Effects of Elevated CO2 on Wheat Yield: Non-Linear Response and Relation to Site Productivity
Abstract
1. Introduction
- Does yield stimulation by eCO2 saturate at high CO2 concentration?
- Is eCO2 yield stimulation dependent on experimental conditions (fumigation technique, rooting environment, wheat type, geographic region)?
- Is there a link between eCO2 yield stimulation and agronomic productivity?
- Are yield components (total aboveground biomass, harvest index, grain mass, grain number, specific grain mass) equally affected by eCO2?
2. Materials and Methods
2.1. Database
2.2. Meta-Analysis
2.3. Response Functions
3. Results
4. Discussion
4.1. Non-Linearity of CO2 Response Function
4.2. Relation to Site Productivity
4.3. Experimental Conditions
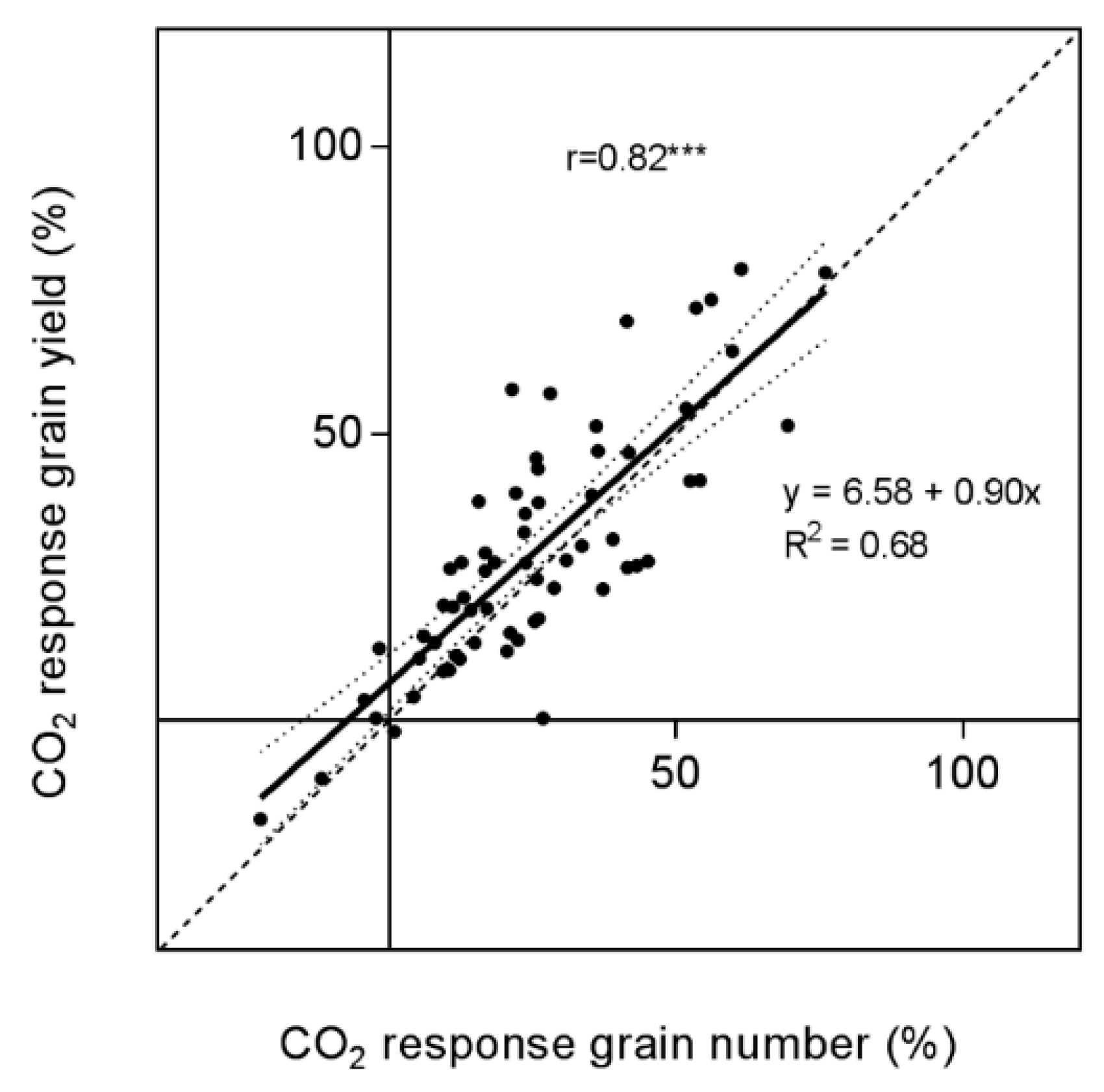
4.4. Yield Components
4.5. Implications for Modelling and Food Security Assessment
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Reference | Grain Yield | Total Aboveground Biomass | Harvest Index | Grain Mass | Grain Number | Specific Grain Mass |
|---|---|---|---|---|---|---|
| Deepak and Agrawal, 1999 [49] | x | x | x | |||
| Dietterich et al., 2015 [60] | x | x | x | x | x | |
| Dijkstra et al., 1999 [61] | x | x | x | x | x | |
| Donnelly et al., 1999 [62] | x | x | x | |||
| Fangmeier et al., 1996 [63] | x | x | x | x | x | |
| Hakala, 1998 [64] | x | x | x | x | x | |
| Houshmandfar et al., 2016 [65] | x | x | x | x | x | |
| Högy, 2002 [66] | x | x | x | x | x | |
| Högy et al., 2009 [44]* | x | x | x | x | x | |
| Högy et al., 2013 [45] | x | x | x | x | x | |
| Kimball et al., 2001 [48] | x | x | x | x | x | |
| Manderscheid and Weigel, 1997 [67] | x | x | x | x | x | |
| Mishra et al., 2013 [68] | x | x | x | x | x | |
| Mortenssen et al. [69]; Fangmeier et al., 1999 [70] | x | x | x | x | ||
| Mulholland et al., 1998 [71] | x | x | x | x | ||
| Fangmeier et al., 1999 [70] Piikki et al., 2008 [72]; | x | x | x | x | x | |
| Pleijel et al., 2000 [73] | x | x | x | x | ||
| Rawson 1995 [74] | x | x | x | |||
| Mulchi et al., 1995 [75]; Rudorff et al., 1996a [76]; Rudorff et al 1996b [77] | x | x | x | x | x | x |
| Van Oijen et al., 1999 [78] | x | x | x | x | x | |
| Weigel and Manderscheid 2012 [79] | x | x | x | |||
| Manderscheid et al., 1995 [80]; Weigel et al., 1994 [81] | x | x | x | x | x | |
| Yang et al., 2007a [82]; Yang et al., 2007b [83] | x | x | x |
References
- FAO. Climate Change and Food Security: Risks and Responses; FAO: Rome, Italy, 2016. [Google Scholar]
- IPCC. Climate Change 2013: The Physical Science Basis; IPCC: Geneva, Switzerland, 2013. [Google Scholar]
- Long, S.P.; Ainsworth, E.A.; Rogers, A.; Ort, D.R. Rising atmospheric carbon dioxide: Plants face the future. Annu. Rev. Plant Biol. 2004, 55, 591–628. [Google Scholar] [CrossRef]
- Wang, L.; Feng, Z.Z.; Schjoerring, J.K. Effects of elevated atmospheric CO2 on physiology and yield of wheat (Triticum aestivum L.): A meta-analytic test of current hypotheses. Agric. Ecosyst. Environ. 2013, 178, 57–63. [Google Scholar] [CrossRef]
- Ainsworth, E.A.; Rogers, A. The response of photosynthesis and stomatal conductance to rising [CO2]: Mechanisms and environmental interactions. Plant Cell Environ. 2007, 30, 258–270. [Google Scholar] [CrossRef]
- Van Der Sleen, P.; Groenendijk, P.; Vlam, M.; Anten, N.P.R.; Boom, A.; Bongers, F.; Pons, T.L.; Terburg, G.; Zuidema, P.A. No growth stimulation of tropical trees by 150 years of CO2 fertilization but water-use efficiency increased. Nat. Geosci. 2015, 8, 24–28. [Google Scholar] [CrossRef]
- O’Leary, G.J.; Christy, B.; Nuttall, J.; Huth, N.; Cammarano, D.; Stöckle, C.; Basso, B.; Shcherbak, I.; Fitzgerald, G.; Luo, Q.; et al. Response of wheat growth, grain yield and water use to elevated CO2 under a free-air CO2 enrichment (face) experiment and modelling in a semi-arid environment. Glob. Chang. Biol. 2015, 21, 2670–2686. [Google Scholar] [CrossRef]
- Long, S.P.; Ainsworth, E.A.; Leakey, A.D.B.; Morgan, P.B. Global food insecurity. Treatment of major food crops with elevated carbon dioxide or ozone under large-scale fully open-air conditions suggests recent models may have overestimated future yields. Philos. Trans. R. Soc. B 2005, 360, 2011–2020. [Google Scholar] [CrossRef]
- Whitehead, D.; Hogan, K.P.; Rogers, G.N.D.; Byers, J.N.; Hunt, J.E.; McSeveny, T.M.; Hollinger, D.Y.; Dungan, R.J.; Earl, W.B.; Bourke, M.P. Performance of large open-top chambers for long-term field investigations of tree response to elevated carbon dioxide concentration. J. Biogeogr. 1995, 22, 307–313. [Google Scholar] [CrossRef]
- Piikki, K.; De Temmerman, L.; Högy, P.; Pleijel, H. The open-top chamber impact on vapour pressure deficit and its consequences for stomatal ozone uptake. Atmos. Environ. 2008, 42, 6513–6522. [Google Scholar] [CrossRef]
- Rawson, H.M. Yield responses of 2 wheat genotypes to carbon-dioxide and temperature in-field studies using temperature-gradient tunnels. Funct. Plant Biol. 1995, 22, 23–32. [Google Scholar] [CrossRef]
- Arp, W.J. Effects of source-sink relations on photosynthetic acclimation to elevated CO2. Plant Cell Environ. 1991, 14, 869–875. [Google Scholar] [CrossRef]
- Kimball, B.A. Crop responses to elevated CO2 and interactions with H2O, N, and temperature. Curr. Opin. Plant Biol. 2016, 31, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Manley, M.; Engelbrecht, M.L.; Williams, P.C.; Kidd, M. Assessment of variance in the measurement of hectolitre mass of wheat, using equipment from different grain producing and exporting countries. Biosyst. Eng. 2009, 103, 176–186. [Google Scholar] [CrossRef]
- Weiss, A.; Moreno-Sotomayer, A. Simulating grain mass and nitrogen concentration in wheat. Eur. J. Agron. 2006, 25, 129–137. [Google Scholar] [CrossRef]
- Calderini, D.F.; Reynolds, M.P.; Slafer, G.A. Wheat: Ecology and Physiology of Yield Determination; Sarratore, G., Slafer, G.A., Eds.; The Haworth Press Inc.: Philadelphia, PA, USA, 1999; p. 503. [Google Scholar]
- van der Kooi, C.J.; Reich, M.; Löw, M.; De Kok, L.; Tausz, M. Growth and yield stimulation under elevated CO2 and drought: A meta-analysis on crops. Envron. Exp. Bot. 2016, 122, 150–157. [Google Scholar] [CrossRef]
- Bustos, D.V.; Hasan, A.K.; Reynolds, M.P.; Calderini, D.F. Combining high grain number and weight through a dh-population to improve grain yield potential of wheat in high-yielding environments. Field Crops Res. 2013, 145, 106–115. [Google Scholar] [CrossRef]
- Amthor, J.S. Effects of atmospheric CO2 concentration on wheat yield: Review of results from experiments using various approaches to control CO2 concentration. Field Crops Res. 2001, 73, 1–34. [Google Scholar] [CrossRef]
- Bishop, K.A.; Leakey, A.D.B.; Ainsworth, E.A. How seasonal temperature or water inputs affect the relative response of C-3 crops to elevated [CO2]: A global analysis of open top chamber and free air CO2 enrichment studies. Food Energy Secur. 2014, 3, 33–45. [Google Scholar] [CrossRef]
- Federov, S. Getdata Graph Digitizer. 2.26.0.20. 2002. Available online: http://getdata-graph-digitizer.com/ (accessed on 20 May 2015).
- NOAA. Available online: http://www.noaa.gov/ (accessed on 19 September 2014).
- Rosenberg, M.S.; Adams, D.C.; Gurevitch, J. Metawin: Statistical Software for Meta-Analysis; Version 2.0; Sinauer Associates, Inc.: Sunderland, MA, USA, 2000. [Google Scholar]
- Feng, Z.Z.; Kobayashi, K.; Ainsworth, E.A. Impact of elevated ozone concentration on growth, physiology, and yield of wheat (Triticum aestivum L.): A meta-analysis. Glob. Chang. Biol. 2008, 14, 2696–2708. [Google Scholar]
- Ainsworth, E.A.; Davey, P.A.; Bernacchi, C.J.; Dermody, O.C.; Heaton, E.A.; Moore, D.J.; Morgan, P.B.; Naidu, S.L.; Ra, H.S.Y.; Zhu, X.G.; et al. A meta-analysis of elevated [CO2] effects on soybean (glycine max) physiology, growth and yield. Glob. Chang. Biol. 2002, 8, 695–709. [Google Scholar] [CrossRef]
- Adams, D.C.; Gurevitch, J.; Rosenberg, M.S. Resampling tests for meta-analysis of ecological data. Ecology 1997, 78, 1277–1283. [Google Scholar] [CrossRef]
- Curtis, P.S.; Wang, X.Z. A meta-analysis of elevated CO2 effects on woody plant mass, form, and physiology. Oecologia 1998, 113, 299–313. [Google Scholar] [CrossRef]
- Motulsky, H.J.; Brown, R.E. Detecting outliers when fitting data with nonlinear regression—A new method based on robust nonlinear regression and the false discovery rate. BMC Bioinform. 2006, 7, 1–20. [Google Scholar] [CrossRef]
- Hogy, P.; Fangmeier, A. Atmospheric co2 enrichment affects potatoes: 1. Aboveground biomass production and tuber yield. Eur. J. Agron. 2009, 30, 78–84. [Google Scholar] [CrossRef]
- Ziska, L.H.; Bunce, J.A. Predicting the impact of changing CO2 on crop yields: Some thoughts on food. New Phytol. 2007, 175, 607–617. [Google Scholar] [CrossRef] [PubMed]
- Högy, P.; Fangmeier, A. Effects of elevated atmospheric CO2 on grain quality of wheat. J. Cereal Sci. 2008, 48, 580–591. [Google Scholar] [CrossRef]
- Long, S.P.; Ainsworth, E.A.; Leakey, A.D.B.; Nosberger, J.; Ort, D.R. Food for thought: Lower-than-expected crop yield stimulation with rising CO2 concentrations. Science 2006, 312, 1918–1921. [Google Scholar] [CrossRef]
- Houshmandfar, A.; Fitzgerald, G.J.; O’Leary, G.; Tausz-Posch, S.; Fletcher, A.; Tausz, M. The relationship between transpiration and nutrient uptake in wheat changes under elevated atmospheric CO2. Physiol. Plant. 2018, 163, 516–529. [Google Scholar] [CrossRef]
- Long, S.P.; Bernacchi, C.J. Gas exchange measurements, what can they tell us about the underlying limitations to photosynthesis? Procedures and sources of error. J. Exp. Bot. 2003, 54, 2393–2401. [Google Scholar] [CrossRef] [PubMed]
- Bloom, A.J. Photorespiration and nitrate assimilation: A major intersection between plant carbon and nitrogen. Photosynth. Res. 2015, 123, 117–128. [Google Scholar] [CrossRef]
- Wujeska-Klause, A.; Crous, K.Y.; Ghannoum, O.; Ellsworth, D.S. Lower photorespiration in elevated CO2 reduces leaf n concentrations in mature eucalyptus trees in the field. Glob. Chang. Biol. 2019, 25, 1282–1295. [Google Scholar] [CrossRef]
- Fitzgerald, G.J.; Tausz, M.; O’Leary, G.; Mollah, M.R.; Tausz-Posch, S.; Seneweera, S.; Mock, I.; Low, M.; Partington, D.L.; McNeil, D.; et al. Elevated atmospheric [CO2] can dramatically increase wheat yields in semi-arid environments and buffer against heat waves. Glob. Chang. Biol. 2016, 22, 2269–2284. [Google Scholar] [CrossRef]
- Taub, D.R.; Miller, B.; Allen, H. Effects of elevated CO2 on the protein concentration of food crops: A meta-analysis. Glob. Chang. Biol. 2008, 14, 565–575. [Google Scholar] [CrossRef]
- Broberg, M.C.; Hogy, P.; Pleijel, H. CO2-induced changes in wheat grain composition: Meta-analysis and response functions. Agronomy 2017, 7, 32. [Google Scholar] [CrossRef]
- Burnett, A.C.; Rogers, A.; Rees, M.; Osborne, C.P. Carbon source-sink limitations differ between two species with contrasting growth strategies. Plant Cell Environ. 2016, 39, 2460–2472. [Google Scholar] [CrossRef] [PubMed]
- Vanuytrecht, E.; Raes, D.; Willems, P. Considering sink strength to model crop production under elevated atmospheric CO2. Agric. For. Meteorol. 2011, 151, 1753–1762. [Google Scholar] [CrossRef]
- Austin, R.B.; Bingham, J.; Blackwell, R.D.; Evans, L.T.; Ford, M.A.; Morgan, C.L.; Taylor, M. Genetic improvements in winter-wheat yields since 1900 and associated physiological-changes. J. Agric. Sci. 1980, 94, 675–689. [Google Scholar] [CrossRef]
- Evans, L.T. Crop Evolution, Adaptation, and Yield; Cambridge University Press: Cambridge, UK, 1993. [Google Scholar]
- Högy, P.; Wieser, H.; Kohler, P.; Schwadorf, K.; Breuer, J.; Franzaring, J.; Muntifering, R.; Fangmeier, A. Effects of elevated CO2 on grain yield and quality of wheat: Results from a 3-year free-air CO2 enrichment experiment. Plant Biol. 2009, 11, 60–69. [Google Scholar] [CrossRef]
- Högy, P.; Brunnbauer, M.; Koehler, P.; Schwadorf, K.; Breuer, J.; Franzaring, J.; Zhunusbayeva, D.; Fangmeier, A. Grain quality characteristics of spring wheat (Triticum aestivum) as affected by free-air CO2 enrichment. Environ. Exp. Bot. 2013, 88, 11–18. [Google Scholar] [CrossRef]
- Kimball, B.A.; Pinter, P.J.; Garcia, R.L.; LaMorte, R.L.; Wall, G.W.; Hunsaker, D.J.; Wechsung, G.; Wechsung, F.; Kartschall, T. Productivity and water use of wheat under free-air CO2 enrichment. Glob. Chang. Biol. 1995, 1, 429–442. [Google Scholar] [CrossRef]
- Miralles, D.J.; Slafer, G.A. Sink limitations to yield in wheat: How could it be reduced? J. Agric. Sci. 2007, 145, 139–149. [Google Scholar] [CrossRef]
- Kimball, B.A.; Morris, C.F.; Pinter, P.J.; Wall, G.W.; Hunsaker, D.J.; Adamsen, F.J.; LaMorte, R.L.; Leavitt, S.W.; Thompson, T.L.; Matthias, A.D.; et al. Elevated CO2, drought and soil nitrogen effects on wheat grain quality. New Phytol. 2001, 150, 295–303. [Google Scholar] [CrossRef]
- Deepak, S.S.; Agrawal, M. Growth and yield responses of wheat plants to elevated levels of CO2 and SO2, singly and in combination. Environ. Pollut. 1999, 104, 411–419. [Google Scholar] [CrossRef]
- Pleijel, H.; Uddling, J. Yield vs. Quality trade-offs for wheat in response to carbon dioxide and ozone. Glob. Chang. Biol. 2012, 18, 596–605. [Google Scholar] [CrossRef]
- Bunce, J. Using face systems to screen wheat cultivars for yield increases at elevated CO2. Agronomy 2017, 7, 20. [Google Scholar] [CrossRef]
- Asseng, S.; Martre, P.; Maiorano, A.; Rötter, R.P.; O’Leary, G.J.; Fitzgerald, G.J.; Girousse, C.; Motzo, R.; Giunta, F.; Babar, M.A.; et al. Climate change impact and adaptation for wheat protein. Glob. Chang. Biol. 2019, 25, 155–173. [Google Scholar] [CrossRef]
- Martre, P.; Wallach, D.; Asseng, S.; Ewert, F.; Jones, J.W.; Rotter, R.P.; Boote, K.J.; Ruane, A.C.; Thorburn, P.J.; Cammarano, D.; et al. Multimodel ensembles of wheat growth: Many models are better than one. Glob. Chang. Biol. 2015, 21, 911–925. [Google Scholar] [CrossRef] [PubMed]
- Lobell, D.B.; Gourdji, S.M. The influence of climate change on global crop productivity. Plant Physiol. 2012, 160, 1686–1697. [Google Scholar] [CrossRef]
- Pleijel, H.; Broberg, M.C.; Hogy, P.; Uddling, J. Nitrogen application is required to realize wheat yield stimulation by elevated CO2 but will not remove the CO2-induced reduction in grain protein concentration. Glob. Chang. Biol. 2019, 25, 1868–1876. [Google Scholar] [CrossRef]
- Myers, S.S.; Zanobetti, A.; Kloog, I.; Huybers, P.; Leakey, A.D.; Bloom, A.J.; Carlisle, E.; Dietterich, L.H.; Fitzgerald, G.; Hasegawa, T.; et al. Increasing CO2 threatens human nutrition. Nature 2014, 510, 139–142. [Google Scholar] [CrossRef]
- Loladze, I. Hidden shift of the ionome of plants exposed to elevated CO2 depletes minerals at the base of human nutrition. Elife 2014, 3, e02245. [Google Scholar] [CrossRef]
- Myers, S.S.; Wessells, K.R.; Kloog, I.; Zanobetti, A.; Schwartz, J. Effect of increased concentrations of atmospheric carbon dioxide on the global threat of zinc deficiency: A modelling study. Lancet Glob. Health 2015, 3, e639–e645. [Google Scholar] [CrossRef]
- Medek, D.E.; Schwartz, J.; Myers, S.S. Estimated effects of future atmospheric CO2 concentrations on protein intake and the risk of protein deficiency by country and region. Environ. Health Perspect. 2017, 125, 087002. [Google Scholar] [CrossRef]
- Dietterich, L.H.; Zanobetti, A.; Huybers, P.; Leakey, A.D.B.; Bloom, A.J.; Carlisle, E.; Fernando, N.; Fitzgerald, G.; Hasegawa, T.; Holbrook, N.M.; et al. Impacts of elevated atmospheric CO2 on nutrient content of important food crops. Sci. Data 2015, 2, 150036. [Google Scholar] [CrossRef]
- Dijkstra, P.; Schapendonk, A.H.M.C.; Groenwold, K.; Jansen, M.; Van de Geijn, S.C. Seasonal changes in the response of winter wheat to elevated atmospheric CO2 concentration grown in open-top chambers and field tracking enclosures. Glob. Chang. Biol. 1999, 5, 563–576. [Google Scholar] [CrossRef]
- Donnelly, A.; Jones, M.B.; Burke, J.I.; Schnieders, B. Does elevated CO2 protect grain yield of wheat from the effects of ozone stress? Z. Nat. C 1999, 54, 802–811. [Google Scholar] [CrossRef]
- Fangmeier, A.; Gruters, U.; Hertstein, U.; SandhageHofmann, A.; Vermehren, B.; Jager, H.J. Effects of elevated CO2, nitrogen supply and tropospheric ozone on spring wheat - 1. Growth and yield. Environ. Pollut. 1996, 91, 381–390. [Google Scholar] [CrossRef]
- Hakala, K. Growth and yield potential of spring wheat in a simulated changed climate with increased CO2 and higher temperature. Eur. J. Agron. 1998, 9, 41–52. [Google Scholar] [CrossRef]
- Houshmandfar, A.; Fitzgerald, G.J.; Macabuhay, A.A.; Armstrong, R.; Tausz-Posch, S.; Low, M.; Tausz, M. Trade-offs between water-use related traits, yield components and mineral nutrition of wheat under free-air CO2 enrichment (face). Eur. J. Agron. 2016, 76, 66–74. [Google Scholar] [CrossRef]
- Högy, P. Wirkungen Erhöhter CO2 -und/oder Ozonkonzentrationen auf den Ertrag und die Qualität Landwirtschaftlicher Nutzpflanzen. Ph.D. Thesis, Justus-Liebig-University, Gießen, Germany, 2002. [Google Scholar]
- Manderscheid, R.; Weigel, H.J. Photosynthetic and growth responses of old and modern spring wheat cultivars to atmospheric CO2 enrichment. Agric. Ecosyst. Environ. 1997, 64, 65–73. [Google Scholar] [CrossRef]
- Mishra, A.K.; Rai, R.; Agrawal, S.B. Differential response of dwarf and tall tropical wheat cultivars to elevated ozone with and without carbon dioxide enrichment: Growth, yield and grain quality. Field Crops Res. 2013, 145, 21–32. [Google Scholar] [CrossRef]
- Mortensen, L.; Egelov, A.H.; Grundahl, L.; Kemp, K.; Manscher, O.H. Final Report of the Espace-Wheat Open-Top Chamber Experiment 1994–1996 at Roskilde, Denmark; National Environmental Research Institute: Roskilde, Denmark, 1998.
- Fangmeier, A.; De Temmerman, L.; Mortensen, L.; Kemp, K.; Burke, J.; Mitchell, R.; van Oijen, M.; Weigel, H.J. Effects on nutrients and on grain quality in spring wheat crops grown under elevated CO2 concentrations and stress conditions in the european, multiple-site experiment ‘espace-wheat’. Eur. J. Agron. 1999, 10, 215–229. [Google Scholar] [CrossRef]
- Mulholland, B.J.; Craigon, J.; Black, C.R.; Colls, J.J.; Atherton, J.; Landon, G. Growth, light interception and yield responses of spring wheat (Triticum aestivum L.) grown under elevated CO2 and O3 in open-top chambers. Glob. Chang. Biol. 1998, 4, 121–130. [Google Scholar] [CrossRef]
- Piikki, K.; De Temmerman, L.; Ojanpera, K.; Danielsson, H.; Pleijel, H. The grain quality of spring wheat (Triticum aestivum L.) in relation to elevated ozone uptake and carbon dioxide exposure. Eur. J. Agron. 2008, 28, 245–254. [Google Scholar] [CrossRef]
- Pleijel, H.; Gelang, J.; Sild, E.; Danielsson, H.; Younis, S.; Karlsson, P.E.; Wallin, G.; Skarby, L.; Sellden, G. Effects of elevated carbon dioxide, ozone and water availability on spring wheat growth and yield. Physiol. Plant. 2000, 108, 61–70. [Google Scholar] [CrossRef]
- Blumenthal, C.; Rawson, H.M.; McKenzie, E.; Gras, P.W.; Barlow, E.W.R.; Wrigley, C.W. Changes in wheat grain quality due to doubling the level of atmospheric CO2. Cereal Chem. 1996, 73, 762–766. [Google Scholar]
- Mulchi, C.; Rudorff, B.; Lee, E.; Rowland, R.; Pausch, R. Morphological responses among crop species to full-season exposures to enhanced concentrations of atmospheric CO2 and O3. Water Air Soil Pollut. 1995, 85, 1379–1386. [Google Scholar] [CrossRef]
- Rudorff, B.F.T.; Mulchi, C.L.; Daughtry, C.S.T.; Lee, E.H. Growth, radiation use efficiency, and canopy reflectance of wheat and corn grown under elevated ozone and carbon dioxide atmospheres. Remote Sens. Environ. 1996, 55, 163–173. [Google Scholar] [CrossRef]
- Rudorff, B.F.T.; Mulchi, C.L.; Lee, E.H.; Rowland, R.; Pausch, R. Effects of enhanced O3 and CO2 enrichment on plant characteristics in wheat and corn. Environ. Pollut. 1996, 94, 53–60. [Google Scholar] [CrossRef]
- Van Oijen, M.; Schapendonk, A.H.C.M.; Jansen, M.J.H.; Pot, C.S.; Maciorowski, R. Do open-top chambers overestimate the effects of rising CO2 on plants? An analysis using spring wheat. Glob. Chang. Biol. 1999, 5, 411–421. [Google Scholar] [CrossRef]
- Weigel, H.J.; Manderscheid, R. Crop growth responses to free air CO2 enrichment and nitrogen fertilization: Rotating barley, ryegrass, sugar beet and wheat. Eur. J. Agron. 2012, 43, 97–107. [Google Scholar] [CrossRef]
- Manderscheid, R.; Bender, J.; Jager, H.J.; Weigel, H.J. Effects of season long co2 enrichment on cereals. 2. Nutrient concentrations and grain quality. Agric. Ecosyst. Env. 1995, 54, 175–185. [Google Scholar] [CrossRef]
- Weigel, H.J.; Manderscheid, R.; Jager, H.J.; Mejer, G.J. Effects of season-long CO2 enrichment on cereals. 1. Growth-performance and yield. Agric. Ecosyst. Environ. 1994, 48, 231–240. [Google Scholar] [CrossRef]
- Yang, L.X.; Wang, Y.L.; Li, S.F.; Huang, J.Y.; Dong, G.C.; Zhu, J.G.; Liu, G.; Han, Y. Effects of free-air CO2 enrichment (face) on dry matter production and allocation in wheat. J. Appl. Ecol. 2007, 18, 339–346. [Google Scholar]
- Yang, L.X.; Huang, J.Y.; Li, S.F.; Yang, H.J.; Zhu, J.G.; Dong, G.C.; Liu, H.J.; Wang, Y.L. Effects of free-air CO2 enrichment on nitrogen uptake and utilization of wheat. J. Appl. Ecol. 2007, 18, 519–525. [Google Scholar]
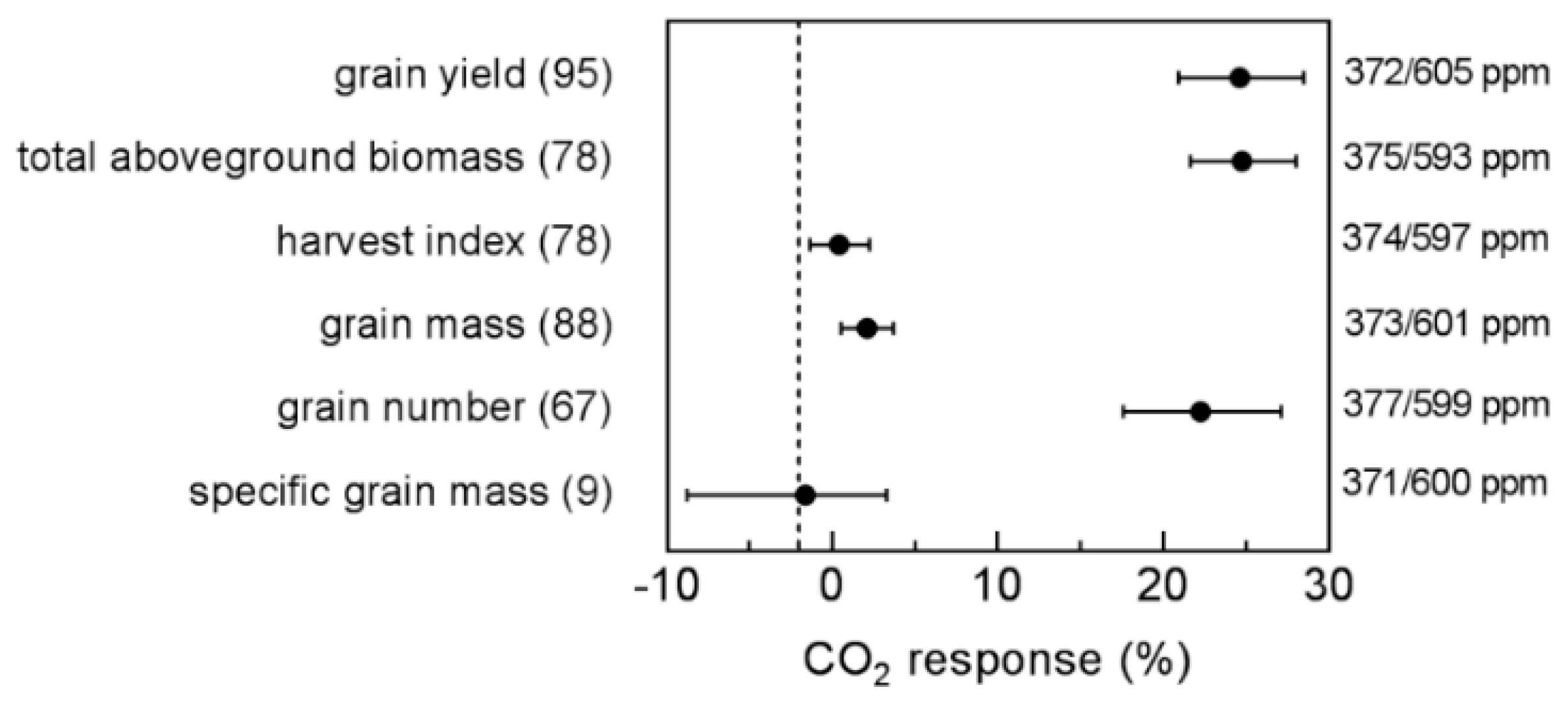
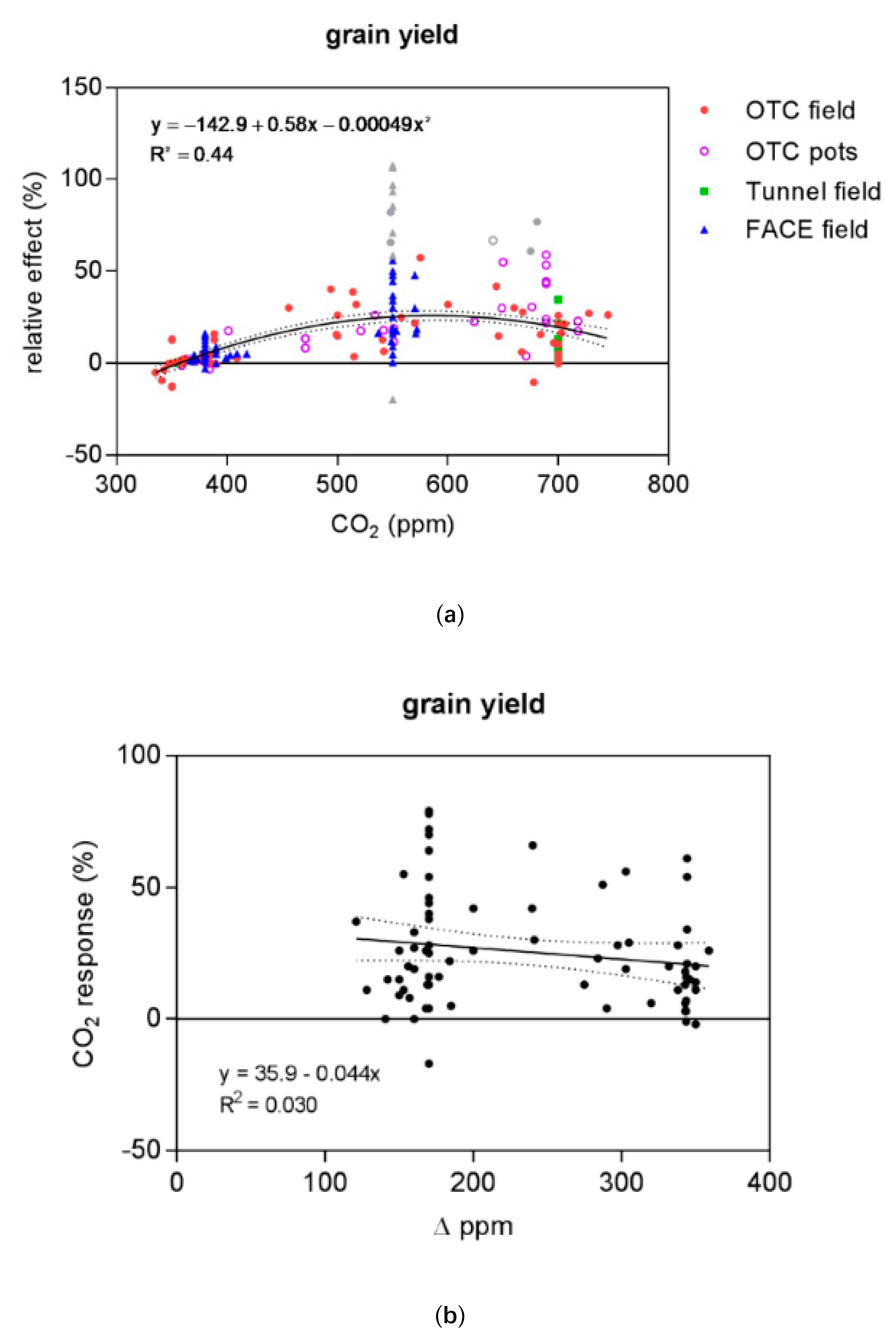
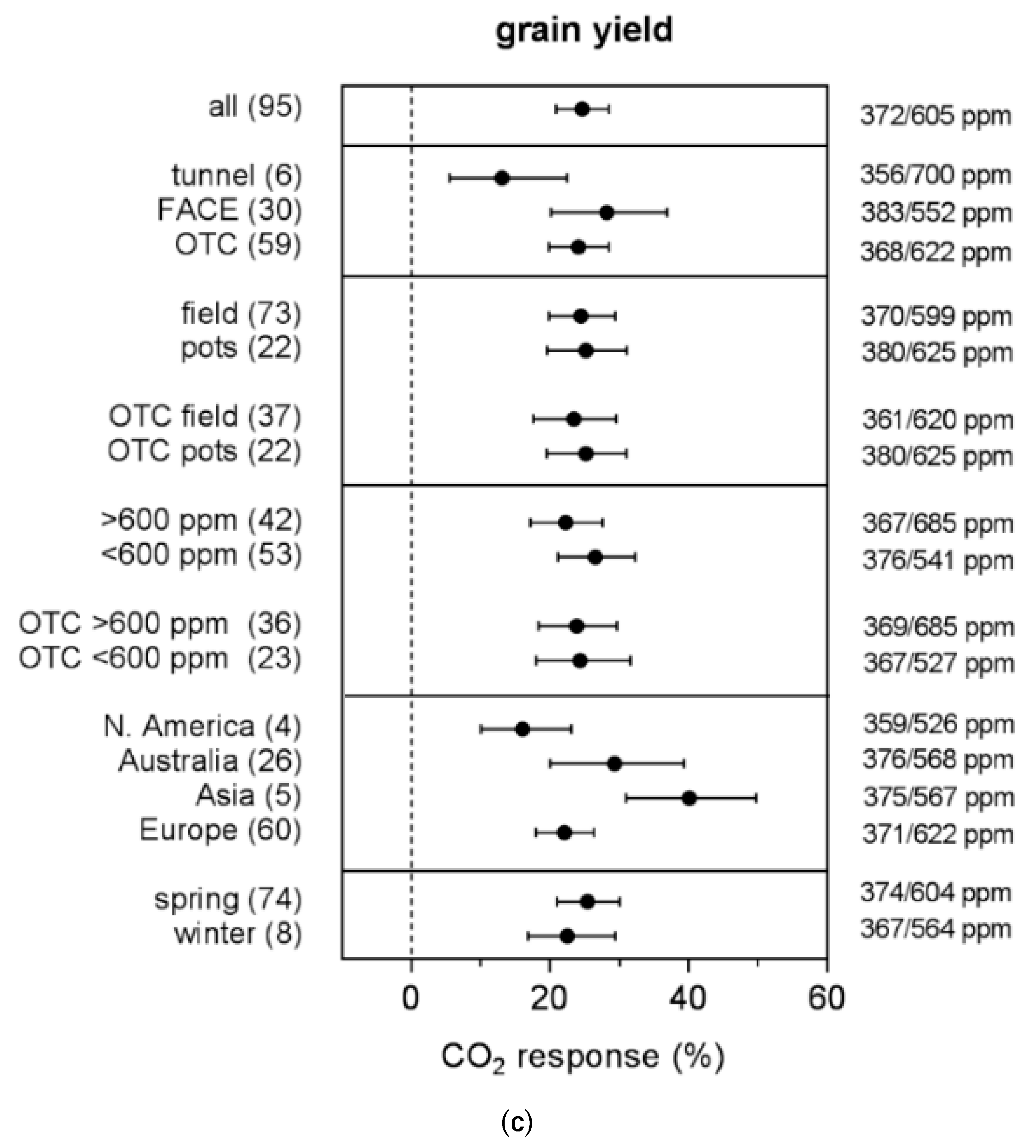
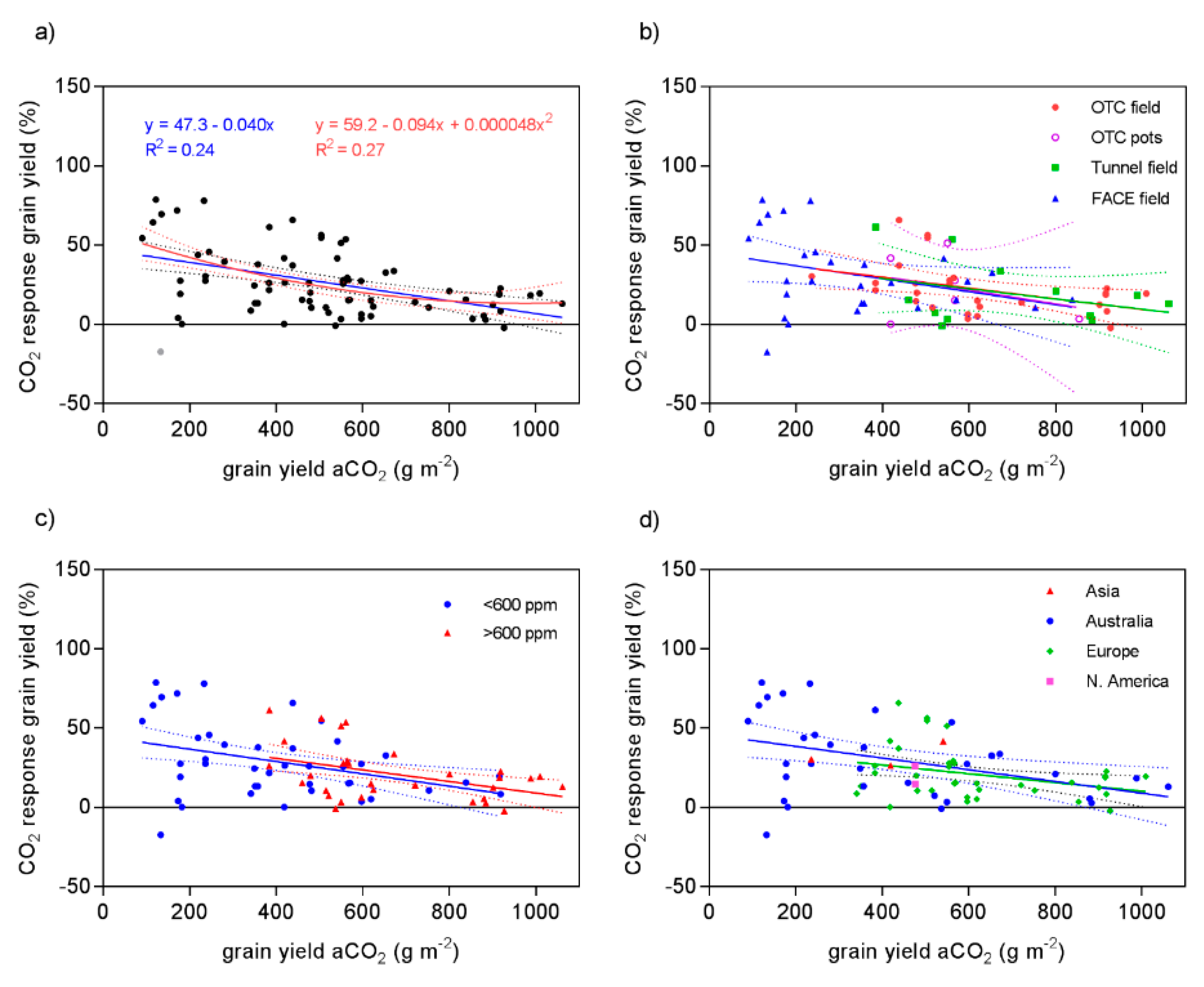
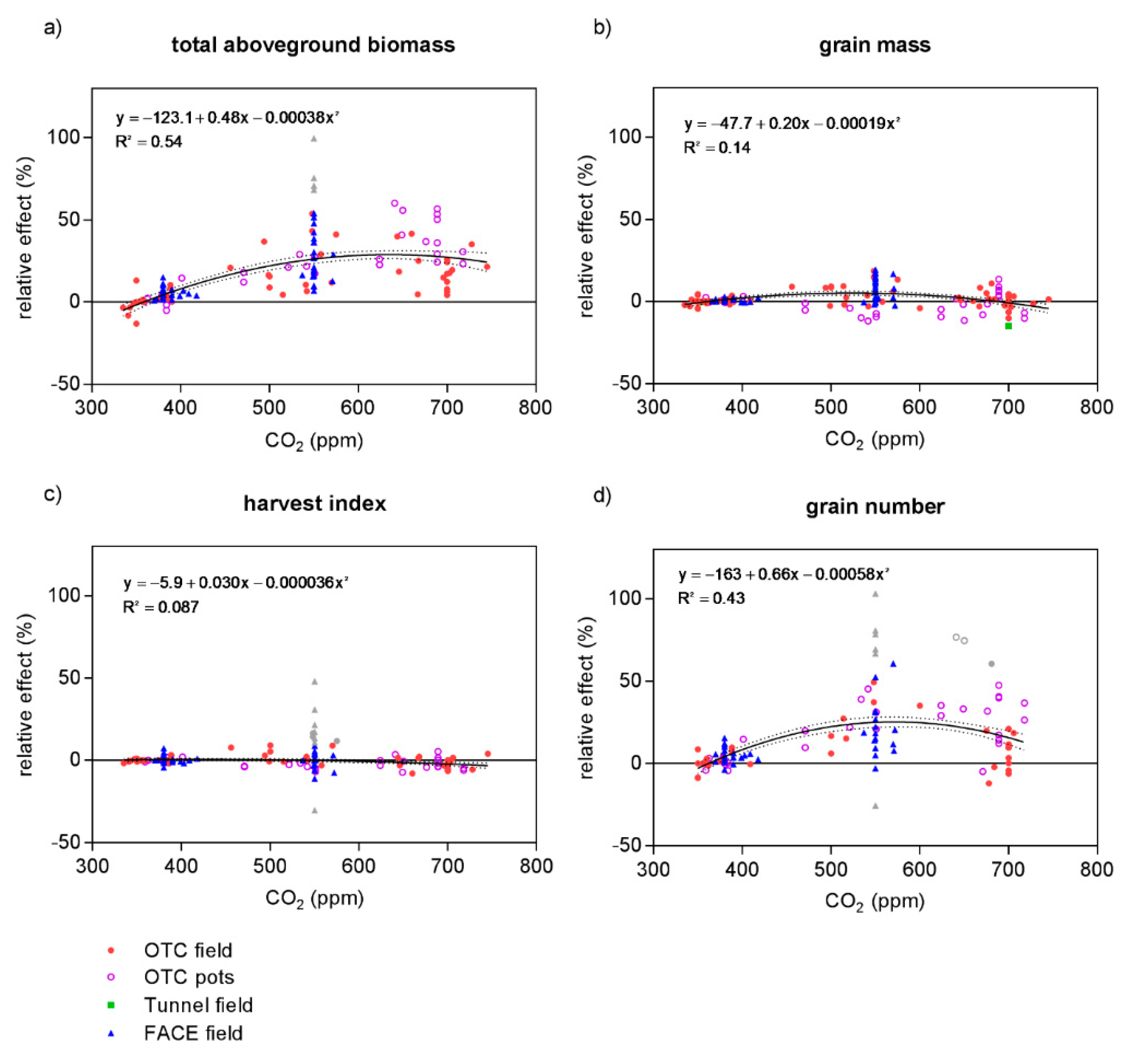
| Variable | Observations (Outliers) | Model | B0 | B1 | B2 | R2 | p-Value | Probability % |
|---|---|---|---|---|---|---|---|---|
| grain yield | 173 (13) | linear | −17.5 | 0.062 | 0.29 | <0.0001 | <0.01 | |
| quadratic | −142.9 | 0.576 | −0.00049 | 0.44 | >99.99 | |||
| total aboveground biomass | 139 (4) | linear | −27.1 | 0.086 | 0.46 | <0.0001 | <0.01 | |
| quadratic | −123.1 | 0.479 | −0.00038 | 0.54 | >99.99 | |||
| harvest index | 139 (10) | linear | 3.2 | −0.007 | 0.07 | 0.0022 | 49.6 | |
| quadratic | −5.9 | 0.030 | −0.00004 | 0.09 | 50.4 | |||
| grain mass | 159 (0) | linear | 1.4 | 0.001 | 0.00 | 0.78 | <0.01 | |
| quadratic | −47.7 | 0.202 | −0.00019 | 0.14 | >99.99 | |||
| grain number | 125 (9) | linear | −17.8 | 0.062 | 0.29 | <0.0001 | <0.01 | |
| quadratic | −151.9 | 0.061 | −0.00053 | 0.47 | >99.99 |
| Group | Observations | Model | B0 | B1 | B2 | R2 | p-Value | Probability % |
|---|---|---|---|---|---|---|---|---|
| all | 73 | linear | 47.3 | −0.040 | 0.24 | 0.00020 | 46.7 | |
| quadratic | 59.2 | −0.094 | 0.000048 | 0.27 | 53.4 | |||
| OTC field | 27 | linear | 43.5 | −0.034 | 0.19 | 0.022 | 74.9 | |
| quadratic | 64.1 | −0.10 | 0.000051 | 0.21 | 25.1 | |||
| OTC pots | 6 | linear | 47.7 | −0.043 | 0.11 | 0.52 | >99.99 | |
| quadratic | −91.3 | 0.42 | −0.00037 | 0.28 | <0.01 | |||
| Tunnel field | 12 | linear | 42.0 | −0.032 | 0.13 | 0.25 | 86.5 | |
| quadratic | 109.8 | −0.24 | 0.00014 | 0.20 | 13.5 | |||
| FACE field | 28 | linear | 45.3 | −0.041 | 0.11 | 0.079 | 73.9 | |
| quadratic | 56.4 | −0.11 | 0.000085 | 0.13 | 26.1 | |||
| <600 ppm | 42 | linear | 44.6 | −0.039 | 0.14 | 0.016 | 72.4 | |
| quadratic | 51.7 | −0.079 | 0.000043 | 0.15 | 27.6 | |||
| >600 ppm | 31 | linear | 45.5 | −0.036 | 0.19 | 0.015 | 56.5 | |
| quadratic | 102.7 | −0.21 | 0.00012 | 0.24 | 43.5 | |||
| Australia | 32 | linear | 46.0 | −0.037 | 0.14 | 0.022 | 76.4 | |
| quadratic | 51.5 | −0.068 | 0.000030 | 0.17 | 23.6 | |||
| Europe | 36 | linear | 37.9 | −0.028 | 0.11 | 0.050 | 78.1 | |
| quadratic | 37.2 | −0.026 | −0.0000015 | 0.11 | 21.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Broberg, M.C.; Högy, P.; Feng, Z.; Pleijel, H. Effects of Elevated CO2 on Wheat Yield: Non-Linear Response and Relation to Site Productivity. Agronomy 2019, 9, 243. https://doi.org/10.3390/agronomy9050243
Broberg MC, Högy P, Feng Z, Pleijel H. Effects of Elevated CO2 on Wheat Yield: Non-Linear Response and Relation to Site Productivity. Agronomy. 2019; 9(5):243. https://doi.org/10.3390/agronomy9050243
Chicago/Turabian StyleBroberg, Malin C., Petra Högy, Zhaozhong Feng, and Håkan Pleijel. 2019. "Effects of Elevated CO2 on Wheat Yield: Non-Linear Response and Relation to Site Productivity" Agronomy 9, no. 5: 243. https://doi.org/10.3390/agronomy9050243
APA StyleBroberg, M. C., Högy, P., Feng, Z., & Pleijel, H. (2019). Effects of Elevated CO2 on Wheat Yield: Non-Linear Response and Relation to Site Productivity. Agronomy, 9(5), 243. https://doi.org/10.3390/agronomy9050243






