Regenerating Agricultural Landscapes with Perennial Groundcover for Intensive Crop Production
Abstract
1. Introduction
2. Agricultural Context
2.1. The Landscape
2.2. The Crops
2.3. Biofuels
3. Environmental Concerns
3.1. Nitrogen Efflux and Water Quality
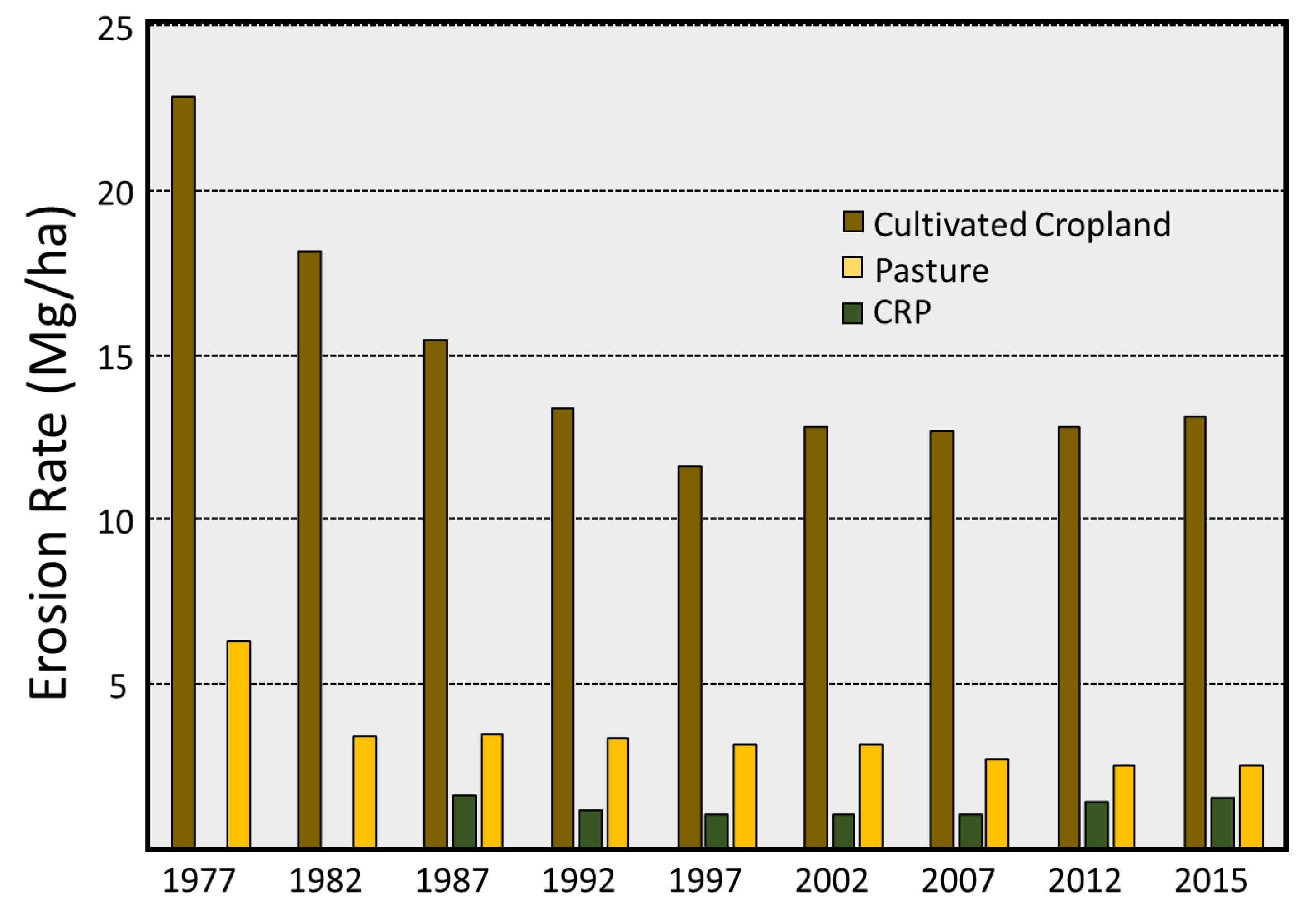
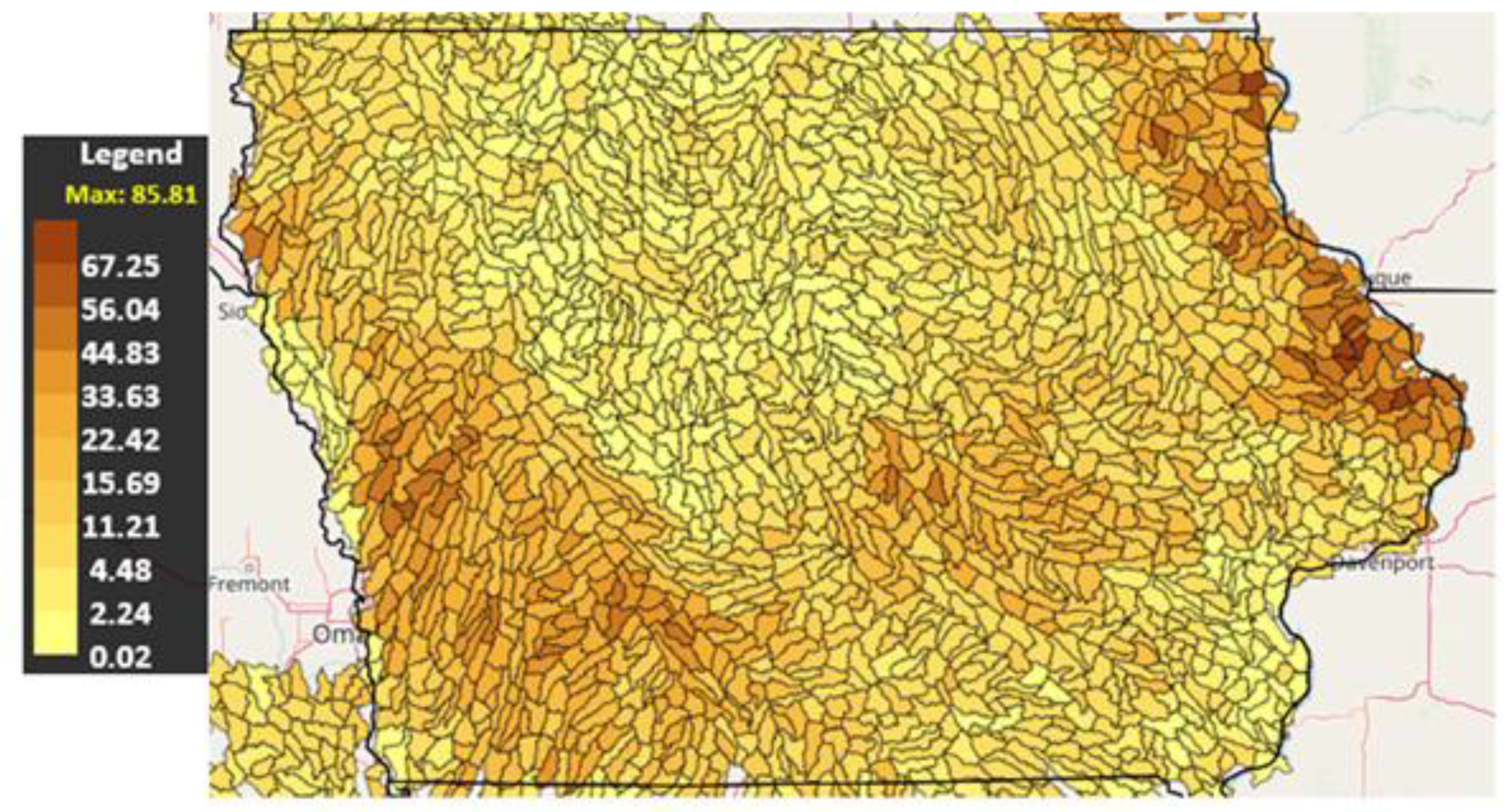
3.2. Soil Erosion
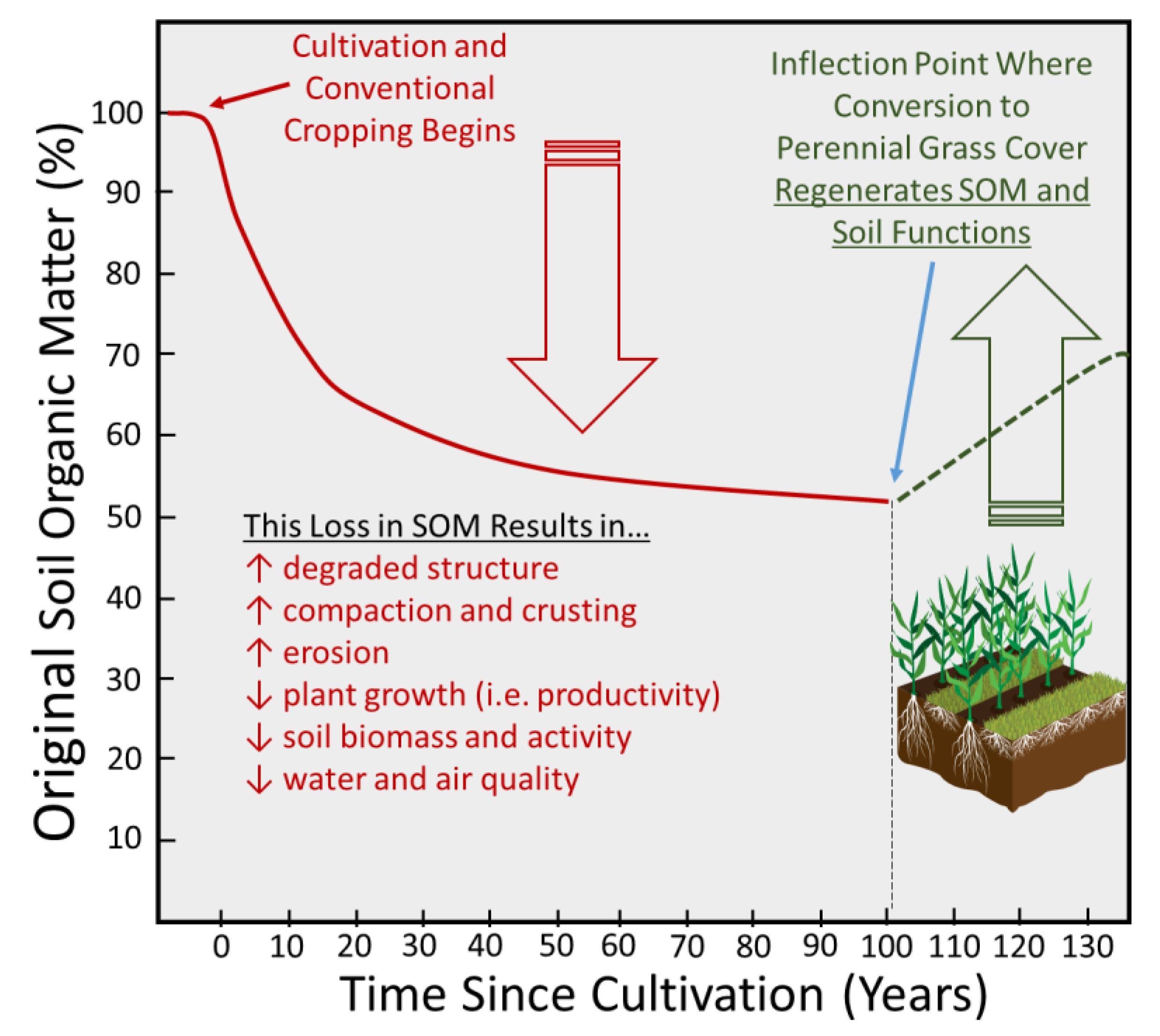
3.3. Soil Quality and Ecosystem Services
4. Mitigating Environmental Impacts of Crop Production
4.1. Annual Cover Crop Systems
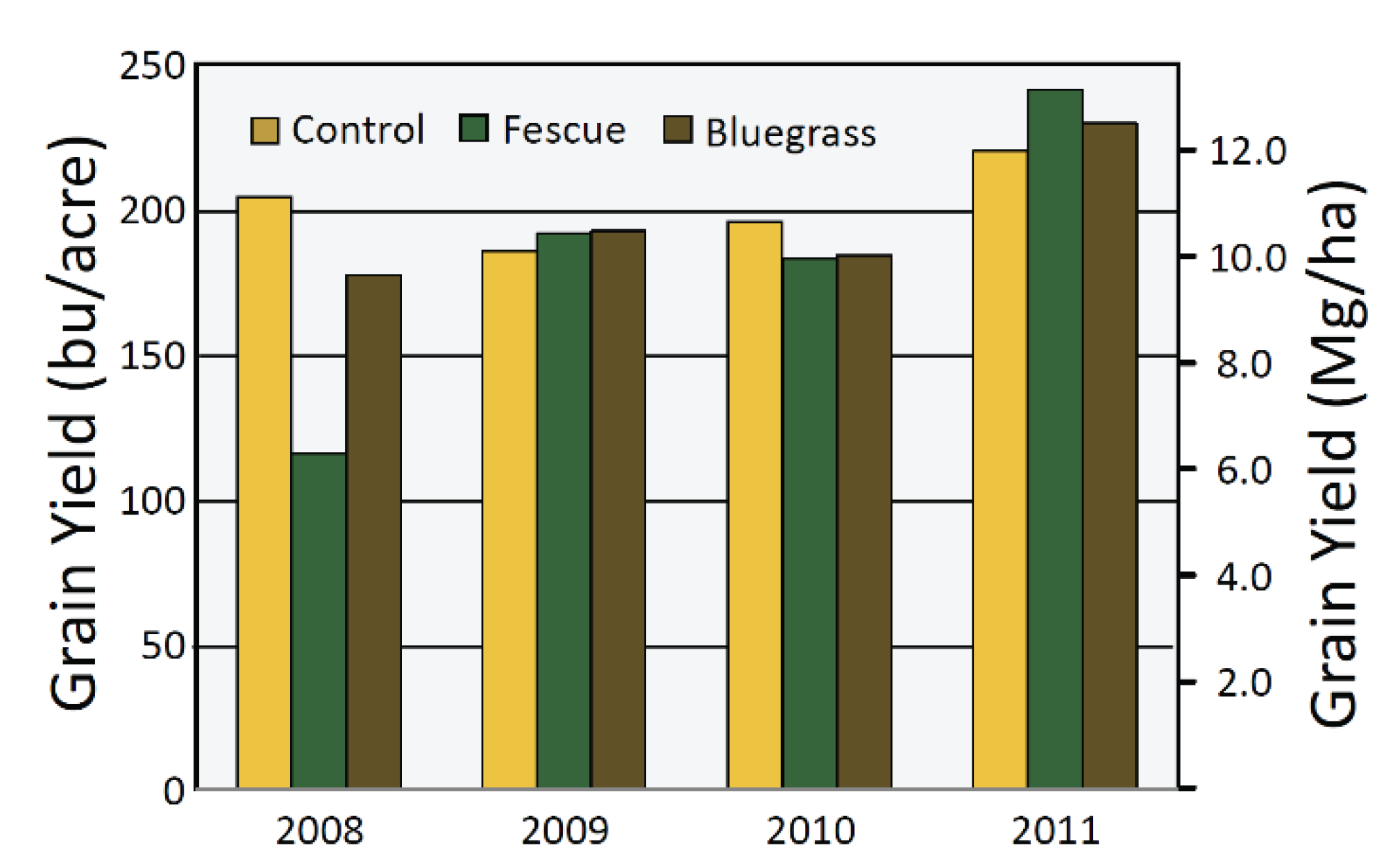
4.2. Perennial Groundcover
5. Designing and Developing Perennial Groundcover Systems
5.1. Crop Ecology and Management
5.1.1. Minimizing Interspecific Competition
5.1.2. Species Complementarity
5.1.3. Managing Competition
5.1.4. Enhancing Crop Competitiveness
5.2. Plant Breeding and Genetics
5.2.1. Developing PGC Cultivars
5.2.2. Developing Maize Hybrids Adapted to PGC Systems
5.3. Soil Health and Nutrient Management
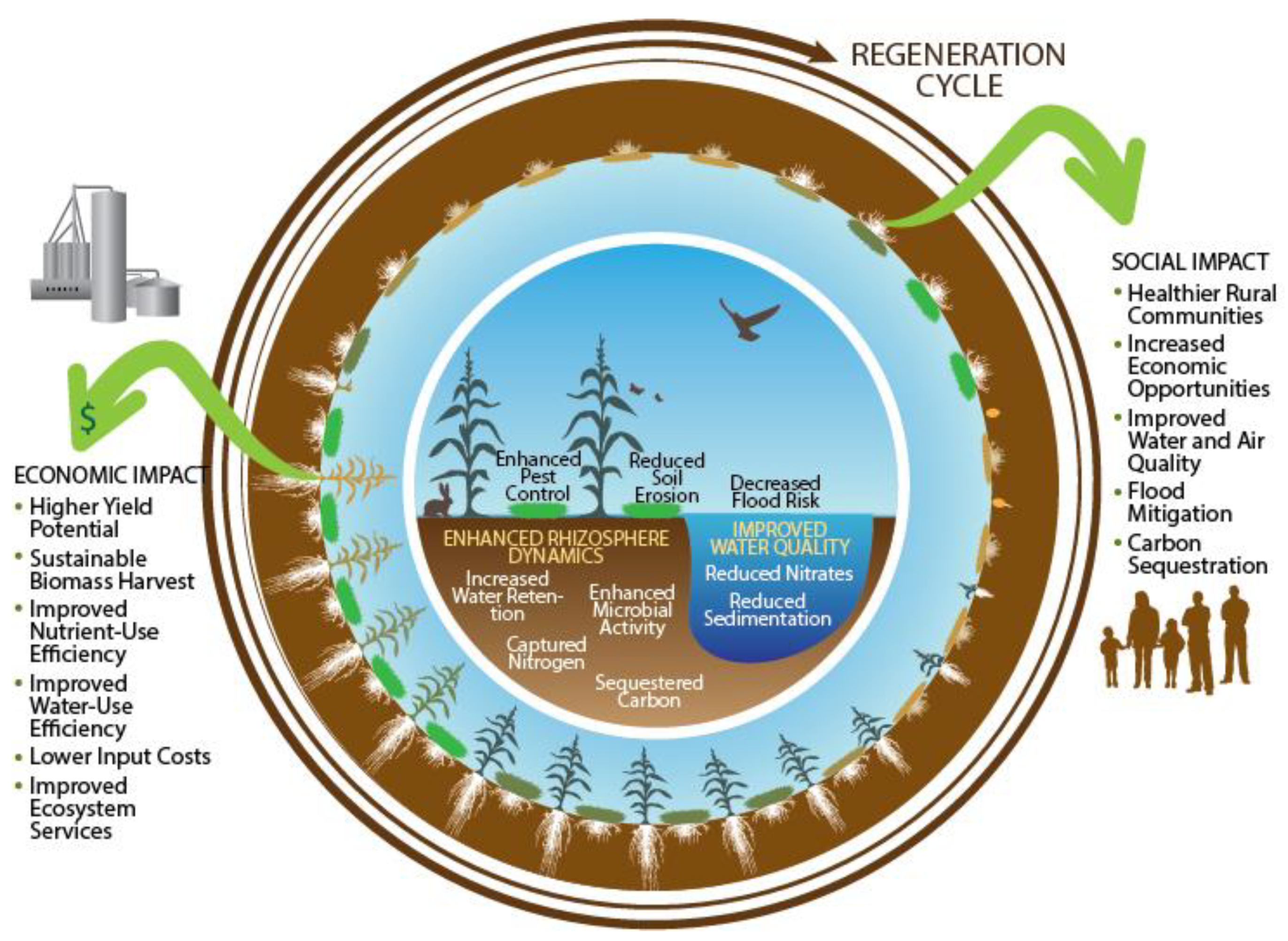
5.4. Ecosystem Services and the Environment
5.5. Economic and Sociological Factors
5.5.1. Socio-Economic Feasibility: Benefits and Costs
5.5.2. Lessons Learned from Cover Crops and Other Conservation Practices
5.5.3. Policies and Mechanisms to Incent Adoption
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kling, C.L.; Chaubey, I.; Cibin, R.; Gassman, P.W.; Panagopoulos, Y. Policy implications from multi-scale watershed models of biofuel crop adoption across the Corn Belt. J. Am. Water Res. Assoc. 2017, 53, 1313–1322. [Google Scholar] [CrossRef]
- Guo, T.; Cibin, R.; Chaubey, I.; Gitau, M.; Arnold, J.G.; Srinivasan, R.; Kiniry, J.R.; Engel, B.A. Evaluation of bioenergy crop growth and the impacts of bioenergy crops on streamflow, tile drain flow and nutrient losses in an extensively tile-drained watershed using SWAT. Sci. Total Environ. 2018, 613, 724–735. [Google Scholar] [CrossRef] [PubMed]
- Teter, J.; Yeh, S.; Khanna, M.; Berndes, G. Water impacts of US biofuels: Insights from an assessment combining economic and biophysical models. PLoS ONE 2018, 13, e0204298. [Google Scholar] [CrossRef] [PubMed]
- Karlen, D.L.; Kovar, J.L.; Cambardella, C.A.; Colvin, T.S. Thirty-year tillage effects on crop yield and soil fertility indicators. Soil Tillage Res. 2013, 130, 24–41. [Google Scholar] [CrossRef]
- Johnson, J.M.F.; Novak, J.M.; Varvel, G.E.; Stott, D.E.; Osborne, S.L.; Karlen, D.L.; Lamb, J.A.; Baker, J.; Adler, P.R. Crop residue mass needed to maintain soil organic carbon levels: Can it be determined? BioEnergy Res. 2014, 7, 481–490. [Google Scholar] [CrossRef]
- Jin, V.L.; Baker, J.M.; Johnson, J.F.; Karlen, D.L.; Lehman, R.M.; Osborne, S.L.; Sauer, T.J.; Stott, D.E.; Varvel, G.E.; Venterea, R.T.; et al. Soil greenhouse gas emissions in response to corn stover removal and tillage management across the US Corn Belt. BioEnergy Res. 2014, 7, 517–527. [Google Scholar] [CrossRef]
- Lehman, R.M.; Cambardella, C.A.; Stott, D.E.; Acosta-Martinez, V.; Manter, D.K.; Buyer, J.S.; Maul, J.E.; Smith, J.L.; Collins, H.P.; Halvorson, J.J.; et al. Understanding and enhancing soil biological health: The solution for reversing soil degradation. Sustainability 2015, 7, 988–1027. [Google Scholar] [CrossRef]
- Porter, P.A.; Mitchell, R.B.; Moore, K.J. Reducing hypoxia in the Gulf of Mexico: An alternative approach. J. Soil Water Conserv. 2015, 70, 63A–68A. [Google Scholar] [CrossRef]
- Cassman, K.; Dobermann, A.; Walters, D. Agroecosystems, N-use efficiency, and N management. AMBIO J. Hum. Environ. 2002, 31, 132–140. [Google Scholar] [CrossRef]
- Cook, B.I.; Ault, T.R.; Smerdon, J.E. Unprecedented 21st Century drought risk in the American Southwest and Central Plains. Sci. Adv. 2015. [Google Scholar] [CrossRef]
- Roesch-McNally, G.E.; Arbuckle, J.G.; Tyndall, J.C. Barriers to implementing climate resilient agricultural strategies: The case of crop diversification in the US Corn Belt. Glob. Environ. Chang. 2018, 48, 206–215. [Google Scholar] [CrossRef]
- Vanloqueren, G.; Baret, P.V. Why are ecological, low-input, multi-resistant wheat cultivars slow to develop commercially? A Belgian agricultural ‘lock-in’ case study. Ecol. Econ. 2008, 66, 436–446. [Google Scholar] [CrossRef]
- Weaver, J.E. North American Prairie; Johnsen Publishing Co.: Lincoln, NE, USA, 1954. [Google Scholar]
- Gallant, A.L.; Sadinski, W.; Roth, M.F.; Rew, C.A. Changes in historical Iowa land cover as context for assessing the environmental benefits of current and future conservation efforts on agricultural lands. J. Soil Water Conserv. 2011, 66, 67A–77A. [Google Scholar] [CrossRef]
- Smith, D.D. Iowa prairie-an endangered ecosystem. Proc. Iowa Acad. Sci. 1981, 88, 7–10. [Google Scholar]
- Lyon, D.J.; Pavlista, A.D.; Hergert, G.W.; Klein, R.N.; Shapiro, C.A.; Knezevic, S.; Mason, S.C.; Nelson, L.A.; Baltensperger, D.D.; Elmore, R.W.; et al. Skip-row planting patterns stabilize corn grain yields in the central Great Plains. Crop Manag. 2009. [Google Scholar] [CrossRef]
- Jenny, H. The Soil Resource, Origin and Behavior; Springer: New York, NY, USA, 1980. [Google Scholar]
- Cambardella, C.A.; Elliott, E.T. Particulate soil organic-matter changes across a grassland cultivation sequence. Soil Sci. Soc. Am. J. 1992, 56, 777–783. [Google Scholar] [CrossRef]
- Aref, S.; Wander, M.M. Long-term trends of corn yield and soil organic matter in different crop sequences and soil fertility treatments on the Morrow Plots. Adv. Agron. 1997, 62, 153–197. [Google Scholar]
- Nafziger, E.D.; Dunker, R.E. Soil organic carbon trends over 100 years in the Morrow plots. Agron. J. 2011, 103, 261–267. [Google Scholar] [CrossRef]
- Huggins, D.R.; Buyanovsky, G.A.; Wagner, G.H.; Brown, J.R.; Darmody, R.G.; Peck, T.R.; Lesoing, G.W.; Vanotti, M.B.; Bundy, L.G. Soil organic C in the tallgrass prairie-derived region of the corn belt: Effects of long-term crop management. Soil Tillage Res. 1998, 47, 219–234. [Google Scholar] [CrossRef]
- Den Biggelaar, C.; Lal, R.; Wiebe, K.; Breneman, V. Impact of soil erosion on crop yields in North America. Adv. Agron. 2001, 72, 1–52. [Google Scholar]
- Dabrowski, J.M.; Peall, S.K.; Van Niekerk, A.; Reinecke, A.J.; Day, J.A.; Schulz, R. Predicting runoff-induced pesticide input in agricultural sub-catchment surface waters: Linking catchment variables and contamination. Water Res. 2002, 36, 4975–4984. [Google Scholar] [CrossRef]
- Lee, K.H.; Isenhart, T.M.; Schultz, R.C. Sediment and nutrient removal in an established multi-species riparian buffer. J. Soil Water Conserv. 2003, 58, 1–8. [Google Scholar]
- Masarik, K.; Norman, J.; Brye, K. Long-term drainage and nitrate leaching below well-drained continuous corn agroecosystems and a prairie. J. Environ. Prot. 2014, 5, 240–254. [Google Scholar] [CrossRef]
- Karlen, D.L.; Hurley, E.G.; Andrews, S.S.; Cambardella, C.A.; Meek, D.W.; Duffy, M.D.; Mallarino, A.P. Crop rotation effects on soil quality at three northern Corn/Soybean Belt locations. Agron. J. 2006, 98, 484–495. [Google Scholar] [CrossRef]
- Wright, C.K.; Wimberly, M.C. Recent land use change in the Western corn belt threatens grasslands and wetlands. Prod. Natl. Acad. Sci. USA 2013, 110, 4134–4139. [Google Scholar] [CrossRef] [PubMed]
- Basche, A.D.; Archontoulis, S.V.; Kaspar, T.C.; Jaynes, D.B.; Parkin, T.B.; Miguez, F.E. Simulating long-term impacts of cover crops and climate change on crop production and environmental outcomes in the Midwestern United States. Agric. Ecosyst. Environ. 2016, 218, 95–106. [Google Scholar] [CrossRef]
- USDA-NASS. Crops and Plants. 2018. Available online: https://www.nass.usda.gov/ (accessed on 11 November 2018).
- H.R.6. Energy Independence and Security Act of 2007; Public Law No: 110-140; Government Printing Office: Washington, DC, USA, 19 December 2007.
- US-DOE. 2016 Billion-Ton Report: Advancing Domestic Resources for a Thriving Bioeconomy, Volume 1: Economic Availability of Feedstocks; ORNL/TM-2016/160; Oak Ridge National Laboratory: Oak Ridge, TN, USA, 2016.
- Perlack, R.D.; Stokes, B.J. Billion-Ton Update: Biomass Supply for a Bioenergy and Bioproducts Industry; ORNL/TM-2011/224; Oak Ridge National Laboratory: Oak Ridge, TN, USA, 2011.
- Karlen, D.L.; Varvel, G.E.; Johnson, J.M.F.; Baker, J.M.; Osborne, S.L.; Novak, J.M.; Adler, P.R.; Roth, G.W.; Birrell, S.J. Monitoring soil quality to assess the sustainability of harvesting corn stover. Agron. J. 2011, 103, 288–295. [Google Scholar] [CrossRef]
- Moore, K.J.; Karlen, D.L.; Lamkey, K.R. Future prospects for corn as a biofuel crop. In Compendium of Bioenergy Crops; Goldman, S.L., Ed.; Science Publishers: Rawalpindi, Pakistan; Taylor & Francis Group, LLC: Abingdon, UK, 2014; pp. 331–352. [Google Scholar]
- Laird, D.A.; Chang, C.W. Long-term impacts of residue harvesting on soil quality. Soil Tillage Res. 2013, 134, 33–40. [Google Scholar] [CrossRef]
- Wilhelm, W.W.; Johnson, J.M.F.; Hatfield, J.L.; Voorhees, W.B.; Linden, D.R. Crop and soil productivity response to corn residue removal: A literature review. Agron. J. 2004, 96, 1–17. [Google Scholar] [CrossRef]
- Wilhelm, W.W.; Johnson, J.M.F.; Karlen, D.L.; Lightle, D.T. Corn stover to sustain soil organic carbon further constrains biomass supply. Agron. J. 2007, 99, 1665–1667. [Google Scholar] [CrossRef]
- Sheehan, J.; Aden, A.; Paustian, K.; Killian, K.; Brenner, J.; Walsh, M.; Nelson, R. Energy and environmental aspects of using corn stover for fuel ethanol. J. Ind. Ecol. 2004, 7, 117–146. [Google Scholar] [CrossRef]
- Karlen, D.L.; Beeler, L.W.; Ong, R.G.; Dale, B.E. Balancing energy, conservation, and soil health requirements for plant biomass. J. Soil Water Conserv. 2015, 70, 279–287. [Google Scholar] [CrossRef]
- Environmental Protection Agency. Reactive Nitrogen in the United States: An Analysis of Inputs, Flows, Consequences, and Management Options; EPA-SAB-11-013; EPA Science Advisory Board, U.S. Environmental Protection Agency: Washington, DC, USA, 2011.
- Hayes, D.; Kling, C.L.; Lawrence, J. Economic Evaluation of Governor Branstad’s Water Quality Initiative; CARD Policy Briefs 2016, 16-PB 19; Center for Agricultural and Rural Development, Iowa State University: Ames, IA, USA, March 2016. [Google Scholar]
- Keiser, D.A. The missing benefits of clean water and the role of mismeasured pollution. J. Assoc. Environ. Resour. Econ. 2019, 6, 669–707. [Google Scholar] [CrossRef]
- Sprague, L.A.; Hirsch, R.M.; Aulenbach, B.T. Nitrate in the Mississippi River and its tributaries, 1980 to 2008: Are we making progress? Environ. Sci. Technol. 2011, 45, 7209–7216. [Google Scholar] [CrossRef] [PubMed]
- Rabalais, N.N.; Turner, R.E.; Scavia, D. Beyond science into policy: Gulf of Mexico hypoxia and the Mississippi river: Nutrient policy development for the Mississippi River watershed reflects the accumulated scientific evidence that the increase in nitrogen loading is the primary factor in the worsening of hypoxia in the northern Gulf of Mexico. BioScience 2002, 52, 129–142. [Google Scholar] [CrossRef]
- Rabotyagov, S.S.; Kling, C.L.; Gassman, P.W.; Rabalais, N.N.; Turner, R.E. The economics of dead zones: Causes, impacts, policy challenges, and a model of the Gulf of Mexico hypoxic zone. Rev. Environ. Econ. Policy 2014, 8, 58–79. [Google Scholar] [CrossRef]
- NOAA, National Oceanic and Atmospheric Administration. Gulf of Mexico ‘Dead Zone’ Is the Largest Ever Measured. 2017. Available online: http://www.noaa.gov/media-release/gulf-of-mexico-dead-zone-is-largest-ever-measured (accessed on 24 October 2018).
- Environmental Protection Agency. Gulf Hypoxia Action Plan. 2008. Available online: https://www.epa.gov/ms-htf/gulf-hypoxia-action-plan (accessed on 24 October 2018).
- Stoner, N. Working in Partnership with States to Address Phosphorus and Nitrogen Pollution through Use of a Framework for State Nutrient Reductions; Office of Water, United States Environmental Protection Agency: Washington, DC, USA, 16 March 2011; p. 6.
- Dinnes, D.L.; Karlen, D.L.; Jaynes, D.B.; Kaspar, T.C.; Hatfield, J.L.; Colvin, T.S.; Cambardella, C.A. Nitrogen management strategies to reduce nitrate leaching in tile-drained Midwestern soils. Agron. J. 2002, 94, 153–171. [Google Scholar] [CrossRef]
- Sugg, Z. Assessing U.S. Farm Drainage: Can GIS Lead to Better Estimates of Subsurface Drainage Extent; World Resources Institute: Washington, DC, USA, 2007; Available online: https://www.wri.org/publication/assessing-us-farm-drainage (accessed on 11 June 2019).
- Arenas Amado, A.; Schilling, K.E.; Jones, C.S.; Thomas, N.; Weber, L.J. Estimation of tile drainage contribution to streamflow and nutrient loads at the watershed scale based on continuously monitored data. Environ. Monit. Assess. 2017, 189, 426. [Google Scholar] [CrossRef]
- David, M.; Drinkwater, L.E.; McIsaac, G.F. Sources of nitrate yields in the Mississippi River Basin. J. Environ. Qual. 2010, 39, 1657–1667. [Google Scholar] [CrossRef]
- Blann, K.L.; Anderson, J.L.; Sands, G.R.; Vondracek, B. Effects of agricultural drainage on aquatic ecosystems: A review. Crit. Rev. Environ. Sci. Technol. 2009, 39, 909–1001. [Google Scholar] [CrossRef]
- Pimentel, D.; Allen, J.; Beers, A.; Guinand, L.; Linder, R.; McLaughlin, P.; Meer, B.; Musonda, D.; Perdue, D.; Poisson, S.; et al. World agriculture and soil erosion. BioScience 1987, 37, 277–283. [Google Scholar] [CrossRef]
- Lal, R. Soil erosion and the global carbon budget. Environ. Int. 2003, 29, 437–450. [Google Scholar] [CrossRef]
- USDA Natural Resources Conservation Service. National Resources Inventory. 2015. Available online: https://www.nrcs.usda.gov/wps/portal/nrcs/main/national/technical/nra/nri/ (accessed on 4 April 2019).
- Nearing, M.A. Potential changes in rainfall erosivity in the U.S. with climate change during the 21st Century. J. Soil Water Conserv. 2001, 56, 229–232. [Google Scholar]
- Nearing, M.A.; Xie, Y.; Liu, B.; Ye, Y. Natural and anthropogenic rates of soil erosion. Int. Soil Water Conserv. Res. 2017, 5, 77–84. [Google Scholar] [CrossRef]
- National Research Council. Soil and Water Quality: An Agenda for Agriculture; National Academy Press: Washington, DC, USA, 1993. [Google Scholar]
- Karlen, D.L.; Rice, C.W. Soil Degradation: Will Humankind Ever Learn? Sustainability 2015, 7, 12490–12501. [Google Scholar] [CrossRef]
- Doetterl, S.; Berhe, A.A.; Nadeu, E.; Wang, Z.; Sommer, M.; Fiener, P. Erosion, deposition and soil carbon: A review of process-level controls, experimental tools and models to address C cycling in dynamic landscapes. Earth Sci. Rev. 2015, 154, 102–122. [Google Scholar] [CrossRef]
- Li, X.; McCarty, G.W.; Karlen, D.L.; Cambardella, C.A. Topographic metric predictions of soil redistribution and organic carbon in Iowa cropland fields. Catena 2017, 160, 222–232. [Google Scholar] [CrossRef]
- Harden, J.W.; Sharpe, J.M.; Parton, W.J.; Ojima, D.S.; Fries, T.L.; Huntington, T.G.; Dabney, S.M. Dynamic replacement and loss of soil carbon on eroding cropland. Glob. Biogeochem. Cycles 1999, 13, 885–901. [Google Scholar] [CrossRef]
- McCarty, G.W.; Ritchie, J.C. Impact of soil movement on carbon sequestration in agricultural ecosystems. Environ. Pollut. 2002, 116, 423–430. [Google Scholar] [CrossRef]
- Montgomery, D.R. Soil erosion and agricultural sustainability. Prod. Natl. Acad. Sci. USA 2007, 104, 13268–13272. [Google Scholar] [CrossRef]
- Weil, R.R.; Brady, N.C. The Nature and Properties of Soils, 15th ed.; Pearson: New York, NY, USA, 2016; ISBN 978-0-13-325448-8. [Google Scholar]
- Moore, K.J.; Karlen, D.L. Double cropping opportunities for biomass crops in the north central USA. Biofuels 2013, 4, 605–615. [Google Scholar] [CrossRef]
- Appelgate, S.R.; Lenssen, A.W.; Wiedenhoeft, M.H.; Kaspar, T.C. Cover crop options and mixes for upper midwest corn–soybean systems. Agron. J. 2017, 109, 968–984. [Google Scholar] [CrossRef]
- Malone, R.W.; Kersebaum, K.C.; Kaspar, T.C.; Ma, L.; Jaynes, D.B.; Gillette, K. Winter rye as a cover crop reduces nitrate loss to subsurface drainage as simulated by HERMES. Agric. Water Manag. 2017, 184, 156–169. [Google Scholar] [CrossRef]
- Blanco-Canqui, H.; Shaver, T.M.; Lindquist, J.L.; Shapiro, C.A.; Elmore, R.A.; Francis, C.A.; Hergert, G.W. Cover crops and ecosystem services: Insights from studies in termperate soils. Agron. J. 2015, 107, 2449–2474. [Google Scholar] [CrossRef]
- Pantoja, J.L.; Woli, K.P.; Sawyer, J.E.; Barker, D.W. Corn nitrogen fertilization requirement and corn–soybean productivity with a rye cover crop. Soil Sci. Soc. Am. J. 2015, 79, 1482–1495. [Google Scholar] [CrossRef]
- Miguez, F.E.; Bollero, G.A. Review of corn yield response under winter cover cropping systems using meta-analytic methods. Crop Sci. 2005, 45, 2318–2329. [Google Scholar] [CrossRef]
- Acharya, J.; Bakker, M.G.; Moorman, T.B.; Kaspar, T.C.; Lenssen, A.W.; Robertson, A.E. Effects of fungicide seed treatments and a winter rye cover crop in no till on the seedling disease complex in corn. Can. J. Plant Pathol. 2018, 40, 481–497. [Google Scholar] [CrossRef]
- Bakker, M.G.; Acharya, J.; Moorman, T.B.; Robertson, A.E.; Kaspar, T.C. The potential for cereal rye cover crops to host corn seedling pathogens. Phytopathology 2016, 106, 591–601. [Google Scholar] [CrossRef]
- Bakker, M.G.; Manter, D.K.; Moorman, T.B.; Kaspar, T.C. Isolation of cultivation-resistant oomycetes, first detected as amplicon sequences, from roots of herbicide-terminated winter rye. Phytobiomes 2017, 1, 24–35. [Google Scholar] [CrossRef]
- Martinez-Feria, R.A.; Jacobs, K.L.; Weidenhoeft, M.H. Estimated Costs of Production for Winter Canola in Iowa; Ag Decision Maker, Information File and Decision Tool, File A1-24; Iowa State University Extension and Outreach: Ames, IA, USA, July 2016. [Google Scholar]
- Wiedenhoeft, M.H.; Martinez-Feria, R.A.; Kaspar, T.C.; Jacobs Keri, L. Suitability of Winter Canola (Brassica napus) for Enhancing Summer Annual Crop Rotations in Iowa. Master’s Thesis, Iowa State University, Ames, IA, USA, 2015. [Google Scholar]
- Arbuckle, J.G. Iowa Farm and Rural Life Poll Summary Report; PM 3075; Iowa State University: Ames, IA, USA, 2018. [Google Scholar]
- Coppess, J.; Schnitkey, G. Farm bill issue review: Crop insurance and cover crops. Farmdoc Daily 2017, 7, 173. Available online: https://farmdocdaily.illinois.edu/2017/09/farm-bill-issue-review-crop-insurance-cover-crops.html (accessed on 27 March 2019).
- USDA RMA. Cover Crops and Crop Insurance; Risk Management Agency, Washington National Office: Washington, DC, USA, 2018. Available online: https://www.rma.usda.gov/Fact-Sheets/National-Fact-Sheets/Cover-Crops-and-Crop-Insurance (accessed on 27 March 2019).
- Wiggans, D.R.; Singer, J.W.; Moore, K.J.; Lamkey, K.R. Response of continuous maize with stover removal to living mulches. Agron. J. 2012, 104, 917–925. [Google Scholar] [CrossRef]
- Wiggans, D.R.; Singer, J.W.; Moore, K.J.; Lamkey, K.R. Continuation of study cited in 81. Unpublished work. 2011. [Google Scholar]
- Maddonni, G.A.; Otegui, M.E.; Andrieu, B.; Chelle, M.; Casal, J.J. Maize leaves turn away from neighbors. Plant Phys. 2002, 130, 1181–1189. [Google Scholar] [CrossRef] [PubMed]
- Dubois, P.G.; Olsefski, G.T.; Flint-Garcia, S.; Setter, T.L.; Hoekenga, O.A.; Brutnell, T.P. Physiological and genetic characterization of end-of-day far-red light response in maize seedlings. Plant Phys. 2010, 154, 173–186. [Google Scholar] [CrossRef] [PubMed]
- Bartel, C.A.; Banik, C.; Lenssen, A.W.; Moore, K.J.; Laird, D.A.; Archontoulis, S.V.; Lamkey, K.R. Establishment of perennial groundcovers for maize-based bioenergy production systems. Agron. J. 2017, 109, 1–14. [Google Scholar] [CrossRef]
- Bartel, C.A.; Banik, C.; Lenssen, A.W.; Moore, K.J.; Laird, D.A.; Archontoulis, S.V.; Lamkey, K.R. Living mulch for sustainable maize stover biomass harvest. Crop Sci. 2017, 57, 3273–3290. [Google Scholar] [CrossRef]
- Elkins, D.; Frederking, D.; Marashi, R.; McVay, B. Living mulch for no-till corn and soybeans. J. Soil Water Conserv. 1983, 38, 431–433. [Google Scholar]
- Elkins, D.M.; Vandeventer, J.W.; Xapusta, G.; Anderson, M.R. No-tillage maize production in chemically suppressed grass sod. Agron. J. 1979, 71, 101–105. [Google Scholar] [CrossRef]
- Sanders, Z.P.; Andrews, J.S.; Saha, U.K.; Vencill, W.; Lee, R.D.; Hill, N.S. Optimizing agronomic practices for clover persistence and corn yield in a white clover–corn living mulch system. Agron. J. 2017, 109, 2025–2032. [Google Scholar] [CrossRef]
- Andrews, J.S.; Sanders, Z.P.; Cabrera, M.L.; Saha, U.K.; Hill, N.S. Nitrogen dynamics in living mulch and annual cover crop corn production systems. Agron. J. 2018, 110, 1309–1317. [Google Scholar] [CrossRef]
- Duiker, S.W.; Hartwig, N.L. Living mulches of legumes in imidazolinone-resistant corn. Agron. J. 2004, 96, 1021–1028. [Google Scholar] [CrossRef]
- Wiggans, D.R.; Singer, J.W.; Moore, K.J.; Lamkey, K.R. Maize water use in living mulch systems with stover removal. Crop Sci. 2012, 52, 327–338. [Google Scholar] [CrossRef]
- Qi, Z.; Helmers, M.J.; Christianson, R.D.; Pederson, C.H. Nitrate-nitrogen losses through subsurface drainage under various agricultural land covers. J. Environ. Qual. 2011, 40, 1578–1585. [Google Scholar] [CrossRef] [PubMed]
- Subbarao, G.V.; Nakahara, K.; Hurtado, M.P.; Ono, H.; Moreta, D.E.; Salcedo, A.F.; Yoshihashi, A.T.; Ishikawa, T.; Ishitani, M.; Ohnishi-Kameyama, M.; et al. Evidence for biological nitrification inhibition in Brachiaria pastures. Prod. Natl. Acad. Sci. USA 2009, 106, 17302–17307. [Google Scholar] [CrossRef] [PubMed]
- Subbarao, G.V.; Sahrawat, K.L.; Nakahara, K.; Ishikawa, T.; Kishii, M.; Rao, I.M.; Hash, C.T.; George, T.S.; Srinivasa Rao, P.; Nardi, P.; et al. Biological nitrification inhibition—A novel strategy to regulate nitrification in agricultural systems. Adv. Agron. 2012, 114, 249–302. [Google Scholar]
- Young, C.A.; Hume, D.E.; McCulley, R.L. Fungal endophytes of tall fescue and perennial ryegrass: Pasture friend or foe? J. Anim. Sci. 2013, 91, 2379–2394. [Google Scholar] [CrossRef]
- Takach, J.E.; Young, C.A. Alkaloid genotype diversity of tall fescue endophytes. Crop Sci. 2014, 54, 667–678. [Google Scholar] [CrossRef]
- Böttinger, S.; Doluschitz, R.; Klaus, J.; Jenane, C.; Samarakoon, N. Agricultural Development and Mechanization in 2013: A Comparative Survey at a Global Level. Presented at the Fourth World Summit on Agriculture Machinery, New Delhi, India, 5–6 December 2013; Available online: http://www.clubofbologna.org/ew/documents/3_1a_KNR_Samarkoon.pdf (accessed on 21 May 2019).
- Honour, E.C. A historical perspective on systems Engineering. Syst. Eng. 2018, 21, 148–151. [Google Scholar] [CrossRef]
- SMC Systems. SMC Systems Engineering Primer & Guide: Concepts, Processes, and Techniques, 2nd ed.; Space & Missile Systems Center, US Air Force: El Segundo, CA, USA, 2004.
- Würschum, T.; Leiser, W.L.; Jähne, F.; Bachteler, K.; Miersch, M.; Hahn, V. The soybean experiment ‘1000 Gardens’: A case study of citizen science for research, education, and beyond. Theor. Appl. Genet. 2019, 132, 617–626. [Google Scholar] [CrossRef]
- Sanders, I.R. Preference, specificity and cheating in the arbuscular mycorrhizal symbiosis. Trends Plant Sci. 2003, 8, 143–145. [Google Scholar] [CrossRef]
- Stachowicz, J.J. Mutualism, facilitation, and the structure of ecological communities: Positive interactions play a critical, but underappreciated, role in ecological communities by reducing physical or biotic stresses in existing habitats and by creating new habitats on which many species depend. BioScience 2001, 51, 235–246. [Google Scholar]
- Schlaeppi, K.; Bulgarelli, D. The plant microbiome at work. MPMI 2015, 28, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Liebman, M.; Dyck, E. Crop rotation and intercropping strategies for weed management. Ecol. Appl. 1993, 3, 92–122. [Google Scholar] [CrossRef] [PubMed]
- Levine, J.M.; HilleRisLambers, J. The importance of niches for the maintenance of species diversity. Nature 2009, 461, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Flynn, E.S.; Moore, K.J.; Singer, J.; Lamkey, K.R. Evaluation of grass and legume species as perennial ground covers in corn production. Crop Sci. 2013, 53, 611–620. [Google Scholar] [CrossRef]
- Franklin, K.A. Shade avoidance. New Phytol. 2008, 179, 930–944. [Google Scholar] [CrossRef] [PubMed]
- Rajcan, I.; Chandler, K.J.; Swanton, C.J. Red-far-red ratio of reflected light: A hypothesis of why early-season weed control is important for corn. Weed Sci. 2017, 52, 774–778. [Google Scholar] [CrossRef]
- Ballaré, C.L.; Scopel, A.L.; Sanchez, R.A. Far-red radiation reflected from adjacent leaves: An early signal of competition in plant canopies. Science 1990, 247, 329–331. [Google Scholar] [CrossRef] [PubMed]
- Markelz, N.H.; Costich, D.E.; Brutnell, T.P. Photomorphogenic responses in maize seedling development. Plant Physiol. 2003, 2003. 133, 1578–1591. [Google Scholar] [CrossRef]
- Page, E.R.; Tollenaar, M.; Lee, E.A.; Lukens, L.; Swanton, C.J. Does the shade avoidance response contribute to the critical period for weed control in maize (Zea mays)? Weed Res. 2009, 49, 563–571. [Google Scholar] [CrossRef]
- Graven, L.M.; Carter, P.R. Seed size/shape and tillage system effect on corn growth and grain yield. J. Prod. Agric. 1990, 3, 445–452. [Google Scholar] [CrossRef]
- Nafziger, E.D. Seed size effects on yield of two corn hybrids. J. Prod. Agric. 1992, 5, 538–540. [Google Scholar] [CrossRef]
- Hunter, R.B.; Kannenberg, L.W. Effect of seed size on emergence, grain yield, and plant height in corn. Can. J. Plant Sci. 1972, 52, 252–256. [Google Scholar] [CrossRef]
- Löf, M.; Welander, N.T. Influence of herbaceous competitors on early growth in direct seeded Fagus sylvatica L. and Quercus robur L. Ann. For. Sci. 2004, 61, 781–788. [Google Scholar] [CrossRef]
- Vandenberghe, C.; Freléchoux, F.; Gadallah, F.; Buttler, A. Competitive effects of herbaceous vegetation on tree seedling emergence, growth, and survival: Does gap size matter. J. Veg. Sci. 2006, 17, 481–488. [Google Scholar] [CrossRef]
- Wilkinson, S.R.; Gross, C.F. Competition for light, soil moisture and nutrients during ladino clover establishment in orchardgrass sod. Agron. J. 1964, 56, 389–392. [Google Scholar] [CrossRef]
- Dear, B.S.; Cocks, P.S.; Wolfe, E.C.; Collins, D.P. Established perennial grasses reduce the growth of emerging subterranean clover seedlings through competition for water, light, and nutrients. Aust. J. Agric. Res. 1998, 49, 41–51. [Google Scholar] [CrossRef]
- Rajcan, I.; Swanton, C.J. Understanding maize-weed competition: Resource competition, light quality and whole plant. Field Crops Res. 2001, 71, 139–150. [Google Scholar] [CrossRef]
- Gibson, K.D.; Fischer, A.J.; Foin, T.C.; Hill, J.E. Implications of delayed Echinochloa spp. germination and duration of competition for integrated weed management in water-seeded rice. Weed Res. 2002, 42, 351–358. [Google Scholar] [CrossRef]
- Sadanandan Nambiar, E.K. Interplay between nutrients, water, root growth and productivity in young plantations. For. Ecol. Manag. 1990, 30, 213–232. [Google Scholar] [CrossRef]
- Kasperbauer, M.J. Far-red light reflection from green leaves and effects on phytochrome-mediated assimilate partitioning under field conditions. Plant Physiol. 1987, 85, 350–354. [Google Scholar] [CrossRef]
- Ferrell, J.; Murphy, T.; Waltz, C.; Yelverton, F. Sulfonylurea herbicides: How do different turfgrasses tolerate them? Turfgrass Trends 2004, 13, 92–96. [Google Scholar]
- Sumner, P.E.; Culpepper, S. How to Set up a Post-Emergence Directed and Shielded Herbicide Sprayer for Cotton; Bulletin 1069; University Georgia Extension: Athens, GA, USA, 2017; p. 4. [Google Scholar]
- Kromp, B. Carabid beetles in sustainable agriculture: A review on pest control efficacy, cultivation impacts and enhancement. Agric. Ecosyst. Environ. 1999, 74, 187–228. [Google Scholar] [CrossRef]
- Gallandt, E.R.; Molloy, T.; Lynch, R.P.; Drummond, F.A. Effect of cover-cropping systems on invertebrate seed predation. Weed Sci. 2005, 53, 69–76. [Google Scholar] [CrossRef]
- Menalled, F.D.; Smith, R.G.; Dauer, J.T.; Fox, T.B. Impact of agricultural management on carabid communities and weed seed predation. Agric. Ecosyst. Environ. 2007, 118, 49–54. [Google Scholar] [CrossRef]
- McCravy, K.W.; Lundgren, J.G. Carabid beetles (Coleoptera: Carabidae) of the Midwestern United States: A review and synthesis of recent research. Terr. Arthropod Rev. 2011, 4, 63–94. [Google Scholar] [CrossRef]
- Liebman, M.; Gallandt, E.R. Many little hammers: Ecological management of crop-weed interactions. In Ecology in Agriculture; Jackson, L.E., Ed.; Academic Press: San Diego, CA, USA, 1997; pp. 291–343. [Google Scholar]
- Norsworthy, J.K.; Ward, S.M.; Shaw, D.R.; Llewellyn, R.S. Reducing the risks of herbicide resistance: Best management practices and recommendations. Weed Sci. 2012, 60, 31–62. [Google Scholar] [CrossRef]
- Lundgren, J.G.; Fergen, J.K. Predator community structure and trophic linkage strength to a focal prey. Mol. Ecol. 2014, 23, 3790–3798. [Google Scholar] [CrossRef]
- Barnard, C.; Frankel, O.H. Grass, grazing animals, and Man in historic perspective. In Grasses and Grasslands; Barnard, C., Ed.; St. Martin’s Press: New York, NY, USA, 1964. [Google Scholar]
- Funk, C.R.; Meyer, W.A. 70 years of turfgrass improvement at the New Jersey Agricultural Experiment Station. USGA Green Sect. Rec. 2001, 39, 19–23. [Google Scholar]
- Sleper, D.A. Breeding tall fescue. Plant Breed. Rev. 1985, 3, 313–342. [Google Scholar]
- Beard, J.B. Turfgrass: Science and Culture; Pearson Prentice Hall, Inc.: Englewood Cliffs, NJ, USA, 1973. [Google Scholar]
- Curley, J.; Jung, G. RAPD-based genetic relationships in Kentucky bluegrass. Crop Sci. 2003, 44, 1299–1306. [Google Scholar] [CrossRef]
- Trammell, M.A.; Butler, T.J.; Young, C.A.; Widdup, K.; Amadeo, J.; Hopkins, A.A.; Nyaupane, N.P.; Biermacher, J.T. Registration of ‘Chisholm’ summer-dormant tall fescue. J. Plant Regist. 2018, 12, 293–299. [Google Scholar] [CrossRef]
- Horton, M.L. The nature of summer dormancy in perennial grasses. Bot. Gaz. 1953, 114, 284–292. [Google Scholar]
- Norton, M.R.; Volaire, F.; Lelievre, F.; Fukai, S. Identification and measurement of summer dormancy in temperate perennial grasses. Crop Sci. 2009, 49, 2347–2352. [Google Scholar] [CrossRef]
- Pepin, G.W.; Funk, C.R. Intraspecific hybridization as a method of breeding kentucky bluegrass (Poa pratenis L.) for turf. Crop Sci. 1971, 11, 445–448. [Google Scholar] [CrossRef]
- Ishikawa, T.; Subbarao, G.V.; Ito, O.; Okada, K. Suppression of nitrification and nitrous oxide emission by the tropical grass Brachiaria humidicola. Plant Soil 2003, 255, 413–419. [Google Scholar] [CrossRef]
- Subbarao, G.V.; Kishii, M.; Nakahara, K.; Ishikawa, T.; Ban, T.; Tsujimoto, H.; George, T.S.; Berry, W.L.; Hash, C.T.; Ito, O. Biological nitrification inhibition (BNI)—Is there potential for genetic interventions in the Triticeae? Breed. Sci. 2009, 59, 529–545. [Google Scholar] [CrossRef]
- Munro, P.E. Inhibition of nitrite-oxidizers by roots of grass. J. Appl. Ecol. 1966, 3, 231–238. [Google Scholar] [CrossRef]
- Moore, D.R.; Waide, J.S. The influence of washings of living roots on nitrification. Soil Biol. Biochem. 1971, 3, 69–83. [Google Scholar] [CrossRef]
- Bonos, S.A.; Meyer, W.A.; Murphy, J.A. Classification of Kentucky bluegrass genotypes grown as spaced-plants. HortScience 2000, 35, 910–913. [Google Scholar] [CrossRef]
- Honig, J.A.; Averello, V.; Bonos, S.A.; Meyer, W.A. Classification of Kentucky bluegrass (Poa pratensis L.) cultivars and accessions based on microsatellite (simple sequence repeat) markers. HortScience 2012, 47, 1356–1366. [Google Scholar] [CrossRef]
- Wieners, R.R.; Fei, S.Z.; Johnson, R.C. Characterization of a USDA Kentucky bluegrass (Poa pratensis L.) core collection for reproductive mode and DNA content by flow cytometry. Gen. Res. Crop Evol. 2006, 53, 1531–1541. [Google Scholar] [CrossRef]
- Rajasekar, S.; Fei, S.Z.; Christians, N.E. Analysis of genetic diversity in colonial bentgrass (Agrostis capillaris L.) using randomly amplified polymorphic DNA (RAPD) markers. Gen. Res. Crop Evol. 2007, 54, 45–53. [Google Scholar] [CrossRef]
- Fei, S.Z. Recent progresses on turfgrass molecular genetics and biotechnology. Acta Hortic. 2008, 783, 247. [Google Scholar]
- Yang, N.; Xu, X.W.; Wang, R.R.; Peng, W.L.; Cai, L.; Song, J.M.; Li, W.; Luo, X.; Niu, L.; Wang, Y.; et al. Contributions of Zea mays subspecies mexicana haplotypes to modern maize. Nat Commun. 2017, 8, 1874. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.; Cooper, M.; Gogerty, J.; Löffler, C.; Borcherding, D.; Wright, K. Maize. In Yield Gains in Major U.S. Field Crops; CSSA Special Publications 33; Smith, S., Specht, J., Diers, B., Carver, B., Eds.; ASA-CSSA-SSSA: Madison, WI, USA, 2014; pp. 125–171. [Google Scholar]
- Schnable, P.S.; Ware, D.; Fulton, R.S.; Stein, J.C.; Wei, F.; Pasternak, S.; Liang, C.; Zhang, J.; Fulton, L.; Graves, T.A.; et al. The B73 maize genome: Complexity, diversity, and dynamics. Science 2009, 326, 1112–1115. [Google Scholar] [CrossRef]
- Hirsch, C.N.; Foerster, J.M.; Johnson, J.M.; Sekhon, R.S.; Muttoni, G.; Vaillancourt, B.; Penagaricano, F.; Lindquist, E.; Pedraza, M.A.; Barry, K.; et al. Insights into the maize pan-genome and pan-transcriptome. Plant Cell 2014, 26, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.; Romay, M.C.; Glaubitz, J.C.; Bradbury, P.J.; Elshire, R.J.; Wang, T.; Li, Y.; Li, Y.; Semagn, K.; Zhang, X.; et al. High-resolution genetic mapping of maize pan-genome sequence anchors. Nat. Commun. 2015, 6, 6914. [Google Scholar] [CrossRef]
- Sun, S.; Zhou, Y.; Chen, J.; Shi, J.; Zhao, H.; Zhao, H.; Song, W.; Zhang, M.; Cui, Y.; Dong, X.; et al. Extensive intraspecific gene order and gene structural variations between Mo17 and other maize genomes. Nat. Genet. 2018, 50, 1289–1295. [Google Scholar] [CrossRef]
- Unterseer, S.; Seidel, M.A.; Bauer, E.; Haberer, G.; Hochholdinger, F.; Opitz, N.; Marcon, C.; Baruch, K.; Spannagl, M.; Mayer, K.F.X.; et al. European Flint reference sequences complement the maize pan-genome. bioRxiv 2017. [Google Scholar] [CrossRef]
- Xu, P.; Wang, L.; Beavis, W.D. An optimization approach to gene stacking. Eur. J. Oper. Res. 2011, 214, 168–178. [Google Scholar] [CrossRef]
- Cooper, M.; Gho, C.; Leafgren, R.; Tang, T.; Messina, C. Breeding drought-tolerant maize hybrids for the US corn-belt: Discovery to product. J. Exp. Bot. 2014, 65, 6191–6204. [Google Scholar] [CrossRef] [PubMed]
- Meuwissen, T.H.E.; Hayes, B.J.; Goddard, M.E. Prediction of total genetic value using genome-wide dense marker maps. Genetics 2001, 157, 1819–1829. [Google Scholar] [PubMed]
- Shi, J.; Gao, H.; Wang, H.; Lafitte, R.; Archibald, R.L.; Yang, M.; Hakimi, S.M.; Mo, H.; Habben, J.E. ARGOS8 variants generated by CRISPR-Cas9 improve maize grain under field drought stress conditions. Plant Biotechnol. J. 2017, 15, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Svitashev, S.; Young, J.K.; Schwartz, C.; Gao, H.; Falco, S.C.; Cigan, M.A. Targeted mutagenesis; precise gene editing; and site-specific gene insertion in maize using Cas9 guide RNA. Plant Physiol. 2015, 169, 931–945. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Rasheed, A.; Hickey, L.T.; He, Z. Fast-forwarding genetic gain trends. Plant Sci. 2018, 23, 184–186. [Google Scholar] [CrossRef]
- Technow, F.; Messina, C.D.; Totir, L.R.; Cooper, M. Integrating crop growth models with whole genome prediction through approximate Bayesian computation. PLoS ONE 2015, 10, e0130855. [Google Scholar] [CrossRef] [PubMed]
- Russell, A.E.; Laird, D.A.; Parkin, T.B.; Mallarino, A.P. Impact of nitrogen fertilization and cropping system on carbon sequestration in Midwestern Mollisols. Soil Sci. Soc. Am. J. 2005, 69, 413–422. [Google Scholar] [CrossRef]
- Schlesinger, W.H. Changes in soil carbon storage and associated properties with disturbance and recovery. In The Changing Carbon Cycle; Springer: New York, NY, USA, 1986; pp. 194–220. [Google Scholar]
- Zajicek, J.M.; Hetrick, B.D.; Owensby, C.E. The influence of soil depth on mycorrhizal colonization of forbs in the tallgrass prairie. Mycologia 1986, 78, 316–320. [Google Scholar] [CrossRef]
- Miller, R.M.; Jastrow, J.D. Mycorrhizal fungi influence soil structure. In Arbuscular Mycorrhizas: Physiology and Function; Springer: Dordrecht, The Netherlands, 2000; pp. 3–18. [Google Scholar]
- Jastrow, J.D.; Miller, R.M.; Lussenhop, J. Contributions of interacting biological mechanisms to soil aggregate stabilization in restored prairie. Soil Biol. Biochem. 1998, 30, 905–916. [Google Scholar] [CrossRef]
- Gale, W.J.; Cambardella, C.A.; Bailey, T.B. Root-derived carbon and the formation and stabilization of aggregates. Soil Sci. Soc. Am. J. 2000, 64, 201–207. [Google Scholar] [CrossRef]
- Fuhlendorf, S.D.; Engle, D.M. Restoring heterogeneity on rangelands: Ecosystem management based on evolutionary grazing patterns. BioScience 2001, 51, 625–632. [Google Scholar] [CrossRef]
- Fierer, N.; Ladau, J.; Clemente, J.C.; Leff, J.W.; Owens, S.M.; Pollard, K.S.; Knight, R.; Gilbert, J.A.; McCulley, R.L. Reconstructing the microbial diversity and function of pre-agricultural tallgrass prairie soils in the United States. Science 2013, 342, 621–623. [Google Scholar] [CrossRef] [PubMed]
- Fisher, B.; Turner, R.K.; Morling, P. Defining and classifying ecosystem services for decision making. Ecol. Econ. 2009, 68, 643–653. [Google Scholar] [CrossRef]
- Keiser, D.A.; Kling, C.L.; Shapiro, J.S. The low but uncertain measured benefits of U.S. water quality policy. Prod. Natl. Acad. Sci. USA 2019, 116, 5262–5269. [Google Scholar] [CrossRef] [PubMed]
- Hoque, M.; Kling, C.L. Economic Valuation of Ecosystem Benefits from Conservation Practices Targeted in Iowa Nutrient Reduction Strategy 2013: A Non Market Valuation Approach; Working Paper 16-WP 561; Center for Agricultural and Rural Development, Iowa State University: Ames, IA, USA, 2016. [Google Scholar]
- Power, A.G. Ecosystem services and agriculture: Tradeoffs and synergies. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 2959–2971. [Google Scholar] [CrossRef]
- Schipanski, M.E.; Barbercheck, M.; Douglas, M.R.; Finney, D.M.; Haider, K.; Kaye, J.P.; Kermanian, A.R.; Mortensen, D.A.; Ryan, M.R.; Tooker, J.; et al. A framework for evaluating ecosystem services provided by cover crops in agroecosystems. Agric. Syst. 2014, 125, 12–22. [Google Scholar] [CrossRef]
- Mayer, P.M.; Reynolds, S.K.; McCutchen, M.D.; Canfield, T.J. Meta-analysis of nitrogen removal in riparian buffers. J. Environ. Qual. 2007, 36, 1172–1180. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, X.; Zhang, M.; Dahlgren, R.A.; Eitzel, M. A review of vegetated buffers and a meta-analysis of their mitigation efficacy in reducing nonpoint source pollution. J. Environ. Qual. 2010, 39, 76–84. [Google Scholar] [CrossRef]
- Blanco-Canqui, H.; Gantzer, C.J.; Anderson, S.H.; Alberts, E.E.; Thompson, A.L. Grass barrier and vegetative filter strip effectiveness in reducing runoff, sediment, nitrogen, and phosphorus loss. Soil Sci. Soc. Am. J. 2004, 68, 1670–1678. [Google Scholar] [CrossRef]
- Blanco-Canqui, H.; Gantzer, C.J.; Anderson, S.H. Performance of grass barriers and filter strips under interrill and concentrated flow. J. Environ. Qual. 2006, 35, 1969–1974. [Google Scholar] [CrossRef]
- Bugg, R.L.; Waddington, C. Using cover crops to manage arthropod pests of orchards: A review. Agric. Ecosyst. Environ. 1994, 50, 11–28. [Google Scholar] [CrossRef]
- Hartwig, N.L.; Ammon, H.U. Cover crops and living mulches. Weed Sci. 2002, 50, 688–699. [Google Scholar] [CrossRef]
- Frank, D.L.; Liburd, O.E. Effects of living and synthetic mulch on the population dynamics of whiteflies and aphids, their associated natural enemies, and insect-transmitted plant diseases in zucchini. Environ. Entomol. 2005, 34, 857–865. [Google Scholar] [CrossRef]
- Hooks, C.R.; Johnson, M.W. Using undersown clovers as living mulches: Effects on yields, lepidopterous pest infestations, and spider densities in a Hawaiian broccoli agroecosystem. Int. J. Pest Manag. 2004, 50, 115–120. [Google Scholar] [CrossRef]
- Bianchi, F.J.; Booij, C.J.H.; Tscharntke, T. Sustainable pest regulation in agricultural landscapes: A review on landscape composition, biodiversity and natural pest control. Proc. R. Soc. B Biol. Sci. 2006, 273, 1715–1727. [Google Scholar] [CrossRef]
- Prasifka, J.R.; Schmidt, N.P.; Kohler, K.A.; O’neal, M.E.; Hellmich, R.L.; Singer, J.W. Effects of living mulches on predator abundance and sentinel prey in a corn–soybean–forage rotation. Environ. Entomol. 2006, 35, 1423–1431. [Google Scholar] [CrossRef]
- Ragsdale, D.W.; Voegtlin, D.J.; O’neil, R.J. Soybean aphid biology in North America. Ann. Entomol. Soc. Am. 2004, 97, 204–208. [Google Scholar] [CrossRef]
- Pratt, M.R.; Tyner, W.E.; Muth, D.J., Jr.; Kladivko, E.J. Synergies between cover crops and corn stover removal. Agric. Syst. 2014, 130, 67–76. [Google Scholar] [CrossRef]
- Bonner, I.J.; Muth, D.J.; Koch, J.B.; Karlen, D.L. Modeled impacts of cover crops and vegetative barriers on corn stover availability and soil quality. BioEnergy Res. 2014, 7, 576–589. [Google Scholar] [CrossRef]
- Rogovska, N.; Laird, D.A.; Rathke, S.J.; Karlen, D.L. Biochar impact on Midwestern Mollisols and maize nutrient availability. Geoderma 2014, 230–231, 340–347. [Google Scholar] [CrossRef]
- Aller, D.; Archontoulis, S.; Zhang, W.; Sawadgo, W.; Laird, D.; Moore, K. Long term biochar effects on corn yield, soil quality, and profitability in the US Midwest. Field Crops Res. 2018, 227, 30–40. [Google Scholar] [CrossRef]
- Zhang, W.; Plastina, A.; Sawadgo, W. Iowa Farmland Ownership and Tenure Survey 1982–2017: A Thirty-Five Year Perspective; Working Paper 18-WP 580; Center for Agricultural and Rural Development, Iowa State University: Ames, IA, USA, 2018. [Google Scholar]
- Bergtold, J.S.; Ramsey, S.; Maddy, L.; Williams, J.R. A review of economic considerations for cover crops as a conservation practice. Renew. Agric. Food Syst. 2017, 34, 62–76. [Google Scholar] [CrossRef]
- Lu, Y.C.; Watkins, K.B.; Teasdale, J.R.; Abdul-Baki, A.A. Cover crops in sustainable food production. Food Rev. Int. 2000, 16, 121–157. [Google Scholar] [CrossRef]
- Borghi, E.; Crusciol, C.A.C.; Nascente, A.S.; Sousa, V.V.; Martins, P.O.; Mateus, G.P.; Costa, C. Sorghum grain yield, forage biomass production and revenue as affected by intercropping time. Eur. J. Agron. 2013, 51, 130–139. [Google Scholar] [CrossRef]
- Gardezi, M.; Arbuckle, J.G. The influence of objective and perceived adaptive capacities on Midwestern farmers’ use of cover crops. Weather Clim. Soc. 2019, 11, 665–679. [Google Scholar] [CrossRef]
- Burnett, E.; Wilson, R.S.; Heeren, A.; Martin, J. Farmer adoption of cover crops in the Western Lake Erie Basin. J. Soil Water Conserv. 2018, 73, 143–155. [Google Scholar] [CrossRef]
- Plastina, A.; Liu, F.; Sawadgo, W.; Miguez, F.; Carlson, S.; Marcillo, G. Annual net returns to cover crops in Iowa. J. Appl. Farm Econ. 2018, 2, 19–36. [Google Scholar]
- Kruger, G.R.; Johnson, W.G.; Weller, S.C.; Owen, M.D.K.; Shaw, D.R.; Wilcut, J.W.; Jordan, D.L.; Wilson, R.G.; Bernards, M.L.; Young, B.G. U.S. grower views on problematic weeds and changes in weed pressure in glyphosate-resistant corn, cotton, and soybean cropping systems. Weed Technol. 2009, 23, 162–166. [Google Scholar] [CrossRef]

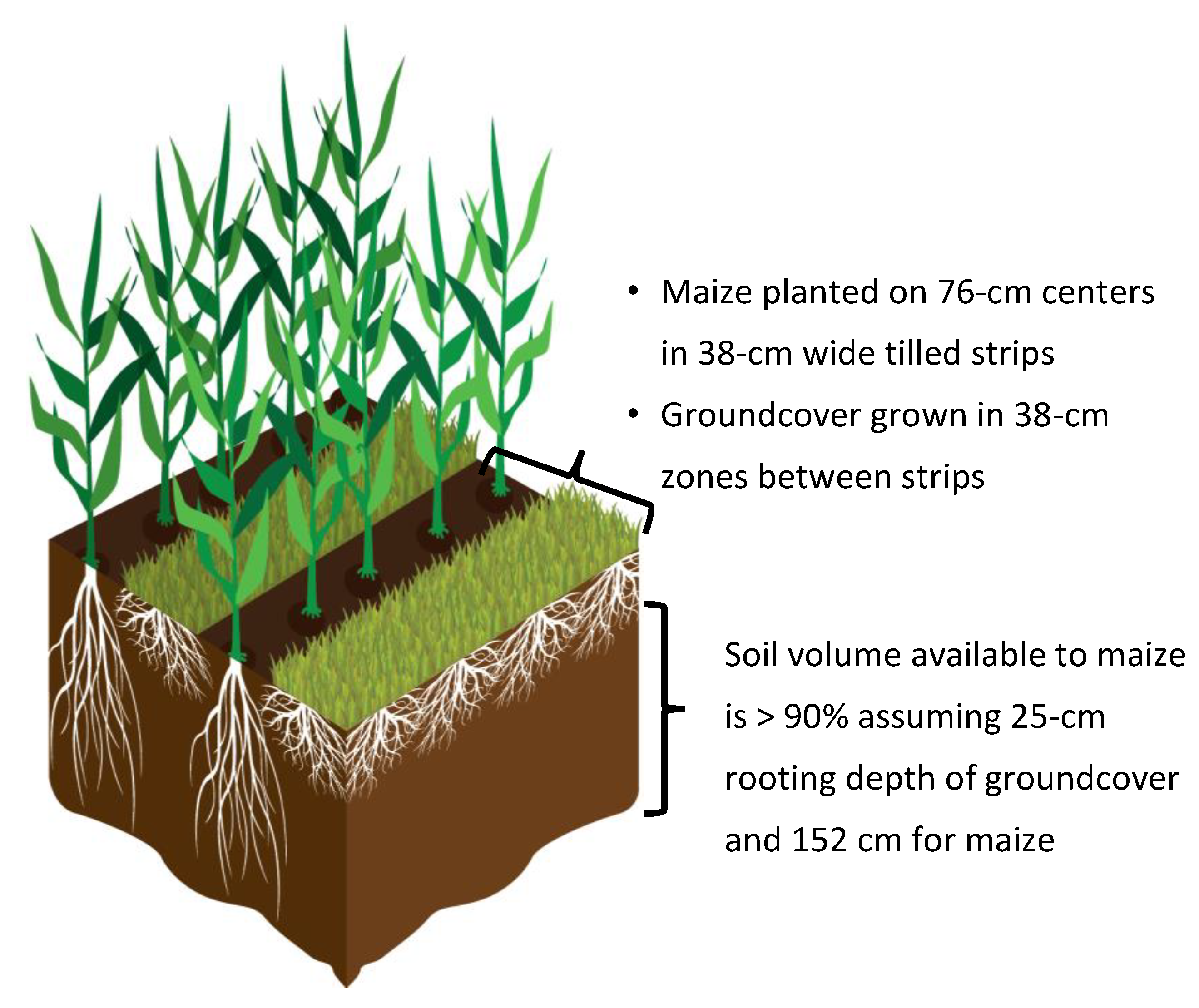
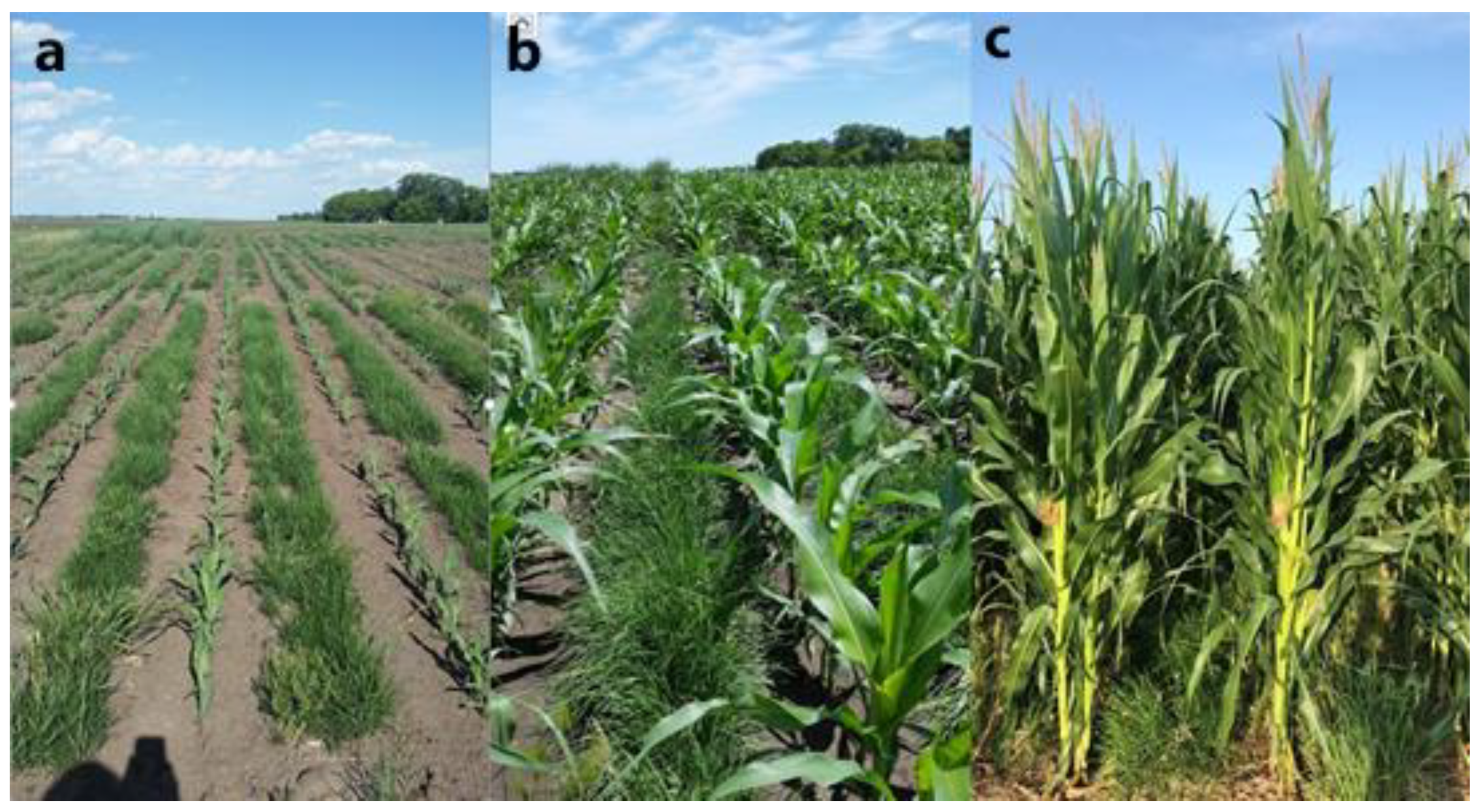

| Site | Prairie | Cropped |
|---|---|---|
| (kg-C/ha) | ||
| Hayden | 182,290 | 126,311 |
| Chipera | 169,809 | 68,170 |
| Larson | 146,590 | 81599 |
| Kalsow | 143,086 | 94,879 |
| Doolittle | 106,766 | 71,665 |
| Ketelsen | 98,362 | 47,726 |
| Average | 141,151 | 81,725 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moore, K.J.; Anex, R.P.; Elobeid, A.E.; Fei, S.; Flora, C.B.; Goggi, A.S.; Jacobs, K.L.; Jha, P.; Kaleita, A.L.; Karlen, D.L.; et al. Regenerating Agricultural Landscapes with Perennial Groundcover for Intensive Crop Production. Agronomy 2019, 9, 458. https://doi.org/10.3390/agronomy9080458
Moore KJ, Anex RP, Elobeid AE, Fei S, Flora CB, Goggi AS, Jacobs KL, Jha P, Kaleita AL, Karlen DL, et al. Regenerating Agricultural Landscapes with Perennial Groundcover for Intensive Crop Production. Agronomy. 2019; 9(8):458. https://doi.org/10.3390/agronomy9080458
Chicago/Turabian StyleMoore, Kenneth J., Robert P. Anex, Amani E. Elobeid, Shuizhang Fei, Cornelia B. Flora, A. Susana Goggi, Keri L. Jacobs, Prashant Jha, Amy L. Kaleita, Douglas L. Karlen, and et al. 2019. "Regenerating Agricultural Landscapes with Perennial Groundcover for Intensive Crop Production" Agronomy 9, no. 8: 458. https://doi.org/10.3390/agronomy9080458
APA StyleMoore, K. J., Anex, R. P., Elobeid, A. E., Fei, S., Flora, C. B., Goggi, A. S., Jacobs, K. L., Jha, P., Kaleita, A. L., Karlen, D. L., Laird, D. A., Lenssen, A. W., Lübberstedt, T., McDaniel, M. D., Raman, D. R., & Weyers, S. L. (2019). Regenerating Agricultural Landscapes with Perennial Groundcover for Intensive Crop Production. Agronomy, 9(8), 458. https://doi.org/10.3390/agronomy9080458








