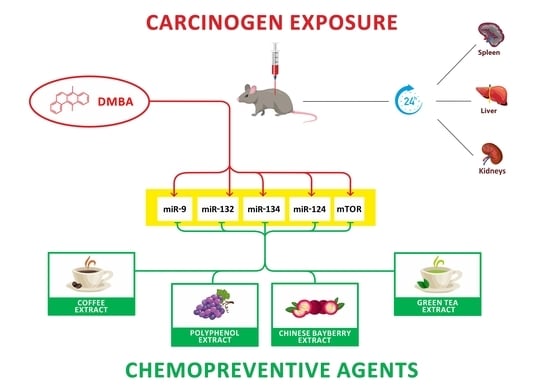
The Chemopreventive Effects of Polyphenols and Coffee, Based upon a DMBA Mouse Model with microRNA and mTOR Gene Expression Biomarkers
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Treatment
2.2. Collective Isolation of RNA
2.3. Polymerase Chain Reaction in Reverse Transcription (RT-PCR)
2.4. Calculations and Statistical Analyses
3. Results
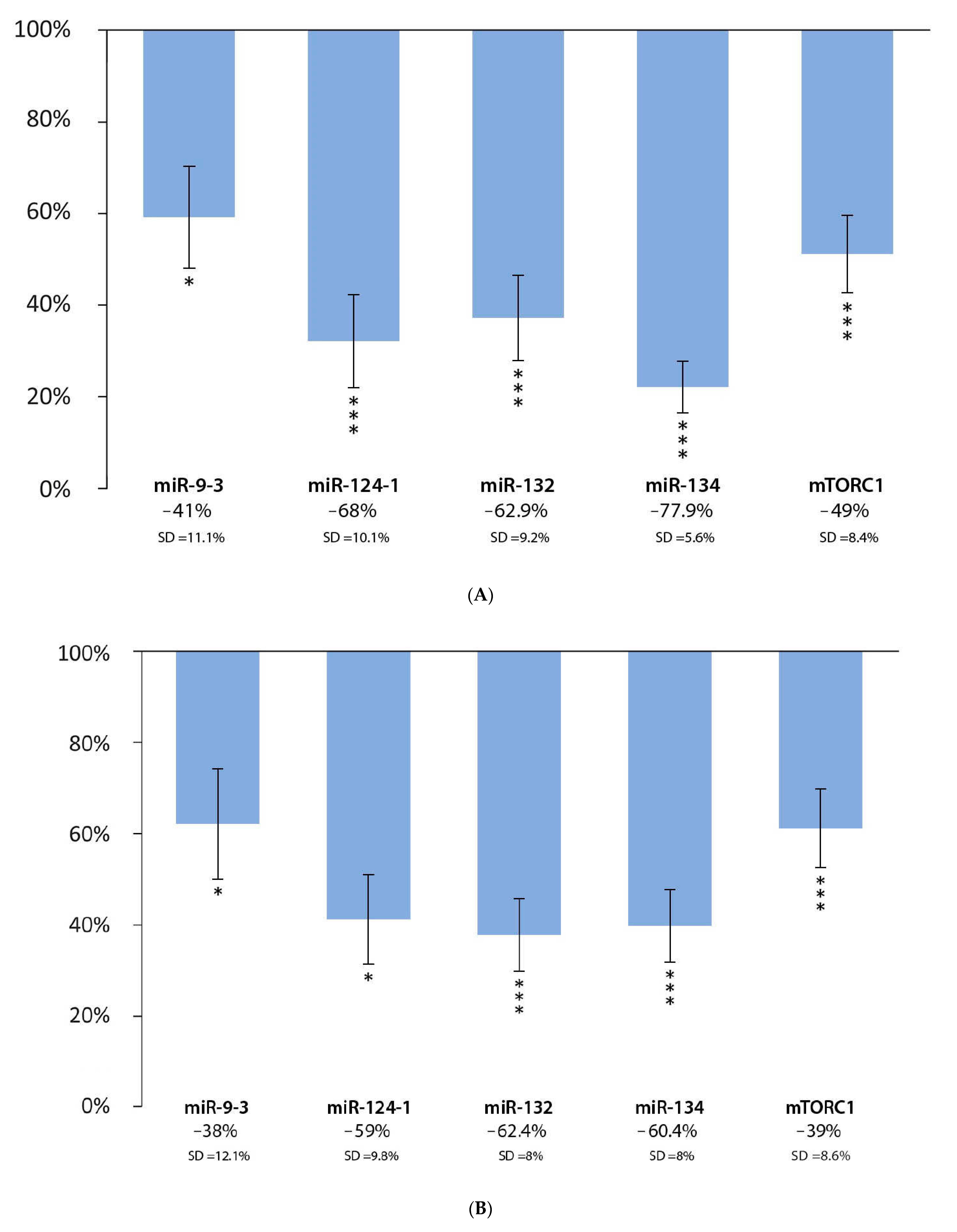
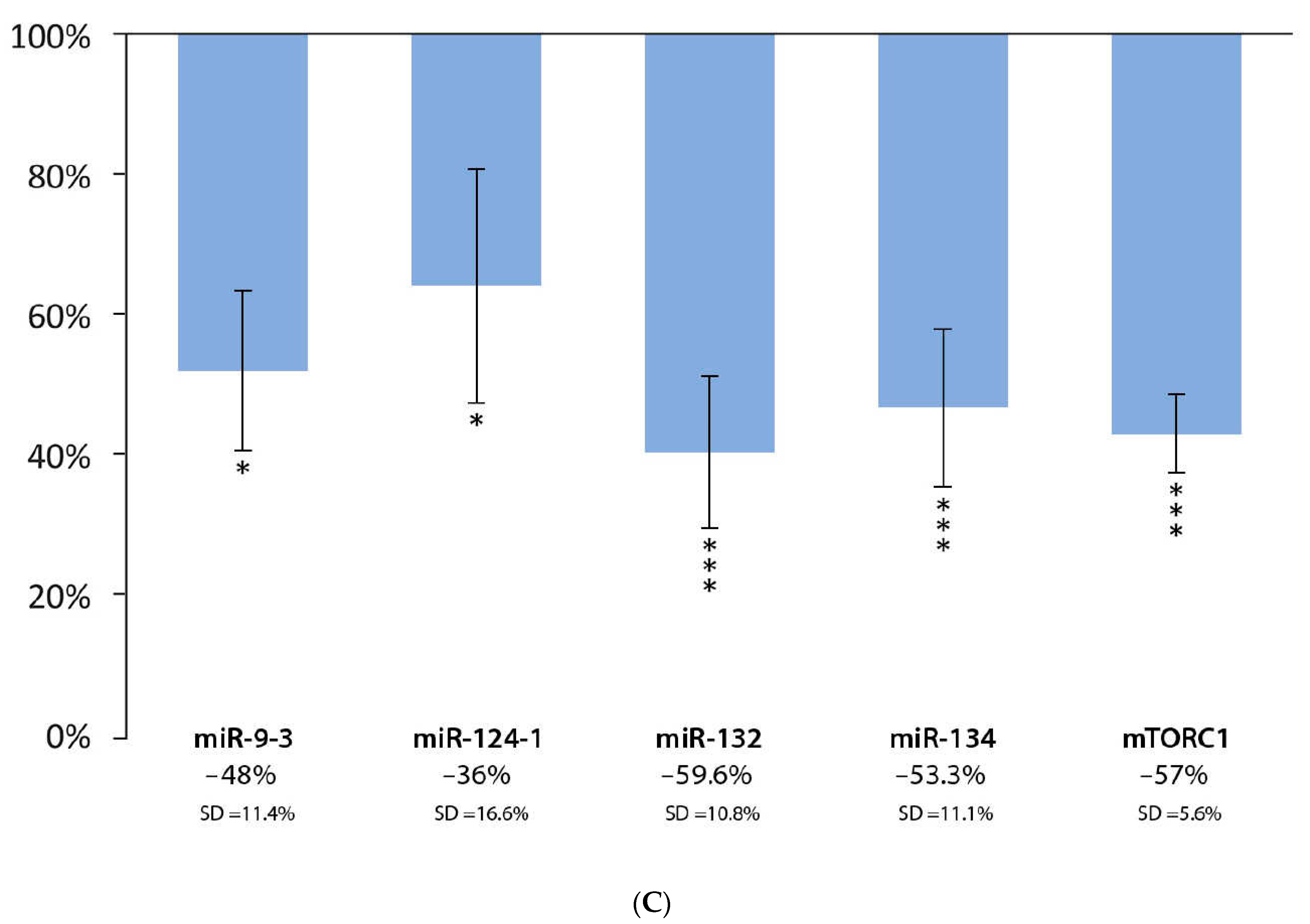
3.1. Effect of Flavonoid Extract and DMBA Treatment in the Liver, Spleen, and Kidneys, Compared to the DMBA Positive Control
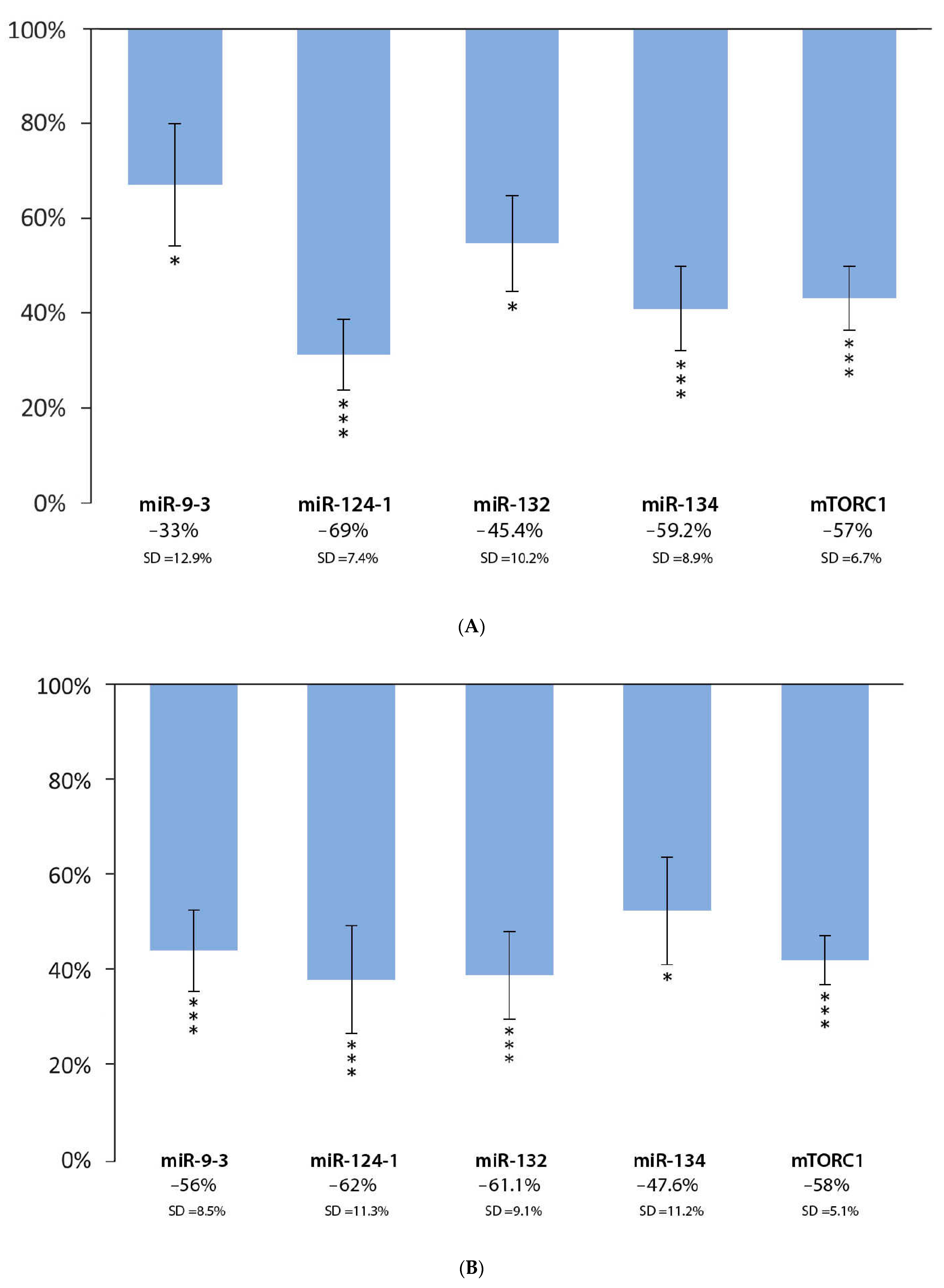
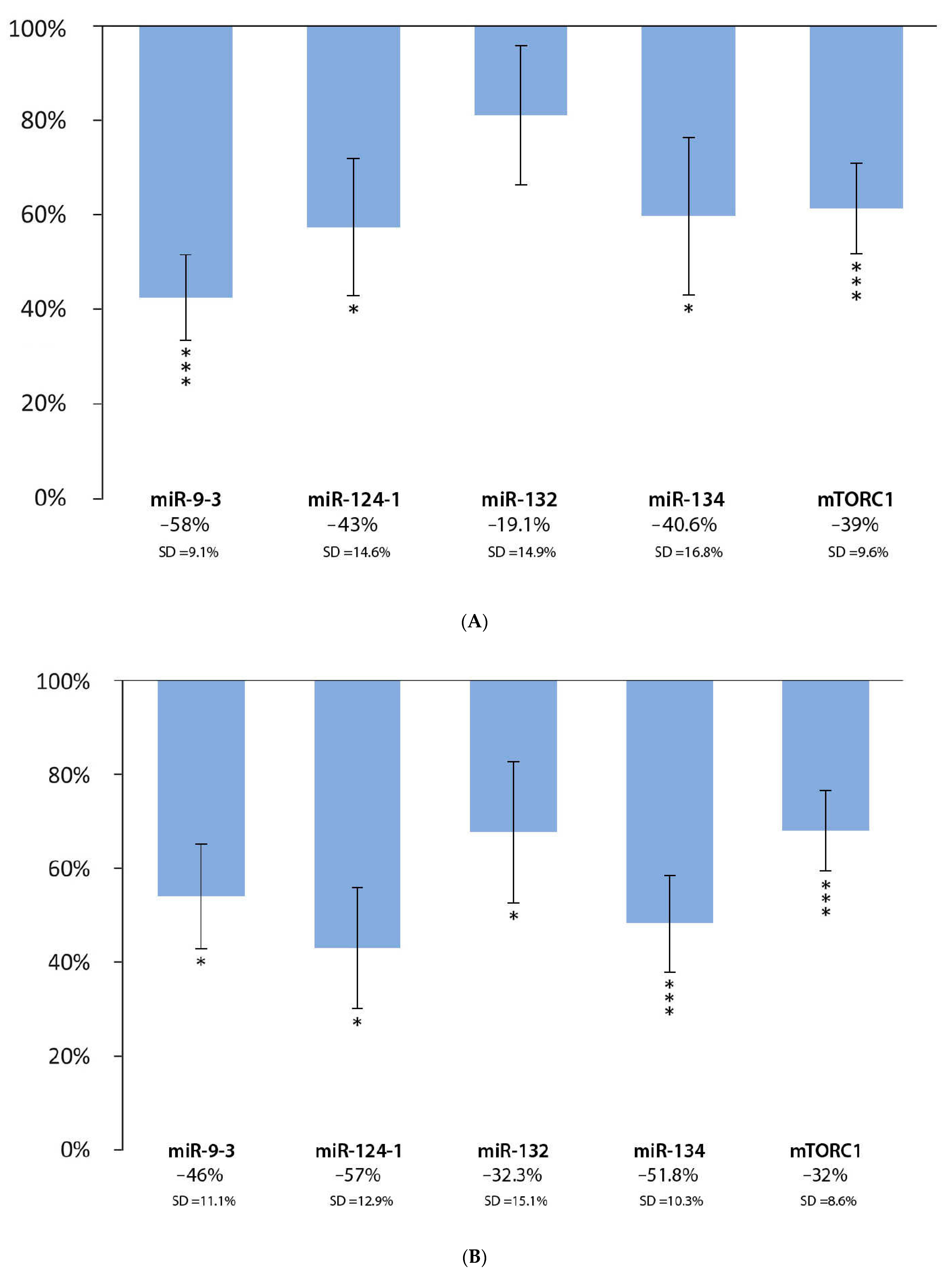
3.2. Effect of Green Tea Extract and DMBA Treatment in the Liver, Spleen, and Kidneys, Compared to the DMBA Positive Control
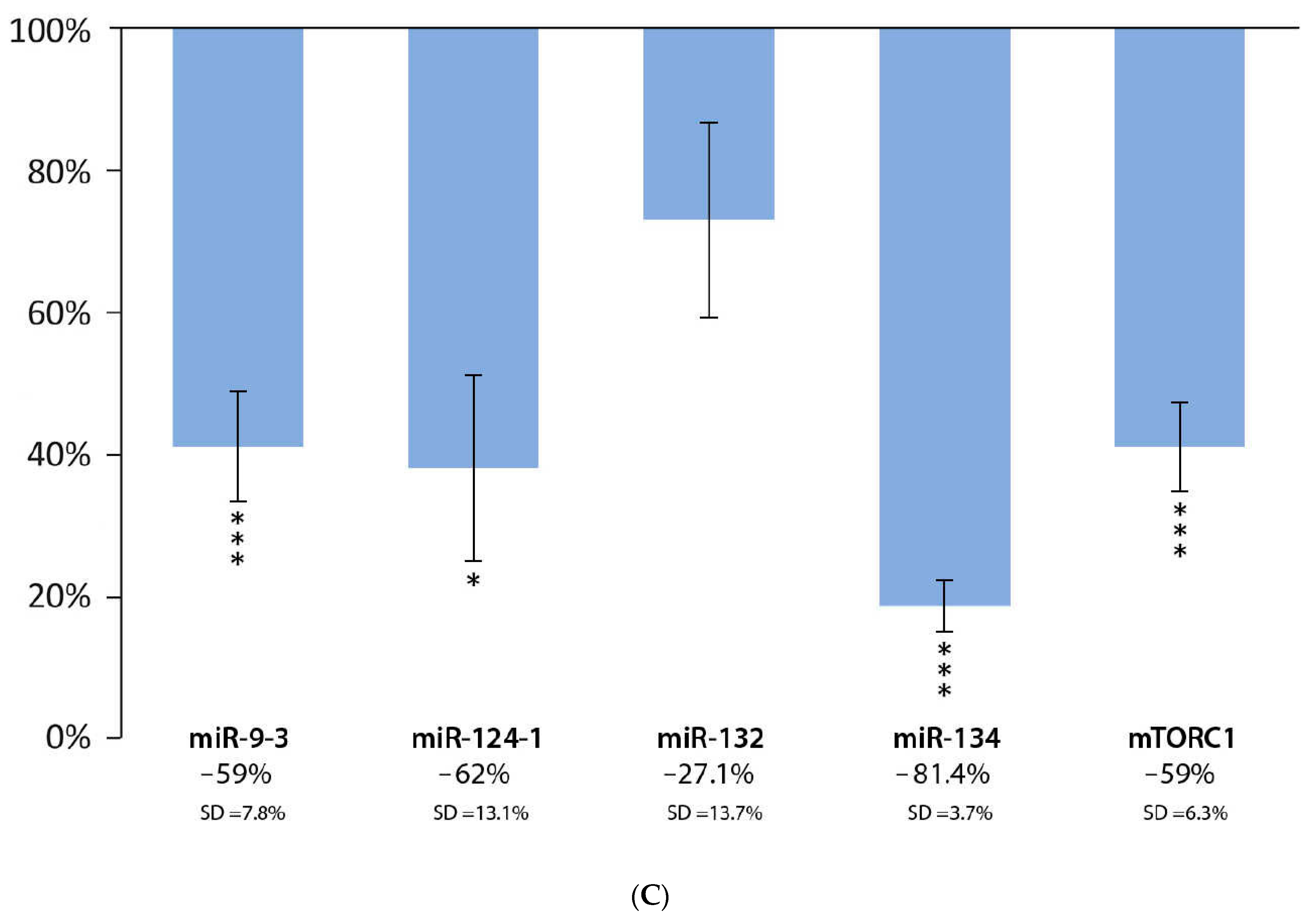
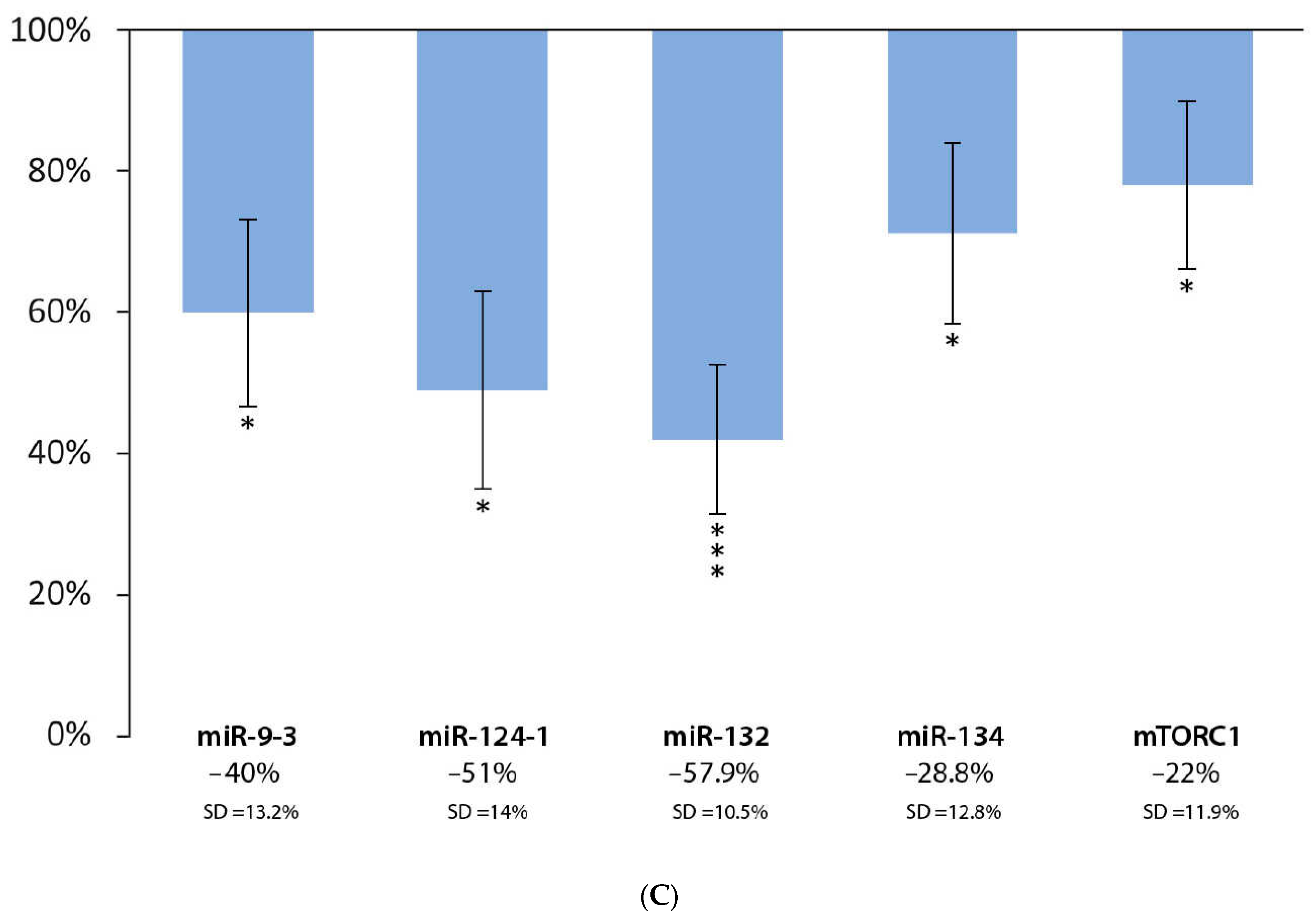
3.3. Effect of Chinese Bayberry Extract and DMBA Treatment in the Liver, Spleen, and Kidneys, Compared to the DMBA Positive Control
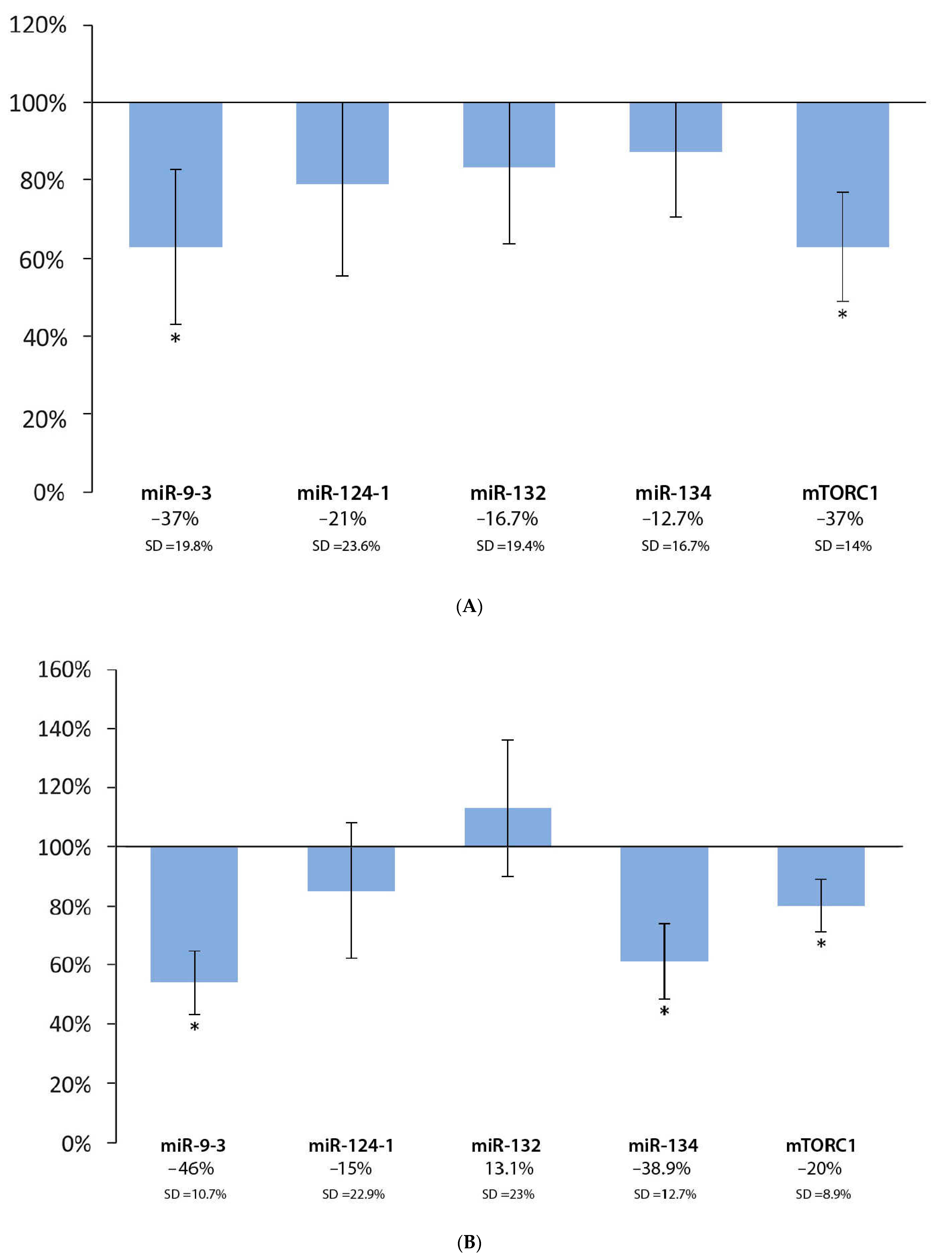
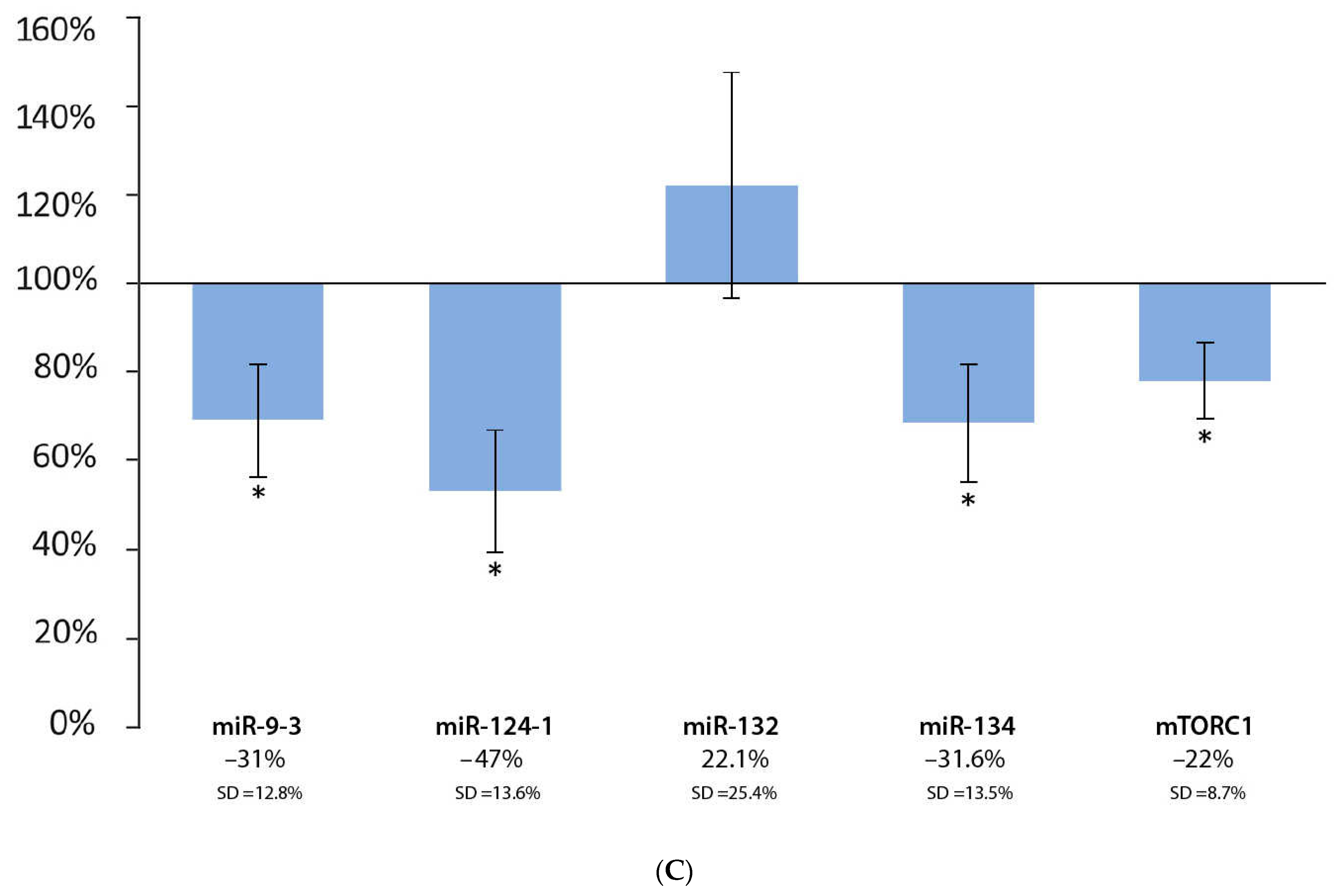
3.4. Effect of Coffee Extract and DMBA Treatment in the Liver, Spleen, and Kidneys, Compared to the DMBA Positive Control
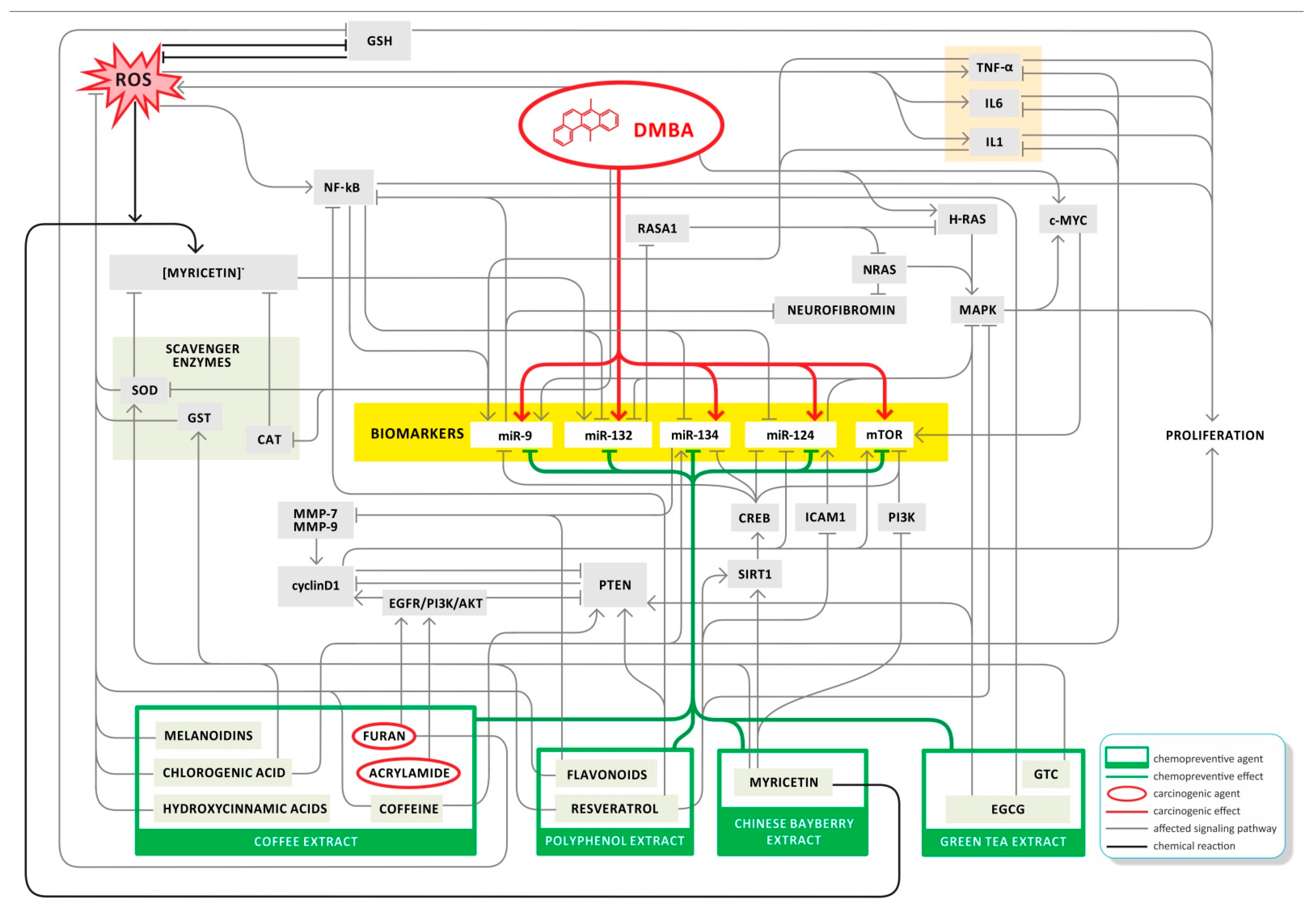
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Torre, L.A.; Siegel, R.L.; Ward, E.M.; Jemal, A. Global Cancer Incidence and Mortality Rates and Trends—An Update. Cancer Epidemiol. Biomark. Prev. 2016, 25, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Hashim, D.; Boffetta, P.; La Vecchia, C.; Rota, M.; Bertuccio, P.; Malvezzi, M.; Negri, E. The global decrease in cancer mortality: Trends and disparities. Ann. Oncol. 2016, 27, 926–933. [Google Scholar] [CrossRef] [PubMed]
- Danaei, G.; Vander Hoorn, S.; Lopez, A.D.; Murray, C.J.; Ezzati, M.; Comparative Risk Assessment Collaborating Group (Cancers). Causes of cancer in the world: Comparative risk assessment of nine behavioural and environmental risk factors. Lancet 2005, 366, 1784–1793. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424, Erratum in CA Cancer J. Clin. 2020, 70, 313. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Budán, F.; Szabó, I.; Varjas, T.; Nowrasteh, G.; Dávid, T.; Gergely, P.; Varga, Z.; Molnár, K.; Kádár, B.; Orsós, Z.; et al. Mixtures of Uncaria and Tabebuia extracts are potentially chemopreventive in CBA/Ca mice: A long-term experiment. Phytother. Res. 2011, 25, 493–500. [Google Scholar] [CrossRef]
- Varjas, T.; Nowrasteh, G.; Budán, F.; Nadasi, E.; Horváth, G.; Makai, S.; Gracza, T.; Cseh, J.; Ember, I. Chemopreventive effect of Panax ginseng. Phytother. Res. 2009, 23, 1399–1403. [Google Scholar] [CrossRef]
- Varjas, T.; Nowrasteh, G.; Budán, F.; Horváth, G.; Cseh, J.; Gyöngyi, Z.; Makai, S.; Ember, I. The effect of fenugreek on the gene expression of arachidonic acid metabolizing enzymes. Phytother. Res. 2011, 25, 221–227. [Google Scholar] [CrossRef]
- Szabo, L.; Molnar, R.; Tomesz, A.; Deutsch, A.; Darago, R.; Nowrasteh, G.; Varjas, T.; Nemeth, B.; Budan, F.; Kiss, I. The effects of flavonoids, green tea polyphenols and coffee on DMBA induced LINE-1 DNA hypomethylation. PLoS ONE 2021, 16, e0250157. [Google Scholar] [CrossRef]
- Szabo, L.; Molnar, R.; Tomesz, A.; Deutsch, A.; Darago, R.; Varjas, T.; Ritter, Z.; Szentpeteri, J.L.; Andreidesz, K.; Mathe, D.; et al. Olive Oil Improves While Trans Fatty Acids Further Aggravate the Hypomethylation of LINE-1 Retrotransposon DNA in an Environmental Carcinogen Model. Nutrients 2022, 14, 908. [Google Scholar] [CrossRef]
- Cui, Y.; Morgenstern, H.; Greenland, S.; Tashkin, D.P.; Mao, J.T.; Cai, L.; Cozen, W.; Mack, T.M.; Lu, Q.Y.; Zhang, Z.F. Dietary flavonoid intake and lung cancer—A population-based case-control study. Cancer 2008, 112, 2241–2248. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, B.B.; Bhardwaj, A.; Aggarwal, R.S.; Seeram, N.P.; Shishodia, S.; Takada, Y. Role of resveratrol in prevention and therapy of cancer: Preclinical and clinical studies. Anticancer Res. 2004, 24, 2783–2840. [Google Scholar] [PubMed]
- Narayanan, B.A. Chemopreventive agents alters global gene expression pattern: Predicting their mode of action and targets. Curr. Cancer Drug Targets 2006, 6, 711–727. [Google Scholar] [CrossRef] [PubMed]
- Landis-Piwowar, K.R.; Huo, C.; Chen, D.; Milacic, V.; Shi, G.; Chan, T.H.; Dou, Q.P. A novel prodrug of the green tea polyphenol (-)-epigallocatechin-3-gallate as a potential anticancer agent. Cancer Res. 2007, 67, 4303–4310. [Google Scholar] [CrossRef]
- Knekt, P.; Kumpulainen, J.; Järvinen, R.; Rissanen, H.; Heliövaara, M.; Reunanen, A.; Hakulinen, T.; Aromaa, A. Flavonoid intake and risk of chronic diseases. Am. J. Clin. Nutr. 2002, 76, 560–568. [Google Scholar] [CrossRef]
- Gaascht, F.; Dicato, M.; Diederich, M. Coffee provides a natural multitarget pharmacopeia against the hallmarks of cancer. Genes Nutr. 2015, 10, 51. [Google Scholar] [CrossRef]
- Hu, G.L.; Wang, X.; Zhang, L.; Qiu, M.H. The sources and mechanisms of bioactive ingredients in coffee. Food Funct. 2019, 10, 3113–3126. [Google Scholar] [CrossRef]
- Blank, I. Current status of acrylamide research in food: Measurement, safety assessment, and formation. Ann. N. Y. Acad. Sci. 2005, 1043, 30–40. [Google Scholar] [CrossRef]
- Xia, E.Q.; Deng, G.F.; Guo, Y.J.; Li, H.B. Biological activities of polyphenols from grapes. Int. J. Mol. Sci. 2010, 11, 622–646. [Google Scholar] [CrossRef]
- Smoliga, J.M.; Baur, J.A.; Hausenblas, H.A. Resveratrol and health—A comprehensive review of human clinical trials. Mol. Nutr. Food Res. 2011, 55, 1129–1141. [Google Scholar] [CrossRef]
- Nguyen, A.V.; Martinez, M.; Stamos, M.J.; Moyer, M.P.; Planutis, K.; Hope, C.; Holcombe, R.F. Results of a phase I pilot clinical trial examining the effect of plant-derived resveratrol and grape powder on Wnt pathway target gene expression in colonic mucosa and colon cancer. Cancer Manag. Res. 2009, 1, 25–37. [Google Scholar] [PubMed]
- Huang, Y.Q.; Lu, X.; Min, H.; Wu, Q.Q.; Shi, X.T.; Bian, K.Q.; Zou, X.P. Green tea and liver cancer risk: A meta-analysis of prospective cohort studies in Asian populations. Nutrition 2016, 32, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Iso, H.; Kubota, Y.; Japan Collaborative Cohort Study for Evaluation of Cancer. Nutrition and disease in the Japan Collaborative Cohort Study for Evaluation of Cancer (JACC). Asian Pac. J. Cancer Prev. 2007, 8, 35–80. [Google Scholar]
- Xing, M.; Cao, Y.; Ren, C.; Liu, Y.; Li, J.; Grierson, D.; Martin, C.; Sun, C.; Chen, K.; Xu, C.; et al. Elucidation of myricetin biosynthesis in Morella rubra of the Myricaceae. Plant J. 2021, 108, 411–425. [Google Scholar] [CrossRef] [PubMed]
- Ferrucci, L.M.; Cartmel, B.; Molinaro, A.M.; Leffell, D.J.; Bale, A.E.; Mayne, S.T. Tea, coffee, and caffeine and early-onset basal cell carcinoma in a case-control study. Eur. J. Cancer Prev. 2014, 23, 296–302. [Google Scholar] [CrossRef]
- Ember, I.; Kiss, I.; Pusztai, Z. Effect of 7,12-dimethylbenz(a)anthracene on onco/suppressor gene action in vivo: A short-term experiment. Anticancer Res. 1998, 18, 445–447. [Google Scholar] [PubMed]
- Stowers, S.J.; Anderson, M.W. Formation and persistence of benzo(a)pyrene metabolite-DNA adducts. Environ. Health Perspect. 1985, 62, 31–39. [Google Scholar] [CrossRef]
- Fuller-Espie, S.L.; Bearoff, F.M.; Minutillo, M.A. Exposure of coelomocytes from the earthworm Eisenia hortensis to Cu, Cd, and dimethylbenz[a]anthracene: An in vitro study examining reactive oxygen species production and immune response inhibition. Pedobiologia 2011, 54, S31–S36. [Google Scholar] [CrossRef]
- Storz, P. Reactive oxygen species in tumor progression. Front. Biosci. 2005, 10, 1881–1896. [Google Scholar] [CrossRef]
- Dreher, D.; Junod, A.F. Role of oxygen free radicals in cancer development. Eur. J. Cancer 1996, 32A, 30–38. [Google Scholar] [CrossRef]
- El-Sohemy, A.; Archer, M.C. Inhibition of N-methyl-N-nitrosourea- and 7,12-dimethylbenz[a] anthracene-induced rat mammary tumorigenesis by dietary cholesterol is independent of Ha-Ras mutations. Carcinogenesis 2000, 21, 827–831. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Perjési, P.; Bayer, Z.; Ember, I. Effect of E-2-(4′-methoxybenzylidene)-1-benzosuberone on the 7,12-dimethylbenz[alpha]anthracene-induced onco/suppressor gene action in vivo. I: A 24-hour experiment. Anticancer Res. 2000, 20, 475–481. [Google Scholar] [PubMed]
- Monostory, K.; Tamási, V.; Vereczkey, L.; Perjési, P. A study on CYP1A inhibitory action of E-2-(4′-methoxybenzylidene)-1-benzosuberone and some related chalcones and cyclic chalcone analogues. Toxicology 2003, 184, 203–210. [Google Scholar] [CrossRef]
- Budán, F.; Varjas, T.; Nowrasteh, G.; Varga, Z.; Boncz, I.; Cseh, J.; Prantner, I.; Antal, T.; Pázsit, E.; Gobel, G.; et al. Early modification of c-myc, Ha-ras and p53 expressions by N-methyl-N-nitrosourea. In Vivo 2008, 22, 793–797. [Google Scholar]
- Budán, F.; Varjas, T.; Nowrasteh, G.; Prantner, I.; Varga, Z.; Ember, A.; Cseh, J.; Gombos, K.; Pázsit, E.; Gobel, G.; et al. Early modification of c-myc, Ha-ras and p53 expressions by chemical carcinogens (DMBA, MNU). In Vivo 2009, 23, 591–598. [Google Scholar] [PubMed]
- Tomesz, A.; Szabo, L.; Molnar, R.; Deutsch, A.; Darago, R.; Mathe, D.; Budan, F.; Ghodratollah, N.; Varjas, T.; Nemeth, B.; et al. Effect of 7,12-Dimethylbenz(α)anthracene on the Expression of miR-330, miR-29a, miR-9-1, miR-9-3 and the mTORC1 Gene in CBA/Ca Mice. In Vivo 2020, 34, 2337–2343. [Google Scholar] [CrossRef] [PubMed]
- Tomesz, A.; Szabo, L.; Molnar, R.; Deutsch, A.; Darago, R.; Raposa, L.B.; Ghodratollah, N.; Varjas, T.; Nemeth, B.; Orsos, Z.; et al. Changes in miR-124-1, miR-212, miR-132, miR-134, and miR-155 Expression Patterns after 7,12-Dimethylbenz(a)anthracene Treatment in CBA/Ca Mice. Cells 2022, 11, 1020. [Google Scholar] [CrossRef]
- Molnar, R.; Szabo, L.; Tomesz, A.; Deutsch, A.; Darago, R.; Ghodratollah, N.; Varjas, T.; Nemeth, B.; Budan, F.; Kiss, I. In vivo effects of olive oil and trans-fatty acids on miR-134, miR-132, miR-124-1, miR-9-3 and mTORC1 gene expression in a DMBA-treated mouse model. PLoS ONE 2021, 16, e0246022. [Google Scholar] [CrossRef]
- Vannini, I.; Fanini, F.; Fabbri, M. MicroRNAs as lung cancer biomarkers and key players in lung carcinogenesis. Clin. Biochem. 2013, 46, 918–925, Erratum in Clin. Biochem. 2019, 63, 162Erratum in Clin. Biochem. 2019, 68, 58. [Google Scholar] [CrossRef]
- Luk, J.M.; Burchard, J.; Zhang, C.; Liu, A.M.; Wong, K.F.; Shek, F.H.; Lee, N.P.; Fan, S.T.; Poon, R.T.; Ivanovska, I.; et al. DLK1-DIO3 genomic imprinted microRNA cluster at 14q32.2 defines a stemlike subtype of hepatocellular carcinoma associated with poor survival. J. Biol. Chem. 2011, 286, 30706–30713. [Google Scholar] [CrossRef]
- Li, M.; Huo, X.; Davuljigari, C.B.; Dai, Q.; Xu, X. MicroRNAs and their role in environmental chemical carcinogenesis. Environ. Geochem. Health 2019, 41, 225–247. [Google Scholar] [CrossRef] [PubMed]
- Oliveto, S.; Mancino, M.; Manfrini, N.; Biffo, S. Role of microRNAs in translation regulation and cancer. World J. Biol. Chem. 2017, 8, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Middleton, J.; Jeon, Y.J.; Magee, P.; Veneziano, D.; Laganà, A.; Leong, H.S.; Sahoo, S.; Fassan, M.; Booton, R.; et al. KRAS induces lung tumorigenesis through microRNAs modulation. Cell Death Dis. 2018, 9, 219. [Google Scholar] [CrossRef] [PubMed]
- Shor, B.; Cavender, D.; Harris, C. A kinase-dead knock-in mutation in mTOR leads to early embryonic lethality and is dispensable for the immune system in heterozygous mice. BMC Immunol. 2009, 10, 28. [Google Scholar] [CrossRef]
- Uchida, S.; Hara, K.; Kobayashi, A.; Funato, H.; Hobara, T.; Otsuki, K.; Yamagata, H.; McEwen, B.S.; Watanabe, Y. Early life stress enhances behavioral vulnerability to stress through the activation of REST4-mediated gene transcription in the medial prefrontal cortex of rodents. J. Neurosci. 2010, 30, 15007–15018. [Google Scholar] [CrossRef]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef]
- Lu, S.C. Regulation of glutathione synthesis. Mol. Asp. Med. 2009, 30, 42–59. [Google Scholar] [CrossRef]
- Ciolino, H.P.; Daschner, P.J.; Yeh, G.C. Resveratrol inhibits transcription of CYP1A1 in vitro by preventing activation of the aryl hydrocarbon receptor. Cancer Res. 1998, 58, 5707–5712. [Google Scholar]
- Satoh, T.; Fujisawa, H.; Nakamura, A.; Takahashi, N.; Watanabe, K. Inhibitory Effects of Eight Green Tea Catechins on Cytochrome P450 1A2, 2C9, 2D6, and 3A4 Activities. J. Pharm. Pharm. Sci. 2016, 19, 188–197. [Google Scholar] [CrossRef]
- Santes-Palacios, R.; Marroquín-Pérez, A.L.; Hernández-Ojeda, S.L.; Camacho-Carranza, R.; Govezensky, T.; Espinosa-Aguirre, J.J. Human CYP1A1 inhibition by flavonoids. Toxicol. In Vitro 2020, 62, 104681. [Google Scholar] [CrossRef]
- Lieke, T.; Steinberg, C.E.W.; Pan, B.; Perminova, I.V.; Meinelt, T.; Knopf, K.; Kloas, W. Phenol-rich fulvic acid as a water additive enhances growth, reduces stress, and stimulates the immune system of fish in aquaculture. Sci. Rep. 2021, 11, 174. [Google Scholar] [CrossRef]
- Hajdrik, P.; Pályi, B.; Kis, Z.; Kovács, N.; Veres, D.S.; Szigeti, K.; Budán, F.; Hegedüs, I.; Kovács, T.; Bergmann, R.; et al. In Vitro Determination of Inhibitory Effects of Humic Substances Complexing Zn and Se on SARS-CoV-2 Virus Replication. Foods 2022, 11, 694. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.A.; Kasum, C.M. Dietary flavonoids: Bioavailability, metabolic effects, and safety. Annu. Rev. Nutr. 2002, 22, 19–34. [Google Scholar] [CrossRef]
- Capek, P.; Paulovičová, E.; Matulová, M.; Mislovičová, D.; Navarini, L.; Suggi-Liverani, F. Coffea arabica instant coffee—Chemical view and immunomodulating properties. Carbohydr. Polym. 2014, 103, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Kandaswami, C.; Middleton, E., Jr. Free radical scavenging and antioxidant activity of plant flavonoids. Adv. Exp. Med. Biol. 1994, 366, 351–376. [Google Scholar] [CrossRef] [PubMed]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956, Erratum in Free Radic. Biol. Med. 1996, 21, 417. [Google Scholar] [CrossRef]
- Middleton, E., Jr.; Kandaswami, C.; Theoharides, T.C. The effects of plant flavonoids on mammalian cells: Implications for inflammation, heart disease, and cancer. Pharmacol. Rev. 2000, 52, 673–751. [Google Scholar]
- Fiander, H.; Schneider, H. Dietary ortho phenols that induce glutathione S-transferase and increase the resistance of cells to hydrogen peroxide are potential cancer chemopreventives that act by two mechanisms: The alleviation of oxidative stress and the detoxification of mutagenic xenobiotics. Cancer Lett. 2000, 156, 117–124. [Google Scholar] [CrossRef]
- Gerszon JRodacka, A.; Puchała, M. Antioxidant Properties of Resveratrol and its Protective Effects in Neurodegenerative Diseases. Med. J. Cell Biol. 2014, 4, 97–117. [Google Scholar]
- Pastore, R.L.; Fratellone, P. Potential health benefits of green tea (Camellia sinensis): A narrative review. Explore 2006, 2, 531–539. [Google Scholar] [CrossRef]
- Liang, N.; Kitts, D.D. Role of Chlorogenic Acids in Controlling Oxidative and Inflammatory Stress Conditions. Nutrients 2015, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Yeung, F.; Hoberg, J.E.; Ramsey, C.S.; Keller, M.D.; Jones, D.R.; Frye, R.A.; Mayo, M.W. Modulation of NF-kappaB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J. 2004, 23, 2369–2380. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Liu, Y.; Xu, L.; Sha, C.; Zhang, H.; Xu, W. Resveratrol alleviates lipopolysaccharide-induced inflammation in PC-12 cells and in rat model. BMC Biotechnol. 2019, 19, 10. [Google Scholar] [CrossRef] [PubMed]
- Kandaswami, C.; Lee, L.T.; Lee, P.P.; Hwang, J.J.; Ke, F.C.; Huang, Y.T.; Lee, M.T. The antitumor activities of flavonoids. In Vivo 2005, 19, 895–909, Erratum in In Vivo 2007, 21, 553Erratum in In Vivo 2007, 21, 1172. [Google Scholar]
- Jung, H.Y.; Lee, D.; Ryu, H.G.; Choi, B.H.; Go, Y.; Lee, N.; Lee, D.; Son, H.G.; Jeon, J.; Kim, S.H.; et al. Myricetin improves endurance capacity and mitochondrial density by activating SIRT1 and PGC-1α. Sci. Rep. 2017, 7, 6237. [Google Scholar] [CrossRef]
- Zhao, Y.N.; Li, W.F.; Li, F.; Zhang, Z.; Dai, Y.D.; Xu, A.L.; Qi, C.; Gao, J.M.; Gao, J. Resveratrol improves learning and memory in normally aged mice through microRNA-CREB pathway. Biochem. Biophys. Res. Commun. 2013, 435, 597–602. [Google Scholar] [CrossRef]
- Shen, J.; Xu, L.; Qu, C.; Sun, H.; Zhang, J. Resveratrol prevents cognitive deficits induced by chronic unpredictable mild stress: Sirt1/miR-134 signalling pathway regulates CREB/BDNF expression in hippocampus in vivo and in vitro. Behav. Brain Res. 2018, 349, 1–7. [Google Scholar] [CrossRef]
- Ghosh, H.S.; McBurney, M.; Robbins, P.D. SIRT1 negatively regulates the mammalian target of rapamycin. PLoS ONE 2010, 5, e9199. [Google Scholar] [CrossRef]
- Chung, S.; Yao, H.; Caito, S.; Hwang, J.W.; Arunachalam, G.; Rahman, I. Regulation of SIRT1 in cellular functions: Role of polyphenols. Arch. Biochem. Biophys. 2010, 501, 79–90. [Google Scholar] [CrossRef]
- Sun, Y.; Ai, X.; Shen, S.; Lu, S. NF-κB-mediated miR-124 suppresses metastasis of non-small-cell lung cancer by targeting MYO10. Oncotarget 2015, 6, 8244–8254. [Google Scholar] [CrossRef]
- De la Rica, L.; García-Gómez, A.; Comet, N.R.; Rodríguez-Ubreva, J.; Ciudad, L.; Vento-Tormo, R.; Company, C.; Álvarez-Errico, D.; García, M.; Gómez-Vaquero, C.; et al. NF-κB-direct activation of microRNAs with repressive effects on monocyte-specific genes is critical for osteoclast differentiation. Genome Biol. 2015, 16, 2. [Google Scholar] [CrossRef] [PubMed]
- Shuang, T.; Wang, M.; Zhou, Y.; Shi, C.; Wang, D. NF-κB1, c-Rel, and ELK1 inhibit miR-134 expression leading to TAB1 upregulation in paclitaxel-resistant human ovarian cancer. Oncotarget 2017, 8, 24853–24868. [Google Scholar] [CrossRef] [PubMed]
- Bazzoni, F.; Rossato, M.; Fabbri, M.; Gaudiosi, D.; Mirolo, M.; Mori, L.; Tamassia, N.; Mantovani, A.; Cassatella, M.A.; Locati, M. Induction and regulatory function of miR-9 in human monocytes and neutrophils exposed to proinflammatory signals. Proc. Natl. Acad. Sci. USA 2009, 106, 5282–5287. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.C.; Li, S.J.; Li, D.J. Hsa-miR-134 suppresses non-small cell lung cancer (NSCLC) development through down-regulation of CCND1. Oncotarget 2016, 7, 35960–35978. [Google Scholar] [CrossRef]
- Merchant, N.; Nagaraju, G.P.; Rajitha, B.; Lammata, S.; Jella, K.K.; Buchwald, Z.S.; Lakka, S.S.; Ali, A.N. Matrix metalloproteinases: Their functional role in lung cancer. Carcinogenesis 2017, 38, 766–780. [Google Scholar] [CrossRef]
- Domitrović, R.; Cvijanović, O.; Šušnić, V.; Katalinić, N. Renoprotective mechanisms of chlorogenic acid in cisplatin-induced kidney injury. Toxicology 2014, 324, 98–107. [Google Scholar] [CrossRef]
- Shankar, S.; Singh, G.; Srivastava, R.K. Chemoprevention by resveratrol: Molecular mechanisms and therapeutic potential. Front. Biosci. 2007, 12, 4839–4854. [Google Scholar] [CrossRef]
- Liu, S.; Wang, X.J.; Liu, Y.; Cui, Y.F. PI3K/AKT/mTOR signaling is involved in (-)-epigallocatechin-3-gallate-induced apoptosis of human pancreatic carcinoma cells. Am. J. Chin. Med. 2013, 41, 629–642. [Google Scholar] [CrossRef]
- Miwa, S.; Sugimoto, N.; Shirai, T.; Hayashi, K.; Nishida, H.; Ohnari, I.; Takeuchi, A.; Yachie, A.; Tsuchiya, H. Caffeine activates tumor suppressor PTEN in sarcoma cells. Int. J. Oncol. 2011, 39, 465–472. [Google Scholar] [CrossRef]
- Radu, A.; Neubauer, V.; Akagi, T.; Hanafusa, H.; Georgescu, M.M. PTEN induces cell cycle arrest by decreasing the level and nuclear localization of cyclin D1. Mol. Cell. Biol. 2003, 23, 6139–6149. [Google Scholar] [CrossRef]
- Szpechcinski, A.; Florczuk, M.; Duk, K.; Zdral, A.; Rudzinski, S.; Bryl, M.; Czyzewicz, G.; Rudzinski, P.; Kupis, W.; Wojda, E.; et al. The expression of circulating miR-504 in plasma is associated with EGFR mutation status in non-small-cell lung carcinoma patients. Cell Mol. Life Sci. 2019, 76, 3641–3656. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Qiu, J.; Wang, X.; Lu, Z.; Wang, D.; Feng, H.; Li, X.; Liu, Q.; Pan, H.; Han, X.; et al. Identification of microRNA-124 in regulation of Hepatocellular carcinoma through BIRC3 and the NF-κB pathway. J. Cancer 2018, 9, 3006–3015. [Google Scholar] [CrossRef]
- Gu, W.; Yao, L.; Li, L.; Zhang, J.; Place, A.T.; Minshall, R.D.; Liu, G. ICAM-1 regulates macrophage polarization by suppressing MCP-1 expression via miR-124 upregulation. Oncotarget 2017, 8, 111882–111901. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, V.; Mandal, A.K.A. Next-Generation Sequencing Reveals the Role of Epigallocatechin-3-Gallate in Regulating Putative Novel and Known microRNAs Which Target the MAPK Pathway in Non-Small-Cell Lung Cancer A549 Cells. Molecules 2019, 24, 368. [Google Scholar] [CrossRef] [PubMed]
- Hall, M.N. mTOR-what does it do? Transpl. Proc. 2008, 40 (Suppl. 10), S5–S8. [Google Scholar] [CrossRef]
- Walker, E.H.; Pacold, M.E.; Perisic, O.; Stephens, L.; Hawkins, P.T.; Wymann, M.P.; Williams, R.L. Structural determinants of phosphoinositide 3-kinase inhibition by wortmannin, LY294002, quercetin, myricetin, and staurosporine. Mol. Cell 2000, 6, 909–919. [Google Scholar] [CrossRef]
- Chappell, W.H.; Steelman, L.S.; Long, J.M.; Kempf, R.C.; Abrams, S.L.; Franklin, R.A.; Bäsecke, J.; Stivala, F.; Donia, M.; Fagone, P.; et al. Ras/Raf/MEK/ERK and PI3K/PTEN/Akt/mTOR inhibitors: Rationale and importance to inhibiting these pathways in human health. Oncotarget 2011, 2, 135–164. [Google Scholar] [CrossRef]
- Masliah-Planchon, J.; Garinet, S.; Pasmant, E. RAS-MAPK pathway epigenetic activation in cancer: miRNAs in action. Oncotarget 2016, 7, 38892–38907. [Google Scholar] [CrossRef]
- Liu, C.W.; Sung, H.C.; Lin, S.R.; Wu, C.W.; Lee, C.W.; Lee, I.T.; Yang, Y.F.; Yu, I.S.; Lin, S.W.; Chiang, M.H.; et al. Resveratrol attenuates ICAM-1 expression and monocyte adhesiveness to TNF-α-treated endothelial cells: Evidence for an anti-inflammatory cascade mediated by the miR-221/222/AMPK/p38/NF-κB pathway. Sci. Rep. 2017, 7, 44689. [Google Scholar] [CrossRef]
- Pathria, G.; Verma, S.; Yin, J.; Scott, D.A.; Ronai, Z.A. MAPK signaling regulates c-MYC for melanoma cell adaptation to asparagine restriction. EMBO Rep. 2021, 22, e51436. [Google Scholar] [CrossRef]
- Zhao, Q.; Assimopoulou, A.N.; Klauck, S.M.; Damianakos, H.; Chinou, I.; Kretschmer, N.; Rios, J.L.; Papageorgiou, V.P.; Bauer, R.; Efferth, T. Inhibition of c-MYC with involvement of ERK/JNK/MAPK and AKT pathways as a novel mechanism for shikonin and its derivatives in killing leukemia cells. Oncotarget 2015, 6, 38934–38951. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Wang, S.; Yang, B.; Zhu, L.; Yin, B.; Chao, T.; Zhao, J.; Yuan, J.; Qiang, B.; Peng, X. The CREB-miR-9 negative feedback minicircuitry coordinates the migration and proliferation of glioma cells. PLoS ONE 2012, 7, e49570. [Google Scholar] [CrossRef] [PubMed]
- Aires, V.; Delmas, D.; Djouadi, F.; Bastin, J.; Cherkaoui-Malki, M.; Latruffe, N. Resveratrol-Induced Changes in MicroRNA Expression in Primary Human Fibroblasts Harboring Carnitine-Palmitoyl Transferase-2 Gene Mutation, Leading to Fatty Acid Oxidation Deficiency. Molecules 2017, 23, 7. [Google Scholar] [CrossRef]
- Laughton, M.J.; Halliwell, B.; Evans, P.J.; Hoult, J.R. Antioxidant and pro-oxidant actions of the plant phenolics quercetin, gossypol and myricetin. Effects on lipid peroxidation, hydroxyl radical generation and bleomycin-dependent damage to DNA. Biochem. Pharmacol. 1989, 38, 2859–2865. [Google Scholar] [CrossRef]
- Stevens, B.; Algar, B.E. Photoperoxidation of unsaturated organic molecules. II. Autoperoxidation of aromatic hydrocarbons. J. Phys. Chem. 1968, 72, 3468–3474. [Google Scholar] [CrossRef]
- Ma, X.; Deng, D.; Chen, W. Inhibitors and Activators of SOD, GSH-Px, and CAT. In Enzyme Inhibitors and Activators; Senturk, M., Ed.; IntechOpen: London, UK; Available online: https://www.intechopen.com/books/enzyme-inhibitors-and-activators/inhibitors-and-activators-of-sod-gsh-px-and-cat (accessed on 29 March 2017). [CrossRef]
- Zhou, Y.; Li, K.S.; Liu, L.; Li, S.L. MicroRNA 132 promotes oxidative stress induced pyroptosis by targeting sirtuin 1 in myocardial ischaemia reperfusion injury. Int. J. Mol. Med. 2020, 45, 1942–1950. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, P.; Xu, C.; Shan, X.; Feng, Q. Acrylamide induces HepG2 cell proliferation through upregulation of miR-21 expression. J. Biomed. Res. 2019, 33, 181–191. [Google Scholar] [CrossRef]
- Chen, T.; Mally, A.; Ozden, S.; Chipman, J.K. Low doses of the carcinogen furan alter cell cycle and apoptosis gene expression in rat liver independent of DNA methylation. Environ. Health Perspect. 2010, 118, 1597–1602. [Google Scholar] [CrossRef]
- Peterson, L.A. Reactive metabolites in the biotransformation of molecules containing a furan ring. Chem. Res. Toxicol. 2013, 26, 6–25. [Google Scholar] [CrossRef]
- Pikarsky, E.; Porat, R.M.; Stein, I.; Abramovitch, R.; Amit, S.; Kasem, S.; Gutkovich-Pyest, E.; Urieli-Shoval, S.; Galun, E.; Ben-Neriah, Y. NF-kappaB functions as a tumour promoter in inflammation-associated cancer. Nature 2004, 431, 461–466. [Google Scholar] [CrossRef]
- Pan, J.Y.; Zhang, F.; Sun, C.C.; Li, S.J.; Li, G.; Gong, F.Y.; Bo, T.; He, J.; Hua, R.X.; Hu, W.D.; et al. miR-134: A Human Cancer Suppressor? Mol. Ther. Nucleic Acids 2017, 6, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Park, E.J.; Pezzuto, J.M. The pharmacology of resveratrol in animals and humans. Biochim. Biophys. Acta 2015, 1852, 1071–1113. [Google Scholar] [CrossRef] [PubMed]
| Group | Ip. DMBA | Daily Dose (of 1 Animal) | Producer | Product and Main Components | Latin/Scientific Names | Quantity |
|---|---|---|---|---|---|---|
| Negative Control (n = 6) | - | |||||
| DMBA Control (n = 6) | + | |||||
| Flavonoid Extract + DMBA (n = 6) | + | 30 mg | Slimbios Ltd. | FruitCafeTM | ||
| Common grape vine seed, peel | Vitis vinifera ‘Cabernet Sauvignon’ | 20 g/100 mL | ||||
| Erithritol | (2R,3S)-Butane-1,2,3,4-tetrol | 12 g/100 mL | ||||
| Resveratrol | trans-3,5,4′-Trihydroxystilbenetrans-3,5,4′-trihydroxystilbene | 4 g/100 mL | ||||
| Blackberry ‘thornfree’ seed, peel | Rubus fruticosus “thorn-free” | 2 g/100 mL | ||||
| Blackcurrant seed, peel | Ribes nigrum | 2 g/100 mL | ||||
| Total polyphenol | 4000–5000 mg/100 mL | |||||
| Green Tea Extract + DMBA (n = 6) | + | 4 mg | Xi’an Longze Biotechnology Co. Ltd. | Green tea | Camellia sinensis | |
| Total polyphenol | 98.53% | |||||
| Total catechins | 80.42% | |||||
| EGCG | Epigallocatechin-3-gallate | 50.45% | ||||
| Caffeine | 1,3,7-Trimethylxanthine | 0.28% | ||||
| CoffeeExtract + DMBA (n = 6) | + | 30 mg | Xi’an Longze Biotechnology Co. Ltd. | Coffee arabica | ||
| Chlorogenic acid | 3-Caffeoylquinic acid | 5.03% | ||||
| Caffeine | 1,3,7-Trimethylxanthine | 1.21% | ||||
| Chinese Bayberry Extract + DMBA (n = 6) | + | 2.5 mg | Xi’an Longze Biotechnology Co. Ltd. | Chinese bayberry | Myrica rubra | |
| Myricetin | 3,5,7,3′,4′,5′-Hexahydroxyflavone | 80.42% |
| miR | Forward | Reverse |
|---|---|---|
| miR-134 | TGTGACTGGTTGACCAGAGG | GTGACTAGGTGGCCCACAG |
| miR-132 | ACCGTGGCTTTCGATTGTTA | CGACCATGGCTGTAGACTGTT |
| miR-124-1 | TCTCTCTCCGTGTTCACAGC | ACCGCGTGCCTTAATTGTAT |
| miR-9-3 | GCCCGTTTCTCTCTTTGGTT | TCTAGCTTTATGACGGCTCTGTGG |
| mTORC1 | AAGGCCTGATGGGATTTGG | TGTCAAGTACACGGGGCAAG |
| mouse U6 | CGCTTCGGCAGCACATATAC | TTCACGAATTTGCGTGTCAT |
| miR-9-3 | miR-124-1 | miR-132 | miR-134 | mTORC1 | |
|---|---|---|---|---|---|
| Polyphenol extract | |||||
| Liver | * | *** | *** | *** | *** |
| Spleen | * | * | *** | *** | *** |
| Kidneys | *** | * | D | *** | *** |
| Green tea | |||||
| Liver | * | *** | * | *** | *** |
| Spleen | *** | *** | *** | * | *** |
| Kidneys | * | * | *** | *** | *** |
| Chinese bayberry | |||||
| Liver | *** | * | D | * | *** |
| Spleen | * | * | * | *** | *** |
| Kidneys | * | * | *** | * | * |
| Coffee extract | |||||
| Liver | * | O | O | O | * |
| Spleen | * | O | I | * | * |
| Kidneys | * | * | I | * | * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Molnar, R.; Szabo, L.; Tomesz, A.; Deutsch, A.; Darago, R.; Raposa, B.L.; Ghodratollah, N.; Varjas, T.; Nemeth, B.; Orsos, Z.; et al. The Chemopreventive Effects of Polyphenols and Coffee, Based upon a DMBA Mouse Model with microRNA and mTOR Gene Expression Biomarkers. Cells 2022, 11, 1300. https://doi.org/10.3390/cells11081300
Molnar R, Szabo L, Tomesz A, Deutsch A, Darago R, Raposa BL, Ghodratollah N, Varjas T, Nemeth B, Orsos Z, et al. The Chemopreventive Effects of Polyphenols and Coffee, Based upon a DMBA Mouse Model with microRNA and mTOR Gene Expression Biomarkers. Cells. 2022; 11(8):1300. https://doi.org/10.3390/cells11081300
Chicago/Turabian StyleMolnar, Richard, Laszlo Szabo, Andras Tomesz, Arpad Deutsch, Richard Darago, Bence L. Raposa, Nowrasteh Ghodratollah, Timea Varjas, Balazs Nemeth, Zsuzsanna Orsos, and et al. 2022. "The Chemopreventive Effects of Polyphenols and Coffee, Based upon a DMBA Mouse Model with microRNA and mTOR Gene Expression Biomarkers" Cells 11, no. 8: 1300. https://doi.org/10.3390/cells11081300
APA StyleMolnar, R., Szabo, L., Tomesz, A., Deutsch, A., Darago, R., Raposa, B. L., Ghodratollah, N., Varjas, T., Nemeth, B., Orsos, Z., Pozsgai, E., Szentpeteri, J. L., Budan, F., & Kiss, I. (2022). The Chemopreventive Effects of Polyphenols and Coffee, Based upon a DMBA Mouse Model with microRNA and mTOR Gene Expression Biomarkers. Cells, 11(8), 1300. https://doi.org/10.3390/cells11081300