Biomarkers Associated with Cardiovascular Disease in COVID-19
Abstract
1. Introduction
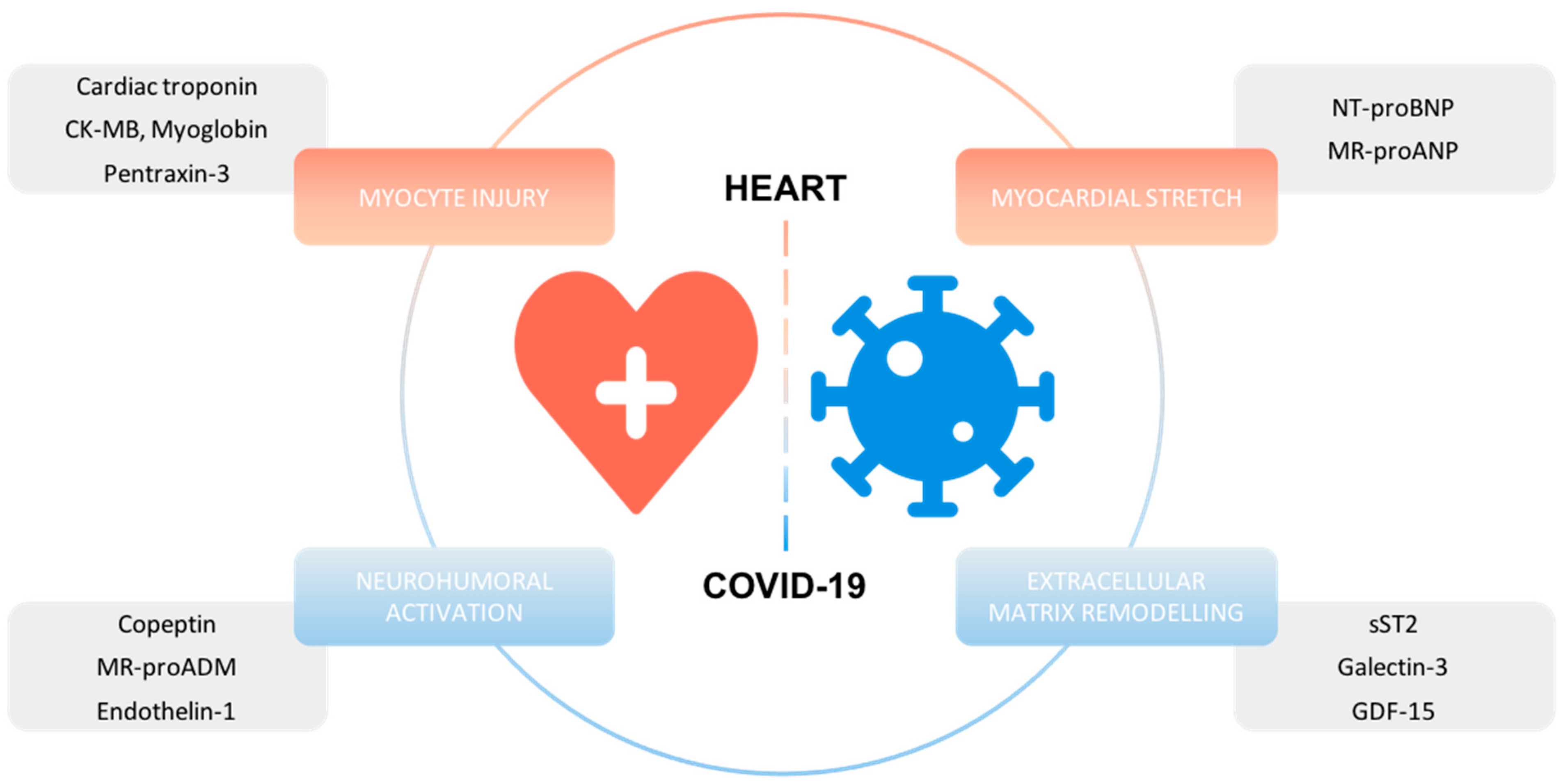
2. Biomarkers of Myocyte Injury
2.1. High-Sensitive Cardiac Troponin, Creatine Kinase and Myoglobin
2.2. Pentraxin-3
3. Biomarkers of Myocardial Stretch
3.1. BNP/NT-proBNP
3.2. MR-proANP
4. Biomarkers of Extracellular Matrix Remodeling
4.1. SST2
4.2. Galectin-3
4.3. GDF-15
5. Biomarkers of Neurohumoral Activation
5.1. Copeptin
5.2. MR-proADM
5.3. Endothelin-1
6. Other Biomarkers
Osteopontin/(A)Symetric Dimethlyarginine (ADMA/SDMA)/Myeloperoxidase
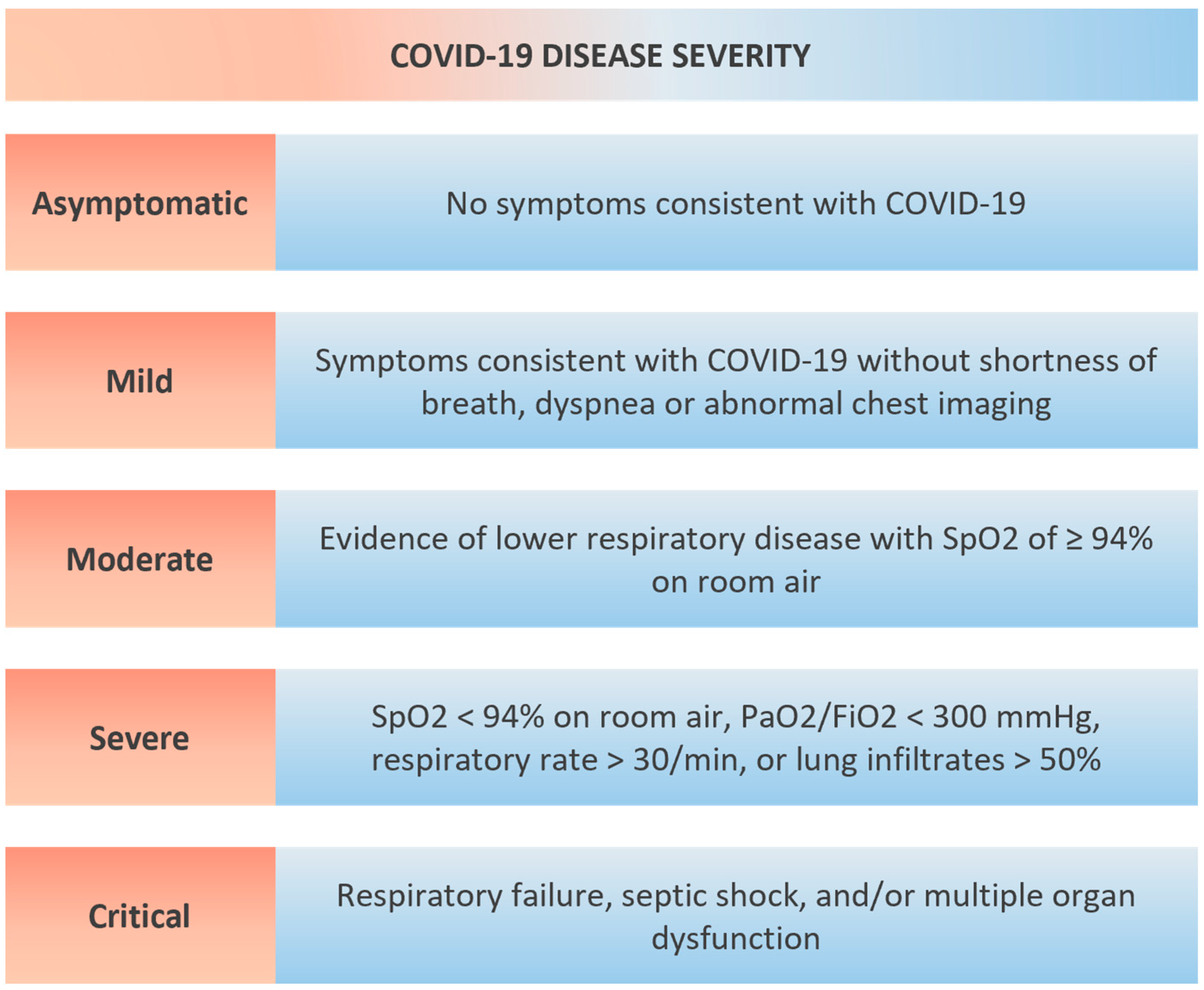
7. Routine Measurement of Cardiac Biomarker in COVID-19?
- While cardiac biomarkers have been proven to provide prognostic information in patients with COVID-19, questions remain about the incremental prognostic value upon addition of these biomarkers to established clinical risk models.
- Asymptomatic increases in biomarkers (e.g., hs-cTn or NT-proBNP) may lead to unnecessary diagnostic work-up and may pose the involved health care personnel to an additional risk of infection.
- In case of myocardial injury, a clear correlation to symptoms and signs of myocardial ischemia (chest pain, ECG and echocardiogram) is necessary to appropriately guide management decisions. If myocardial ischemia is found, patients should be treated as acute coronary syndromes in line with current guidelines with a special focus on the safety of associated health care personnel.
- Future research on the potential pathophysiological mechanisms linking cardiovascular disease to COVID-19 is necessary to further define and improve knowledge on the prognostic value of cardiac biomarkers in these patients.
- There is a significant lack of direct comparisons of cardiac biomarkers in patients with COVID-19, which is a necessity to ultimately define the “best” biomarker for risk prediction in this patient population.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fauci, A.S.; Lane, H.C.; Redfield, R.R. COVID-19—Navigating the uncharted. N. Engl. J. Med. 2020, 382, 1268–1269. [Google Scholar] [CrossRef] [PubMed]
- Parasher, A. COVID-19: Current understanding of its pathophysiology, clinical presentation and treatment. Postgrad. Med. J. 2020, 97, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Peeri, N.C.; Shrestha, N.; Rahman, M.S.; Zaki, R.; Tan, Z.; Bibi, S.; Baghbanzadeh, M.; Aghamohammadi, N.; Zhang, W.; Haque, U. The SARS, MERS and novel coronavirus (COVID-19) epidemics, the newest and biggest global health threats: What lessons have we learned? Int. J. Epidemiol. 2020, 49, 717–726. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 10 January 2022).
- World Health Organization (WHO). Tracking SARS-CoV-2 Variants. Available online: https://www.who.int/emergencies/what-we-do/tracking-SARS-CoV-2-variants (accessed on 10 January 2022).
- National Institutes of Health. COVID-19. Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. Available online: https://www.covid19treatmentguidelines.nih.gov (accessed on 10 January 2022).
- Fox, S.E.; Akmatbekov, A.; Harbert, J.L.; Li, G.; Brown, J.Q.; Heide, R.S.V. Pulmonary and cardiac pathology in African American patients with COVID-19: An autopsy series from New Orleans. Lancet Respir. Med. 2020, 8, 681–686. [Google Scholar] [CrossRef]
- Tavazzi, G.; Pellegrini, C.; Maurelli, M.; Belliato, M.; Sciutti, F.; Bottazzi, A.; Sepe, P.A.; Resasco, T.; Camporotondo, R.; Bruno, R.; et al. Myocardial localization of coronavirus in COVID-19 cardiogenic shock. Eur. J. Heart Fail. 2020, 22, 911–915. [Google Scholar] [CrossRef] [PubMed]
- Schaller, T.; Hirschbühl, K.; Burkhardt, K.; Braun, G.; Trepel, M.; Märkl, B.; Claus, R. Postmortem examination of patients with COVID-19. JAMA 2020, 323, 2518–2520. [Google Scholar] [CrossRef]
- Ruan, Q.; Yang, K.; Wang, W.; Jiang, L.; Song, J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020, 46, 846–848. [Google Scholar] [CrossRef]
- Shi, S.; Qin, M.; Shen, B.; Cai, Y.; Liu, T.; Yang, F.; Gong, W.; Liu, X.; Liang, J.; Zhao, Q.; et al. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China. JAMA Cardiol. 2020, 5, 802. [Google Scholar] [CrossRef]
- Lindner, D.; Fitzek, A.; Bräuninger, H.; Aleshcheva, G.; Edler, C.; Meissner, K.; Scherschel, K.; Kirchhof, P.; Escher, F.; Schultheiss, H.-P.; et al. Association of cardiac infection with SARS-CoV-2 in confirmed COVID-19 autopsy cases. JAMA Cardiol. 2020, 5, 1281–1285. [Google Scholar] [CrossRef]
- Chung, M.K.; Zidar, D.A.; Bristow, M.R.; Cameron, S.J.; Chan, T.; Iii, C.V.H.; Kwon, D.H.; Singh, T.; Tilton, J.C.; Tsai, E.J.; et al. COVID-19 and cardiovascular disease: From bench to bedside. Circ. Res. 2021, 128, 1214–1236. [Google Scholar] [CrossRef]
- Baigent, C.; Windecker, S.; Andreini, D.; Arbelo, E.; Barbato, E.; Bartorelli, A.L.; Baumbach, A.; Behr, E.R.; Berti, S.; The Task Force for the Management of COVID-19 of the European Society of Cardiology; et al. European Society of Cardiology guidance for the diagnosis and management of cardiovascular disease during the COVID-19 pandemic: Part 1—Epidemiology, pathophysiology, and diagnosis. Eur. Heart J. 2021, ehab696. [Google Scholar] [CrossRef]
- Brown, C.C.; Young, S.G.; Pro, G.C. COVID-19 vaccination rates vary by community vulnerability: A county-level analysis. Vaccine 2021, 39, 4245–4249. [Google Scholar] [CrossRef] [PubMed]
- Kaur, R.J.; Dutta, S.; Bhardwaj, P.; Charan, J.; Dhingra, S.; Mitra, P.; Singh, K.; Yadav, D.; Sharma, P.; Misra, S. Adverse events reported from COVID-19 vaccine trials: A systematic review. Indian J. Clin. Biochem. 2021, 36, 427–439. [Google Scholar] [CrossRef] [PubMed]
- Klein, N.P.; Lewis, N.; Goddard, K.; Fireman, B.; Zerbo, O.; Hanson, K.E.; Donahue, J.G.; Kharbanda, E.O.; Naleway, A.; Nelson, J.C.; et al. Surveillance for adverse events after COVID-19 mRNA vaccination. JAMA 2021, 326, 1390–1399. [Google Scholar] [CrossRef]
- Diaz, G.A.; Parsons, G.T.; Gering, S.K.; Meier, A.R.; Hutchinson, I.V.; Robicsek, A. Myocarditis and pericarditis after vaccination for COVID-19. JAMA 2021, 326, 1210. [Google Scholar] [CrossRef]
- Witberg, G.; Barda, N.; Hoss, S.; Richter, I.; Wiessman, M.; Aviv, Y.; Grinberg, T.; Auster, O.; Dagan, N.; Balicer, R.D.; et al. Myocarditis after Covid-19 vaccination in a large health care organization. N. Engl. J. Med. 2021, 385, 2132–2139. [Google Scholar] [CrossRef]
- Cabaniss, C.D. Creatine Kinase; Butterworths: Kidlington, UK, 1990. [Google Scholar]
- Stone, M.J.; Waterman, M.R.; Harimoto, D.; Murray, G.; Willson, N.; Platt, M.R.; Blomqvist, G.; Willerson, J.T. Serum myoglobin level as diagnostic test in patients with acute myocardial infarction. Heart 1977, 39, 375–380. [Google Scholar] [CrossRef]
- Yamasaki, K.; Kurimura, M.; Kasai, T.; Sagara, M.; Kodama, T.; Inoue, K. Determination of physiological plasma pentraxin 3 (PTX3) levels in healthy populations. Clin. Chem. Lab. Med. (CCLM) 2009, 47, 471–477. [Google Scholar] [CrossRef]
- Fradley, M.G.; Larson, M.; Cheng, S.; McCabe, E.; Coglianese, E.; Shah, R.V.; Levy, D.; Vasan, R.S.; Wang, T.J. Reference limits for N-terminal-pro-B-type natriuretic peptide in healthy individuals (from the Framingham heart study). Am. J. Cardiol. 2011, 108, 1341–1345. [Google Scholar] [CrossRef]
- Tzikas, S.; Keller, T.; Wild, P.S.; Schulz, A.; Zwiener, I.; Zeller, T.; Schnabel, R.B.; Sinning, C.; Lubos, E.; Kunde, J.; et al. Midregional pro-atrial natriuretic peptide in the general population/Insights from the Gutenberg health study. Clin. Chem. Lab. Med. (CCLM) 2012, 51, 1125–1133. [Google Scholar] [CrossRef]
- Lu, J.; Snider, J.V.; Grenache, D.G. Establishment of reference intervals for soluble ST2 from a United States population. Clin. Chim. Acta 2010, 411, 1825–1826. [Google Scholar] [CrossRef] [PubMed]
- Iurisci, I.; Tinari, N.; Natoli, C.; Angelucci, D.; Cianchetti, E.; Iacobelli, S. Concentrations of galectin-3 in the sera of normal controls and cancer patients. Clin. Cancer Res. 2000, 6, 1389–1393. [Google Scholar] [PubMed]
- Hamon, S.M.; Griffin, T.P.; Islam, N.; Wall, D.; Griffin, M.; O’Shea, P.M. Defining reference intervals for a serum growth differentiation factor-15 (GDF-15) assay in a Caucasian population and its potential utility in diabetic kidney disease (DKD). Clin. Chem. Lab. Med. (CCLM) 2018, 57, 510–520. [Google Scholar] [CrossRef]
- Morgenthaler, N.G.; Struck, J.; Alonso, C.; Bergmann, A. Assay for the measurement of copeptin, a stable peptide derived from the precursor of vasopressin. Clin. Chem. 2006, 52, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Lorubbio, M.; Conti, A.A.; Ognibene, A. Midregional pro-adrenomedullin (MR-ProADM) reference values in serum. Clin. Biochem. 2018, 53, 173–174. [Google Scholar] [CrossRef]
- Bossard, M.; Pumpol, K.; van der Lely, S.; Aeschbacher, S.; Schoen, T.; Krisai, P.; Lam, T.; Todd, J.; Estis, J.; Risch, M.; et al. Plasma endothelin-1 and cardiovascular risk among young and healthy adults. Atherosclerosis 2015, 239, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Collet, J.-P.; Thiele, H.; Barbato, E.; Barthélémy, O.; Bauersachs, J.; Bhatt, D.L.; Dendale, P.; Dorobantu, M.; Edvardsen, T.; Folliguet, T.; et al. 2020 ESC guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: The Task Force for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation of the European Society of Cardiology (ESC). Eur. Heart J. 2021, 42, 1289–1367. [Google Scholar] [CrossRef]
- Chapman, A.; Lee, K.K.; McAllister, D.A.; Cullen, L.; Greenslade, J.H.; Parsonage, W.; Worster, A.; Kavsak, P.A.; Blankenberg, S.; Neumann, J.; et al. Association of high-sensitivity cardiac troponin I concentration with cardiac outcomes in patients with suspected acute coronary syndrome. JAMA 2017, 318, 1913–1924. [Google Scholar] [CrossRef]
- Kvisvik, B.; Mørkrid, L.; Røsjø, H.; Cvancarova, M.; Rowe, A.D.; Eek, C.; Bendz, B.; Edvardsen, T.; Gravning, J. High-sensitivity troponin T vs. I in acute coronary syndrome: Prediction of significant coronary lesions and long-term prognosis. Clin. Chem. 2017, 63, 552–562. [Google Scholar] [CrossRef]
- Alcalai, R.; Planer, D.; Culhaoglu, A.; Osman, A.; Pollak, A.; Lotan, C. Acute coronary syndrome vs. nonspecific troponin elevation: Clinical predictors and survival analysis. Arch. Intern. Med. 2007, 167, 276–281. [Google Scholar] [CrossRef]
- Landesberg, G.; Jaffe, A.S.; Gilon, D.; Levin, P.D.; Goodman, S.; Abu-Baih, A.; Beeri, R.; Weissman, C.; Sprung, C.L.; Landesberg, A. Troponin elevation in severe sepsis and septic shock: The role of left ventricular diastolic dysfunction and right ventricular dilatation. Crit. Care Med. 2014, 42, 790–800. [Google Scholar] [CrossRef] [PubMed]
- Lala, A.; Johnson, K.W.; Januzzi, J.L.; Russak, A.J.; Paranjpe, I.; Richter, F.; Zhao, S.; Somani, S.; Van Vleck, T.; Vaid, A.; et al. Prevalence and impact of myocardial injury in patients hospitalized with COVID-19 infection. J. Am. Coll. Cardiol. 2020, 76, 533–546. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Baigent, C.; Windecker, S.; Andreini, D.; Arbelo, E.; Barbato, E.; Bartorelli, A.L.; Baumbach, A.; Behr, E.R.; Berti, S.; The Task Force for the Management of COVID-19 of the European Society of Cardiology; et al. ESC guidance for the diagnosis and management of cardiovascular disease during the COVID-19 pandemic: Part 2—Care pathways, treatment, and follow-up. Eur. Heart J. 2021, ehab697. [Google Scholar] [CrossRef]
- Tian, W.; Jiang, W.; Yao, J.; Nicholson, C.J.; Li, R.; Sigurslid, H.; Wooster, L.; Rotter, J.I.; Guo, X.; Malhotra, R. Predictors of mortality in hospitalized COVID-19 patients: A systematic review and meta-analysis. J. Med. Virol. 2020, 92, 1875–1883. [Google Scholar] [CrossRef]
- Li, X.; Guan, B.; Su, T.; Liu, W.; Chen, M.; Waleed, K.B.; Guan, X.; Gary, T.; Zhu, Z. Impact of cardiovascular disease and cardiac injury on in-hospital mortality in patients with COVID-19: A systematic review and meta-analysis. Heart 2020, 106, 1142–1147. [Google Scholar] [CrossRef]
- Zhu, Z.; Wang, M.; Lin, W.; Cai, Q.; Zhang, L.; Chen, D.; Liu, F.; Xiong, X.; Chu, J.; Peng, J.; et al. Cardiac biomarkers, cardiac injury, and comorbidities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): A systematic review and meta-analysis. Immun. Inflamm. Dis. 2021, 9, 1071–1100. [Google Scholar] [CrossRef]
- Wungu, C.D.K.; Khaerunnisa, S.; Putri, E.A.C.; Hidayati, H.B.; Qurnianingsih, E.; Lukitasari, L.; Humairah, I.; Soetjipto. Meta-analysis of cardiac markers for predictive factors on severity and mortality of COVID-19. Int. J. Infect. Dis. 2021, 105, 551–559. [Google Scholar] [CrossRef]
- An, W.; Kang, J.-S.; Wang, Q.; Kim, T.-E. Cardiac biomarkers and COVID-19: A systematic review and meta-analysis. J. Infect. Public Health 2021, 14, 1191–1197. [Google Scholar] [CrossRef]
- Wibowo, A.; Pranata, R.; Akbar, M.R.; Purnomowati, A.; Martha, J.W. Prognostic performance of troponin in COVID-19: A diagnostic meta-analysis and meta-regression. Int. J. Infect. Dis. 2021, 105, 312–318. [Google Scholar] [CrossRef]
- Orsucci, D.; Trezzi, M.; Anichini, R.; Blanc, P.; Barontini, L.; Biagini, C.; Capitanini, A.; Comeglio, M.; Corsini, P.; Gemignani, F.; et al. Increased creatine kinase may predict a worse COVID-19 outcome. J. Clin. Med. 2021, 10, 1734. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Li, P.; Ma, L.; Liang, H.; Lei, J.; Li, W.; Wang, K.; Song, Y.; Li, S.; Yang, W.; et al. Clinical characteristics and short-term outcomes of severe patients with COVID-19 in Wuhan, China. Front. Med. 2020, 7, 491. [Google Scholar] [CrossRef] [PubMed]
- Martins-Filho, P.R.; Barreto-Filho, J.A.S.; Santos, V.S. Myocardial injury biomarkers and cardiac complications associated with mortality in patients with COVID-19. Arq. Bras. Cardiol. 2020, 115, 273–277. [Google Scholar] [CrossRef]
- Yu, J.-S.; Chen, R.-D.; Zeng, L.-C.; Yang, H.-K.; Li, H. Myoglobin offers higher accuracy than other cardiac-specific biomarkers for the prognosis of COVID-19. Front. Cardiovasc. Med. 2021, 8, 686328. [Google Scholar] [CrossRef]
- Ali, A.; Noman, M.; Guo, Y.; Liu, X.; Zhang, R.; Zhou, J.; Zheng, Y.; Zhang, X.-E.; Qi, Y.; Chen, X.; et al. Myoglobin and C-reactive protein are efficient and reliable early predictors of COVID-19 associated mortality. Sci. Rep. 2021, 11, 5975. [Google Scholar] [CrossRef]
- Yang, J.; Liao, X.; Yin, W.; Wang, B.; Yue, J.; Bai, L.; Liu, D.; Zhu, T.; Huang, Z.; Kang, Y. Elevated cardiac biomarkers may be effective prognostic predictors for patients with COVID-19: A multicenter, observational study. Am. J. Emerg. Med. 2020, 39, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.-J.; Cheng, X.; Zhou, F.; Lei, F.; Akolkar, G.; Cai, J.; Zhang, X.-J.; Blet, A.; Xie, J.; Zhang, P.; et al. Redefining cardiac biomarkers in predicting mortality of inpatients with COVID-19. Hypertension 2020, 76, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Garlanda, C.; Bottazzi, B.; Magrini, E.; Inforzato, A.; Mantovani, A. PTX3, a humoral pattern recognition molecule, in innate immunity, tissue repair, and cancer. Physiol. Rev. 2018, 98, 623–639. [Google Scholar] [CrossRef]
- Quismorio, F.P.; Quismorio, A.V. 46—Clinical application of serologic tests, serum protein abnormalities, and other clinical laboratory tests in systemic lupus erythematosus. In Dubois’ Lupus Erythematosus and Related Syndromes, 9th ed.; Wallace, D.J., Hahn, B.H., Eds.; Elsevier: London, UK, 2019; pp. 579–597. ISBN 978-0-323-47927-1. [Google Scholar]
- Salio, M.; Chimenti, S.; De Angelis, N.; Molla, F.; Maina, V.; Nebuloni, M.; Pasqualini, F.; Latini, R.; Garlanda, C.; Mantovani, A. Cardioprotective function of the long pentraxin PTX3 in acute myocardial infarction. Circulation 2008, 117, 1055–1064. [Google Scholar] [CrossRef]
- Maugeri, N.; Rovere-Querini, P.; Slavich, M.; Coppi, G.; Doni, A.; Bottazzi, B.; Garlanda, C.; Cianflone, D.; Maseri, A.; Mantovani, A.; et al. Early and transient release of leukocyte pentraxin 3 during acute myocardial infarction. J. Immunol. 2011, 187, 970–979. [Google Scholar] [CrossRef]
- Ristagno, G.; Fumagalli, F.; Bottazzi, B.; Mantovani, A.; Olivari, D.; Novelli, D.; Latini, R. Pentraxin 3 in cardiovascular disease. Front. Immunol. 2019, 10, 823. [Google Scholar] [CrossRef] [PubMed]
- Brunetta, E.; Folci, M.; Bottazzi, B.; De Santis, M.; Gritti, G.; Protti, A.; Mapelli, S.N.; Bonovas, S.; Piovani, D.; Leone, R.; et al. Macrophage expression and prognostic significance of the long pentraxin PTX3 in COVID-19. Nat. Immunol. 2020, 22, 19–24. [Google Scholar] [CrossRef] [PubMed]
- De Bruin, S.; Bos, L.D.; van Roon, M.A.; Tuip-de Boer, A.M.; Schuurman, A.R.; Koel-Simmelinck, M.J.; Bogaard, H.J.; Tuinman, P.R.; van Agtmael, M.A.; Hamann, J.; et al. Clinical features and prognostic factors in Covid-19: A prospective cohort study. eBioMedicine 2021, 67, 103378. [Google Scholar] [CrossRef] [PubMed]
- Tong, M.; Xiong, Y.; Zhu, C.; Xu, H.; Zheng, Q.; Hu, C.; Jiang, Y.; Zou, L.; Xiao, X.; Chen, F.; et al. Elevated serum pentraxin-3 levels is positively correlated to disease severity and coagulopathy in COVID-19 patients. Mediterr. J. Hematol. Infect. Dis. 2020, 13, e2021015. [Google Scholar] [CrossRef]
- Protti, A.; Meessen, J.; Bottazzi, B.; Garlanda, C.; Milani, A.; Bacci, M.; Mantovani, A.; Cecconi, M.; Latini, R.; Caironi, P. Circulating pentraxin 3 in severe COVID-19 or other pulmonary sepsis. Eur. J. Clin. Investig. 2021, 51, e13530. [Google Scholar] [CrossRef]
- Genç, A.B.; Yaylaci, S.; Dheir, H.; Genç, A.C.; Işsever, K.; Çekiç, D.; Kocayiğit, H.; Çokluk, E.; Karacan, A.; Şekeroğlu, M.R.; et al. The predictive and diagnostic accuracy of long pentraxin-3 in COVID-19 pneumonia. Turk. J. Med. Sci. 2021, 51, 448–453. [Google Scholar] [CrossRef]
- Kukla, M.; Menżyk, T.; Dembiński, M.; Winiarski, M.; Garlicki, A.; Bociąga-Jasik, M.; Skonieczna, M.; Hudy, D.; Maziarz, B.; Kuśnierz-Cabala, B.; et al. Fetuin-A deficiency but not pentraxin 3, FGF-21, or irisin, predisposes to more serious COVID-19 course. Biomolecules 2021, 11, 1422. [Google Scholar] [CrossRef]
- Gritti, G.; Raimondi, F.; Bottazzi, B.; Ripamonti, D.; Riva, I.; Landi, F.; Alborghetti, L.; Frigeni, M.; Damiani, M.; Micò, C.; et al. Siltuximab downregulates interleukin-8 and pentraxin 3 to improve ventilatory status and survival in severe COVID-19. Leukemia 2021, 35, 2710–2714. [Google Scholar] [CrossRef]
- Hall, C. Essential biochemistry and physiology of (NT-pro) BNP. Eur. J. Heart Fail. 2004, 6, 257–260. [Google Scholar] [CrossRef]
- Sawada, Y.; Suda, M.; Yokoyama, H.; Kanda, T.; Sakamaki, T.; Tanaka, S.; Nagai, R.; Abe, S.; Takeuchi, T. Stretch-induced hypertrophic growth of cardiocytes and processing of brain-type natriuretic peptide are controlled by proprotein-processing endoprotease furin. J. Biol. Chem. 1997, 272, 20545–20554. [Google Scholar] [CrossRef]
- de Lemos, J.A.; McGuire, D.K.; Drazner, M.H. B-type natriuretic peptide in cardiovascular disease. Lancet 2003, 362, 316–322. [Google Scholar] [CrossRef]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Galvani, M.; Ferrini, D.; Ottani, F. Natriuretic peptides for risk stratification of patients with acute coronary syndromes. Eur. J. Heart Fail. 2004, 6, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.-L.; Hu, B.-L.; Wen, H.-C.; Zhang, Y.-L.; Zhu, J.-J. Prognostic value of plasma brain natriuretic peptide value for patientswith sepsis: A meta-analysis. J. Crit. Care 2018, 48, 145–152. [Google Scholar] [CrossRef]
- Cunningham, J.W.; Claggett, B.L.; Jering, K.S.; Vaduganathan, M.; Bhatt, A.S.; Rosenthal, N.; Solomon, S.D. Prognostic value of natriuretic peptides and cardiac troponins in COVID-19. Circulation 2021, 144, 177–179. [Google Scholar] [CrossRef]
- Zwaenepoel, B.; Dhont, S.; Hoste, E.; Gevaert, S.; Schaubroeck, H. The prognostic value of cardiac biomarkers and echocardiography in critical COVID-19. Front. Cardiovasc. Med. 2021, 8, 752237. [Google Scholar] [CrossRef]
- Stefanini, G.G.; Chiarito, M.; Ferrante, G.; Cannata, F.; Azzolini, E.; Viggiani, G.; De Marco, A.; Briani, M.; Bocciolone, M.; Bragato, R.; et al. Early detection of elevated cardiac biomarkers to optimise risk stratification in patients with COVID-19. Heart 2020, 106, 1512–1518. [Google Scholar] [CrossRef]
- Gao, L.; Jiang, D.; Wen, X.-S.; Cheng, X.-C.; Sun, M.; He, B.; You, L.-N.; Lei, P.; Tan, X.-W.; Qin, S.; et al. Prognostic value of NT-proBNP in patients with severe COVID-19. Respir. Res. 2020, 21, 83. [Google Scholar] [CrossRef]
- Inciardi, R.M.; Adamo, M.; Lupi, L.; Cani, D.S.; Di Pasquale, M.; Tomasoni, D.; Italia, L.; Zaccone, G.; Tedino, C.; Fabbricatore, D.; et al. Characteristics and outcomes of patients hospitalized for COVID-19 and cardiac disease in Northern Italy. Eur. Heart J. 2020, 41, 1821–1829. [Google Scholar] [CrossRef]
- Caro-Codón, J.; Rey, J.R.; Buño, A.; Iniesta, A.M.; Rosillo, S.O.; Castrejon-Castrejon, S.; Rodriguez-Sotelo, L.; Martinez, L.A.; Marco, I.; Merino, C.; et al. Characterization of NT-proBNP in a large cohort of COVID-19 patients. Eur. J. Heart Fail. 2021, 23, 456–464. [Google Scholar] [CrossRef]
- Sheth, A.; Modi, M.; Dawson, D.; Dominic, P. Prognostic value of cardiac biomarkers in COVID-19 infection. Sci. Rep. 2021, 11, 4930. [Google Scholar] [CrossRef] [PubMed]
- Sorrentino, S.; Cacia, M.; Leo, I.; Polimeni, A.; Sabatino, J.; Spaccarotella, C.A.M.; Mongiardo, A.; De Rosa, S.; Indolfi, C. B-type natriuretic peptide as biomarker of COVID-19 disease severity—A meta-analysis. J. Clin. Med. 2020, 9, 2957. [Google Scholar] [CrossRef] [PubMed]
- Pranata, R.; Huang, I.; Lukito, A.A.; Raharjo, S.B. Elevated N-terminal pro-brain natriuretic peptide is associated with increased mortality in patients with COVID-19: Systematic review and meta-analysis. Postgrad. Med. J. 2020, 96, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Curry, F.-R.E. Atrial natriuretic peptide: An essential physiological regulator of transvascular fluid, protein transport, and plasma volume. J. Clin. Investig. 2005, 115, 1458–1461. [Google Scholar] [CrossRef] [PubMed]
- Morgenthaler, N.G.; Struck, J.; Thomas, B.; Bergmann, A. Immunoluminometric assay for the midregion of pro-atrial natriuretic peptide in human plasma. Clin. Chem. 2004, 50, 234–236. [Google Scholar] [CrossRef]
- Shah, R.V.; Truong, Q.A.; Gaggin, H.K.; Pfannkuche, J.; Hartmann, O.; Januzzi, J.L. Mid-regional pro-atrial natriuretic peptide and pro-adrenomedullin testing for the diagnostic and prognostic evaluation of patients with acute dyspnoea. Eur. Heart J. 2012, 33, 2197–2205. [Google Scholar] [CrossRef]
- von Haehling, S.; Jankowska, E.A.; Morgenthaler, N.G.; Vassanelli, C.; Zanolla, L.; Rozentryt, P.; Filippatos, G.S.; Doehner, W.; Koehler, F.; Papassotiriou, J.; et al. Comparison of midregional pro-atrial natriuretic peptide with N-terminal pro-B-type natriuretic peptide in predicting survival in patients with chronic heart failure. J. Am. Coll. Cardiol. 2007, 50, 1973–1980. [Google Scholar] [CrossRef]
- Krüger, S.; Ewig, S.; Kunde, J.; Hartmann, O.; Suttorp, N.; Welte, T.; The CAPNETZ Study Group. Pro-atrial natriuretic peptide and pro-vasopressin for predicting short-term and long-term survival in community-acquired pneumonia: Results from the German Competence Network CAPNETZ. Thorax 2010, 65, 208–214. [Google Scholar] [CrossRef]
- Pervez, M.O.; Winther, J.A.; Brynildsen, J.; Strand, H.; Christensen, G.; Høiseth, A.D.; Myhre, P.L.; Røysland, R.; Lyngbakken, M.N.; Omland, T.; et al. Prognostic and diagnostic significance of mid-regional pro-atrial natriuretic peptide in acute exacerbation of chronic obstructive pulmonary disease and acute heart failure: Data from the ACE 2 Study. Biomarkers 2018, 23, 654–663. [Google Scholar] [CrossRef]
- Kaufmann, C.C.; Ahmed, A.; Kassem, M.; Freynhofer, M.K.; Jäger, B.; Aicher, G.-C.; Equiluz-Bruck, S.; Spiel, A.O.; Funk, G.; Gschwantler, M.; et al. Mid-regional pro-atrial natriuretic peptide independently predicts short-term mortality in COVID-19. Eur. J. Clin. Investig. 2021, 51, e13531. [Google Scholar] [CrossRef]
- Villacorta, H.; Maisel, A.S. Soluble ST2 testing: A promising biomarker in the management of heart failure. Arq. Bras. Cardiol. 2015, 106, 145–152. [Google Scholar] [CrossRef]
- Pascual-Figal, D.A.; Januzzi, J.L. The biology of ST2: The International ST2 Consensus Panel. Am. J. Cardiol. 2015, 115, 3B–7B. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, J.; Owyang, A.; Oldham, E.; Song, Y.; Murphy, E.; McClanahan, T.K.; Zurawski, G.; Moshrefi, M.; Qin, J.; Li, X.; et al. IL-33, an Interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity 2005, 23, 479–490. [Google Scholar] [CrossRef]
- Ip, C.; Luk, K.S.; Yuen, V.L.C.; Chiang, L.; Chan, C.K.; Ho, K.; Gong, M.; Lee, T.T.L.; Leung, K.S.K.; Roever, L.; et al. Soluble suppression of tumorigenicity 2 (sST2) for predicting disease severity or mortality outcomes in cardiovascular diseases: A systematic review and meta-analysis. IJC Heart Vasc. 2021, 37, 100887. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Hong, X.-Y.; Li, Y.; Chen, W.; Ye, G.; Li, Y.; Luo, Y. Serum-soluble ST2 as a novel biomarker reflecting inflammatory status and illness severity in patients with COVID-19. Biomark. Med. 2020, 14, 1619–1629. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Marteles, M.; Rubio-Gracia, J.; Peña-Fresneda, N.; Garcés-Horna, V.; Gracia-Tello, B.; Martínez-Lostao, L.; Crespo-Aznárez, S.; Pérez-Calvo, J.I.; Giménez-López, I. Early measurement of blood sST2 is a good predictor of death and poor outcomes in patients admitted for COVID-19 infection. J. Clin. Med. 2021, 10, 3534. [Google Scholar] [CrossRef] [PubMed]
- Wendt, R.; Lingitz, M.-T.; Laggner, M.; Mildner, M.; Traxler, D.; Graf, A.; Krotka, P.; Moser, B.; Hoetzenecker, K.; Kalbitz, S.; et al. Clinical relevance of elevated soluble ST2, HSP27 and 20S proteasome at hospital admission in patients with COVID-19. Biology 2021, 10, 1186. [Google Scholar] [CrossRef]
- Abers, M.S.; Delmonte, O.M.; Ricotta, E.E.; Fintzi, J.; Fink, D.L.; de Jesus, A.A.A.; Zarember, K.A.; Alehashemi, S.; Oikonomou, V.; Desai, J.V.; et al. An immune-based biomarker signature is associated with mortality in COVID-19 patients. JCI Insight 2021, 6, 144455. [Google Scholar] [CrossRef]
- Díaz-Alvarez, L.; Ortega, E. The many roles of galectin-3, a multifaceted molecule, in innate immune responses against pathogens. Mediat. Inflamm. 2017, 2017, 9247574. [Google Scholar] [CrossRef]
- Indonesian Journal of Clinical Pathology and Medical Laboratory. Homepage. Available online: https://indonesianjournalofclinicalpathology.org/index.php/patologi/index (accessed on 9 January 2022).
- Blanda, V.; Bracale, U.M.; Di Taranto, M.D.; Fortunato, G. Galectin-3 in cardiovascular diseases. Int. J. Mol. Sci. 2020, 21, 9232. [Google Scholar] [CrossRef]
- Wu, C.; Lv, Z.; Li, X.; Zhou, X.; Mao, W.; Zhu, M. Galectin-3 in predicting mortality of heart failure: A systematic review and meta-analysis. Heart Surg. Forum 2021, 24, E327–E332. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Cao, S.; Yang, H.; Li, J. Prognostic impact of galectin-3 in chronic kidney disease patients: A systematic review and meta-analysis. Int. Urol. Nephrol. 2019, 51, 1005–1011. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.E.; Gao, W.; Levy, D.; Santhanakrishnan, R.; Araki, T.; Rosas, I.O.; Hatabu, H.; Latourelle, J.C.; Nishino, M.; Dupuis, J.; et al. Galectin-3 is associated with restrictive lung disease and interstitial lung abnormalities. Am. J. Respir. Crit. Care Med. 2016, 194, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Portacci, A.; Diaferia, F.; Santomasi, C.; Dragonieri, S.; Boniello, E.; Di Serio, F.; Carpagnano, G.E. Galectin-3 as prognostic biomarker in patients with COVID-19 acute respiratory failure. Respir. Med. 2021, 187, 106556. [Google Scholar] [CrossRef] [PubMed]
- Kazancioglu, S.; Yilmaz, F.M.; Bastug, A.; Ozbay, B.O.; Aydos, O.; Yücel, Ç.; Bodur, H.; Yilmaz, G. Assessment of galectin-1, galectin-3, and prostaglandin E2 levels in patients with COVID-19. Jpn. J. Infect. Dis. 2021, 74, 530–536. [Google Scholar] [CrossRef]
- Chen, P.-K.; Lan, J.-L.; Huang, P.-H.; Hsu, J.-L.; Chang, C.-K.; Tien, N.; Lin, H.-J.; Chen, D.-Y. Interleukin-18 is a potential biomarker to discriminate active adult-onset still’s disease from COVID-19. Front. Immunol. 2021, 12, 719544. [Google Scholar] [CrossRef]
- Bootcov, M.R.; Bauskin, A.R.; Valenzuela, S.M.; Moore, A.G.; Bansal, M.; He, X.Y.; Zhang, H.P.; Donnellan, M.; Mahler, S.; Pryor, K.; et al. MIC-1, a novel macrophage inhibitory cytokine, is a divergent member of the TGF-β superfamily. Proc. Natl. Acad. Sci. USA 1997, 94, 11514–11519. [Google Scholar] [CrossRef]
- Savic-Radojevic, A.; Pljesa-Ercegovac, M.; Matic, M.; Simic, D.; Radovanovic, S.; Simic, T. Chapter four—Novel biomarkers of heart failure. In Advances in Clinical Chemistry; Makowski, G.S., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; Volume 79, pp. 93–152. [Google Scholar]
- Schlittenhardt, D.; Schober, A.; Strelau, J.; Bonaterra, G.A.; Schmiedt, W.; Unsicker, K.; Metz, J.; Kinscherf, R. Involvement of growth differentiation factor-15/macrophage inhibitory cytokine-1 (GDF-15/MIC-1) in oxLDL-induced apoptosis of human macrophages in vitro and in arteriosclerotic lesions. Cell Tissue Res. 2004, 318, 325–333. [Google Scholar] [CrossRef]
- Luo, J.-W.; Duan, W.-H.; Song, L.; Yu, Y.-Q.; Shi, D.-Z. A meta-analysis of growth differentiation factor-15 and prognosis in chronic heart failure. Front. Cardiovasc. Med. 2021, 8, 630818. [Google Scholar] [CrossRef]
- Chirinos, J.A.; Orlenko, A.; Zhao, L.; Basso, M.D.; Cvijic, M.E.; Li, Z.; Spires, T.E.; Yarde, M.; Wang, Z.; Seiffert, D.A.; et al. Multiple plasma biomarkers for risk stratification in patients with heart failure and preserved ejection fraction. J. Am. Coll. Cardiol. 2020, 75, 1281–1295. [Google Scholar] [CrossRef]
- Verhamme, F.M.; Freeman, C.M.; Brusselle, G.; Bracke, K.; Curtis, J.L. GDF-15 in pulmonary and critical care medicine. Am. J. Respir. Cell Mol. Biol. 2019, 60, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Buendgens, L.; Yagmur, E.; Bruensing, J.; Herbers, U.; Baeck, C.; Trautwein, C.; Koch, A.; Tacke, F. Growth differentiation factor-15 is a predictor of mortality in critically ill patients with sepsis. Dis. Markers 2017, 2017, 5271203. [Google Scholar] [CrossRef] [PubMed]
- Myhre, P.L.; Prebensen, C.; Strand, H.; Røysland, R.; Jonassen, C.M.; Rangberg, A.; Sørensen, V.; Søvik, S.; Røsjø, H.; Svensson, M.; et al. Growth differentiation factor 15 provides prognostic information superior to established cardiovascular and inflammatory biomarkers in unselected patients hospitalized with COVID-19. Circulation 2020, 142, 2128–2137. [Google Scholar] [CrossRef] [PubMed]
- de Guadiana Romualdo, L.G.; Mulero, M.D.R.; Olivo, M.H.; Rojas, C.R.; Arenas, V.R.; Morales, M.G.; Abellán, A.B.; Conesa-Zamora, P.; García-García, J.; Hernández, A.C.; et al. Circulating levels of GDF-15 and calprotectin for prediction of in-hospital mortality in COVID-19 patients: A case series. J. Infect. 2020, 82, e40–e42. [Google Scholar] [CrossRef]
- Notz, Q.; Schmalzing, M.; Wedekink, F.; Schlesinger, T.; Gernert, M.; Herrmann, J.; Sorger, L.; Weismann, D.; Schmid, B.; Sitter, M.; et al. Pro- and anti-inflammatory responses in severe COVID-19-induced acute respiratory distress syndrome—An observational pilot study. Front. Immunol. 2020, 11, 581338. [Google Scholar] [CrossRef]
- Teng, X.; Zhang, J.; Shi, Y.; Liu, Y.; Yang, Y.; He, J.; Luo, S.; Huang, Y.; Liu, Y.; Liu, D.; et al. Comprehensive profiling of inflammatory factors revealed that growth differentiation factor-15 is an indicator of disease severity in COVID-19 patients. Front. Immunol. 2021, 12, 662465. [Google Scholar] [CrossRef]
- Alserawan, L.; Peñacoba, P.; Orozco Echevarría, S.E.; Castillo, D.; Ortiz, E.; Martínez-Martínez, L.; Moga Naranjo, E.; Domingo, P.; Castellví, I.; Juárez, C.; et al. Growth differentiation factor 15 (GDF-15): A novel biomarker associated with poorer respiratory function in COVID-19. Diagnostics 2021, 11, 1998. [Google Scholar] [CrossRef]
- Bolignano, D.; Cabassi, A.; Fiaccadori, E.; Ghigo, E.; Pasquali, R.; Peracino, A.; Peri, A.; Plebani, M.; Santoro, A.; Settanni, F.; et al. Copeptin (CTproAVP), a new tool for understanding the role of vasopressin in pathophysiology. Clin. Chem. Lab. Med. (CCLM) 2014, 52, 1447–1456. [Google Scholar] [CrossRef]
- Rotondo, F.; Butz, H.; Syro, L.V.; Yousef, G.M.; Di Ieva, A.; Restrepo, L.M.; Quintanar-Stephano, A.; Berczi, I.; Kovacs, K. Arginine vasopressin (AVP): A review of its historical perspectives, current research and multifunctional role in the hypothalamo-hypophysial system. Pituitary 2016, 19, 345–355. [Google Scholar] [CrossRef]
- Struck, J.; Morgenthaler, N.G.; Bergmann, A. Copeptin, a stable peptide derived from the vasopressin precursor, is elevated in serum of sepsis patients. Peptides 2005, 26, 2500–2504. [Google Scholar] [CrossRef]
- Kolditz, M.; Halank, M.; Schulte-Hubbert, B.; Bergmann, S.; Albrecht, S.; Höffken, G. Copeptin predicts clinical deterioration and persistent instability in community-acquired pneumonia. Respir. Med. 2012, 106, 1320–1328. [Google Scholar] [CrossRef] [PubMed]
- Seligman, R.; Papassotiriou, J.; Morgenthaler, N.G.; Meisner, M.; Teixeira, P.J. Copeptin, a novel prognostic biomarker in ventilator-associated pneumonia. Crit. Care 2008, 12, R11. [Google Scholar] [CrossRef] [PubMed]
- Hellenkamp, K.; Schwung, J.; Rossmann, H.; Kaeberich, A.; Wachter, R.; Hasenfuß, G.; Konstantinides, S.; Lankeit, M. Risk stratification of normotensive pulmonary embolism: Prognostic impact of copeptin. Eur. Respir. J. 2015, 46, 1701–1710. [Google Scholar] [CrossRef] [PubMed]
- Winther, J.A.; Brynildsen, J.; Høiseth, A.D.; Strand, H.; Følling, I.; Christensen, G.; Nygård, S.; Røsjø, H.; Omland, T. Prognostic and diagnostic significance of copeptin in acute exacerbation of chronic obstructive pulmonary disease and acute heart failure: Data from the ACE 2 study. Respir. Res. 2017, 18, 184. [Google Scholar] [CrossRef] [PubMed]
- Mueller, C.; Möckel, M.; Giannitsis, E.; Huber, K.; Mair, J.; Plebani, M.; Thygesen, K.; Jaffe, A.S.; Lindahl, B.; ESC Study Group on Biomarkers in Cardiology of the Acute Cardiovascular Care Association. Use of copeptin for rapid rule-out of acute myocardial infarction. Eur. Heart J. Acute Cardiovasc. Care 2017, 7, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Neumann, J.T.; Weimann, J.; Sörensen, N.A.; Hartikainen, T.S.; Haller, P.M.; Lehmacher, J.; Brocks, C.; Tenhaeff, S.; Karakas, M.; Renné, T.; et al. A biomarker model to distinguish types of myocardial infarction and injury. J. Am. Coll. Cardiol. 2021, 78, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, C.C.; Ahmed, A.; Kassem, M.; Freynhofer, M.K.; Jäger, B.; Aicher, G.; Equiluz-Bruck, S.; Spiel, A.O.; Vafai-Tabrizi, F.; Gschwantler, M.; et al. Improvement of outcome prediction of hospitalized patients with COVID-19 by a dual marker strategy using high-sensitive cardiac troponin I and copeptin. Clin. Res. Cardiol. 2021, 111, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Gregoriano, C.; Molitor, A.; Haag, E.; Kutz, A.; Koch, D.; Haubitz, S.; Conen, A.; Bernasconi, L.; Hammerer-Lercher, A.; Fux, C.A.; et al. Activation of vasopressin system during COVID-19 is associated with adverse clinical outcomes: An observational study. J. Endocr. Soc. 2021, 5, bvab045. [Google Scholar] [CrossRef]
- In, E.; Kuluöztürk, M.; Telo, S.; Toraman, Z.A.; Karabulut, E. Can copeptin predict the severity of coronavirus disease 2019 infection? Rev. Assoc. Med. Bras. 2021, 67, 1137–1142. [Google Scholar] [CrossRef]
- Jougasaki, M.; Burnett, J.C. Adrenomedullin: Potential in physiology and pathophysiology. Life Sci. 2000, 66, 855–872. [Google Scholar] [CrossRef]
- Potocki, M.; Ziller, R.; Mueller, C. Mid-regional pro-adrenomedullin in acute heart failure: A better biomarker or just another biomarker? Curr. Heart Fail. Rep. 2012, 9, 244–251. [Google Scholar] [CrossRef] [PubMed]
- Lopes, D.; Menezes Falcão, L. Mid-regional pro-adrenomedullin and ST2 in heart failure: Contributions to diagnosis and prognosis. Rev. Port. Cardiol. 2017, 36, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela-Sánchez, F.; Valenzuela-Méndez, B.; Rodríguez-Gutiérrez, J.F.; Estella-García, Á.; González-García, M. New role of biomarkers: Mid-regional pro-adrenomedullin, the biomarker of organ failure. Ann. Transl. Med. 2016, 4, 329. [Google Scholar] [CrossRef]
- Maisel, A.; Mueller, C.; Nowak, R.; Peacock, W.F.; Landsberg, J.W.; Ponikowski, P.; Mockel, M.; Hogan, C.; Wu, A.H.B.; Richards, M.; et al. Mid-region pro-hormone markers for diagnosis and prognosis in acute dyspnea: Results from the BACH (Biomarkers in Acute Heart Failure) trial. J. Am. Coll. Cardiol. 2010, 55, 2062–2076. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Taub, P.; Iqbal, N.; Fard, A.; Clopton, P.; Maisel, A. Mid-region pro-adrenomedullin adds predictive value to clinical predictors and Framingham risk score for long-term mortality in stable outpatients with heart failure. Eur. J. Heart Fail. 2013, 15, 1343–1349. [Google Scholar] [CrossRef]
- Andaluz-Ojeda, D.; Nguyen, H.B.; Meunier-Beillard, N.; Cicuéndez, R.; Quenot, J.-P.; Calvo, D.; Dargent, A.; Zarca, E.; Andrés, C.; Nogales, L.; et al. Superior accuracy of mid-regional proadrenomedullin for mortality prediction in sepsis with varying levels of illness severity. Ann. Intensive Care 2017, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.T.; Angus, D.C.; Kellum, J.A.; Pugh, N.A.; Weissfeld, L.A.; Struck, J.; Delude, R.L.; Rosengart, M.R.; Yealy, D.M. Midregional proadrenomedullin as a prognostic tool in community-acquired pneumonia. Chest 2009, 136, 823–831. [Google Scholar] [CrossRef]
- Gregoriano, C.; Koch, D.; Kutz, A.; Haubitz, S.; Conen, A.; Bernasconi, L.; Hammerer-Lercher, A.; Saeed, K.; Mueller, B.; Schuetz, P. The vasoactive peptide MR-pro-adrenomedullin in COVID-19 patients: An observational study. Clin. Chem. Lab. Med. (CCLM) 2020, 59, 995–1004. [Google Scholar] [CrossRef]
- García de Guadiana-Romualdo, L.; Martínez Martínez, M.; Rodríguez Mulero, M.D.; Esteban-Torrella, P.; Hernández Olivo, M.; Alcaraz García, M.J.; Campos-Rodríguez, V.; Sancho-Rodríguez, N.; Galindo Martínez, M.; Alcaraz, A.; et al. Circulating MR-proADM levels, as an indicator of endothelial dysfunction, for early risk stratification of mid-term mortality in COVID-19 patients. Int. J. Infect. Dis. 2021, 111, 211–218. [Google Scholar] [CrossRef]
- Minieri, M.; Di Lecce, V.N.; Lia, M.S.; Maurici, M.; Bernardini, S.; Legramante, J.M. Role of MR-proADM in the risk stratification of COVID-19 patients assessed at the triage of the emergency department. Crit. Care 2021, 25, 407. [Google Scholar] [CrossRef]
- Honore, P.M.; Redant, S.; Moorthamers, S.; Preseau, T.; Kaefer, K.; Barreto Gutierrez, L.; Attou, R.; Gallerani, A.; De Bels, D. MR-proADM has a good ability to predict 28-day mortality in critically ill patients with SARS-CoV-2 pneumonia: Beware of some potential confounders! J. Crit. Care 2021, 67, 212–213. [Google Scholar] [CrossRef] [PubMed]
- Zaninotto, M.; Mion, M.M.; Marchioro, L.; Padoan, A.; Plebani, M. Endothelial dysfunction and mid-regional proAdrenomedullin: What role in SARS-CoV-2 infected patients? Clin. Chim. Acta 2021, 523, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Van Oers, J.A.H.; Kluiters, Y.; Bons, J.A.P.; de Jongh, M.; Pouwels, S.; Ramnarain, D.; de Lange, D.W.; de Grooth, H.-J.; Girbes, A.R.J. Endothelium-associated biomarkers mid-regional proadrenomedullin and C-terminal proendothelin-1 have good ability to predict 28-day mortality in critically ill patients with SARS-CoV-2 pneumonia: A prospective cohort study. J. Crit. Care 2021, 66, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Lo Sasso, B.; Gambino, C.M.; Scichilone, N.; Giglio, R.V.; Bivona, G.; Scazzone, C.; Muratore, R.; Milano, S.; Barbagallo, M.; Agnello, L.; et al. Clinical utility of midregional proadrenomedullin in patients with COVID-19. Lab. Med. 2021, 52, 493–498. [Google Scholar] [CrossRef]
- Roedl, K.; Jarczak, D.; Fischer, M.; Haddad, M.; Boenisch, O.; de Heer, G.; Burdelski, C.; Frings, D.; Sensen, B.; Karakas, M.; et al. MR-proAdrenomedullin as a predictor of renal replacement therapy in a cohort of critically ill patients with COVID-19. Biomarkers 2021, 26, 417–424. [Google Scholar] [CrossRef]
- Sozio, E.; Tascini, C.; Fabris, M.; D’Aurizio, F.; De Carlo, C.; Graziano, E.; Bassi, F.; Sbrana, F.; Ripoli, A.; Pagotto, A.; et al. MR-proADM as prognostic factor of outcome in COVID-19 patients. Sci. Rep. 2021, 11, 5121. [Google Scholar] [CrossRef]
- Benedetti, I.; Spinelli, D.; Callegari, T.; Bonometti, R.; Molinaro, E.; Novara, E.; Cassinari, M.; Frino, C.; Guaschino, R.; Boverio, R.; et al. High levels of mid-regional proadrenomedullin in ARDS COVID-19 patients: The experience of a single, Italian Center. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 1743–1751. [Google Scholar] [CrossRef]
- Montrucchio, G.; Sales, G.; Rumbolo, F.; Palmesino, F.; Fanelli, V.; Urbino, R.; Filippini, C.; Mengozzi, G.; Brazzi, L. Effectiveness of mid-regional pro-adrenomedullin (MR-proADM) as prognostic marker in COVID-19 critically ill patients: An observational prospective study. PLoS ONE 2021, 16, e0246771. [Google Scholar] [CrossRef]
- García de Guadiana-Romualdo, L.; Calvo Nieves, M.D.; Rodríguez Mulero, M.D.; Calcerrada Alises, I.; Hernández Olivo, M.; Trapiello Fernández, W.; González Morales, M.; Bolado Jiménez, C.; Albaladejo-Otón, M.D.; Fernández Ovalle, H.; et al. MR-proADM as marker of endotheliitis predicts COVID-19 severity. Eur. J. Clin. Investig. 2021, 51, e13511. [Google Scholar] [CrossRef]
- Spoto, S.; Agrò, F.E.; Sambuco, F.; Travaglino, F.; Valeriani, E.; Fogolari, M.; Mangiacapra, F.; Costantino, S.; Ciccozzi, M.; Angeletti, S. High value of mid-regional proadrenomedullin in COVID-19: A marker of widespread endothelial damage, disease severity, and mortality. J. Med. Virol. 2020, 93, 2820–2827. [Google Scholar] [CrossRef]
- Hickey, K.A.; Rubanyi, G.; Paul, R.J.; Highsmith, R.F. Characterization of a coronary vasoconstrictor produced by cultured endothelial cells. Am. J. Physiol. Physiol. 1985, 248, C550–C556. [Google Scholar] [CrossRef] [PubMed]
- Freeman, B.D.; Machado, F.S.; Tanowitz, H.B.; Desruisseaux, M.S. Endothelin-1 and its role in the pathogenesis of infectious diseases. Life Sci. 2014, 118, 110–119. [Google Scholar] [CrossRef] [PubMed]
- Kedzierski, R.M.; Yanagisawa, M. Endothelin system: The double-edged sword in health and disease. Annu. Rev. Pharmacol. Toxicol. 2001, 41, 851–876. [Google Scholar] [CrossRef] [PubMed]
- Jankowich, M.; Choudhary, G. Endothelin-1 levels and cardiovascular events. Trends Cardiovasc. Med. 2020, 30, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bonaventura, A.; Vecchié, A.; Dagna, L.; Martinod, K.; Dixon, D.L.; Van Tassell, B.W.; Dentali, F.; Montecucco, F.; Massberg, S.; Levi, M.; et al. Endothelial dysfunction and immunothrombosis as key pathogenic mechanisms in COVID-19. Nat. Rev. Immunol. 2021, 21, 319–329. [Google Scholar] [CrossRef] [PubMed]
- PubMed. Endothelin-1 Contributes to Endothelial Dysfunction and Enhanced Vasoconstriction through Augmented Superoxide Production in Penile Arteries from Insulin-Resistant Obese Rats: Role of ET(A) and ET(B) Receptors. Available online: https://pubmed-ncbi-nlm-nih-gov.ez.srv.meduniwien.ac.at/25091502/ (accessed on 10 January 2022).
- Gregoriano, C.; Damm, D.; Kutz, A.; Koch, D.; Wolfisberg, S.; Haubitz, S.; Conen, A.; Bernasconi, L.; Hammerer-Lercher, A.; Fux, C.A.; et al. Association of endothelial activation assessed through endothelin-I precursor peptide measurement with mortality in COVID-19 patients: An observational analysis. Respir. Res. 2021, 22, 148. [Google Scholar] [CrossRef] [PubMed]
- Icer, M.A.; Gezmen-Karadag, M. The multiple functions and mechanisms of osteopontin. Clin. Biochem. 2018, 59, 17–24. [Google Scholar] [CrossRef]
- Hayek, S.S.; Roderburg, C.; Blakely, P.; Launius, C.; Eugen-Olsen, J.; Tacke, F.; Ktena, S.; Keitel, V.; Luedde, M.; Giamarellos-Bourboulis, E.J.; et al. Circulating osteopontin levels and outcomes in patients hospitalized for COVID-19. J. Clin. Med. 2021, 10, 3907. [Google Scholar] [CrossRef]
- Bai, G.; Furushima, D.; Niki, T.; Matsuba, T.; Maeda, Y.; Takahashi, A.; Hattori, T.; Ashino, Y. High levels of the cleaved form of galectin-9 and osteopontin in the plasma are associated with inflammatory markers that reflect the severity of COVID-19 pneumonia. Int. J. Mol. Sci. 2021, 22, 4978. [Google Scholar] [CrossRef]
- Varım, C.; Demirci, T.; Cengiz, H.; Hacıbekiroğlu, I.; Tuncer, F.B.; Çokluk, E.; Toptan, H.; Karabay, O.; Yıldırım, I. Relationship between serum osteopontin levels and the severity of COVID-19 infection. Wien. Klin. Wochenschr. 2020, 133, 298–302. [Google Scholar] [CrossRef]
- Sibal, L.; Agarwal, S.C.; Home, P.D.; Boger, R.H. The role of asymmetric dimethylarginine (ADMA) in endothelial dysfunction and cardiovascular disease. Curr. Cardiol. Rev. 2010, 6, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Hannemann, J.; Balfanz, P.; Schwedhelm, E.; Hartmann, B.; Ule, J.; Müller-Wieland, D.; Dahl, E.; Dreher, M.; Marx, N.; Böger, R. Elevated serum SDMA and ADMA at hospital admission predict in-hospital mortality of COVID-19 patients. Sci. Rep. 2021, 11, 9895. [Google Scholar] [CrossRef] [PubMed]
- Koeth, R.A.; Haselden, V.; Tang, W.H.W. Chapter one—Myeloperoxidase in cardiovascular disease. In Advances in Clinical Chemistry; Makowski, G.S., Ed.; Elsevier: Amsterdam, The Netherlands, 2013; Volume 62, pp. 1–32. [Google Scholar]
- Pincemail, J.; Cavalier, E.; Charlier, C.; Cheramy–Bien, J.-P.; Brevers, E.; Courtois, A.; Fadeur, M.; Meziane, S.; Goff, C.; Misset, B.; et al. Oxidative stress status in COVID-19 patients hospitalized in intensive care unit for severe pneumonia. A pilot study. Antioxidants 2021, 10, 257. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, S.; Chelluboina, S.; Jedge, P.; Doke, P.; Palkar, S.; Mishra, A.C.; Arankalle, V.A. Elevated levels of neutrophil activated proteins, alpha-defensins (DEFA1), calprotectin (S100A8/A9) and myeloperoxidase (MPO) are associated with disease severity in COVID-19 patients. Front. Cell. Infect. Microbiol. 2021, 11, 751232. [Google Scholar] [CrossRef] [PubMed]
- Otto Walter, L.; Cardoso, C.C.; Santos-Pirath, M.; Costa, H.Z.; Gartner, R.; Werle, I.; Mohr, E.T.B.; da Rosa, J.S.; Felisberto, M.; Kretzer, I.F.; et al. The relationship between peripheral immune response and disease severity in SARS-CoV-2-infected subjects: A cross-sectional study. Immunology 2022. [Google Scholar] [CrossRef] [PubMed]
- Bermejo-Martin, J.F.; González-Rivera, M.; Almansa, R.; Micheloud, D.; Tedim, A.P.; Domínguez-Gil, M.; Resino, S.; Martín-Fernández, M.; Murua, P.R.; Pérez-García, F.; et al. Viral RNA load in plasma is associated with critical illness and a dysregulated host response in COVID-19. Crit. Care 2020, 24, 691. [Google Scholar] [CrossRef]
- Mueller, C.; Giannitsis, E.; Jaffe, A.S.; Huber, K.; Mair, J.; Cullen, L.; Hammarsten, O.; Mills, N.L.; Möckel, M.; Krychtiuk, K.; et al. Cardiovascular biomarkers in patients with COVID-19. Eur. Heart J. Acute Cardiovasc. Care 2021, 10, 310–319. [Google Scholar] [CrossRef]
| Biomarkers | Primary Origin | Primary Release Mode | Molecular Weight (kD) | Normal Range | Mortality in COVID-19 |
|---|---|---|---|---|---|
| Biomarkers of Myocyte Injury | |||||
| Hs-cTn | Cardiac myocytes | Myocardial injury | 24 (Trop I) 35 (Trop T) | Assay specific cut-offs | I |
| CK-MB | Cardiac myocytes, skeletal muscle | Muscle injury (primarily heart) | 84 | 5–25 IU/L [20] | I |
| Myoglobin | Cardiac myocytes, skeletal muscle | Muscle injury | 17.7 | Mean 31 ng/mL (+/− 1.3) [21] | I |
| Pentraxin-3 | Smooth muscle cells, vascular endothelial cells, macrophages | Vascular injury/inflammation | 40.2 | M: 1.87 ng/mL (CI, 1.81–1.94) [22] F: 2.12 ng/mL (CI, 2.05–2.19) [22] | I |
| Biomarkers of Myocardial Stretch | |||||
| NT-proBNP | Ventricular myocytes | Ventricular volume or pressure overload | 8.5 | M: 42.5–106.4 pg/mL (97.5 percentile) [23] F: 111.0–215.9 pg/mL (97.5 percentile) [23] | I |
| MR-proANP | Atrial myocytes | Atrial volume or pressure overload | 2.6 (ANP) | M: 90.2–228 pmol/mL (97.5 percentile) [24] F: 118–214 pmol/mL (97.5 percentile) [24] | III |
| Biomarkers of Extracellular Matrix Remodeling | |||||
| sST2 | Cardiac and lung cells | Inflammation/fibrosis/vascular congestion | 37 | M: mean 24.9 ng/mL (95% CI, 23.7–26.2) [25] F: mean 16.9 ng/mL (95% CI, 16.1–17.7) [25] | II |
| Galectin-3 | Mostly non-cardiac: macrophages/ fibroblasts | Fibrosis | 30 | Median 62 ng/mL (IQR, 20–313) [26] | II |
| GDF-15 | Unknown | Inflammation | 16.7 | 2.5 percentile: 399 ng/L (90% CI, 399–399) [27] 97.5 percentile: 1335 ng/L (90% CI, 1152–1445) [27] | I |
| Biomarkers of Neurohumoral Activation | |||||
| Copeptin | Pituitary gland | Osmotic stimulation/stress | 4021 | Median 4.2 pmol/L (IQR, 1.0–13.8) [28] | II |
| MR-proADM | Ubiquitary | Neurohumoral activation/inflammation | 5.1 | 2.5 percentile: 0.21 nmol/L (90% CI, 0.19–0.23) [29] 97.5 percentile: 0.57 nmol/L (90% CI, 0.55–0.59) [29] | I |
| Endothelin-1 | Vascular endothelial cells | Pleiotropic (pulsatile stretch, hypoxemia…) | 2492 | M: 2.64 ng/L (IQR, 2.21–3.17) [30] F: 2.46 ng/L (IQR, 2.05–2.94) [30] | III |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaufmann, C.C.; Ahmed, A.; Burger, A.L.; Muthspiel, M.; Jäger, B.; Wojta, J.; Huber, K. Biomarkers Associated with Cardiovascular Disease in COVID-19. Cells 2022, 11, 922. https://doi.org/10.3390/cells11060922
Kaufmann CC, Ahmed A, Burger AL, Muthspiel M, Jäger B, Wojta J, Huber K. Biomarkers Associated with Cardiovascular Disease in COVID-19. Cells. 2022; 11(6):922. https://doi.org/10.3390/cells11060922
Chicago/Turabian StyleKaufmann, Christoph C., Amro Ahmed, Achim Leo Burger, Marie Muthspiel, Bernhard Jäger, Johann Wojta, and Kurt Huber. 2022. "Biomarkers Associated with Cardiovascular Disease in COVID-19" Cells 11, no. 6: 922. https://doi.org/10.3390/cells11060922
APA StyleKaufmann, C. C., Ahmed, A., Burger, A. L., Muthspiel, M., Jäger, B., Wojta, J., & Huber, K. (2022). Biomarkers Associated with Cardiovascular Disease in COVID-19. Cells, 11(6), 922. https://doi.org/10.3390/cells11060922





