Scaffold-Mediated Immunoengineering as Innovative Strategy for Tendon Regeneration
Abstract
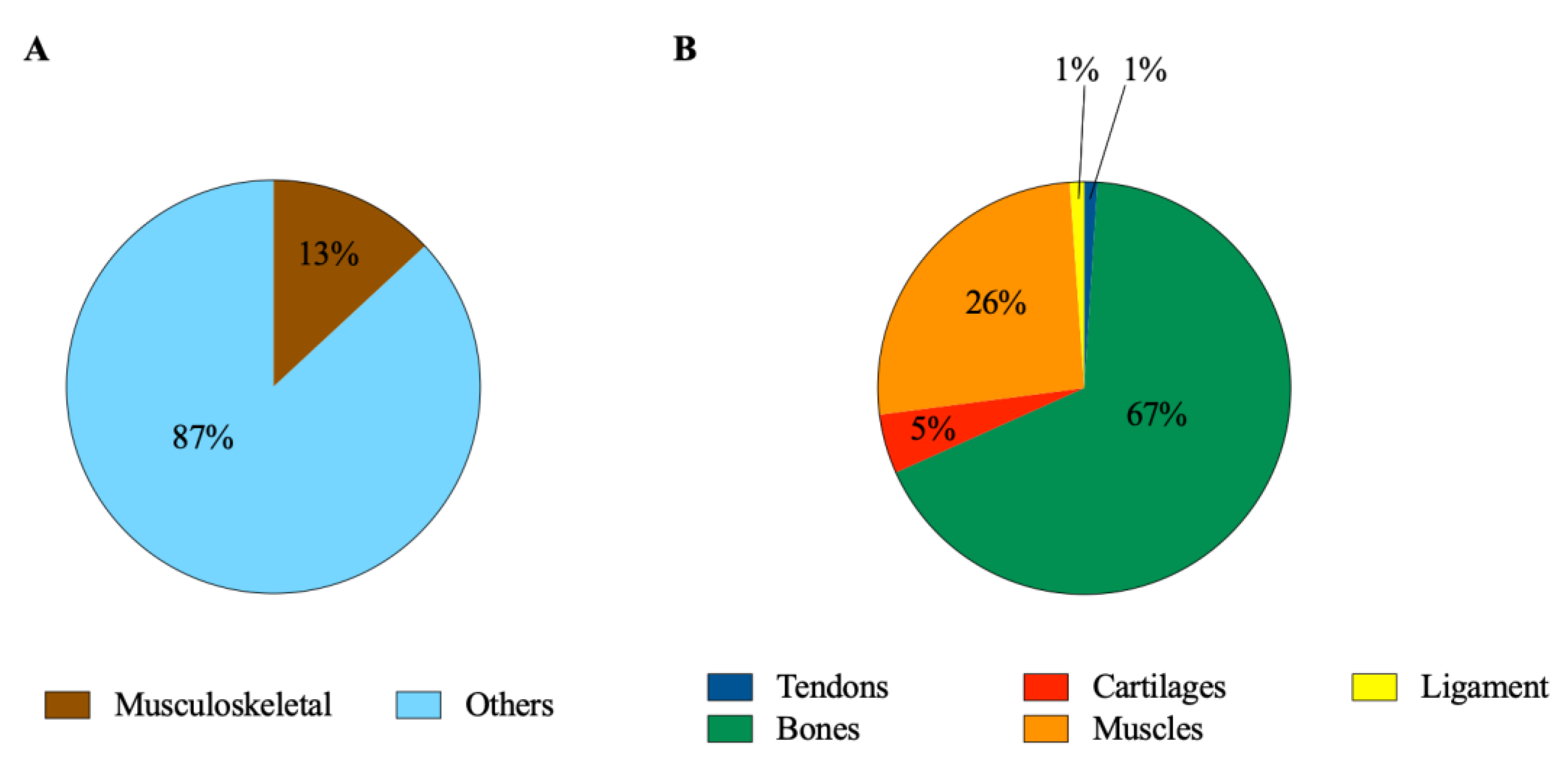
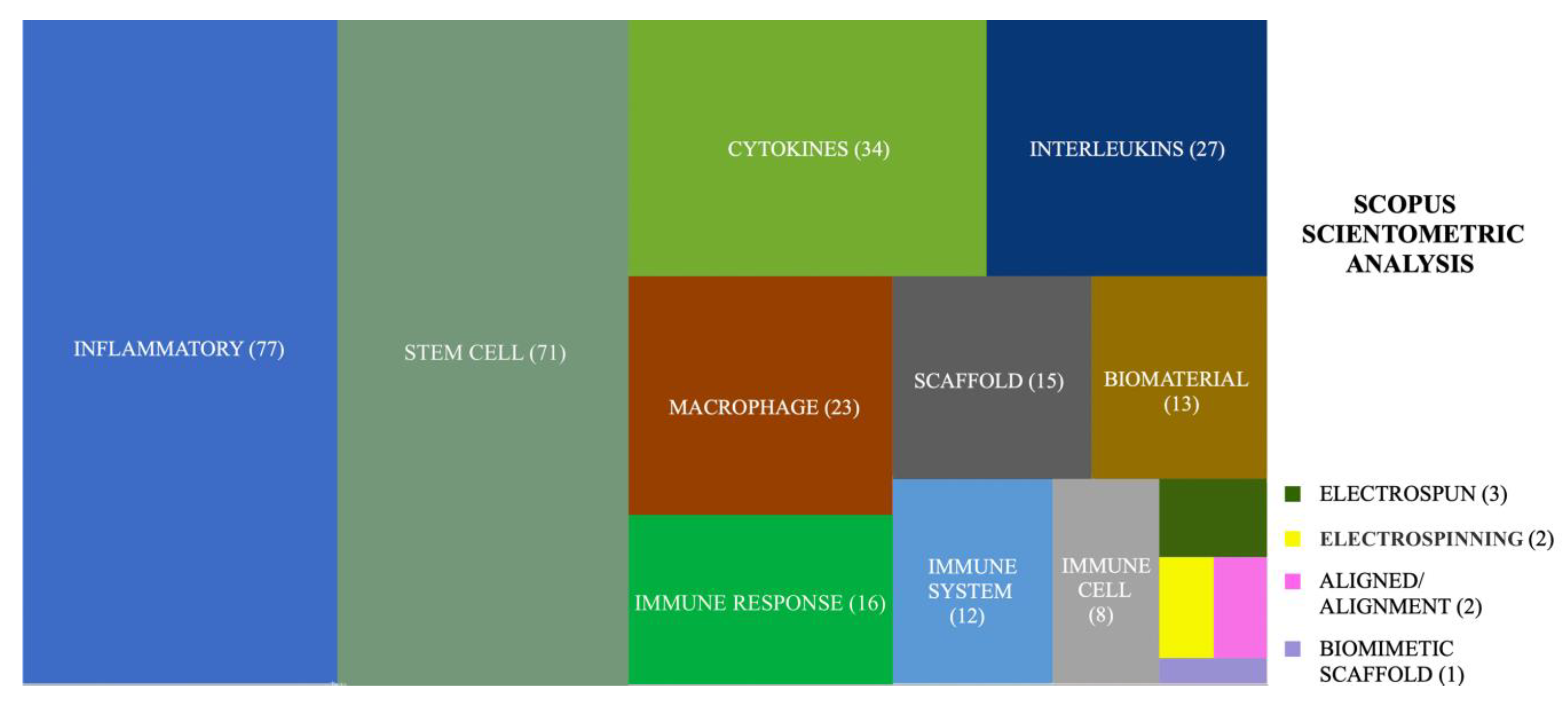
1. Introduction
2. Biomimetic Scaffolds Applied for Tendon TE
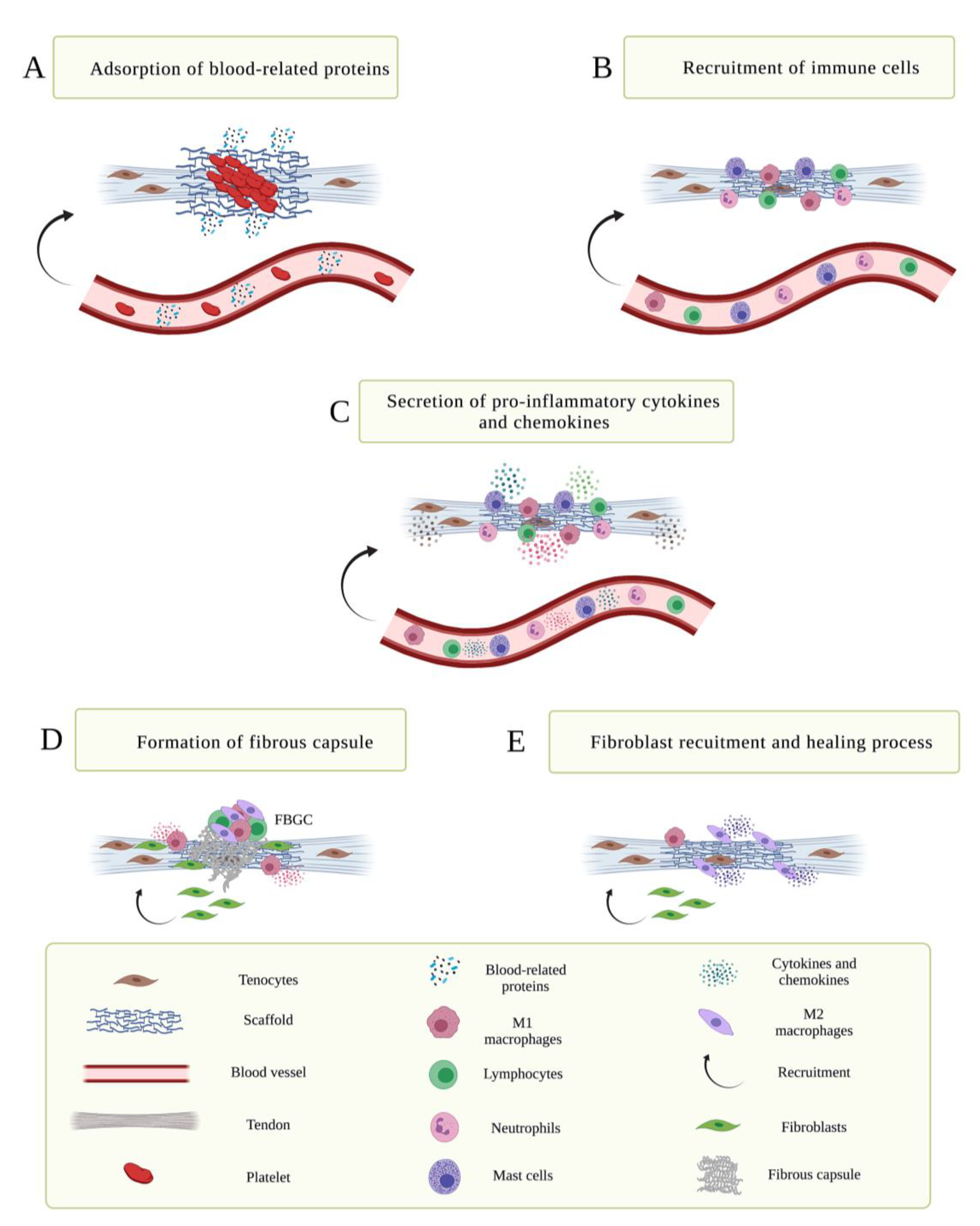
3. Immune Response Induced by Scaffold Implantation
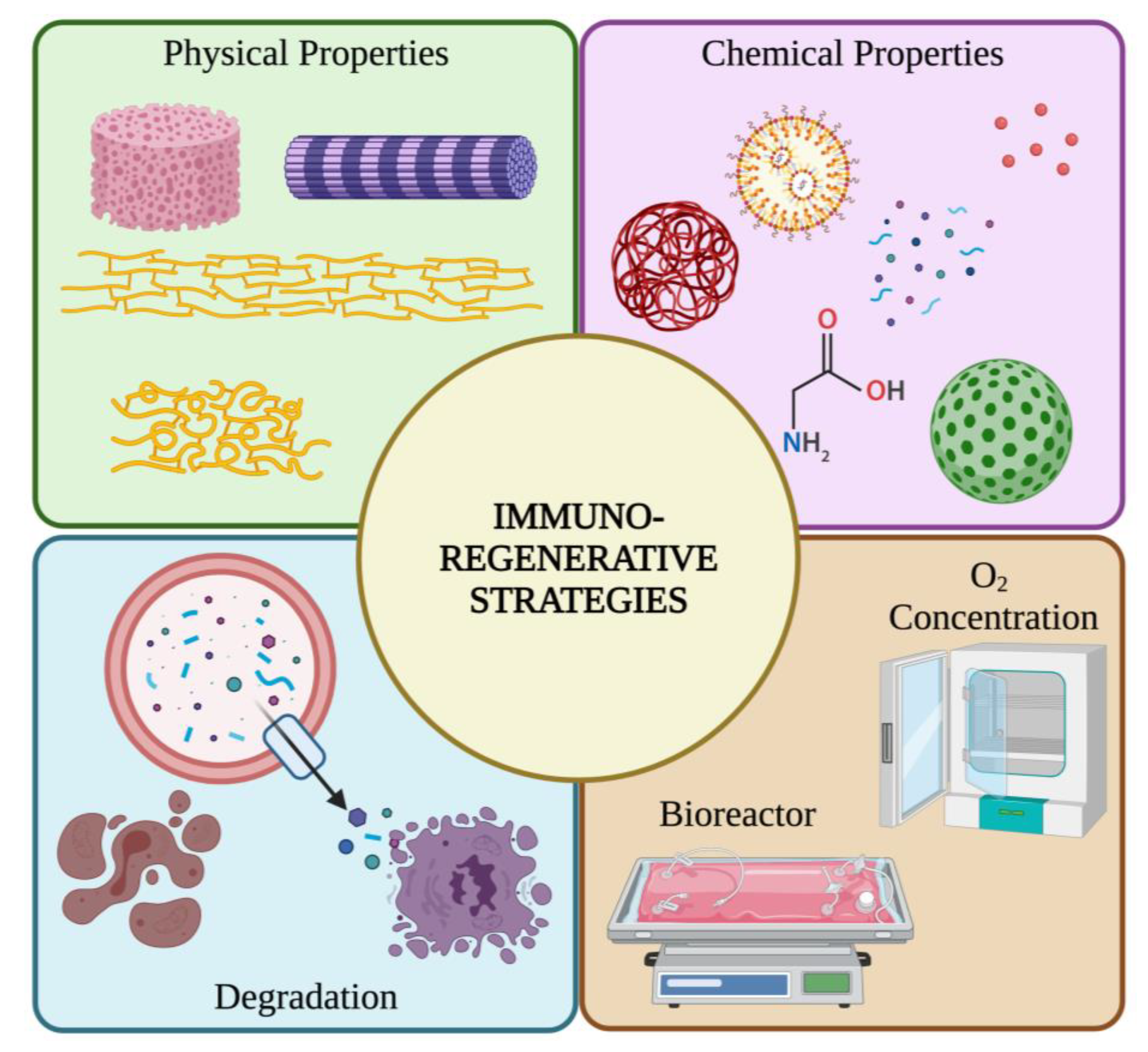
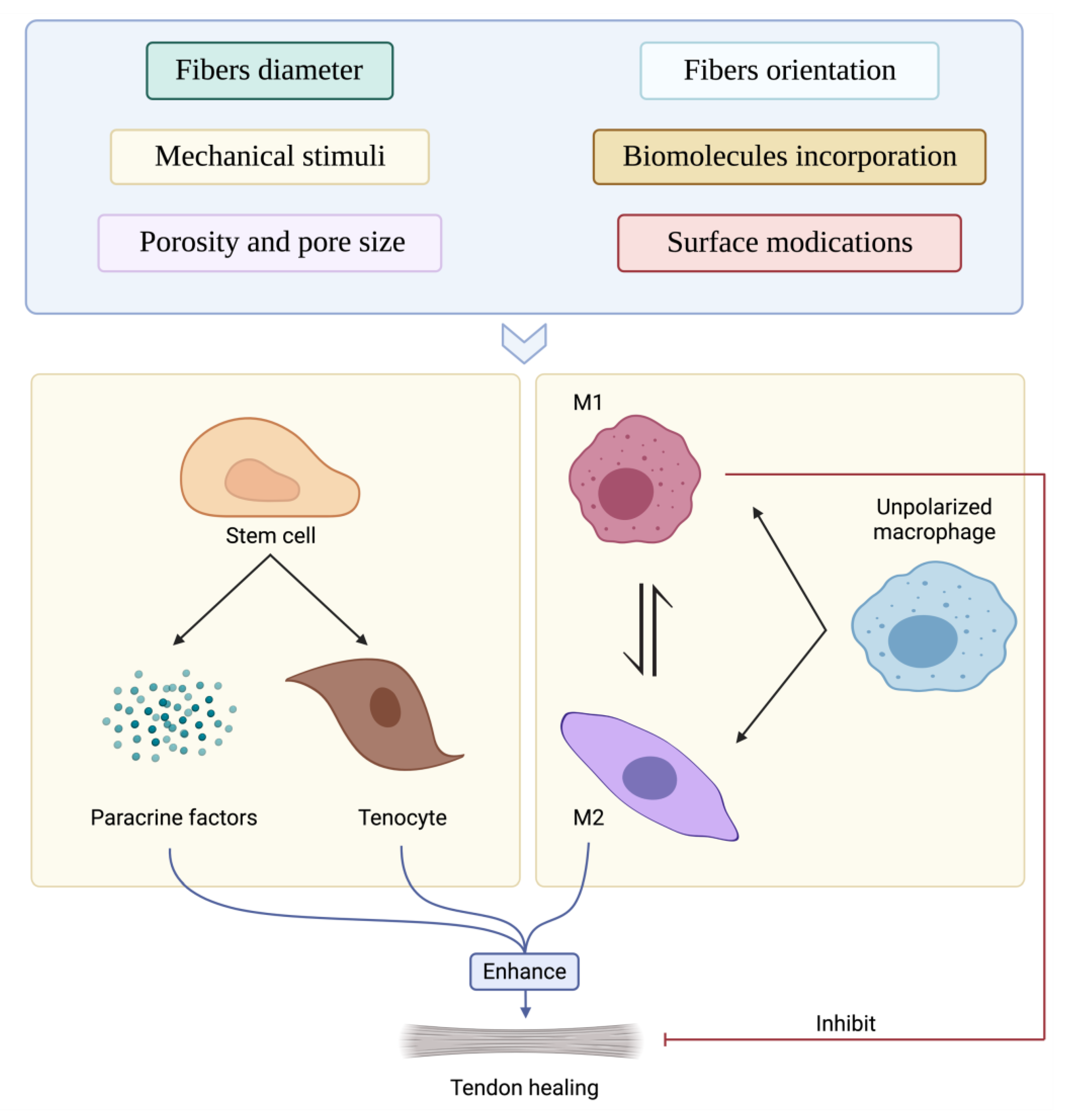
4. Scaffold Immunoengineering Strategies for Tendon TE Applications
4.1. Immuno-Induction of Scaffold on Immune Cells
4.1.1. Intrinsic Properties of the Scaffold on Immune Cells
Topography Effect on Immune Cells
Influence of Porosity and Pore Size on Immune Cells
Effect of Surface Modification and Biomaterial Chemistry on Immune Cells
4.1.2. Effect of Scaffold Degradation on Immune Cells
4.1.3. Effect of Environment Scaffold Subjection on Immune Cells
Mechanical and Electromagnetic Stimuli Influence on Immune Cells
How Oxygen Concentration Influences Immune Cells
4.2. Immuno-Induction of Scaffold on Stem Cells
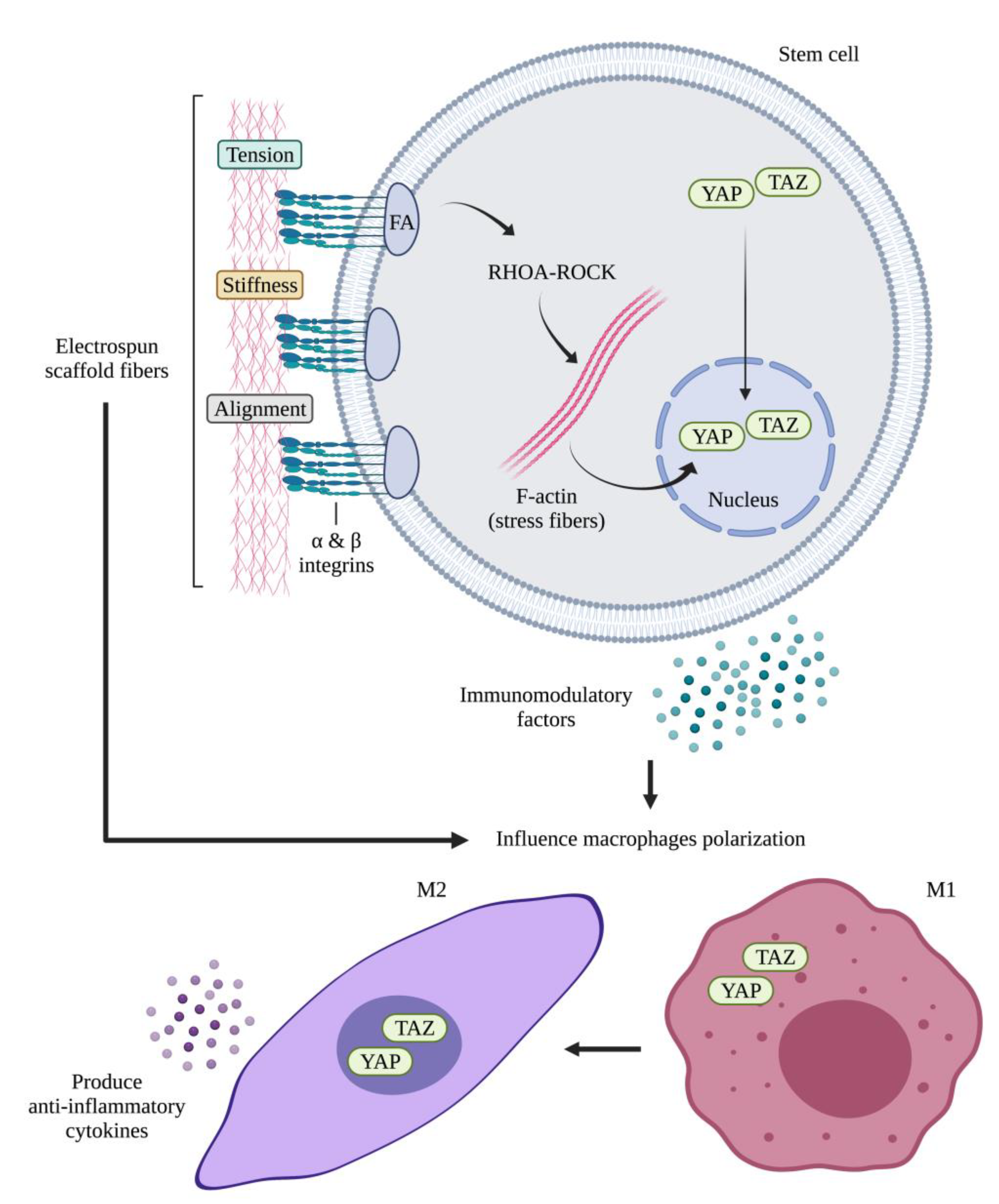
4.2.1. Intrinsic Properties of the Scaffold on Stem Cells
Topography Effect on Stem Cells: Fiber Alignment and Diameter Size
4.2.2. Effect of Environment Scaffold Subjection on Stem Cells
Mechanical Stimuli Influence on the Immunomodulation of Stem Cells
4.3. Biological Strategies to Enhance the Immunoregenerative Potential of the Scaffolds
5. Insights in the Molecular Pathways Regulating the Scaffold’s Mediated Immunomodulation
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmed, A.S.; Li, J.; Abdul, A.M.D.; Ahmed, M.; Östenson, C.-G.; Salo, P.T.; Hewitt, C.; Hart, D.A.; Ackermann, P.W. Compromised Neurotrophic and Angiogenic Regenerative Capability during Tendon Healing in a Rat Model of Type-II Diabetes. PLOS ONE 2017, 12, e0170748. [Google Scholar] [CrossRef]
- Castro, A.D.A.E.; Skare, T.L.; Nassif, P.A.N.; Sakuma, A.K.; Barros, W.H. Tendinopathy and Obesity. ABCD Arq. Bras. Cir. Dig. 2016, 29, 107–110. [Google Scholar] [CrossRef]
- Rees, J.D.; Maffulli, N.; Cook, J. Management of Tendinopathy. Am. J. Sports Med. 2009, 37, 1855–1867. [Google Scholar] [CrossRef] [PubMed]
- Lomas, A.J.; Ryan, C.N.M.; Sorushanova, A.; Shologu, N.; Sideri, A.I.; Tsioli, V.; Fthenakis, G.C.; Tzora, A.; Skoufos, I.; Quinlan, L.R.; et al. The Past, Present and Future in Scaffold-Based Tendon Treatments. Adv. Drug Deliv. Rev. 2015, 84, 257–277. [Google Scholar] [CrossRef] [PubMed]
- Patterson-Kane, J.C.; Rich, T. Achilles Tendon Injuries in Elite Athletes: Lessons in Pathophysiology From Their Equine Counterparts. ILAR J. 2014, 55, 86–99. [Google Scholar] [CrossRef]
- Sharma, P.; Maffulli, N. Tendon Injury and Tendinopathy: Healing and Repair. J. Bone Joint Surg. Am. 2005, 87, 187–202. [Google Scholar] [CrossRef]
- Millar, N.L.; Hueber, A.J.; Reilly, J.H.; Xu, Y.; Fazzi, U.G.; Murrell, G.A.C.; McInnes, I.B. Inflammation Is Present in Early Human Tendinopathy. Am. J. Sports Med. 2010, 38, 2085–2091. [Google Scholar] [CrossRef] [PubMed]
- D’Addona, A.; Maffulli, N.; Formisano, S.; Rosa, D. Inflammation in Tendinopathy. Surgeon 2017, 15, 297–302. [Google Scholar] [CrossRef]
- Stolk, M.; Klatte-Schulz, F.; Schmock, A.; Minkwitz, S.; Wildemann, B.; Seifert, M. New Insights into Tenocyte-Immune Cell Interplay in an in Vitro Model of Inflammation. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Millar, N.L.; Murrell, G.A.C.; Mcinnes, I.B. Inflammatory Mechanisms in Tendinopathy—Towards Translation. Nat. Rev. Rheumatol. 2017, 13, 110–122. [Google Scholar] [CrossRef]
- Millar, N.L.; Silbernagel, K.G.; Thorborg, K.; Kirwan, P.D.; Galatz, L.M.; Abrams, G.D.; Murrell, G.A.C.; McInnes, I.B.; Rodeo, S.A. Tendinopathy. Nat. Rev. Dis. Primer 2021, 7, 1. [Google Scholar] [CrossRef]
- Jomaa, G.; Kwan, C.-K.; Fu, S.-C.; Ling, S.K.-K.; Chan, K.-M.; Yung, P.S.-H.; Rolf, C. A Systematic Review of Inflammatory Cells and Markers in Human Tendinopathy. BMC Musculoskelet. Disord. 2020, 21, 78. [Google Scholar] [CrossRef] [PubMed]
- Alshomer, F.; Chaves, C.; Kalaskar, D.M. Advances in Tendon and Ligament Tissue Engineering: Materials Perspective. J. Mater. 2018, 2018, 1–17. [Google Scholar] [CrossRef]
- Sayegh, E.T.; Sandy, J.D.; Virk, M.S.; Romeo, A.A.; Wysocki, R.W.; Galante, J.O.; Trella, K.J.; Plaas, A.; Wang, V.M. Recent Scientific Advances Towards the Development of Tendon Healing Strategies. Curr. Tissue Eng. 2015, 4, 128–143. [Google Scholar] [CrossRef][Green Version]
- Ratcliffe, A.; Butler, D.L.; Dyment, N.A.; Cagle, P.J.; Proctor, C.S.; Ratcliffe, S.S.; Flatow, E.L. Scaffolds for Tendon and Ligament Repair and Regeneration. Ann. Biomed. Eng. 2015, 43, 819–831. [Google Scholar] [CrossRef]
- Reverchon, E.; Baldino, L.; Cardea, S.; De Marco, I. Biodegradable Synthetic Scaffolds for Tendon Regeneration. Muscles Ligaments Tendons J. 2012, 2, 181–186. [Google Scholar] [CrossRef]
- Russo, V.; El Khatib, M.; di Marcantonio, L.; Ancora, M.; Wyrwa, R.; Mauro, A.; Walter, T.; Weisser, J.; Citeroni, M.R.; Lazzaro, F.; et al. Tendon Biomimetic Electrospun PLGA Fleeces Induce an Early Epithelial-Mesenchymal Transition and Tenogenic Differentiation on Amniotic Epithelial Stem Cells. Cells 2020, 9, 303. [Google Scholar] [CrossRef]
- El Khatib, M.; Mauro, A.; Di Mattia, M.; Wyrwa, R.; Schweder, M.; Ancora, M.; Lazzaro, F.; Berardinelli, P.; Valbonetti, L.; Di Giacinto, O.; et al. Electrospun PLGA Fiber Diameter and Alignment of Tendon Biomimetic Fleece Potentiate Tenogenic Differentiation and Immunomodulatory Function of Amniotic Epithelial Stem Cells. Cells 2020, 9, 1207. [Google Scholar] [CrossRef]
- El Khatib, M.; Mauro, A.; Wyrwa, R.; Di Mattia, M.; Turriani, M.; Di Giacinto, O.; Kretzschmar, B.; Seemann, T.; Valbonetti, L.; Berardinelli, P.; et al. Fabrication and Plasma Surface Activation of Aligned Electrospun PLGA Fiber Fleeces with Improved Adhesion and Infiltration of Amniotic Epithelial Stem Cells Maintaining Their Teno-Inductive Potential. Molecules 2020, 25, 3176. [Google Scholar] [CrossRef]
- Santos, M.L.; Rodrigues, M.T.; Domingues, R.M.A.; Reis, R.L.; Gomes, M.E. Biomaterials as Tendon and Ligament Substitutes: Current Developments. In Regenerative Strategies for the Treatment of Knee Joint Disabilities; Studies in Mechanobiology, Tissue Engineering and Biomaterials; Oliveira, J.M., Reis, R.L., Eds.; Springer International Publishing: Cham, Switzerland, 2017; Volume 21, pp. 349–371. ISBN 978-3-319-44783-4. [Google Scholar]
- Desai, S.; Jayasuriya, C.T. Implementation of Endogenous and Exogenous Mesenchymal Progenitor Cells for Skeletal Tissue Regeneration and Repair. Bioengineering 2020, 7, 86. [Google Scholar] [CrossRef] [PubMed]
- Mariani, E.; Lisignoli, G.; Borzì, R.M.; Pulsatelli, L. Biomaterials: Foreign Bodies or Tuners for the Immune Response? Int. J. Mol. Sci. 2019, 20, 636. [Google Scholar] [CrossRef]
- Cun, X.; Hosta-Rigau, L. Topography: A Biophysical Approach to Direct the Fate of Mesenchymal Stem Cells in Tissue Engineering Applications. Nanomaterials 2020, 10, 2070. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.; Tang, L. Influence of Scaffold Design on Host Immune and Stem Cell Responses. Semin. Immunol. 2017, 29, 62–71. [Google Scholar] [CrossRef]
- Green, J.J. Immunoengineering Has Arrived. J. Biomed. Mater. Res. Part A 2021, 109, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Sridharan, R.; Cameron, A.R.; Kelly, D.J.; Kearney, C.J.; O’Brien, F.J. Biomaterial Based Modulation of Macrophage Polarization: A Review and Suggested Design Principles. Mater. Today 2015, 18, 313–325. [Google Scholar] [CrossRef]
- Sadtler, K.; Allen, B.W.; Estrellas, K.; Housseau, F.; Pardoll, D.M.; Elisseeff, J.H. The Scaffold Immune Microenvironment: Biomaterial-Mediated Immune Polarization in Traumatic and Nontraumatic Applications. Tissue Eng. Part A 2017, 23, 1044–1053. [Google Scholar] [CrossRef]
- Sadtler, K.; Wolf, M.T.; Ganguly, S.; Moad, C.A.; Chung, L.; Majumdar, S.; Housseau, F.; Pardoll, D.M.; Elisseeff, J.H. Divergent Immune Responses to Synthetic and Biological Scaffolds. Biomaterials 2019, 192, 405–415. [Google Scholar] [CrossRef]
- Saino, E.; Focarete, M.L.; Gualandi, C.; Emanuele, E.; Cornaglia, A.I.; Imbriani, M.; Visai, L. Effect of Electrospun Fiber Diameter and Alignment on Macrophage Activation and Secretion of Proinflammatory Cytokines and Chemokines. Biomacromolecules 2011, 12, 1900–1911. [Google Scholar] [CrossRef] [PubMed]
- Ogle, M.E.; Segar, C.E.; Sridhar, S.; Botchwey, E.A. Monocytes and Macrophages in Tissue Repair: Implications for Immunoregenerative Biomaterial Design. Exp. Biol. Med. 2016, 241, 1084–1097. [Google Scholar] [CrossRef]
- Motamedi, A.S.; Mirzadeh, H.; Hajiesmaeilbaigi, F.; Bagheri-Khoulenjani, S.; Shokrgozar, M. Effect of Electrospinning Parameters on Morphological Properties of PVDF Nanofibrous Scaffolds. Prog. Biomater. 2017, 6, 113–123. [Google Scholar] [CrossRef]
- Chen, Y.; Shu, Z.; Qian, K.; Wang, J.; Zhu, H. Harnessing the Properties of Biomaterial to Enhance the Immunomodulation of Mesenchymal Stem Cells. Tissue Eng. Part B Rev. 2019, 25, 492–499. [Google Scholar] [CrossRef]
- Tomás, A.R.; Gonçalves, A.I.; Paz, E.; Freitas, P.; Domingues, R.M.A.; Gomes, M.E. Magneto-Mechanical Actuation of Magnetic Responsive Fibrous Scaffolds Boosts Tenogenesis of Human Adipose Stem Cells. Nanoscale 2019, 11, 18255–18271. [Google Scholar] [CrossRef]
- Citeroni, M.R.; Ciardulli, M.C.; Russo, V.; Della Porta, G.; Mauro, A.; El Khatib, M.; Di Mattia, M.; Galesso, D.; Barbera, C.; Forsyth, N.R.; et al. In Vitro Innovation of Tendon Tissue Engineering Strategies. Int. J. Mol. Sci. 2020, 21, 6726. [Google Scholar] [CrossRef] [PubMed]
- Ilaltdinov, A.W.; Gong, Y.; Leong, D.J.; Fung, D.T.; Sun, H.B. Tendon Homeostasis: Overview. In Encyclopedia of Bone Biology; Elsevier: Amsterdam, The Netherlands, 2020; pp. 270–293. ISBN 978-0-12-814082-6. [Google Scholar]
- Thorpe, C.T.; Birch, H.L.; Clegg, P.D.; Screen, H.R.C. The Role of the Non-Collagenous Matrix in Tendon Function. Int. J. Exp. Pathol. 2013, 94, 248–259. [Google Scholar] [CrossRef]
- Kannus, P. Structure of the Tendon Connective Tissue. Scand. J. Med. Sci. Sports 2000, 10, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Nichols, A.E.C.; Best, K.T.; Loiselle, A.E. The Cellular Basis of Fibrotic Tendon Healing: Challenges and Opportunities. Transl. Res. 2019, 209, 156–168. [Google Scholar] [CrossRef] [PubMed]
- Docheva, D.; Müller, S.A.; Majewski, M.; Evans, C.H. Biologics for Tendon Repair. Adv. Drug Deliv. Rev. 2015, 84, 222–239. [Google Scholar] [CrossRef] [PubMed]
- Sensini, A.; Cristofolini, L. Biofabrication of Electrospun Scaffolds for the Regeneration of Tendons and Ligaments. Materials 2018, 11, 1963. [Google Scholar] [CrossRef]
- Moshiri, A.; Oryan, A.; Meimandi-Parizi, A. Role of Tissue-Engineered Artificial Tendon in Healing of a Large Achilles Tendon Defect Model in Rabbits. J. Am. Coll. Surg. 2013, 217, 421–441. [Google Scholar] [CrossRef]
- Rothrauff, B.B.; Lauro, B.B.; Yang, G.; Debski, R.E.; Musahl, V.; Tuan, R.S. Braided and Stacked Electrospun Nanofibrous Scaffolds for Tendon and Ligament Tissue Engineering. Tissue Eng. Part A 2017, 23, 378–389. [Google Scholar] [CrossRef]
- Zhang, B.; Luo, Q.; Deng, B.; Morita, Y.; Ju, Y.; Song, G. Construction of Tendon Replacement Tissue Based on Collagen Sponge and Mesenchymal Stem Cells by Coupled Mechano-Chemical Induction and Evaluation of Its Tendon Repair Abilities. Acta Biomater. 2018, 74, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Müller, S.A.; Dürselen, L.; Heisterbach, P.; Evans, C.; Majewski, M. Effect of a Simple Collagen Type I Sponge for Achilles Tendon Repair in a Rat Model. Am. J. Sports Med. 2016, 44, 1998–2004. [Google Scholar] [CrossRef] [PubMed]
- Gabler, C.; Saß, J.-O.; Gierschner, S.; Lindner, T.; Bader, R.; Tischer, T. In Vivo Evaluation of Different Collagen Scaffolds in an Achilles Tendon Defect Model. BioMed Res. Int. 2018, 2018, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Qian, S.; Wang, Z.; Zheng, Z.; Ran, J.; Zhu, J.; Chen, W. A Collagen and Silk Scaffold for Improved Healing of the Tendon and Bone Interface in a Rabbit Model. Med. Sci. Monit. 2019, 25, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Seo, Y.-K.; Kim, J.-H.; Eo, S.-R. Co-Effect of Silk and Amniotic Membrane for Tendon Repair. J. Biomater. Sci. Polym. Ed. 2016, 27, 1232–1247. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.-Y.; Chung, J.-W.; Park, H.-J.; Jiang, Y.-Y.; Park, J.-K.; Seo, Y.-K. Silk and Collagen Scaffolds for Tendon Reconstruction. Proc. Inst. Mech. Eng. 2014, 228, 388–396. [Google Scholar] [CrossRef]
- Zafar, A.; Neil, R. Extruded Collagen Fibers and Biphasic Collagen Sponge for Rotator Cuff Tendon Repair Application: An Ovine in-Vivo Study. Muscles Ligaments Tendons J. 2018, 8, 544–551. [Google Scholar] [CrossRef]
- Kishore, V.; Bullock, W.; Sun, X.; Van Dyke, W.S.; Akkus, O. Tenogenic Differentiation of Human MSCs Induced by the Topography of Electrochemically Aligned Collagen Threads. Biomaterials 2012, 33, 2137–2144. [Google Scholar] [CrossRef]
- Younesi, M.; Islam, A.; Kishore, V.; Anderson, J.M.; Akkus, O. Tenogenic Induction of Human MSCs by Anisotropically Aligned Collagen Biotextiles. Adv. Funct. Mater. 2014, 24, 5762–5770. [Google Scholar] [CrossRef]
- Islam, A.; Mbimba, T.; Younesi, M.; Akkus, O. Effects of Substrate Stiffness on the Tenoinduction of Human Mesenchymal Stem Cells. Acta Biomater. 2017, 58, 244–253. [Google Scholar] [CrossRef]
- Meimandi-Parizi, A.; Oryan, A.; Moshiri, A. Role of Tissue Engineered Collagen Based Tridimensional Implant on the Healing Response of the Experimentally Induced Large Achilles Tendon Defect Model in Rabbits: A Long Term Study with High Clinical Relevance. J. Biomed. Sci. 2013, 20, 28. [Google Scholar] [CrossRef] [PubMed]
- Full, S.M.; Delman, C.; Gluck, J.M.; Abdmaulen, R.; Shemin, R.J.; Heydarkhan-Hagvall, S. Effect of Fiber Orientation of Collagen-Based Electrospun Meshes on Human Fibroblasts for Ligament Tissue Engineering Applications. J. Biomed. Mater. Res. Part B Appl. Biomater. 2015, 103, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Zhi, Y.; Liu, W.; Zhang, P.; Jiang, J.; Chen, S. Electrospun Silk Fibroin Mat Enhances Tendon-Bone Healing in a Rabbit Extra-Articular Model. Biotechnol. Lett. 2016, 38, 1827–1835. [Google Scholar] [CrossRef]
- Sensini, A.; Gualandi, C.; Cristofolini, L.; Tozzi, G.; Dicarlo, M.; Teti, G.; Mattioli-Belmonte, M.; Focarete, M.L. Biofabrication of Bundles of Poly(Lactic Acid)-Collagen Blends Mimicking the Fascicles of the Human Achille Tendon. Biofabrication 2017, 9, 015025. [Google Scholar] [CrossRef]
- Sensini, A.; Gualandi, C.; Zucchelli, A.; Boyle, L.A.; Kao, A.P.; Reilly, G.C.; Tozzi, G.; Cristofolini, L.; Focarete, M.L. Tendon Fascicle-Inspired Nanofibrous Scaffold of Polylactic Acid/Collagen with Enhanced 3D-Structure and Biomechanical Properties. Sci. Rep. 2018, 8, 17167. [Google Scholar] [CrossRef] [PubMed]
- Lannutti, J.; Reneker, D.; Ma, T.; Tomasko, D.; Farson, D. Electrospinning for Tissue Engineering Scaffolds. Mater. Sci. Eng. C 2007, 27, 504–509. [Google Scholar] [CrossRef]
- Jiang, T.; Carbone, E.J.; Lo, K.W.-H.; Laurencin, C.T. Electrospinning of Polymer Nanofibers for Tissue Regeneration. Prog. Polym. Sci. 2015, 46, 1–24. [Google Scholar] [CrossRef]
- Chen, M.; Patra, P.K.; Warner, S.B.; Bhowmick, S. Optimization of Electrospinning Process Parameters For Tissue Engineering Scaffolds. Biophys. Rev. Lett. 2006, 01, 153–178. [Google Scholar] [CrossRef]
- Sahoo, S.; Toh, S.L.; Goh, J.C.H. A BFGF-Releasing Silk/PLGA-Based Biohybrid Scaffold for Ligament/Tendon Tissue Engineering Using Mesenchymal Progenitor Cells. Biomaterials 2010, 31, 2990–2998. [Google Scholar] [CrossRef]
- Erisken, C.; Zhang, X.; Moffat, K.L.; Levine, W.N.; Lu, H.H. Scaffold Fiber Diameter Regulates Human Tendon Fibroblast Growth and Differentiation. Tissue Eng. Part A 2013, 19, 519–528. [Google Scholar] [CrossRef]
- Wu, S.; Wang, Y.; Streubel, P.N.; Duan, B. Living Nanofiber Yarn-Based Woven Biotextiles for Tendon Tissue Engineering Using Cell Tri-Culture and Mechanical Stimulation. Acta Biomater. 2017, 62, 102–115. [Google Scholar] [CrossRef]
- Wang, X.; Ding, B.; Li, B. Biomimetic Electrospun Nanofibrous Structures for Tissue Engineering. Mater. Today 2013, 16, 229–241. [Google Scholar] [CrossRef]
- Lin, J.; Wang, X.; Ding, B.; Yu, J.; Sun, G.; Wang, M. Biomimicry via Electrospinning. Crit. Rev. Solid State Mater. Sci. 2012, 37, 94–114. [Google Scholar] [CrossRef]
- Nam, J.; Huang, Y.; Agarwal, S.; Lannutti, J. Improved Cellular Infiltration in Electrospun Fiber via Engineered Porosity. Tissue Eng. 2007, 13, 2249–2257. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.M.; Erisken, C.; Iskratsch, T.; Sheetz, M.; Levine, W.N.; Lu, H.H. Polymer Fiber-Based Models of Connective Tissue Repair and Healing. Biomaterials 2017, 112, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Bashur, C.A.; Shaffer, R.D.; Dahlgren, L.A.; Guelcher, S.A.; Goldstein, A.S. Effect of Fiber Diameter and Alignment of Electrospun Polyurethane Meshes on Mesenchymal Progenitor Cells. Tissue Eng. Part A 2009, 15, 2435–2445. [Google Scholar] [CrossRef] [PubMed]
- Moffat, K.L.; Kwei, A.S.-P.; Spalazzi, J.P.; Doty, S.B.; Levine, W.N.; Lu, H.H. Novel Nanofiber-Based Scaffold for Rotator Cuff Repair and Augmentation. Tissue Eng. Part A 2009, 15, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Leung, M.; Jana, S.; Tsao, C.-T.; Zhang, M. Tenogenic Differentiation of Human Bone Marrow Stem Cells via a Combinatory Effect of Aligned Chitosan–Poly-Caprolactone Nanofibers and TGF-Β3. J. Mater. Chem. B 2013, 1, 6516–6524. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yuan, H.; Liu, H.; Chen, X.; Lu, P.; Zhu, T.; Yang, L.; Yin, Z.; Heng, B.C.; Zhang, Y.; et al. Well-Aligned Chitosan-Based Ultrafine Fibers Committed Teno-Lineage Differentiation of Human Induced Pluripotent Stem Cells for Achilles Tendon Regeneration. Biomaterials 2015, 53, 716–730. [Google Scholar] [CrossRef]
- Nitti, P.; Gallo, N.; Natta, L.; Scalera, F.; Palazzo, B.; Sannino, A.; Gervaso, F. Influence of Nanofiber Orientation on Morphological and Mechanical Properties of Electrospun Chitosan Mats. J. Healthc. Eng. 2018, 2018, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Orr, S.B.; Chainani, A.; Hippensteel, K.J.; Kishan, A.; Gilchrist, C.; Garrigues, N.W.; Ruch, D.S.; Guilak, F.; Little, D. Aligned Multilayered Electrospun Scaffolds for Rotator Cuff Tendon Tissue Engineering. Acta Biomater. 2015, 24, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Ran, J.; Chen, W.; Hu, Y.; Zhu, T.; Chen, X.; Yin, Z.; Heng, B.C.; Feng, G.; Le, H.; et al. Alignment of Collagen Fiber in Knitted Silk Scaffold for Functional Massive Rotator Cuff Repair. Acta Biomater. 2017, 51, 317–329. [Google Scholar] [CrossRef]
- Chen, J.L.; Yin, Z.; Shen, W.L.; Chen, X.; Heng, B.C.; Zou, X.H.; Ouyang, H.W. Efficacy of HESC-MSCs in Knitted Silk-Collagen Scaffold for Tendon Tissue Engineering and Their Roles. Biomaterials 2010, 31, 9438–9451. [Google Scholar] [CrossRef]
- Gammon, J.M.; Jewell, C.M. Engineering Immune Tolerance with Biomaterials. Adv. Healthc. Mater. 2019, 8, e1801419. [Google Scholar] [CrossRef]
- Schmidt, D.R.; Waldeck, H.; Kao, W.J. Protein Adsorption to Biomaterials. In Biological Interactions on Materials Surfaces; Puleo, D.A., Bizios, R., Eds.; Springer US: New York, NY, USA, 2009; pp. 1–18. ISBN 978-0-387-98160-4. [Google Scholar]
- Felgueiras, H.P.; Antunes, J.C.; Martins, M.C.L.; Barbosa, M.A. Fundamentals of Protein and Cell Interactions in Biomaterials. In Peptides and Proteins as Biomaterials for Tissue Regeneration and Repair; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Arvind, V.; Huang, A.H. Reparative and Maladaptive Inflammation in Tendon Healing. Front. Bioeng. Biotechnol. 2021, 9, 719047. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Wu, Y.; Zhou, K.; Wu, D.; Yao, X.; Heng, B.C.; Zhou, J.; Liu, H.; Ouyang, H. Interplay of Forces and the Immune Response for Functional Tendon Regeneration. Front. Cell Dev. Biol. 2021, 9, 657621. [Google Scholar] [CrossRef]
- Noah, A.C.; Li, T.M.; Martinez, L.M.; Wada, S.; Swanson, J.B.; Disser, N.P.; Sugg, K.B.; Rodeo, S.A.; Lu, T.T.; Mendias, C.L. Adaptive and Innate Immune Cell Responses in Tendons and Lymph Nodes after Tendon Injury and Repair. J. Appl. Physiol. 2020, 128, 473–482. [Google Scholar] [CrossRef]
- Mauro, A.; Russo, V.; Di Marcantonio, L.; Berardinelli, P.; Martelli, A.; Muttini, A.; Mattioli, M.; Barboni, B. M1 and M2 Macrophage Recruitment during Tendon Regeneration Induced by Amniotic Epithelial Cell Allotransplantation in Ovine. Res. Vet. Sci. 2016, 105, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Su, N.; Gao, P.-L.; Wang, K.; Wang, J.-Y.; Zhong, Y.; Luo, Y. Fibrous Scaffolds Potentiate the Paracrine Function of Mesenchymal Stem Cells: A New Dimension in Cell-Material Interaction. Biomaterials 2017, 141, 74–85. [Google Scholar] [CrossRef]
- Wan, S.; Fu, X.; Ji, Y.; Li, M.; Shi, X.; Wang, Y. FAK- and YAP/TAZ Dependent Mechanotransduction Pathways Are Required for Enhanced Immunomodulatory Properties of Adipose-Derived Mesenchymal Stem Cells Induced by Aligned Fibrous Scaffolds. Biomaterials 2018, 171, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yang, Y.; Yildirimer, L.; Xu, T.; Zhao, X. Advanced Technology-Driven Therapeutic Interventions for Prevention of Tendon Adhesion: Design, Intrinsic and Extrinsic Factor Considerations. Acta Biomater. 2021, 124, 15–32. [Google Scholar] [CrossRef] [PubMed]
- Longo, U.G.; Lamberti, A.; Petrillo, S.; Maffulli, N.; Denaro, V. Scaffolds in Tendon Tissue Engineering. Stem Cells Int. 2012, 2012, 1–8. [Google Scholar] [CrossRef]
- Liang, X.; Ding, Y.; Zhang, Y.; Tse, H.-F.; Lian, Q. Paracrine Mechanisms of Mesenchymal Stem Cell-Based Therapy: Current Status and Perspectives. Cell Transplant. 2014, 23, 1045–1059. [Google Scholar] [CrossRef]
- Barboni, B.; Russo, V.; Berardinelli, P.; Mauro, A.; Valbonetti, L.; Sanyal, H.; Canciello, A.; Greco, L.; Muttini, A.; Gatta, V.; et al. Placental Stem Cells from Domestic Animals: Translational Potential and Clinical Relevance. Cell Transplant. 2018, 27, 93–116. [Google Scholar] [CrossRef]
- Ackermann, P.W. Tendinopathies in Sports: From Basic Research to the Field. In Sports Injuries: Prevention, Diagnosis, Treatment and Rehabilitation; Doral, M.N., Karlsson, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 1–15. ISBN 9783642368011. [Google Scholar]
- Garg, K.; Pullen, N.A.; Oskeritzian, C.A.; Ryan, J.J.; Bowlin, G.L. Macrophage Functional Polarization (M1/M2) in Response to Varying Fiber and Pore Dimensions of Electrospun Scaffolds. Biomaterials 2013, 34, 4439–4451. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Cui, Y.; Wang, J.; Yang, X.; Wu, Y.; Wang, K.; Gao, X.; Li, D.; Li, Y.; Zheng, X.L.; et al. The Effect of Thick Fibers and Large Pores of Electrospun Poly(ϵ-Caprolactone) Vascular Grafts on Macrophage Polarization and Arterial Regeneration. Biomaterials 2014, 35, 5700–5710. [Google Scholar] [CrossRef]
- Jiang, J.; Li, Z.; Wang, H.; Wang, Y.; Carlson, M.A.; Teusink, M.J.; MacEwan, M.R.; Gu, L.; Xie, J. Expanded 3D Nanofiber Scaffolds: Cell Penetration, Neovascularization, and Host Response. Adv. Healthc. Mater. 2016, 5, 2993–3003. [Google Scholar] [CrossRef] [PubMed]
- Schoenenberger, A.D.; Tempfer, H.; Lehner, C.; Egloff, J.; Mauracher, M.; Bird, A.; Widmer, J.; Maniura-Weber, K.; Fucentese, S.F.; Traweger, A.; et al. Macromechanics and Polycaprolactone Fiber Organization Drive Macrophage Polarization and Regulate Inflammatory Activation of Tendon in Vitro and in Vivo. Biomaterials 2020, 249, 120034. [Google Scholar] [CrossRef]
- Cao, H.; Mchugh, K.; Chew, S.Y.; Anderson, J.M. The Topographical Effect of Electrospun Nanofibrous Scaffolds on the in Vivo and in Vitro Foreign Body Reaction. J. Biomed. Mater. Res. Part A 2010, 93, 1151–1159. [Google Scholar] [CrossRef]
- Ballotta, V.; Driessen-Mol, A.; Bouten, C.V.; Baaijens, F.P.T. Strain-Dependent Modulation of Macrophage Polarization within Scaffolds. Biomaterials 2014, 35, 4919–4928. [Google Scholar] [CrossRef] [PubMed]
- Bonito, V.; de Kort, B.J.; Bouten, C.V.; Smits, A.I. Cyclic Strain Affects Macrophage Cytokine Secretion and Extracellular Matrix Turnover in Electrospun Scaffolds. Tissue Eng. Part A 2019, 25, 1310–1325. [Google Scholar] [CrossRef]
- Deepthi, S.; Nivedhitha Sundaram, M.; Deepti Kadavan, J.; Jayakumar, R. Layered Chitosan-Collagen Hydrogel/Aligned PLLA Nanofiber Construct for Flexor Tendon Regeneration. Carbohydr. Polym. 2016, 153, 492–500. [Google Scholar] [CrossRef]
- Shen, Y.; Tu, T.; Yi, B.; Wang, X.; Tang, H.; Liu, W.; Zhang, Y. Electrospun Acid-Neutralizing Fibers for the Amelioration of Inflammatory Response. Acta Biomater. 2019, 97, 200–215. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; He, N.; Yao, Z.; Wang, X.; Wang, H.; He, M.; Li, Y.; Qian, Y. An Integrative Dual-Layer Poly-L-Lactic Acid Fibrous Membrane Prevents Peritendinous Adhesions. Front. Bioeng. Biotechnol. 2020, 8, 387. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Byun, H.; Madhurakkat Perikamana, S.K.; Lee, S.; Shin, H. Current Advances in Immunomodulatory Biomaterials for Bone Regeneration. Adv. Healthc. Mater. 2019, 8, 1801106. [Google Scholar] [CrossRef] [PubMed]
- Andorko, J.I.; Jewell, C.M. Designing Biomaterials with Immunomodulatory Properties for Tissue Engineering and Regenerative Medicine. Bioeng. Transl. Med. 2017, 2, 139–155. [Google Scholar] [CrossRef]
- Kunzler, T.P.; Drobek, T.; Schuler, M.; Spencer, N.D. Systematic Study of Osteoblast and Fibroblast Response to Roughness by Means of Surface-Morphology Gradients. Biomaterials 2007, 28, 2175–2182. [Google Scholar] [CrossRef]
- Prasad, B.R.; Brook, M.A.; Smith, T.; Zhao, S.; Chen, Y.; Sheardown, H.; D’souza, R.; Rochev, Y. Controlling Cellular Activity by Manipulating Silicone Surface Roughness. Colloids Surf. B Biointerfaces 2010, 78, 237–242. [Google Scholar] [CrossRef]
- Vance, R.J.; Miller, D.C.; Thapa, A.; Haberstroh, K.M.; Webster, T.J. Decreased Fibroblast Cell Density on Chemically Degraded Poly-Lactic-Co-Glycolic Acid, Polyurethane, and Polycaprolactone. Biomaterials 2004, 25, 2095–2103. [Google Scholar] [CrossRef]
- Kim, H.; Murakami, H.; Chehroudi, B.; Textor, M.; Brunette, D.M. Effects of Surface Topography on the Connective Tissue Attachment to Subcutaneous Implants. Int. J. Oral Maxillofac. Implants 2006, 21, 354–365. [Google Scholar]
- Cheng, L.; Wang, Y.; Sun, G.; Wen, S.; Deng, L.; Zhang, H.; Cui, W. Hydration-Enhanced Lubricating Electrospun Nanofibrous Membranes Prevent Tissue Adhesion. Research 2020, 2020, 12. [Google Scholar] [CrossRef]
- Saidina, D.S.; Zubir, S.A.; Fontana, S.; Hérold, C.; Mariatti, M. Synthesis and Characterization of Graphene-Based Inks for Spray-Coating Applications. J. Electron. Mater. 2019, 48, 5757–5770. [Google Scholar] [CrossRef]
- Zhou, H.; Zhao, K.; Li, W.; Yang, N.; Liu, Y.; Chen, C.; Wei, T. The Interactions between Pristine Graphene and Macrophages and the Production of Cytokines/Chemokines via TLR- and NF-ΚB-Related Signaling Pathways. Biomaterials 2012, 33, 6933–6942. [Google Scholar] [CrossRef]
- Peixoto, T.; Paiva, M.C.; Marques, A.T.; Lopes, M.A. Potential of Graphene–Polymer Composites for Ligament and Tendon Repair: A Review. Adv. Eng. Mater. 2020, 22, 2000492. [Google Scholar] [CrossRef]
- Lasocka, I.; Jastrzebska, E.; Szulc-Dabrowska, L.; Skibniewski, M.; Pasternak, I.; Kalbacova, M.H.; Skibniewska, E.M. The Effects of Graphene and Mesenchymal Stem Cells in Cutaneous Wound Healing and Their Putative Action Mechanism. Int. J. Nanomed. 2019, 14, 2281–2299. [Google Scholar] [CrossRef]
- Ma, J.; Liu, R.; Wang, X.; Liu, Q.; Chen, Y.; Valle, R.P.; Zuo, Y.Y.; Xia, T.; Liu, S. Crucial Role of Lateral Size for Graphene Oxide in Activating Macrophages and Stimulating Pro-Inflammatory Responses in Cells and Animals. ACS Nano 2015, 9, 10498–10515. [Google Scholar] [CrossRef]
- Orecchioni, M.; Bedognetti, D.; Newman, L.; Fuoco, C.; Spada, F.; Hendrickx, W.; Marincola, F.M.; Sgarrella, F.; Rodrigues, A.F.; Ménard-Moyon, C.; et al. Single-Cell Mass Cytometry and Transcriptome Profiling Reveal the Impact of Graphene on Human Immune Cells. Nat. Commun. 2017, 8, 1109. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.P.; Lazzaretto, B.; Hultenby, K.; Newman, L.; Rodrigues, A.F.; Lozano, N.; Kostarelos, K.; Malmberg, P.; Fadeel, B. Graphene Oxide Elicits Membrane Lipid Changes and Neutrophil Extracellular Trap Formation. Chem 2018, 4, 334–358. [Google Scholar] [CrossRef]
- Zhang, Y.; Nayak, T.R.; Hong, H.; Cai, W. Graphene: A Versatile Nanoplatform for Biomedical Applications. Nanoscale 2012, 4, 3833–3842. [Google Scholar] [CrossRef] [PubMed]
- Pattnaik, S.; Swain, K.; Lin, Z. Graphene and Graphene-Based Nanocomposites: Biomedical Applications and Biosafety. J. Mater. Chem. B 2016, 4, 7813–7831. [Google Scholar] [CrossRef]
- Shin, S.R.; Li, Y.-C.; Jang, H.L.; Khoshakhlagh, P.; Akbari, M.; Nasajpour, A.; Zhang, Y.S.; Tamayol, A.; Khademhosseini, A. Graphene-Based Materials for Tissue Engineering. Adv. Drug Deliv. Rev. 2016, 105, 255–274. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Wan, Q.; Pei, X. Graphene Family Materials in Bone Tissue Regeneration: Perspectives and Challenges. Nanoscale Res. Lett. 2018, 13, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Wahab, I.F.; Razak, S.I.A.; Azmi, N.S.; Dahli, F.N.; Yusof, A.H.M.; Nayan, N.H.M. Electrospun Graphene Oxide-Based Nanofibres. In Advances in Carbon Nanostructures; Silva, A.M.T., Carabineiro, S.A.C., Eds.; InTech: Rijeka, Croatia, 2016; ISBN 978-953-51-2642-3. [Google Scholar]
- Fong, D.; Hoemann, C.D. Chitosan Immunomodulatory Properties: Perspectives on the Impact of Structural Properties and Dosage. Future Sci. OA 2018, 4, FSO225. [Google Scholar] [CrossRef]
- Van-Pham, D.-T.; Quyen, T.T.B.; Toan, P.V.; Nguyen, C.-N.; Ho, M.H.; Thien, D.V.H. Temperature Effects on Electrospun Chitosan Nanofibers. Green Process. Synth. 2020, 9, 488–495. [Google Scholar] [CrossRef]
- Nguyen, P.K.; Baek, K.; Deng, F.; Criscione, J.D.; Tuan, R.S.; Kuo, C.K. Tendon Tissue-Engineering Scaffolds. In Biomaterials Science; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1351–1371. ISBN 978-0-12-816137-1. [Google Scholar]
- Liu, W.; Cao, Y. Application of Scaffold Materials in Tissue Reconstruction in Immunocompetent Mammals: Our Experience and Future Requirements. Biomaterials 2007, 28, 5078–5086. [Google Scholar] [CrossRef]
- Yang, D.; Xiao, J.; Wang, B.; Li, L.; Kong, X.; Liao, J. The Immune Reaction and Degradation Fate of Scaffold in Cartilage/Bone Tissue Engineering; Elsevier Ltd: Amsterdam, The Netherlands, 2019; Volume 104. [Google Scholar]
- Zhang, X.; Wu, Y.; Pan, Z.; Sun, H.; Wang, J.; Yu, D.; Zhu, S.; Dai, J.; Chen, Y.; Tian, N.; et al. The Effects of Lactate and Acid on Articular Chondrocytes Function: Implications for Polymeric Cartilage Scaffold Design. Acta Biomater. 2016, 42, 329–340. [Google Scholar] [CrossRef]
- Landes, C.A.; Ballon, A.; Roth, C. In-Patient Versusin Vitro Degradation of P(L/DL)LA and PLGA. J. Biomed. Mater. Res. Part B Appl. Biomater. 2006, 76B, 403–411. [Google Scholar] [CrossRef]
- Ayyoob, M.; Kim, Y. Effect of Chemical Composition Variant and Oxygen Plasma Treatments on the Wettability of PLGA Thin Films, Synthesized by Direct Copolycondensation. Polymers 2018, 10, 1132. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.-W.; Su, S.-H.; Eberhart, R.C.; Tang, L. Phagocyte Responses to Degradable Polymers. J. Biomed. Mater. Res. Part A 2007, 82A, 492–497. [Google Scholar] [CrossRef]
- Tabata, Y.; Ikada, Y. Macrophage Phagocytosis of Biodegradable Microspheres Composed OfL-Lactic Acid/Glycolic Acid Homo- and Copolymers. J. Biomed. Mater. Res. 1988, 22, 837–858. [Google Scholar] [CrossRef]
- Fuchs, A.-K.; Syrovets, T.; Haas, K.A.; Loos, C.; Musyanovych, A.; Mailänder, V.; Landfester, K.; Simmet, T. Carboxyl- and Amino-Functionalized Polystyrene Nanoparticles Differentially Affect the Polarization Profile of M1 and M2 Macrophage Subsets. Biomaterials 2016, 85, 78–87. [Google Scholar] [CrossRef]
- Adams, S.; Wuescher, L.M.; Worth, R.; Yildirim-Ayan, E. Mechano-Immunomodulation: Mechanoresponsive Changes in Macrophage Activity and Polarization. Ann. Biomed. Eng. 2019, 47, 2213–2231. [Google Scholar] [CrossRef]
- Mevoy, A.; Jeyam, M.; Ferrier, G.; Evans, C.E.; Andrew, J.G. Synergistic Effect of Particles and Cyclic Pressure on Cytokine Production in Human Monocyte/Macrophages: Proposed Role in Periprosthetic Osteolysis. Bone 2002, 30, 171–177. [Google Scholar] [CrossRef]
- Vinhas, A.; Almeida, A.F.; Gonçalves, A.I.; Rodrigues, M.T.; Gomes, M.E. Magnetic Stimulation Drives Macrophage Polarization in Cell to–Cell Communication with Il-1β Primed Tendon Cells. Int. J. Mol. Sci. 2020, 21, 5441. [Google Scholar] [CrossRef]
- Benjamin, M.; Ralphs, J.R. Tendons and Ligaments--an Overview. Histol. Histopathol. 1997, 12, 1135–1144. [Google Scholar] [PubMed]
- Vailas, A.C.; Tipton, C.M.; Laughlin, H.L.; Tcheng, T.K.; Matthes, R.D. Physical Activity and Hypophysectomy on the Aerobic Capacity of Ligaments and Tendons. J. Appl. Physiol. 1978, 44, 542–546. [Google Scholar] [CrossRef]
- Hirai, D.M.; Colburn, T.D.; Craig, J.C.; Hotta, K.; Kano, Y.; Musch, T.I.; Poole, D.C. Skeletal Muscle Interstitial O2 Pressures: Bridging the Gap between the Capillary and Myocyte. Microcirculation 2019, 26, e12497. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Prado, E.; Dunn, J.F.; Vasconez, J.; Castillo, D.; Viscor, G. Partial Pressure of Oxygen in the Human Body: A General Review. Am. J. Blood Res. 2019, 9, 1–14. [Google Scholar] [PubMed]
- Cardwell, R.D.; Dahlgren, L.A.; Goldstein, A.S. Electrospun Fibre Diameter, Not Alignment, Affects Mesenchymal Stem Cell Differentiation into the Tendon/Ligament Lineage. J. Tissue Eng. Regen. Med. 2014, 8, 937–945. [Google Scholar] [CrossRef]
- Tu, T.; Shen, Y.; Wang, X.; Zhang, W.; Zhou, G.; Zhang, Y.; Wang, W.; Liu, W. Tendon ECM Modified Bioactive Electrospun Fibers Promote MSC Tenogenic Differentiation and Tendon Regeneration. Appl. Mater. Today 2020, 18, 100495. [Google Scholar] [CrossRef]
- Ciardulli, M.C.; Marino, L.; Lovecchio, J.; Giordano, E.; Forsyth, N.R.; Selleri, C.; Maffulli, N.; Porta, G.D. Tendon and Cytokine Marker Expression by Human Bone Marrow Mesenchymal Stem Cells in a Hyaluronate/Poly-Lactic-Co-Glycolic Acid (PLGA)/Fibrin Three-Dimensional (3D) Scaffold. Cells 2020, 9, 1268. [Google Scholar] [CrossRef]
- Barboni, B.; Russo, V.; Gatta, V.; Bernabò, N.; Berardinelli, P.; Mauro, A.; Martelli, A.; Valbonetti, L.; Muttini, A.; Di Giacinto, O.; et al. Therapeutic Potential of HAECs for Early Achilles Tendon Defect Repair through Regeneration. J. Tissue Eng. Regen. Med. 2018, 12. [Google Scholar] [CrossRef] [PubMed]
- Barboni, B.; Russo, V.; Curini, V.; Mauro, A.; Martelli, A.; Muttini, A.; Bernabò, N.; Valbonetti, L.; Marchisio, M.; Di Giacinto, O.; et al. Achilles Tendon Regeneration Can Be Improved by Amniotic Epithelial Cell Allotransplantation. Cell Transplant. 2012, 21, 2377–2395. [Google Scholar] [CrossRef]
- Citeroni, M.R.; Mauro, A.; Ciardulli, M.C.; Di Mattia, M.; El Khatib, M.; Russo, V.; Turriani, M.; Santer, M.; Della Porta, G.; Maffulli, N.; et al. Amnion-Derived Teno-Inductive Secretomes: A Novel Approach to Foster Tendon Differentiation and Regeneration in an Ovine Model. Front. Bioeng. Biotechnol. 2021, 9, 649288. [Google Scholar] [CrossRef]
- Lange-Consiglio, A.; Lazzari, B.; Perrini, C.; Pizzi, F.; Stella, A.; Cremonesi, F.; Capra, E. MicroRNAs of Equine Amniotic Mesenchymal Cell-Derived Microvesicles and Their Involvement in Anti-Inflammatory Processes. Cell Transplant. 2018, 27, 45–54. [Google Scholar] [CrossRef]
- Magatti, M.; Vertua, E.; Cargnoni, A.; Silini, A.; Parolini, O. The Immunomodulatory Properties of Amniotic Cells: The Two Sides of the Coin. Cell Transplant. 2018, 27, 31–44. [Google Scholar] [CrossRef]
- Parolini, O.; Souza-Moreira, L.; O’Valle, F.; Magatti, M.; Hernandez-Cortes, P.; Gonzalez-Rey, E.; Delgado, M. Therapeutic Effect of Human Amniotic Membrane-Derived Cells on Experimental Arthritis and Other Inflammatory Disorders. Arthritis Rheumatol. 2014, 66, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Barboni, B.; Russo, V.; Curini, V.; Martelli, A.; Berardinelli, P.; Mauro, A.; Mattioli, M.; Marchisio, M.; Bonassi Signoroni, P.; Parolini, O.; et al. Gestational Stage Affects Amniotic Epithelial Cells Phenotype, Methylation Status, Immunomodulatory and Stemness Properties. Stem Cell Rev. Rep. 2014, 10, 725–741. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, X.; Zhang, E.; Yang, L.; Yuan, H.; Tu, W.; Zhang, H.; Yin, Z.; Shen, W.; Chen, X.; et al. An Epigenetic Bioactive Composite Scaffold with Well-Aligned Nanofibers for Functional Tendon Tissue Engineering. Acta Biomater. 2018, 66, 141–156. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Lee, R.H.; Bazhanov, N.; Oh, J.Y.; Prockop, D.J. Anti-Inflammatory Protein TSG-6 Secreted by Activated MSCs Attenuates Zymosan-Induced Mouse Peritonitis by Decreasing TLR2/NF-ΚB Signaling in Resident Macrophages. Blood 2011, 118, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Dyer, D.P.; Salanga, C.L.; Johns, S.C.; Valdambrini, E.; Fuster, M.M.; Milner, C.M.; Day, A.J.; Handel, T.M. The Anti-Inflammatory Protein TSG-6 Regulates Chemokine Function by Inhibiting Chemokine/Glycosaminoglycan Interactions. J. Biol. Chem. 2016, 291, 12627–12640. [Google Scholar] [CrossRef] [PubMed]
- Harris, S.G.; Padilla, J.; Koumas, L.; Ray, D.; Phipps, R.P. Prostaglandins as Modulators of Immunity. Trends Immunol. 2002, 23, 144–150. [Google Scholar] [CrossRef]
- Manuelpillai, U.; Lourensz, D.; Vaghjiani, V.; Tchongue, J.; Lacey, D.; Tee, J.-Y.; Murthi, P.; Chan, J.; Hodge, A.; Sievert, W. Human Amniotic Epithelial Cell Transplantation Induces Markers of Alternative Macrophage Activation and Reduces Established Hepatic Fibrosis. PLoS ONE 2012, 7, e38631. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.E.; Gumucio, J.P.; Sugg, K.B.; Bedi, A.; Mendias, C.L. MMP Inhibition as a Potential Method to Augment the Healing of Skeletal Muscle and Tendon Extracellular Matrix. J. Appl. Physiol. 2013, 115, 884–891. [Google Scholar] [CrossRef]
- Qian, Y.; Li, L.; Song, Y.; Dong, L.; Chen, P.; Li, X.; Cai, K.; Germershaus, O.; Yang, L.; Fan, Y. Surface Modification of Nanofibrous Matrices via Layer-by-Layer Functionalized Silk Assembly for Mitigating the Foreign Body Reaction. Biomaterials 2018, 164, 22–37. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Ge, L.; Wang, H.; Cheng, Y.; Gorantla, S.; Poluektova, L.Y.; Gombart, A.F.; Xie, J. Eluted 25-Hydroxyvitamin D3 from Radially Aligned Nanofiber Scaffolds Enhances Cathelicidin Production While Reducing Inflammatory Response in Human Immune System-Engrafted Mice. Acta Biomater. 2019, 97, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Chen, H.; Wu, T.; Pan, G.; Fan, C.; Xu, Y.; Cui, W. Macrophage Infiltration of Electrospun Polyester Fibers. Biomater. Sci. 2017, 5, 1579–1587. [Google Scholar] [CrossRef]
- Su, N.; Hao, Y.; Wang, F.; Hou, W.; Chen, H.; Luo, Y. Mesenchymal Stromal Exosome–Functionalized Scaffolds Induce Innate and Adaptive Immunomodulatory Responses toward Tissue Repair. Sci. Adv. 2021, 7, eabf7207. [Google Scholar] [CrossRef]
- Xi, K.; Gu, Y.; Tang, J.; Chen, H.; Xu, Y.; Wu, L.; Cai, F.; Deng, L.; Yang, H.; Shi, Q.; et al. Microenvironment-Responsive Immunoregulatory Electrospun Fibers for Promoting Nerve Function Recovery. Nat. Commun. 2020, 11, 1–18. [Google Scholar] [CrossRef]
- Song, W.; Ma, Z.; Wang, C.; Li, H.; He, Y. Pro-Chondrogenic and Immunomodulatory Melatonin-Loaded Electrospun Membranes for Tendon-to-Bone Healing. J. Mater. Chem. B 2019, 7, 6564–6575. [Google Scholar] [CrossRef]
- Dong, L.; Li, L.; Song, Y.; Fang, Y.; Liu, J.; Chen, P.; Wang, S.; Wang, C.; Xia, T.; Liu, W.; et al. MSC-Derived Immunomodulatory Extracellular Matrix Functionalized Electrospun Fibers for Mitigating Foreign-Body Reaction and Tendon Adhesion. Acta Biomater. 2021, 133, 280–296. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cao, Z.; Cheng, R.; Qin, M.; Zhang, D.; Deng, L.; Chen, X.; Cui, W. Immunomodulated Electrospun Fibrous Scaffolds via BFGF Camouflage for Pelvic Regeneration. Appl. Mater. Today 2019, 15, 570–581. [Google Scholar] [CrossRef]
- Kim, O.-H.; Yoon, O.J.; Lee, H.J. Silk Fibroin Scaffolds Potentiate Immunomodulatory Function of Human Mesenchymal Stromal Cells. Biochem. Biophys. Res. Commun. 2019, 519, 323–329. [Google Scholar] [CrossRef] [PubMed]
- De la Ossa, J.G.; Fusco, A.; Azimi, B.; Esposito Salsano, J.; Digiacomo, M.; Coltelli, M.-B.; De Clerck, K.; Roy, I.; Macchia, M.; Lazzeri, A.; et al. Immunomodulatory Activity of Electrospun Polyhydroxyalkanoate Fiber Scaffolds Incorporating Olive Leaf Extract. Appl. Sci. 2021, 11, 4006. [Google Scholar] [CrossRef]
- Barrachina, L.; Remacha, A.R.; Romero, A.; Vázquez, F.J.; Albareda, J.; Prades, M.; Gosálvez, J.; Roy, R.; Zaragoza, P.; Martín-Burriel, I.; et al. Priming Equine Bone Marrow-Derived Mesenchymal Stem Cells with Proinflammatory Cytokines: Implications in Immunomodulation–Immunogenicity Balance, Cell Viability, and Differentiation Potential. Stem Cells Dev. 2017, 26, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Andia, I.; Rubio-Azpeitia, E.; Maffulli, N. Platelet-Rich Plasma Modulates the Secretion of Inflammatory/Angiogenic Proteins by Inflamed Tenocytes. Clin. Orthop. Relat. Res. 2015, 473, 1624–1634. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Li, Y.; Zhang, T.; Shi, M.; Song, X.; Yang, S.; Liu, H.; Zhang, M.; Cui, Q.; Li, Z. Hepatocyte Growth Factor-Induced Tendon Stem Cell Conditioned Medium Promotes Healing of Injured Achilles Tendon. Front. Cell Dev. Biol. 2021, 9, 654084. [Google Scholar] [CrossRef]
- Zarychta-Wiśniewska, W.; Burdzinska, A.; Kulesza, A.; Gala, K.; Kaleta, B.; Zielniok, K.; Siennicka, K.; Sabat, M.; Paczek, L. Bmp-12 Activates Tenogenic Pathway in Human Adipose Stem Cells and Affects Their Immunomodulatory and Secretory Properties. BMC Cell Biol. 2017, 18, 13. [Google Scholar] [CrossRef]
- Barker, T.; Martins, T.B.; Hill, H.R.; Kjeldsberg, C.R.; Dixon, B.M.; Schneider, E.D.; Henriksen, V.T.; Weaver, L.K. Vitamin D Sufficiency Associates with an Increase in Anti-Inflammatory Cytokines after Intense Exercise in Humans. Cytokine 2014, 65, 134–137. [Google Scholar] [CrossRef]
- Topilski, I.; Flaishon, L.; Naveh, Y.; Harmelin, A.; Levo, Y.; Shachar, I. The Anti-Inflammatory Effects of 1,25-Dihydroxyvitamin D3 on Th2 Cells in Vivo Are Due in Part to the Control of Integrin-Mediated T Lymphocyte Homing. Eur. J. Immunol. 2004, 34, 1068–1076. [Google Scholar] [CrossRef]
- Zhang, Y.; Leung, D.Y.M.; Richers, B.N.; Liu, Y.; Remigio, L.K.; Riches, D.W.; Goleva, E. Vitamin D Inhibits Monocyte/Macrophage Proinflammatory Cytokine Production by Targeting MAPK Phosphatase-1. J. Immunol. 2012, 188, 2127–2135. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Xu, Y.; Chen, S.; Tan, Z.; Xiong, K.; Li, Y.; Ye, Y.; Luo, Z.-P.; He, F.; Gong, Y. Rescue of Proinflammatory Cytokine-Inhibited Chondrogenesis by the Antiarthritic Effect of Melatonin in Synovium Mesenchymal Stem Cells via Suppression of Reactive Oxygen Species and Matrix Metalloproteinases. Free Radic. Biol. Med. 2014, 68, 234–246. [Google Scholar] [CrossRef]
- Xia, Y.; Chen, S.; Zeng, S.; Zhao, Y.; Zhu, C.; Deng, B.; Zhu, G.; Yin, Y.; Wang, W.; Hardeland, R.; et al. Melatonin in Macrophage Biology: Current Understanding and Future Perspectives. J. Pineal Res. 2019, 66, e12547. [Google Scholar] [CrossRef]
- Bonito, V.; Smits, A.I.; Goor, O.J.G.M.; Ippel, B.D.; Driessen-Mol, A.; Münker, T.J.A.G.; Bosman, A.W.; Mes, T.; Dankers, P.Y.W.; Bouten, C.V.C. Modulation of Macrophage Phenotype and Protein Secretion via Heparin-IL-4 Functionalized Supramolecular Elastomers. Acta Biomater. 2018, 71, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Boehler, R.M.; Kuo, R.; Shin, S.; Goodman, A.G.; Pilecki, M.A.; Leonard, J.N.; Shea, L.D. Lentivirus Delivery of IL-10 to Promote and Sustain Macrophage Polarization towards an Anti-Inflammatory Phenotype: Lentivirus Delivery of IL-10. Biotechnol. Bioeng. 2014, 111, 1210–1221. [Google Scholar] [CrossRef] [PubMed]
- Spiller, K.L.; Nassiri, S.; Witherel, C.E.; Anfang, R.R.; Ng, J.; Nakazawa, K.R.; Yu, T.; Vunjak-Novakovic, G. Sequential Delivery of Immunomodulatory Cytokines to Facilitate the M1-to-M2 Transition of Macrophages and Enhance Vascularization of Bone Scaffolds. Biomaterials 2015, 37, 194–207. [Google Scholar] [CrossRef]
- Kearney, C.J.; Mooney, D.J. Macroscale Delivery Systems for Molecular and Cellular Payloads. Nat. Mater. 2013, 12, 1004–1017. [Google Scholar] [CrossRef] [PubMed]
- McWhorter, F.Y.; Wang, T.; Nguyen, P.; Chung, T.; Liu, W.F. Modulation of Macrophage Phenotype by Cell Shape. Proc. Natl. Acad. Sci. USA 2013, 110, 17253–17258. [Google Scholar] [CrossRef]
- Ryan, J.M.; Barry, F.; Murphy, J.M.; Mahon, B.P. Interferon-γ Does Not Break, but Promotes the Immunosuppressive Capacity of Adult Human Mesenchymal Stem Cells. Clin. Exp. Immunol. 2007, 149, 353–363. [Google Scholar] [CrossRef]
- Zhang, D.; Lin, Z.Y.; Cheng, R.; Wu, W.; Yu, J.; Zhao, X.; Chen, X.; Cui, W. Reinforcement of Transvaginal Repair Using Polypropylene Mesh Functionalized with Basic Fibroblast Growth Factor. Colloids Surf. B Biointerfaces 2016, 142, 10–19. [Google Scholar] [CrossRef]
- Lockyer, S.; Rowland, I.; Spencer, J.P.E.; Yaqoob, P.; Stonehouse, W. Impact of Phenolic-Rich Olive Leaf Extract on Blood Pressure, Plasma Lipids and Inflammatory Markers: A Randomised Controlled Trial. Eur. J. Nutr. 2017, 56, 1421–1432. [Google Scholar] [CrossRef] [PubMed]
- Kurian, T.K.; Banik, S.; Gopal, D.; Chakrabarti, S.; Mazumder, N. Elucidating Methods for Isolation and Quantification of Exosomes: A Review. Mol. Biotechnol. 2021, 63, 249–266. [Google Scholar] [CrossRef] [PubMed]
- Lo Sicco, C.; Reverberi, D.; Balbi, C.; Ulivi, V.; Principi, E.; Pascucci, L.; Becherini, P.; Bosco, M.C.; Varesio, L.; Franzin, C.; et al. Mesenchymal Stem Cell-Derived Extracellular Vesicles as Mediators of Anti-Inflammatory Effects: Endorsement of Macrophage Polarization: MSC-Derived EVs Promote Macrophage Polarization. STEM CELLS Transl. Med. 2017, 6, 1018–1028. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, C.S.; Clements, A.E.B.; Kink, J.A.; Choi, U.; Baer, G.S.; Halanski, M.A.; Hematti, P.; Vanderby, R. Extracellular Vesicle-Educated Macrophages Promote Early Achilles Tendon Healing. Stem Cells 2019, 37, 652–662. [Google Scholar] [CrossRef]
- Chamberlain, C.S.; Kink, J.A.; Wildenauer, L.A.; McCaughey, M.; Henry, K.; Spiker, A.M.; Halanski, M.A.; Hematti, P.; Vanderby, R. Exosome-educated Macrophages and Exosomes Differentially Improve Ligament Healing. Stem Cells 2021, 39, 55–61. [Google Scholar] [CrossRef]
- Shen, H.; Yoneda, S.; Abu-Amer, Y.; Guilak, F.; Gelberman, R.H. Stem Cell-derived Extracellular Vesicles Attenuate the Early Inflammatory Response after Tendon Injury and Repair. J. Orthop. Res. 2020, 38, 117–127. [Google Scholar] [CrossRef]
- Best, K.T.; Nichols, A.E.C.; Knapp, E.; Hammert, W.C.; Ketonis, C.; Jonason, J.H.; Awad, H.A.; Loiselle, A.E. NF-ΚB Activation Persists into the Remodeling Phase of Tendon Healing and Promotes Myofibroblast Survival. Sci. Signal. 2020, 13, eabb7209. [Google Scholar] [CrossRef]
- Best, K.T.; Lee, F.K.; Knapp, E.; Awad, H.A.; Loiselle, A.E. Deletion of NFKB1 Enhances Canonical NF-ΚB Signaling and Increases Macrophage and Myofibroblast Content during Tendon Healing. Sci. Rep. 2019, 9, 10926. [Google Scholar] [CrossRef]
- Peng, Y.; Chen, B.; Zhao, J.; Peng, Z.; Xu, W.; Yu, G. Effect of Intravenous Transplantation of HUCB-MSCs on M1/M2 Subtype Conversion in Monocyte/Macrophages of AMI Mice. Biomed. Pharmacother. 2019, 111, 624–630. [Google Scholar] [CrossRef]
- Yin, Y.; Hao, H.; Cheng, Y.; Gao, J.; Liu, J.; Xie, Z.; Zhang, Q.; Zang, L.; Han, W.; Mu, Y. The Homing of Human Umbilical Cord-Derived Mesenchymal Stem Cells and the Subsequent Modulation of Macrophage Polarization in Type 2 Diabetic Mice. Int. Immunopharmacol. 2018, 60, 235–245. [Google Scholar] [CrossRef]
- Müller, L.; Tunger, A.; Wobus, M.; von Bonin, M.; Towers, R.; Bornhäuser, M.; Dazzi, F.; Wehner, R.; Schmitz, M. Immunomodulatory Properties of Mesenchymal Stromal Cells: An Update. Front. Cell Dev. Biol. 2021, 9, 637725. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Virumbrales, M.; Menta, R.; Pérez, L.M.; Lucchesi, O.; Mancheño-Corvo, P.; Avivar-Valderas, Á.; Palacios, I.; Herrero-Mendez, A.; Dalemans, W.; de la Rosa, O.; et al. Human Adipose Mesenchymal Stem Cells Modulate Myeloid Cells toward an Anti-Inflammatory and Reparative Phenotype: Role of IL-6 and PGE2. Stem Cell Res. Ther. 2020, 11, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Gao, H.; Chen, L.; Fang, N.; Chen, H.; Song, G.; Yu, L.; Zhang, Q.; Zhang, T. Effect of Peripheral Blood-Derived Mesenchymal Stem Cells on Macrophage Polarization and Th17/Treg Balance in Vitro. Regen. Ther. 2020, 14, 275–283. [Google Scholar] [CrossRef]
- Chan, K.-M.; Fu, S.-C.; Wong, Y.-P.; Hui, W.-C.; Cheuk, Y.-C.; Wong, M.W.-N. Expression of Transforming Growth Factor β Isoforms and Their Roles in Tendon Healing. Wound Repair Regen. 2008, 16, 399–407. [Google Scholar] [CrossRef]
- Chegini, N. TGF-Beta System: The Principal Profibrotic Mediator of Peritoneal Adhesion Formation. Semin. Reprod. Med. 2008, 26, 298–312. [Google Scholar] [CrossRef] [PubMed]
- Aktas, E.; Chamberlain, C.S.; Saether, E.E.; Duenwald-Kuehl, S.E.; Kondratko-Mittnacht, J.; Stitgen, M.; Lee, J.S.; Clements, A.E.; Murphy, W.L.; Vanderby, R. Immune Modulation with Primed Mesenchymal Stem Cells Delivered via Biodegradable Scaffold to Repair an Achilles Tendon Segmental Defect. J. Orthop. Res. 2017, 35, 269–280. [Google Scholar] [CrossRef]
- Lyras, D.N.; Kazakos, K.; Verettas, D.; Polychronidis, A.; Tryfonidis, M.; Botaitis, S.; Agrogiannis, G.; Simopoulos, C.; Kokka, A.; Patsouris, E. The Influence of Platelet-Rich Plasma on Angiogenesis During the Early Phase of Tendon Healing. Foot Ankle Int. 2009, 30, 1101–1106. [Google Scholar] [CrossRef]
- Lou, J.; Tu, Y.; Burns, M.; Silva, M.J.; Manske, P. BMP-12 Gene Transfer Augmentation of Lacerated Tendon Repair. J. Orthop. Res. 2001, 19, 1199–1202. [Google Scholar] [CrossRef]
- McCarthy, D.P.; Hunter, Z.N.; Chackerian, B.; Shea, L.D.; Miller, S.D. Targeted Immunomodulation Using Antigen-Conjugated Nanoparticles: Targeted Immunomodulation. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2014, 6, 298–315. [Google Scholar] [CrossRef]
- Fathi-Achachelouei, M.; Knopf-Marques, H.; Ribeiro da Silva, C.E.; Barthès, J.; Bat, E.; Tezcaner, A.; Vrana, N.E. Use of Nanoparticles in Tissue Engineering and Regenerative Medicine. Front. Bioeng. Biotechnol. 2019, 7, 113. [Google Scholar] [CrossRef] [PubMed]
- Arango-Ospina, M.; Nawaz, Q.; Boccaccini, A.R. Silicate-Based Nanoceramics in Regenerative Medicine. In Nanostructured Biomaterials for Regenerative Medicine; Elsevier: Amsterdam, The Netherlands, 2020; pp. 255–273. ISBN 978-0-08-102594-9. [Google Scholar]
- Vinhas, A.; Gonçalves, A.I.; Rodrigues, M.T.; Gomes, M.E. Human Tendon-Derived Cell Sheets Created by Magnetic Force-Based Tissue Engineering Hold Tenogenic and Immunomodulatory Potential. Acta Biomater. 2021, 131, 236–247. [Google Scholar] [CrossRef] [PubMed]
- Vinhas, A.; Rodrigues, M.T.; Gonçalves, A.I.; Reis, R.L.; Gomes, M.E. Magnetic Responsive Materials Modulate the Inflammatory Profile of IL-1β Conditioned Tendon Cells. Acta Biomater. 2020, 117, 235–245. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-ΚB Signaling in Inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Yoon, J.S.; Lee, J.Y.; Kim, H.J.; Park, K.; Kim, S.E. Long-Term Local PDGF Delivery Using Porous Microspheres Modified with Heparin for Tendon Healing of Rotator Cuff Tendinitis in a Rabbit Model. Carbohydr. Polym. 2019, 209, 372–381. [Google Scholar] [CrossRef]
- Jeong, C.; Kim, S.; Shim, K.-S.; Kim, H.-J.; Song, M.; Park, K.; Song, H.-R. Exploring the In Vivo Anti-Inflammatory Actions of Simvastatin-Loaded Porous Microspheres on Inflamed Tenocytes in a Collagenase-Induced Animal Model of Achilles Tendinitis. Int. J. Mol. Sci. 2018, 19, 820. [Google Scholar] [CrossRef] [PubMed]
- Eliasson, P.; Svensson, R.B.; Giannopoulos, A.; Eismark, C.; Kjær, M.; Schjerling, P.; Heinemeier, K.M. Simvastatin and Atorvastatin Reduce the Mechanical Properties of Tendon Constructs in Vitro and Introduce Catabolic Changes in the Gene Expression Pattern. PLOS ONE 2017, 12, e0172797. [Google Scholar] [CrossRef]
- Choi, H.J.; Choi, S.; Kim, J.G.; Song, M.H.; Shim, K.-S.; Lim, Y.-M.; Kim, H.-J.; Park, K.; Kim, S.E. Enhanced Tendon Restoration Effects of Anti-Inflammatory, Lactoferrin-Immobilized, Heparin-Polymeric Nanoparticles in an Achilles Tendinitis Rat Model. Carbohydr. Polym. 2020, 241, 116284. [Google Scholar] [CrossRef]
- Dohnert, M.B.; Ferreira, G.K.; Silveira, P.C.L.; Zanoni, E.T.; Dohnert, L.H.; de Souza, C.T.; Paula, M.M.S. Inflammatory Cytokines Content in Achilles Tendinopathy after Phonophoresis Treatment Combined with Gold Nanoparticles and Diclophenac Diethylammonium in Rats. Inflammation 2015, 38, 1044–1049. [Google Scholar] [CrossRef]
- Keller, T.C.; Hogan, M.V.; Kesturu, G.; James, R.; Balian, G.; Chhabra, A.B. Growth/Differentiation Factor-5 Modulates the Synthesis and Expression of Extracellular Matrix and Cell-Adhesion-Related Molecules of Rat Achilles Tendon Fibroblasts. Connect. Tissue Res. 2011, 52, 353–364. [Google Scholar] [CrossRef]
- Martino, F.; Perestrelo, A.R.; Vinarský, V.; Pagliari, S.; Forte, G. Cellular Mechanotransduction: From Tension to Function. Front. Physiol. 2018, 9, 824. [Google Scholar] [CrossRef]
- Arnoczky, S.P.; Tian, T.; Lavagnino, M.; Gardner, K. Ex Vivo Static Tensile Loading Inhibits MMP-1 Expression in Rat Tail Tendon Cells through a Cytoskeletally Based Mechanotransduction Mechanism. J. Orthop. Res. 2004, 22, 328–333. [Google Scholar] [CrossRef]
- Killian, M.L.; Cavinatto, L.; Galatz, L.M.; Thomopoulos, S. The Role of Mechanobiology in Tendon Healing. J. Shoulder Elbow Surg. 2012, 21, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.-C. Mechanobiology of Tendon. J. Biomech. 2006, 39, 1563–1582. [Google Scholar] [CrossRef]
- Kamkin, A. Mechanosensitivity in Cells and Tissues; Academic Publishing House: Moscow, Russia, 2005; ISBN 978-5-7695-2590-2. [Google Scholar]
- Arnoczky, S.P.; Tian, T.; Lavagnino, M.; Gardner, K.; Schuler, P.; Morse, P. Activation of Stress-Activated Protein Kinases (SAPK) in Tendon Cells Following Cyclic Strain: The Effects of Strain Frequency, Strain Magnitude, and Cytosolic Calcium. J. Orthop. Res. 2002, 20, 947–952. [Google Scholar] [CrossRef]
- Giori, N.J.; Beaupré, G.S.; Carter, D.R. Cellular Shape and Pressure May Mediate Mechanical Control of Tissue Composition in Tendons. J. Orthop. Res. 1993, 11, 581–591. [Google Scholar] [CrossRef] [PubMed]
- Hannafin, J.A.; Arnoczky, S.P.; Hoonjan, A.; Torzilli, P.A. Effect of Stress Deprivation and Cyclic Tensile Loading on the Material and Morphologic Properties of Canine Flexor Digitorum Profundus Tendon: An in Vitro Study. J. Orthop. Res. 1995, 13, 907–914. [Google Scholar] [CrossRef]
- Hannafin, J.A.; Attia, E.A.; Henshaw, R.; Warren, R.F.; Bhargava, M.M. Effect of Cyclic Strain and Plating Matrix on Cell Proliferation and Integrin Expression by Ligament Fibroblasts. J. Orthop. Res. 2006, 24, 149–158. [Google Scholar] [CrossRef]
- Magnusson, S.P.; Langberg, H.; Kjaer, M. The Pathogenesis of Tendinopathy: Balancing the Response to Loading. Nat. Rev. Rheumatol. 2010, 6, 262–268. [Google Scholar] [CrossRef]
- Robbins, J.R.; Evanko, S.P.; Vogel, K.G. Mechanical Loading and TGF-Beta Regulate Proteoglycan Synthesis in Tendon. Arch. Biochem. Biophys. 1997, 342, 203–211. [Google Scholar] [CrossRef]
- Vogel, K.G. The Effect of Compressive Loading on Proteoglycan Turnover in Cultured Fetal Tendon. Connect. Tissue Res. 1996, 34, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Vogel, K.G.; Ordög, A.; Pogány, G.; Oláh, J. Proteoglycans in the Compressed Region of Human Tibialis Posterior Tendon and in Ligaments. J. Orthop. Res. Off. Publ. Orthop. Res. Soc. 1993, 11, 68–77. [Google Scholar] [CrossRef]
- Waggett, A.D.; Benjamin, M.; Ralphs, J.R. Connexin 32 and 43 Gap Junctions Differentially Modulate Tenocyte Response to Cyclic Mechanical Load. Eur. J. Cell Biol. 2006, 85, 1145–1154. [Google Scholar] [CrossRef] [PubMed]
- Wall, M.E.; Banes, A.J. Early Responses to Mechanical Load in Tendon: Role for Calcium Signaling, Gap Junctions and Intercellular Communication. J. Musculoskelet. Neuronal Interact. 2005, 5, 70–84. [Google Scholar]
- Yamamoto, E.; Tokura, S.; Hayashi, K. Effects of Cyclic Stress on the Mechanical Properties of Cultured Collagen Fascicles from the Rabbit Patellar Tendon. J. Biomech. Eng. 2003, 125, 893–901. [Google Scholar] [CrossRef]
- Wang, T.; Gardiner, B.S.; Lin, Z.; Rubenson, J.; Kirk, T.B.; Wang, A.; Xu, J.; Smith, D.W.; Lloyd, D.G.; Zheng, M.H. Bioreactor Design for Tendon/Ligament Engineering. Tissue Eng. Part B Rev. 2013, 19, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Galatz, L.M.; Charlton, N.; Das, R.; Kim, H.M.; Havlioglu, N.; Thomopoulos, S. Complete Removal of Load Is Detrimental to Rotator Cuff Healing. J. Shoulder Elbow Surg. 2009, 18, 669–675. [Google Scholar] [CrossRef]
- Hettrich, C.M.; Rodeo, S.A.; Hannafin, J.A.; Ehteshami, J.; Shubin Stein, B.E. The Effect of Muscle Paralysis Using Botox on the Healing of Tendon to Bone in a Rat Model. J. Shoulder Elbow Surg. 2011, 20, 688–697. [Google Scholar] [CrossRef] [PubMed]
- Thomopoulos, S.; Williams, G.R.; Soslowsky, L.J. Tendon to Bone Healing: Differences in Biomechanical, Structural, and Compositional Properties Due to a Range of Activity Levels. J. Biomech. Eng. 2003, 125, 106–113. [Google Scholar] [CrossRef]
- Ballotta, V.; Smits, A.I.P.M.; Driessen-Mol, A.; Bouten, C.V.C.; Baaijens, F.P.T. Synergistic Protein Secretion by Mesenchymal Stromal Cells Seeded in 3D Scaffolds and Circulating Leukocytes in Physiological Flow. Biomaterials 2014, 35, 9100–9113. [Google Scholar] [CrossRef] [PubMed]
- Hansen, C.A.; Schroering, A.G.; Carey, D.J.; Robishaw, J.D. Localization of a Heterotrimeric G Protein Gamma Subunit to Focal Adhesions and Associated Stress Fibers. J. Cell Biol. 1994, 126, 811–819. [Google Scholar] [CrossRef]
- Hoon, J.L.; Tan, M.H.; Koh, C.-G. The Regulation of Cellular Responses to Mechanical Cues by Rho GTPases. Cells 2016, 5, 17. [Google Scholar] [CrossRef] [PubMed]
- Lehoux, S.; Tedgui, A. Signal Transduction of Mechanical Stresses in the Vascular Wall. Hypertension 1998, 32, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Burridge, K.; Chrzanowska-Wodnicka, M. Focal Adhesions, Contractility, and Signaling. Annu. Rev. Cell Dev. Biol. 1996, 12, 463–519. [Google Scholar] [CrossRef]
- Ridley, A.J.; Hall, A. The Small GTP-Binding Protein Rho Regulates the Assembly of Focal Adhesions and Actin Stress Fibers in Response to Growth Factors. Cell 1992, 70, 389–399. [Google Scholar] [CrossRef]
- Riveline, D.; Zamir, E.; Balaban, N.Q.; Schwarz, U.S.; Ishizaki, T.; Narumiya, S.; Kam, Z.; Geiger, B.; Bershadsky, A.D. Focal Contacts as Mechanosensors: Externally Applied Local Mechanical Force Induces Growth of Focal Contacts by an mDia1-Dependent and ROCK-Independent Mechanism. J. Cell Biol. 2001, 153, 1175–1186. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.R.; Bole, M.; Chen, C.; Hardin, C.C.; Kho, A.T.; Mih, J.; Deng, L.; Butler, J.; Tschumperlin, D.; Fredberg, J.J.; et al. Cell Elasticity Determines Macrophage Function. PLoS ONE 2012, 7, e41024. [Google Scholar] [CrossRef]
- Beningo, K.A.; Wang, Y. Fc-Receptor-Mediated Phagocytosis Is Regulated by Mechanical Properties of the Target. J. Cell Sci. 2002, 115, 849–856. [Google Scholar] [CrossRef]
- Vining, K.H.; Mooney, D.J. Mechanical Forces Direct Stem Cell Behaviour in Development and Regeneration. Nat. Rev. Mol. Cell Biol. 2017, 18, 728–742. [Google Scholar] [CrossRef]
- Dupont, S.; Morsut, L.; Aragona, M.; Enzo, E.; Giulitti, S.; Cordenonsi, M.; Zanconato, F.; Le Digabel, J.; Forcato, M.; Bicciato, S.; et al. Role of YAP/TAZ in Mechanotransduction. Nature 2011, 474, 179–183. [Google Scholar] [CrossRef]
- Oka, T.; Sudol, M. Nuclear Localization and Pro-Apoptotic Signaling of YAP2 Require Intact PDZ-Binding Motif. Genes Cells 2009, 14, 607–615. [Google Scholar] [CrossRef]
- Piccolo, S.; Dupont, S.; Cordenonsi, M. The Biology of YAP/TAZ: Hippo Signaling and Beyond. Physiol. Rev. 2014, 94, 1287–1312. [Google Scholar] [CrossRef] [PubMed]
- Aragona, M.; Panciera, T.; Manfrin, A.; Giulitti, S.; Michielin, F.; Elvassore, N.; Dupont, S.; Piccolo, S. A Mechanical Checkpoint Controls Multicellular Growth through YAP/TAZ Regulation by Actin-Processing Factors. Cell 2013, 154, 1047–1059. [Google Scholar] [CrossRef] [PubMed]
- Driscoll, T.P.; Cosgrove, B.D.; Heo, S.-J.; Shurden, Z.E.; Mauck, R.L. Cytoskeletal to Nuclear Strain Transfer Regulates YAP Signaling in Mesenchymal Stem Cells. Biophys. J. 2015, 108, 2783–2793. [Google Scholar] [CrossRef] [PubMed]
- Panciera, T.; Azzolin, L.; Cordenonsi, M.; Piccolo, S. Mechanobiology of YAP and TAZ in Physiology and Disease. Nat. Rev. Mol. Cell Biol. 2017, 18, 758–770. [Google Scholar] [CrossRef] [PubMed]
- Totaro, A.; Castellan, M.; Battilana, G.; Zanconato, F.; Azzolin, L.; Giulitti, S.; Cordenonsi, M.; Piccolo, S. YAP/TAZ Link Cell Mechanics to Notch Signalling to Control Epidermal Stem Cell Fate. Nat. Commun. 2017, 8, 15206. [Google Scholar] [CrossRef]
- Wada, K.-I.; Itoga, K.; Okano, T.; Yonemura, S.; Sasaki, H. Hippo Pathway Regulation by Cell Morphology and Stress Fibers. Development 2011, 138, 3907–3914. [Google Scholar] [CrossRef] [PubMed]
- Sorrentino, G.; Ruggeri, N.; Specchia, V.; Cordenonsi, M.; Mano, M.; Dupont, S.; Manfrin, A.; Ingallina, E.; Sommaggio, R.; Piazza, S.; et al. Metabolic Control of YAP and TAZ by the Mevalonate Pathway. Nat. Cell Biol. 2014, 16, 357–366. [Google Scholar] [CrossRef]
- Alarcón, C.; Zaromytidou, A.-I.; Xi, Q.; Gao, S.; Yu, J.; Fujisawa, S.; Barlas, A.; Miller, A.N.; Manova-Todorova, K.; Macias, M.J.; et al. Nuclear CDKs Drive Smad Transcriptional Activation and Turnover in BMP and TGF-Beta Pathways. Cell 2009, 139, 757–769. [Google Scholar] [CrossRef]
- Varelas, X.; Sakuma, R.; Samavarchi-Tehrani, P.; Peerani, R.; Rao, B.M.; Dembowy, J.; Yaffe, M.B.; Zandstra, P.W.; Wrana, J.L. TAZ Controls Smad Nucleocytoplasmic Shuttling and Regulates Human Embryonic Stem-Cell Self-Renewal. Nat. Cell Biol. 2008, 10, 837–848. [Google Scholar] [CrossRef]
- Varelas, X.; Samavarchi-Tehrani, P.; Narimatsu, M.; Weiss, A.; Cockburn, K.; Larsen, B.G.; Rossant, J.; Wrana, J.L. The Crumbs Complex Couples Cell Density Sensing to Hippo-Dependent Control of the TGF-β-SMAD Pathway. Dev. Cell 2010, 19, 831–844. [Google Scholar] [CrossRef]
- Liu, C.; Tian, S.; Bai, J.; Yu, K.; Liu, L.; Liu, G.; Dong, R.; Tian, D. Regulation of ERK1/2 and SMAD2/3 Pathways by Using Multi-Layered Electrospun PCL-Amnion Nanofibrous Membranes for the Prevention of Post-Surgical Tendon Adhesion. Int. J. Nanomed. 2020, 15, 927–942. [Google Scholar] [CrossRef] [PubMed]
| Scaffold Properties | Material | Parameters | Immune Response | Reference |
|---|---|---|---|---|
| Diameter size | PDO | Different fiber diameter size (0.35, 2.20, and 2.80 µm) | Increasing fiber diameter → ↑ M2 macrophages expression | [90] |
| PCL | Different fiber diameters (0.69 and 5.59 μm) | Increased fiber diameter size (5.59 µm) → ↑ M2 macrophages expression | [91] | |
| Alignment | PCL | Random and aligned fiber orientation; scaffolds unmodified or extended to macro-scale thicknesses of 3 or 10 mm | Expanded scaffolds ↑ regenerative answer and thinner collagen fibrous capsule compared to unexpanded nanofiber scaffolds Aligned fibers (expanded to 3 mm) fewest ↓ number of giant cells | [92] |
| PLLA | Five different types of scaffolds: aligned microfibers, aligned nanofibers, random microfibers, random nanofibers, and on film | Nanofibrous PLLA scaffolds ↓ inflammatory response than films and microfibrous scaffolds PLLA film ↑ number of foreign body giant cells than the micro and nanofibrous scaffolds | [29] | |
| PCL | Random and aligned fiber orientation | Random fibers ↑ pro-inflammatory response compared to aligned fibers | [93] | |
| PCL | Random and aligned fiber orientation | Aligned fibers → least amount of monocyte adhesion with a thinner fibrous capsule and more fibroblasts infiltration compared to randomly oriented fibers | [94] | |
| Pore size | PDO | Different pore size (0.96, 10.57, and 14.73 µm) | 14.73 µm pore size → M2 macrophage polarization, ↑Arginase I and ↓iNOS | [90] |
| Mechanical stimulus | PCL | 7 and 12% cyclic uniaxial strains (0.8 Hz) | 7% mechanical strain → ↑ MCP-1, IL-6, IL-10, and MMP-9 (M2 markers) 12% strain → M1 proinflammatory phenotype | [95] |
| CE-UPy-PCL | Cyclic strains: 0%, 8% and 14% strain at 0.8 Hz | High strains addressed a pro-inflammatory condition | [96] | |
| PCL | Static culture (1% constant strain) and dynamic loading (7% cyclic strain at 1 Hz) | Dynamic loading → ↑ CCR7 (M1 marker) | [93] | |
| Surface modification | PLLA | Lubricating layer of chitosan collagen and alginate hydrogel | ↓ Protein adsorption | [97] |
| PLGA | CTS layer coating | ↓ inflammatory cells recruitment and FBGCs formation | [98] | |
| PLLA | Two layers of PLLA membranes combined into a single layer | ↓ Adhesion to the tissues | [99] |
| Material | Stem Cell Type | Propriety | Outcomes | Reference |
|---|---|---|---|---|
| PCL | Rat ADMSCs | Randomly oriented, aligned and mesh-like electrospun fibers | gene expression of PGE2, iNOS, and VEGF within ADMSCs engineered within aligned and mesh-like fibers | [83] |
| PLLA | Human ADMSCs | Randomly oriented and highly aligned electrospun fibers | gene expression of COX-2, TGF-β, TSG-6, and M-CSF in ADMSCs cultured within aligned fibers ↑ protein expression of COX-2 and TSG-6 and ↑ secreted levels of PGE2 in ADMSCs on aligned fibers | [84] |
| PLGA | Ovine AECs | Electrospun PLGA scaffolds with two different diameter size (1.27 and 2.50 µm) | ↑ gene expression of IL-4 and IL-10 and ↓ gene expression of IL-12 and IL-6 within small fiber diameter size (1.27 µm) | [18] |
| PCL | Human ADMSCs | Electrospun PCL-DT-NPs yarns cultivated under static and magnetic stimulation conditions | ↑ gene expression of MMP-1, MMP-2, MMP-3, TIMPs, IL-10, and IL-4 with ↓ gene expression of IL-6 and COX-2 under magnetic stimulation condition | [33] |
| Bioactive Molecule | Scaffold Material | Cell Type | Outcomes in the Studied Cell Type | Reference |
|---|---|---|---|---|
| NSAIDs | PELA | Macrophages | ↓ inflammatory response and ↓TA | [85] |
| IL-4 | CE-UPy-PCL | Macrophages | ↑ IL-10, TGF-β1 and MMP-9 ↓ IL-6 | [2] |
| IL-4 | PCL | Macrophages | ↑M2 macrophage markers (Arginase I, CD206…) | [153] |
| 25-hydroxyvitamin D3 | PCL | Macrophages | ↓ TNF-a, IL-6 and ↑ IL-4, IL-10 | [154] |
| Ibuprofen | PLA | Macrophages | ↓TNF-α expression and collagen III deposition | [155] |
| Mesenchymal stromal exosomes | PEF | BM Macrophages | ↑ CD206+ M2 macrophages and the concentration of IL-4, IL-10 and IL13 ↓ concentration of TNF-α and IFN-γ | [156] |
| IL-4 plasmid-loaded liposomes (aL/p) | MSaP | BM Macrophages | ↑ levels of IL-10 and TGF-β ↓ levels of IL-1 and TNF-α | [157] |
| Melatonin | PCL | Human BMSCs | Inhibition of macrophage (CD68-positive cell) accumulation at the tendon-to-bone interface. | [158] |
| MSCs-derived ECM | PCL/SF | Human BMSCs | In vitro: ↑ M2 macrophage polarization and ↓ IL -1β, IL-6, CXCL11, IL-10, IL-1R2 and TGF-β1 In vivo: ↓ FBR, thinner fibrotic capsule formation and ↑ M2 macrophage polarization | [159] |
| bFGF | PLLA | Human vaginal fibroblasts | ↑ concentration of TGF-β1 and concentration of TNF-α | [160] |
| IFN-γ | SF/PLGA | Human BMSCs | transcription levels of COX-2 and IDO transcription levels of TNF-α | [161] |
| OLE | PHA | Human HaCaTs | concentration of IL-1, IL-6, IL-8 and TNF-α | [162] |
| IFN-γ and TNF-α | No scaffold | Human BMSCs | gene expression of IDO, iNOS, IL-6, COX-2 and VCAM-1 | [163] |
| PRP | No scaffold | Human tenocytes | concentration of VEGF, RANTES and HGF gene expression of IL-6, IL-6R, and IL-8 | [164] |
| HGF | No scaffold | Tendon fibroblasts | concentration of MMP-2 and MMP-9, α-SMA, TIMP-1, VEGF and IL-10 gene expression of IL-6 | [165] |
| BMP-12 | No scaffold | Human ASCs | concentration of VEGF, MMP1, MMP8 and IL6 | [166] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Russo, V.; El Khatib, M.; Prencipe, G.; Cerveró-Varona, A.; Citeroni, M.R.; Mauro, A.; Berardinelli, P.; Faydaver, M.; Haidar-Montes, A.A.; Turriani, M.; et al. Scaffold-Mediated Immunoengineering as Innovative Strategy for Tendon Regeneration. Cells 2022, 11, 266. https://doi.org/10.3390/cells11020266
Russo V, El Khatib M, Prencipe G, Cerveró-Varona A, Citeroni MR, Mauro A, Berardinelli P, Faydaver M, Haidar-Montes AA, Turriani M, et al. Scaffold-Mediated Immunoengineering as Innovative Strategy for Tendon Regeneration. Cells. 2022; 11(2):266. https://doi.org/10.3390/cells11020266
Chicago/Turabian StyleRusso, Valentina, Mohammad El Khatib, Giuseppe Prencipe, Adrián Cerveró-Varona, Maria Rita Citeroni, Annunziata Mauro, Paolo Berardinelli, Melisa Faydaver, Arlette A. Haidar-Montes, Maura Turriani, and et al. 2022. "Scaffold-Mediated Immunoengineering as Innovative Strategy for Tendon Regeneration" Cells 11, no. 2: 266. https://doi.org/10.3390/cells11020266
APA StyleRusso, V., El Khatib, M., Prencipe, G., Cerveró-Varona, A., Citeroni, M. R., Mauro, A., Berardinelli, P., Faydaver, M., Haidar-Montes, A. A., Turriani, M., Di Giacinto, O., Raspa, M., Scavizzi, F., Bonaventura, F., Liverani, L., Boccaccini, A. R., & Barboni, B. (2022). Scaffold-Mediated Immunoengineering as Innovative Strategy for Tendon Regeneration. Cells, 11(2), 266. https://doi.org/10.3390/cells11020266
















