Cell Type-Specific Role of RNA Nuclease SMG6 in Neurogenesis
Abstract
1. Introduction
2. Material and Methods
2.1. Mice
2.2. Immunoblot Analysis
2.3. RT-qPCR
2.4. Neurosphere Formation Assay and In Vitro Neuronal Stem Cell Differentiation
2.5. Histology, TUNEL Reaction and Immunofluorescent Staining
2.6. Microscopy and Image Analysis
2.7. RNA-Seq and Bioinformatic Analysis
2.8. Statistical Analysis
3. Results
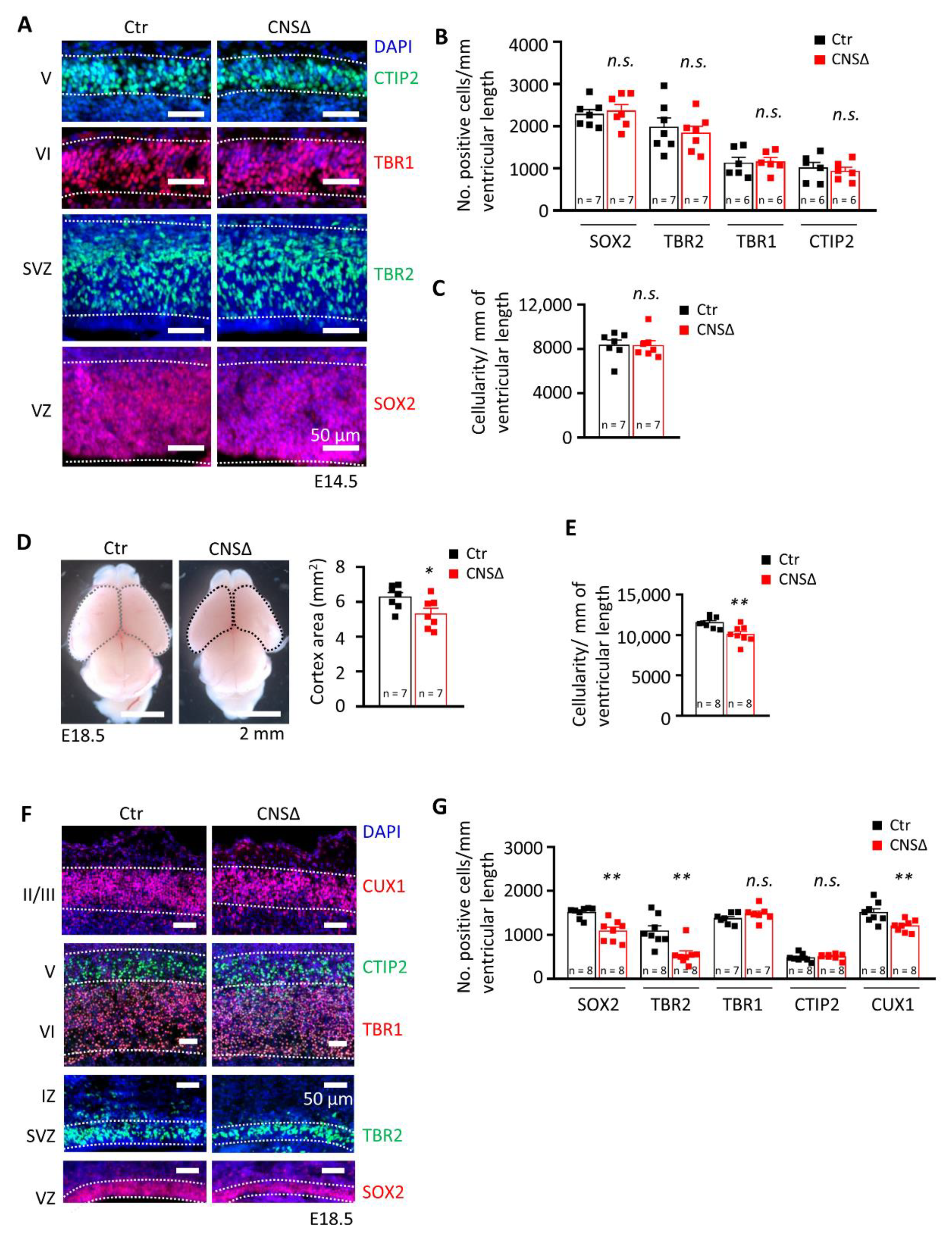
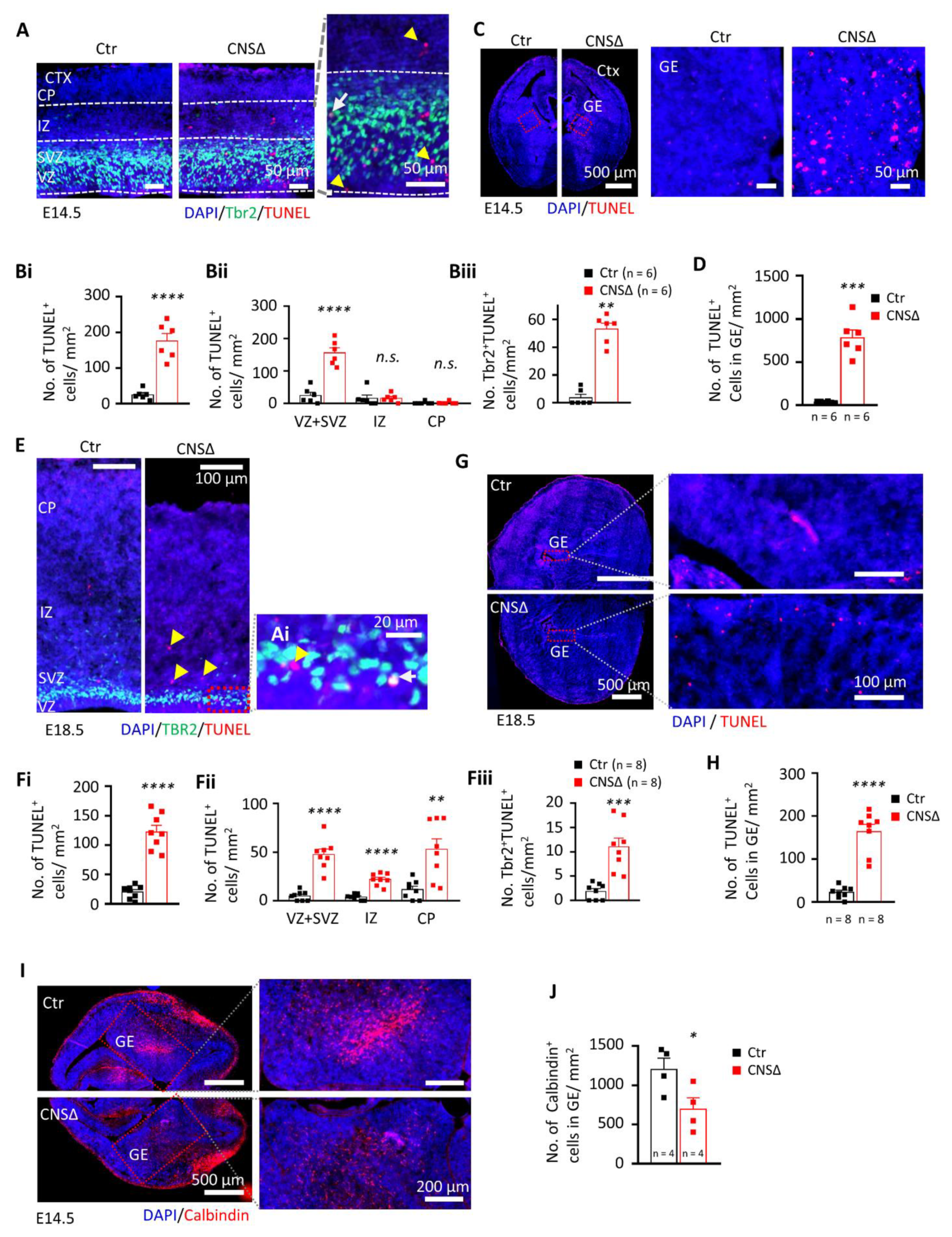
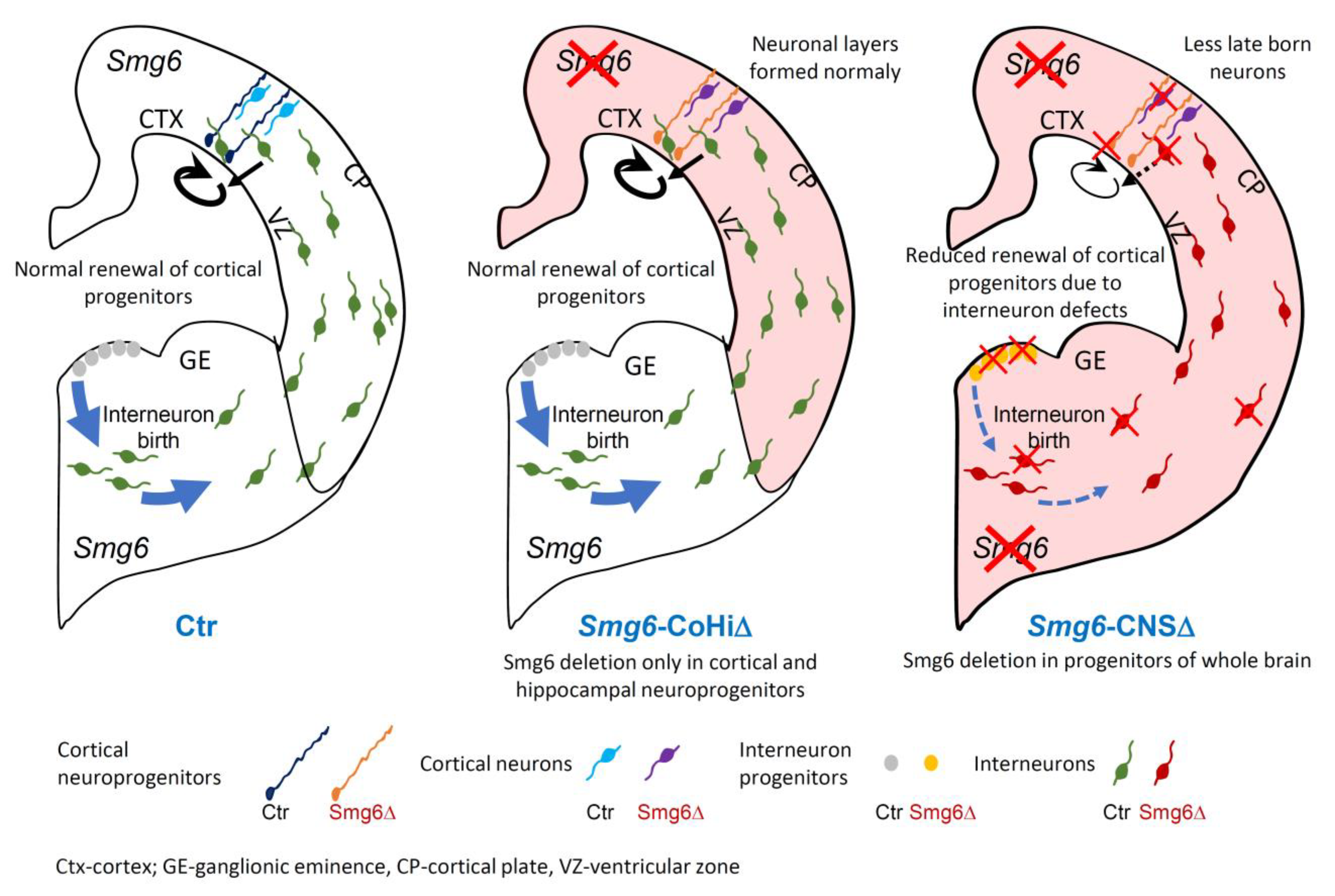
3.1. Smg6 Deletion in CNS Compromises Embryonic Neurogenesis and Newborn Viability
3.2. SMG6 Is Essential for Survival of Neural Cells
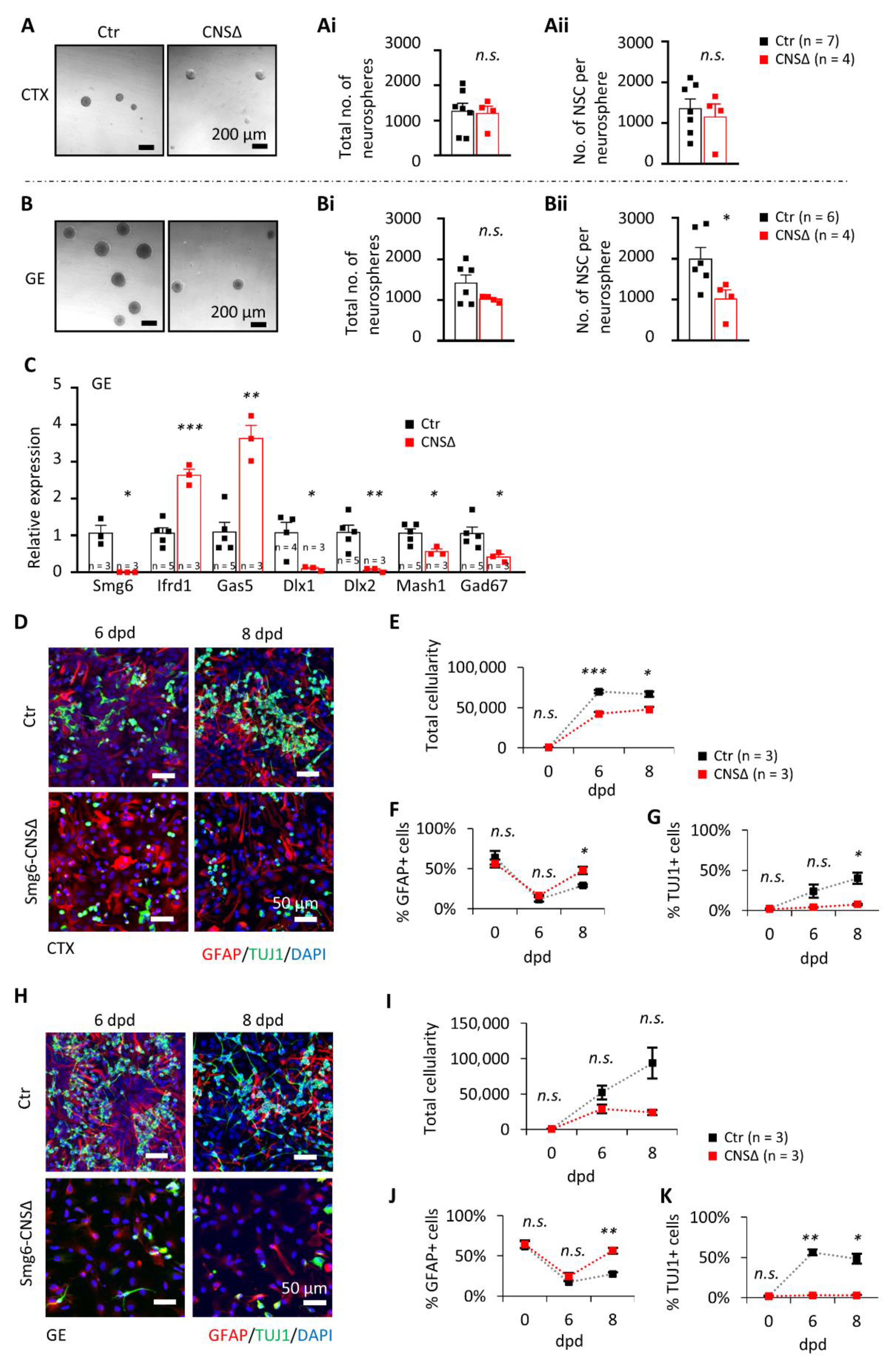
3.3. Smg6 Deletion Compromises the Self-Renewal and Differentiation Capacity of Neuroprogenitors
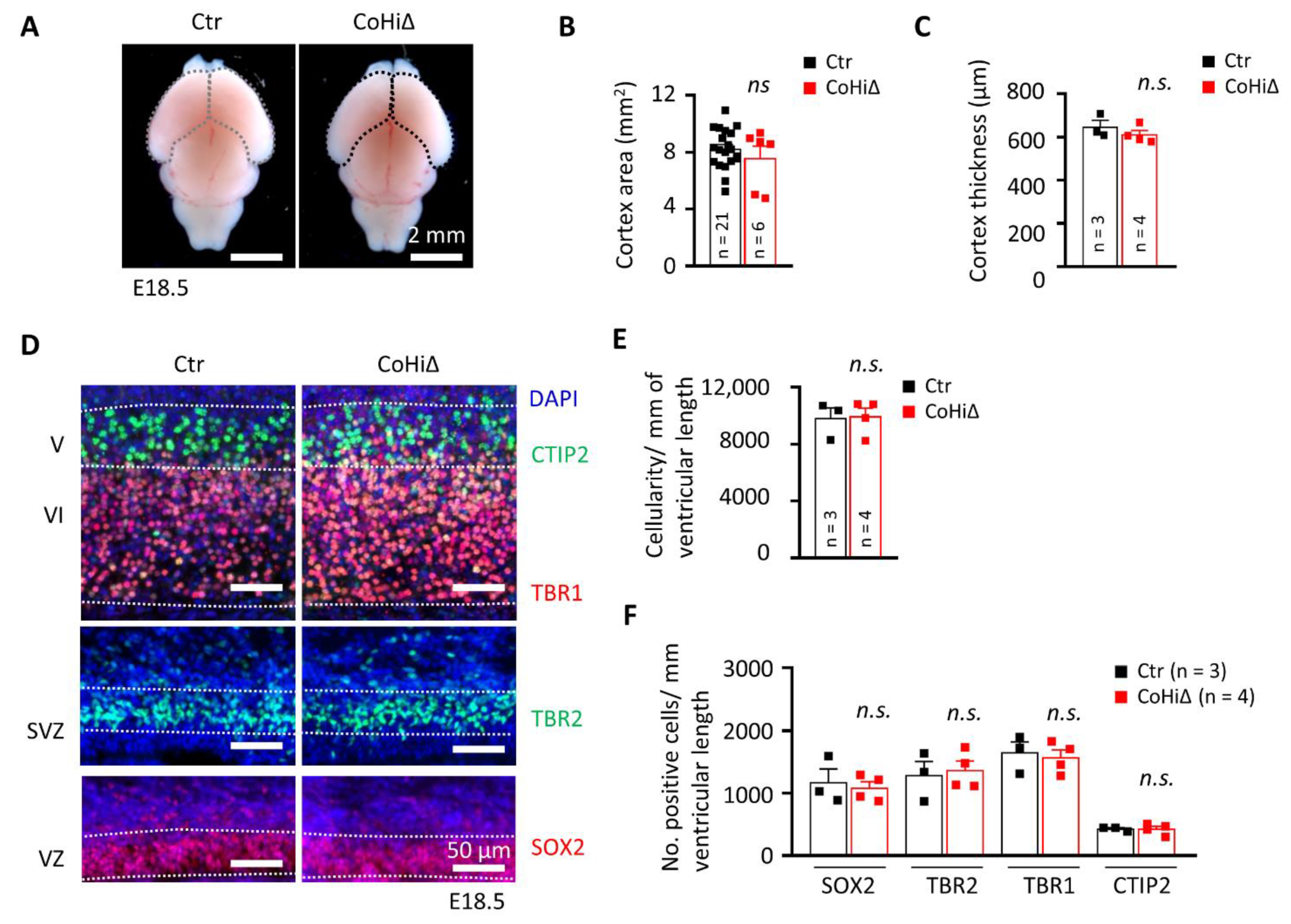
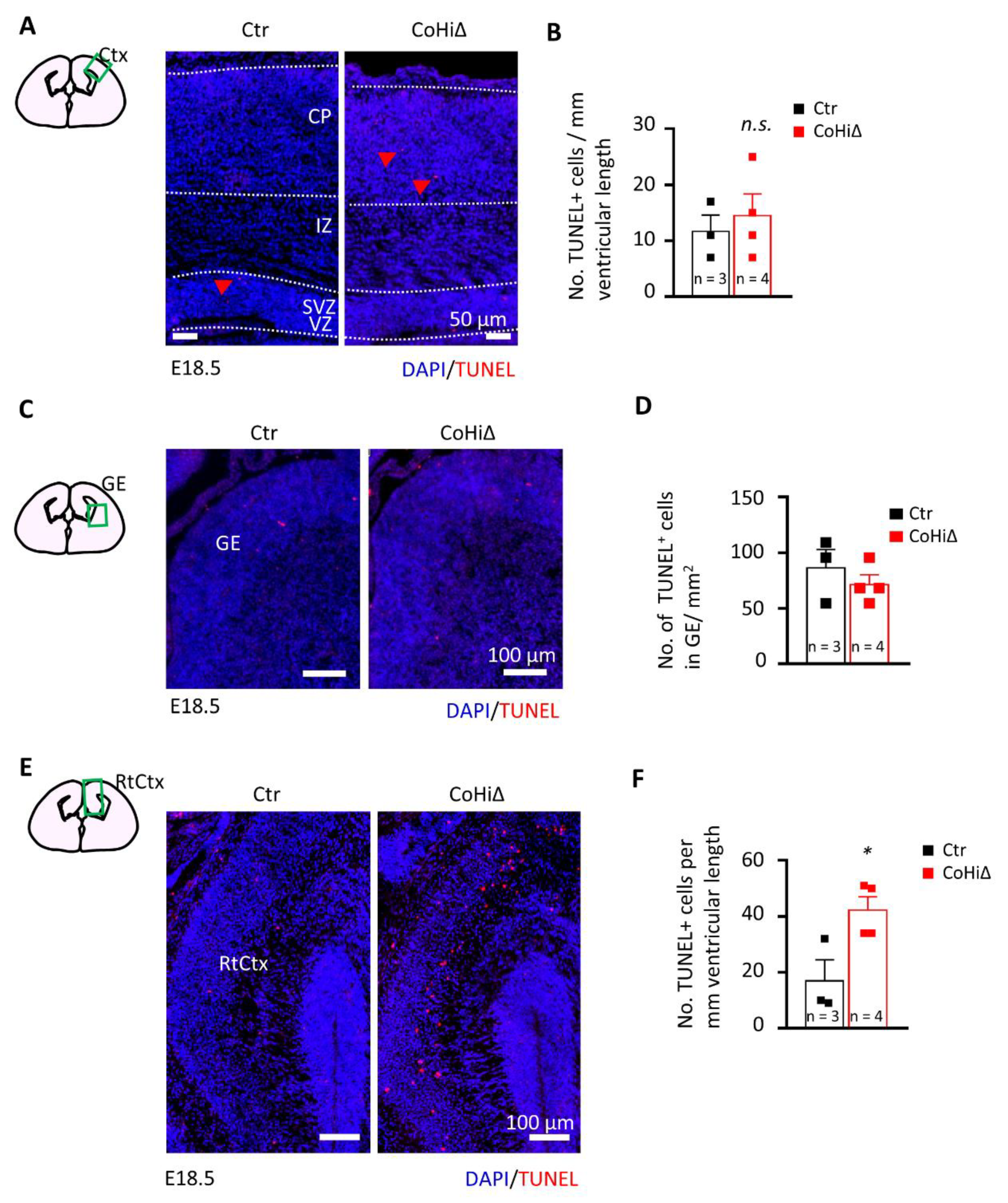
3.4. SMG6 Deficiency Exclusively in the Cortex Does Not Inhibit Corticogenesis
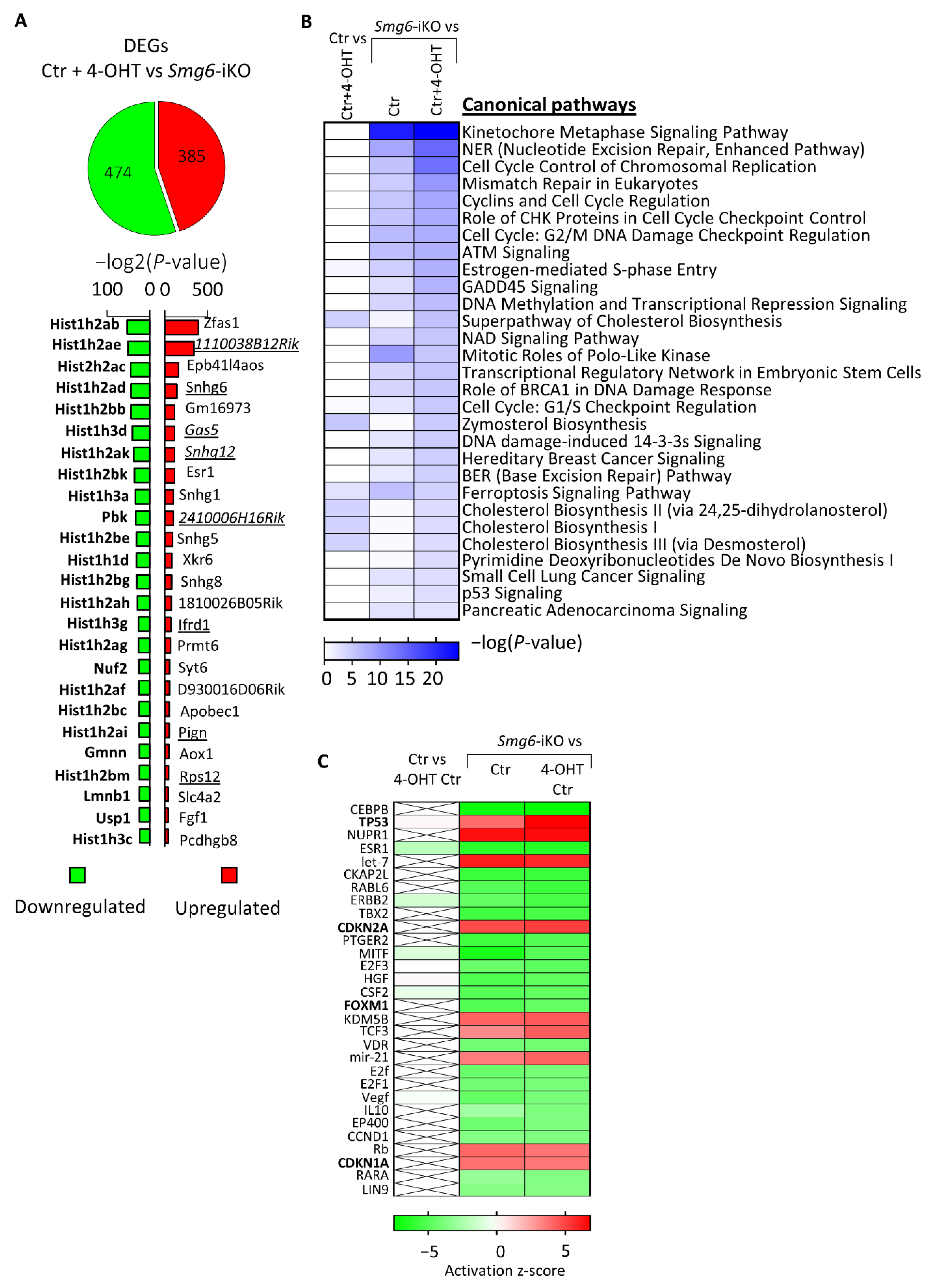
3.5. SMG6 Null Mutation Activates DNA Repair and p53 Pathways Causing Cell Cycle Dysregulation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lykke-Andersen, S.; Jensen, T.H. Nonsense-mediated mRNA decay: An intricate machinery that shapes transcriptomes. Nat. Rev. Mol. Cell Biol. 2015, 16, 665–677. [Google Scholar] [CrossRef] [PubMed]
- Palacios, I.M. Nonsense-mediated mRNA decay: From mechanistic insights to impacts on human health. Brief. Funct. Genom. 2013, 12, 25–36. [Google Scholar] [CrossRef]
- Han, X.; Wei, Y.L.; Wang, H.; Wang, F.L.; Ju, Z.Y.; Li, T.L. Nonsense-mediated mRNA decay: A ‘nonsense’ pathway makes sense in stem cell biology. Nucleic Acids Res. 2018, 46, 1038–1051. [Google Scholar] [CrossRef]
- Colombo, M.; Karousis, E.D.; Bourquin, J.; Bruggmann, R.; Muhlemann, O. Transcriptome-wide identification of NMD-targeted human mRNAs reveals extensive redundancy between SMG6-and SMG7-mediated degradation pathways. RNA 2017, 23, 189–201. [Google Scholar] [CrossRef]
- Boehm, V.; Kueckelmann, S.; Gerbracht, J.V.; Kallabis, S.; Britto-Borges, T.; Altmuller, J.; Kruger, M.; Dieterich, C.; Gehring, N.H. SMG5-SMG7 authorize nonsense-mediated mRNA decay by enabling SMG6 endonucleolytic activity. Nat. Commun. 2021, 12, 3965. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, C.; Peixeiro, I.; Romao, L. Gene Expression Regulation by Upstream Open Reading Frames and Human Disease. PLoS Genet. 2013, 9, e1003529. [Google Scholar] [CrossRef] [PubMed]
- Frischmeyer, P.A.; Dietz, H.C. Nonsense-mediated mRNA decay in health and disease. Hum. Mol. Genet. 1999, 8, 1893–1900. [Google Scholar] [CrossRef] [PubMed]
- Nickless, A.; Bailis, J.M.; You, Z.S. Control of gene expression through the nonsense-mediated RNA decay pathway. Cell Biosci. 2017, 7, 26. [Google Scholar] [CrossRef]
- Pawlicka, K.; Kalathiya, U.; Alfaro, J. Nonsense-Mediated mRNA Decay: Pathologies and the Potential for Novel Therapeutics. Cancers 2020, 12, 765. [Google Scholar] [CrossRef]
- Medghalchi, S.M.; Frischmeyer, P.A.; Mendell, J.T.; Kelly, A.G.; Lawler, A.M.; Dietz, H.C. Rent1, a trans-effector of nonsense-mediated mRNA decay, is essential for mammalian embryonic viability. Hum. Mol. Genet. 2001, 10, 99–105. [Google Scholar] [CrossRef]
- Weischenfeldt, J.; Damgaard, I.; Bryder, D.; Theilgaard-Monch, K.; Thoren, L.A.; Nielsen, F.C.; Jacobsen, S.E.W.; Nerlov, C.; Porse, B.T. NMD is essential for hematopoietic stem and progenitor cells and for eliminating by-products of programmed DNA rearrangements. Genes Dev. 2008, 22, 1381–1396. [Google Scholar] [CrossRef]
- McIlwain, D.R.; Pan, Q.; Reilly, P.T.; Elia, A.J.; McCracken, S.; Wakeham, A.C.; Itie-Youten, A.; Blencowe, B.J.; Mak, T.W. Smg1 is required for embryogenesis and regulates diverse genes via alternative splicing coupled to nonsense-mediated mRNA decay. Proc. Natl. Acad. Sci. USA 2010, 107, 12186–12191. [Google Scholar] [CrossRef]
- Li, T.L.; Shi, Y.; Wang, P.; Guachalla, L.M.; Sun, B.F.; Joerss, T.; Chen, Y.S.; Groth, M.; Krueger, A.; Platzer, M.; et al. Smg6/Est1 licenses embryonic stem cell differentiation via nonsense-mediated mRNA decay. EMBO J. 2015, 34, 1630–1647. [Google Scholar] [CrossRef] [PubMed]
- Jaffrey, S.R.; Wilkinson, M.F. Nonsense-mediated RNA decay in the brain: Emerging modulator of neural development and disease. Nat. Rev. Neurosci. 2018, 19, 715–728. [Google Scholar] [CrossRef] [PubMed]
- Addington, A.M.; Gauthier, J.; Piton, A.; Hamdan, F.F.; Raymond, A.; Gogtay, N.; Miller, R.; Tossell, J.; Bakalar, J.; Germain, G.; et al. A novel frameshift mutation in UPF3B identified in brothers affected with childhood onset schizophrenia and autism spectrum disorders. Mol. Psychiatry 2011, 16, 238–239. [Google Scholar] [CrossRef] [PubMed]
- Tarpey, P.S.; Raymond, F.L.; Nguyen, L.S.; Rodriguez, J.; Hackett, A.; Vandeleur, L.; Smith, R.; Shoubridge, C.; Edkins, S.; Stevens, C.; et al. Mutations in UPF3B, a member of the nonsense-mediated mRNA decay complex, cause syndromic and nonsyndromic mental retardation. Nat. Genet. 2007, 39, 1127–1133. [Google Scholar] [CrossRef]
- Nguyen, L.S.; Kim, H.G.; Rosenfeld, J.A.; Shen, Y.P.; Gusella, J.F.; Lacassie, Y.; Layman, L.C.; Shaffer, L.G.; Gecz, J. Contribution of copy number variants involving nonsense-mediated mRNA decay pathway genes to neuro-developmental disorders. Hum. Mol. Genet. 2013, 22, 1816–1825. [Google Scholar] [CrossRef]
- Sartor, F.; Anderson, J.; McCaig, C.; Miedzybrodzka, Z.; Muller, B. Mutation of genes controlling mRNA metabolism and protein synthesis predisposes to neurodevelopmental disorders. Biochem. Soc. Trans. 2015, 43, 1259–1265. [Google Scholar] [CrossRef]
- Shaheen, R.; Anazi, S.; Ben-Omran, T.; Seidahmed, M.Z.; Caddle, L.B.; Palmer, K.; Ali, R.; Alshidi, T.; Hagos, S.; Goodwin, L.; et al. Mutations in SMG9, Encoding an Essential Component of Nonsense-Mediated Decay Machinery, Cause a Multiple Congenital Anomaly Syndrome in Humans and Mice. Am. J. Hum. Genet. 2016, 98, 643–652. [Google Scholar] [CrossRef]
- Colak, D.; Ji, S.J.; Porse, B.T.; Jaffrey, S.R. Regulation of Axon Guidance by Compartmentalized Nonsense-Mediated mRNA Decay. Cell 2013, 153, 1252–1265. [Google Scholar] [CrossRef]
- Huang, L.; Shum, E.Y.; Jones, S.H.; Lou, C.H.; Dumdie, J.; Kim, H.; Roberts, A.J.; Jolly, L.A.; Espinoza, J.L.; Skarbrevik, D.M.; et al. A Upf3b-mutant mouse model with behavioral and neurogenesis defects. Mol. Psychiatry 2018, 23, 1773–1786. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.L.; Stoica, L.; Liu, Y.W.; Zhu, P.J.; Bhattacharya, A.; Buffington, S.A.; Huq, R.; Eissa, N.T.; Larsson, O.; Porse, B.T.; et al. Inhibition of Upf2-Dependent Nonsense-Mediated Decay Leads to Behavioral and Neurophysiological Abnormalities by Activating the Immune Response. Neuron 2019, 104, 665–679.e8. [Google Scholar] [CrossRef] [PubMed]
- Jolly, L.A.; Homan, C.C.; Jacob, R.; Barry, S.; Gecz, J. The UPF3B gene, implicated in intellectual disability, autism, ADHD and childhood onset schizophrenia regulates neural progenitor cell behaviour and neuronal outgrowth. Hum. Mol. Genet. 2013, 22, 4673–4687. [Google Scholar] [CrossRef]
- Long, A.A.; Mahapatra, C.T.; Woodruff, E.A.; Rohrbough, J.; Leung, H.T.; Shino, S.; An, L.L.; Doerge, R.W.; Metzstein, M.M.; Pak, W.L.; et al. The nonsense-mediated decay pathway maintains synapse architecture and synaptic vesicle cycle efficacy. J. Cell Sci. 2010, 123, 3303–3315. [Google Scholar] [CrossRef]
- Mooney, C.M.; Jimenez-Mateos, E.M.; Engel, T.; Mooney, C.; Diviney, M.; Veno, M.T.; Kjems, J.; Farrell, M.A.; O’Brien, D.F.; Delanty, N.; et al. RNA sequencing of synaptic and cytoplasmic Upf1-bound transcripts supports contribution of nonsense-mediated decay to epileptogenesis. Sci. Rep. 2017, 7, 41517. [Google Scholar] [CrossRef]
- Notaras, M.; Allen, M.; Longo, F.; Volk, N.; Toth, M.; Jeon, N.L.; Klann, E.; Colak, D. UPF2 leads to degradation of dendritically targeted mRNAs to regulate synaptic plasticity and cognitive function. Mol. Psychiatry 2020, 25, 3360–3379. [Google Scholar] [CrossRef]
- Tronche, F.; Kellendonk, C.; Kretz, O.; Gass, P.; Anlag, K.; Orban, P.C.; Bock, R.; Klein, R.; Schutz, G. Disruption of the glucocorticoid receptor gene in the nervous system results in reduced anxiety. Nat. Genet. 1999, 23, 99–103. [Google Scholar] [CrossRef]
- Gorski, J.A.; Talley, T.; Qiu, M.S.; Puelles, L.; Rubenstein, J.L.R.; Jones, K.R. Cortical excitatory neurons and glia, but not GABAergic neurons, are produced in the Emx1-expressing lineage. J. Neurosci. 2002, 22, 6309–6314. [Google Scholar] [CrossRef]
- Frappart, P.O.; Tong, W.M.; Demuth, I.; Radovanovic, I.; Herceg, Z.; Aguzzi, A.; Digweed, M.; Wang, Z.Q. An essential function for NBS1 in the prevention of ataxia and cerebellar defects. Nat. Med. 2005, 11, 538–544. [Google Scholar] [CrossRef]
- Park, G.; Horie, T.; Kanayama, T.; Fukasawa, K.; Iezaki, T.; Onishi, Y.; Ozaki, K.; Nakamura, Y.; Yoneda, Y.; Takarada, T.; et al. The transcriptional modulator Ifrd1 controls PGC-1alpha expression under short-term adrenergic stimulation in brown adipocytes. FEBS J. 2017, 284, 784–795. [Google Scholar] [CrossRef] [PubMed]
- Spandidos, A.; Wang, X.W.; Wang, H.J.; Seed, B. PrimerBank: A resource of human and mouse PCR primer pairs for gene expression detection and quantification. Nucleic Acids Res. 2010, 38, D792–D799. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, Q.; Yang, P.; Wang, C.; Liu, J.; Ding, W.; Liu, W.; Bai, Y.; Yang, Y.; Wang, H.; et al. LSD1 co-repressor Rcor2 orchestrates neurogenesis in the developing mouse brain. Nat. Commun. 2016, 7, 10481. [Google Scholar] [CrossRef] [PubMed]
- Kraus, P.; Sivakamasundari, V.; Lim, S.L.; Xing, X.; Lipovich, L.; Lufkin, T. Making sense of Dlx1 antisense RNA. Dev. Biol. 2013, 376, 224–235. [Google Scholar] [CrossRef][Green Version]
- Grigaravicius, P.; Kaminska, E.; Hubner, C.A.; McKinnon, P.J.; von Deimling, A.; Frappart, P.O. Rint1 inactivation triggers genomic instability, ER stress and autophagy inhibition in the brain. Cell Death Differ. 2016, 23, 454–468. [Google Scholar] [CrossRef] [PubMed]
- Gruber, R.; Zhou, Z.; Sukchev, M.; Joerss, T.; Frappart, P.O.; Wang, Z.Q. MCPH1 regulates the neuroprogenitor division mode by coupling the centrosomal cycle with mitotic entry through the Chk1-Cdc25 pathway. Nat. Cell Biol. 2011, 13, 1325–1334. [Google Scholar] [CrossRef]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef]
- Liao, Y.; Smyth, G.K.; Shi, W. featureCounts: An efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 2014, 30, 923–930. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [PubMed]
- Cauli, B.; Audinat, E.; Lambolez, B.; Angulo, M.C.; Ropert, N.; Tsuzuki, K.; Hestrin, S.; Rossier, J. Molecular and physiological diversity of cortical nonpyramidal cells. J. Neurosci. 1997, 17, 3894–3906. [Google Scholar] [CrossRef] [PubMed]
- Gonchar, Y.; Burkhalter, A. Three distinct families of GABAergic neurons in rat visual cortex. Cereb. Cortex 1997, 7, 347–358. [Google Scholar] [CrossRef]
- Kubota, Y.; Kawaguchi, Y. 3 Classes of Gabaergic Interneurons in Neocortex and Neostriatum. Jpn. J. Physiol. 1994, 44, S145–S148. [Google Scholar] [PubMed]
- Anderson, S.A.; Qiu, M.S.; Bulfone, A.; Eisenstat, D.D.; Meneses, J.; Pedersen, R.; Rubenstein, J.L.R. Mutations of the homeobox genes Dlx-1 and Dlx-2 disrupt the striatal subventricular zone and differentiation of late born striatal neurons. Neuron 1997, 19, 27–37. [Google Scholar] [CrossRef]
- Cobos, I.; Borello, U.; Rubenstein, J.L.R. Dlx transcription factors promote migration through repression of axon and dendrite growth. Neuron 2007, 54, 873–888. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.J.; Iwasaki, H.; Watanabe, M.; Okabe, S. Dlx1 transcription factor regulates dendritic growth and postsynaptic differentiation through inhibition of neuropilin-2 and PAK3 expression. Eur. J. Neurosci. 2014, 39, 531–547. [Google Scholar] [CrossRef] [PubMed]
- Le, T.N.; Du, G.Y.; Fonseca, M.; Zhou, Q.P.; Wigle, J.T.; Eisenstat, D.D. Dlx homeobox genes promote cortical interneuron migration from the basal forebrain by direct repression of the semaphorin receptor Neuropilin-2. J. Biol. Chem. 2007, 282, 19071–19081. [Google Scholar] [CrossRef]
- Long, J.E.; Garel, S.; Alvarez-Dolado, M.; Yoshikawa, K.; Osumi, N.; Alvarez-Buylla, A.; Rubenstein, J.L.R. Dlx-dependent and -independent regulation of olfactory bulb interneuron differentiation. J. Neurosci. 2007, 27, 3230–3243. [Google Scholar] [CrossRef]
- Stuhmer, T.; Puelles, L.; Ekker, M.; Rubenstein, J.L. Expression from a Dlx gene enhancer marks adult mouse cortical GABAergic neurons. Cereb. Cortex 2002, 12, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.W.; Hui, W.W.Y.; Wang, J.J.; Yung, M.M.H.; Hui, L.M.N.; Qin, Y.; Liang, R.R.; Leung, T.H.Y.; Xu, D.; Chan, K.K.L.; et al. DLX1 acts as a crucial target of FOXM1 to promote ovarian cancer aggressiveness by enhancing TGF-beta/SMAD4 signaling. Oncogene 2017, 36, 1404–1416. [Google Scholar] [CrossRef]
- Nagao, M.; Campbell, K.; Burns, K.; Kuan, C.Y.; Trumpp, A.; Nakafuku, M. Coordinated control of self-renewal and differentiation of neural stem cells by Myc and the p19(ARF)-p53 pathway. J. Cell Biol. 2008, 183, 1243–1257. [Google Scholar] [CrossRef]
- Zinin, N.; Adameyko, I.; Wilhelm, M.; Fritz, N.; Uhlen, P.; Ernfors, P.; Henriksson, M.A. MYC proteins promote neuronal differentiation by controlling the mode of progenitor cell division. EMBO Rep. 2014, 15, 383–391. [Google Scholar] [CrossRef]
- Liang, H.X.; Hippenmeyer, S.; Ghashghaei, H.T. A Nestin-cre transgenic mouse is insufficient for recombination in early embryonic neural progenitors. Biol. Open 2012, 1, 1200–1203. [Google Scholar] [CrossRef]
- Lee, Y.; Shull, E.R.P.; Frappart, P.O.; Katyal, S.; Enriquez-Rios, V.; Zhao, J.F.; Russell, H.R.; Brown, E.J.; McKinnon, P.J. ATR maintains select progenitors during nervous system development. EMBO J. 2012, 31, 1177–1189. [Google Scholar] [CrossRef] [PubMed]
- Haydar, T.F.; Wang, F.; Schwartz, M.L.; Rakic, P. Differential modulation of proliferation in the neocortical ventricular and subventricular zones. J. Neurosci. 2000, 20, 5764–5774. [Google Scholar] [CrossRef] [PubMed]
- LoTurco, J.J.; Owens, D.F.; Heath, M.J.S.; Davis, M.B.E.; Kriegstein, A.R. GABA and glutamate depolarize cortical progenitor cells and inhibit DNA synthesis. Neuron 1995, 15, 1287–1298. [Google Scholar] [CrossRef]
- McSweeney, C.; Dong, F.P.; Chen, M.; Vitale, J.; Xu, L.; Crowley, N.; Luscher, B.; Zou, D.H.; Mao, Y.W. Full function of exon junction complex factor, Rbm8a, is critical for interneuron development. Transl. Psychiatry 2020, 10, 379. [Google Scholar] [CrossRef] [PubMed]
- Qiu, M.S.; Bulfone, A.; Ghattas, I.; Meneses, J.J.; Christensen, L.; Sharpe, P.T.; Presley, R.; Pedersen, R.A.; Rubenstein, J.L.R. Role of the Dlx homeobox genes in proximodistal patterning of the branchial arches: Mutations of Dlx-1, Dlx-2, and Dlx-1 and -2 alter morphogenesis of proximal skeletal and soft tissue structures derived from the first and second arches. Dev. Biol. 1997, 185, 165–184. [Google Scholar] [CrossRef]
- Asada, H.; Kawamura, Y.; Maruyama, K.; Kume, H.; Ding, R.G.; Kanbara, N.; Kuzume, H.; Sanbo, M.; Yagi, T.; Obata, K. Cleft palate and decreased brain gamma-aminobutyric acid in mice lacking the 67-kDa isoform of glutamic acid decarboxylase. Proc. Natl. Acad. Sci. USA 1997, 94, 6496–6499. [Google Scholar] [CrossRef]
- Kash, S.F.; Tecott, L.H.; Hodge, C.; Baekkeskov, S. Increased anxiety and altered responses to anxiolytics in mice deficient in the 65-kDa isoform of glutamic acid decarboxylase. Proc. Natl. Acad. Sci. USA 1999, 96, 1698–1703. [Google Scholar] [CrossRef]
- Sussel, L.; Marin, O.; Kimura, S.; Rubenstein, J.L.R. Loss of Nkx2.1 homeobox gene function results in a ventral to dorsal molecular respecification within the basal telencephalon: Evidence for a transformation of the pallidum into the striatum. Development 1999, 126, 3359–3370. [Google Scholar] [CrossRef]
- Butt, S.J.B.; Sousa, V.H.; Fuccillo, M.V.; Hjerling-Leffler, J.; Miyoshi, G.; Kimura, S.; Fishell, G. The requirement of Nkx2-1 in the temporal specification of cortical interneuron subtypes. Neuron 2008, 59, 722–732. [Google Scholar] [CrossRef]
- Azim, E.; Jabaudon, D.; Fame, R.M.; Macklis, J.D. SOX6 controls dorsal progenitor identity and interneuron diversity during neocortical development. Nat. Neurosci. 2009, 12, 1238–1247. [Google Scholar] [CrossRef] [PubMed]
- Smits, P.; Li, P.; Mandel, J.; Zhang, Z.P.; Deng, J.M.; Behringer, R.R.; de Crombrugghe, B.; Lefebvre, V. The transcription factors L-sox5 and sox6 are essential for cartilage formation. Dev. Cell 2001, 1, 277–290. [Google Scholar] [CrossRef]
- Cowen, L.E.; Tang, Y. Identification of nonsense-mediated mRNA decay pathway as a critical regulator of p53 isoform beta. Sci. Rep. 2017, 7, 17535. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Huici, V.; Wang, B.; Gartner, A. A Role for the Nonsense-Mediated mRNA Decay Pathway in Maintaining Genome Stability in Caenorhabditis elegans. Genetics 2017, 206, 1853–1864. [Google Scholar] [CrossRef][Green Version]
- Gudikote, J.P.; Cascone, T.; Poteete, A.; Sitthideatphaiboon, P.; Wu, Q.; Morikawa, N.; Zhang, F.; Peng, S.; Tong, P.; Li, L.; et al. Inhibition of nonsense-mediated decay rescues p53beta/gamma isoform expression and activates the p53 pathway in MDM2-overexpressing and select p53-mutant cancers. J. Biol. Chem. 2021, 297, 101163. [Google Scholar] [CrossRef] [PubMed]
- Janke, R.; Kong, J.; Braberg, H.; Cantin, G.; Yates, J.R.; Krogan, N.J.; Heyer, W.D. Nonsense-mediated decay regulates key components of homologous recombination. Nucleic Acids Res. 2016, 44, 5218–5230. [Google Scholar] [CrossRef][Green Version]
- Thoren, L.A.; Norgaard, G.A.; Weischenfeldt, J.; Waage, J.; Jakobsen, J.S.; Damgaard, I.; Bergstrom, F.C.; Blom, A.M.; Borup, R.; Bisgaard, H.C.; et al. UPF2 Is a Critical Regulator of Liver Development, Function and Regeneration. PLoS ONE 2010, 5, e11650. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.; Jones, S.H.; Lake, B.B.; Dumdie, J.N.; Shum, E.Y.; Zhang, L.J.; Chen, S.; Sohni, A.; Pandya, S.; Gallo, R.L.; et al. The role of the NMD factor UPF3B in olfactory sensory neurons. eLife 2020, 9, e57525. [Google Scholar] [CrossRef]
| Transcript | Primer Sequence (5′ to 3′) | Reference |
|---|---|---|
| Smg6 | AGGAATTGGACAGCCAACAG | Li et al., 2015 [13] |
| TCTCGGTTTTATCCGGTTTG | ||
| Gas5 | TTTCCGGTCCTTCATTCTGA | Weischenfeldt et al., 2008 [11] |
| TCTTCTATTTGAGCCTCCATCCA | ||
| Ifrd1 | ATCGGACTGTTCAACCTTTCAG | Park et al., 2017 [30] |
| GCACTCTTATCAAGGGTTAGGTC | ||
| Gad67 | CACAGGTCACCCTCGATTTTT | MGH primerbank [31] |
| ACCATCCAACGATCTCTCTCATC | ||
| Dlx1 | GGCTGTGTTTATGGAGTTTGGG | Wang et al., 2016 [32] |
| CCTGGGTTTACGGATCTTTTTC | ||
| Dlx2 | GTGGCTGATATGCACTCGACC | MGH primerbank [31] |
| GCTGGTTGGTGTAGTAGCTGC | ||
| Mash1 | TCTCCTGGGAATGGACTTTG | Kraus et al., 2013 [33] |
| GGTTGGCTGTCTGGTTTGTT | ||
| Actin | AGAGGGAAATCGTGCGTGAC | Li et al., 2015 [13] |
| CAATAGTGATGACCTGGCCGT | ||
| Gapdh | GTGTTCCTACCCCCAATGTGT | Li et al., 2015 [13] |
| ATTGTCATACCAGGAAATGAGCTT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guerra, G.M.; May, D.; Kroll, T.; Koch, P.; Groth, M.; Wang, Z.-Q.; Li, T.-L.; Grigaravičius, P. Cell Type-Specific Role of RNA Nuclease SMG6 in Neurogenesis. Cells 2021, 10, 3365. https://doi.org/10.3390/cells10123365
Guerra GM, May D, Kroll T, Koch P, Groth M, Wang Z-Q, Li T-L, Grigaravičius P. Cell Type-Specific Role of RNA Nuclease SMG6 in Neurogenesis. Cells. 2021; 10(12):3365. https://doi.org/10.3390/cells10123365
Chicago/Turabian StyleGuerra, Gabriela Maria, Doreen May, Torsten Kroll, Philipp Koch, Marco Groth, Zhao-Qi Wang, Tang-Liang Li, and Paulius Grigaravičius. 2021. "Cell Type-Specific Role of RNA Nuclease SMG6 in Neurogenesis" Cells 10, no. 12: 3365. https://doi.org/10.3390/cells10123365
APA StyleGuerra, G. M., May, D., Kroll, T., Koch, P., Groth, M., Wang, Z.-Q., Li, T.-L., & Grigaravičius, P. (2021). Cell Type-Specific Role of RNA Nuclease SMG6 in Neurogenesis. Cells, 10(12), 3365. https://doi.org/10.3390/cells10123365







