Insulin-Degrading Enzyme, an Under-Estimated Potential Target to Treat Cancer?
Abstract
1. Introduction
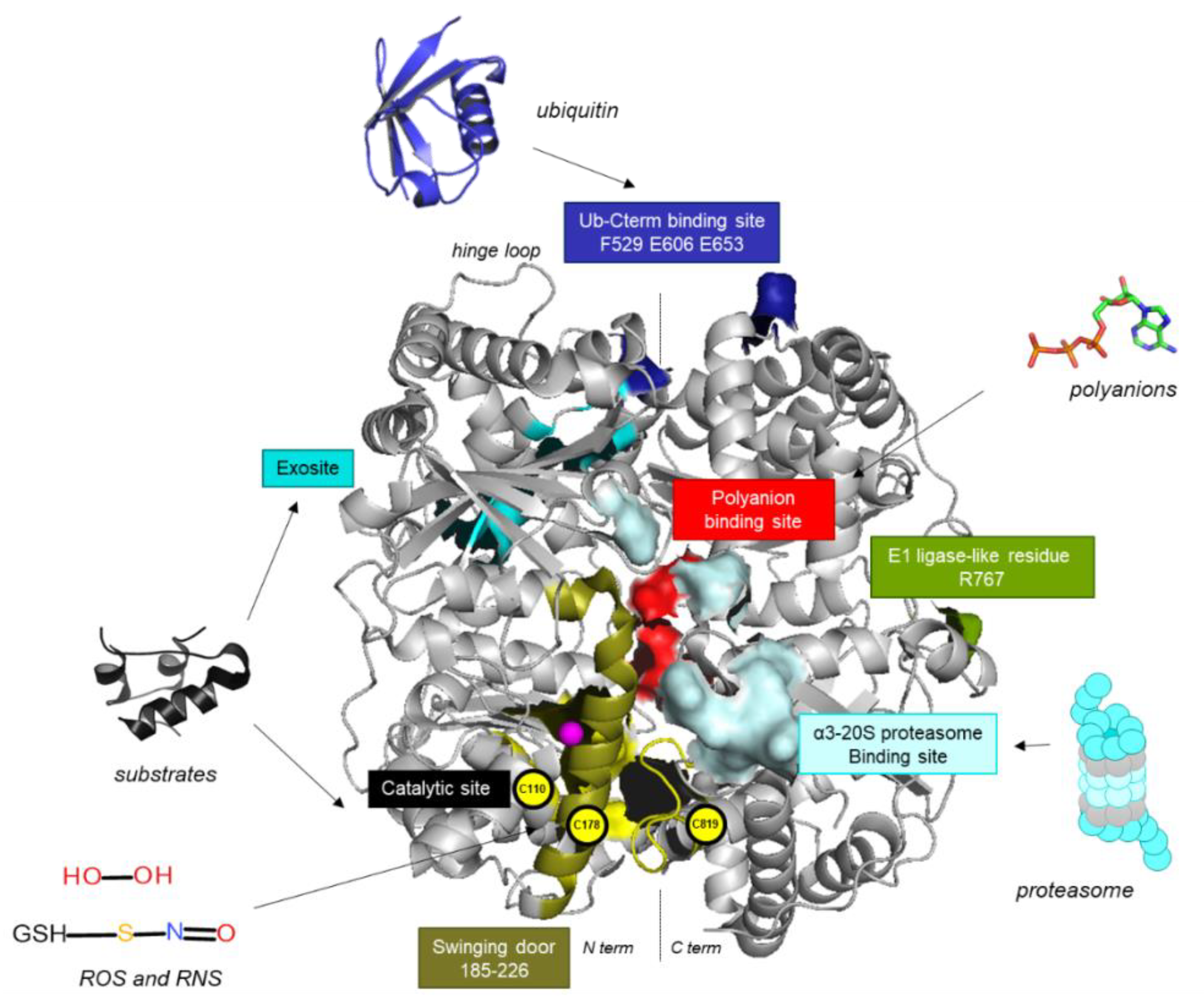
2. An Atypical Structure at the Basis of IDE’s Multiple Roles
3. A Long List of Substrates
4. IDE: A Chaperone-Like Protein?
5. IDE’s Close Link with the Proteasome
6. Expression of IDE in Human Cancers
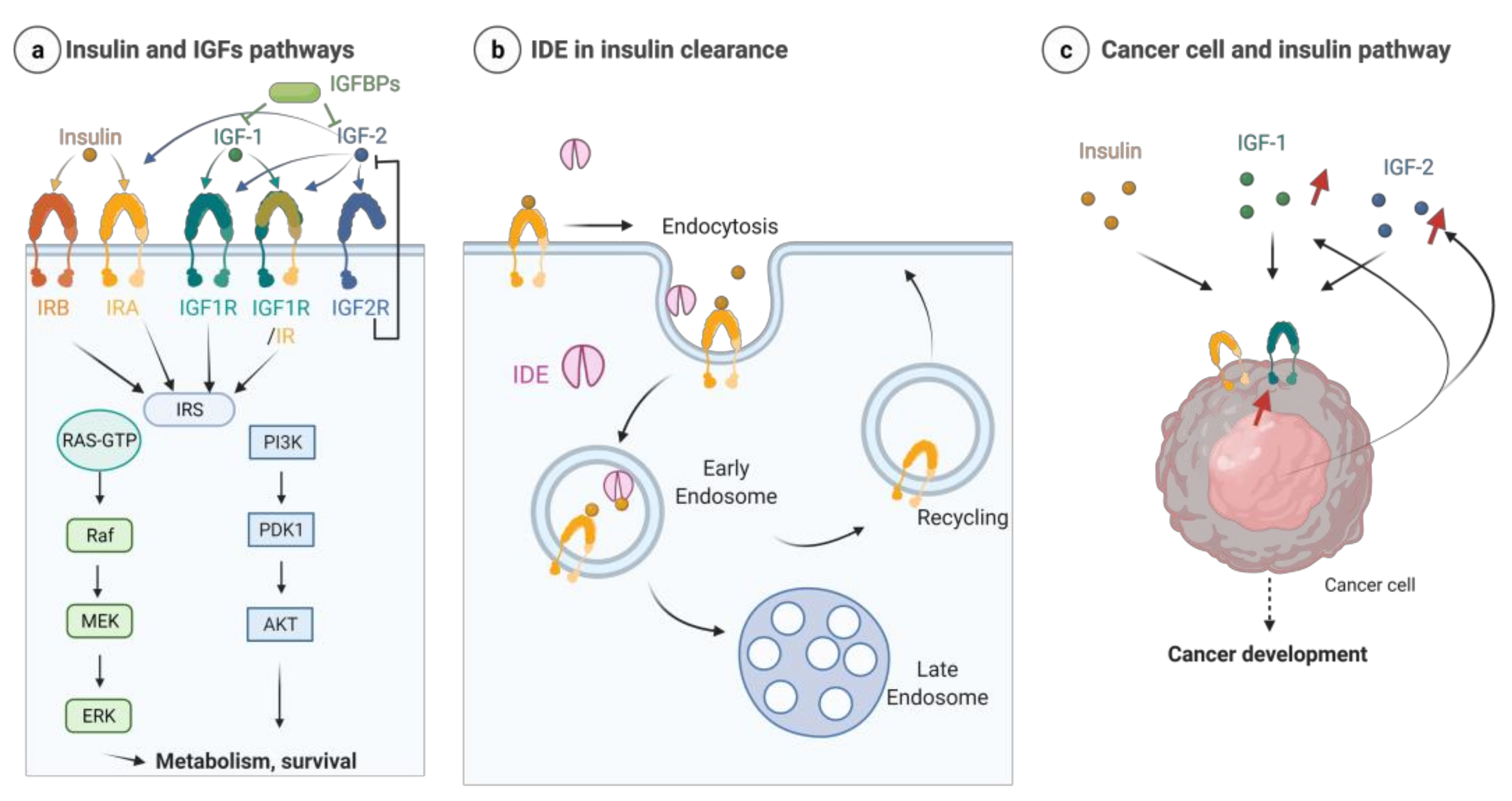
7. IDE and the Insulin/Insulin-Growth Factor Signaling (IIS) Pathway
8. IDE and Sex Hormones
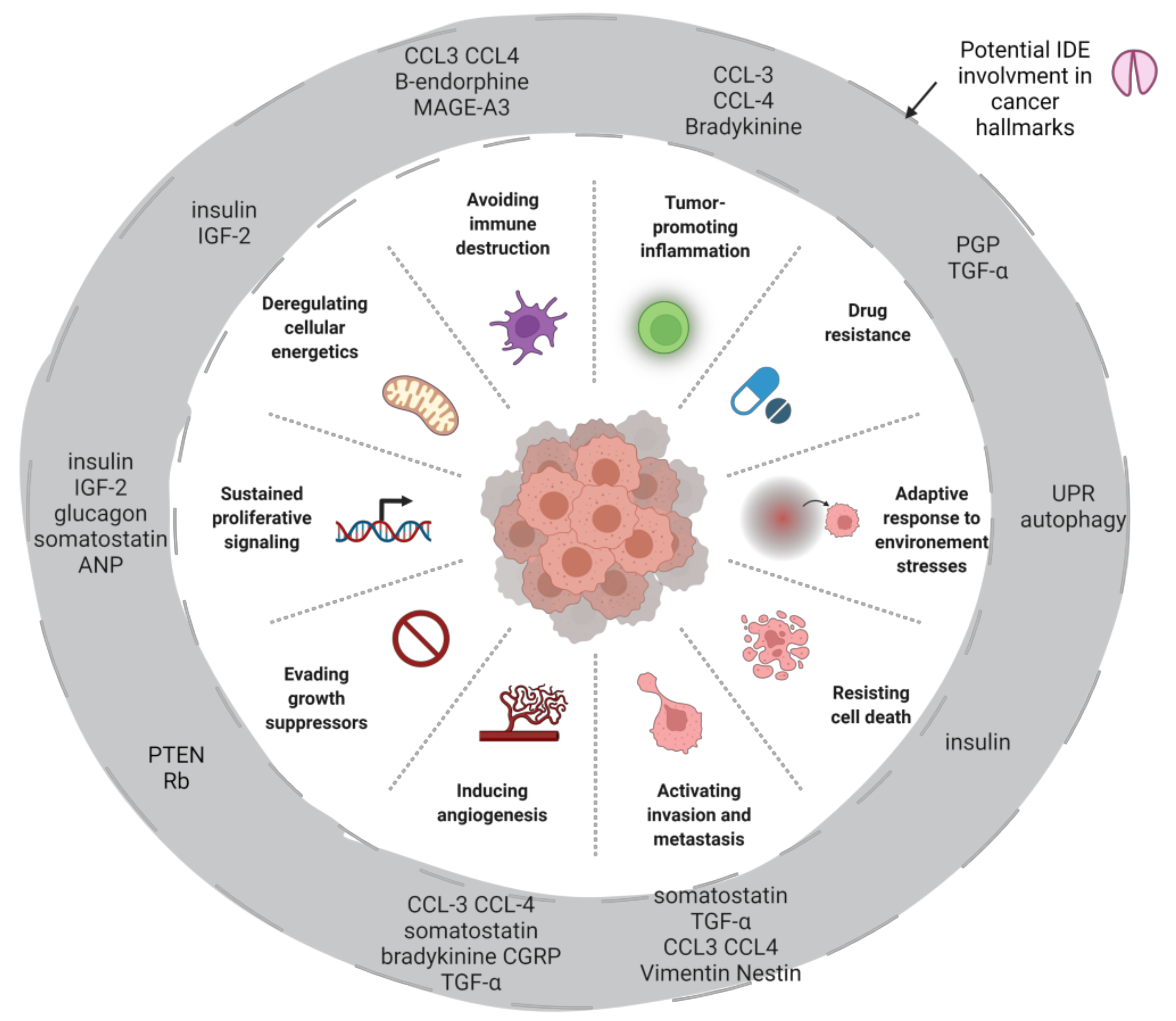
9. IDE, Tumor Suppression and Proliferation
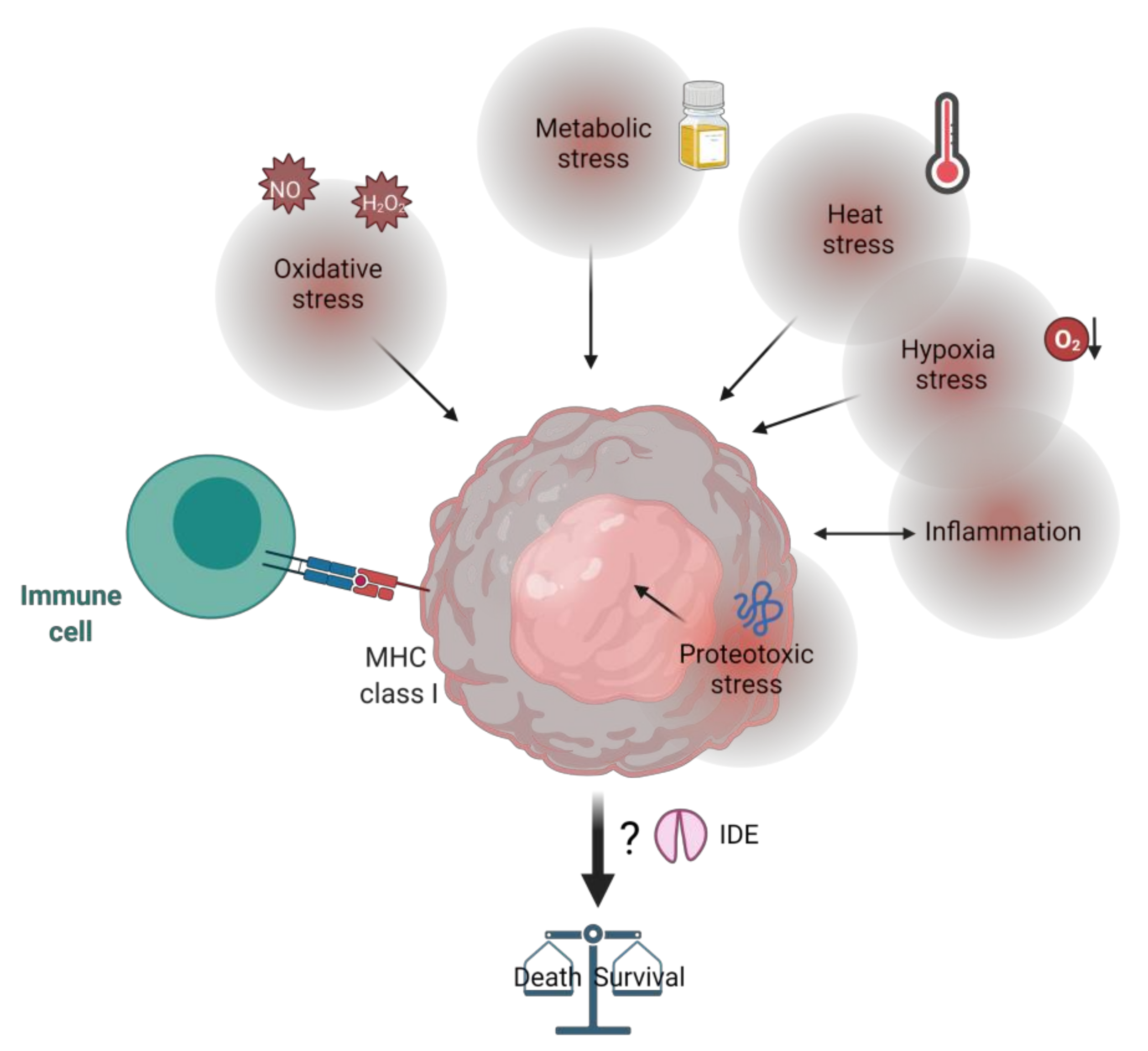
10. IDE, Tumor Microenvironment and Stress
11. IDE Is a Druggable Target
12. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pivovarova, O.; Höhn, A.; Grune, T.; Pfeiffer, A.F.H.; Rudovich, N. Insulin-Degrading Enzyme: New Therapeutic Target for Diabetes and Alzheimer’s Disease? Ann. Med. 2016, 48, 614–624. [Google Scholar] [CrossRef] [PubMed]
- Leissring, M.A.; González-Casimiro, C.M.; Merino, B.; Suire, C.N.; Perdomo, G. Targeting Insulin-Degrading Enzyme in Insulin Clearance. Int. J. Mol. Sci. 2021, 22, 2235. [Google Scholar] [CrossRef] [PubMed]
- Sousa, L.; Guarda, M.; Meneses, M.J.; Macedo, M.P.; Vicente Miranda, H. Insulin-Degrading Enzyme: An Ally against Metabolic and Neurodegenerative Diseases. J. Pathol. 2021, 255, 346–361. [Google Scholar] [CrossRef] [PubMed]
- Leissring, M.A. Insulin-Degrading Enzyme: Paradoxes and Possibilities. Cells 2021, 10, 2445. [Google Scholar] [CrossRef]
- Hamel, F.G.; Mahoney, M.J.; Duckworth, W.C. Degradation of Intraendosomal Insulin by Insulin-Degrading Enzyme without Acidification. Diabetes 1991, 40, 436–443. [Google Scholar] [CrossRef]
- Song, E.S.; Jang, H.; Guo, H.-F.; Juliano, M.A.; Juliano, L.; Morris, A.J.; Galperin, E.; Rodgers, D.W.; Hersh, L.B. Inositol Phosphates and Phosphoinositides Activate Insulin-Degrading Enzyme, While Phosphoinositides Also Mediate Binding to Endosomes. Proc. Natl. Acad. Sci. USA 2017, 114, E2826–E2835. [Google Scholar] [CrossRef]
- Leissring, M.A.; Farris, W.; Wu, X.; Christodoulou, D.C.; Haigis, M.C.; Guarente, L.; Selkoe, D.J. Alternative Translation Initiation Generates a Novel Isoform of Insulin-Degrading Enzyme Targeted to Mitochondria. Biochem. J. 2004, 383, 439–446. [Google Scholar] [CrossRef]
- Morita, M.; Kurochkin, I.V.; Motojima, K.; Goto, S.; Takano, T.; Okamura, S.; Sato, R.; Yokota, S.; Imanaka, T. Insulin-Degrading Enzyme Exists inside of Rat Liver Peroxisomes and Degrades Oxidized Proteins. Cell Struct. Funct. 2000, 25, 309–315. [Google Scholar] [CrossRef][Green Version]
- Kupfer, S.R.; Wilson, E.M.; French, F.S. Androgen and Glucocorticoid Receptors Interact with Insulin Degrading Enzyme. J. Biol. Chem. 1994, 269, 20622–20628. [Google Scholar] [CrossRef]
- Seta, K.A.; Roth, R.A. Overexpression of Insulin Degrading Enzyme: Cellular Localization and Effects on Insulin Signaling. Biochem. Biophys. Res. Commun. 1997, 231, 167–171. [Google Scholar] [CrossRef]
- Bulloj, A.; Leal, M.C.; Xu, H.; Castaño, E.M.; Morelli, L. Insulin-Degrading Enzyme Sorting in Exosomes: A Secretory Pathway for a Key Brain Amyloid-Beta Degrading Protease. J. Alzheimers Dis. JAD 2010, 19, 79–95. [Google Scholar] [CrossRef]
- Son, S.M.; Cha, M.-Y.; Choi, H.; Kang, S.; Choi, H.; Lee, M.-S.; Park, S.A.; Mook-Jung, I. Insulin-Degrading Enzyme Secretion from Astrocytes Is Mediated by an Autophagy-Based Unconventional Secretory Pathway in Alzheimer Disease. Autophagy 2016, 12, 784–800. [Google Scholar] [CrossRef]
- Zhao, J.; Li, L.; Leissring, M.A. Insulin-Degrading Enzyme Is Exported via an Unconventional Protein Secretion Pathway. Mol. Neurodegener. 2009, 4, 4. [Google Scholar] [CrossRef]
- Li, Q.; Ali, M.A.; Cohen, J.I. Insulin Degrading Enzyme Is a Cellular Receptor Mediating Varicella-Zoster Virus Infection and Cell-to-Cell Spread. Cell 2006, 127, 305–316. [Google Scholar] [CrossRef]
- Tundo, G.R.; Sbardella, D.; Ciaccio, C.; Grasso, G.; Gioia, M.; Coletta, A.; Polticelli, F.; Di Pierro, D.; Milardi, D.; Van Endert, P.; et al. Multiple Functions of Insulin-Degrading Enzyme: A Metabolic Crosslight? Crit. Rev. Biochem. Mol. Biol. 2017, 52, 1–29. [Google Scholar] [CrossRef]
- Malito, E.; Hulse, R.E.; Tang, W.-J. Amyloid Beta-Degrading Cryptidases: Insulin Degrading Enzyme, Presequence Peptidase, and Neprilysin. Cell. Mol. Life Sci. CMLS 2008, 65, 2574–2585. [Google Scholar] [CrossRef]
- Perlman, R.K.; Gehm, B.D.; Kuo, W.L.; Rosner, M.R. Functional Analysis of Conserved Residues in the Active Site of Insulin-Degrading Enzyme. J. Biol. Chem. 1993, 268, 21538–21544. [Google Scholar] [CrossRef]
- McCord, L.A.; Liang, W.G.; Dowdell, E.; Kalas, V.; Hoey, R.J.; Koide, A.; Koide, S.; Tang, W.-J. Conformational States and Recognition of Amyloidogenic Peptides of Human Insulin-Degrading Enzyme. Proc. Natl. Acad. Sci. USA 2013, 110, 13827–13832. [Google Scholar] [CrossRef]
- Zhang, Z.; Liang, W.G.; Bailey, L.J.; Tan, Y.Z.; Wei, H.; Wang, A.; Farcasanu, M.; Woods, V.A.; McCord, L.A.; Lee, D.; et al. Ensemble CryoEM Elucidates the Mechanism of Insulin Capture and Degradation by Human Insulin Degrading Enzyme. eLife 2018, 7, e33572. [Google Scholar] [CrossRef]
- Shen, Y.; Joachimiak, A.; Rosner, M.R.; Tang, W.-J. Structures of Human Insulin-Degrading Enzyme Reveal a New Substrate Recognition Mechanism. Nature 2006, 443, 870–874. [Google Scholar] [CrossRef]
- Stefanidis, L.; Fusco, N.D.; Cooper, S.E.; Smith-Carpenter, J.E.; Alper, B.J. Molecular Determinants of Substrate Specificity in Human Insulin-Degrading Enzyme. Biochemistry 2018, 57, 4903–4914. [Google Scholar] [CrossRef]
- Manolopoulou, M.; Guo, Q.; Malito, E.; Schilling, A.B.; Tang, W.-J. Molecular Basis of Catalytic Chamber-Assisted Unfolding and Cleavage of Human Insulin by Human Insulin-Degrading Enzyme. J. Biol. Chem. 2009, 284, 14177–14188. [Google Scholar] [CrossRef]
- Malito, E.; Ralat, L.A.; Manolopoulou, M.; Tsay, J.L.; Wadlington, N.L.; Tang, W.-J. Molecular Bases for the Recognition of Short Peptide Substrates and Cysteine-Directed Modifications of Human Insulin-Degrading Enzyme. Biochemistry 2008, 47, 12822–12834. [Google Scholar] [CrossRef]
- Noinaj, N.; Bhasin, S.K.; Song, E.S.; Scoggin, K.E.; Juliano, M.A.; Juliano, L.; Hersh, L.B.; Rodgers, D.W. Identification of the Allosteric Regulatory Site of Insulysin. PLoS ONE 2011, 6, e20864. [Google Scholar] [CrossRef][Green Version]
- Noinaj, N.; Song, E.S.; Bhasin, S.; Alper, B.J.; Schmidt, W.K.; Hersh, L.B.; Rodgers, D.W. Anion Activation Site of Insulin-Degrading Enzyme. J. Biol. Chem. 2012, 287, 48–57. [Google Scholar] [CrossRef]
- Im, H.; Manolopoulou, M.; Malito, E.; Shen, Y.; Zhao, J.; Neant-Fery, M.; Sun, C.-Y.; Meredith, S.C.; Sisodia, S.S.; Leissring, M.A.; et al. Structure of Substrate-Free Human Insulin-Degrading Enzyme (IDE) and Biophysical Analysis of ATP-Induced Conformational Switch of IDE. J. Biol. Chem. 2007, 282, 25453–25463. [Google Scholar] [CrossRef]
- Sbardella, D.; Tundo, G.R.; Coletta, A.; Marcoux, J.; Koufogeorgou, E.I.; Ciaccio, C.; Santoro, A.M.; Milardi, D.; Grasso, G.; Cozza, P.; et al. The Insulin-Degrading Enzyme Is an Allosteric Modulator of the 20S Proteasome and a Potential Competitor of the 19S. Cell. Mol. Life Sci. CMLS 2018, 75, 3441–3456. [Google Scholar] [CrossRef]
- Grasso, G.; Lanza, V.; Malgieri, G.; Fattorusso, R.; Pietropaolo, A.; Rizzarelli, E.; Milardi, D. The Insulin Degrading Enzyme Activates Ubiquitin and Promotes the Formation of K48 and K63 Diubiquitin. Chem. Commun. Camb. Engl. 2015, 51, 15724–15727. [Google Scholar] [CrossRef]
- Bellia, F.; Lanza, V.; Ahmed, I.M.M.; Garcia-Vinuales, S.; Veiss, E.; Arizzi, M.; Calcagno, D.; Milardi, D.; Grasso, G. Site Directed Mutagenesis of Insulin-Degrading Enzyme Allows Singling out the Molecular Basis of Peptidase versus E1-like Activity: The Role of Metal Ions. Met. Integr. Biomet. Sci. 2019, 11, 278–281. [Google Scholar] [CrossRef]
- Ralat, L.A.; Ren, M.; Schilling, A.B.; Tang, W.-J. Protective Role of Cys-178 against the Inactivation and Oligomerization of Human Insulin-Degrading Enzyme by Oxidation and Nitrosylation. J. Biol. Chem. 2009, 284, 34005–34018. [Google Scholar] [CrossRef]
- Duckworth, W.C.; Bennett, R.G.; Hamel, F.G. Insulin Degradation: Progress and Potential. Endocr. Rev. 1998, 19, 608–624. [Google Scholar] [CrossRef] [PubMed]
- Vigneri, R.; Sciacca, L.; Vigneri, P. Rethinking the Relationship between Insulin and Cancer. Trends Endocrinol. Metab. 2020, 31, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Duckworth, W.C.; Kitabchi, A.E. Insulin and Glucagon Degradation by the Same Enzyme. Diabetes 1974, 23, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Yagi, T.; Kubota, E.; Koyama, H.; Tanaka, T.; Kataoka, H.; Imaeda, K.; Joh, T. Glucagon Promotes Colon Cancer Cell Growth via Regulating AMPK and MAPK Pathways. Oncotarget 2018, 9, 10650–10664. [Google Scholar] [CrossRef][Green Version]
- Ciaccio, C.; Tundo, G.R.; Grasso, G.; Spoto, G.; Marasco, D.; Ruvo, M.; Gioia, M.; Rizzarelli, E.; Coletta, M. Somatostatin: A Novel Substrate and a Modulator of Insulin-Degrading Enzyme Activity. J. Mol. Biol. 2009, 385, 1556–1567. [Google Scholar] [CrossRef][Green Version]
- Pyronnet, S.; Bousquet, C.; Najib, S.; Azar, R.; Laklai, H.; Susini, C. Antitumor Effects of Somatostatin. Mol. Cell. Endocrinol. 2008, 286, 230–237. [Google Scholar] [CrossRef]
- Müller, D.; Baumeister, H.; Buck, F.; Richter, D. Atrial Natriuretic Peptide (ANP) Is a High-Affinity Substrate for Rat Insulin-Degrading Enzyme. Eur. J. Biochem. 1991, 202, 285–292. [Google Scholar] [CrossRef]
- Xu, M.; Liu, X.; Li, P.; Yang, Y.; Zhang, W.; Zhao, S.; Zeng, Y.; Zhou, X.; Zeng, L.-H.; Yang, G. Modified Natriuretic Peptides and Their Potential Role in Cancer Treatment. Biomed. J. 2021, 14, 1051–1064. [Google Scholar] [CrossRef]
- Mezzasoma, L.; Peirce, M.J.; Minelli, A.; Bellezza, I. Natriuretic Peptides: The Case of Prostate Cancer. Mol. Basel Switz. 2017, 22, E1680. [Google Scholar] [CrossRef]
- Ralat, L.A.; Guo, Q.; Ren, M.; Funke, T.; Dickey, D.M.; Potter, L.R.; Tang, W.-J. Insulin-Degrading Enzyme Modulates the Natriuretic Peptide-Mediated Signaling Response. J. Biol. Chem. 2011, 286, 4670–4679. [Google Scholar] [CrossRef]
- Safavi, A.; Miller, B.C.; Cottam, L.; Hersh, L.B. Identification of Gamma-Endorphin-Generating Enzyme as Insulin-Degrading Enzyme. Biochemistry 1996, 35, 14318–14325. [Google Scholar] [CrossRef]
- Scheau, C.; Draghici, C.; Ilie, M.A.; Lupu, M.; Solomon, I.; Tampa, M.; Georgescu, S.R.; Caruntu, A.; Constantin, C.; Neagu, M.; et al. Neuroendocrine Factors in Melanoma Pathogenesis. Cancers 2021, 13, 2277. [Google Scholar] [CrossRef]
- Banerjee, J.; Papu John, A.M.S.; Schuller, H.M. Regulation of Nonsmall-Cell Lung Cancer Stem Cell like Cells by Neurotransmitters and Opioid Peptides. Int. J. Cancer 2015, 137, 2815–2824. [Google Scholar] [CrossRef]
- Da Costa, P.L.N.; Sirois, P.; Tannock, I.F.; Chammas, R. The Role of Kinin Receptors in Cancer and Therapeutic Opportunities. Cancer Lett. 2014, 345, 27–38. [Google Scholar] [CrossRef]
- Kim, Y.-G.; Lone, A.M.; Nolte, W.M.; Saghatelian, A. Peptidomics Approach to Elucidate the Proteolytic Regulation of Bioactive Peptides. Proc. Natl. Acad. Sci. USA 2012, 109, 8523–8527. [Google Scholar] [CrossRef]
- Zhang, Y.; Lin, C.; Wang, X.; Ji, T. Calcitonin Gene-Related Peptide: A Promising Bridge between Cancer Development and Cancer-Associated Pain in Oral Squamous Cell Carcinoma. Oncol. Lett. 2020, 20, 253. [Google Scholar] [CrossRef]
- Misbin, R.I.; Almira, E.C. Degradation of Insulin and Insulin-Like Growth Factors by Enzyme Purified From Human Erythrocytes: Comparison of Degradation Products Observed With A14- and B26-[125I]Monoiodoinsulin. Diabetes 1989, 38, 152–158. [Google Scholar] [CrossRef]
- Livingstone, C. IGF2 and Cancer. Endocr. Relat. Cancer 2013, 20, R321–R339. [Google Scholar] [CrossRef]
- Garcia, J.V.; Gehm, B.D.; Rosner, M.R. An Evolutionarily Conserved Enzyme Degrades Transforming Growth Factor-Alpha as Well as Insulin. J. Cell Biol. 1989, 109, 1301–1307. [Google Scholar] [CrossRef]
- Guo, Q.; Manolopoulou, M.; Bian, Y.; Schilling, A.B.; Tang, W.-J. Molecular Basis for the Recognition and Cleavages of IGF-II, TGF-Alpha, and Amylin by Human Insulin-Degrading Enzyme. J. Mol. Biol. 2010, 395, 430–443. [Google Scholar] [CrossRef]
- Sasaki, T.; Hiroki, K.; Yamashita, Y. The Role of Epidermal Growth Factor Receptor in Cancer Metastasis and Microenvironment. BioMed Res. Int. 2013, 2013, e546318. [Google Scholar] [CrossRef]
- Liang, W.G.; Ren, M.; Zhao, F.; Tang, W.-J. Structures of Human CCL18, CCL3, and CCL4 Reveal Molecular Determinants for Quaternary Structures and Sensitivity to Insulin-Degrading Enzyme. J. Mol. Biol. 2015, 427, 1345–1358. [Google Scholar] [CrossRef]
- Ren, M.; Guo, Q.; Guo, L.; Lenz, M.; Qian, F.; Koenen, R.R.; Xu, H.; Schilling, A.B.; Weber, C.; Ye, R.D.; et al. Polymerization of MIP-1 Chemokine (CCL3 and CCL4) and Clearance of MIP-1 by Insulin-Degrading Enzyme. EMBO J. 2010, 29, 3952–3966. [Google Scholar] [CrossRef] [PubMed]
- Korbecki, J.; Grochans, S.; Gutowska, I.; Barczak, K.; Baranowska-Bosiacka, I. CC Chemokines in a Tumor: A Review of Pro-Cancer and Anti-Cancer Properties of Receptors CCR5, CCR6, CCR7, CCR8, CCR9, and CCR10 Ligands. Int. J. Mol. Sci. 2020, 21, E7619. [Google Scholar] [CrossRef]
- Ralat, L.A.; Kalas, V.; Zheng, Z.; Goldman, R.D.; Sosnick, T.R.; Tang, W.-J. Ubiquitin Is a Novel Substrate for Human Insulin-Degrading Enzyme. J. Mol. Biol. 2011, 406, 454–466. [Google Scholar] [CrossRef] [PubMed]
- Mansour, M.A. Ubiquitination: Friend and Foe in Cancer. Int. J. Biochem. Cell Biol. 2018, 101, 80–93. [Google Scholar] [CrossRef] [PubMed]
- Kurochkin, I.V.; Goto, S. Alzheimer’s Beta-Amyloid Peptide Specifically Interacts with and Is Degraded by Insulin Degrading Enzyme. FEBS Lett. 1994, 345, 33–37. [Google Scholar] [CrossRef]
- Qin, H.; Wang, J.; Ren, J.; Qu, X. Amyloid β and Tumorigenesis: Amyloid β-Induced Telomere Dysfunction in Tumor Cells. CCS Chem. 2019, 1, 313–325. [Google Scholar]
- Majd, S.; Power, J.; Majd, Z. Alzheimer’s Disease and Cancer: When Two Monsters Cannot Be Together. Front. Neurosci. 2019, 13, 155. [Google Scholar] [CrossRef]
- Pavliukeviciene, B.; Zentelyte, A.; Jankunec, M.; Valiuliene, G.; Talaikis, M.; Navakauskiene, R.; Niaura, G.; Valincius, G. Amyloid β Oligomers Inhibit Growth of Human Cancer Cells. PLoS ONE 2019, 14, e0221563. [Google Scholar] [CrossRef]
- Bennett, R.G.; Duckworth, W.C.; Hamel, F.G. Degradation of Amylin by Insulin-Degrading Enzyme. J. Biol. Chem. 2000, 275, 36621–36625. [Google Scholar] [CrossRef]
- Deprez-Poulain, R.; Hennuyer, N.; Bosc, D.; Liang, W.G.; Enée, E.; Marechal, X.; Charton, J.; Totobenazara, J.; Berte, G.; Jahklal, J.; et al. Catalytic Site Inhibition of Insulin-Degrading Enzyme by a Small Molecule Induces Glucose Intolerance in Mice. Nat. Commun. 2015, 6, 8250. [Google Scholar] [CrossRef]
- Kurochkin, I.V. Amyloidogenic Determinant as a Substrate Recognition Motif of Insulin-Degrading Enzyme. FEBS Lett. 1998, 427, 153–156. [Google Scholar] [CrossRef]
- Ramaraju, B.; Nelson, S.L.; Zheng, W.; Ghirlando, R.; Deshmukh, L. Quantitative NMR Study of Insulin-Degrading Enzyme Using Amyloid-β and HIV-1 P6 Elucidates Its Chaperone Activity. Biochemistry 2021, 60, 2519–2523. [Google Scholar] [CrossRef]
- Tundo, G.R.; Sbardella, D.; Ciaccio, C.; Bianculli, A.; Orlandi, A.; Desimio, M.G.; Arcuri, G.; Coletta, M.; Marini, S. Insulin-Degrading Enzyme (IDE): A Novel Heat Shock-like Protein. J. Biol. Chem. 2013, 288, 2281–2289. [Google Scholar] [CrossRef]
- Sbardella, D.; Tundo, G.R.; Sciandra, F.; Bozzi, M.; Gioia, M.; Ciaccio, C.; Tarantino, U.; Brancaccio, A.; Coletta, M.; Marini, S. Proteasome Activity Is Affected by Fluctuations in Insulin-Degrading Enzyme Distribution. PLoS ONE 2015, 10, e0132455. [Google Scholar] [CrossRef]
- Carpenter, J.E.; Jackson, W.; de Souza, G.A.; Haarr, L.; Grose, C. Insulin-Degrading Enzyme Binds to the Nonglycosylated Precursor of Varicella-Zoster Virus GE Protein Found in the Endoplasmic Reticulum. J. Virol. 2010, 84, 847–855. [Google Scholar] [CrossRef]
- Li, Q.; Ali, M.A.; Wang, K.; Sayre, D.; Hamel, F.G.; Fischer, E.R.; Bennett, R.G.; Cohen, J.I. Insulin Degrading Enzyme Induces a Conformational Change in Varicella-Zoster Virus GE, and Enhances Virus Infectivity and Stability. PLoS ONE 2010, 5, e11327. [Google Scholar] [CrossRef]
- De Tullio, M.B.; Morelli, L.; Castaño, E.M. The Irreversible Binding of Amyloid Peptide Substrates to Insulin-Degrading Enzyme. Prion 2008, 2, 51–56. [Google Scholar] [CrossRef]
- Sharma, S.K.; Chorell, E.; Steneberg, P.; Vernersson-Lindahl, E.; Edlund, H.; Wittung-Stafshede, P. Insulin-Degrading Enzyme Prevents α-Synuclein Fibril Formation in a Nonproteolytical Manner. Sci. Rep. 2015, 5, 12531. [Google Scholar] [CrossRef]
- De Tullio, M.B.; Castelletto, V.; Hamley, I.W.; Martino Adami, P.V.; Morelli, L.; Castaño, E.M. Proteolytically Inactive Insulin-Degrading Enzyme Inhibits Amyloid Formation Yielding Non-Neurotoxic Aβ Peptide Aggregates. PLoS ONE 2013, 8, e59113. [Google Scholar] [CrossRef] [PubMed]
- Ejma, M.; Madetko, N.; Brzecka, A.; Guranski, K.; Alster, P.; Misiuk-Hojło, M.; Somasundaram, S.G.; Kirkland, C.E.; Aliev, G. The Links between Parkinson’s Disease and Cancer. Biomedicines 2020, 8, E416. [Google Scholar] [CrossRef] [PubMed]
- Chou, Y.-H.; Kuo, W.-L.; Rosner, M.R.; Tang, W.-J.; Goldman, R.D. Structural Changes in Intermediate Filament Networks Alter the Activity of Insulin-Degrading Enzyme. FASEB J. 2009, 23, 3734–3742. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Alsharif, S.; Fallatah, A.; Chung, B.M. Intermediate Filaments as Effectors of Cancer Development and Metastasis: A Focus on Keratins, Vimentin, and Nestin. Cells 2019, 8, E497. [Google Scholar] [CrossRef]
- Rousseau, A.; Bertolotti, A. Regulation of Proteasome Assembly and Activity in Health and Disease. Nat. Rev. Mol. Cell Biol. 2018, 19, 697–712. [Google Scholar] [CrossRef]
- Thibaudeau, T.A.; Smith, D.M. A Practical Review of Proteasome Pharmacology. Pharmacol. Rev. 2019, 71, 170–197. [Google Scholar] [CrossRef]
- Grasso, G.; Rizzarelli, E.; Spoto, G. How the Binding and Degrading Capabilities of Insulin Degrading Enzyme Are Affected by Ubiquitin. Biochim. Biophys. Acta BBA-Proteins Proteom. 2008, 1784, 1122–1126. [Google Scholar] [CrossRef]
- Saric, T.; Müller, D.; Seitz, H.-J.; Pavelic, K. Non-Covalent Interaction of Ubiquitin with Insulin-Degrading Enzyme. Mol. Cell. Endocrinol. 2003, 204, 11–20. [Google Scholar] [CrossRef]
- Bennett, R.G.; Hamel, F.G.; Duckworth, W.C. Insulin Inhibits the Ubiquitin-Dependent Degrading Activity of the 26S Proteasome. Endocrinology 2000, 141, 2508–2517. [Google Scholar] [CrossRef]
- Fawcett, J.; Permana, P.A.; Levy, J.L.; Duckworth, W.C. Regulation of Protein Degradation by Insulin-Degrading Enzyme: Analysis by Small Interfering RNA-Mediated Gene Silencing. Arch. Biochem. Biophys. 2007, 468, 128–133. [Google Scholar] [CrossRef]
- Gandolfi, S.; Laubach, J.P.; Hideshima, T.; Chauhan, D.; Anderson, K.C.; Richardson, P.G. The Proteasome and Proteasome Inhibitors in Multiple Myeloma. Cancer Metastasis Rev. 2017, 36, 561–584. [Google Scholar] [CrossRef]
- Weirich, G.; Mengele, K.; Yfanti, C.; Gkazepis, A.; Hellmann, D.; Welk, A.; Giersig, C.; Kuo, W.-L.; Rosner, M.R.; Tang, W.-J.; et al. Immunohistochemical Evidence of Ubiquitous Distribution of the Metalloendoprotease Insulin-Degrading Enzyme (IDE; Insulysin) in Human Non-Malignant Tissues and Tumor Cell Lines. Biol. Chem. 2008, 389, 1441–1445. [Google Scholar] [CrossRef]
- Yfanti, C.; Mengele, K.; Gkazepis, A.; Weirich, G.; Giersig, C.; Kuo, W.-L.; Tang, W.-J.; Rosner, M.; Schmitt, M. Expression of Metalloprotease Insulin-Degrading Enzyme (Insulysin) in Normal and Malignant Human Tissues. Int. J. Mol. Med. 2008, 22, 421–431. [Google Scholar]
- Radulescu, R.T.; Hufnagel, C.; Luppa, P.; Hellebrand, H.; Kuo, W.-L.; Rosner, M.R.; Harbeck, N.; Giersig, C.; Meindl, A.; Schmitt, M.; et al. Immunohistochemical Demonstration of the Zinc Metalloprotease Insulin-Degrading Enzyme in Normal and Malignant Human Breast: Correlation with Tissue Insulin Levels. Int. J. Oncol. 2007, 30, 73–80. [Google Scholar] [CrossRef][Green Version]
- Gennari, A.; Sormani, M.; Puntoni, M.; Martini, V.; Amaro, A.; Bruzzi, P.; Pfeffer, U. Identification of a Prognostic Signature Based on the Expression of Genes Related to the Insulin Pathway in Early Breast Cancer. Breast Care Basel Switz. 2021, 16, 299–306. [Google Scholar] [CrossRef]
- Pollak, M. Insulin and Insulin-like Growth Factor Signalling in Neoplasia. Nat. Rev. Cancer 2008, 8, 915–928. [Google Scholar] [CrossRef]
- Vigneri, P.G.; Tirrò, E.; Pennisi, M.S.; Massimino, M.; Stella, S.; Romano, C.; Manzella, L. The Insulin/IGF System in Colorectal Cancer Development and Resistance to Therapy. Front. Oncol. 2015, 5, 230. [Google Scholar] [CrossRef]
- Kasprzak, A. Insulin-Like Growth Factor 1 (IGF-1) Signaling in Glucose Metabolism in Colorectal Cancer. Int. J. Mol. Sci. 2021, 22, 6434. [Google Scholar] [CrossRef]
- Pu, X.; Chen, D. Targeting Adipokines in Obesity-Related Tumors. Front. Oncol. 2021, 11, 685923. [Google Scholar] [CrossRef]
- Lauby-Secretan, B.; Scoccianti, C.; Loomis, D.; Grosse, Y.; Bianchini, F.; Straif, K. Body Fatness and Cancer—Viewpoint of the IARC Working Group. Available online: https://www.nejm.org/doi/10.1056/NEJMsr1606602 (accessed on 20 January 2022).
- Calle, E.E. Overweight, Obesity, and Mortality from Cancer in a Prospectively Studied Cohort of U.S. Adults. N. Engl. J. Med. 2003, 348, 1625–1638. [Google Scholar] [CrossRef]
- Scully, T.; Ettela, A.; LeRoith, D.; Gallagher, E.J. Obesity, Type 2 Diabetes, and Cancer Risk. Front. Oncol. 2020, 10, 615375. [Google Scholar] [CrossRef]
- Vigneri, P.; Frasca, F.; Sciacca, L.; Pandini, G.; Vigneri, R. Diabetes and Cancer. Endocr. Relat. Cancer 2009, 16, 1103–1123. [Google Scholar] [CrossRef]
- Tokarz, V.L.; MacDonald, P.E.; Klip, A. The Cell Biology of Systemic Insulin Function. J. Cell Biol. 2018, 217, 2273–2289. [Google Scholar] [CrossRef]
- Iraburu, M.J.; Garner, T.; Montiel-Duarte, C. Revising Endosomal Trafficking under Insulin Receptor Activation. Int. J. Mol. Sci. 2021, 22, 6978. [Google Scholar] [CrossRef]
- Miller, B.C.; Eckman, E.A.; Sambamurti, K.; Dobbs, N.; Chow, K.M.; Eckman, C.B.; Hersh, L.B.; Thiele, D.L. Amyloid-Beta Peptide Levels in Brain Are Inversely Correlated with Insulysin Activity Levels in Vivo. Proc. Natl. Acad. Sci. USA 2003, 100, 6221–6226. [Google Scholar] [CrossRef]
- Farris, W.; Mansourian, S.; Chang, Y.; Lindsley, L.; Eckman, E.A.; Frosch, M.P.; Eckman, C.B.; Tanzi, R.E.; Selkoe, D.J.; Guénette, S. Insulin-Degrading Enzyme Regulates the Levels of Insulin, Amyloid β-Protein, and the β-Amyloid Precursor Protein Intracellular Domain in Vivo. Proc. Natl. Acad. Sci. USA 2003, 100, 4162–4167. [Google Scholar] [CrossRef]
- Villa-Pérez, P.; Merino, B.; Fernández-Díaz, C.M.; Cidad, P.; Lobatón, C.D.; Moreno, A.; Muturi, H.T.; Ghadieh, H.E.; Najjar, S.M.; Leissring, M.A.; et al. Liver-Specific Ablation of Insulin-Degrading Enzyme Causes Hepatic Insulin Resistance and Glucose Intolerance, without Affecting Insulin Clearance in Mice. Metabolism 2018, 88, 1–11. [Google Scholar] [CrossRef]
- Najjar, S.M.; Perdomo, G. Hepatic Insulin Clearance: Mechanism and Physiology. Physiology 2019, 34, 198–215. [Google Scholar] [CrossRef]
- Calinescu, A.; Turcu, G.; Nedelcu, R.I.; Brinzea, A.; Hodorogea, A.; Antohe, M.; Diaconu, C.; Bleotu, C.; Pirici, D.; Jilaveanu, L.B.; et al. On the Dual Role of Carcinoembryonic Antigen-Related Cell Adhesion Molecule 1 (CEACAM1) in Human Malignancies. J. Immunol. Res. 2018, 2018, 7169081. [Google Scholar] [CrossRef]
- Zhao, L.; Teter, B.; Morihara, T.; Lim, G.P.; Ambegaokar, S.S.; Ubeda, O.J.; Frautschy, S.A.; Cole, G.M. Insulin-Degrading Enzyme as a Downstream Target of Insulin Receptor Signaling Cascade: Implications for Alzheimer’s Disease Intervention. J. Neurosci. 2004, 24, 11120–11126. [Google Scholar] [CrossRef]
- Pivovarova, O.; Gögebakan, Ö.; Pfeiffer, A.F.H.; Rudovich, N. Glucose Inhibits the Insulin-Induced Activation of the Insulin-Degrading Enzyme in HepG2 Cells. Diabetologia 2009, 52, 1656–1664. [Google Scholar] [CrossRef] [PubMed]
- Vella, V.; Milluzzo, A.; Scalisi, N.M.; Vigneri, P.; Sciacca, L. Insulin Receptor Isoforms in Cancer. Int. J. Mol. Sci. 2018, 19, 3615. [Google Scholar] [CrossRef] [PubMed]
- Riedemann, J.; Macaulay, V.M. IGF1R Signalling and Its Inhibition. Endocr. Relat. Cancer 2006, 13 (Suppl. S1), S33–S43. [Google Scholar] [CrossRef] [PubMed]
- Nosho, K.; Yamamoto, H.; Taniguchi, H.; Adachi, Y.; Yoshida, Y.; Arimura, Y.; Endo, T.; Hinoda, Y.; Imai, K. Interplay of Insulin-like Growth Factor-II, Insulin-like Growth Factor-I, Insulin-like Growth Factor-I Receptor, COX-2, and Matrix Metalloproteinase-7, Play Key Roles in the Early Stage of Colorectal Carcinogenesis. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2004, 10, 7950–7957. [Google Scholar] [CrossRef]
- Thackeray, J.T.; Pietzsch, S.; Stapel, B.; Ricke-Hoch, M.; Lee, C.-W.; Bankstahl, J.P.; Scherr, M.; Heineke, J.; Scharf, G.; Haghikia, A.; et al. Insulin Supplementation Attenuates Cancer-Induced Cardiomyopathy and Slows Tumor Disease Progression. JCI Insight 2017, 2, 93098. [Google Scholar] [CrossRef]
- González-Casimiro, C.M.; Cámara-Torres, P.; Merino, B.; Diez-Hermano, S.; Postigo-Casado, T.; Leissring, M.A.; Cózar-Castellano, I.; Perdomo, G. Effects of Fasting and Feeding on Transcriptional and Posttranscriptional Regulation of Insulin-Degrading Enzyme in Mice. Cells 2021, 10, 2446. [Google Scholar] [CrossRef]
- Zhao, L.; Yao, J.; Mao, Z.; Chen, S.; Wang, Y.; Brinton, R.D. 17β-Estradiol Regulates Insulin-Degrading Enzyme Expression via an ERβ/PI3-K Pathway in Hippocampus: Relevance to Alzheimer’s Prevention. Neurobiol. Aging 2011, 32, 1949–1963. [Google Scholar] [CrossRef]
- Udrisar, D.P.; Wanderley, M.I.; Porto, R.C.C.; Cardoso, C.L.P.; Barbosa, M.C.L.; Camberos, M.C.; Cresto, J.C. Androgen- and Estrogen-Dependent Regulation of Insulin-Degrading Enzyme in Subcellular Fractions of Rat Prostate and Uterus. Exp. Biol. Med. 2005, 230, 479–486. [Google Scholar] [CrossRef]
- Vieira, J.S.B.C.; Saraiva, K.L.A.; Barbosa, M.C.L.; Porto, R.C.C.; Cresto, J.C.; Peixoto, C.A.; Wanderley, M.I.; Udrisar, D.P. Effect of Dexamethasone and Testosterone Treatment on the Regulation of Insulin-Degrading Enzyme and Cellular Changes in Ventral Rat Prostate after Castration. Int. J. Exp. Pathol. 2011, 92, 272–280. [Google Scholar] [CrossRef]
- Radulescu, R.T.; Duckworth, W.C.; Levy, J.L.; Fawcett, J. Retinoblastoma Protein Co-Purifies with Proteasomal Insulin-Degrading Enzyme: Implications for Cell Proliferation Control. Biochem. Biophys. Res. Commun. 2010, 395, 196–199. [Google Scholar] [CrossRef]
- Radulescu, R.T. Intracellular Insulin in Human Tumors: Examples and Implications. Diabetol. Metab. Syndr. 2011, 3, 5. [Google Scholar] [CrossRef][Green Version]
- Liu, M.; Wang, Z.; Ren, M.; Yang, X.; Liu, B.; Qi, H.; Yu, M.; Song, S.; Chen, S.; Liu, L.; et al. SIRT4 Regulates PTEN Stability through IDE in Response to Cellular Stresses. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2019, 33, 5535–5547. [Google Scholar] [CrossRef]
- Pivovarova, O.; von Loeffelholz, C.; Ilkavets, I.; Sticht, C.; Zhuk, S.; Murahovschi, V.; Lukowski, S.; Döcke, S.; Kriebel, J.; de las Heras Gala, T.; et al. Modulation of Insulin Degrading Enzyme Activity and Liver Cell Proliferation. Cell Cycle Georget. Tex 2015, 14, 2293–2300. [Google Scholar] [CrossRef][Green Version]
- Minchenko, D.O.; Khita, O.O.; Tsymbal, D.O.; Danilovskyi, S.V.; Rudnytska, O.V.; Halkin, O.V.; Kryvdiuk, I.V.; Smeshkova, M.V.; Yakymchuk, M.M.; Bezrodnyi, B.H.; et al. Expression of IDE and PITRM1 Genes in ERN1 Knockdown U87 Glioma Cells: Effect of Hypoxia and Glucose Deprivation. Endocr. Regul. 2020, 54, 183–195. [Google Scholar] [CrossRef]
- Urra, H.; Dufey, E.; Avril, T.; Chevet, E.; Hetz, C. Endoplasmic Reticulum Stress and the Hallmarks of Cancer. Trends Cancer 2016, 2, 252–262. [Google Scholar] [CrossRef]
- Beuzelin, C.; Evnouchidou, I.; Rigolet, P.; Cauvet-Burgevin, A.; Girard, P.-M.; Dardalhon, D.; Culina, S.; Gdoura, A.; van Endert, P.; Francesconi, S. Deletion of the Fission Yeast Homologue of Human Insulinase Reveals a TORC1-Dependent Pathway Mediating Resistance to Proteotoxic Stress. PLoS ONE 2013, 8, e67705. [Google Scholar] [CrossRef]
- Levy, J.M.M.; Towers, C.G.; Thorburn, A. Targeting Autophagy in Cancer. Nat. Rev. Cancer 2017, 17, 528–542. [Google Scholar] [CrossRef]
- New, J.; Thomas, S.M. Autophagy-Dependent Secretion: Mechanism, Factors Secreted, and Disease Implications. Autophagy 2019, 15, 1682–1693. [Google Scholar] [CrossRef]
- Parmentier, N.; Stroobant, V.; Colau, D.; de Diesbach, P.; Morel, S.; Chapiro, J.; van Endert, P.; Van den Eynde, B.J. Production of an Antigenic Peptide by Insulin-Degrading Enzyme. Nat. Immunol. 2010, 11, 449–454. [Google Scholar] [CrossRef]
- Culina, S.; Mauvais, F.-X.; Hsu, H.-T.; Burgevin, A.; Guénette, S.; Moser, A.; van Endert, P. No Major Role for Insulin-Degrading Enzyme in Antigen Presentation by MHC Molecules. PLoS ONE 2014, 9, e88365. [Google Scholar] [CrossRef]
- Suire, C.N.; Nainar, S.; Fazio, M.; Kreutzer, A.G.; Paymozd-Yazdi, T.; Topper, C.L.; Thompson, C.R.; Leissring, M.A. Peptidic Inhibitors of Insulin-Degrading Enzyme with Potential for Dermatological Applications Discovered via Phage Display. PLoS ONE 2018, 13, e0193101. [Google Scholar] [CrossRef]
- Singh, N.; Baby, D.; Rajguru, J.P.; Patil, P.B.; Thakkannavar, S.S.; Pujari, V.B. Inflammation and Cancer. Ann. Afr. Med. 2019, 18, 121–126. [Google Scholar] [CrossRef]
- Kurauti, M.A.; Costa-Júnior, J.M.; Ferreira, S.M.; Santos, G.J.; Sponton, C.H.G.; Carneiro, E.M.; Telles, G.D.; Chacon-Mikahil, M.P.T.; Cavaglieri, C.R.; Rezende, L.F.; et al. Interleukin-6 Increases the Expression and Activity of Insulin-Degrading Enzyme. Sci. Rep. 2017, 7, 46750. [Google Scholar] [CrossRef]
- Nash, Y.; Ganoth, A.; Borenstein-Auerbach, N.; Levy-Barazany, H.; Goldsmith, G.; Kopelevich, A.; Pozyuchenko, K.; Sakhneny, L.; Lazdon, E.; Blanga-Kanfi, S.; et al. From Virus to Diabetes Therapy: Characterization of a Specific Insulin-Degrading Enzyme Inhibitor for Diabetes Treatment. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2021, 35, e21374. [Google Scholar] [CrossRef]
- Caravaggio, J.W.; Hasu, M.; MacLaren, R.; Thabet, M.; Raizman, J.E.; Veinot, J.P.; Marcel, Y.L.; Milne, R.W.; Whitman, S.C. Insulin-Degrading Enzyme Deficiency in Bone Marrow Cells Increases Atherosclerosis in LDL Receptor-Deficient Mice. Cardiovasc. Pathol. Off. J. Soc. Cardiovasc. Pathol. 2013, 22, 458–464. [Google Scholar] [CrossRef]
- Huang, H.; Li, X.; Zhuang, Y.; Li, N.; Zhu, X.; Hu, J.; Ben, J.; Yang, Q.; Bai, H.; Chen, Q. Class A Scavenger Receptor Activation Inhibits Endoplasmic Reticulum Stress-Induced Autophagy in Macrophage. J. Biomed. Res. 2014, 28, 213–221. [Google Scholar] [CrossRef]
- Neyen, C.; Plüddemann, A.; Mukhopadhyay, S.; Maniati, E.; Bossard, M.; Gordon, S.; Hagemann, T. Macrophage Scavenger Receptor a Promotes Tumor Progression in Murine Models of Ovarian and Pancreatic Cancer. J. Immunol. 2013, 190, 3798–3805. [Google Scholar] [CrossRef]
- Cao, J.; Liu, J.; Xu, R.; Zhu, X.; Zhao, X.; Qian, B.-Z. Prognostic Role of Tumour-Associated Macrophages and Macrophage Scavenger Receptor 1 in Prostate Cancer: A Systematic Review and Meta-Analysis. Oncotarget 2017, 8, 83261–83269. [Google Scholar] [CrossRef]
- Tada, Y.; Matsumi, Y.; Hara, K.; Miyauchi, W.; Sugesawa, K.; Uejima, C.; Tanio, A.; Kihara, K.; Yamamoto, M.; Takano, S.; et al. Infiltration of CD204-Overexpressing Macrophages Contributes to the Progression of Stage II and III Colorectal Cancer. Anticancer Res. 2021, 41, 4857–4865. [Google Scholar] [CrossRef]
- Gottesman, M.M.; Pastan, I.H. The Role of Multidrug Resistance Efflux Pumps in Cancer: Revisiting a JNCI Publication Exploring Expression of the MDR1 (P-Glycoprotein) Gene. J. Natl. Cancer Inst. 2015, 107, djv222. [Google Scholar] [CrossRef]
- Radulescu, R.T.; Poznic, M.; Pavelic, K. Complex Formation between Metabolic Enzymes in Tumor Cells: Unfolding the MDR1-IDE Paradigm. Mol. Cancer Ther. 2009, 8, 3171. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, Z. Protein S-Nitrosylation and Cancer. Cancer Lett. 2012, 320, 123–129. [Google Scholar] [CrossRef]
- Sharma, V.; Fernando, V.; Letson, J.; Walia, Y.; Zheng, X.; Fackelman, D.; Furuta, S. S-Nitrosylation in Tumor Microenvironment. Int. J. Mol. Sci. 2021, 22, 4600. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Hay, S.O.; Kang, D.; McBride, M.; Li, L.; Zhao, J.; Leissring, M.A. Deletion of Insulin-Degrading Enzyme Elicits Antipodal, Age-Dependent Effects on Glucose and Insulin Tolerance. PLoS ONE 2011, 6, e20818. [Google Scholar] [CrossRef]
- Farris, W.; Mansourian, S.; Leissring, M.A.; Eckman, E.A.; Bertram, L.; Eckman, C.B.; Tanzi, R.E.; Selkoe, D.J. Partial Loss-of-Function Mutations in Insulin-Degrading Enzyme That Induce Diabetes Also Impair Degradation of Amyloid Beta-Protein. Am. J. Pathol. 2004, 164, 1425–1434. [Google Scholar] [CrossRef]
- Fernández-Díaz, C.M.; Merino, B.; López-Acosta, J.F.; Cidad, P.; de la Fuente, M.A.; Lobatón, C.D.; Moreno, A.; Leissring, M.A.; Perdomo, G.; Cózar-Castellano, I. Pancreatic β-Cell-Specific Deletion of Insulin-Degrading Enzyme Leads to Dysregulated Insulin Secretion and β-Cell Functional Immaturity. Am. J. Physiol. Endocrinol. Metab. 2019, 317, E805–E819. [Google Scholar] [CrossRef] [PubMed]
- Leissring, M.A.; Malito, E.; Hedouin, S.; Reinstatler, L.; Sahara, T.; Abdul-Hay, S.O.; Choudhry, S.; Maharvi, G.M.; Fauq, A.H.; Huzarska, M.; et al. Designed Inhibitors of Insulin-Degrading Enzyme Regulate the Catabolism and Activity of Insulin. PLoS ONE 2010, 5, e10504. [Google Scholar] [CrossRef] [PubMed]
- Maianti, J.P.; Tan, G.A.; Vetere, A.; Welsh, A.J.; Wagner, B.K.; Seeliger, M.A.; Liu, D.R. Substrate-Selective Inhibitors That Reprogram the Activity of Insulin-Degrading Enzyme. Nat. Chem. Biol. 2019, 15, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Charton, J.; Gauriot, M.; Guo, Q.; Hennuyer, N.; Marechal, X.; Dumont, J.; Hamdane, M.; Pottiez, V.; Landry, V.; Sperandio, O.; et al. Imidazole-Derived 2-[N-Carbamoylmethyl-Alkylamino]Acetic Acids, Substrate-Dependent Modulators of Insulin-Degrading Enzyme in Amyloid-β Hydrolysis. Eur. J. Med. Chem. 2014, 79, 184–193. [Google Scholar] [CrossRef]
- Charton, J.; Gauriot, M.; Totobenazara, J.; Hennuyer, N.; Dumont, J.; Bosc, D.; Marechal, X.; Elbakali, J.; Herledan, A.; Wen, X.; et al. Structure-Activity Relationships of Imidazole-Derived 2-[N-Carbamoylmethyl-Alkylamino]Acetic Acids, Dual Binders of Human Insulin-Degrading Enzyme. Eur. J. Med. Chem. 2015, 90, 547–567. [Google Scholar] [CrossRef]
- Adamek, R.N.; Suire, C.N.; Stokes, R.W.; Brizuela, M.K.; Cohen, S.M.; Leissring, M.A. Hydroxypyridinethione Inhibitors of Human Insulin-Degrading Enzyme. ChemMedChem 2021, 16, 1775–1787. [Google Scholar] [CrossRef]
- Leroux, F.; Bosc, D.; Beghyn, T.; Hermant, P.; Warenghem, S.; Landry, V.; Pottiez, V.; Guillaume, V.; Charton, J.; Herledan, A.; et al. Identification of Ebselen as a Potent Inhibitor of Insulin Degrading Enzyme by a Drug Repurposing Screening. Eur. J. Med. Chem. 2019, 179, 557–566. [Google Scholar] [CrossRef]
- Yang, D.; Qin, W.; Shi, X.; Zhu, B.; Xie, M.; Zhao, H.; Teng, B.; Wu, Y.; Zhao, R.; Yin, F.; et al. Stabilized β-Hairpin Peptide Inhibits Insulin Degrading Enzyme. J. Med. Chem. 2018, 61, 8174–8185. [Google Scholar] [CrossRef]
- Tundo, G.R.; Sbardella, D.; De Pascali, S.A.; Ciaccio, C.; Coletta, M.; Fanizzi, F.P.; Marini, S. Novel Platinum(II) Compounds Modulate Insulin-Degrading Enzyme Activity and Induce Cell Death in Neuroblastoma Cells. J. Biol. Inorg. Chem. JBIC Publ. Soc. Biol. Inorg. Chem. 2015, 20, 101–108. [Google Scholar] [CrossRef]
- Cabrol, C.; Huzarska, M.A.; Dinolfo, C.; Rodriguez, M.C.; Reinstatler, L.; Ni, J.; Yeh, L.-A.; Cuny, G.D.; Stein, R.L.; Selkoe, D.J.; et al. Small-Molecule Activators of Insulin-Degrading Enzyme Discovered through High-Throughput Compound Screening. PLoS ONE 2009, 4, e5274. [Google Scholar] [CrossRef]
- Herledan, A.; Andres, M.; Lejeune-Dodge, A.; Leroux, F.; Biela, A.; Piveteau, C.; Warenghem, S.; Couturier, C.; Deprez, B.; Deprez-Poulain, R. Drug Target Engagement Using Coupled Cellular Thermal Shift Assay-Acoustic Reverse-Phase Protein Array. SLAS Discov. Adv. Life Sci. R D 2020, 25, 207–214. [Google Scholar] [CrossRef]
| Substrates | Substrate Characterization | Cancer Effect 1 | |||||
|---|---|---|---|---|---|---|---|
| Length (Residues) | Affinity (Km in µM) | PDB Codes | References | Pro | Anti | Ref. | |
| Hormones | |||||||
| Insulin | 51 | 0.070 | 2G54; 2G56; 2WBY; 6BF8; 6BFC; 6B3Q | [31] | X | [32] | |
| Glucagon | 29 | 3.5 | 2G49 | [33] | X | [34] | |
| Somatostatin | 14/28 | 7.5 | - | [35] | X | [36] | |
| ANP | 28 | nd | 3N57 | [37] | X | [38,39] | |
| CNP | 22 | nd | - | [40] | X | [38,39] | |
| Neuropeptides | |||||||
| Β-endorphin | 31 | 13 | - | [41] | X | X | [42] |
| Dynorphin (B9) | 9 | 27 | - | [41] | X | [43] | |
| Bradykinin | 9 | 4200 | 3CWW | [23] | X | [44] | |
| CGRP | 37 | nd | - | [45] | X | [46] | |
| Growth Factors | |||||||
| IGF-2 | 67 | nd | 3E4Z | [47] | X | [48] | |
| TGF-α | 50 | nd | 3E50 | [49,50] | X | [51] | |
| Cytokines | |||||||
| CCL3 | 70 | nd | 3H44 | [52,53] | X | X | [54] |
| CCL4 | 69 | nd | 4RAL | [52,53] | X | X | [54] |
| Others | |||||||
| Ubiquitin | 76 | nd | 3OFI | [55] | X 2 | [56] | |
| Amyloid β | 40–42 | 2 | 2G47; 2WK3 | [57] | X | [58,59,60] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lesire, L.; Leroux, F.; Deprez-Poulain, R.; Deprez, B. Insulin-Degrading Enzyme, an Under-Estimated Potential Target to Treat Cancer? Cells 2022, 11, 1228. https://doi.org/10.3390/cells11071228
Lesire L, Leroux F, Deprez-Poulain R, Deprez B. Insulin-Degrading Enzyme, an Under-Estimated Potential Target to Treat Cancer? Cells. 2022; 11(7):1228. https://doi.org/10.3390/cells11071228
Chicago/Turabian StyleLesire, Laetitia, Florence Leroux, Rebecca Deprez-Poulain, and Benoit Deprez. 2022. "Insulin-Degrading Enzyme, an Under-Estimated Potential Target to Treat Cancer?" Cells 11, no. 7: 1228. https://doi.org/10.3390/cells11071228
APA StyleLesire, L., Leroux, F., Deprez-Poulain, R., & Deprez, B. (2022). Insulin-Degrading Enzyme, an Under-Estimated Potential Target to Treat Cancer? Cells, 11(7), 1228. https://doi.org/10.3390/cells11071228






