Folic Acid and Autism: A Systematic Review of the Current State of Knowledge
Abstract
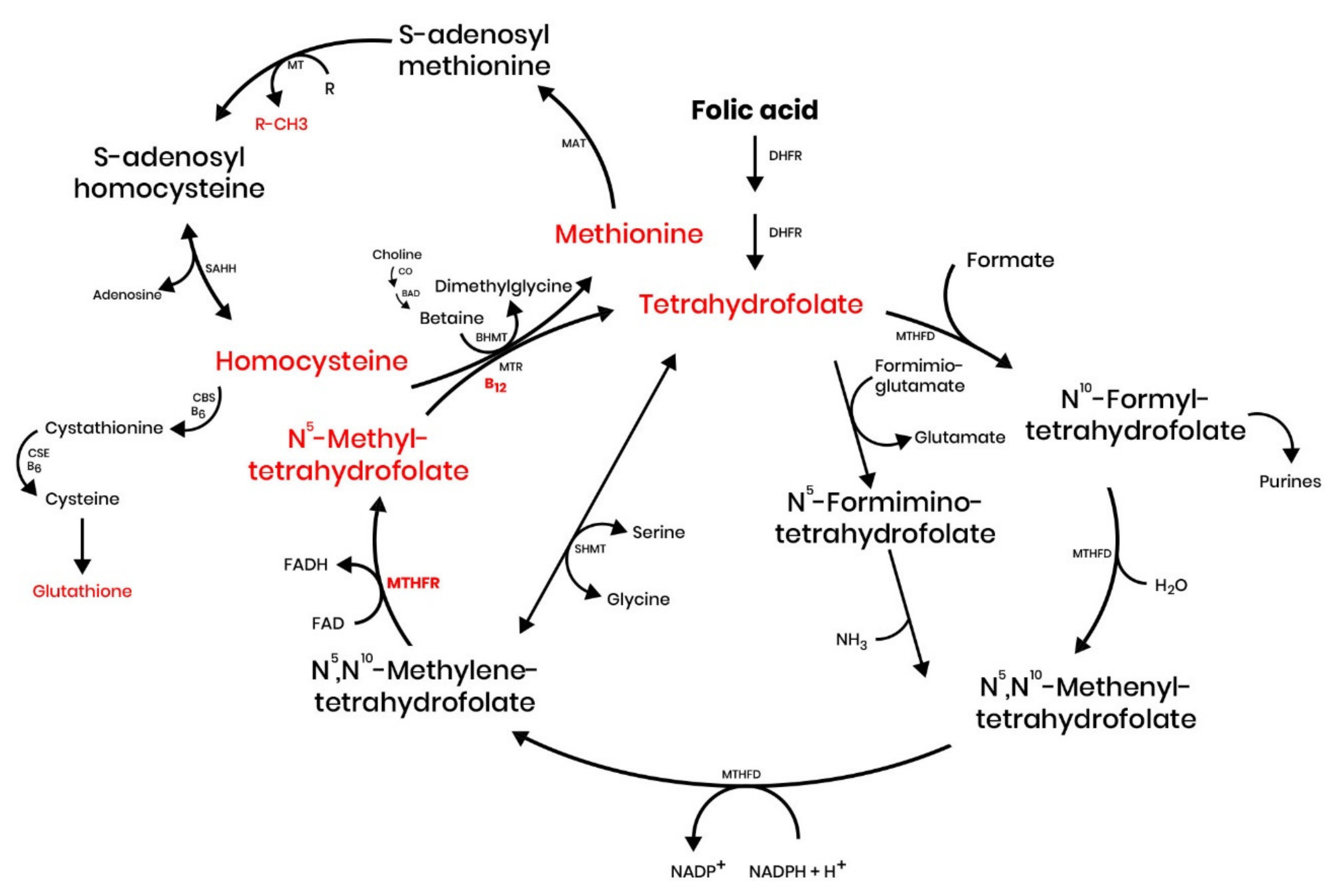
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Eligibility Criteria
2.3. Literature Search and Selection of Articles
2.4. Data Extraction
2.5. Risk of Bias across Studies
3. Results
3.1. Overview of Literature Search Results
3.2. Summary of the Results Reported by Human Clinical Trials Included in the Systematic Review
3.3. Studies on the Association between Maternal FA Supplementation in Reducing the Risk of Asds in Offspring
3.4. Studies of Folate Supplementation or Folate Levels in ASD Diagnosed Children
3.5. Studies on MTHFR Polymorphisms and ASDs
3.6. Studies on the Frequency of Serum FRAA in ASD Children
3.7. Summary of the Results Reported by Animal Clinical Studies Included in the Systematic Review
4. Discussion
4.1. Summary of Evidence
4.2. Limitations
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Christensen, D.L.; Maenner, M.J.; Bilder, D.; Constantino, J.N.; Daniels, J.; Durkin, M.S.; Fitzgerald, R.T.; Kurzius-Spencer, M.; Pettygrove, S.D.; Robinson, C.; et al. Prevalence and characteristics of autism spectrum disorder among children aged 4 years-Early Autism and Developmental Disabilities Monitoring Network, Seven Sites, United States, 2010, 2012, and 2014. MMWR Surveill. Summ. 2019, 68, 1–19. [Google Scholar] [CrossRef]
- Dias, C.M.; Walsh, C.A. Recent Advances in Understanding the Genetic Architecture of Autism. Annu. Rev. Genom. Hum. Genet. 2020, 21, 289–304. [Google Scholar] [CrossRef]
- Hertz-Picciotto, I.; Schmidt, R.; Krakowiak, P. Understanding environmental contributions to autism: Causal concepts and the state of science. Autism Res. 2018, 11, 554–586. [Google Scholar] [CrossRef]
- Bölte, S.; Girdler, S.; Marschik, P.B. The contribution of environmental exposure to the etiology of autism spectrum disorder. Cell. Mol. Life Sci. 2018, 76, 1275–1297. [Google Scholar] [CrossRef]
- Alam, R.; Abdolmaleky, H.M.; Zhou, J.-R. Microbiome, inflammation, epigenetic alterations, and mental diseases. Am. J. Med. Genet. B Neuropsychiatr. Genet. 2017, 174, 651–660. [Google Scholar] [CrossRef]
- Waye, M.M.Y.; Cheng, H.Y. Genetics and epigenetics of autism: A Review. Psychiatry Clin. Neurosci. 2017, 72, 228–244. [Google Scholar] [CrossRef]
- Chaste, P.; Leboyer, M. Autism risk factors: Genes, environment, and gene-environment interactions. Dialogues Clin. Neurosci. 2012, 14, 281–292. [Google Scholar]
- Modabbernia, A.; Velthorst, E.; Reichenberg, A. Environmental risk factors for autism: An evidence-based review of systematic reviews and meta-analyses. Mol. Autism 2017, 8, 1–16. [Google Scholar] [CrossRef]
- Dominguez-Salas, P.; Cox, S.E.; Prentice, A.M.; Hennig, B.J.; Moore, S.E. Maternal nutritional status, C1 metabolism and offspring DNA methylation: A review of current evidence in human subjects. Proc. Nutr. Soc. 2011, 71, 154–165. [Google Scholar] [CrossRef]
- Vargas, D.L.; Nascimbene, C.; Krishnan, C.; Zimmerman, A.W.; Pardo, C.A. Neuroglial activation and neuroinflammation in the brain of patients with autism. Ann. Neurol. 2004, 57, 67–81. [Google Scholar] [CrossRef]
- Tabouy, L.; Getselter, D.; Ziv, O.; Karpuj, M.; Tabouy, T.; Lukic, I.; Maayouf, R.; Werbner, N.; Ben-Amram, H.; Nuriel-Ohayon, M.; et al. Dysbiosis of microbiome and probiotic treatment in a genetic model of autism spectrum disorders. Brain Behav. Immun. 2018, 73, 310–319. [Google Scholar] [CrossRef]
- Hollis, F.; Kanellopoulos, A.K.; Bagni, C. Mitochondrial dysfunction in Autism Spectrum Disorder: Clinical features and perspectives. Curr. Opin. Neurobiol. 2017, 45, 178–187. [Google Scholar] [CrossRef]
- Chauhan, A.; Gu, F.; Essa, M.M.; Wegiel, J.; Kaur, K.; Brown, W.T.; Chauhan, V. Brain region-specific deficit in mitochondrial electron transport chain complexes in children with autism. J. Neurochem. 2011, 117, 209–220. [Google Scholar] [CrossRef]
- Devilbiss, E.A.; Gardner, R.; Newschaffer, C.J.; Lee, B. Maternal folate status as a risk factor for autism spectrum disorders: A review of existing evidence. Br. J. Nutr. 2015, 114, 663–672. [Google Scholar] [CrossRef]
- Schmidt, R.J.; Schroeder, D.I.; Crary-Dooley, F.K.; Barkoski, J.M.; Tancredi, D.J.; Walker, C.K.; Ozonoff, S.; Hertz-Picciotto, I.; LaSalle, J.M. Self-reported pregnancy exposures and placental DNA methylation in the MARBLES prospective autism sibling study. Environ. Epigenetics 2016, 2. [Google Scholar] [CrossRef]
- Ciernia, A.V.; LaSalle, J. The landscape of DNA methylation amid a perfect storm of autism aetiologies. Nat. Rev. Neurosci. 2016, 17, 411–423. [Google Scholar] [CrossRef]
- Ducker, G.; Rabinowitz, J.D. One-Carbon Metabolism in Health and Disease. Cell Metab. 2017, 25, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Tisato, V.; Silva, J.A.; Longo, G.; Gallo, I.; Singh, A.V.; Milani, D.; Gemmati, D. Genetics and Epigenetics of One-Carbon Metabolism Pathway in Autism Spectrum Disorder: A Sex-Specific Brain Epigenome? Genes 2021, 12, 782. [Google Scholar] [CrossRef]
- De-Regil, L.M.; Peña-Rosas, J.P.; Fernández-Gaxiola, A.C.; Rayco-Solon, P. Effects and safety of periconceptional oral folate supplementation for preventing birth defects. Cochrane Database Syst. Rev. 2015, CD007950. [Google Scholar] [CrossRef]
- Djukic, A. Folate-Responsive Neurologic Diseases. Pediatr. Neurol. 2007, 37, 387–397. [Google Scholar] [CrossRef]
- Bailey, L.B.; Berry, R.J. Folic acid supplementation and the occurrence of congenital heart defects, orofacial clefts, multiple births, and miscarriage. Am. J. Clin. Nutr. 2005, 81, 1213S–1217S. [Google Scholar] [CrossRef] [PubMed]
- Moat, S.J.; Lang, D.; McDowell, I.F.; Clarke, Z.L.; Madhavan, A.K.; Lewis, M.J.; Goodfellow, J. Folate, homocysteine, endothelial function and cardiovascular disease. J. Nutr. Biochem. 2004, 15, 64–79. [Google Scholar] [CrossRef]
- LaSalle, J.M. A genomic point-of-view on environmental factors influencing the human brain methylome. Epigenetics 2011, 6, 862–869. [Google Scholar] [CrossRef] [PubMed]
- Goyette, P.; Sumner, J.S.; Milos, R.; Duncan, A.M.; Rosenblatt, D.S.; Matthews, R.G.; Rozen, R. Human methylene tetrahydrofolate reductase: Isolation of cDNA, mapping and mutation identification. Nat. Genet. 1994, 7, 195–200. [Google Scholar] [CrossRef]
- Wuerthele, S.; Yasuda, R.; Freed, W.; Hoffer, B. The effect of local application of homocysteine on neuronal activity in the central nervous system of the rat. Life Sci. 1982, 31, 2683–2691. [Google Scholar] [CrossRef]
- Singer, H.S.; Butler, I.; Rothenberg, S.; Valle, O.; Freeman, J. Interrelationships among serum folate, CSF folate, neurotransmitters, and neuropsychiatric symptoms. Neurology 1980, 30, 419. [Google Scholar]
- Orenbuch, A.; Fortis, K.; Taesuwan, S.; Yaffe, R.; Caudill, M.A.; Golan, H.M. Prenatal Nutritional Intervention Reduces Autistic-Like Behavior Rates Among Mthfr-Deficient Mice. Front. Neurosci. 2019, 13, 383. [Google Scholar] [CrossRef]
- Dufficy, L.; Naumovski, N.; Ng, X.; Blades, B.; Yates, Z.; Travers, C.; Lewis, P.; Sturm, J.; Veysey, M.; Roach, P.D.; et al. G80A reduced folate carrier SNP influences the absorption and cellular translocation of dietary folate and its association with blood pressure in an elderly population. Life Sci. 2006, 79, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Chango, A.; Emery-Fillon, N.; de Courcy, G.P.; Lambert, D.; Pfister, M.; Rosenblatt, D.S.; Nicolas, J.-P. A Polymorphism (80G->A) in the Reduced Folate Carrier Gene and Its Associations with Folate Status and Homocysteinemia. Mol. Genet. Metab. 2000, 70, 310–315. [Google Scholar] [CrossRef]
- Alam, C.; Hoque, T.; Finnell, R.; Goldman, I.D.; Bendayan, R. Regulation of Reduced Folate Carrier (RFC) by Vitamin D Receptor at the Blood-Brain Barrier. Mol. Pharm. 2017, 14, 3848–3858. [Google Scholar] [CrossRef]
- Ramaekers, V.T.; Blau, N.; Sequeira, J.M.; Nassogne, M.-C.; Quadros, E.V. Folate Receptor Autoimmunity and Cerebral Folate Deficiency in Low-Functioning Autism with Neurological Deficits. Neuropediatrics 2007, 38, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.D.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.F.; Devereaux, P.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. Ann. Intern. Med. 2009, 151, W-65–W-94. [Google Scholar] [CrossRef] [PubMed]
- Moretti, P.; Sahoo, T.; Hyland, K.; Bottiglieri, T.; Peters, S.; Del Gaudio, D.; Roa, B.; Curry, S.; Zhu, H.; Finnell, R.; et al. Cerebral folate deficiency with developmental delay, autism, and response to folinic acid. Neurology 2005, 64, 1088–1090. [Google Scholar] [CrossRef] [PubMed]
- Virk, J.; Liew, Z.; Olsen, J.; A Nohr, E.; Catov, J.M.; Ritz, B. Preconceptional and prenatal supplementary folic acid and multivitamin intake and autism spectrum disorders. Autism 2015, 20, 710–718. [Google Scholar] [CrossRef]
- Levine, S.Z.; Kodesh, A.; Viktorin, A.; Smith, L.; Uher, R.; Reichenberg, A.; Sandin, S. Association of Maternal Use of Folic Acid and Multivitamin Supplements in the Periods Before and During Pregnancy With the Risk of Autism Spectrum Disorder in Offspring. JAMA Psychiatry 2018, 75, 176–184. [Google Scholar] [CrossRef]
- Surén, P.; Roth, C.; Bresnahan, M.; Haugen, M.; Hornig, M.; Hirtz, D.; Lie, K.K.; Lipkin, W.I.; Magnus, P.; Reichborn-Kjennerud, T.; et al. Association Between Maternal Use of Folic Acid Supplements and Risk of Autism Spectrum Disorders in Children. JAMA 2013, 309, 570–577. [Google Scholar] [CrossRef]
- DeVilbiss, E.A.; Magnusson, C.; Gardner, R.M.; Rai, D.; Newschaffer, C.J.; Lyall, K.; Dalman, C.; Lee, B.K. Antenatal nutritional supplementation and autism spectrum disorders in the Stockholm youth cohort: Population based cohort study. BMJ 2017, 359, j4273. [Google Scholar] [CrossRef]
- Raghavan, R.; Riley, A.W.; Volk, H.; Caruso, D.; Hironaka, L.; Sices, L.; Hong, X.; Wang, G.; Ji, Y.; Brucato, M.; et al. Maternal multivitamin intake, plasma folate and vitamin B12 levels and autism spectrum disorder risk in offspring. Paediatr. Perinat. Epidemiol. 2018, 32, 100–111. [Google Scholar] [CrossRef]
- Schmidt, R.; Iosif, A.-M.; Angel, E.G.; Ozonoff, S. Association of Maternal Prenatal Vitamin Use with Risk for Autism Spectrum Disorder Recurrence in Young Siblings. JAMA Psychiatry 2019, 76, 391–398. [Google Scholar] [CrossRef]
- Graaff, J.S.-D.; Ghassabian, A.; Jaddoe, V.W.; Tiemeier, H.; Roza, S.J. Folate concentrations during pregnancy and autistic traits in the offspring. The Generation R Study. Eur. J. Public Health 2014, 25, 431–433. [Google Scholar] [CrossRef]
- Nilsen, R.M.; Surén, P.; Gunnes, N.; Alsaker, E.R.; Bresnahan, M.; Hirtz, D.; Hornig, M.; Lie, K.K.; Lipkin, W.I.; Reichborn-Kjennerud, T.; et al. Analysis of self-selection bias in a population-based cohort study of autism spectrum disorders. Paediatr. Périnat. Epidemiol. 2013, 27, 553–563. [Google Scholar] [CrossRef]
- Christian, P.; Murray-Kolb, L.E.; Khatry, S.K.; Katz, J.; Schaefer, B.A.; Cole, P.M.; LeClerq, S.C.; Tielsch, J.M. Prenatal Micronutrient Supplementation and Intellectual and Motor Function in Early School-aged Children in Nepal. JAMA 2010, 304, 2716–2723. [Google Scholar] [CrossRef] [PubMed]
- Roth, C.; Magnus, P.; Schjølberg, S.; Stoltenberg, C.; Surén, P.; McKeague, I.W.; Davey Smith, G.; Reichborn-Kjennerud, T.; Susser, E. Folic Acid Supplements in Pregnancy and Severe Language Delay in Children. JAMA 2011, 30, 1566. [Google Scholar] [CrossRef]
- Sun, C.; Zou, M.; Zhao, D.; Xia, W.; Wu, L. Efficacy of Folic Acid Supplementation in Autistic Children Participating in Structured Teaching: An Open-Label Trial. Nutrients 2016, 8, 337. [Google Scholar] [CrossRef]
- James, S.J.; Melnyk, S.; Fuchs, G.; Reid, T.; Jernigan, S.; Pavliv, O.; Hubanks, A.; Gaylor, D.W. Efficacy of methylcobalamin and folinic acid treatment on glutathione redox status in children with autism. Am. J. Clin. Nutr. 2008, 89, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Kałużna-Czaplińska, J.; Michalska, M.; Rynkowski, J. Vitamin supplementation reduces the level of homocysteine in the urine of autistic children. Nutr. Res. 2011, 31, 318–321. [Google Scholar] [CrossRef]
- Amanat, A.; Mostafa, I.W.; Yahya, M.A.; Musthafa, M.E.; Marwan, M.A.; Richard, C.D. Hyperhomocysteinemia among Omani autistic children: A case-control study. Acta Biochim. Pol. 2011, 58, 547–551. [Google Scholar]
- Guo, M.; Li, L.; Zhang, Q.; Chen, L.; Dai, Y.; Liu, L.; Feng, J.; Cai, X.; Cheng, Q.; Chen, J.; et al. Vitamin and mineral status of children with autism spectrum disorder in Hainan Province of China: Associations with symptoms. Nutr. Neurosci. 2018, 23, 803–810. [Google Scholar] [CrossRef]
- Adams, J.B.; Audhya, T.; Geis, E.; Gehn, E.; Fimbres, V.; Pollard, E.L.; Mitchell, J.; Ingram, J.; Hellmers, R.; Laake, D.; et al. Comprehensive nutritional and dietary intervention for autism spectrum disorder-a randomized, controlled 12-Month trial. Nutrients 2018, 10, 369. [Google Scholar] [CrossRef]
- Shoffner, J.; Trommer, B.; Thurm, A.; Farmer, C.; Iii, W.A.L.; Soskey, L.; Rodriguez, A.N.; D’Souza, P.; Spence, S.J.; Hyland, K.; et al. CSF concentrations of 5-methyltetrahydrofolate in a cohort of young children with autism. Neurology 2016, 86, 2258–2263. [Google Scholar] [CrossRef]
- Li, Q.; Yang, T.; Chen, L.; Dai, Y.; Wu, L.-J.; Jia, F.-Y.; Hao, Y.; Li, L.; Zhang, J.; Ke, X.-Y.; et al. Serum Folate Status Is Primarily Associated with Neurodevelopment in Children with Autism Spectrum Disorders Aged Three and Under—A Multi-Center Study in China. Front. Nutr. 2021, 8. [Google Scholar] [CrossRef]
- Guo, T.; Chen, H.; Liu, B.; Ji, W.; Yang, C. Methylenetetrahydrofolate Reductase Polymorphisms C677T and Risk of Autism in the Chinese Han Population. Genet. Test. Mol. Biomark. 2012, 16, 968–973. [Google Scholar] [CrossRef]
- Goin-Kochel, R.P.; Porter, A.E.; Peters, S.U.; Shinawi, M.; Sahoo, T.; Beaudet, A.L. TheMTHFR 677C→T polymorphism and behaviors in children with autism: Exploratory genotype-phenotype correlations. Autism Res. 2009, 2, 98–108. [Google Scholar] [CrossRef]
- Dos Santos, P.A.C.; Longo, D.; Brandalize, A.P.C.; Schuler-Faccini, L. MTHFR C677T is not a risk factor for autism spectrum disorders in South Brazil. Psychiatr. Genet. 2010, 20, 187–189. [Google Scholar] [CrossRef]
- Mohammad, N.S.; Shruti, P.S.; Bharathi, V.; Prasad, C.K.; Hussain, T.; Alrokayan, S.; Naik, U.; Devi, A.R.R. Clinical utility of folate pathway genetic polymorphisms in the diagnosis of autism spectrum disorders. Psychiatr. Genet. 2016, 26, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Ismail, S.; Senna, A.A.; Behiry, E.G.; Ashaat, E.A.; Zaki, M.S.; Ashaat, N.A.; Salah, D.M. Study of C677T variant of methylene tetrahydrofolate reductase gene in autistic spectrum disorder Egyptian children. Am. J. Med. Genet. Part. B Neuropsychiatr. Genet. 2019, 180, 305–309. [Google Scholar] [CrossRef]
- Zhang, Z.; Yu, L.; Li, S.; Liu, J. Association Study of Polymorphisms in Genes Relevant to Vitamin B12 and Folate Metabolism with Childhood Autism Spectrum Disorder in a Han Chinese Population. Med. Sci. Monit. 2018, 24, 370–376. [Google Scholar] [CrossRef]
- Sener, E.F.; Oztop, D.B.; Ozkul, Y. MTHFR Gene C677T Polymorphism in Autism Spectrum Disorders. Genet. Res. Int. 2014, 2014, 1–5. [Google Scholar] [CrossRef]
- Mohammad, N.S.; Jain, J.M.N.; Chintakindi, K.P.; Singh, R.P.; Naik, U.; Akella, R.R.D. Aberrations in folate metabolic pathway and altered susceptibility to autism. Psychiatr. Genet. 2009, 19, 171–176. [Google Scholar] [CrossRef]
- El-Baz, F.; El-Aal, M.A.; Kamal, T.M.; Sadek, A.A.; Othman, A.A. Study of the C677T and 1298AC polymorphic genotypes of MTHFR Gene in autism spectrum disorder. Electron. Physician 2017, 9, 5287–5293. [Google Scholar] [CrossRef] [PubMed]
- James, S.J.; Melnyk, S.; Jernigan, S.; Pavliv, O.; Trusty, T.; Lehman, S.; Seidel, L.; Gaylor, D.W.; Cleves, M.A. A functional polymorphism in the reduced folate carrier gene and DNA hypomethylation in mothers of children with autism. Am. J. Med. Genet. Part. B: Neuropsychiatr. Genet. 2010, 153B, 1209–1220. [Google Scholar] [CrossRef]
- Zhou, J.; Liu, A.; He, F.; Jin, Y.; Zhou, S.; Xu, R.; Guo, H.; Zhou, W.; Wei, Q.; Wang, M. High prevalence of serum folate receptor autoantibodies in children with autism spectrum disorders. Biomarkers 2018, 23, 622–624. [Google Scholar] [CrossRef] [PubMed]
- Frye, R.E.; Slattery, J.; Delhey, L.; Furgerson, B.; Strickland, T.; Tippett, M.; Sailey, A.; Wynne, R.; Rose, S.; Melnyk, S.; et al. Folinic acid improves verbal communication in children with autism and language impairment: A randomized double-blind placebo-controlled trial. Mol. Psychiatry 2016, 23, 247–256. [Google Scholar] [CrossRef]
- Degroote, S.; Hunting, D.; Takser, L. Periconceptional folate deficiency leads to autism-like traits in Wistar rat offspring. Neurotoxicol. Teratol. 2018, 66, 132–138. [Google Scholar] [CrossRef]
- Barua, S.; Kuizon, S.; Brown, W.T.; Junaid, M.A. High Gestational Folic Acid Supplementation Alters Expression of Imprinted and Candidate Autism Susceptibility Genes in a sex-Specific Manner in Mouse Offspring. J. Mol. Neurosci. 2015, 58, 277–286. [Google Scholar] [CrossRef]
- Kezurer, N.; Galron, D.; Golan, H. Increased susceptibility to mild neonatal stress in MTHFR deficient mice. Behav. Brain Res. 2013, 253, 240–252. [Google Scholar] [CrossRef]
- Barua, S.; Kuizon, S.; Chadman, K.K.; Brown, W.T.; Junaid, M.A. Microarray Analysis Reveals Higher Gestational Folic Acid Alters Expression of Genes in the Cerebellum of Mice Offspring—A Pilot Study. Brain Sci. 2015, 5, 14–31. [Google Scholar] [CrossRef] [PubMed]
- Sadigurschi, N.; Golan, H.M. Maternal and offspring MTHFR genotypes interact in a mouse model to induce ASD-like behavior. Genes Brain Behav. 2018, 18, e12547. [Google Scholar] [CrossRef] [PubMed]
- Sequeira, J.M.; Desai, A.; Berrocal-Zaragoza, M.I.; Murphy, M.M.; Fernández-Ballart, J.D.; Quadros, E.V. Exposure to Folate Receptor Alpha Antibodies during Gestation and Weaning Leads to Severe Behavioral Deficits in Rats: A Pilot Study. PLoS ONE 2016, 11, e0152249. [Google Scholar] [CrossRef]
- Chu, D.; Li, L.; Jiang, Y.; Tan, J.; Ji, J.; Zhang, Y.; Jin, N.; Liu, F. Excess Folic Acid Supplementation Before and During Pregnancy and Lactation Activates Fos Gene Expression and Alters Behaviors in Male Mouse Offspring. Front. Neurosci. 2019, 13, 313. [Google Scholar] [CrossRef]
- Desai, A.; Sequeira, J.M.; Quadros, E.V. Prevention of behavioral deficits in rats exposed to folate receptor antibodies: Implication in autism. Mol. Psychiatry 2016, 22, 1291–1297. [Google Scholar] [CrossRef]
- Wiens, D.; DeSoto, M.C. Is High Folic Acid Intake a Risk Factor for Autism?—A Review. Brain Sci. 2017, 7, 149. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Sheng, C.; Xie, R.; Sun, W.; Asztalos, E.; Moddemann, D.; Zwaigenbaum, L.; Walker, M.; Wen, S.W. New perspective on mpact of folic acid supplementation during pregnancy on neurodevelopment/autism in the offspring children–A systematic review. PLoS ONE 2016, 11, e0165626. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.-Q.; Li, H.-B.; Zhai, D.-S.; Ding, S.-B. Association of maternal prenatal folic acid intake with subsequent risk of autism spectrum disorder in children: A systematic review and meta-analysis. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2019, 94, 109650. [Google Scholar] [CrossRef]
- Pu, D.; Shen, Y.; Wu, J. Association between MTHFR Gene Polymorphisms and the Risk of Autism Spectrum Disorders: A Meta-Analysis. Autism Res. 2013, 6, 384–392. [Google Scholar] [CrossRef]
- Castro, K.; Klein, L.S.; Baronio, D.; Gottfried, C.; Riesgo, R.; Perry, I.S. Folic acid and autism: What do we know? Nutr. Neurosci. 2016, 19, 310–317. [Google Scholar] [CrossRef]
- Schaevitz, L.R.; Berger-Sweeney, J.E. Gene-Environment Interactions and Epigenetic Pathways in Autism: The Importance of One-Carbon Metabolism. ILAR J. 2012, 53, 322–340. [Google Scholar] [CrossRef]
- Paul, L.; Selhub, J. Interaction between excess folate and low vitamin B12 status. Mol. Asp. Med. 2017, 53, 43–47. [Google Scholar] [CrossRef]
- Neggers, Y. The Relationship between Folic Acid and Risk of Autism Spectrum Disorders. Health 2014, 2, 429–444. [Google Scholar] [CrossRef]
- Frye, R.E.; Slattery, J.C.; Quadros, E.V. Folate metabolism abnormalities in autism: Potential biomarkers. Biomark. Med. 2017, 11, 687–699. [Google Scholar] [CrossRef]
- Schmidt, R.; Tancredi, D.; Ozonoff, S.; Hansen, R.L.; Hartiala, J.; Allayee, H.; Schmidt, L.C.; Tassone, F.; Hertz-Picciotto, I. Maternal periconceptional folic acid intake and risk of autism spectrum disorders and developmental delay in the CHARGE (CHildhood Autism Risks from Genetics and Environment) case-control study. Am. J. Clin. Nutr. 2012, 96, 80–89. [Google Scholar] [CrossRef]
- Braun, J.M.; Froehlich, T.; Kalkbrenner, A.; Pfeiffer, C.M.; Fazili, Z.; Yolton, K.; Lanphear, B.P. Brief Report: Are autistic-behaviors in children related to prenatal vitamin use and maternal whole blood folate concentrations? J. Autism Dev. Disord. 2014, 44, 2602–2607. [Google Scholar] [CrossRef]
- Bjørk, M.; Riedel, B.; Spigset, O.; Veiby, G.; Kolstad, E.; Daltveit, A.K.; Gilhus, N.E. Association of Folic Acid Supplementation During Pregnancy with the Risk of Autistic Traits in Children Exposed to Antiepileptic Drugs In Utero. JAMA Neurol. 2018, 75, 160–168. [Google Scholar] [CrossRef]
- Barua, S.; Kuizon, S.; Brown, W.T.; Junaid, M.A. DNA Methylation Profiling at Single-Base Resolution Reveals Gestational Folic Acid Supplementation Influences the Epigenome of Mouse Offspring Cerebellum. Front. Neurosci. 2016, 10, 168. [Google Scholar] [CrossRef]
- Egorova, O.; Myte, R.; Schneede, J.; Hägglöf, B.; Bölte, S.; Domellöf, E.; A’Roch, B.I.; Elgh, F.; Ueland, P.M.; Silfverdal, S.A. Maternal blood folate status during early pregnancy and occurrence of autism spectrum disorder in offspring: A study of 62 serum biomarkers. Mol. Autism 2020, 11, 1–15. [Google Scholar] [CrossRef]
- Valera-Gran, D.; de la Hera, M.G.; Navarrete-Muñoz, E.M.; Fernandez-Somoano, A.; Tardon, A.; Julvez, J.; Forns, J.; Lertxundi, N.; Ibarluzea, J.; Murcia, M.; et al. Folic Acid Supplements During Pregnancy and Child Psychomotor Development After the First Year of Life. JAMA Pediatr. 2014, 168, e142611. [Google Scholar] [CrossRef] [PubMed]
- Rogers, E.J. Has enhanced folate status during pregnancy altered natural selection and possibly Autism prevalence? A closer look at a possible link. Med. Hypotheses 2008, 71, 406–410. [Google Scholar] [CrossRef]
- Strøm, M.; Granström, C.; Lyall, K.; Ascherio, A.; Olsen, S.F. Research Letter: Folic acid supplementation and intake of folate in pregnancy in relation to offspring risk of autism spectrum disorder. Psychol. Med. 2018, 48, 1048–1054. [Google Scholar] [CrossRef] [PubMed]
- Ramaekers, V.; Sequeira, J.; Quadros, E. The basis for folinic acid treatment in neuro-psychiatric disorders. Biochimie 2016, 126, 79–90. [Google Scholar] [CrossRef]
- Ladd-Acosta, C.; Hansen, K.; Briem, E.; Fallin, M.D.; Kaufmann, W.E.; Feinberg, A.P. Common DNA methylation alterations in multiple brain regions in autism. Mol. Psychiatry 2013, 19, 862–871. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-H.; Yates, Z.; Veysey, M.; Heo, Y.-R.; Lucock, M. Contemporary Issues Surrounding Folic Acid Fortification Initiatives. Prev. Nutr. Food Sci. 2014, 19, 247–260. [Google Scholar] [CrossRef]
- Sawaengsri, H.; Wang, J.; Reginaldo, C.; Steluti, J.; Wu, D.; Meydani, S.N.; Selhub, J.; Paul, L. High folic acid intake reduces natural killer cell cytotoxicity in aged mice. J. Nutr. Biochem. 2016, 30, 102–107. [Google Scholar] [CrossRef]
- Morris, M.S.; Jacques, P.F.; Rosenberg, I.H.; Selhub, J. Circulating unmetabolized folic acid and 5-methyltetrahydrofolate in relation to anemia, macrocytosis, and cognitive test performance in American seniors. Am. J. Clin. Nutr. 2010, 91, 1733–1744. [Google Scholar] [CrossRef] [PubMed]
- Girotto, F.; Scott, L.; Avchalumov, Y.; Harris, J.; Iannattone, S.; Drummond-Main, C.; Tobias, R.; Bello-Espinosa, L.; Rho, J.M.; Davidsen, J.; et al. High dose folic acid supplementation of rats alters synaptic transmission and seizure susceptibility in offspring. Sci. Rep. 2013, 3, srep01465. [Google Scholar] [CrossRef]
- Kao, T.T.; Wang, K.C.; Chang, W.N.; Lin, C.Y.; Chen, B.H.; Wu, H.L.; Shi, G.Y.; Tsai, J.N.; Fu, T.F. Characterization and comparative studies of zebrafish and human recombinant dihydrofolate reductases--inhibition by folic scid and polyphenols. Drug Metab. Dispos. 2008, 36, 508–516. [Google Scholar] [CrossRef]
- Kelly, P.; McPartlin, J.; Goggins, M.; Weir, D.G.; Scott, J.M. Unmetabolized folic acid in serum: Acute studies in subjects consuming fortified food and supplements. Am. J. Clin. Nutr. 1997, 65, 1790–1795. [Google Scholar] [CrossRef]
- Arabi, M.; Christensen, K.E.; Chan, D.; Leclerc, D.; Landry, M.; Ly, L.; Rozen, R.; Trasler, J. Testicular MTHFR deficiency may explain sperm DNA hypomethilation associated with high dose folic acid supplementation. Hum. Mol. Genet. 2018, 27, 1123–1135. [Google Scholar] [CrossRef] [PubMed]
- Sie, K.K.Y.; Medline, A.; Van Weel, J.; Sohn, K.-J.; Choi, S.-W.; Croxford, R.; Kim, Y.-I. Effect of maternal and postweaning folic acid supplementation on colorectal cancer risk in the offspring. Gut 2011, 60, 1687–1694. [Google Scholar] [CrossRef]
- Morris, M.S.; Jacques, P. Folate and neurological functions: Epidemiological perspectives. In Folate in Health and Disease, 2nd ed.; Bailey, L.B., Ed.; CRC Press: New York, NY, USA, 2010; pp. 325–346. [Google Scholar]
- Reynolds, E. Vitamin B12, folic acid, and the nervous system. Lancet Neurol. 2006, 5, 949–960. [Google Scholar] [CrossRef]

| No | Study | Type of Study | Sample Size | Supplement Used | Period of Intake | ASDs Cases | Study Outcome Parameters | Outcomes |
|---|---|---|---|---|---|---|---|---|
| 1 | Virk J et al., (2016) [34] | Population-based cohort study | n = 35,059 women | FA and/or multivitamin supplements (containing at last 400 μg of FA) | 4 weeks prior to 8 weeks after pregnancy | 552 (1.6%) | Autism and Asperger’s syndrome. Pervasive developmental disorder—not otherwise specified (PDD-NOS) | No association has been found between early folate or multivitamin intake for autism spectrum disorder [34]. |
| 2 | Levine SZ et al., (2018) [35] | Case-control cohort study | n = 45,300 children | FA and/or multivitamin supplements | Before but not during pregnancy/during but not before pregnancy/before and during pregnancy from 4 weeks before and 8 weeks into pregnancy2 years before pregnancy | 572 (1.3%) | Risk of ASDs in offspring | Maternal exposure to FA and multivitamin supplements before and during pregnancy is associated with a reduced risk of ASDs in the offspring compared with the offspring of mothers without such exposure [35]. |
| 3 | Surén Pet al., (2013) [36] | Cohort study | n = 85,176 children | Multivitamin supplements (400 μg of FA) fish oil and FA | 4 weeks before to 8 weeks after the start of pregnancy | 270 (0.32%) | Autistic disorder risk. Asperger syndrome. PDD-NOS | Maternal use of supplemental folic acid before conception and early in pregnancy was associated with a lower risk of autism spectrum disorders in children [36]. |
| 4 | DeVilbiss EA et al., (2017) [37] | Population- based cohort study | n = 273,107 mother–child pairs | Multivitamins supplements FA supplements (400 μg) | First trimester (400μg/day) | 158 (0.26%) in the maternal multivitamin use group 430 (0.48%) in the no-nutritional supplementation use group | Risk of ASDs with and without intellectual disability in offspring | Maternal multivitamin supplementation during pregnancy may be inversely associated with ASDs with intellectual disability in offspring [37]. |
| 5 | Raghavan R et al., (2018) [38] | Cohort study | n = 1257 mother–infant pairs | Multivitamin Supplements | During pregnancy≤2 times/week3–5 times/week>5 times/week | 86 (6.8%) | Risk of ASDs in offspring.Maternal B12 levels 2–3 days after birthMTHFR genotype | The results show that moderate intake (3–5 times/week) of multivitamin supplements during pregnancy is associated with decreased risk of ASDs in offspring. Low (≤2 times/week) and high (>5 times/week) supplementation was associated with increased risk of ASDs. There was a “U” shaped relationship between maternal multivitamin supplementation frequency and ASDs risk [38]. |
| 6 | Schmidt RJ et al., (2019) [39] | Cohort study | n = 332 children and theirn = 305 mothers | FA (600 μg) and iron (27 mg) supplements | First month of pregnancy 6 months before and each month during the pregnancy | 18 (14.1%) following mother supplementation and 37 (32.7%) from mothers receiving no supplementation | ASD recurrence in siblings of children with ASDs in high-risk families | Maternal prenatal vitamin intake during the first month of pregnancy may reduce ASDs recurrence in siblings of children with ASDs in high-risk families [39]. |
| 7 | Steenweg-de Graaff J et al., (2015) [40] | Population-based birth cohort study | n = 5591 mothers of single live-born neonates | FA supplements | Preconceptional within the first 10 weeks of pregnancy After the first 10 weeks of pregnancy | 3893 (70%) | Maternal plasma folate concentrations at 13 weeks of gestation. Autistic traits in the offspring at the age of six years | Maternal folate was not associated with autistic traits in the offspring. In contrast, prenatal folic acid use was associated with less child autistic traits [40]. |
| 8 | Nilsen RM et al., (2013) [41] | Population-based cohort study | n = 507,856 Medical Birth Registry of Norway nationwide population children n = 89 836 MoBa cohort sample children | FA supplements | Prenatal exposure | 234 (0.26%) in the cohort and 2072 (0.41%) in the nationwide population | Specialist-confirmed diagnosis ASDs.Estimated the risk of ASDs associated with: primipara pregnancy (no, yes), prenatal folic acid use (no, yes), prenatal smoking (no, yes), low birth weight (no, yes), preterm birth (no, yes), offspring sex (female, male), and caesarean section history (no, yes). | Was observed that maternal prenatal folic acid supplement use was associated with a 14–17% adjusted risk reduction for ASDs [41]. |
| 9 | Christian P et al., (2010) [42] | Cohort study | n = 676 children | FA (400 μg) FA and iron (60 mg) FA, iron and zinc (30 mg) FA, iron, zinc and vitamins D (10 μg), E (10 mg), B1 (1.6 mg), B2 (1.8 mg), B6 (2.2 mg), B12 (2.6 μg), C (100 mg), and K (65 μg); niacin (20 mg); copper (2.0 mg); magnesium (100 mg), with 1000-μg vitamin A | early pregnancy for 3 months postpartum | N.S | Estimation of children’s intellectual functioning:universal nonverbal intelligence test (UNIT); tests of executive function, including go/no-go, the Stroop test, backward digit span test; movement assessment battery for children (MABC); finger-tapping test | Maternal prenatal FA and iron supplementation was associated with better test scores in offspring at 7 to 9 years compared to the control group with only vitamin A supplementation [42]. |
| 10 | Roth C et al., (2011) [43] | Cohort study | n = 38,954 children | FA and/or multivitamin supplements | 4 weeks before to 8 weeks after conception | 204 (0.5%) severe language delay and 1290 (3.3%) moderate language delay | Risk of child with severe language delay. Assessment of severe language delay and motor delay | Maternal FA use was associated with a reduced risk of severe language delay in offspring at age 3 years. Instead, no significant association with motor skills delay risk was found [43]. |
| No | Study | Type of Study | Sample Size | Age | Supplement Used | Period of Intake | Study Outcome Parameters | Outcomes |
|---|---|---|---|---|---|---|---|---|
| 1 | Sun Cet al., (2016) [44] | Open-Label Trial | n = 66 ASDs children | 4.5 ± 1.1 years old | FA supplements (400 μg) | Two times/daily3 months | Plasma levels of: FA homocysteine glutathione metabolism before and after treatment. Improvement of autism symptoms | Intervention improved autism symptoms towards sociability, cognitive verbal/preverbal, receptive language, and affective expression and communication. Furthermore, this treatment also improved the concentrations of folic acid, homocysteine, and normalized glutathione redox metabolism [44]. |
| 2 | Moretti P et al., (2005) [33] | Single study subject | n = one girl | 6 years old | Folinic acid supplements (0.5 mg/kg) | One time/daily doubled after 2 weeks1 year | CSF levels of 5-methyltetrahydrofolate (5-MTHF), pterins and neurotransmitter metabolites | Treatment with folinic acid corrected CSF abnormalities and improved motor skills. The evaluation showed that despite improvement of motor skills and parental reports of increased responsivity, her cognitive, language, and socialization skills remained delayed [33]. |
| 3 | James SJ et al., (2009) [45] | Open-label trial | n = 40 ASDs children | 2–7 years old | Methylcobalamin (75 μg/kg) Folinic acid (400μg) | 2 times/week (methylcobalamin) 2 times/day (folinic acid) 3 months | Plasma levels of: methionineSAMSAHSAM:SAH ratio. Homocysteine, cysteine, cysteinylglycine, tGSH, fGSH, GSSG | The significant improvements observed in transmethylation metabolites and glutathione redox status after treatment suggest that targeted nutritional intervention with methylcobalamin and folinic acid may be of clinical benefit in some children who have autism [45]. |
| 4 | Kałużna-Czaplińska J et al., (2011) [46] | Case-control study | n = 30 ASDs children vs.21 non ASDs children | 4–11 years old | FA supplements (400 μg) Vitamin B6 (200 mg) Vitamin B12 (1.2 μg) (sugar-free diet) | 3 months | Urine homocysteine levels in ASDs children before vitamin supplementation and 3 months after | The study showed a significant improvement in sleep and gastrointestinal problems compared with the placebo group. The intake of vitamins B6 and B12, together with folic acid, was found to be more effective in lowering the levels of urinary homocysteine than the intake of vitamins B6 and B12 alone [46]. |
| 5 | Amanat A et al., (2011) [47] | Case-control study | n = 40 ASDs children vs 40 TD children of their age and gender matched controls | 3–5 years old | No supplements | N.A | Fasting serum homocysteine levels. Serum folate and vitamin B12 levels. Follow up time: December 2009–August 2010 | The results indicated that mean serum Hcy levels were significantly higher in autistic children as compared to controls. Significantly lower serum folate and vitamin B12 levels were observed in autistic children as compared to controls. The levels of homocysteine in autistic children were also much higher as compared to normal reference values (5–15 μmol/L) [47]. |
| 6 | Guo M et al., (2020) [48] | Clinical Trial | n = 274 ASDs children vs. 97 TD age-matched children | 4.24 ± 1.20 years old | No supplements | N.A | Vitamin and mineral concentrations in ASDs and TD children. Influence of vitamin and mineral status on the Autism Behavior Checklist (ABC) score, Social Responsiveness Scale (SRS), and correlation analysis between the Gesell Developmental Scale (GDS) scores | Results showed a significant insufficiency of vitamin and mineral, especially of folate and vitamin D, in ASDs children. The analysis correlated these lower nutrient levels with ASDs traits [48]. |
| 7 | Adams JB et al., (2018) [49] | Randomized controlled single-blind study | n = 67 children and adults with ASDs from Arizona vs.50 non-sibling neurotypical controls of similar age and gender | 3–58 years old | Special vitamin/mineral supplements (~600 mcg FA)Essential fatty acids (omega-3 and omega-6 fatty acids).Carnitine supplement | 12 months | Blood and urine levels of: vitamins, biomarkers of vitamin status, minerals, plasma amino acids, plasma glutathione, neurotransmitters, and biomarkers of oxidative stress, methylation, sulfation and energy production | There was a significant improvement in nonverbal intellectual ability in the treatment group compared to the non-treatment group based on a blinded clinical assessment. Based on semi-blinded assessment, the treatment group, compared to the non-treatment group, had significantly greater improvement in autism symptoms and developmental age. The treatment group had significantly greater increases in EPA; DHA; carnitine; and vitamins A, B2, B5, B6, B12, folic acid, and coenzyme Q10 [49]. |
| 8 | Shoffner J et al., (2016) [50] | Cohort study | n = 67 children with a diagnosis of DSM-IV-TR autistic disorder at last one lumbar puncture (LP) | 2–6 years old | No supplements | N.A | 5-methyltetrahydrofolate concentration in CSF and blood samples (follow up time: 30 ±8 months). | CSF 5-MTHF levels vary significantly over time in an unpredictable fashion and do not show a significant relationship to typical clinical features of autism [50]. |
| 9 | Li Q et al., (2021) [51] | Multi-center study | n= 1300 ASDs children vs. 1246 TD children | <7 years old | No supplements | N.A | Serum folate levels. Effect of serum folate level on symptoms assessed with ABC, SRS, and Childhood Autism Rating Scale (CARS) | The results showed that serum folate levels were lower in ASDs children comparing to the levels found in typically developing children. Moreover, the author underlined the necessity to evaluate folate status in children with ASDs aged three and under [51]. |
| No | Study | Type of Study | Sample Size | Age | Aim of Study | Study Outcome Parameters | Outcomes |
| 1 | Guo T et al., (2012) [52] | Population-based case-control study | n = 186 ASDs children vs. 186 control children | 8.1 (±4.3) years old | Role of the MTHFR C677T polymorphism on the autism risk in the population | Frequency of genotype MTHFR 677TT in children | The frequency of genotype MTHFR 677TT in children with autism was significantly higher than those in controls. This study suggested that MTHFR C677T is a risk factor of autism in Chinese Han children [52]. |
| 2 | Goin-Kochel RP et al., (2009) [53] | Exploratory genotype-phenotype correlations study | n = 147 ASDs children | 7.9 years old | Potential differences among MTHFR genotypes for specific behaviors | Blood samples genotyped for the MTHFR 677C-T polymorphism | The results provide preliminary evidence supporting a relationship between MTHFR 677C-T genotype and specific behaviors among children with autism [53]. |
| 3 | Santos PAC dos et al., (2010) [54] | Case-control study | n = 151 ASDs children vs. 100 healthy control children | <3 years old | Association between C677Tpolymorphism and ASDs | ADI-R criteria used for the evaluation of patient’s behavior genotype distribution of the MTHFR C667T polymorphism | The frequency of the T allele was 0.38 for the case group and 0.35 for the control group (p = 0.77). The genotypic distribution did not show significant differences between cases and controls (p = 0.72) nor association between the T allele and selected behaviors [54]. |
| 4 | Mohammad NS et al., (2016) [55] | Case-control study | n = 138 ASDs children vs. 138 non-autistic children of matched age | (4.4±1.7) years old vs. (4.4±1.6) years old | Development of an artificial neural network (ANN) model from the data of 138 autistic and 138 non-autistic children using GCPII C1561T, SHMT1 C1420T, MTHFR C677T, MTR A2756G, and MTRR A66G as the predictors of autism risk | Genetic analyses:GCPII C1561T, SHMT1 C1420T, MTHFR C677T, MTR A2756G, and MTRR A66G as predictors of autism risk.Plasma homocysteine determination | Genetic polymorphisms of the folate pathway were moderate predictors of autism risk. MTHFR C677T and hyperhomocysteinemia have been identified as risk factors for autism worldwide. Synergistic interactions between MTHFR C677T and MTRR A66G increase homocysteine [55]. |
| 5 | Ismail S et al., (2019) [56] | Case-control study | n = 78 ASDs children vs. 80matched healthy control children | 3–6 years old | Investigate the association of MTHFR gene rs1801133 (C677T) variant among ASDs children | Full clinical and radiological examinations DNA genotyped for MTHFR genetic variant (C677T) | MTHFR (C677T) allele frequency was found to be higher significantly in ASDs cases compared with non-autistic children. Additionally, there was a higher distribution of combined CT + TT genotypes among autistic patients with consanguinity and family history of psychological disease [56]. |
| 6 | Zhang Z et al., (2018) [57] | Case-control study | n = 201 ASDs children vs. 200 healthy control children | - | Association between childhood ASDs and single-nucleotide polymorphisms (SNPs) in genes involved with vitamin B12 and folate metabolism | Genotypes of transcobalamin 2 (TCN2) rs1801198, methionine synthase (MTR) rs1805087, methionine synthase reductase (MTRR) rs1801394, and methylene tetrahydrofolate reductase (MTHFR) rs1801133 were examined | Results showed no association of all examined single-nucleotide polymorphisms SNPs with childhood ASDs and its severity [57]. |
| 7 | Sener EF et al., (2014) [58] | Cohort study | n = 98 ASDs children vs. 70 age and sex-matched non-autistic children | ≦3 years old | Investigate the possible effect of C677T polymorphisms in a population cohort | DNA tested for MTHFR C677T polymorphism | MTHFR 677T-allele frequency was found to be higher in autistic children compared with non-autistic children, but it was not found statistically significant [58]. |
| 8 | Mohammad NS et al., (2009) [59] | Population study | n = 138 ASDs children vs. 138 age and sex matched nonautistic children | 2–10 years old | Investigate whether genetic polymorphisms are the underlying causes for aberrations in folate pathway reported in autistic children | DNA tested for five genetic polymorphisms: cytosolic serine hydroxyl methyl transferase (SHMT1 C1420T), methylene tetrahydrofolate reductase (MTHFR C677T, and MTHFR A1298C), methionine synthase reductase (MTRR A66G), methionine synthase (MS A2756G) | MTHFR C677T is a risk factor, whereas MTRR A66G and SHMT C1420T polymorphismsreduce the risk for autism. MTHFR A1298C acts additively in increasing the risk for autism [59]. |
| 9 | El-Baz F et al., (2017) [60] | Case-control study | n = 31 ASDs children vs. 39 children normal control group | 4.5 ± 2 years old | Identification of C677T and 1298AC polymorphic genotypes of MTHFR gene among a sample of children with autism | Identification of C677T and 1298AC polymorphic genotypes of MTHFR gene | There is a significant association between severity and occurrence of autism with MTHFR gene polymorphisms C677T and A1298C. Further studies are needed on a larger scale to explore other gene polymorphisms that may be associated with autism to correlate the genetic basis of autism [60]. |
| 10 | James SJ et al., (2010) [61] | Population-basedcase-control study | n = 529 case-parent triosvs. 566 TD controls | 3–10 years old | Investigate the frequency of common functional polymorphisms in the folate pathway | Allele frequencies of MTHFR C677T, MTHFR A1298C, TCII C776G, or MTRR A66G among mothers, fathers, or affected child compared to population controls. Determination of percent 5-methylcystosine/ total cytosine in DNA plasma transmethylation metabolites genetic relative risk and likelihood ratio test, transmission disequilibrium test, maternal plasma transmethylation metabolites and plasma folate concentrationslobal DNA methylation density and RFC1genotype association among Arkansas mothers | The results showed a significant increase in the reduced folate carrier (RFC1) G allele frequency among case mothers but not among fathers or affected children. Subsequent log linear analysis of the RFC1 A80G genotype within family trios revealed that the maternal G allele was associated with a significant increase in risk of autism, whereas the inherited genotype of the child was not.Results suggest that the maternal genetics/epigenetics may influence fetal predisposition to autism [61]. |
| No | Study | Type of Study | Sample Size | Treatment | Age | Study Outcome Parameters | Outcomes |
|---|---|---|---|---|---|---|---|
| 1 | Ramaekers V et al., (2007) [31] | Case-control study | n = 25 patients early-onset low-functioning autism vs. 25 controls age match | Folinic acid supplementation dose of 1–3 mg/kg/day3–6 months following | 2.8–12.3 years old | Serum and cerebrospinal fluid (CSF) folate level analysis and autoantibodies against FRs assay. Serum FA, vitamin B12, homocysteine, and amino acids concentrationFR1 and FR2 genotyping | Reduced CSF folate levels were observed in 19 of these 23 patients. Oral folinic acid supplementation led to normal CSF 5-MTHF levels and partial or complete clinical recovery after 12 months. Serum FR autoimmunity appears to represent an important factor in the pathogenesis of reduced folate transport to the nervous system among children with early-onset low-functioning autism associated with or without neurological deficits [31]. |
| 2 | Zhou J et al., (2018) [62] | Cohort study | n = 40 ASDs children vs. 42 gender and age matched TD children | No supplementation | Younger than 14 years old (2–6 years old) | Serum FRAA concentrations | Serum FRAA are more prevalent in children with ASDs than in TD children, suggesting that children with ASDs may have defects in folic acid absorption that play a role in the onset of ASDs [62]. |
| 3 | Frye RE et al., (2018) [63] | Double-blind randomized placebo-controlled parallel study | n = 48 ASDs children | Folinic acid (2 mg/kg) (maximum 50 mg) per day for 12 weeks | ±7 years old | Improvement of verbal communication. Development of language and communication skills | Improvement in verbal communication was significantly greater for the participants on folinic acid compared with participants on placebo with a medium-to-large effect size, particularly in those participants who were positive for FRAAs [63]. |
| No | Study | Type of Animal | Treatment Used | Study Outcome Parameters | Sample Size | Outcomes |
|---|---|---|---|---|---|---|
| 1 | Degroote S et al., (2018) [64] | Female Wistar rat | 0.2 mg/kg FA | Blood homocysteine levels. Behavioral alterations relevant to autism-like in offspring | n = 12 female, n = 8 male | Maternal periconceptional deficit in folate provokes alterations in the behavior of offspring relevant to the autistic-like phenotype [64]. |
| 2 | Barua S et al., (2016) [65] | Female C57BL/6J Mice | 2 mg/kg control diet FA (CD) 20 mg/kg high maternal FA (HMFA) | Expression of genes in the cerebral hemispheres (CHs) of 1-day-old pups, FA concentration in CH Tissue | n = 12 CD female, n = 12 HMFA female | The results showed that HMFA supplementation alters offsprings’ CH gene expression in a sex-specific manner. These changes may influence infants’ brain development. In addition, it was also found that HMFA had no impact on global DNA methylation levels of the offspring epigenome [65]. |
| 3 | Barua S et al., (2015) [67] | C57BL/6 J mice | 2 mg/kg FA,20 mg/kg FA | Expression of genes in the cerebellum of offspring from day old pups | n = 6,n = 6 | Results revealed that exposure to the higher dose FA diet during gestation dysregulated expression of several genes in the cerebellum of both male and female pups [67]. |
| 4 | Kezurer N et al., (2013) [66] | Mice on a Balb/cAnNCrlBR backgroundMTHFR heterozygote | Mild neonatal stress (25 μL PBS at postnatal days 4–10) | Offspring genotyped of DNA isolated from toe clips. Reflex analysis and newborn development. Reflex development in newborn mice. Anxiety-related behavior.Adult behavior. Corticosterone levels | n = 97 mice (7–14 in each group) were tested | Overall, the results support an interaction between mild neonatal stress, the MTHFR genotype, and sex [66]. |
| 5 | Orenbuch A et al., (2019) [27] | Mice with a Balb/cAnNCrlBR backgroundheterozygous for the Mthfr-knockout | 9 mg/mL FA in the enriched food group | Tissue concentrations of C1 metabolites in the liver, cerebral cortex, and basal forebrainMthfr/ Genotypingbehaviors | 5 groups of mice from each sex | The study suggests that MTHFR deficiency can increase the risk of ASD-like behavior in mice and that prenatal dietary intervention focused on MTHFR genotypes can reduce the risk of ASDs-like behavior. Findings emphasize the critical role of in utero C1 metabolism in developmental trajectories that lead to the presentation of autistic behavior. Aberrations in both the GABAergic and glutamatergic pathways suggest that Mthfr deficiency is linked to deleterious alterations in the basal cortical circuit activities in the affected mice [27]. |
| 6 | Sadigurschi N et al., (2019) [68] | Mice on a Balb/cAnNCrlBR backgroundheterozygote Mthfr-KO mice | No treatment | Genotyping of DNA isolated from toe clips. Immuno-fluorescence analysis of brain tissue. Morphogenic and behavioral assessments | N.S (not specified) | The study provides evidence for the profound impact of a genetic deficiency in the MTHFR gene on the induction of autistic features. In the mouse model of ASDs, this deficiency directly regulates metabolite availability and indirectly controls the environment of the developing embryonic brain [68]. |
| 7 | Sequeira JM et al., (2016) [69] | Long Evans hooded Rat | Normal diet containing 2 mg/kg FAFRα- Ab at a dose of 4μg/embryo mixed intra-peritoneal (IP) injection FRα-Ab (35μg per pup per day) on post-natal days (PND) 10, 11 and 12 (GST-PRW group). Single dose of antibody at 4.0 μg/embryo on GD8. Antibody (50μg in 0.2ml rat serum IP) on PND 26, 27, and 28 (POW group) | Behavioral tests investigated the effect of exposure to FRα antibodies (Ab) during gestation (GST), the pre-weaning (PRW), and the post weaning (POW) periods on learning and behavior in adulthood | N.S | Deficits in rats exposed to Ab during gestation and pre-weaning (GST+PRW) included indications of increased levels of anxiety. None of these rats learned the active place avoidance task, indicating severe learning deficits and cognitive impairment. Similar but less severe deficits were observed in rats exposed to Ab during GST alone or only during the PRW period, suggesting the extreme sensitivity of the fetal as well as the neonatal rat brain to the deleterious effects of exposure to Ab during this period [69]. |
| 8 | Chu D et al., (2019) [70] | ICR mice | NIH-31 open formula diet containing 2 mg /kg FA.Deionized water for 1week containing: no FA (control) 3.75 mg/L FA (MFA) 22.5 mg/L FA (HFA) | Behavioral testsRNA sequencing for postnatal day 21 mice brain. Expression of genes in adult mice brain 5 months postpartum of each group.Body weight of the offspring for 5 months | n = 6 male, n = 12 female | Results demonstrated a change in gene expression profile in weaning mice during early life FA supplementation. Long-term behavioral effects were more evident in adult male mouse offspring in a dose-sensitive mode [70]. |
| 9 | Desai A et al., (2017) [71] | Rat | Normal diet containing 2 mg/kg FAFRα Ab at a dose of 4 or 12 μg per embryo in 1 mL normal rat serum was administered by (IP) injection1 mg of folinic acid (GD7–GD12) IP and/or 0.5 mg | Folate uptake and FRα Ab localization studies. Behavioral tests | 50 | Findings suggest severe behavioral and cognitive changes mirroring ASD symptoms following gestational Ab exposure in a rat model and protection afforded by folinic acid and dexamethasone treatment [71]. |
| No | Study | Type of Study | Aim of Study | Parameters Assessed in the Study | Outcomes |
|---|---|---|---|---|---|
| 1 | DeVilbiss EA et al., (2015) [14] | Review | Overview and summaries of the folate role in neurodevelopmental disorders; relationship between maternal folate and ASDs | Maternal folate and autism spectrum disorders and related traits. Self-reported maternal folate and autism spectrum disorder traits. Maternal folate biomarker and autism spectrum disorder traits. Folate supplementation and ASDs | Inconclusive evidences underline the need for future studies of maternal folate status during the pre- and peri-conceptional periods. In addition, an incorporation of genetic data could complete better these assessments [14]. |
| 2 | Wiens D et al., (2017) [72] | Review | Examination of folic acid (FA) effects on neuronal development from tissue culture experiments, understanding ASDs metabolic causes and alternative folinic acid treatment | Unmetabolized FA neural development metabolic abnormalities.Autoantibodies in ASD autism risk | Evidence concludes that optimal levels are important for healthy development, but over-supplementation can lead to negative outcomes [72]. |
| 3 | Gao Y et al., (2016) [73] | Systematic Review | Evaluation of evidence of FA impact on neurodevelopment | FA supplementation.Maternal red blood cell (RBC) folate levels. Plasma folate | The review suggests a beneficial effect of folic acid supplementation in pregnancy on children’s neurodevelopment [73]. |
| 4 | Guo B-Q et al., (2019) [74] | Systematic Review and Meta-Analysis | Elucidate the association of maternal FA intake during the prenatal period and ASD risk in offspring | FA intake.Period of FA intake. FA intake and risk of ASDs subtypes. FA supplementation (excluding diet consumption) and risk of ASDs. Geographical area and risk of ASDs | Findings do not support the link between FA supplementation during prenatal period and ASD reduced risk in offspring. In addition, more investigation is needed because of many study limitations [74]. |
| 5 | Pu D et al., (2013) [75] | Meta-Analysis | Investigation of the MTHFR polymorphisms (C677T and A1298C) and the ASD risk | Meta-Analysis of MTHFR Polymorphisms between ASD children and controls. Distributions of MTHFR C677T/A1298C genotypes. Meta-Analysis of MTHFR C677T/A1298C polymorphisms on risk of ASD patient population based on whether they were from a country with food fortification of FA or not | This meta-analysis found that periconceptional FA supplementation may reduce ASD risk in those with MTHFR 677C>T polymorphisms where an increased risk of ASDs was indicated. The C677T polymorphism was found to be associated with ASDSs only in children from countries without food fortification [75]. |
| 6 | Cierna AV et al., (2016) [16] | Review | Investigating the methylation of cytosine bases as one of the most stable and crucial forms of epigenetic regulation of the genome | DNA methylation at different regulatory genomic elements across tissues and cell types and during different developmental stages | In genetically susceptible individuals with altered DNA-methylation patterns, a potential protective effect of supplementation taken before conception was suggested [16]. |
| 7 | Modabbernia A et al., (2017) [8] | Review | Investigating environmental risk factors for ASDs. | Advanced parental age. Pregnancy-related complications and conditions. Environmental risk factors for ASDs, Genetic and epigenetic-related effects | Studies of environmental risk factors were inconclusive as a result of significant methodological limitations [8]. |
| 8 | Dias CM et al., (2020) [2] | Review | Elucidate how genetic risk affects cellular functioning and clinical phenotypes | Roles of de novo copy number variants and single-nucleotide variants—causing loss-of-function or missense changes. Mosaic single-nucleotide variants. Inherited variants (including common variants). Rare recessive inherited variants. Noncoding variants, both inherited and de novo | Findings underline the need of whole-exome sequencing and further genome studies with increased sample size for better understanding of neuro-developmental disorders [2]. |
| 9 | Castro K et al., (2016) [76] | Review | Evaluation of serum nutrient levels and nutritional interventions targeting ASDs. | Folate intake.Serum homocysteine and folate levels.Oral folinic acid supplementation.Urine homocysteine level in children | Inconsistent conclusions were found regarding the association of FA supplementation during pregnancy and ASDs [76]. |
| 10 | Waye MMY et al., (2017) [6] | Review | Summary of genetic and epigenetic ASDs studies | Environmental risk factors. Genetic risk factors | Study evaluation concluded that although Fragile X, SHANK3, CASPR2 has been linked to ASDs risk, also folate based dietary intervention and environmental pollutants reduction might help to suppress epigenetic changes during maternity. Further study of autoantibodies against Caspr2 and folate receptor alpha has been proposed as important therapeutic targets [6]. |
| 11 | Schaevitz LR et al., (2012) [77] | Review | Focus on DNA methylation | Genetic polymorphisms. Levels of nutrients in parents and children with ASDs | Evidence underlined the important role of both nutrition and genetic components of the C1 metabolic pathway on increasing susceptibility to ASDs. Further studies are needed to better understand the different risk factors and the critical periods most essential for normal development of the brain [77]. |
| 12 | Paul L et al., (2017) [78] | Review | Summarize interaction between folate and vitamin B12 on health consequences | Folate status and biochemical markers of vitamin B12 insufficiency. Clinical outcomes associated with vitamin B12 deficiency, B vitamin imbalance during pregnancy | Negative health consequences, especially in women during pregnancy and their offspring, have been associated with impaired folate status or intake and vitamin B12 status or intake [78]. |
| 13 | Neggers Y et al., (2014) [79] | Review | Investigation of FA and autism risk | Frequency of MTHFR alleles 677C→T1298A→C in cases and controls. Plasma levels of folate metabolites. Serum folate, cerebrospinal folate, CSF 5MTHF folate receptor (FR) autoantibodies. Consumption of prenatal multivitamins and nutrients from 3 months before conception during pregnancy. Maternal FAintake. Serum FR blocking. Autoimmune antibody levels | Although results show a positive link of perinatal FA supplementation in reducing ASD incidence, recent studies underline the importance of further studies to understand the modulative role of high maternal FA intake in DNA methylation in ASD and ASD-related traits [79]. |
| 14 | Chaste P et al., (2012) [7] | Review | Summary of genetic, epigenetic, and environmental risk factors related to autism | Genetic risk factors. Environmental risk factors. Gene-environment interaction | Conclusions are inconsistent as further studies are needed to better characterize the impact of environmental factors, although an additive or multiplicative effect has been indicated [7]. |
| 15 | Frye RE et al., (2017) [80] | Review | Investigation on biomarkers used to detect folate abnormalities | Polymorphisms in folate genes related to autism environment—genome interaction. Folate metabolism FRAAs in pregnancy. Folate and ASDs | Evidence underlined the need of further studies for specific biomarkers of the folate pathway that might help to detect ASDs early and diagnose ASDs, as the abnormality of FA metabolism has a potential impact in ASD offspring. Thus, specific type and dose of folate and other cofactors could be used for treating or preventing ASD traits [80]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoxha, B.; Hoxha, M.; Domi, E.; Gervasoni, J.; Persichilli, S.; Malaj, V.; Zappacosta, B. Folic Acid and Autism: A Systematic Review of the Current State of Knowledge. Cells 2021, 10, 1976. https://doi.org/10.3390/cells10081976
Hoxha B, Hoxha M, Domi E, Gervasoni J, Persichilli S, Malaj V, Zappacosta B. Folic Acid and Autism: A Systematic Review of the Current State of Knowledge. Cells. 2021; 10(8):1976. https://doi.org/10.3390/cells10081976
Chicago/Turabian StyleHoxha, Bianka, Malvina Hoxha, Elisa Domi, Jacopo Gervasoni, Silvia Persichilli, Visar Malaj, and Bruno Zappacosta. 2021. "Folic Acid and Autism: A Systematic Review of the Current State of Knowledge" Cells 10, no. 8: 1976. https://doi.org/10.3390/cells10081976
APA StyleHoxha, B., Hoxha, M., Domi, E., Gervasoni, J., Persichilli, S., Malaj, V., & Zappacosta, B. (2021). Folic Acid and Autism: A Systematic Review of the Current State of Knowledge. Cells, 10(8), 1976. https://doi.org/10.3390/cells10081976






