Overexpression of MET4 Leads to the Upregulation of Stress-Related Genes and Enhanced Sulfite Tolerance in Saccharomyces uvarum
Abstract
1. Introduction
2. Materials and Methods
2.1. Construction of the MET4 Gene Expression Vector
2.2. Genetic Transformation
2.3. Sulfite Tolerant Trait Typing
2.4. PCR Analysis
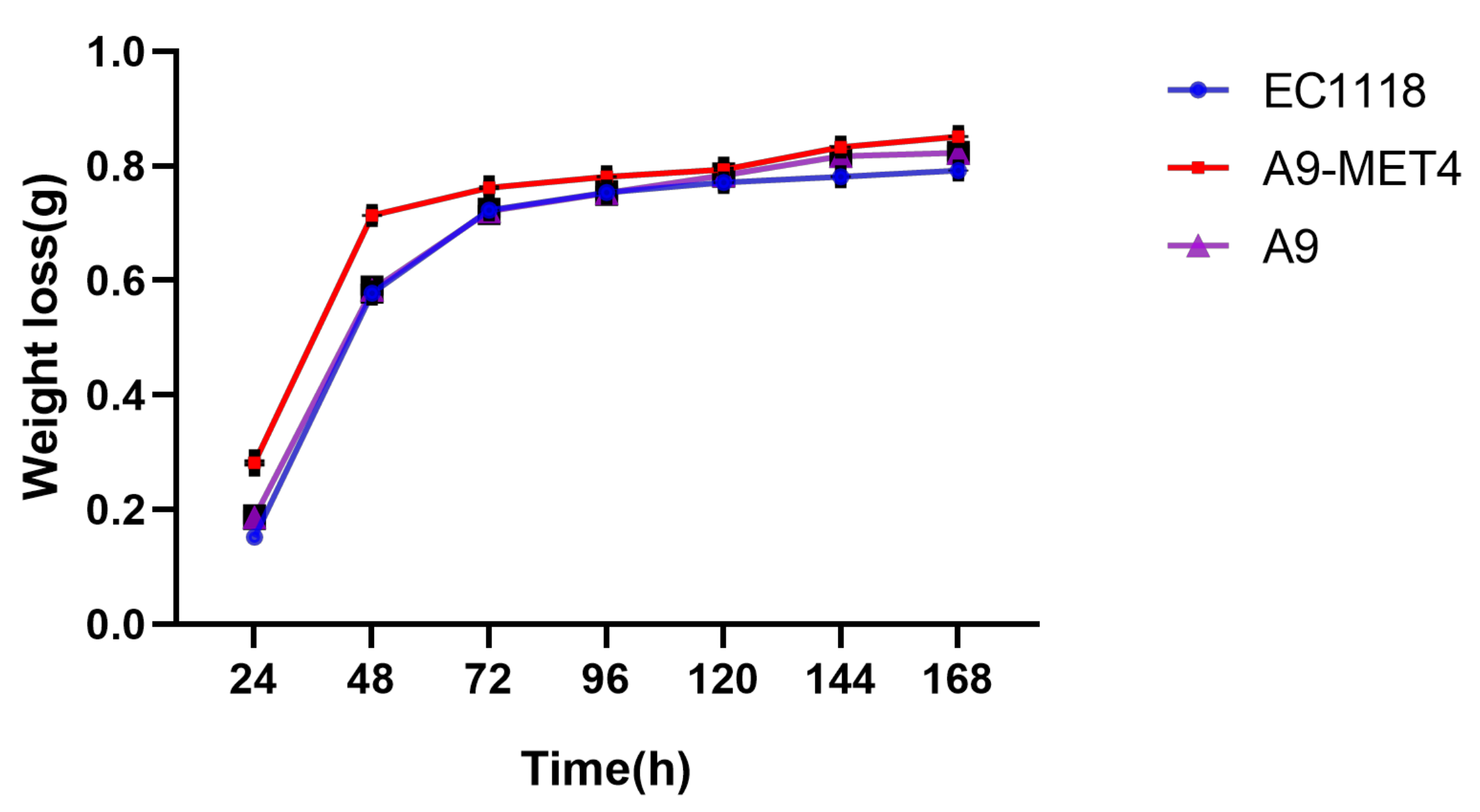
2.5. Fermentation Weight Loss Analysis
2.6. Ribonucleic Acid (RNA) Extraction and cDNA Synthesis
2.7. RT-qPCR Analysis
2.8. Transcriptome Analysis
2.9. Data Analysis
3. Results
3.1. Detection of Transgenic Strains
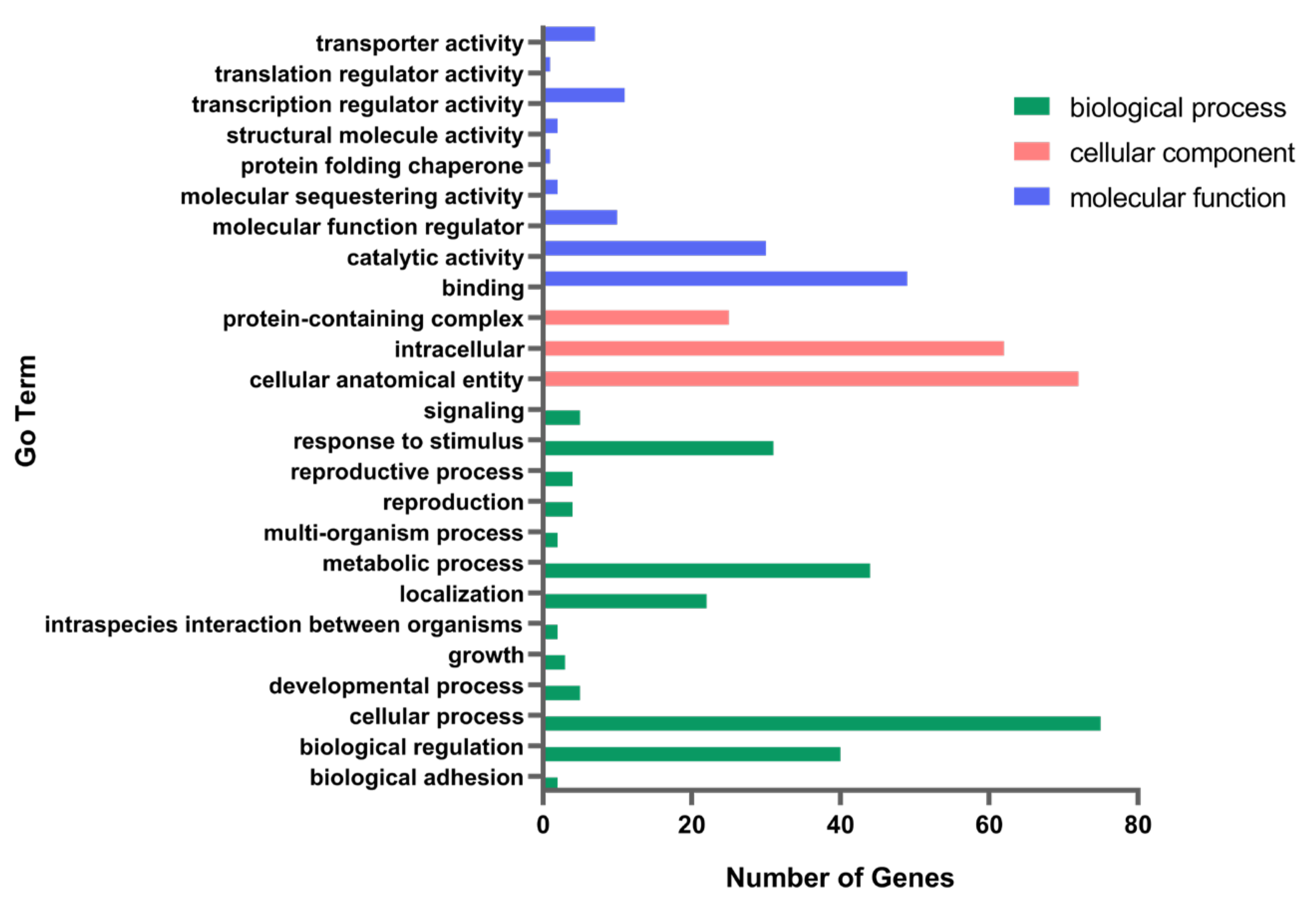
3.2. Transcriptome Differential Gene Analysis
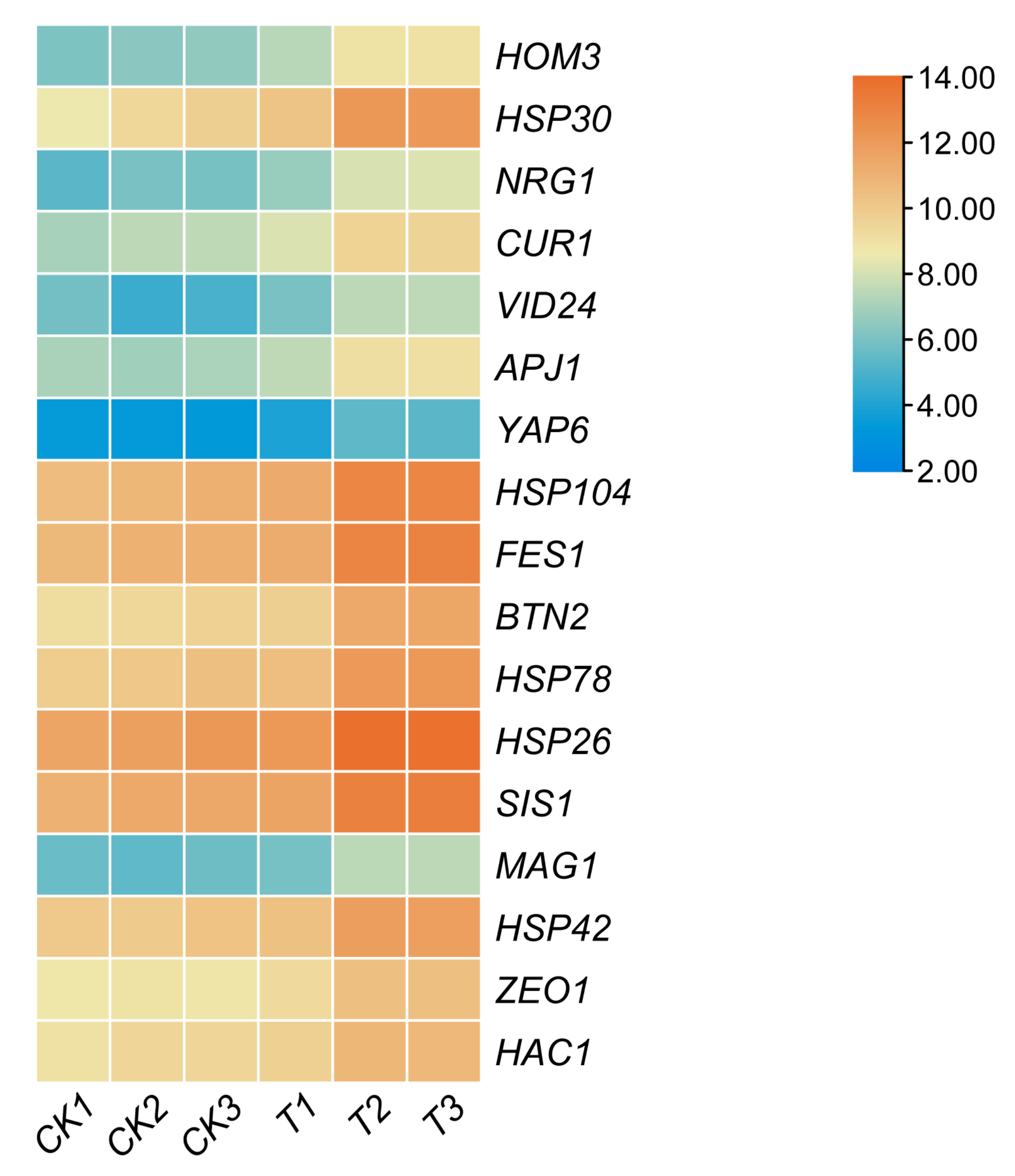
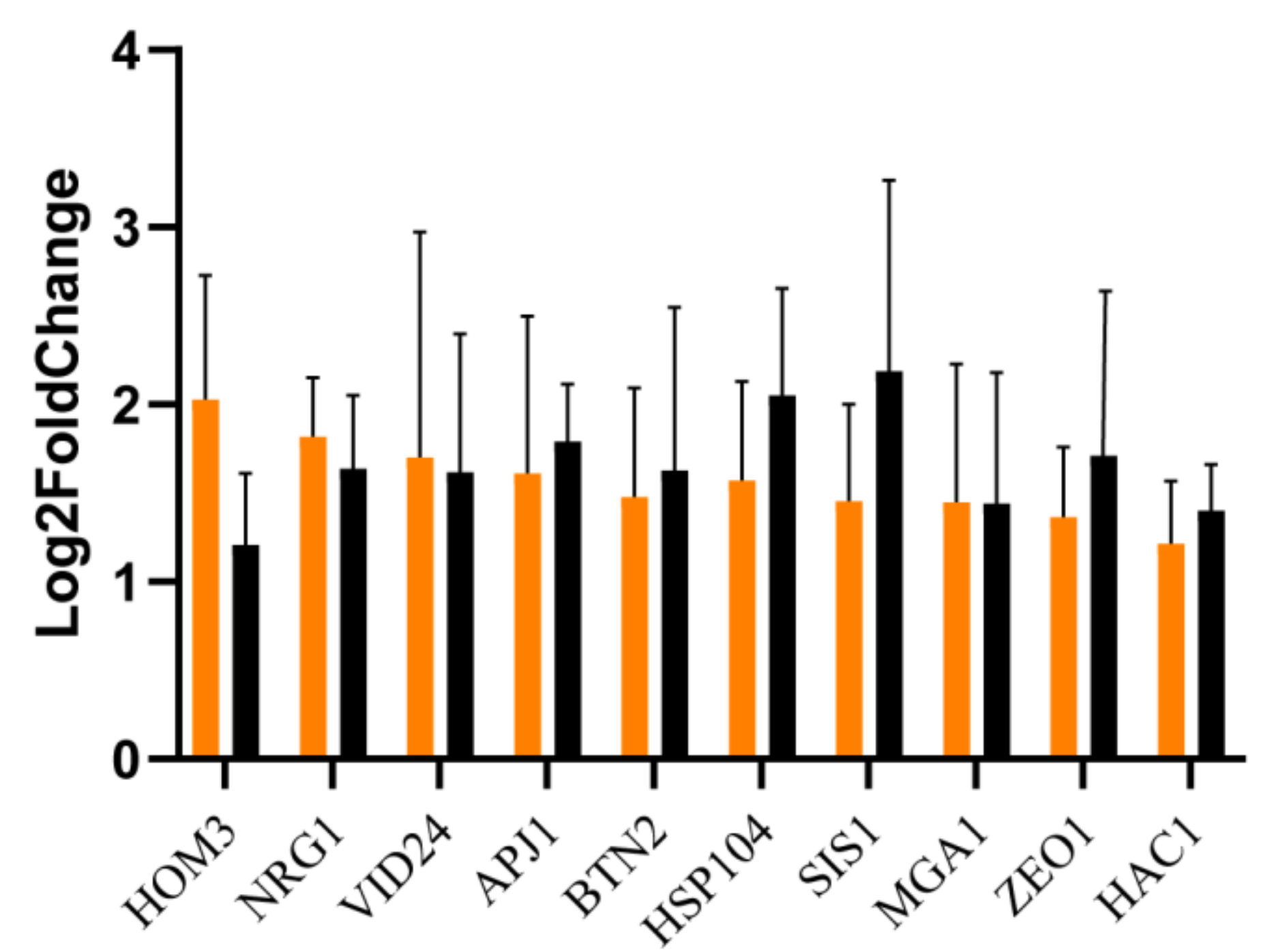
3.3. RT-qPCR Validation
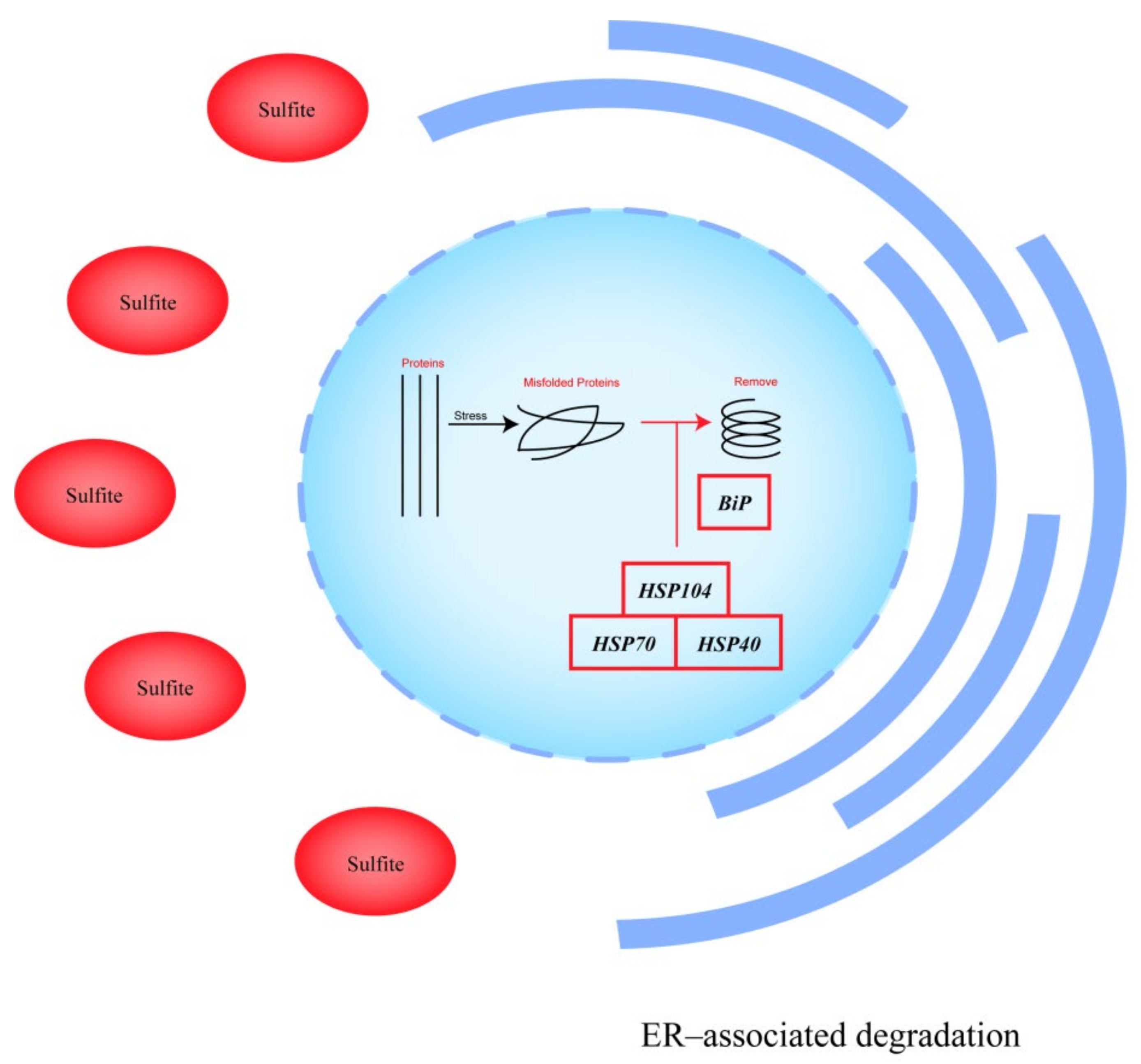
4. Discussion
5. Conclusions
- (1)
- We suggest that a mechanism of sulfite tolerance exists in S. uvarum, i.e., tolerance to higher sulfite concentrations is achieved by depleting the sulfite in the culture environment. S. uvarum may be able to tolerate higher concentrations of sulfite by continuously growing and dividing, and by upregulating the expression of genes, such as HOM3, which consume part of the sulfite in the culture medium or fermentation broth.
- (2)
- S. uvarum may be able to tolerate higher concentrations of sulfite by increasing the expression of MET4, and thus the stress-related genes. The hypothesis that the ‘MET4 gene can regulate sulfite tolerance in S. uvarum’ is proved.
- (3)
- Overexpression of the MET4 gene resulted in a significant increase in sulfite tolerance, an upregulation of fermentation-stress-related gene expression, and a significant change in the transcriptome profile of the strain. The transformants could broaden the application of S. uvarum in the winemaking industry.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, H.Y.; Lee, S.A.; Bradbury, J.E.; Warren, R.N.; Sheth, H.; Hooks, D.O.; Richards, K.D.; Gardner, R.C. Yeasts isolated from New Zealand vineyards and wineries. Aust. J. Grape Wine Res. 2010, 16, 491–496. [Google Scholar] [CrossRef]
- Zhang, H.; Richards, K.D.; Wilson, S.; Lee, S.A.; Sheehan, H.; Roncoroni, M.; Gardner, R.C. Genetic characterization of strains of Saccharomyces uvarum from New Zealand wineries. Food Microbiol. 2015, 46, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Nadai, C.; Treu, L.; Campanaro, S.; Giacomini, A.; Corich, V. Different mechanisms of resistance modulate sulfite tolerance in wine yeasts. Appl. Microbiol. Biotechnol. 2015, 100, 797–813. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y. Formation and characteristics of wine bouquet produced by wine yeasts. Microbiology 2007, 34, 757–760. (In Chinese) [Google Scholar]
- Liu, X.Z.; Zhang, Z.M.; Zhang, H.Y. Cross breeding and hybrid identification of sulphite-tolerant hybrids of Saccharomyces uvarum. S. Afr. J. Enol. Vitic. 2017, 38, 125–131. [Google Scholar] [CrossRef][Green Version]
- Vargas-Maya, N.I.; González-Hernández, G.A.; Padilla-Guerrero, I.E.; Torres-Guzmán, J.C. Overexpression of smORF YNR034W-A/EGO4 in Saccharomyces cerevisiae increases the fermentative efficiency of Agave tequilana Weber must. J. Ind. Microbiol. Biotechnol. 2017, 44, 63–74. [Google Scholar] [CrossRef]
- Bergman, A.; Vitay, D.; Hellgren, J.; Chen, Y.; Nielsen, J.; Siewers, V. Effects of overexpression of STB5 in Saccharomyces cerevisiae on fatty acid biosynthesis, physiology and transcriptome. FEMS Yeast Res. 2019, 19, foz027. [Google Scholar] [CrossRef]
- Liu, X.Z.; Sang, M.; Zhang, X.A.; Zhang, T.K.; Zhang, H.Y.; He, X.; Li, S.X.; Sun, X.D.; Zhang, Z.M. Enhancing expression of SSU1 genes in Saccharomyces uvarum leads to an increase in sulfite tolerance and a transcriptome profile change. FEMS Yeast Res. 2017, 17, fox023. [Google Scholar] [CrossRef]
- Liu, X.; Liu, X.; Zhang, Z.; Sang, M.; Sun, X.D.; He, C.Z.; Xin, P.Y.; Zhang, H.Y. Functional analysis of the FZF1 genes of Saccharomyces uvarum. Front. Microbiol. 2018, 9, 96. [Google Scholar] [CrossRef]
- Hansen, J.; Johannesen, P.F. Cysteine is essential for transcriptional regulation of the sulfur assimilation genes in Saccharomyces cerevisiae. Mol. Gen. Genet. 2000, 263, 535–542. [Google Scholar] [CrossRef]
- Thomas, D.; Surdinkerjan, Y. Metabolism of sulfur amino acids in Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev. 1997, 61, 503–532. [Google Scholar] [PubMed]
- Kuras, L.; Thomas, D. Functional analysis of MET4, a yeast transcriptional activator responsive to S-adenosylmethionine. Mol. Cell. Biol. 1995, 15, 208. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Zhang, X.A.; Liu, X.Z.; Li, S.X.; Zhang, H.Y. Cloning and sequence analysis of the FZF1 gene concerning sulfur tolerance from Saccharomyces bayanus. Sci. Technol. Food Ind. 2015, 36, 166–172. (In Chinese) [Google Scholar]
- Thomas, D.; Jacquemin, I.; Surdin-Kerjan, Y. MET4, a leucine zipper protein, and centromere-binding factor 1 are both required for transcriptional activation of sulfur metabolism in Saccharomyces cerevisiae. Mol. Cell. Biol. 1992, 12, 1719–1727. [Google Scholar] [PubMed]
- Kuras, L.; Cherest, H.; Surdin-Kerjan, Y.; Thomas, D. A heteromeric complex containing the centromere binding factor 1 and two basic leucine zipper factors, MET4 and Met28, mediates the transcription activation of yeast sulfur metabolism. EMBO J. 1996, 15, 2519–2529. [Google Scholar] [CrossRef] [PubMed]
- Su, N.Y.; Ouni, I.; Papagiannis, C.V.; Kaiser, P. A dominant suppressor mutation of the Met30 cell cycle defect suggests regulation of the Saccharomyces cerevisiae MET4-Cbf1 transcription complex by Met32. J. Biol. Chem. 2008, 283, 11615–11624. [Google Scholar] [CrossRef] [PubMed]
- Kuras, L.; Barbey, R.; Thomas, D. Assembly of a bZIP-bHLH transcription activation complex: Formation of the yeast Cbf1-MET4-Met28 complex is regulated through Met28 stimulation of Cbf1 DNA binding. EMBO J. 1997, 116, 2441–2451. [Google Scholar] [CrossRef] [PubMed]
- Blaiseau, P.L.; Thomas, D. Multiple transcriptional activation complexes tether the yeast activator MET4 to DNA. EMBO J. 2014, 17, 6327–6336. [Google Scholar] [CrossRef]
- Sambrook, J.; Fritsch, E.F.; Maniatis, T. Molecular Cloning: A Laboratory Manual, New York: Cold Spring Harbor Laboratory Press. BioScience 1989, 33, 721–722. [Google Scholar]
- Nardi, T.; Corich, V.; Giacomini, A.; Blondin, B. A sulphite-inducible form of the sulphite efflux gene SSU1 in a Saccharomyces cerevisiae wine yeast. Microbiology 2010, 156, 1686–1696. [Google Scholar] [CrossRef]
- Bian, W.; Liu, X.; Zhang, Z.; Zhang, H. Transcriptome analysis of diploid and triploid Populus tomentosa. Peer J. 2020, 8, e10204. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Wakasa, Y.; Yasuda, H.; Oono, Y.; Kawakatsu, T.; Hirose, S.; Takahashi, H.; Hayashi, S.; Yang, L.; Takaiwa, F. Expression of ER quality control-related genes in response to changes in BiP1 levels in developing rice endosperm. Plant J. Cell Mol. Biol. 2015, 65, 675–689. [Google Scholar] [CrossRef] [PubMed]
- Sathyanarayanan, U.; Musa, M.; Dib, P.B.; Raimundo, N.; Milosevic, I.; Krisko, A. ATP hydrolysis by yeast Hsp104 determines protein aggregate dissolution and size in vivo. Nat. Commun. 2020, 11, 5226. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Hu, X.; Guo, Y.; Wang, L.; Ji, J.; Li, J.; Zhang, Z.R. A technique for delineating the unfolding requirements for substrate entry into retrotranslocons during endoplasmic reticulum-associated degradation. J. Biol. Chem. 2019, 294, 20084–20096. [Google Scholar] [CrossRef]
- Glover, J.R.; Lindquist, S. Hsp104, Hsp70, and Hsp40: A novel chaperone system that rescues previously aggregated proteins. Cell 1998, 94, 73. [Google Scholar] [CrossRef]
- Rafalski, J.A.; Falco, S.C. Structure of the yeast HOM3 gene which encodes aspartokinase. J. Biol. Chem. 1988, 263, 2146–2151. [Google Scholar] [CrossRef]
- Lee, T.A.; Jorgensen, P.; Bognar, A.L.; Peyraud, C.; Thomas, D.; Tyers, M. Dissection of combinatorial control by the MET4 transcriptional complex. Mol. Biol. Cell 2010, 21, 456. [Google Scholar] [CrossRef]
- Park, H.; Bakalinsky, A.T. SSU1 mediates sulphite efflux in Saccharomyces cerevisiae. Yeast 2000, 16, 881–888. [Google Scholar] [CrossRef]
- Thorsen, M.; Lagniel, G.; Kristiansson, E.; Junot, C.; Nerman, O.; Labarre, J.; Tamas, M.J. Quantitative transcriptome, proteome, and sulfur metabolite profiling of the Saccharomyces cerevisiae response to arsenite. Physiol. Genom. 2007, 30, 35–43. [Google Scholar] [CrossRef]
- Youn, J.Y.; Friesen, H.; Nguyen Ba, A.N.; Liang, W.; Messier, V.; Cox, M.J.; Moses, A.M.; Andrews, B. Functional analysis of kinases and transcription factors in Saccharomyces cerevisiae using an integrated overexpression library. G3 Genes|Genomes|Genetics 2017, 7, 911–921. [Google Scholar] [CrossRef]
- Kobayashi, K.; Yoshimoto, A. Studies on yeast sulfite reductase. IV. Structure and steady-state kinetics. Biochim. Biophys. Acta 1982, 705, 348–356. [Google Scholar] [CrossRef]
- McIsaac, R.S.; Petti, A.A.; Bussemaker, H.J.; Bussemaker, D. Perturbation-based analysis and modeling of combinatorial regulation in the yeast sulfur assimilation pathway. Mol. Biol. Cell 2012, 23, 2993–3007. [Google Scholar] [CrossRef] [PubMed]
- Arévalo-Rodríguez, M.; Calderón, I.L.; Holmberg, S. Mutations that cause threonine sensitivity identify catalytic and regulatory regions of the aspartate kinase of Saccharomyces cerevisiae. Yeast 2010, 15, 1331–1345. [Google Scholar] [CrossRef]
- Chen, J.; Gao, T.; Wan, S.; Zhang, Y.H.; Yang, J.K.; Yu, Y.B.; Wang, W.D. Genome-wide identification, classification and expression analysis of the HSP gene superfamily in tea plant (Camellia sinensis). Int. J. Mol. Sci. 2018, 19, 2633. [Google Scholar] [CrossRef]

| Gene | Sequence (5′–3′) | Product Length |
|---|---|---|
| HOM3 | TAA ATG GTG TCG GTC GTG | 235 |
| TTG GCT CTG ATA ACT TGC T | ||
| NRG1 | ATT TCG GCG TTT GAT AGA | 304 |
| CAT TCA GTT GGG ATA GCG | ||
| VID24 | GTA GAC AGT TTG GCG GAG AA | 155 |
| CGG TCA ACG AGA CGG AAT | ||
| APJ1 | CTT TGA CAC GGG AGG AGT | 146 |
| GGA AGT TGG GCG TAG AGC | ||
| BTN2 | GTT GAA CCA TTC TAT CCC TC | 218 |
| GAT TCC TTC TTG GCT TTT | ||
| Hsp104 | AAG AAT TGA CTC CCG TGG TG | 193 |
| ACC TGG CTC ACC AAT CAA AC | ||
| SIS1 | GCC AAC AGG GGA TAC TGA AA | 227 |
| TGA AAG CGT CTT CAT TGC TG | ||
| MGA1 | TCT GAA ACC GTA TGA CCC | 273 |
| TTA CCA TCT TTG CCC ACA | ||
| ZEO1 | AGC TGG ATG AAA CTA AGG A | 174 |
| TGG TGG TGA CTT CGG TCT | ||
| HAC1 | CAA GAC GGA GAA CAT ACA AGA | 177 |
| ATC GTA ATC ACG GCT GGA | ||
| MET4 | TCG CAG TAT GAC CAA TCC AA | 163 |
| CAG CCG TGC TTA CAG GAA AT | ||
| ACT1 | AGC GCA ATC CAA GAG AGG TA | 153 |
| GCT TCG GTC AAA AGA ACA GG |
| Strain | HYG | Sodium Sulfite (mM) | ||||
|---|---|---|---|---|---|---|
| 5 | 10 | 20 | 40 | 60 | ||
| A9 | − | + | + | + | − | − |
| A9-MET4 | + | + | + | + | + | − |
| Sample | CK1 | CK2 | CK3 | T1 | T2 | T3 | |
|---|---|---|---|---|---|---|---|
| FPKM | |||||||
| 0–1 | 1.2% | 1.1% | 1.2% | 1.1% | 2.5% | 2.5% | |
| 1–10 | 12.7% | 13.5% | 13.1% | 12.1% | 21.6% | 20.9% | |
| ≥10 | 86.1% | 85.4% | 85.7% | 86.9% | 75.9% | 76.6% | |
| KEGG Category | Gene | Log2 | Decribution |
|---|---|---|---|
| Amino sugar and nucleotide sugar metabolism | CHS2 (CHitin Synthase 2) | 1.54 | Chitin synthase |
| LPX1 (Lipase of PeroXisomes 1) | 1.24 | Chitinase | |
| Autophagy-yeast | LST8 * (Lethal with Sec Thirteen 8) | 1.62 | Target of rapamycin complex subunit LST8 |
| Endocytosis | SSA4 * (Stress-Seventy subfamily A 4) | 1.43 | Heat shock 70 kDa protein |
| ENT2 (Epsin N-Terminal homology 2) | 1.40 | Epsin-like protein required for endocytosis and actin patch assembly | |
| RNA transport | BTN2 (BaTteN disease 2) | 1.48 | Translation initiation factor |
| DI49_2214 (Unannotated) | 1.23 | Polyadenylate-binding protein | |
| Roibosome biogenesis in eukaryotes | NOP4 (NucleOlar Protein 4) | 1.29 | Nucleolar protein 4 |
| Spliceosome | SLU7 (Synergistic Lethal with U5 snRNA 7) | 1.37 | Pre-mRNA-processing factor |
| SSA4 * (Stress-Seventy subfamily A 4) | 1.43 | Heat shock 70 kDa protein | |
| Mitogen-Activated Protein Kinase (MAPK) signaling pathway-yeast | TEC1 (Transposon Enhancement Control 1) | 1.41 | Transcriptional enhancer factor |
| DI49_4478 (Unannotated) | 0.64 | Cytokinesis protein | |
| Non-homologous end-joining | DNL4 (DNA Ligase 4) | 0.86 | DNA ligase 4 |
| ATP-binding cassette (ABC) transporters | PDR12 (Pleiotropic Drug Resistance 12) | 0.87 | ATP-binding cassette |
| Vitamin B6 metabolism | SNO1 (SNZ proximal Open reading frame 1 | 1.20 | 5′-Phosphate synthase pdxT subunit |
| Sphingolipid metabolism | LCB5 (Long-Chain Base 5) | 1.43 | Sphingosine kinase |
| Mannose type O-glycan biosynthesis | ZEO1 * (ZEOcin resistance 1) | 1.36 | Dolichyl-phosphate-mannose-protein mannosyltransferase |
| Glycosylphosphatidylinositol (GPI)-anchor biosynthesis | MCD4 (Morphogenesis Checkpoint Dependent 4) | 1.27 | Phosphatidylinositol glycan |
| GPI12 (GlycosylPhosphatidylInositol anchor biosynthesis 12) | 1.54 | N-acetylglucosaminylphosphatidylinositol deacetylase | |
| Protein processing in the endoplasmic reticulum | APJ1 * (Anti-Prion DnaJ gene 1) | 1.61 | DnaJ homolog subfamily A member 2 |
| FES1 * (Factor Exchange for Ssa1p 1) | 1.50 | Hsp70-interacting protein | |
| HSP26 * (Heat Shock Protein 26) | 1.47 | HSP20 family protein | |
| SSA4 * (Stress-Seventy subfamily A 4) | 1.43 | Heat shock 70 kDa protein | |
| SSE1 * (Stress Seventy subfamily E 1) | 1.41 | Heat shock protein 110 kDa | |
| HSP42 * (Heat Shock Protein 42) | 1.37 | HSP20 family protein | |
| KAR2 * (KARyogamy 2) | 1.29 | Heat shock 70 kDa protein 5 | |
| DER1 * (Degradation in the Endoplasmic Reticulum 1) | 1.26 | Derlin-2/3 | |
| MPD1 * (Multicopy suppressor of PDI1 deletion 1) | 1.08 | Protein disulfide-isomerase A6 | |
| Meiosis-yeast | RPI1 * (Ras-cAMP Pathway Inhibitor 1) | 1.33 | Mediator of RNA polymerase II transcription subunit |
| PHD1 (PseudoHyphal Determinant 1) | 1.29 | Enhanced filamentous growth protein 1 | |
| Cysteine and methionine metabolism | HOM3 * (HOMoserine requiring gene 3) | 2.03 | Aspartate kinase |
| Lysine degradation | DI49_2072 (Unannotated) | 1.31 | Lysine N-acetyltransferase |
| SET2 (SET domain-containing 2) | 1.34 | Histone-lysine N-methyltransferase SETD2 | |
| Longevity regulating pathway-multiple species | Hsp104 * (Heat Shock Protein 104) | 1.57 | ATP-dependent Clp protease ATP-binding subunit ClpB |
| HSP78 * (Heat Shock Protein 78) | 1.48 | ATP-dependent Clp protease ATP-binding subunit ClpB | |
| SSA4 * (Stress-Seventy subfamily A 4) | 1.43 | Heat shock 70 kDa protein | |
| RPI1 * (Ras-cAMP pathway inhibitor 1) | 1.33 | Mediator of RNA polymerase II transcription subunit |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, Z.; Zhang, Z.; Zhao, W.; Yin, T.; Liu, X.; Zhang, H. Overexpression of MET4 Leads to the Upregulation of Stress-Related Genes and Enhanced Sulfite Tolerance in Saccharomyces uvarum. Cells 2022, 11, 636. https://doi.org/10.3390/cells11040636
Wei Z, Zhang Z, Zhao W, Yin T, Liu X, Zhang H. Overexpression of MET4 Leads to the Upregulation of Stress-Related Genes and Enhanced Sulfite Tolerance in Saccharomyces uvarum. Cells. 2022; 11(4):636. https://doi.org/10.3390/cells11040636
Chicago/Turabian StyleWei, Zhuo, Zhiming Zhang, Wenjuan Zhao, Tuo Yin, Xiaozhen Liu, and Hanyao Zhang. 2022. "Overexpression of MET4 Leads to the Upregulation of Stress-Related Genes and Enhanced Sulfite Tolerance in Saccharomyces uvarum" Cells 11, no. 4: 636. https://doi.org/10.3390/cells11040636
APA StyleWei, Z., Zhang, Z., Zhao, W., Yin, T., Liu, X., & Zhang, H. (2022). Overexpression of MET4 Leads to the Upregulation of Stress-Related Genes and Enhanced Sulfite Tolerance in Saccharomyces uvarum. Cells, 11(4), 636. https://doi.org/10.3390/cells11040636






