Basophils Orchestrating Eosinophils’ Chemotaxis and Function in Allergic Inflammation
Abstract
1. Eosinophils and Basophils Are Pivotal Players in the Progression of Allergic Inflammation
2. Chemotaxis
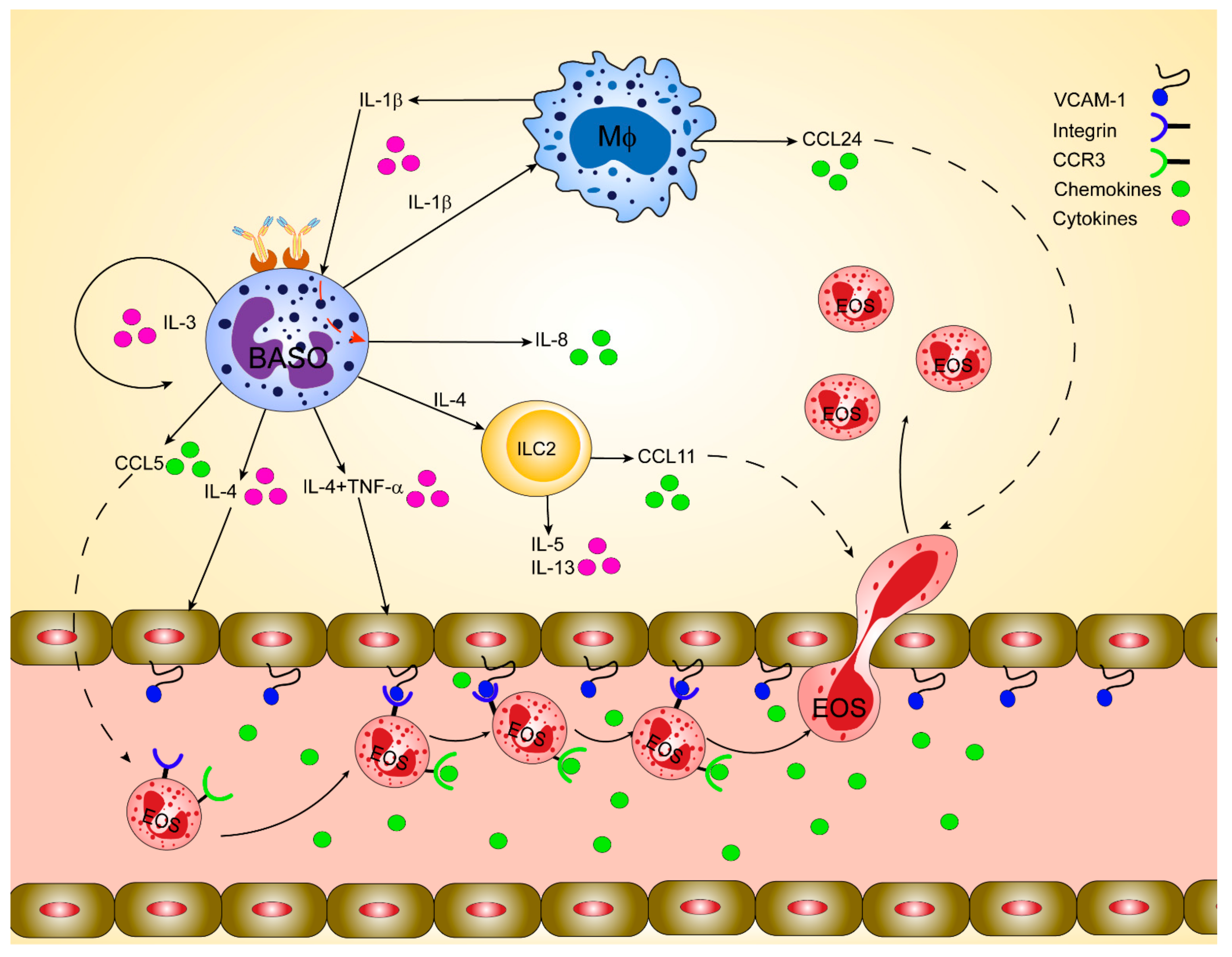
2.1. Contribution of Basophils to Regulate Eosinophil Infiltration to Inflamed Skin
2.2. The Controversial Role of Basophils in Affecting Eosinophil Infiltration to Sites of Inflammation in the Airways
2.3. Overview of the General Mechanisms of Eosinophil and Basophil Chemotaxis
2.4. The CCR3/CCL11/CCL24 Axis
2.5. The GM-CSF Cytokine Family Axis
2.6. Mechanisms of How Basophil Imprint Eosinophil Migration to Inflamed Skin and Lung
3. Cell-to-Cell Interaction at Sites of Inflammation
4. Common Molecular Targets on Basophils and Eosinophils for Novel Therapies in Allergic Inflammation
5. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Akdis, C.A.; Arkwright, P.D.; Brüggen, M.-C.; Busse, W.; Gadina, M.; Guttman-Yassky, E.; Kabashima, K.; Mitamura, Y.; Vian, L.; Wu, J.; et al. Type 2 immunity in the skin and lungs. Allergy 2020, 75, 1582–1605. [Google Scholar] [CrossRef]
- Stone, K.D.; Prussin, C.; Metcalfe, D.D. IgE, mast cells, basophils, and eosinophils. J. Allergy Clin. Immunol. 2010, 125, S73–S80. [Google Scholar] [CrossRef]
- Siracusa, M.C.; Kim, B.S.; Spergel, J.M.; Artis, D. Basophils and allergic inflammation. J. Allergy Clin. Immunol. 2013, 132, 789–801. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.B.; Liu, M.C.; Galli, S.J.; Bochner, B.S.; Kagey-Sobotka, A.; Lichtenstein, L.M. Identification of IgE-bearing cells in the late-phase response to antigen in the lung as basophils. Am. J. Respir. Cell Mol. Biol. 1994, 10, 384–390. [Google Scholar] [CrossRef]
- Devouassoux, G.; Foster, B.; Scott, L.M.; Metcalfe, D.D.; Prussin, C. Frequency and characterization of antigen-specific IL-4– and IL-13– producing basophils and T cells in peripheral blood of healthy and asthmatic subjects. J. Allergy Clin. Immunol. 1999, 104, 811–819. [Google Scholar] [CrossRef]
- Marone, G.; Granata, F.; Pucino, V.; Pecoraro, A.; Heffler, E.; Loffredo, S.; Scadding, G.W.; Varricchi, G. The Intriguing Role of Interleukin 13 in the Pathophysiology of Asthma. Front. Pharmacol. 2019, 10, 1387. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, C.; Otsuka, A.; Kitoh, A.; Honda, T.; Egawa, G.; Nakajima, S.; Nakamizo, S.; Arita, M.; Kubo, M.; Miyachi, Y.; et al. Basophils regulate the recruitment of eosinophils in a murine model of irritant contact dermatitis. J. Allergy Clin. Immunol. 2014, 134, 100–107.e12. [Google Scholar] [CrossRef]
- Cheng, L.E.; Sullivan, B.M.; Retana, L.E.; Allen, C.D.; Liang, H.-E.; Locksley, R.M. IgE-activated basophils regulate eosinophil tissue entry by modulating endothelial function. J. Exp. Med. 2015, 212, 513–524. [Google Scholar] [CrossRef]
- Iype, J.; Odermatt, A.; Bachmann, S.; Coeudevez, M.; Fux, M. IL-1b Promotes Immunoregulatory Responses in Human Blood Basophils. Allergy 2021, 1. [Google Scholar] [CrossRef]
- Perrigoue, J.G.; Saenz, S.A.; Siracusa, M.C.; Allenspach, E.J.; Taylor, B.C.; Giacomin, P.R.; Nair, M.G.; Du, Y.; Zaph, C.; Van Rooijen, N.; et al. MHC class II–dependent basophil–CD4+ T cell interactions promote TH2 cytokine–dependent immunity. Nat. Immunol. 2009, 10, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Yoshimoto, T.; Yasuda, K.; Tanaka, H.; Nakahira, M.; Imai, Y.; Fujimori, Y.; Nakanishi, K. Basophils contribute to TH2-IgE responses in vivo via IL-4 production and presentation of peptide–MHC class II complexes to CD4+ T cells. Nat. Immunol. 2009, 10, 706–712. [Google Scholar] [CrossRef] [PubMed]
- Sokol, C.L.; Chu, N.-Q.; Yu, S.; Nish, S.A.; Laufer, T.M.; Medzhitov, R. Basophils function as antigen-presenting cells for an allergen-induced T helper type 2 response. Nat. Immunol. 2009, 10, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Van Beek, A.A.; Knol, E.F.; De Vos, P.; Smelt, M.J.; Savelkoul, H.F.J.; Van Neerven, R.J.J. Recent Developments in Basophil Research: Do Basophils Initiate and Perpetuate Type 2 T-Helper Cell Responses? Int. Arch. Allergy Immunol. 2013, 160, 7–17. [Google Scholar] [CrossRef]
- Hogan, S.P.; Rosenberg, H.F.; Moqbel, R.; Phipps, S.; Foster, P.S.; Lacy, P.; Kay, A.B.; Rothenberg, M.E. Eosinophils: Biological Properties and Role in Health and Disease. Clin. Exp. Allergy 2008, 38, 709–750. [Google Scholar] [CrossRef]
- O’Donnell, M.C.; Ackerman, S.J.; Gleich, G.J.; Thomas, L.L. Activation of basophil and mast cell histamine release by eosinophil granule major basic protein. J. Exp. Med. 1983, 157, 1981–1991. [Google Scholar] [CrossRef]
- Koshino, T.; Teshima, S.; Fukushima, N.; Takaishi, T.; Hirai, K.; Miyamoto, Y.; Arai, Y.; Sano, Y.; Ito, K.; Morita, Y. Identification of basophils by immunohistochemistry in the airways of post-mortem cases of fatal asthma. Clin. Exp. Allergy 1993, 23, 919–925. [Google Scholar] [CrossRef]
- Macfarlane, A.J.; Kon, O.M.; Smith, S.J.; Zeibecoglou, K.; Khan, L.; Barata, L.T.; McEuen, A.R.; Buckley, M.G.; Walls, A.F.; Meng, Q.; et al. Basophils, eosinophils, and mast cells in atopic and nonatopic asthma and in late-phase allergic reactions in the lung and skin. J. Allergy Clin. Immunol. 2000, 105, 99–107. [Google Scholar] [CrossRef]
- Irani, A.-M.A.; Huang, C.; Xia, H.-Z.; Kepley, C.; Nafie, A.; Fouda, E.D.; Craig, S.; Zweiman, B.; Schwartz, L.B. Immunohistochemical detection of human basophils in late-phase skin reactions. J. Allergy Clin. Immunol. 1998, 101, 354–362. [Google Scholar] [CrossRef]
- Gauvreau, G.M.; Lee, J.M.; Watson, R.M.; Irani, A.-M.A.; Schwartz, L.B.; O’Byrne, P.M. Increased Numbers of Both Airway Basophils and Mast Cells in Sputum after Allergen Inhalation Challenge of Atopic Asthmatics. Am. J. Respir. Crit. Care Med. 2000, 161, 1473–1478. [Google Scholar] [CrossRef] [PubMed]
- Dijkstra, D.; Hennig, C.; Hansen, G.; Biller, H.; Krug, N.; Hohlfeld, J.M. Identification and quantification of basophils in the airways of asthmatics following segmental allergen challenge. Cytom. Part. A 2014, 85, 580–587. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Satoh, T.; Takayama, K.; Miyagishi, C.; Walls, A.F.; Yokozeki, H. Basophil recruitment and activation in inflammatory skin diseases. Allergy 2011, 66, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Ying, S.; Robinson, D.S.; Meng, Q.; Barata, L.T.; McEuen, A.R.; Buckley, M.G.; Walls, A.F.; Askenase, P.W.; Kay, A.B. C-C Chemo-kines in Allergen-Induced Late-Phase Cutaneous Responses in Atopic Subjects: Association of Eotaxin with Early 6-Hour Eosinophils, and of Eotaxin-2 and Monocyte Chemoattractant Protein-4 with the Later 24-Hour Tissue Eosinophilia, and Relationship to Basophils and Other C-C Chemokines (Monocyte Chemoattractant Protein-3 and RANTES). J. Immunol. 1999, 163, 3976–3984. [Google Scholar]
- Satoh, T.; Ito, Y.; Miyagishi, C.; Yokozeki, H. Basophils Infiltrate Skin Lesions of Eosinophilic Pustular Folliculitis (Ofuji’s Disease). ACTA Derm. Venereol. 2011, 91, 371–372. [Google Scholar] [CrossRef] [PubMed]
- Wardlaw, A.J.; Dunnette, S.; Gleich, G.J.; Collins, J.V.; Kay, A.B. Eosinophils and Mast Cells in Bronchoalveolar Lavage in Subjects with Mild Asthma: Relationship to Bronchial Hyperreactivity. Am. Rev. Respir. Dis. 1988, 137, 62–69. [Google Scholar] [CrossRef]
- Metcalfe, D.D.; Pawankar, R.; Ackerman, S.J.; Akin, C.; Clayton, F.; Falcone, F.H.; Gleich, G.J.; Irani, A.-M.; Johansson, M.W.; Klion, A.D.; et al. Biomarkers of the involvement of mast cells, basophils and eosinophils in asthma and allergic diseases. World Allergy Organ. J. 2016, 9, 7. [Google Scholar] [CrossRef] [PubMed]
- Motomura, Y.; Morita, H.; Moro, K.; Nakae, S.; Artis, D.; Endo, T.A.; Kuroki, Y.; Ohara, O.; Koyasu, S.; Kubo, M. Basophil-Derived Interleukin-4 Controls the Function of Natural Helper Cells, a Member of ILC2s, in Lung Inflammation. Immunology 2014, 40, 758–771. [Google Scholar] [CrossRef]
- Thomas, L.L. Basophil and Eosinophil Interactions in Health and Disease. Chem. Immunol. 1995, 61, 186–207. [Google Scholar]
- Shakoory, B.; Fitzgerald, S.M.; Lee, S.A.; Chi, D.S.; Krishnaswamy, G. The Role of Human Mast Cell-Derived Cytokines in Eosinophil Biology. J. Interf. Cytokine Res. 2004, 24, 271–281. [Google Scholar] [CrossRef]
- Wardlaw, A.J. Eosinophil trafficking in asthma. Clin. Med. 2001, 1, 214–218. [Google Scholar] [CrossRef]
- Van Oosterhout, A.J.; Fattah, D.; Van Ark, I.; Hofman, G.; Buckley, T.L.; Nijkamp, F.P. Eosinophil infiltration precedes development of airway hyperreactivity and mucosal exudation after intranasal administration of interleukin-5 to mice. J. Allergy Clin. Immunol. 1995, 96, 104–112. [Google Scholar] [CrossRef]
- Wang, J.; Palmer, K.; Lotvall, J.; Milan, S.; Lei, X.F.; Matthaei, I.K.; Gauldie, J.; Inman, M.D.; Jordana, M.; Xing, Z. Circulating, but not local lung, IL-5 is required for the development of antigen-induced airways eosinophilia. J. Clin. Investig. 1998, 102, 1132–1141. [Google Scholar] [CrossRef] [PubMed]
- Mukai, K.; Matsuoka, K.; Taya, C.; Suzuki, H.; Yokozeki, H.; Nishioka, K.; Hirokawa, K.; Etori, M.; Yamashita, M.; Kubota, T.; et al. Basophils Play a Critical Role in the Development of IgE-Mediated Chronic Allergic Inflammation Independently of T Cells and Mast Cells. Immunology 2005, 23, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Obata, K.; Mukai, K.; Tsujimura, Y.; Ishiwata, K.; Kawano, Y.; Minegishi, Y.; Watanabe, N.; Karasuyama, H. Basophils are essential initiators of a novel type of chronic allergic inflammation. Blood 2007, 110, 913–920. [Google Scholar] [CrossRef] [PubMed]
- Ohnmacht, C.; Schwartz, C.; Panzer, M.; Schiedewitz, I.; Naumann, R.; Voehringer, D. Basophils Orchestrate Chronic Allergic Dermatitis and Protective Immunity against Helminths. Immunology 2010, 33, 364–374. [Google Scholar] [CrossRef]
- Matsuoka, K.; Shitara, H.; Taya, C.; Kohno, K.; Kikkawa, Y.; Yonekawa, H. Novel Basophil- or Eosinophil-Depleted Mouse Models for Functional Analyses of Allergic Inflammation. PLoS ONE 2013, 8, e60958. [Google Scholar] [CrossRef] [PubMed]
- Mashiko, S.; Mehta, H.; Bissonnette, R.; Sarfati, M. Increased frequencies of basophils, type 2 innate lymphoid cells and Th2 cells in skin of patients with atopic dermatitis but not psoriasis. J. Dermatol. Sci. 2017, 88, 167–174. [Google Scholar] [CrossRef]
- Nabe, T.; Zindl, C.L.; Jung, Y.W.; Stephens, R.; Sakamoto, A.; Kohno, S.; Atkinson, T.P.; Chaplin, D.D. Induction of a late asthmatic response associated with airway inflammation in mice. Eur. J. Pharmacol. 2005, 521, 144–155. [Google Scholar] [CrossRef]
- Zhong, W.; Su, W.; Zhang, Y.; Liu, Q.; Wu, J.; Di, C.; Zhang, Z.; Xia, Z. Basophils as a primary inducer of the T helper type 2 immunity in ovalbumin-induced allergic airway inflammation. Immunology 2014, 142, 202–215. [Google Scholar] [CrossRef]
- Hammad, H.; Plantinga, M.; Deswarte, K.; Pouliot, P.; Willart, M.A.; Kool, M.; Muskens, F.; Lambrecht, B.N. Inflammatory dendritic cells—Not basophils—Are necessary and sufficient for induction of Th2 immunity to inhaled house dust mite allergen. J. Exp. Med. 2010, 207, 2097–2111. [Google Scholar] [CrossRef]
- Matsuoka, K.; Taya, C.; Kubo, S.; Toyama-Sorimachi, N.; Kitamura, F.; Ra, C.; Yonekawa, H.; Karasuyama, H. Establishment of antigen-specific IgE transgenic mice to study pathological and immunobiological roles of IgE in vivo. Int. Immunol. 1999, 11, 987–994. [Google Scholar] [CrossRef]
- Simon, D.; Braathen, L.R.; Simon, H.-U. Eosinophils and atopic dermatitis. Allergy 2004, 59, 561–570. [Google Scholar] [CrossRef] [PubMed]
- Wakita, H.; Sakamoto, T.; Tokura, Y.; Takigawa, M. E-selectin and vascular cell adhesion molecule-1 as critical adhesion molecules for infiltration of T lymphocytes and eosinophils in atopic dermatitis. J. Cutan. Pathol. 1994, 21, 33–39. [Google Scholar] [CrossRef]
- Yawalkar, N.; Uguccioni, M.; Schärer, J.; Braunwalder, J.; Karlen, S.; Dewald, B.; Braathen, L.R.; Baggiolini, M. Enhanced Expression of Eotaxin and CCR3 in Atopic Dermatitis. J. Investig. Dermatol. 1999, 113, 43–48. [Google Scholar] [CrossRef]
- Nabe, T.; Matsuya, K.; Akamizu, K.; Fujita, M.; Nakagawa, T.; Shioe, M.; Kida, H.; Takiguchi, A.; Wakamori, H.; Fujii, M.; et al. Roles of basophils and mast cells infiltrating the lung by multiple antigen challenges in asthmatic responses of mice. Br. J. Pharmacol. 2013, 169, 462–476. [Google Scholar] [CrossRef]
- Fattouh, R.; Al-Garawi, A.; Fattouh, M.; Arias, K.; Walker, T.D.; Goncharova, S.; Coyle, A.J.; Humbles, A.A.; Jordana, M. Eosinophils Are Dispensable for Allergic Remodeling and Immunity in a Model of House Dust Mite–induced Airway Disease. Am. J. Respir. Crit. Care Med. 2011, 183, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Graham, M.T.; Nadeau, K.C. Lessons learned from mice and man: Mimicking human allergy through mouse models. Clin. Immunol. 2014, 155, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.-L.; Sen, A.I.; Kitaura, M.; Yoshie, O.; Rothenberg, M.E.; Murphy, P.M.; Luster, A.D. Identification of a Mouse Eosinophil Receptor for the CC Chemokine Eotaxin. Biochem. Biophys. Res. Commun. 1996, 223, 679–684. [Google Scholar] [CrossRef] [PubMed]
- Daugherty, B.L.; Siciliano, S.J.; DeMartino, A.J.; Malkowitz, L.; Sirotina, A.; Springer, M.S. Cloning, expression, and characterization of the human eosinophil eotaxin receptor. J. Exp. Med. 1996, 183, 2349–2354. [Google Scholar] [CrossRef] [PubMed]
- Uguccioni, M.; Mackay, C.R.; Ochensberger, B.; Loetscher, P.; Rhis, S.; LaRosa, G.J.; Rao, P.; Ponath, P.D.; Baggiolini, M.; Dahinden, A.C. High expression of the chemokine receptor CCR3 in human blood basophils. Role in activation by eotaxin, MCP-4, and other chemokines. J. Clin. Investig. 1997, 100, 1137–1143. [Google Scholar] [CrossRef]
- Ma, W.; Bryce, P.J.; Humbles, A.A.; Laouini, D.; Yalcindag, A.; Alenius, H.; Friend, D.S.; Oettgen, H.C.; Gerard, C.; Geha, R.S. CCR3 is essential for skin eosinophilia and airway hyperresponsiveness in a murine model of allergic skin inflammation. J. Clin. Investig. 2002, 109, 621–628. [Google Scholar] [CrossRef]
- Humbles, A.A.; Lu, B.; Friend, D.S.; Okinaga, S.; Lora, J.; Al-Garawi, A.; Martin, T.R.; Gerard, N.P.; Gerard, C. The murine CCR3 receptor regulates both the role of eosinophils and mast cells in allergen-induced airway inflammation and hyperresponsiveness. Proc. Natl. Acad. Sci. USA 2002, 99, 1479–1484. [Google Scholar] [CrossRef] [PubMed]
- Pope, S.M.; Zimmermann, N.; Stringer, K.F.; Karow, M.L.; Rothenberg, M.E. The Eotaxin Chemokines and CCR3 Are Fundamental Regulators of Allergen-Induced Pulmonary Eosinophilia. J. Immunol. 2005, 175, 5341–5350. [Google Scholar] [CrossRef] [PubMed]
- Sénéchal, S.; Fahy, O.; Gentina, T.; Vorng, H.; Capron, M.; Walls, A.F.; McEuen, A.R.; Buckley, M.G.; Hamid, Q.; Wallaert, B.; et al. CCR3-Blocking Antibody Inhibits Allergen-Induced Eosinophil Recruitment in Human Skin Xenografts from Allergic Patients. Lab. Investig. 2002, 82, 929–939. [Google Scholar] [CrossRef] [PubMed]
- Kaburagi, Y.; Shimada, Y.; Nagaoka, T.; Hasegawa, M.; Takehara, K.; Sato, S. Enhanced production of CC-chemokines (RANTES, MCP-1, MIP-1α, MIP-1β, and eotaxin) in patients with atopic dermatitis. Arch. Dermatol. Res. 2001, 293, 350–355. [Google Scholar] [CrossRef]
- Lilly, C.M.; Woodruff, P.G.; Camargo, C.A.; Nakamura, H.; Drazen, J.M.; Nadel, E.S.; Hanrahan, J.P. Elevated plasma eotaxin levels in patients with acute asthma. J. Allergy Clin. Immunol. 1999, 104, 786–790. [Google Scholar] [CrossRef]
- Nakamura, H.; Weiss, S.T.; Israel, E.; Luster, A.D.; Drazen, J.M.; Lilly, C.M. Eotaxin and Impaired Lung Function in Asthma. Am. J. Respir. Crit. Care Med. 1999, 160, 1952–1956. [Google Scholar] [CrossRef]
- Berkman, N.; Ohnona, S.; Chung, F.K.; Breuer, R. Eotaxin-3 but Not Eotaxin Gene Expression Is Upregulated in Asthmatics 24 Hours after Allergen Challenge. Am. J. Respir. Cell Mol. Biol. 2001, 24, 682–687. [Google Scholar] [CrossRef] [PubMed]
- Kagami, S.; Kakinuma, T.; Saeki, H.; Tsunemi, Y.; Fujita, H.; Nakamura, K.; Takekoshi, T.; Kishimoto, M.; Mitsui, H.; Torii, H.; et al. Significant elevation of serum levels of eotaxin-3/CCL26, but not of eotaxin-2/CCL24, in patients with atopic dermatitis: Serum eotaxin-3/CCL26 levels reflect the disease activity of atopic dermatitis. Clin. Exp. Immunol. 2003, 134, 309–313. [Google Scholar] [CrossRef]
- Kitaura, M.; Nakajima, T.; Imai, T.; Harada, S.; Combadiere, C.; Tiffany, H.L.; Murphy, P.M.; Yoshie, O. Molecular Cloning of Human Eotaxin, an Eosinophil-selective CC Chemokine, and Identification of a Specific Eosinophil Eotaxin Receptor, CC Chemokine Receptor 3. J. Biol. Chem. 1996, 271, 7725–7730. [Google Scholar] [CrossRef] [PubMed]
- Ponath, P.D.; Qin, S.; Ringler, D.J.; Clark-Lewis, I.; Wang, J.; Kassam, N.; Smith, H.; Shi, X.; Gonzalo, A.J.; Newman, W.; et al. Cloning of the human eosinophil chemoattractant, eotaxin. Expression, receptor binding, and functional properties suggest a mechanism for the selective recruitment of eosinophils. J. Clin. Investig. 1996, 97, 604–612. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Zepeda, E.A.; Rothenberg, M.E.; Ownbey, R.T.; Celestin, J.; Leder, P.; Luster, A.D. Human eotaxin is a specific chemoattractant for eosinophil cells and provides a new mechanism to explain tissue eosinophilia. Nat. Med. 1996, 2, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Matthews, A.N.; Friend, D.S.; Zimmermann, N.; Sarafi, M.N.; Luster, A.D.; Pearlman, E.; Wert, S.E.; Rothenberg, M.E. Eotaxin is required for the baseline level of tissue eosinophils. Proc. Natl. Acad. Sci. USA 1998, 95, 6273–6278. [Google Scholar] [CrossRef] [PubMed]
- Pope, S.M.; Fulkerson, P.C.; Blanchard, C.; Akei, H.S.; Nikolaidis, N.M.; Zimmermann, N.; Molkentin, J.D.; Rothenberg, M.E. Identification of a Cooperative Mechanism Involving Interleukin-13 and Eotaxin-2 in Experimental Allergic Lung Inflammation. J. Biol. Chem. 2005, 280, 13952–13961. [Google Scholar] [CrossRef] [PubMed]
- Rothenberg, M.E.; MacLean, J.A.; Pearlman, E.; Luster, A.D.; Leder, P. Targeted Disruption of the Chemokine Eotaxin Partially Reduces Antigen-induced Tissue Eosinophilia. J. Exp. Med. 1997, 185, 785–790. [Google Scholar] [CrossRef] [PubMed]
- Yamada, H.; Hirai, K.; Miyamasu, M.; Iikura, M.; Misaki, Y.; Shoji, S.; Takaishi, T.; Kasahara, T.; Morita, Y.; Ito, K. Eotaxin Is a Potent Chemotaxin for Human Basophils. Biochem. Biophys. Res. Commun. 1997, 231, 365–368. [Google Scholar] [CrossRef]
- Forssmann, U.; Uguccioni, M.; Loetscher, P.; Dahinden, C.A.; Langen, H.; Thelen, M.; Baggiolini, M. Eotaxin-2, a Novel CC Chemokine that Is Selective for the Chemokine Receptor CCR3, and Acts Like Eotaxin on Human Eosinophil and Basophil Leukocytes. J. Exp. Med. 1997, 185, 2171–2176. [Google Scholar] [CrossRef]
- Alam, R.; York, J.; Boyars, M.; Stafford, S.; Grant, A.J.; Lee, J.; Forsythe, P.; Šim, T.; Ida, N. Increased MCP-1, RANTES, and MIP-1alpha in bronchoalveolar lavage fluid of allergic asthmatic patients. Am. J. Respir. Crit. Care Med. 1996, 153, 1398–1404. [Google Scholar] [CrossRef]
- Ahmadi, Z.; Hassanshahi, G.; Khorramdelazad, H.; Zainodini, N.; Koochakzadeh, L. An Overlook to the Characteristics and Roles Played by Eotaxin Network in the Pathophysiology of Food Allergies: Allergic Asthma and Atopic Dermatitis. Inflammation 2016, 39, 1–15. [Google Scholar] [CrossRef]
- Foster, P.S.; Maltby, S.; Rosenberg, H.F.; Tay, H.L.; Hogan, S.P.; Collison, A.M.; Yang, M.; Kaiko, G.E.; Hansbro, P.M.; Kumar, R.K.; et al. Modeling TH2 responses and airway inflammation to understand fundamental mechanisms regulating the pathogenesis of asthma. Immunol. Rev. 2017, 278, 20–40. [Google Scholar] [CrossRef]
- Esnault, S.; Kelly, E.A. Essential mechanisms of differential activation of eosinophils by IL-3 compared to GM-CSF and IL-5. Crit. Rev. Immunol. 2017, 36, 429–444. [Google Scholar] [CrossRef]
- Broide, D.H.; Paine, M.M.; Firestein, G.S. Eosinophils express interleukin 5 and granulocyte macrophage-colony-stimulating factor mRNA at sites of allergic inflammation in asthmatics. J. Clin. Investig. 1992, 90, 1414–1424. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.W.; Kelly, E.A.B.; Busse, W.W.; Jarjour, N.N.; Mosher, D.F. Up-Regulation and Activation of Eosinophil Integrins in Blood and Airway after Segmental Lung Antigen Challenge. J. Immunol. 2008, 180, 7622–7635. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Liu, M.C.; Stealey, A.B.; Friedman, B.; Lichtenstein, L.M.; Permutt, S.; Schleimer, R.P. Production of granulocyte/macrophage colony-stimulating factor in human airways during allergen-induced late-phase reactions in atopic subjects. Lymphokine Cytokine Res. 1992, 11, 287–292. [Google Scholar] [PubMed]
- Moser, R.; Fehr, J.; Olgiati, L.; Bruijnzeel, P.L. Migration of primed human eosinophils across cytokine-activated endothelial cell monolayers. Blood 1992, 79, 2937–2945. [Google Scholar] [CrossRef]
- Dougan, M.; Dranoff, G.; Dougan, S.K. GM-CSF, IL-3, and IL-5 Family of Cytokines: Regulators of Inflammation. Immunology 2019, 50, 796–811. [Google Scholar] [CrossRef] [PubMed]
- Kämpfer, S.S.; Odermatt, A.; Dahinden, C.A.; Fux, M. Late IL-3–induced phenotypic and functional alterations in human basophils require continuous IL-3 receptor signaling. J. Leukoc. Biol. 2016, 101, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Koketsu, R.; Suzukawa, M.; Kawakami, A.; Iikura, M. Human Basophils and Cytokines/Chemokines. Allergol. Int. 2009, 58, 1–10. [Google Scholar] [CrossRef]
- Bochner, B.S.; McKelvey, A.A.; Sterbinsky, A.S.; Hildreth, E.J.; Derse, C.P.; Klunk, A.D.; Lichtenstein, L.M.; Schleimer, R.P. IL-3 augments adhesiveness for endothelium and CD11b expression in human basophils but not neutrophils. J. Immunol. 1990, 145, 1832–1837. [Google Scholar]
- Yoshimura-Uchiyama, C.; Yamaguchi, M.; Nagase, H.; Fujisawa, T.; Ra, C.; Matsushima, K.; Iwata, T.; Igarashi, T.; Yamamoto, K.; Hirai, K. Comparative effects of basophil-directed growth factors. Biochem. Biophys. Res. Commun. 2003, 302, 201–206. [Google Scholar] [CrossRef]
- Ebisawa, M.; Yamada, T.; Bickel, C.; Klunk, D.; Schleimer, R.P. Eosinophil transendothelial migration induced by cytokines. III. Effect of the chemokine RANTES. J. Immunol. 1994, 153, 2153–2160. [Google Scholar]
- Tanimoto, Y.; Takahashi, K.; Kimura, I. Effects of cytokines on human basophil chemotaxis. Clin. Exp. Allergy 1992, 22, 1020–1025. [Google Scholar] [CrossRef]
- Schroeder, J.T.; Chichester, K.L.; Bieneman, A.P. Human Basophils Secrete IL-3: Evidence of Autocrine Priming for Phenotypic and Functional Responses in Allergic Disease. J. Immunol. 2009, 182, 2432–2438. [Google Scholar] [CrossRef]
- Simson, L.; Foster, P.S. Chemokine and cytokine cooperativity: Eosinophil migration in the asthmatic response. Immunol. Cell Biol. 2000, 78, 415–422. [Google Scholar] [CrossRef]
- Mould, A.W.; Matthaei, I.K.; Young, I.G.; Foster, P.S. Relationship between interleukin-5 and eotaxin in regulating blood and tissue eosinophilia in mice. J. Clin. Investig. 1997, 99, 1064–1071. [Google Scholar] [CrossRef] [PubMed]
- Mould, A.W.; Ramsay, A.J.; Matthaei, K.I.; Young, I.G.; Rothenberg, M.E.; Foster, P.S. The Effect of IL-5 and Eotaxin Expression in the Lung on Eosinophil Trafficking and Degranulation and the Induction of Bronchial Hyperreactivity. J. Immunol. 2000, 164, 2142–2150. [Google Scholar] [CrossRef] [PubMed]
- Collins, P.D.; Marleau, S.; Griffiths-Johnson, A.D.; Jose, P.J.; Williams, T.J. Cooperation between interleukin-5 and the chemokine eotaxin to induce eosinophil accumulation in vivo. J. Exp. Med. 1995, 182, 1169–1174. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.W. Activation states of blood eosinophils in asthma. Clin. Exp. Allergy 2014, 44, 482–498. [Google Scholar] [CrossRef] [PubMed]
- Henderson, W.R.; Chi, E.Y.; Maliszewski, C.R. Soluble IL-4 Receptor Inhibits Airway Inflammation Following Allergen Challenge in a Mouse Model of Asthma. J. Immunol. 2000, 164, 1086–1095. [Google Scholar] [CrossRef] [PubMed]
- Brusselle, G.; Kips, J.; Joos, G.; Bluethmann, H.; Pauwels, R. Allergen-induced airway inflammation and bronchial responsiveness in wild-type and interleukin-4-deficient mice. Am. J. Respir. Cell Mol. Biol. 1995, 12, 254–259. [Google Scholar] [CrossRef]
- Mochizuki, M.; Schröder, J.-M.; Christophers, E.; Yamamoto, S. IL-4 Induces Eotaxin in Human Dermal Fibroblasts. Int. Arch. Allergy Immunol. 1999, 120, 19–23. [Google Scholar] [CrossRef]
- Mochizuki, M.; Bartels, J.; Mallet, I.A.; Christophers, E.; Schröder, J.M. IL-4 induces eotaxin: A possible mechanism of selective eosinophil recruitment in helminth infection and atopy. J. Immunol. 1998, 160, 60–68. [Google Scholar]
- Zimmermann, N.; Hogan, S.P.; Mishra, A.; Brandt, E.B.; Bodette, T.R.; Pope, S.M.; Finkelman, F.D.; Rothenberg, M.E. Murine Eotaxin-2: A Constitutive Eosinophil Chemokine Induced by Allergen Challenge and IL-4 Overexpression. J. Immunol. 2000, 165, 5839–5846. [Google Scholar] [CrossRef]
- Shinkai, A.; Yoshisue, H.; Koike, M.; Shoji, E.; Nakagawa, S.; Saito, A.; Takeda, T.; Imabeppu, S.; Kato, Y.; Hanai, N.; et al. A novel human CC chemokine, eotaxin-3, which is expressed in IL-4-stimulated vascular endothelial cells, exhibits potent activity toward eosinophils. J. Immunol. 1999, 163, 1602–1610. [Google Scholar] [PubMed]
- Van Panhuys, N.; Prout, M.; Forbes, E.; Min, B.; Paul, W.E.; Le Gros, G. Basophils Are the Major Producers of IL-4 during Primary Helminth Infection. J. Immunol. 2011, 186, 2719–2728. [Google Scholar] [CrossRef]
- Min, B.; Prout, M.; Hu-Li, J.; Zhu, J.; Jankovic, D.; Morgan, E.S.; Urban, J.F.; Dvorak, A.M.; Finkelman, F.D.; Legros, G.; et al. Basophils Produce IL-4 and Accumulate in Tissues after Infection with a Th2-inducing Parasite. J. Exp. Med. 2004, 200, 507–517. [Google Scholar] [CrossRef] [PubMed]
- Voehringer, D.; Shinkai, K.; Locksley, R.M. Type 2 Immunity Reflects Orchestrated Recruitment of Cells Committed to IL-4 Production. Immunology 2004, 20, 267–277. [Google Scholar] [CrossRef]
- Nouri-Aria, K.T.; Irani, A.-M.A.; Jacobson, M.R.; O’Brien, F.; Varga, E.M.; Till, S.J.; Durham, S.R.; Schwartz, L.B. Basophil recruitment and IL-4 production during human allergen-induced late asthma. J. Allergy Clin. Immunol. 2001, 108, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Hoeck, J.; Woisetschläger, M. Activation of Eotaxin-3/CCL26 Gene Expression in Human Dermal Fibroblasts Is Mediated by STAT6. J. Immunol. 2001, 167, 3216–3222. [Google Scholar] [CrossRef] [PubMed]
- Hoeck, J.; Woisetschläger, M. STAT6 Mediates Eotaxin-1 Expression in IL-4 or TNF-α-Induced Fibroblasts. J. Immunol. 2001, 166, 4507–4515. [Google Scholar] [CrossRef]
- Ochensberger, B.; Daepp, G.C.; Rihs, S. Dahinden Human Blood Basophils Produce Interleukin-13 in Response to IgE-Receptor-Dependent and -Independent Activation. Blood 1996, 88, 3028–3037. [Google Scholar] [CrossRef]
- Gibbs, B.F.; Haas, H.; Falcone, F.H.; Albrecht, C.; Vollrath, I.B.; Noll, T.; Wolff, H.H.; Amon, U. Purified human peripheral blood basophils release interleukin-13 and preformed interleukin-4 following immunological activation. Eur. J. Immunol. 1996, 26, 2493–2498. [Google Scholar] [CrossRef]
- Rot, A.; Krieger, M.; Brunner, T.; Bischoff, S.C.; Schall, T.J.; Dahinden, A.C. RANTES and macrophage inflammatory protein 1 alpha induce the migration and activation of normal human eosinophil granulocytes. J. Exp. Med. 1992, 176, 1489–1495. [Google Scholar] [CrossRef] [PubMed]
- Brunner, T.; Heusser, C.H. Dahinden Human Peripheral Blood Basophils Primed by Interleukin 3 (IL-3) Produce IL-4 in Response to Immunoglobulin E Receptor Stimulation. J. Exp. Med. 1993, 177, 605–611. [Google Scholar] [CrossRef]
- Hagmann, B.R.; Odermatt, A.; Kaufmann, T.; Dahinden, C.A.; Fux, M.; Hagmann, B.R. Balance between IL-3 and type Iinterferons and their interrelationship with FasL dictates lifespan and effector functions of human basophils. Clin. Exp. Allergy 2017, 47, 71–84. [Google Scholar] [CrossRef] [PubMed]
- Sehmi, R.; Cromwell, O.; Wardlaw, A.J.; Moqbel, R.; Kay, A.B. Interleukin-8 is a chemo-attractant for eosinophils purified from subjects with a blood eosinophilia but not from normal healthy subjects. Clin. Exp. Allergy 1993, 23, 1027–1036. [Google Scholar] [CrossRef] [PubMed]
- Karasuyama, H.; Miyake, K.; Yoshikawa, S.; Kawano, Y.; Yamanishi, Y. How do basophils contribute to Th2 cell differentiation and allergic responses? Int. Immunol. 2018, 30, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Tschopp, C.M.; Spiegl, N.; Didichenko, S.; Lutmann, W.; Julius, P.; Virchow, J.C.; Hack, C.E.; Dahinden, C.A. Granzyme B, a novel mediator of allergic inflammation: Its induction and release in blood basophils and human asthma. Blood 2006, 108, 2290–2299. [Google Scholar] [CrossRef] [PubMed]
- Landolina, N.; Gangwar, R.S.; Levi-Schaffer, F. Mast Cells’ Integrated Actions with Eosinophils and Fibroblasts in Allergic Inflammation. Adv. Immunol. 2015, 125, 41–85. [Google Scholar] [CrossRef]
- Jiao, D.; Wong, C.-K.; Qiu, H.-N.; Dong, J.; Cai, Z.; Chu, M.; Hon, K.-L.; Tsang, M.S.-M.; Lam, C.W.-K. NOD2 and TLR2 ligands trigger the activation of basophils and eosinophils by interacting with dermal fibroblasts in atopic dermatitis-like skin inflammation. Cell. Mol. Immunol. 2015, 13, 535–550. [Google Scholar] [CrossRef]
- Schroeder, J.T.; Bieneman, A.P. Activation of Human Basophils by A549 Lung Epithelial Cells Reveals a Novel IgE-Dependent Response Independent of Allergen. J. Immunol. 2017, 199, 855–865. [Google Scholar] [CrossRef]
- Yoshimoto, T. Basophils as Th2-inducing antigen-presenting cells. Int. Immunol. 2010, 22, 543–550. [Google Scholar] [CrossRef]
- Kim, S.; Shen, T.; Min, B. Basophils Can Directly Present or Cross-Present Antigen to CD8 Lymphocytes and Alter CD8 T Cell Differentiation into IL-10-Producing Phenotypes. J. Immunol. 2009, 183, 3033–3039. [Google Scholar] [CrossRef] [PubMed]
- Voskamp, A.L.; Prickett, S.R.; Mackay, F.; Rolland, J.M.; O’Hehir, R.E. MHC Class II Expression in Human Basophils: Induction and Lack of Functional Significance. PLoS ONE 2013, 8, e81777. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, B.M.; Liang, H.-E.; Bando, J.K.; Wu, D.; Cheng, L.E.; McKerrow, J.K.; Allen, C.D.C.; Locksley, R.M. Genetic analysis of basophil function in vivo. Nat. Immunol. 2011, 12, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Wakahara, K.; Van, V.Q.; Baba, N.; Bégin, P.; Rubio, M.; Delespesse, G.; Sarfati, M. Basophils are recruited to inflamed lungs and exacerbate memory Th2 responses in mice and humans. Allergy 2012, 68, 180–189. [Google Scholar] [CrossRef]
- Sarfati, M.; Wakahara, K.; Chapuy, L.; Delespesse, G. Mutual Interaction of Basophils and T Cells in Chronic Inflammatory Diseases. Front. Immunol. 2015, 6, 399. [Google Scholar] [CrossRef] [PubMed]
- Jogdand, P.; Siddhuraj, P.; Mori, M.; Sanden, C.; Jönsson, J.; Walls, A.F.; Kearley, J.; Humbles, A.A.; Kolbeck, R.; Bjermer, L.; et al. Eosinophils, basophils and type 2 immune microenvironments in COPD-affected lung tissue. Eur. Respir. J. 2020, 55, 1900110. [Google Scholar] [CrossRef]
- Davoine, F.; Lacy, P. Eosinophil Cytokines, Chemokines, and Growth Factors: Emerging Roles in Immunity. Front. Immunol. 2014, 5, 570. [Google Scholar] [CrossRef]
- Simon, H.-U.; Yousefi, S.; Germic, N.; Arnold, I.C.; Haczku, A.; Karaulov, A.V.; Simon, D.; Rosenberg, H.F. The Cellular Functions of Eosinophils: Collegium Internationale Allergologicum (CIA) Update 2020. Int. Arch. Allergy Immunol. 2019, 181, 11–23. [Google Scholar] [CrossRef]
- Büttner, C.; Lun, A.; Splettstoesser, T.; Kunkel, G.; Renz, H. Monoclonal anti-interleukin-5 treatment suppresses eosinophil but not T-cell functions. Eur. Respir. J. 2003, 21, 799–803. [Google Scholar] [CrossRef]
- Wright, A.K.A.; Diver, S.; McCarthy, J.; Marvin, A.; Soares, M.; Thornton, T.; Bourne, M.; Craner, M.; Evans, H.; Edwards, S.; et al. Mepolizumab does not alter the blood basophil count in severe asthma. Allergy 2019, 74, 2488–2490. [Google Scholar] [CrossRef]
- Kips, J.C.; O’Connor, B.J.; Langley, S.J.; Woodcock, A.; Kerstjens, H.A.M.; Postma, D.S.; Danzig, M.; Cuss, F.; Pauwels, R.A. Effect of SCH55700, a Humanized Anti-Human Interleukin-5 Antibody, in Severe Persistent Asthma. Am. J. Respir. Crit. Care Med. 2003, 167, 1655–1659. [Google Scholar] [CrossRef]
- Roufosse, F. Targeting the Interleukin-5 Pathway for Treatment of Eosinophilic Conditions Other than Asthma. Front. Med. 2018, 5, 49. [Google Scholar] [CrossRef]
- Laviolette, M.; Gossage, D.L.; Gauvreau, G.; Leigh, R.; Olivenstein, R.; Katial, R.; Busse, W.W.; Wenzel, S.; Wu, Y.; Datta, V.; et al. Effects of benralizumab on airway eosinophils in asthmatic patients with sputum eosinophilia. J. Allergy Clin. Immunol. 2013, 132, 1086–1096.e5. [Google Scholar] [CrossRef] [PubMed]
- Herzog, E.; Busfield, S.; Biondo, M.; Vairo, G.; Dewitte, M.; Pragst, I.; Dickneite, G.; Nash, A.; Zollner, S. Pharmacodynamic Activity and Preclinical Safety of CSL362, a Novel Humanised, Affinity Matured Monoclonal Antibody Against Human Interleukin 3 Receptor. Blood 2012, 120, 1524. [Google Scholar] [CrossRef]
- Pecaric-Petkovic, T.; Didichenko, S.A.; Kaempfer, S.; Spiegl, N.; Dahinden, C.A. Human basophils and eosinophils are the direct target leukocytes of the novel IL-1 family member IL-33. Blood 2009, 113, 1526–1534. [Google Scholar] [CrossRef]
- Porsbjerg, C.M.; Sverrild, A.; Lloyd, C.M.; Menzies-Gow, A.N.; Bel, E.H. Anti-alarmins in asthma: Targeting the airway epithelium with next-generation biologics. Eur. Respir. J. 2020, 56, 2000260. [Google Scholar] [CrossRef]
- Lee, H.Y.; Rhee, C.K.; Kang, J.Y.; Byun, J.H.; Choi, J.Y.; Kim, S.J.; Kim, Y.K.; Kwon, S.S.; Lee, S.Y. Blockade of IL-33/ST2 ameliorates airway inflammation in a murine model of allergic asthma. Exp. Lung Res. 2014, 40, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Allinne, J.; Scott, G.; Lim, W.K.; Birchard, D.; Erjefält, J.S.; Sandén, C.; Ben, L.-H.; Agrawal, A.; Kaur, N.; Kim, J.H.; et al. IL-33 blockade affects mediators of persistence and exacerbation in a model of chronic airway inflammation. J. Allergy Clin. Immunol. 2019, 144, 1624–1637.e10. [Google Scholar] [CrossRef]
- Fulkerson, P.C.; Fischetti, C.A.; McBride, M.L.; Hassman, L.M.; Hogan, S.P.; Rothenberg, M.E. A central regulatory role for eosinophils and the eotaxin/CCR3 axis in chronic experimental allergic airway inflammation. Proc. Natl. Acad. Sci. USA 2006, 103, 16418–16423. [Google Scholar] [CrossRef] [PubMed]
- Gauvreau, G.M.; Fitzgerald, J.M.; Boulet, L.P.; Watson, R.M.; Hui, L.; Villineuve, H.; Scime, T.X.; Schlatman, A.R.; Obminski, C.; Kum, J.; et al. The effects of a CCR3 inhibitor, AXP1275, on allergen-induced airway responses in adults with mild-to-moderate atopic asthma. Clin. Exp. Allergy 2018, 48, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Grozdanovic, M.; Laffey, K.G.; Abdelkarim, H.; Hitchinson, B.; Harijith, A.; Moon, H.-G.; Park, G.Y.; Rousslang, L.K.; Masterson, J.C.; Furuta, G.T.; et al. Novel peptide nanoparticle–biased antagonist of CCR3 blocks eosinophil recruitment and airway hyperresponsiveness. J. Allergy Clin. Immunol. 2019, 143, 669–680.e12. [Google Scholar] [CrossRef] [PubMed]
- Royer, J.F.; Schratl, P.; Carrillo, J.J.; Jupp, R.; Barker, J.; Weyman-Jones, C.; Beri, R.; Sargent, C.; Schmidt, J.A.; Lang-Loidolt, D.; et al. A novel antagonist of prostaglandin D2blocks the locomotion of eosinophils and basophils. Eur. J. Clin. Investig. 2008, 38, 663–671. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, S.; Ford, L.; Pearlman, D.; Spector, S.; Sher, L.; Skobieranda, F.; Wang, L.; Kirkesseli, S.; Rocklin, R.; Bock, B.; et al. Dupilumab in Persistent Asthma with Elevated Eosinophil Levels. N. Engl. J. Med. 2013, 368, 2455–2466. [Google Scholar] [CrossRef] [PubMed]
- Menzella, F.; Galeone, C.; Lusuardi, M.; Simonazzi, A.; Castagnetti, C.; Ruggiero, P.; Facciolongo, N. Near-fatal asthma responsive to mepolizumab after failure of omalizumab and bronchial thermoplasty. Ther. Clin. Risk Manag. 2017, ume 13, 1489–1493. [Google Scholar] [CrossRef]
- Fulkerson, P.C.; Rothenberg, M.E. Targeting eosinophils in allergy, inflammation and beyond. Nat. Rev. Drug Discov. 2013, 12, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.; Corren, J.; Pavord, I.D.; Maspero, J.; Wenzel, S.; Rabe, K.F.; Busse, W.W.; Ford, L.; Sher, L.; Fitzgerald, J.M.; et al. Dupilumab Efficacy and Safety in Moderate-to-Severe Uncontrolled Asthma. N. Engl. J. Med. 2018, 378, 2486–2496. [Google Scholar] [CrossRef]
- Beck, L.A.; Thaçi, D.; Hamilton, J.D.; Graham, N.M.; Bieber, T.; Rocklin, R.; Ming, J.E.; Ren, H.; Kao, R.; Simpson, E.; et al. Dupilumab Treatment in Adults with Moderate-to-Severe Atopic Dermatitis. N. Engl. J. Med. 2014, 371, 130–139. [Google Scholar] [CrossRef]
- De Wijs, L.; Bosma, A.; Erler, N.; Hollestein, L.; Gerbens, L.; Middelkamp-Hup, M.; Kunkeler, A.; Nijsten, T.; Spuls, P.; Hijnen, D. Effectiveness of dupilumab treatment in 95 patients with atopic dermatitis: Daily practice data. Br. J. Dermatol. 2020, 182, 418–426. [Google Scholar] [CrossRef]
- Yamauchi, T.; Sasaki, S.; Lee, E.S.; Tamura, T.; Seki, M.; Miwa, T.; Kobayashi, K.; Saruta, Y.; Kitami, Y.; Sueki, H.; et al. Dupilumab treatment ameliorates clinical and hematological symptoms, including blood eosinophilia, in patients with atopic dermatitis. Int. J. Dermatol. 2020, 60, 190–195. [Google Scholar] [CrossRef]
- Rohner, L.; Reinhart, R.; Hagmann, B.; Odermatt, A.; Babirye, A.; Kaufmann, T.; Fux, M. FcεRI cross-linking and IL-3 protect human basophils from intrinsic apoptotic stress. J. Allergy Clin. Immunol. 2018, 142, 1647–1650.e3. [Google Scholar] [CrossRef] [PubMed]
- Rohner, L.; Reinhart, R.; Iype, J.; Bachmann, S.; Kaufmann, T.; Fux, M. Impact of BH3-mimetics on Human and Mouse Blood Leukocytes: A Comparative Study. Sci. Rep. 2020, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Souers, A.J.; Leverson, J.D.; Boghaert, E.R.; Ackler, S.L.; Catron, N.D.; Chen, J.; Dayton, B.D.; Ding, H.; Enschede, S.H.; Fairbrother, W.J.; et al. ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat. Med. 2013, 19, 202–208. [Google Scholar] [CrossRef] [PubMed]
| Model | Sensitization | Challenge | Way of Basophil Assessment | Diminished Eosinophil Infiltration | Reference |
|---|---|---|---|---|---|
| IgE-CAI | i.v. TNP-specific IgE | s.q. TNP-OVA | CD49bpos cell fraction | Yes | [32] |
| Anti-CD200R3 (B103) | Yes | [33] | |||
| Mcpt8Cre BAC | Yes | [34] | |||
| BasoDTR mice | Yes | [35] | |||
| ICD | i.p NAC | NAC and croton oil on skin | Anti-FcεRIα (MAR-1) | Yes | [36] |
| BasTRECK | Yes | [36] |
| Model | Sensitization | Challenge | Way of Basophil Assessment | Diminished Eosinophil Infiltration | Reference |
|---|---|---|---|---|---|
| OVA-induced asthma | i.p. OVA-alum | i.t. OVA | Anti-FcεRIα (MAR-1) | No | [37] |
| i.p. OVA-alum | i.n. OVA | Mcpt8Cre BAC | No | [34] | |
| Anti-FcεRIα (MAR-1) | Yes | [38] | |||
| Anti-CD200R3 (B103) | Yes | [38] | |||
| HDM-induced asthma | i.n. HDM | i.n. HDM | Anti-FcεRIα (MAR-1) | Yes | [39] |
| i.n. HDM | i.n. HDM | Anti-CD200R3 (B103) | Yes | [39] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iype, J.; Fux, M. Basophils Orchestrating Eosinophils’ Chemotaxis and Function in Allergic Inflammation. Cells 2021, 10, 895. https://doi.org/10.3390/cells10040895
Iype J, Fux M. Basophils Orchestrating Eosinophils’ Chemotaxis and Function in Allergic Inflammation. Cells. 2021; 10(4):895. https://doi.org/10.3390/cells10040895
Chicago/Turabian StyleIype, Joseena, and Michaela Fux. 2021. "Basophils Orchestrating Eosinophils’ Chemotaxis and Function in Allergic Inflammation" Cells 10, no. 4: 895. https://doi.org/10.3390/cells10040895
APA StyleIype, J., & Fux, M. (2021). Basophils Orchestrating Eosinophils’ Chemotaxis and Function in Allergic Inflammation. Cells, 10(4), 895. https://doi.org/10.3390/cells10040895





