Therapeutic Potential of MRGPRX2 Inhibitors on Mast Cells
Abstract
1. Introduction
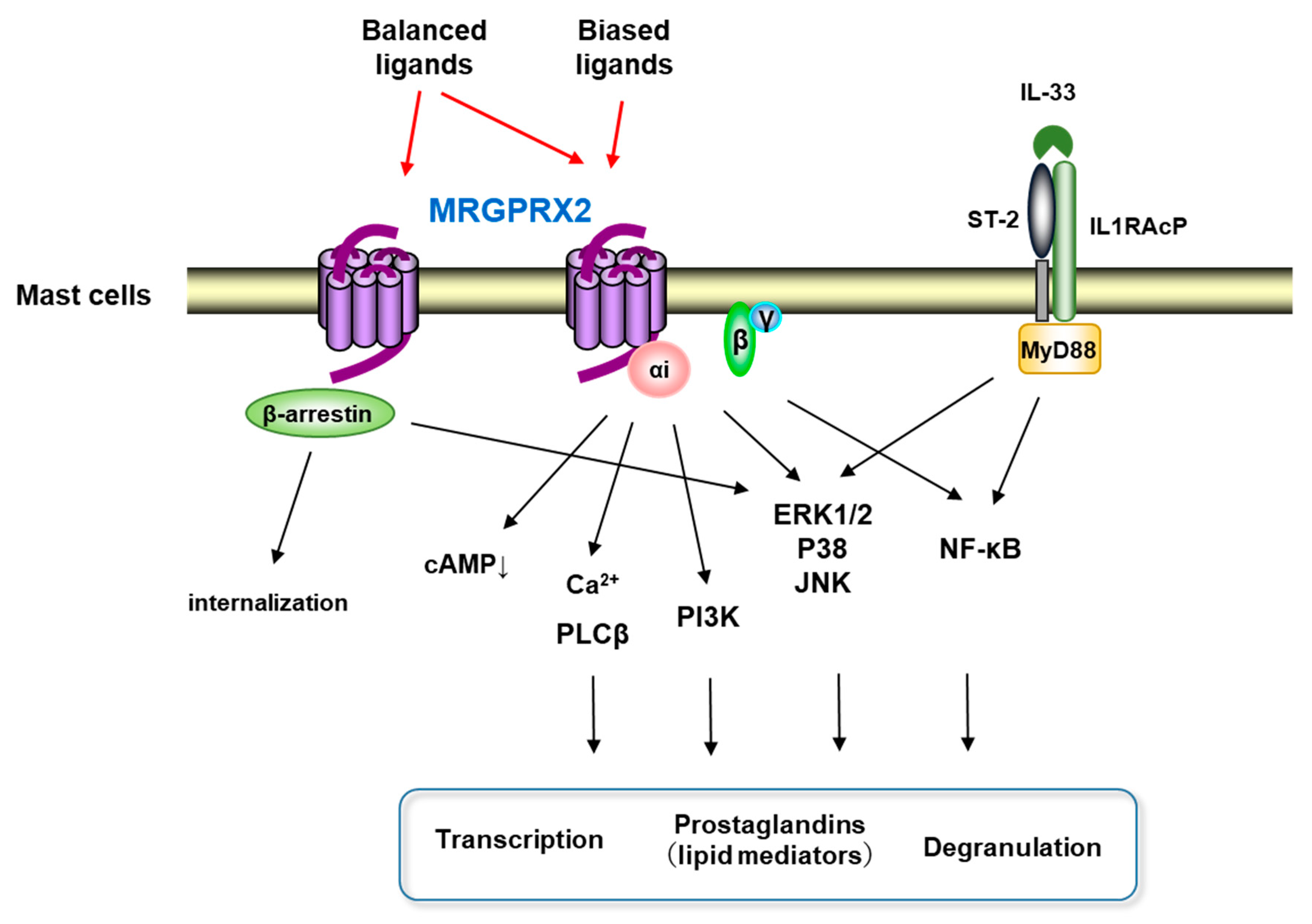
2. MRGPRX2
3. Neurogenic Inflammation in Postoperative Pain and Migraine
4. Type 2 Inflammation
4.1. Atopic Dermatitis (AD)
4.2. Chronic Urticaria (CU)
4.3. Allergic Contact Dermatitis (ACD)
4.4. Rheumatoid Arthritis (RA)
4.5. Ulcerative Colitis (UC)
5. Non-Histaminergic Itch
6. Drug-Induced Pseudoallergic Reaction
7. Host Defense
8. MRGPRX2 Inhibitors
8.1. Direct Inhibition of MRGPRX2
8.2. Inhibition of MRGPRX2 Downstream Signaling
8.3. Others
9. Future Prospects
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- St John, A.L.; Abraham, S.N. Innate immunity and its regulation by mast cells. J. Immunol. 2013, 190, 4458–4463. [Google Scholar] [CrossRef]
- Abraham, N.S.; St John, K.A. Mast cell-orchestrated immunity to pathogens. Nat. Rev. Immunol. 2010, 10, 440–452. [Google Scholar] [CrossRef] [PubMed]
- Younan, G.; Suber, F.; Xing, W.; Shi, T.; Kunori, Y.; Abrink, M.; Pejler, G.; Schlenner, S.M.; Rodewald, H.R.; Moore, F.D.; et al. The inflammatory response after an epidermal burn depends on the activities of mouse mast cell proteases 4 and 5. J. Immunol. 2010, 185, 7681–7690. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, D.F.; Barrett, N.A.; Austen, K.F.; Immunological Genome Project Consortium. Expression profiling of constitutive mast cells reveals a unique identity within the immune system. Nat. Immunol. 2016, 17, 878–887. [Google Scholar] [CrossRef] [PubMed]
- Metcalfe, D.D.; Baram, D.; Mekori, Y.A. Mast cells. Physiol. Rev. 1997, 77, 1033–1079. [Google Scholar] [CrossRef] [PubMed]
- Ferry, X.; Brehin, S.; Kamel, R.; Landry, Y. G protein-dependent activation of mast cell by peptides and basic secretagogues. Peptides 2002, 23, 1507–1515. [Google Scholar] [CrossRef]
- Kinet, J.P. The high-affinity IgE receptor (Fc epsilon RI): From physiology to pathology. Annu. Rev. Immunol. 1999, 17, 931–972. [Google Scholar] [CrossRef]
- Nadler, M.J.; Matthews, S.A.; Turner, H.; Kinet, J.P. Signal transduction by the high-affinity immunoglobulin E receptor Fc epsilon RI: Coupling form to function. Adv. Immunol. 2001, 76, 325–355. [Google Scholar] [CrossRef]
- Siraganian, R.P. Mast cell signal transduction from the high-affinity IgE receptor. Curr. Opin. Immunol. 2003, 15, 639–646. [Google Scholar] [CrossRef]
- Varricchi, G.; de Paulis, A.; Marone, G.; Galli, S.J. Future Needs in Mast Cell Biology. Int. J. Mol. Sci. 2019, 20, 4397. [Google Scholar] [CrossRef]
- Harvima, I.T.; Levi-Schaffer, F.; Draber, P.; Friedman, S.; Polakovicova, I.; Gibbs, B.F.; Blank, U.; Nilsson, G.; Maurer, M. Molecular targets on mast cells and basophils for novel therapies. J. Allergy Clin. Immunol. 2014, 134, 530–544. [Google Scholar] [CrossRef]
- Yoshimoto, T.; Matsushita, K. Innate-type and acquired-type allergy regulated by IL-33. Allergol. Int. 2014, 1, 3–11. [Google Scholar] [CrossRef]
- Bjerkan, L.; Sonesson, A.; Schenck, K. Multiple Functions of the New Cytokine-Based Antimicrobial Peptide Thymic Stromal Lymphopoietin (TSLP). Pharmaceuticals 2016, 9, 41. [Google Scholar] [CrossRef]
- Ferry, X.; Eichwald, V.; Daeffler, L.; Landry, Y. Activation of betagamma subunits of G(i2) and G(i3) proteins by basic secretagogues induces exocytosis through phospholipase Cbeta and arachidonate release through phospholipase Cgamma in MCs. J. Immunol. 2001, 167, 4805–4813. [Google Scholar] [CrossRef]
- Mousli, M.; Bronner, C.; Bueb, J.L.; Tschirhart, E.; Gies, J.P.; Landry, Y. Activation of rat peritoneal MCs by SP and mastoparan. J. Pharmacol. Exp. Ther. 1989, 250, 329–335. [Google Scholar]
- Mousli, M.; Bronner, C.; Bockaert, J.; Rouot, B.; Landry, Y. Interaction of SP, compound 48/80 and mastoparan with the alpha-subunit C-terminus of G protein. Immunol. Lett. 1990, 25, 355–357. [Google Scholar] [CrossRef]
- Tatemoto, K.; Nozaki, Y.; Tsuda, R.; Konno, S.; Tomura, K.; Furuno, M.; Ogasawara, H.; Edamura, K.; Takagi, H.; Iwamura, H.; et al. Immunoglobulin E-independent activation of mast cell is mediated by Mrg receptors. Biochem. Biophys. Res. Commun. 2006, 349, 1322–1328. [Google Scholar] [CrossRef]
- Fujisawa, D.; Kashiwakura, J.; Kita, H.; Kikukawa, Y.; Fujitani, Y.; Sasaki-Sakamoto, T.; Kuroda, K.; Nunomura, S.; Hayama, K.; Terui, T.; et al. Expression of Mas-related gene X2 on mast cells is upregulated in the skin of patients with severe chronic urticaria. J. Allergy Clin. Immunol. 2014, 134, 622–633. [Google Scholar] [CrossRef] [PubMed]
- Ray, P.; Torck, A.; Quigley, L.; Wangzhou, A.; Neiman, M.; Rao, C.; Lam, T.; Kim, J.Y.; Kim, T.H.; Zhang, M.Q.; et al. Comparative transcriptome profiling of the human and mouse dorsal root ganglia: An RNA-seq-based resource for pain and sensory neuroscience research. Pain 2018, 159, 1325–1345. [Google Scholar] [CrossRef] [PubMed]
- McNeil, B.D.; Pundir, P.; Meeker, S.; Han, L.; Undem, B.J.; Kulka, M.; Dong, X. Identification of a MC specific receptor crucial for pseudo-allergic drug reactions. Nature 2015, 519, 237–241. [Google Scholar] [CrossRef]
- Subramanian, H.; Gupta, K.; Lee, D.; Bayir, A.K.; Ahn, H.; Ali, H. β-Defensins Activate Human Mast Cells via Mas-Related Gene X2. J. Immunol. 2013, 191, 345–352. [Google Scholar] [CrossRef]
- Lembo, P.M.C.; Grazzini, E.; Groblewski, T.; O′Donnell, D.; Roy, M.-O.; Zhang, J.; Hoffert, C.; Cao, J.; Schmidt, R.; Pelletier, M.; et al. Proenkephalin A gene products activate a new family of sensory neuron-specific GPCRs. Nat. Neurosci. 2002, 5, 201–209. [Google Scholar] [CrossRef]
- Dong, X.; Han, S.; Zylka, M.J.; Simon, M.I.; Anderson, D.J. A diverse family of GPCRs expressed in specific subsets of nociceptive sensory neurons. Cell 2001, 106, 619–632. [Google Scholar] [CrossRef]
- Han, S.K.; Dong, X.; Hwang, J.; Zylka, M.J.; Anderson, D.J.; Simon, M.I. Orphan G protein-coupled receptors MrgA1 and MrgC11 are distinctively activated by RF-amide-related peptides through the Galpha q/11 pathway. Proc. Natl. Acad. Sci. USA 2002, 99, 14740–14745. [Google Scholar] [CrossRef] [PubMed]
- Robas, N.; Mead, E.; Fidock, M. MrgX2 is a high potency cortistatin receptor expressed in dorsal root ganglion. J. Biol. Chem. 2003, 278, 44400–44404. [Google Scholar] [CrossRef] [PubMed]
- Kamohara, M.; Matsuo, A.; Takasaki, J.; Kohda, M.; Matsumoto, M.; Matsumoto, S.; Soga, T.; Hiyama, H.; Kobori, M.; Katou, M. Identification of MrgX2 as a human G-protein-coupled receptor for proadrenomedullin N-terminal peptides. Biochem. Biophys. Res. Commun. 2005, 330, 1146–1152. [Google Scholar] [CrossRef]
- Nunn, C.; Langenegger, D.; Hurth, K.; Schmidt, K.; Fehlmann, D.; Hoyer, D. Agonist properties of putative small-molecule somatostatin sst2 receptor-selective antagonists. Eur. J. Pharmacol. 2003, 465, 211–218. [Google Scholar] [CrossRef]
- Regoli, D.; Dion, S.; Rhaleb, N.E.; Rouissi, N.; Tousignant, C.; Jukic, D.; D′Orleans-Juste, P.; Drapeau, G. Selective agonists for receptors of substance P and related neurokinins. Biopolymers 1989, 28, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Arifuzzaman, M.; Mobley, Y.R.; Choi, H.W.; Bist, P.; Salinas, C.A.; Brown, Z.D.; Chen, S.L.; Staats, H.F.; Abraham, S.N. MRGPR-mediated activation of local mast cells clears cutaneous bacterial infection and protects against reinfection. Sci. Adv. 2019, 5, eaav0216. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, H.; Gupta, K.; Guo, Q.; Price, R.; Ali, H. Mas-related gene X2 (MrgX2) is a novel G protein-coupled receptor for the antimicrobial peptide LL-37 in human mast cells: Resistance to receptor phosphorylation, desensitization, and internalization. J. Biol. Chem. 2011, 286, 44739–44749. [Google Scholar] [CrossRef]
- Tatemoto, K.; Nozaki, Y.; Tsuda, R.; Kaneko, S.; Tomura, K.; Furuno, M.; Ogasawara, H.; Edamura, K.; Takagi, H.; Iwamura, H.; et al. Endogenous protein and enzyme fragments induce immunoglobulin E-independent activation of mast cells via a G protein-coupled receptor, MRGPRX2. Scand. J. Immunol. 2018, 87, e12655. [Google Scholar] [CrossRef]
- Ogasawara, H.; Furuno, M.; Edamura, K.; Noguchi, M. Peptides of major basic protein and eosinophil cationic protein activate human mast cells. Biochem. Biophys. Rep. 2020, 21, 100719. [Google Scholar] [CrossRef]
- McNeil, D.B. Mas-related G protein-coupled receptor X2 activation by therapeutic drugs. Neurosci. Lett. 2021, 751, 135746. [Google Scholar] [CrossRef] [PubMed]
- Boix, E.; Leonidas, D.D.; Nikolovski, Z.; Nogués, M.V.; Cuchillo, C.M.; Acharya, K.R. Crystal structure of eosinophil cationic protein at 2.4 A resolution. Biochemistry 1999, 38, 16794–16801. [Google Scholar] [CrossRef] [PubMed]
- Swaminathan, G.J.; Weaver, A.J.; Loegering, D.A.; Checkel, J.L.; Leonidas, D.D.; Gleich, G.J.; Acharya, K.R. Crystal structure of the eosinophil major basic protein at 1.8 A. An atypical lectin with a paradigm shift in specificity. J. Biol. Chem. 2001, 276, 26197–26203. [Google Scholar] [CrossRef]
- Azimi, E.; Reddy, V.B.; Shade, K.C.; Anthony, R.M.; Talbot, S.; Pereira, P.J.S.; Lerner, E.A. Dual action of neurokinin-1 antagonists on Mas-related GPCRs. JCI Insight 2016, 1, e89362. [Google Scholar] [CrossRef]
- Pedersen, S.H.; la Cour, S.H.; Calloe, K.; Hauser, F.; Olesen, J.; Klaerke, D.A.; Jansen-Olesen, I. PACAP-38 and PACAP(6-38) degranulate rat meningeal mast cells via the orphan MrgB(3)-Receptor. Front. Cell Neurosci. 2019, 13, 114. [Google Scholar] [CrossRef] [PubMed]
- Haitina, T.; Fredriksson, R.; Foord, S.M.; Schioth, H.B.; Gloriam, D.E. The G protein-coupled receptor subset of the dog genome is more similar to that in humans than rodents. BMC Genom. 2009, 10, 24. [Google Scholar] [CrossRef] [PubMed]
- Hamamura-Yasuno, E.; Iguchi, T.; Kumagai, K.; Tsuchiya, Y.; Mori, K. Identification of the dog orthologue of human MAS-related G protein coupled receptor X2 (MRGPRX2) essential for drug-induced pseudo-allergic reactions. Sci. Rep. 2020, 10, 16146. [Google Scholar] [CrossRef]
- Whalen, E.J.; Rajagopal, S.; Lefkowitz, R.J. Therapeutic potential of beta-arrestin- and G protein-biased agonists. Trends Mol. Med. 2011, 17, 126–139. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, Y.; Zhang, Y.; Lai, Y.; Chen, W.; Xiao, Z.; Zhang, W.; Jin, M.; Yu, B. LL-37-induced human mast cell activation through G protein-coupled receptor MrgX2. Int. Immunopharmacol. 2017, 49, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, H.; Furuno, M.; Edamura, K.; Noguchi, M. Novel MRGPRX2 antagonists inhibit IgE-independent activation of human umbilical cord blood-derived mast cells. J. Leukoc. Biol. 2019, 106, 1069–1077. [Google Scholar] [CrossRef]
- Occhiuto, C.J.; Kammala, A.K.; Yang, C.; Nellutla, R.; Garcia, M.; Gomez, G.; Subramanian, H. Store-Operated Calcium Entry via STIM1 Contributes to MRGPRX2 Induced Mast Cell Functions. Front. Immunol. 2020, 10, 3143. [Google Scholar] [CrossRef] [PubMed]
- Metcalfe, D.D.; Peavy, D.R.; Gilfillan, M.A. Mechanisms of mast cell signaling in anaphylaxis. J. Allergy Clin. Immunol. 2009, 124, 639–646. [Google Scholar] [CrossRef]
- Zhang, C.; Baumgartner, R.A.; Yamada, K.; Beaven, M.A. Mitogen-activated protein (MAP) kinase regulates production of tumor necrosis factor-alpha and release of arachidonic acid in mast cells. Indications of communication between p38 and p42 MAP kinases. J. Biol. Chem. 1997, 272, 13397–13402. [Google Scholar] [CrossRef]
- Gu, Y.; Yang, D.K.; Spinas, E.; Kritas, S.K.; Saggini, A.; Caraffa, A.; Antinolfi, P.; Saggini, R.; Conti, P. Role of TNF in mast cell neuroinflammation and pain. J. Biol. Regul. Homeost. Agents. 2015, 29, 787–791. [Google Scholar]
- Okayama, Y.; Tkaczyk, C.; Metcalfe, D.D.; Gilfillan, A.M. Comparison of Fc epsilon RI- and Fc gamma RI-mediated degranulation and TNF-alpha synthesis in human mast cells: Selective utilization of phosphatidylinositol-3-kinase for Fc gamma RI-induced degranulation. Eur. J. Immunol. 2003, 33, 1450–1459. [Google Scholar] [CrossRef]
- Azzolina, A.; Guarneri, P.; Lampiasi, N. Involvement of p38 and JNK MAPKs pathways in Substance P-induced production of TNF-alpha by peritoneal mast cells. Cytokine 2002, 18, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Azzolina, A.; Bongiovanni, A.; Lampiasi, N. Substance P induces TNF-alpha and IL-6 production through NF kappa B in peritoneal mast cells. Biochim. Biophys. Acta. 2003, 1643, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Wang, J.; Zhang, Y.; Zeng, Y.; Hu, S.; Bai, H.; Hou, Y.; Wang, C.; He, H.; He, L. Imperatorin ameliorates mast cell-mediated allergic airway inflammation by inhibiting MRGPRX2 and CamKII/ERK signaling pathway. Biochem. Pharmacol. 2021, 184, 114401. [Google Scholar] [CrossRef]
- Kanazawa, K.; Okumura, K.; Ogawa, H.; Niyonsaba, F. An antimicrobial peptide with angiogenic properties, AG-30/5C, activates human mast cells through the MAPK and NF-kappaB pathways. Immunol. Res. 2016, 64, 594–603. [Google Scholar] [CrossRef]
- Franke, K.; Wang, Z.; Zuberbier, T.; Babina, M. Cytokines Stimulated by IL-33 in Human Skin Mast Cells: Involvement of NF-kappaB and p38 at Distinct Levels and Potent Co-Operation with FcepsilonRI and MRGPRX2. Int. J. Mol. Sci. 2021, 22, 3580. [Google Scholar] [CrossRef] [PubMed]
- Benyon, R.C.; Robinson, C.; Church, M.K. Differential release of histamine and eicosanoids from human skin MCs activated by IgE-dependent and non-immunological stimuli. Br. J. Pharmacol. 1989, 97, 898–904. [Google Scholar] [CrossRef] [PubMed]
- Babina, M.; Wang, Z.; Roy, S.; Guhl, S.; Franke, K.; Artuc, M.; Ali, H.; Zuberbier, T. MRGPRX2 is the codeine receptor of human skin mast cells: Desensitization through beta-arrestin and lack of correlation with the FcepsilonRI pathway. J. Investig. Dermatol. 2021, 141, 1286–1296. [Google Scholar] [CrossRef]
- Chompunud Na Ayudhya, C.; Amponnawarat, A.; Ali, H. Substance P serves as a balanced agonist for MRGPRX2 and a single tyrosine residue is required for beta-arrestin recruitment and receptor internalization. Int. J. Mol. Sci. 2021, 22, 5318. [Google Scholar] [CrossRef]
- Roy, S.; Ganguly, A.; Haque, M.; Ali, H. Angiogenic host defense peptide AG-30/5C and bradykinin B(2) receptor antagonist icatibant are G Protein biased agonists for MRGPRX2 in mast cells. J. Immunol. 2019, 202, 1229–1238. [Google Scholar] [CrossRef]
- Shelburne, C.P.; Nakano, H.; St John, A.L.; Chan, C.; McLachlan, J.B.; Gunn, M.D.; Staats, H.F.; Abraham, S.N. Mast cells augment adaptive immunity by orchestrating dendritic cell trafficking through infected tissues. Cell Host Microbe 2009, 6, 331–342. [Google Scholar] [CrossRef]
- Dothel, G.; Barbaro, M.R.; Boudin, H.; Vasina, V.; Cremon, C.; Gargano, L.; Bellacosa, L.; De Giorgio, R.; Le Berre-Scoul, C.; Aubert, P.; et al. Nerve fiber outgrowth is increased in the intestinal mucosa of patients with irritable bowel syndrome. Gastroenterology 2015, 148, 1002–1101. [Google Scholar] [CrossRef]
- Gupta, K.; Harvima, I.T. Mast cell-neural interactions contribute to pain and itch. Immunol. Rev. 2018, 282, 168–187. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, M.; Kido, K.; Ohtani, N.; Masaki, E. Mast cell stabilization promotes antinociceptive effects in a mouse model of postoperative pain. J. Pain Res. 2013, 6, 161–166. [Google Scholar] [CrossRef]
- Williamson, D.J.; Hargreaves, R.J. Neurogenic inflammation in the context of migraine. Microsc. Res. Tech. 2001, 53, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Kehlet, H.; Jensen, T.S.; Woolf, C.J. Persistent postsurgical pain: Risk factors and prevention. Lancet 2006, 367, 1618–1625. [Google Scholar] [CrossRef]
- Marchand, F.; Perretti, M.; McMahon, S.B. Role of the immune system in chronic pain. Nat. Rev. Neurosci. 2005, 6, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, S.M.; Drewes, C.C.; Silva, C.R.; Trevisan, G.; Boschen, S.L.; Moreira, C.G.; de Almeida Cabrini, D.; Da Cunha, C.; Ferreira, J. Involvement of mast cells in a mouse model of postoperative pain. J. Eur. J. Pharmacol. 2011, 672, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Lisowska, B.; Siewruk, K.; Lisowski, A. Substance P and acute pain in patients undergoing orthopedic surgery. PLoS ONE 2016, 11, e0146400. [Google Scholar] [CrossRef]
- Green, D.P.; Limjunyawong, N.; Gour, N.; Pundir, P.; Dong, X. A mast-cell-specific receptor mediates neurogenic inflammation and pain. Neuron 2019, 101, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Borsook, D.; Hargreaves, R.; Bountra, C.; Porreca, F. Lost but making progress—Where will new analgesic drugs come from? Sci. Transl. Med. 2014, 6, 249sr3. [Google Scholar] [CrossRef]
- Borsook, D.; Upadhyay, J.; Klimas, M.; Schwarz, A.J.; Coimbra, A.; Baumgartner, R.; George, E.; Potter, W.Z.; Large, T.; Bleakman, D.; et al. Decision-making using fMRI in clinical drug development: Revisiting NK-1 receptor antagonists for pain. Drug. Discov. Today 2012, 17, 964–973. [Google Scholar] [CrossRef]
- Pietrobon, D.; Striessnig, J. Neurobiology of migraine. Nat. Rev. Neurosci. 2003, 4, 386–398. [Google Scholar] [CrossRef]
- Ramachandra, R. Neurogenic inflammation and its role in migraine. Semin. Immunopathol. 2018, 40, 301–314. [Google Scholar] [CrossRef]
- Okragly, A.J.; Morin, S.M.; DeRosa, D.; Martin, A.P.; Johnson, K.W.; Johnson, M.P.; Benschop, R.J. Human mast cells release the migraine-inducing factor pituitary adenylate cyclase-activating polypeptide (PACAP). Cephalalgia 2018, 38, 1564–1574. [Google Scholar] [CrossRef]
- Levy, D. Migraine pain, meningeal inflammation, and mast cells. Curr. Pain Headache Rep. 2009, 13, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Fusayasu, E.; Kowa, H.; Takeshima, T.; Nakaso, N.; Nakashima, K. Increased plasma substance P and CGRP levels, and high ACE activity in migraineurs during headache-free periods. Pain 2007, 128, 209–214. [Google Scholar] [CrossRef]
- Malhotra, R. Understanding migraine: Potential role of neurogenic inflammation. Ann. Indian Acad. Neurol. 2016, 19, 175–182. [Google Scholar] [CrossRef]
- Leung, D.Y.; Bieber, T. Atopic dermatitis. Lancet 2003, 361, 151–160. [Google Scholar] [CrossRef]
- Oyoshi, M.K.; He, R.; Kumar, L.; Yoon, J.; Geha, R.S. Cellular and molecular mechanisms in atopic dermatitis. Adv. Immunol. 2009, 102, 135–226. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, T.; Ando, T.; Kimura, M.; Wilson, B.S.; Kawakami, Y. Mast cells in atopic dermatitis. Curr. Opin. Immunol. 2009, 21, 666–678. [Google Scholar] [CrossRef]
- Mori, T.; Ishida, K.; Mukumoto, S.; Yamada, Y.; Imokawa, G.; Kabashima, K.; Kobayashi, M.; Bito, T.; Nakamura, M.; Ogasawara, K.; et al. Comparison of skin barrier function and sensory nerve electric current perception threshold between IgE-high extrinsic and IgE-normal intrinsic types of atopic dermatitis. Br. J. Dermatol. 2010, 162, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.T.; Goodarzi, H.; Chen, H.Y. IgE, mast cells, and eosinophils in atopic dermatitis. Clin. Rev. Allergy Immunol. 2011, 41, 298–310. [Google Scholar] [CrossRef]
- Hon, K.L.; Lam, M.C.; Wong, K.Y.; Leung, T.F.; Ng, P.C. Pathophysiology of nocturnal scratching in childhood atopic dermatitis: The role of brain-derived neurotrophic factor and substance P. Br. J. Dermatol. 2007, 157, 922–925. [Google Scholar] [CrossRef]
- Salomon, J.; Baran, E. The role of selected neuropeptides in pathogenesis of atopic dermatitis. J. Eur. Acad. Dermatol. Venereol. 2008, 22, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Toyoda, M.; Nakamura, M.; Makino, T.; Hino, T.; Kagoura, M.; Morohashi, M. Nerve growth factor and substance P are useful plasma markers of disease activity in atopic dermatitis. Br. J. Dermatol. 2002, 147, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Thapaliya, M.; Chompunud Na Ayudhya, C.; Amponnawarat, A.; Roy, S.; Ali, H. Mast Cell-Specific MRGPRX2: A Key Modulator of Neuro-Immune Interaction in Allergic Diseases. Curr. Allergy Asthma Rep. 2021, 21, 3. [Google Scholar] [CrossRef]
- Serhan, N.; Basso, L.; Sibilano, R.; Petitfils, C.; Meixiong, J.; Bonnart, C.; Reber, L.L.; Marichal, T.; Starkl, P.; Cenac, N.; et al. House dust mites activate nociceptor-mast cell clusters to drive type 2 skin inflammation. Nat. Immunol. 2019, 20, 1435–1443. [Google Scholar] [CrossRef] [PubMed]
- Kiehl, P.; Falkenberg, K.; Vogelbruch, M.; Kapp, A. Tissue eosinophilia in acute and chronic atopic dermatitis: A morphometric approach using quantitative image analysis of immunostaining. Br. J. Dermatol. 2001, 145, 720–729. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Babina, M. MRGPRX2 signals its importance in cutaneous mast cell biology: Does MRGPRX2 connect mast cells and atopic dermatitis? Exp. Dermatol. 2020, 29, 1104–1111. [Google Scholar] [CrossRef] [PubMed]
- Ferrer, M.; Nuñez-Córdoba, J.M.; Luquin, E.; Grattan, C.E.; De la Borbolla, J.M.; Sanz, M.; Schwartz, L.B. Serum total tryptase levels are increased in patients with active chronic urticaria. Clin. Exp. Allergy 2010, 40, 1760–1766. [Google Scholar] [CrossRef]
- Bracken, S.J.; Abraham, S.; MacLeod, A.S. Autoimmune Theories of Chronic Spontaneous Urticaria. Front. Immunol. 2019, 10, 627. [Google Scholar] [CrossRef]
- Borici-Mazi, R.; Kouridakis, S.; Kontou-Fili, K. Cutaneous responses to substance P and calcitonin gene-related peptide in chronic urticaria: The effect of cetirizine and dimethindene. Allergy 1999, 54, 46–56. [Google Scholar] [CrossRef]
- Smith, C.H.; Atkinson, B.; Morris, R.W.; Hayes, N.; Foreman, J.C.; Lee, T.H. Cutaneous responses to vasoactive intestinal polypeptide in chronic idiopathic urticaria. Lancet 1992, 339, 91–93. [Google Scholar] [CrossRef]
- Kühn, H.; Kolkhir, P.; Babina, M.; Düll, M.; Frischbutter, S.; Fok, J.S.; Jiao, Q.; Metz, M.; Scheffel, J.; Wolf, K.; et al. Mas-related G protein-coupled receptor X2 and its activators in dermatologic allergies. J. Allergy Clin. Immunol. 2021, 147, 456–469. [Google Scholar] [CrossRef] [PubMed]
- Ying, S.; Kikuchi, Y.; Meng, Q.; Kay, A.B.; Kaplan, A.P. TH1/TH2 cytokines and inflammatory cells in skin biopsy specimens from patients with chronic idiopathic urticaria: Comparison with the allergen-induced late-phase cutaneous reaction. J. Allergy Clin. Immunol. 2002, 109, 694–700. [Google Scholar] [CrossRef]
- Peters, M.S.; Schroeter, A.L.; Kephart, G.M.; Gleich, G.J. Localization of eosinophil granule major basic protein in chronic urticaria. J. Investig. Dermatol. 1983, 81, 39–43. [Google Scholar] [CrossRef]
- Spry, C.J.; Tai, P.C.; Barkans, J. Tissue localization of human eosinophil cationic proteins in allergic diseases. Int. Arch. Allergy Appl. Immunol. 1985, 77, 252–254. [Google Scholar] [CrossRef] [PubMed]
- Vocanson, M.; Hennino, A.; Rozières, A.; Poyet, G.; Nicolas, J.F. Effector and regulatory mechanisms in allergic contact dermatitis. Allergy 2009, 64, 1699–1714. [Google Scholar] [CrossRef]
- Saint-Mezard, P.; Rosieres, A.; Krasteva, M.; Berard, F.; Dubois, B.; Kaiserlian, D.; Nicolas, J.F. Allergic contact dermatitis. Eur. J. Dermatol. 2004, 14, 284–295. [Google Scholar]
- Meixiong, J.; Anderson, M.; Limjunyawong, N.; Sabbagh, M.F.; Hu, E.; Mack, M.R.; Oetjen, L.K.; Wang, F.; Kim, B.S.; Dong, X. Activation of Mast-Cell-Expressed Mas-Related G-Protein-Coupled Receptors Drives Non-histaminergic Itch. Immunity 2019, 50, 1163–1171. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Wang, J.; Zhang, Y.; Ge, S.; Wu, Y.; Fan, T.; Wang, N. Gold induces a pseudo-allergic reaction via MRGPRX2 both in vitro and in vivo. Cell. Immunol. 2019, 341, 103923. [Google Scholar] [CrossRef]
- Peng, B.; Che, D.; Hao, Y.; Zheng, Y.; Liu, R.; Qian, Y.; Cao, J.; Wang, J.; Zhang, Y.; He, L.; et al. Thimerosal induces skin pseudo-allergic reaction via Mas-related G-protein coupled receptor B2. J. Dermatol. Sci. 2019, 95, 99–106. [Google Scholar] [CrossRef]
- Rivellese, F.; Nerviani, A.; Rossi, F.W.; Marone, G.; Matucci-Cerinic, M.; de Paulis, A.; Pitzalis, C. Mast cells in rheumatoid arthritis: Friends or foes? Autoimmun. Rev. 2017, 16, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Tetlow, L.C.; Woolley, D.E. Distribution, activation and tryptase/chymase phenotype of mast cells in the rheumatoid lesion. Ann. Rheum. Dis. 1995, 54, 549–555. [Google Scholar] [CrossRef]
- Malone, D.G.; Irani, A.M.; Schwartz, L.B.; Barrett, K.E.; Metcalfe, D.D. Mast cell numbers and histamine levels in synovial fluids from patients with diverse arthritides. Arthritis Rheum. 1986, 29, 956–963. [Google Scholar] [CrossRef] [PubMed]
- Olsson, N.; Ulfgren, A.K.; Nilsson, G. Demonstration of mast cell chemotactic activity in synovial fluid from rheumatoid patients. Ann. Rheum. Dis. 2001, 60, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Kashiwakura, J.; Matsuda, A.; Watanabe, Y.; Sakamoto-Sasaki, T.; Matsumoto, K.; Hashimoto, N.; Saito, S.; Ohmori, K.; Nagaoka, M.; et al. Activation of human synovial mast cells from rheumatoid arthritis or osteoarthritis patients in response to aggregated IgG through Fcgamma receptor I and Fcgamma receptor II. Arthritis Rheum. 2013, 65, 109–119. [Google Scholar] [CrossRef]
- Marabini, S.; Matucci-Cerinic, M.; Geppetti, P.; Del Bianco, E.; Marchesoni, A.; Tosi, S.; Cagnoni, M.; Partsch, G. Substance P and somatostatin levels in rheumatoid arthritis, osteoarthritis, and psoriatic arthritis synovial fluid. Ann. N. Y. Acad. Sci. 1991, 632, 435–436. [Google Scholar] [CrossRef]
- Dirmeier, M.; Capellino, S.; Schubert, T.; Angele, P.; Anders, S.; Straub, R.H. Lower density of synovial nerve fibres positive for calcitonin gene-related peptide relative to substance P in rheumatoid arthritis but not in osteoarthritis. Rheumatology 2008, 47, 36–40. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Okamura, Y.; Mishima, S.; Kashiwakura, J.I.; Sasaki-Sakamoto, T.; Toyoshima, S.; Kuroda, K.; Saito, S.; Tokuhashi, Y.; Okayama, Y. The dual regulation of substance P-mediated inflammation via human synovial mast cells in rheumatoid arthritis. Allergol. Int. 2017, 66 (Suppl. 1), S9–S20. [Google Scholar] [CrossRef]
- Suurmond, J.; van der Velden, D.; Kuiper, J.; Bot, I.; Toes, R.E. Mast cells in rheumatic disease. Eur. J. Pharmacol. 2016, 778, 116–124. [Google Scholar] [CrossRef]
- Porter, R.J.; Kalla, R.; Ho, G.T. Ulcerative colitis: Recent advances in the understanding of disease pathogenesis. F1000Research 2020, 9, 294. [Google Scholar] [CrossRef]
- Stasikowska-Kanicka, O.; Danilewicz, M.; Głowacka, A.; Wągrowska-Danilewicz, M. Mast cells and eosinophils are involved in activation of ulcerative colitis. Adv. Med. Sci. 2012, 57, 230–236. [Google Scholar] [CrossRef]
- Bischoff, S.C. Mast cells in gastrointestinal disorders. Eur. J. Pharmacol. 2016, 778, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Chen, E.; Chuang, L.S.; Giri, M.; Villaverde, N.; Hsu, N.Y.; Sabic, K.; Joshowitz, S.; Gettler, K.; Nayar, S.; Chai, Z.; et al. Inflamed ulcerative colitis regions associated with MRGPRX2-mediated mast cell degranulation and cell activation modules, defining a new therapeutic target. Gastroenterology 2021, 160, 1709–1724. [Google Scholar] [CrossRef]
- Watanabe, T.; Kubota, Y.; Muto, T. Substance P containing nerve fibers in ulcerative colitis. Int. J. Colorectal. Dis. 1998, 13, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, C.N.; Robert, M.E.; Eysselein, V.E. Rectal substance P concentrations are increased in ulcerative colitis but not in Crohn’s disease. Am. J. Gastroenterol. 1993, 88, 908–913. [Google Scholar]
- Kamo, A.; Negi, O.; Tengara, S.; Kamata, Y.; Noguchi, A.; Ogawa, H.; Tominaga, M.; Takamori, K. Histamine H(4) receptor antagonists ineffective against itch and skin inflammation in atopic dermatitis mouse model. J. Investig. Dermatol. 2014, 134, 546–548. [Google Scholar] [CrossRef] [PubMed]
- Sanders, K.M.; Nattkemper, L.A.; Yosipovitch, G. Advances in understanding itching and scratching: A new era of targeted treatments. F1000Research 2016, 5, F1000. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Steinhoff, M.; Neisius, U.; Ikoma, A.; Fartasch, M.; Heyer, G.; Skov, P.S.; Luger, T.A.; Schmelz, M. Proteinase-activated receptor-2 mediates itch: A novel pathway for pruritus in human skin. J. Neurosci. 2003, 23, 6176. [Google Scholar] [CrossRef]
- Gaudenzio, N.; Sibilano, R.; Marichal, T.; Starkl, P.; Reber, L.L.; Cenac, N.; McNeil, B.D.; Dong, X.; Hernandez, J.D.; Sagi-Eisenberg, R.; et al. Different activation signals induce distinct mast cell degranulation strategies. J. Clin. Investig. 2016, 126, 3981–3998. [Google Scholar] [CrossRef]
- Takamori, A.; Nambu, A.; Sato, K.; Yamaguchi, S.; Matsuda, K.; Numata, T.; Sugawara, T.; Yoshizaki, T.; Arae, K.; Morita, H.; et al. IL-31 is crucial for induction of pruritus, but not inflammation, in contact hypersensitivity. Sci. Rep. 2018, 8, 6639. [Google Scholar] [CrossRef] [PubMed]
- Kido-Nakahara, M.; Buddenkotte, J.; Kempkes, C.; Ikoma, A.; Cevikbas, F.; Akiyama, T.; Nunes, F.; Seeliger, S.; Hasdemir, B.; Mess, C.; et al. Neural peptidase endothelin-converting enzyme 1 regulates endothelin 1-induced pruritus. J. Clin. Investig. 2014, 124, 2683–2695. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Park, C.K.; Chen, G.; Han, Q.; Xie, R.G.; Liu, T.; Ji, R.R.; Lee, S.Y. A monoclonal antibody that targets a NaV1.7 channel voltage sensor for pain and itch relief. Cell 2014, 157, 1393–1404. [Google Scholar] [CrossRef]
- Farnam, K.; Chang, C.; Teuber, S.; Gershwin, M.E. Nonallergic drug hypersensitivity reactions. Int. Arch. Allergy Immunol. 2012, 159, 327–345. [Google Scholar] [CrossRef]
- Kelesidis, T.; Fleisher, J.; Tsiodras, S. Anaphylactoid reaction considered ciprofloxacin related: A case report and literature review. Clin. Ther. 2010, 32, 515–526. [Google Scholar] [CrossRef]
- Lumry, W.R.; Li, H.H.; Levy, R.J.; Potter, P.C.; Farkas, H.; Moldovan, D.; Riedl, M.; Li, H.; Craig, T.; Bloom, B.J.; et al. Randomized placebo-controlled trial of the bradykinin B2 receptor antagonist icatibant for the treatment of acute attacks of hereditary angioedema: The FAST-3 trial. Ann. Allergy Asthma Immunol. 2011, 107, 529–537. [Google Scholar] [CrossRef]
- Sivagnanam, S.; Deleu, D. Red man syndrome. Crit. Care 2003, 7, 119–120. [Google Scholar] [CrossRef]
- Lieberman, P.; Garvey, L.H. Mast cells and anaphylaxis. Curr. Allergy Asthma Rep. 2016, 16, 20. [Google Scholar] [CrossRef]
- Fisher, M.M.; Munro, I. Life-threatening anaphylactoid reactions to muscle relaxants. Anesth. Analg. 1983, 62, 559–564. [Google Scholar] [CrossRef]
- Kumar, M.; Duraisamy, K.; Chow, B.K. Unlocking the Non-IgE-Mediated Pseudo-Allergic Reaction Puzzle with Mas-Related G-Protein Coupled Receptor Member X2 (MRGPRX2). Cells 2021, 10, 1033. [Google Scholar] [CrossRef]
- Ali, H. Revisiting the role of MRGPRX2 on hypersensitivity reactions to neuromuscular blocking drugs. Curr. Opin. Immunol. 2021, 72, 65–71. [Google Scholar] [CrossRef]
- Wei, D.; Hu, T.; Hou, Y.J.; Wang, X.J.; Lu, J.Y.; Ge, S.; Wang, C.; He, H.Z. MRGPRX2 is critical for clozapine induced pseudo-allergic reactions. Immunopharmacol. Immunotoxicol. 2021, 43, 77–84. [Google Scholar] [CrossRef]
- Zschaler, J.; Schlorke, D.; Arnhold, J. Differences in innate immune response between man and mouse. Crit. Rev. Immunol. 2014, 34, 433–454. [Google Scholar] [CrossRef]
- Scott, D.; Miller, W. Idiosyncratic cutaneous adverse drug reactions in the dog: Literature review and report of 101 cases (1990–1996). Canine Pract. 1999, 24, 16–22. [Google Scholar]
- Pearce, F.L. Functional heterogeneity of mast cells from different species and tissues. Klin. Wochenschr. 1982, 60, 954–957. [Google Scholar] [CrossRef]
- Robinson, E.P.; Faggella, A.M.; Henry, D.P.; Russell, W.L. Comparison of histamine release induced by morphine and oxymorphone administration in dogs. Am. J. Vet. Res. 1988, 49, 1699–1701. [Google Scholar]
- Mori, K.; Maru, C.; Takasuna, K. Characterization of histamine release induced by fluoroquinolone antibacterial agents in-vivo and in-vitro. J. Pharm. Pharmacol. 2000, 52, 577–584. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, F.; Chen, X.; Zhu, X.; Uetrecht, J. Involvement of the immune system in idiosyncratic drug reactions. Drug Metab. Pharmacokinet. 2011, 26, 47–59. [Google Scholar] [CrossRef]
- Postow, M.A.; Sidlow, R.; Hellmann, M.D. Immune-Related Adverse Events Associated with Immune Checkpoint Blockade. N. Engl. J. Med. 2018, 378, 158–168. [Google Scholar] [CrossRef]
- Chompunud Na Ayudhya, C.; Roy, S.; Thapaliya, M.; Ali, H. Roles of a Mast Cell-Specific Receptor MRGPRX2 in Host Defense and Inflammation. J. Dent. Res. 2020, 99, 882–890. [Google Scholar] [CrossRef]
- Roy, S.; Chompunud Na Ayudhya, C.; Thapaliya, M.; Deepak, V.; Ali, H. Multifaceted MRGPRX2: New insight into the role of mast cells in health and disease. J. Allergy Clin. Immunol. 2021, 148, 293–308. [Google Scholar] [CrossRef]
- Pundir, P.; Liu, R.; Vasavda, C.; Serhan, N.; Limjunyawong, N.; Yee, R.; Zhan, Y.; Dong, X.; Wu, X.; Zhang, Y.; et al. A Connective Tissue Mast-Cell-Specific Receptor Detects Bacterial Quorum-Sensing Molecules and Mediates Antibacterial Immunity. Cell Host Microbe 2019, 26, 114–122. [Google Scholar] [CrossRef]
- Qiao, C.; Hu, S.; Che, D.; Wang, J.; Gao, J.; Ma, R.; Jiang, W.; Zhang, T.; Liu, R. The anti-anaphylactoid effects of Piperine through regulating MAS-related G protein-coupled receptor X2 activation. Phytother. Res. 2020, 34, 1409–1420. [Google Scholar] [CrossRef]
- Hou, Y.; Che, D.; Ma, P.; Zhao, T.; Zeng, Y.; Wang, N. Anti-pseudo-allergy effect of isoliquiritigenin is MRGPRX2-dependent. Immunol. Lett. 2018, 198, 52–59. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.; Li, C.; Ding, Y.; Hu, S.; An, H. Inhibitory function of Shikonin on MRGPRX2-mediated pseudo-allergic reactions induced by the secretagogue. Phytomedicine 2020, 68, 153149. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, J.; Ge, S.; Zeng, Y.; Wang, N.; Wu, Y. Roxithromycin inhibits compound 48/80-induced pseudo-allergy via the MrgprX2 pathway both in vitro and in vivo. Cell. Immunol. 2020, 358, 104239. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.; Wang, J.; Liu, R.; Zhang, G.; Dong, K.; Zhang, T. Paeoniflorin inhibits MRGPRX2-mediated pseudo-allergic reaction via calcium signaling pathway. Phytother. Res. 2020, 34, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Che, D.; Li, C.; Cao, J.; Wang, J.; Ma, P.; Zhao, T.; An, H.; Zhang, T. Quercetin inhibits Mrgprx2-induced pseudo-allergic reaction via PLCgamma-IP3R related Ca(2+) fluctuations. Int. Immunopharmacol. 2019, 66, 185–197. [Google Scholar] [CrossRef]
- Kumar, M.; Singh, K.; Duraisamy, K.; Allam, A.A.; Ajarem, J.; Kwok Chong Chow, B. Protective Effect of Genistein against Compound 48/80 Induced Anaphylactoid Shock via Inhibiting MAS Related G Protein-Coupled Receptor X2 (MRGPRX2). Molecules 2020, 25, 1028. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Liu, S.; Ogasawara, T.; Sawasaki, T.; Takasaki, Y.; Yorozuya, T.; Mogi, M. A novel MRGPRX2-targeting antagonistic DNA aptamer inhibits histamine release and prevents mast cell-mediated anaphylaxis. Eur. J. Pharmacol. 2020, 878, 173104. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.; Hu, S.; Ge, S.; Jia, M.; Wang, N. Resveratrol inhibits MRGPRX2-mediated mast cell activation via Nrf2 pathway. Int. Immunopharmacol. 2021, 93, 107426. [Google Scholar] [CrossRef]
- Huang, Y.; Huang, L.; Zhu, G.; Pei, Z.; Zhang, W. Downregulated microRNA-27b attenuates lipopolysaccharide-induced acute lung injury via activation of NF-E2-related factor 2 and inhibition of nuclear factor kappaB signaling pathway. J. Cell. Physiol. 2019, 234, 6023–6032. [Google Scholar] [CrossRef]
- Xue, Z.; Zhang, Y.; Zeng, Y.; Hu, S.; Bai, H.; Wang, J.; Jing, H.; Wang, N. Licochalcone A inhibits MAS-related GPR family member X2-induced pseudo-allergic reaction by suppressing nuclear migration of nuclear factor-kappaB. Phytother Res. 2021. [Google Scholar] [CrossRef]
- Lv, H.; Yang, H.; Wang, Z.; Feng, H.; Deng, X.; Cheng, G.; Ci, X. Nrf2 signaling and autophagy are complementary in protecting lipopolysaccharide/d-galactosamine-induced acute liver injury by licochalcone A. Cell Death Dis. 2019, 10, 313. [Google Scholar] [CrossRef]
- Callahan, B.N.; Kammala, A.K.; Syed, M.; Yang, C.; Occhiuto, C.J.; Nellutla, R.; Chumanevich, A.P.; Oskeritzian, C.A.; Das, R.; Subramanian, H. Osthole, a Natural Plant Derivative Inhibits MRGPRX2 Induced Mast Cell Responses. Front. Immunol. 2020, 11, 703. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Lv, Y.; Kong, L.; Sun, Y.; Fu, J.; Li, L.; He, L. Simultaneous identification of the anaphylactoid components from traditional Chinese medicine injections using rat basophilic leukemia 2H3 and laboratory of allergic disease 2 dual-mixed/cell membrane chromatography model. Electrophoresis 2018, 39, 1181–1189. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Sato, H.; Sakamaki, K.; Kamada, M.; Okuno, Y.; Fukuishi, N.; Furuta, K.; Tanaka, S. Suppression of IgE-Independent Degranulation of Murine Connective Tissue-Type Mast Cells by Dexamethasone. Cells 2019, 8, 112. [Google Scholar] [CrossRef]
- Syed, M.; Kammala, A.K.; Callahan, B.; Oskeritzian, C.A.; Subramanian, H. Lactic acid suppresses MRGPRX2 mediated mast cell responses. Cell. Immunol. 2021, 368, 104422. [Google Scholar] [CrossRef]
- Izawa, K.; Isobe, M.; Matsukawa, T.; Ito, S.; Maehara, A.; Takahashi, M.; Yamanishi, Y.; Kaitani, A.; Oki, T.; Okumura, K.; et al. Sphingomyelin and ceramide are physiological ligands for human LMIR3/CD300f, inhibiting FcepsilonRI-mediated mast cell activation. J. Allergy Clin. Immunol. 2014, 133, 270–273. [Google Scholar] [CrossRef]
- Dondalska, A.; Rönnberg, E.; Ma, H.; Pålsson, S.A.; Magnusdottir, E.; Gao, T.; Adam, L.; Lerner, E.A.; Nilsson, G.; Lagerström, M.; et al. Amelioration of Compound 48/80-Mediated Itch and LL-37-Induced Inflammation by a Single-Stranded Oligonucleotide. Front. Immunol. 2020, 11, 559589. [Google Scholar] [CrossRef]
- Fernandopulle, N.A.; Zhang, S.S.; Soeding, P.F.; Mackay, G.A. MRGPRX2 activation in mast cells by neuromuscular blocking agents and other agonists: Modulation by sugammadex. Clin. Exp. Allergy 2021, 51, 685–695. [Google Scholar] [CrossRef]
- Meghwal, M.; Goswami, T.K. Piper nigrum and piperine: An update. Phytother. Res. 2013, 27, 1121–1130. [Google Scholar] [CrossRef]
- Peng, F.; Du, Q.; Peng, C.; Wang, N.; Tang, H.; Xie, X.; Shen, J.; Chen, J. A review: The pharmacology of isoliquiritigenin. Phytother. Res. 2015, 29, 969–977. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.B.; Graham, T.A.; Azimi, E.; Lerner, E.A. A single amino acid in MRGPRX2 necessary for binding and activation by pruritogens. J. Allergy Clin. Immunol. 2017, 140, 1726–1728. [Google Scholar] [CrossRef]
- Lansu, K.; Karpiak, J.; Liu, J.; Huang, X.P.; McCorvy, J.D.; Kroeze, W.K.; Che, T.; Nagase, H.; Carroll, F.I.; Jin, J.; et al. In silico design of novel probes for the atypical opioid receptor MRGPRX2. Nat. Chem. Biol. 2017, 13, 529–536. [Google Scholar] [CrossRef]
- Mencarelli, A.; Gunawan, M.; Yong, K.S.M.; Bist, P.; Tan, W.W.S.; Tan, S.Y.; Liu, M.; Huang, E.K.; Fan, Y.; Chan, J.K.Y.; et al. A humanized mouse model to study mast cells mediated cutaneous adverse drug reactions. J. Leukoc. Biol. 2020, 107, 797–807. [Google Scholar] [CrossRef] [PubMed]
- Takamori, A.; Izawa, K.; Kaitani, A.; Ando, T.; Okamoto, Y.; Maehara, A.; Tanabe, A.; Nagamine, M.; Yamada, H.; Uchida, S.; et al. Identification of inhibitory mechanisms in pseudo-allergy involving Mrgprb2/MRGPRX2-mediated mast cell activation. J. Allergy Clin. Immunol. 2019, 143, 1231–1235. [Google Scholar] [CrossRef]
- Shimada, I.; Ueda, T.; Kofuku, Y.; Eddy, M.T.; Wüthrich, K. GPCR drug discovery: Integrating solution NMR data with crystal and cryo-EM structures. Nat. Rev. Drug Discov. 2019, 18, 59–82. [Google Scholar] [CrossRef]
- Sasaki, E.; Suemizu, H.; Shimada, A.; Hanazawa, K.; Oiwa, R.; Kamioka, M.; Tomioka, I.; Sotomaru, Y.; Hirakawa, R.; Eto, T.; et al. Generation of transgenic non-human primates with germline transmission. Nature 2009, 459, 523–527. [Google Scholar] [CrossRef]
- Sato, K.; Oiwa, R.; Kumita, W.; Henry, R.; Sakuma, T.; Ito, R.; Nozu, R.; Inoue, T.; Katano, I.; Sato, K.; et al. Generation of a Nonhuman Primate Model of Severe Combined Immunodeficiency Using Highly Efficient Genome Editing. Cell Stem Cell 2016, 19, 127–138. [Google Scholar] [CrossRef] [PubMed]

| Type | Inhibitor | Mechanism | References |
|---|---|---|---|
| Direct inhibition | QWF | Dual action to MRGPRX2 and NK-1 receptor Competitive inhibition of SP binding to MRGPRX2 Inhibition of intracellular Ca2+ mobilization and mast cell degranulation Reduction of SP-induced itch | [36] |
| Compound 1, 2 | MRGPRX2 antagonist, not for NK-1 or M2R Competitive inhibition of SP binding to MRGPRX2 Inhibition of intracellular Ca2+ mobilization, ERK signaling, GTP binding, MC degranulation, and de novo PGD2 production | [32,42] | |
| Piperine | Direct interaction to MRGPRX2 determined by binding to MRGPRX2-expressing cell membrane Inhibition of intracellular Ca2+ mobilization, degranulation, histamine release, cytokines release, PLCγ1, PKC, inositol 1,4,5-triphate receptor, p38, PKB, and ERK Reduction of C48/80-induced anaphylactoid reactions | [141] | |
| Isoliquiritigenin | Predicted direct interaction to MRGPRX2 Binding to MRGPRX2 by molecular docking assay Inhibition of intracellular Ca2+ mobilization, cytokines release, MC degranulation, and histamine release Reduction of C48/80-induced anaphylactoid reactions | [142] | |
| Shikonin | Direct interaction to MRGPRX2 determined by surface plasmon resonance and molecular docking analysis Inhibition of intracellular Ca2+ mobilization, MC degranulation, histamine release, cytokines release, PLCγ1, PKC, inositol 1,4,5-triphate receptor, and ERK Reduction of C48/80-induced anaphylactoid reaction | [143] | |
| Imperatorin | Direct interaction to MRGPRX2 determined by surface plasmon resonance and molecular docking analysis Inhibition of intracellular Ca2+ mobilization, MC degranulation, histamine release, cytokines release, CamKII, and ERK Reduction of SP-induced anaphylactoid reaction and OVA-induced lung inflammtion | [50] | |
| Roxithromysin | Direct interaction to MRGPRX2 by surface plasmon resonance Inhibition of intracellular Ca2+ mobilization, MC degranulation, histamine release, cytokines release, PLCγ1, inositol 1,4,5-triphate receptor, and p38 Reduction of C48/80-induced anaphylactoid reaction | [144] | |
| Paeoniflorin | Predicted direct interaction to MRGPRX2 Binding to MRGPRX2 by molecular docking analysis Inhibition of intracellular Ca2+ mobilization, MC degranulation, histamine release, cytokines release, PLCγ1, inositol 1,4,5-triphate receptor, p38, ERK, AKT, and PKC Reduction of C48/80-induced anaphylactoid reaction | [145] | |
| Quercetin | Predicted direct interaction to MRGPRX2 Binding to MRGPRX2 by molecular docking analysis Inhibition of intracellular Ca2+ mobilization, MC degranulation, histamine release, cytokines release, PLCγ1, inositol 1,4,5-triphate receptor, and ERK Reduction of C48/80-induced anaphylactoid reaction | [146] | |
| Genistein | Predicted direct interaction to MRGPRX2 Binding to MRGPRX2 by molecular docking analysis Inhibition of intracellular Ca2+ mobilization, β-arrestin recruitment, and MC degranulation Reduction of C48/80-induced anaphylactoid reaction | [147] | |
| Aptamer-X35 | ssDNA aptamer Direct interaction to MRGPRX2 determined by binding to MRGPRX2 expressing cells Inhibition of MC degranulation and histamine release Reduction of SP-induced anaphylactoid reaction in MRGPRX2-expressing cell engraft rat | [148] | |
| MRGPRX2 signal inhibitor | Resveratrol | Inhibition of MRGPRX2 downstream signal, NF-κB via Nrf2/HO-1 pathway activation Inhibition of intracellular Ca2+ mobilization, MC degranulation, histamine release, and cytokines release Reduction of C48/80-induced anaphylactoid reaction | [149,150] |
| Licochalcone A | Inhibition of MRGPRX2 downstream signal, NF-κB Inhibition of intracellular Ca2+ mobilization, MC degranulation, and cytokines release Reduction of SP-induced anaphylactoid reaction | [151,152] | |
| Osthole | Inhibition of MRGPRX2-mediated intracellular Ca2+ mobilization, ERK, MC degranulation, and cytokines release Reduction of C48/80-induced paw edema and LL-37-induced inflammation in vivo | [153] | |
| Dexamethasone | Downregulation of Gαi and MRGPRX2 downstream signals | [154] | |
| Lactic acid | Inhibition of MRGPRX2-mediated intracellular Ca2+ mobilization, MC degranulation, and cytokines release Reduction of LL-37-induced inflammation in vivo | [155] | |
| Others | Ceramide, sphingomyelin | Inhibition of MRGPRX2-mediated mast cell activation by CD300f activation Reduction of MRGPRX2 ligands-induced inflammation in vivo | [156,157] |
| ssDNA | Unknown mechanism Reduction of MRGPRX2 ligands-induced inflammation in vivo | [158] | |
| Sugammadex | Encapsulation of MRGPRX2 ligand Inhibition of intracellular Ca2+ mobilization, MC degranulation, and CCL2 release | [159] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ogasawara, H.; Noguchi, M. Therapeutic Potential of MRGPRX2 Inhibitors on Mast Cells. Cells 2021, 10, 2906. https://doi.org/10.3390/cells10112906
Ogasawara H, Noguchi M. Therapeutic Potential of MRGPRX2 Inhibitors on Mast Cells. Cells. 2021; 10(11):2906. https://doi.org/10.3390/cells10112906
Chicago/Turabian StyleOgasawara, Hiroyuki, and Masato Noguchi. 2021. "Therapeutic Potential of MRGPRX2 Inhibitors on Mast Cells" Cells 10, no. 11: 2906. https://doi.org/10.3390/cells10112906
APA StyleOgasawara, H., & Noguchi, M. (2021). Therapeutic Potential of MRGPRX2 Inhibitors on Mast Cells. Cells, 10(11), 2906. https://doi.org/10.3390/cells10112906






