Effect of Selenium Enrichment and Type of Application on Yield, Functional Quality and Mineral Composition of Curly Endive Grown in a Hydroponic System
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Growing Conditions
2.2. Yield and Morphological Traits
2.3. Functional Quality Analysis
2.4. Mineral Composition
2.5. Statistical Analysis
3. Results
3.1. Plant Se Content
3.2. Crop Performance
3.3. Antioxidant Properties and Nutritional Value
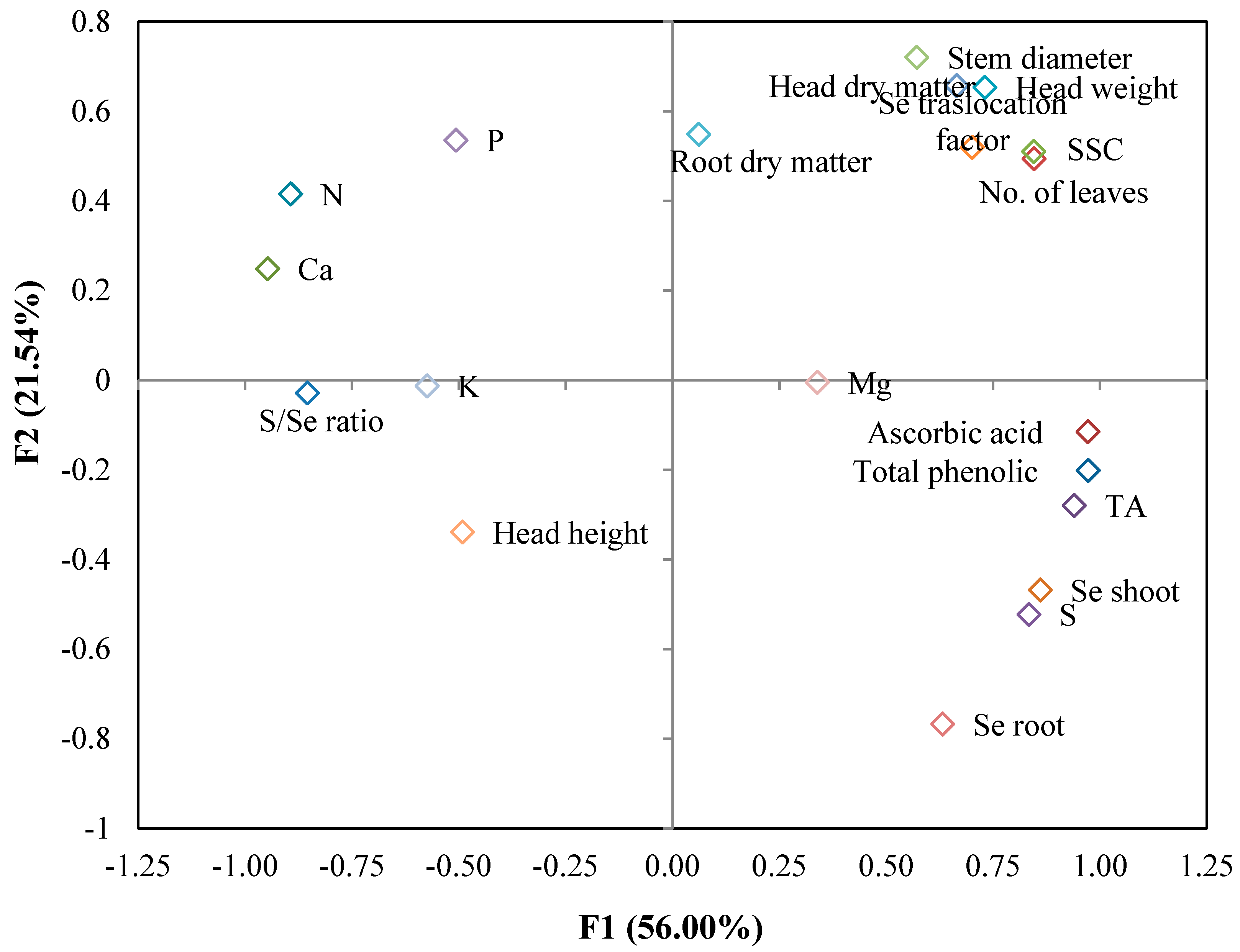
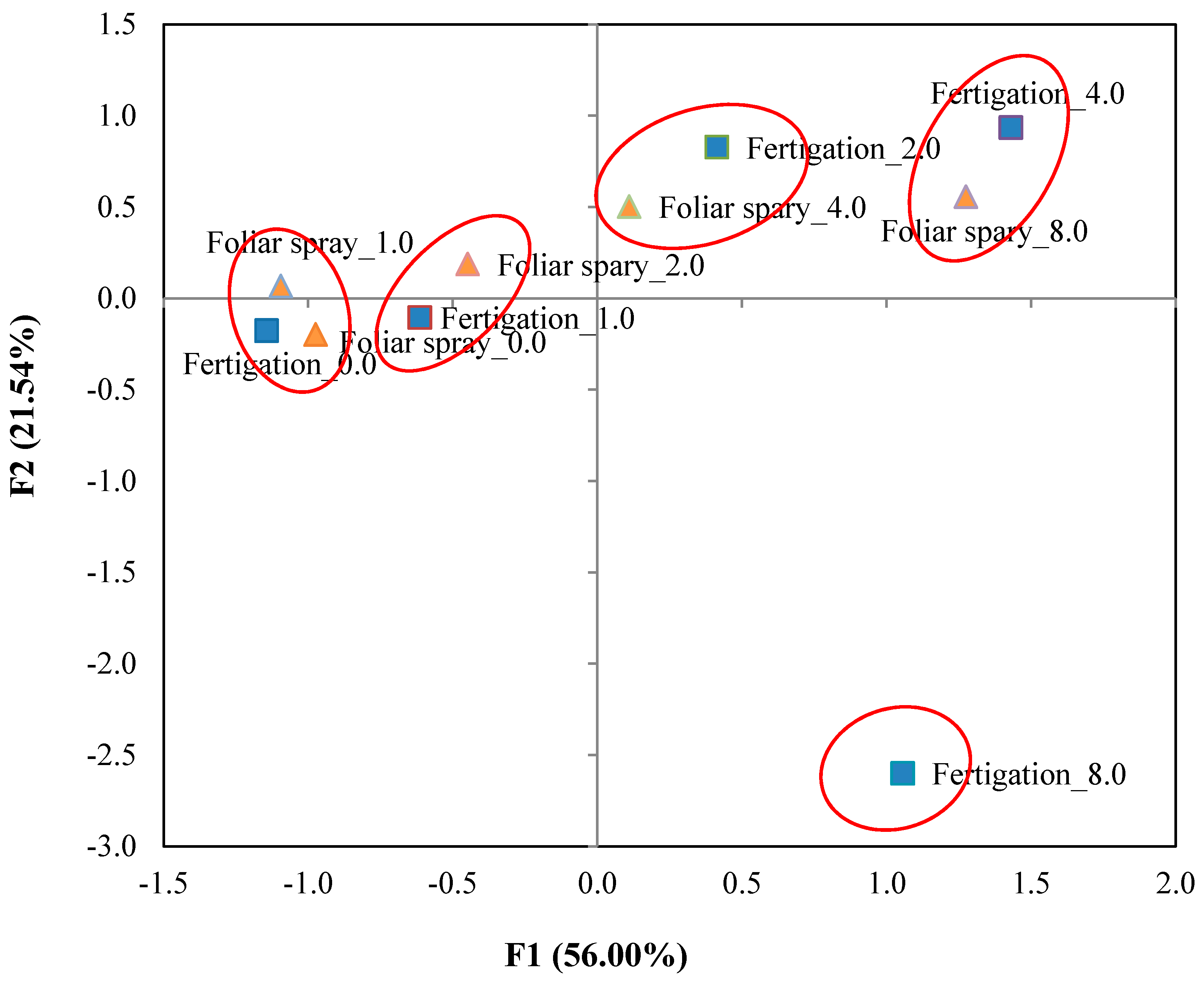
3.4. Principal Component Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hawrylak-Nowak, B. Comparative effects of selenite and selenate on growth and selenium accumulation in lettuce plants under hydroponic conditions. Plant Growth Regul. 2013, 70, 149–157. Available online: https://link.springer.com/article/10.1007/s10725-013-9788-5 (accessed on 14 February 2019). [CrossRef]
- White, P.J.; Broadley, M.R. Biofortification of crops with seven mineral elements often lacking human diets-iron, zinc, copper, calcium, magnesium, selenium and iodine. New Phytol. 2009, 182, 49–84. Available online: https://www.ncbi.nlm.nih.gov/pubmed/19192191 (accessed on 14 February 2019). [CrossRef]
- Moncada, A.; Miceli, A.; Sabatino, L.; Iapichino, G.; D’Anna, F.; Vetrano, F. Effect of molybdenum rate on yield and quality of lettuce, escarole, and curly endive grown in a floating system. Agronomy 2018, 8, 171. Available online: https://www.mdpi.com/2073-4395/8/9/171/htm (accessed on 14 February 2019). [CrossRef]
- Sabatino, L.; D’Anna, F.; D’Anna, F.; Iapichino, G.; Moncada, A.; D’Anna, E.; De Pasquale, C. Interactive effects of genotype and molybdenum supply on yield and overall fruit quality of tomato. Front. Plant Sci. 2019, 9, 1922. Available online: https://www.frontiersin.org/articles/10.3389/fpls.2018.01922/full (accessed on 14 February 2019). [CrossRef] [PubMed]
- U.S. Department of Agriculture. Dietary Reference Intakes: Elements. 2003. Available online: http://warp.nal.usda.gov/fnic/etextA/000105.html (accessed on 14 February 2019).
- Kieliszek, M.; Blažejak, S. Selenium: Significance, and outlook for supplementation. Nutrition 2013, 29, 713–718. Available online: https://www.ncbi.nlm.nih.gov/pubmed/23422539 (accessed on 14 February 2019). [CrossRef]
- Gissel-Nielsen, G.; Gupta, U.C.; Lamand, M.; Westermarck, T. Selenium in soils and plants and its importance in livestock and human nutrition. Adv. Agron. 1984, 37, 397–460. Available online: https://www.sciencedirect.com/science/article/pii/S0065211308604599 (accessed on 14 February 2019).
- Marschner, P. Marschner’s Mineral Nutrition of Higher Plants, 3rd ed.; Elsevier/Academic Press: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. Available online: https://www.ncbi.nlm.nih.gov/pubmed/22381456 (accessed on 14 February 2019). [CrossRef]
- Drutel, A.; Archambeaud, F.; Caron, P. Selenium and the thyroid gland: More good news for clinicians. Clin. Endocrinol. (Oxford) 2013, 78, 155–164. Available online: https://www.ncbi.nlm.nih.gov/pubmed/23046013 (accessed on 14 February 2019). [CrossRef] [PubMed]
- Mistry, H.D.; Broughton Pipkin, F.; Redman, C.W.; Poston, L. Selenium in reproductive health. Am. J. Obstet. Gynecol. 2012, 206, 21–30. Available online: https://www.ncbi.nlm.nih.gov/pubmed/21963101 (accessed on 14 February 2019). [CrossRef]
- Dennert, G.; Zwahlen, M.; Brinkman, M.; Vinceti, M.; Zeegers, M.P.; Horneber, M. Selenium for preventing cancer. Cochrane Database Syst. Rev. 2011, 5, CD005195. [Google Scholar] [CrossRef]
- Chen, L.; Yang, F.; Xu, J.; Yun, H.; Hu, Q.; Zhang, Y.; Pan, G. Determination of selenium concentration of rice in China and effect of fertilization of selenite and selenate on Se content of rice. J. Agric. Food Chem. 2002, 50, 5128–5130. Available online: https://www.ncbi.nlm.nih.gov/pubmed/12188618 (accessed on 14 February 2019). [CrossRef]
- Hawkesford, M.J.; Zhao, F.J. Strategies for increasing the selenium content of wheat. J. Cereal Sci. 2007, 46, 282–292. Available online: https://www.sciencedirect.com/science/article/pii/S0733521007000379 (accessed on 14 February 2019). [CrossRef]
- Lyons, G.H.; Stangoulis, J.C.R.; Graham, R.D. Exploiting micronutrient interaction to optimize biofortification programs: The case for inclusion of selenium and iodine in the Harvest Plust programs. Nutr. Rev. 2004, 62, 247–252. Available online: https://www.ncbi.nlm.nih.gov/pubmed/15291398 (accessed on 14 February 2019). [CrossRef]
- Schiavon, M.; dall’Acqua, S.; Mietto, A.; Pilon-Smits, E.A.H.; Sambo, P.; Masi, A.; Malagoli, M. Selenium Fertilization Alters the Chemical Composition and Antioxidant Constituents of Tomato (Solanum lycopersicon L.). J. Agric. Food Chem. 2013, 61, 10542–10554. Available online: https://www.ncbi.nlm.nih.gov/pubmed/24079300 (accessed on 14 February 2019). [CrossRef]
- Ramos, S.J.; Faquin, V.; Guilherme, L.R.G.; Castro, E.M.; Ávila, F.W.; Carvalho, G.S.; Bastos, C.E.A.; Oliveira, C. Selenium biofortification and antioxidant activity in lettuce plants fed with selenate and selenite. Plant Soil Environ. 2010, 56, 584–588. Available online: https://www.agriculturejournals.cz/publicFiles/31992.pdf (accessed on 14 February 2019). [CrossRef]
- Koudela, M.; Petříková, K. Nutritional composition and yield of endive cultivars—Cichorium endivia L. Hort. Sci. 2007, 34, 6–10. Available online: https://www.agriculturejournals.cz/publicFiles/00115.pdf (accessed on 14 February 2019). [CrossRef]
- Sonneveld, C.; Voogt, W. Plant Nutrition of Greenhouse Crops; Springer: New York, NY, USA, 2009. [Google Scholar]
- Goto, E.; Both, A.J.; Albright, L.D.; Langhans, R.W.; Leed, A.R. Effect of dissolved oxygen concentration on lettuce rowth in floating hydroponics. Acta Hortic. 1996, 440, 205–210. Available online: https://www.ncbi.nlm.nih.gov/pubmed/11541573 (accessed on 14 February 2019). [CrossRef] [PubMed]
- Han, C.; Zhao, Y.; Leonard, S.W.; Traber, M. Edible coatings to improve storability and enhance nutritional value of fresh and frozen strawberries (Fragaria × ananassa) and raspberries (Rubus ideaus). Postharvest Biol. Technol. 2008, 33, 67–78. Available online: https://www.sciencedirect.com/science/article/abs/pii/S0925521404000274 (accessed on 14 February 2019). [CrossRef]
- Rivero, R.M.; Ruiz, J.M.; Garcia, P.C.; López-Lefebre, L.R.; Sánchez, E.; Romero, L. Resistance to cold and heat stress: Accumulation of phenolic compounds in tomato and watermelon plants. Plant Sci. 2001, 160, 315–321. Available online: https://www.sciencedirect.com/science/article/pii/S0168945200003952 (accessed on 14 February 2019). [CrossRef]
- Morand, P.; Gullo, J.L. Mineralisation des tissus vegetaux en vue du dosage de P, Ca, Mg, Na, K. Ann. Agron. 1970, 21, 229–236. [Google Scholar]
- Fogg, D.N.; Wilkinson, N.T. The colorimetric determination of phosphorus. Analist 1958, 83, 406–414. [Google Scholar] [CrossRef]
- Pedrero, Z.; Madrid, Y.; Camara, C. Selenium species bioaccessibility in enriched radish (Raphanus sativus): A potential dietary source of selenium. J. Agric. Food Chem. 2006, 54, 2412–2417. Available online: https://www.ncbi.nlm.nih.gov/pubmed/16536627 (accessed on 14 February 2019). [CrossRef]
- Hatfield, D.L.; Tsuji, P.A.; Carlson, B.A.; Gladyshev, V.N. Selenium and selenocysteine: Roles in cancer, health, and development. Trends Biochem. Sci. 2014, 39, 112–120. Available online: https://www.ncbi.nlm.nih.gov/pubmed/24485058 (accessed on 14 February 2019). [CrossRef] [PubMed]
- Roman, M.; Jitaru, P.; Barbante, C. Selenium biochemistry and its role for human health. Metallomics 2014, 6, 25–54. Available online: https://www.ncbi.nlm.nih.gov/pubmed/24185753 (accessed on 14 February 2019). [CrossRef]
- Malagoli, M.; Schiavon, M.; dell’Acqua, S.; Pilon-Smits, E.A.H. Effects of selenium biofortification on crop nutritional quality. Front. Plant Sci. 2015, 6, 280. Available online: https://www.ncbi.nlm.nih.gov/pubmed/25954299 (accessed on 14 February 2019). [CrossRef]
- Golubkina, N.A.; Kosheleva, O.V.; Krivenkova, L.V.; Dobrutskaya, H.G.; Nadezhkin, S.; Caruso, G. Intersexual differences in plant growth, yield, mineral composition and antioxidants of spinach (Spinacia oleracea L.) as affected by selenium form. Sci. Hortic. 2017, 225, 350–358. Available online: https://www.sciencedirect.com/science/article/pii/S0304423817304107 (accessed on 14 February 2019). [CrossRef]
- Hawrylak-Nowak, B. Beneficial effects of exogenous selenium in cucumber seedlings subjected to salt stress. Biol. Trace Elem. Res. 2009, 132, 259–269. Available online: https://www.ncbi.nlm.nih.gov/pubmed/19434374 (accessed on 14 February 2019). [CrossRef]
- Pilon-Smits, E.A.H.; Quinn, C.F.; Tapken, W.; Malagoli, M.; Schiavon, M. Physiological functions of beneficial elements. Curr. Opin. Plant Biol. 2009, 12, 267–274. Available online: https://www.sciencedirect.com/science/article/pii/S1369526609000326 (accessed on 14 February 2019). [CrossRef]
- Hajiboland, R.; Amjad, L. Does antioxidant capacity of leaves play a role in growth response to selenium at different sulfur nutritional status? Plant Soil Environ. 2007, 53, 207–215. Available online: https://www.agriculturejournals.cz/publicFiles/00158.pdf (accessed on 14 February 2019). [CrossRef]
- Ferrarese, M.; Mahmoodi, M.; Quattrini, E.; Schiavi, M.; Ferrante, A. Biofortification of spinach plants applying selenium in the nutrient solution of floating system. VCRB 2012, 76, 127–136. Available online: https://content.sciendo.com/view/journals/vcrb/76/1/article-p127.xml (accessed on 14 February 2019). [CrossRef]
- Ávila, F.V.; Yang, Y.; Faquin, V.; Ramos, S.J.; Guilherme, L.R.G.; Thannhauser, T.W.; Li, L. Impact of selenium supply on Se-methylselenocysteine and glucosinolate accumulation in selenium-biofortified Brassica sprouts. Food Chem. 2014, 165, 578–586. Available online: https://www.ncbi.nlm.nih.gov/pubmed/25038715 (accessed on 14 February 2019). [CrossRef]
- Institute of Medicine (US) Panel on Dietary Antioxidants and Related Compounds. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academy Press: Washington, DC, USA, 2000. Available online: https://www.ncbi.nlm.nih.gov/books/NBK225483/ (accessed on 14 February 2019).
- Feist, L.J.; Parker, D.R. Ecotypic variation in selenium accumulation among populations of Stanleya pinnata. New Phytol. 2001, 149, 61–69. Available online: https://nph.onlinelibrary.wiley.com/doi/full/10.1046/j.1469-8137.2001.00004.x (accessed on 14 February 2019). [CrossRef]
- Suarez, D.L.; Grieve, C.M.; Poss, J.A. Irrigation method affects selenium accumulation in forage Brassica species. J. Plant Nutr. 2003, 26, 191–201. Available online: https://www.tandfonline.com/doi/abs/10.1081/PLN-120016504 (accessed on 14 February 2019). [CrossRef]
- White, P.J.; Bowen, H.C.; Parmaguru, P.; Fritz, M.; Spracklen, W.P.; Spiby, R.E.; Meacham, M.C.; Mead, A.; Harriman, M.; Trueman, L.J.; Smith, B.M.; Thomas, B.; Broadley, M.R. Interactions between selenium and sulphur nutrition in Arabidopsis thaliana. J. Exp. Bot. 2004, 55, 1927–1937. Available online: https://www.ncbi.nlm.nih.gov/pubmed/15258164 (accessed on 14 February 2019). [CrossRef] [PubMed]
- Ríos, J.J.; Rosales, M.A.; Blasco, B.; Cervilha, L.; Romero, L.; Ruiz, J.M. Biofortification of Se and induction of the antioxidant capacity in lettuce plants. Sci. Hortic. 2008, 116, 248–255. Available online: https://www.sciencedirect.com/science/article/pii/S0304423808000113 (accessed on 14 February 2019). [CrossRef]
- Boldrin, P.F.; Faquin, V.; Ramos, S.J.; Boldrin, K.V.F.; Ávila, F.W.; Guilherme, L.R.G. Soil and foliar application of selenium in rice biofortification. J. Food Compost. Anal. 2013, 31, 238–244. Available online: https://www.sciencedirect.com/science/article/pii/S0889157513000744 (accessed on 14 February 2019). [CrossRef]
- Ghasemzadeh, A.; Ghasemzadeh, N. Flavonoids and phenolic acids: Role and biochemical activity in plants and human. J. Med. Plant Res. 2011, 5, 6697–6703. Available online: http://www.academicjournals.org/app/webroot/article/article1380724896_Ghasemzadeh%20and%20Ghasemzadeh.pdf (accessed on 14 February 2019). [CrossRef]
- Dixon, R.A.; Paiva, N. Stress-induced phenylpropanoid metabolism. Plant Cell 1995, 7, 1085–1097. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC160915/ (accessed on 14 February 2019). [CrossRef]
- Moglia, A.; Lanteri, S.; Comino, C.; Acquadro, A.; De Vos, R.; Beekwilder, J. Stress-induced biosynthesis of dicaffeoylquinic acids in globe artichoke. J. Agric. Food Chem. 2008, 56, 8641–8649. Available online: https://www.ncbi.nlm.nih.gov/pubmed/18710252 (accessed on 14 February 2019). [CrossRef]
- Lee, M.J.; Lee, G.P.; Park, K.W. Effect of selenium on growth and quality in hydroponically-grown Korean Mint (Agastache rugosa). J. Korean Soc. Hortic. Sci. 2001, 42, 483–486. [Google Scholar]
- Xu, J.; Zhu, S.; Yang, F.; Cheng, L.; Hu, Y.; Pan, G.; Hu, Q. The influence of selenium on the antioxidant activity of green tea. J. Sci. Food Agric. 2003, 83, 451–455. Available online: https://onlinelibrary.wiley.com/doi/pdf/10.1002/jsfa.1405 (accessed on 14 February 2019). [CrossRef]
- Golubkina, N.; Kekina, H.; Caruso, G. Foliar biofortification of Indian mustard (Brassica juncea L.) with selenium and iodine. Plants 2018, 7, 80. [Google Scholar] [CrossRef] [PubMed]
- Wright, M.J.; Davison, K.L. Nitrate accumulation in crops and nitrate poisoning in animals. Adv. Agron. 1964, 16, 197–247. Available online: https://www.sciencedirect.com/science/article/pii/S0065211308600255 (accessed on 14 February 2019).
- Prasad, S.; Chetty, A.A. Nitrate-N determination in leafy vegetables: Study of the effects of cooking and reezing. Food Chem. 2008, 106, 772–780. Available online: https://www.sciencedirect.com/science/article/pii/S0308814607005602 (accessed on 14 February 2019). [CrossRef]
- Lei, B.; Bian, Z.; Yang, Q.; Wang, J.; Cheng, R.; Li, U.; Liu, W.; Zhang, Y.; Fang, H.; Tong, Y. The positive function of selenium supplementation on reducing nitrate accumulation in hydroponic lettuce (Lactuca sativa L.). J. Integr. Agric. 2018, 17, 837–846. Available online: https://www.sciencedirect.com/science/article/pii/S2095311917617593 (accessed on 14 February 2019). [CrossRef]
| Treatments | Se Concentration (mg kg−1 DW) | Se Translocation Factor (Shoot/Root Se ratio) | ||||
|---|---|---|---|---|---|---|
| Shoot | Root | |||||
| Fertigation × 0.0 μmol Se L−1 | 0.71 | i | 1.08 | g | 0.66 | g |
| Fertigation × 1.0 μmol Se L−1 | 1.94 | g | 1.25 | f | 1.55 | c |
| Fertigation × 2.0 μmol Se L−1 | 3.91 | e | 1.88 | e | 2.08 | a |
| Fertigation × 4.0 μmol Se L−1 | 8.83 | c | 8.87 | c | 1.00 | e |
| Fertigation × 8.0 μmol Se L−1 | 17.61 | a | 33.45 | a | 0.53 | h |
| Foliar spray × 0.0 μmol Se L−1 | 0.72 | i | 1.07 | g | 0.67 | g |
| Foliar spray × 1.0 μmol Se L−1 | 0.95 | h | 1.07 | g | 0.89 | f |
| Foliar spray × 2.0 μmol Se L−1 | 2.56 | f | 1.26 | f | 2.03 | a |
| Foliar spray × 4.0 μmol Se L−1 | 5.76 | d | 3.28 | d | 1.75 | b |
| Foliar spray × 8.0 μmol Se L−1 | 12.67 | b | 10.42 | b | 1.22 | d |
| Statistical significance | ||||||
| Type of application | *** | *** | *** | |||
| Se concentration | *** | *** | *** | |||
| Type of application × Se concentration | *** | *** | *** | |||
| Treatments | HFW (g) | HH (cm) | SD (mm) | NL | HDMC (%) | RDMC (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fertigation × 0.0 μmol Se L−1 | 219.6 | e | 27.7 | 25.0 | c | 43.8 | f | 9.5 | b | 2.2 | b | |
| Fertigation × 1.0 μmol Se L−1 | 222.8 | de | 27.5 | 24.6 | c | 43.7 | f | 9.5 | b | 2.2 | b | |
| Fertigation × 2.0 μmol Se L−1 | 276.5 | b | 27.4 | 27.1 | b | 58.1 | c | 10.6 | a | 2.5 | a | |
| Fertigation × 4.0 μmol Se L−1 | 313.2 | a | 27.2 | 29.3 | a | 72.8 | a | 10.7 | a | 2.5 | a | |
| Fertigation × 8.0 μmol Se L−1 | 214.0 | e | 27.5 | 23.5 | d | 47.8 | e | 9.3 | b | 2.2 | b | |
| Foliar spray × 0.0 μmol Se L−1 | 218.4 | e | 27.4 | 24.9 | c | 42.2 | f | 9.5 | b | 2.2 | b | |
| Foliar spray × 1.0 μmol Se L−1 | 219.2 | e | 27.3 | 24.1 | cd | 43.0 | f | 9.5 | b | 2.2 | b | |
| Foliar spray × 2.0 μmol Se L−1 | 233.4 | d | 27.9 | 27.5 | ab | 47.9 | e | 9.5 | b | 2.2 | b | |
| Foliar spray × 4.0 μmol Se L−1 | 251.2 | c | 27.2 | 28.5 | a | 54.1 | d | 9.7 | b | 2.2 | b | |
| Foliar spray × 8.0 μmol Se L−1 | 279.2 | b | 27.0 | 29.0 | a | 63.3 | b | 10.5 | a | 2.6 | a | |
| Statistical significance | ||||||||||||
| Type of application | ** | NS | * | ** | ** | NS | ||||||
| Se concentration | *** | NS | *** | *** | *** | ** | ||||||
| Type of application × Se concentration | *** | NS | *** | *** | *** | *** | ||||||
| Treatments | SSC (°Brix) | TA (%) | Ascorbic Acid (mg kg−1fw) | Total Phenolic (mg of Caffeic Acid g−1f.w.) | ||||
|---|---|---|---|---|---|---|---|---|
| Fertigation × 0.0 μmol Se L−1 | 4.2 | c | 0.6 | e | 76.1 | e | 0.56 | g |
| Fertigation × 1.0 μmol Se L−1 | 4.4 | bc | 0.7 | d | 82.6 | d | 0.62 | fg |
| Fertigation × 2.0 μmol Se L−1 | 4.6 | b | 0.8 | c | 94.9 | c | 0.77 | d |
| Fertigation × 4.0 μmol Se L−1 | 4.9 | a | 1.0 | b | 101.4 | b | 0.89 | b |
| Fertigation × 8.0 μmol Se L−1 | 4.3 | c | 1.1 | a | 104.7 | a | 0.95 | a |
| Foliar spray × 0.0 μmol Se L−1 | 4.2 | c | 0.6 | e | 75.1 | e | 0.55 | g |
| Foliar spray × 1.0 μmol Se L−1 | 4.3 | c | 0.6 | e | 75.3 | e | 0.57 | g |
| Foliar spray × 2.0 μmol Se L−1 | 4.3 | c | 0.6 | e | 82.0 | d | 0.64 | f |
| Foliar spray × 4.0 μmol Se L−1 | 4.5 | b | 0.7 | d | 94.2 | c | 0.73 | e |
| Foliar spray × 8.0 μmol Se L−1 | 4.8 | a | 1.0 | b | 100.4 | b | 0.85 | c |
| Statistical significance | ||||||||
| Type of application | NS | *** | *** | *** | ||||
| Se concentration | *** | *** | *** | *** | ||||
| Type of application × Se concentration | *** | *** | *** | *** | ||||
| Treatments | N (mg g−1 DW) | P (mg g−1 DW) | K (mg g−1 DW) | Ca (mg g−1 DW) | Mg (mg g−1 DW) | S (mg g−1 DW) | S/Se Ratio | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fertigation × 0.0 μmol Se L−1 | 5.60 | a | 0.66 | 3.11 | 0.71 | a | 0.33 | 3.82 | e | 5.35 | a | |||
| Fertigation × 1.0 μmol Se L−1 | 5.24 | bc | 0.65 | 3.07 | 0.70 | a | 0.33 | 3.86 | e | 1.99 | c | |||
| Fertigation × 2.0 μmol Se L−1 | 4.99 | c | 0.63 | 3.11 | 0.55 | cd | 0.33 | 4.24 | c | 1.09 | e | |||
| Fertigation × 4.0 μmol Se L−1 | 4.32 | d | 0.65 | 3.04 | 0.52 | d | 0.35 | 4.67 | b | 0.53 | g | |||
| Fertigation × 8.0 μmol Se L−1 | 3.39 | f | 0.61 | 3.07 | 0.42 | f | 0.34 | 5.47 | a | 0.31 | h | |||
| Foliar spray × 0.0 μmol Se L−1 | 5.60 | a | 0.64 | 3.10 | 0.72 | a | 0.36 | 3.80 | e | 5.30 | a | |||
| Foliar spray × 1.0 μmol Se L−1 | 5.40 | b | 0.68 | 3.12 | 0.73 | a | 0.32 | 3.79 | e | 4.00 | b | |||
| Foliar spray × 2.0 μmol Se L−1 | 5.24 | bc | 0.64 | 3.05 | 0.66 | b | 0.35 | 3.74 | e | 1.46 | d | |||
| Foliar spray × 4.0 μmol Se L−1 | 4.91 | c | 0.66 | 3.06 | 0.58 | c | 0.32 | 4.07 | d | 0.71 | f | |||
| Foliar spray × 8.0 μmol Se L−1 | 4.00 | e | 0.65 | 3.08 | 0.49 | e | 0.36 | 4.57 | b | 0.36 | h | |||
| Statistical significance | ||||||||||||||
| Type of application | *** | NS | NS | *** | NS | *** | *** | |||||||
| Se concentration | *** | NS | NS | *** | NS | *** | *** | |||||||
| Type of application × Se concentration | *** | NS | NS | * | NS | *** | *** | |||||||
| Variable | PC1 | PC2 | PC3 | PC4 |
|---|---|---|---|---|
| Head weight | 0.731 | 0.654 | 0.064 | 0.053 |
| Head height | −0.491 | −0.338 | −0.609 | 0.226 |
| No. of leaves | 0.846 | 0.494 | 0.067 | 0.027 |
| Stem diameter | 0.571 | 0.721 | −0.217 | −0.003 |
| Head dry matter | 0.665 | 0.658 | 0.173 | 0.159 |
| Root dry matter | 0.701 | 0.520 | 0.255 | 0.325 |
| SSC | 0.844 | 0.511 | 0.077 | 0.004 |
| TA | 0.940 | −0.279 | 0.120 | 0.037 |
| Ascorbic acid | 0.972 | −0.115 | −0.036 | −0.168 |
| Total phenolic | 0.972 | −0.201 | 0.001 | −0.093 |
| N | −0.894 | 0.416 | −0.047 | 0.068 |
| P | −0.507 | 0.536 | 0.334 | −0.387 |
| K | −0.574 | −0.012 | 0.606 | 0.076 |
| Ca | −0.947 | 0.249 | 0.028 | 0.088 |
| Mg | 0.338 | −0.005 | −0.242 | 0.840 |
| S | 0.834 | −0.522 | 0.148 | −0.026 |
| Se shoot | 0.860 | −0.468 | 0.063 | −0.047 |
| Se root | 0.632 | −0.767 | 0.073 | −0.026 |
| Se translocation factor | 0.061 | 0.549 | −0.669 | −0.288 |
| S/Se ratio | −0.854 | −0.028 | 0.369 | 0.309 |
| Eigenvalue | 11.201 | 4.308 | 1.698 | 1.279 |
| Variance % | 56.006 | 21.539 | 8.488 | 6.393 |
| Cumulative % | 56.006 | 77.545 | 86.033 | 92.426 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabatino, L.; Ntatsi, G.; Iapichino, G.; D’Anna, F.; De Pasquale, C. Effect of Selenium Enrichment and Type of Application on Yield, Functional Quality and Mineral Composition of Curly Endive Grown in a Hydroponic System. Agronomy 2019, 9, 207. https://doi.org/10.3390/agronomy9040207
Sabatino L, Ntatsi G, Iapichino G, D’Anna F, De Pasquale C. Effect of Selenium Enrichment and Type of Application on Yield, Functional Quality and Mineral Composition of Curly Endive Grown in a Hydroponic System. Agronomy. 2019; 9(4):207. https://doi.org/10.3390/agronomy9040207
Chicago/Turabian StyleSabatino, Leo, Georgia Ntatsi, Giovanni Iapichino, Fabio D’Anna, and Claudio De Pasquale. 2019. "Effect of Selenium Enrichment and Type of Application on Yield, Functional Quality and Mineral Composition of Curly Endive Grown in a Hydroponic System" Agronomy 9, no. 4: 207. https://doi.org/10.3390/agronomy9040207
APA StyleSabatino, L., Ntatsi, G., Iapichino, G., D’Anna, F., & De Pasquale, C. (2019). Effect of Selenium Enrichment and Type of Application on Yield, Functional Quality and Mineral Composition of Curly Endive Grown in a Hydroponic System. Agronomy, 9(4), 207. https://doi.org/10.3390/agronomy9040207









