Differential Expression of miRNAs and Their Predicted Target Pathways in Cochlear Nucleus Following Chronic Noise Exposure in Rats
Abstract
1. Introduction
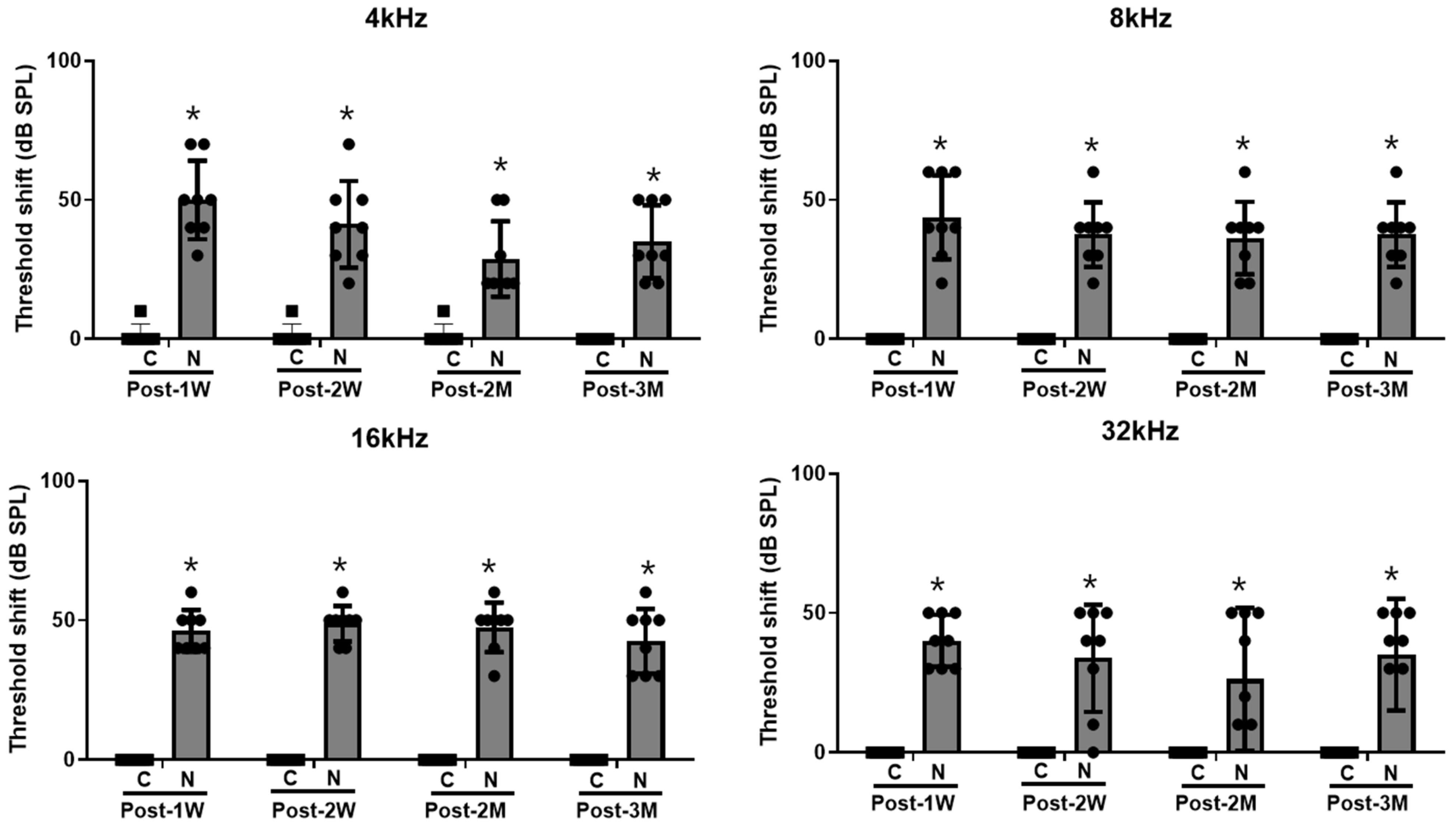
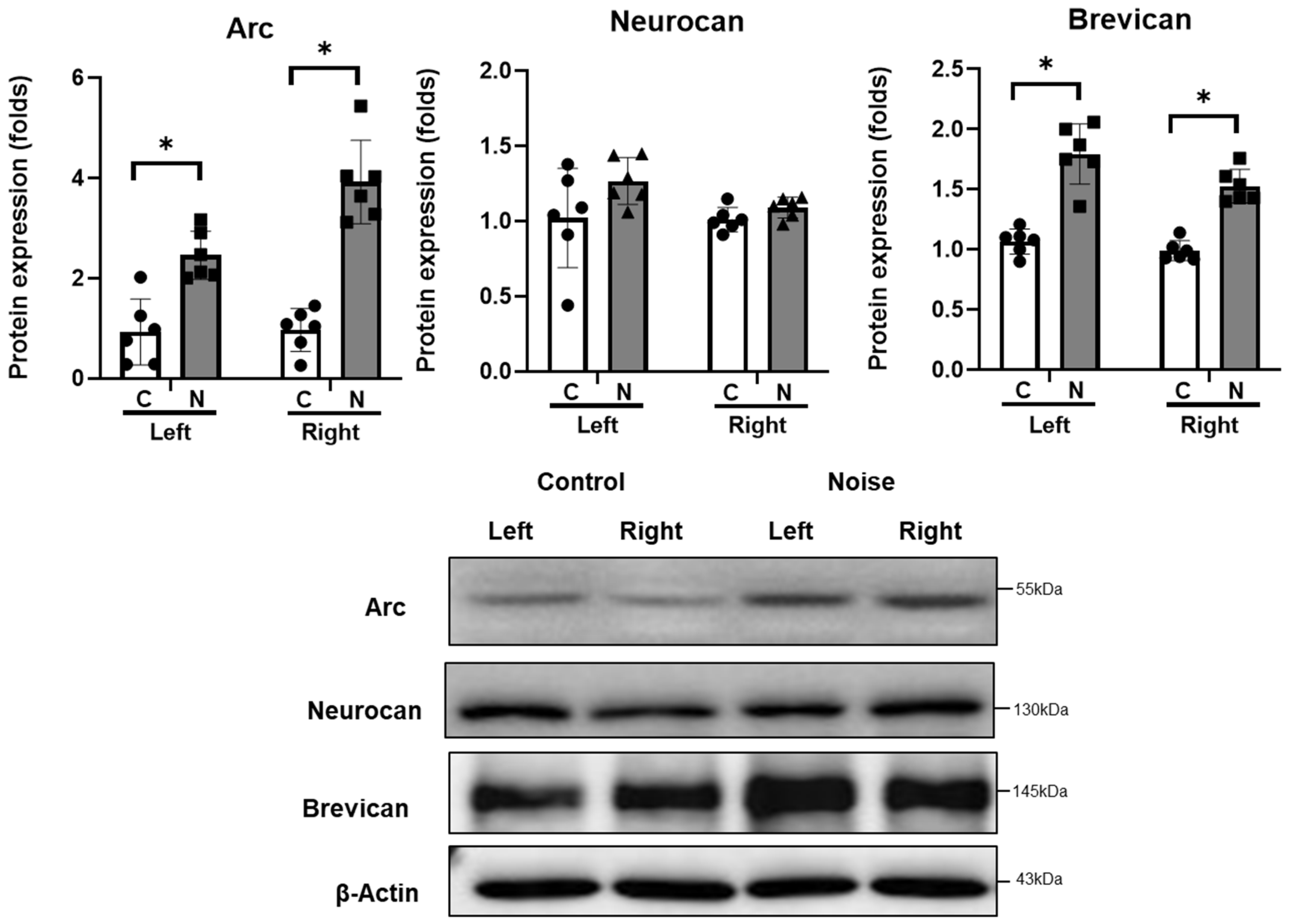
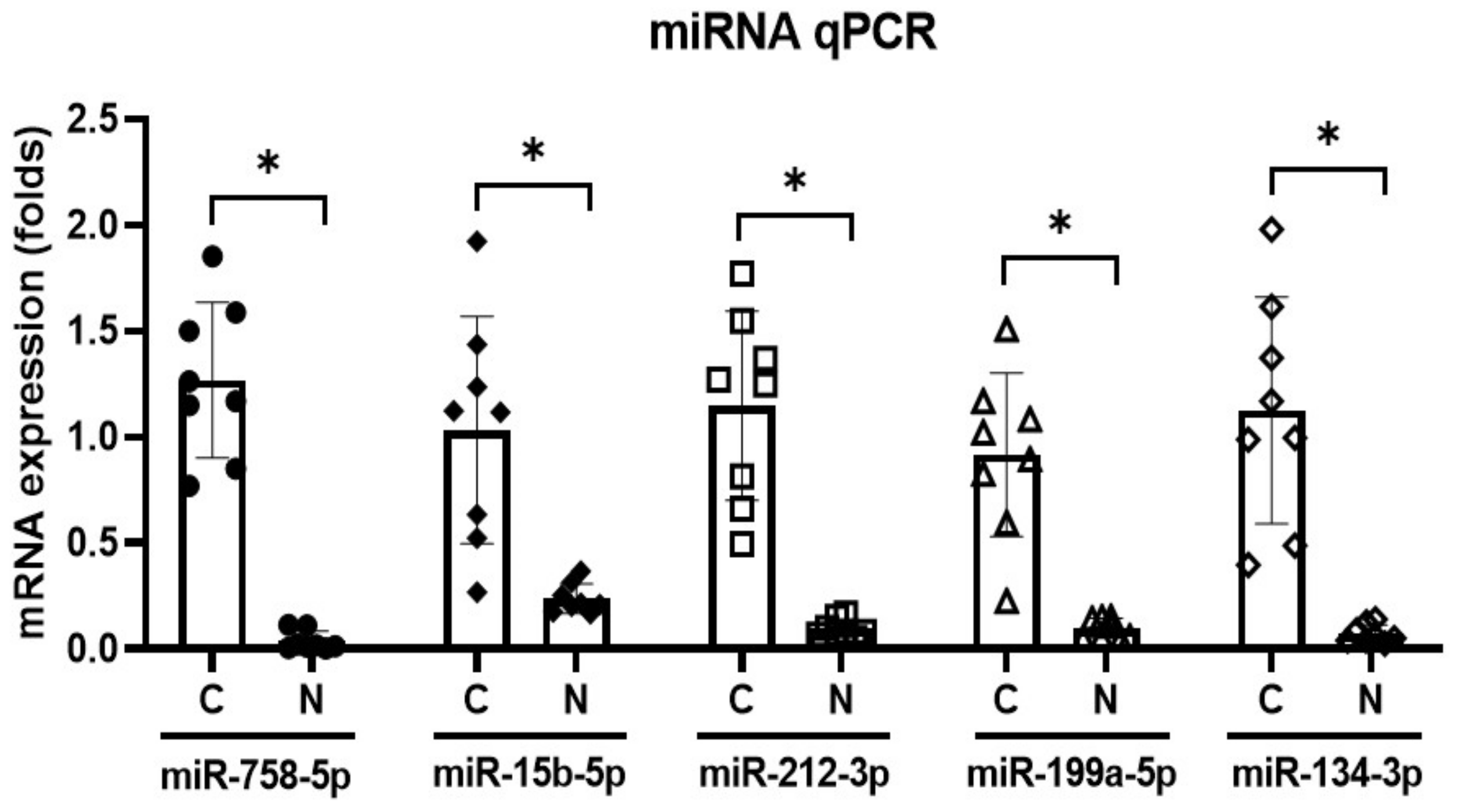
2. Results
3. Discussion
4. Materials and Methods
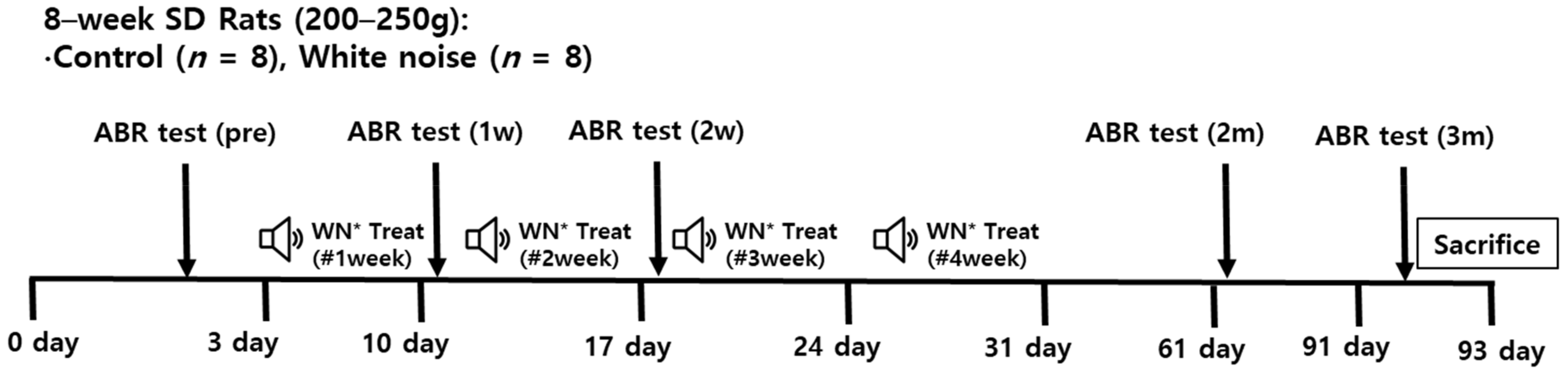
4.1. Noise and Comparison Groups
4.2. Hearing Level Measurement
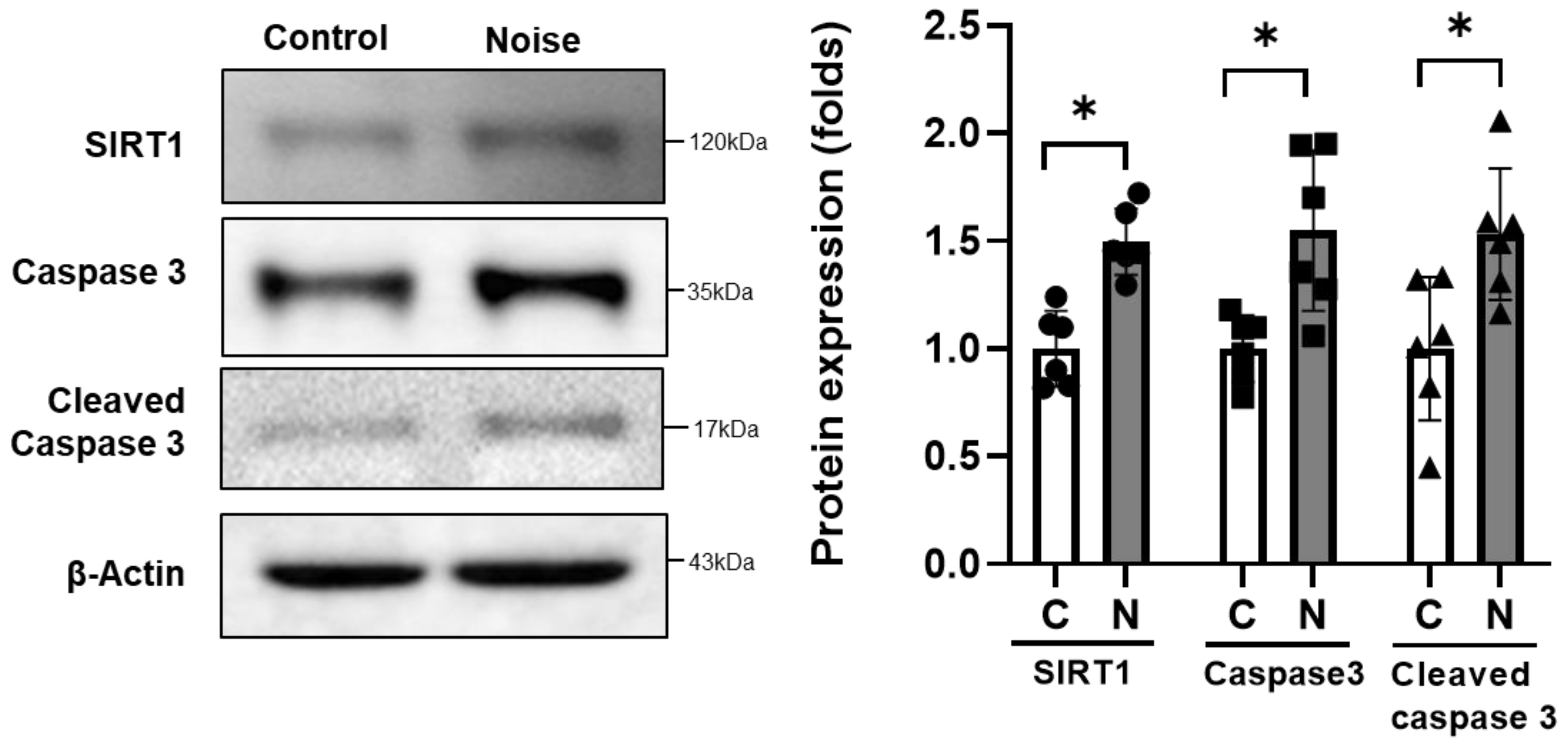
4.3. Protein Expression in CN
4.4. miRNA Expression in the CN
4.5. The Predicted Target Genes for miRNAs
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Manohar, S.; Ramchander, P.; Salvi, R.; Seigel, G. Synaptic Reorganization Response in the Cochlear Nucleus Following Intense Noise Exposure. Neuroscience 2018, 399, 184–198. [Google Scholar] [CrossRef] [PubMed]
- Manley, G.A. The mammalian Cretaceous cochlear revolution. Hear. Res. 2017, 352, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Weinrich, L.; Sonntag, M.; Arendt, T.; Morawski, M. Neuroanatomical characterization of perineuronal net components in the human cochlear nucleus and superior olivary complex. Hear. Res. 2018, 367, 32–47. [Google Scholar] [CrossRef]
- Favuzzi, E.; Marques-Smith, A.; Deogracias, R.; Winterflood, C.M.; Sánchez-Aguilera, A.; Mantoan, L.; Maeso, P.; Fernandes, C.; Ewers, H.; Rico, B. Activity-Dependent Gating of Parvalbumin Interneuron Function by the Perineuronal Net Protein Brevican. Neuron 2017, 95, 639–655.e10. [Google Scholar] [CrossRef]
- Nguyen, A.; Khaleel, H.M.; Razak, K.A. Effects of noise-induced hearing loss on parvalbumin and perineuronal net expression in the mouse primary auditory cortex. Hear. Res. 2017, 350, 82–90. [Google Scholar] [CrossRef]
- Park, S.S.; Lee, D.H.; Lee, S.M.; Lee, C.H.; Kim, S.Y. Noise exposure alters MMP9 and brevican expression in the rat primary auditory cortex. BMC Neurosci. 2020, 21, 16. [Google Scholar] [CrossRef] [PubMed]
- Krohs, C.; Bordeynik-Cohen, M.; Messika-Gold, N.; Elkon, R.; Avraham, K.B.; Nothwang, H.G. Expression pattern of cochlear microRNAs in the mammalian auditory hindbrain. Cell Tissue Res. 2021, 383, 655–666. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Han, S.H.; Kim, B.G.; Suh, M.W.; Lee, J.H.; Oh, S.H.; Park, M.K. Changes in microRNA Expression in the Cochlear Nucleus and Inferior Colliculus after Acute Noise-Induced Hearing Loss. Int. J. Mol. Sci. 2020, 21, 8792. [Google Scholar] [CrossRef]
- Giraldez, A.J.; Cinalli, R.M.; Glasner, M.E.; Enright, A.; Thomson, J.M.; Baskerville, S.; Hammond, S.M.; Bartel, D.P.; Schier, A.F. MicroRNAs Regulate Brain Morphogenesis in Zebrafish. Science 2005, 308, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Schratt, G.M.; Tuebing, F.; Nigh, E.A.; Kane, C.G.; Sabatini, M.E.; Kiebler, M.; Greenberg, M.E. A brain-specific microRNA regulates dendritic spine development. Nature 2006, 439, 283–289. [Google Scholar] [CrossRef]
- Higuchi, F.; Uchida, S.; Yamagata, H.; Abe-Higuchi, N.; Hobara, T.; Hara, K.; Kobayashi, A.; Shintaku, T.; Itoh, Y.; Suzuki, T.; et al. Hippocampal MicroRNA-124 Enhances Chronic Stress Resilience in Mice. J. Neurosci. 2016, 36, 7253–7267. [Google Scholar] [CrossRef] [PubMed]
- Schlüter, T.; Berger, C.; Rosengauer, E.; Fieth, P.; Krohs, C.; Ushakov, K.; Steel, K.P.; Avraham, K.B.; Hartmann, A.K.; Felmy, F.; et al. miR-96 is required for normal development of the auditory hindbrain. Hum. Mol. Genet. 2018, 27, 860–874. [Google Scholar] [CrossRef] [PubMed]
- Friedman, L.M.; Dror, A.A.; Mor, E.; Tenne, T.; Toren, G.; Satoh, T.; Biesemeier, D.J.; Shomron, N.; Fekete, D.M.; Hornstein, E.; et al. MicroRNAs are essential for development and function of inner ear hair cells in vertebrates. Proc. Natl. Acad. Sci. USA 2009, 106, 7915–7920. [Google Scholar] [CrossRef] [PubMed]
- Napoli, D.; Lupori, L.; Mazziotti, R.; Sagona, G.; Bagnoli, S.; Samad, M.; Sacramento, E.K.; Kirkpartick, J.; Putignano, E.; Chen, S.; et al. MiR-29 coordinates age-dependent plasticity brakes in the adult visual cortex. EMBO Rep. 2020, 21, e50431. [Google Scholar] [CrossRef] [PubMed]
- Hill, K.; Yuan, H.; Wang, X.; Sha, S.H. Noise-Induced Loss of Hair Cells and Cochlear Synaptopathy Are Mediated by the Activation of AMPK. J. Neurosci. 2016, 36, 7497–7510. [Google Scholar] [CrossRef] [PubMed]
- Nagashima, R.; Yamaguchi, T.; Kuramoto, N.; Ogita, K. Acoustic overstimulation activates 5′-AMP-activated protein kinase through a temporary decrease in ATP level in the cochlear spiral ligament prior to permanent hearing loss in mice. Neurochem. Int. 2011, 59, 812–820. [Google Scholar] [CrossRef]
- Noack, F.; Calegari, F. Micro- RNA s meet epigenetics to make for better brains. EMBO Rep. 2014, 15, 1224–1225. [Google Scholar] [CrossRef][Green Version]
- Wu, H.Z.Y.; Thalamuthu, A.; Cheng, L.; Fowler, C.; Masters, C.L.; Sachdev, P.; Mather, K.A. Differential blood miRNA expression in brain amyloid imaging-defined Alzheimer’s disease and controls. Alzheimers Res. Ther. 2020, 12, 59. [Google Scholar] [CrossRef]
- Aten, S.; Page, C.E.; Kalidindi, A.; Wheaton, K.; Niraula, A.; Godbout, J.P.; Hoyt, K.; Obrietan, K. miR-132/212 is induced by stress and its dysregulation triggers anxiety-related behavior. Neuropharmacology 2018, 144, 256–270. [Google Scholar] [CrossRef]
- Cha, D.J.; Mengel, D.; Mustapic, M.; Liu, W.; Selkoe, D.J.; Kapogiannis, D.; Galasko, D.; Rissman, R.A.; Bennett, D.A.; Walsh, D.M. miR-212 and miR-132 Are Downregulated in Neurally Derived Plasma Exosomes of Alzheimer’s Patients. Front. Neurosci. 2019, 13, 1208. [Google Scholar] [CrossRef]
- Song, D.; Li, G.; Hong, Y.; Zhang, P.; Zhu, J.; Yang, L.; Huang, J. miR-199a decreases Neuritin expression involved in the development of Alzheimer’s disease in APP/PS1 mice. Int. J. Mol. Med. 2020, 46, 384–396. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.; Zhu, X.; Song, Q.; Wang, P.; Liu, Z.; Yu, S.Y. MiR-134 modulates chronic stress-induced structural plasticity and depression-like behaviors via downregulation of Limk1/cofilin signaling in rats. Neuropharmacology 2018, 131, 364–376. [Google Scholar] [CrossRef] [PubMed]
- Ruderman, N.B.; Xu, X.J.; Nelson, L.; Cacicedo, J.M.; Saha, A.K.; Lan, F.; Ido, Y. AMPK and SIRT1: A long-standing partnership? Am. J. Physiol. Endocrinol. Metab. 2010, 298, E751–E760. [Google Scholar] [CrossRef]
- Lin, J.Y.; Kuo, W.W.; Baskaran, R.; Kuo, C.H.; Chen, Y.A.; Chen, W.S.; Ho, T.J.; Day, C.H.; Mahalakshmi, B.; Huang, C.Y. Swimming exercise stimulates IGF1/PI3K/Akt and AMPK/SIRT1/PGC1α survival signaling to suppress apoptosis and inflammation in aging hippocampus. Aging 2020, 12, 6852–6864. [Google Scholar] [CrossRef]
- Ramadori, G.; Lee, C.E.; Bookout, A.L.; Lee, S.; Williams, K.; Anderson, J.; Elmquist, J.K.; Coppari, R. Brain SIRT1: Anatomical Distribution and Regulation by Energy Availability. J. Neurosci. 2008, 28, 9989–9996. [Google Scholar] [CrossRef]
- Vaziri, H.; Dessain, S.K.; Eaton, E.N.; Imai, S.-I.; Frye, R.A.; Pandita, T.K.; Guarente, L.; Weinberg, R.A. hSIR2SIRT1 Functions as an NAD-Dependent p53 Deacetylase. Cell 2001, 107, 149–159. [Google Scholar] [CrossRef]
- Jeong, H.; Cohen, D.E.; Cui, L.; Supinski, A.; Savas, J.N.; Mazzulli, J.R.; Yates, J.R.; Bordone, L.; Guarente, L.; Krainc, D. Sirt1 mediates neuroprotection from mutant huntingtin by activation of the TORC1 and CREB transcriptional pathway. Nat. Med. 2011, 18, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Ferland, C.L.; Hawley, W.R.; Puckett, R.E.; Wineberg, K.; Lubin, F.D.; Dohanich, G.P.; Schrader, L.A. Sirtuin Activity in Dentate Gyrus Contributes to Chronic Stress-Induced Behavior and Extracellular Signal-Regulated Protein Kinases 1 and 2 Cascade Changes in the Hippocampus. Biol. Psychiatry 2013, 74, 927–935. [Google Scholar] [CrossRef]
- Prozorovski, T.; Schulze-Topphoff, U.; Glumm, R.; Baumgart, J.; Schröter, F.; Ninnemann, O.; Siegert, E.; Bendix, I.; Brüstle, O.; Nitsch, R.; et al. Sirt1 contributes critically to the redox-dependent fate of neural progenitors. Nat. Cell Biol. 2008, 10, 385–394. [Google Scholar] [CrossRef]
- Li, X.H.; Chen, C.; Tu, Y.; Sun, H.T.; Zhao, M.L.; Cheng, S.X.; Qu, Y.; Zhang, S. Sirt1 Promotes Axonogenesis by Deacetylation of Akt and Inactivation of GSK3. Mol. Neurobiol. 2013, 48, 490–499. [Google Scholar] [CrossRef]
- Teertam, S.K.; Babu, P.P. Differential role of SIRT1/MAPK pathway during cerebral ischemia in rats and humans. Sci. Rep. 2021, 11, 6339. [Google Scholar] [CrossRef] [PubMed]
- Selivanova, O.; Brieger, J.; Heinrich, U.-R.; Mann, W. Akt and c-Jun N-terminal kinase are regulated in response to moderate noise exposure in the cochlea of guinea pigs. ORL J. Otorhinolaryngol. Relat. Spec. 2007, 69, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Plas, D.R.; Thompson, C.B. Akt-dependent transformation: There is more to growth than just surviving. Oncogene 2005, 24, 7435–7442. [Google Scholar] [CrossRef]
- Duan, J.; Cui, J.; Zheng, H.; Xi, M.; Guo, C.; Weng, Y.; Yin, Y.; Wei, G.; Cao, J.; Wang, Y.; et al. Aralia taibaiensis Protects against I/R-Induced Brain Cell Injury through the Akt/SIRT1/FOXO3a Pathway. Oxidative Med. Cell. Longev. 2019, 2019, 7609765. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Yuan, H.; Talaska, A.E.; Hill, K.; Sha, S.-H. Increased Sensitivity to Noise-Induced Hearing Loss by Blockade of Endogenous PI3K/Akt Signaling. J. Assoc. Res. Otolaryngol. 2015, 16, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Sha, S.-H.; Chen, F.-Q.; Schacht, J. PTEN attenuates PIP3/Akt signaling in the cochlea of the aging CBA/J mouse. Hear. Res. 2010, 264, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Chung, W.-H.; Pak, K.; Lin, B.; Webster, N.; Ryan, A.F. A PI3K Pathway Mediates Hair Cell Survival and Opposes Gentamicin Toxicity in Neonatal Rat Organ of Corti. J. Assoc. Res. Otolaryngol. 2006, 7, 373–382. [Google Scholar] [CrossRef]
- Lai, R.; Li, W.; Hu, P.; Xie, D.; Wen, J. Role of Hsp90/Akt pathway in the pathogenesis of gentamicin-induced hearing loss. Int. J. Clin. Exp. Pathol. 2018, 11, 4431–4438. [Google Scholar]
- Kim, Y.; Kang, H.; Powathil, G.; Kim, H.; Trucu, D.; Lee, W.; Lawler, S.; Chaplain, M. Role of extracellular matrix and microenvironment in regulation of tumor growth and LAR-mediated invasion in glioblastoma. PLoS ONE 2018, 13, e0204865. [Google Scholar] [CrossRef]
- Jahan, N.; Hannila, S.S. Transforming growth factor β-induced expression of chondroitin sulfate proteoglycans is mediated through non-Smad signaling pathways. Exp. Neurol. 2015, 263, 372–384. [Google Scholar] [CrossRef]
- Silver, J.; Miller, J.H. Regeneration beyond the glial scar. Nat. Rev. Neurosci. 2004, 5, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Bradbury, E.J.; Moon, L.; Popat, R.J.; King, V.R.; Bennett, G.S.; Patel, P.N.; Fawcett, J.; McMahon, S. Chondroitinase ABC promotes functional recovery after spinal cord injury. Nature 2002, 416, 636–640. [Google Scholar] [CrossRef] [PubMed]
- Han, K.H.; Cho, H.; Han, K.R.; Mun, S.K.; Kim, Y.K.; Park, I.; Chang, M. Role of microRNA-375-3p-mediated regulation in tinnitus development. Int. J. Mol. Med. 2021, 48, 136. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Heo, H.; Kim, D.H.; Kim, H.J.; Oh, S.H. Neural Plastic Changes in the Subcortical Auditory Neural Pathway after Single-Sided Deafness in Adult Mice: A MEMRI Study. BioMed Res. Int. 2018, 2018, 8624745. [Google Scholar] [CrossRef] [PubMed]
- Alvarado, J.C.; Fuentes-Santamaría, V.; Jareño-Flores, T.; Blanco, J.L.; Juiz, J.M. Normal variations in the morphology of auditory brainstem response (ABR) waveforms: A study in wistar rats. Neurosci. Res. 2012, 73, 302–311. [Google Scholar] [CrossRef]
- Vlachos, I.S.; Zagganas, K.; Paraskevopoulou, M.D.; Georgakilas, G.; Karagkouni, D.; Vergoulis, T.; Dalamagas, T.; Hatzigeorgiou, A.G. DIANA-miRPath v3.0: Deciphering microRNA function with experimental support. Nucleic Acids Res. 2015, 43, W460–W466. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Kim, J.K.; Park, S.H.; Kim, B.G.; Jang, A.S.; Oh, S.H.; Lee, J.H.; Suh, M.W.; Park, M.K. Effects of inhaled particulate matter on the central nervous system in mice. NeuroToxicology 2018, 67, 169–177. [Google Scholar] [CrossRef]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE Guidelines: Minimum Information for Publication of Quantitative Real-Time PCR Experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef]
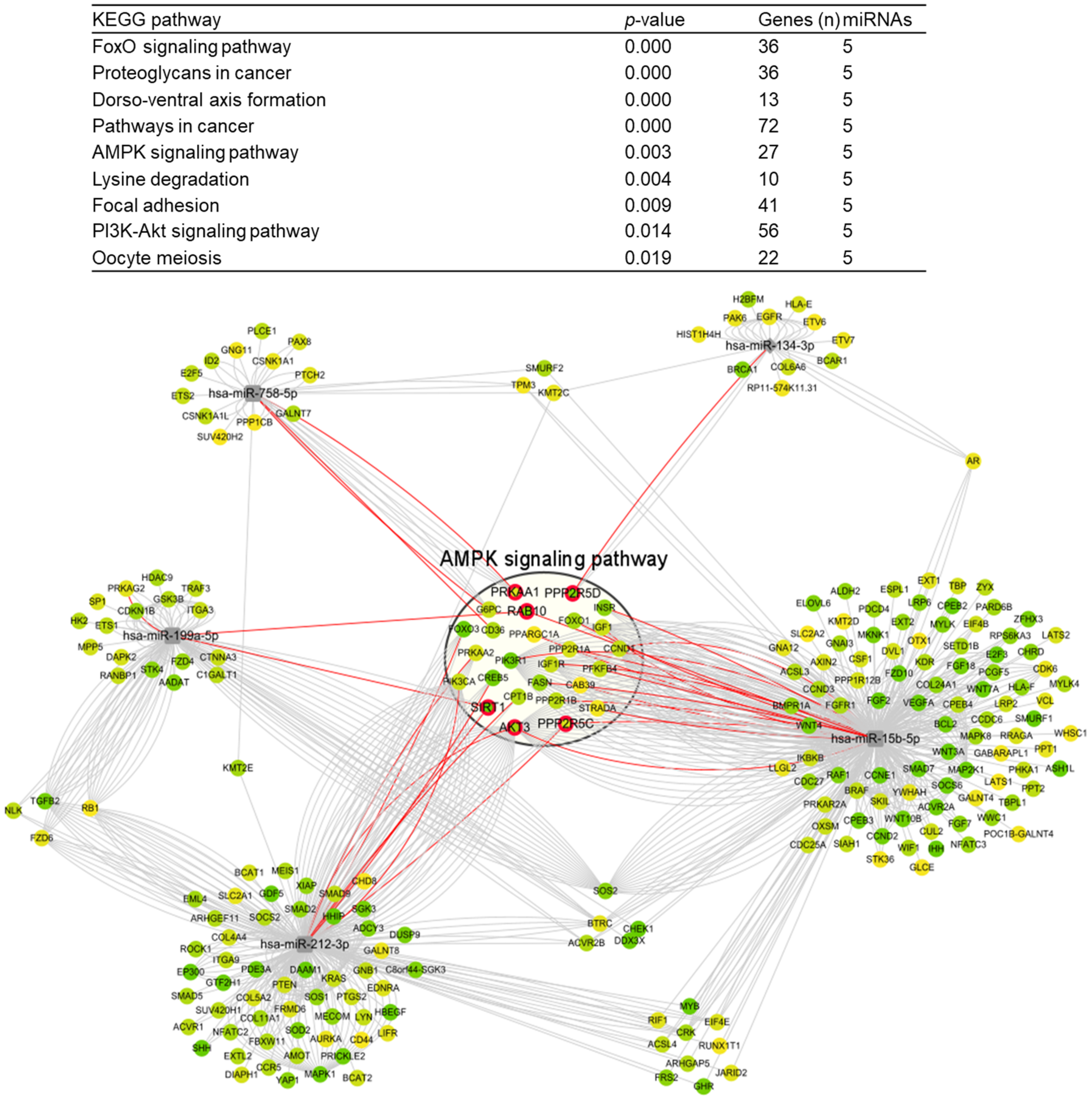
| Related miRNA | Gene Name | Score | |
|---|---|---|---|
| hsa-miR-15b-5p | FASN | fatty acid synthase | 0.935 |
| IGF1R | insulin like growth factor 1 receptor | 0.866 | |
| PPP2R5C | protein phosphatase 2 regulatory subunit B’gamma | 0.845 | |
| CCND1 | cyclin D1 | 0.873 | |
| PPP2R1A | protein phosphatase 2 scaffold subunit Aalpha | 0.847 | |
| PIK3R1 | phosphoinositide-3-kinase regulatory subunit 1 | 0.989 | |
| INSR | insulin receptor | 0.947 | |
| IGF1 | insulin like growth factor 1 | 0.859 | |
| AKT3 | AKT serine/threonine kinase 3 | 1 | |
| PPARGC1A | PPARG coactivator 1 alpha | 0.806 | |
| FOXO1 | forkhead box O1 | 0.913 | |
| CAB39 | calcium binding protein 39 | 0.8 | |
| PPP2R1B | protein phosphatase 2 scaffold subunit Abeta | 0.885 | |
| RAB10 | RAB10, member RAS oncogene family | 0.941 | |
| PFKFB4 | 6-phosphofructo-2-kinase/fructose-2,6-biphosphatase 4 | 0.872 | |
| STRADA | STE20 related adaptor alpha | 0.81 | |
| hsa-miR-758-5p | CD36 | CD36 molecule | 0.901 |
| G6PC | glucose-6-phosphatase catalytic subunit 1 | 0.865 | |
| PRKAA1 | protein kinase AMP-activated catalytic subunit alpha 1 | 0.84 | |
| hsa-miR-199a-5p | SIRT1 | sirtuin 1 | 0.9 |
| PRKAG2 | protein kinase AMP-activated non-catalytic subunit gamma 2 | 0.831 | |
| RAB10 | RAB10, member RAS oncogene family | 0.954 | |
| hsa-miR-212-3p| | PRKAA2 | protein kinase AMP-activated catalytic subunit alpha 2 | 0.844 |
| SIRT1 | sirtuin 1 | 1 | |
| CREB5 | cAMP responsive element binding protein 5 | 0.982 | |
| PPP2R5C | protein phosphatase 2 regulatory subunit B’gamma | 0.889 | |
| AKT3 | AKT serine/threonine kinase 3 | 0.92 | |
| PIK3CA | phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha | 0.864 | |
| FOXO3 | forkhead box O3 | 0.997 | |
| CPT1B | carnitine palmitoyltransferase 1B | 0.892 | |
| hsa-miR-134-3p | PPP2R5D | protein phosphatase 2 regulatory subunit B’delta | 0.965 |
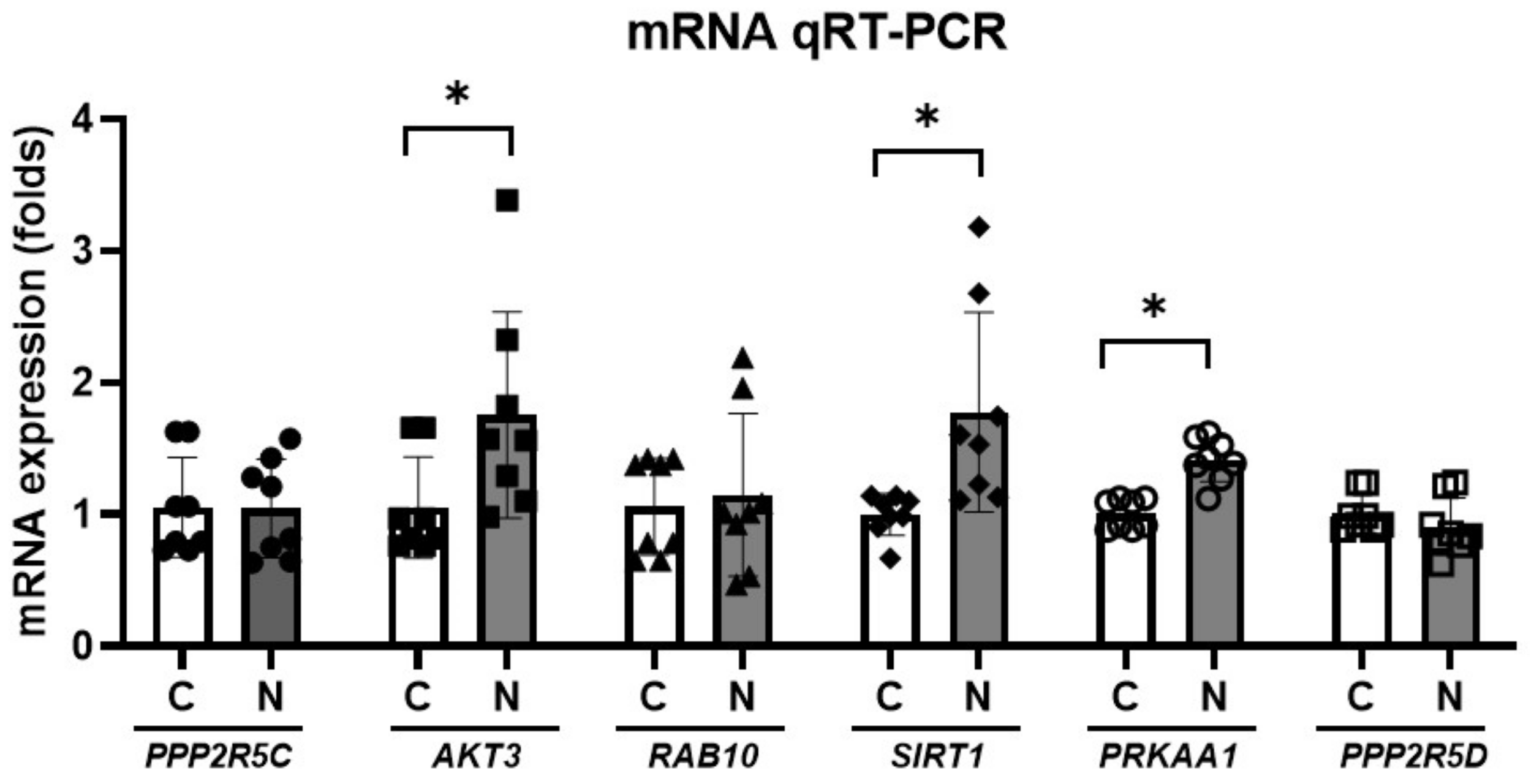
| Gene | Primer Sequence (Forward) | Primer Sequence (Reverse) | Annealing Temperature (°C) | Product Size (bp) |
|---|---|---|---|---|
| PPP2R5C | 5′-CTAGCCAAAGCGAATCCCCA-3′ | 5′-GAGTCTCGTCGCTCACTGTC-3′ | 60 | 93 |
| AKT3 | 5′-CCACCTGAAAAGTATGACGACG-3′ | 5′-TAAGAGCGAGGACTGGTGGA-3′ | 60 | 133 |
| RAB10 | 5′-TTGTTTGCCCCCACTACTCC-3′ | 5′-TAAATGAGGGGCTGACACCG-3′ | 60 | 185 |
| SIRT1 | 5′-GCAGGTTGCGGGAATCCAA-3′ | 5′-GGCAAGATGCTGTTGCAAA-3′ | 60 | 155 |
| PRKAA1 | 5′-GGGTGAAGATCGGCCACTAC-3′ | 5′-CTCTCTGCGGATTTTCCCGA-3′ | 60 | 62 |
| PPP2R5D | 5′-CGAGTCGGGTCGCTAAGAAG-3′ | 5′-ACACTCAGAGTCAAAGGGCG-3′ | 60 | 164 |
| GAPDH | 5′-AGTGCCAGCCTCGTCTCATA-3′ | 5′-AAGAGAAGGCAGCCCTGGTA-3′ | 60 | 93 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, C.H.; Jeon, J.; Lee, S.M.; Kim, S.Y. Differential Expression of miRNAs and Their Predicted Target Pathways in Cochlear Nucleus Following Chronic Noise Exposure in Rats. Cells 2022, 11, 2266. https://doi.org/10.3390/cells11152266
Lee CH, Jeon J, Lee SM, Kim SY. Differential Expression of miRNAs and Their Predicted Target Pathways in Cochlear Nucleus Following Chronic Noise Exposure in Rats. Cells. 2022; 11(15):2266. https://doi.org/10.3390/cells11152266
Chicago/Turabian StyleLee, Chang Ho, Jiwon Jeon, So Min Lee, and So Young Kim. 2022. "Differential Expression of miRNAs and Their Predicted Target Pathways in Cochlear Nucleus Following Chronic Noise Exposure in Rats" Cells 11, no. 15: 2266. https://doi.org/10.3390/cells11152266
APA StyleLee, C. H., Jeon, J., Lee, S. M., & Kim, S. Y. (2022). Differential Expression of miRNAs and Their Predicted Target Pathways in Cochlear Nucleus Following Chronic Noise Exposure in Rats. Cells, 11(15), 2266. https://doi.org/10.3390/cells11152266





