Breeding Strategies to Improve Miscanthus as a Sustainable Source of Biomass for Bioenergy and Biorenewable Products
Abstract
1. Introduction
2. Breeding Strategies
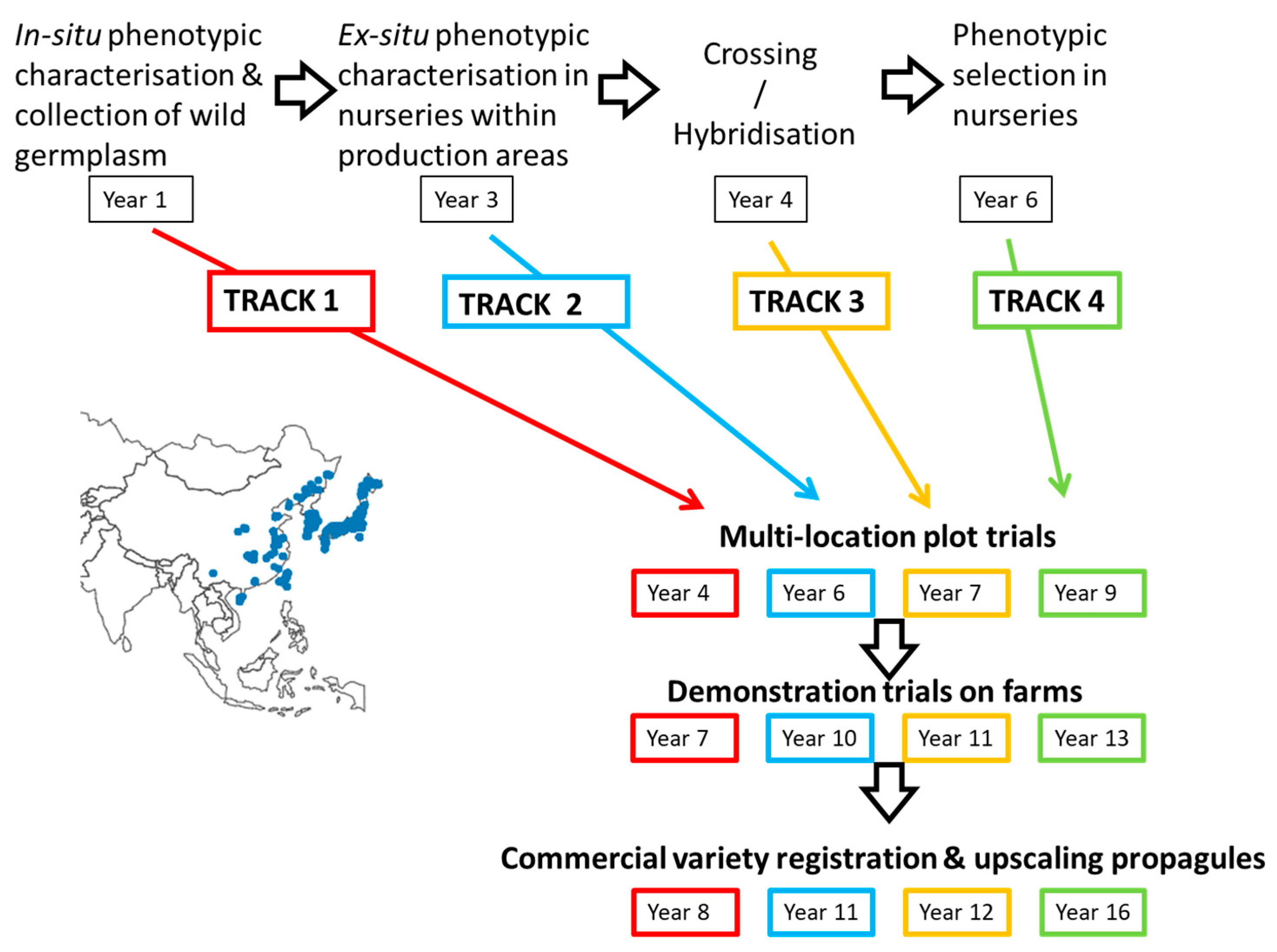
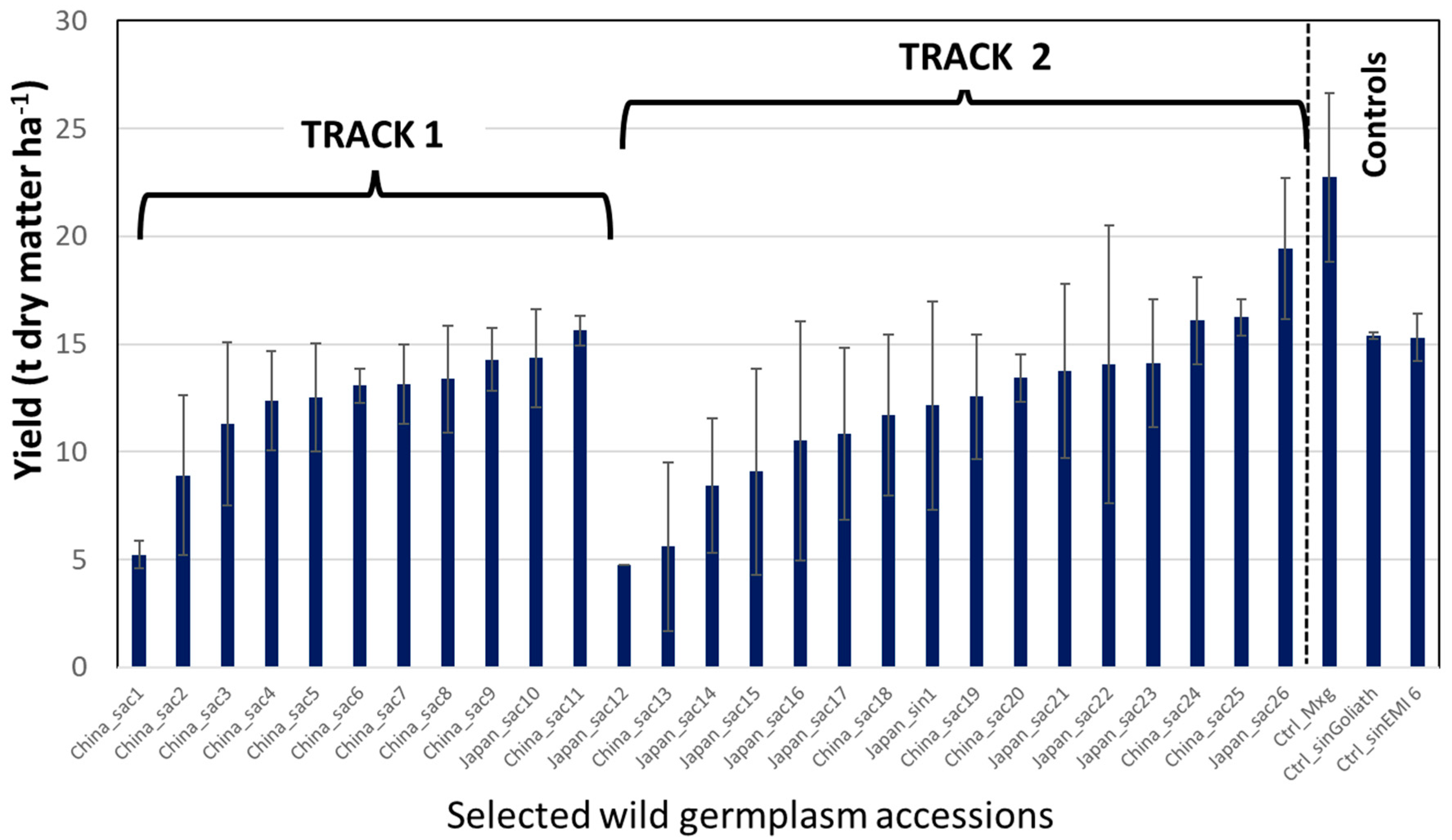
3. Aberystwyth Experience of Implementing Fast Tracks
4. Tracks 3 and 4: Improvement through Hybridisation
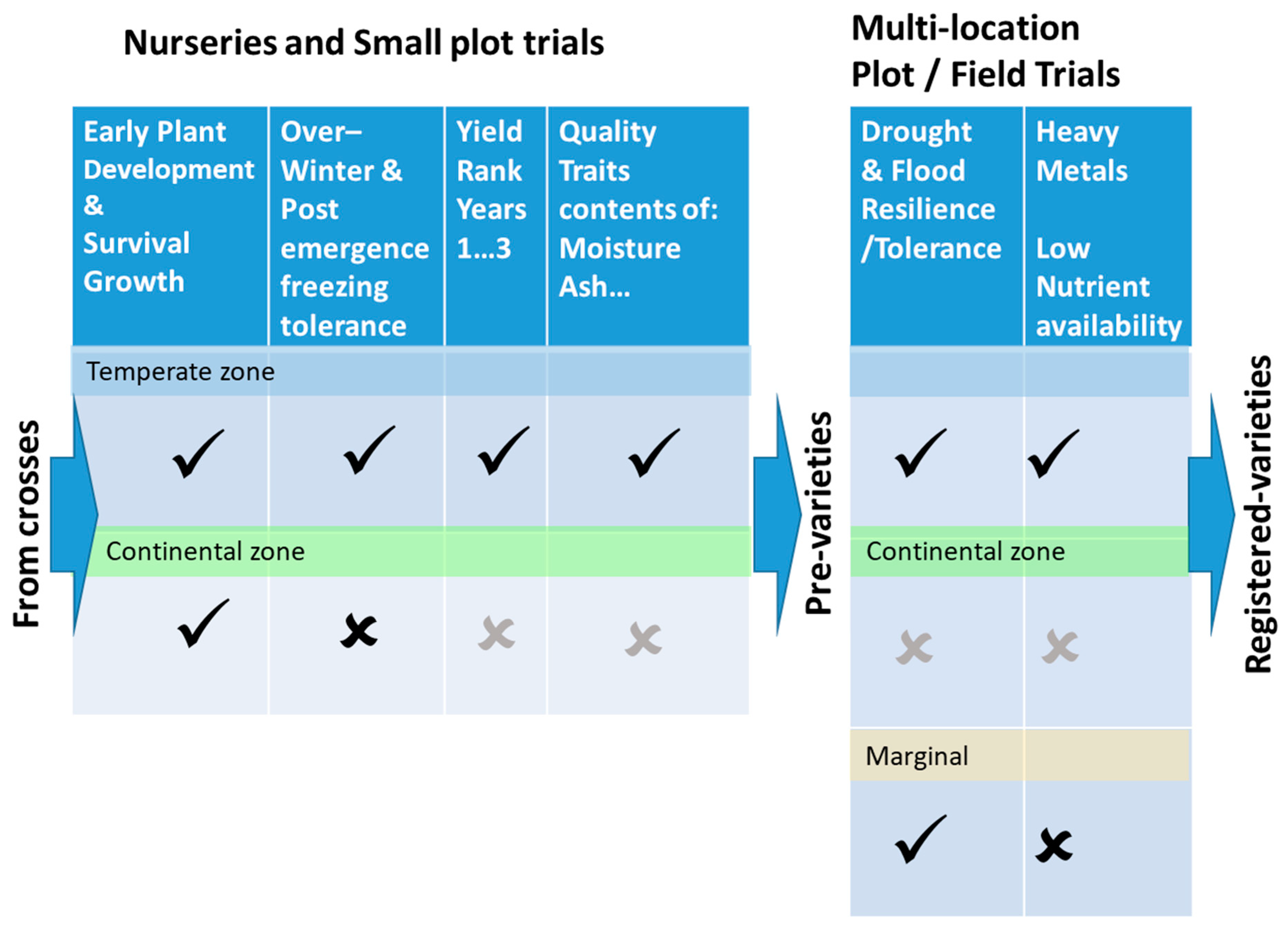
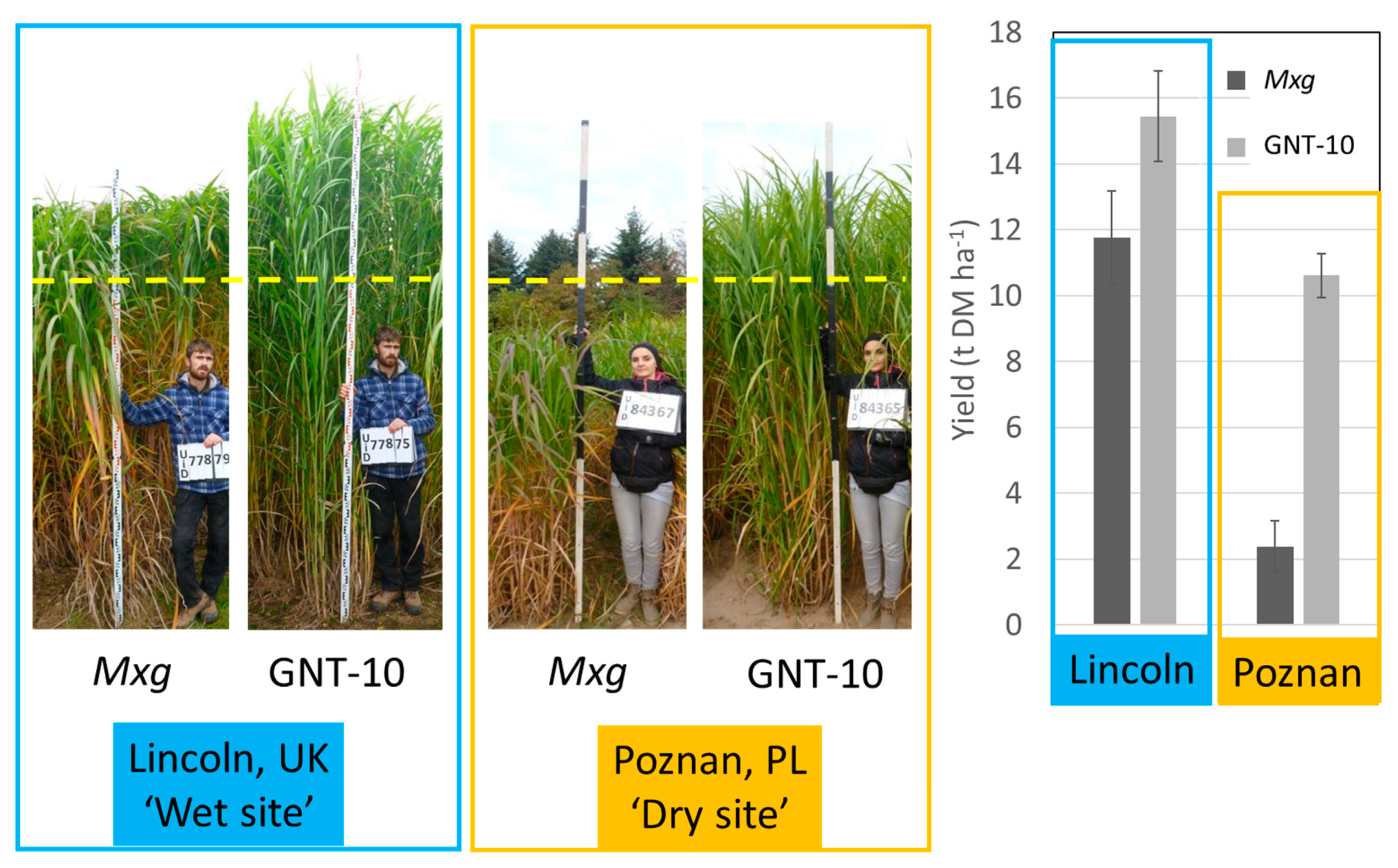
5. Phenotyping in Multi-Location Plot Trials
6. Selection Criteria and Methodology
7. Conclusions and Future Improvements in Miscanthus Breeding
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Henderson, G.; Shah, N.; Azapagic, A.; Beerling, D.; Cheeseman, C.; Hepburn, C.; House, J.I.; Le Quéré, C.; Markusson, N.; Shepherd, J.; et al. Greenhouse Gas Removal; The Royal Society and Royal Academy of Engineering: London, UK, 2018; p. 136. Available online: royalsociety.org/greenhouse-gas-removal (accessed on 28 May 2019).
- Robson, P.; Hastings, A.; Clifton-Brown, J.; McCalmont, J. Chapter 15: Sustainable use of Miscanthus for biofuel. In Achieving Carbon-Negative Bioenergy Systems from Plant Materials; Saffron, E., Ed.; Burleigh Dodds Science Publishing Ltd.: Cambridge, UK, 2019. [Google Scholar]
- Stewart, J.R.; Toma, Y.; Fernandez, F.G.; Nishiwaki, A.; Yamada, T.; Bollero, G. The ecology and agronomy of Miscanthus sinensis, a species important to bioenergy crop development, in its native range in Japan: A review. Glob. Chang. Biol. Bioenergy 2009, 1, 126–153. [Google Scholar] [CrossRef]
- Kalinina, O.; Nunn, C.; Sanderson, R.; Hastings, A.F.S.; van der Weijde, T.; Ozguven, M.; Tarakanov, I.; Schule, H.; Trindade, L.M.; Dolstra, O.; et al. Extending Miscanthus Cultivation with Novel Germplasm at Six Contrasting Sites. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef]
- Greef, J.M.; Deuter, M.; Jung, C.; Schondelmaier, J. Genetic diversity of European Miscanthus species revealed by AFLP fingerprinting. Genet. Resour. Crop Evol. 1997, 44, 185–195. [Google Scholar] [CrossRef]
- Clifton-Brown, J.C.; Lewandowski, I.; Andersson, B.; Basch, G.; Christian, D.G.; Kjeldsen, J.B.; Jorgensen, U.; Mortensen, J.V.; Riche, A.B.; Schwarz, K.U.; et al. Performance of 15 Miscanthus genotypes at five sites in Europe. Agron. J. 2001, 93, 1013–1019. [Google Scholar] [CrossRef]
- Lewandowski, I.; Clifton-Brown, J.C.; Scurlock, J.M.O.; Huisman, W. Miscanthus: European experience with a novel energy crop. Biomass Bioenergy 2000, 19, 209–277. [Google Scholar] [CrossRef]
- Heaton, E.A.; Dohleman, F.G.; Miguez, A.F.; Juvik, J.A.; Lozovaya, V.; Widholm, J.; Zabotina, O.A.; Mcisaac, G.F.; David, M.B.; Voigt, T.B.; et al. Miscanthus: A Promising Biomass Crop. Adv. Bot. Res. 2010, 56, 75–137. [Google Scholar] [CrossRef]
- Ings, J.; Mur, L.A.J.; Robson, P.R.H.; Bosch, M. Physiological and growth responses to water deficit in the bioenergy crop Miscanthus x giganteus. Front. Plant Sci. 2013, 4. [Google Scholar] [CrossRef]
- Clifton-Brown, J.C.; Lewandowski, I.; Bangerth, F.; Jones, M.B. Comparative responses to water stress in stay-green, rapid- and slow senescing genotypes of the biomass crop, Miscanthus. New Phytol. 2002, 154, 335–345. [Google Scholar] [CrossRef]
- Hastings, A.; Smith, P.; Clifton-Brown, J.; Taylor, G. Future biomass supply for low carbon European energy provision in a changing world. In Proceedings of the TERRAenVISION Environmental Issues Today: Scientific Solutions for Societal Issues, Barcelona, Spain, 27 January–2 February 2018. [Google Scholar]
- Hastings, A.; Clifton-Brown, J.; Wattenbach, M.; Mitchell, C.P.; Stampfl, P.; Smith, P. Future energy potential of Miscanthus in Europe. Glob. Chang. Biol. Bioenergy 2009, 1, 180–196. [Google Scholar] [CrossRef]
- Clifton-Brown, J.; Schwarz, K.U.; Hastings, A. History of the Development of Miscanthus as a Bioenergy Crop: From Small Beginnings to Potential Realisation. Biol. Environ. 2015, 115B, 45–57. [Google Scholar] [CrossRef]
- Clifton-Brown, J.; Harfouche, A.; Casler, M.D.; Jones, H.D.; Macalpine, W.J.; Murphy-Bokern, D.; Smart, L.B.; Adler, A.; Ashman, C.; Awty-Carroll, D.; et al. Breeding progress and preparedness for mass-scale deployment of perennial lignocellulosic biomass crops switchgrass, Miscanthus, willow and poplar. Glob. Chang. Biol. Bioenergy 2019, 11, 118–151. [Google Scholar] [CrossRef]
- Clifton-Brown, J.; Hastings, A.; Mos, M.; Mccalmont, J.P.; Ashman, C.; Awty-Carroll, D.; Cerazy, J.; Chiang, Y.C.; Cosentino, S.; Cracroft-Eley, W.; et al. Progress in upscaling Miscanthus biomass production for the European bio-economy with seed-based hybrids. Glob. Chang. Biol. Bioenergy 2017, 9, 6–17. [Google Scholar] [CrossRef]
- Robson, P.; Jensen, E.; Hawkins, S.; White, S.R.; Kenobi, K.; Clifton-Brown, J.; Donnison, I.; Farrar, K. Accelerating the domestication of a bioenergy crop: Identifying and modelling morphological targets for sustainable yield increase in Miscanthus. J. Exp. Bot. 2013, 64, 4143–4155. [Google Scholar] [CrossRef]
- Slavov, G.; Robson, P.; Jensen, E.; Hodgson, E.; Farrar, K.; Allison, G.; Hawkins, S.; Thomas-Jones, S.; Ma, X.F.; Huang, L.; et al. Contrasting geographic patterns of genetic variation for molecular markers vs. phenotypic traits in the energy grass Miscanthus sinensis. Glob. Chang. Biol. Bioenergy 2013, 5, 562–571. [Google Scholar] [CrossRef]
- Xi, Q.; Jezowkski, S. Plant resources of Triarrhena and Miscanthus species in China and its meaning for Europe. Plant Breed. Seed Sci. 2004, 49, 63–77. [Google Scholar]
- Huang, L.S.; Flavell, R.; Donnison, I.S.; Chiang, Y.-C.; Hastings, A.; Hayes, C.; Heidt, C.; Hong, H.; Hsu, T.-W.; Humphreys, M.; et al. Collecting wild Miscanthus germplasm in Asia for crop improvement and conservation in Europe whilst adhering to the guidelines of the United Nations’ Convention on Biological Diversity. Ann. Bot.-Lond. 2018. [Google Scholar] [CrossRef]
- Hayes, C.; Virk, D.S. Assessing the Relative Efficacy of Polyester Pollination Bags and Crossing Tents and Isolation Chambers for Seed Harvest in Miscanthus Crosses. Int. J. Plant Breed. Genet. 2016, 10, 79–90. [Google Scholar] [CrossRef][Green Version]
- Clifton-Brown, J.C.; Senior, H.; Purdy, S.J.; Horsnell, R.; Lankamp, B.; Müennekhoff, A.-K.; Virk, D.; Guillemois, E.; Chetty, V.; Cookson, A. Investigating the potential of novel non-woven fabrics for efficient pollination control in plant breeding. PLoS ONE 2018, 13, e0204728. [Google Scholar] [CrossRef]
- Lewandowski, I.; Clifton-Brown, J.; Trindade, L.M.; van der Linden, G.C.; Schwarz, K.U.; Muller-Samann, K.; Anisimov, A.; Chen, C.L.; Doistre, O.; Donnison, I.S.; et al. Progress on Optimizing Miscanthus Biomass Production for the European Bioeconomy: Results of the EU FP7 Project OPTIMISC. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef]
- Van der Weijde, T.; Huxley, L.M.; Hawkins, S.; Sembiring, E.H.; Farrar, K.; Dolstra, O.; Visser, R.G.; Trindade, L.M. Impact of drought stress on growth and quality of Miscanthus for biofuel production. Glob. Chang. Biol. Bioenergy 2017, 9, 770–782. [Google Scholar] [CrossRef]
- Malinowska, M.; Donnison, I.S.; Robson, P.R.H. Phenomics analysis of drought responses in Miscanthus collected from different geographical locations. Glob. Chang. Biol. Bioenergy 2016. [Google Scholar] [CrossRef]
- Stavridou, E.; Hastings, A.; Webster, R.; Robson, P. The impact of soil salinity on the yield, composition and physiology of the bioenergy grass Miscanthus x giganteus. Glob. Chang. Biol. Bioenergy 2016. [Google Scholar] [CrossRef]
- Robson, P.R.; Donnison, I.S.; Clifton-Brown, J.C. Stem growth characteristics of high yielding Miscanthus correlate with yield, development and intraspecific competition within plots. GCB Bioenergy 2019. [Google Scholar] [CrossRef]
- Wagner, M.; Mangold, A.; Lask, J.; Petig, E.; Kiesel, A.; Lewandowski, I. Economic and environmental performance of Miscanthus cultivated on marginal land for biogas production. Glob. Chang. Biol. Bioenergy 2019, 11, 34–49. [Google Scholar] [CrossRef]
- Knörzer, H.; Hartung, K.; Piepho, H.P.; Lewandowski, I. Assessment of variability in biomass yield and quality: What is an adequate size of sampling area for Miscanthus? Glob. Chang. Biol. Bioenergy 2013, 5, 572–579. [Google Scholar] [CrossRef]
- Ashman, C.; Awty-Carroll, D.; Mos, M.; Robson, P.; Clifton-Brown, J. Assessing seed priming, sowing date, and mulch film to improve the germination and survival of direct-sown Miscanthus sinensis in the United Kingdom. Glob. Chang. Biol. Bioenergy 2018, 10, 612–627. [Google Scholar] [CrossRef]
- Clifton-Brown, J.C.; Lewandowski, I. Overwintering problems of newly established Miscanthus plantations can be overcome by identifying genotypes with improved rhizome cold tolerance. New Phytol. 2000, 148, 287–294. [Google Scholar] [CrossRef]
- Rusinowski, S.; Krzyżak, J.; Clifton-Brown, J.; Jensen, E.; Mos, M.; Webster, R.; Sitko, K.; Pogrzeba, M. New Miscanthus hybrids cultivated at a Polish metal(loid)-contaminated site demonstrate superior stomatal regulation and reduced shoot Pb and Cd concentrations. Environ. Pollut. 2019. (In revision) [Google Scholar] [CrossRef]
- Chae, W.B.; Hong, S.J.; Gifford, J.M.; Rayburn, A.L.; Widholm, J.M.; Juvik, J.A. Synthetic polyploid production of Miscanthus sacchariflorus, Miscanthus sinensis, and Miscanthus x giganteus. Glob. Chang. Biol. Bioenergy 2013, 5, 338–350. [Google Scholar] [CrossRef]
- Wang, X.; Yamada, T.; Kong, F.J.; Abe, Y.; Hoshino, Y.; Sato, H.; Takamizo, T.; Kanazawa, A.; Yamada, T. Establishment of an efficient in vitro culture and particle bombardment-mediated transformation systems in Miscanthus sinensis Anderss., a potential bioenergy crop. Glob. Chang. Biol. Bioenergy 2011, 3, 322–332. [Google Scholar] [CrossRef]
- Kim, C.; Wang, X.Y.; Lee, T.H.; Jakob, K.; Lee, G.J.; Paterson, A.H. Comparative Analysis of Miscanthus and Saccharum Reveals a Shared Whole-Genome Duplication but Different Evolutionary Fates. Plant Cell 2014, 26, 2420–2429. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.; Zhang, D.; Auckland, S.A.; Rainville, L.K.; Jakob, K.; Kronmiller, B.; Sacks, E.J.; Deuter, M.; Paterson, A.H. SSR-based genetic maps of Miscanthus sinensis and M. sacchariflorus, and their comparison to sorghum. Theor. Appl. Genet. 2012, 124, 1325–1338. [Google Scholar] [CrossRef]
- Swaminathan, K.; Alabady, M.; Varala, K.; De Paoli, E.; Ho, I.; Rokhsar, D.; Arumuganathan, A.; Ming, R.; Green, P.; Meyers, B.; et al. Genomic and small RNA sequencing of Miscanthus × giganteus shows the utility of sorghum as a reference genome sequence for Andropogoneae grasses. Genome Biol. 2010, 11, 2–18. [Google Scholar] [CrossRef]
- Ma, X.F.; Jensen, E.; Alexandrov, N.; Troukhan, M.; Zhang, L.P.; Thomas-Jones, S.; Farrar, K.; Clifton-Brown, J.; Donnison, I.; Swaller, T.; et al. High Resolution Genetic Mapping by Genome Sequencing Reveals Genome Duplication and Tetraploid Genetic Structure of the Diploid Miscanthus sinensis. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Davey, C.L.; Robson, P.; Hawkins, S.; Farrar, K.; Clifton-Brown, J.C.; Donnison, I.S.; Slavov, G.T. Genetic relationships between spring emergence, canopy phenology, and biomass yield increase the accuracy of genomic prediction in Miscanthus. J. Exp. Bot. 2017, 68, 5093–5102. [Google Scholar] [CrossRef]
- Slavov, G.T.; Nipper, R.; Robson, P.; Farrar, K.; Allison, G.G.; Bosch, M.; Clifton-Brown, J.C.; Donnison, I.S.; Jensen, E. Genome-wide association studies and prediction of 17 traits related to phenology, biomass and cell wall composition in the energy grass Miscanthus sinensis. New Phytol. 2014, 201, 1227–1239. [Google Scholar] [CrossRef]
- Clark, L.V.; Dwiyanti, M.S.; Anzoua, K.G.; Brummer, J.E.; Ghimire, B.K.; Głowacka, K.; Hall, M.; Heo, K.; Jin, X.; Lipka, A.E. Genome-wide association and genomic prediction for biomass yield in a genetically diverse Miscanthus sinensis germplasm panel phenotyped at five locations in Asia and North America. GCB Bioenergy 2019. [Google Scholar] [CrossRef]
- Slavov, G.; Davey, C.; Bosch, M.; Robson, P.; Donnison, I.; Mackay, I. Genomic index selection provides a pragmatic framework for setting and refining multi-objective breeding targets in Miscanthus. Ann Bot-Lond. 2018. [Google Scholar] [CrossRef]


| Phenotyping Schema Name | Use | Measurement Level | Protocol | When Performed |
|---|---|---|---|---|
| SurvivingPlants_pc | To compare establishment and overwintering survival. | plot/quadrat | 100 × Surviving plant count divided by planted plants. | July after planting and October (year 1) and following June (year 2) |
| EHeight_cm | Used to accurately calculate the start of growth. | plant | Measure from first emergence, from soil level to shoot tip/leaf tip. | Weekly March to May (until longest shoot is >40 cm). |
| FrostDamagePlantParts_0–9 | To assess leaf/canopy damage by frosts occurring after spring emergence. | plant | 0–9 scale of damage to plant part e.g. leaf where 0 is undamaged. | Weekly March to June. |
| FrostDamage PlantsinPlot_pc | No. of plants with score of 8, based on above frost score, to assess season start and stop. | plot | Using quadrat of 16 plants with 7 plants affected; 100 × 7/16 = 44%. | Weekly March to June. |
| CanopyHeight_cm | To compare morphology and create growth curves for models. | plant/plot/ quadrat | From ground to where most of the leaves bend to create a canopy. | Monthly, or higher frequency if resources allow. |
| TotalStems_ countperplant | To compare plant morphologies contributing to yield & quality. | plant | Count all stems over 10 cm. | Late autumn (normally October) |
| BigStems_countperplant | To compare plant morphologies making yield and quality. | plant | Count the number of stems which reach 60% of the CanopyHeight. For spreading plants count within 0.5 m2 (use a frame of 71 × 71 cm). | Late autumn (normally October) |
| Greenness_0–9 | To compare senescence from drought, cold and nutrient deficiency. This helps interpret G × E effects. | plant/plot/ quadrat | Visual score where 9 = all leaves dark green, 5 = half of canopy green, 1= totally brown. | When environmental stress events result in differences. At least in late autumn and again just before spring harvest. |
| FloweringScore_0–5 | This visual score helps identify the window of flowering to understand cross pollination possibilities of different hybrids, resultant seed set, shattering and establishment of volunteer seedlings. | plant/plot/ quadrat | Flowering Score (FS) 0-no flowering 1-Flag leaf visible 2-Panicle > 1 cm 3-Start of anthesis (yellow anthers) 4-Main flowering period (flowering intensity >= 50% of ‘big’ stems that contribute to the canopy) 5-Flowering complete (no anthesis on any stems). | Monthly from July and at higher frequencies once flowering has begun. |
| PlantBasal Diameter_cm | To assess plant maturity and or a measure of potential for invasion by rhizome creep. | plant | The diameter of the ‘tuft’ at plant base. | Late autumn, or just after spring harvest. |
| PanicleHeight_ cm | To understand morphological differences. | plant/plot/ quadrat | The height from the ground to the base of the panicle of the tallest flowering stem. | Late autumn when growth has ceased. |
| Light Interception_pc | Assessing when and to what extend the leaf canopy is intercepting available incident light. | plot/quadrat | The light intercepted by the canopy as measured by a ‘line ceptometer’. 0% no interception. >95% full canopy closure. | March to October. |
| Phenotyping Schema Name | Use | Protocol |
|---|---|---|
| QuadratArea_m2 | Yield assessments are based on sampling area - here called a ‘QuadratArea’. | Minimum sample area for Miscanthus is 6 m2. In practice quadrats may not be used and the harvested area is calculated from the number of rows and columns of plants harvested in a plot and the plant density. |
| FWQuadrat _kg | Bulk ‘fresh weight’ from the ‘QuadratArea_m2’ harvested. | The fresh weight in kg of the stems cut within the QuadratArea_m2 at a cutting height of 5 cm above ground level. Often measured immediately ‘on field’ using a tripod and hanging balance method. |
| FWSS_g | Fresh Weight of a subsample to calculate the moisture content and dry matter yield; samples may be used for compositional analysis. | A typical subsample is ~200 g fresh weight of randomly selected whole stems, or from well mixed chip. FWSS_g should be determined as quickly as possible after the FWQuadrat_kg to avoid moisture losses. |
| DWSS_g | Needed to calculate the moisture content and dry matter yield. | Oven dry subsample to constant weight, at 105 °C is most reliable, but lower temperatures are acceptable, as long as drying is complete, for certain compositional analysis. |
| Moisture Content_pc | MoistureContent_pc is a key biomass quality characteristic. | = 100 × (FWSS_g − DWSS_g)/ FWSS_g. |
| QuadratDW _kg | QuadratDW_kg is an intermediate in upscaling measurement of yield. | = FWQuadrat_kg − FWQuadrat_kg × (MoistureContent_pc/100) |
| Yield_tDWperha | Expressed as tonnes dry matter per ha, or in SI units ‘Mg ha−1’. | = (10,000/QuadratArea_m2 × QuadratDW_kg)/1000 |
| FWLeafSS_g | Improves yield assessment by calculating ‘leaf share’. | Quantify leaf lamina, not leaf sheath which is difficult to separate from the stems. Should be determined as quickly as possible after the FWQuadrat_kg. |
| DWLeafSS_g | Improves yield assessment by calculating ‘leaf share’. | FWLeafSS is dried to constant weight at 105 °C. 40–60 °C may be used if carbohydrate analysis is required. |
| MoistureContent Leaf_pc | A key biomass quality characteristic. | = 100 × (FWLeafSS_g − DWLeafSS_g)/ FWLeafSS_g. |
| FWStemSS_g | To assess proportion of yield due to stem. | Includes stems (culm) and leaf sheath; measured as soon as possible after the FWQuadrat_kg to increase accuracy of moisture content data. |
| DWStemSS_g | To assess proportion of yield due to stem. | The FWStemSS is dried to constant weight at temperatures 105 °C. 40–60 °C is recommended for analysis of cell wall carbohydrates (lignin and cellulose). |
| MoistureContent Stem_pc | To assess proportion of yield due to stem. | = 100 × (FWStemSS_g − DWStemSS_g)/FWStemSS_g |
| LeafBiomass Proportion_pc | To assess proportion of yield due to leaf. | = 100 × (DWLeafSS_g/(DWLeafSS_g + DWStemSS_g)) |
| StemBiomass Proportion_pc | To assess proportion of yield due to stem. | = 100*(DWStemSS_g/(DWLeafSS_g + DWStemSS_g)) |
| StemBasal Diameter_mm | Impacts senescence rate and moisture content, harvesting and baling efficiency. | Measure the basal diameter of ~five randomly selected stems. |
| StemWallAt Base_mm | Impacts senescence rate and moisture content, harvesting and baling efficiency. | Measure at base of ~five randomly selected stems. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Clifton-Brown, J.; Schwarz, K.-U.; Awty-Carroll, D.; Iurato, A.; Meyer, H.; Greef, J.; Gwyn, J.; Mos, M.; Ashman, C.; Hayes, C.; et al. Breeding Strategies to Improve Miscanthus as a Sustainable Source of Biomass for Bioenergy and Biorenewable Products. Agronomy 2019, 9, 673. https://doi.org/10.3390/agronomy9110673
Clifton-Brown J, Schwarz K-U, Awty-Carroll D, Iurato A, Meyer H, Greef J, Gwyn J, Mos M, Ashman C, Hayes C, et al. Breeding Strategies to Improve Miscanthus as a Sustainable Source of Biomass for Bioenergy and Biorenewable Products. Agronomy. 2019; 9(11):673. https://doi.org/10.3390/agronomy9110673
Chicago/Turabian StyleClifton-Brown, John, Kai-Uwe Schwarz, Danny Awty-Carroll, Antonella Iurato, Heike Meyer, Jörg Greef, Jeff Gwyn, Michal Mos, Christopher Ashman, Charlotte Hayes, and et al. 2019. "Breeding Strategies to Improve Miscanthus as a Sustainable Source of Biomass for Bioenergy and Biorenewable Products" Agronomy 9, no. 11: 673. https://doi.org/10.3390/agronomy9110673
APA StyleClifton-Brown, J., Schwarz, K.-U., Awty-Carroll, D., Iurato, A., Meyer, H., Greef, J., Gwyn, J., Mos, M., Ashman, C., Hayes, C., Huang, L., Norris, J., Rodgers, C., Scordia, D., Shafiei, R., Squance, M., Swaller, T., Youell, S., Cosentino, S., ... Robson, P. (2019). Breeding Strategies to Improve Miscanthus as a Sustainable Source of Biomass for Bioenergy and Biorenewable Products. Agronomy, 9(11), 673. https://doi.org/10.3390/agronomy9110673







