Molecular Biomarkers for Adrenoleukodystrophy: An Unmet Need
Abstract
:1. Introduction
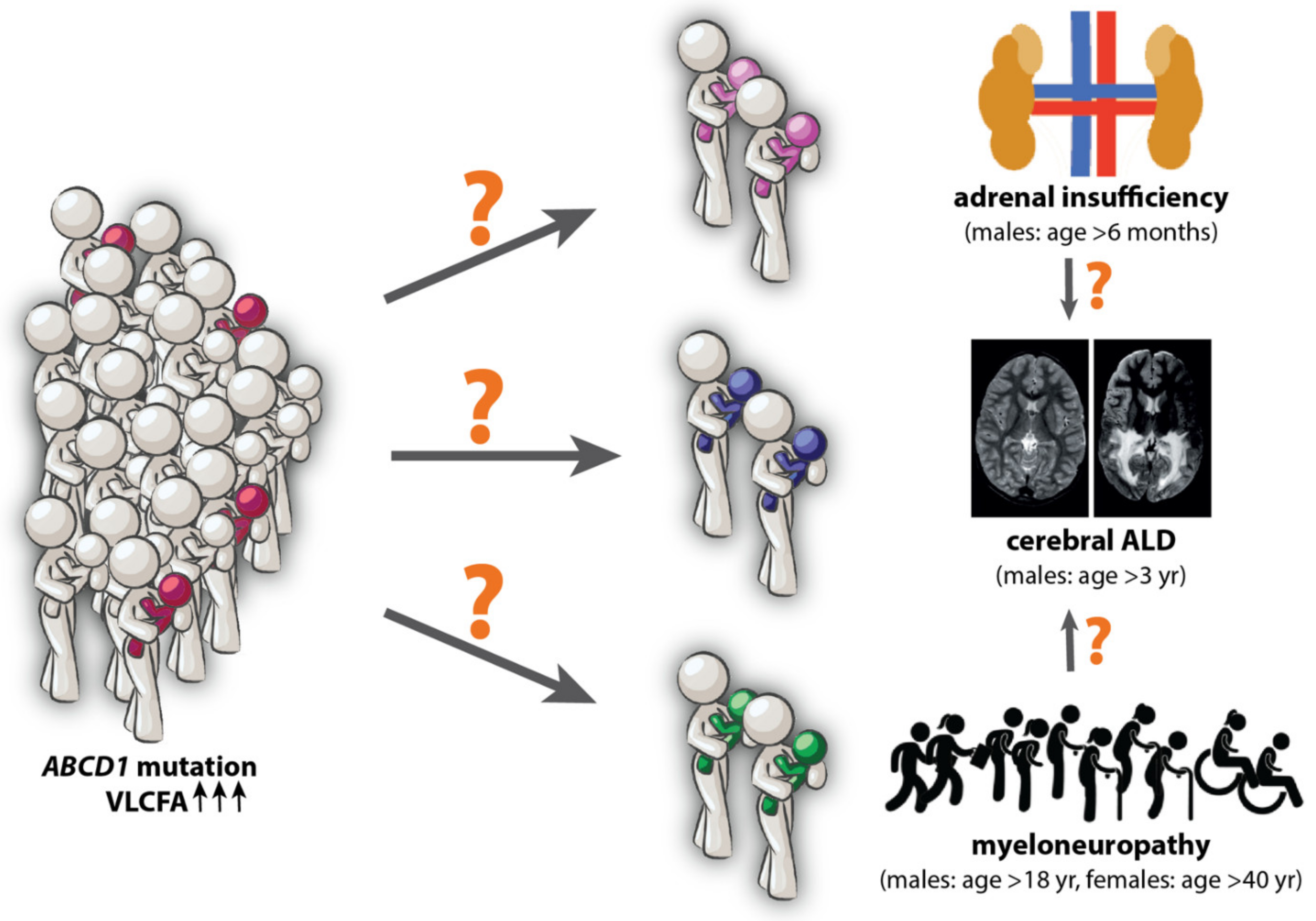
2. Molecular Biomarkers for ALD: An Unmet Need
- (1)
- As a diagnostic marker to identify the onset of leukodystrophy in patients with ALD. Currently, an MRI of the brain is used to detect white matter lesions in an asymptomatic stage so that treatment (hematopoietic cell transplant; HCT) is still an option [18,19]. However, an MRI of the brain is relatively costly and for young children invasive because of the need for general anesthesia.
- (2)
- As a predictive marker to identify individuals at high risk for cerebral ALD before its onset, particularly with respect to newborn screening positives [20,21]. Currently, all male ALD patients undergo the same intensive follow-up [22,23,24]. However, only an estimated 40% will develop cerebral ALD before the age of 18 years. If markers were identified to stratify patients in “high” and “low” risk groups for the development of cerebral ALD, personalized follow-up could be offered.
- (3)
- As a prognostic biomarker to predict fast versus slow progression of myelopathy is needed as this would allow for: (1) better counselling of individual patients and (2) for the stratification of patients in clinical trials.
- (4)
- As a disease monitoring biomarker that correlates with disease severity (for example the myelopathy) and is dynamic (i.e., changes within a reasonable timeframe as the disease progresses). This would be extremely useful as a surrogate.
3. Setting the Stage
4. Biomarker Studies So Far
4.1. Very Long-Chain Fatty Acids
4.2. Complex Lipids and Lysophosphatidylcholine (lysoPC)
4.3. Oxidative Damage Markers
4.4. Inflammatory Markers
4.5. Neuro-Axonal and Astroglial Injury Markers
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Moser, A.B.; Jones, R.O.; Hubbard, W.C.; Tortorelli, S.; Orsini, J.; Caggana, M.; Vogel, B.H.; Raymond, G.V. Newborn screening for X-linked adrenoleukodystrophy. Int. J. Neonatal. Screen. 2016, 2, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Mosser, J.; Douar, A.M.; Sarde, C.O.; Kioschis, P.; Feil, R.; Moser, H.; Poustka, A.M.; Mandel, J.L.; Aubourg, P. Putative X-linked adrenoleukodystrophy gene shares unexpected homology with ABC transporters. Nature 1993, 361, 726–730. [Google Scholar] [CrossRef] [PubMed]
- Kemp, S.; Theodoulou, F.L.; Wanders, R.J. Mammalian peroxisomal ABC transporters: From endogenous substrates to pathology and clinical significance. Br. J. Pharmacol. 2011, 164, 1753–1766. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kemp, S.; Pujol, A.; Waterham, H.R.; van Geel, B.M.; Boehm, C.D.; Raymond, G.V.; Cutting, G.R.; Wanders, R.J.A.; Moser, H.W. ABCD1 mutations and the X-linked adrenoleukodystrophy mutation database: Role in diagnosis and clinical correlations. Hum. Mutat. 2001, 18, 499–515. [Google Scholar] [CrossRef]
- Singh, I.; Moser, H.W.; Moser, A.B.; Kishimoto, Y. Adrenoleukodystrophy: Impaired oxidation of long chain fatty acids in cultured skin fibroblasts an adrenal cortex. Biochem. Biophys. Res. Commun. 1981, 102, 1223–1229. [Google Scholar] [CrossRef]
- Igarashi, M.; Schaumburg, H.H.; Powers, J.; Kishmoto, Y.; Kolodny, E.; Suzuki, K.; Kishimoto, Y.; Koilodny, E.; Suzuki, K. Fatty acid abnormality in adrenoleukodystrophy. J. Neurochem. 1976, 26, 851–860. [Google Scholar] [CrossRef]
- Kemp, S.; Wanders, R. Biochemical aspects of X-linked adrenoleukodystrophy. Brain Pathol. 2010, 20, 831–837. [Google Scholar] [CrossRef]
- Kemp, S.; Huffnagel, I.C.; Linthorst, G.E.; Wanders, R.J.A.; Engelen, M. Adrenoleukodystrophy - Neuroendocrine pathogenesis and redefinition of natural history. Nat. Rev. Endocrinol. 2016, 12, 606–615. [Google Scholar] [CrossRef]
- Huffnagel, I.C.; Laheji, F.K.; Aziz-Bose, R.; Tritos, N.A.; Marino, R.; Linthorst, G.E.; Kemp, S.; Engelen, M.; Eichler, F. The Natural History of Adrenal Insufficiency in X-Linked Adrenoleukodystrophy: An International Collaboration. J. Clin. Endocrinol. Metab. 2019, 104, 118–126. [Google Scholar] [CrossRef] [Green Version]
- Huffnagel, I.C.; van Ballegoij, W.J.C.; van Geel, B.M.; Vos, J.M.B.W.; Kemp, S.; Engelen, M. Progression of myelopathy in males with adrenoleukodystrophy: Towards clinical trial readiness. Brain 2019, 142, 334–343. [Google Scholar] [CrossRef]
- Engelen, M.; Barbier, M.; Dijkstra, I.M.E.; Schür, R.; de Bie, R.M.A.; Verhamme, C.; Dijkgraaf, M.G.W.; Aubourg, P.A.; Wanders, R.J.A.; van Geel, B.M.; et al. X-linked adrenoleukodystrophy in women: A cross-sectional cohort study. Brain 2014, 137, 693–706. [Google Scholar] [CrossRef] [Green Version]
- Huffnagel, I.C.; Dijkgraaf, M.G.W.; Janssens, G.E.; van Weeghel, M.; van Geel, B.M.; Poll-The, B.T.; Kemp, S.; Engelen, M. Disease progression in women with X-linked adrenoleukodystrophy is slow. Orphanet J. Rare Dis. 2019, 14, 30. [Google Scholar] [CrossRef] [Green Version]
- FDA-NIH Biomarkers Definitions Working Group. Biomarker Qualification: Evidentiary Framework Guidance for Industry and FDA Staff (December 2018). Available online: https://www.fda.gov/media/122319/download. (accessed on 15 November 2021).
- Berger, J.; Molzer, B.; Fae, I.; Bernheimer, H. X-linked adrenoleukodystrophy (ALD): A novel mutation of the ALD gene in 6 members of a family presenting with 5 different phenotypes. Biochem. Biophys. Res. Commun. 1994, 205, 1638–1643. [Google Scholar] [CrossRef]
- Korenke, G.C.; Fuchs, S.; Krasemann, E.; Doerr, H.G.; Wilichowski, E.; Hunneman, D.H.; Hanefeld, F. Cerebral adrenoleukodystrophy (ALD) in only one of monozygotic twins with an identical ALD genotype. Ann. Neurol. 1996, 40, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Di Rocco, M.; Doria-Lamba, L.; Caruso, U. Monozygotic twins with X-linked adrenoleukodystrophy and different phenotypes. Ann. Neurol. 2001, 50, 424. [Google Scholar] [CrossRef] [PubMed]
- Wiesinger, C.; Eichler, F.S.; Berger, J. The genetic landscape of X-linked adrenoleukodystrophy: Inheritance, mutations, modifier genes, and diagnosis. Appl. Clin. Genet. 2015, 8, 109–121. [Google Scholar]
- Miller, W.P.; Rothman, S.M.; Nascene, D.; Kivisto, T.; DeFor, T.E.; Ziegler, R.S.; Eisengart, J.; Leiser, K.; Raymond, G.; Lund, T.C.; et al. Outcomes after allogeneic hematopoietic cell transplantation for childhood cerebral adrenoleukodystrophy: The largest single-institution cohort report. Blood 2011, 118, 1971–1978. [Google Scholar] [CrossRef]
- Pierpont, E.I.; Eisengart, J.B.; Shanley, R.; Nascene, D.; Raymond, G.V.; Shapiro, E.G.; Ziegler, R.S.; Orchard, P.J.; Miller, W.P. Neurocognitive trajectory of boys who received a hematopoietic stem cell transplant at an early stage of childhood cerebral adrenoleukodystrophy. JAMA Neurol. 2017, 74, 710–717. [Google Scholar] [CrossRef] [Green Version]
- van de Stadt, S.I.W.; Mooyer, P.A.W.; Dijkstra, I.M.E.; Dekker, C.J.M.; Vats, D.; Vera, M.; Ruzhnikov, M.R.Z.; van Haren, K.; Tang, N.; Koop, K.; et al. Biochemical Studies in Fibroblasts to Interpret Variants of Unknown Significance in the ABCD1 Gene. Genes 2021, 12, 1930. [Google Scholar] [CrossRef]
- Eichler, F.; Duncan, C.; Musolino, P.L.; Orchard, P.J.; De Oliveira, S.; Thrasher, A.J.; Armant, M.; Dansereau, C.; Lund, T.C.; Miller, W.P.; et al. Hematopoietic stem-cell gene therapy for cerebral adrenoleukodystrophy. N. Engl. J. Med. 2017, 377, 1630–1638. [Google Scholar] [CrossRef] [Green Version]
- Barendsen, R.W.; Dijkstra, I.M.E.; Visser, W.F.; Alders, M.; Bliek, J.; Boelen, A.; Bouva, M.J.; van der Crabben, S.N.; Elsinghorst, E.; van Gorp, A.G.M.; et al. Adrenoleukodystrophy Newborn Screening in the Netherlands (SCAN Study): The X-Factor. Front. Cell Dev. Biol. 2020, 8, 499. [Google Scholar] [CrossRef]
- Mallack, E.J.; Turk, B.R.; Yan, H.; Price, C.; Demetres, M.; Moser, A.B.; Becker, C.; Hollandsworth, K.; Adang, L.; Vanderver, A.; et al. MRI surveillance of boys with X-linked adrenoleukodystrophy identified by newborn screening: Meta-analysis and consensus guidelines. J. Inherit. Metab. Dis. 2021, 44, 728–739. [Google Scholar] [CrossRef]
- Engelen, M.; Kemp, S.; de Visser, M.; van Geel, B.M.; Wanders, R.J.; Aubourg, P.; Poll-The, B. X-linked adrenoleukodystrophy (X-ALD): Clinical presentation and guidelines for diagnosis, follow-up and management. Orphanet J. Rare Dis. 2012, 7, 51. [Google Scholar] [CrossRef]
- Noseworthy, J.H.; Vandervoort, M.K.; Wong, C.J.; Ebers, G.C. Interrater variability with the Expanded Disability Status Scale (EDSS) and Functional Systems (FS) in a multiple sclerosis clinical trial. The Canadian Cooperation MS Study Group. Neurology 1990, 40, 971–975. [Google Scholar] [CrossRef] [PubMed]
- Van Ballegoij, W.J.C.; Huffnagel, I.C.; van de Stadt, S.I.W.; Weinstein, H.C.; Bennebroek, C.A.M.; Engelen, M.; Verbraak, F.D. Optical coherence tomography to measure the progression of myelopathy in adrenoleukodystrophy. Ann. Clin. Transl. Neurol. 2021, 8, 1064–1072. [Google Scholar] [CrossRef]
- Van Ballegoij, W.J.C.; van de Stadt, S.I.W.; Huffnagel, I.C.; Kemp, S.; van der Knaap, M.S.; Engelen, M. Postural Body Sway as Surrogate Outcome for Myelopathy in Adrenoleukodystrophy. Front. Physiol. 2020, 11, 786. [Google Scholar] [CrossRef] [PubMed]
- Moser, H.W.; Moser, A.E.; Singh, I.; O’Neill, B.P. Adrenoleukodystrophy: Survey of 303 cases: Biochemistry, diagnosis, and therapy. Ann. Neurol. 1984, 16, 628–641. [Google Scholar] [CrossRef]
- Boles, D.J.; Craft, D.A.; Padgett, D.A.; Loria, R.M.; Rizzo, W.B. Clinical variation in X-linked adrenoleukodystrophy: Fatty acid and lipid metabolism in cultured fibroblasts. Biochem. Med. Metab. Biol. 1991, 45, 74–91. [Google Scholar] [CrossRef]
- Moser, A.B.; Kreiter, N.; Bezman, L.; Lu, S.; Raymond, G.V.; Naidu, S.; Moser, H.W. Plasma very long chain fatty acids in 3,000 peroxisome disease patients and 29,000 controls. Ann. Neurol. 1999, 45, 100–110. [Google Scholar] [CrossRef]
- Stradomska, T.J.; Tylki-Szymańska, A. Serum very-long-chain fatty acids levels determined by gas chromatography in the diagnosis of peroxisomal disorders in Poland. Folia Neuropathol. 2009, 47, 306–313. [Google Scholar]
- Antoku, Y.; Koike, F.; Ohtsuka, Y.; Sakai, T.; Tsukamoto, K.; Nagara, H.; Iwashita, H.; Goto, I. Adrenoleukodystrophy: A correlation between saturated very long-chain fatty acids in mononuclear cells and phenotype. Ann. Neurol. 1991, 30, 101–103. [Google Scholar] [CrossRef] [PubMed]
- Theda, C.; Moser, A.B.; Powers, J.M.; Moser, H.W. Phospholipids in X-linked adrenoleukodystrophy white matter: Fatty acid abnormalities before the onset of demyelination. J. Neurol. Sci. 1992, 110, 195–204. [Google Scholar] [CrossRef]
- Khan, M.; Pahan, K.; Singh, A.K.; Singh, I. Cytokine-induced accumulation of very long-chain fatty acids in rat C6 glial cells: Implication for X-adrenoleukodystrophy. J. Neurochem. 1998, 71, 78–87. [Google Scholar] [CrossRef] [Green Version]
- Berger, J.; Forss-Petter, S.; Eichler, F.S. Pathophysiology of X-linked adrenoleukodystrophy. Biochimie 2014, 98, 135–142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asheuer, M.; Bieche, I.; Laurendeau, I.; Moser, A.; Hainque, B.; Vidaud, M.; Aubourg, P. Decreased expression of ABCD4 and BG1 genes early in the pathogenesis of X-linked adrenoleukodystrophy. Hum. Mol. Genet. 2005, 14, 1293–1303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paintlia, A.S.; Gilg, A.G.; Khan, M.; Singh, A.K.; Barbosa, E.; Singh, I. Correlation of very long chain fatty acid accumulation and inflammatory disease progression in childhood X-ALD: Implications for potential therapies. Neurobiol. Dis. 2003, 14, 425–439. [Google Scholar] [CrossRef] [PubMed]
- Van de Beek, M.-C.; Ofman, R.; Dijkstra, I.; Wijburg, F.; Engelen, M.; Wanders, R.; Kemp, S. Lipid-induced endoplasmic reticulum stress in X-linked adrenoleukodystrophy. Biochim. Biophys. Acta 2017, 1863, 2255–2265. [Google Scholar] [CrossRef] [PubMed]
- Raas, Q.; van de Beek, M.-C.; Forss-Petter, S.; Dijkstra, I.M.; Deschiffart, A.; Freshner, B.C.; Stevenson, T.J.; Jaspers, Y.R.; Nagtzaam, L.; Wanders, R.J.; et al. Metabolic rerouting via SCD1 induction impacts X-linked adrenoleukodystrophy. J. Clin. Investig. 2021, 131, e142500. [Google Scholar] [CrossRef]
- Law, S.-H.; Chan, M.-L.; Marathe, G.K.; Parveen, F.; Chen, C.-H.; Ke, L.-Y. An Updated Review of Lysophosphatidylcholine Metabolism in Human Diseases. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef] [Green Version]
- Hubbard, W.C.; Moser, A.B.; Liu, A.C.; Jones, R.O.; Steinberg, S.J.; Lorey, F.; Panny, S.R.; Vogt, R.F., Jr.; Macaya, D.; Turgeon, C.T.; et al. Newborn screening for X-linked adrenoleukodystrophy (X-ALD): Validation of a combined liquid chromatography-tandem mass spectrometric (LC-MS/MS) method. Mol. Genet. Metab. 2009, 97, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, W.C.; Moser, A.B.; Tortorelli, S.; Liu, A.; Jones, D.; Moser, H. Combined liquid chromatography-tandem mass spectrometry as an analytical method for high throughput screening for X-linked adrenoleukodystrophy and other peroxisomal disorders: Preliminary findings. Mol. Genet. Metab. 2006, 89, 185–187. [Google Scholar] [CrossRef]
- Huffnagel, I.C.; van de Beek, M.-C.; Showers, A.L.; Orsini, J.J.; Klouwer, F.C.C.; Dijkstra, I.M.E.; Schielen, P.C.; van Lenthe, H.; Wanders, R.J.A.; Vaz, F.M.; et al. Comparison of C26:0-carnitine and C26:0-lysophosphatidylcholine as diagnostic markers in dried blood spots from newborns and patients with adrenoleukodystrophy. Mol. Genet. Metab. 2017, 122, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Jaspers, Y.R.J.; Ferdinandusse, S.; Dijkstra, I.M.E.; Barendsen, R.W.; van Lenthe, H.; Kulik, W.; Engelen, M.; Goorden, S.M.I.; Vaz, F.M.; Kemp, S. Comparison of the Diagnostic Performance of C26:0-Lysophosphatidylcholine and Very Long-Chain Fatty Acids Analysis for Peroxisomal Disorders. Front. Cell Dev. Biol. 2020, 8, 690. [Google Scholar] [CrossRef] [PubMed]
- Eichler, F.S.; Ren, J.Q.; Cossoy, M.; Rietsch, A.M.; Nagpal, S.; Moser, A.B.; Frosch, M.P.; Ransohoff, R.M. Is microglial apoptosis an early pathogenic change in cerebral X-linked adrenoleukodystrophy? Ann. Neurol. 2008, 63, 729–742. [Google Scholar] [CrossRef]
- Kettwig, M.; Klemp, H.; Nessler, S.; Streit, F.; Krätzner, R.; Rosewich, H.; Gärtner, J. Targeted metabolomics revealed changes in phospholipids during the development of neuroinflammation in Abcd1tm1Kds mice and X-linked adrenoleukodystrophy patients. J. Inherit. Metab. Dis. 2021. [CrossRef]
- Vaz, F.M.; Pras-Raves, M.; Bootsma, A.H.; van Kampen, A.H. Principles and practice of lipidomics. J. Inherit. Metab. Dis. 2015, 38, 41–52. [Google Scholar] [CrossRef]
- Lee, D.-K.; Long, N.P.; Jung, J.; Kim, T.J.; Na, E.; Kang, Y.P.; Kwon, S.W.; Jang, J. Integrative lipidomic and transcriptomic analysis of X-linked adrenoleukodystrophy reveals distinct lipidome signatures between adrenomyeloneuropathy and childhood cerebral adrenoleukodystrophy. Biochem. Biophys. Res. Commun. 2019, 508, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, Y.; Hama, K.; Shimozawa, N.; Yokoyama, K. Glycosphingolipids with Very Long-Chain Fatty Acids Accumulate in Fibroblasts from Adrenoleukodystrophy Patients. Int. J. Mol. Sci. 2021, 22. [Google Scholar] [CrossRef] [PubMed]
- Richmond, P.A.; van der Kloet, F.; Vaz, F.M.; Lin, D.; Uzozie, A.; Graham, E.; Kobor, M.; Mostafavi, S.; Moerland, P.D.; Lange, P.F.; et al. Multi-Omic Approach to Identify Phenotypic Modifiers Underlying Cerebral Demyelination in X-Linked Adrenoleukodystrophy. Front. Cell Dev. Biol. 2020, 8, 520. [Google Scholar] [CrossRef]
- Fourcade, S.; Lopez-Erauskin, J.; Galino, J.; Duval, C.; Naudi, A.; Jove, M.; Kemp, S.; Villarroya, F.; Ferrer, I.; Pamplona, R.; et al. Early oxidative damage underlying neurodegeneration in X-adrenoleukodystrophy. Hum. Mol. Genet. 2008, 17, 1762–1773. [Google Scholar] [CrossRef] [Green Version]
- Galino, J.; Ruiz, M.; Fourcade, S.; Schluter, A.; Lopez-Erauskin, J.; Guilera, C.; Jove, M.; Naudi, A.; Garcia-Arumi, E.; Andreu, A.L.; et al. Oxidative damage compromises energy metabolism in the axonal degeneration mouse model of X-adrenoleukodystrophy. Antioxid. Redox Signal. 2011, 15, 2095–2107. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Erauskin, J.; Fourcade, S.; Galino, J.; Ruiz, M.; Schluter, A.; Naudi, A.; Jove, M.; Portero-Otin, M.; Pamplona, R.; Ferrer, I.; et al. Antioxidants halt axonal degeneration in a mouse model of X-adrenoleukodystrophy. Ann. Neurol. 2011, 70, 84–92. [Google Scholar] [CrossRef]
- Schafer, F.Q.; Buettner, G.R. Redox environment of the cell as viewed through the redox state of the glutathione disulfide/glutathione couple. Free Radic. Biol. Med. 2001, 30, 1191–1212. [Google Scholar] [CrossRef]
- Petrillo, S.; Piemonte, F.; Pastore, A.; Tozzi, G.; Aiello, C.; Pujol, A.; Cappa, M.; Bertini, E. Glutathione imbalance in patients with X-linked adrenoleukodystrophy. Mol. Genet. Metab. 2013, 109, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Nury, T.; Zarrouk, A.; Ragot, K.; Debbabi, M.; Riedinger, J.-M.; Vejux, A.; Aubourg, P.; Lizard, G. 7-Ketocholesterol is increased in the plasma of X-ALD patients and induces peroxisomal modifications in microglial cells: Potential roles of 7-ketocholesterol in the pathophysiology of X-ALD. J. Steroid Biochem. Mol. Biol. 2017, 169, 123–136. [Google Scholar] [CrossRef]
- Sinclair, A.J. Docosahexaenoic acid and the brain- what is its role? Asia Pac. J. Clin. Nutr. 2019, 28, 675–688. [Google Scholar] [CrossRef]
- Ungurianu, A.; Zanfirescu, A.; Nițulescu, G.; Margină, D. Vitamin E beyond Its Antioxidant Label. Antioxidants 2021, 10, 634. [Google Scholar] [CrossRef] [PubMed]
- Fourcade, S.; López-Erauskin, J.; Ruiz, M.; Ferrer, I.; Pujol, A. Mitochondrial dysfunction and oxidative damage cooperatively fuel axonal degeneration in X-linked adrenoleukodystrophy. Biochimie 2014, 98, 143–149. [Google Scholar] [CrossRef]
- Casasnovas, C.; Ruiz, M.; Schlüter, A.; Naudí, A.; Fourcade, S.; Veciana, M.; Castañer, S.; Albertí, A.; Bargalló, N.; Johnson, M.; et al. Biomarker Identification, Safety, and Efficacy of High-Dose Antioxidants for Adrenomyeloneuropathy: A Phase II Pilot Study. Neurotherapeutics 2019, 16, 1167–1182. [Google Scholar] [CrossRef] [Green Version]
- Turk, B.R.; Theisen, B.E.; Nemeth, C.L.; Marx, J.S.; Shi, X.; Rosen, M.; Jones, R.O.; Moser, A.B.; Watkins, P.A.; Raymond, G.V.; et al. Antioxidant capacity and superoxide dismutase activity in adrenoleukodystrophy. JAMA Neurol. 2017, 74, 519–524. [Google Scholar] [CrossRef]
- Brose, R.D.; Avramopoulos, D.; Smith, K.D. SOD2 as a potential modifier of X-linked adrenoleukodystrophy clinical phenotypes. J. Neurol. 2012, 259, 1440–1447. [Google Scholar] [CrossRef] [PubMed]
- Powers, J.M.; Liu, Y.; Moser, A.B.; Moser, H.W. The inflammatory myelinopathy of adreno-leukodystrophy: Cells, effector molecules, and pathogenetic implications. J. Neuropathol. Exp. Neurol. 1992, 51, 630–643. [Google Scholar] [CrossRef] [PubMed]
- Marchetti, D.P.; Donida, B.; Jacques, C.E.; Deon, M.; Hauschild, T.C.; Koehler-Santos, P.; de Moura Coelho, D.; Coitinho, A.S.; Jardim, L.B.; Vargas, C.R. Inflammatory profile in X-linked adrenoleukodystrophy patients: Understanding disease progression. J. Cell. Biochem. 2018, 119, 1223–1233. [Google Scholar] [CrossRef]
- Cappa, M.; Bizzarri, C.; Petroni, A.; Carta, G.; Cordeddu, L.; Valeriani, M.; Vollono, C.; De Pasquale, L.; Blasevich, M.; Banni, S. A mixture of oleic, erucic and conjugated linoleic acids modulates cerebrospinal fluid inflammatory markers and improve somatosensorial evoked potential in X-linked adrenoleukodystrophy female carriers. J. Inherit. Metab. Dis. 2012, 35, 899–907. [Google Scholar] [CrossRef] [Green Version]
- Gaetani, L.; Paolini Paoletti, F.; Bellomo, G.; Mancini, A.; Simoni, S.; Di Filippo, M.; Parnetti, L. CSF and Blood Biomarkers in Neuroinflammatory and Neurodegenerative Diseases: Implications for Treatment. Trends Pharmacol. Sci. 2020, 41, 1023–1037. [Google Scholar] [CrossRef]
- Akira, S.; Kishimoto, T. IL-6 and NF-IL6 in acute-phase response and viral infection. Immunol. Rev. 1992, 127, 25–50. [Google Scholar] [CrossRef]
- Lund, T.C.; Stadem, P.S.; Panoskaltsis-Mortari, A.; Raymond, G.; Miller, W.P.; Tolar, J.; Orchard, P.J. Elevated cerebral spinal fluid cytokine levels in boys with cerebral adrenoleukodystrophy correlates with MRI severity. PLoS ONE 2012, 7, e32218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thibert, K.A.; Raymond, G.V.; Nascene, D.R.; Miller, W.P.; Tolar, J.; Orchard, P.J.; Lund, T.C. Cerebrospinal fluid matrix metalloproteinases are elevated in cerebral adrenoleukodystrophy and correlate with MRI severity and neurologic dysfunction. PLoS ONE 2012, 7, e50430. [Google Scholar] [CrossRef] [Green Version]
- Loes, D.J.; Hite, S.; Moser, H.; Stillman, A.E.; Shapiro, E.; Lockman, L.; Latchaw, R.E.; Krivit, W. Adrenoleukodystrophy: A scoring method for brain MR observations. AJNR Am. J. Neuroradiol. 1994, 15, 1761–1766. [Google Scholar]
- Yu, C. Lysosomal storage disorders: Sphingolipidoses. In Biomarkers in Inborn Errors of Metabolism—Clinical Aspects and Laboratory Determination; Garg, U., Smith, L.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 211–233. ISBN 9780128028964. [Google Scholar]
- Orchard, P.J.; Lund, T.; Miller, W.; Rothman, S.M.; Raymond, G.; Nascene, D.; Basso, L.; Cloyd, J.; Tolar, J. Chitotriosidase as a biomarker of cerebral adrenoleukodystrophy. J. Neuroinflamm. 2011, 8, 144. [Google Scholar] [CrossRef] [Green Version]
- Lund, T.C.; Ng, M.; Orchard, P.J.; Loes, D.J.; Raymond, G.V.; Gupta, A.; Kenny-Jung, D.; Nascene, D.R. Volume of Gadolinium Enhancement and Successful Repair of the Blood-Brain Barrier in Cerebral Adrenoleukodystrophy. Biol. Blood Marrow Transplant. 2020, 26, 1894–1899. [Google Scholar] [CrossRef]
- Orchard, P.J.; Markowski, T.W.; Higgins, L.; Raymond, G.V.; Nascene, D.R.; Miller, W.P.; Pierpont, E.I.; Lund, T.C. Association between APOE4 and biomarkers in cerebral adrenoleukodystrophy. Sci. Rep. 2019, 9, 7858. [Google Scholar] [CrossRef]
- Orchard, P.J.; Nascene, D.R.; Gupta, A.; Taisto, M.E.; Higgins, L.; Markowski, T.W.; Lund, T.C. Cerebral adrenoleukodystrophy is associated with loss of tolerance to profilin. Eur. J. Immunol. 2019, 49, 947–953. [Google Scholar] [CrossRef]
- Miller, W.P.; Mantovani, L.F.; Muzic, J.; Rykken, J.B.; Gawande, R.S.; Lund, T.C.; Shanley, R.M.; Raymond, G.V.; Orchard, P.J.; Nascene, D.R. Intensity of MRI Gadolinium Enhancement in Cerebral Adrenoleukodystrophy: A Biomarker for Inflammation and Predictor of Outcome following Transplantation in Higher Risk Patients. AJNR. Am. J. Neuroradiol. 2016, 37, 367–372. [Google Scholar] [CrossRef] [Green Version]
- Van Ballegoij, W.J.C.; van de Stadt, S.I.W.; Huffnagel, I.C.; Kemp, S.; Willemse, E.A.J.; Teunissen, C.E.; Engelen, M. Plasma NfL and GFAP as biomarkers of spinal cord degeneration in adrenoleukodystrophy. Ann. Clin. Transl. Neurol. 2020, 7, 2127–2136. [Google Scholar] [CrossRef] [PubMed]
- Weinhofer, I.; Rommer, P.; Zierfuss, B.; Altmann, P.; Foiani, M.; Heslegrave, A.; Zetterberg, H.; Gleiss, A.; Musolino, P.L.; Gong, Y.; et al. Neurofilament light chain as a potential biomarker for monitoring neurodegeneration in X-linked adrenoleukodystrophy. Nat. Commun. 2021, 12, 1816. [Google Scholar] [CrossRef] [PubMed]
- Craig-Schapiro, R.; Perrin, R.J.; Roe, C.M.; Xiong, C.; Carter, D.; Cairns, N.J.; Mintun, M.A.; Peskind, E.R.; Li, G.; Galasko, D.R.; et al. YKL-40: A novel prognostic fluid biomarker for preclinical Alzheimer’s disease. Biol. Psychiatry 2010, 68, 903–912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olsson, B.; Malmeström, C.; Basun, H.; Annas, P.; Höglund, K.; Lannfelt, L.; Andreasen, N.; Zetterberg, H.; Blennow, K. Extreme stability of chitotriosidase in cerebrospinal fluid makes it a suitable marker for microglial activation in clinical trials. J. Alzheimers Dis. 2012, 32, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Manouchehrinia, A.; Piehl, F.; Hillert, J.; Kuhle, J.; Alfredsson, L.; Olsson, T.; Kockum, I. Confounding effect of blood volume and body mass index on blood neurofilament light chain levels. Ann. Clin. Transl. Neurol. 2020, 7, 139–143. [Google Scholar] [CrossRef] [Green Version]
- Beerepoot, S.; Heijst, H.; Roos, B.; Wamelink, M.M.C.; Boelens, J.J.; Lindemans, C.A.; van Hasselt, P.M.; Jacobs, E.H.; van der Knaap, M.S.; Teunissen, C.E.; et al. Neurofilament light chain and glial fibrillary acidic protein levels in metachromatic leukodystrophy. Brain 2021, awab304. [Google Scholar] [CrossRef]
| Biomarker | Fluid | Change in ALD | Interpretation | Potential Application |
|---|---|---|---|---|
| C26:0-lysoPC | Plasma and dried blood spots | Increased in all ALD phenotypes, including ALD women with normal plasma VLCFA levels. | Data is required to investigate correlations with disease severity. | As a diagnostic biomarker in newborns, male and female ALD patients. |
| Reduced Glutathione (GSH) | Plasma | Decreased in all ALD phenotypes compared to controls. | Data is required to investigate correlations with disease severity and longitudinal changes in GSH levels. | As a disease-activity biomarker to quantify oxidative stress. |
| Monocyte Chemoattractant Protein 1 (MCP1) | CSF | Increased in cerebral ALD patients compared to controls. | MCP1 correlates with MRI severity score, chitotriosidase activity and total protein levels. Longitudinal data over the course of treatment is needed. | As a screening biomarker to detect neuroinflammation. As a prognostic biomarker to predict post-treatment outcome. |
| Total protein levels | CSF | Increased in cerebral ALD patients compared to controls. | Total protein levels strongly correlate to pre-transplant MRI severity scores and pre- and post- HSCT neurological functional scores. | As a prognostic biomarker to predict post-treatment outcome. |
| Chitotriosidase | CSF and plasma | Increased in cerebral ALD patients compared to controls. | Chitotriosidase correlates with pre- and post- HSCT neurological functional scores, post-transplant MRI severity score and the change in functional status. | As a screening biomarker to detect neuroinflammation. As a prognostic biomarker to predict post-treatment outcome. |
| Superoxide dismutase (SOD) | Plasma | SOD activity decreases in a stepwise manner for ALD phenotypes; control > myelopathy > cerebral ALD | SOD activity inversely correlates with clinical MRI severity score in cerebral ALD patients. Longitudinal data to the onset of cerebral ALD is needed. | As a disease-activity biomarker to quantify oxidative stress. As a screening biomarker to detect the onset of cerebral ALD. |
| Autoantibodies against profilin 1 (PFN1) | Plasma | Increased in boys with cerebral ALD compared to non-cerebral cases and controls. | Longitudinal data pre- and post-onset of cerebral ALD and over the course of HSCT treatment is needed. | As a screening biomarker to detect the onset of cerebral ALD. |
| Neurofilament light chain (NfL) | Plasma | Increased in ALD patients compared to controls. Increased in boys with cerebral ALD compared to non-cerebral cases and controls. | NfL correlates with clinical measures of myelopathy severity in male ALD patients, and with MRI severity score in cerebral ALD patients. | As a prognostic biomarker to predict the onset of cerebral ALD. As a treatment-responsive biomarker to reflect neuronal damage. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Honey, M.I.J.; Jaspers, Y.R.J.; Engelen, M.; Kemp, S.; Huffnagel, I.C. Molecular Biomarkers for Adrenoleukodystrophy: An Unmet Need. Cells 2021, 10, 3427. https://doi.org/10.3390/cells10123427
Honey MIJ, Jaspers YRJ, Engelen M, Kemp S, Huffnagel IC. Molecular Biomarkers for Adrenoleukodystrophy: An Unmet Need. Cells. 2021; 10(12):3427. https://doi.org/10.3390/cells10123427
Chicago/Turabian StyleHoney, Madison I. J., Yorrick R. J. Jaspers, Marc Engelen, Stephan Kemp, and Irene C. Huffnagel. 2021. "Molecular Biomarkers for Adrenoleukodystrophy: An Unmet Need" Cells 10, no. 12: 3427. https://doi.org/10.3390/cells10123427
APA StyleHoney, M. I. J., Jaspers, Y. R. J., Engelen, M., Kemp, S., & Huffnagel, I. C. (2021). Molecular Biomarkers for Adrenoleukodystrophy: An Unmet Need. Cells, 10(12), 3427. https://doi.org/10.3390/cells10123427






