Infertility in Men: Advances towards a Comprehensive and Integrative Strategy for Precision Theranostics
Abstract
:1. Introduction
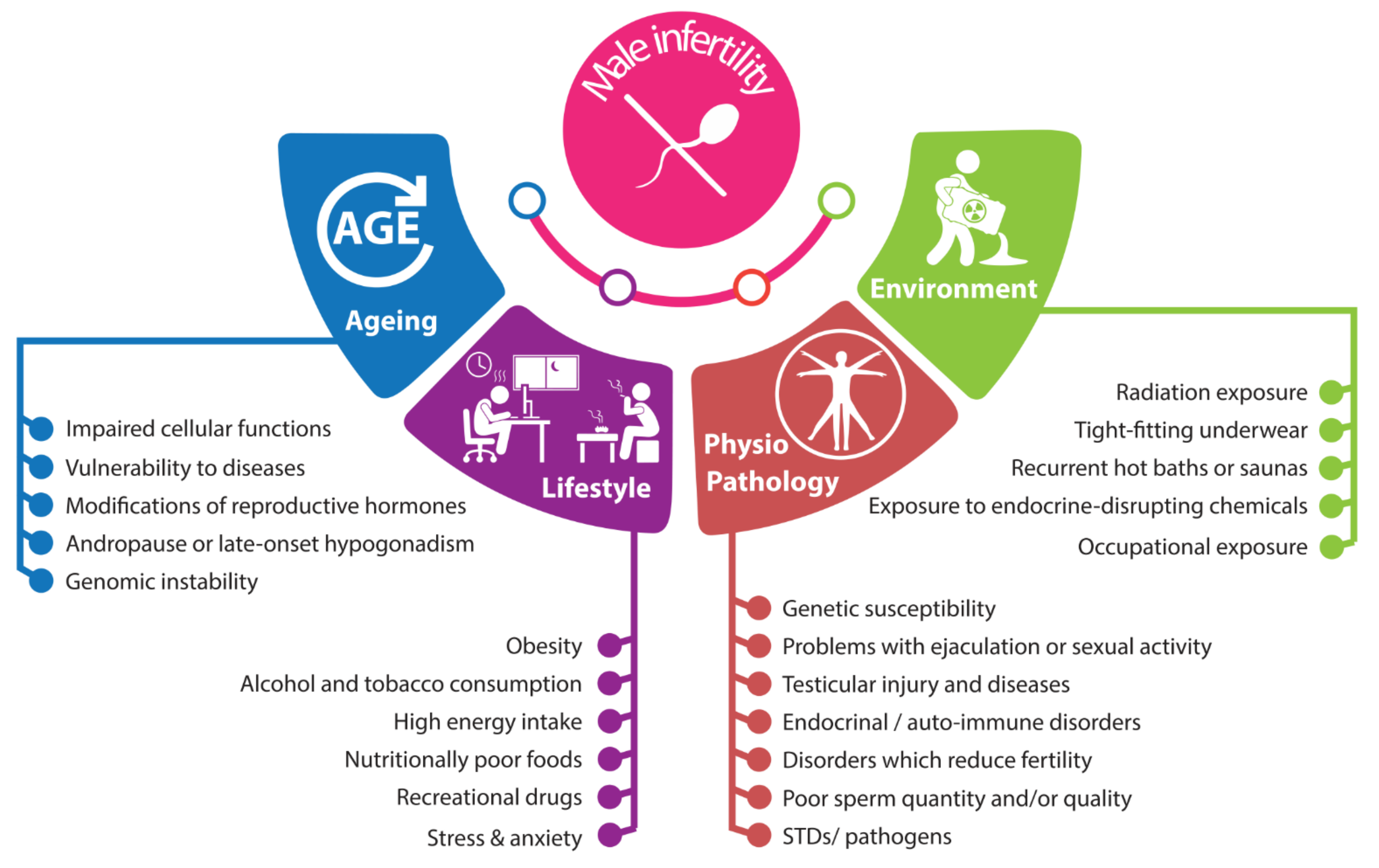
2. Male Infertility: Complex and Interconnected Roots
2.1. Anatomo-Pathophysiological Factors
2.2. Environmental Factors
2.3. Lifestyle
2.4. Aging and Male Infertility
3. Markers of Male Infertility
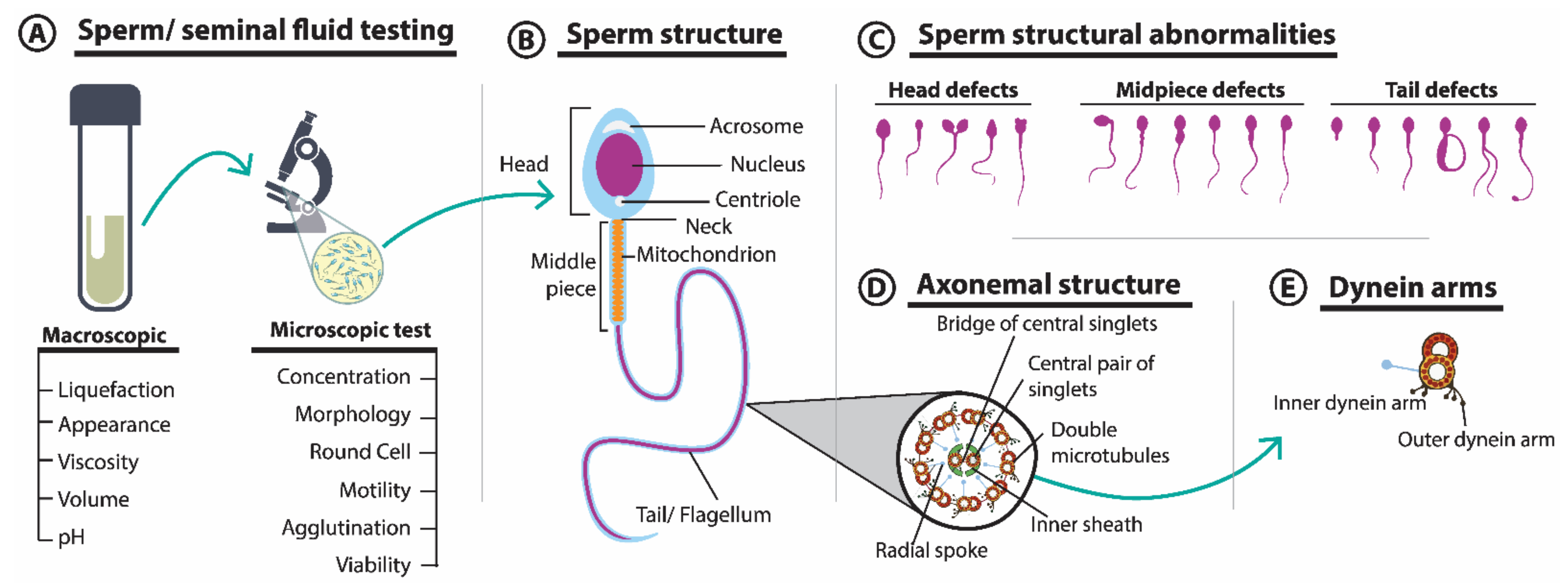
3.1. Seminal Fluid Parameters and Sperm Morphology
3.2. Reactive Oxygen Species
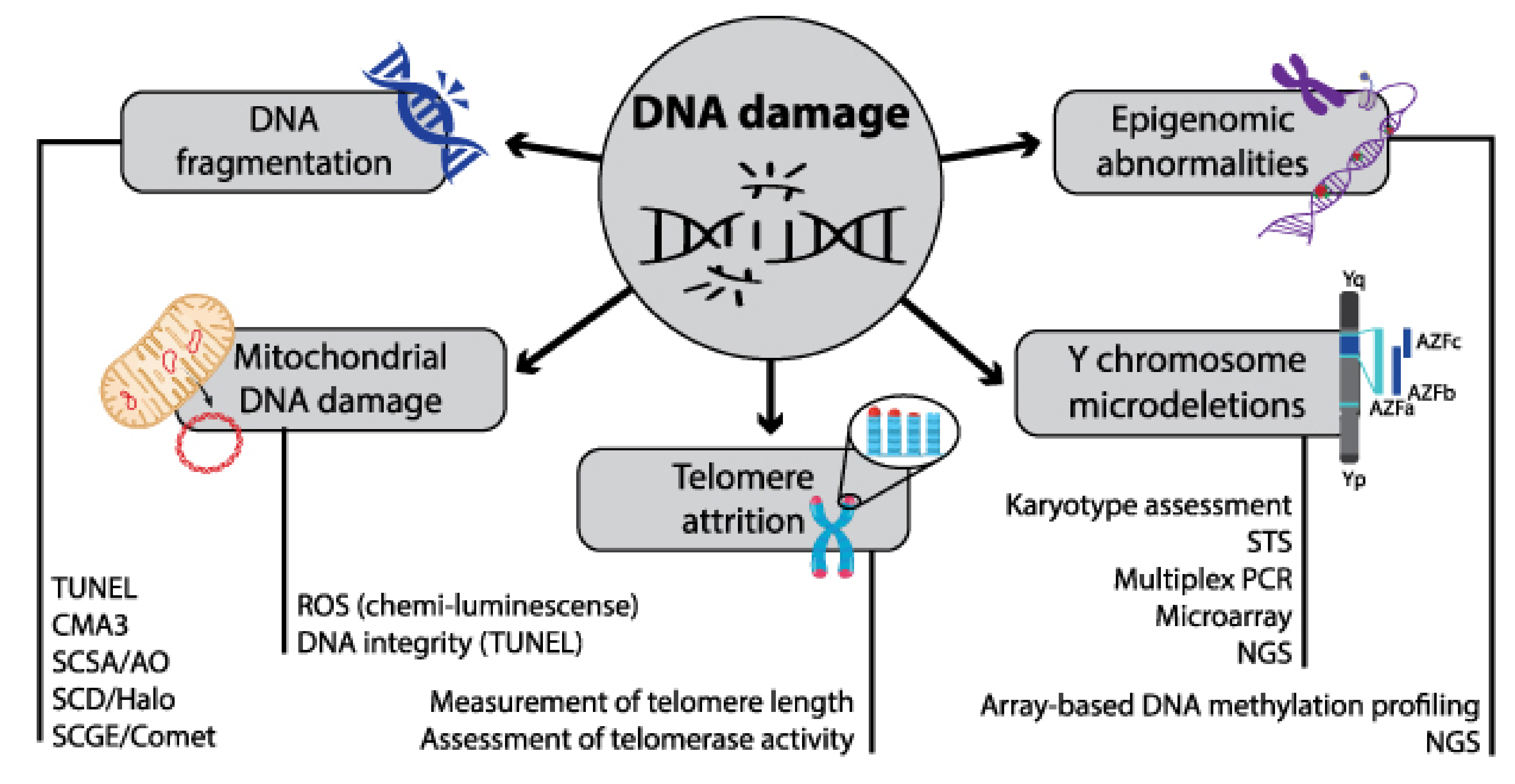
3.3. Sperm DNA Fragmentation (SDF)
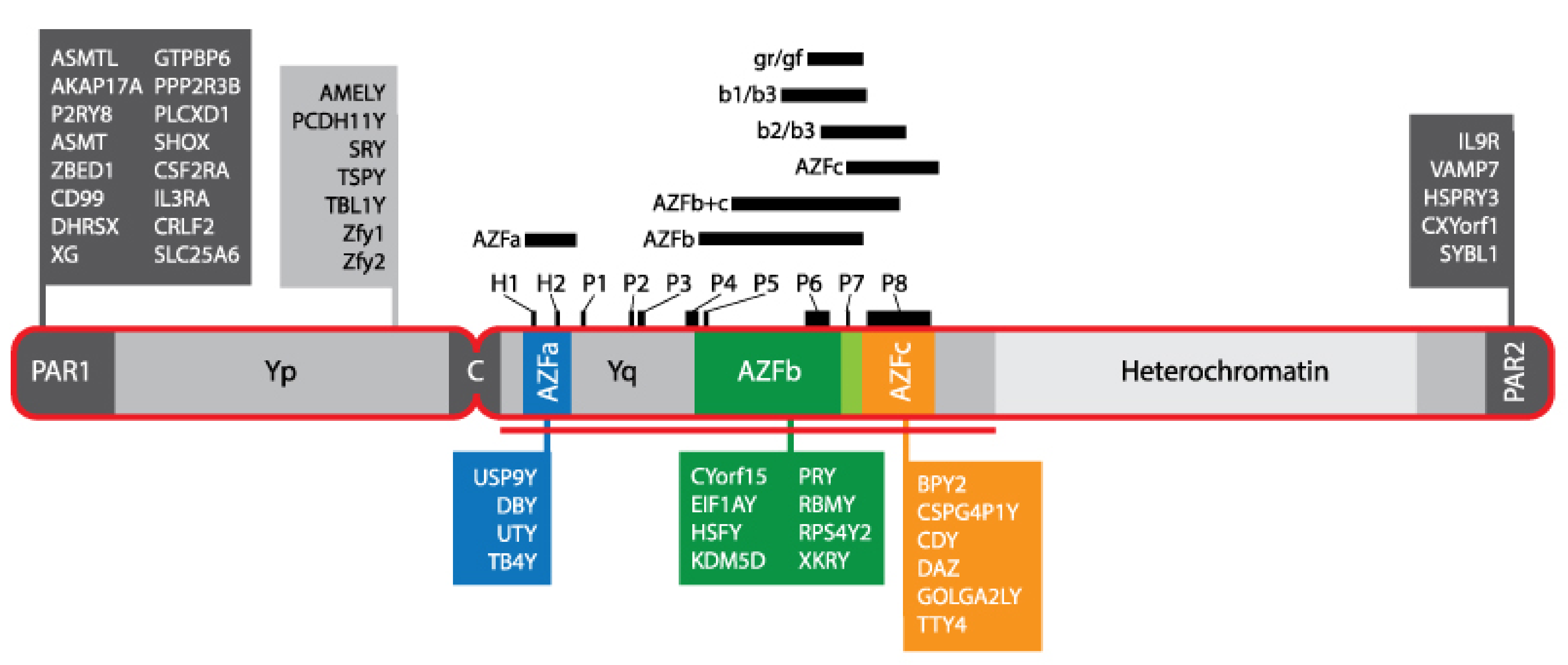
3.4. Genomic Markers
3.5. Transcriptomic and Epigenomic Markers
| miRNA/Transcriptomic and Epigenomic Factors | Regulation | Association with | Ref |
|---|---|---|---|
| miR-196a-2, miR-196a-5p, miR-141, miR-429, and miR-7-1-3p | Up-regulation | Idiopathic male infertility | [97,106] |
| miR-424 | Down-regulation | Idiopathic male infertility | [107] |
| MiR-371a-3p | Up-regulation | Sperm concentration and total sperm count | [108] |
| piR-31068, piR-31098, piR-31925, piR-43771, and piR-43773 | Differentially expressed/ down-regulation | Asthenozoospermia | [109] |
| miR-19b and let-7a | Up-regulation | Idiopathic infertility | [110] |
| hsa-let-7b-5p | Down-regulation | Asthenozoospermia/idiopathic male infertility | [111] |
| miR-192a | Up-regulation | Germ cell apoptosis | [112] |
| miR-23b, miR-146a, miR-155, miR-223, miR-17-92, and miR-34a | Down-regulation | Miscarriage, pre-eclampsia, and small for gestational age fetuses | [113] |
| MTHFR promoter | Hypermethylation | Abnormal concentration/motility of sperm | [114,115,116] |
3.6. Proteomic and Metabolomic Markers
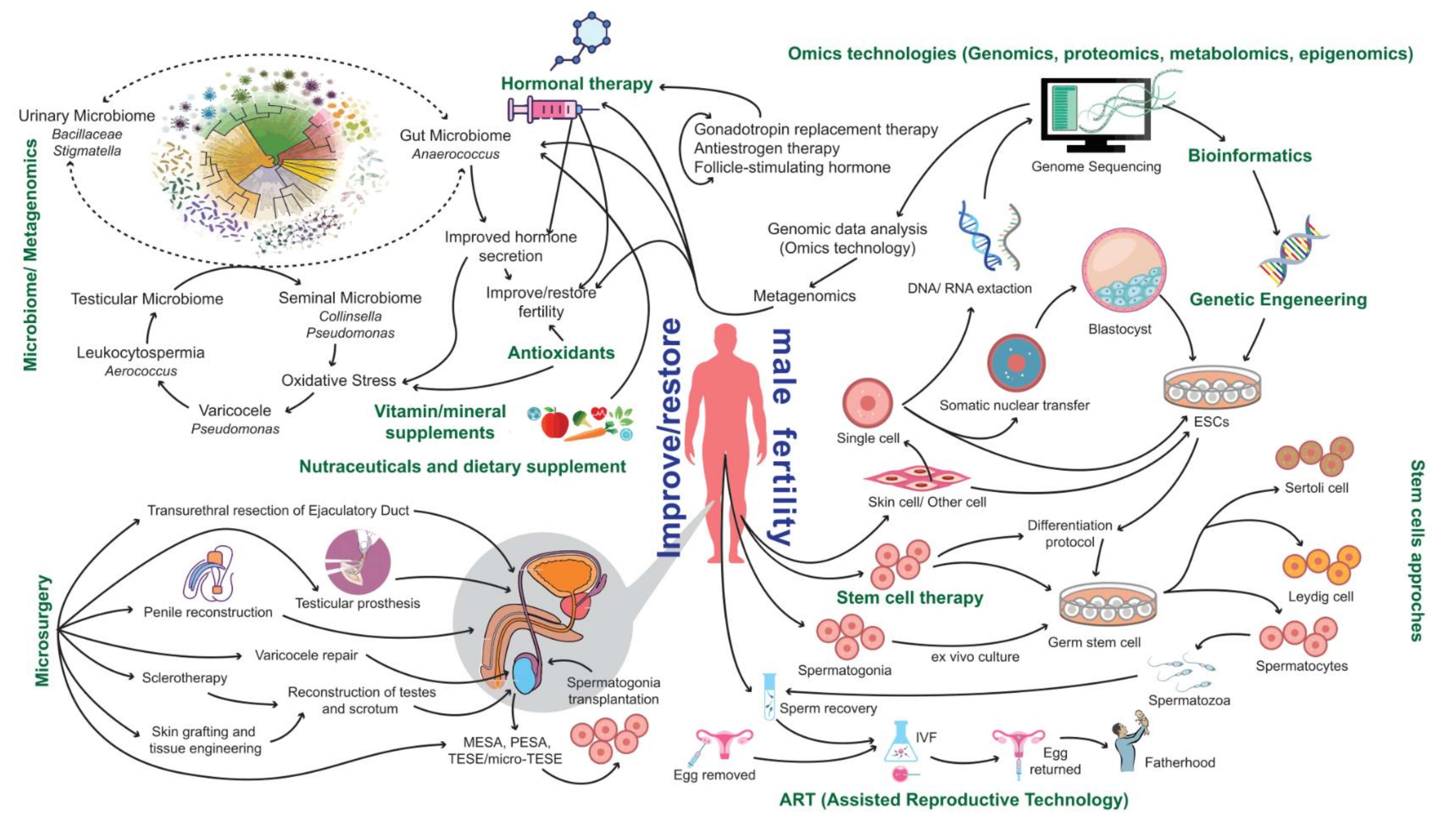
4. Current Therapeutic Options
4.1. Assisted Reproductive Technologies (ARTs)
4.2. Surgical Approaches
4.3. Antioxidants
4.4. Vitamin and Mineral Supplementation
4.5. Hormonal-Based Therapies
5. Promotion of a Healthy Lifestyle: A Promising but Underexplored Approach
6. The Potential of Multiomics
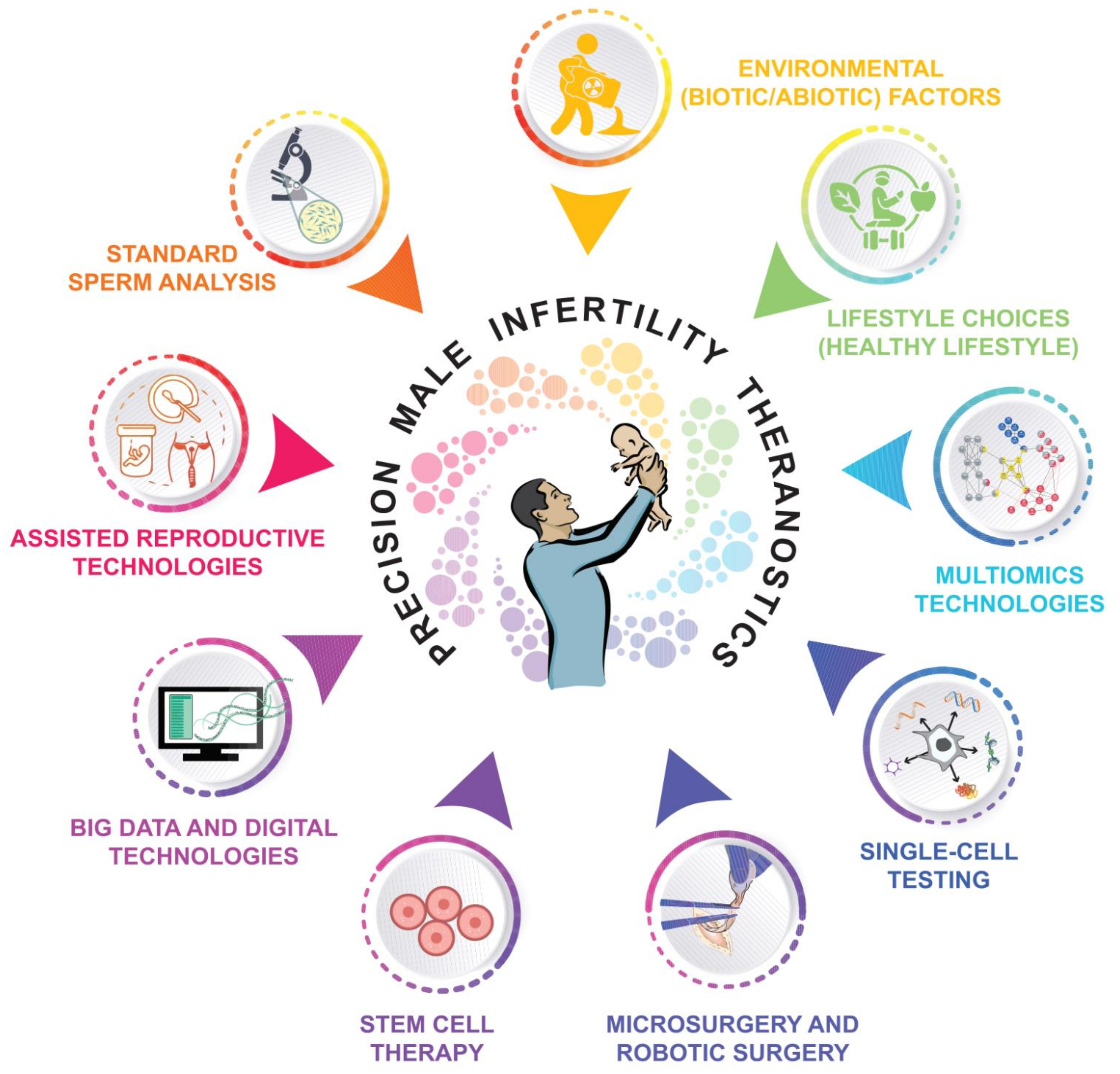
7. Advances towards Precision Male Reproductive Medicine
8. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. International Classification of Diseases, 11th Revision (ICD-11). Available online: https://www.who.int/news-room/fact-sheets/detail/infertility (accessed on 10 January 2022).
- Agarwal, A.; Baskaran, S.; Parekh, N.; Cho, C.-L.; Henkel, R.; Vij, S.; Arafa, M.; Selvam, M.K.P.; Shah, R. Male infertility. Lancet 2021, 397, 319–333. [Google Scholar] [CrossRef]
- Agarwal, A.; Mulgund, A.; Hamada, A.; Chyatte, M.R. A unique view on male infertility around the globe. Reprod. Biol. Endocrinol. RBE 2015, 13, 37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, H.; Gong, T.-T.; Jiang, Y.-T.; Zhang, S.; Zhao, Y.-H.; Wu, Q.-J. Global, regional, and national prevalence and disability-adjusted life-years for infertility in 195 countries and territories, 1990–2017: Results from a global burden of disease study, 2017. Aging 2019, 11, 10952. [Google Scholar] [CrossRef]
- WHO. WHO Laboratory Manual for the Examination and Processing of Human Semen; WHO: Geneva, Switzerland, 2021; Volume 276. [Google Scholar]
- Sharma, R.; Agarwal, A.; Rohra, V.K.; Assidi, M.; Abu-Elmagd, M.; Turki, R.F. Effects of increased paternal age on sperm quality, reproductive outcome and associated epigenetic risks to offspring. Reprod. Biol. Endocrinol. RBE 2015, 13, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Achermann, A.P.P.; Esteves, S.C. Diagnosis and management of infertility due to ejaculatory duct obstruction: Summary evidence. Int. Braz. J. Urol Off. J. Braz. Soc. Urol. 2021, 47, 868–881. [Google Scholar] [CrossRef] [PubMed]
- Saab, M.M.; Landers, M.; Hegarty, J. Males’ awareness of benign testicular disorders: An integrative review. Am. J. Men’s Health 2018, 12, 556–566. [Google Scholar] [CrossRef] [PubMed]
- Babakhanzadeh, E.; Nazari, M.; Ghasemifar, S.; Khodadadian, A. Some of the factors involved in male infertility: A prospective review. Int. J. Gen. Med. 2020, 13, 29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lucas, X. Clinical use of deslorelin (GnRH agonist) in companion animals: A review. Reprod. Domest. Anim. 2014, 49, 64–71. [Google Scholar] [CrossRef] [Green Version]
- Tsevat, D.G.; Wiesenfeld, H.C.; Parks, C.; Peipert, J.F. Sexually transmitted diseases and infertility. Am. J. Obstet. Gynecol. 2017, 216, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Farsimadan, M.; Motamedifar, M. Bacterial infection of the male reproductive system causing infertility. J. Reprod. Immunol. 2020, 142, 103183. [Google Scholar] [CrossRef]
- Liu, W.; Han, R.; Wu, H.; Han, D. Viral threat to male fertility. Andrologia 2018, 50, e13140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dutta, S.; Majzoub, A.; Agarwal, A. Oxidative stress and sperm function: A systematic review on evaluation and management. Arab J. Urol. 2019, 17, 87–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bisht, S.; Faiq, M.; Tolahunase, M.; Dada, R. Oxidative stress and male infertility. Nat. Rev. Urol. 2017, 14, 470–485. [Google Scholar] [CrossRef] [PubMed]
- Hart, K.; Tadros, N.N. The role of environmental factors and lifestyle on male reproductive health, the epigenome, and resulting offspring. Panminerva Med. 2019, 61, 187–195. [Google Scholar] [CrossRef]
- Cescon, M.; Chianese, R.; Tavares, R.S. Environmental impact on male (In) fertility via epigenetic route. J. Clin. Med. 2020, 9, 2520. [Google Scholar] [CrossRef]
- Marić, T.; Fučić, A.; Aghayanian, A. Environmental and occupational exposures associated with male infertility. Arch. Ind. Hyg. Toxicol. 2021, 72, 101–113. [Google Scholar] [CrossRef]
- Giulioni, C.; Maurizi, V.; Scarcella, S.; Di Biase, M.; Iacovelli, V.; Galosi, A.B.; Castellani, D. Do environmental and occupational exposure to pyrethroids and organophosphates affect human semen parameters? Results of a systematic review and meta—analysis. Andrologia 2021, 53, e14215. [Google Scholar] [CrossRef]
- Martelli, M.; Zingaretti, L.; Salvio, G.; Bracci, M.; Santarelli, L. Influence of Work on Andropause and Menopause: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 10074. [Google Scholar] [CrossRef]
- Leisegang, K.; Dutta, S. Do lifestyle practices impede male fertility? Andrologia 2021, 53, e13595. [Google Scholar] [CrossRef]
- Yu, G.; Bai, Z.; Song, C.; Cheng, Q.; Wang, G.; Tang, Z.; Yang, S. Current progress on the effect of mobile phone radiation on sperm quality: An updated systematic review and meta-analysis of human and animal studies. Environ. Pollut. 2021, 282, 116952. [Google Scholar] [CrossRef]
- Kim, S.; Han, D.; Ryu, J.; Kim, K.; Kim, Y.H. Effects of mobile phone usage on sperm quality–No time-dependent relationship on usage: A systematic review and updated meta-analysis. Environ. Res. 2021, 202, 111784. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xu, A.; Gong, L.; Chen, Z.; Zhang, B.; Li, X. The Microbiome, an Important Factor That Is Easily Overlooked in Male Infertility. Front Microbiol. 2022, 13, 831272. [Google Scholar] [CrossRef] [PubMed]
- Venneri, M.A.; Franceschini, E.; Sciarra, F.; Rosato, E.; D’Ettorre, G.; Lenzi, A. Human genital tracts microbiota: Dysbiosis crucial for infertility. J. Endocrinol. Investig. 2022, 45, 1151–1160. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xie, Z. Exploring the role of gut microbiome in male reproduction. Andrology 2022, 10, 441–450. [Google Scholar] [CrossRef]
- Campbell, J.M.; Lane, M.; Owens, J.A.; Bakos, H.W. Paternal obesity negatively affects male fertility and assisted reproduction outcomes: A systematic review and meta-analysis. Reprod. Biomed. Online 2015, 31, 593–604. [Google Scholar] [CrossRef] [Green Version]
- Mir, J.; Franken, D.; Andrabi, S.; Ashraf, M.; Rao, K. Impact of weight loss on sperm DNA integrity in obese men. Andrologia 2018, 50, e12957. [Google Scholar] [CrossRef]
- Ding, N.; Zhang, X.; Di Zhang, X.; Jing, J.; Liu, S.S.; Mu, Y.P.; Peng, L.L.; Yan, Y.J.; Xiao, G.M.; Bi, X.Y. Impairment of spermatogenesis and sperm motility by the high-fat diet-induced dysbiosis of gut microbes. Gut 2020, 69, 1608–1619. [Google Scholar] [CrossRef] [Green Version]
- Skoracka, K.; Eder, P.; Łykowska-Szuber, L.; Dobrowolska, A.; Krela-Kaźmierczak, I. Diet and nutritional factors in male (in) fertility—underestimated factors. J. Clin. Med. 2020, 9, 1400. [Google Scholar] [CrossRef]
- Toledo, E.; Lopez-del Burgo, C.; Ruiz-Zambrana, A.; Donazar, M.; Navarro-Blasco, Í.; Martínez-González, M.A.; de Irala, J. Dietary patterns and difficulty conceiving: A nested case–control study. Fertil. Steril. 2011, 96, 1149–1153. [Google Scholar] [CrossRef] [Green Version]
- Eslamian, G.; Amirjannati, N.; Rashidkhani, B.; Sadeghi, M.R.; Hekmatdoost, A. Nutrient patterns and asthenozoospermia: A case–control study. Andrologia 2017, 49, e12624. [Google Scholar] [CrossRef]
- Wise, L.A.; Wesselink, A.K.; Tucker, K.L.; Saklani, S.; Mikkelsen, E.M.; Cueto, H.; Riis, A.H.; Trolle, E.; McKinnon, C.J.; Hahn, K.A. Dietary fat intake and fecundability in 2 preconception cohort studies. Am. J. Epidemiol. 2018, 187, 60–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fontana, R.; Torre, S.D. The deep correlation between energy metabolism and reproduction: A view on the effects of nutrition for women fertility. Nutrients 2016, 8, 87. [Google Scholar] [CrossRef] [PubMed]
- Salas-Huetos, A.; James, E.R.; Aston, K.I.; Jenkins, T.G.; Carrell, D.T. Diet and sperm quality: Nutrients, foods and dietary patterns. Reprod. Biol. 2019, 19, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Salas-Huetos, A.; Bulló, M.; Salas-Salvadó, J. Dietary patterns, foods and nutrients in male fertility parameters and fecundability: A systematic review of observational studies. Hum. Reprod. Update 2017, 23, 371–389. [Google Scholar] [CrossRef]
- Giahi, L.; Mohammadmoradi, S.; Javidan, A.; Sadeghi, M.R. Nutritional modifications in male infertility: A systematic review covering 2 decades. Nutr. Rev. 2016, 74, 118–130. [Google Scholar] [CrossRef] [Green Version]
- Showell, M.G.; Mackenzie—Proctor, R.; Brown, J.; Yazdani, A.; Stankiewicz, M.T.; Hart, R.J. Antioxidants for male subfertility. Cochrane Database Syst. Rev. 2014, 12, CD007411. [Google Scholar] [CrossRef]
- Danielewicz, A.; Przybyłowicz, K.E.; Przybyłowicz, M. Dietary patterns and poor semen quality risk in men: A cross-sectional study. Nutrients 2018, 10, 1162. [Google Scholar] [CrossRef] [Green Version]
- Salas-Huetos, A.; Babio, N.; Carrell, D.T.; Bulló, M.; Salas-Salvadó, J. Adherence to the Mediterranean diet is positively associated with sperm motility: A cross-sectional analysis. Sci. Rep. 2019, 9, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Ricci, E.; Viganò, P.; Cipriani, S.; Somigliana, E.; Chiaffarino, F.; Bulfoni, A.; Parazzini, F. Coffee and caffeine intake and male infertility: A systematic review. Nutr. J. 2017, 16, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Kumar, N.; Singh, A.K. Impact of Faulty Lifestyle Behaviors on Male Fertility: A Narrative Review. Eurasian J. Med. Biol. Sci. 2021, 1, 70–79. [Google Scholar]
- Gentiluomo, M.; Luddi, A.; Cingolani, A.; Fornili, M.; Governini, L.; Lucenteforte, E.; Baglietto, L.; Piomboni, P.; Campa, D. Telomere length and male fertility. Int. J. Mol. Sci. 2021, 22, 3959. [Google Scholar] [CrossRef] [PubMed]
- Mazur, D.J.; Lipshultz, L.I. Infertility in the aging male. Curr. Urol. Rep. 2018, 19, 1–9. [Google Scholar] [CrossRef] [PubMed]
- du Fossé, N.A.; Van der Hoorn, M.-L.P.; van Lith, J.M.; le Cessie, S.; Lashley, E.E. Advanced paternal age is associated with an increased risk of spontaneous miscarriage: A systematic review and meta-analysis. Hum. Reprod. Update 2020, 26, 650–669. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Wang, Y.; Peng, M.; Xu, J.; Fan, Z.; Liu, C.; Zhao, K.; Zhang, H. Effect of paternal age on offspring birth defects: A systematic review and meta-analysis. Aging 2020, 12, 25373. [Google Scholar] [CrossRef]
- Shay, J.W.; Wright, W.E. Telomeres and telomerase: Three decades of progress. Nat. Rev. Genet. 2019, 20, 299–309. [Google Scholar] [CrossRef]
- Brandt, J.S.; Cruz Ithier, M.A.; Rosen, T.; Ashkinadze, E. Advanced paternal age, infertility, and reproductive risks: A review of the literature. Prenat. Diagn. 2019, 39, 81–87. [Google Scholar] [CrossRef] [Green Version]
- Nguyen-Powanda, P.; Robaire, B. Oxidative stress and reproductive function in the aging male. Biology 2020, 9, 282. [Google Scholar] [CrossRef]
- Coutton, C.; Vargas, A.S.; Amiri-Yekta, A.; Kherraf, Z.-E.; Mustapha, S.F.B.; Le Tanno, P.; Wambergue-Legrand, C.; Karaouzène, T.; Martinez, G.; Crouzy, S. Mutations in CFAP43 and CFAP44 cause male infertility and flagellum defects in Trypanosoma and human. Nat. Commun. 2018, 9, 1–18. [Google Scholar] [CrossRef]
- Whitfield, M.; Thomas, L.; Bequignon, E.; Schmitt, A.; Stouvenel, L.; Montantin, G.; Tissier, S.; Duquesnoy, P.; Copin, B.; Chantot, S. Mutations in DNAH17, encoding a sperm-specific axonemal outer dynein arm heavy chain, cause isolated male infertility due to asthenozoospermia. Am. J. Hum. Genet. 2019, 105, 198–212. [Google Scholar] [CrossRef] [Green Version]
- Sironen, A.; Shoemark, A.; Patel, M.; Loebinger, M.R.; Mitchison, H.M. Sperm defects in primary ciliary dyskinesia and related causes of male infertility. Cell. Mol. Life Sci. 2020, 77, 2029–2048. [Google Scholar] [CrossRef] [Green Version]
- Agarwal, A.; Parekh, N.; Selvam, M.K.P.; Henkel, R.; Shah, R.; Homa, S.T.; Ramasamy, R.; Ko, E.; Tremellen, K.; Esteves, S. Male oxidative stress infertility (MOSI): Proposed terminology and clinical practice guidelines for management of idiopathic male infertility. World J. Men’s Health 2019, 37, 296–312. [Google Scholar] [CrossRef] [PubMed]
- Brunner, R.J.; Demeter, J.H.; Sindhwani, P. Review of guidelines for the evaluation and treatment of leukocytospermia in male infertility. World J. Men’s Health 2019, 37, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Villaverde, A.I.S.B.; Netherton, J.; Baker, M.A. From past to present: The link between reactive oxygen species in sperm and male infertility. Antioxidants 2019, 8, 616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takeshima, T.; Usui, K.; Mori, K.; Asai, T.; Yasuda, K.; Kuroda, S.; Yumura, Y. Oxidative stress and male infertility. Reprod. Med. Biol. 2021, 20, 41–52. [Google Scholar] [CrossRef]
- Agarwal, A.; Panner Selvam, M.K.; Baskaran, S.; Cho, C.-L. Sperm DNA damage and its impact on male reproductive health: A critical review for clinicians, reproductive professionals and researchers. Expert Rev. Mol. Diagn. 2019, 19, 443–457. [Google Scholar] [CrossRef]
- Le, M.T.; Nguyen, T.A.T.; Nguyen, H.T.T.; Nguyen, T.T.T.; Nguyen, V.T.; Le, D.D.; Nguyen, V.Q.H.; Cao, N.T. Does sperm DNA fragmentation correlate with semen parameters? Reprod. Med. Biol. 2019, 18, 390–396. [Google Scholar] [CrossRef] [Green Version]
- Chatterjee, N.; Walker, G.C. Mechanisms of DNA damage, repair, and mutagenesis. Environ. Mol. Mutagenesis 2017, 58, 235–263. [Google Scholar] [CrossRef] [Green Version]
- Muratori, M.; Tamburrino, L.; Marchiani, S.; Cambi, M.; Olivito, B.; Azzari, C.; Forti, G.; Baldi, E. Investigation on the origin of sperm DNA fragmentation: Role of apoptosis, immaturity and oxidative stress. Mol. Med. 2015, 21, 109–122. [Google Scholar] [CrossRef]
- Bui, A.; Sharma, R.; Henkel, R.; Agarwal, A. Reactive oxygen species impact on sperm DNA and its role in male infertility. Andrologia 2018, 50, e13012. [Google Scholar] [CrossRef]
- Tahmasbpour Marzouni, E.; Ilkhani, H.; Beigi Harchegani, A.; Shafaghatian, H.; Layali, I.; Shahriary, A. Epigenetic Modifications, A New Approach to Male Infertility Etiology: A Review. Int. J. Fertil. Steril. 2022, 16, 1–9. [Google Scholar] [CrossRef]
- Nawaz, S.; Ullah, M.I.; Hamid, B.S.; Nargis, J.; Nawaz, M.; Hussain, S.; Ahmad, W. A loss-of-function variant in DNA mismatch repair gene MLH3 underlies severe oligozoospermia. J. Hum. Genet. 2021, 66, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Hekim, N.; Gunes, S.; Asci, R.; Henkel, R.; Abur, U. Semiquantitative promoter methylation of MLH1 and MSH2 genes and their impact on sperm DNA fragmentation and chromatin condensation in infertile men. Andrologia 2021, 53, e13827. [Google Scholar] [CrossRef] [PubMed]
- Witherspoon, L.; Dergham, A.; Flannigan, R. Y-microdeletions: A review of the genetic basis for this common cause of male infertility. Transl. Androl. Urol. 2021, 10, 1383. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, S.; Sharma, R.; Gupta, S.; Cakar, Z.; De Geyter, C.; Agarwal, A. Inter—and intra—laboratory standardization of TUNEL assay for assessment of sperm DNA fragmentation. Andrology 2017, 5, 477–485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zini, A.; Agarwal, A. A Clinician’s Guide to Sperm DNA and Chromatin Damage; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Dutta, S.; Henkel, R.; Agarwal, A. Comparative analysis of tests used to assess sperm chromatin integrity and DNA fragmentation. Andrologia 2021, 53, e13718. [Google Scholar] [CrossRef]
- Krausz, C.; Rosta, V.; Swerdloff, R.S.; Wang, C. Genetics of male infertility. In Emery and Rimoin’s Principles and Practice of Medical Genetics and Genomics; Pyeritz, R.E., Korf, B.R., Grody, W.W., Eds.; Elsevier: Berkeley, CA, USA, 2022; pp. 121–147. [Google Scholar]
- Oud, M.S.; Volozonoka, L.; Smits, R.M.; Vissers, L.E.; Ramos, L.; Veltman, J.A. A systematic review and standardized clinical validity assessment of male infertility genes. Hum. Reprod. 2019, 34, 932–941. [Google Scholar] [CrossRef] [Green Version]
- Rabinowitz, M.J.; Huffman, P.J.; Haney, N.M.; Kohn, T.P. Y-chromosome microdeletions: A review of prevalence, screening, and clinical considerations. Appl. Clin. Genet. 2021, 14, 51. [Google Scholar] [CrossRef]
- Punjani, N.; Kang, C.; Schlegel, P.N. Clinical implications of Y chromosome microdeletions among infertile men. Best Pract. Res. Clin. Endocrinol. Metab. 2020, 34, 101471. [Google Scholar] [CrossRef]
- Colaco, S.; Modi, D. Genetics of the human Y chromosome and its association with male infertility. Reprod. Biol. Endocrinol. 2018, 16, 1–24. [Google Scholar] [CrossRef] [Green Version]
- Stahl, P.J.; Masson, P.; Mielnik, A.; Marean, M.B.; Schlegel, P.N.; Paduch, D.A. A decade of experience emphasizes that testing for Y microdeletions is essential in American men with azoospermia and severe oligozoospermia. Fertil. Steril. 2010, 94, 1753–1756. [Google Scholar] [CrossRef]
- Yuen, R.K.; Merkoulovitch, A.; MacDonald, J.R.; Vlasschaert, M.; Lo, K.; Grober, E.; Marshall, C.R.; Jarvi, K.A.; Kolomietz, E.; Scherer, S.W. Development of a high-resolution Y-chromosome microarray for improved male infertility diagnosis. Fertil. Steril. 2014, 101, 1079–1085.e3. [Google Scholar] [CrossRef] [PubMed]
- Thirumavalavan, N.; Gabrielsen, J.S.; Lamb, D.J. Where are we going with gene screening for male infertility? Fertil. Steril. 2019, 111, 842–850. [Google Scholar] [CrossRef] [PubMed]
- Kruger, A.N.; Ellison, Q.; Brogley, M.A.; Gerlinger, E.R.; Mueller, J.L. Male mice with large inversions or deletions of X-chromosome palindrome arms are fertile and express their associated genes during post-meiosis. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Vockel, M.; Riera-Escamilla, A.; Tüttelmann, F.; Krausz, C. The X chromosome and male infertility. Hum. Genet. 2021, 140, 203–215. [Google Scholar] [CrossRef] [Green Version]
- Tuttelmann, F. Aberrations on the X-chromosome as cause of male infertility. Endocrine Abstracts 2016, 41, S17.1. [Google Scholar] [CrossRef] [Green Version]
- Signore, F.; Gulìa, C.; Votino, R.; De Leo, V.; Zaami, S.; Putignani, L.; Gigli, S.; Santini, E.; Bertacca, L.; Porrello, A. The role of number of copies, structure, behavior and copy number variations (CNV) of the Y chromosome in male infertility. Genes 2020, 11, 40. [Google Scholar] [CrossRef] [Green Version]
- Ji, J.; Qin, Y.; Wang, R.; Huang, Z.; Zhang, Y.; Zhou, R.; Song, L.; Ling, X.; Hu, Z.; Miao, D. Copy number gain of VCX, X-linked multi-copy gene, leads to cell proliferation and apoptosis during spermatogenesis. Oncotarget 2016, 7, 78532. [Google Scholar] [CrossRef] [Green Version]
- Salas-Huetos, A.; Aston, K.I. Defining new genetic etiologies of male infertility: Progress and future prospects. Transl. Androl. Urol. 2021, 10, 1486. [Google Scholar] [CrossRef]
- Xavier, M.; Salas-Huetos, A.; Oud, M.; Aston, K.; Veltman, J. Disease gene discovery in male infertility: Past, present and future. Hum. Genet. 2021, 140, 7–19. [Google Scholar] [CrossRef]
- Tüttelmann, F.; Rajpert-De Meyts, E.; Nieschlag, E.; Simoni, M. Gene polymorphisms and male infertility–a meta-analysis and literature review. Reprod. Biomed. Online 2007, 15, 643–658. [Google Scholar] [CrossRef]
- Hong, H.-h.; Hu, Y.; Yu, X.-q.; Zhou, L.; Lv, M.-q.; Sun, Y.; Ren, W.-j.; Zhou, D.-x. Associations of C677T polymorphism in methylenetetrahydrofolate reductase (MTHFR) gene with male infertility risk: A meta-analysis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2017, 212, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Han, L.-J.; He, X.-F.; Ye, X.-H. Methylenetetrahydrofolate reductase C677T and A1298C polymorphisms and male infertility risk: An updated meta-analysis. Medicine 2020, 99, e23662. [Google Scholar] [CrossRef] [PubMed]
- Karimian, M.; Parvaresh, L.; Behjati, M. Genetic variations as molecular diagnostic factors for idiopathic male infertility: Current knowledge and future perspectives. Expert Rev. Mol. Diagn. 2021, 21, 1191–1210. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, T.; Abdel-Hamid, I.; Taymour, M.; Ali, O. Genetic variants in varicocele-related male infertility: A systematic review and future directions. Hum. Fertil. 2021, 24, 1–17. [Google Scholar] [CrossRef]
- Metin Mahmutoglu, A.; Gunes, S.; Asci, R.; Henkel, R.; Aydin, O. Association of XRCC1 and ERCC2 promoters’ methylation with chromatin condensation and sperm DNA fragmentation in idiopathic oligoasthenoteratozoospermic men. Andrologia 2021, 53, e13925. [Google Scholar] [CrossRef]
- Gunes, S.; Agarwal, A.; Henkel, R.; Mahmutoglu, A.; Sharma, R.; Esteves, S.; Aljowair, A.; Emirzeoglu, D.; Alkhani, A.; Pelegrini, L. Association between promoter methylation of MLH 1 and MSH 2 and reactive oxygen species in oligozoospermic men—A pilot study. Andrologia 2018, 50, e12903. [Google Scholar] [CrossRef] [Green Version]
- Aitken, R.J.; Drevet, J.R. The importance of oxidative stress in determining the functionality of mammalian spermatozoa: A two-edged sword. Antioxidants 2020, 9, 111. [Google Scholar] [CrossRef] [Green Version]
- Thomas, H.I.; Chen, Y.-S.; Hung, C.-H.; Sreerangaraja Urs, D.B.; Liao, T.-L.; Lai, Y.-C.; Komrskova, K.; Postlerová, P.; Lin, Y.-F.; Kao, S.-H. Genetic Association in the Maintenance of the Mitochondrial Microenvironment and Sperm Capacity. Oxidative Med. Cell. Longev. 2021, 2021, 5561395. [Google Scholar] [CrossRef]
- Dzudzor, B.; Bimah, B.; Amarh, V.; Ocloo, A. Sperm parameters and mitochondrial DNA sequence variants among patients at a fertility clinic in Ghana. PLoS ONE 2021, 16, e0252923. [Google Scholar]
- Giacone, F.; Cannarella, R.; Mongioì, L.M.; Alamo, A.; Condorelli, R.A.; Calogero, A.E.; La Vignera, S. Epigenetics of male fertility: Effects on assisted reproductive techniques. World J. Men’s Health 2019, 37, 148–156. [Google Scholar] [CrossRef]
- Cannarella, R.; Condorelli, R.A.; Mongioì, L.M.; La Vignera, S.; Calogero, A.E. Molecular biology of spermatogenesis: Novel targets of apparently idiopathic male infertility. Int. J. Mol. Sci. 2020, 21, 1728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, U.; Conine, C.C.; Shea, J.M.; Boskovic, A.; Derr, A.G.; Bing, X.Y.; Belleannee, C.; Kucukural, A.; Serra, R.W.; Sun, F. Biogenesis and function of tRNA fragments during sperm maturation and fertilization in mammals. Science 2016, 351, 391–396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, J.; Gu, H.; Tang, Q.; Wu, W.; Yuan, B.; Guo, D.; Wei, Y.; Sun, H.; Xia, Y.; Ding, H. Common SNP in hsa-miR-196a-2 increases hsa-miR-196a-5p expression and predisposes to idiopathic male infertility in Chinese Han population. Sci. Rep. 2016, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Barceló, M.; Mata, A.; Bassas, L.; Larriba, S. Exosomal microRNAs in seminal plasma are markers of the origin of azoospermia and can predict the presence of sperm in testicular tissue. Hum. Reprod. 2018, 33, 1087–1098. [Google Scholar] [CrossRef] [PubMed]
- Manfrevola, F.; Ferraro, B.; Sellitto, C.; Rocco, D.; Fasano, S.; Pierantoni, R.; Chianese, R. CRISP2, CATSPER1 and PATE1 Expression in Human Asthenozoospermic Semen. Cells 2021, 10, 1956. [Google Scholar] [CrossRef]
- Fend-Guella, D.L.; von Kopylow, K.; Spiess, A.-N.; Schulze, W.; Salzbrunn, A.; Diederich, S.; El Hajj, N.; Haaf, T.; Zechner, U.; Linke, M. The DNA methylation profile of human spermatogonia at single-cell-and single-allele-resolution refutes its role in spermatogonial stem cell function and germ cell differentiation. Mol. Hum. Reprod. 2019, 25, 283–294. [Google Scholar] [CrossRef]
- Franzago, M.; Rovere, M.L.; Franchi, P.G.; Vitacolonna, E.; Stuppia, L. Epigenetics and human reproduction: The primary prevention of the noncommunicable diseases. Epigenomics 2019, 11, 1441–1460. [Google Scholar] [CrossRef]
- Carrell, D.T. The sperm epigenome: Implications for assisted reproductive technologies. In Genetic Damage in Human Spermatozoa; Springer: Berlin/Heidelberg, Germany, 2019; pp. 47–56. [Google Scholar]
- McSwiggin, H.; O’Doherty, A. Epigenetic reprogramming during spermatogenesis and male factor infertility. Reproduction 2018, 156, R9–R21. [Google Scholar] [CrossRef]
- Alkhaled, Y.; Laqqan, M.; Tierling, S.; Lo Porto, C.; Hammadeh, M. DNA methylation level of spermatozoa from subfertile and proven fertile and its relation to standard sperm parameters. Andrologia 2018, 50, e13011. [Google Scholar] [CrossRef]
- Coppedè, F.; Stoccoro, A.; Tannorella, P.; Gallo, R.; Nicolì, V.; Migliore, L. Association of polymorphisms in genes involved in one-carbon metabolism with MTHFR methylation levels. Int. J. Mol. Sci. 2019, 20, 3754. [Google Scholar] [CrossRef] [Green Version]
- Wu, W.; Qin, Y.; Li, Z.; Dong, J.; Dai, J.; Lu, C.; Guo, X.; Zhao, Y.; Zhu, Y.; Zhang, W. Genome-wide microRNA expression profiling in idiopathic non-obstructive azoospermia: Significant up-regulation of miR-141, miR-429 and miR-7-1-3p. Hum. Reprod. 2013, 28, 1827–1836. [Google Scholar] [CrossRef] [PubMed]
- Che, Q.; Wang, W.; Duan, P.; Fang, F.; Liu, C.; Zhou, T.; Li, H.; Xiong, C.; Zhao, K. Downregulation of miR-322 promotes apoptosis of GC-2 cell by targeting Ddx3x. Reprod. Biol. Endocrinol. 2019, 17, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Radtke, A.; Dieckmann, K.P.; Grobelny, F.; Salzbrunn, A.; Oing, C.; Schulze, W.; Belge, G. Expression of miRNA—371a—3p in seminal plasma and ejaculate is associated with sperm concentration. Andrology 2019, 7, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Wang, C.; Fu, Z.; Liang, H.; Zhang, S.; Lu, M.; Sun, W.; Ye, C.; Zhang, C.-Y.; Zen, K. Systematic characterization of seminal plasma piRNAs as molecular biomarkers for male infertility. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Wu, W.; Hu, Z.; Qin, Y.; Dong, J.; Dai, J.; Lu, C.; Zhang, W.; Shen, H.; Xia, Y.; Wang, X. Seminal plasma microRNAs: Potential biomarkers for spermatogenesis status. Mol. Hum. Reprod. 2012, 18, 489–497. [Google Scholar] [CrossRef]
- Zhu, Y.; Wu, Y.; Jin, K.; Lu, H.; Liu, F.; Guo, Y.; Yan, F.; Shi, W.; Liu, Y.; Cao, X. Differential proteomic profiling in human spermatozoa that did or did not result in pregnancy via IVF and AID. Proteomics–Clin. Appl. 2013, 7, 850–858. [Google Scholar] [CrossRef]
- Zhi, E.-L.; Liang, G.-Q.; Li, P.; Chen, H.-X.; Tian, R.-H.; Xu, P.; Li, Z. Seminal plasma miR-192a: A biomarker predicting successful resolution of nonobstructive azoospermia following varicocele repair. Asian J. Androl. 2018, 20, 396. [Google Scholar]
- Schjenken, J.E.; Zhang, B.; Chan, H.Y.; Sharkey, D.J.; Fullston, T.; Robertson, S.A. mi RNA regulation of immune tolerance in early pregnancy. Am. J. Reprod. Immunol. 2016, 75, 272–280. [Google Scholar] [CrossRef]
- Rotondo, J.; Bosi, S.; Bazzan, E.; Di Domenico, M.; De Mattei, M.; Selvatici, R.; Patella, A.; Marci, R.; Tognon, M.; Martini, F. Methylenetetrahydrofolate reductase gene promoter hypermethylation in semen samples of infertile couples correlates with recurrent spontaneous abortion. Hum. Reprod. 2012, 27, 3632–3638. [Google Scholar] [CrossRef] [Green Version]
- Karaca, M.; Konac, E.; Yurteri, B.; Bozdag, G.; Sogutdelen, E.; Bilen, C. Association between methylenetetrahydrofolate reductase (MTHFR) gene promoter hypermethylation and the risk of idiopathic male infertility. Andrologia 2017, 49, e12698. [Google Scholar] [CrossRef]
- Botezatu, A.; Socolov, R.; Socolov, D.; Iancu, I.V.; Anton, G. Methylation pattern of methylene tetrahydrofolate reductase and small nuclear ribonucleoprotein polypeptide N promoters in oligoasthenospermia: A case-control study. Reprod. Biomed. Online 2014, 28, 225–231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ly, L.; Chan, D.; Trasler, J.M. Developmental windows of susceptibility for epigenetic inheritance through the male germline. Semin. Cell Dev. Biol. 2015, 43, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.P.; Jenkins, T.G.; Aston, K.I.; Guo, J.; Pastuszak, A.W.; Hanson, H.A.; Hotaling, J.M. Harnessing the full potential of reproductive genetics and epigenetics for male infertility in the era of “big data”. Fertil. Steril. 2020, 113, 478–488. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, S.; Sujit, K.M.; Singh, V.; Pandey, R.; Trivedi, S.; Singh, K.; Gupta, G.; Rajender, S. Array-based DNA methylation profiling reveals peripheral blood differential methylation in male infertility. Fertil. Steril. 2019, 112, 61–72.e61. [Google Scholar] [CrossRef]
- Candenas, L.; Chianese, R. Exosome composition and seminal plasma proteome: A promising source of biomarkers of male infertility. Int. J. Mol. Sci. 2020, 21, 7022. [Google Scholar] [CrossRef]
- Panner Selvam, M.K.; Finelli, R.; Agarwal, A.; Henkel, R. Proteomics and metabolomics—Current and future perspectives in clinical andrology. Andrologia 2021, 53, e13711. [Google Scholar] [CrossRef]
- Liu, X.; Wang, W.; Zhu, P.; Wang, J.; Wang, Y.; Wang, X.; Liu, J.; Li, N.; Wang, X.; Lin, C. In-depth quantitative proteome analysis of seminal plasma from men with oligoasthenozoospermia and normozoospermia. Reprod. Biomed. Online 2018, 37, 467–479. [Google Scholar] [CrossRef]
- Panner Selvam, M.K.; Agarwal, A.; Pushparaj, P.N.; Baskaran, S.; Bendou, H. Sperm proteome analysis and identification of fertility-associated biomarkers in unexplained male infertility. Genes 2019, 10, 522. [Google Scholar] [CrossRef] [Green Version]
- Kanannejad, Z.; Gharesi—Fard, B. Difference in the seminal plasma protein expression in unexplained infertile men with successful and unsuccessful in vitro fertilisation outcome. Andrologia 2019, 51, e13158. [Google Scholar] [CrossRef]
- Salehi, M.; Akbari, H.; Heidari, M.H.; Molouki, A.; Murulitharan, K.; Moeini, H.; Novin, M.G.; Aabed, F.; Taheri, H.; Fadaei, F. Correlation between human clusterin in seminal plasma with sperm protamine deficiency and DNA fragmentation. Mol. Reprod. Dev. 2013, 80, 718–724. [Google Scholar] [CrossRef]
- Longo, V.; Forleo, A.; Provenzano, S.P.; Coppola, L.; Zara, V.; Ferramosca, A.; Siciliano, P.; Capone, S. HS-SPME-GC-MS metabolomics approach for sperm quality evaluation by semen volatile organic compounds (VOCs) analysis. Biomed. Phys. Eng. Express 2018, 5, 015006. [Google Scholar] [CrossRef]
- Murgia, F.; Corda, V.; Serrenti, M.; Usai, V.; Santoru, M.L.; Hurt, K.J.; Passaretti, M.; Monni, M.C.; Atzori, L.; Monni, G. Seminal Fluid Metabolomic Markers of Oligozoospermic Infertility in Humans. Metabolites 2020, 10, 64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, Q.; Liu, L.; Wu, Y.; Wang, X.; Luo, L.; Nan, B.; Zhang, J.; Tian, M.; Shen, H. Seminal plasma metabolites mediate the associations of multiple environmental pollutants with semen quality in Chinese men. Environ. Int. 2019, 132, 105066. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Zhang, J.; Xu, Z.; Xu, Y.; Xu, A.; Chen, W.; Miao, C.; Liu, S.; Wang, Z.; Jia, R. Metabolomic profiling of human spermatozoa in idiopathic asthenozoospermia patients using gas chromatography-mass spectrometry. BioMed Res. Int. 2018, 2018, 8327506. [Google Scholar] [CrossRef] [PubMed]
- Engel, K.M.; Baumann, S.; Rolle-Kampczyk, U.; Schiller, J.; von Bergen, M.; Grunewald, S. Metabolomic profiling reveals correlations between spermiogram parameters and the metabolites present in human spermatozoa and seminal plasma. PLoS ONE 2019, 14, e0211679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiao, S.; Wu, W.; Chen, M.; Tang, Q.; Xia, Y.; Jia, W.; Wang, X. Seminal plasma metabolomics approach for the diagnosis of unexplained male infertility. PLoS ONE 2017, 12, e0181115. [Google Scholar] [CrossRef] [Green Version]
- Tournaye, H. Male factor infertility and ART. Asian J. Androl. 2012, 14, 103–108. [Google Scholar] [CrossRef] [Green Version]
- Chambers, G.M.; Sullivan, E.A.; Ishihara, O.; Chapman, M.G.; Adamson, G.D. The economic impact of assisted reproductive technology: A review of selected developed countries. Fertil. Steril. 2009, 91, 2281–2294. [Google Scholar] [CrossRef]
- Tournaye, H.; Krausz, C.; Oates, R.D. Concepts in diagnosis and therapy for male reproductive impairment. Lancet Diabetes Endocrinol. 2017, 5, 554–564. [Google Scholar] [CrossRef]
- Inhorn, M.C.; Patrizio, P. Infertility around the globe: New thinking on gender, reproductive technologies and global movements in the 21st century. Hum. Reprod. Update 2015, 21, 411–426. [Google Scholar] [CrossRef] [Green Version]
- Shevell, T.; Malone, F.D.; Vidaver, J.; Porter, T.F.; Luthy, D.A.; Comstock, C.H.; Hankins, G.D.; Eddleman, K.; Dolan, S.; Dugoff, L. Assisted reproductive technology and pregnancy outcome. Obstet. Gynecol. 2005, 106, 1039–1045. [Google Scholar] [CrossRef] [PubMed]
- Schieve, L.A.; Meikle, S.F.; Ferre, C.; Peterson, H.B.; Jeng, G.; Wilcox, L.S. Low and very low birth weight in infants conceived with use of assisted reproductive technology. N. Engl. J. Med. 2002, 346, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.; Kurinczuk, J.J.; Bower, C.; Webb, S. The risk of major birth defects after intracytoplasmic sperm injection and in vitro fertilization. N. Engl. J. Med. 2002, 346, 725–730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Corona, G.; Minhas, S.; Giwercman, A.; Bettocchi, C.; Dinkelman-Smit, M.; Dohle, G.; Fusco, F.; Kadioglou, A.; Kliesch, S.; Kopa, Z. Sperm recovery and ICSI outcomes in men with non-obstructive azoospermia: A systematic review and meta-analysis. Hum. Reprod. Update 2019, 25, 733–757. [Google Scholar] [CrossRef] [PubMed]
- Eliveld, J.; van Wely, M.; Meißner, A.; Repping, S.; van der Veen, F.; van Pelt, A.M. The risk of TESE-induced hypogonadism: A systematic review and meta-analysis. Hum. Reprod. Update 2018, 24, 442–454. [Google Scholar] [CrossRef]
- Doudt, A.D.; Christman, M.S. Management of Pediatric and Adolescent Varicocele. In Varicocele and Male Infertility; Springer: Berlin/Heidelberg, Germany, 2019; pp. 343–355. [Google Scholar]
- Yao, A.; Ingargiola, M.; Lopez, C.; Sanati-Mehrizy, P.; Burish, N.; Jablonka, E.; Taub, P. Total penile reconstruction: A systematic review. J. Plast. Reconstr. Aesthetic Surg. 2018, 71, 788–806. [Google Scholar] [CrossRef]
- LeQuang, J.A. Updates on Psoriasis and Cutaneous Oncology: Proceedings from the 2017 MauiDerm Meeting. J. Clin. Aesthetic Dermatol. 2017, 10, S8. [Google Scholar]
- Geerkens, M.J.; Al-Itejawi, H.H.; Nieuwenhuijzen, J.A.; Meuleman, E.J.; Lissenberg-Witte, B.I.; van Moorselaar, R.; Vis, A.N. Sexual dysfunction and bother due to erectile dysfunction in the healthy elderly male population: Prevalence from a systematic review. Eur. Urol. Focus 2020, 6, 776–790. [Google Scholar] [CrossRef]
- Parnham, A.S.; Albersen, M.; Sahdev, V.; Christodoulidou, M.; Nigam, R.; Malone, P.; Freeman, A.; Muneer, A. Glansectomy and split-thickness skin graft for penile cancer. Eur. Urol. 2018, 73, 284–289. [Google Scholar] [CrossRef]
- Mannelli, G.; Gazzini, L.; Comini, L.V.; Parrinello, G.; Nocini, R.; Marchioni, D.; Molteni, G. Double free flaps in oral cavity and oropharynx reconstruction: Systematic review, indications and limits. Oral Oncol. 2020, 104, 104637. [Google Scholar] [CrossRef]
- De Leo, V.; Tosti, C.; Morgante, G.; Ponchia, R.; Luddi, A.; Governini, L.; Piomboni, P. Positive Effect of a New Combination of Antioxidants and Natural Hormone Stimulants for the Treatment of Oligoasthenoteratozoospermia. J. Clin. Med. 2022, 11, 1991. [Google Scholar] [CrossRef] [PubMed]
- Younus, H. Therapeutic potentials of superoxide dismutase. Int. J. Health Sci. 2018, 12, 88. [Google Scholar]
- Torres-Arce, E.; Vizmanos, B.; Babio, N.; Márquez-Sandoval, F.; Salas-Huetos, A. Dietary Antioxidants in the Treatment of Male Infertility: Counteracting Oxidative Stress. Biology 2021, 10, 241. [Google Scholar] [CrossRef] [PubMed]
- Makarova, N.; Romanov, Y.A.; Dolgushina, N.; Parker, M.; Krasnyi, A. Comparative analysis of the expression of glutathione peroxidase and glutathione reductase genes in human sperm after cryopreservation. Bull. Exp. Biol. Med. 2018, 165, 166–170. [Google Scholar] [CrossRef]
- Governini, L.; Ponchia, R.; Artini, P.G.; Casarosa, E.; Marzi, I.; Capaldo, A.; Luddi, A.; Piomboni, P. Respiratory Mitochondrial Efficiency and DNA Oxidation in Human Sperm after In Vitro Myo-Inositol Treatment. J. Clin. Med. 2020, 9, 1638. [Google Scholar] [CrossRef]
- Condorelli, R.A.; Barbagallo, F.; Calogero, A.E.; Cannarella, R.; Crafa, A.; La Vignera, S. D-chiro-inositol improves sperm mitochondrial membrane potential: In vitro evidence. J. Clin. Med. 2020, 9, 1373. [Google Scholar] [CrossRef]
- Vishvkarma, R.; Alahmar, A.T.; Gupta, G.; Rajender, S. Coenzyme Q10 effect on semen parameters: Profound or meagre? Andrologia 2020, 52, e13570. [Google Scholar] [CrossRef]
- Nouri, M.; Amani, R.; Nasr—Esfahani, M.; Tarrahi, M.J. The effects of lycopene supplement on the spermatogram and seminal oxidative stress in infertile men: A randomized, double—blind, placebo—controlled clinical trial. Phytother. Res. 2019, 33, 3203–3211. [Google Scholar] [CrossRef]
- Balercia, G.; Buldreghini, E.; Vignini, A.; Tiano, L.; Paggi, F.; Amoroso, S.; Ricciardo-Lamonica, G.; Boscaro, M.; Lenzi, A.; Littarru, G. Coenzyme Q10 treatment in infertile men with idiopathic asthenozoospermia: A placebo-controlled, double-blind randomized trial. Fertil. Steril. 2009, 91, 1785–1792. [Google Scholar] [CrossRef]
- Nadjarzadeh, A.; Sadeghi, M.; Amirjannati, N.; Vafa, M.; Motevalian, S.; Gohari, M.; Akhondi, M.; Yavari, P.; Shidfar, F. Coenzyme Q 10 improves seminal oxidative defense but does not affect on semen parameters in idiopathic oligoasthenoteratozoospermia: A randomized double-blind, placebo controlled trial. J. Endocrinol. Investig. 2011, 34, e224–e228. [Google Scholar]
- Moradi, M.; Moradi, A.; Alemi, M.; Ahmadnia, H.; Abdi, H.; Ahmadi, A.; Bazargan-Hejazi, S. Safety and efficacy of clomiphene citrate and L-carnitine in idiopathic male infertility: A comparative study. Urol. J. 2010, 7, 188. [Google Scholar] [PubMed]
- Ahmed, S.H.; Ahsan, S.; Iqbal, T.; Burney, S.A. Relationship of seminal free L-Carnitine with functional spermatozoal characteristics: Results from an observational study conducted in a tertiary care hospital of Karachi, Pakistan. J. Pak. Med. Assoc. 2017, 67, 280–284. [Google Scholar]
- Sheikh, N.; Goodarzi, M.; Bab Al-Havaejee, H.; Safari, M.; Amiri, I.; Najafi, R.; Hadeie, J. L-carnitine level in seminal plasma of fertile and infertile men. J. Res. Health Sci. 2007, 7, 43–48. [Google Scholar]
- Filipcikova, R.; Oborna, I.; Brezinova, J.; Novotny, J.; Wojewodka, G.; De Sanctis, J.B.; Radova, L.; Hajduch, M.; Radzioch, D. Lycopene improves the distorted ratio between AA/DHA in the seminal plasma of infertile males and increases the likelihood of successful pregnancy. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc. Czech Repub. 2015, 159, 77–82. [Google Scholar] [CrossRef] [Green Version]
- Oborna, I.; Malickova, K.; Fingerova, H.; Brezinova, J.; Horka, P.; Novotny, J.; Bryndova, H.; Filipcikova, R.; Svobodova, M. A randomized controlled trial of lycopene treatment on soluble receptor for advanced glycation end products in seminal and blood plasma of normospermic men. Am. J. Reprod. Immunol. 2011, 66, 179–184. [Google Scholar] [CrossRef]
- Ciftci, H.; Verit, A.; Savas, M.; Yeni, E.; Erel, O. Effects of N-acetylcysteine on semen parameters and oxidative/antioxidant status. Urology 2009, 74, 73–76. [Google Scholar] [CrossRef]
- Jannatifar, R.; Parivar, K.; Roodbari, N.H.; Nasr-Esfahani, M.H. Effects of N-acetyl-cysteine supplementation on sperm quality, chromatin integrity and level of oxidative stress in infertile men. Reprod. Biol. Endocrinol. 2019, 17, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jannatifar, R.; Parivar, K.; Roodbari, N.H.; Nasr-Esfahani, M.H. The effect of N-acetyl-cysteine on NRF2 antioxidant gene expression in asthenoteratozoospermia men: A clinical trial study. Int. J. Fertil. Steril. 2020, 14, 171. [Google Scholar]
- Malmir, M.; Naderi Noreini, S.; Ghafarizadeh, A.; Faraji, T.; Asali, Z. Ameliorative effect of melatonin on apoptosis, DNA fragmentation, membrane integrity and lipid peroxidation of spermatozoa in the idiopathic asthenoteratospermic men: In vitro. Andrologia 2021, 53, e13944. [Google Scholar] [CrossRef]
- Awad, H.; Halawa, F.; Mostafa, T.; Atta, H. Melatonin hormone profile in infertile males. Int. J. Androl. 2006, 29, 409–413. [Google Scholar] [CrossRef]
- Hassan, M.H.; El-Taieb, M.A.; Fares, N.N.; Fayed, H.M.; Toghan, R.; Ibrahim, H.M. Men with idiopathic oligoasthenoteratozoospermia exhibit lower serum and seminal plasma melatonin levels: Comparative effect of night-light exposure with fertile males. Exp. Ther. Med. 2020, 20, 235–242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ortiz, A.; Espino, J.; Bejarano, I.; Lozano, G.; Monllor, F.; García, J.; Pariente, J.; Rodríguez, A. High endogenous melatonin concentrations enhance sperm quality and short—term in vitro exposure to melatonin improves aspects of sperm motility. J. Pineal Res. 2011, 50, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Espino, J.; Bejarano, I.; Ortiz, Á.; Lozano, G.M.; García, J.F.; Pariente, J.A.; Rodríguez, A.B. Melatonin as a potential tool against oxidative damage and apoptosis in ejaculated human spermatozoa. Fertil. Steril. 2010, 94, 1915–1917. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, B.; Molavi, N.; Tavalaee, M.; Abbasi, H.; Nasr-Esfahani, M.H. Alpha-lipoic acid improves sperm motility in infertile men after varicocelectomy: A triple-blind randomized controlled trial. Reprod. Biomed. Online 2020, 41, 1084–1091. [Google Scholar] [CrossRef] [PubMed]
- Haghighian, H.K.; Haidari, F.; Mohammadi-Asl, J.; Dadfar, M. Randomized, triple-blind, placebo-controlled clinical trial examining the effects of alpha-lipoic acid supplement on the spermatogram and seminal oxidative stress in infertile men. Fertil. Steril. 2015, 104, 318–324. [Google Scholar] [CrossRef]
- Taherian, S.s.; Khayamabed, R.; Tavalaee, M.; Nasr—Esfahani, M.H. Alpha—lipoic acid minimises reactive oxygen species—induced damages during sperm processing. Andrologia 2019, 51, e13314. [Google Scholar] [CrossRef]
- Asa, E.; Ahmadi, R.; Mahmoodi, M.; Mohammadniya, A. Supplementation of freezing media with alpha lipoic acid preserves the structural and functional characteristics of sperm against cryodamage in infertile men with asthenoteratozoospermia. Cryobiology 2020, 96, 166–174. [Google Scholar] [CrossRef]
- Safarinejad, M. Effect of omega—3 polyunsaturated fatty acid supplementation on semen profile and enzymatic anti—oxidant capacity of seminal plasma in infertile men with idiopathic oligoasthenoteratospermia: A double—blind, placebo—controlled, randomised study. Andrologia 2011, 43, 38–47. [Google Scholar] [CrossRef]
- Jensen, T.K.; Priskorn, L.; Holmboe, S.A.; Nassan, F.L.; Andersson, A.-M.; Dalgård, C.; Petersen, J.H.; Chavarro, J.E.; Jørgensen, N. Associations of fish oil supplement use with testicular function in young men. JAMA Netw. Open 2020, 3, e1919462. [Google Scholar] [CrossRef]
- Martínez-Soto, J.C.; Domingo, J.C.; Cordobilla, B.; Nicolás, M.; Fernández, L.; Albero, P.; Gadea, J.; Landeras, J. Dietary supplementation with docosahexaenoic acid (DHA) improves seminal antioxidant status and decreases sperm DNA fragmentation. Syst. Biol. Reprod. Med. 2016, 62, 387–395. [Google Scholar] [CrossRef] [Green Version]
- Ghafarizadeh, A.; Malmir, M.; Naderi Noreini, S.; Faraji, T. Antioxidant effects of N—acetylcysteine on the male reproductive system: A systematic review. Andrologia 2021, 53, e13898. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.-C.; Li, H.-Y.; Li, X.-Y.; Yu, K.; Deng, S.-L.; Tian, L. Protective effects of melatonin on male fertility preservation and reproductive system. Cryobiology 2020, 95, 1–8. [Google Scholar] [CrossRef]
- Di Tucci, C.; Galati, G.; Mattei, G.; Bonanni, V.; Capri, O.; D’Amelio, R.; Muzii, L.; Benedetti Panici, P. The role of alpha lipoic acid in female and male infertility: A systematic review. Gynecol. Endocrinol. 2021, 37, 497–505. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, B.; Nourmohamadi, M.; Hajipour, S.; Taghizadeh, M.; Asemi, Z.; Keshavarz, S.A.; Jafarnejad, S. The effect of omega-3 fatty acids, EPA, and/or DHA on male infertility: A systematic review and meta-analysis. J. Diet. Suppl. 2019, 16, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Angulo, C.; Maldonado, R.; Pulgar, E.; Mancilla, H.; Córdova, A.; Villarroel, F.; Castro, M.A.; Concha, I.I. Vitamin C and oxidative stress in the seminiferous epithelium. Biol. Res. 2011, 44, 169–180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, M.-C.; Chiu, Y.-H.; Gaskins, A.J.; Mínguez-Alarcón, L.; Nassan, F.L.; Williams, P.L.; Petrozza, J.; Hauser, R.; Chavarro, J.E. Men’s Intake of Vitamin C and β-Carotene Is Positively Related to Fertilization Rate but Not to Live Birth Rate in Couples Undergoing Infertility Treatment. J. Nutr. 2019, 149, 1977–1984. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, T.; Burdeos, G.C.; Itaya, M.; Nakagawa, K.; Miyazawa, T. Vitamin E: Regulatory redox interactions. IUBMB Life 2019, 71, 430–441. [Google Scholar] [CrossRef]
- Irani, M.; Amirian, M.; Sadeghi, R.; Le Lez, J.; Roudsari, R.L. The effect of folate and folate plus zinc supplementation on endocrine parameters and sperm characteristics in sub-fertile men: A systematic review and meta-analysis. Urol. J. 2017, 14, 4069–4078. [Google Scholar]
- Rafiee, B.; Morowvat, M.H.; Rahimi-Ghalati, N. Comparing the effectiveness of dietary vitamin C and exercise interventions on fertility parameters in normal obese men. Urol. J. 2016, 13, 2635–2639. [Google Scholar]
- Suleiman, S.A.; Ali, M.E.; Zaki, Z.; El—Malik, E.; Nasr, M. Lipid peroxidation and human sperm motility: Protective role of vitamin E. J. Androl. 1996, 17, 530–537. [Google Scholar]
- Wong, W.Y.; Merkus, H.M.; Thomas, C.M.; Menkveld, R.; Zielhuis, G.A.; Steegers-Theunissen, R.P. Effects of folic acid and zinc sulfate on male factor subfertility: A double-blind, randomized, placebo-controlled trial. Fertil. Steril. 2002, 77, 491–498. [Google Scholar] [CrossRef]
- Raigani, M.; Yaghmaei, B.; Amirjannti, N.; Lakpour, N.; Akhondi, M.; Zeraati, H.; Hajihosseinal, M.; Sadeghi, M. The micronutrient supplements, zinc sulphate and folic acid, did not ameliorate sperm functional parameters in oligoasthenoteratozoospermic men. Andrologia 2014, 46, 956–962. [Google Scholar] [CrossRef] [PubMed]
- Colagar, A.H.; Marzony, E.T.; Chaichi, M.J. Zinc levels in seminal plasma are associated with sperm quality in fertile and infertile men. Nutr. Res. 2009, 29, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Alahmar, A.T.; Sengupta, P. Impact of coenzyme Q10 and selenium on seminal fluid parameters and antioxidant status in men with idiopathic infertility. Biol. Trace Elem. Res. 2021, 199, 1246–1252. [Google Scholar] [CrossRef]
- Safarinejad, M.R.; Safarinejad, S. Efficacy of selenium and/or N-acetyl-cysteine for improving semen parameters in infertile men: A double-blind, placebo controlled, randomized study. J. Urol. 2009, 181, 741–751. [Google Scholar] [CrossRef]
- de Castro Pacheco, L.M.S.; da Cunha, M.Y.P.T.; Daltro, A.F.C.S.; Torquato Filho, S.E.; de Assis, R.C.; Celedonio, R.F.; Pires, L.V.; Maia, C.S.C.; Guedes, M.I.F. Unbalance of Se and nutritional status in male infertility. JBRA Assist. Reprod. 2021, 25, 202. [Google Scholar]
- Liu, P.; Yuan, G.; Zhou, Q.; Liu, Y.; He, X.; Zhang, H.; Guo, Y.; Wen, Y.; Huang, S.; Ke, Y. The association between metal exposure and semen quality in Chinese males: The mediating effect of androgens. Environ. Pollut. 2020, 264, 113975. [Google Scholar] [CrossRef]
- Wu, S.; Wang, M.; Deng, Y.; Qiu, J.; Zhang, X.; Tan, J. Associations of toxic and essential trace elements in serum, follicular fluid, and seminal plasma with In vitro fertilization outcomes. Ecotoxicol. Environ. Saf. 2020, 204, 110965. [Google Scholar] [CrossRef]
- Kerns, K.; Zigo, M.; Sutovsky, P. Zinc: A necessary ion for mammalian sperm fertilization competency. Int. J. Mol. Sci. 2018, 19, 4097. [Google Scholar] [CrossRef] [Green Version]
- Salas-Huetos, A.; Rosique-Esteban, N.; Becerra-Tomás, N.; Vizmanos, B.; Bulló, M.; Salas-Salvadó, J. The effect of nutrients and dietary supplements on sperm quality parameters: A systematic review and meta-analysis of randomized clinical trials. Adv. Nutr. 2018, 9, 833–848. [Google Scholar] [CrossRef]
- Zhao, J.; Dong, X.; Hu, X.; Long, Z.; Wang, L.; Liu, Q.; Sun, B.; Wang, Q.; Wu, Q.; Li, L. Zinc levels in seminal plasma and their correlation with male infertility: A systematic review and meta-analysis. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Majzoub, A.; Agarwal, A. Systematic review of antioxidant types and doses in male infertility: Benefits on semen parameters, advanced sperm function, assisted reproduction and live-birth rate. Arab J. Urol. 2018, 16, 113–124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, P.; Kaur, P.; Ghanghas, P.; Kaur, J.; Kaushal, N. Selenium Ameliorates Ibuprofen Induced Testicular Toxicity by Redox Regulation: Running Head: Se protects against NSAID induced testicular toxicity. Reprod. Toxicol. 2020, 96, 349–358. [Google Scholar] [CrossRef]
- Chyra-Jach, D.; Kaletka, Z.; Dobrakowski, M.; Machoń-Grecka, A.; Kasperczyk, S.; Bellanti, F.; Birkner, E.; Kasperczyk, A. Levels of macro-and trace elements and select cytokines in the semen of infertile men. Biol. Trace Elem. Res. 2020, 197, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Khourdaji, I.; Lee, H.; Smith, R.P. Frontiers in hormone therapy for male infertility. Transl. Androl. Urol. 2018, 7, S353. [Google Scholar] [CrossRef] [PubMed]
- Attia, A.M.; Abou-Setta, A.M.; Al-Inany, H.G. Gonadotrophins for idiopathic male factor subfertility. Cochrane Database Syst. Rev. 2013, 8, CD005071. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jungwirth, A.; Giwercman, A.; Tournaye, H.; Diemer, T.; Kopa, Z.; Dohle, G.; Krausz, C.; Infertility, E.W.G.o.M. European Association of Urology guidelines on Male Infertility: The 2012 update. Eur. Urol. 2012, 62, 324–332. [Google Scholar] [CrossRef]
- Okwuosa, T.M.; Morgans, A.; Rhee, J.-W.; Reding, K.W.; Maliski, S.; Plana, J.-C.; Volgman, A.S.; Moseley, K.F.; Porter, C.B.; Ismail-Khan, R. Impact of hormonal therapies for treatment of hormone-dependent cancers (Breast and Prostate) on the cardiovascular system: Effects and modifications: A scientific statement from the American Heart Association. Circ. Genom. Precis. Med. 2021, 14, e000082. [Google Scholar] [CrossRef]
- Vander Borght, M.; Wyns, C. Fertility and infertility: Definition and epidemiology. Clin. Biochem. 2018, 62, 2–10. [Google Scholar] [CrossRef]
- Gnoth, C.; Godehardt, E.; Frank-Herrmann, P.; Friol, K.; Tigges, J.; Freundl, G. Definition and prevalence of subfertility and infertility. Hum. Reprod. 2005, 20, 1144–1147. [Google Scholar] [CrossRef] [Green Version]
- Wei, Y.; Chen, Q.; Qian, W. Effect of bariatric surgery on semen parameters: A systematic review and meta-analysis. Med. Sci. Monit. Basic Res. 2018, 24, 188. [Google Scholar] [CrossRef] [PubMed]
- Ibañez-Perez, J.; Santos-Zorrozua, B.; Lopez-Lopez, E.; Matorras, R.; Garcia-Orad, A. An update on the implication of physical activity on semen quality: A systematic review and meta-analysis. Arch. Gynecol. Obstet. 2019, 299, 901–921. [Google Scholar] [CrossRef] [PubMed]
- Liese, A.D.; Roach, A.K.; Sparks, K.C.; Marquart, L.; D’Agostino Jr, R.B.; Mayer-Davis, E.J. Whole-grain intake and insulin sensitivity: The Insulin Resistance Atherosclerosis Study. Am. J. Clin. Nutr. 2003, 78, 965–971. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mínguez—Alarcón, L.; Afeiche, M.C.; Chiu, Y.H.; Vanegas, J.C.; Williams, P.L.; Tanrikut, C.; Toth, T.L.; Hauser, R.; Chavarro, J.E. Male soy food intake was not associated with in vitro fertilization outcomes among couples attending a fertility center. Andrology 2015, 3, 702–708. [Google Scholar] [CrossRef]
- Lyngsø, J.; Ramlau-Hansen, C.H.; Bay, B.; Ingerslev, H.J.; Hulman, A.; Kesmodel, U.S. Association between coffee or caffeine consumption and fecundity and fertility: A systematic review and dose–response meta-analysis. Clin. Epidemiol. 2017, 9, 699. [Google Scholar] [CrossRef] [Green Version]
- Salas-Huetos, A.; Moraleda, R.; Giardina, S.; Anton, E.; Blanco, J.; Salas-Salvadó, J.; Bulló, M. Effect of nut consumption on semen quality and functionality in healthy men consuming a Western-style diet: A randomized controlled trial. Am. J. Clin. Nutr. 2018, 108, 953–962. [Google Scholar] [CrossRef] [Green Version]
- Seymen, C.M. The other side of COVID—19 pandemic: Effects on male fertility. J. Med. Virol. 2021, 93, 1396–1402. [Google Scholar] [CrossRef]
- Aydın, T.; Karadağ, M.A.; Demir, A.; Çeçen, K.; Karasu, Y.; Ülker, K. Effect of modification of lifestyle on reproductive potential. Kafkas J. Med. Sci. 2014, 4, 27–35. [Google Scholar] [CrossRef]
- Lalinde-Acevedo, P.C.; Mayorga-Torres, B.J.M.; Agarwal, A.; Du Plessis, S.S.; Ahmad, G.; Cadavid, Á.P.; Maya, W.D.C. Physically active men show better semen parameters than their sedentary counterparts. Int. J. Fertil. Steril. 2017, 11, 156. [Google Scholar]
- Silvestris, E.; Lovero, D.; Palmirotta, R. Nutrition and female fertility: An interdependent correlation. Front. Endocrinol. 2019, 10, 346. [Google Scholar] [CrossRef] [Green Version]
- Wise, L.A.; Rothman, K.J.; Mikkelsen, E.M.; Sørensen, H.T.; Riis, A.H.; Hatch, E.E. A prospective cohort study of physical activity and time to pregnancy. Fertil. Steril. 2012, 97, 1136–1142.e4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hajizadeh Maleki, B.; Tartibian, B.; Chehrazi, M. Effectiveness of Exercise Training on Male Factor Infertility: A Systematic Review and Network Meta-analysis. Sports Health 2021, 14, 19417381211055399. [Google Scholar] [CrossRef] [PubMed]
- Maleki, B.H.; Tartibian, B. High-intensity interval training modulates male factor infertility through anti-inflammatory and antioxidative mechanisms in infertile men: A randomized controlled trial. Cytokine 2020, 125, 154861. [Google Scholar] [CrossRef] [PubMed]
- Maleki, B.H.; Tartibian, B. Resistance exercise modulates male factor infertility through anti-inflammatory and antioxidative mechanisms in infertile men: A RCT. Life Sci. 2018, 203, 150–160. [Google Scholar] [CrossRef]
- Wise, L.A.; Rothman, K.J.; Wesselink, A.K.; Mikkelsen, E.M.; Sorensen, H.T.; McKinnon, C.J.; Hatch, E.E. Male sleep duration and fecundability in a North American preconception cohort study. Fertil. Steril. 2018, 109, 453–459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goldstein, C.A.; Smith, Y.R. Sleep, circadian rhythms, and fertility. Curr. Sleep Med. Rep. 2016, 2, 206–217. [Google Scholar] [CrossRef] [Green Version]
- Baker, F.C.; Sassoon, S.A.; Kahan, T.; Palaniappan, L.; Nicholas, C.L.; Trinder, J.; Colrain, I.M. Perceived poor sleep quality in the absence of polysomnographic sleep disturbance in women with severe premenstrual syndrome. J. Sleep Res. 2012, 21, 535–545. [Google Scholar] [CrossRef] [Green Version]
- Sharkey, K.M.; Crawford, S.L.; Kim, S.; Joffe, H. Objective sleep interruption and reproductive hormone dynamics in the menstrual cycle. Sleep Med. 2014, 15, 688–693. [Google Scholar] [CrossRef] [Green Version]
- Dada, R.; Tolahunase, M. Yoga Meditation Lifestyle Intervention: Impact on Male Reproductive Health. In Bioenvironmental Issues Affecting Men’s Reproductive and Sexual Health; Elsevier: Amsterdam, The Netherlands, 2018; pp. 135–156. [Google Scholar]
- Patel, A.; Sharma, P.; Kumar, P. Application of mindfulness-based psychological interventions in infertility. J. Hum. Reprod. Sci. 2020, 13, 3. [Google Scholar] [CrossRef]
- Hosseini, M.S.; Mousavi, P.; Hekmat, K.; Haghighyzadeh, M.H.; Fard, R.J.; Jafari, R.M. Effects of a short-term mindfulness-based stress reduction program on the quality of life of women with infertility: A randomized controlled clinical trial. Complementary Ther. Med. 2020, 50, 102403. [Google Scholar] [CrossRef]
- Tolahunase, M.; Sagar, R.; Chaurasia, P.; Dada, R. Impact of yoga-and meditation-based lifestyle intervention on depression, quality of life, and cellular aging in infertile couples. Fertil. Steril. 2018, 110, e67. [Google Scholar] [CrossRef]
- Wang, M.; Liu, X.; Chang, G.; Chen, Y.; An, G.; Yan, L.; Gao, S.; Xu, Y.; Cui, Y.; Dong, J. Single-cell RNA sequencing analysis reveals sequential cell fate transition during human spermatogenesis. Cell Stem Cell 2018, 23, 599–614.e594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- La, H.; Yoo, H.; Lee, E.J.; Thang, N.X.; Choi, H.J.; Oh, J.; Park, J.H.; Hong, K. Insights from the Applications of Single-Cell Transcriptomic Analysis in Germ Cell Development and Reproductive Medicine. Int. J. Mol. Sci. 2021, 22, 823. [Google Scholar] [CrossRef] [PubMed]
- Hajiesmailpoor, A.; Emami, P.; Kondori, B.J.; Ghorbani, M. Stem cell therapy as a recent advanced approach in male infertility. Tissue Cell 2021, 73, 101634. [Google Scholar] [CrossRef]
- Punjani, N.; Kang, C.; Lee, R.K.; Goldstein, M.; Li, P.S. Technological Advancements in Male Infertility Microsurgery. J. Clin. Med. 2021, 10, 4259. [Google Scholar] [CrossRef]
- Darves-Bornoz, A.; Panken, E.; Brannigan, R.E.; Halpern, J.A. Robotic Surgery for Male Infertility. Urol. Clin. 2021, 48, 127–135. [Google Scholar] [CrossRef]
- Chang, T.C.; Seufert, C.; Eminaga, O.; Shkolyar, E.; Hu, J.C.; Liao, J.C. Current trends in artificial intelligence application for endourology and robotic surgery. Urol. Clin. 2021, 48, 151–160. [Google Scholar] [CrossRef]
- Hameed, B.; Dhavileswarapu, S.; Aiswarya, V.; Raza, S.Z.; Karimi, H.; Khanuja, H.S.; Shetty, D.K.; Ibrahim, S.; Shah, M.J.; Naik, N. Artificial intelligence and its impact on urological diseases and management: A comprehensive review of the literature. J. Clin. Med. 2021, 10, 1864. [Google Scholar] [CrossRef]
- Krassowski, M.; Das, V.; Sahu, S.K.; Misra, B.B. State of the Field in Multi-Omics Research: From Computational Needs to Data Mining and Sharing. Front. Genet. 2020, 11. [Google Scholar] [CrossRef]
- Alahmar, A.T. Role of oxidative stress in male infertility: An updated review. J. Hum. Reprod. Sci. 2019, 12, 4. [Google Scholar] [CrossRef]
- Irez, T.; Bicer, S.; Sahin, E.; Dutta, S.; Sengupta, P. Cytokines and adipokines in the regulation of spermatogenesis and semen quality. Chem. Biol. Lett. 2020, 7, 131–139. [Google Scholar]
- Haidl, G.; Haidl, F.; Allam, J.P.; Schuppe, H.C. Therapeutic options in male genital tract inflammation. Andrologia 2019, 51, e13207. [Google Scholar] [CrossRef] [PubMed]
- Unemo, M.; Seifert, H.S.; Hook, E.W.; Hawkes, S.; Ndowa, F.; Dillon, J.-A.R. Gonorrhoea. Nat. Rev. Dis. Primers 2019, 5, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Elieh Ali Komi, D.; Shafaghat, F.; Haidl, G. Significance of mast cells in spermatogenesis, implantation, pregnancy, and abortion: Cross talk and molecular mechanisms. Am. J. Reprod. Immunol. 2020, 83, e13228. [Google Scholar] [CrossRef] [Green Version]
- De Rose, A.; Baldi, M.; Gallo, F.; Rossi, P.; Gattuccio, I.; Marino, A.; Canepa, P.; Mantica, G.; Paraboschi, I.; Gattuccio, F. The management of male infertility: From nutraceuticals to diagnostics. Int. J. Med. Device Adjuv. Treat. 2018, 1, e110. [Google Scholar]
| Nutritional Factors | Findings | References |
|---|---|---|
| Dietary patterns | Unhealthy diets (western diet) increased the risk of infertility. | [31,32] |
| Dietary fats | High-fat dairy products may increase the risk of infertility. Trans fatty acids may increase the risk of metabolic disorders that negatively affect ovarian function. | [33,34,35] |
| Vegetables and fruits | Although vegetables and fruits were associated with improved semen quality and fertility related to antioxidants, folic acid, fiber, and minerals, pesticide residues may modify the beneficial effects. | [35,36,37,38,39,40] |
| Beverages | High intake of sugary beverages increased infertility risk. |
| Test | Purpose | Method | Principle | Result |
|---|---|---|---|---|
| TUNEL | To identify single- and double-strand DNA breaks | Fluorescence-labeled nucleotides are added to the site of DNA fragmentation | Quantifies the enzymatic incorporation of dUTP in DNA breaks | Sperm with fragmented DNA fluorescence |
| CMA3 staining | To determine DNA integrity | Staining by CMA3 | Competes with protamine for the same binding site in DNA | A positive test indicates a low DNA protamination state associated with poorly packaged sperm chromatin |
| SCSA/AO test | To detect breaks in single-stranded DNA (ssDNA) | Acid denaturation, followed by staining with AO | AO emits fluorescence after binding with breaks | Denatured DNA emit an orange-red fluorescence, normal DNA emits green |
| SCD/Halo test | To detect DNA breaks in lysed sperm nuclei | Agarose-embedded sperm are subjected to a denaturing solution to remove nuclear proteins | Assesses dispersion of DNA fragments after denaturation | Sperm with fragmented DNA do not produce the halo characteristic; halo of dispersed DNA loops are observed in sperm with non-fragmented DNA |
| SCGE/ Comet assay | To detect DNA single-strand breaks, ALS, and cross-linking | Gel electrophoresis | Electrophoretic assessment of DNA fragments | Comet tail size represents the amount of DNA fragments |
| Antioxidant | Dose | Effects on Sperm Parameters/Quality | References |
|---|---|---|---|
| CoQ10 | 200–300 mg/day | Improved sperm motility and TAC concentrations and decreased MDA levels. | [153,155,156] |
| Carnitines | 25 mg/day | Improved sperm count, motility, and morphology. | [157,158,159] |
| Lycopene | 20–25 mg/day | Increased seminal omega-3; improved sperm count, concentration, motility; and improved TAC; decreased seminal oxidative stress. | [154,160,161] |
| NAC | 600 mg/day | Reduced apoptotic rate; improved sperm morphology, volume, motility, viscosity, TAC, DNA fragmentation, and protamine deficiency; decreased ROS production. | [162,163,164] |
| Melatonin | N/A | Sperm melatonin incubation was positively correlated with reduced DNA damage, MDA levels and higher sperm viability and motility. | [165,166,167,168,169] |
| Alpha-lipoic acid | 600 mg/day | Improved sperm viability, motility, count, concentration, and TAC; decreased DNA damage and MDA levels. | [170,171,172,173] |
| Omega-3 | 1.5–2.0 g/day | Improved sperm volume, count, concentration, motility, and morphology; improved testis size, TAC, and reduced DNA fragmentation. | [174,175,176] |
| Vitamins/ Minerals | Dose | Main Conclusions | References |
|---|---|---|---|
| Vitamin C | 1.0 g/day | Improved semen agglutination and sperm concentration, motility, and viability; positively associated with higher fertilization rates. | [182,185] |
| Vitamin E | 100–600 mg/day | Decreased MDA levels and increased fertilization rates. | [186] |
| Vitamin B9 | 5 mg/day | Improved sperm count. | [187] |
| Zinc | 200–500 mg/day | Improved sperm count, motility, and fertilization rates and reduced the incidence of anti-sperm antibodies; improved sperm chromatin integrity. | [188,189] |
| Selenium | 200–1000 μg/day | Improved TAC and sperm count, concentration, motility, and morphology; positively associated with pregnancy and live birth. | [190,191,192,193,194] |
| Factors | Findings | References |
|---|---|---|
| Dietary patterns | Healthy dietary patterns (Mediterranean and paleo diet) with low-fat and high non-dairy protein (i.e., fish and white meat) has an important influence on fertility. Dairy products rich in calcium and protein are beneficial. Diets with a low-glycemic load and high amounts of whole grains may benefit fecundity. | [31,32,33,40,209,210] |
| Oily sea fish, olive oil, and rapeseed oil intake | Fish and seafood are the main sources of omega-3 and fat-soluble vitamins A, D, E, and K; therefore, their incorporation into the diet may improve semen quality. Vegetable oils containing unsaturated acid residues, alpha-linolenic acid, vitamin E, and polyphenols can benefit fertility. | [13,37,211] |
| Vegetable, fruit, nut, and seed intake | Vegetables and fruits provide antioxidants, folic acid, fiber, and minerals associated with improved semen quality and fertility. Nuts and unroasted unsalted seeds provide fiber, tocopherols, phytosterols, polyphenols, and minerals that have a beneficial effect on sperm quality. | [35,36,37,196,212] |
| Whole-grain products in the diet | It is recommended that refined flour products be limited in the diet, with whole-grain products that are rich in fiber being more beneficial for fertility. | [36,213] |
| Physical exercise | Along with a healthy diet, regular exercise can improve BMI and fertility. It affects general health and well-being and protects against cardiovascular disease, type 2 diabetes, and psychological stress, among others. | [214,215,216,217] |
| Resistance training | Combined aerobic and resistance training, moderate-intensity continuous training, high-intensity continuous training, resistance training, and high-intensity interval training strategies improved semen quality parameters, seminal markers of oxidative stress, seminal markers of inflammation, as well as measures of body composition. | [218,219,220] |
| Sleep | Adequate sleep is crucial for general health and well-being. The relationship between sleep and reproductive hormones is bidirectional; reproductive hormones may modify sleep, and sleep disruption may alter the profile of reproductive hormone secretion. Multiple pathways exist by which sleep and circadian rhythms influence fertility. Additionally, good sleep can reduce mental stress. | [221,222,223,224] |
| Proactive stress reduction | Yoga and mindfulness training benefits fertility by reversing cellular dysfunctions in male reproductive organs and alleviates mental disturbances that influence reproduction. | [225,226,227,228] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Assidi, M. Infertility in Men: Advances towards a Comprehensive and Integrative Strategy for Precision Theranostics. Cells 2022, 11, 1711. https://doi.org/10.3390/cells11101711
Assidi M. Infertility in Men: Advances towards a Comprehensive and Integrative Strategy for Precision Theranostics. Cells. 2022; 11(10):1711. https://doi.org/10.3390/cells11101711
Chicago/Turabian StyleAssidi, Mourad. 2022. "Infertility in Men: Advances towards a Comprehensive and Integrative Strategy for Precision Theranostics" Cells 11, no. 10: 1711. https://doi.org/10.3390/cells11101711
APA StyleAssidi, M. (2022). Infertility in Men: Advances towards a Comprehensive and Integrative Strategy for Precision Theranostics. Cells, 11(10), 1711. https://doi.org/10.3390/cells11101711






