White Matter Interstitial Neurons in the Adult Human Brain: 3% of Cortical Neurons in Quest for Recognition
Abstract
1. Introduction
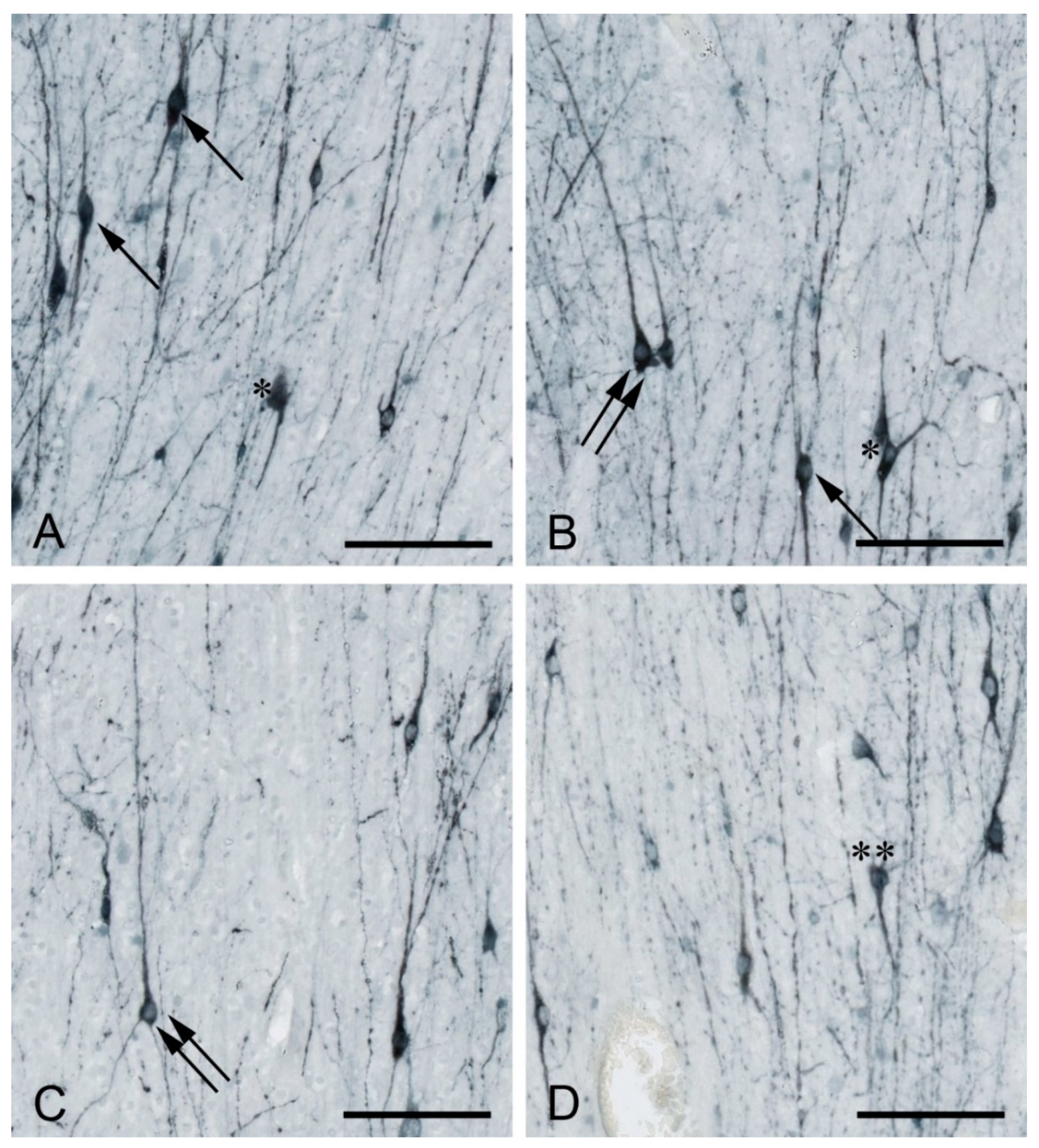
2. Developmental Origin, Morphology, and Molecular Profile of WMIN
3. Total Neuronal Number, Density, and Spatial Distribution of WMIN
4. Functional Importance of WMIN
5. Pathology of WMIN
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Colombo, J.A. Cellular complexity in subcortical white matter: A distributed control circuit. Brain Struct. Funct. 2018, 223, 981–985. [Google Scholar] [CrossRef] [PubMed]
- Meynert, T. Der Bau der Grosshirnrinde und seine ortlichen Verschiedenheiten, nebst einem pathologisch-anatomischen Corollarium. Vierteljschr. Psychiat. 1867, 1, 77–93, 126–170, 198–217. [Google Scholar]
- Judaš, M.; Sedmak, G.; Pletikos, M. Early history of subplate and interstitial neurons: From Theodor Meynert (1867) to the discovery of the subplate zone (1974). J. Anat. 2010, 217, 344–367. [Google Scholar] [CrossRef] [PubMed]
- Ranke, O. Beitrage zur Kenntnis der normalen und pathologischen Hirnrindenbildung. Beitr. Pathol. Anat. 1910, 47, 51–125. [Google Scholar]
- Kostović, I.; Molliver, M. A new interpretation of the laminar development of cerebral cortex: Synaptogenesis in different layers of neopallium in the human fetus. Anat. Rec. 1974, 178, 395. [Google Scholar]
- Kostović, I.; Rakic, P. Developmental history of the transient subplate zone in visual and somatosensory cortex of the macaque monkey and human brain. J. Comp. Neurol. 1990, 297, 441–470. [Google Scholar] [CrossRef]
- Allendoerfer, K.L.; Shatz, C.J. The subplate, a transient neocortical structure: Its role in the development of connections between thalamus and cortex. Annu. Rev. Neurosci. 1994, 17, 185–218. [Google Scholar] [CrossRef]
- Kanold, P.O.; Luhmann, H.J. The subplate and early cortical circuits. Annu. Rev. Neurosci. 2010, 33, 23–48. [Google Scholar] [CrossRef]
- Kostović, I.; Rakic, P. Cytology and time origin of interstitial neurons in the white matter in infant and adult human and monkey telencephalon. J. Neurocytol. 1980, 9, 219–242. [Google Scholar] [CrossRef]
- Luskin, M.B.; Shatz, C.J. Studies of the earliest generated cells of the cat’s visual cortex: Cogeneration of subplate and marginal zone. J. Neurosci. 1985, 4, 1062–1075. [Google Scholar] [CrossRef]
- Chun, J.J.M.; Shatz, C.J. The earliest-generated neurons of the cat cerebral cortex: Characterization by MAP2 and neurotransmitter immunohistochemistry during fetal life. J. Neurosci. 1989, 9, 1648–1667. [Google Scholar] [CrossRef]
- Chun, J.J.M.; Shatz, C.J. Interstitial cells of the adult neocortical white matter are the remnant of the early generated subplate neuron population. J. Comp. Neurol. 1989, 282, 555–569. [Google Scholar] [CrossRef] [PubMed]
- Valverde, F.; Facal-Valverde, M.V.; Santacana, M.; Heredia, M. Development and differentiation of early generated cells of sublayer IVb in the somatosensory cortex of the rat: A correlated Golgi and autoradiographic study. J. Comp. Neurol. 1989, 290, 118–140. [Google Scholar] [CrossRef] [PubMed]
- Mrzljak, L.; Uylings, H.B.; Kostovic, I.; Van Eden, C.G. Prenatal development of neurons in the human prefrontal cortex: I. A qualitative Golgi study. J. Comp. Neurol. 1988, 271, 355–386. [Google Scholar] [CrossRef] [PubMed]
- Valverde, F.; Facal-Valverde, M.V. Postnatal development of interstitial (subplate) cells in the white matter of the temporal cortex of kittens: A correlated Golgi and electron microscopy study. J. Comp. Neurol. 1988, 269, 168–192. [Google Scholar] [CrossRef]
- Meyer, G.; Wahle, P.; Castaneyra-Perdomo, A.; Ferres-Torres, R. Morphology of neurons in the white matter of the adult human neocortex. Exp. Brain Res. 1992, 88, 204–212. [Google Scholar] [CrossRef]
- Garcia-Marin, V.; Blazquez-Llorca, L.; Rodriguez, J.R.; Gonzalez-Soriano, J.; DeFelipe, J. Differential distribution of neurons in the gyral white matter of the human cerebral cortex. J. Comp. Neurol. 2010, 518, 4740–4759. [Google Scholar] [CrossRef]
- Judaš, M.; Sedmak, G.; Pletikos, M.; Jovanov-Milošević, N. Population of subplate and interstitial neurons in fetal and adult human telencephalon. J. Anat. 2010, 217, 381–399. [Google Scholar] [CrossRef]
- Hoerder-Suabedissen, A.; Molnar, Z. Molecular diversity of early-born subplate neurons. Cereb. Cortex 2013, 23, 1473–1483. [Google Scholar] [CrossRef]
- Kostović, I.; Rakic, P. Development of prestriate visual projections in the monkey and human fetal cerebrum revealed by transient cholinesterase staining. J. Neurosci. 1984, 4, 25–42. [Google Scholar] [CrossRef]
- Valverde, F.; Lopez-Mascaraque, L.; De Carlos, J.A.S. Persistence of early-generated neurons in the rodent subplate: Assessment of cell death in neocortex during the early postnatal period. J. Neurosci. 1995, 15, 5014–5024. [Google Scholar] [CrossRef] [PubMed]
- Robertson, R.T.; Annis, C.M.; Baratta, J.; Haraldson, S.; Ingeman, J.; Kageyama, G.H.; Kimm, E.; Yu, J. Do subplate neurons comprise a transient population of cells in developing neocortex of rats? J. Comp. Neurol. 2000, 426, 632–650. [Google Scholar] [CrossRef]
- Kostović, I.; Sedmak, G.; Judaš, M. Neural histology and neurogenesis of the human fetal and infant brain. Neuroimage 2019, 188, 743–773. [Google Scholar] [CrossRef] [PubMed]
- Pedraza, M.; Hoerder-Suabedissen, A.; Albert-Maestro, M.A.; Molnar, Z.; De Carlos, J.A. Extracortical origin of some murine subplate cell population. Proc. Natl. Acad. Sci. USA 2014, 111, 8613–8618. [Google Scholar] [CrossRef] [PubMed]
- Frazer, S.; Prados, J.; Niquille, M.; Cadilhac, C.; Markopoulos, F.; Gomez, L.; Tomasello, U.; Telley, L.; Holtmaat, A.; Jabaudon, D.; et al. Transcriptomic and anatomic parcellation of 5-HT3AR expressing cortical interneuron subtypes revealed by single-cell RNA sequencing. Nat. Commun. 2017, 8, 14219. [Google Scholar] [CrossRef]
- Viswanathan, S.; Sheikh, A.; Looger, L.L.; Kanold, P.O. Molecularly defined subplate neurons project both to thalamocortical recipient layers and thalamus. Cereb. Cortex 2017, 27, 4759–4768. [Google Scholar] [CrossRef]
- Judaš, M.; Sedmak, G.; Kostović, I. The significance of the subplate for evolution and developmental plasticity of the human brain. Front. Hum. Neurosci. 2013, 7, 423. [Google Scholar] [CrossRef]
- Duque, A.; Krsnik, Ž.; Kostović, I.; Rakic, P. Secondary expansion of the transient subplate zone in the developing cerebrum of human and nonhuman primates. Proc. Natl. Acad. Sci. USA 2016, 113, 9892. [Google Scholar] [CrossRef]
- Reep, R.L. Cortical layer VII and persistent subplate cells in mammalian brains. Brain Behav. Evol. 2000, 56, 212–234. [Google Scholar] [CrossRef]
- Clancy, B.; Silva-Filho, M.; Friedlander, M.J. Structure and projections of white matter neurons in the postnatal rat visual cortex. J. Comp. Neurol. 2001, 434, 233–252. [Google Scholar] [CrossRef]
- Suarez-Sola, M.L.; Gonzales-Delgado, F.J.; Pueyo-Morlans, M.; Medina-Bolivar, O.C.; Hernandez-Acosta, N.C.; Gonzales-Gomez, M.; Meyer, G. Neurons in the white matter of the adult human neocortex. Front. Neuroanat. 2009, 3, 7. [Google Scholar] [CrossRef]
- Sedmak, G.; Judaš, M. The total number of white matter interstitial neurons in the human brain. J. Anat. 2019, 235, 626–636. [Google Scholar] [CrossRef] [PubMed]
- Okhotin, V.E.; Kalinichenko, S.G. Subcortical white matter interstitial cells: Their connections, neurochemical specialization, and role in the histogenesis of the cortex. Neurosci. Behav. Physiol. 2003, 33, 177–194. [Google Scholar] [CrossRef] [PubMed]
- Eastwood, S.L.; Harrison, P.J. Interstitial white matter neurons express less reelin and are abnormally distributed in schizophrenia: Towards an integration of molecular and morphologic aspects of the neurodevelopmental hypothesis. Mol. Psychiatry 2003, 8, 769, 821–831. [Google Scholar] [CrossRef][Green Version]
- Anderson, S.A.; Volk, D.W.; Lewis, D.A. Increased density of microtubule associated protein 2-immunoreactive neurons in the prefrontal white matter of schizophrenic subjects. Schizophr. Res. 1996, 19, 111–119. [Google Scholar] [CrossRef]
- Chan-Palay, V.; Allen, Y.S.; Lang, W.; Haesler, U.; Polak, J.M. I. Cytology and distribution in normal human cerebral cortex of neurons immunoreactive with antisera against neuropeptide Y. J. Comp. Neurol. 1985, 238, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Berman, N.E.J.; Fredrickson, E. Morphology and laminar distribution of neuropeptide Y immunoreactive neurons in the human striate cortex. Synapse 1992, 11, 20–27. [Google Scholar] [CrossRef]
- Fischer, H.C.; Kuljis, R.O. Multiple types of nitrogen monoxide synthase-/NADPH diaphorase-containing neurons in the human cerebral neocortex. Brain Res. 1994, 654, 105–117. [Google Scholar] [CrossRef]
- Delalle, I.; Evers, P.; Kostović, I.; Uylings, H.B.M. Laminar distribution of neuropeptide Y-immunoreactive neurons in human prefrontal cortex during development. J. Comp. Neurol. 1997, 379, 515–522. [Google Scholar] [CrossRef]
- Uylings, H.B.M.; Delalle, I. Morphology of neuropeptide Y-immunoreactive neurons and fibers in human prefrontal cortex during prenatal and postnatal development. J. Comp. Neurol. 1997, 379, 523–540. [Google Scholar] [CrossRef]
- Ang, L.C.; Shul, D.D. Peptidergic neurons of subcortical white matter in aging and Alzheimer’s brain. Brain Res. 1995, 674, 329–335. [Google Scholar] [CrossRef]
- Kostović, I.; Štefulj-Fučić, A.; Mrzljak, L.; Jukić, S.; Delalle, I. Prenatal and perinatal development of the somatostatin-immunoreactive neurons in the human prefrontal cortex. Neurosci. Lett. 1991, 124, 153–156. [Google Scholar] [CrossRef]
- Kowall, N.W.; Quigley, B.J.; Krause, J.E.; Lu, F.; Kosofsky, B.E.; Ferrante, R.J. Substance P and substance P receptor histochemistry in human neurodegenerative disease. Reg. Pep. 1993, 46, 174–185. [Google Scholar] [CrossRef]
- Akbarian, S.; Bunney, W.E.; Potkin, S.G.; Wigal, S.B.; Hagman, J.O.; Sandman, C.A.; Jones, E.G. Altered distribution of nicotinamide adenine dinucleotide phosphate-diaphorase cells in frontal lobe of schizophrenics implies disturbances of cortical development. Arch. Gen. Psychiat. 1993, 50, 169–177. [Google Scholar] [CrossRef]
- Akbarian, S.; Vinuela, A.; Kim, J.J.; Potkin, S.G.; Bunney, W.E.; Jones, E.G. Distorted distribution of nicotinamide-adenine dinucleotide phosphate-diaphorase neurons in temporal lobe of schizophrenics implies anomalous cortical development. Arch. Gen. Psychiat. 1993, 50, 178–187. [Google Scholar] [CrossRef]
- DeFelipe, J. A study of NADPH diaphorase-positive axonal plexuses in the human temporal cortex. Brain Res. 1993, 615, 342–346. [Google Scholar] [CrossRef][Green Version]
- Smiley, J.F.; Levey, A.I.; Mesulam, M.M. Infracortical interstitial cells concurrently expression m2-muscarinic receptors, acetylcholinesterase and nicotinamide adenine dinucleotide phosphate-diaphorase in the human and monkey cerebral cortex. Neuroscience 1998, 84, 755–769. [Google Scholar] [CrossRef]
- Judaš, M.; Šestan, N.; Kostović, I. Nitrinergic neurons in the developing and adult human telencephalon: Transient and permanent patterns of expression in comparison to other mammals. Microsc. Res. Tech. 1999, 45, 401–419. [Google Scholar] [CrossRef]
- Judaš, M.; Šimić, G.; Petanjek, Z.; Jovanov-Milošević, N.; Pletikos, M.; Vasung, L.; Vukšić, M.; Kostović, I. The Zagreb Collection of human brains: A unique, versatile, but underexploited resource for the neuroscience community. Ann. N. Y. Acad. Sci. 2011, 1225 (Suppl. 1), E105–E130. [Google Scholar] [CrossRef]
- Shering, A.F.; Lowenstein, P.R. Neocortex provides direct synaptic input to interstitial neurons of the Intermediate zone of kittens and white matter of cats: A light and electron microscopy study. J. Comp. Neurol. 1994, 347, 433–443. [Google Scholar] [CrossRef]
- Meyer, G.; Gonzales-Hernandez, T.; Galindo-Mireles, D.; Castaneyra-Perdomo, A.; Ferres-Torres, R. The efferent projections of neurons in the white matter of different cortical areas of the adult rat. Anat. Embryol. 1991, 184, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Clancy, B.; Cauller, L.J. Widespread projections form subgriseal neurons (layer VII) to layer I in adult rat cortex. J. Comp. Neurol. 1999, 407, 275–286. [Google Scholar] [CrossRef]
- Von Monakow, C. Gehirnpathologie; Alferd Holder: Wien, Austria, 1905. [Google Scholar]
- Rojiani, A.M.; Emery, J.A.; Anderson, K.J.; Massey, J.K. Distribution of heterotopic neurons in normal hemispheric white matter: A morphometric analysis. J. Neuropathol. Exp. Neurol. 1996, 55, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Richter, Z.; Janszky, J.; Setalo, G.; Horvath, R.; Horvath, Z.; Doczi, T.; Seress, L.; Abraham, H. Characterization of neurons in the cortical white matter in human temporal lobe epilepsy. Neuroscience 2016, 333, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Mortazavi, F.; Wang, X.; Rosene, D.L.; Rockland, K.S. White matter neurons in young adult and aged rhesus monkey. Front. Neuroanat. 2016, 10, 15. [Google Scholar] [CrossRef]
- Nykjaer, C.H.; Brudek, T.; Salvesen, L.; Pakkenberg, B. Changes in the cell population in brain white matter in multiple system atrophy. Mov. Disord. 2017, 32, 1074–1082. [Google Scholar] [CrossRef]
- Swiegers, J.; Bhagwandin, A.; Sherwood, C.C.; Bertelsen, M.F.; Maseko, B.C.; Hemingway, J.; Rockland, K.S.; Molnar, Z.; Manger, P.R. The distribution, number and certain neurochemical identities of infracortical white matter neurons in a lar gibbon (Hylobater lar) brain. J. Comp. Neurol. 2019, 527, 1633–1653. [Google Scholar] [CrossRef]
- Meencke, H.J. The density of dystopic neurons in the white matter of the gyrus frontalis in epilepsies. J. Neurol. 1983, 230, 171–181. [Google Scholar] [CrossRef]
- Eastwood, S.L.; Harrison, P.J. Interstitial white matter neuron density in the dorsolateral prefrontal cortex and parahippocampal gyrus in schizophrenia. Schizophr. Res. 2005, 79, 181–188. [Google Scholar] [CrossRef]
- Wegiel, J.; Flory, M.; Kuchna, I.; Nowicki, K.; Ma, S.Y.; Imaki, H.; Wegiel, J.; Cohen, I.L.; London, E.; Wisniewski, T.; et al. Stereological study of the neuronal number and volume of 38 brain subdivisions of subjects diagnosed with autism reveals significant alterations restricted to the striatum, amygdala and cerebellum. Acta Neuropathol. Commun. 2014, 2, 141. [Google Scholar] [CrossRef]
- Herculano-Houzel, S. The Human Advantage. A New Understanding of How Our Brain Became Remarkable; The MIT Press: Cambridge, MA, USA, 2016; pp. 217–226. [Google Scholar]
- von Bartheld, C.S.; Bahney, J.; Herculano-Houzel, S. The search for true numbers of neurons and glial cells in the human brain: A review of 150 years of cell counting. J. Comp. Neurol. 2016, 524, 3865–3895. [Google Scholar] [CrossRef] [PubMed]
- Dallerac, G.; Zapata, J.; Rouach, N. Versatile control of synaptic circuits by astrocytes: Where, when and how? Nat. Rev. Neurosci. 2018, 19, 729–743. [Google Scholar] [CrossRef] [PubMed]
- Marinelli, S.; Basilico, B.; Marrone, M.C.; Ragozzino, D. Microglia-neuron crosstalk: Signaling mechanism and control of synaptic transmission. Semin. Cell Dev. Biol. 2019, 94, 138–151. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.D.; Risher, W.C.; Risher, M.L. Regulation of synaptic development by astrocyte signaling factors and their emerging roles in substance abuse. Cells 2020, 9, 297. [Google Scholar] [CrossRef]
- Kim, Y.S.; Choi, J.; Yoon, B.E. Neuron-Glia interactions in neurodevelopmental disorders. Cells 2020, 9, 2176. [Google Scholar] [CrossRef] [PubMed]
- Judaš, M.; Radoš, M.; Jovanov-Milošević, N.; Hrabač, P.; Štern-Padovan, R.; Kostović, I. Structural, immunocytochemical, and MR imaging properties of periventricular crossroads of growing cortical pathways in preterm infants. AJNR Am. J. Neuroradiol. 2005, 26, 2671–2684. [Google Scholar] [PubMed]
- Tomioka, R.; Okamoto, K.; Furuta, T.; Fujiyama, F.; Iwasato, T.; Yanagawa, Y.; Obata, K.; Kaneko, T.; Tamamaki, N. Demonstration of long-range GABAergic connections distributed throughout the mouse neocortex. Eur. J. Neurosci. 2005, 21, 1587–1600. [Google Scholar] [CrossRef]
- Tomioka, R.; Rockland, K.S. Long-distance corticocortical GABAergic neurons in the adult monkey white and grey matter. J. Comp. Neurol. 2007, 505, 526–538. [Google Scholar] [CrossRef]
- Torres-Reveron, J.; Friedlander, M.J. Properties of persistent postnatal cortical subplate neurons. J. Neurosci. 2007, 27, 9962–9974. [Google Scholar] [CrossRef]
- Von Engelhardt, J.; Khrulev, S.; Eliava, M.; Wahlster, S.; Monyer, H. 5-HT(3A) receptor-bearing white matter interstitial GABAergic interneurons are functionally integrated into cortical and subcortical networks. J. Neurosci. 2011, 31, 16844–16854. [Google Scholar] [CrossRef]
- Regidor, J.; Edvinsson, L.; Divac, I. NOS neurones lie near branching of cortical arteriolae. Neuroreport 1993, 4, 112–114. [Google Scholar] [CrossRef] [PubMed]
- Okhotin, V.E.; Kupriyanov, V.V. Neurovascular relationships in the human neocortex. Neurosci. Behav. Physiol. 1997, 27, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Estrada, C.; De Felipe, J. Nitric oxide-producing neurons in the neocortex: Morphological and functional relationship with intraparenchymal microvasculature. Cereb. Cortex 1998, 8, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Cauli, B.; Hamel, E. Revisiting the role of neurons in neurovascular coupling. Front. Neuroenerg. 2010, 2, 9. [Google Scholar] [CrossRef]
- Kilduff, T.S.; Cauli, B.; Gerashchenko, D. Activation of cortical interneurons during sleep: An anatomical link to homeostatic sleep regulation. Trends Neurosci. 2011, 34, 10–19. [Google Scholar] [CrossRef]
- Kostović, I.; Judaš, M.; Sedmak, G. Developmental history of the subplate zone, subplate neurons and interstitial white matter neurons: Relevance for schizophrenia. Int. J. Dev. Neurosci. 2011, 29, 193–205. [Google Scholar] [CrossRef]
- Hoerder-Suabedissen, A.; Hayashi, S.; Upton, L.; Nolan, Z.; Casas-Torremocha, D.; Grant, E.; Viswanathan, S.; Kanold, P.O.; Clasca, F.; Kim, Y.; et al. Subset of cortical layer 6b neurons selectively innervate higher order thalamic nuclei in mice. Cereb. Cortex 2018, 28, 1882–1897. [Google Scholar] [CrossRef]
- Hardiman, O.; Burke, T.; Philips, J.; Murphy, S.; O’Moore, B.; Staunton, H.; Farrell, M.A. Microdysgenesis in resected temporal neocortex: Incidence and clinical significance in focal epilepsy. Neurology 1988, 38, 1041–1047. [Google Scholar] [CrossRef]
- Emery, J.A.; Roper, S.N.; Rojiani, A.M. White matter neuronal heterotopia in temporal lobe epilepsy: A morphometric and immunohistochemical study. J. Neuropathol. Exp. Neurol. 1997, 56, 1276–1282. [Google Scholar] [CrossRef]
- Kasper, B.S.; Stefan, H.; Buchfelder, M.; Paulus, W. Temporal lobe microdysgenesis in epilepsy versus control brains. J. Neuropathol. Exp. Neurol. 1999, 58, 22–28. [Google Scholar] [CrossRef][Green Version]
- Thom, M.; Sisodiya, S.; Harkness, W.; Scaravilli, F. Microdysgenesis in temporal lobe epilepsy. A quantitative and immunohistochemical study of white matter neurons. Brain 2001, 124, 2299–2309. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.Y.; Ellis, M.; Brooke-Ball, H.; de Tisi, J.; Eriksson, S.H.; Brandner, S.; Sisodiya, S.M.; Thom, M. High-throughput, automated, quantification of white matter neurons in mild malformation of cortical development in epilepsy. Acta Neuropathol. Commun. 2014, 2, 72. [Google Scholar] [CrossRef] [PubMed]
- Akbarian, S.; Kim, J.J.; Potkin, S.G. Maldistribution of interstitial neurons in prefrontal white matter of the brains of schizophrenic patients. Arch. Gen. Psychiatry 1996, 53, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Kirkpatrick, B.; Conley, R.C.; Kakoyannis, A.; Reep, R.L.; Roberts, R.C. Interstitial cells of the white matter in the inferior parietal cortex in schizophrenia: An unbiased cell-counting study. Synapse 1999, 34, 95–102. [Google Scholar] [CrossRef]
- Kirkpatrick, B.; Messias, N.C.; Conley, R.R.; Roberts, R.C. Interstitial cells of the white matter in the dorsolateral prefrontal cortex in deficit and nondeficit schizophrenia. J. Nerv. Ment. Dis. 2003, 191, 563–567. [Google Scholar] [CrossRef]
- Beasley, C.L.; Cotter, D.R.; Everall, P. Density and distribution of white matter neurons in schizophrenia, bipolar disorder and major depressive disorder: No evidence for abnormalities of neuronal migration. Mol. Psychiatry 2002, 7, 564–570. [Google Scholar] [CrossRef]
- Eastwood, S.L.; Harrison, P.J. Cellular basis of reduced cortical reelin expression in schizophrenia. Am. J. Psychiatry 2006, 163, 540–542. [Google Scholar] [CrossRef]
- Rioux, L.; Nissanov, J.; Katherine, L.; Bilker, W.B.; Arnold, S.E. Distribution of microtubule-associated protein MAP2-immunoreactive interstitial neurons in the parahippocampal white matter in subjects with schizophrenia. Am. J. Psychiatry 2003, 160, 149–155. [Google Scholar] [CrossRef]
- Connor, C.M.; Guo, Y.; Akbarian, S. Cingulate white matter neurons in schizophrenia and bipolar disorder. Biol. Psyhciatry 2009, 66, 486–493. [Google Scholar] [CrossRef]
- Fung, S.J.; Joshi, D.; Fillman, S.G.; Wieckert, C.S. High white matter neuron density with elevated cortical cytokine expression in schizophrenia. Biol. Psychiatry 2014, 75, e5–e7. [Google Scholar] [CrossRef]
- McFadden, W.C.; Jaffe, A.E.; Ye, T.; Paltan-Ortiz, J.D.; Hyde, T.M.; Kleinman, J.E. Assessment of genetic risk for distribution of total white matter neurons in dorsolateral prefrontal cortex: Role in schizophrenia. Schizophr. Res. 2016, 176, 141–143. [Google Scholar] [CrossRef] [PubMed]
- Molnar, M.; Potkin, S.G.; Bunney, W.E.; Jones, E.G. mRNA expression patterns and distribution of white matter neurons in dorsolateral prefrontal cortex of depressed patients differ from those in schizophrenia patients. Biol. Psychiatry 2003, 53, 39–47. [Google Scholar] [CrossRef]
- Bailey, A.; Luthert, P.; Dean, A.; Harding, B.; Janota, I.; Montgomery, M.; Rutter, M.; Lantos, P. A clinicopathological study of autism. Brain 1998, 121, 889–905. [Google Scholar] [CrossRef] [PubMed]
- Chan-Palay, V. Somatostatin immunoreactive neurons in the human hippocampus and cortex shown by immunogold/silver intensification of vibratome sections: Coexistence with neuropeptide Y neurons, and effects in Alzheimer dementia. J. Comp. Neurol. 1987, 260, 201–223. [Google Scholar] [CrossRef]
- Chan-Palay, V.; Lang, W.; Allen, Y.S.; Haesler, U.; Polak, J.M. II. Cortical neurons immunoreactive with antisera against neuropeptide Y are altered in Alzheimer’s-type dementia. J. Comp. Neurol. 1985, 238, 390–400. [Google Scholar] [CrossRef] [PubMed]
- Van de Nes, J.A.P.; Sandmann-Keil, D.; Braak, H. Interstitial cells subjacent to the entorhinal region expressing somatostatin-28 immunoreactivity are susceptible to development of Alzheimer’s disease-related cytoskeletal changes. Acta Neuropathol. 2002, 104, 351–356. [Google Scholar] [CrossRef]
- Bunney, B.G.; Potkin, S.G.; Bunney, W.E. Neuropathological studies of brain tissue in schizophrenia. J. Psychiat. Res. 1997, 2, 159–173. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sedmak, G.; Judaš, M. White Matter Interstitial Neurons in the Adult Human Brain: 3% of Cortical Neurons in Quest for Recognition. Cells 2021, 10, 190. https://doi.org/10.3390/cells10010190
Sedmak G, Judaš M. White Matter Interstitial Neurons in the Adult Human Brain: 3% of Cortical Neurons in Quest for Recognition. Cells. 2021; 10(1):190. https://doi.org/10.3390/cells10010190
Chicago/Turabian StyleSedmak, Goran, and Miloš Judaš. 2021. "White Matter Interstitial Neurons in the Adult Human Brain: 3% of Cortical Neurons in Quest for Recognition" Cells 10, no. 1: 190. https://doi.org/10.3390/cells10010190
APA StyleSedmak, G., & Judaš, M. (2021). White Matter Interstitial Neurons in the Adult Human Brain: 3% of Cortical Neurons in Quest for Recognition. Cells, 10(1), 190. https://doi.org/10.3390/cells10010190




