Store-Operated Calcium Entry in Skeletal Muscle: What Makes It Different?
Abstract
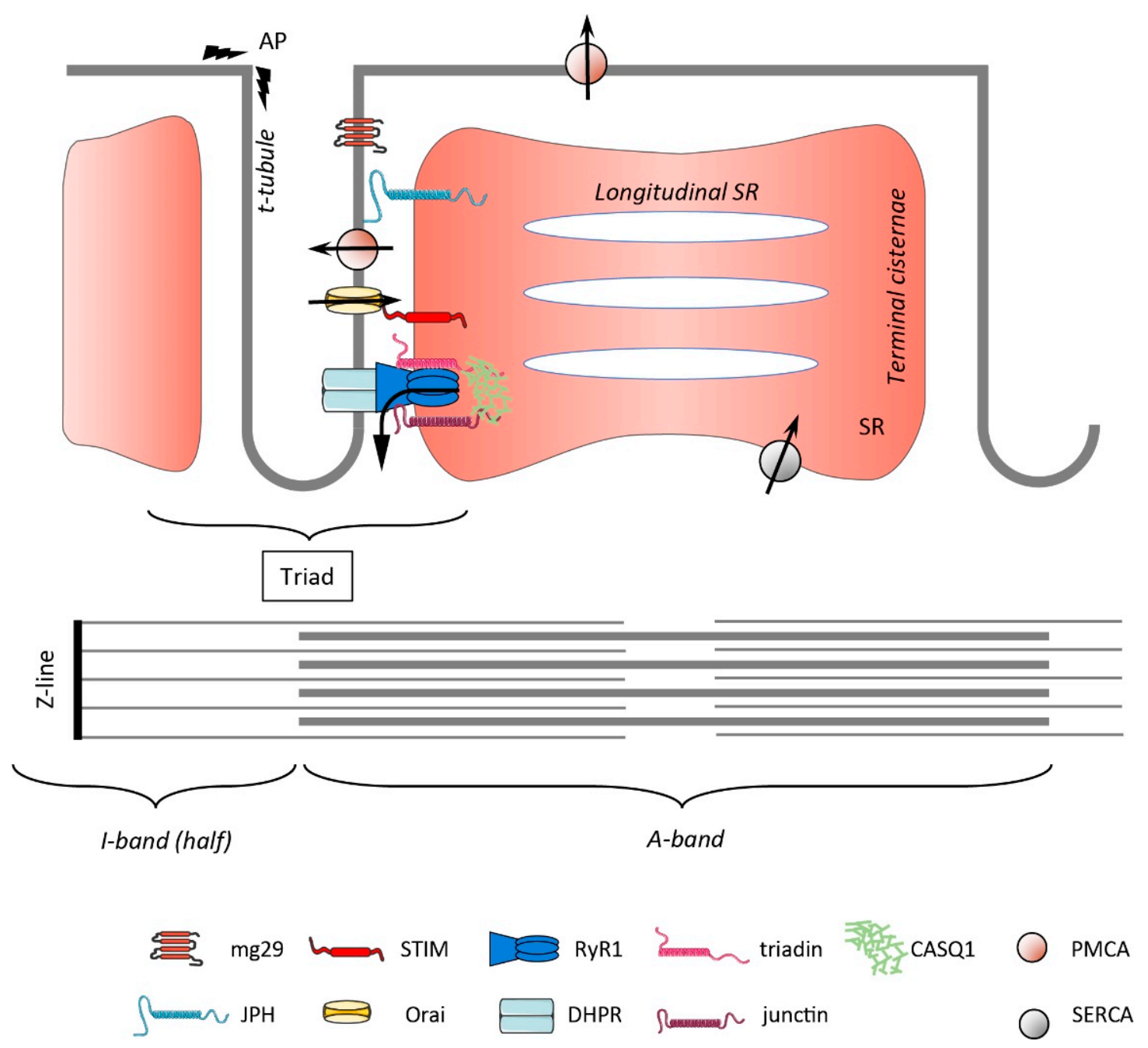
1. Overview of the Muscle Structure and Ca2+ Handling
2. Localization and Role of SOCE Molecules in Cells with “Constrained” Architecture
2.1. Localization and Movements of STIM and Orai
2.2. Regulation of SOCE in Skeletal Muscle
3. Muscle-Specific Fast Activation Kinetics of SOCE
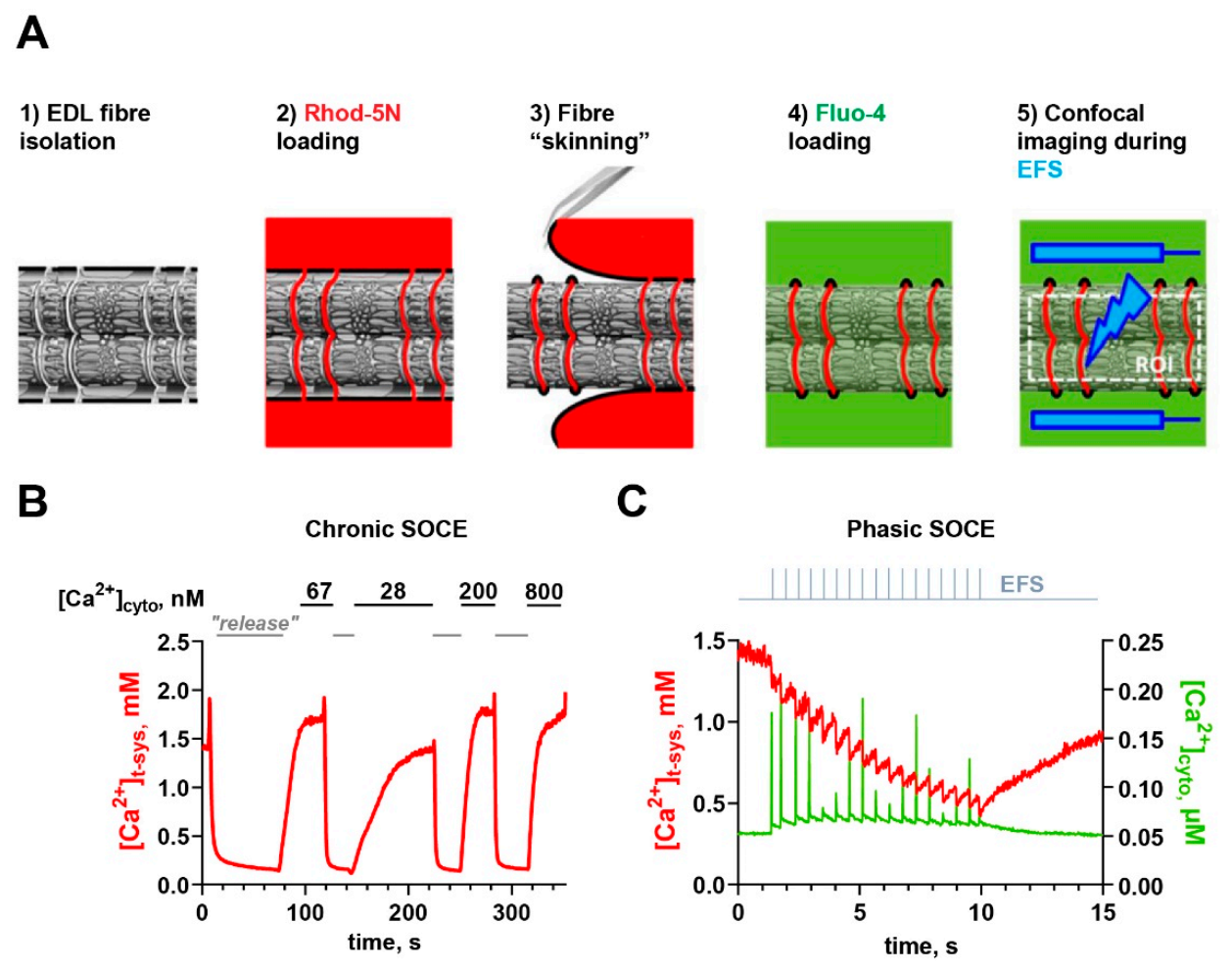
3.1. Using “Skinned” Fibers to Measure SOCE
3.2. How to Measure SOCE during EC Coupling?
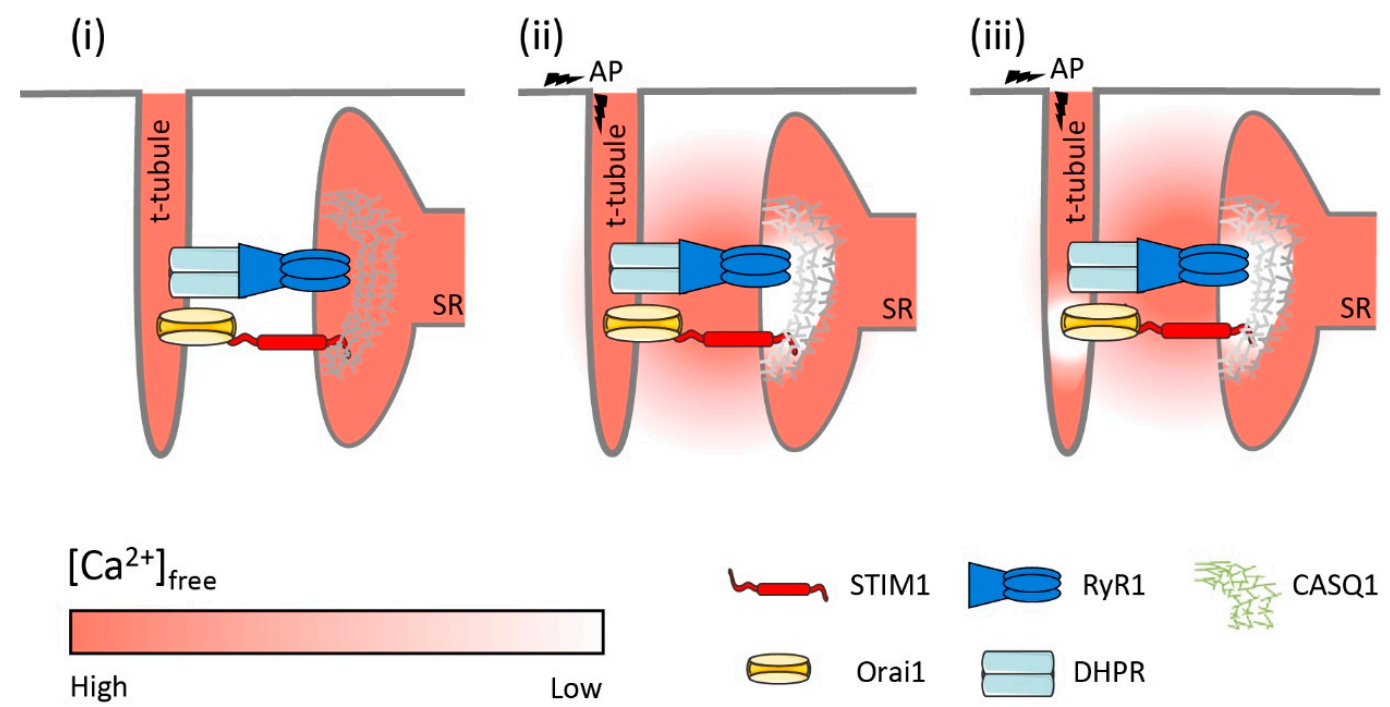
3.3. A Potential Model of Phasic SOCE Activation
3.4. What Is the Physiological Role of Phasic SOCE?
4. Pharmacology of SOCE in Skeletal Muscle: Recent Advances
Recent Drug Developments and Future Perspectives
5. Variety of STIM Molecules Expressed in Adult Tissue
6. STIM and Orai in Different Fiber Types
7. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AP | Action potential |
| APACC | AP-activated Ca2+ current |
| CAD | Ca2+-activated domain |
| CASQ | Calsequestrin |
| CDI | Ca2+-dependent inactivation |
| cER | Cortical ER |
| CEU | Ca2+ entry unit |
| CPA | Cyclopiasonic acid |
| CRACR2A | CRAC regulator 2A |
| DHPR | Dihydropyridine receptor |
| EB1 | End-binding protein 1 |
| EC | Excitation–contraction |
| EDL | Extensor digitorum longus muscle |
| EFS | Electrical field stimulation |
| FDB | Flexor digitorum brevis muscle |
| FRAP | Fluorescence recovery after photobleaching |
| HIT | High intensity |
| ICRAC | Ca2+ release activated Ca2+ current |
| IP3 | Inositol 1,4,5-trisphosphate |
| IP3R | IP3 receptor |
| JPH | Junctophilin |
| jSR | Junctional SR |
| lSR | Longitudinal SR |
| MAP6 | Microtubule-associated protein 6 |
| MCS | Membrane contact site |
| MEF | Mouse embryonic fibroblast |
| MEF2 | Myocyte enhancer factor-2 |
| Mg29 | Mitsugumin 29 |
| MyHC | Myosin heavy chain |
| NCX | Na+/Ca2+ exchanger |
| NFAT | Nuclear factor of activated T-cells |
| NMJ | Neuromuscular junction |
| PiP2 | Phosphatidylinositol 4,5-bisphosphate |
| PM | Plasma membrane |
| PMCA | Plasma membrane Ca2+ ATPase |
| POST | Partner of STIM |
| RyR1 | Ryanodine receptor 1 |
| SARAF | SOCE-associated regulatory factor |
| SERCA | Sarco-endoplasmic Ca2+ ATPase |
| SR | Sarcoplasmic reticulum |
| STIM | Stromal interaction molecule |
| STIMATE | STIM-activating enhancer |
| TA | Tibialis anterior muscle |
| TAs | Tubular aggregates |
| TC | Terminal cisternae |
| Tg | Thapsigargin |
| TRP | Transient receptor potential |
References
- Boncompagni, S.; Pozzer, D.; Viscomi, C.; Ferreiro, A.; Zito, E. Physical and Functional Cross Talk Between Endo-Sarcoplasmic Reticulum and Mitochondria in Skeletal Muscle. Antioxid. Redox. Signal. 2020, 32, 873–883. [Google Scholar] [CrossRef] [PubMed]
- Al-Qusairi, L.; Laporte, J. T-Tubule Biogenesis and Triad Formation in Skeletal Muscle and Implication in Human Diseases. Skelet. Muscle 2011, 1, 26. [Google Scholar] [CrossRef]
- Sorrentino, V. Sarcoplasmic Reticulum: Structural Determinants and Protein Dynamics. Int. J. Biochem. Cell Biol. 2011, 43, 1075–1078. [Google Scholar] [CrossRef] [PubMed]
- Schiaffino, S.; Reggiani, C. Fiber Types in Mammalian Skeletal Muscles. Physiol. Rev. 2011, 91, 1447–1531. [Google Scholar] [CrossRef] [PubMed]
- Flucher, B.E.; Tuluc, P. How and Why Are Calcium Currents Curtailed in the Skeletal Muscle Voltage-Gated Calcium Channels?: Curtailed Skeletal Muscle Calcium Currents. J. Physiol. 2017, 595, 1451–1463. [Google Scholar] [CrossRef] [PubMed]
- Putney, J.W. A Model for Receptor-Regulated Calcium Entry. Cell Calcium 1986, 7, 1–12. [Google Scholar] [CrossRef]
- Liou, J.; Kim, M.L.; Heo, W.D.; Jones, J.T.; Myers, J.W.; Ferrell, J.E.; Meyer, T. STIM Is a Ca2+ Sensor Essential for Ca2+-Store-Depletion-Triggered Ca2+ Influx. Curr. Biol. 2005, 15, 1235–1241. [Google Scholar] [CrossRef]
- Zhang, S.L.; Yu, Y.; Roos, J.; Kozak, J.A.; Deerinck, T.J.; Ellisman, M.H.; Stauderman, K.A.; Cahalan, M.D. STIM1 Is a Ca2+ Sensor That Activates CRAC Channels and Migrates from the Ca2+ Store to the Plasma Membrane. Nature 2005, 437, 902–905. [Google Scholar] [CrossRef]
- Roos, J.; DiGregorio, P.J.; Yeromin, A.V.; Ohlsen, K.; Lioudyno, M.; Zhang, S.; Safrina, O.; Kozak, J.A.; Wagner, S.L.; Cahalan, M.D.; et al. STIM1, an Essential and Conserved Component of Store-Operated Ca2+ Channel Function. J. Cell. Biol. 2005, 169, 435–445. [Google Scholar] [CrossRef]
- Feske, S.; Gwack, Y.; Prakriya, M.; Srikanth, S.; Puppel, S.-H.; Tanasa, B.; Hogan, P.G.; Lewis, R.S.; Daly, M.; Rao, A. A Mutation in Orai1 Causes Immune Deficiency by Abrogating CRAC Channel Function. Nature 2006, 441, 179–185. [Google Scholar] [CrossRef]
- Vig, M.; Peinelt, C.; Beck, A.; Koomoa, D.L.; Rabah, D.; Koblan-Huberson, M.; Kraft, S.; Turner, H.; Fleig, A.; Penner, R.; et al. CRACM1 Is a Plasma Membrane Protein Essential for Store-Operated Ca2+ Entry. Science 2006, 312, 1220–1223. [Google Scholar] [CrossRef]
- Yeromin, A.V.; Zhang, S.L.; Jiang, W.; Yu, Y.; Safrina, O.; Cahalan, M.D. Molecular Identification of the CRAC Channel by Altered Ion Selectivity in a Mutant of Orai. Nature 2006, 443, 226–229. [Google Scholar] [CrossRef]
- Huang, G.N.; Zeng, W.; Kim, J.Y.; Yuan, J.P.; Han, L.; Muallem, S.; Worley, P.F. STIM1 Carboxyl-Terminus Activates Native SOC, Icrac and TRPC1 Channels. Nat. Cell. Biol. 2006, 8, 1003–1010. [Google Scholar] [CrossRef]
- Liou, J.; Fivaz, M.; Inoue, T.; Meyer, T. Live-Cell Imaging Reveals Sequential Oligomerization and Local Plasma Membrane Targeting of Stromal Interaction Molecule 1 after Ca2+ Store Depletion. Proc. Natl. Acad. Sci. USA 2007, 104, 9301–9306. [Google Scholar] [CrossRef]
- Park, C.Y.; Hoover, P.J.; Mullins, F.M.; Bachhawat, P.; Covington, E.D.; Raunser, S.; Walz, T.; Garcia, K.C.; Dolmetsch, R.E.; Lewis, R.S. STIM1 Clusters and Activates CRAC Channels via Direct Binding of a Cytosolic Domain to Orai1. Cell 2009, 136, 876–890. [Google Scholar] [CrossRef]
- Lewis, R.S. Store-Operated Calcium Channels: New Perspectives on Mechanism and Function. Cold Spring Harb. Perspect. Biol. 2011, 3, a003970. [Google Scholar] [CrossRef]
- Orci, L.; Ravazzola, M.; Le Coadic, M.; Shen, W.-W.; Demaurex, N.; Cosson, P. STIM1-Induced Precortical and Cortical Subdomains of the Endoplasmic Reticulum. Proc. Natl. Acad. Sci. USA 2009, 106, 19358–19362. [Google Scholar] [CrossRef]
- Perni, S.; Dynes, J.L.; Yeromin, A.V.; Cahalan, M.D.; Franzini-Armstrong, C. Nanoscale Patterning of STIM1 and Orai1 during Store-Operated Ca 2+ Entry. Proc. Natl. Acad. Sci. USA 2015, 112, E5533–E5542. [Google Scholar] [CrossRef] [PubMed]
- Ercan, E.; Momburg, F.; Engel, U.; Temmerman, K.; Nickel, W.; Seedorf, M. A Conserved, Lipid-Mediated Sorting Mechanism of Yeast Ist2 and Mammalian STIM Proteins to the Peripheral ER. Traffic 2009, 10, 1802–1818. [Google Scholar] [CrossRef]
- Bhardwaj, R.; Müller, H.-M.; Nickel, W.; Seedorf, M. Oligomerization and Ca2+/Calmodulin Control Binding of the ER Ca2+-Sensors STIM1 and STIM2 to Plasma Membrane Lipids. Biosci. Rep. 2013, 33, e00077. [Google Scholar] [CrossRef]
- Berna-Erro, A.; Jardin, I.; Salido, G.M.; Rosado, J.A. Role of STIM2 in Cell Function and Physiopathology: STIM2 in Cell Function and Physiopathology. J. Physiol. 2017, 595, 3111–3128. [Google Scholar] [CrossRef] [PubMed]
- Muik, M.; Schindl, R.; Fahrner, M.; Romanin, C. Ca2+ Release-Activated Ca2+ (CRAC) Current, Structure, and Function. Cell. Mol. Life. Sci. 2012, 69, 4163–4176. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Choi, S.; Maléth, J.J.; Park, S.; Ahuja, M.; Muallem, S. The ER/PM Microdomain, PI(4,5)P2 and the Regulation of STIM1–Orai1 Channel Function. Cell Calcium 2015, 58, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Prakriya, M.; Lewis, R.S. Store-operated calcium channels. Physiol. Rev. 2015, 95, 54. [Google Scholar] [CrossRef]
- Armstrong, C.M.; Bezanilla, F.M.; Horowicz, P. Twitches in the Presence of Ethylene Glycol Bis(β-Aminoethyl Ether)-N,N′-Tetraacetic Acid. Biochim. Biophys. Acta (BBA)—Bioenerg. 1972, 267, 605–608. [Google Scholar] [CrossRef]
- Launikonis, B.S.; Murphy, R.M.; Edwards, J.N. Toward the Roles of Store-Operated Ca2+ Entry in Skeletal Muscle. Pflügers Arch.—Eur. J. Physiol. 2010, 460, 813–823. [Google Scholar] [CrossRef]
- Ogawa, Y.; Kurebayashi, N.; Murayama, T. Ryanodine Receptor Isoforms in Excitation-Contraction Coupling. Adv. Biophys. 1999, 36, 27–64. [Google Scholar] [CrossRef]
- Murayama, T.; Kurebayashi, N.; Ogawa, Y. Role of Mg(2+) in Ca(2+)-Induced Ca(2+) Release through Ryanodine Receptors of Frog Skeletal Muscle: Modulations by Adenine Nucleotides and Caffeine. Biophys. J. 2000, 78, 1810–1824. [Google Scholar] [CrossRef]
- Stiber, J.; Hawkins, A.; Zhang, Z.-S.; Wang, S.; Burch, J.; Graham, V.; Ward, C.C.; Seth, M.; Finch, E.; Malouf, N.; et al. STIM1 Signalling Controls Store-Operated Calcium Entry Required for Development and Contractile Function in Skeletal Muscle. Nat. Cell. Biol. 2008, 10, 688–697. [Google Scholar] [CrossRef]
- Yarotskyy, V.; Dirksen, R.T. Temperature and RyR1 Regulate the Activation Rate of Store-Operated Ca2+ Entry Current in Myotubes. Biophys. J. 2012, 103, 202–211. [Google Scholar] [CrossRef][Green Version]
- Mallouk, N.; Allard, B. Ca(2+) Influx and Opening of Ca(2+)-Activated K(+) Channels in Muscle Fibers from Control and Mdx Mice. Biophys. J. 2002, 82, 3012–3021. [Google Scholar] [CrossRef]
- Berbey, C.; Allard, B. Electrically Silent Divalent Cation Entries in Resting and Active Voltage-Controlled Muscle Fibers. Biophys J. 2009, 96, 2648–2657. [Google Scholar] [CrossRef]
- Kurebayashi, N.; Ogawa, Y. Depletion of Ca2+ in the Sarcoplasmic Reticulum Stimulates Ca2+ Entry into Mouse Skeletal Muscle Fibres. J. Physiol. 2001, 15. [Google Scholar] [CrossRef]
- Lacruz, R.S.; Feske, S. Diseases Caused by Mutations in ORAI1 and STIM1: Mutations in ORAI1 and STIM1. Ann. N. Y. Acad. Sci. 2015, 1356, 45–79. [Google Scholar] [CrossRef]
- Grigoriev, I.; Gouveia, S.M.; van der Vaart, B.; Demmers, J.; Smyth, J.T.; Honnappa, S.; Splinter, D.; Steinmetz, M.O.; Putney, J.W.; Hoogenraad, C.C.; et al. STIM1 Is a MT-Plus-End-Tracking Protein Involved in Remodeling of the ER. Curr. Biol. 2008, 18, 177–182. [Google Scholar] [CrossRef]
- Chang, C.-L.; Chen, Y.-J.; Quintanilla, C.G.; Hsieh, T.-S.; Liou, J. EB1 Binding Restricts STIM1 Translocation to ER–PM Junctions and Regulates Store-Operated Ca2+ Entry. J. Cell. Biol. 2018, 217, 2047–2058. [Google Scholar] [CrossRef]
- Barone, V.; Randazzo, D.; Del Re, V.; Sorrentino, V.; Rossi, D. Organization of Junctional Sarcoplasmic Reticulum Proteins in Skeletal Muscle Fibers. J. Muscle. Res. Cell. Motil. 2015, 36, 501–515. [Google Scholar] [CrossRef]
- Wei-LaPierre, L.; Carrell, E.M.; Boncompagni, S.; Protasi, F.; Dirksen, R.T. Orai1-Dependent Calcium Entry Promotes Skeletal Muscle Growth and Limits Fatigue. Nat. Commun. 2013, 4. [Google Scholar] [CrossRef]
- Koenig, X.; Choi, R.H.; Launikonis, B.S. Store-Operated Ca2+ Entry Is Activated by Every Action Potential in Skeletal Muscle. Commun. Biol. 2018, 1. [Google Scholar] [CrossRef]
- Lee, K.J.; Hyun, C.; Woo, J.S.; Park, C.S.; Kim, D.H.; Lee, E.H. Stromal Interaction Molecule 1 (STIM1) Regulates Sarcoplasmic/Endoplasmic Reticulum Ca2+-ATPase 1a (SERCA1a) in Skeletal Muscle. Pflügers Arch.—Eur. J. Physiol. 2014, 466, 987–1001. [Google Scholar] [CrossRef]
- Boncompagni, S.; Michelucci, A.; Pietrangelo, L.; Dirksen, R.T.; Protasi, F. Exercise-Dependent Formation of New Junctions That Promote STIM1-Orai1 Assembly in Skeletal Muscle. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Sztretye, M.; Singlár, Z.; Balogh, N.; Kis, G.; Szentesi, P.; Angyal, Á.; Balatoni, I.; Csernoch, L.; Dienes, B. The Role of Orai1 in Regulating Sarcoplasmic Calcium Release, Mitochondrial Morphology and Function in Myostatin Deficient Skeletal Muscle. Front. Physiol. 2020, 11, 601090. [Google Scholar] [CrossRef] [PubMed]
- Cusimano, V.; Pampinella, F.; Giacomello, E.; Sorrentino, V. Assembly and Dynamics of Proteins of the Longitudinal and Junctional Sarcoplasmic Reticulum in Skeletal Muscle Cells. Proc. Natl. Acad. Sci. USA 2009, 106, 4695–4700. [Google Scholar] [CrossRef] [PubMed]
- Sébastien, M.; Aubin, P.; Brocard, J.; Brocard, J.; Marty, I.; Fauré, J. Dynamics of Triadin, a Muscle-Specific Triad Protein, within Sarcoplasmic Reticulum Subdomains. MBoC 2020, 31, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Launikonis, B.S.; Zhou, J.; Royer, L.; Shannon, T.R.; Brum, G.; Rios, E. Depletion “Skraps” and Dynamic Buffering inside the Cellular Calcium Store. Proc. Natl. Acad. Sci. USA 2006, 103, 2982–2987. [Google Scholar] [CrossRef]
- Canato, M.; Scorzeto, M.; Giacomello, M.; Protasi, F.; Reggiani, C.; Stienen, G.J.M. Massive Alterations of Sarcoplasmic Reticulum Free Calcium in Skeletal Muscle Fibers Lacking Calsequestrin Revealed by a Genetically Encoded Probe. Proc. Natl. Acad. Sci. USA 2010, 107, 22326–22331. [Google Scholar] [CrossRef]
- Blaauw, B.; del Piccolo, P.; Rodriguez, L.; Hernandez Gonzalez, V.-H.; Agatea, L.; Solagna, F.; Mammano, F.; Pozzan, T.; Schiaffino, S. No Evidence for Inositol 1,4,5-Trisphosphate–Dependent Ca2+ Release in Isolated Fibers of Adult Mouse Skeletal Muscle. J. Gen. Physiol. 2012, 140, 235–241. [Google Scholar] [CrossRef]
- Jaimovich, E.; Reyes, R.; Liberona, J.L.; Powell, J.A. IP3 Receptors, IP3 Transients, and Nucleus-Associated Ca2+ Signals in Cultured Skeletal Muscle. Am. J. Physiol. Cell. Physiol. 2000, 278, C998–C1010. [Google Scholar] [CrossRef]
- Casas, M.; Figueroa, R.; Jorquera, G.; Escobar, M.; Molgó, J.; Jaimovich, E. IP3-Dependent, Post-Tetanic Calcium Transients Induced by Electrostimulation of Adult Skeletal Muscle Fibers. J. Gen. Physiol. 2010, 136, 455–467. [Google Scholar] [CrossRef]
- Díaz-Vegas, A.R.; Cordova, A.; Valladares, D.; Llanos, P.; Hidalgo, C.; Gherardi, G.; De Stefani, D.; Mammucari, C.; Rizzuto, R.; Contreras-Ferrat, A.; et al. Mitochondrial Calcium Increase Induced by RyR1 and IP3R Channel Activation After Membrane Depolarization Regulates Skeletal Muscle Metabolism. Front. Physiol. 2018, 9, 791. [Google Scholar] [CrossRef]
- Powell, J.A.; Molgo, J.; Adams, D.S.; Colasante, C.; Williams, A.; Bohlen, M.; Jaimovich, E. IP3 Receptors and Associated Ca2+ Signals Localize to Satellite Cells and to Components of the Neuromuscular Junction in Skeletal Muscle. J. Neurosci. 2003, 23, 8185–8192. [Google Scholar] [CrossRef]
- Zhu, H.; Bhattacharyya, B.J.; Lin, H.; Gomez, C.M. Skeletal Muscle IP3R1 Receptors Amplify Physiological and Pathological Synaptic Calcium Signals. J. Neurosci. 2011, 31, 15269–15283. [Google Scholar] [CrossRef]
- Porter, K.R.; Palade, G.E. Studies on the Endoplasmic Reticulum. III. Its Form and Distribution in Striated Muscle Cells. J. Biophys. Biochem. Cytol. 1957, 3, 269–300. [Google Scholar] [CrossRef]
- Li, C.; Qian, T.; He, R.; Wan, C.; Liu, Y.; Yu, H. Endoplasmic Reticulum–Plasma Membrane Contact Sites: Regulators, Mechanisms, and Physiological Functions. Front. Cell Dev. Biol. 2021, 9, 627700. [Google Scholar] [CrossRef]
- Hirata, Y.; Brotto, M.; Weisleder, N.; Chu, Y.; Lin, P.; Zhao, X.; Thornton, A.; Komazaki, S.; Takeshima, H.; Ma, J.; et al. Uncoupling Store-Operated Ca2+ Entry and Altered Ca2+ Release from Sarcoplasmic Reticulum through Silencing of Junctophilin Genes. Biophys. J. 2006, 90, 4418–4427. [Google Scholar] [CrossRef]
- van Oort, R.J.; Garbino, A.; Wang, W.; Dixit, S.S.; Landstrom, A.P.; Gaur, N.; De Almeida, A.C.; Skapura, D.G.; Rudy, Y.; Burns, A.R.; et al. Disrupted Junctional Membrane Complexes and Hyperactive Ryanodine Receptors After Acute Junctophilin Knockdown in Mice. Circulation 2011, 123, 979–988. [Google Scholar] [CrossRef]
- Powell, J.A.; Petherbridge, L.; Flucher, B.E. Formation of Triads without the Dihydropyridine Receptor Alpha Subunits in Cell Lines from Dysgenic Skeletal Muscle. J. Cell. Biol. 1996, 134, 375–387. [Google Scholar] [CrossRef]
- Felder, E.; Protasi, F.; Hirsch, R.; Franzini-Armstrong, C.; Allen, P.D. Morphology and Molecular Composition of Sarcoplasmic Reticulum Surface Junctions in the Absence of DHPR and RyR in Mouse Skeletal Muscle. Biophys. J. 2002, 82, 3144–3149. [Google Scholar] [CrossRef]
- Pan, Z.; Yang, D.; Nagaraj, R.Y.; Nosek, T.A.; Nishi, M.; Takeshima, H.; Cheng, H.; Ma, J. Dysfunction of Store-Operated Calcium Channel in Muscle Cells Lacking Mg29. Nat. Cell Biol. 2002, 4, 379–383. [Google Scholar] [CrossRef]
- Thevenon, D.; Smida-Rezgui, S.; Chevessier, F.; Groh, S.; Henry-Berger, J.; Beatriz Romero, N.; Villaz, M.; DeWaard, M.; Marty, I. Human Skeletal Muscle Triadin: Gene Organization and Cloning of the Major Isoform, Trisk 51. Biochem. Biophys. Res. Commun. 2003, 303, 669–675. [Google Scholar] [CrossRef]
- Marty, I. Triadin Regulation of the Ryanodine Receptor Complex: Triadin Regulation of the Ryanodine Receptor Complex. J. Physiol. 2015, 593, 3261–3266. [Google Scholar] [CrossRef] [PubMed]
- Treves, S.; Vukcevic, M.; Maj, M.; Thurnheer, R.; Mosca, B.; Zorzato, F. Minor Sarcoplasmic Reticulum Membrane Components That Modulate Excitation-Contraction Coupling in Striated Muscles: Sarcoplasmic Reticulum Membrane Components. J. Physiol. 2009, 587, 3071–3079. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, X.; Duan, H.; Fulton, T.R.; Eu, J.P.; Meissner, G. Altered Stored Calcium Release in Skeletal Myotubes Deficient of Triadin and Junctin. Cell Calcium 2009, 45, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Treves, S.; Feriotto, G.; Moccagatta, L.; Gambari, R.; Zorzato, F. Molecular Cloning, Expression, Functional Characterization, Chromosomal Localization, and Gene Structure of Junctate, a Novel Integral Calcium Binding Protein of Sarco(Endo)Plasmic Reticulum Membrane. J. Biol. Chem. 2000, 275, 39555–39568. [Google Scholar] [CrossRef]
- Divet, A.; Paesante, S.; Grasso, C.; Cavagna, D.; Tiveron, C.; Paolini, C.; Protasi, F.; Huchet-Cadiou, C.; Treves, S.; Zorzato, F. Increased Ca2+ Storage Capacity of the Skeletal Muscle Sarcoplasmic Reticulum of Transgenic Mice Over-Expressing Membrane Bound Calcium Binding Protein Junctate. J. Cell. Physiol. 2007, 213, 464–474. [Google Scholar] [CrossRef]
- Srikanth, S.; Jew, M.; Kim, K.-D.; Yee, M.-K.; Abramson, J.; Gwack, Y. Junctate Is a Ca2+-Sensing Structural Component of Orai1 and Stromal Interaction Molecule 1 (STIM1). Proc. Natl. Acad. Sci. USA 2012, 109, 8682–8687. [Google Scholar] [CrossRef]
- Shin, D.W.; Pan, Z.; Kim, E.K.; Lee, J.M.; Bhat, M.B.; Parness, J.; Kim, D.H.; Ma, J. A Retrograde Signal from Calsequestrin for the Regulation of Store-Operated Ca2+ Entry in Skeletal Muscle. J. Biol. Chem. 2003, 278, 3286–3292. [Google Scholar] [CrossRef]
- Zhao, X.; Min, C.K.; Ko, J.-K.; Parness, J.; Kim, D.H.; Weisleder, N.; Ma, J. Increased Store-Operated Ca2+ Entry in Skeletal Muscle with Reduced Calsequestrin-1 Expression. Biophys. J. 2010, 99, 1556–1564. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, L.; Li, S.; Zheng, Y.; Yan, X.; Chen, M.; Wang, H.; Putney, J.W.; Luo, D. Retrograde Regulation of STIM1-Orai1 Interaction and Store-Operated Ca2+ Entry by Calsequestrin. Sci. Rep. 2015, 5, 11349. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, L.; Li, S.; Xue, J.; Luo, D. Calsequestrin-1 Regulates Store-Operated Ca2+ Entry by Inhibiting STIM1 Aggregation. Cell. Physiol. Biochem. 2016, 38, 2183–2193. [Google Scholar] [CrossRef]
- Shimuta, M.; Komazaki, S.; Nishi, M.; Iino, M.; Nakagawara, K.; Takeshima, H. Structure and Expression of Mitsugumin29 Gene. Febs Lett. 1998, 431, 263–267. [Google Scholar] [CrossRef]
- Nishi, M.; Komazaki, S.; Kurebayashi, N.; Ogawa, Y.; Noda, T.; Iino, M.; Takeshima, H. Abnormal Features in Skeletal Muscle from Mice Lacking Mitsugumin29. J. Cell. Biol. 1999, 147, 1473–1480. [Google Scholar] [CrossRef]
- Zhao, X.; Weisleder, N.; Thornton, A.; Oppong, Y.; Campbell, R.; Ma, J.; Brotto, M. Compromised Store-Operated Ca 2 Entry in Aged Skeletal Muscle. Aging Cell 2008, 7, 561–568. [Google Scholar] [CrossRef]
- Nagaraj, R.Y.; Nosek, C.M.; Brotto, M.A.; Nishi, M.; Takeshima, H.; Nosek, T.M.; Ma, J. Increased Susceptibility to Fatigue of Slow- and Fast-Twitch Muscles from Mice Lacking the MG29 Gene. Physiol Genom. 2000, 4, 43–49. [Google Scholar] [CrossRef]
- Kurebayashi, N.; Takeshima, H.; Nishi, M.; Murayama, T.; Suzuki, E.; Ogawa, Y. Changes in Ca2+ Handling in Adult MG29-Deficient Skeletal Muscle. Biochem. Biophys. Res. Commun. 2003, 310, 1266–1272. [Google Scholar] [CrossRef]
- Edwards, J.N.; Blackmore, D.G.; Gilbert, D.F.; Murphy, R.M.; Launikonis, B.S. Store-Operated Calcium Entry Remains Fully Functional in Aged Mouse Skeletal Muscle despite a Decline in STIM1 Protein Expression: Store-Operated Ca2+ Entry in Aged Skeletal Muscle. Aging Cell 2011, 10, 675–685. [Google Scholar] [CrossRef]
- Kang, F.; Zhou, M.; Huang, X.; Fan, J.; Wei, L.; Boulanger, J.; Liu, Z.; Salamero, J.; Liu, Y.; Chen, L. E-Syt1 Re-Arranges STIM1 Clusters to Stabilize Ring-Shaped ER-PM Contact Sites and Accelerate Ca2+ Store Replenishment. Sci. Rep. 2019, 9, 3975. [Google Scholar] [CrossRef]
- Berlansky, S.; Humer, C.; Sallinger, M.; Frischauf, I. More Than Just Simple Interaction between STIM and Orai Proteins: CRAC Channel Function Enabled by a Network of Interactions with Regulatory Proteins. IJMS 2021, 22, 471. [Google Scholar] [CrossRef]
- Derler, I.; Jardin, I.; Stathopulos, P.B.; Muik, M.; Fahrner, M.; Zayats, V.; Pandey, S.K.; Poteser, M.; Lackner, B.; Absolonova, M.; et al. Cholesterol Modulates Orai1 Channel Function. Sci. Signal. 2016, 9, ra10. [Google Scholar] [CrossRef]
- Rosemblatt, M.; Hidalgo, C.; Vergara, C.; Ikemoto, N. Immunological and Biochemical Properties of Transverse Tubule Membranes Isolated from Rabbit Skeletal Muscle. J. Biol. Chem. 1981, 256, 8140–8148. [Google Scholar] [CrossRef]
- Carozzi, A.J.; Ikonen, E.; Lindsay, M.R.; Parton, R.G. Role of Cholesterol in Developing T-Tubules: Analogous Mechanisms for T-Tubule and Caveolae Biogenesis. Traffic 2000, 1, 326–341. [Google Scholar] [CrossRef]
- Maléth, J.; Choi, S.; Muallem, S.; Ahuja, M. Translocation between PI(4,5)P2-Poor and PI(4,5)P2-Rich Microdomains during Store Depletion Determines STIM1 Conformation and Orai1 Gating. Nat. Commun. 2014, 5, 5843. [Google Scholar] [CrossRef]
- Allard, B.; Couchoux, H.; Pouvreau, S.; Jacquemond, V. Sarcoplasmic Reticulum Ca 2+ Release and Depletion Fail to Affect Sarcolemmal Ion Channel Activity in Mouse Skeletal Muscle: Ca2+ Release and Ion Channel Activity in Skeletal Muscle. J. Physiol. 2006, 575, 69–81. [Google Scholar] [CrossRef]
- Protasi, F.; Pietrangelo, L.; Boncompagni, S. Calcium Entry Units (CEUs): Perspectives in Skeletal Muscle Function and Disease. J. Muscle Res. Cell. Motil. 2020. [Google Scholar] [CrossRef]
- Michelucci, A.; Boncompagni, S.; Pietrangelo, L.; García-Castañeda, M.; Takano, T.; Malik, S.; Dirksen, R.T.; Protasi, F. Transverse Tubule Remodeling Enhances Orai1-Dependent Ca2+ Entry in Skeletal Muscle. eLife 2019, 8, e47576. [Google Scholar] [CrossRef]
- Silva-Rojas, R.; Laporte, J.; Böhm, J. STIM1/ORAI1 Loss-of-Function and Gain-of-Function Mutations Inversely Impact on SOCE and Calcium Homeostasis and Cause Multi-Systemic Mirror Diseases. Front. Physiol. 2020, 11, 604941. [Google Scholar] [CrossRef]
- Michelucci, A.; Boncompagni, S.; Pietrangelo, L.; Takano, T.; Protasi, F.; Dirksen, R.T. Pre-Assembled Ca2+ Entry Units and Constitutively Active Ca2+ Entry in Skeletal Muscle of Calsequestrin-1 Knockout Mice. J. Physiol. 2020, 152, e202012617. [Google Scholar] [CrossRef]
- Jayasinghe, I.D.; Munro, M.; Baddeley, D.; Launikonis, B.S.; Soeller, C. Observation of the Molecular Organization of Calcium Release Sites in Fast- and Slow-Twitch Skeletal Muscle with Nanoscale Imaging. J. R Soc. Interface. 2014, 11, 20140570. [Google Scholar] [CrossRef]
- Lyfenko, A.D.; Dirksen, R.T. Differential Dependence of Store-Operated and Excitation-Coupled Ca2+ Entry in Skeletal Muscle on STIM1 and Orai1: Differential Dependence of SOCE and ECCE on STIM1 and Orai1. J. Physiol. 2008, 586, 4815–4824. [Google Scholar] [CrossRef]
- Cully, T.R.; Choi, R.H.; Bjorksten, A.R.; Stephenson, D.G.; Murphy, R.M.; Launikonis, B.S. Junctional Membrane Ca2+ Dynamics in Human Muscle Fibers Are Altered by Malignant Hyperthermia Causative RyR Mutation. Proc. Natl. Acad. Sci. USA 2018, 115, 8215–8220. [Google Scholar] [CrossRef]
- Boncompagni, S.; Thomas, M.; Lopez, J.R.; Allen, P.D.; Yuan, Q.; Kranias, E.G.; Franzini-Armstrong, C.; Perez, C.F. Triadin/Junctin Double Null Mouse Reveals a Differential Role for Triadin and Junctin in Anchoring CASQ to the JSR and Regulating Ca(2+) Homeostasis. PLoS ONE 2012, 7, e39962. [Google Scholar] [CrossRef] [PubMed]
- Sébastien, M.; Giannesini, B.; Aubin, P.; Brocard, J.; Chivet, M.; Pietrangelo, L.; Boncompagni, S.; Bosc, C.; Brocard, J.; Rendu, J.; et al. Deletion of the Microtubule-Associated Protein 6 (MAP6) Results in Skeletal Muscle Dysfunction. Skelet. Muscle 2018, 8, 30. [Google Scholar] [CrossRef] [PubMed]
- Cully, T.R.; Murphy, R.M.; Roberts, L.; Raastad, T.; Fassett, R.G.; Coombes, J.S.; Jayasinghe, I.D.; Launikonis, B.S. Human Skeletal Muscle Plasmalemma Alters Its Structure to Change Its Ca2+-Handling Following Heavy-Load Resistance Exercise. Nat. Commun. 2017, 8, 14266. [Google Scholar] [CrossRef] [PubMed]
- Launikonis, B.S.; Barnes, M.; Stephenson, D.G. Identification of the Coupling between Skeletal Muscle Store-Operated Ca2+ Entry and the Inositol Trisphosphate Receptor. Proc. Natl. Acad. Sci. USA 2003, 100, 2941–2944. [Google Scholar] [CrossRef]
- Launikonis, B.S.; Ríos, E. Store-Operated Ca2+ Entry during Intracellular Ca2+ Release in Mammalian Skeletal Muscle: SOCE during Ca 2+ Release in Muscle. J. Physiol. 2007, 583, 81–97. [Google Scholar] [CrossRef]
- Launikonis, B.S.; Stephenson, D.G.; Friedrich, O. Rapid Ca2+ Flux through the Transverse Tubular Membrane, Activated by Individual Action Potentials in Mammalian Skeletal Muscle: Action Potential-Activated Ca2+ Flux. J. Physiol. 2009, 587, 2299–2312. [Google Scholar] [CrossRef]
- Cully, T.R.; Edwards, J.N.; Murphy, R.M.; Launikonis, B.S. A Quantitative Description of Tubular System Ca2+ Handling in Fast- and Slow-Twitch Muscle Fibres: Quantitating t-System Ca2+ Handling. J. Physiol. 2016, 594, 2795–2810. [Google Scholar] [CrossRef]
- Stienen, G.J.M. Chronicle of Skinned Muscle Fibres. J. Physiol. 2000, 527, 1. [Google Scholar] [CrossRef]
- Posterino, G.S. “Current” Advances in Mechanically Skinned Skeletal Muscle Fibres. Clin. Exp. Pharmacol. Physiol. 2001, 28, 668–674. [Google Scholar] [CrossRef]
- Lamb, G.D.; Stephenson, D.G. Measurement of Force and Calcium Release Using Mechanically Skinned Fibers from Mammalian Skeletal Muscle. J. Appl. Physiol. 2018, 125, 1105–1127. [Google Scholar] [CrossRef]
- Endo, M. ENTRY OF A DYE INTO THE SARCOTUBULAR SYSTEM OF MUSCLE. Nature 1964, 202, 1115–1116. [Google Scholar] [CrossRef]
- Lamb, G.D.; Junankar, P.R.; Stephenson, D.G. Raised Intracellular [Ca2+] Abolishes Excitation-Contraction Coupling in Skeletal Muscle Fibres of Rat and Toad. J. Physiol. 1995, 489 Pt 2, 349–362. [Google Scholar] [CrossRef]
- Lamb, G.D.; Stephenson, D.G. Calcium Release in Skinned Muscle Fibres of the Toad by Transverse Tubule Depolarization or by Direct Stimulation. J. Physiol. 1990, 423, 495–517. [Google Scholar] [CrossRef]
- Posterino, G.S.; Lamb, G.D. Effect of Sarcoplasmic Reticulum Ca2+ Content on Action Potential-Induced Ca2+ Release in Rat Skeletal Muscle Fibres. J. Physiol. 2003, 551, 219–237. [Google Scholar] [CrossRef]
- Posterino, G.S.; Lamb, G.D.; Stephenson, D.G. Twitch and Tetanic Force Responses and Longitudinal Propagation of Action Potentials in Skinned Skeletal Muscle Fibres of the Rat. J. Physiol. 2000, 527 Pt 1, 131–137. [Google Scholar] [CrossRef]
- Lamb, G.D.; Cellini, M.A.; Stephenson, D.G. Different Ca2+ Releasing Action of Caffeine and Depolarisation in Skeletal Muscle Fibres of the Rat. J. Physiol. 2001, 531, 715–728. [Google Scholar] [CrossRef]
- Lamb, G.D.; Stephenson, D.G. Effects of Intracellular PH and [Mg2+] on Excitation-Contraction Coupling in Skeletal Muscle Fibres of the Rat. J. Physiol. 1994, 478 Pt 2, 331–339. [Google Scholar] [CrossRef]
- Cully, T.R.; Edwards, J.N.; Launikonis, B.S. Activation and Propagation of Ca2+ Release from inside the Sarcoplasmic Reticulum Network of Mammalian Skeletal Muscle: Ca2+ Waves in Mammalian Muscle. J. Physiol. 2014, 592, 3727–3746. [Google Scholar] [CrossRef]
- Edwards, J.N.; Murphy, R.M.; Cully, T.R.; von Wegner, F.; Friedrich, O.; Launikonis, B.S. Ultra-Rapid Activation and Deactivation of Store-Operated Ca2+ Entry in Skeletal Muscle. Cell Calcium 2010, 47, 458–467. [Google Scholar] [CrossRef]
- Li, H.; Ding, X.; Lopez, J.R.; Takeshima, H.; Ma, J.; Allen, P.D.; Eltit, J.M. Impaired Orai1-Mediated Resting Ca2+ Entry Reduces the Cytosolic [Ca 2+ ] and Sarcoplasmic Reticulum Ca2+ Loading in Quiescent Junctophilin 1 Knock-out Myotubes. J. Biol. Chem. 2010, 285, 39171–39179. [Google Scholar] [CrossRef]
- Koenig, X.; Choi, R.H.; Schicker, K.; Singh, D.P.; Hilber, K.; Launikonis, B.S. Mechanistic Insights into Store-Operated Ca2+ Entry during Excitation-Contraction Coupling in Skeletal Muscle. Biochim. Biophys. Acta (BBA)—Mol. Cell Res. 2019, 1866, 1239–1248. [Google Scholar] [CrossRef] [PubMed]
- Rudolf, R.; Magalhães, P.J.; Pozzan, T. Direct in Vivo Monitoring of Sarcoplasmic Reticulum Ca2+ and Cytosolic CAMP Dynamics in Mouse Skeletal Muscle. J. Cell. Biol. 2006, 173, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Lamb, G.D.; Stephenson, D.G. Effect of Mg2+ on the Control of Ca2+ Release in Skeletal Muscle Fibres of the Toad. J. Physiol. 1991, 434, 507–528. [Google Scholar] [CrossRef] [PubMed]
- Choi, R.H.; Koenig, X.; Launikonis, B.S. Dantrolene Requires Mg2+ to Arrest Malignant Hyperthermia. Proc. Natl. Acad. Sci. USA 2017, 114, 4811–4815. [Google Scholar] [CrossRef] [PubMed]
- Manno, C.; Sztretye, M.; Figueroa, L.; Allen, P.D.; Ríos, E. Dynamic Measurement of the Calcium Buffering Properties of the Sarcoplasmic Reticulum in Mouse Skeletal Muscle: Ca2+ Buffering in the SR. J. Physiol. 2013, 591, 423–442. [Google Scholar] [CrossRef] [PubMed]
- Luik, R.M.; Wang, B.; Prakriya, M.; Wu, M.M.; Lewis, R.S. Oligomerization of STIM1 Couples ER Calcium Depletion to CRAC Channel Activation. Nature 2008, 454, 538–542. [Google Scholar] [CrossRef]
- Soboloff, J.; Rothberg, B.S.; Madesh, M.; Gill, D.L. STIM Proteins: Dynamic Calcium Signal Transducers. Nat. Rev. Mol. Cell. Biol. 2012, 13, 549–565. [Google Scholar] [CrossRef]
- MacLennan, D.H.; Wong, P.T.S. Isolation of a Calcium-Sequestering Protein from Sarcoplasmic Reticulum. Proc. Natl. Acad. Sci. USA 1971, 68, 1231–1235. [Google Scholar] [CrossRef]
- Ikemoto, N.; Bhatnagar, G.M.; Nagy, B.; Gergely, J. Interaction of Divalent Cations with the 55,000-Dalton Protein Component of the Sarcoplasmic Reticulum: Studies of fluorescence and circular dichroism. J. Biol. Chem. 1972, 247, 7835–7837. [Google Scholar] [CrossRef]
- Murphy, R.M.; Larkins, N.T.; Mollica, J.P.; Beard, N.A.; Lamb, G.D. Calsequestrin Content and SERCA Determine Normal and Maximal Ca2+ Storage Levels in Sarcoplasmic Reticulum of Fast- and Slow-Twitch Fibres of Rat. J. Physiol. 2009, 587, 443–460. [Google Scholar] [CrossRef]
- Beard, N.A.; Laver, D.R.; Dulhunty, A.F. Calsequestrin and the Calcium Release Channel of Skeletal and Cardiac Muscle. Prog. Biophys. Mol. Biol 2004, 85, 33–69. [Google Scholar] [CrossRef]
- Royer, L.; Ríos, E. Deconstructing Calsequestrin. Complex Buffering in the Calcium Store of Skeletal Muscle. J. Physiol. 2009, 587, 3101–3111. [Google Scholar] [CrossRef]
- Woo, J.S.; Jeong, S.Y.; Park, J.H.; Choi, J.H.; Lee, E.H. Calsequestrin: A Well-Known but Curious Protein in Skeletal Muscle. Exp. Mol. Med. 2020, 52, 1908–1925. [Google Scholar] [CrossRef]
- MacLennan, D.H.; Reithmeier, R.A. Ion Tamers. Nat. Struct. Biol. 1998, 5, 409–411. [Google Scholar] [CrossRef]
- Manno, C.; Figueroa, L.C.; Gillespie, D.; Fitts, R.; Kang, C.; Franzini-Armstrong, C.; Rios, E. Calsequestrin Depolymerizes When Calcium Is Depleted in the Sarcoplasmic Reticulum of Working Muscle. Proc. Natl. Acad. Sci. USA 2017, 114, E638–E647. [Google Scholar] [CrossRef]
- Ivarsson, N.; Mattsson, C.M.; Cheng, A.J.; Bruton, J.D.; Ekblom, B.; Lanner, J.T.; Westerblad, H. SR Ca2+ Leak in Skeletal Muscle Fibers Acts as an Intracellular Signal to Increase Fatigue Resistance. J. Gen. Physiol. 2019, 151, 567–577. [Google Scholar] [CrossRef]
- Bellinger, A.M.; Reiken, S.; Carlson, C.; Mongillo, M.; Liu, X.; Rothman, L.; Matecki, S.; Lacampagne, A.; Marks, A.R. Hypernitrosylated Ryanodine Receptor Calcium Release Channels Are Leaky in Dystrophic Muscle. Nat. Med. 2009, 15, 325–330. [Google Scholar] [CrossRef]
- Reddish, F.N.; Miller, C.L.; Deng, X.; Dong, B.; Patel, A.A.; Ghane, M.A.; Mosca, B.; McBean, C.; Wu, S.; Solntsev, K.M.; et al. Rapid Subcellular Calcium Responses and Dynamics by Calcium Sensor G-CatchER+. iScience 2021, 24, 102129. [Google Scholar] [CrossRef]
- Carrell, E.M.; Coppola, A.R.; McBride, H.J.; Dirksen, R.T. Orai1 Enhances Muscle Endurance by Promoting Fatigue-Resistant Type I Fiber Content but Not through Acute Store-Operated Ca2+ Entry. FASEB J. 2016, 30, 4109–4119. [Google Scholar] [CrossRef]
- Lee, E.H.; Cherednichenko, G.; Pessah, I.N.; Allen, P.D. Functional Coupling between TRPC3 and RyR1 Regulates the Expressions of Key Triadic Proteins. J. Biol. Chem. 2006, 281, 10042–10048. [Google Scholar] [CrossRef]
- Dirksen, R.T. Checking Your SOCCs and Feet: The Molecular Mechanisms of Ca2+ Entry in Skeletal Muscle: Ca2+ Entry in Skeletal Muscle. J. Physiol. 2009, 587, 3139–3147. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Erxleben, C.; Abramowitz, J.; Flockerzi, V.; Zhu, M.X.; Armstrong, D.L.; Birnbaumer, L. Functional Interactions among Orai1, TRPCs, and STIM1 Suggest a STIM-Regulated Heteromeric Orai/TRPC Model for SOCE/Icrac Channels. Proc. Natl. Acad. Sci. USA 2008, 105, 2895–2900. [Google Scholar] [CrossRef] [PubMed]
- Antigny, F.; Sabourin, J.; Saüc, S.; Bernheim, L.; Koenig, S.; Frieden, M. TRPC1 and TRPC4 Channels Functionally Interact with STIM1L to Promote Myogenesis and Maintain Fast Repetitive Ca2+ Release in Human Myotubes. Biochim. Biophys. Acta (BBA)—Mol. Cell Res. 2017, 1864, 806–813. [Google Scholar] [CrossRef] [PubMed]
- Cheng, K.T.; Liu, X.; Ong, H.L.; Ambudkar, I.S. Functional Requirement for Orai1 in Store-Operated TRPC1-STIM1 Channels. J. Biol. Chem. 2008, 283, 12935–12940. [Google Scholar] [CrossRef] [PubMed]
- Ong, H.L.; Cheng, K.T.; Liu, X.; Bandyopadhyay, B.C.; Paria, B.C.; Soboloff, J.; Pani, B.; Gwack, Y.; Srikanth, S.; Singh, B.B.; et al. Dynamic Assembly of TRPC1-STIM1-Orai1 Ternary Complex Is Involved in Store-Operated Calcium Influx. Evidence for Similarities in Store-Operated and Calcium Release-Activated Calcium Channel Components. J. Biol. Chem. 2007, 282, 9105–9116. [Google Scholar] [CrossRef]
- Jardin, I.; Lopez, J.J.; Salido, G.M.; Rosado, J.A. Orai1 Mediates the Interaction between STIM1 and HTRPC1 and Regulates the Mode of Activation of HTRPC1-Forming Ca2+ Channels. J. Biol. Chem. 2008, 283, 25296–25304. [Google Scholar] [CrossRef]
- Berridge, M.J. Capacitative Calcium Entry. Biochem. J. 1995, 11, 312. [Google Scholar] [CrossRef]
- Sampieri, A.; Diaz-Muñoz, M.; Antaramian, A.; Vaca, L. The Foot Structure from the Type 1 Ryanodine Receptor Is Required for Functional Coupling to Store-Operated Channels. J. Biol. Chem. 2005, 280, 24804–24815. [Google Scholar] [CrossRef]
- Kiselyov, K.I.; Shin, D.M.; Wang, Y.; Pessah, I.N.; Allen, P.D.; Muallem, S. Gating of Store-Operated Channels by Conformational Coupling to Ryanodine Receptors. Mol. Cell. 2000, 6, 421–431. [Google Scholar] [CrossRef]
- Estrada, M.; Espinosa, A.; Gibson, C.J.; Uhlen, P.; Jaimovich, E. Capacitative Calcium Entry in Testosterone-Induced Intracellular Calcium Oscillations in Myotubes. J. Endocrinol. 2005, 184, 371–379. [Google Scholar] [CrossRef]
- Powell, J.A.; Carrasco, M.A.; Adams, D.S.; Drouet, B.; Rios, J.; Müller, M.; Estrada, M.; Jaimovich, E. IP(3) Receptor Function and Localization in Myotubes: An Unexplored Ca2+ Signaling Pathway in Skeletal Muscle. J. Cell. Sci. 2001, 114, 3673–3683. [Google Scholar] [CrossRef]
- Lilliu, E.; Hilber, K.; Launikonis, B.S.; Koenig, X. Phasic Store-Operated Ca2+ Entry During Excitation-Contraction Coupling in Skeletal Muscle Fibers From Exercised Mice. Front. Physiol. 2020, 11, 597647. [Google Scholar] [CrossRef]
- Baylor, S.M.; Hollingworth, S. Sarcoplasmic Reticulum Calcium Release Compared in Slow-Twitch and Fast-Twitch Fibres of Mouse Muscle. J. Physiol. 2003, 551, 125–138. [Google Scholar] [CrossRef]
- Adams, R.J.; Schwartz, A. Comparative Mechanisms for Contraction of Cardiac and Skeletal Muscle. Chest 1980, 78, 123–139. [Google Scholar] [CrossRef]
- Vergara, J.L.; DiFranco, M.; Novo, D. Dimensions of Calcium Release Domains in Frog Skeletal Muscle Fibers; Bearman, G.H., Bornhop, D.J., Levenson, R.M., Eds.; International Society for Optics and Photonics: San Jose, CA, USA, 2001; pp. 133–143. [Google Scholar] [CrossRef]
- Darbellay, B.; Arnaudeau, S.; Bader, C.R.; Konig, S.; Bernheim, L. STIM1L Is a New Actin-Binding Splice Variant Involved in Fast Repetitive Ca2+ Release. J. Cell. Biol. 2011, 194, 335–346. [Google Scholar] [CrossRef]
- Merritt, J.E.; Armstrong, W.P.; Benham, C.D.; Hallam, T.J.; Jacob, R.; Jaxa-Chamiec, A.; Leigh, B.K.; McCarthy, S.A.; Moores, K.E.; Rink, T.J. SK&F 96365, a Novel Inhibitor of Receptor-Mediated Calcium Entry. Biochem. J. 1990, 271, 515–522. [Google Scholar] [CrossRef]
- Singh, A.; Hildebrand, M.E.; Garcia, E.; Snutch, T.P. The Transient Receptor Potential Channel Antagonist SKF96365 Is a Potent Blocker of Low-Voltage-Activated T-Type Calcium Channels. Br. J. Pharmacol. 2010, 160, 1464–1475. [Google Scholar] [CrossRef]
- Maruyama, T.; Kanaji, T.; Nakade, S.; Kanno, T.; Mikoshiba, K. 2APB, 2-Aminoethoxydiphenyl Borate, a Membrane-Penetrable Modulator of Ins(1,4,5)P3-Induced Ca2+ Release. J. Biochem. 1997, 122, 498–505. [Google Scholar] [CrossRef]
- Djuric, S.W.; BaMaung, N.Y.; Basha, A.; Liu, H.; Luly, J.R.; Madar, D.J.; Sciotti, R.J.; Tu, N.P.; Wagenaar, F.L.; Wiedeman, P.E.; et al. 3,5-Bis(Trifluoromethyl)Pyrazoles: A Novel Class of NFAT Transcription Factor Regulator. J. Med. Chem. 2000, 43, 2975–2981. [Google Scholar] [CrossRef]
- Trevillyan, J.M.; Chiou, X.G.; Chen, Y.W.; Ballaron, S.J.; Sheets, M.P.; Smith, M.L.; Wiedeman, P.E.; Warrior, U.; Wilkins, J.; Gubbins, E.J.; et al. Potent Inhibition of NFAT Activation and T Cell Cytokine Production by Novel Low Molecular Weight Pyrazole Compounds. J. Biol. Chem. 2001, 276, 48118–48126. [Google Scholar] [CrossRef]
- Chen, Y.-W.; Smith, M.L.; Chiou, G.X.; Ballaron, S.; Sheets, M.P.; Gubbins, E.; Warrior, U.; Wilkins, J.; Surowy, C.; Nakane, M.; et al. TH1 and TH2 Cytokine Inhibition by 3,5-Bis(Trifluoromethyl)Pyrazoles, a Novel Class of Immunomodulators. Cell. Immunol. 2002, 220, 134–142. [Google Scholar] [CrossRef]
- Putney, J.W. Pharmacology of Store-Operated Calcium Channels. Mol. Interv. 2010, 10, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Bakowski, D.; Murray, F.; Parekh, A.B. Store-Operated Ca2+ Channels: Mechanism, Function, Pharmacology, and Therapeutic Targets. Annu. Rev. Pharmacol. Toxicol. 2021, 61, 629–654. [Google Scholar] [CrossRef] [PubMed]
- Tian, C.; Du, L.; Zhou, Y.; Li, M. Store-Operated CRAC Channel Inhibitors: Opportunities and Challenges. Future Med. Chem. 2016, 8, 817–832. [Google Scholar] [CrossRef]
- Zhang, X.; Xin, P.; Yoast, R.E.; Emrich, S.M.; Johnson, M.T.; Pathak, T.; Benson, J.C.; Azimi, I.; Gill, D.L.; Monteith, G.R.; et al. Distinct Pharmacological Profiles of ORAI1, ORAI2, and ORAI3 Channels. Cell Calcium 2020, 91, 102281. [Google Scholar] [CrossRef]
- Vassilopoulos, S.; Brocard, J.; Garcia, L.; Marty, I.; Bouron, A. Retrograde Regulation of Store-Operated Calcium Channels by the Ryanodine Receptor-Associated Protein Triadin 95 in Rat Skeletal Myotubes. Cell Calcium 2007, 41, 179–185. [Google Scholar] [CrossRef]
- Eltit, J.M.; Ding, X.; Pessah, I.N.; Allen, P.D.; Lopez, J.R. Nonspecific Sarcolemmal Cation Channels Are Critical for the Pathogenesis of Malignant Hyperthermia. FASEB J. 2013, 27, 991–1000. [Google Scholar] [CrossRef]
- Gutierrez-Martin, Y.; Martin-Romero, F.J.; Henao, F. Store-Operated Calcium Entry in Differentiated C2C12 Skeletal Muscle Cells. Biochim. Biophys. Acta 2005, 1711, 33–40. [Google Scholar] [CrossRef][Green Version]
- Vazquez, G.; de Boland, A.R.; Boland, R.L. 1alpha,25-Dihydroxy-Vitamin-D3-Induced Store-Operated Ca2+ Influx in Skeletal Muscle Cells. Modulation by Phospholipase c, Protein Kinase c, and Tyrosine Kinases. J. Biol. Chem. 1998, 273, 33954–33960. [Google Scholar] [CrossRef]
- Reichling, D.B.; MacDermott, A.B. Lanthanum Actions on Excitatory Amino Acid-Gated Currents and Voltage-Gated Calcium Currents in Rat Dorsal Horn Neurons. J. Physiol. 1991, 441, 199–218. [Google Scholar] [CrossRef]
- Leffler, A.; Linte, R.M.; Nau, C.; Reeh, P.; Babes, A. A High-Threshold Heat-Activated Channel in Cultured Rat Dorsal Root Ganglion Neurons Resembles TRPV2 and Is Blocked by Gadolinium. Eur. J. Neurosci. 2007, 26, 12–22. [Google Scholar] [CrossRef]
- Oz, M.; Tchugunova, Y.B.; Dunn, S.M. Direct Inhibition of Voltage-Dependent Ca2+ Fluxes by Ethanol and Higher Alcohols in Rabbit T-Tubule Membranes. Eur. J. Pharmacol. 2001, 418, 169–176. [Google Scholar] [CrossRef]
- Lee, E.H.; Lopez, J.R.; Li, J.; Protasi, F.; Pessah, I.N.; Kim, D.H.; Allen, P.D. Conformational Coupling of DHPR and RyR1 in Skeletal Myotubes Is Influenced by Long-Range Allosterism: Evidence for a Negative Regulatory Module. Am. J. Physiol. Cell. Physiol. 2004, 286, C179–C189. [Google Scholar] [CrossRef][Green Version]
- Mosca, B.; Eckhardt, J.; Bergamelli, L.; Treves, S.; Bongianino, R.; De Negri, M.; Priori, S.G.; Protasi, F.; Zorzato, F. Role of the JP45-Calsequestrin Complex on Calcium Entry in Slow Twitch Skeletal Muscles. J. Biol. Chem. 2016, 291, 14555–14565. [Google Scholar] [CrossRef]
- Bannister, R.A.; Pessah, I.N.; Beam, K.G. The Skeletal L-Type Ca 2+ Current Is a Major Contributor to Excitation-Coupled Ca 2+ Entry. J. Gen. Physiol. 2009, 133, 79–91. [Google Scholar] [CrossRef]
- Chung, S.C.; McDonald, T.V.; Gardner, P. Inhibition by SK&F 96365 of Ca2+ Current, IL-2 Production and Activation in T Lymphocytes. Br. J. Pharmacol. 1994, 113, 861–868. [Google Scholar]
- Olivera, J.F.; Fernando Olivera, J.; Pizarro, G. Two Inhibitors of Store Operated Ca2+ Entry Suppress Excitation Contraction Coupling in Frog Skeletal Muscle. J. Muscle Res. Cell. Motil. 2010, 31, 127–139. [Google Scholar] [CrossRef]
- Ho, T.C.; Horn, N.A.; Huynh, T.; Kelava, L.; Lansman, J.B. Evidence TRPV4 Contributes to Mechanosensitive Ion Channels in Mouse Skeletal Muscle Fibers. Channels 2012, 6, 246–254. [Google Scholar] [CrossRef]
- Tanahashi, Y.; Wang, B.; Murakami, Y.; Unno, T.; Matsuyama, H.; Nagano, H.; Komori, S. Inhibitory Effects of SKF96365 on the Activities of K(+) Channels in Mouse Small Intestinal Smooth Muscle Cells. J. Vet. Med. Sci. 2016, 78, 203–211. [Google Scholar] [CrossRef]
- Iouzalen, L.; Lantoine, F.; Pernollet, M.G.; Millanvoye-Van Brussel, E.; Devynck, M.A.; David-Dufilho, M. SK&F 96365 Inhibits Intracellular Ca2+ Pumps and Raises Cytosolic Ca2+ Concentration without Production of Nitric Oxide and von Willebrand Factor. Cell Calcium 1996, 20, 501–508. [Google Scholar] [CrossRef]
- Song, M.; Chen, D.; Yu, S.P. The TRPC Channel Blocker SKF 96365 Inhibits Glioblastoma Cell Growth by Enhancing Reverse Mode of the Na(+) /Ca(2+) Exchanger and Increasing Intracellular Ca(2+). Br. J. Pharmacol. 2014, 171, 3432–3447. [Google Scholar] [CrossRef] [PubMed]
- Cherednichenko, G.; Hurne, A.M.; Fessenden, J.D.; Lee, E.H.; Allen, P.D.; Beam, K.G.; Pessah, I.N. Conformational Activation of Ca2+ Entry by Depolarization of Skeletal Myotubes. Proc. Natl. Acad. Sci. USA 2004, 101, 15793–15798. [Google Scholar] [CrossRef] [PubMed]
- Lanner, J.T.; Katz, A.; Tavi, P.; Sandström, M.E.; Zhang, S.-J.; Wretman, C.; James, S.; Fauconnier, J.; Lännergren, J.; Bruton, J.D.; et al. The Role of Ca2+ Influx for Insulin-Mediated Glucose Uptake in Skeletal Muscle. Diabetes 2006, 55, 2077–2083. [Google Scholar] [CrossRef] [PubMed]
- Juretić, N.; Jorquera, G.; Caviedes, P.; Jaimovich, E.; Riveros, N. Electrical Stimulation Induces Calcium-Dependent up-Regulation of Neuregulin-1β in Dystrophic Skeletal Muscle Cell Lines. Cell Physiol. Biochem. 2012, 29, 919–930. [Google Scholar] [CrossRef]
- Missiaen, L.; Callewaert, G.; De Smedt, H.; Parys, J.B. 2-Aminoethoxydiphenyl Borate Affects the Inositol 1,4,5-Trisphosphate Receptor, the Intracellular Ca2+ Pump and the Non-Specific Ca2+ Leak from the Non-Mitochondrial Ca2+ Stores in Permeabilized A7r5 Cells. Cell Calcium 2001, 29, 111–116. [Google Scholar] [CrossRef]
- Carrasco, M.A.; Riveros, N.; Ríos, J.; Müller, M.; Torres, F.; Pineda, J.; Lantadilla, S.; Jaimovich, E. Depolarization-Induced Slow Calcium Transients Activate Early Genes in Skeletal Muscle Cells. Am. J. Physiol. Cell. Physiol. 2003, 284, C1438–C1447. [Google Scholar] [CrossRef]
- Liberona, J.L.; Cárdenas, J.C.; Reyes, R.; Hidalgo, J.; Molgó, J.; Jaimovich, E. Sodium-Dependent Action Potentials Induced by Brevetoxin-3 Trigger Both IP3 Increase and Intracellular Ca2+ Release in Rat Skeletal Myotubes. Cell. Calcium. 2008, 44, 289–297. [Google Scholar] [CrossRef]
- Gregory, R.B.; Rychkov, G.; Barritt, G.J. Evidence That 2-Aminoethyl Diphenylborate Is a Novel Inhibitor of Store-Operated Ca2+ Channels in Liver Cells, and Acts through a Mechanism Which Does Not Involve Inositol Trisphosphate Receptors. Biochem. J. 2001, 354, 285–290. [Google Scholar] [CrossRef]
- Prakriya, M.; Lewis, R.S. Potentiation and Inhibition of Ca(2+) Release-Activated Ca(2+) Channels by 2-Aminoethyldiphenyl Borate (2-APB) Occurs Independently of IP(3) Receptors. J. Physiol. 2001, 536, 3–19. [Google Scholar] [CrossRef]
- Bootman, M.D.; Collins, T.J.; Mackenzie, L.; Roderick, H.L.; Berridge, M.J.; Peppiatt, C.M. 2-Aminoethoxydiphenyl Borate (2-APB) Is a Reliable Blocker of Store-Operated Ca2+ Entry but an Inconsistent Inhibitor of InsP3-Induced Ca2+ Release. Faseb J. 2002, 16, 1145–1150. [Google Scholar] [CrossRef]
- DeHaven, W.I.; Smyth, J.T.; Boyles, R.R.; Bird, G.S.; Putney, J.W. Complex Actions of 2-Aminoethyldiphenyl Borate on Store-Operated Calcium Entry. J. Biol. Chem. 2008, 283, 19265–19273. [Google Scholar] [CrossRef]
- Peinelt, C.; Lis, A.; Beck, A.; Fleig, A.; Penner, R. 2-Aminoethoxydiphenyl Borate Directly Facilitates and Indirectly Inhibits STIM1-Dependent Gating of CRAC Channels. J. Physiol. 2008, 586, 3061–3073. [Google Scholar] [CrossRef]
- Lis, A.; Peinelt, C.; Beck, A.; Parvez, S.; Monteilh-Zoller, M.; Fleig, A.; Penner, R. CRACM1, CRACM2, and CRACM3 Are Store-Operated Ca2+ Channels with Distinct Functional Properties. Curr. Biol. 2007, 17, 794–800. [Google Scholar] [CrossRef]
- Ishikawa, J.; Ohga, K.; Yoshino, T.; Takezawa, R.; Ichikawa, A.; Kubota, H.; Yamada, T. A Pyrazole Derivative, YM-58483, Potently Inhibits Store-Operated Sustained Ca2+ Influx and IL-2 Production in T Lymphocytes. J. Immunol. 2003, 170, 4441–4449. [Google Scholar] [CrossRef]
- Zitt, C.; Strauss, B.; Schwarz, E.C.; Spaeth, N.; Rast, G.; Hatzelmann, A.; Hoth, M. Potent Inhibition of Ca2+ Release-Activated Ca2+ Channels and T-Lymphocyte Activation by the Pyrazole Derivative BTP2. J. Biol. Chem. 2004, 279, 12427–12437. [Google Scholar] [CrossRef]
- Steinckwich, N.; Frippiat, J.-P.; Stasia, M.-J.; Erard, M.; Boxio, R.; Tankosic, C.; Doignon, I.; Nüsse, O. Potent Inhibition of Store-Operated Ca2+ Influx and Superoxide Production in HL60 Cells and Polymorphonuclear Neutrophils by the Pyrazole Derivative BTP2. J. Leukoc. Biol. 2007, 81, 1054–1064. [Google Scholar] [CrossRef]
- Takezawa, R.; Cheng, H.; Beck, A.; Ishikawa, J.; Launay, P.; Kubota, H.; Kinet, J.-P.; Fleig, A.; Yamada, T.; Penner, R. A Pyrazole Derivative Potently Inhibits Lymphocyte Ca2+ Influx and Cytokine Production by Facilitating Transient Receptor Potential Melastatin 4 Channel Activity. Mol. Pharmacol. 2006, 69, 1413–1420. [Google Scholar] [CrossRef]
- He, L.-P.; Hewavitharana, T.; Soboloff, J.; Spassova, M.A.; Gill, D.L. A Functional Link between Store-Operated and TRPC Channels Revealed by the 3,5-Bis(Trifluoromethyl)Pyrazole Derivative, BTP2. J. Biol. Chem. 2005, 280, 10997–11006. [Google Scholar] [CrossRef]
- Thornton, A.M.; Zhao, X.; Weisleder, N.; Brotto, L.S.; Bougoin, S.; Nosek, T.M.; Reid, M.; Hardin, B.; Pan, Z.; Ma, J.; et al. Store-Operated Ca2+ Entry (SOCE) Contributes to Normal Skeletal Muscle Contractility in Young but Not in Aged Skeletal Muscle. Aging 2011, 3, 621–634. [Google Scholar] [CrossRef]
- Zhao, X.; Moloughney, J.G.; Zhang, S.; Komazaki, S.; Weisleder, N. Orai1 Mediates Exacerbated Ca2+ Entry in Dystrophic Skeletal Muscle. PLoS ONE 2012, 7, 13. [Google Scholar] [CrossRef]
- Meizoso-Huesca, A.; Launikonis, B.S. The Orai1 Inhibitor BTP2 Has Multiple Effects on Ca2+ Handling in Skeletal Muscle. J. Gen. Physiol. 2021, 153, e202012747. [Google Scholar] [CrossRef]
- Ashmole, I.; Duffy, S.M.; Leyland, M.L.; Morrison, V.S.; Begg, M.; Bradding, P. CRACM/Orai Ion Channel Expression and Function in Human Lung Mast Cells. J. Allergy Clin. Immunol. 2012, 129, 1628–1635.e2. [Google Scholar] [CrossRef]
- Derler, I.; Schindl, R.; Fritsch, R.; Heftberger, P.; Riedl, M.C.; Begg, M.; House, D.; Romanin, C. The Action of Selective CRAC Channel Blockers Is Affected by the Orai Pore Geometry. Cell Calcium 2013, 53, 139–151. [Google Scholar] [CrossRef]
- Rice, L.V.; Bax, H.J.; Russell, L.J.; Barrett, V.J.; Walton, S.E.; Deakin, A.M.; Thomson, S.A.; Lucas, F.; Solari, R.; House, D.; et al. Characterization of Selective Calcium-Release Activated Calcium Channel Blockers in Mast Cells and T-Cells from Human, Rat, Mouse and Guinea-Pig Preparations. Eur. J. Pharmacol. 2013, 704, 49–57. [Google Scholar] [CrossRef]
- Bulla, M.; Gyimesi, G.; Kim, J.H.; Bhardwaj, R.; Hediger, M.A.; Frieden, M.; Demaurex, N. ORAI1 Channel Gating and Selectivity Is Differentially Altered by Natural Mutations in the First or Third Transmembrane Domain. J. Physiol. 2019, 597, 561–582. [Google Scholar] [CrossRef]
- Waldherr, L.; Tiffner, A.; Mishra, D.; Sallinger, M.; Schober, R.; Frischauf, I.; Schmidt, T.; Handl, V.; Sagmeister, P.; Köckinger, M.; et al. Blockage of Store-Operated Ca2+ Influx by Synta66 Is Mediated by Direct Inhibition of the Ca2+ Selective Orai1 Pore. Cancers 2020, 12, 2876. [Google Scholar] [CrossRef]
- Li, J.; McKeown, L.; Ojelabi, O.; Stacey, M.; Foster, R.; O’Regan, D.; Porter, K.E.; Beech, D.J. Nanomolar Potency and Selectivity of a Ca2+ Release-Activated Ca2+ Channel Inhibitor against Store-Operated Ca2+ Entry and Migration of Vascular Smooth Muscle Cells. Br. J. Pharmacol. 2011, 164, 382–393. [Google Scholar] [CrossRef]
- Di Sabatino, A.; Rovedatti, L.; Kaur, R.; Spencer, J.P.; Brown, J.T.; Morisset, V.D.; Biancheri, P.; Leakey, N.A.B.; Wilde, J.I.; Scott, L.; et al. Targeting Gut T Cell Ca2+ Release-Activated Ca2+ Channels Inhibits T Cell Cytokine Production and T-Box Transcription Factor T-Bet in Inflammatory Bowel Disease. J. Immunol. 2009, 183, 3454–3462. [Google Scholar] [CrossRef]
- Riva, B.; Griglio, A.; Serafini, M.; Cordero-Sanchez, C.; Aprile, S.; Di Paola, R.; Gugliandolo, E.; Alansary, D.; Biocotino, I.; Lim, D.; et al. Pyrtriazoles, a Novel Class of Store-Operated Calcium Entry Modulators: Discovery, Biological Profiling, and in Vivo Proof-of-Concept Efficacy in Acute Pancreatitis. J. Med. Chem. 2018, 61, 9756–9783. [Google Scholar] [CrossRef]
- Serafini, M.; Cordero-Sanchez, C.; Di Paola, R.; Bhela, I.P.; Aprile, S.; Purghè, B.; Fusco, R.; Cuzzocrea, S.; Genazzani, A.A.; Riva, B.; et al. Store-Operated Calcium Entry as a Therapeutic Target in Acute Pancreatitis: Discovery and Development of Drug-Like SOCE Inhibitors. J. Med. Chem. 2020, 63, 14761–14779. [Google Scholar] [CrossRef]
- Azimi, I.; Stevenson, R.J.; Zhang, X.; Meizoso-Huesca, A.; Xin, P.; Johnson, M.; Flanagan, J.U.; Chalmers, S.B.; Yoast, R.E.; Kapure, J.S.; et al. A New Selective Pharmacological Enhancer of the Orai1 Ca2+ Channel Reveals Roles for Orai1 in Smooth and Skeletal Muscle Functions. ACS Pharmacol. Transl. Sci. 2020, 3, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Thomas, N.L.; Williams, A.J. Pharmacology of Ryanodine Receptors and Ca2+-Induced Ca2+ Release. Wires Membr. Transp. Signal. 2012, 1, 383–397. [Google Scholar] [CrossRef]
- Horinouchi, T.; Higashi, T.; Higa, T.; Terada, K.; Mai, Y.; Aoyagi, H.; Hatate, C.; Nepal, P.; Horiguchi, M.; Harada, T.; et al. Different Binding Property of STIM1 and Its Novel Splice Variant STIM1L to Orai1, TRPC3, and TRPC6 Channels. Biochem. Biophys. Res. Commun. 2012, 428, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Saüc, S.; Bulla, M.; Nunes, P.; Orci, L.; Marchetti, A.; Antigny, F.; Bernheim, L.; Cosson, P.; Frieden, M.; Demaurex, N. STIM1L Traps and Gates Orai1 Channels without Remodeling the Cortical ER. J. Cell. Sci. 2015, jcs.164228. [Google Scholar] [CrossRef]
- Dyrda, A.; Koenig, S.; Frieden, M. STIM1 Long and STIM1 Gate Differently TRPC1 during Store-Operated Calcium Entry. Cell Calcium 2020, 86, 102134. [Google Scholar] [CrossRef]
- Antigny, F.; Koenig, S.; Bernheim, L.; Frieden, M. During Post-Natal Human Myogenesis, Normal Myotube Size Requires TRPC1- and TRPC4-Mediated Ca2+ Entry. J. Cell. Sci. 2013, 126, 2525–2533. [Google Scholar] [CrossRef]
- Saüc, S.; Frieden, M. Neurological and Motor Disorders: TRPC in the Skeletal Muscle. Adv. Exp. Med. Biol. 2017, 993, 557–575. [Google Scholar] [CrossRef]
- Darbellay, B.; Arnaudeau, S.; Ceroni, D.; Bader, C.R.; Konig, S.; Bernheim, L. Human Muscle Economy Myoblast Differentiation and Excitation-Contraction Coupling Use the Same Molecular Partners, STIM1 and STIM2. J. Biol. Chem. 2010, 285, 22437–22447. [Google Scholar] [CrossRef]
- Brandman, O.; Liou, J.; Park, W.S.; Meyer, T. STIM2 Is a Feedback Regulator That Stabilizes Basal Cytosolic and Endoplasmic Reticulum Ca2+ Levels. Cell 2007, 131, 1327–1339. [Google Scholar] [CrossRef]
- Berna-Erro, A.; Braun, A.; Kraft, R.; Kleinschnitz, C.; Schuhmann, M.K.; Stegner, D.; Wultsch, T.; Eilers, J.; Meuth, S.G.; Stoll, G.; et al. STIM2 Regulates Capacitive Ca2+ Entry in Neurons and Plays a Key Role in Hypoxic Neuronal Cell Death. Sci. Signal. 2009, 2, ra67. [Google Scholar] [CrossRef]
- Oh, M.R.; Lee, K.J.; Huang, M.; Kim, J.O.; Kim, D.H.; Cho, C.-H.; Lee, E.H. STIM2 Regulates Both Intracellular Ca2+ Distribution and Ca2+ Movement in Skeletal Myotubes. Sci. Rep. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Rana, A.; Yen, M.; Sadaghiani, A.M.; Malmersjö, S.; Park, C.Y.; Dolmetsch, R.E.; Lewis, R.S. Alternative Splicing Converts STIM2 from an Activator to an Inhibitor of Store-Operated Calcium Channels. J. Cell. Biol. 2015, 209, 653–670. [Google Scholar] [CrossRef]
- Miederer, A.-M.; Alansary, D.; Schwär, G.; Lee, P.-H.; Jung, M.; Helms, V.; Niemeyer, B.A. A STIM2 Splice Variant Negatively Regulates Store-Operated Calcium Entry. Nat. Commun. 2015, 6. [Google Scholar] [CrossRef]
- Kim, K.M.; Rana, A.; Park, C.Y. Orai1 Inhibitor STIM2β Regulates Myogenesis by Controlling SOCE Dependent Transcriptional Factors. Sci. Rep. 2019, 9, 10794. [Google Scholar] [CrossRef]
- Böhm, J.; Laporte, J. Gain-of-Function Mutations in STIM1 and ORAI1 Causing Tubular Aggregate Myopathy and Stormorken Syndrome. Cell Calcium 2018, 76, 1–9. [Google Scholar] [CrossRef]
- Eberstein, A.; Goodgold, J. Slow and Fast Twitch Fibers in Human Skeletal Muscle. Am. J. Physiol.—Leg. Content 1968, 215, 535–541. [Google Scholar] [CrossRef]
- Schiaffino, S.; Rossi, A.C.; Smerdu, V.; Leinwand, L.A.; Reggiani, C. Developmental Myosins: Expression Patterns and Functional Significance. Skelet. Muscle 2015, 5, 22. [Google Scholar] [CrossRef]
- Calderón, J.C.; Bolaños, P.; Caputo, C. Myosin Heavy Chain Isoform Composition and Ca(2+) Transients in Fibres from Enzymatically Dissociated Murine Soleus and Extensor Digitorum Longus Muscles. J. Physiol. 2010, 588, 267–279. [Google Scholar] [CrossRef]
- Luff, A.R.; Atwood, H.L. Changes in the Sarcoplasmic Reticulum and Transverse Tubular System of Fast and Slow Skeletal Muscles of the Mouse during Postnatal Development. J. Cell. Biol. 1971, 51, 369–383. [Google Scholar] [CrossRef]
- McCarl, C.-A.; Picard, C.; Khalil, S.; Kawasaki, T.; Röther, J.; Papolos, A.; Kutok, J.; Hivroz, C.; LeDeist, F.; Plogmann, K.; et al. ORAI1 Deficiency and Lack of Store-Operated Ca2+ Entry Cause Immunodeficiency, Myopathy, and Ectodermal Dysplasia. J. Allergy Clin. Immunol. 2009, 124, 1311–1318.e7. [Google Scholar] [CrossRef]
- Boncompagni, S.; Pecorai, C.; Michelucci, A.; Pietrangelo, L.; Protasi, F. Long-Term Exercise Reduces Formation of Tubular Aggregates and Promotes Maintenance of Ca2+ Entry Units in Aged Muscle. Front. Physiol. 2021, 11, 601057. [Google Scholar] [CrossRef]
- Agbulut, O.; Destombes, J.; Thiesson, D.; Butler-Browne, G. Age-Related Appearance of Tubular Aggregates in the Skeletal Muscle of Almost All Male Inbred Mice. Histochem. Cell. Biol. 2000, 114, 477–481. [Google Scholar] [CrossRef]
- Schiaffino, S.; Reggiani, C. Molecular Diversity of Myofibrillar Proteins: Gene Regulation and Functional Significance. Physiol. Rev. 1996, 76, 371–423. [Google Scholar] [CrossRef]
- Dos Santos, M.; Backer, S.; Saintpierre, B.; Izac, B.; Andrieu, M.; Letourneur, F.; Relaix, F.; Sotiropoulos, A.; Maire, P. Single-Nucleus RNA-Seq and FISH Identify Coordinated Transcriptional Activity in Mammalian Myofibers. Nat. Commun. 2020, 11, 5102. [Google Scholar] [CrossRef]
- Petrany, M.J.; Swoboda, C.O.; Sun, C.; Chetal, K.; Chen, X.; Weirauch, M.T.; Salomonis, N.; Millay, D.P. Single-Nucleus RNA-Seq Identifies Transcriptional Heterogeneity in Multinucleated Skeletal Myofibers. Nat. Commun. 2020, 11, 6374. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lilliu, E.; Koenig, S.; Koenig, X.; Frieden, M. Store-Operated Calcium Entry in Skeletal Muscle: What Makes It Different? Cells 2021, 10, 2356. https://doi.org/10.3390/cells10092356
Lilliu E, Koenig S, Koenig X, Frieden M. Store-Operated Calcium Entry in Skeletal Muscle: What Makes It Different? Cells. 2021; 10(9):2356. https://doi.org/10.3390/cells10092356
Chicago/Turabian StyleLilliu, Elena, Stéphane Koenig, Xaver Koenig, and Maud Frieden. 2021. "Store-Operated Calcium Entry in Skeletal Muscle: What Makes It Different?" Cells 10, no. 9: 2356. https://doi.org/10.3390/cells10092356
APA StyleLilliu, E., Koenig, S., Koenig, X., & Frieden, M. (2021). Store-Operated Calcium Entry in Skeletal Muscle: What Makes It Different? Cells, 10(9), 2356. https://doi.org/10.3390/cells10092356





