Enhancing Zinc Accumulation and Bioavailability in Wheat Grains by Integrated Zinc and Pesticide Application
Abstract
:1. Introduction
2. Materials and Methods
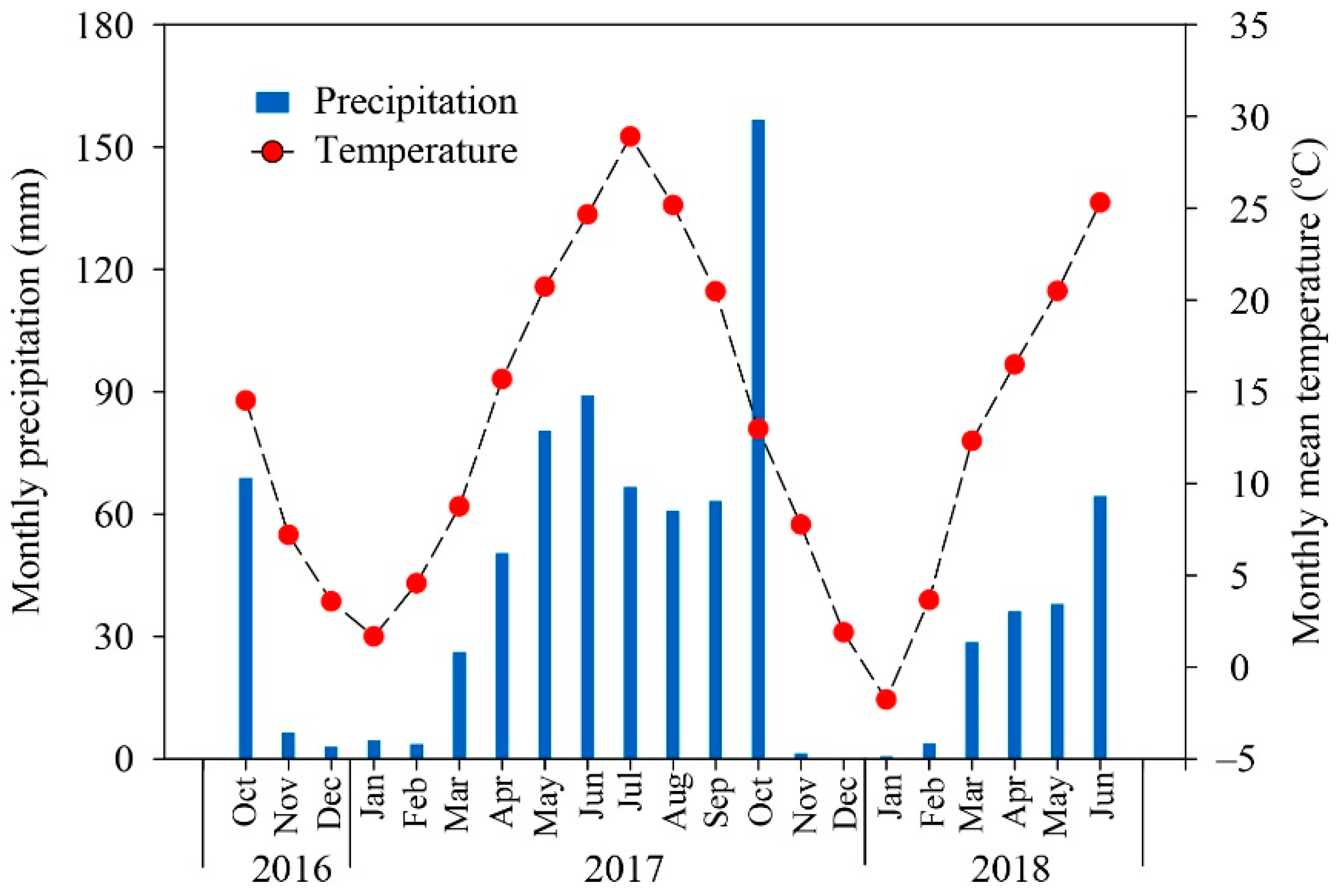
2.1. Site Description
2.2. Experimental Design
2.3. Plant Sampling and Analyses
2.4. Estimation of Zn Bioavailability
2.5. Statistical Analyses
3. Results
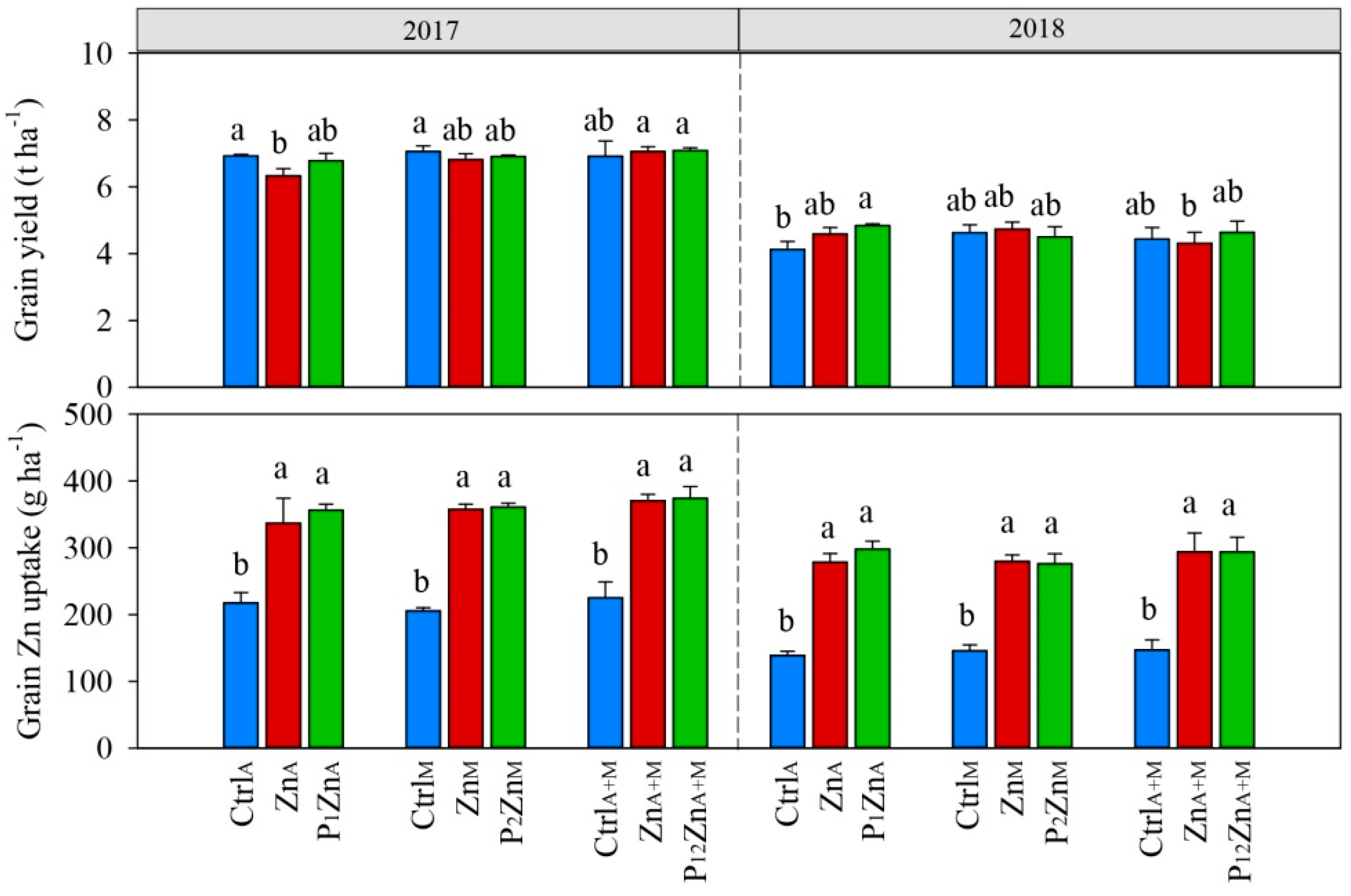
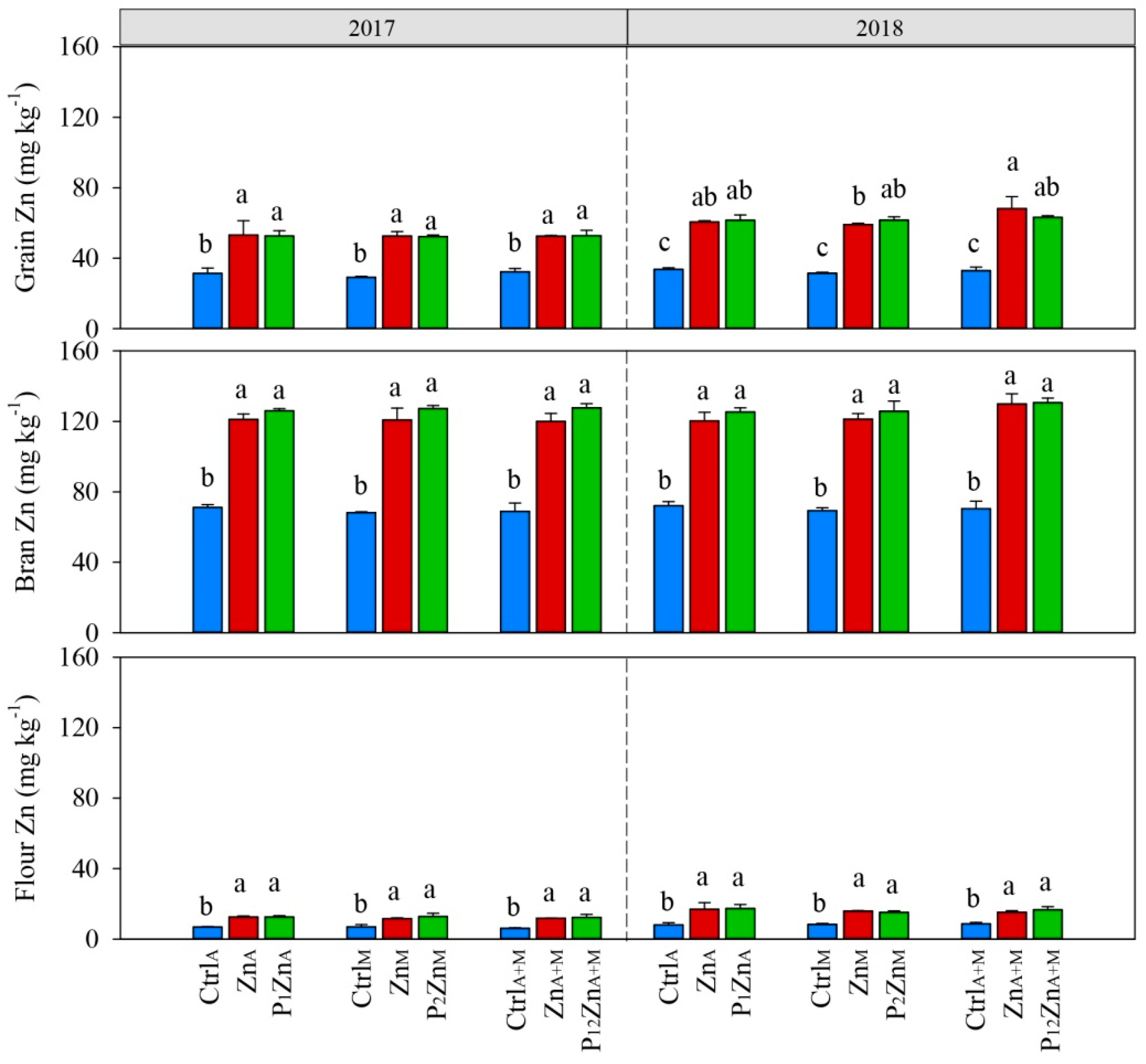
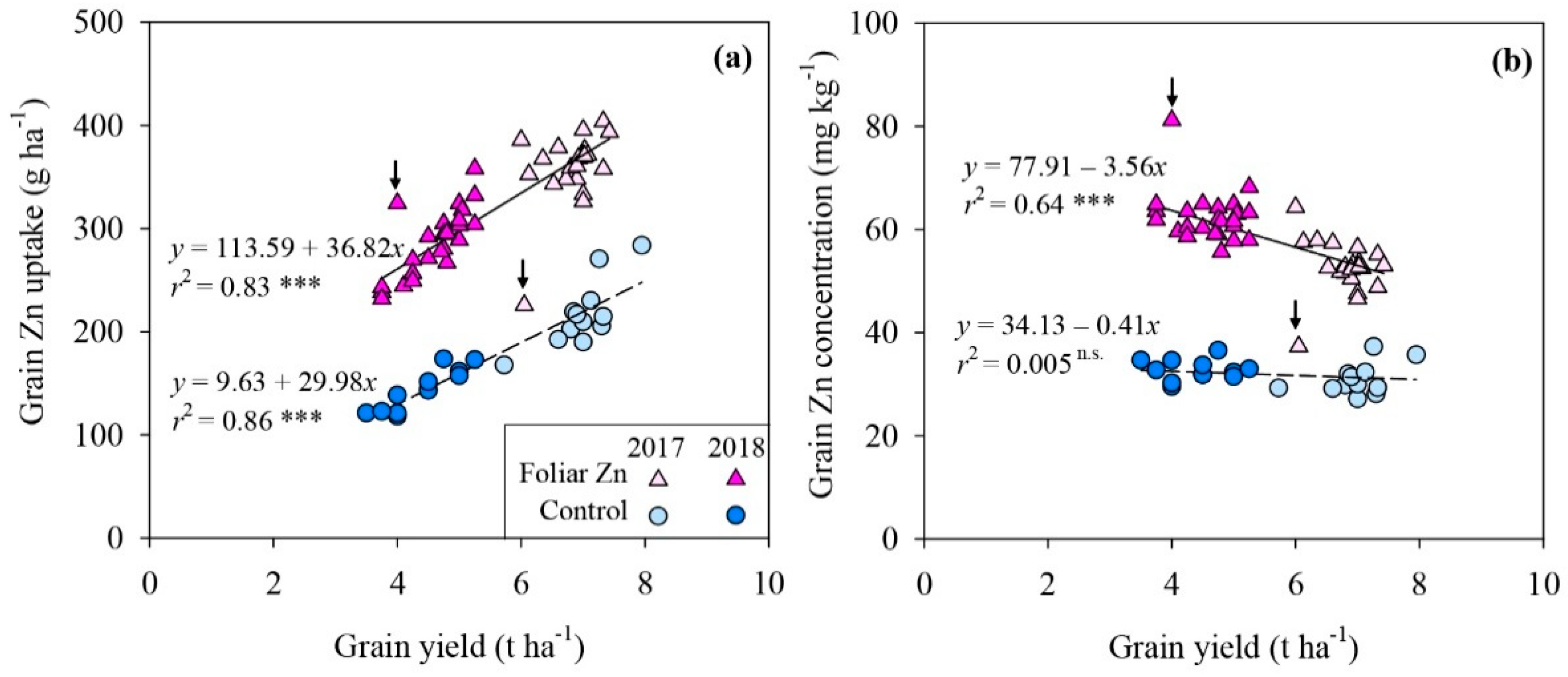
3.1. Grain Yield and Grain Zn Uptake and Concentration
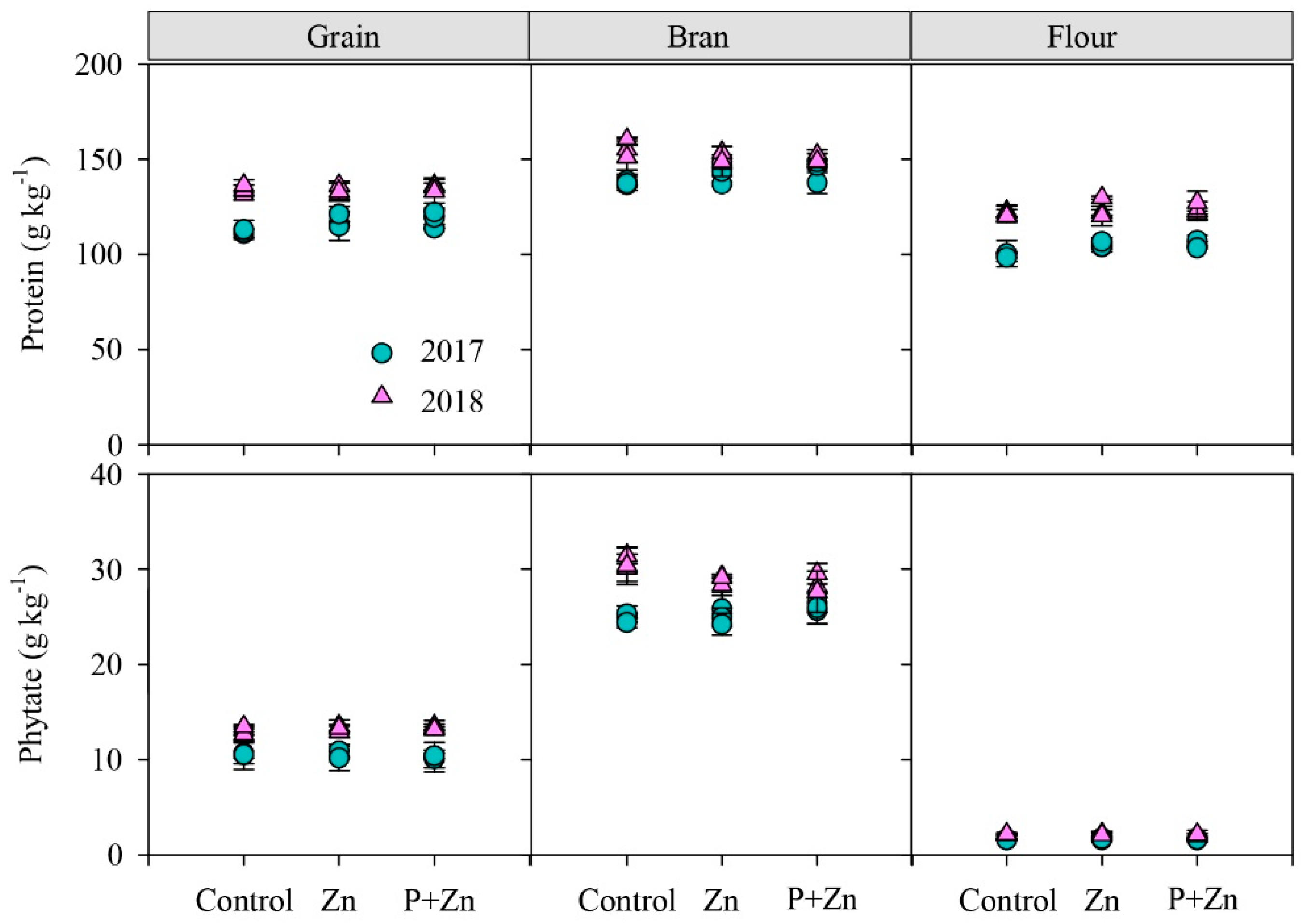
3.2. Zn-Binding Ligands in Grain and Its Fractions
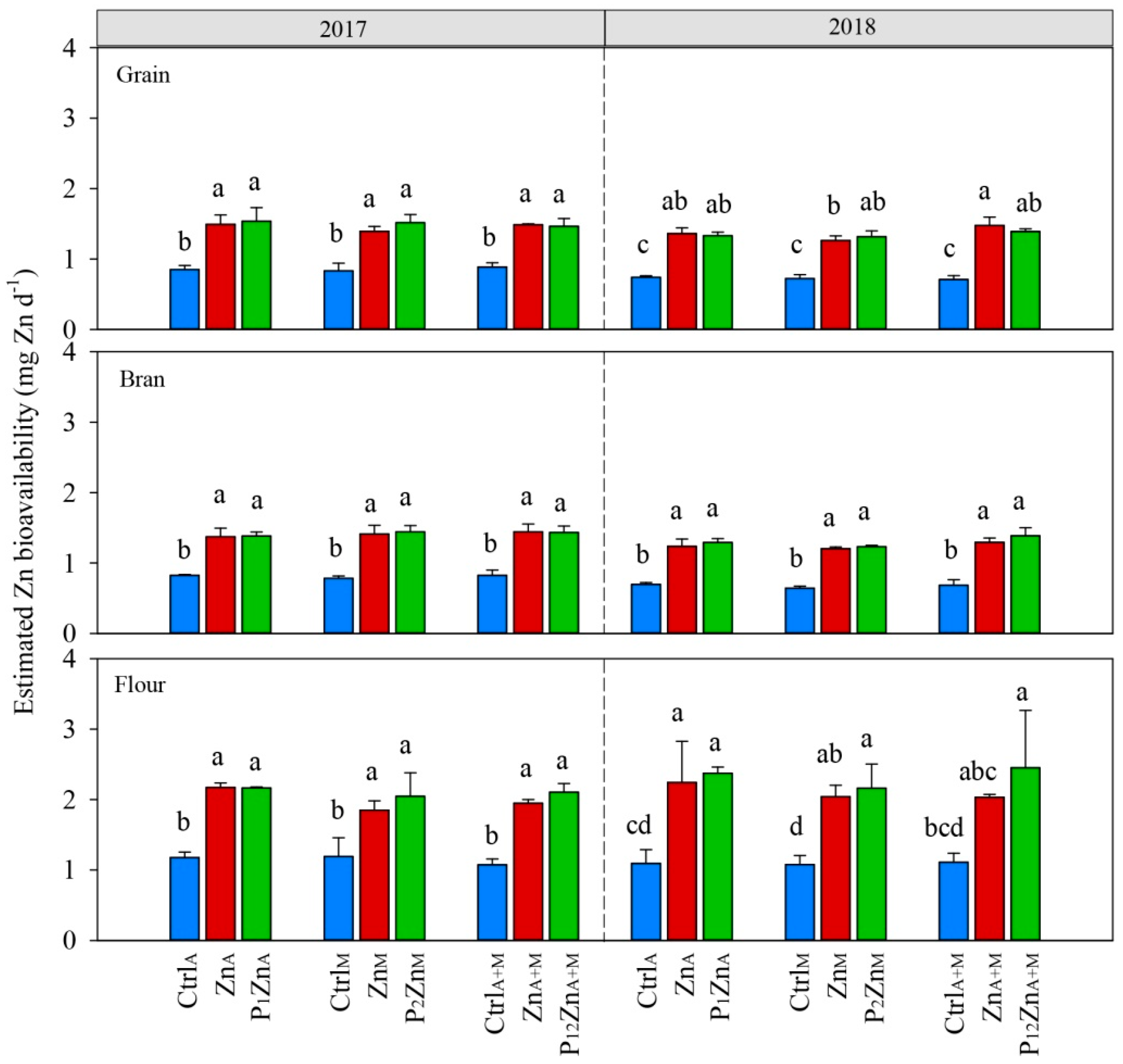
3.3. Zinc Bioavailability
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hotz, C.; Brown, K.H. Assessment of the risk of zinc deficiency in populations. Food Nutr. Bull. 2004, 25, S130–S162. [Google Scholar]
- Cakmak, I.; McLaughlin, M.J.; White, P. Zinc for better crop production and human health. Plant Soil 2017, 411, 1–4. [Google Scholar] [CrossRef]
- Cakmak, I. Enrichment of cereal grains with zinc: Agronomic or genetic biofortification? Plant Soil 2008, 302, 1–17. [Google Scholar] [CrossRef]
- Cakmak, I.; Kutman, U.B. Agronomic biofortification of cereals with zinc: A review. Eur. J. Soil Sci. 2018, 69, 172–180. [Google Scholar] [CrossRef]
- Bouis, H.E.; Hotz, C.; McClafferty, B.; Meenakshi, J.V.; Pfeiffer, W.H. Biofortification: A new tool to reduce micronutrient malnutrition. Food Nutr. Bull. 2011, 1, s31–s40. [Google Scholar] [CrossRef]
- Fan, M.; Zhao, F.; Fairweather-Tait, S.J.; Poulton, P.R.; Dunham, S.J.; McGrath, S.P. Evidence of decreasing mineral density in wheat grain over the last 160 years. J. Trace Elem. Med. Biol. 2008, 22, 315–324. [Google Scholar] [CrossRef]
- Zhao, F.J.; Su, Y.H.; Dunham, S.J.; Rakszegi, M.; Bedo, Z.; McGrath, S.P.; Shewry, P.R. Variation in mineral micronutrient concentrations in grain of wheat lines of diverse origin. J. Cereal Sci. 2009, 49, 290–295. [Google Scholar] [CrossRef]
- Morgounov, A.I.; Belan, I.; Zelenskiy, Y.; Roseeva, L.; Toemoeskoezi, S.; Bekes, F.; Abugalieve, A.; Cakmak, I.; Vargas, M.; Crossa, J. Historical changes in grain yield and quality of spring wheat varieties cultivated in Siberia from 1900 to 2010. Can. J. Plant Sci. 2013, 93, 425–433. [Google Scholar] [CrossRef]
- Graham, R.D.; Welch, R.M.; Saunders, D.A.; Ortiz Monasterio, I.; Bouis, H.E.; Bonierbale, M.; de Haan, S.; Burgos, G.; Thiele, G.; Liria, R.; et al. Nutritious subsistence food systems. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: Cambridge, MA, USA, 2007; Volume 92, pp. 1–74. [Google Scholar]
- Venegas, J.P.; Graybosch, R.A.; Wienhold, B.; Rose, D.J.; Waters, B.M.; Baenziger, P.S.; Eskridge, K.; Bai, G.; Amand, P.S. Biofortification of hard red winter wheat by genes conditioning low phytate and high grain protein concentration. Crop Sci. 2018, 58, 1–12. [Google Scholar] [CrossRef]
- Persson, D.P.; de Bang, T.C.; Pedas, P.R.; Kutman, U.B.; Cakmak, I.; Andersen, B.; Finnie, C.; Schjoerring, J.K.; Husted, S. Molecular speciation and tissue compartmentation of zinc in durum wheat grains with contrasting nutritional status. New Phytol. 2016, 211, 1255–1265. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Sun, Y.; Ye, Y.; Karim, M.R.; Xue, Y.; Yan, P.; Meng, Q.; Cui, Z.; Cakmak, I.; Zhang, F.; et al. Zinc biofortification of wheat through fertilizer applications in different locations of China. Field Crop Res. 2012, 125, 1–7. [Google Scholar] [CrossRef]
- Zou, C.Q.; Zhang, Y.Q.; Rashid, A.; Ram, H.; Savasli, E.; Arisoy, R.Z.; Ortiz-Monasterio, I.; Simunji, S.; Wang, Z.H.; Sohu, V.; et al. Biofortification of wheat with zinc through zinc fertilization in seven countries. Plant Soil 2012, 361, 119–130. [Google Scholar] [CrossRef]
- Wang, Y.; Zou, C.; Mirza, Z.; Li, H.; Zhang, Z.; Li, D.; Xu, C.; Zhou, X.; Shi, X.; Xie, D.; et al. Cost of agronomic biofortification of wheat with zinc in China. Agron. Sustain. Dev. 2016, 36, 44. [Google Scholar] [CrossRef]
- Wang, X.; Liu, D.; Zhang, W.; Wang, C.; Cakmak, I.; Zou, C. An effective strategy to improve grain zinc concentration of winter wheat, Aphids prevention and farmers’ income. Field Crop Res. 2015, 184, 74–79. [Google Scholar] [CrossRef]
- Ram, H.; Rashid, A.; Zhang, W.; Duarte, A.P.; Phattarakul, N.; Simunji, S.; Kalayci, M.; Freitas, R.; Rerkasem, B.; Bal, R.S.; et al. Biofortification of wheat, rice and common bean by applying foliar zinc fertilizer along with pesticides in seven countries. Plant Soil 2016, 403, 389–401. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, X.; Liu, K.; Fei, P.; Chen, J.; Li, X.; Ning, P.; Chen, Y.; Shi, J.; Tian, X. Improving zinc concentration and bioavailability of wheat grain through combined foliar applications of zinc and pesticides. Agron. J. 2019, 111, 1478–1487. [Google Scholar] [CrossRef]
- Cakmak, I.; Kalayci, M.; Kaya, Y.; Torun, A.A.; Aydin, N.; Wang, Y.; Arisoy, Z.; Erdem, H.; Yazici, A.; Gokmen, O.; et al. Biofortification and localization of zinc in wheat grain. J. Agric. Food Chem. 2010, 58, 9092–9102. [Google Scholar] [CrossRef]
- Nelson, D.W.; Sommers, L.E. Determination of total nitrogen in plant material. Agron. J. 1973, 65, 109–112. [Google Scholar] [CrossRef]
- Miller, L.V.; Krebs, N.F.; Hambidge, M.K. A mathematical model of zinc absorption in humans as a function of dietary zinc and phytate. J. Nutr. 2007, 137, 135–141. [Google Scholar] [CrossRef]
- Ram, H.; Sohu, V.S.; Cakmak, I.; Singh, K.; Buttar, G.S.; Sodhi, G.P.S.; Gill, H.S.; Bhagat, I.; Singh, P.; Dhaliwal, S.S.; et al. Agronomic fortification of rice and wheat grains with zinc for nutritional security. Curr. Sci. 2015, 109, 1171–1176. [Google Scholar] [CrossRef]
- Cakmak, I.; Hoffland, E. Zinc for the improvement of crop production and human health. Plant Soil 2012, 361, 1–2. [Google Scholar] [CrossRef] [Green Version]
- Kutman, U.B.; Yildiz, B.; Ozturk, L.; Cakmak, I. Biofortification of durum wheat with zinc through soil and foliar applications of nitrogen. Cereal Chem. 2010, 87, 1–9. [Google Scholar] [CrossRef]
- Montalvo, D.; Degryse, F.; Silva, R.C.D.; Baird, R.; McLaughlin, M.J. Agronomic effectiveness of zinc sources as micronutrient fertilizer. Adv. Agron. 2016, 139, 215–267. [Google Scholar]
- Khokhar, J.S.; Sareen, S.; Tyagi, B.S.; Singh, G.; Wilson, L.; King, I.P.; Young, S.D.; Broadley, M.R. Variation in grain Zn concentration, and the grain ionome, in field-grown Indian wheat. PLoS ONE 2018, 13, e192026. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhang, Y.; Tong, Y.; Xue, Y.; Liu, D.; Zhang, W.; Deng, Y.; Meng, Q.; Yue, S.; Yan, P.; et al. Harvesting more grain zinc of wheat for human health. Sci. Rep. 2017, 7, 7016. [Google Scholar] [CrossRef]
- Li, M.; Tian, X.; Li, X.; Wang, S. Effect of Zn application methods on Zn distribution and bioavailability in wheat pearling fractions of two wheat genotypes. J. Integr. Agric. 2017, 16, 1617–1623. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Wang, S.; Tian, X.; Zhao, J.; Li, H.; Guo, C.; Chen, Y.; Zhao, A. Zn distribution and bioavailability in whole grain and grain fractions of winter wheat as affected by applications of soil N and foliar Zn combined with N or P. J. Cereal Sci. 2015, 61, 26–32. [Google Scholar] [CrossRef]
- Li, M.; Wang, S.; Tian, X.; Li, S.; Chen, Y.; Jia, Z.; Liu, K.; Zhao, A. Zinc and iron concentrations in grain milling fractions through combined foliar applications of Zn and macronutrients. Field Crop Res. 2016, 187, 135–141. [Google Scholar] [CrossRef]
- Liu, N.; Zhang, Y.; Wang, B.; Xue, Y.; Yu, P.; Zhang, Q.; Wang, Z. Is grain zinc concentration in wheat limited by source? Aust. J. Crop Sci. 2014, 8, 1534–1541. [Google Scholar]
- Stomph, T.J.; Choi, E.Y.; Stangoulis, J.C.R. Temporal dynamics in wheat grain zinc distribution: Is sink limitation the key? Ann. Bot. 2011, 107, 927–937. [Google Scholar] [CrossRef]
- Wang, Y.X.; Specht, A.; Horst, W.J. Stable isotope labelling and zinc distribution in grains studied by laser ablation ICP-MS in an ear culture system reveals zinc transport barriers during grain filling in wheat. New Phytol. 2011, 189, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Ajiboye, B.; Cakmak, I.; Paterson, D.; de Jonge, M.D.; Howard, D.L.; Stacey, S.P.; Torun, A.A.; Aydin, N.; McLaughlin, M.J. X-ray fluorescence microscopy of zinc localization in wheat grains biofortified through foliar zinc applications at different growth stages under field conditions. Plant Soil 2015, 392, 357–370. [Google Scholar] [CrossRef]
- Erenoglu, E.B.; Kutman, U.B.; Ceylan, Y.; Yildiz, B.; Cakmak, I. Improved nitrogen nutrition enhances root uptake, root-to-shoot translocation and remobilization of zinc (65Zn) in wheat. New Phytol. 2011, 189, 438–448. [Google Scholar] [CrossRef] [PubMed]

| Measured Variable | Source of Variation | ||
|---|---|---|---|
| Year (Yr) | Treatment (T) | Yr × T | |
| Grain yield (t ha−1) | <0.0001 | n.s. | n.s. |
| Grain Zn uptake (g ha−1) | <0.0001 | <0.0001 | n.s. |
| Grain Zn concentration (mg kg−1) | <0.0001 | <0.0001 | n.s. |
| Bran Zn concentration (mg kg−1) | n.s. 1 | <0.0001 | n.s. |
| Flour Zn concentration (mg kg−1) | <0.0001 | <0.0001 | n.s. |
| Grain protein concentration (g kg−1) | <0.0001 | n.s. | n.s. |
| Bran protein concentration (g kg−1) | <0.0001 | n.s. | n.s. |
| Flour protein concentration (g kg−1) | <0.0001 | n.s. | n.s. |
| Grain phytate concentration (g kg−1) | <0.0001 | n.s. | n.s. |
| Bran phytate concentration (g kg−1) | <0.0001 | n.s. | n.s. |
| Flour phytate concentration (g kg−1) | <0.0001 | n.s. | n.s. |
| Zn bioavailability in grain (mg Zn d−1) | 0.0041 | <0.0001 | n.s. |
| Zn bioavailability in bran (mg Zn d−1) | 0.0005 | <0.0001 | n.s. |
| Zn bioavailability in flour (mg Zn d−1) | n.s. | <0.0001 | n.s. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ning, P.; Wang, S.; Fei, P.; Zhang, X.; Dong, J.; Shi, J.; Tian, X. Enhancing Zinc Accumulation and Bioavailability in Wheat Grains by Integrated Zinc and Pesticide Application. Agronomy 2019, 9, 530. https://doi.org/10.3390/agronomy9090530
Ning P, Wang S, Fei P, Zhang X, Dong J, Shi J, Tian X. Enhancing Zinc Accumulation and Bioavailability in Wheat Grains by Integrated Zinc and Pesticide Application. Agronomy. 2019; 9(9):530. https://doi.org/10.3390/agronomy9090530
Chicago/Turabian StyleNing, Peng, Shaoxia Wang, Peiwen Fei, Xiaoyuan Zhang, Jinjin Dong, Jianglan Shi, and Xiaohong Tian. 2019. "Enhancing Zinc Accumulation and Bioavailability in Wheat Grains by Integrated Zinc and Pesticide Application" Agronomy 9, no. 9: 530. https://doi.org/10.3390/agronomy9090530
APA StyleNing, P., Wang, S., Fei, P., Zhang, X., Dong, J., Shi, J., & Tian, X. (2019). Enhancing Zinc Accumulation and Bioavailability in Wheat Grains by Integrated Zinc and Pesticide Application. Agronomy, 9(9), 530. https://doi.org/10.3390/agronomy9090530






