Glyphosate as a Tool for the Incorporation of New Herbicide Options in Integrated Weed Management in Maize: A Weed Dynamics Evaluation
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
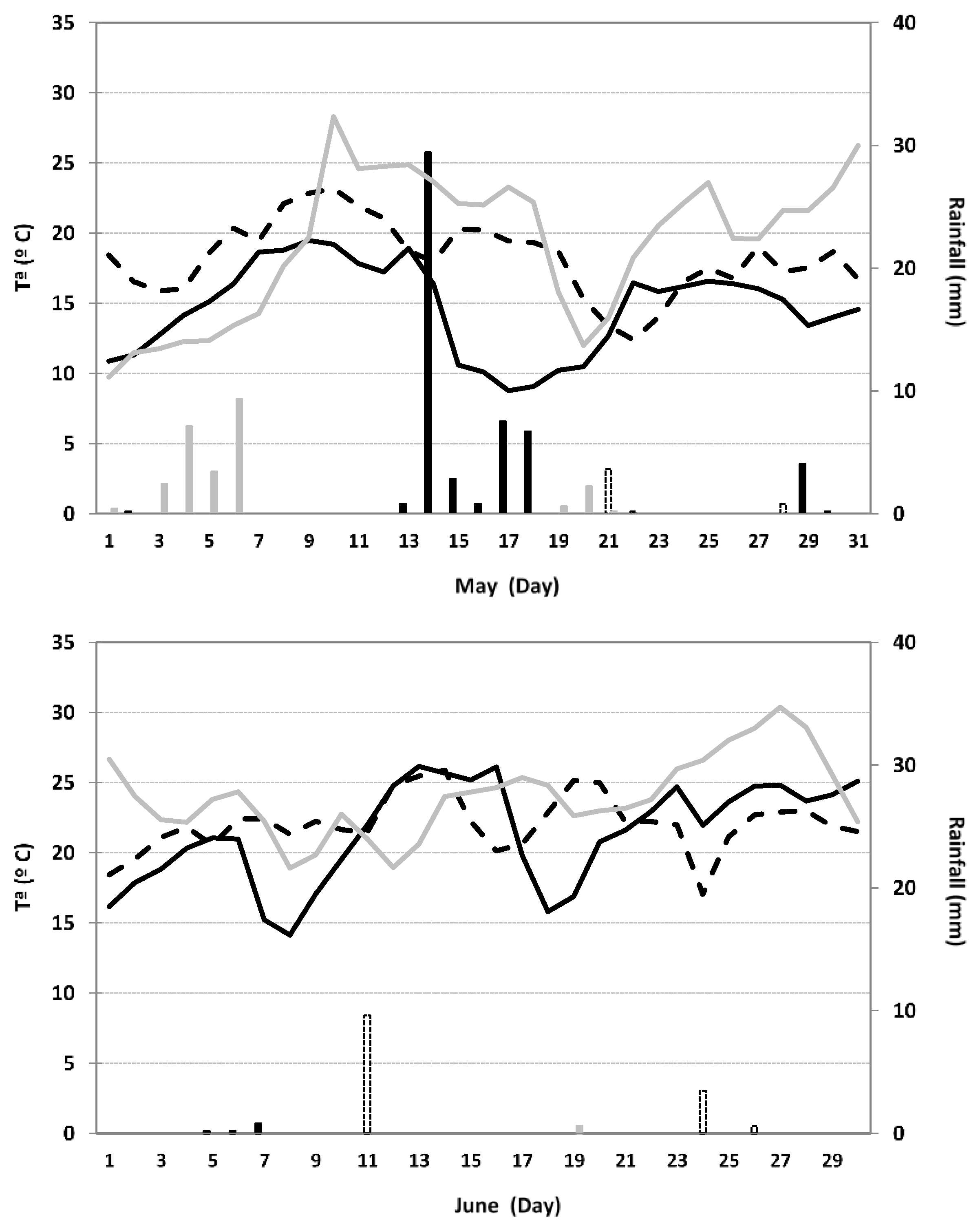
2.2. Experimental Site and Field Layout
2.3. Data Collection
2.4. Data Analysis
3. Results
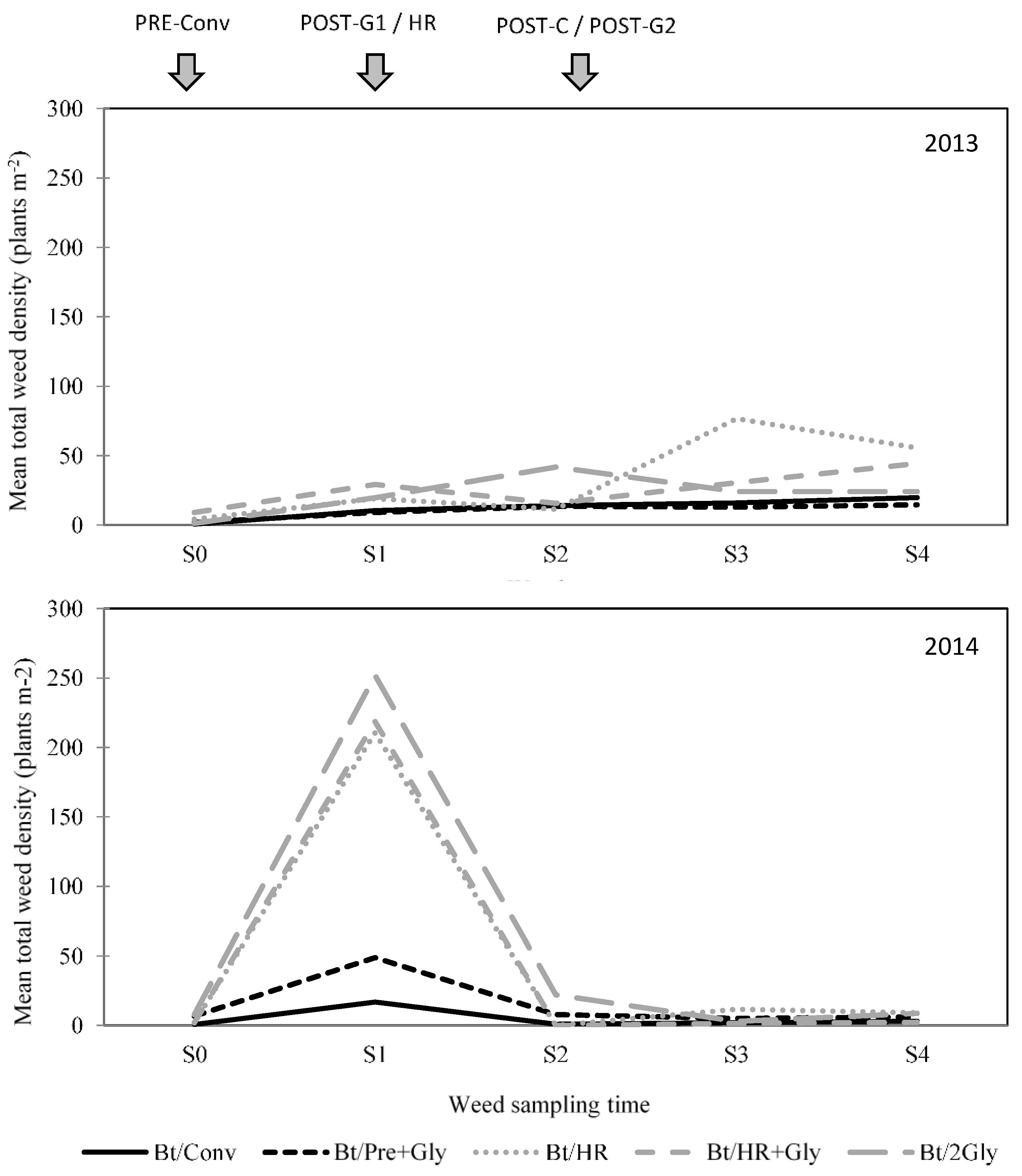
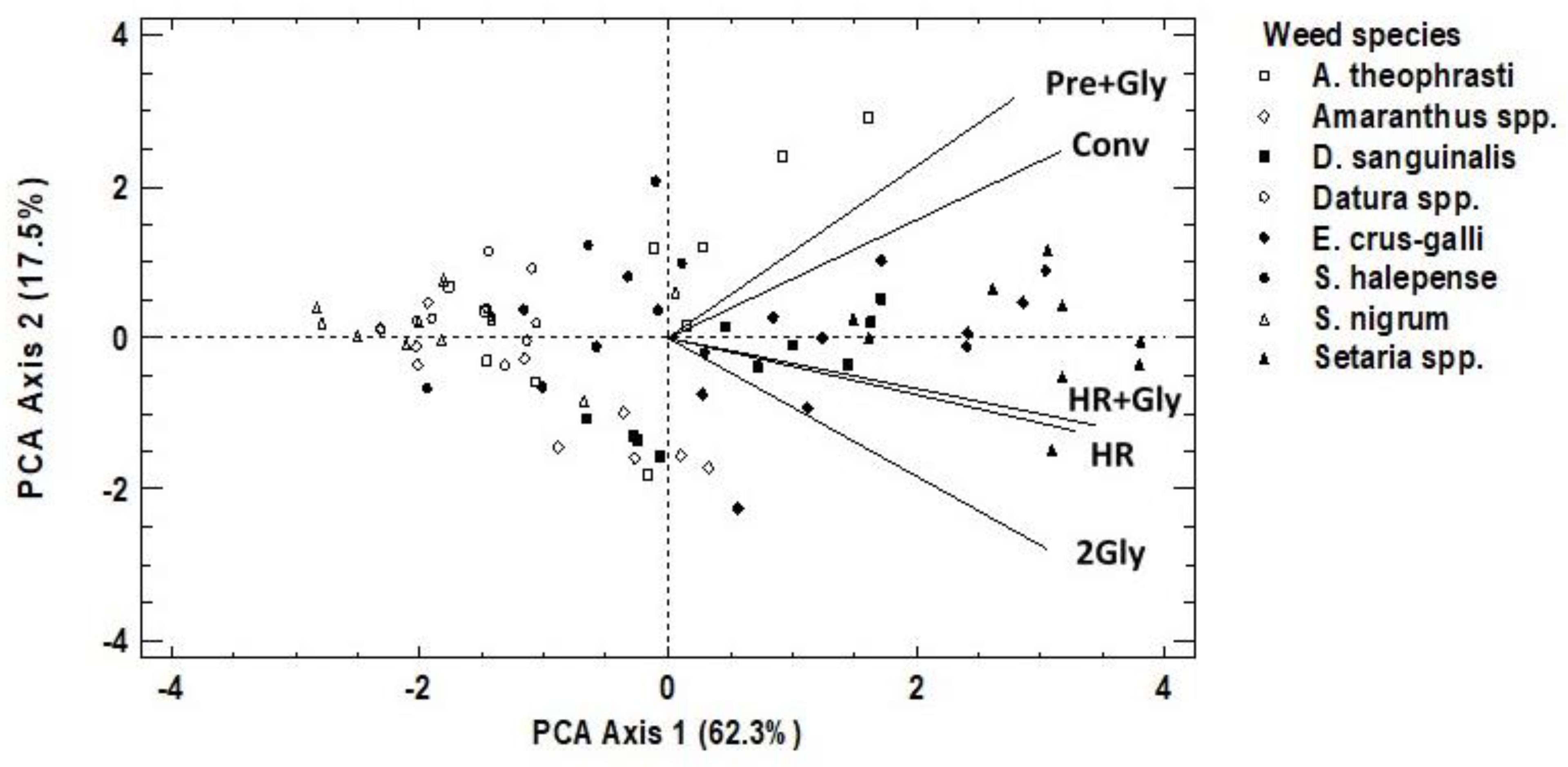
3.1. Weed Species Composition
3.2. Weed Species Abundance, Richness, and Diversity
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Oerke, E.C.; Dehne, H.W. Safeguarding production-losses in major crops and the role of crop protection. Crop. Prot. 2004, 23, 275–285. [Google Scholar] [CrossRef]
- Eurostat. The use of plant protection products in the European Union. Data 1992–2003. 2007. Available online: http://ec.europa.eu/eurostat/en/web/products-statistical-books/-/KS-76-06-669 (accessed on 17 September 2019).
- Birch, A.N.E.; Begg, G.S.; Squire, G.R. How agro-ecological research helps to address food security issues under new IPM and pesticide reduction policies for global crop production systems. J. Exp. Bot. 2011, 62, 3251–3261. [Google Scholar] [CrossRef] [PubMed]
- Meissle, M.; Mouron, P.; Musa, T.; Bigler, F.; Pons, X.; Vasileiadis, V.P.; Otto, S.; Antichi, D.; Kiss, J.; Pálinkás, Z.; et al. Pests, pesticide use and alternative options in European maize production: Current status and future prospects. J. Appl. Entomol. 2010, 134, 357–375. [Google Scholar] [CrossRef]
- Popp, J.; Pető, K.; Nagy, J. Pesticide productivity and food security. A review. Agron. Sust. Dev. 2013, 33, 243–255. [Google Scholar] [CrossRef]
- Directive 2009/128/EC of the European Parliament and of the Council Establishing a Framework for Community Action to Achieve the Sustainable Use of Pesticides. 2009. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=celex%3A32009L0128 (accessed on 17 September 2019).
- Pretty, J. Sustainability in agriculture: Recent progress and emergent challenges. Sustainability in agriculture. Issues Env. Sci. Technol. 2005, 21, 1–15. [Google Scholar]
- James, C. Global Status of Commercialized Biotech/GM Crops: 2016; No. 53; ISAAA: Ithaca, NY, USA, 2017. [Google Scholar]
- Brookes, G.; Barefoot, P. Global impact of biotech crops environmental effects, 1996–2010. GM Crops Food 2012, 3, 1–9. [Google Scholar]
- Cortet, J.; Griffiths, B.S.; Bohanec, M.; Demsar, D.; Andersen, M.N.; Caul, S.; Birch, A.N.E.; Pernin, C.; Tabone, E.; de Vaufleury, A.; et al. Evaluation of effects of transgenic Bt maize on microarthropods in a European multi-site experiment. Pedobiologia 2007, 51, 207–218. [Google Scholar] [CrossRef]
- Romeis, J.; Bartsch, D.; Bigler, F.; Candolfi, M.P.; Gielkens, M.M.C.; Hartley, S.E.; Hellmich, R.L.; Huesing, J.E.; Jepson, P.C.; Layton, R.; et al. Assessment of risk of insect-resistant transgenic crops to non-target arthropods. Nat. Biotechnol. 2008, 26, 203–208. [Google Scholar] [CrossRef]
- Farinós, G.P.; Hernández-Crespo, P.; Ortego, F.; Castañera, P. Monitoring of Sesamia nonagrioides resistance to MON 810 maize in the European Union: Lessons from a long-term harmonized plan. Pest. Manag. Sci. 2018, 74, 557–568. [Google Scholar] [CrossRef]
- Camargo, A.M.; Andow, D.A.; Castañera, P.; Farinós, G.P. First detection of a Sesamia nonagrioides resistance allele to Bt maize in Europe. Sci. Rep. 2018, 8, 3977. [Google Scholar] [CrossRef]
- Pilgrim, E.S.; Macleod, C.J.A.; Blackwell, M.S.A.; Bol, R.; Hogan, D.V.; Chadwick, D.R.; Cardenas, L.; Misselbrook, T.H.; Haygarth, P.M.; Brazier, R.E.; et al. Interactions among agricultural production and other ecosystem services delivered from European grasslands. Adv. Agron. 2010, 109, 117–154. [Google Scholar]
- Duffy, J.E.; Godwin, C.M.; Cardinale, B.J. Biodiversity effects in the wild are common and as strong as key drivers of productivity. Nature 2017, 549, 261–264. [Google Scholar] [CrossRef] [PubMed]
- Benbrook, C.M. Trends in glyphosate herbicide use in the United States and globally. Environ. Sci. Eur. 2016, 28, 3. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.G.; Davis, V.M.; Kruger, G.R.; Weller, S.C. Influence of glyphosate-resistant cropping systems on weed species shifts and glyphosate-resistant weed populations. Eur. J. Agron. 2009, 31, 162–172. [Google Scholar] [CrossRef]
- Bonny, S. Genetically modified herbicide-tolerant crops, weeds, and herbicides: Overview and impact. Environ. Manag. 2016, 57, 31–48. [Google Scholar] [CrossRef]
- García-Ruiz, E.; Loureiro, I.; Farinós, G.P.; Gómez, P.; Gutiérrez, E.; Sánchez, F.J.; Escorial, M.C.; Ortego, F.; Chueca, M.C.; Castañera, P. Weeds and ground-dwelling predators’ response to two different weed management systems in glyphosate-tolerant cotton: A farm-scale study. PLoS ONE 2018, 13, e0191408. [Google Scholar] [CrossRef]
- Gage, K.L.; Krausz, R.F.; Walters, S.A. Emerging challenges for weed management in herbicide-resistant crops. Agriculture 2019, 9, 8. [Google Scholar] [CrossRef]
- Beckie, H.J.; Ashworth, M.B.; Flower, K.C. Herbicide resistance management: Recent developments and trends. Plants 2019, 8, 161. [Google Scholar] [CrossRef]
- Seedek, K.E.M.; Mahas, A.; Mahfouz, M. Plant genome engineering for targeted improvement of crop traits. Front. Plant. Sci. 2019, 10. [Google Scholar] [CrossRef]
- Schütte, G.; Eckerstorfer, M.; Rastelli, V.; Reichenbecher, W.; Restrepo-Vassalli, S.; Ruohonen-Lehto, M.; Saucy, A.-G.W.; Mertens, M. Herbicide resistance and biodiversity: Agronomic and environmental aspects of genetically modified herbicide-resistant plants. Environ. Sci. Eur. 2017, 29. [Google Scholar] [CrossRef]
- Risch, S.J. Agricultural ecology and insect outbreaks. In Insect Outbreaks; Barbos, A.P., Schultz, J.C., Eds.; Academic Press: New York, NY, USA, 1987; pp. 217–238. [Google Scholar]
- Van Emden, H.F. Plant diversity and natural enemy efficiency in agroecosystems. In Critical Issues in Biological Control; Mackauer, M., Ehler, L.E., Roland, J., Eds.; Intercept: Andover, Hants, UK, 1990; pp. 63–80. [Google Scholar]
- Marshall, E.J.P.; Brown, V.K.; Boatman, N.D.; Lutman, P.J.W.; Squire, G.R.; Ward, L.K. The role of weeds in supporting biological diversity within crop fields. Weed Res. 2003, 43, 77–89. [Google Scholar] [CrossRef]
- Storkey, J.; Westbury, D.B. Managing arable weeds for biodiversity. Pest. Manag. Sci. 2007, 63, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Fried, G.; Petit, S.; Dessaint, F.; Reboud, X. Arable weed decline in Northern France: Crop edges as refugia for weed conservation? Biol. Conserv. 2009, 142, 238–243. [Google Scholar] [CrossRef]
- Haddad, N.M.; Crutsinger, G.M.; Gross, K.; Haarstad, J.; Tilman, D. Plant diversity and the stability of foodwebs. Ecol. Lett. 2011, 14, 42–46. [Google Scholar] [CrossRef]
- Isbell, F.; Calcagno, V.; Hector, A.; Connolly, J.; Harpole, W.S.; Reich, P.B.; Scherer-Lorenzen, M.; Schmid, B.; Tilman, D.; van Ruijven, J.; et al. High plant diversity is needed to maintain ecosystem services. Nature 2011, 477, 199–202. [Google Scholar] [CrossRef]
- Petit, S.; Boursault, A.; Le Guilloux, M.; Munier-Jolain, N.; Reboud, X. Weeds in agricultural landscapes: A review. Agron Sustain. Dev. 2011, 31, 309–317. [Google Scholar] [CrossRef]
- Dewar, A.M. Weed control in glyphosate-tolerant maize in Europe. Pest. Manag. Sci. 2009, 65, 1047–1058. [Google Scholar] [CrossRef]
- Derksen, D.; Thomas, A.; Lafond, G.; Loeppky, H.; Swanton, C. Impact of post-emergence herbicides on weed community diversity within conservation-tillage systems. Weed Res. 1995, 35, 311–320. [Google Scholar] [CrossRef]
- Svobodová, Z.; Skokova-Habustova, O.; Holec, Z.; Bohac, J.; Jursik, M.; Soukup, J. Split application of glyphosate herbicide in herbicide-tolerant maize provides efficient weed control and favors beneficial epigeic arthropods. Agric. Ecosyst. Environ. 2018, 251, 171–179. [Google Scholar] [CrossRef]
- Liebman, M.; Mohler, C.L.; Staver, C.P. Ecological Management of Agricultural Weeds; Cambridge University Press: Cambridge, UK, 2001. [Google Scholar]
- Hawes, J.; Haughton, A.J.; Osborne, J.L.; Roy, D.B.; Clark, S.J.; Perry, J.N.; Rothery, P.; Bohan, D.A.; Brooks, D.R.; Champion, G.T.; et al. Responses of plants and invertebrate trophic groups to contrasting herbicide regimes in the farm scale evaluations of genetically modified herbicide-tolerant crops. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2003, 358, 1899–1913. [Google Scholar]
- Birch, A.N.E.; Begg, G.S. The future central role of IPM in EU crop protection: How can ecological research be put into practice? In Proceedings of the Crop Protection in Northern Britain 2010, Dundee, UK, 27–28 February 2018. [Google Scholar]
- Arpaia, S.; Messéan, A.; Birch, A.N.E.; Hokannen, H.; Härtel, S.; van Loon, J.; Lovei, G.; Park, J.; Spreafico, H.; Squire, G.R.; et al. Assessing and monitoring impacts of genetically modified plants on agro-ecosystems: The approach of AMIGA Project. Entomologia 2014, 2, 79–86. [Google Scholar] [CrossRef]
- Vasileiadis, V.P.; Otto, S.; van Dijk, W.; Urek, G.; Leskovšek, R.; Verschwele, A. On-farm evaluation of integrated weed management tools for maize production in three different agro-environments in Europe: Agronomic efficacy, herbicide use reduction, and economic sustainability. Eur. J. Agron. 2015, 63, 71–78. [Google Scholar] [CrossRef]
- Lamichhane, J.R.; Devos, Y.; Beckie, H.J.; Owen, M.D.K.; Tillie, P.; Messéan, A.; Kudsk, P. Integrated weed management systems with herbicide-tolerant crops in the European Union: Lessons learnt from home and abroad. Crit. Rev. Biotechnol. 2017, 37, 459–475. [Google Scholar] [CrossRef]
- SiAR. Agroclimatic Information System for Irrigation Network of the Spanish Ministry of Agriculture and Fisheries, Food and Environment (MAPAMA). Available online: http://eportal.mapama.gob.es/websiar/Inicio.aspx (accessed on 17 September 2019).
- Rozier, C.; Hamzaoui, J.; Lemoine, D.; Czarnes, S.; Legendre, L. Field-based assessment of the mechanism of maize yield enhancement by Azospirillum lipoferum CRT1. Sci. Rep. 2017, 7, 7416. [Google Scholar] [CrossRef]
- Littell, R.C.; Henry, P.R.; Ammerman, C.B. Statistical analysis of repeated measures data using SAS procedures. J. Anim. Sci. 1998, 76, 1216–1231. [Google Scholar] [CrossRef]
- Wang, Z.; Goonewardene, L.A. The use of MIXED models in the analysis of animal experiments with repeated measures data. Can. J. Anim. Sci. 2004, 84, 1–11. [Google Scholar] [CrossRef]
- San Martín, C.; Andújar, D.; Fernández-Quintanilla, C.; Dorado, J. Spatial distribution patterns of weed communities in corn fields of central Spain. Weed Sci. 2015, 63, 936–945. [Google Scholar] [CrossRef]
- Jensen, P.K.; Bibard, V.; Czembor, E.; Dumitru, S.; Foucart, G.; Froud-Williams, R.J.; Jensen, J.E.; Saavedra, M.; Sattin, M.; Soukup, J.; et al. Survey of weeds in maize crops in Europe. Aarhus University. 2011. [cited 2018 April 4]. Available online: https://www.researchgate.net/publication/232775702_survey_of_weeds_in_maize_cops_in_europe (accessed on 17 September 2019).
- Carey, J.B.; Kells, J.J. Timing of total postemergence herbicide applications to maximize weed control and corn (Zea mays) yield. Weed Technol. 1995, 9, 356–361. [Google Scholar] [CrossRef]
- Heard, M.S.; Hawes, C.; Champion, G.T.; Clark, S.J.; Firbank, L.G.; Haughton, A.J.; Parish, A.M.; Perry, J.N.; Rothery, P.; Scott, R.J.; et al. Weeds in fields with contrasting conventional and genetically modified herbicide-tolerant crops. 1. Effects on abundance and diversity. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2003, 358, 1819–1832. [Google Scholar]
- Dieleman, J.A.; Mortensen, D.A. Characterizing the spatial pattern of Abutilon theophrasti seedling patches. Weed Res. 1999, 39, 455–467. [Google Scholar] [CrossRef]
- Armel, G.R.; Wilson, H.P.; Richardson, R.J.; Hines, T.E. Mesotrione alone and in mixture with glyphosate in glyphosate-resistant corn (Zea mays). Weed Technol. 2003, 17, 680–685. [Google Scholar] [CrossRef]
- Hartzler, R.G.; Battles, B.A. Reduced fitness of velvetleaf (Abutilon theophrasti) surviving glyphosate. Weed Technol. 2001, 15, 492–496. [Google Scholar] [CrossRef]
- Schwartz-Lazaro, L.M.; Copes, J.T. A review of the soil seedbank from a weed scientist’s perspective. Agronomy 2019, 9, 7. [Google Scholar] [CrossRef]
- Champion, G.T.; May, M.J.; Bennett, S.; Brooks, D.R.; Clark, S.J.; Daniels, R.E.; Firbank, L.G.; Haughton, A.J.; Hawes, C.; Heard, M.S.; et al. Crop management and agronomic context of the Farm Scale Evaluations of genetically modified herbicide-tolerant crops. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2003, 358, 1801–1818. [Google Scholar]
- Clements, D.; Weise, S.; Swanton, C. Integrated weed management and weed species diversity. Phytoprotection 1994, 75, 1–18. [Google Scholar] [CrossRef]
- Bàrberi, P.; Bocci, G.; Carlesi, S.; Armengot, L.; Blanco-Moreno, J.M.; Sans, F.X. Linking species traits to agroecosystem services: A functional analysis of weed communities. Weed Res. 2018, 58, 76–88. [Google Scholar] [CrossRef]
- Storkey, J.; Brooks, D.; Haughton, A.; Hawes, C.; Smith, B.M.; Holland, J.M. Using functional traits to quantify the value of plant communities to invertebrate ecosystem service providers in arable landscapes. J. Ecol. 2013, 101, 38–46. [Google Scholar] [CrossRef]
- Armengot, L.; Blanco-Moreno, J.M.; Bàrberi, P.; Bocci, G.; Carlesi, S.; Aendekerk, R.; Berner, A.; Celette, F.; Grosse, M.; Huiting, H.; et al. Tillage as a driver of change in weed communities: A functional perspective. Agric. Ecosyst. Environ. 2016, 222, 276–285. [Google Scholar] [CrossRef]
- Burger, J.; Darmency, H.; Granger, S.; Guyot, S.H.M.; Messéan, A.; Colbach, N. Simulation study of the impact of changed cropping practices in conventional and GM maize on weeds and associated biodiversity. Agric. Syst. 2015, 137, 51–63. [Google Scholar] [CrossRef]
- Gabriel, D.; Sait, S.M.; Hodgson, J.A.; Schmutz, U.; Kunin, W.E.; Benton, T.G. Scale matters: The impact of organic farming on biodiversity at different spatial scales. Ecol. Lett. 2010, 13, 858–869. [Google Scholar] [CrossRef]
- Waltz, E. Glyphosate resistance threatens Roundup hegemony. Nat. Biotechnol. 2010, 28, 537–538. [Google Scholar] [CrossRef] [PubMed]
- Owen, M.D.K. Weed species shifts in glyphosate-resistant crops. Pest. Manag. Sci. 2008, 64, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Powles, S.B. Evolved glyphosate-resistant weeds around the world: Lessons to be learnt. Pest. Manag. Sci. 2008, 64, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Bàrberi, P.; Burgio, G.; Dinelli, G.; Moonen, A.C.; Otto, S.; Vazzana, C.; Zanin, G. Functional biodiversity in the agricultural landscape: Relationships between weeds and arthropod fauna. Weed Res. 2010, 50, 388–401. [Google Scholar] [CrossRef]
- Brooks, D.R.; Storkey, J.; Clark, S.J.; Firbank, L.G.; Petit, S.; Woiwod, I.P. Trophic links between functional groups of arable plants and beetles are stable at a national scale. J. Anim. Ecol. 2012, 81, 4–13. [Google Scholar] [CrossRef]
| Field Operations | Year | |||
|---|---|---|---|---|
| 2012 | 2013 | 2014 | ||
| Soil preparation | Disk harrowing | 24 and 27 January | 20 March | 6 and 10 March |
| Subsoiling | 31 January | 9 April | 14 March | |
| Rotovating | 14 February | 16 April | 29 March | |
| Sowing date | 11 May | 9 May | 19 May | |
| Maize seed variety | DKC6450 DKC6451YG | DKC6450 DKC6451YG | DKC6450 DKC6451YG | |
| Planting density | 80,000 seeds ha−1 | 80,000 seeds ha−1 | 80,000 seeds ha−1 | |
| Row width | 0.75 m | 0.75 m | 0.75 m | |
| Fertilization | Basal dressing (NPK) | 13 February 8–15–15, 300 kg ha−1 | 15 April 8–15–15, 300 kg ha−1 | 27 March 8–15–15, 300 kg ha−1 |
| Topdressing | 8 June Urea-46, 300 kg ha−1 | 15 June CAN 27% *, 500 kg ha−1 | 1 July Urea-46, 600 kg ha−1 | |
| First irrigation | 9 June | 2 June | 17 June | |
| Plant height | 2.60 m | 2.70 m | 2.75 m | |
| Flowering | 24 July | 3 August | 7 August | |
| Harvest | 5 December | 9 January | 30 December | |
| Weed Management Program | Herbicide Treatments (N°) | Application Time (Maize Growth Stage) | Herbicide Active Ingredients | Rate (g a.i.ha−1) |
|---|---|---|---|---|
| Conventional (Conv.) | 2 | PRE | S-metolachlor 31.25% + terbuthylazine 18.75% | 1250 750 |
| POST (6–8 leaf stage) | nicosulfuron 6% + mesotrione 10% | 39 100 | ||
| Herbicide Reduced (HR) | 1 | POST (6–8 leaf stage) | S-metolachlor 31.25% + terbuthylazine 18.75% + nicosulfuron 6% + mesotrione 10% | 938 562 45 50 |
| Pre + Gly | 2 | PRE | S-metolachlor 31.25% + terbuthylazine 18.75% | 1250 750 |
| POST (6–8 leaf stage) | glyphosate 36% | 1080 | ||
| HR + Gly | 2 | POST (4–6 leaf stage) | S-metholachlor 31.25% + terbuthylazine 18.75% + nicosulfuron 6% + mesotrione 10% | 938 562 45 50 |
| POST (8–10 leaf stage) | glyphosate 36% | 1080 | ||
| 2 Gly | 2 | POST (4–6 leaf stage) | glyphosate 36% | 1080 |
| POST (8–10 leaf stage) | glyphosate 36% | 1080 |
| Weed | Weed Management Program | Treatment (T) (F2,14 (P)) | ||
|---|---|---|---|---|
| Bt/HR | Bt/HR + Gly | Bt/2 Gly | ||
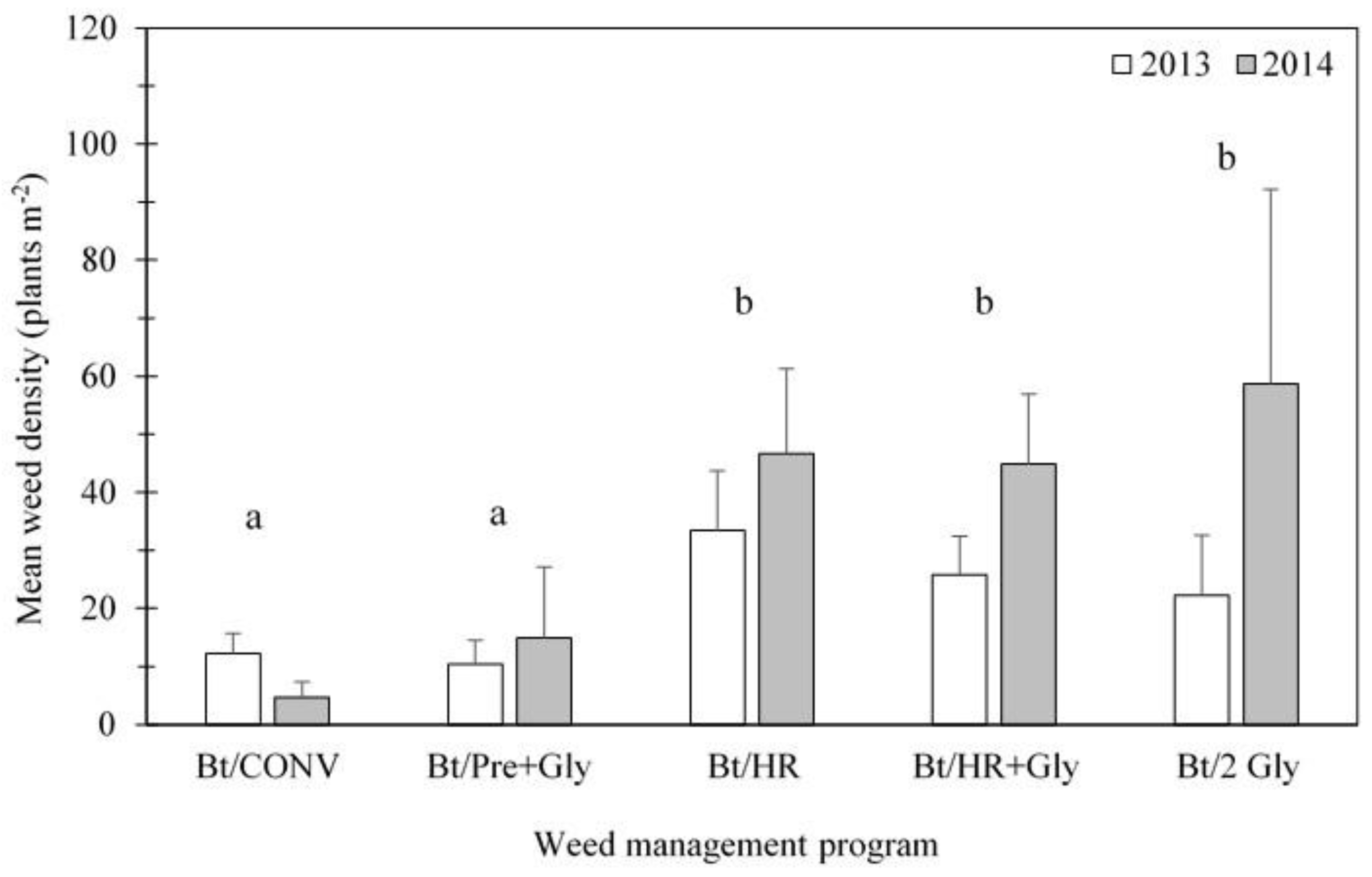
| Abundance (plants m−2) | 9.7 ± 4.1 | 31.3 ± 10.6 | 4.9 ± 3.0 | 119.26 * (0.00) |
| Species Richness (S) | 3.9 ± 0.3 | 6.5 ± 0.6 | 3.9 ± 0.9 | 23.06 * (0.00) |
| Shannon-Wiener diversity index (H′) | 0.9 ± 0.1 | 1.1 ± 0.2 | 1.0 ± 0.2 | 1.81 (0.21) |
| Weed Management Program | Plant Density | |||||||
|---|---|---|---|---|---|---|---|---|
| Dicotyledonous | Monocotyledonous | |||||||
| Abutilon theophrasti | Amaranthus spp. | Datura spp. | Solanum nigrum | Digitaria sanguinalis | Echinochloa crus-galli | Setaria spp. | Sorghum halepense | |
| Conv | 2.8 ± 4.2 a | 0.2 ± 0.3 a | 0.6 ± 0.5 | 0.4 ± 0.4 | 3.6 ± 5.0 a | 5.6 ± 5.0 a | 8.6 ± 5.9 a | 1.9 ± 2.0 |
| Pre + Gly | 10.9 ± 19.8 ab | 0.4 ± 0.5 a | 1.4 ± 1.4 | 0.8 ± 1.7 | 1.7 ± 2.2 a | 5.9 ± 5.3 a | 11.0 ± 7.6 a | 2.3 ± 3.1 |
| HR | 1.2 ± 0.8 a | 5.4 ± 7.6 b | 1.6 ± 1.3 | 1.5 ± 1.4 | 24.5 ± 11.6 d | 16.8 ± 9.2 b | 62.1 ± 40.5 b | 4.6 ± 3.3 |
| HR + Gly | 12.8 ± 11.9 b | 3.9 ± 3.8 b | 1.4 ± 0.9 | 2.3 ± 2.1 | 6.4 ± 4.4 b | 28.8 ± 22.0 b | 44.2 ± 40.0 b | 4.9 ± 3.8 |
| 2 Gly | 4.1 ± 6.3 ab | 16.2 ± 18.5 b | 2.1 ± 2.7 | 0.8 ± 1.2 | 11.3 ± 6.6 c | 18.4 ± 15.2 b | 61.9 ± 63.7 b | 2.7 ± 2.7 |
| Y (F1,40 (P)) | 2.42 (0.12) | 11.46 * (0.00) | 0.89 (0.35) | 1.04 (0.31) | 14.26 * (0.00) | 4.11 * (0.04) | 16.83 * (0.00) | 2.15 (0.15) |
| T (F4,40 (P)) | 4.00 * (0.01) | 10.09 * (0.01) | 1.02 (0.41) | 2.59 (0.05) | 29.201 * (0.00) | 9.31 * (0.00) | 18.37 * (0.00) | 1.53 (0.21) |
| Y × T (F4,40 (P)) | 0.35 (0.84) | 2.51 (0.06) | 1.14 (0.96) | 0.74 (0.57) | 3.43 * (0.01) | 0.91 (0.46) | 6.03 * (0.00) | 2.35 (0.07) |
| Year | Weed Management Program | Year (Y) (F1,40 (P)) | Treatment (T) (F4,40 (P)) | Y × T (F4,40 (P)) | |||||
|---|---|---|---|---|---|---|---|---|---|
| Conv | Pre + Gly | HR | HR + Gly | 2 Gly | |||||
| Species Richness (S) | 2013 | 4.8 ± 0.9 | 4.7 ± 1.5 | 5.0 ± 1.5 | 5.1 ± 0.3 | 5.1 ± 0.7 | 35.19 * (0.00) | 2.23 (0.08) | 1.21 (0.32) |
| 2014 | 2.9 ± 0.6 | 3.2 ± 1.2 | 3.5 ± 0.3 | 3.1 ± 0.3 | 4.6 ± 0.4 | ||||
| Diversity index (H′) | 2013 | 1.0 ± 0.1 | 1.1 ± 0.4 | 0.9 ± 0.1 | 1.0 ± 0.2 | 1.0 ± 0.2 | 36.04 * (0.00) | 2.11 (0.09) | 1.53 (0.21) |
| 2014 | 0.7 ± 0.1 | 0.7 ± 0.3 | 0.6 ± 0.1 | 0.5 ± 0.1 | 0.9 ± 0.2 | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loureiro, I.; Santin-Montanyá, I.; Escorial, M.-C.; García-Ruiz, E.; Cobos, G.; Sánchez-Ramos, I.; Pascual, S.; González-Núñez, M.; Chueca, M.-C. Glyphosate as a Tool for the Incorporation of New Herbicide Options in Integrated Weed Management in Maize: A Weed Dynamics Evaluation. Agronomy 2019, 9, 876. https://doi.org/10.3390/agronomy9120876
Loureiro I, Santin-Montanyá I, Escorial M-C, García-Ruiz E, Cobos G, Sánchez-Ramos I, Pascual S, González-Núñez M, Chueca M-C. Glyphosate as a Tool for the Incorporation of New Herbicide Options in Integrated Weed Management in Maize: A Weed Dynamics Evaluation. Agronomy. 2019; 9(12):876. https://doi.org/10.3390/agronomy9120876
Chicago/Turabian StyleLoureiro, Iñigo, Inés Santin-Montanyá, María-Concepción Escorial, Esteban García-Ruiz, Guillermo Cobos, Ismael Sánchez-Ramos, Susana Pascual, Manuel González-Núñez, and María-Cristina Chueca. 2019. "Glyphosate as a Tool for the Incorporation of New Herbicide Options in Integrated Weed Management in Maize: A Weed Dynamics Evaluation" Agronomy 9, no. 12: 876. https://doi.org/10.3390/agronomy9120876
APA StyleLoureiro, I., Santin-Montanyá, I., Escorial, M.-C., García-Ruiz, E., Cobos, G., Sánchez-Ramos, I., Pascual, S., González-Núñez, M., & Chueca, M.-C. (2019). Glyphosate as a Tool for the Incorporation of New Herbicide Options in Integrated Weed Management in Maize: A Weed Dynamics Evaluation. Agronomy, 9(12), 876. https://doi.org/10.3390/agronomy9120876







