Expression of Immuno-Oncologic Biomarkers Is Enriched in Colorectal Cancers and Other Solid Tumors Harboring the A59T Variant of KRAS
Abstract
1. Introduction
2. Methods
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Neumann, J.; Zeindl-Eberhart, E.; Kirchner, T.; Jung, A. Frequency and type of KRAS mutations in routine diagnostic analysis of metastatic colorectal cancer. Pathol. Res. Pract. 2009, 205, 858–862. [Google Scholar] [CrossRef]
- Lou, E.; D’Souza, D.; Nelson, A.C. Therapeutic Response of Metastatic Colorectal Cancer Harboring aKRASMissense Mutation After Combination Chemotherapy With the EGFR Inhibitor Panitumumab. J. Natl. Compr. Cancer Netw. 2017, 15, 427–432. [Google Scholar] [CrossRef][Green Version]
- Henzler, C.; Schomaker, M.; Yang, R.; Lambert, A.P.; LaRue, R.; Kincaid, R.; Beckman, K.; Kemmer, T.; Wilson, J.; Yohe, S.; et al. Optimization of a microfluidics-based next generation sequencing assay for clinical oncology diagnostics. Ann. Transl. Med. 2018, 6, 162. [Google Scholar] [CrossRef]
- Loree, J.M.; Kopetz, S. Why a One Size Fits All Approach to RAS Might Not Fit Colorectal Cancer. J. Natl. Compr. Cancer Netw. 2017, 15, 545–547. [Google Scholar] [CrossRef][Green Version]
- Stadler, Z.K.; Battaglin, F.; Middha, S.; Hechtman, J.F.; Tran, C.; Cercek, A.; Yaeger, R.; Segal, N.H.; Varghese, A.M.; Reidy-Lagunes, D.L.; et al. Reliable Detection of Mismatch Repair Deficiency in Colorectal Cancers Using Mutational Load in Next-Generation Sequencing Panels. J. Clin. Oncol. 2016, 34, 2141–2147. [Google Scholar] [CrossRef]
- VanderWalde, A.; Spetzler, D.; Xiao, N.; Gatalica, Z.; Marshall, J. Microsatellite instability status determined by next-generation sequencing and compared with PD-L1 and tumor mutational burden in 11,348 patients. Cancer Med. 2018, 7, 746–756. [Google Scholar] [CrossRef]
- Costigan, D.C.; Dong, F. The extended spectrum of RAS-MAPK pathway mutations in colorectal cancer. Genes Chromosom. Cancer 2020, 59, 152–159. [Google Scholar] [CrossRef]
- Hamarsheh, S.; Groß, O.; Brummer, T.; Zeiser, R. Immune modulatory effects of oncogenic KRAS in cancer. Nat. Commun. 2020, 11, 1–11. [Google Scholar] [CrossRef]
- Tokunaga, R.; Xiu, J.; Goldberg, R.M.; Philip, P.A.; Seeber, A.; Battaglin, F.; Arai, H.; Lo, J.H.; Naseem, M.; Puccini, A.; et al. The impact of ARID1A mutation on molecular characteristics in colorectal cancer. Eur. J. Cancer 2020, 140, 119–129. [Google Scholar] [CrossRef]
- Malapelle, U.; Passiglia, F.; Cremolini, C.; Reale, M.L.; Pepe, F.; Pisapia, P.; Avallone, A.; Cortinovis, D.; De Stefano, A.; Fassan, M.; et al. RAS as a positive predictive biomarker: Focus on lung and colorectal cancer patients. Eur. J. Cancer 2021, 146, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Alcantara, K.M.M.; Malapit, J.R.P.; Yu, R.T.D.; Garrido, J.A.M.G.; Rigor, J.P.T.; Angeles, A.K.J.; La Paz, E.M.C.-D.; Garcia, R.L. Non-Redundant and Overlapping Oncogenic Readouts of Non-Canonical and Novel Colorectal Cancer KRAS and NRAS Mutants. Cells 2019, 8, 1557. [Google Scholar] [CrossRef]
- Haigis, K.M. KRAS Alleles: The Devil Is in the Detail. Trends Cancer 2017, 3, 686–697. [Google Scholar] [CrossRef]
- Iwata, S.; Masuhara, K.; Umeki, N.; Sako, Y.; Maruta, S. Interaction of a novel fluorescent GTP analogue with the small G-protein K-Ras. J. Biochem. 2015, 159, 41–48. [Google Scholar] [CrossRef]
- John, J.; Frech, M.; Wittinghofer, A. Biochemical properties of Ha-ras encoded p21 mutants and mechanism of the autophosphorylation reaction. J. Biol. Chem. 1988, 263, 11792–11799. [Google Scholar] [CrossRef]
- Lacal, J.C.; Srivastava, S.K.; Anderson, P.S.; Aaronson, S.A. Ras p21 proteins with high or low GTPase activity can efficiently transform NIH3T3 cells. Cell 1986, 44, 609–617. [Google Scholar] [CrossRef]
- Lacal, J.C.; Aaronson, S.A. Activation of ras p21 transforming properties associated with an increase in the release rate of bound guanine nucleotide. Mol. Cell. Biol. 1986, 6, 4214–4220. [Google Scholar] [CrossRef]
- Muñoz-Maldonado, C.; Zimmer, Y.; Medová, M. A Comparative Analysis of Individual RAS Mutations in Cancer Biology. Front. Oncol. 2019, 9, 1088. [Google Scholar] [CrossRef]
- Douillard, J.-Y.; Oliner, K.S.; Siena, S.; Tabernero, J.; Burkes, R.; Barugel, M.; Humblet, Y.; Bodoky, G.; Cunningham, D.; Jassem, J.; et al. Panitumumab–FOLFOX4 Treatment and RAS Mutations in Colorectal Cancer. N. Engl. J. Med. 2013, 369, 1023–1034. [Google Scholar] [CrossRef]
- Sepulveda, A.R.; Hamilton, S.R.; Allegra, C.J.; Grody, W.; Cushman-Vokoun, A.M.; Funkhouser, W.K.; Kopetz, S.E.; Lieu, C.; Lindor, N.M.; Minsky, B.D.; et al. Molecular Biomarkers for the Evaluation of Colorectal Cancer: Guideline From the American Society for Clinical Pathology, College of American Pathologists, Association for Molecular Pathology, and the American Society of Clinical Oncology. J. Clin. Oncol. 2017, 35, 1453–1486. [Google Scholar] [CrossRef]
- Sorich, M.J.; Wiese, M.D.; Rowland, A.; Kichenadasse, G.; McKinnon, R.A.; Karapetis, C.S. Extended RAS mutations and anti-EGFR monoclonal antibody survival benefit in metastatic colorectal cancer: A meta-analysis of randomized, controlled trials. Ann. Oncol. 2015, 26, 13–21. [Google Scholar] [CrossRef]
- Van Cutsem, E.; Lenz, H.-J.; Köhne, C.-H.; Heinemann, V.; Tejpar, S.; Melezínek, I.; Beier, F.; Stroh, C.; Rougier, P.; Van Krieken, J.H.; et al. Fluorouracil, Leucovorin, and Irinotecan Plus Cetuximab Treatment and RAS Mutations in Colorectal Cancer. J. Clin. Oncol. 2015, 33, 692–700. [Google Scholar] [CrossRef]
- Loree, J.M.; Dowers, A.; Tu, D.; Jonker, D.J.; Edelstein, D.L.; Quinn, H.; Holtrup, F.; Price, T.; Zalcberg, J.R.; Moore, M.J.; et al. Expanded Low Allele Frequency RAS and BRAF V600E Testing in Metastatic Colorectal Cancer as Predictive Biomarkers for Cetuximab in the Randomized CO.17 Trial. Clin. Cancer Res. 2021, 27, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Segelov, E.; Thavaneswaran, S.; Waring, P.M.; Desai, J.; Robledo, K.P.; Gebski, V.J.; Elez, E.; Nott, L.M.; Karapetis, C.S.; Lunke, S.; et al. Response to Cetuximab With or Without Irinotecan in Patients With Refractory Metastatic Colorectal Cancer Harboring the KRAS G13D Mutation: Australasian Gastro-Intestinal Trials Group ICECREAM Study. J. Clin. Oncol. 2016, 34, 2258–2264. [Google Scholar] [CrossRef]
- McFall, T.; Stites, E.C. A mechanism for the response of KRASG13D expressing colorectal cancers to EGFR inhibitors. Mol. Cell. Oncol. 2020, 7, 1701914. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Yan, W.-Y.; Xie, L.; Cheng, L.; Yang, M.; Li, L.; Shi, J.; Liu, B.-R.; Qian, X.-P. Coexistence of MSI with KRAS mutation is associated with worse prognosis in colorectal cancer. Medicine 2016, 95, e5649. [Google Scholar] [CrossRef]
- Watson, R.; Liu, T.-C.; Ruzinova, M.B. High frequency of KRAS mutation in early onset colorectal adenocarcinoma: Implications for pathogenesis. Hum. Pathol. 2016, 56, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Fearon, E.R.; Vogelstein, B. A genetic model for colorectal tumorigenesis. Cell 1990, 61, 759–767. [Google Scholar] [CrossRef]
- Kondelin, J.; Salokas, K.; Saarinen, L.; Ovaska, K.; Rauanheimo, H.; Plaketti, R.; Hamberg, J.; Liu, X.; Yadav, L.; Gylfe, A.E.; et al. Comprehensive evaluation of coding region point mutations in microsatellite-unstable colorectal cancer. EMBO Mol. Med. 2018, 10, e8552. [Google Scholar] [CrossRef]
- Marabelle, A.; Fakih, M.; Lopez, J.; Shah, M.; Shapira-Frommer, R.; Nakagawa, K.; Chung, H.C.; Kindler, H.L.; Lopez-Martin, J.A.; Miller, W.H.; et al. Association of tumour mutational burden with outcomes in patients with advanced solid tumours treated with pembrolizumab: Prospective biomarker analysis of the multicohort, open-label, phase 2 KEYNOTE-158 study. Lancet Oncol. 2020, 21, 1353–1365. [Google Scholar] [CrossRef]
- FDA Approves Pembrolizumab for Adults and Children with TMB-H Solid Tumors. Available online: https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-pembrolizumab-adults-and-children-tmb-h-solid-tumors (accessed on 16 November 2020).
- Passaro, A.; Stenzinger, A.; Peters, S. Tumor Mutational Burden as a Pan-cancer Biomarker for Immunotherapy: The Limits and Potential for Convergence. Cancer Cell 2020, 38, 624–625. [Google Scholar] [CrossRef]
- Adashek, J.J.; Subbiah, V.; Kurzrock, R. From Tissue-Agnostic to N-of-One Therapies: (R)Evolution of the Precision Paradigm. Trends Cancer 2021, 7, 15–28. [Google Scholar] [CrossRef]
- Prasad, V.; Addeo, A. The FDA approval of pembrolizumab for patients with TMB >10 mut/Mb: Was it a wise decision? No. Ann. Oncol. 2020, 31, 1112–1114. [Google Scholar] [CrossRef] [PubMed]
- Hause, R.J.; Pritchard, C.C.; Shendure, R.J.H.J.; Salipante, C.C.P.S.J. Classification and characterization of microsatellite instability across 18 cancer types. Nat. Med. 2016, 22, 1342–1350. [Google Scholar] [CrossRef] [PubMed]
- Coelho, M.A.; Trécesson, S.D.C.; Rana, S.; Zecchin, D.; Moore, C.; Molina-Arcas, M.; East, P.; Spencer-Dene, B.; Nye, E.; Barnouin, K.; et al. Oncogenic RAS Signaling Promotes Tumor Immunoresistance by Stabilizing PD-L1 mRNA. Immunity 2017, 47, 1083–1099.e6. [Google Scholar] [CrossRef]
- Vijayvergia, N.; Innocent, J.; Bhatia, A.K.; Shameem, R.; Xiu, J.; Gatalica, Z.; Grivennikov, S.; El-Deiry, W.S. Reduced PD-1/PD-L1 expression in KRAS-mutant versus wild-type microsatellite instable (MSI-H) colorectal cancer (CRC) and association of wnt pathway corepressor TLE-3. J. Clin. Oncol. 2015, 33, 3611. [Google Scholar] [CrossRef]
- Jeanson, A.; Tomasini, P.; Souquet-Bressand, M.; Brandone, N.; Boucekine, M.; Grangeon, M.; Chaleat, S.; Khobta, N.; Milia, J.; Mhanna, L.; et al. Efficacy of Immune Checkpoint Inhibitors in KRAS-Mutant Non-Small Cell Lung Cancer (NSCLC). J. Thorac. Oncol. 2019, 14, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
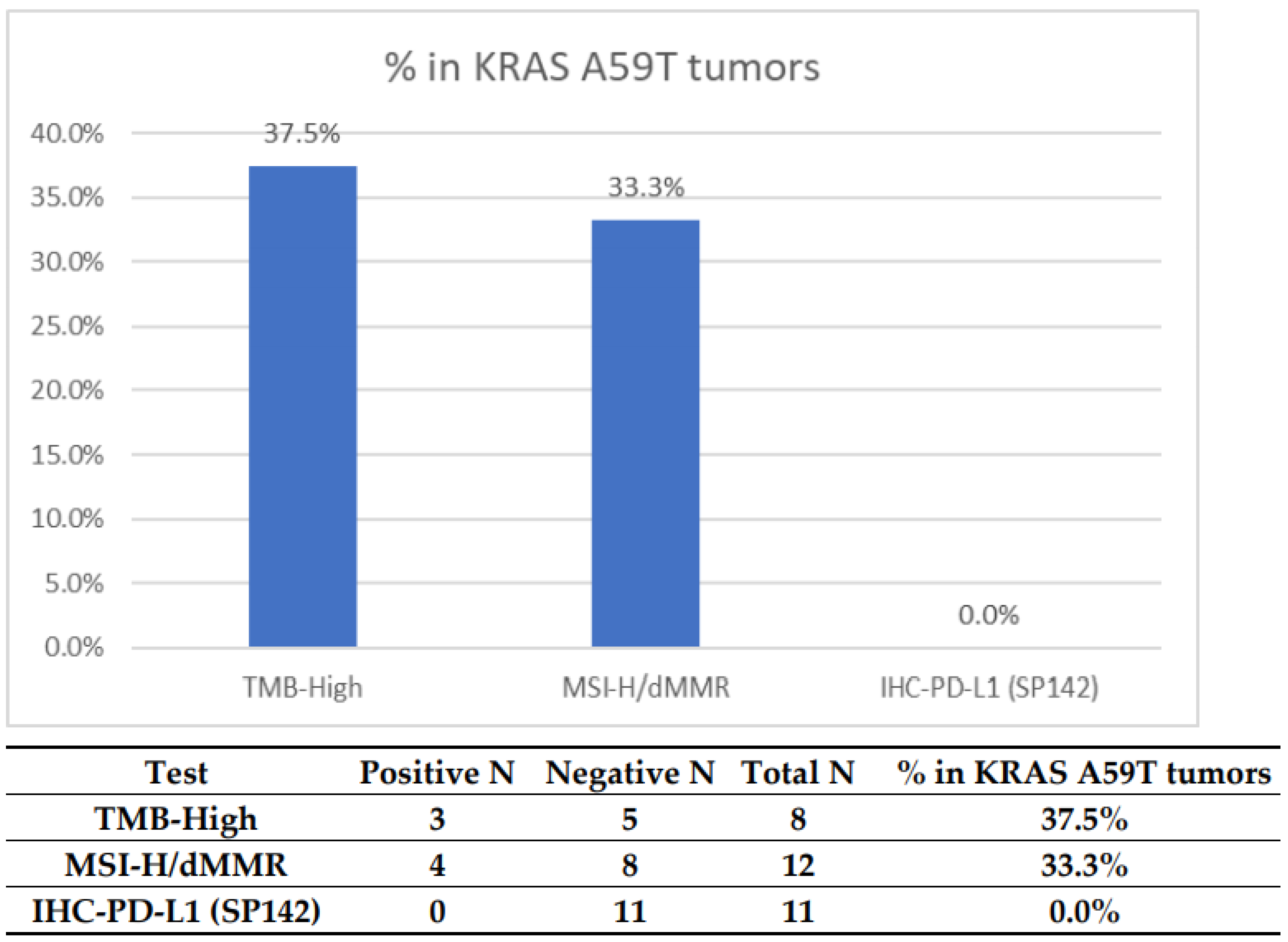
| Total N (%) | Female N (%) | Male N (%) | Median Age Female | Median Age Male | Age Range | |
|---|---|---|---|---|---|---|
| Primary | 10 (71.4) | 5 (35.7) | 5 (35.7) | 63.0 | 62.0 | 32–83 |
| Metastatic | 3 (21.4) | 2 (14.3) | 1 (7.1) | 63.0 | 83.0 | 61–83 |
| Unclear | 1 (7.1) | 0 (0.0) | 1 (7.1) | NA | 31.0 | 31 |
| Left | 5 (35.7) | 3 (21.4) | 2 (14.3) | 50.0 | 48.5 | 31–68 |
| Right | 5 (35.7) | 1 (7.1) | 4 (28.6) | 63.0 | 72.0 | 49–83 |
| Transverse | 1 (7.1) | 1 (7.1) | 0 (0.0) | 63.0 | NA | 63 |
| Unclear | 3 (21.4) | 2 (14.3) | 1 (7.1) | 63.0 | 62.0 | 61–65 |
| Total | 14 | 7 (50) | 7 (50) | 63.0 | 62.0 | 31–83 |
| Case ID | MSI/MMR | NGS-MSI | FA-MSI | MMR Deficiency (Four IHCs) |
|---|---|---|---|---|
| 1 | Not Tested | Not Tested | Not Tested | Not Tested |
| 2 | Not Tested | Not Tested | Not Tested | Not Tested |
| 3 | MSS/pMMR | Not Tested | stable | proficient |
| 4 | MSS/pMMR | Not Tested | Not Tested | proficient |
| 5 | MSS/pMMR | stable | Not Tested | proficient |
| 6 | MSS/pMMR | Not Tested | stable | proficient |
| 7 | MSS/pMMR | stable | stable | proficient |
| 8 | MSS/pMMR | stable | stable | proficient |
| 9 | MSI-H/dMMR | MSI-H | Not Tested | deficient |
| 10 | MSI-H/dMMR | MSI-H | Not Tested | Not Tested |
| 11 | MSS/pMMR | stable | Not Tested | proficient |
| 12 | MSI-H/dMMR | MSI-H | Not Tested | deficient |
| 13 | MSS/pMMR | stable | Not Tested | proficient |
| 14 | MSI-H/dMMR | MSI-H | Not Tested | Not Tested |
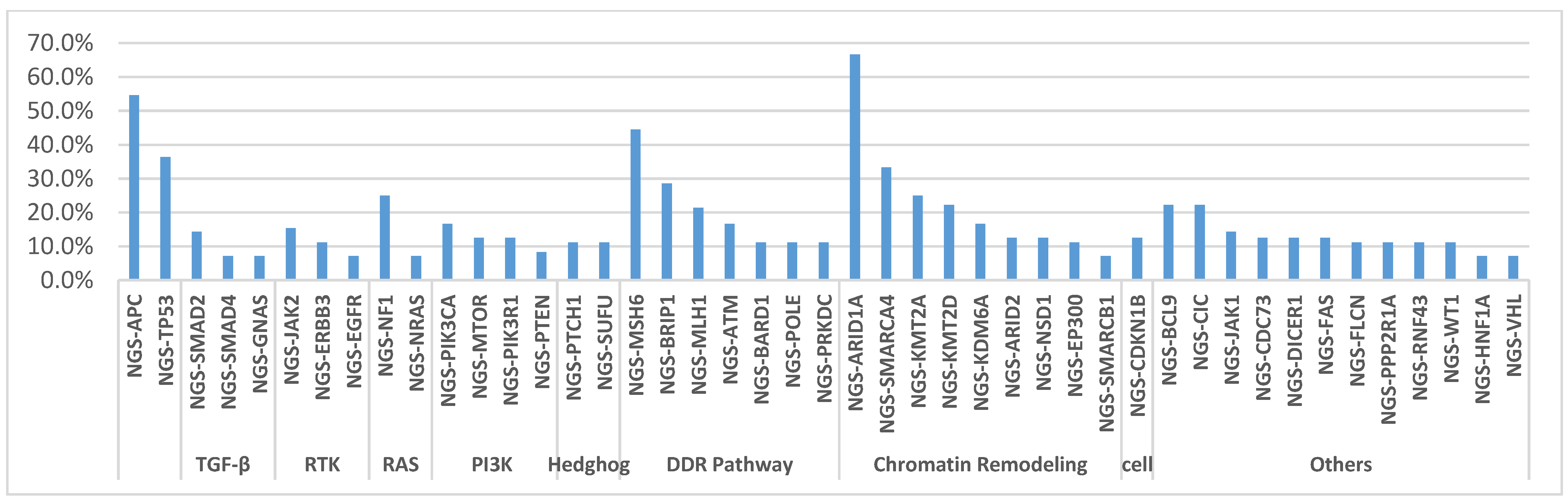
| Pathway | Test (NGS) | Positive | Negative | Total | % KRAS MT A59T |
|---|---|---|---|---|---|
| WNT Signaling Pathway | APC | 6 | 5 | 11 | 54.5% |
| TP53 Pathway | TP53 | 4 | 7 | 11 | 36.4% |
| TGF-β signaling pathway | SMAD2 | 1 | 6 | 7 | 14.3% |
| SMAD4 | 1 | 13 | 14 | 7.1% | |
| Signal Transduction | GNAS | 1 | 13 | 14 | 7.1% |
| Receptor Tyrosine Kinases/Co-factors | JAK2 | 2 | 11 | 13 | 15.4% |
| ERBB3 | 1 | 8 | 9 | 11.1% | |
| EGFR | 1 | 13 | 14 | 7.1% | |
| RAS Pathway | NF1 | 1 | 3 | 4 | 25.0% |
| NRAS | 1 | 13 | 14 | 7.1% | |
| PI3K Pathway | PIK3CA | 2 | 10 | 12 | 16.7% |
| MTOR | 1 | 7 | 8 | 12.5% | |
| PIK3R1 | 1 | 7 | 8 | 12.5% | |
| PTEN | 1 | 11 | 12 | 8.3% | |
| Hedgehog Signaling Pathway | PTCH1 | 1 | 8 | 9 | 11.1% |
| SUFU | 1 | 8 | 9 | 11.1% | |
| DDR Pathway | MSH6 | 4 | 5 | 9 | 44.4% |
| BRIP1 | 2 | 5 | 7 | 28.6% | |
| MLH1 | 3 | 11 | 14 | 21.4% | |
| ATM | 2 | 10 | 12 | 16.7% | |
| BARD1 | 1 | 8 | 9 | 11.1% | |
| POLE | 1 | 8 | 9 | 11.1% | |
| PRKDC | 1 | 8 | 9 | 11.1% | |
| Chromatin Remodeling | ARID1A | 4 | 2 | 6 | 66.7% |
| SMARCA4 | 3 | 6 | 9 | 33.3% | |
| KMT2A | 2 | 6 | 8 | 25.0% | |
| KMT2D | 2 | 7 | 9 | 22.2% | |
| KDM6A | 1 | 5 | 6 | 16.7% | |
| ARID2 | 1 | 7 | 8 | 12.5% | |
| NSD1 | 1 | 7 | 8 | 12.5% | |
| EP300 | 1 | 8 | 9 | 11.1% | |
| SMARCB1 | 1 | 13 | 14 | 7.1% | |
| Cell Cycle | CDKN1B | 1 | 7 | 8 | 12.5% |
| Other biomarkers | BCL9 | 2 | 7 | 9 | 22.2% |
| CIC | 2 | 7 | 9 | 22.2% | |
| JAK1 | 1 | 6 | 7 | 14.3% | |
| CDC73 | 1 | 7 | 8 | 12.5% | |
| DICER1 | 1 | 7 | 8 | 12.5% | |
| FAS | 1 | 7 | 8 | 12.5% | |
| FLCN | 1 | 8 | 9 | 11.1% | |
| PPP2R1A | 1 | 8 | 9 | 11.1% | |
| RNF43 | 1 | 8 | 9 | 11.1% | |
| WT1 | 1 | 8 | 9 | 11.1% | |
| HNF1A | 1 | 13 | 14 | 7.1% | |
| VHL | 1 | 13 | 14 | 7.1% |
| Pathway | Tumor | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MSI Status | High | High | High | High | Low | Low | Low | Low | Low | Low | Low | Low | |||
| NGS-POLE | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | ||||||
| TMB per Mb | 49 | IND | 14 | 19 | 294 | 5 | 16 | 11 | 9 | ||||||
| RAS Pathway | NGS-KRAS | A59T | A59T | A59T | A59T | A59T | A59T | A59T | A59T | A59T | A59T | A59T | A59T | A59T | A59T |
| NGS-BRAF | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| NGS-NRAS | 0 | 0 | 0 | 0 | A18T | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Cell Cycle | NGS-CDKN1B | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | ||||||
| Chromatin Remodeling | NGS-ARID1A | 1 | 1 | 1 | 0 | 1 | 0 | ||||||||
| NGS-ARID2 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||
| NGS-EP300 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | ||||||
| NGS-KDM6A | 1 | 0 | 0 | 0 | 0 | 0 | |||||||||
| NGS-KMT2A | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | |||||||
| NGS-KMT2D | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| NGS-NSD1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||||
| NGS-SMARCA4 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| NGS-SMARCB1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| DDR Pathway | NGS-ATM | 1 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
| NGS-BARD1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | ||||||
| NGS-BRIP1 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | ||||||||
| NGS-MLH1 | 1 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| NGS-MSH6 | 1 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | 0 | ||||||
| NGS-POLE | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | ||||||
| NGS-PRKDC | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | ||||||
| Hedgehog Signaling Pathway | NGS-PTCH1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| NGS-SUFU | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | ||||||
| PI3K Pathway | NGS-MTOR | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | ||||||
| NGS-PIK3CA | 0 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||
| NGS-PIK3R1 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | |||||||
| NGS-PTEN | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||
| NGS-NF1 | 0 | 1 | 0 | 0 | |||||||||||
| NGS-NRAS | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Receptor Tyrosine Kinases/Co-factors | NGS-EGFR | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| NGS-ERBB2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| NGS-JAK2 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||
| Signal Transduction | NGS-GNAS | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| TGF-β signaling pathway | NGS-SMAD2 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | |||||||
| NGS-SMAD4 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| TP53 Pathway | NGS-TP53 | 1 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 0 | 0 | 0 | |||
| WNT Signaling Pathway | NGS-APC | 0 | 0 | 1 | 1 | 1 | 1 | 1 | 0 | 1 | 0 | 0 | |||
| Other biomarkers | NGS-BCL9 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |||||
| NGS-CDC73 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | |||||||
| NGS-CIC | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| NGS-DICER1 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | |||||||
| NGS-FAS | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | |||||||
| NGS-FLCN | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| NGS-HNF1A | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | |
| NGS-JAK1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||||
| NGS-PPP2R1A | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| NGS-RNF43 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||
| NGS-VHL | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| NGS-WT1 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | ||||||
| CNA-FGFR4 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| Cancer Types | N | MSI-H/dMMR | MSS/pMMR | Unknown |
|---|---|---|---|---|
| Endometrial | 18 | 10 | 5 | 3 |
| Esophageal Junction | 5 | 3 | 2 | |
| Ovarian Surface Epithelial Carcinomas | 4 | 2 | 1 | 1 |
| Cancer of Unknown Primary | 2 | 2 | ||
| Bladder | 1 | 1 | ||
| Cervical Cancer | 1 | 1 | ||
| Cholangiocarcinoma | 1 | 1 | ||
| Gastric Adenocarcinoma | 1 | 1 | ||
| Lung Non-small cell lung cancer (NSCLC) | 1 | 1 | ||
| Penile Cancer | 1 | 1 | ||
| Small Intestinal Malignancies | 1 | 1 | ||
| Total | 36 | 21 | 9 | 6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lou, E.; Xiu, J.; Baca, Y.; Nelson, A.C.; Weinberg, B.A.; Beg, M.S.; Salem, M.E.; Lenz, H.-J.; Philip, P.; El-Deiry, W.S.; et al. Expression of Immuno-Oncologic Biomarkers Is Enriched in Colorectal Cancers and Other Solid Tumors Harboring the A59T Variant of KRAS. Cells 2021, 10, 1275. https://doi.org/10.3390/cells10061275
Lou E, Xiu J, Baca Y, Nelson AC, Weinberg BA, Beg MS, Salem ME, Lenz H-J, Philip P, El-Deiry WS, et al. Expression of Immuno-Oncologic Biomarkers Is Enriched in Colorectal Cancers and Other Solid Tumors Harboring the A59T Variant of KRAS. Cells. 2021; 10(6):1275. https://doi.org/10.3390/cells10061275
Chicago/Turabian StyleLou, Emil, Joanne Xiu, Yasmine Baca, Andrew C. Nelson, Benjamin A. Weinberg, Muhammad Shaalan Beg, Mohamed E. Salem, Heinz-Josef Lenz, Philip Philip, Wafik S. El-Deiry, and et al. 2021. "Expression of Immuno-Oncologic Biomarkers Is Enriched in Colorectal Cancers and Other Solid Tumors Harboring the A59T Variant of KRAS" Cells 10, no. 6: 1275. https://doi.org/10.3390/cells10061275
APA StyleLou, E., Xiu, J., Baca, Y., Nelson, A. C., Weinberg, B. A., Beg, M. S., Salem, M. E., Lenz, H.-J., Philip, P., El-Deiry, W. S., & Korn, W. M. (2021). Expression of Immuno-Oncologic Biomarkers Is Enriched in Colorectal Cancers and Other Solid Tumors Harboring the A59T Variant of KRAS. Cells, 10(6), 1275. https://doi.org/10.3390/cells10061275






