Modeling Retinal Ganglion Cell Dysfunction in Optic Neuropathies
Abstract
1. Introduction
2. To Be or Not to Be, This Is the Question
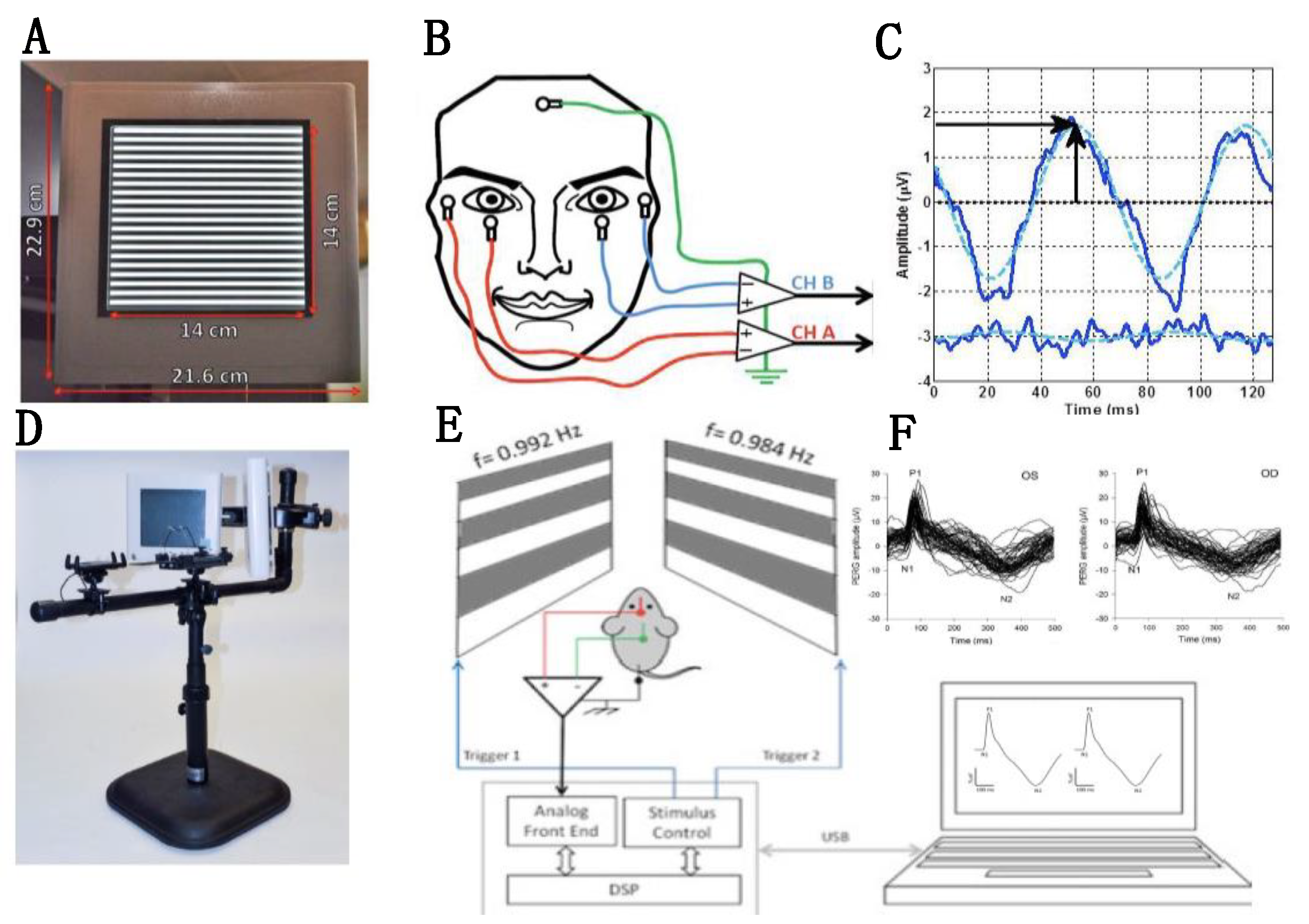
2.1. Non-Invasive Assessment of RGC Function
2.2. Physiological Significance of Altered PERG Signal
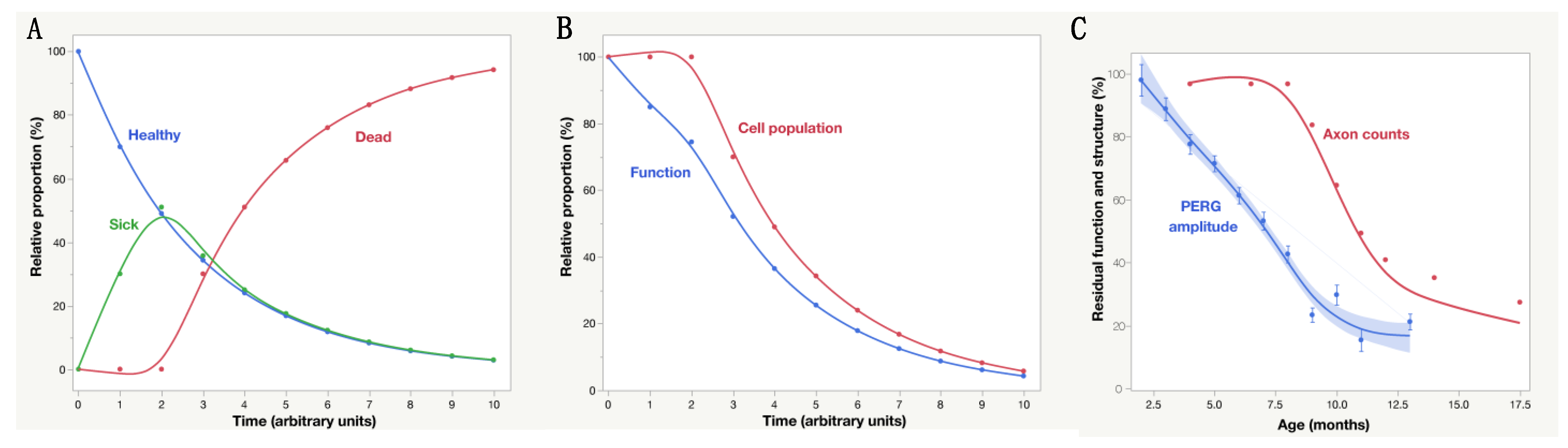
2.3. PERG Dynamics and RGC Functional Properties
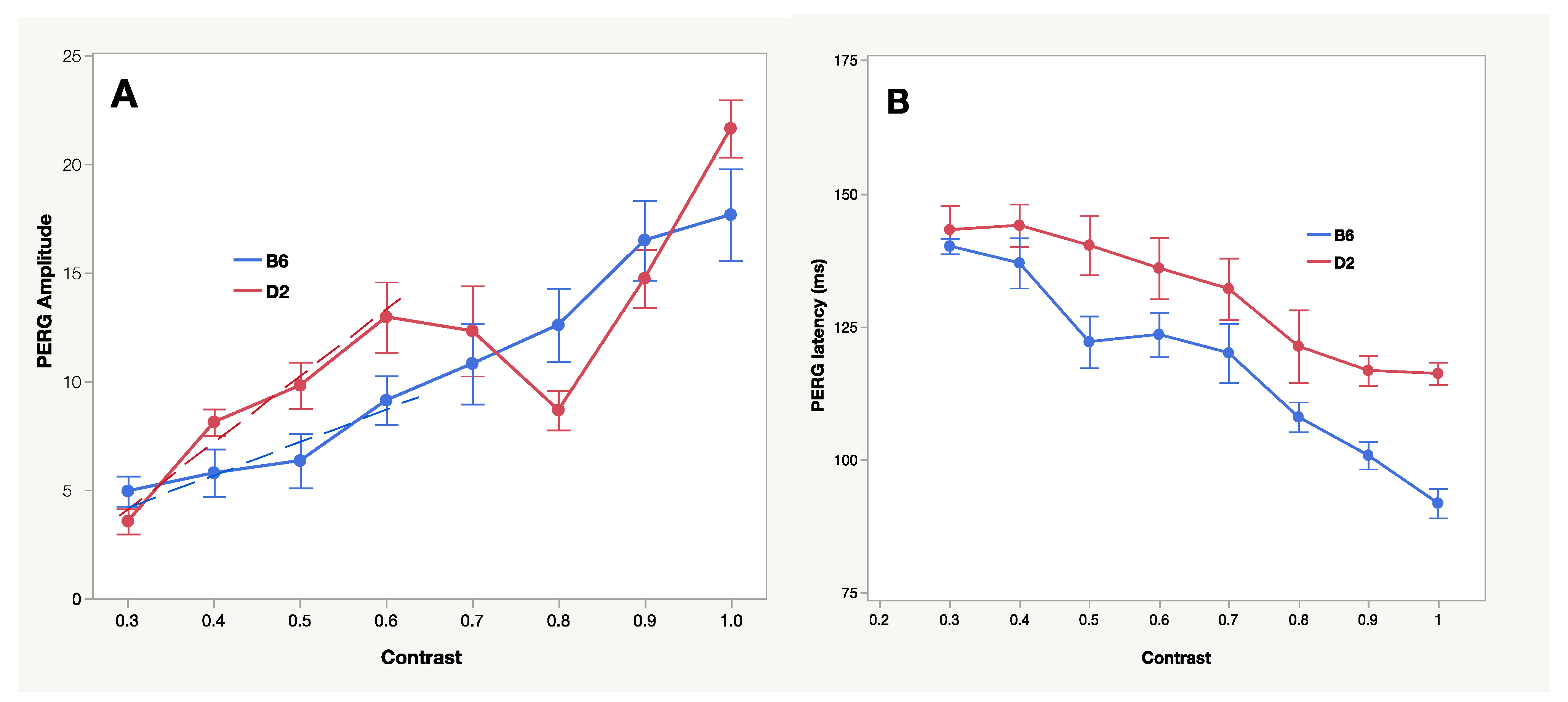
2.4. PERG Dynamics—Physiological Approaches
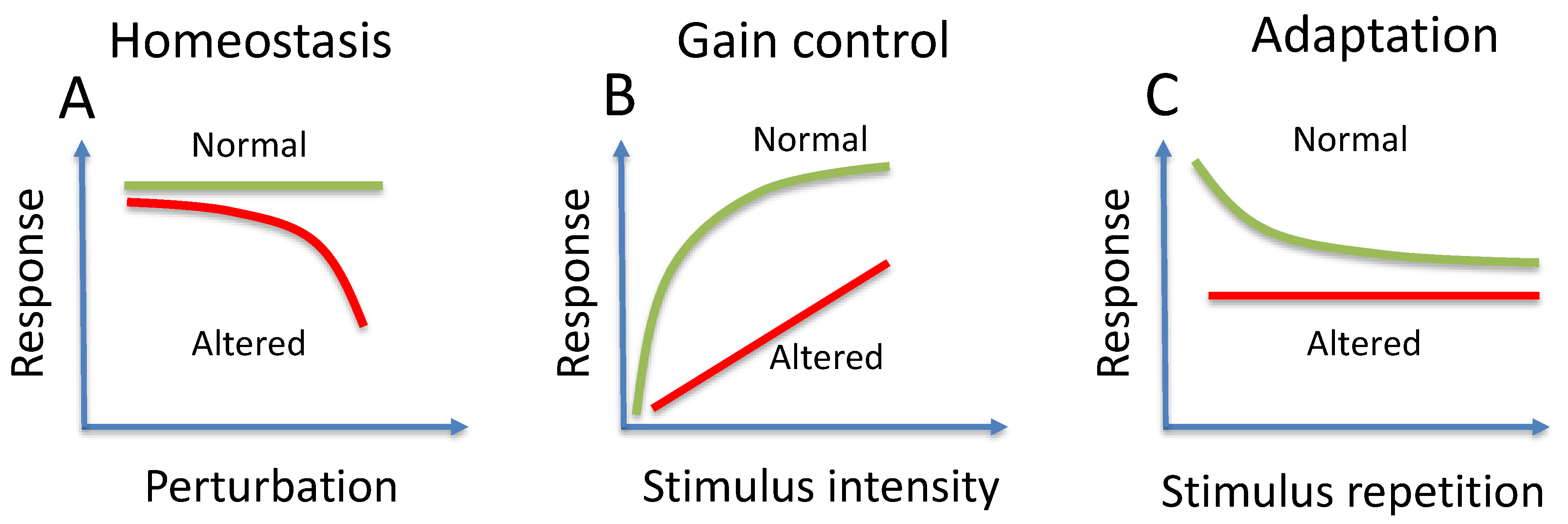
3. Homeostasis
4. Gain Control
5. Adaptation
6. PERG Dynamics—Interventional Approaches
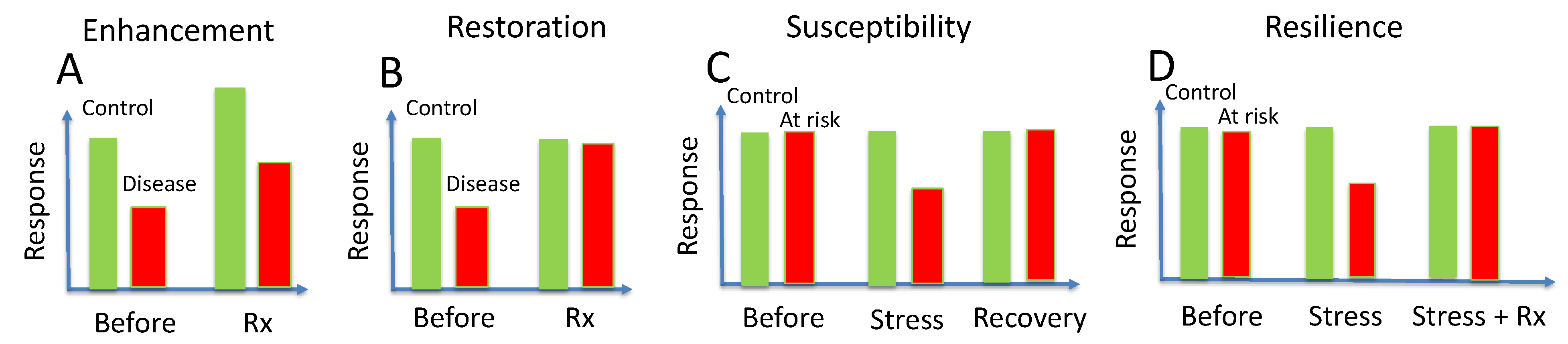
7. Enhancement
8. Restoration
9. Susceptibility
10. Acquired Resilience
11. Functional Phenotyping
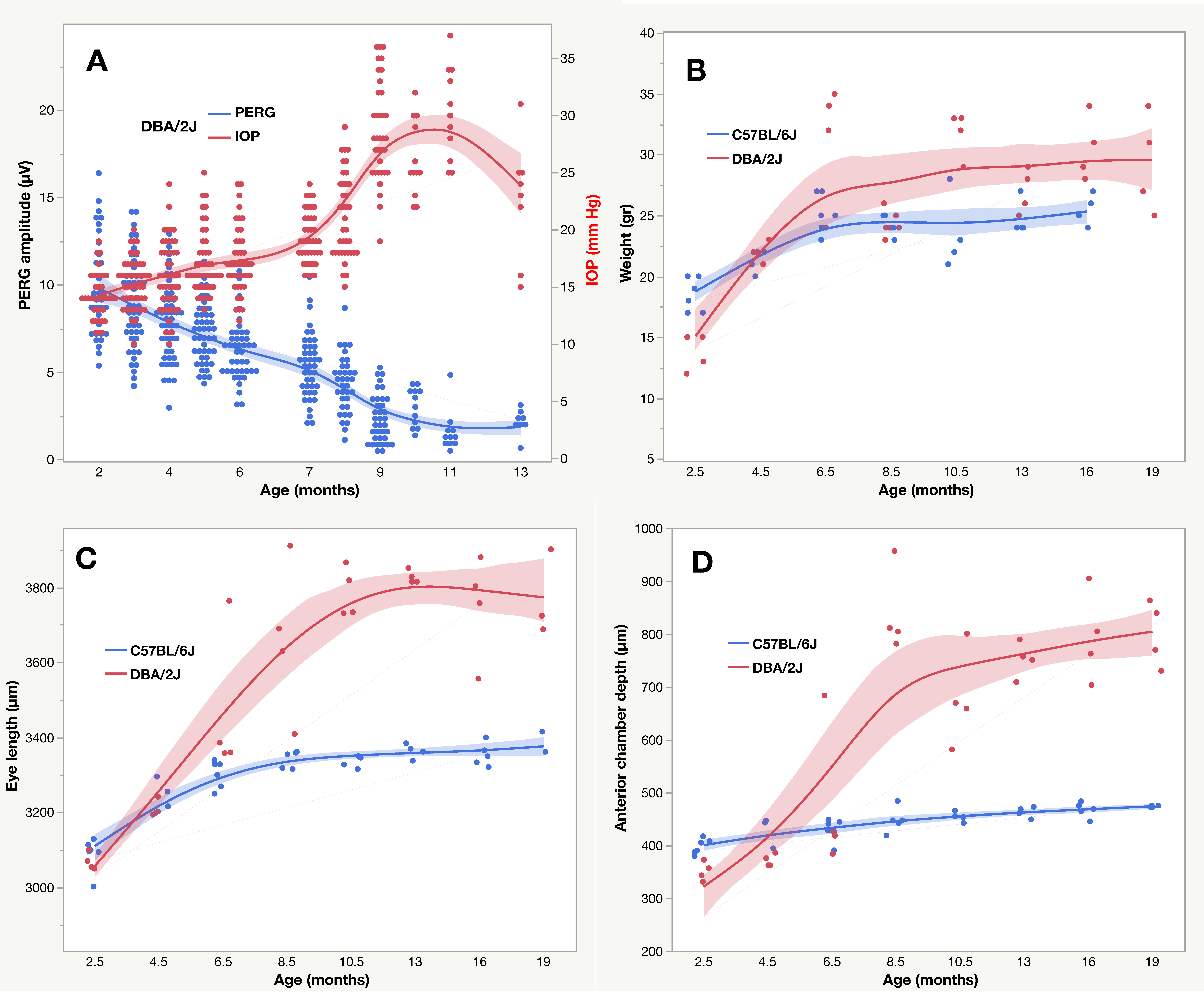
12. Age-Related Factors
13. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Financial Disclosure
References
- Almasieh, M.; Levin, L.A. Neuroprotection in Glaucoma: Animal Models and Clinical Trials. Annu. Rev. Vis. Sci. 2017, 3, 91–120. [Google Scholar] [CrossRef]
- Wareham, L.K.; Risner, M.L.; Calkins, D.J. Protect, Repair, and Regenerate: Towards Restoring Vision in Glaucoma. Curr. Ophthalmol. Rep. 2020, 8, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Khatib, T.Z.; Martin, K.R. Neuroprotection in Glaucoma: Towards Clinical Trials and Precision Medicine. Curr. Eye Res. 2020, 45, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Tsai, J.C. Innovative IOP-Independent Neuroprotection and Neuroregeneration Strategies in the Pipeline for Glaucoma. J. Ophthalmol. 2020, 2020, 1–6. [Google Scholar] [CrossRef]
- Levin, L.A. Neuroprotection in Optic Neuropathy. Asia-Pacific J. Ophthalmol. 2018, 7, 246–250. [Google Scholar] [CrossRef]
- Guymer, C.; Wood, J.P.; Chidlow, G.; Casson, R.J. Neuroprotection in glaucoma: Recent advances and clinical translation. Clin. Exp. Ophthalmol. 2019, 47, 88–105. [Google Scholar] [CrossRef]
- Pearson, C.; Martin, K. Stem cell approaches to glaucoma: From aqueous outflow modulation to retinal neuroprotection. Prog. Brain Res. 2015, 220, 241–256. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Wang, Y.; Yao, K. Protection of retinal ganglion cells in glaucoma: Current status and future. Exp. Eye Res. 2021, 205, 108506. [Google Scholar] [CrossRef] [PubMed]
- Howell, G.R.; Libby, R.T.; Jakobs, T.; Smith, R.S.; Phalan, F.C.; Barter, J.W.; Barbay, J.M.; Marchant, J.K.; Mahesh, N.; Porciatti, V.; et al. Axons of retinal ganglion cells are insulted in the optic nerve early in DBA/2J glaucoma. J. Cell Biol. 2007, 179, 1523–1537. [Google Scholar] [CrossRef]
- Williams, P.A.; Harder, J.M.; Foxworth, N.E.; Cochran, K.E.; Philip, V.M.; Porciatti, V.; Smithies, O.; John, S.W.M. Vitamin B3modulates mitochondrial vulnerability and prevents glaucoma in aged mice. Science 2017, 355, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Crish, S.D.; Sappington, R.M.; Inman, D.; Horner, P.J.; Calkins, D.J. Distal axonopathy with structural persistence in glaucomatous neurodegeneration. Proc. Natl. Acad. Sci. USA 2010, 107, 5196–5201. [Google Scholar] [CrossRef] [PubMed]
- Buckingham, B.P.; Inman, D.; Lambert, W.; Oglesby, E.; Calkins, D.J.; Steele, M.R.; Vetter, M.L.; Marsh-Armstrong, N.; Horner, P.J. Progressive Ganglion Cell Degeneration Precedes Neuronal Loss in a Mouse Model of Glaucoma. J. Neurosci. 2008, 28, 2735–2744. [Google Scholar] [CrossRef]
- Enriquez-Algeciras, M.; Ding, D.; Chou, T.-H.; Wang, J.; Padgett, K.R.; Porciatti, V.; Bhattacharya, S.K. Evaluation of a Transgenic Mouse Model of Multiple Sclerosis with Noninvasive Methods. Investig. Opthalmology Vis. Sci. 2011, 52, 2405–2411. [Google Scholar] [CrossRef]
- Porciatti, V.; Ventura, L.M. Retinal ganglion cell functional plasticity and optic neuropathy: A comprehensive model. J. Neuro-Ophthalmol. 2012, 32, 354–358. [Google Scholar] [CrossRef]
- Siebert, U.; Alagoz, O.; Bayoumi, A.M.; Jahn, B.; Owens, D.K.; Cohen, D.J.; Kuntz, K.M. State-Transition Modeling: A report of the ISPOR-SMDM Modeling Good Research Practices Task Force-3. Med. Decis. Mak. 2012, 32, 690–700. [Google Scholar] [CrossRef]
- Cordeiro, M.F.; Migdal, C.; Bloom, P.; Fitzke, F.W.; E Moss, S. Imaging apoptosis in the eye. Eye 2011, 25, 545–553. [Google Scholar] [CrossRef] [PubMed]
- Yap, T.E.; Donna, P.; Almonte, M.T.; Cordeiro, M.F. Real-Time Imaging of Retinal Ganglion Cell Apoptosis. Cells 2018, 7, 60. [Google Scholar] [CrossRef] [PubMed]
- Leinonen, H.; Tanila, H. Vision in laboratory rodents—Tools to measure it and implications for behavioral research. Behav. Brain Res. 2018, 352, 172–182. [Google Scholar] [CrossRef] [PubMed]
- Porciatti, V. Electrophysiological assessment of retinal ganglion cell function. Exp. Eye Res. 2015, 141, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Thompson, D.; Drasdo, N. The origins of luminance and pattern responses of the pattern electroretinogram. Int. J. Psychophysiol. 1994, 16, 219–227. [Google Scholar] [CrossRef]
- Hess, R.F.; Baker, C.L., Jr. Human pattern-evoked electroretinogram. J. Neurophysiol. 1984, 51, 939–951. [Google Scholar] [CrossRef]
- Porciatti, V.; Saleh, M.; Nagaraju, M. The Pattern Electroretinogram as a Tool to Monitor Progressive Retinal Ganglion Cell Dysfunction in the DBA/2J Mouse Model of Glaucoma. Investig. Opthalmology Vis. Sci. 2007, 48, 745–751. [Google Scholar] [CrossRef] [PubMed]
- Porciatti, V. The mouse pattern electroretinogram. Doc. Ophthalmol. 2007, 115, 145–153. [Google Scholar] [CrossRef]
- Chou, T.-H.; Bohorquez, J.; Toft-Nielsen, J.; Ozdamar, O.; Porciatti, V. Robust mouse pattern electroretinograms derived simultaneously from each eye using a common snout electrode. Investig. Opthalmology Vis. Sci. 2014, 55, 2469–2475. [Google Scholar] [CrossRef] [PubMed]
- Sehi, M.; Grewal, D.; Goodkin, M.L.; Greenfield, D.S. Reversal of Retinal Ganglion Cell Dysfunction after Surgical Reduction of Intraocular Pressure. Ophthalmology 2010, 117, 2329–2336. [Google Scholar] [CrossRef] [PubMed]
- Ventura, L.M.; Porciatti, V. Restoration of retinal ganglion cell function in early glaucoma after intraocular pressure reduction: A pilot study. Ophthalmology 2005, 112, 20–27. [Google Scholar] [CrossRef]
- Yang, E.; Al-Mugheiry, T.S.; Normando, E.M.; Cordeiro, M.F. Real-Time Imaging of Retinal Cell Apoptosis by Confocal Scanning Laser Ophthalmoscopy and Its Role in Glaucoma. Front. Neurol. 2018, 9, 338. [Google Scholar] [CrossRef] [PubMed]
- Saleh, M.; Nagaraju, M.; Porciatti, V. Longitudinal evaluation of retinal ganglion cell function and IOP in the DBA/2J mouse model of glaucoma. Investig. Opthalmology Vis. Sci. 2007, 48, 4564–4572. [Google Scholar] [CrossRef] [PubMed]
- Banitt, M.R.; Ventura, L.M.; Feuer, W.J.; Savatovsky, E.; Luna, G.; Shif, O.; Bosse, B.; Porciatti, V. Progressive Loss of Retinal Ganglion Cell Function Precedes Structural Loss by Several Years in Glaucoma Suspects. Investig. Opthalmology Vis. Sci. 2013, 54, 2346–2352. [Google Scholar] [CrossRef]
- Nagaraju, M.; Saleh, M.; Porciatti, V. IOP-Dependent Retinal Ganglion Cell Dysfunction in Glaucomatous DBA/2J Mice. Investig. Opthalmology Vis. Sci. 2007, 48, 4573–4579. [Google Scholar] [CrossRef]
- Ventura, L.M.; Golubev, I.; Lee, W.; Nose, I.; Parel, J.-M.; Feuer, W.J.; Porciatti, V. Head-down posture induces PERG alterations in early glaucoma. J. Glaucoma 2013, 22, 255–264. [Google Scholar] [CrossRef]
- Porciatti, V.; Feuer, W.J.; Monsalve, P.; Triolo, G.; Vazquez, L.; McSoley, J.; Ventura, L.M. Head-down Posture in Glaucoma Suspects Induces Changes in IOP, Systemic Pressure, and PERG That Predict Future Loss of Optic Nerve Tissue. J. Glaucoma 2017, 26, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.-H.; Feuer, W.J.; Schwartz, O.; Rojas, M.J.; Roebber, J.K.; Porciatti, V. Integrative properties of retinal ganglion cell electrical responsiveness depend on neurotrophic support and genotype in the mouse. Exp. Eye Res. 2016, 145, 68–74. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Porciatti, V.; Sorokac, N.; Buchser, W. Habituation of retinal ganglion cell activity in response to steady state pattern visual stimuli in normal subjects. Investig. Opthalmology Vis. Sci. 2005, 46, 1296–1302. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.-H.; Toft-Nielsen, J.; Porciatti, V. Adaptation of retinal ganglion cell function during flickering light in the mouse. Sci. Rep. 2019, 9, 18396. [Google Scholar] [CrossRef] [PubMed]
- Monsalve, P.; Ren, S.; Triolo, G.; Vazquez, L.; Henderson, A.D.; Kostic, M.; Gordon, P.; Feuer, W.J.; Porciatti, V. Steady-state PERG adaptation: A conspicuous component of response variability with clinical significance. Doc. Ophthalmol. 2018, 136, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Monsalve, P.; Triolo, G.; Toft-Nielsen, J.; Bohorquez, J.; Henderson, A.D.; Delgado, R.; Miskiel, E.; Ozdamar, O.; Feuer, W.J.; Porciatti, V. Next Generation PERG Method: Expanding the Response Dynamic Range and Capturing Response Adaptation. Transl. Vis. Sci. Technol. 2017, 6, 5. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Chou, T.-H.; Romano, G.L.; Amato, R.; Porciatti, V. Nicotinamide-Rich Diet in DBA/2J Mice Preserves Retinal Ganglion Cell Metabolic Function as Assessed by PERG Adaptation to Flicker. Nutrients. 2020, 12, 1910. [Google Scholar] [CrossRef]
- Gandolfi, S.; Marchini, G.; Caporossi, A.; Scuderi, G.; Tomasso, L.; Brunoro, A. Cytidine 5′-Diphosphocholine (Citicoline): Evidence for a Neuroprotective Role in Glaucoma. Nutrients. 2020, 12, 793. [Google Scholar] [CrossRef]
- Parisi, V.; Barbano, L.; Di Renzo, A.; Coppola, G.; Ziccardi, L. Neuroenhancement and neuroprotection by oral solution citicoline in non-arteritic ischemic optic neuropathy as a model of neurodegeneration: A randomized pilot study. PLoS ONE 2019, 14, e0220435. [Google Scholar] [CrossRef]
- Gottlob, I.; Weghaupt, H.; Vass, C.; Auff, E. Effect of levodopa on the human pattern electroretinogram and pattern visual evoked potentials. Graefe’s Arch. Clin. Exp. Ophthalmol. 1989, 227, 421–427. [Google Scholar] [CrossRef]
- Libby, R.T.; Anderson, M.; Pang, I.-H.; Robinson, Z.H.; Savinova, O.V.; Cosma, I.M.; Snow, A.; Wilson, L.A.; Smith, R.S.; Clark, A.F.; et al. Inherited glaucoma in DBA/2J mice: Pertinent disease features for studying the neurodegeneration. Vis. Neurosci. 2005, 22, 637–648. [Google Scholar] [CrossRef] [PubMed]
- Libby, R.T.; Gould, D.B.; Anderson, M.G.; John, S.W. Complex genetics of glaucoma susceptibility. Annu. Rev. Genom. Hum. Genet. 2005, 6, 15–44. [Google Scholar] [CrossRef]
- Carelli, V.; Achilli, A.; Valentino, M.L.; Rengo, C.; Semino, O.; Pala, M.; Olivieri, A.; Mattiazzi, M.; Pallotti, F.; Carrara, F.; et al. Haplogroup effects and recombination of mitochondrial DNA: Novel clues from the analysis of Leber hereditary optic neuropathy pedigrees. Am. J. Hum. Genet. 2006, 78, 564–574. [Google Scholar] [CrossRef] [PubMed]
- Hudson, G.; Yu-Wai-Man, P.; Griffiths, P.G.; Caporali, L.; Salomao, S.S.; Berezovsky, A.; Carelli, V.; Zeviani, M.; Chinnery, P.F. Variation in OPA1 does not explain the incomplete penetrance of Leber hereditary optic neuropathy. Mol. Vis. 2010, 16, 2760–2764. [Google Scholar]
- Crowston, J.G.; Kong, Y.X.G.; Trounce, I.A.; Dang, T.M.; Fahy, E.T.; Bui, B.; Morrison, J.C.; Chrysostomou, V. An acute intraocular pressure challenge to assess retinal ganglion cell injury and recovery in the mouse. Exp. Eye Res. 2015, 141, 3–8. [Google Scholar] [CrossRef]
- Chou, T.-H.; Park, K.K.; Luo, X.; Porciatti, V. Retrograde signaling in the optic nerve is necessary for electrical responsiveness of retinal ganglion cells. Investig. Opthalmology Vis. Sci. 2013, 54, 1236–1243. [Google Scholar] [CrossRef]
- Chou, T.-H.; Musada, G.R.; Romano, G.L.; Bolton, E.; Porciatti, V. Anesthetic Preconditioning as Endogenous Neuroprotection in Glaucoma. Int. J. Mol. Sci. 2018, 19, 237. [Google Scholar] [CrossRef] [PubMed]
- Stone, J.; Mitrofanis, J.; Johnstone, D.M.; Falsini, B.; Bisti, S.; Adam, P.; Nuevo, A.B.; George-Weinstein, M.; Mason, R.; Eells, J. Acquired Resilience: An Evolved System of Tissue Protection in Mammals. Dose-Response 2018, 16, 1559325818803428. [Google Scholar] [CrossRef] [PubMed]
- Calkins, D.J. Adaptive responses to neurodegenerative stress in glaucoma. Prog. Retin. Eye Res. 2021, 2021, 100953. [Google Scholar] [CrossRef]
- Gameiro, G.; Monsalve, P.; Golubev, I.; Ventura, L.; Porciatti, V. Neurovascular Changes Associated with the Water Drinking Test. J. Glaucoma 2018, 27, 429–432. [Google Scholar] [CrossRef]
- Anderson, M.G.; Libby, R.T.; Mao, M.; Cosma, I.M.; A Wilson, L.; Smith, R.S.; John, S.W.M. Genetic context determines susceptibility to intraocular pressure elevation in a mouse pigmentary glaucoma. BMC Biol. 2006, 4, 20. [Google Scholar] [CrossRef]
- Geisert, E.E.; Williams, R.W. Using BXD mouse strains in vision research: A systems genetics approach. Mol. Vis. 2020, 26, 173–187. [Google Scholar]
- Porciatti, V.; Chou, T.H.; Feuer, W.J. C57BL/6J, DBA/2J, and DBA/2J.Gpnmb mice have different visual signal processing in the inner retina. Mol. Vis. 2010, 16, 2939–2947. [Google Scholar] [PubMed]
- Williams, R.W.; Strom, R.C.; Rice, D.S.; Goldowitz, D. Genetic and environmental control of variation in retinal ganglion cell number in mice. J. Neurosci. 1996, 16, 7193–7205. [Google Scholar] [CrossRef]
- Schuettauf, F.; Quinto, K.; Naskar, R.; Zurakowski, D. Effects of anti-glaucoma medications on gangion cell survival: The DBA/2J mouse model. Vis. Res. 2002, 42, 2333–2337. [Google Scholar] [CrossRef]
- Harder, J.M.; Guymer, C.; Wood, J.P.M.; Daskalaki, E.; Chidlow, G.; Zhang, C.; Balasubramanian, R.; Cardozo, B.H.; Foxworth, N.E.; Deering, K.E.; et al. Disturbed glucose and pyruvate metabolism in glaucoma with neuroprotection by pyruvate or rapamycin. Proc. Natl. Acad. Sci. USA 2020, 117, 33619–33627. [Google Scholar] [CrossRef] [PubMed]
- Chou, T.-H.; Kocaoglu, O.P.; Borja, D.; Ruggeri, M.; Uhlhorn, S.R.; Manns, F.; Porciatti, V. Postnatal Elongation of Eye Size in DBA/2J Mice Compared with C57BL/6J Mice: In Vivo Analysis with Whole-Eye OCT. Investig. Opthalmology Vis. Sci. 2011, 52, 3604–3612. [Google Scholar] [CrossRef] [PubMed]
- Fiedorowicz, M.; Wełniak-Kamińska, M.; Świątkiewicz, M.; Orzeł, J.; Chorągiewicz, T.; Toro, M.D.; Rejdak, R.; Bogorodzki, P.; Grieb, P. Changes of Ocular Dimensions as a Marker of Disease Progression in a Murine Model of Pigmentary Glaucoma. Front. Pharmacol. 2020, 11. [Google Scholar] [CrossRef]
- Cone, F.E.; Gelman, S.E.; Son, J.L.; Pease, M.; Quigley, H.A. Differential susceptibility to experimental glaucoma among 3 mouse strains using bead and viscoelastic injection. Exp. Eye Res. 2010, 91, 415–424. [Google Scholar] [CrossRef] [PubMed]
| Hypothetical Transitional Stages of RGCs in Degenerative Optic Neuropathies | ||||||
| 0 | 1 | 2 | 3 | 4 | 5 | 6 |
| Healthy | At Risk | Sick | Very sick | Dying | Dead | Effaced |
| Non-Invasive Electrophysiology | ||||||
| Normal | ±Abnormal | Abnormal, Reversible | Abnormal, Reversible | Floor, Irreversible | Floor | Floor |
| Non-Invasive Imaging | ||||||
| Normal | Normal | Normal | Normal | Abnormal | Abnormal | Abnormal |
| Neuronal Processes Altering PERG Dynamics | |
|---|---|
| Homeostasis | Stable function despite perturbance |
| Gain control | Reduction of sensitivity for high intensity stimuli |
| Adaptation | Reduction of response for repeated stimuli |
| Susceptibility | Temporary loss of function upon stress |
| Resilience | Reduced susceptibility to stress |
| Enhancement | Nonspecific improvement of function |
| Restoration | Recovery of lost function |
| Protection | Prevention of future loss of function |
| Rescue | Salvage of residual function |
| Plasticity | Reconfigured function |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Porciatti, V.; Chou, T.-H. Modeling Retinal Ganglion Cell Dysfunction in Optic Neuropathies. Cells 2021, 10, 1398. https://doi.org/10.3390/cells10061398
Porciatti V, Chou T-H. Modeling Retinal Ganglion Cell Dysfunction in Optic Neuropathies. Cells. 2021; 10(6):1398. https://doi.org/10.3390/cells10061398
Chicago/Turabian StylePorciatti, Vittorio, and Tsung-Han Chou. 2021. "Modeling Retinal Ganglion Cell Dysfunction in Optic Neuropathies" Cells 10, no. 6: 1398. https://doi.org/10.3390/cells10061398
APA StylePorciatti, V., & Chou, T.-H. (2021). Modeling Retinal Ganglion Cell Dysfunction in Optic Neuropathies. Cells, 10(6), 1398. https://doi.org/10.3390/cells10061398






