Trace Amine-Associated Receptor 1 Trafficking to Cilia of Thyroid Epithelial Cells
Abstract
1. Introduction
2. Materials and Methods
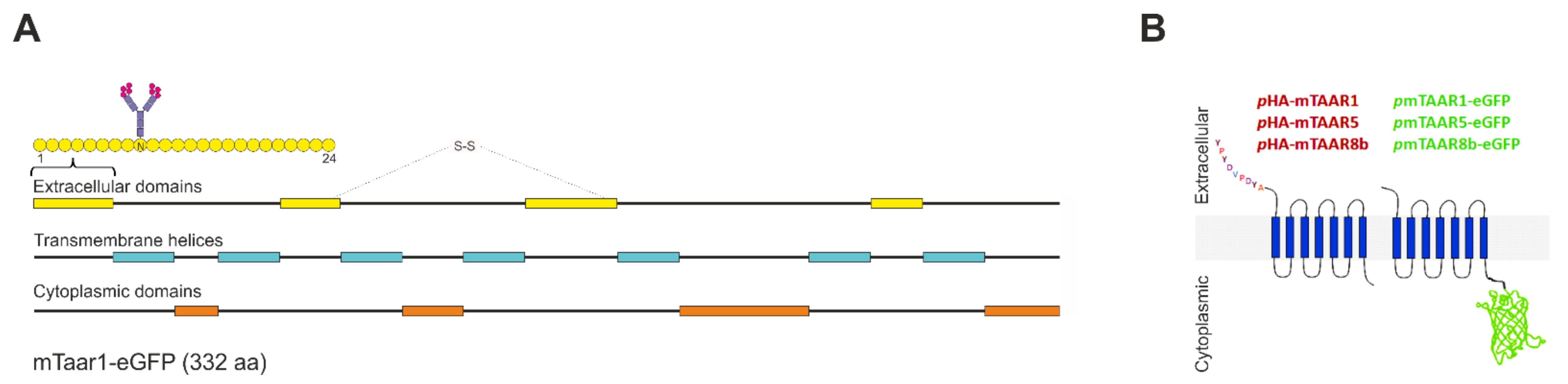
2.1. Vector Construction
2.2. Cell Culture
2.3. Transient Transfection by Electroporation
2.4. Lentiviral Transduction
2.5. Cytochemistry and Indirect Immunofluorescence
2.6. Cell Lysate Preparation, SDS-PAGE and Immunoblotting
2.7. Cell Surface Biotinylation and Streptavidin Pull-Down Experiments
2.8. Molecular Mass Calculation
3. Results
3.1. Constructs and Cell Lines Used for Stable and Transient Expression in Thyrocytes In Vitro
3.2. Chimeric mTaar1-eGFP Is Abundant in High Molecular Mass Form in KTC-Z and Nthy-Z Cells, but Primarily Monomeric Chimeras Reach the Surface of KTC-Z Cells
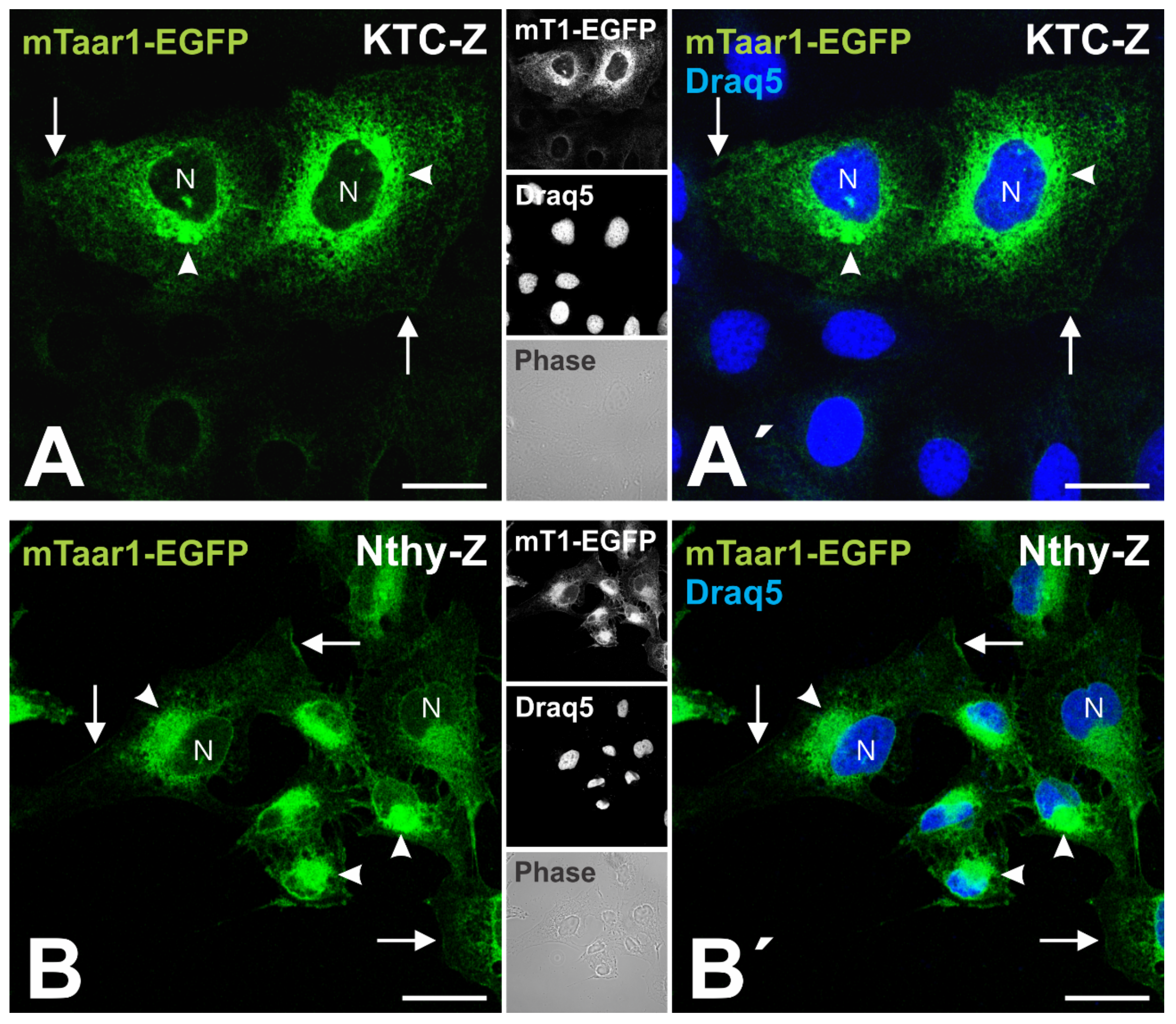
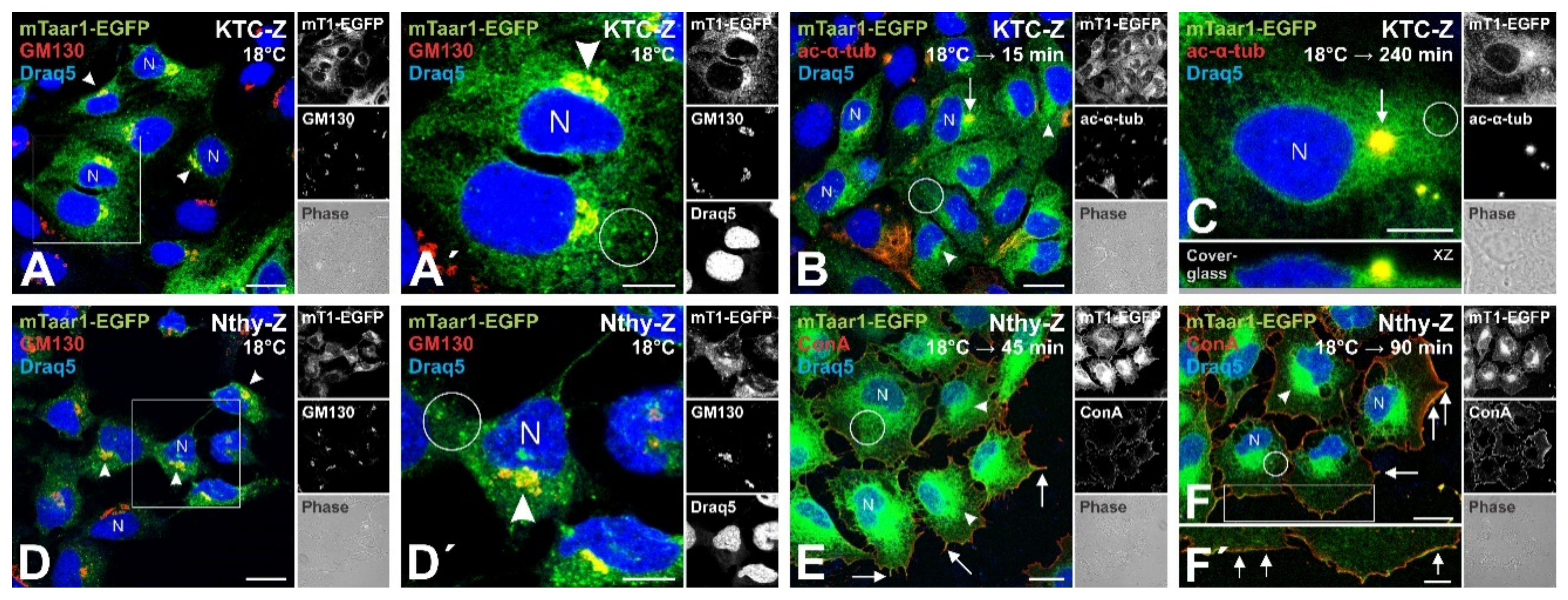
3.3. Transport of mTaar1-eGFP in Transduced, Polarized Thyroid Epithelial Cells Results in Its Targeting to and Localization at Procilia
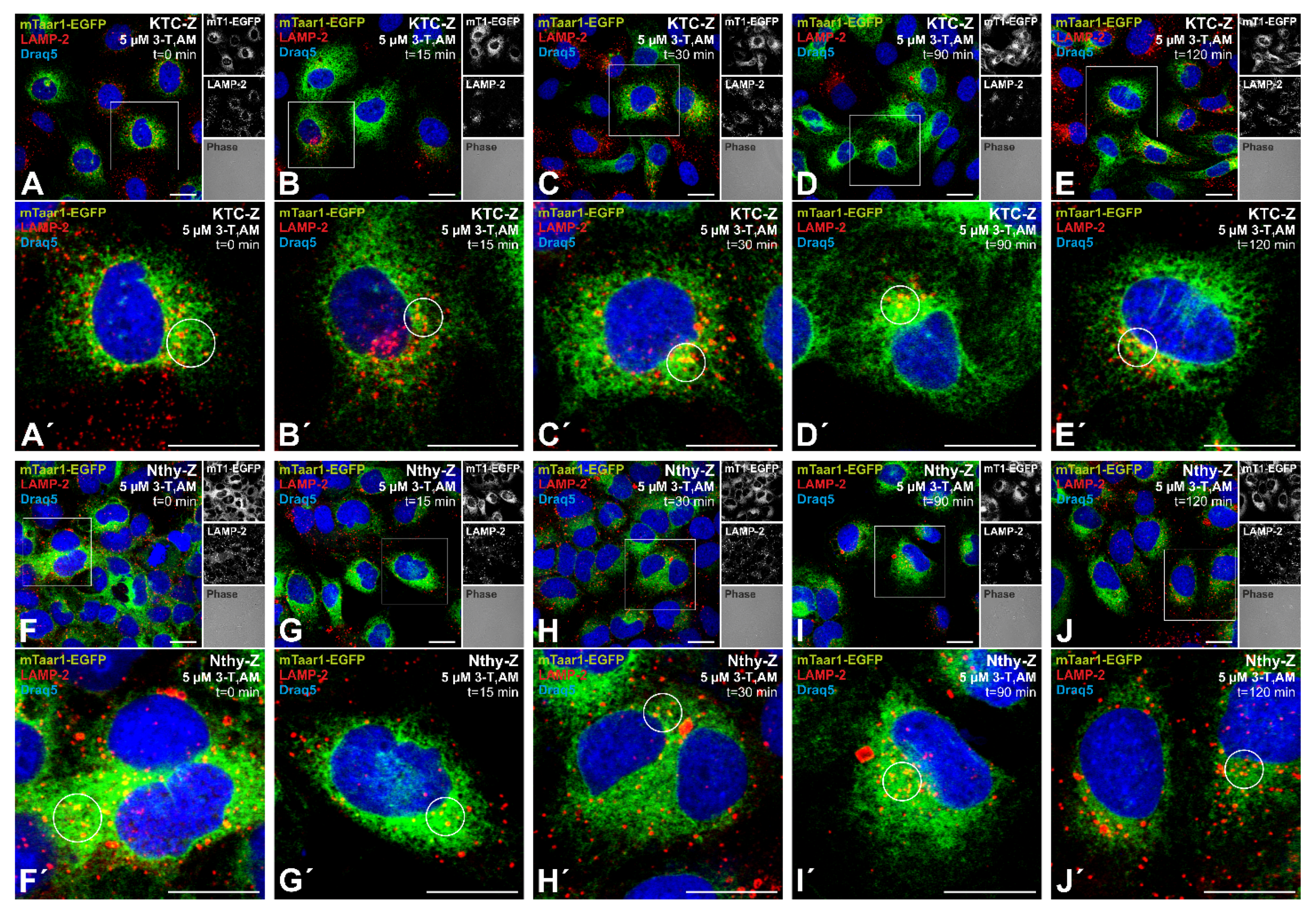
3.4. Incubation with the Putative Ligand 3-Iodothyronamine Does Not Result in Downregulation of mTaar1-EGFP from Procilia or the Cell Surface
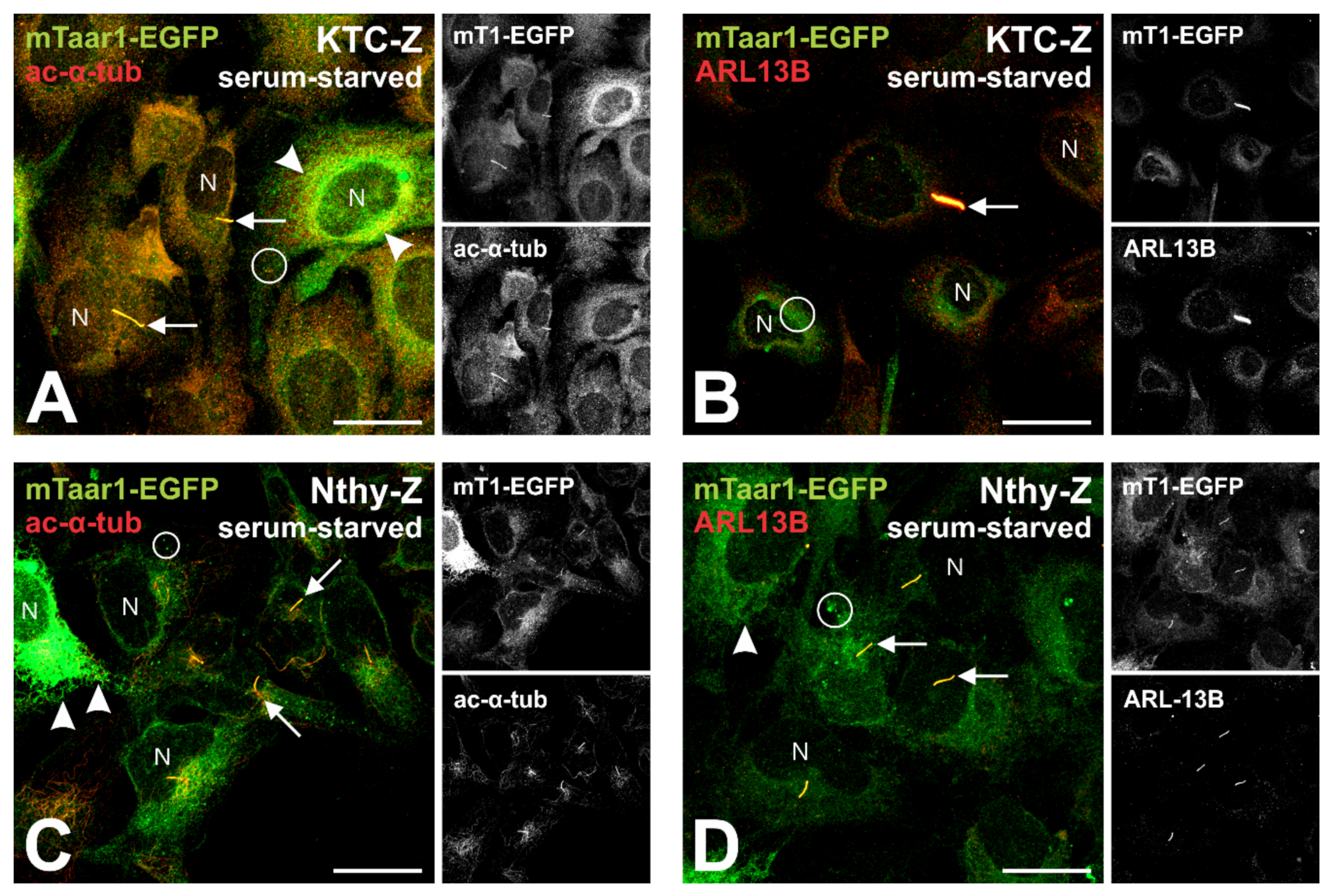
3.5. Serum-Starvation Reveals Transport of mTaar1-EGFP to Cilia of Transduced Human Thyrocytes Arrested at the G1/S-Transition
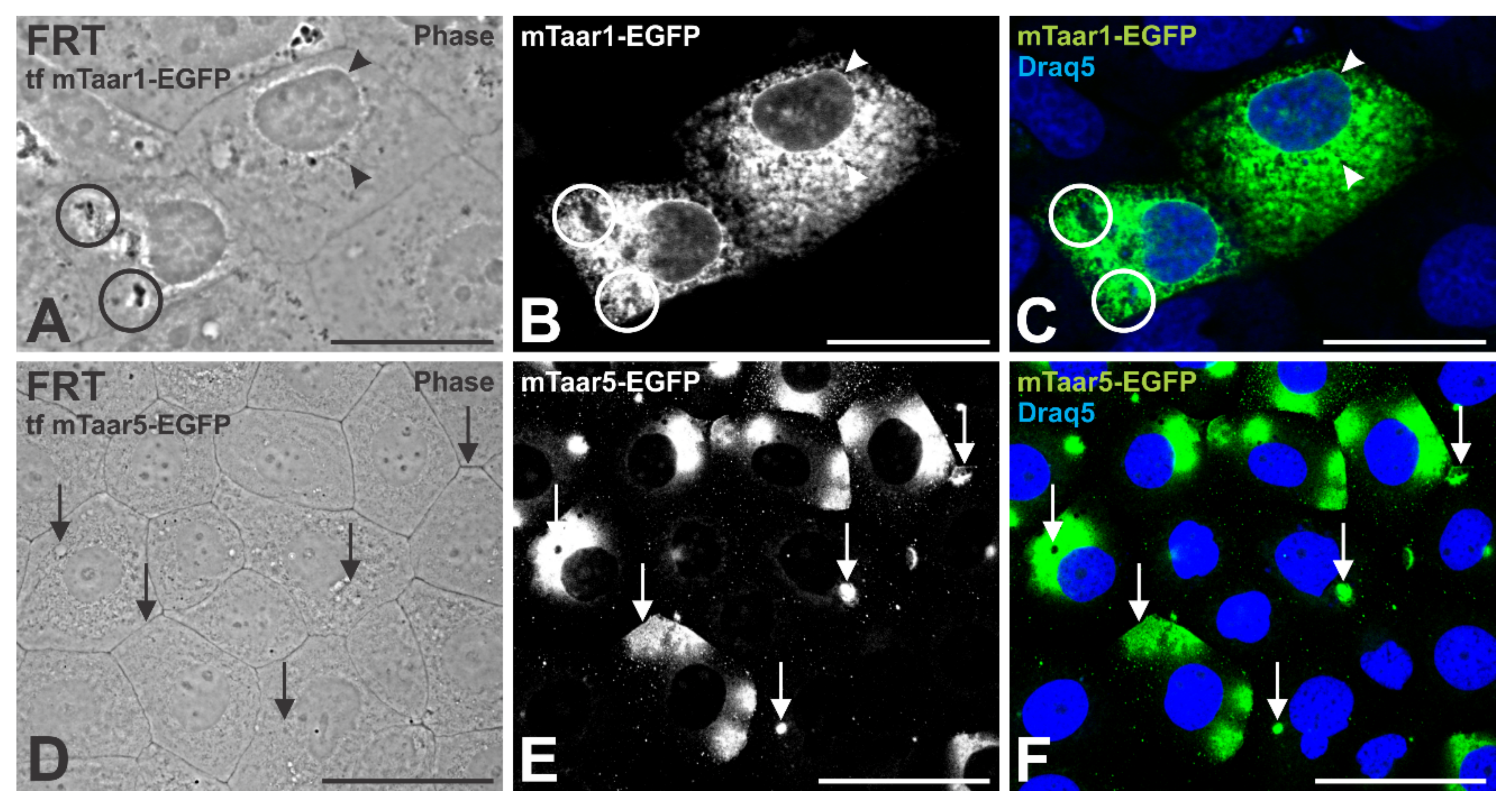
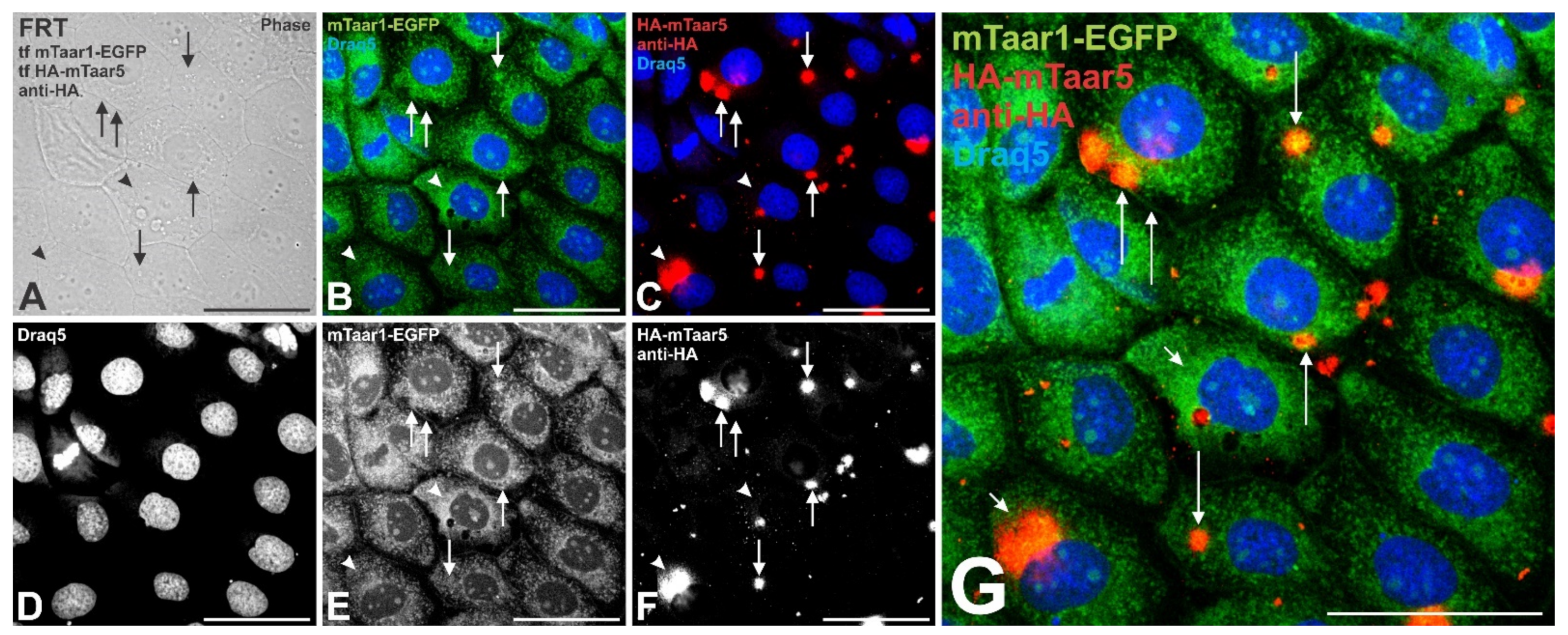
3.6. Transient Expression of a Related mTaar Protein Results in Trafficking of mTaar1-EGFP to Cilia of FRT Cells
4. Discussion
4.1. Significance of Taar1 Trafficking to Cilia of Well-Polarized Thyroid Epithelial Cells
4.2. Cilia on Human and Rodent Thyrocytes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Szumska, J.; Qatato, M.; Rehders, M.; Führer, D.; Biebermann, H.; Grandy, D.K.; Köhrle, J.; Brix, K. Trace Amine-Associated Receptor 1 Localization at the Apical Plasma Membrane Domain of Fisher Rat Thyroid Epithelial Cells Is Confined to Cilia. Eur. Thyroid J. 2015, 4 (Suppl. 1), 30–41. [Google Scholar] [CrossRef]
- Fernandez-Santos, J.M.; Utrilla, J.C.; Vazquez-Roman, V.; Villar-Rodriguez, J.L.; Gutierrez-Aviles, L.; Martin-Lacave, I. Primary Cilium in the Human Thyrocyte: Changes in Frequency and Length in Relation to the Functional Pathology of the Thyroid Gland. Thyroid 2019, 29, 595–606. [Google Scholar] [CrossRef]
- Lee, J.; Yi, S.; Chang, J.Y.; Kim, J.T.; Sul, H.J.; Park, K.C.; Zhu, X.; Cheng, S.-Y.; Kero, J.; Kim, J.; et al. Loss of Primary Cilia Results in the Development of Cancer in the Murine Thyroid Gland. Mol. Cells 2019, 42, 113–122. [Google Scholar] [PubMed]
- Szumska, J.; Batool, Z.; Al-Hashimi, A.; Venugopalan, V.; Skripnik, V.; Schaschke, N.; Bogyo, M.; Brix, K. Treatment of rat thyrocytes in vitro with cathepsin B and L inhibitors results in disruption of primary cilia leading to redistribution of the trace amine associated receptor 1 to the endoplasmic reticulum. Biochimie 2019, 166, 270–285. [Google Scholar] [CrossRef]
- Brix, K.; Szumska, J.; Weber, J.; Qatato, M.; Venugopalan, V.; Al-Hashimi, A.; Rehders, M. Auto-Regulation of the Thyroid Gland Beyond Classical Pathways. Exp. Clin. Endocrinol. Diabetes Off. J. Ger. Soc. Endocrinol. Ger. Diabetes Assoc. 2020, 128, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, M.; Hirata, N.; Tanaka, T.; Suizu, F.; Nakajima, H.; Chiorini, J.A. Autophagy as a modulator of cell death machinery. Cell Death Dis. 2020, 11, 517. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Park, K.C.; Sul, H.J.; Hong, H.J.; Kim, K.H.; Kero, J.; Shong, M. Loss of primary cilia promotes mitochondria-dependent apoptosis in thyroid cancer. Sci. Rep. 2021, 11, 4181. [Google Scholar] [CrossRef]
- Qatato, M.; Szumska, J.; Skripnik, V.; Rijntjes, E.; Köhrle, J.; Brix, K. Canonical TSH Regulation of Cathepsin-Mediated Thyroglobulin Processing in the Thyroid Gland of Male Mice Requires Taar1 Expression. Front. Pharmacol. 2018, 9, 221. [Google Scholar] [CrossRef]
- Borowsky, B.; Adham, N.; Jones, K.A.; Raddatz, R.; Artymyshyn, R.; Ogozalek, K.L.; Durkin, M.M.; Lakhlani, P.P.; Bonini, J.A.; Pathirana, S.; et al. Trace amines: Identification of a family of mammalian G protein-coupled receptors. Proc. Natl. Acad. Sci. UAS 2001, 98, 8966–8971. [Google Scholar] [CrossRef] [PubMed]
- Bunzow, J.R.; Sonders, M.S.; Arttamangkul, S.; Harrison, L.M.; Zhang, G.; Quigley, D.I.; Darland, T.; Suchland, K.L.; Pasumamula, S.; Kennedy, J.L.; et al. Amphetamine, 3,4-methylenedioxymethamphetamine, lysergic acid diethylamide, and metabolites of the catecholamine neurotransmitters are agonists of a rat trace amine receptor. Mol. Pharmacol. 2001, 60, 1181–1188. [Google Scholar] [CrossRef] [PubMed]
- Grandy, D.K. Trace amine-associated receptor 1-Family archetype or iconoclast? Pharmacol. Ther. 2007, 116, 355–390. [Google Scholar] [CrossRef]
- Köhrle, J.; Biebermann, H. 3-Iodothyronamine-A Thyroid Hormone Metabolite With Distinct Target Profiles and Mode of Action. Endocr. Rev. 2019, 40, 602–630. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.M. The emerging role of trace amine-associated receptor 1 in the functional regulation of monoamine transporters and dopaminergic activity. J. Neurochem. 2011, 116, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Al-Hashimi, A.; Venugopalan, V.; Sereesongsaeng, N.; Tedelind, S.; Pinzaru, A.M.; Hein, Z.; Springer, S.; Weber, E.; Führer, D.; Scott, C.J.; et al. Significance of nuclear cathepsin V in normal thyroid epithelial and carcinoma cells. Biochim. Et Biophys. Acta. Mol. Cell Res. 2020, 1867, 118846. [Google Scholar] [CrossRef] [PubMed]
- Lindemann, L.; Ebeling, M.; Kratochwil, N.A.; Bunzow, J.R.; Grandy, D.K.; Hoener, M.C. Trace amine-associated receptors form structurally and functionally distinct subfamilies of novel G protein-coupled receptors. Genomics 2005, 85, 372–385. [Google Scholar] [CrossRef]
- Dinter, J.; Mühlhaus, J.; Wienchol, C.L.; Yi, C.X.; Nürnberg, D.; Morin, S.; Grüters, A.; Köhrle, J.; Schöneberg, T.; Tschöp, M.; et al. Inverse agonistic action of 3-iodothyronamine at the human trace amine-associated receptor 5. PLoS ONE 2015, 10, e0117774. [Google Scholar] [CrossRef] [PubMed]
- Mühlhaus, J.; Dinter, J.; Nürnberg, D.; Rehders, M.; Depke, M.; Golchert, J.; Homuth, G.; Yi, C.X.; Morin, S.; Köhrle, J.; et al. Analysis of human TAAR8 and murine Taar8b mediated signaling pathways and expression profile. Int. J. Mol. Sci. 2014, 15, 20638–20655. [Google Scholar] [CrossRef] [PubMed]
- Hanenberg, H.; Xiao, X.L.; Dilloo, D.; Hashino, K.; Kato, I.; Williams, D.A. Colocalization of retrovirus and target cells on specific fibronectin fragments increases genetic transduction of mammalian cells. Nat. Med. 1996, 2, 876–882. [Google Scholar] [CrossRef] [PubMed]
- Hein, Z.; Uchtenhagen, H.; Abualrous, E.T.; Saini, S.K.; Janßen, L.; Van Hateren, A.; Wiek, C.; Hanenberg, H.; Momburg, F.; Achour, A.; et al. Peptide-independent stabilization of MHC class I molecules breaches cellular quality control. J. Cell Sci. 2014, 127, 2885–2897. [Google Scholar] [CrossRef]
- Kurebayashi, J.; Tanaka, K.; Otsuki, T.; Moriya, T.; Kunisue, H.; Uno, M.; Sonoo, H. All-trans-retinoic acid modulates expression levels of thyroglobulin and cytokines in a new human poorly differentiated papillary thyroid carcinoma cell line, KTC-1. J. Clin. Endocrinol. Metab. 2000, 85, 2889–2896. [Google Scholar]
- Tedelind, S.; Jordans, S.; Resemann, H.; Blum, G.; Bogyo, M.; Führer, D.; Brix, K. Cathepsin B trafficking in thyroid carcinoma cells. Thyroid Res. 2011, 4 (Suppl. 1), S2. [Google Scholar] [CrossRef]
- Lemoine, N.R.; Mayall, E.S.; Jones, T.; Sheer, D.; McDermid, S.; Kendall-Taylor, P.; Wynford-Thomas, D. Characterisation of human thyroid epithelial cells immortalised in vitro by simian virus 40 DNA transfection. Br. J. Cancer 1989, 60, 897–903. [Google Scholar] [CrossRef] [PubMed]
- van den Hoff, M.J.; Moorman, A.F.; Lamers, W.H. Electroporation in ‘intracellular’ buffer increases cell survival. Nucleic Acids Res. 1992, 20, 2902. [Google Scholar] [CrossRef] [PubMed]
- Halenius, A.; Hauka, S.; Dölken, L.; Stindt, J.; Reinhard, H.; Wiek, C.; Hanenberg, H.; Koszinowski, U.H.; Momburg, F.; Hengel, H. Human cytomegalovirus disrupts the major histocompatibility complex class I peptide-loading complex and inhibits tapasin gene transcription. J. Virol. 2011, 85, 3473–3485. [Google Scholar] [CrossRef] [PubMed]
- Neuhoff, V.; Philipp, K.; Zimmer, H.G.; Mesecke, S. A simple, versatile, sensitive and volume-independent method for quantitative protein determination which is independent of other external influences. Hoppe Seylers Z. Physiol. Chem. 1979, 360, 1657–1670. [Google Scholar] [CrossRef]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Kyhse-Andersen, J. Electroblotting of multiple gels: A simple apparatus without buffer tank for rapid transfer of proteins from polyacrylamide to nitrocellulose. J. Biochem. Biophys. Methods 1984, 10, 203–209. [Google Scholar] [CrossRef]
- Brix, K.; Summa, W.; Lottspeich, F.; Herzog, V. Extracellularly occurring histone H1 mediates the binding of thyroglobulin to the cell surface of mouse macrophages. J. Clin. Investig. 1998, 102, 283–293. [Google Scholar] [CrossRef]
- Schweppe, R.E.; Klopper, J.P.; Korch, C.; Pugazhenthi, U.; Benezra, M.; Knauf, J.A.; Fagin, J.A.; Marlow, L.A.; Copland, J.A.; Smallridge, R.C.; et al. Deoxyribonucleic acid profiling analysis of 40 human thyroid cancer cell lines reveals cross-contamination resulting in cell line redundancy and misidentification. J. Clin. Endocrinol. Metab. 2008, 93, 4331–4341. [Google Scholar] [CrossRef]
- Suzuki, K.; Mitsutake, N.; Saenko, V.; Suzuki, M.; Matsuse, M.; Ohtsuru, A.; Kumagai, A.; Uga, T.; Yano, H.; Nagayama, Y.; et al. Dedifferentiation of human primary thyrocytes into multilineage progenitor cells without gene introduction. PLoS ONE 2011, 6, e19354. [Google Scholar] [CrossRef]
- Gierke, S.; Kumar, P.; Wittmann, T. Analysis of microtubule polymerization dynamics in live cells. Methods Cell Biol. 2010, 97, 15–33. [Google Scholar] [PubMed]
- Calebiro, D.; Godbole, A. Internalization of G-protein-coupled receptors: Implication in receptor function, physiology and diseases. Best Pract. Res. Clin. Endocrinol. Metab. 2018, 32, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Schanze, N.; Jacobi, S.F.; Rijntjes, E.; Mergler, S.; Del Olmo, M.; Hoefig, C.S.; Khajavi, N.; Lehmphul, I.; Biebermann, H.; Mittag, J.; et al. 3-Iodothyronamine Decreases Expression of Genes Involved in Iodide Metabolism in Mouse Thyroids and Inhibits Iodide Uptake in PCCL3 Thyrocytes. Thyroid 2017, 27, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Wolinsky, T.D.; Swanson, C.J.; Smith, K.E.; Zhong, H.; Borowsky, B.; Seeman, P.; Branchek, T.; Gerald, C.P. The Trace Amine 1 receptor knockout mouse: An animal model with relevance to schizophrenia. Genes Brain Behav. 2007, 6, 628–639. [Google Scholar] [CrossRef] [PubMed]
- Lindemann, L.; Meyer, C.A.; Jeanneau, K.; Bradaia, A.; Ozmen, L.; Bluethmann, H.; Bettler, B.; Wettstein, J.G.; Borroni, E.; Moreau, J.L.; et al. Trace amine-associated receptor 1 modulates dopaminergic activity. J. Pharmacol. Exp. Ther. 2008, 324, 948–956. [Google Scholar] [CrossRef]
- Revel, F.G.; Moreau, J.L.; Gainetdinov, R.R.; Bradaia, A.; Sotnikova, T.D.; Mory, R.; Durkin, S.; Zbinden, K.G.; Norcross, R.; Meyer, C.A.; et al. TAAR1 activation modulates monoaminergic neurotransmission, preventing hyperdopaminergic and hypoglutamatergic activity. Proc. Natl. Acad. Sci. USA 2011, 108, 8485–8490. [Google Scholar] [CrossRef]
- Xie, Z.; Miller, G.M. Trace amine-associated receptor 1 as a monoaminergic modulator in brain. Biochem. Pharmacol. 2009, 78, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Jing, L.; Li, J.X. Trace amine-associated receptor 1: A promising target for the treatment of psychostimulant addiction. Eur. J. Pharmacol. 2015, 761, 345–352. [Google Scholar] [CrossRef]
- Pei, Y.; Asif-Malik, A.; Canales, J.J. Trace Amines and the Trace Amine-Associated Receptor 1: Pharmacology, Neurochemistry, and Clinical Implications. Front. Neurosci. 2016, 10, 148. [Google Scholar] [CrossRef]
- Panetta, R.; Greenwood, M.T. Physiological relevance of GPCR oligomerization and its impact on drug discovery. Drug Discov. Today 2008, 13, 1059–1066. [Google Scholar] [CrossRef]
- Milligan, G. G protein-coupled receptor hetero-dimerization: Contribution to pharmacology and function. Br. J. Pharmacol. 2009, 158, 5–14. [Google Scholar] [CrossRef]
- Smith, N.J.; Milligan, G. Allostery at G protein-coupled receptor homo- and heteromers: Uncharted pharmacological landscapes. Pharmacol. Rev. 2010, 62, 701–725. [Google Scholar] [CrossRef]
- Babusyte, A.; Kotthoff, M.; Fiedler, J.; Krautwurst, D. Biogenic amines activate blood leukocytes via trace amine-associated receptors TAAR1 and TAAR2. J. Leukoc. Biol. 2013, 93, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Espinoza, S.; Salahpour, A.; Masri, B.; Sotnikova, T.D.; Messa, M.; Barak, L.S.; Caron, M.G.; Gainetdinov, R.R. Functional interaction between trace amine-associated receptor 1 and dopamine D2 receptor. Mol. Pharmacol. 2011, 80, 416–425. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.M.; Verrico, C.D.; Jassen, A.; Konar, M.; Yang, H.; Panas, H.; Bahn, M.; Johnson, R.; Madras, B.K. Primate trace amine receptor 1 modulation by the dopamine transporter. J. Pharmacol. Exp. Ther. 2005, 313, 983–994. [Google Scholar] [CrossRef] [PubMed]
- Barak, L.S.; Salahpour, A.; Zhang, X.; Masri, B.; Sotnikova, T.D.; Ramsey, A.J.; Violin, J.D.; Lefkowitz, R.J.; Caron, M.G.; Gainetdinov, R.R. Pharmacological characterization of membrane-expressed human trace amine-associated receptor 1 (TAAR1) by a bioluminescence resonance energy transfer cAMP biosensor. Mol. Pharmacol. 2008, 74, 585–594. [Google Scholar] [CrossRef]
- Piperno, G.; LeDizet, M.; Chang, X.J. Microtubules containing acetylated alpha-tubulin in mammalian cells in culture. J. Cell Biol. 1987, 104, 289–302. [Google Scholar] [CrossRef]
- Dacheux, D.; Roger, B.; Bosc, C.; Landrein, N.; Roche, E.; Chansel, L.; Trian, T.; Andrieux, A.; Papaxanthos-Roche, A.; Marthan, R.; et al. Human FAM154A (SAXO1) is a microtubule-stabilizing protein specific to cilia and related structures. J. Cell Sci. 2015, 128, 1294–1307. [Google Scholar] [CrossRef]
- Duvernay, M.T.; Dong, C.; Zhang, X.; Zhou, F.; Nichols, C.D.; Wu, G. Anterograde trafficking of G protein-coupled receptors: Function of the C-terminal F(X)6LL motif in export from the endoplasmic reticulum. Mol. Pharmacol. 2009, 75, 751–761. [Google Scholar] [CrossRef]
- Brix, K.; Qatato, M.; Szumska, J.; Venugopalan, V.; Rehders, M. Thyroglobulin Storage, Processing and Degradation for Thyroid Hormone Liberation. In The Thyroid and Its Diseases; Luster, M., Duntas, L.H., Wartofsky, L., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2019; pp. 25–48. [Google Scholar]
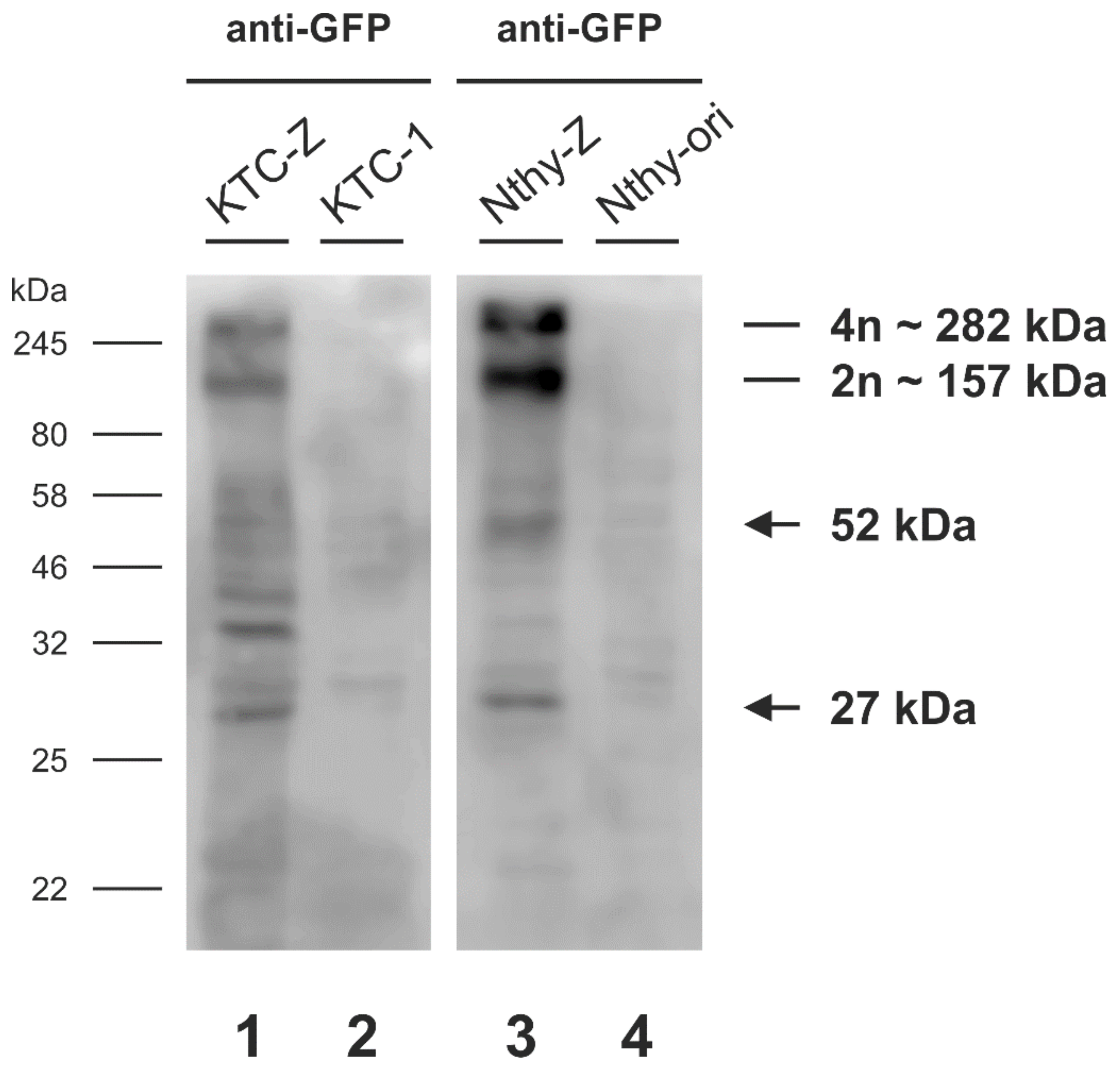
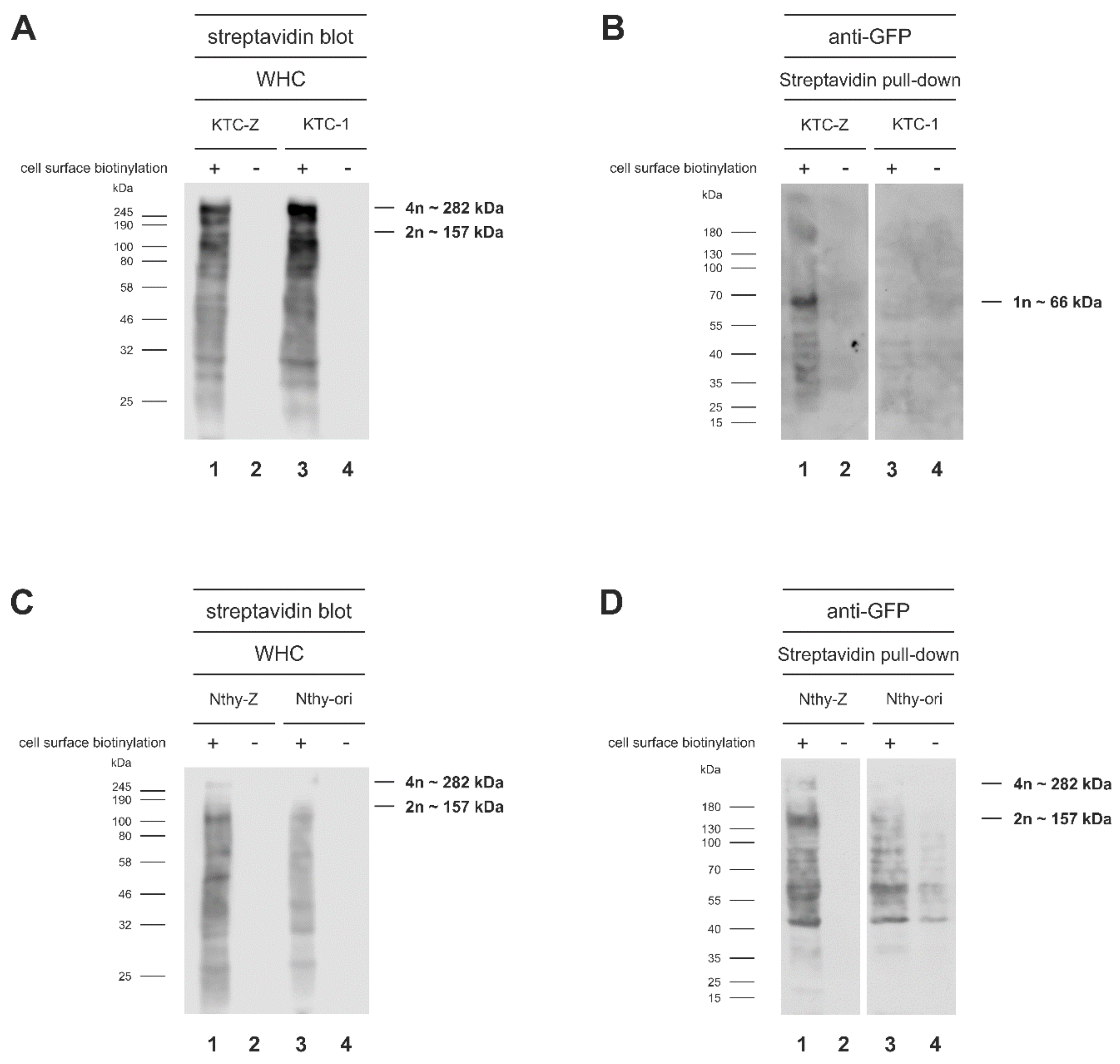
| Immunoblot | HMM Band 1 | HMM Band 2 |
|---|---|---|
| KTC-Z Blot 1 | 182 | 289.5 |
| KTC-Z Blot 2 | 135 | 286.5 |
| KTC-Z Blot 3 | 164 | 289 |
| Means for KTC-Z | 160.3 | 288.3 |
| per 65.8 kDa monomer | 2.437 | 4.382 |
| Nthy-Z Blot 1 | 135 | 274 |
| Nthy-Z Blot 2 | 153 | 307 |
| Nthy-Z Blot 3 | 167 | 296 |
| Nthy-Z Blot 4 | 156 | 222 |
| Means for Nthy-Z | 152.8 | 274.8 |
| per 65.8 kDa monomer | 2.321 | 4.176 |
| Average across cell lines | 157 | 282 |
| per 65.8 kDa monomer | 2.4 | 4.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qatato, M.; Venugopalan, V.; Al-Hashimi, A.; Rehders, M.; Valentine, A.D.; Hein, Z.; Dallto, U.; Springer, S.; Brix, K. Trace Amine-Associated Receptor 1 Trafficking to Cilia of Thyroid Epithelial Cells. Cells 2021, 10, 1518. https://doi.org/10.3390/cells10061518
Qatato M, Venugopalan V, Al-Hashimi A, Rehders M, Valentine AD, Hein Z, Dallto U, Springer S, Brix K. Trace Amine-Associated Receptor 1 Trafficking to Cilia of Thyroid Epithelial Cells. Cells. 2021; 10(6):1518. https://doi.org/10.3390/cells10061518
Chicago/Turabian StyleQatato, Maria, Vaishnavi Venugopalan, Alaa Al-Hashimi, Maren Rehders, Aaron D. Valentine, Zeynep Hein, Uillred Dallto, Sebastian Springer, and Klaudia Brix. 2021. "Trace Amine-Associated Receptor 1 Trafficking to Cilia of Thyroid Epithelial Cells" Cells 10, no. 6: 1518. https://doi.org/10.3390/cells10061518
APA StyleQatato, M., Venugopalan, V., Al-Hashimi, A., Rehders, M., Valentine, A. D., Hein, Z., Dallto, U., Springer, S., & Brix, K. (2021). Trace Amine-Associated Receptor 1 Trafficking to Cilia of Thyroid Epithelial Cells. Cells, 10(6), 1518. https://doi.org/10.3390/cells10061518






