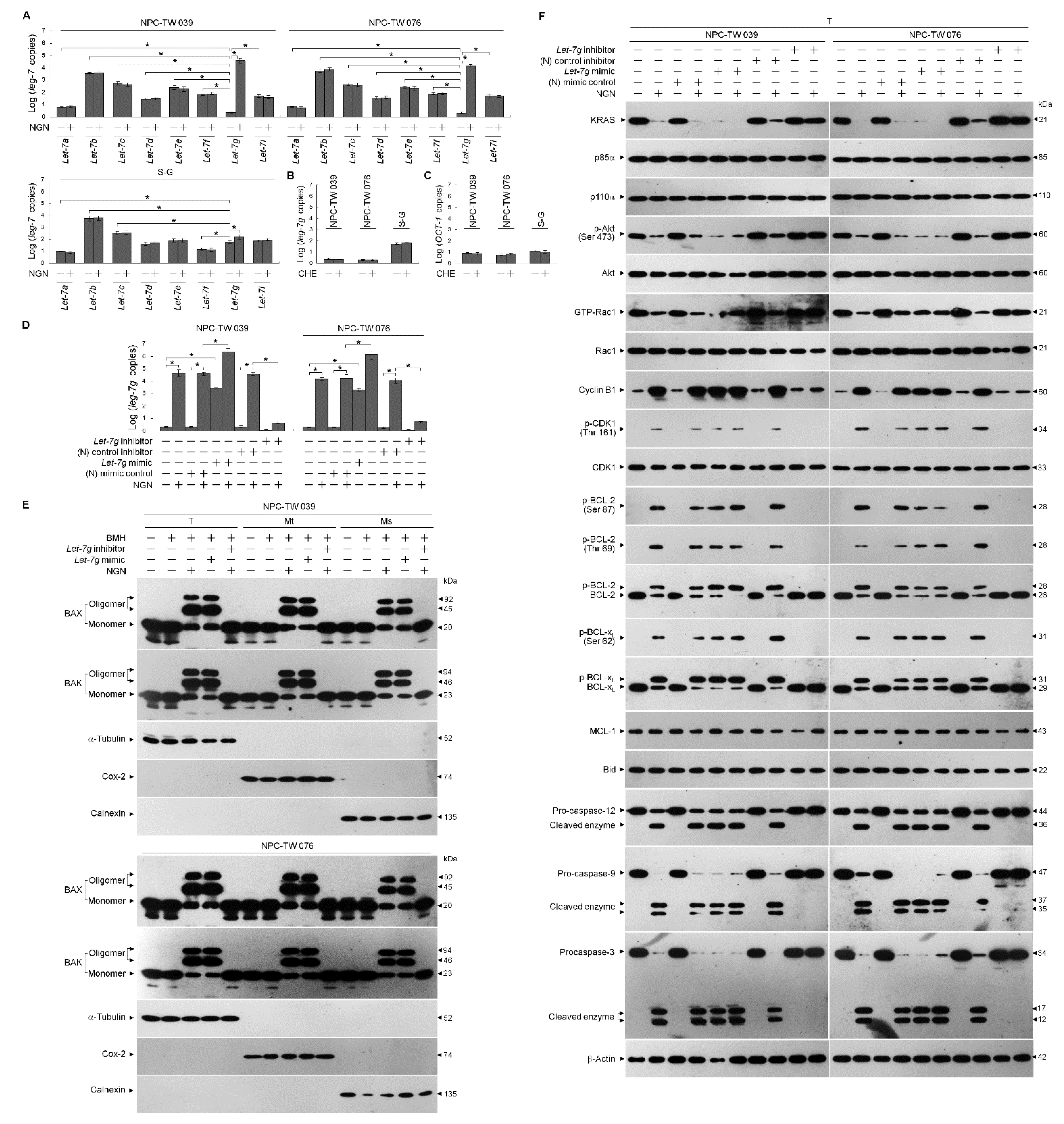
3.3. NGN-Induced KRAS–PI3K–Rac1–Akt-Modulated Metabolic Dysfunction of Glycolysis and Mitochondrial OXPHOS Associated with the Up-Regulation of let-7g
3.4. Let-7g Upregulation-Induced Suppression of Lipid Raft-Associated GLUT-1 Is Involved in the Inhibition of Glycolytic and Mitochondrial OXPHOS Activity
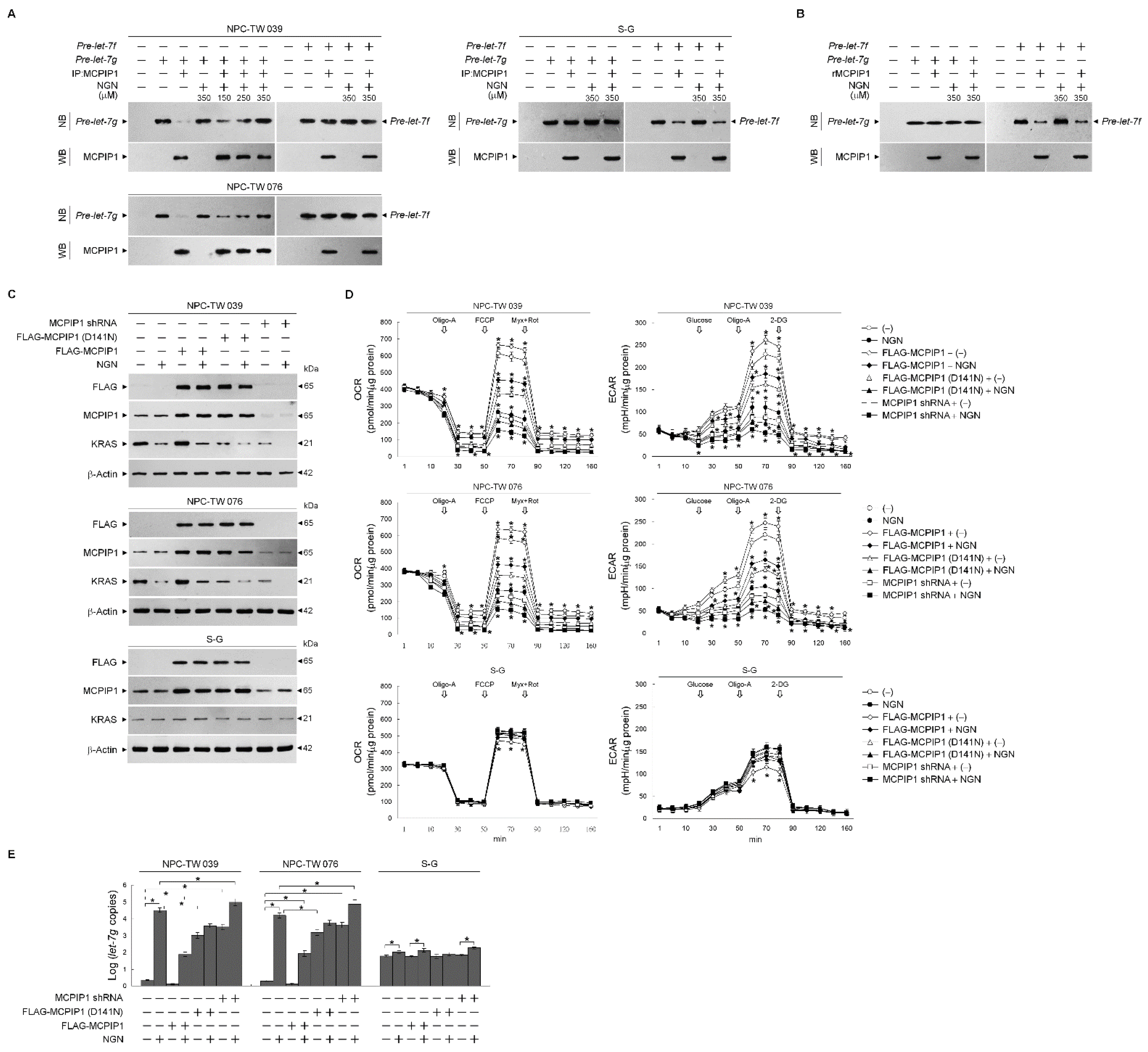
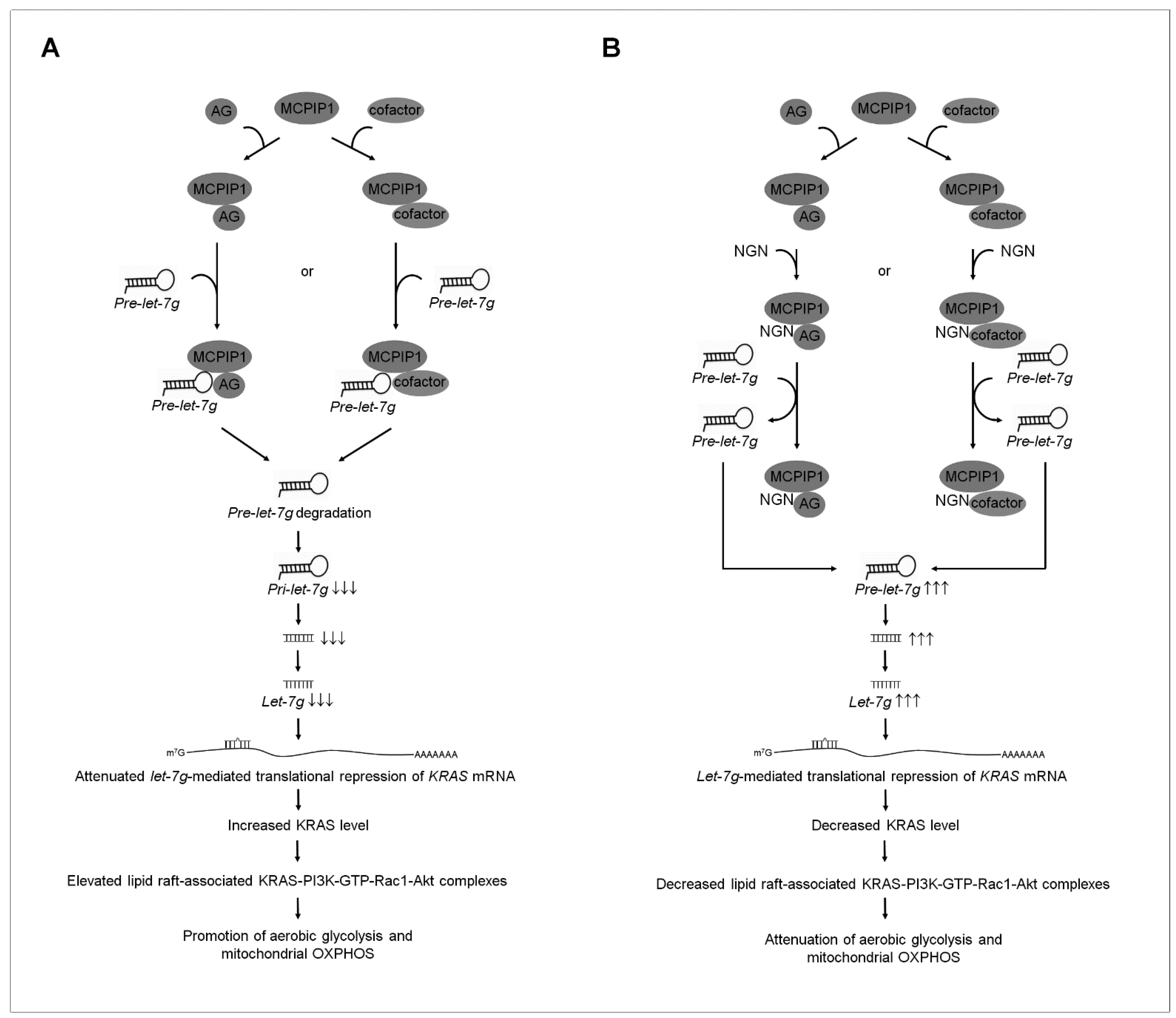
3.5. NGN Inhibited the MCPIP1-Mediated Degradation of let-7g
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Adenosine triphosphate | ATP |
| B cell lymphoma 2 | BCL-2 |
| Bcl-2-antagonist of cell death | BAD |
| Bcl-2 antagonist killer 1 | BAK |
| Bcl-2-associated x protein | BAX |
| BCL-2/B-cell lymphoma-extra large | BCL-xL |
| Cyclin dependent kinase 1 | CDK1 |
| Detergent-resistant membranes | DRM |
| Detergent-soluble | DS |
| Endoplasmic reticulum | ER |
| Epstein–Barr virus | EBV |
| Extracellular acidification rate | ECAR |
| Glucose transporter-1 | GLUT-1 |
| Hexokinase II | HK-II |
| Hypoxia-inducible factor 1α | HIF-α |
| Latent membrane protein 1 | LAMP1 |
| Lethal-7 | let-7 |
| MicroRNAs | miRNAs |
| Mitochondrial DNA | mtDNA |
| Mitochondrial oxidative phosphorylation | OXPHOS |
| Monocyte chemoattractant protein-induced protein-1 | MCPIP1 |
| Nasopharyngeal Carcinoma | NPC |
| Oxygen consumption rate | OCR |
| Phosphatase and tensin homolog deleted from chromosome 10 | PTEN |
| Phosphatidylinositol 3-kinase | PI3K |
| Phosphatidylinositol-4,5-bisphosphate | PIP2 |
| Phosphatidylinositol-3,4,5-trisphosphate | PIP3 |
| Poly (ADP-ribose) polymerase | PARP |
| Protein kinase B | Akt |
| Protein kinase C | PKC |
| Pyruvate dehydrogenase kinase 1 | PDK1 |
| Pyruvate kinase type M2— | PKM2 |
| Ras-related C3 botulinum toxin substrate 1 | Rac1 |
| Short hairpin RNA | shRNA |
| Succinate dehydrogenase | SDH |
References
- Gatenby, R.A.; Gillies, R.J. Why do cancers have high aerobic glycolysis? Nat. Rev. Cancer 2004, 4, 891–899. [Google Scholar] [CrossRef]
- Zhao, Y.; Butler, E.B.; Tan, M. Targeting cellular metabolism to improve cancer therapeutics. Cell Death Dis. 2013, 4, e532. [Google Scholar] [CrossRef] [PubMed]
- Ward, P.S.; Thompson, C.B. Metabolic reprogramming: A cancer hallmark even warburg did not anticipate. Cancer Cell 2012, 21, 297–308. [Google Scholar] [CrossRef]
- Goetzman, E.S.; Prochownik, E.V. The Role for Myc in Coordinating Glycolysis, Oxidative Phosphorylation, Glutaminolysis, and Fatty Acid Metabolism in Normal and Neoplastic Tissues. Front. Endocrinol. 2018, 9, 129. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J. Energy metabolism of cancer: Glycolysis versus oxidative phosphorylation (Review). Oncol. Lett. 2012, 4, 1151–1157. [Google Scholar] [CrossRef] [PubMed]
- Mathupala, S.P.; Ko, Y.H.; Pedersen, P.L. The pivotal roles of mitochondria in cancer: Warburg and beyond and encouraging prospects for effective therapies. Biochim. Biophys. Acta 2010, 1797, 1225–1230. [Google Scholar] [CrossRef]
- Szymanski, J.; Janikiewicz, J.; Michalska, B.; Patalas-Krawczyk, P.; Perrone, M.; Ziolkowski, W.; Duszynski, J.; Pinton, P.; Dobrzyn, A.; Wieckowski, M.R. Interaction of Mitochondria with the Endoplasmic Reticulum and Plasma Membrane in Calcium Homeostasis, Lipid Trafficking and Mitochondrial Structure. Int. J. Mol. Sci. 2017, 18, 1576. [Google Scholar] [CrossRef]
- Singh, P.K.; Mehla, K.; Hollingsworth, M.A.; Johnson, K.R. Regulation of Aerobic Glycolysis by microRNAs in Cancer. Mol. Cell. Pharmacol. 2011, 3, 125–134. [Google Scholar] [PubMed]
- Tomasetti, M.; Neuzil, J.; Dong, L. MicroRNAs as regulators of mitochondrial function: Role in cancer suppression. Biochim. Biophys. Acta 2014, 1840, 1441–1453. [Google Scholar] [CrossRef]
- Nguyen, L.H.; Zhu, H. Lin28 and let-7 in cell metabolism and cancer. Transl. Pediatr. 2015, 4, 4–11. [Google Scholar]
- Balzeau, J.; Menezes, M.R.; Cao, S.; Hagan, J.P. The LIN28/let-7 Pathway in Cancer. Front. Genet. 2017, 8, 31. [Google Scholar] [CrossRef]
- Luan, J.; Wang, J.; Su, Q.; Chen, X.; Jiang, G.; Xu, X. Meta-analysis of the differentially expressed microRNA profiles in nasopharyngeal carcinoma. Oncotarget 2016, 7, 10513–10521. [Google Scholar] [CrossRef]
- Shimizu, S.; Takehara, T.; Hikita, H.; Kodama, T.; Miyagi, T.; Hosui, A.; Tatsumi, T.; Ishida, H.; Noda, T.; Nagano, H.; et al. The let-7 family of microRNAs inhibits Bcl-xL expression and potentiates sorafenib-induced apoptosis in human hepatocellular carcinoma. J. Hepatol. 2010, 52, 698–704. [Google Scholar] [CrossRef]
- Hu, H.; Zhang, L.; Teng, G.; Wu, Y.; Chen, Y. A variant in 3′-untranslated region of KRAS compromises its interaction with hsa-let-7g and contributes to the development of lung cancer in patients with COPD. Int. J. Chronic Obstr. Pulm. Dis. 2015, 10, 1641–1649. [Google Scholar] [CrossRef]
- Chen, K.J.; Hou, Y.; Wang, K.; Li, J.; Xia, Y.; Yang, X.Y.; Lv, G.; Xing, X.L.; Shen, F. Reexpression of Let-7g microRNA inhibits the proliferation and migration via K-Ras/HMGA2/snail axis in hepatocellular carcinoma. BioMed Res. Int. 2014, 2014, 742417. [Google Scholar] [CrossRef] [PubMed]
- Deng, M.; Tang, H.; Zhou, Y.; Zhou, M.; Xiong, W.; Zheng, Y.; Ye, Q.; Zeng, X.; Liao, Q.; Guo, X.; et al. miR-216b suppresses tumor growth and invasion by targeting KRAS in nasopharyngeal carcinoma. J. Cell Sci. 2011, 124, 2997–3005. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhao, Y.; Liu, L.; Su, H.; Dong, D.; Wang, J.; Zhang, Y.; Chen, Q.; Li, C. MicroRNA-19b Promotes Nasopharyngeal Carcinoma More Sensitive to Cisplatin by Suppressing KRAS. Technol. Cancer Res. Treat. 2018, 17, 1533033818793652. [Google Scholar] [CrossRef]
- Castellano, E.; Sheridan, C.; Thin, M.Z.; Nye, E.; Spencer-Dene, B.; Diefenbacher, M.E.; Moore, C.; Kumar, M.S.; Murillo, M.M.; Gronroos, E.; et al. Requirement for interaction of PI3-kinase p110α with RAS in lung tumor maintenance. Cancer Cell 2013, 24, 617–630. [Google Scholar] [CrossRef] [PubMed]
- Murga, C.; Zohar, M.; Teramoto, H.; Gutkind, J.S. Rac1 and RhoG promote cell survival by the activation of PI3K and Akt, independently of their ability to stimulate JNK and NF-kappaB. Oncogene 2002, 21, 207–216. [Google Scholar] [CrossRef]
- Tolias, K.F.; Cantley, L.C.; Carpenter, C.L. Rho family GTPases bind to phosphoinositide kinases. J. Biol. Chem. 1995, 270, 17656–17659. [Google Scholar] [CrossRef] [PubMed]
- Cairns, R.A.; Harris, I.S.; Mak, T.W. Regulation of cancer cell metabolism. Nat. Rev. Cancer 2011, 11, 85–95. [Google Scholar] [CrossRef]
- DeBerardinis, R.J.; Lum, J.J.; Hatzivassiliou, G.; Thompson, C.B. The biology of cancer: Metabolic reprogramming fuels cell growth and proliferation. Cell Metab. 2008, 7, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Hoxhaj, G.; Manning, B.D. The PI3K-AKT network at the interface of oncogenic signalling and cancer metabolism. Nat. Rev. Cancer 2020, 20, 74–88. [Google Scholar] [CrossRef] [PubMed]
- Gross, A.; Katz, S.G. Non-apoptotic functions of BCL-2 family proteins. Cell Death Differ. 2017, 24, 1348–1358. [Google Scholar] [CrossRef]
- Alam, M.A.; Subhan, N.; Rahman, M.M.; Uddin, S.J.; Reza, H.M.; Sarker, S.D. Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanisms of action. Adv. Nutr. 2014, 5, 404–417. [Google Scholar] [CrossRef]
- Cheng, H.; Jiang, X.; Zhang, Q.; Ma, J.; Cheng, R.; Yong, H.; Shi, H.; Zhou, X.; Ge, L.; Gao, G. Naringin inhibits colorectal cancer cell growth by repressing the PI3K/AKT/mTOR signaling pathway. Exp. Ther. Med. 2020, 19, 3798–3804. [Google Scholar] [CrossRef]
- Raha, S.; Yumnam, S.; Hong, G.E.; Lee, H.J.; Saralamma, V.V.; Park, H.S.; Heo, J.D.; Lee, S.J.; Kim, E.H.; Kim, J.A.; et al. Naringin induces autophagy-mediated growth inhibition by downregulating the PI3K/Akt/mTOR cascade via activation of MAPK pathways in AGS cancer cells. Int. J. Oncol. 2015, 47, 1061–1069. [Google Scholar] [CrossRef] [PubMed]
- Purushotham, A.; Tian, M.; Belury, M.A. The citrus fruit flavonoid naringenin suppresses hepatic glucose production from Fao hepatoma cells. Mol. Nutr. Food Res. 2009, 53, 300–307. [Google Scholar] [CrossRef]
- Lin, D.C.; Meng, X.; Hazawa, M.; Nagata, Y.; Varela, A.M.; Xu, L.; Sato, Y.; Liu, L.Z.; Ding, L.W.; Sharma, A.; et al. The genomic landscape of nasopharyngeal carcinoma. Nat. Genet. 2014, 46, 866–871. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.G.; Tien, N.; Chang, Y.C.; Lin, M.L.; Chen, S.S. Oxidative Stress-Induced Unscheduled CDK1-Cyclin B1 Activity Impairs ER-Mitochondria-Mediated Bioenergetic Metabolism. Cells 2021, 10, 1280. [Google Scholar] [CrossRef]
- Reddy, S.P.; Raslan, W.F.; Gooneratne, S.; Kathuria, S.; Marks, J.E. Prognostic significance of keratinization in nasopharyngeal carcinoma. Am. J. Otolaryngol. 1995, 16, 103–108. [Google Scholar] [CrossRef]
- Shi, W.; Pataki, I.; MacMillan, C.; Pintilie, M.; Payne, D.; O’Sullivan, B.; Cummings, B.J.; Warde, P.; Liu, F.F. Molecular pathology parameters in human nasopharyngeal carcinoma. Cancer 2002, 94, 1997–2006. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.L.; Wang, S.G.; Chan, W.L.; Lee, C.H.; Wu, T.S.; Lin, M.L.; Chen, S.S. Impairment of Membrane Lipid Homeostasis by Bichalcone Analog TSWU-BR4 Attenuates Function of GRP78 in Regulation of the Oxidative Balance and Invasion of Cancer Cells. Cells 2020, 9, 371. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.L.; Lu, Y.C.; Chung, J.G.; Li, Y.C.; Wang, S.G.; Ng, S.H.; Wu, C.Y.; Su, H.L.; Chen, S.S. Aloe-emodin induces apoptosis of human nasopharyngeal carcinoma cells via caspase-8-mediated activation of the mitochondrial death pathway. Cancer Lett. 2010, 291, 46–58. [Google Scholar] [CrossRef] [PubMed]
- Smulow, J.B.; Glickman, I. An epithelial-like cell line in continuous culture from normal adult human gingiva. Proc. Soc. Exp. Biol. Med. Soc. Exp. Biol. Med. 1966, 121, 1294–1296. [Google Scholar] [CrossRef]
- Xia, Y.; Chu, W.; Qi, Q.; Xun, L. New insights into the QuikChange process guide the use of Phusion DNA polymerase for site-directed mutagenesis. Nucleic Acids Res. 2015, 43, e12. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.I.; Katsura, A.; Miyazono, K. A role of uridylation pathway for blockade of let-7 microRNA biogenesis by Lin28B. Cancer Sci. 2015, 106, 1174–1181. [Google Scholar] [CrossRef]
- Wang, Y.; Lu, M.; Xiong, L.; Fan, J.; Zhou, Y.; Li, H.; Peng, X.; Zhong, Z.; Wang, Y.; Huang, F.; et al. Drp1-mediated mitochondrial fission promotes renal fibroblast activation and fibrogenesis. Cell Death Dis. 2020, 11, 29. [Google Scholar] [CrossRef] [PubMed]
- Bulua, A.C.; Simon, A.; Maddipati, R.; Pelletier, M.; Park, H.; Kim, K.Y.; Sack, M.N.; Kastner, D.L.; Siegel, R.M. Mitochondrial reactive oxygen species promote production of proinflammatory cytokines and are elevated in TNFR1-associated periodic syndrome (TRAPS). J. Exp. Med. 2011, 208, 519–533. [Google Scholar] [CrossRef]
- Cui, F.; Zhou, Q.; Xiao, K.; Ma, S. The MicroRNA hsa-let-7g Promotes Proliferation and Inhibits Apoptosis in Lung Cancer by Targeting HOXB1. Yonsei Med. J. 2020, 61, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Wan, G.; Lim, Q.E.; Too, H.P. High-performance quantification of mature microRNAs by real-time RT-PCR using deoxyuridine-incorporated oligonucleotides and hemi-nested primers. RNA 2010, 16, 1436–1445. [Google Scholar] [CrossRef]
- Dobosz, E.; Wilamowski, M.; Lech, M.; Bugara, B.; Jura, J.; Potempa, J.; Koziel, J. MCPIP-1, Alias Regnase-1, Controls Epithelial Inflammation by Posttranscriptional Regulation of IL-8 Production. J. Innate Immun. 2016, 8, 564–578. [Google Scholar] [CrossRef]
- Jin, X.; Zhang, W.; Wang, Y.; Liu, J.; Hao, F.; Li, Y.; Tian, M.; Shu, H.; Dong, J.; Feng, Y.; et al. Pyruvate Kinase M2 Promotes the Activation of Dendritic Cells by Enhancing IL-12p35 Expression. Cell Rep. 2020, 31, 107690. [Google Scholar] [CrossRef] [PubMed]
- Mollinedo, F.; Gajate, C. Lipid rafts as signaling hubs in cancer cell survival/death and invasion: Implications in tumor progression and therapy: Thematic Review Series: Biology of Lipid Rafts. J. Lipid Res. 2020, 61, 611–635. [Google Scholar] [CrossRef]
- Brown, D.A.; London, E. Functions of lipid rafts in biological membranes. Annu. Rev. Cell Dev. Biol. 1998, 14, 111–136. [Google Scholar] [CrossRef]
- Drevot, P.; Langlet, C.; Guo, X.J.; Bernard, A.M.; Colard, O.; Chauvin, J.P.; Lasserre, R.; He, H.T. TCR signal initiation machinery is pre-assembled and activated in a subset of membrane rafts. EMBO J. 2002, 21, 1899–1908. [Google Scholar] [CrossRef]
- Chen, K.C.; Hsieh, I.C.; Hsi, E.; Wang, Y.S.; Dai, C.Y.; Chou, W.W.; Juo, S.H. Negative feedback regulation between microRNA let-7g and the oxLDL receptor LOX-1. J. Cell Sci. 2011, 124, 4115–4124. [Google Scholar] [CrossRef] [PubMed]
- Sung, W.W.; Chen, P.R.; Liao, M.H.; Lee, J.W. Enhanced aerobic glycolysis of nasopharyngeal carcinoma cells by Epstein-Barr virus latent membrane protein 1. Exp. Cell Res. 2017, 359, 94–100. [Google Scholar] [CrossRef]
- Zhang, M.X.; Hua, Y.J.; Wang, H.Y.; Zhou, L.; Mai, H.Q.; Guo, X.; Zhao, C.; Huang, W.L.; Hong, M.H.; Chen, M.Y. Long-term prognostic implications and therapeutic target role of hexokinase II in patients with nasopharyngeal carcinoma. Oncotarget 2016, 7, 21287–21297. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Reyes, I.; Chandel, N.S. Mitochondrial TCA cycle metabolites control physiology and disease. Nat. Commun. 2020, 11, 102. [Google Scholar] [CrossRef]
- Zhang, H. HIF-1 suppresses lipid catabolism to promote cancer progression. Mol. Cell. Oncol. 2015, 2, e980184. [Google Scholar] [CrossRef]
- Dervartanian, D.V.; Veeger, C. Studies on Succinate Dehydrogenase. I. Spectral Properties of the Purified Enzyme and Formation of Enzyme-Competitive Inhibitor Complexes. Biochim. Biophys. Acta 1964, 92, 233–247. [Google Scholar]
- Beloribi-Djefaflia, S.; Vasseur, S.; Guillaumond, F. Lipid metabolic reprogramming in cancer cells. Oncogenesis 2016, 5, e189. [Google Scholar] [CrossRef]
- Wu, C.W.; Wang, S.G.; Lee, C.H.; Chan, W.L.; Lin, M.L.; Chen, S.S. Enforced C-Src Activation Causes Compartmental Dysregulation of PI3K and PTEN Molecules in Lipid Rafts of Tongue Squamous Carcinoma Cells by Attenuating Rac1-Akt-GLUT-1-Mediated Sphingolipid Synthesis. Int. J. Mol. Sci. 2020, 21, 5812. [Google Scholar] [CrossRef] [PubMed]
- Xie, B.; Wang, S.; Jiang, N.; Li, J.J. Cyclin B1/CDK1-regulated mitochondrial bioenergetics in cell cycle progression and tumor resistance. Cancer Lett. 2019, 443, 56–66. [Google Scholar] [CrossRef]
- Lindqvist, A.; van Zon, W.; Karlsson Rosenthal, C.; Wolthuis, R.M. Cyclin B1-Cdk1 activation continues after centrosome separation to control mitotic progression. PLoS Biol. 2007, 5, e123. [Google Scholar] [CrossRef]
- Morgan, D.O. Principles of CDK regulation. Nature 1995, 374, 131–134. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.Y.; Chen, C.C.; Conte, D., Jr.; Moresco, J.J.; Chaves, D.A.; Mitani, S.; Yates, J.R., 3rd; Tsai, M.D.; Mello, C.C. A ribonuclease coordinates siRNA amplification and mRNA cleavage during RNAi. Cell 2015, 160, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.W.; Tchernyshyov, I.; Semenza, G.L.; Dang, C.V. HIF-1-mediated expression of pyruvate dehydrogenase kinase: A metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 2006, 3, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Papandreou, I.; Cairns, R.A.; Fontana, L.; Lim, A.L.; Denko, N.C. HIF-1 mediates adaptation to hypoxia by actively downregulating mitochondrial oxygen consumption. Cell Metab. 2006, 3, 187–197. [Google Scholar] [CrossRef]
- Singh, S.; Raza, W.; Parveen, S.; Meena, A.; Luqman, S. Flavonoid display ability to target microRNAs in cancer pathogenesis. Biochem. Pharmacol. 2021, 189, 114409. [Google Scholar] [CrossRef] [PubMed]
- Fan, W.; Shi, R.; Guan, M.; Chen, P.; Wu, H.; Su, W.; Wang, Y.; Li, P. The Effects of Naringenin on miRNA-mRNA Profiles in HepaRG Cells. Int. J. Mol. Sci. 2021, 22, 2292. [Google Scholar] [CrossRef]
- Rajabi, S.; Noori, S.; Ashrafi, M.R.; Movahed, M.A.; Farzaneh, S.; Zarghi, A. Naringenin enhances anti-proliferation effect of 1-ferrocenyl-3-(4-methylsulfonylphenyl) propen-1-one on two different cells via targeting calmodulin signaling pathway. Mol. Biol. Rep. 2022, 49, 1027–1036. [Google Scholar] [CrossRef] [PubMed]
- Qi, Z.; Kong, S.; Zhao, S.; Tang, Q. Naringenin inhibits human breast cancer cells (MDA-MB-231) by inducing programmed cell death, caspase stimulation, G2/M phase cell cycle arrest and suppresses cancer metastasis. Cell. Mol. Biol. 2021, 67, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Zeya, B.; Nafees, S.; Imtiyaz, K.; Uroog, L.; Fakhri, K.U.; Rizvi, M.M.A. Diosmin in combination with naringenin enhances apoptosis in colon cancer cells. Oncol. Rep. 2022, 47, 4. [Google Scholar] [CrossRef] [PubMed]
- Sargazi, M.L.; Juybari, K.B.; Tarzi, M.E.; Amirkhosravi, A.; Nematollahi, M.H.; Mirzamohammdi, S.; Mehrbani, M.; Mehrabani, M.; Mehrabani, M. Naringenin attenuates cell viability and migration of C6 glioblastoma cell line: A possible role of hedgehog signaling pathway. Mol. Biol. Rep. 2021, 48, 6413–6421. [Google Scholar] [CrossRef] [PubMed]
- Otsu, M.; Hiles, I.; Gout, I.; Fry, M.J.; Ruiz-Larrea, F.; Panayotou, G.; Thompson, A.; Dhand, R.; Hsuan, J.; Totty, N.; et al. Characterization of two 85 kd proteins that associate with receptor tyrosine kinases, middle-T/pp60c-src complexes, and PI3-kinase. Cell 1991, 65, 91–104. [Google Scholar] [CrossRef]
- Shepherd, P.R.; Withers, D.J.; Siddle, K. Phosphoinositide 3-kinase: The key switch mechanism in insulin signalling. Biochem. J. 1998, 333, 471–490. [Google Scholar] [CrossRef]
- Ueki, K.; Fruman, D.A.; Brachmann, S.M.; Tseng, Y.H.; Cantley, L.C.; Kahn, C.R. Molecular balance between the regulatory and catalytic subunits of phosphoinositide 3-kinase regulates cell signaling and survival. Mol. Cell. Biol. 2002, 22, 965–977. [Google Scholar] [CrossRef]
- Cheung, L.W.; Walkiewicz, K.W.; Besong, T.M.; Guo, H.; Hawke, D.H.; Arold, S.T.; Mills, G.B. Regulation of the PI3K pathway through a p85α monomer-homodimer equilibrium. eLife 2015, 4, e06866. [Google Scholar] [CrossRef]
- Wu, C.W.; Wang, S.G.; Lin, M.L.; Chen, S.S. Downregulation of miR-144 by triptolide enhanced p85α-PTEN complex formation causing S phase arrest of human nasopharyngeal carcinoma cells. Eur. J. Pharmacol. 2019, 855, 137–148. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, L.M.; Spangle, J.M.; Ohlson, C.E.; Cheng, H.; Roberts, T.M.; Cantley, L.C.; Zhao, J.J. PI3K-p110α mediates the oncogenic activity induced by loss of the novel tumor suppressor PI3K-p85α. Proc. Natl. Acad. Sci. USA 2017, 114, 7095–7100. [Google Scholar] [CrossRef] [PubMed]
- Murillo, M.M.; Rana, S.; Spencer-Dene, B.; Nye, E.; Stamp, G.; Downward, J. Disruption of the Interaction of RAS with PI 3-Kinase Induces Regression of EGFR-Mutant-Driven Lung Cancer. Cell Rep. 2018, 25, 3545–3553.e2. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hung, K.-C.; Tien, N.; Bau, D.-T.; Yao, C.-H.; Chen, C.-H.; Yang, J.-L.; Lin, M.-L.; Chen, S.-S. Let-7g Upregulation Attenuated the KRAS–PI3K–Rac1–Akt Axis-Mediated Bioenergetic Functions. Cells 2023, 12, 2313. https://doi.org/10.3390/cells12182313
Hung K-C, Tien N, Bau D-T, Yao C-H, Chen C-H, Yang J-L, Lin M-L, Chen S-S. Let-7g Upregulation Attenuated the KRAS–PI3K–Rac1–Akt Axis-Mediated Bioenergetic Functions. Cells. 2023; 12(18):2313. https://doi.org/10.3390/cells12182313
Chicago/Turabian StyleHung, Kuang-Chen, Ni Tien, Da-Tian Bau, Chun-Hsu Yao, Chan-Hung Chen, Jiun-Long Yang, Meng-Liang Lin, and Shih-Shun Chen. 2023. "Let-7g Upregulation Attenuated the KRAS–PI3K–Rac1–Akt Axis-Mediated Bioenergetic Functions" Cells 12, no. 18: 2313. https://doi.org/10.3390/cells12182313
APA StyleHung, K.-C., Tien, N., Bau, D.-T., Yao, C.-H., Chen, C.-H., Yang, J.-L., Lin, M.-L., & Chen, S.-S. (2023). Let-7g Upregulation Attenuated the KRAS–PI3K–Rac1–Akt Axis-Mediated Bioenergetic Functions. Cells, 12(18), 2313. https://doi.org/10.3390/cells12182313







