Raman Spectroscopy as a Neuromonitoring Tool in Traumatic Brain Injury: A Systematic Review and Clinical Perspectives
Abstract
:1. Introduction
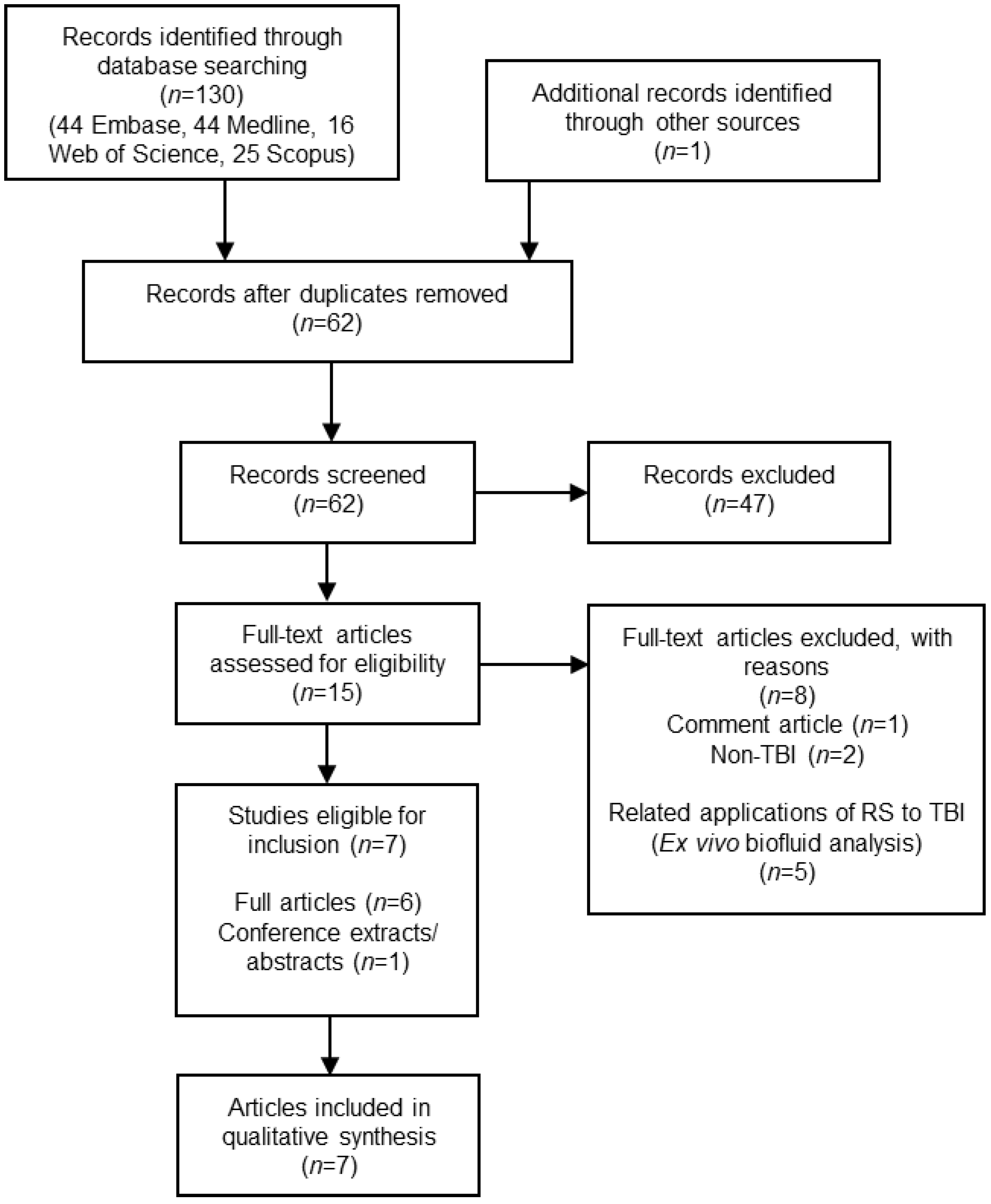
2. Materials and Methods
- Exp Traumatic brain injury;
- Exp craniocerebral trauma;
- Keywords 1 or 2;
- Raman spectroscopy;
- Exp spectrum analysis, Raman;
- Keywords 4 or 5;
- Keywords 3 and 6.
2.1. Study Selection
2.2. Data Extraction
2.3. Risk of Bias
2.4. Synthesis of Results
3. Results
3.1. Animal Models of TBI
3.2. Risk of Bias
3.3. Analogous Animal Studies with Applicable Adverse Tissue Conditions
3.4. Biofluid Diagnostics
3.5. Spectral Evidence from Animal Models
4. Discussion
4.1. Translational Considerations
4.2. Cranial Access
4.3. Probe Development
4.4. Wavelength Selection
4.5. Potential Clinical Applications
4.5.1. In Vivo Monitoring: Detailed Biochemical Assessment
4.5.2. Microdialysis Online Analysis
4.5.3. Pitchside/Bedside Concussion Assessment
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CCI | Controlled cortical impact |
| FTIR | Fourier transform infrared spectroscopy |
| GCS | Glasgow coma scale |
| GFAP | Glial fibrillary acidic protein |
| ICP | Intracranial pressure |
| mTBI | Mild TBI |
| NAA | n-Acetyle aspartate |
| NSE | Neuron-specific enolase |
| RS | Raman spectroscopy |
| SCAT5 | Sports concussion assessment tool 5 |
| SERS | Surface-enhanced Raman scattering |
| SKiNET | Self-optimising Kohonen index network |
| sTBI | Severe traumatic brain injury |
| TBI | Traumatic brain injury |
| UCHL1 | Ubiquitin C-terminal hydrolase-L1 |
References
- Langlois, J.A.; Rutland-Brown, W.; Wald, M.M. The epidemiology and impact of traumatic brain injury: A brief overview. J. Head Trauma Rehabil. 2006, 21, 375–378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jazayeri, S.B.; Beygi, S.; Shokraneh, F.; Hagen, E.M.; Rahimi-Movaghar, V. Incidence of traumatic spinal cord injury worldwide: A systematic review. Eur. Spine J. 2015, 24, 905–918. [Google Scholar] [CrossRef] [PubMed]
- Dewan, M.; Rattani, A.; Gupta, S.; Baticulon, R.; Hung, Y.; Punchak, M.; Agrawal, A.; Adeleye, A.; Shrime, M.; Rubiano, A.; et al. Estimating the global incidence of traumatic brain injury. J. Neurosurg. 2018, 130, 1080–1097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiong, Y.; Mahmood, A.; Chopp, M. Emerging treatments for traumatic brain injury. Expert Opin. Emerg. Drugs 2009, 14, 67–84. [Google Scholar] [CrossRef]
- Bullock, M.R.; Chesnut, R.; Ghajar, J.; Gordon, D.; Hartl, R.; Newell, D.W.; Servadei, F.; Walters, B.C.; Wilberger, J. Surgical management of traumatic brain injury. Neurosurgery 2006, 58, 16–24. [Google Scholar]
- Faul, M.; Coronado, V. Epidemiology of traumatic brain injury. Handb. Clin. Neurol. 2015, 127, 3–13. [Google Scholar] [CrossRef]
- Polinder, S.; Haagsma, J.A.; van Klaveren, D.; Steyerberg, E.W.; van Beeck, E.F. Health-related quality of life after TBI: A systematic review of study design, instruments, measurement properties, and outcome. Popul. Health Metr. 2015, 13, 4. [Google Scholar] [CrossRef] [Green Version]
- Pavlovic, D.; Pekic, S.; Stojanovic, M.; Popovic, V. Traumatic brain injury: Neuropathological, neurocognitive and neurobehavioral sequelae. Pituitary 2019, 22, 270–282. [Google Scholar] [CrossRef]
- Graham, N.S.N.; Sharp, D.J. Understanding neurodegeneration after traumatic brain injury: From mechanisms to clinical trials in dementia. J. Neurol. Neurosurg. Psychiatry 2019, 90, 1221–1233. [Google Scholar] [CrossRef] [Green Version]
- Chesnut, R.M.; Chesnut, R.M.; Marshall, L.F.; Klauber, M.R.; Blunt, B.A.; Baldwin, N.; Eisenberg, H.M.; Jane, J.A.; Marmarou, A.; Foulkes, M.A. The role of secondary brain injury in determining outcome from severe head injury. J. Trauma 1993, 34, 216. [Google Scholar] [CrossRef]
- Brain Trauma Foundation Guidelines for the Management of Severe TBI, 4th Ed. Available online: https://braintrauma.org/guidelines/guidelines-for-the-management-of-severe-tbi-4th-ed#/ (accessed on 26 August 2021).
- Ng, S.Y.; Lee, A.Y.W. Traumatic Brain Injuries: Pathophysiology and Potential Therapeutic Targets. Front. Cell. Neurosci. 2019, 13, 528. [Google Scholar] [CrossRef] [PubMed]
- Cheng, G.; Kong, R.H.; Zhang, L.M.; Zhang, J.N. Mitochondria in traumatic brain injury and mitochondrial-targeted multipotential therapeutic strategies. Br. J. Pharmacol. 2012, 167, 699–719. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belli, A.; Sen, J.; Petzold, A.; Russo, S.; Kitchen, N.; Smith, M. Clinical Article Metabolic failure precedes intracranial pressure rises in traumatic brain injury: A microdialysis study. Acta Neurochir. 2008, 150, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Chamoun, R.; Suki, D.; Gopinath, S.P.; Goodman, J.C.; Robertson, C. Role of extracellular glutamate measured by cerebral microdialysis in severe traumatic brain injury: Clinical article. J. Neurosurg. 2010, 113, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Thompson, B.M.; Gao, X.; Hall, E.D. Temporal relationship of peroxynitrite-induced oxidative damage, calpain-mediated cytoskeletal degradation and neurodegeneration after traumatic brain injury. Exp. Neurol. 2007, 205, 154–165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Semple, B.D.; Bye, N.; Rancan, M.; Ziebell, J.M.; Morganti-Kossmann, M.C. Role of CCL2 (MCP-1) in traumatic brain injury (TBI): Evidence from severe TBI patients and CCL2−/− mice. J. Cereb. Blood Flow Metab. 2010, 30, 769–782. [Google Scholar] [CrossRef]
- Okonkwo, D.O.; Shutter, L.A.; Moore, C.; Temkin, N.R.; Puccio, A.M.; Madden, C.J.; Andaluz, N.; Chesnut, R.M.; Bullock, M.R.; Grant, G.A.; et al. Brain tissue oxygen monitoring and management in severe traumatic brain injury (BOOST-II): A phase II randomized trial. Crit. Care Med. 2017, 45, 1907. [Google Scholar] [CrossRef]
- Chesnut, R.; Aguilera, S.; Buki, A.; Bulger, E.; Citerio, G.; Cooper, D.; Arrastia, R.; Diringer, M.; Figaji, A.; Gao, G.; et al. A management algorithm for adult patients with both brain oxygen and intracranial pressure monitoring: The Seattle International Severe Traumatic Brain Injury Consensus Conference (SIBICC). Intensive Care Med. 2020, 46, 919–929. [Google Scholar] [CrossRef] [Green Version]
- Carpenter, K.; Young, A.; Hutchinson, P. Advanced monitoring in traumatic brain injury: Microdialysis. Curr. Opin. Crit. Care 2017, 23, 103–109. [Google Scholar] [CrossRef]
- Ercole, A.; Magnoni, S.; Vegliante, G.; Pastorelli, R.; Surmacki, J.; Bohndiek, S.; Zanier, E. Current and Emerging Technologies for Probing Molecular Signatures of Traumatic Brain Injury. Front. Neurol. 2017, 8, 450. [Google Scholar] [CrossRef] [Green Version]
- Cordero, E.; Latka, I.; Matthäus, C.; Schie, I.; Popp, J. In-vivo Raman spectroscopy: From basics to applications. J. Biomed. Opt. 2018, 23, 071210. [Google Scholar] [CrossRef] [PubMed]
- Hanlon, E.B.; Manoharan, R.; Koo, T.W.; Shafer, K.E.; Motz, J.T.; Fitzmaurice, M.; Kramer, J.R.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Prospects for in vivo Raman spectroscopy. Phys. Med. Biol. 2000, 45, R1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demtröder, W. Laser Raman Spectroscopy. In Laser Spectroscopy2; Springer: Berlin/Heidelberg, Germany, 2015; pp. 149–181. [Google Scholar] [CrossRef]
- Bik, E.; Mateuszuk, L.; Stojak, M.; Chlopicki, S.; Baranska, M.; Majzner, K. Menadione-induced endothelial inflammation detected by Raman spectroscopy. Biochim. Biophys. Acta Mol. Cell Res. 2021, 1868, 118911. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Rhodes, J.S.; Garg, A.; Takemoto, J.Y.; Qi, X.; Harihar, S.; Tom Chang, C.W.; Moon, K.R.; Zhou, A. Label-free discrimination and quantitative analysis of oxidative stress induced cytotoxicity and potential protection of antioxidants using Raman micro-spectroscopy and machine learning. Anal. Chim. Acta 2020, 1128, 221–230. [Google Scholar] [CrossRef]
- Lalonde, J.W.; O’Connor, S.P.; Noojin, G.D.; Gonzales, C.C.; Yakovlev, V.V.; Denton, M.L. Measuring cytochrome c redox state using resonance Raman spectroscopy to determine metabolic rates in electron transport chain when exposed to light. In Mechanisms of Photobiomodulation Therapy XIV; Hamblin, M.R., Carroll, J.D., Arany, P., Eds.; SPIE: Bellingham, WA, USA, 2019; Volume 10861, p. 18. [Google Scholar] [CrossRef]
- Jung, G.; Kang, S.; Lee, G.; Kim, D. Biochemical Characterization of the Brain Hippocampal Areas after Cerebral Ischemia-Reperfusion Using Raman Spectroscopy. Appl. Spectrosc. 2018, 72, 1479–1486. [Google Scholar] [CrossRef]
- Chaichi, A.; Hasan, S.; Mehta, N.; Donnarumma, F.; Ebenezer, P.; Murray, K.; Francis, J.; Gartia, M. Label-free lipidome study of paraventricular thalamic nucleus (PVT) of rat brain with post-traumatic stress injury by Raman imaging. Analyst 2021, 146, 170–183. [Google Scholar] [CrossRef]
- Lakshmi, R.; Kartha, V.; Krishna Murali, C.; Solomon, J.; Uma Devi, P. Tissue Raman Spectroscopy for the Study of Radiation Damage: Brain Irradiation of Mice. Radiat. Res. 2002, 157, 175–182. [Google Scholar] [CrossRef]
- Devitt, G.; Howard, K.; Mudher, A.; Mahajan, S. Raman Spectroscopy: An Emerging Tool in Neurodegenerative Disease Research and Diagnosis. ACS Chem. Neurosci. 2018, 9, 404–420. [Google Scholar] [CrossRef]
- Aguiar, R.P.; Silveira, L.; Falcão, E.T.; Pacheco, M.T.T.; Zângaro, R.A.; Pasqualucci, C.A. Discriminating Neoplastic and Normal Brain Tissues in Vitro Through Raman Spectroscopy: A Principal Components Analysis Classification Model. Photomed. Laser Surg. 2013, 31, 595–604. [Google Scholar] [CrossRef]
- Desroches, J.; Jermyn, M.; Pinto, M.; Picot, F.; Tremblay, M.; Obaid, S.; Marple, E.; Urmey, K.; Trudel, D.; Soulez, G.; et al. A new method using Raman spectroscopy for in vivo targeted brain cancer tissue biopsy. Sci. Rep. 2018, 8, 1792. [Google Scholar] [CrossRef]
- Livermore, L.; Isabelle, M.; Bell, I.; Edgar, O.; Voets, N.; Stacey, R.; Ansorge, O.; Vallance, C.; Plaha, P. Raman spectroscopy to differentiate between fresh tissue samples of glioma and normal brain: A comparison with 5-ALA-induced fluorescence-guided surgery. J. Neurosurg. 2020, 135, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Koljenović, S.; Schut, T.; Vincent, A.; Kros, J.; Puppels, G. Detection of meningioma in dura mater by Raman spectroscopy. Anal. Chem. 2005, 77, 7958–7965. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Fan, Y.; He, M.; Ma, X.; Song, Y.; Liu, M.; Xu, J. Accuracy of Raman spectroscopy in differentiating brain tumor from normal brain tissue. Oncotarget 2017, 8, 36824–36831. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hollon, T.; Lewis, S.; Freudiger, C.; Sunney Xie, X.; Orringer, D. Improving the accuracy of brain tumor surgery via Raman-based technology. Neurosurg. Focus 2016, 40, E9. [Google Scholar] [CrossRef] [PubMed]
- Depciuch, J.; Tołpa, B.; Witek, P.; Szmuc, K.; Kaznowska, E.; Osuchowski, M.; Król, P.; Cebulski, J. Raman and FTIR spectroscopy in determining the chemical changes in healthy brain tissues and glioblastoma tumor tissues. Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 2020, 225, 117526. [Google Scholar] [CrossRef]
- Kalkanis, S.; Kast, R.; Rosenblum, M.; Mikkelsen, T.; Yurgelevic, S.; Nelson, K.; Raghunathan, A.; Poisson, L.; Auner, G. Raman spectroscopy to distinguish grey matter, necrosis, and glioblastoma multiforme in frozen tissue sections. J. Neurooncol. 2014, 116, 477–485. [Google Scholar] [CrossRef]
- Jermyn, M.; Mok, K.; Mercier, J.; Desroches, J.; Pichette, J.; Saint-Arnaud, K.; Bernstein, L.; Guiot, M.; Petrecca, K.; Leblond, F. Intraoperative brain cancer detection with Raman spectroscopy in humans. Sci. Transl. Med. 2015, 7, 274ra19. [Google Scholar] [CrossRef]
- Jermyn, M.; Desroches, J.; Mercier, J.; St-Arnaud, K.; Guiot, M.; Leblond, F.; Petrecca, K. Raman spectroscopy detects distant invasive brain cancer cells centimeters beyond MRI capability in humans. Biomed. Opt. Express 2016, 7, 5129. [Google Scholar] [CrossRef] [Green Version]
- Desroches, J.; Jermyn, M.; Mok, K.; Lemieux-Leduc, C.; Mercier, J.; St-Arnaud, K.; Urmey, K.; Guiot, M.; Marple, E.; Petrecca, K.; et al. Characterization of a Raman spectroscopy probe system for intraoperative brain tissue classification. Biomed. Opt. Express 2015, 6, 2380. [Google Scholar] [CrossRef] [Green Version]
- Galli, R.; Meinhardt, M.; Koch, E.; Schackert, G.; Steiner, G.; Kirsch, M.; Uckermann, O. Rapid Label-Free Analysis of Brain Tumor Biopsies by Near Infrared Raman and Fluorescence Spectroscopy—A Study of 209 Patients. Front. Oncol. 2019, 9, 1165. [Google Scholar] [CrossRef]
- Hollon, T.; Pandian, B.; Adapa, A.; Urias, E.; Save, A.; Khalsa, S.; Eichberg, D.; D’Amico, R.; Farooq, Z.; Lewis, S.; et al. Near real-time intraoperative brain tumor diagnosis using stimulated Raman histology and deep neural networks. Nat. Med. 2020, 26, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, 332–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hooijmans, C.R.; Rovers, M.M.; De Vries, R.B.M.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banbury, C.; Styles, I.; Eisenstein, N.; Zanier, E.R.; Vegliante, G.; Belli, A.; Logan, A.; Oppenheimer, P.G. Spectroscopic detection of traumatic brain injury severity and biochemistry from the retina. Biomed. Opt. Express 2020, 11, 6249–6261. [Google Scholar] [CrossRef] [PubMed]
- Banbury, C.; Mason, R.; Styles, I.; Eisenstein, N.; Clancy, M.; Belli, A.; Logan, A.; Goldberg Oppenheimer, P. Development of the Self Optimising Kohonen Index Network (SKiNET) for Raman Spectroscopy Based Detection of Anatomical Eye Tissue. Sci. Rep. 2019, 9, 10812. [Google Scholar] [CrossRef]
- Ercole, A.; Surmaki, J.; Ansel-Bollepalli, L.; Pischiutta, F.; Bohndiek, S.; Zanier, E. Mapping spatial biochemical changes in a mouse model of traumatic brain injury with Raman imaging. Intensive Care Med. Exp. 2017, 5. [Google Scholar]
- Surmacki, J.M.; Ansel-Bollepalli, L.; Pischiutta, F.; Zanier, E.R.; Ercole, A.; Bohndiek, S.E. Label-free monitoring of tissue biochemistry following traumatic brain injury using Raman spectroscopy. Analyst 2017, 142, 132. [Google Scholar] [CrossRef]
- Mowbray, M.; Banbury, C.; Rickard, J.J.S.; Davies, D.J.; Goldberg Oppenheimer, P. Development and Characterization of a Probe Device toward Intracranial Spectroscopy of Traumatic Brain Injury. ACS Biomater. Sci. Eng. 2021, 7, 1252–1262. [Google Scholar] [CrossRef]
- Kawon, K.; Setkowicz, Z.; Drozdz, A.; Janeczko, K.; Chwiej, J. The methods of vibrational microspectroscopy reveals long-term biochemical anomalies within the region of mechanical injury within the rat brain. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2021, 263, 120214. [Google Scholar] [CrossRef]
- Hu, F.; Lamprecht, M.; Wei, L.; Morrison, B.; Min, W. Bioorthogonal chemical imaging of metabolic activities in live mammalian hippocampal tissues with stimulated Raman scattering. Sci. Rep. 2016, 6, 39660. [Google Scholar] [CrossRef] [Green Version]
- Du Sert, N.P.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; Emerson, M.; et al. Reporting animal research: Explanation and elaboration for the ARRIVE guidelines 2.0. PLoS Biol. 2020, 18, e3000411. [Google Scholar] [CrossRef]
- Hackett, M.J.; Desouza, M.; Caine, S.; Bewer, B.; Nichol, H.; Paterson, P.G.; Colbourne, F. A New Method To Image Heme-Fe, Total Fe, and Aggregated Protein Levels after Intracerebral Hemorrhage. ACS Chem. Neurosci. 2015, 6, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Williamson, M.R.; Dietrich, K.; Hackett, M.J.; Caine, S.; Nadeau, C.A.; Aziz, J.R.; Nichol, H.; Paterson, P.G.; Colbourne, F. Rehabilitation Augments Hematoma Clearance and Attenuates Oxidative Injury and Ion Dyshomeostasis After Brain Hemorrhage. Stroke 2017, 48, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Dutta, A.; Gautam, R.; Chatterjee, S.; Ariese, F.; Sikdar, S.K.; Umapathy, S. Ascorbate Protects Neurons against Oxidative Stress: A Raman Microspectroscopic Study. ACS Chem. Neurosci. 2015, 6, 1794–1801. [Google Scholar] [CrossRef]
- Khatri, N.; Thakur, M.; Pareek, V.; Kumar, S.; Sharma, S.; Datusalia, A.K. Oxidative Stress: Major Threat in Traumatic Brain Injury. CNS Neurol. Disord. Drug Targets 2018, 17, 689–695. [Google Scholar] [CrossRef]
- Mehta, T.; Fayyaz, M.; Giler, G.E.; Kaur, H.; Raikwar, S.P.; Kempuraj, D.; Selvakumar, G.P.; Ahmed, M.E.; Thangavel, R.; Zaheer, S.; et al. Current Trends in Biomarkers for Traumatic Brain Injury. Open access J. Neurol. Neurosurg. 2020, 12, 86. [Google Scholar]
- Wang, K.K.; Yang, Z.H.; Zhu, T.; Shi, Y.; Rubenstein, R.; Tyndall, J.A.; Manley, G.T. An update on diagnostic and prognostic biomarkers for traumatic brain injury. Expert Rev. Mol. Diagn. 2018, 18, 165–180. [Google Scholar] [CrossRef]
- Calcagnile, O.; Anell, A.; Undén, J. The addition of S100B to guidelines for management of mild head injury is potentially cost saving. BMC Neurol. 2016, 16, 200. [Google Scholar] [CrossRef] [Green Version]
- Granger, J.H.; Schlotter, N.E.; Crawford, A.C.; Porter, M.D. Prospects for point-of-care pathogen diagnostics using surface-enhanced Raman scattering (SERS). Chem. Soc. Rev. 2016, 45, 3865–3882. [Google Scholar] [CrossRef]
- McNay, G.; Eustace, D.; Smith, W.E.; Faulds, K.; Graham, D. Surface-enhanced Raman scattering (SERS) and surface-enhanced resonance raman scattering (SERRS): A review of applications. Appl. Spectrosc. 2011, 65, 825–837. [Google Scholar] [CrossRef]
- Rickard, J.J.; Di-Pietro, V.; Smith, D.; Davies, D.; Belli, A.; Goldberg Oppenheimer, P. Rapid optofluidic detection of biomarkers for traumatic brain injury via surface-enhanced Raman spectroscopy. Nat. Biomed. Eng. 2020, 4, 610–623. [Google Scholar] [CrossRef] [PubMed]
- Osier, N.; Ziari, M.; Puccio, A.; Poloyac, S.; Okonkwo, D.; Minnigh, M.; Beers, S.; Conley, Y. Elevated cerebrospinal fluid concentrations of N-acetylaspartate correlate with poor outcome in a pilot study of severe brain trauma. Brain Inj. 2019, 33, 1364–1371. [Google Scholar] [CrossRef] [PubMed]
- Toman, E.; Harrisson, S.; Belli, T. Biomarkers in traumatic brain injury: A review. J.R. Army Med. Corps 2016, 162, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Zheng, P.; Kasani, S.; Wu, S.; Yang, F.; Lewis, S.; Nayeem, S.; Engler-Chiurazzi, E.; Wigginton, J.; Simpkins, J.; et al. Paper-Based Surface-Enhanced Raman Scattering Lateral Flow Strip for Detection of Neuron-Specific Enolase in Blood Plasma. Anal. Chem. 2017, 89, 10104–10110. [Google Scholar] [CrossRef]
- Gao, X.; Boryczka, J.; Zheng, P.; Kasani, S.; Yang, F.; Engler-Chiurazzi, E.; Simpkins, J.; Wigginton, J.; Wu, N. A “hot Spot”-Enhanced paper lateral flow assay for ultrasensitive detection of traumatic brain injury biomarker S-100β in blood plasma. Biosens. Bioelectron. 2021, 177, 112967. [Google Scholar] [CrossRef]
- Li, D.; Yang, M.; Li, H.; Mao, L.; Wang, Y.; Sun, B. SERS based protocol using flow glass-hemostix for detection of neuron-specific enolase in blood plasma. New J. Chem. 2019, 43, 5925–5931. [Google Scholar] [CrossRef]
- O’Neal, P.; Motamedi, M.; Lin, W.; Chen, J.; Coté, G. Feasibility study using surface-enhanced Raman spectroscopy for the quantitative detection of excitatory amino acids. J. Biomed. Opt. 2003, 8, 33. [Google Scholar] [CrossRef]
- O’Neal, D.P.; Motamedi, M.; Chen, J.; Cote, G.L. Surface-enhanced Raman spectroscopy for the in-vitro and ex-vivo detection of excitatory amino acids. In Biomedical Applications of Raman Spectroscopy; SPIE: Bellingham, WA, USA, 1999; Volume 3608, pp. 211–216. [Google Scholar] [CrossRef]
- Larkin, P. IR and Raman Spectra-Structure Correlations. In Infrared Raman Spectrosc; Elsevier: Amsterdam, The Netherlands, 2011; pp. 73–115. [Google Scholar] [CrossRef]
- Kang, D.G.; Woo, K.C.; Kang, D.H.; Park, C.; Kim, S.K. Improved spectral resolution of the femtosecond stimulated Raman spectroscopy achieved by the use of the 2nd-order diffraction method. Sci. Rep. 2021, 11, 3361. [Google Scholar] [CrossRef]
- Lemler, P.; Premasiri, W.R.; DelMonaco, A.; Ziegler, L.D. NIR Raman spectra of whole human blood: Effects of laser-induced and in vitro hemoglobin denaturation. Anal. Bioanal. Chem. 2014, 406, 193–200. [Google Scholar] [CrossRef]
- Thomas, D.B.; Hauber, M.E.; Hanley, D.; Waterhouse, G.I.N.; Fraser, S.; Gordon, K.C. Analysing avian eggshell pigments with Raman spectroscopy. J. Exp. Biol. 2015, 218, 2670–2674. [Google Scholar] [CrossRef] [Green Version]
- Talari, A.C.S.; Movasaghi, Z.; Rehman, S.; Rehman, I.U. Raman Spectroscopy of Biological Tissues. Appl. Spectrosc. Rev. 2014, 50, 46–111. [Google Scholar] [CrossRef]
- Tay, L.; Tremblay, R.; Hulse, J.; Zurakowski, B.; Thompson, M.; Bani-Yaghoub, M. Detection of acute brain injury by Raman spectral signature. Analyst 2011, 136, 1620–1626. [Google Scholar] [CrossRef] [PubMed]
- Girard, A.; Cooper, A.; Mabbott, S.; Bradley, B.; Asiala, S.; Jamieson, L.; Clucas, C.; Capewell, P.; Marchesi, F.; Gibbins, M.P.; et al. Raman spectroscopic analysis of skin as a diagnostic tool for Human African Trypanosomiasis. PLoS Pathog. 2021, 17, e1010060. [Google Scholar] [CrossRef]
- Journal, A.I.; Lu, M.; Zhao, L.; Wang, Y.; You, G.; Kan, X.; Zhang, Y.; Zhang, N.; Wang, B.; Guo, Y.-J.; et al. Measurement of the methemoglobin concentration using Raman spectroscopy, Artificial Cells. Nanomed. Biotechnol. 2014, 42, 63–69. [Google Scholar] [CrossRef]
- Sacharz, J.; Wesełucha-Birczyńska, A.; Zięba-Palus, J.; Lewandowski, M.H.; Kowalski, R.; Palus, K.; Chrobok, Ł.; Moskal, P.; Birczyńska, M.; Sozańska, A. Epileptic rat brain tissue analyzed by 2D correlation Raman spectroscopy. Spectrochim. Acta-Part A Mol. Biomol. Spectrosc. 2018, 188, 581–588. [Google Scholar] [CrossRef]
- Shin, H.J.; Lee, J.H.; Kim, Y.D.; Shin, I.; Sim, T.; Lim, D.K. Raman-Based in Situ Monitoring of Changes in Molecular Signatures during Mitochondrially Mediated Apoptosis. ACS Omega 2019, 4, 8188–8195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Codrich, D. Applications and Limits of Raman Spectroscopy in the Study of Colonic and Pulmonary Malformations. Ph.D. Thesis, Universita’ Degli Studi di Trieste, Trieste, Italy, 2006. [Google Scholar]
- Wood, B.R.; Caspers, P.; Puppels, G.J.; Pandiancherri, S.; McNaughton, D. Resonance Raman spectroscopy of red blood cells using near-infrared laser excitation. Anal. Bioanal. Chem. 2007, 387, 1691–1703. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.A.; Pereira, F.J.; Aller, A.J.; Littlejohn, D. Raman spectrometry as a screening tool for solvent-extracted azo dyes from polyester-based textile fibres. Polym. Test. 2020, 91, 106765. [Google Scholar] [CrossRef]
- Bankapur, A.; Krishnamurthy, R.S.; Zachariah, E.; Santhosh, C.; Chougule, B.; Praveen, B.; Valiathan, M.; Mathur, D. Micro-Raman Spectroscopy of Silver Nanoparticle Induced Stress on Optically-Trapped Stem Cells. PLoS ONE 2012, 7, e35075. [Google Scholar] [CrossRef]
- Yao, H.; Tao, Z.; Ai, M.; Peng, L.; Wang, G.; He, B.; Li, Y.-Q. Raman spectroscopic analysis of apoptosis of single human gastric cancer cells. Vib. Spect. 2009, 50, 193–197. [Google Scholar] [CrossRef]
- Notingher, I.; Green, C.; Dyer, C.; Perkins, E.; Hopkins, N.; Lindsay, C.; Hench, L. Discrimination between ricin and sulphur mustard toxicity in vitro using Raman spectroscopy. J.R. Soc. Interface 2004, 1, 79–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krafft, C.; Knetschke, T.; Funk, R.; Salzer, R. Studies on stress-induced changes at the subcellular level by Raman microspectroscopic mapping. Anal. Chem. 2006, 78, 4424–4429. [Google Scholar] [CrossRef] [PubMed]
- Muthuselvi, C.; Pandiaraja, S.S.; Ravikumar, B.; Athimoolam, S.; Srinivasan, N.; Krishnakum, R.V. FT-IR and FT-Raman Spectroscopic Analyzes of Indeno Quinoxaline Derivative Crystal. Asian J. Appl. Sci. 2018, 11, 83–91. [Google Scholar] [CrossRef] [Green Version]
- Kumar, V.; Panikar, Y.; Palafox, M.A.; Vats, J.; Kostova, I.; Lang, K.; Rastogi, V. Ab-initio calculations, FT-IR and FT-Raman spectra of 2-chloro-6-methyl benzonitrile. Indian J. Pure Appl. Phys. 2010, 48, 85–94. [Google Scholar]
- Krafft, C.; Neudert, L.; Simat, T.; Salzer, R. Near infrared Raman spectra of human brain lipids. Spectrochim. Acta-Part A Mol. Biomol. Spectrosc. 2005, 61, 1529–1535. [Google Scholar] [CrossRef] [PubMed]
- Farquharson, S.; Gift, A.; Shende, C.; Inscore, F.; Ordway, B.; Farquharson, C.; Murren, J. Surface-enhanced Raman Spectral Measurements of 5-Fluorouracil in Saliva. Molecules 2008, 13, 2608. [Google Scholar] [CrossRef] [PubMed]
- De Gelder, J.; De Gussem, K.; Vandenabeele, P.; Moens, L. Reference database of Raman spectra of biological molecules. J. Raman Spectrosc. 2007, 38, 1133–1147. [Google Scholar] [CrossRef]
- Staniszewska-Slezak, E.; Malek, K.; Baranska, M. Complementary analysis of tissue homogenates composition obtained by Vis and NIR laser excitations and Raman spectroscopy. Spectrochim. Acta-Part A Mol. Biomol. Spectrosc. 2015, 147, 245–256. [Google Scholar] [CrossRef]
- Clemens, G.; Butler, H.J.; Ashton, K.M.; Brodbelt, A.; Dawson, T.P.; Fullwood, L.M.; Jenkinson, M.D.; Stables, R.; Baker, M.J.; North, L.; et al. Feature Driven Classification of Raman Spectra for Real-Time Spectral Brain Tumour Diagnosis Using Sound. Analyst 2017, 142, 98–109. [Google Scholar]
- Acosta-Maeda, T.E. Raman Spectroscopy for Planetary Exploration and Characterization of Extraterrestrial Materials. Ph.D. Thesis, University of Hawai’i at Manoa, Honolulu, HI, USA, 2016. [Google Scholar]
- Mccreery, R.L.; Chalmers, J.M.; Griffiths, P.R. Photometric Standards for Raman Spectroscopy. In Handbook of Vibrational Spectroscopy Photometric Standards for Raman Spectroscopy; John Wiley & Sons Ltd: Chichester, UK, 2002. [Google Scholar]
- Demediuk, P.; Faden, A.I.; Romhanyi, R.; Vink, R.; Mcintosh, T.K. Traumatic Brain Injury in the Rat: Effects on Lipid Metabolism, Tissue Magnesium, and Water Content. J. Neurotrauma 1988, 5, 105–119. [Google Scholar] [CrossRef]
- Lamade, A.M.; Anthonymuthu, T.S.; Hier, Z.E.; Gao, Y.; Kagan, V.E.; Bayır, H. Mitochondrial Damage & Lipid Signaling in Traumatic Brain Injury HHS Public Access. Exp. Neurol. 2020, 329, 113307. [Google Scholar] [CrossRef] [PubMed]
- Mallah, K.; Quanico, J.; Trede, D.; Kobeissy, F.; Zibara, K.; Salzet, M.; Fournier, I. Lipid Changes Associated with Traumatic Brain Injury Revealed by 3D MALDI-MSI. Anal. Chem. 2018, 90, 10568–10576. [Google Scholar] [CrossRef] [PubMed]
- Tashibu, K. Analysis of water content in rat brain using Raman spectroscopy. Brain Nerve 1990, 42, 999–1004. [Google Scholar]
- Kitajima, T.; Tashibu, K.; Tani, S.; Mizuno, A.; Nakamura, N. Analysis of water content in young rats brain edema by Raman spectroscopy. No Shinkei 1993, 45, 519–524. [Google Scholar]
- Wolthuis, R.; van Aken, M.; Fountas, K.; Robinson, J.S.; Bruining, H.A.; Puppels, G.J. Determination of water concentration in brain tissue by Raman spectroscopy. Anal. Chem. 2001, 73, 3915–3920. [Google Scholar] [CrossRef] [PubMed]
- Desroches, J.; Lemoine, É.; Pinto, M.; Marple, E.; Urmey, K.; Diaz, R.; Guiot, M.; Wilson, B.; Petrecca, K.; Leblond, F. Development and first in-human use of a Raman spectroscopy guidance system integrated with a brain biopsy needle. J. Biophotonics 2019, 12, e201800396. [Google Scholar] [CrossRef]
- Seule, M.; Sikorski, C.; Sakowitz, O.; von Campe, G.; Santos, E.; Orakcioglu, B.; Unterberg, A.; Keller, E. Evaluation of a New Brain Tissue Probe for Intracranial Pressure, Temperature, and Cerebral Blood Flow Monitoring in Patients with Aneurysmal Subarachnoid Hemorrhage. Neurocrit. Care 2016, 25, 193–200. [Google Scholar] [CrossRef] [Green Version]
- Tuschel, D. Selecting an Excitation Wavelength for Raman Spectroscopy. Spectroscopy 2016, 31, 14–23. [Google Scholar]
- Signoretti, S.; Marmarou, A.; Aygok, G.; Fatouros, P.; Portella, G.; Bullock, R. Assessment of mitochondrial impairment in traumatic brain injury using high-resolution proton magnetic resonance spectroscopy. J. Neurosurg. 2008, 108, 42–52. [Google Scholar] [CrossRef]
- Adjunctive therapies in lifethreatening CNS infections. Eur. J. Neurol. 2015, 22, 19. [CrossRef]
- Nordström, C.; Nielsen, T.; Schalén, W.; Reinstrup, P.; Ungerstedt, U. Biochemical indications of cerebral ischaemia and mitochondrial dysfunction in severe brain trauma analysed with regard to type of lesion. Acta Neurochir. 2016, 158, 1231–1240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stovell, M.; Mada, M.; Helmy, A.; Carpenter, T.; Thelin, E.; Yan, J.; Guilfoyle, M.; Jalloh, I.; Howe, D.; Grice, P.; et al. The effect of succinate on brain NADH/NAD + redox state and high energy phosphate metabolism in acute traumatic brain injury. Sci. Rep. 2018, 8, 11140. [Google Scholar] [CrossRef] [PubMed]
- Sonig, A.; Jumah, F.; Raju, B.; Patel, N.V.; Gupta, G.; Nanda, A. The Historical Evolution of Intracranial Pressure Monitoring. World Neurosurg. 2020, 138, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Tas, J.; Beqiri, E.; van Kaam, C.; Ercole, A.; Bellen, G.; Bruyninckx, D.; Cabeleira, M.; Czosnyka, M.; Depreitere, B.; Donnelly, J.; et al. An Update on the COGiTATE Phase II Study: Feasibility and Safety of Targeting an Optimal Cerebral Perfusion Pressure as a Patient-Tailored Therapy in Severe Traumatic Brain Injury. Acta Neurochir. Suppl. 2021, 131, 143–147. [Google Scholar] [CrossRef] [PubMed]
- DePaoli, D.; Lemoine, E.; Ember, K.; Parent, M.; Prud’homme, M.; Cantin, L.; Petrecca, K.; Leblond, F.; Côté, D. Rise of Raman spectroscopy in neurosurgery: A review. J. Biomed. Opt. 2020, 25, 050901. [Google Scholar] [CrossRef] [PubMed]
- Stevens, A.R.; Ng, I.H.X.; Helmy, A.; Hutchinson, P.J.A.; Menon, D.K.; Ercole, A. Glucose Dynamics of Cortical Spreading Depolarization in Acute Brain Injury: A Systematic Review. J. Neurotrauma 2019, 36, 2153–2166. [Google Scholar] [CrossRef]
- Teasdale, G.; Maas, A.; Lecky, F.; Manley, G.; Stocchetti, N.; Murray, G. The Glasgow Coma Scale at 40 years: Standing the test of time. Lancet Neurol. 2014, 13, 844–854. [Google Scholar] [CrossRef]
- Echemendia, R.J.; Meeuwisse, W.; McCrory, P.; Davis, G.A.; Putukian, M.; Leddy, J.; Makdissi, M.; Sullivan, S.J.; Broglio, S.P.; Raftery, M.; et al. The sport concussion assessment tool 5th edition (SCAT5): Background and rationale. Br. J. Sports Med. 2017, 51, 851–858. [Google Scholar] [CrossRef] [Green Version]
- Gill, M.R.; Reiley, D.G.; Green, S.M. Interrater reliability of Glasgow Coma Scale scores in the emergency department. Ann. Emerg. Med. 2004, 43, 215–223. [Google Scholar] [CrossRef]
- Stevens, A.R.; Su, Z.; Toman, E.; Belli, A.; Davies, D. Optical pupillometry in traumatic brain injury: Neurological pupil index and its relationship with intracranial pressure through significant event analysis. Brain Inj. 2019, 33, 1032–1038. [Google Scholar] [CrossRef]
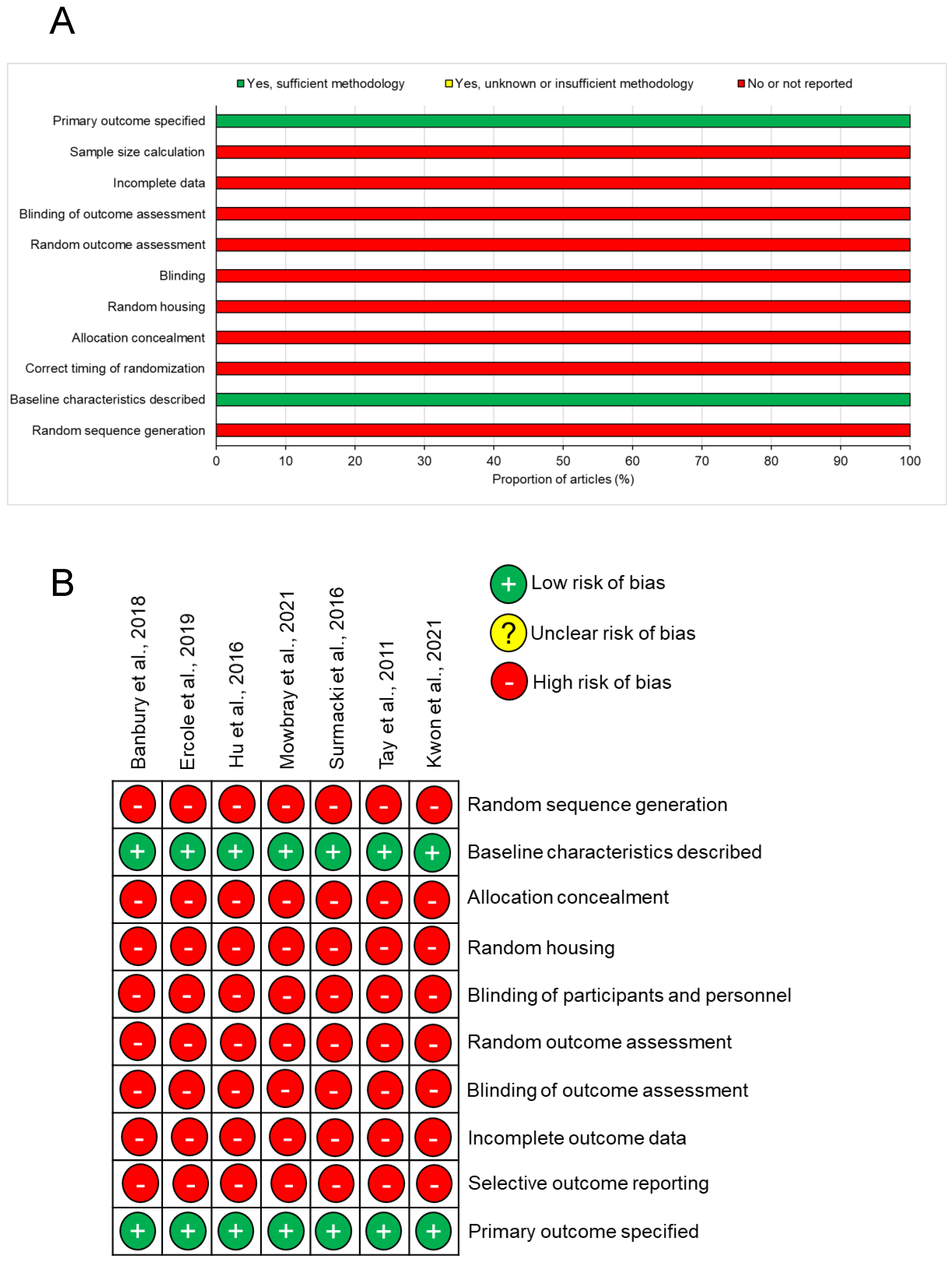

| Author | Year | Type | Animal | Model Used | Raman Technique (Wavelength) | Overall Risk of Bias |
|---|---|---|---|---|---|---|
| Banbury et al. | 2020 | Article | Mouse | In vivo CCI mTBI/sTBI | Confocal RS (785 nm) | Moderate |
| Ercole et al. | 2017 | Abstract | Mouse | In vivo CCI sTBI | Confocal RS imaging (785 nm) | Moderate |
| Hu et al. | 2016 | Article | Rat | Stretch organotypic hippocampal slice cultures | Stimulated Raman scattering (720–990 nm pump beam, 1064 nm Stokes laser) | Moderate |
| Mowbray et al. | 2021 | Article | Rat | In vivo CCI sTBI | Confocal RS (785 nm) | Moderate |
| Surmacki et al. | 2016 | Article | Mouse | In vivo CCI | Confocal RS (785 nm) | Moderate |
| Tay et al. | 2011 | Article | Mouse | In vivo CCI | Confocal RS (785 nm) | Moderate |
| Kawon et al. | 2021 | Article | Rat | In vivo drill focal injury | Confocal RS (532 nm)—Additionally, FTIR-only Raman findings reported in this article | Moderate |
| Peak Wavenumber (cm−1) | Appropriation | TBI |
|---|---|---|
| 426 | Hb [50] Cholesterol [50] | Increasing in contusion core between 2 and 7 days [50] |
| 491-2 | Combination modes of the uracil ring plus ribose vibrations [92] | Increasing in contusion core between 2 and 7 days [50] |
| 605 | Cholesterol [50] | Increased in contusion core vs. contralateral hemisphere [50] |
| 675 | Hb [50], glycerol [93] | Decreasing in contusion core between 2 and 7 days [50] |
| 701-2 | Lipids (701 cm−1) [50], cholesterol, cholesterol ester | Increasing in contusion core between 2 and 7 days [50] |
| 718 | Symmetric and anti-symmetric stretch vibrations of the choline group N+(CH3)3 in phospholipid, lipids [50] | Decreased in contusion core vs. contralateral hemisphere (2 and 7 days) [50] |
| 725 | nucleic acid (A) [57,94] | Reduced oxidative stress [57] |
| 754 | Hb [50] | Increased in contusion core vs. contralateral hemisphere (2 and 7 days) [50], decreasing in contusion core between 2 and 7 days [50] |
| 782 | DNA peak, cytosine (C), uracil (U), thymine (T), C, T, U-ring breathing [57,95] | Reduced oxidative stress [57] |
| 801 | Cyclohexane [74,75,98] (532 nm [96], 575 nm [73] and 785 nm [97] excitation) | Increased in contusion core vs. contralateral hemisphere [50] |
| 850-2 | C-H wagging [72] (850 cm−1), C−C stretch in proline (collagen) [94]; ring breathing (tyrosine) [94]; C−O−C stretching (glycogen, polysaccharides) [57,94], albumin (852 cm−1) [50] | Increased in mTBI and sTBI (3 days) [47] |
| 970 | Intralipid [50] haem aggregation marker bands, fibrin components [74]; C-C stretching mixed with C-H rocking, biliverdin, protoporphyrin IX [75] | Decreasing in contusion core between 2 and 7 days [50] |
| 1002 | Ring breathing mode of phenylalanine (1002 cm−1) [50] and (1005 cm−1) [57] | Decreasing in contusion core between 2 and 7 days [50], decreased in sTBI (3 days) [47,51], decreasing in contusion core between 2 and 7 days [50], weak or absent in injured samples [77] |
| 1003 | C-C skeletal [72], phenylalanine [76,94], Hb, albumin [50] | Decreasing in contusion core between 2 and 7 days [50], strong signal in pericontusional tissue (days 2 and 7) [49] |
| 1079 | Intralipid [50] | Decreasing in contusion core between 2 and 7 days [50] |
| 1090 | PO2 stretch, phospholipids and nucleic acid [77] | Stable in oxidative stress [57] |
| 1096-8 | Phosphodioxy PO2 [78], nucleic acid (1097 cm−1) [94] | Increased in mTBI and sTBI (3 days) [47] |
| 1154 | Phenylalanine, tryptophan, hypro, tyr, phe, m(CC/CN) proteins (1155 cm−1) [94] and (1156 cm−1) [57] | Decreasing in contusion core between 2 and 7 days [50] |
| 1175 | Amide III vibration, cholesterol [77] | Band appears after injury [77] |
| 1224 | Hb [50] | Increased in contusion core vs. contralateral hemisphere (2 days) [50], decreasing in contusion core between 2 and 7 days [50] |
| 1227-8 | Amide III vibration, phospholipid [77] (1227 cm−1); C-H methine bending vibration with changing methaemaglobin content (1228 cm−1) [79] | Increased in contusion core vs. contralateral hemisphere [50], band after injury [77] |
| 1266 | CH bending modes [72]: amide groups in lipids and proteins [76], cardiolipin [47] | Decreased in mTBI and sTBI (3 days) [47,51], decreased in contusion core vs. contralateral hemisphere (2 and 7 days) [50] |
| 1301 | CH2 twist/wag/deformation: phospholipid, mixed fatty acid chains, mixed amide III protein vibration [77], lipids [50] | Increasing in contusion core between 2 and 7 days [50], reduction in peak in ipsilateral corpus callosum and internal capsule (distant from lesion) at 2 days, partially recovering by 7 days [49] |
| 1320 | CH2 CH3 twisting; Proteins/lipids nucleic acid [57] | Reduced oxidative stress [57] |
| 1337-1340 | Nucleic acid (A,G) [57]; b protein CH2 deformation [72], amide III [76] (1337 cm−1); collagen (1338 cm−1) [94]; albumin(1339 cm−1) [50]; CH2 scissors, C-OH bending [80] | Reduced in oxidative stress [57], increased in sTBI (3 days) [47,51] |
| 1420 | DNA peak, nucleic acid (A, G) [57]; -CH2 bending mode for proteins and lipids, indicates change in cytochrome c, intracellular environment, and mitochondrial membrane structures [81] | Reduced oxidative stress [57] |
| 1440 | CH2 twisting and bending [51,82,96], Lipid, cholesterol, phospholipid, mixed amide I protein, tyrosine [80,94] | Initially decreased in contusion core vs. contralateral hemisphere (2 and 7 days), increasing progressively in contralateral hemisphere and contusion core between 2 and 7 days, to above control levels [50], reduced after ischaemic injury |
| 1447-50 | CH2 bending [72], mixed proteins and lipids [76] (1447 cm−1) [94], albumin, CH2 deformation from lipids and proteins (1450 cm−1) [77] | Increased in sTBI (3 days) [47,51] |
| 1462 | Lipids [50] | Increasing in contralateral hemisphere between 2 and 7 days [50] |
| 1547 | Hb [50] | Decreased in contusion core vs. contralateral hemisphere (2 and 7 days), decreasing progressively over time [50] |
| 1560 | Associated with mitochondrial activity of cells [77] | Strong signal in pericontusional tissue (days 2 and 7) [49] |
| 1562 | Hb [50] | Increased in contusion core vs. contralateral hemisphere (2 days resolving by 7 days) [50] |
| 1576 | DNA peak, nucleic acid (A, G) [57]; Hb in red blood cells [82] (though not identified by dedicated erythrocyte RS at 785 nm [83]; N = N stretching vibration [84] | Reduced oxidative stress [57] |
| 1586 | Albumin [50] | Sharp band after injury [77] |
| 1618-1620 | Predominantly C = O stretch in protein, Hb, Amide I (1618 cm−1) [77], tyrosine/tryptophan [94] | Sharp band after injury [77] |
| 1620 | Hb [50] | Increased in contusion core vs. contralateral hemisphere (2 days, resolving by 7 days) [50] |
| 1648 | Predominantly C = O stretch in protein, Amide I [57] | Decreased in contusion core vs. contralateral hemisphere (2 and 7 days), increasing progressively in contralateral hemisphere over time [50] |
| 1660 | Lipids [50], C = C stretching [72,76,82], amide I vibration [57,77], Tyrosine [80,94], mixed lipids and proteins [76], alpha-helix/random coil [94], cardiolipin [47] | Decreased in mTBI and sTBI (3 days) [47,51], decreased in contusion core vs. contralateral hemisphere (2 and 7 days) increasing progressively in contralateral hemisphere and contusion core between 2 and 7 days [50,77]. Increased after ischaemic injury. |
| 1670 | Cholesterol, C = C stretching [50] | Marginal increase in contusion core vs. contralateral to lesion [50] |
| Metrics cm−1/cm−1 | Remark | Changes in Animal Models |
|---|---|---|
| 701/718 | Cholesterol/phospholipid | Statistically significant difference between the contralateral and pericontusional/contusional regions at 7 days after TBI [50] |
| 1301/1250 | Mixed fatty acid chains/amide III protein | Statistically significant difference between the contralateral and pericontusional/contusional regions at 7 days after TBI [50] |
| 1440/1660 | CH deformation/mixed amide I protein and C=C stretching of lipids | Statistically significant difference between the contralateral and pericontusional/contusional regions at 2, 3, and 7 days after TBI [48,51,52] |
| 1301/1620 | Mixed amide III protein and fatty acid chains/haemoglobin | Statistically significant difference between the contralateral and pericontusional/contusional regions at 7 days after TBI [50] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stevens, A.R.; Stickland, C.A.; Harris, G.; Ahmed, Z.; Goldberg Oppenheimer, P.; Belli, A.; Davies, D.J. Raman Spectroscopy as a Neuromonitoring Tool in Traumatic Brain Injury: A Systematic Review and Clinical Perspectives. Cells 2022, 11, 1227. https://doi.org/10.3390/cells11071227
Stevens AR, Stickland CA, Harris G, Ahmed Z, Goldberg Oppenheimer P, Belli A, Davies DJ. Raman Spectroscopy as a Neuromonitoring Tool in Traumatic Brain Injury: A Systematic Review and Clinical Perspectives. Cells. 2022; 11(7):1227. https://doi.org/10.3390/cells11071227
Chicago/Turabian StyleStevens, Andrew R., Clarissa A. Stickland, Georgia Harris, Zubair Ahmed, Pola Goldberg Oppenheimer, Antonio Belli, and David J. Davies. 2022. "Raman Spectroscopy as a Neuromonitoring Tool in Traumatic Brain Injury: A Systematic Review and Clinical Perspectives" Cells 11, no. 7: 1227. https://doi.org/10.3390/cells11071227
APA StyleStevens, A. R., Stickland, C. A., Harris, G., Ahmed, Z., Goldberg Oppenheimer, P., Belli, A., & Davies, D. J. (2022). Raman Spectroscopy as a Neuromonitoring Tool in Traumatic Brain Injury: A Systematic Review and Clinical Perspectives. Cells, 11(7), 1227. https://doi.org/10.3390/cells11071227








