Autonomy in the Development of Stem Cell-Derived Embryoids: Sprouting Blastocyst-Like Cysts, and Ethical Implications
Abstract
1. Introduction
2. Some Relevant Facts about Mammalian Embryogenesis
3. Autonomy in the Morphogenesis of Stem Cell-Derived Embryoids
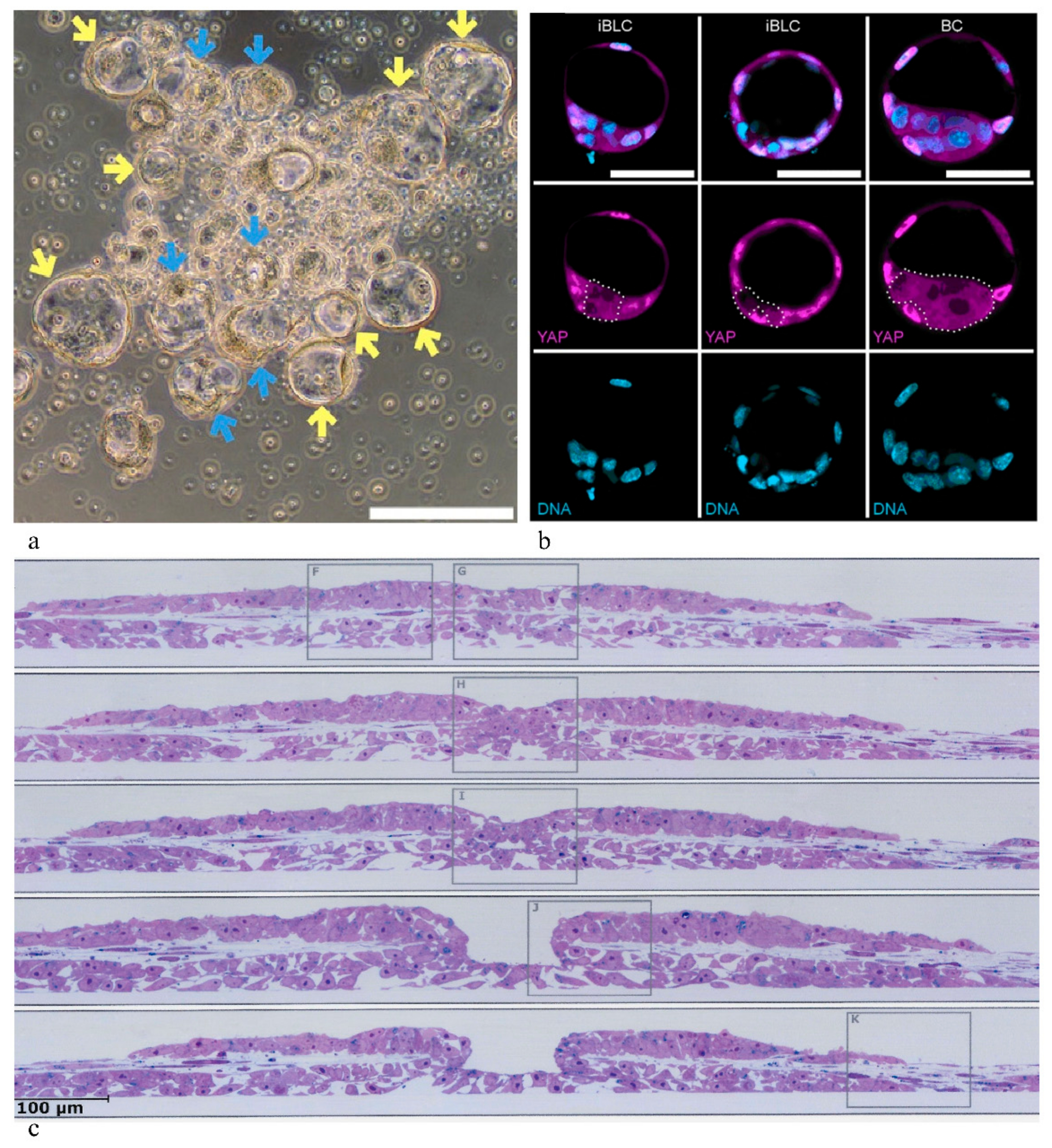
4. Spontaneous Budding of Blastocyst-Like Cysts in Stem Cell Cultures
4.1. Implantation Potential of Blastocyst-Like Cysts, and Endometrial Response
4.2. Extended/Expanded Potential Stem Cells, 2C-Like Cells
5. Conclusions and Ethical Implications
6. Note Added in Proof
Funding
Acknowledgments
Conflicts of Interest
References
- Matthews, K.R.W.; Wagner, D.S.; Warmflash, A. Stem cell-based models of embryos: The need for improved naming conventions. Stem Cell Rep. 2021, 16, 1014–1020. [Google Scholar] [CrossRef]
- Gupta, A.; Lutolf, M.P.; Hughes, A.J.; Sonnen, K.F. Bioengineering in vitro models of embryonic development. Stem Cell Rep. 2021, 16, 1104–1116. [Google Scholar] [CrossRef]
- White, M.D.; Plachta, N. Specification of the First Mammalian Cell Lineages In Vivo and In Vitro. Cold Spring Harb. Perspect. Biol. 2020, 12, a035634. [Google Scholar] [CrossRef]
- Posfai, E.; Schell, J.P.; Janiszewski, A.; Rovic, I.; Murray, A.; Bradshaw, B.; Yamakawa, T.; Pardon, T.; El Bakkali, M.; Talon, I.; et al. Evaluating totipotency using criteria of increasing stringency. Nat. Cell Biol. 2021, 23, 49–60. [Google Scholar] [CrossRef]
- Shahbazi, M.N.; Siggia, E.D.; Zernicka-Goetz, M. Self-organization of stem cells into embryos: A window on early mammalian development. Science 2019, 364, 948–951. [Google Scholar] [CrossRef]
- Rossant, J.; Tam, P.P.L. Opportunities and Challenges with Stem Cell-Based Embryo Models. Stem Cell Rep. 2021, 16, 1031–1038. [Google Scholar] [CrossRef]
- Rivron, N.; Fu, J. It takes a village to form embryo models. Stem Cell Rep. 2021, 16, 1011–1013. [Google Scholar] [CrossRef]
- Baillie-Benson, P.; Moris, N.; Martinez Arias, A. Pluripotent stem cell models of early mammalian development. Curr. Opin. Cell Biol. 2020, 66, 89–96. [Google Scholar] [CrossRef]
- Posfai, E.; Lanner, F.; Mulas, C.; Leitch, H.G. All models are wrong, but some are useful: Establishing standards for stem cell-based embryo models. Stem Cell Rep. 2021, 16, 1117–1141. [Google Scholar] [CrossRef]
- Denker, H.-W. Cell lineage, determination and differentiation in earliest developmental stages in mammals. In Problems of the Keimbahn. New Work on Mammalian Germ Cell Lineage; Hilscher, W., Ed.; S. Karger AG: Basel, Switzerland, 1983; Volume 24, pp. 22–58. [Google Scholar]
- Aach, J.; Lunshof, J.; Iyer, E.; Church, G.M. Addressing the ethical issues raised by synthetic human entities with embryo-like features. eLife 2017, 6, e20674. [Google Scholar] [CrossRef]
- Denker, H.-W. Embryonic stem cells: An exciting field for basic research and tissue engineering, but also an ethical dilemma? Cells Tissues Organs 1999, 165, 246–249. [Google Scholar] [CrossRef]
- Denker, H.-W. Potentiality of embryonic stem cells: An ethical problem even with alternative stem cell sources. J. Med. Ethics 2006, 32, 665–671. [Google Scholar] [CrossRef]
- Denker, H.-W. Early human development: New data raise important embryological and ethical questions relevant for stem cell research. Naturwissenschaften 2004, 91, 1–21. [Google Scholar] [CrossRef]
- Pera, M.F.; de Wert, G.; Dondorp, W.; Lovell-Badge, R.; Mummery, C.L.; Munsie, M.; Tam, P.P. What if stem cells turn into embryos in a dish? Nat. Methods 2015, 12, 917–919. [Google Scholar] [CrossRef] [PubMed]
- Deglincerti, A.; Croft, G.F.; Pietila, L.N.; Zernicka-Goetz, M.; Siggia, E.D.; Brivanlou, A.H. Self-organization of the in vitro attached human embryo. Nature 2016, 533, 251–254. [Google Scholar] [CrossRef]
- Shahbazi, M.N.; Jedrusik, A.; Vuoristo, S.; Recher, G.; Hupalowska, A.; Bolton, V.; Fogarty, N.M.; Campbell, A.; Devito, L.G.; Ilic, D.; et al. Self-organization of the human embryo in the absence of maternal tissues. Nat. Cell Biol. 2016, 18, 700–708. [Google Scholar] [CrossRef]
- Turner, D.A.; Girgin, M.; Alonso-Crisostomo, L.; Trivedi, V.; Baillie-Johnson, P.; Glodowski, C.R.; Hayward, P.C.; Collignon, J.; Gustavsen, C.; Serup, P.; et al. Anteroposterior polarity and elongation in the absence of extra-embryonic tissues and of spatially localised signalling in gastruloids: Mammalian embryonic organoids. Development 2017, 144, 3894–3906. [Google Scholar] [CrossRef] [PubMed]
- ten Berge, D.; Koole, W.; Fuerer, C.; Fish, M.; Eroglu, E.; Nusse, R. Wnt signaling mediates self-organization and axis formation in embryoid bodies. Cell Stem Cell 2008, 3, 508–518. [Google Scholar] [CrossRef]
- Van den Brink, S.C.; Baillie-Johnson, P.; Balayo, T.; Hadjantonakis, A.K.; Nowotschin, S.; Turner, D.A.; Martinez Arias, A. Symmetry breaking, germ layer specification and axial organisation in aggregates of mouse embryonic stem cells. Development 2014, 141, 4231–4242. [Google Scholar] [CrossRef]
- Van den Brink, S.C.; Alemany, A.; van Batenburg, V.; Moris, N.; Blotenburg, M.; Vivié, J.; Baillie-Johnson, P.; Nichols, J.; Sonnen, K.F.; Martinez Arias, A.; et al. Single-cell and spatial transcriptomics reveal somitogenesis in gastruloids. Nature 2020, 582, 402–409. [Google Scholar] [CrossRef]
- Harrison, S.E.; Sozen, B.; Christodoulou, N.; Kyprianou, C.; Zernicka-Goetz, M. Assembly of embryonic and extra-embryonic stem cells to mimic embryogenesis in vitro. Science 2017, 356, eaal1810. [Google Scholar] [CrossRef]
- Harrison, S.E.; Sozen, B.; Zernicka-Goetz, M. In vitro generation of mouse polarized embryo-like structures from embryonic and trophoblast stem cells. Nat. Protoc. 2018, 13, 1586–1602. [Google Scholar] [CrossRef]
- Sozen, B.; Amadei, G.; Cox, A.; Wang, R.; Na, E.; Czukiewska, S.; Chappell, L.; Voet, T.; Michel, G.; Jing, N.; et al. Self-assembly of embryonic and two extra-embryonic stem cell types into gastrulating embryo-like structures. Nat. Cell Biol. 2018, 20, 979–989. [Google Scholar] [CrossRef]
- Sozen, B.; Cox, A.L.; De Jonghe, J.; Bao, M.; Hollfelder, F.; Glover, D.M.; Zernicka-Goetz, M. Self-Organization of Mouse Stem Cells into an Extended Potential Blastoid. Dev. Cell 2019, 51, 698.e8–712.e8. [Google Scholar] [CrossRef]
- Zhang, S.; Chen, T.; Chen, N.; Gao, D.; Shi, B.; Kong, S.; West, R.C.; Yuan, Y.; Zhi, M.; Wei, Q.; et al. Implantation initiation of self-assembled embryo-like structures generated using three types of mouse blastocyst-derived stem cells. Nat. Commun. 2019, 10, 496. [Google Scholar] [CrossRef]
- Rivron, N.C.; Frias-Aldeguer, J.; Vrij, E.J.; Boisset, J.C.; Korving, J.; Vivie, J.; Truckenmuller, R.K.; van Oudenaarden, A.; van Blitterswijk, C.A.; Geijsen, N. Blastocyst-like structures generated solely from stem cells. Nature 2018, 557, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Moris, N.; Anlas, K.; van den Brink, S.C.; Alemany, A.; Schroder, J.; Ghimire, S.; Balayo, T.; van Oudenaarden, A.; Martinez Arias, A. An in vitro model of early anteroposterior organization during human development. Nature 2020, 582, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Taniguchi, K.; Townshend, R.F.; Miki, T.; Gumucio, D.L.; Fu, J. A pluripotent stem cell-based model for post-implantation human amniotic sac development. Nat. Commun. 2017, 8, 208. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Wang, P.; Sozen, B.; Qiu, H.; Zhu, Y.; Zhang, X.; Ming, J.; Zernicka-Goetz, M.; Na, J. Machine learning-assisted high-content analysis of pluripotent stem cell-derived embryos in vitro. Stem Cell Rep. 2021, 16, 1331–1346. [Google Scholar] [CrossRef] [PubMed]
- Hyun, I.; Wilkerson, A.; Johnston, J. Embryology policy: Revisit the 14-day rule. Nature 2016, 533, 169–171. [Google Scholar] [CrossRef]
- Matthews, K.R.; Morali, D. National human embryo and embryoid research policies: A survey of 22 top research-intensive countries. Regen. Med. 2020, 15, 1905–1917. [Google Scholar] [CrossRef]
- Hyun, I.; Munsie, M.; Pera, M.F.; Rivron, N.C.; Rossant, J. Toward Guidelines for Research on Human Embryo Models Formed from Stem Cells. Stem Cell Rep. 2020, 14, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Nicolas, P.; Etoc, F.; Brivanlou, A.H. The ethics of human-embryoids model: A call for consistency. J. Mol. Med. 2021, 99, 569–579. [Google Scholar] [CrossRef]
- Hyun, I.; Bredenoord, A.L.; Briscoe, J.; Klipstein, S.; Tan, T. Human embryo research beyond the primitive streak. Science 2021, 371, 998–1000. [Google Scholar] [CrossRef]
- Pereira Daoud, A.M.; Popovic, M.; Dondorp, W.J.; Trani Bustos, M.; Bredenoord, A.L.; Chuva de Sousa Lopes, S.M.; van den Brink, S.C.; Roelen, B.A.J.; de Wert, G.; Heindryckx, B. Modelling human embryogenesis: Embryo-like structures spark ethical and policy debate. Hum. Reprod. Update 2020, 26, 779–798. [Google Scholar] [CrossRef]
- Barzegari, A.; Gueguen, V.; Omidi, Y.; Ostadrahimi, A.; Nouri, M.; Pavon-Djavid, G. The role of Hippo signaling pathway and mechanotransduction in tuning embryoid body formation and differentiation. J. Cell Physiol. 2020, 235, 5072–5083. [Google Scholar] [CrossRef]
- Liu, X.; Tan, J.P.; Schröder, J.; Aberkane, A.; Ouyang, J.F.; Mohenska, M.; Lim, S.M.; Sun, Y.B.Y.; Chen, J.; Sun, G.; et al. Modelling human blastocysts by reprogramming fibroblasts into iBlastoids. Nature 2021, 591, 627–632. [Google Scholar] [CrossRef]
- Li, R.; Zhong, C.; Yu, Y.; Liu, H.; Sakurai, M.; Yu, L.; Min, Z.; Shi, L.; Wei, Y.; Takahashi, Y.; et al. Generation of Blastocyst-like Structures from Mouse Embryonic and Adult Cell Cultures. Cell 2019, 179, 687–702 e18. [Google Scholar] [CrossRef]
- Junyent, S.; Garcin, C.L.; Szczerkowski, J.L.A.; Trieu, T.J.; Reeves, J.; Habib, S.J. Specialized cytonemes induce self-organization of stem cells. Proc. Natl. Acad. Sci. USA 2020, 117, 7236–7244. [Google Scholar] [CrossRef]
- Veenvliet, J.V.; Bolondi, A.; Kretzmer, H.; Haut, L.; Scholze-Wittler, M.; Schifferl, D.; Koch, F.; Guignard, L.; Kumar, A.S.; Pustet, M.; et al. Mouse embryonic stem cells self-organize into trunk-like structures with neural tube and somites. Science 2020, 370, eaba4937. [Google Scholar] [CrossRef]
- Zheng, Y.; Shao, Y.; Fu, J. A microfluidics-based stem cell model of early post-implantation human development. Nat. Protoc. 2021, 16, 309–326. [Google Scholar] [CrossRef]
- Beccari, L.; Moris, N.; Girgin, M.; Turner, D.A.; Baillie-Johnson, P.; Cossy, A.C.; Lutolf, M.P.; Duboule, D.; Martinez Arias, A. Multi-axial self-organization properties of mouse embryonic stem cells into gastruloids. Nature 2018, 562, 272–276. [Google Scholar] [CrossRef]
- Vrij, E.J.; Scholte op Reimer, Y.S.; Aldeguer, J.F.; Guerreiro, I.M.; Kind, J.; Koo, B.-K.; Van Blitterswijk, C.A.; Rivron, N.C. Chemically-defined induction of a primitive endoderm and epiblast-like niche supports post-implantation progression from blastoids. bioRxiv 2019. [Google Scholar] [CrossRef]
- Yu, L.; Wei, Y.; Duan, J.; Schmitz, D.A.; Sakurai, M.; Wang, L.; Wang, K.C.; Zhao, S.; Hon, G.C.; Wu, J. Blastocyst-like structures generated from human pluripotent stem cells. Nature 2021, 591, 620–626. [Google Scholar] [CrossRef] [PubMed]
- Sozen, B.; Jorgensen, V.; Zhu, M.; Cui, T.; Zernicka-Goetz, M. Reconstructing human early embryogenesis in vitro with pluripotent stem cells. bioRxiv 2021. [Google Scholar] [CrossRef]
- Mori, S.; Sakakura, E.; Tsunekawa, Y.; Hagiwara, M.; Suzuki, T.; Eiraku, M. Self-organized formation of developing appendages from murine pluripotent stem cells. Nat. Commun. 2019, 10, 3802. [Google Scholar] [CrossRef]
- Girgin, M.U.; Broguiere, N.; Mattolini, L.; Lutolf, M.P. Gastruloids generated without exogenous Wnt activation develop anterior neural tissues. Stem Cell Rep. 2021, 16, 1143–1155. [Google Scholar] [CrossRef] [PubMed]
- Bertels, S.; Jaggy, M.; Richter, B.; Keppler, S.; Weber, K.; Genthner, E.; Fischer, A.C.; Thiel, M.; Wegener, M.; Greiner, A.M.; et al. Geometrically defined environments direct cell division rate and subcellular YAP localization in single mouse embryonic stem cells. Sci. Rep. 2021, 11, 9269. [Google Scholar] [CrossRef]
- Shahbazi, M.N.; Zernicka-Goetz, M. Deconstructing and reconstructing the mouse and human early embryo. Nat. Cell Biol. 2018, 20, 878–887. [Google Scholar] [CrossRef]
- Yang, J.; Fu, H.; Tam, C.; Liu, P. Expanded potential: The key to synthetic embryo? Curr. Opin. Genet. Dev. 2020, 64, 72–77. [Google Scholar] [CrossRef]
- Posfai, E.; Schell, J.P.; Janiszewski, A.; Rovic, I.; Murray, A.; Bradshaw, B.; Pardon, T.; El Bakkali, M.; Talon, I.; De Geest, N.; et al. Defining totipotency using criteria of increasing stringency. bioRxiv 2020. [Google Scholar] [CrossRef]
- Shahbazi, M.N. Mechanisms of human embryo development: From cell fate to tissue shape and back. Development 2020, 147. [Google Scholar] [CrossRef]
- Piotrowska, M. Research guidelines for embryoids. J. Med. Ethics 2021. [Google Scholar] [CrossRef] [PubMed]
- Denker, H.-W. Self-Organization of Stem Cell Colonies and of Early Mammalian Embryos: Recent Experiments Shed New Light on the Role of Autonomy vs. External Instructions in Basic Body Plan Development. Cells 2016, 5, 39. [Google Scholar] [CrossRef] [PubMed]
- Denker, H.-W. Induced pluripotent stem cells: How to deal with the developmental potential. Reprod. Biomed. Online 2009, 19 (Suppl. S1), 34–37. [Google Scholar] [CrossRef]
- Aguilera-Castrejon, A.; Oldak, B.; Shani, T.; Ghanem, N.; Itzkovich, C.; Slomovich, S.; Tarazi, S.; Bayerl, J.; Chugaeva, V.; Ayyash, M.; et al. Ex utero mouse embryogenesis from pre-gastrulation to late organogenesis. Nature 2021, 593, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Gardner, R.L. The early blastocyst is bilaterally symmetrical and its axis of symmetry is aligned with the animal-vegetal axis of the zygote in the mouse. Development 1997, 124, 289–301. [Google Scholar] [CrossRef]
- Gardner, R.L. Polarity in early mammalian development. Curr. Opin. Genet. Dev. 1999, 9, 417–421. [Google Scholar] [CrossRef]
- Takaoka, K.; Yamamoto, M.; Hamada, H. Origin and role of distal visceral endoderm, a group of cells that determines anterior-posterior polarity of the mouse embryo. Nat. Cell Biol. 2011, 13, 743–752. [Google Scholar] [CrossRef]
- Takaoka, K.; Hamada, H. Cell fate decisions and axis determination in the early mouse embryo. Development 2012, 139, 3–14. [Google Scholar] [CrossRef]
- Hsu, Y.C. In vitro development of individually cultured whole mouse embryos from blastocyst to early somite stage. Dev. Biol. 1979, 68, 453–461. [Google Scholar] [CrossRef]
- Chen, L.T.; Hsu, Y.C. Development of mouse embryos in vitro: Preimplantation to the limb bud stage. Science 1982, 218, 66–68. [Google Scholar] [CrossRef]
- Denker, H.-W. Formation of the blastocyst: Determination of trophoblast and embryonic knot. In Developmental Biology and Pathology; Gropp, A., Benirschke, K., Eds.; Springer: Berlin/Heidelberg Germany, 1976; Volume 62, pp. 59–79. [Google Scholar]
- Boiani, M.; Casser, E.; Fuellen, G.; Christians, E.S. Totipotency continuity from zygote to early blastomeres—A model under revision. Reproduction 2019, 158, R49–R65. [Google Scholar] [CrossRef]
- Casser, E.; Wdowik, S.; Israel, S.; Witten, A.; Schlatt, S.; Nordhoff, V.; Boiani, M. Differences in blastomere totipotency in 2-cell mouse embryos are a maternal trait mediated by asymmetric mRNA distribution. Mol. Hum. Reprod. 2019, 25, 729–744. [Google Scholar] [CrossRef] [PubMed]
- Denker, H.-W. Recent embryo twinning data prompt reconsideration of theories on a crucial role of segregation of oocyte cytoplasmic constituents in mammals. Mol. Hum. Reprod. 2020, 26, 193–198. [Google Scholar] [CrossRef]
- Dalcq, A.-M. Nouvelles donées structurales et cytochimiques sur l’oeuf des mammifères. Rev. Gén. Sci. 1954, 61, 19–41. [Google Scholar]
- Seidel, F. Die Entwicklungspotenzen einer isolierten Blastomere des Zweizellenstadiums im Säugetierei. Naturwissenschaften 1952, 39, 355–356. [Google Scholar] [CrossRef]
- Seidel, F. Die Entwicklungsfähigkeiten isolierter Furchungszellen aus dem Ei des Kaninchens Oryctolagus cuniculus. Wilhelm Roux Arch. Dev. Mech. Org. 1960, 152, 43–130. [Google Scholar] [CrossRef]
- Kime, C.; Kiyonari, H.; Ohtsuka, S.; Kohbayashi, E.; Asahi, M.; Yamanaka, S.; Takahashi, M.; Tomoda, K. Induced 2C Expression and Implantation-Competent Blastocyst-like Cysts from Primed Pluripotent Stem Cells. Stem Cell Rep. 2019, 13, 485–498. [Google Scholar] [CrossRef]
- Thomson, J.A.; Kalishman, J.; Golos, T.G.; Durning, M.; Harris, C.P.; Hearn, J.P. Pluripotent Cell Lines Derived from Common Marmoset (Callithrix jacchus) Blastocysts. Biol. Reprod. 1996, 55, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Behr, R.; Heneweer, C.; Viebahn, C.; Denker, H.W.; Thie, M. Epithelial-mesenchymal transition in colonies of rhesus monkey embryonic stem cells: A model for processes involved in gastrulation. Stem Cells 2005, 23, 805–816. [Google Scholar] [CrossRef]
- Warmflash, A.; Sorre, B.; Etoc, F.; Siggia, E.D.; Brivanlou, A.H. A method to recapitulate early embryonic spatial patterning in human embryonic stem cells. Nat. Methods 2014, 11, 847–854. [Google Scholar] [CrossRef]
- Kime, C.; Kiyonari, H.; Ohtsuka, S.; Kohbayashi, E.; Asahi, M.; Yamanaka, S.; Takahashi, M.; Tomoda, K. Implantation-Competent Blastocyst-Like Structures from Mouse Pluripotent Stem Cells. bioRxiv 2018. [Google Scholar] [CrossRef]
- Kime, C.; Sakaki-Yumoto, M.; Goodrich, L.; Hayashi, Y.; Sami, S.; Derynck, R.; Asahi, M.; Panning, B.; Yamanaka, S.; Tomoda, K. Autotaxin-mediated lipid signaling intersects with LIF and BMP signaling to promote the naive pluripotency transcription factor program. Proc. Natl. Acad. Sci. USA 2016, 113, 12478–12483. [Google Scholar] [CrossRef] [PubMed]
- Pour, M.; Nachman, I. Building Blastocysts from Stem Cells. Stem Cell Rep. 2019, 13, 437–439. [Google Scholar] [CrossRef] [PubMed]
- Ishiuchi, T.; Enriquez-Gasca, R.; Mizutani, E.; Boskovic, A.; Ziegler-Birling, C.; Rodriguez-Terrones, D.; Wakayama, T.; Vaquerizas, J.M.; Torres-Padilla, M.E. Early embryonic-like cells are induced by downregulating replication-dependent chromatin assembly. Nat. Struct. Mol. Biol. 2015, 22, 662–671. [Google Scholar] [CrossRef]
- Kolodziejczyk, A.A.; Kim, J.K.; Tsang, J.C.; Ilicic, T.; Henriksson, J.; Natarajan, K.N.; Tuck, A.C.; Gao, X.; Buhler, M.; Liu, P.; et al. Single Cell RNA-Sequencing of Pluripotent States Unlocks Modular Transcriptional Variation. Cell Stem Cell 2015, 17, 471–485. [Google Scholar] [CrossRef] [PubMed]
- Hendrickson, P.G.; Dorais, J.A.; Grow, E.J.; Whiddon, J.L.; Lim, J.W.; Wike, C.L.; Weaver, B.D.; Pflueger, C.; Emery, B.R.; Wilcox, A.L.; et al. Conserved roles of mouse DUX and human DUX4 in activating cleavage-stage genes and MERVL/HERVL retrotransposons. Nat. Genet. 2017, 49, 925–934. [Google Scholar] [CrossRef]
- Rodriguez-Terrones, D.; Gaume, X.; Ishiuchi, T.; Weiss, A.; Kopp, A.; Kruse, K.; Penning, A.; Vaquerizas, J.M.; Brino, L.; Torres-Padilla, M.E. A molecular roadmap for the emergence of early-embryonic-like cells in culture. Nat. Genet. 2017, 50, 106–119. [Google Scholar] [CrossRef]
- Wu, K.; Liu, H.; Wang, Y.; He, J.; Xu, S.; Chen, Y.; Kuang, J.; Liu, J.; Guo, L.; Li, D.; et al. SETDB1-Mediated Cell Fate Transition between 2C-Like and Pluripotent States. Cell Rep. 2020, 30, 25.e6–36.e6. [Google Scholar] [CrossRef]
- Rodriguez-Terrones, D.; Hartleben, G.; Gaume, X.; Eid, A.; Guthmann, M.; Iturbide, A.; Torres-Padilla, M.E. A distinct metabolic state arises during the emergence of 2-cell-like cells. EMBO Rep. 2019, 21, e48354. [Google Scholar] [CrossRef]
- Hu, Z.; Tan, D.E.K.; Chia, G.; Tan, H.; Leong, H.F.; Chen, B.J.; Lau, M.S.; Tan, K.Y.S.; Bi, X.; Yang, D.; et al. Maternal factor NELFA drives a 2C-like state in mouse embryonic stem cells. Nat. Cell Biol. 2020, 22, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Baker, C.L.; Pera, M.F. Capturing Totipotent Stem Cells. Cell Stem Cell 2018, 22, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Genet, M.; Torres-Padilla, M.E. The molecular and cellular features of 2-cell-like cells: A reference guide. Development 2020, 147, dev189688. [Google Scholar] [CrossRef]
- Tomoda, K.; Hu, H.; Sahara, Y.; Sanyal, H.; Takasato, M.; Kime, C. Reprogramming Epiblast Stem Cells into Pre-Implantation Blastocyst Cell-like Cells. bioRxiv 2020. [Google Scholar] [CrossRef]
- Yang, J.; Ryan, D.J.; Wang, W.; Tsang, J.C.; Lan, G.; Masaki, H.; Gao, X.; Antunes, L.; Yu, Y.; Zhu, Z.; et al. Establishment of mouse expanded potential stem cells. Nature 2017, 550, 393–397. [Google Scholar] [CrossRef]
- Macfarlan, T.S.; Gifford, W.D.; Driscoll, S.; Lettieri, K.; Rowe, H.M.; Bonanomi, D.; Firth, A.; Singer, O.; Trono, D.; Pfaff, S.L. Embryonic stem cell potency fluctuates with endogenous retrovirus activity. Nature 2012, 487, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Hübner, K.; Fuhrmann, G.; Christenson, L.K.; Kehler, J.; Reinbold, R.; De La Fuente, R.; Wood, J.; Strauss, J.F., III; Boiani, M.; Schöler, H.R. Derivation of oocytes from mouse embryonic stem cells. Science 2003, 300, 1251–1256. [Google Scholar] [CrossRef]
- Denker, H.-W. Stem cell terminology and ‘synthetic’ embryos: A new debate on totipotency, omnipotency, and pluripotency and how it relates to recent experimental data. Cells Tissues Organs 2014, 199, 221–227. [Google Scholar] [CrossRef]
- Maranca-Hüwel, B.; Denker, H.-W. Epithelial-mesenchymal transition in rhesus monkey embryonic stem cell colonies: The role of culturing conditions. In Vitro Cell Dev. Biol. Anim. 2010, 46, 516–528. [Google Scholar] [CrossRef]
- Shiozawa, S.; Nakajima, M.; Okahara, J.; Kuortaki, Y.; Kisa, F.; Yoshimatsu, S.; Nakamura, M.; Koya, I.; Yoshimura, M.; Sasagawa, Y.; et al. Primed to Naive-Like Conversion of the Common Marmoset Embryonic Stem Cells. Stem Cells Dev. 2020, 29, 761–773. [Google Scholar] [CrossRef] [PubMed]
- Hancock, G.V.; Wamaitha, S.E.; Peretz, L.; Clark, A.T. Mammalian primordial germ cell specification. Development 2021, 148. [Google Scholar] [CrossRef] [PubMed]
- Stauske, M.; Rodriguez Polo, I.; Haas, W.; Knorr, D.Y.; Borchert, T.; Streckfuss-Bomeke, K.; Dressel, R.; Bartels, I.; Tiburcy, M.; Zimmermann, W.H.; et al. Non-Human Primate iPSC Generation, Cultivation, and Cardiac Differentiation under Chemically Defined Conditions. Cells 2020, 9, 1349. [Google Scholar] [CrossRef]
- Gilbert, S.F.; Sarkar, S. Embracing complexity: Organicism for the 21st century. Dev. Dyn. 2000, 219, 1–9. [Google Scholar] [CrossRef]
- Olmsted, Z.T.; Paluh, J.L. Co-development of central and peripheral neurons with trunk mesendoderm in human elongating multi-lineage organized gastruloids. Nat. Commun. 2021, 12, 1–9. [Google Scholar] [CrossRef]
- Xu, P.-F.; Borges, R.M.; Fillatre, J.; de Oliveira-Melo, M.; Cheng, T.; Thisse, B.; Thisse, D. Construction of a mammalian embryo model from stem cells organized by a morphogen signalling centre. Nat. Commun. 2021, 12, 3277. [Google Scholar] [CrossRef] [PubMed]
- Lovell-Badge, R.; Anthony, E.; Barker, R.A.; Bubela, T.; Brivanlou, A.H.; Carpenter, M.; Charo, R.A.; Clark, A.; Clayton, E.; Cong, Y.; et al. ISSCR Guidelines for Stem Cell Research and Clinical Translation: The 2021 update. Stem Cell Rep. 2021. [Google Scholar] [CrossRef]
- Clark, A.T.; Brivanlou, A.; Fu, J.; Kato, K.; Mathews, D.; Niakan, K.K.; Rivron, N.; Saitou, M.; Surani, A.; Tang, F.; et al. Human embryo research, stem cell-derived embryo models and in vitro gametogenesis: Considerations leading to the revised ISSCR guidelines. Stem Cell Rep. 2021. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Denker, H.-W. Autonomy in the Development of Stem Cell-Derived Embryoids: Sprouting Blastocyst-Like Cysts, and Ethical Implications. Cells 2021, 10, 1461. https://doi.org/10.3390/cells10061461
Denker H-W. Autonomy in the Development of Stem Cell-Derived Embryoids: Sprouting Blastocyst-Like Cysts, and Ethical Implications. Cells. 2021; 10(6):1461. https://doi.org/10.3390/cells10061461
Chicago/Turabian StyleDenker, Hans-Werner. 2021. "Autonomy in the Development of Stem Cell-Derived Embryoids: Sprouting Blastocyst-Like Cysts, and Ethical Implications" Cells 10, no. 6: 1461. https://doi.org/10.3390/cells10061461
APA StyleDenker, H.-W. (2021). Autonomy in the Development of Stem Cell-Derived Embryoids: Sprouting Blastocyst-Like Cysts, and Ethical Implications. Cells, 10(6), 1461. https://doi.org/10.3390/cells10061461





