Impact of Climatic Variables on Carbon Content in Sugar Beet Root
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field Trials
2.2. Analyses
2.3. Statistics
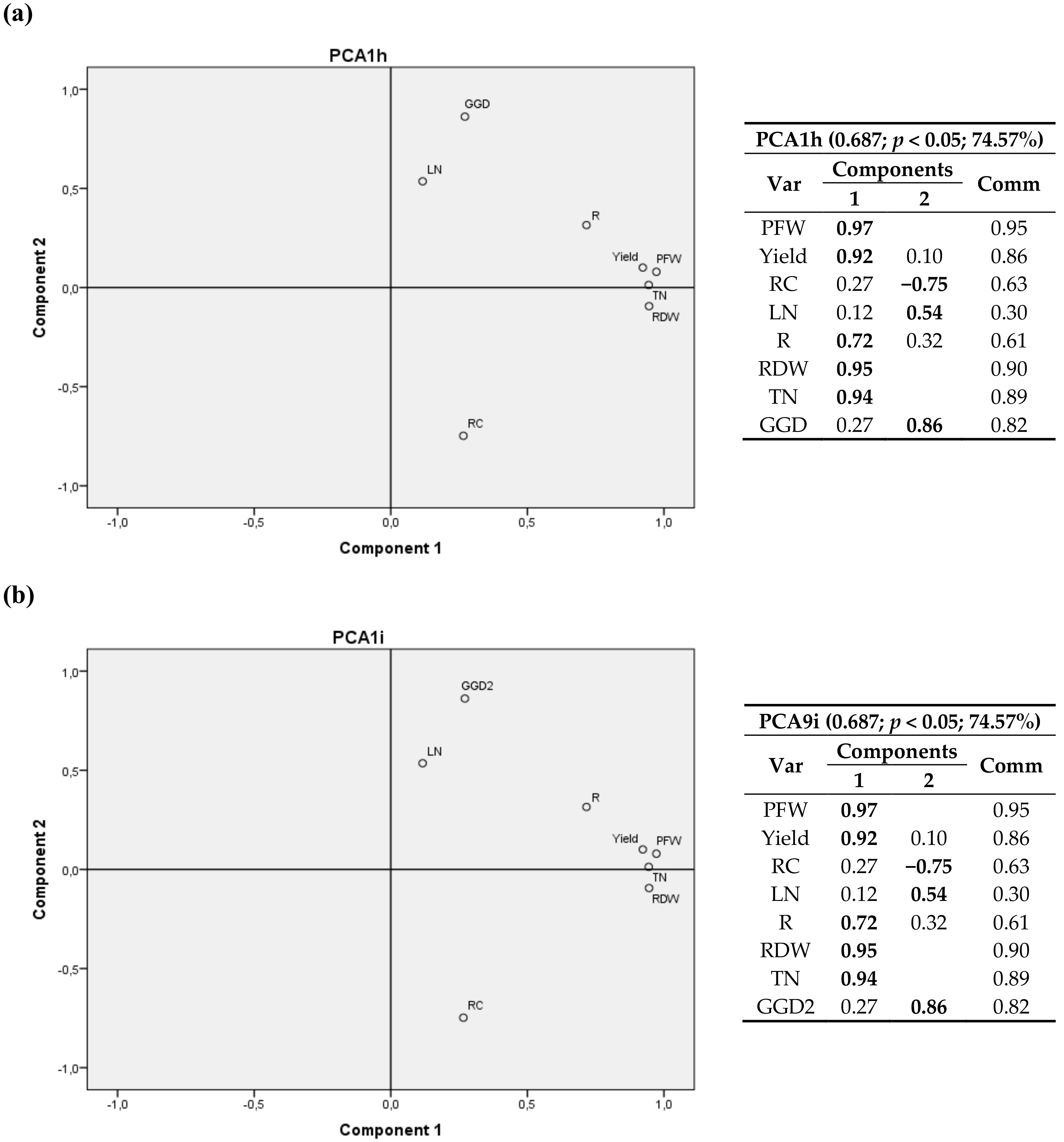
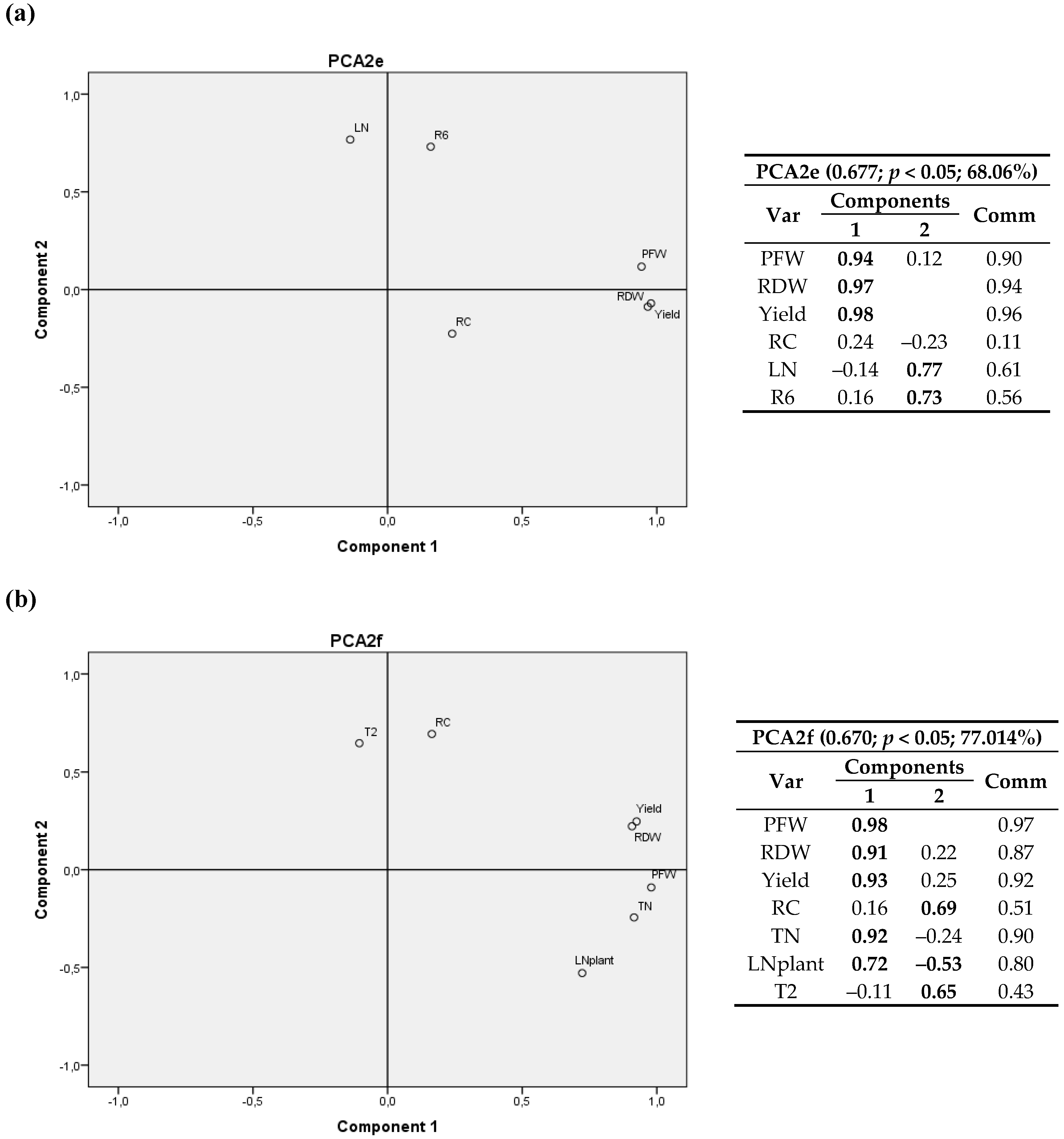
3. Results
3.1. Plant Variables at Harvest
3.1.1. Fresh Weight and Yield
3.1.2. Root Carbon Content, Dry Matter and CO2
3.1.3. Nitrogen Content in the Leaves, Total Nitrogen Absorption and Carbon to Nitrogen Ratio
3.1.4. Tukey’s Test
3.2. Climatic Variables
3.2.1. First Cultivation Year
3.2.2. Second Cultivation Year
4. Discussion
4.1. Plant Variables at Harvest
4.1.1. Fresh Weight and Yield
4.1.2. Carbon Content in the Roots, Dry Matter and CO2
4.1.3. Nitrogen Content in Leaves, Total Nitrogen Absorption and Carbon to Nitrogen Ratio
4.2. Climatic Variables
4.2.1. First Cultivation Year
4.2.2. Second Cultivation Year
4.3. Practical Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Smith, P.; Bustamante, M.; Ahammad, H.; Clark, H.; Dong, H.; Elsiddig, E.A.; Haberl, H.; Harper, R.; House, J.; Jafari, M.; et al. Agriculture, forestry and other land use (AFOLU). In Climate Change 2014: Mitigation of Climate Change. Contribution of Working Group III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Edenhofer, O., Pichs-Madruga, R., Sokona, Y., Farahani, E., Kadner, S., Seyboth, K., Adler, A., Baum, I., Brunner, S., Eickemeier, P., et al., Eds.; Cambridge University Press: Cambridge, UK, 2014; pp. 811–922. [Google Scholar]
- Smith, P.; Martino, D.; Cai, Z.; Gwary, D.; Janzen, H.; Kumar, P.; McCarl, B.; Ogle, S.; O’Mara, F.; Rice, C.; et al. Agriculture. In Climate Change 2007, Mitigation of Climate Change WGIII—IPCC Fourth Assessment Report; Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2007; pp. 498–540. [Google Scholar]
- Taiz, L.; Zeiger, E.; Moller, I.M.; Murphy, A. Plant Physiology, 6th ed.; Sinauer Associates Inc.: Sunderland, MA, USA, 2015. [Google Scholar]
- McKendry, P. Energy production from biomass (part 1): Overview of biomass. Bioresour. Technol. 2002, 83, 37–46. [Google Scholar] [CrossRef]
- Carvajal, M.; Mota, C.; Alcaraz-López, C.; Iglesias, M.; Martínez-Ballesta, M.C. Investigación Sobre la Absorción de CO2 por los Cultivos más Representativos; Consejo Superior de Investigaciones Científicas: Murcia, Spain, 2009. [Google Scholar]
- Gardner, F.P.; Pearce, R.B.; Mitchell, R.L. Carbon fixation by crop canopies. In Physiology of Crop Plants; Iowa State University Press: Iowa, IA, USA, 1985; pp. 31–57. [Google Scholar]
- Intergovernmental Panel on Climate Change (IPCC). Climate Change 2014: Synthesis Report. In Fifth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2014; p. 151. [Google Scholar]
- Victoria Jumilla, F.; Costa Gómez, I.; Castro Corbalán, T.; García Cárdenas, R.; Romojaro Casado, M.C.; Mesa del Castillo Navarro, M.L.; Motos Alarcón, M.I. La Iniciativa de Ecorresponsabilidad Agricultura Murciana como Sumidero de CO2. Marca LESSCO2; Observatorio Regional de Cambio Climático, Región de Murcia: Murcia, Spain, 2011. [Google Scholar]
- Klenk, I.; Landquist, B.; Ruiz de Imaña, O. The product carbon footprint of EU beet sugar. Summary of key findings. Sugar Ind. J. 2012, 137, 169–177. [Google Scholar]
- Crutzen, P.J.; Mosier, A.R.; Smith, K.A.; Winiwarter, W. N2O release from agro-biofuel production negates global warming reduction by replacing fossil fuels. Atmos. Chem. Phys. 2008, 8, 389–395. [Google Scholar] [CrossRef] [Green Version]
- Jaggard, K.W.; Clark, C.J.A.; Draycott, A.P. The weight and processing quality of components of the storage roots of sugar beet (Beta vulgaris L). J. Sci. Food Agric. 1999, 79, 1389–1398. [Google Scholar] [CrossRef]
- Milford, G.F.J.; Houghton, B.J. An analysis of the variation in crown size in sugar-beet (Beta vulgaris) grown in England. Ann. Appl. Biol. 1999, 134, 225–232. [Google Scholar] [CrossRef]
- Van Heemst, H.D.J. Physiological principles. In Modelling of Agricultural Production: Weather, Soils and Crops; van Keulen, H., Wolf, J., Eds.; Pudoc: Wageningen, The Netherlands, 1986; pp. 13–26. [Google Scholar]
- Scott, R.; Jaggard, K. Crop physiology and agronomy. In The Sugar Beet Crop; Springer: Dordrecht, The Netherlands, 1993; pp. 179–237. [Google Scholar]
- Kenter, C.; Hoffmann, C.M.; Märländer, B. Effects of weather variables on sugar beet yield development (Beta vulgaris L.). Eur. J. Agron. 2006, 24, 62–69. [Google Scholar] [CrossRef]
- Echevarría Ruiz de Vargas, C.; Feria Bourrellier, A.B.; Terencio Jiménez, E. Conceptos generales del metabolismo y del transporte de sacarosa. In Aspectos Fisiológicos de la Remolacha de Siembra Otoñal; Junta de Andalucia, Consejeria de Agricultura y Pesca: Sevilla, Spain, 2005; pp. 11–22. [Google Scholar]
- Gordo-Ingelmo, L.F. Composición Química y Control Agrícola de los no-Azúcares en la Remolacha Azucarera; Caja de Ahorros Municipal de Burgos: Burgos, Spain, 1994; p. 205. [Google Scholar]
- Jiménez, E.T.; García-Mauriño, S.; Morillo-Velarde, R.; Echevarría, C. Actividades enzimáticas de degradación de la sacarosa producción de azúcares reductores en la remolacha de siembra otoñal. In Aspectos Fisiológicos de la Remolacha de Siembra Otoñal; Junta de Andalucia, Consejeria de Agricultura y Pesca: Sevilla, Spain, 2005; pp. 45–69. [Google Scholar]
- Thomas, T.H. Sugar beet. In Crop Yield: Physiology and Processes; Smith, D.L., Hamel, C., Eds.; Springer: Berlin, Germany, 2000; pp. 311–332. [Google Scholar]
- Milford, G.F.J. The growth and development of the storage root of sugar beet. Ann. Appl. Biol. 1973, 75, 427–438. [Google Scholar] [CrossRef]
- Bell, C.I.; Milford, G.F.J.; Leigh, R.A. Sugar beet. In Photoassimilate Distribution in Plants and Crops: Source-Sink Relationships; Zamski, E., Schaffer, A.A., Eds.; Marcel Dekker Inc.: New York, NY, USA, 1996; Volume 48, pp. 691–707. [Google Scholar]
- Demmers-Derks, H.; Mitchell, R.A.C.; Mitchell, V.J.; Lawlor, D.W. Response of sugar beet (Beta vulgaris L.) yield and biochemical composition to elevated CO2 and temperature at two nitrogen applications. Plant Cell Environ. 1998, 21, 829–836. [Google Scholar] [CrossRef]
- Burkart, S.; Manderscheid, R.; Weigel, H.J. Canopy CO2 exchange of sugar beet under different CO2 concentrations and nitrogen supply: Results from a free-air CO2 enrichment study. Plant Biol. 2009, 11, 109–123. [Google Scholar] [CrossRef] [PubMed]
- Manderscheid, R.; Pacholski, A.; Weigel, H.-J. Effect of free air carbon dioxide enrichment combined with two nitrogen levels on growth, yield and yield quality of sugar beet: Evidence for a sink limitation of beet growth under elevated CO2. Eur. J. Agron. 2010, 32, 228–239. [Google Scholar] [CrossRef]
- Hoffmann, C.M.; Kluge-Severin, S. Light absorption and radiation use efficiency of autumn and spring sown sugar beets. Field Crop. Res. 2010, 119, 238–244. [Google Scholar] [CrossRef]
- Milford, G.F.J.; Pocock, T.O.; Riley, J. An analysis of leaf growth in sugar beet. I. Leaf appearance and expansion in relation to temperature under controlled conditions. Ann. Appl. Biol. 1985, 106, 163–172. [Google Scholar] [CrossRef]
- Villarías-Moradillo, J.L.; de Liñán y Vicente, C. La Remolacha Azucarera; Ediciones Agrotécnicas: Madrid, Spain, 1999. [Google Scholar]
- Hull, R.; Webb, D.J. The effect of sowing date and harvesting date on the yield of sugar beet. J. Agric. Sci. 1970, 75, 223. [Google Scholar] [CrossRef]
- Freckleton, R.P.; Watkinson, A.R.; Webb, D.J.; Thomas, T.H. Yield of sugar beet in relation to weather and nutrients. Agric. For. Meteorol. 1999, 93, 39–51. [Google Scholar] [CrossRef]
- Clover, G.R.G.; Jaggard, K.W.; Smith, H.G.; Azam-Ali, S.N. The use of radiation interception and transpiration to predict the yield of healthy, droughted and virus-infected sugar beet. J. Agric. Sci. 2001, 136, 169–178. [Google Scholar] [CrossRef]
- Gordo, L.F.; Morillo-Velarde, R.; Martínez, J.J.; Echevarría Ruiz de Vargas, C.; García-Mauriño, S. Crecimiento y desarrollo de la remolacha azucarera de siembra otoñal. In Aspectos Fisiológicos de la Remolacha de Siembra Otoñal; Junta de Andalucia; Consejeria de Agricultura y Pesca: Sevilla, Spain, 2005; pp. 23–44. [Google Scholar]
- Petkeviciene, B. The effects of climate factors on sugar beet early sowing timing. Agron. Res. 2009, 7, 436–443. [Google Scholar]
- Hoffmann, C.M.; Kluge-Severin, S. Growth analysis of autumn and spring sown sugar beet. Eur. J. Agron. 2011, 34, 1–9. [Google Scholar] [CrossRef]
- Loel, J.; Hoffmann, C.M. Importance of growth stage and weather conditions for the winter hardiness of autumn sown sugar beet. Field Crop. Res. 2014, 162, 70–76. [Google Scholar] [CrossRef]
- Jaggard, K.W.; Dewar, A.M.; Pidgeon, J.D. The relative effects of drought stress and virus yellows on the yield of sugarbeet in the UK, 1980–95. J. Agric. Sci. 1998, 130, 337–343. [Google Scholar] [CrossRef]
- Tanner, C.B.; Sinclair, T.R. Efficient water use in crop production: Research or re-search? In Limitations to Efficient Water Use in Crop Production; Taylor, H.M., Jordan, W.R., Sinclair, T.R., Eds.; American Society of Agronomy: Madison, WI, USA, 1983; pp. 1–27. [Google Scholar]
- Rinaldi, M.; Vonella, A.V. The response of autumn and spring sown sugar beet (Beta vulgaris L.) to irrigation in Southern Italy: Water and radiation use efficiency. Field Crop. Res. 2006, 95, 103–114. [Google Scholar] [CrossRef]
- Fabeiro, C.; Martín de Santa Olalla, F.; López, R.; Domínguez, A. Production and quality of the sugar beet (Beta vulgaris L.) cultivated under controlled deficit irrigation conditions in a semi-arid climate. Agric. Water Manag. 2003, 62, 215–227. [Google Scholar] [CrossRef]
- Asociación de investigación para la mejora del cultivo de la remolacha azucarera (AIMCRA). Plan 2020 para la sostenibilidad de la remolacha azucarera. Rev. AIMCRA 2015, 120, 29–31. [Google Scholar]
- Malnou, C.S.; Jaggard, K.W.; Sparkes, D.L. Nitrogen fertilizer and the efficiency of the sugar beet crop in late summer. Eur. J. Agron. 2008, 28, 47–56. [Google Scholar] [CrossRef]
- Grzebisz, W.; Szczepaniak, W.; Pepliński, K.; Barłóg, P.; Cyna, K. Impact of nitrogen concentration variability in sugar beet plant organs throughout the growing season on dry matter accumulation patterns. J. Elemntol. 2012. [Google Scholar] [CrossRef]
- Malnou, C.S.; Jaggard, K.W.; Sparkes, D.L. A canopy approach to nitrogen fertilizer recommendations for the sugar beet crop. Eur. J. Agron. 2006, 25, 254–263. [Google Scholar] [CrossRef]
- Kiymaz, S.; Ertek, A. Yield and quality of sugar beet (Beta vulgaris L.) at different water and nitrogen levels under the climatic conditions of Kırsehir, Turkey. Agric. Water Manag. 2015, 158, 156–165. [Google Scholar] [CrossRef]
- Giaquinta, R.T. Sucrose translocation and storage in the sugar beet. Plant Phys. 1979, 63, 828–832. [Google Scholar] [CrossRef]
- Hoffmann, C.M. Changes in N composition of sugar beet varieties in response to increasing N supply. J. Agron. Crop Sci. 2005, 191, 138–145. [Google Scholar] [CrossRef]
- Hoffmann, C.M.; Kenter, C.; Bloch, D. Marc concentration of sugar beet (Beta vulgaris L) in relation to sucrose storage. J. Sci. Food Agric. 2005, 85, 459–465. [Google Scholar] [CrossRef]
- Ma, S.; He, F.; Tian, D.; Zou, D.; Yan, Z.; Yang, Y.; Zhou, T.; Huang, K.; Shen, H.; Fang, J. Variations and determinants of carbon content in plants: A global synthesis. Biogeosciences 2018, 15, 693–702. [Google Scholar] [CrossRef]
- Ministerio de Agricultura, Pesca y Alimentación (MAGRAMA). Available online: www.magrama.gov/agricultura/ (accessed on 15 August 2015).
- Boiffin, J.; Durr, C.; Fleury, A.; Marin-Laflèche, A.; Maillet, I. Analysis of the variability of sugar beet (Beta vulgaris L.) growth during the early stages. I. Influence of various conditions on crop establishment. Agronomie 1992, 12, 515–525. [Google Scholar] [CrossRef]
- Derscheid, L.A.; Lytle, W.F. Growing Degree Days (GDD); Cooperative Extension Service, South Dakota State University: Brookings, SD, USA, 1981; p. 4. [Google Scholar]
- Milford, G.F.J.; Pocock, T.O.; Jaggard, K.W.; Biscoe, P.V.; Armstrong, M.J.; Last, P.J.; Goodman, P.J. An analysis of leaf growth in sugar beet. IV. The expansion of the leaf canopy in relation to temperature and nitrogen. Ann. Appl. Biol. 1985, 107, 335–347. [Google Scholar] [CrossRef]
- Asociación de Investigación para la Mejora del Cultivo de la Remolacha Azucarera (AIMCRA). Recomendaciones de Cultivo: Zona Norte. Available online: http://www.aimcra.es/Recomendaciones/RecomendacionesNorte.aspx (accessed on 14 January 2015).
- Kenter, C.; Hoffmann, C.M. Seasonal patterns of sucrose concentration in relation to other quality parameters of sugar beet (Beta vulgaris L.). J. Sci. Food Agric. 2006, 86, 62–70. [Google Scholar] [CrossRef]
- O'Rourke, N.; Psych, R.; Hatcher, L. A Step-by-Step Approach to Using SAS for Factor Analysis and Structural Equation Modeling, 2nd ed.; SAS Institute: Cary, NC, USA, 2013. [Google Scholar]
- Pérez López, C. Técnicas de Análisis Multivariante de Datos: Aplicaciones Con SPSS; Pearson Educación: Madrid, Spain, 2004. [Google Scholar]
- Harrigan, G.G.; Goodacre, R. Metabolic Profiling: Its Role in Biomarker Discovery and Gene Function Analysis; Springer: New York, NY, USA, 2003; p. 335. [Google Scholar]
- Asociación de investigación para la mejora del cultivo de la remolacha azucarera (AIMCRA). Memoria Campaña 2012/2013 Siembra Primavera 2012; AIMCRA: Valladolid, Spain, 2012; p. 124. [Google Scholar]
- Abdel-Motagally, F.M.; Attia, K.K. Response of sugar beet plants to nitrogen and potassium fertilization in sandy calcareous soil. Int. J. Agric. Biol. 2009, 11, 695–700. [Google Scholar]
- Martínez Quesada, J.J.; Morillo Velarde, R.; Aguilera García, Y.; Infante Vázquez, J.M. Growth of sugar beet under limited nitrogen conditions. In Sugar Beet Growth and Growth Modelling. Advances in Sugar Beet Research; Institut International de Recherches Betteravieres: Brussels, Belgium, 2003; Volume 5, pp. 33–45. [Google Scholar]
- Allison, M.F.; Armstrong, M.J.; Jaggard, K.W.; Todd, A.D.; Milford, G.F.J. An analysis of the agronomic, economic and environmental effects of applying N fertilizer to sugarbeet (Beta vulgaris). J. Agric. Sci. 1996, 127, 475. [Google Scholar] [CrossRef]
- Monteith, J.L.; Moss, C.J. Climate and the efficiency of crop production in Britain [and discussion]. Philos. Trans. R. Soc. B Biol. Sci. 1977, 281, 277–294. [Google Scholar] [CrossRef]
- Shock, C.C.; Seddigh, M.; Saunders, L.D.; Stieber, T.D.; Miller, J.G. Sugarbeet nitrogen uptake and performance following heavily fertilized onion. Agron. J. 2000, 92, 10–15. [Google Scholar] [CrossRef]
- Pocock, T.O.; Milford, G.F.J.; Armstrong, M.J. Storage root quality in sugarbeet in relation to nitrogen uptake. J. Agric. Sci. 2009, 115, 355. [Google Scholar] [CrossRef]
- Draycott, A.P.; Christenson, D.R. Nutrients for Sugarbeet Production: Soil-Plant Relationships; CABI Publishing: Wallingford, UK, 2003; p. 242. [Google Scholar]
- Ouda, M.M.S. Effect of nitrogen and sulphur fertilizers levels on sugar beet in newly cultivated sandy soil. Zagazig J. Agric. Res. 2002, 29, 33–50. [Google Scholar]
- Tabourel-Tayot, F.; Gastal, F. MecaNiCAL, a supply–demand model of carbon and nitrogen partitioning applied to defoliated grass: 2. Parameter estimation and model evaluation. Eur. J. Agron. 1998, 9, 243–258. [Google Scholar] [CrossRef]
- Dixon, R.K.; Solomon, A.M.; Brown, S.; Houghton, R.A.; Trexier, M.C.; Wisniewski, J. Carbon Pools and Flux of Global Forest Ecosystems. Science 1994, 263, 185–190. [Google Scholar] [CrossRef] [PubMed]
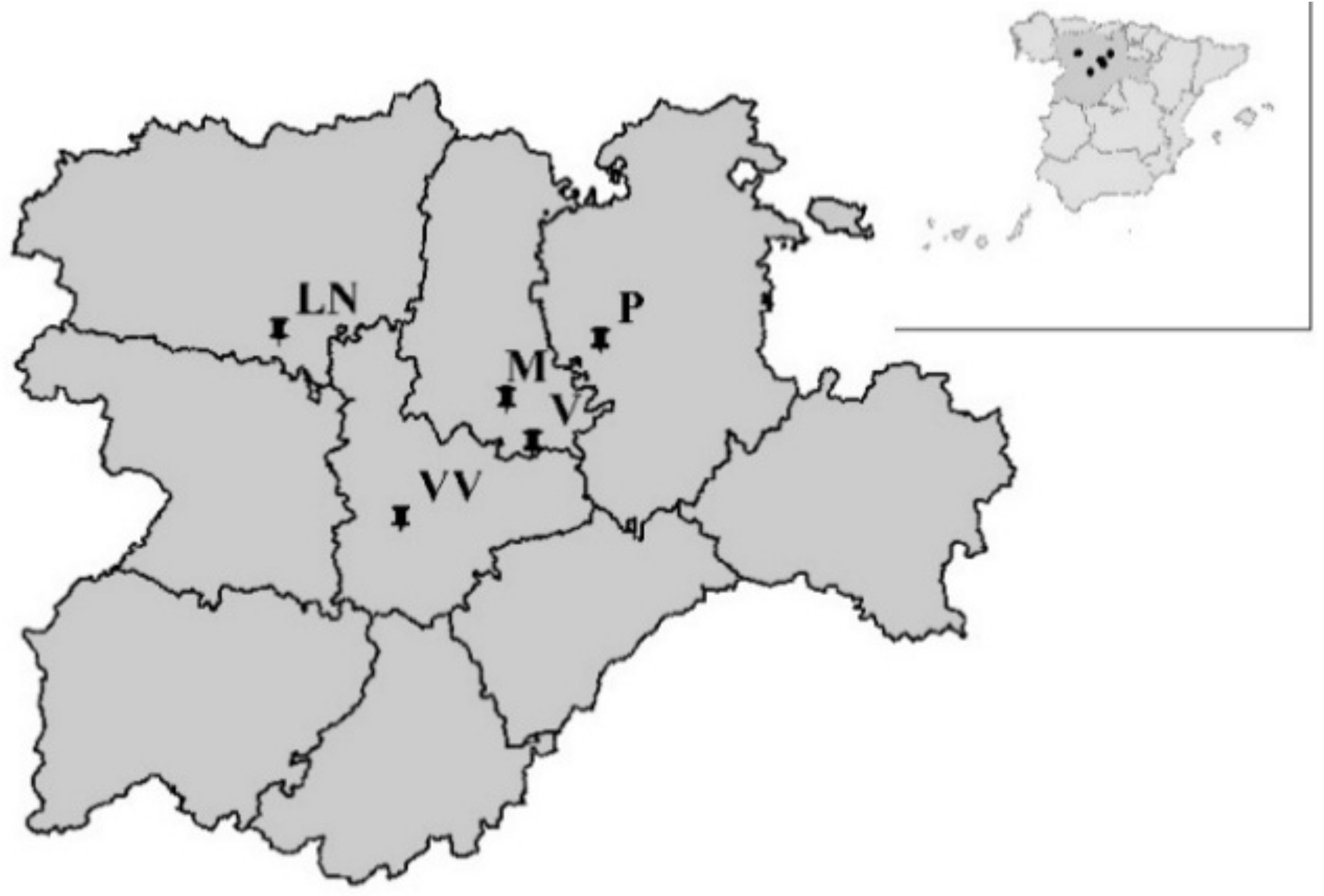
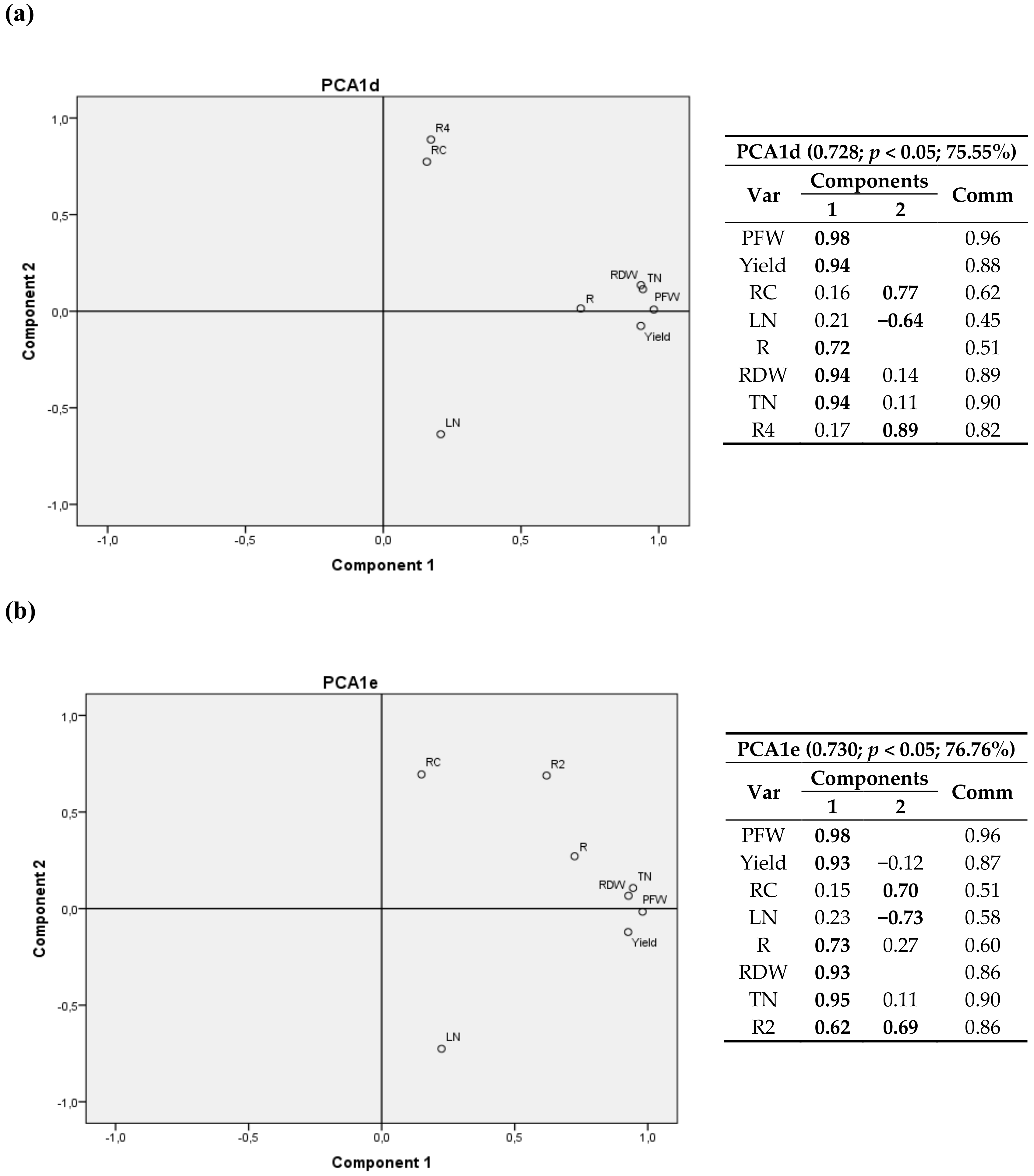
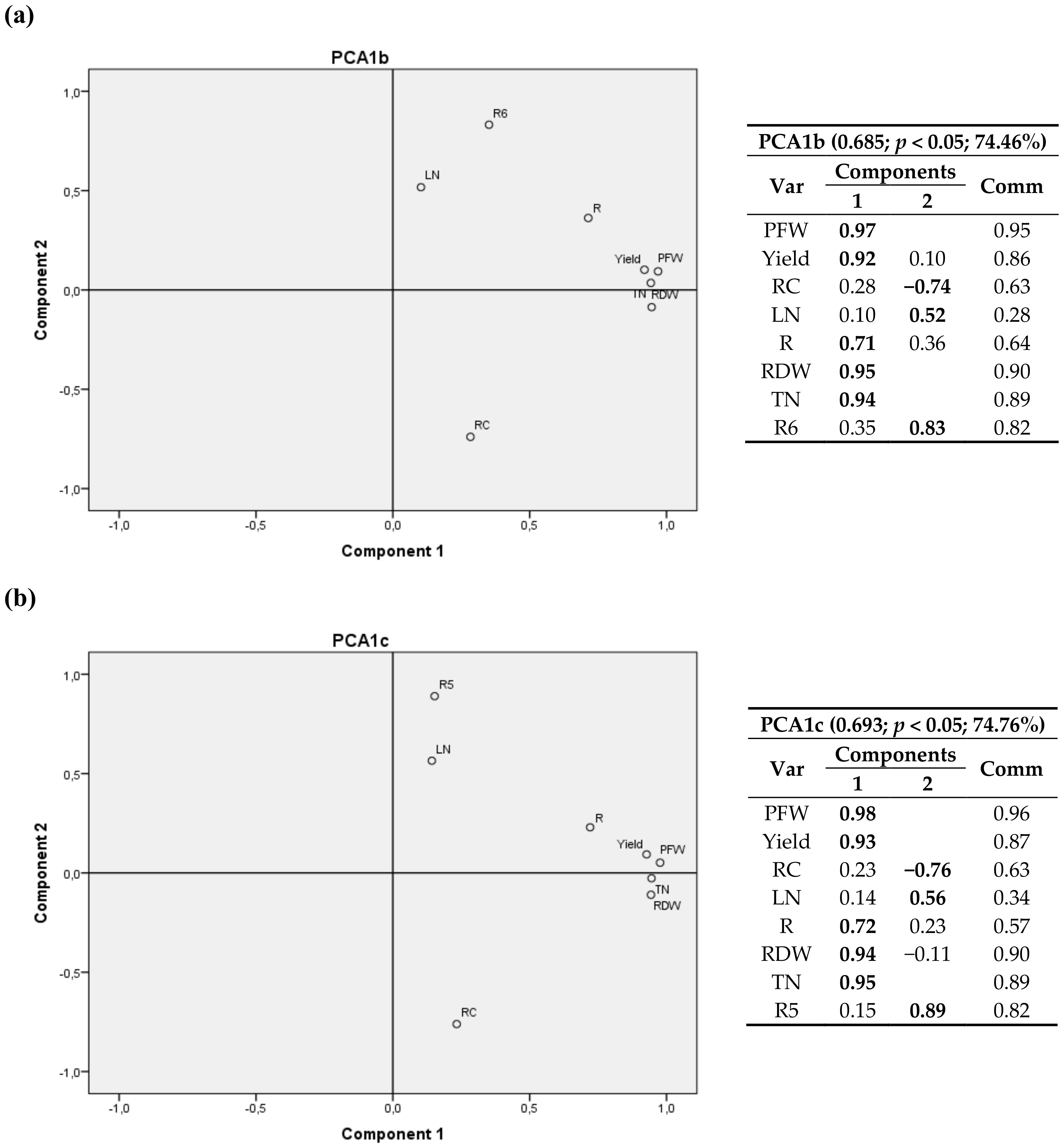


| Site | Sowing Date | Harvesting Date | Cultivation Period (d) |
|---|---|---|---|
| M | 23 March 2011 | 29 September 2011 | 190 |
| LN-I | 13 April 2011 | 10 October 2011 | 180 |
| VV-I | 10 March 2011 | 27 September 2011 | 201 |
| P | 16 March 2012 | 8 October 2012 | 206 |
| LN-II | 21 March 2012 | 10 October 2012 | 203 |
| V | 26 March 2012 | 3 October 2012 | 191 |
| VV-II | 5 March 2012 | 17 October 2012 | 226 |
| Site | Year | Coordinates | Meteorological Data (Cultivation Period) | Irrigation Period | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Latitude | Longitude | Altitude | T | P | ETo | R | ETo | Dose | ||
| Magaz (M) | 2011 | N 41° 59′ 24′′ | W 04° 24′ 36′′ | 737 | 17.0 | 100 | 792 | 4125 | 531 | 619 |
| Laguna de Negrillos–I (LN-I) | N 42° 13′ 56′′ | W 05° 37′ 37′′ | 782 | 16.8 | 182 | 805 | 4312 | 553 | 637 | |
| Villavieja-I (VV-I) | N 41° 29′ 59′′ | W 05° 03′ 53′′ | 674 | 16.9 | 134 | 872 | 4679 | 623 | 718 | |
| Pampliega (P) | 2012 | N 42° 12′ 26′′ | W 04° 00′ 02′′ | 764 | 15.5 | 178 | 941 | 4662 | 575 | 663 |
| Laguna de Negrillos-II (LN-II) | N 42° 15′ 47′′ | W 05° 07′ 57′′ | 778 | 15.5 | 199 | 898 | 4684 | 550 | 633 | |
| Vertavilo (V) | N 41° 50′ 52′′ | W 04° 19′ 23′′ | 785 | 16.0 | 147 | 798 | 3949 | 593 | 684 | |
| Villavieja-II (VV-II) | N 41° 31′ 39′′ | W 05° 01′ 30′′ | 710 | 15.8 | 100 | 908 | 4853 | 652 | 753 | |
| Variable | Description | Units |
|---|---|---|
| R1 | Accumulated radiation a.e. | MJ·m−2 |
| R2 | Accumulated radiation until 1200 °C·d a.s. | MJ·m−2 |
| R3 | Accumulated radiation after 1200 °C·d a.s. | MJ·m−2 |
| R4 | Accumulated radiation in the first 65 days a.s. | MJ·m−2 |
| R5 | Accumulated radiation in the last 25 days | MJ·m−2 |
| R6 | Accumulated radiation after day 175 a.s. | MJ·m−2 |
| T1 | Mean temperature during the first 65 days | °C |
| T2 | Mean temperature during the first 1200 °C·d a.s. | °C |
| T3 | Mean temperature deviation from 18 °C after 1200 °C·d a.s. | °C |
| GDD | Growth degree-day a.s. | °C·d |
| GDD1 | Growth degree-day a.e. | °C·d |
| GDD2 | Growth degree-day from 1200 °C·d | °C·d |
| Site | Soil Analysis | Fertilization | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Type | Texture | AW (%) | pH | SOM (%) | P (ppm) | K (ppm) | Mg (ppm) | Ca (ppm) | Na (ppm) | CO32− (%) | N (kg ha−1) | P2O5 (kg ha−1) | K2O (kg ha−1) | |
| 2011 | ||||||||||||||
| M | Entisol | Clay-Loam | 12.9 | 8.8 | 1.7 | 20 | 166 | 300 | 4570 | 71 | 24.3 | 140 | 125 | 100 |
| LN-I | Inceptisol | Loam | 11.2 | 6.7 | 1.1 | 25 | 137 | 80 | 950 | 35 | 1.1 | 180 | 75 | 0 |
| VV-I | Entisol | Loam | 11.3 | 8.5 | 1.2 | 19 | 162 | 590 | 2480 | 118 | 4.2 | 180 | 75 | 0 |
| 2012 | ||||||||||||||
| P | Entisol | Silty-Clay | 14.8 | 7.9 | 3.6 | 55 | 410 | 520 | 3710 | 58 | 15 | 110 | 84 | 28 |
| LN-II | Inceptisol | Sandy-Clay-Loam | 12.4 | 7.0 | 0.8 | 15 | 111 | 220 | 1100 | 29 | 0.8 | 183 | 70 | 0 |
| V | Inceptisol | Clay | 17.3 | 8.1 | 2.2 | 23 | 633 | 290 | 5220 | 13 | 25.3 | 155 | 208 | 92 |
| VV-II | Entisol | Sandy-Clay-Loam | 12.6 | 8.5 | 1.0 | 10 | 195 | 320 | 3610 | 35 | 13.9 | 180 | 115 | 0 |
| Fresh Matter | Dry Matter | ||||
| Variable | Description | Units | Variable | Description | Units |
| PFW | Plant fresh weight | g | PDW | Plant dry weight | g |
| RFW | Root fresh weight | g | RDW | Root dry weight | g |
| LFW | Leaves fresh weight | g | LDW | Leaves dry weight | g |
| CFW | Crown fresh weight | g | CDW | Crown dry weight | g |
| RTLR | Root to leaves ratio | - | RDM | Root dry matter content | g·kg−1 |
| Yield | Root fresh weight per ha | t·ha−1 | LDM | Leaves dry matter content | g·kg−1 |
| RBio | Root dry weight per ha | t·ha−1 | |||
| LBio | Leaves dry weight per ha | t·ha−1 | |||
| CBio | Crown dry weight per ha | t·ha−1 | |||
| PBio | Plant dry weight per ha | t·ha−1 | |||
| Carbon | Nitrogen | ||||
| Variable | Description | Units | Variable | Description | Units |
| RC | Root carbon content | g·kg−1 | RN | Root nitrogen content | g·kg−1 |
| LC | Leaves carbon content | g·kg−1 | LN | Leaves nitrogen content | g·kg−1 |
| CC | Crown carbon content | g·kg−1 | CN | Crown nitrogen content | g·kg−1 |
| RC/plant | Root carbon weight per plant | g | RN/plant | Root nitrogen weight per plant | g |
| LC/plant | Leaves carbon weight per plant | g | LN/plant | Leaves nitrogen weight per plant | g |
| CC/plant | Crown carbon weight per plant | g | CN/plant | Crown nitrogen weight per plant | g |
| C/plant | Total carbon per plant | g | TN/plant | Total nitrogen absorbed per plant | g |
| TC | Total carbon per ha | t·ha−1 | TN | Total nitrogen absorbed per ha | kg·ha−1 |
| CO2/plant | CO2 per plant | g | |||
| TCO2 | CO2 per ha | t·ha−1 | CNR | RC/LN | - |
| Var | Treatments | Effects | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Site | Variety | Fertilization | Site | Variety | Fertilization | S × F | |||||
| M | LN-I | VV-I | Dulzata | Sandrina | N | 2 × N | |||||
| PFW | 888.64 | 1377.69 | 1808.29 | 1288.21 | 1428.20 | 1252.67 | 1463.74 | *** (87.54) | n.s. | * (71.48) | n.s. |
| YIELD | 74.73 | 92.51 | 120.94 | 95.14 | 96.98 | 92.50 | 99.62 | *** (6.72) | n.s. | n.s. | n.s. |
| LFW | 182.24 | 499.19 | 524.29 | 351.69 | 452.12 | 343.41 | 460.40 | *** (25.426) | * (20.76) | *** (20.76) | *** (35.95) |
| RTLR | 3.58 | 1.68 | 1.99 | 2.72 | 2.11 | 2.76 | 2.07 | *** (0.14) | *** (0.121) | *** (0.121) | *** (0.209 |
| RC | 423.24 | 449.65 | 429.26 | 434.26 | 433.84 | 432.98 | 435.12 | *** (4.71) | n.s. | n.s. | n.s. |
| LN | 22.38 | 19.61 | 21.89 | 20.64 | 21.95 | 20.00 | 22.55 | *** (0.39) | ** (0.32) | *** (0.32) | *** (0.56) |
| CNR | 19.09 | 23.74 | 19.73 | 21.25 | 19.88 | 21.92 | 19.21 | *** (0.45) | * (0.367) | *** (0.37) | *** (0.64) |
| PDW | 168.09 | 266.20 | 332.77 | 244.47 | 266.91 | 243.47 | 267.90 | ***(0.47) | n.s | n.s | n.s |
| RDM | 198.70 | 251.26 | 220.35 | 220.05 | 226.81 | 224.99 | 221.87 | *** (3.42) | n.s. | n.s. | n.s. |
| TN | 195.43 | 386.09 | 500.14 | 345.15 | 375.95 | 325.46 | 395.64 | *** (22.92) | n.s. | * (18.71) | * (32.41) |
| TCO2 | 31.53 | 52.53 | 63.84 | 46.65 | 51.09 | 46.35 | 51.40 | *** (3.27) | n.s. | n.s. | n.s. |
| Var | Treatments | Effects | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Site | Variety | Site | Variety | S × V | |||||
| P | LN-II | V | VV-II | Amalia | Sandrina | ||||
| PFW | 1789.46 | - | 1534.67 | 1754.70 | 1579.32 | 1806.58 | n.s. | n.s. | n.s. |
| YIELD | 110.03 | 96.41 | 119.69 | 124.38 | 104.45 | 120.80 | * (7.47) | * (5.28) | n.s. |
| LFW | 639.52 | - | 367.92 | 482.26 | 465.29 | 527.83 | *** (31.27) | n.s. | *** (44.22) |
| RTLR | 1.43 | - | 2.83 | 2.23 | 2.11 | 2.22 | *** (0.153) | n.s. | *** (0.216) |
| RC | 423.51 | 446.25 | 441.00 | 439.10 | 437.12 | 437.81 | * (5.59) | n.s. | n.s. |
| LN | 22.46 | - | 19.18 | 20.41 | 19.99 | 21.38 | *** (0.47) | * (0.38) | n.s. |
| CNR | 19.02 | - | 23.34 | 21.97 | 22.01 | 20.73 | *** (0.59) | * (0.48) | n.s. |
| PDW | 352.23 | - | 314.98 | 330.01 | 317.14 | 347.14 | n.s. | n.s. | n.s. |
| RDM | 229.43 | 243.62 | 230.39 | 231.13 | 240.13 | 227.16 | * (3.81) | ** (2.69) | * (5.390) |
| TN | 494.02 | - | 370.72 | 362.79 | 381.71 | 436.64 | *** (24.597) | n.s. | n.s. |
| TCO2 | 69.42 | - | 62.37 | 65.98 | 62.85 | 68.99 | n.s. | n.s. | n.s. |
| Site | Cultivation Period (CP) and Calculated Meteorological Data | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Emergence | CP | R1 | R2 | R3 | R4 | R5 | R6 | T1 | T2 | T3 | GDD | GDD1 | GDD2 | |
| d (°C·d) | d | MJ·m−2 | MJ·m−2 | MJ·m−2 | MJ·m−2 | MJ·m−2 | MJ·m−2 | °C | °C | °C | °C·d | °C·d | °C·d | |
| 2011 | ||||||||||||||
| M | - | 190 | - | 1595 | 2530 | 1153 | 506 | 285 | 13.49 | 13.97 | 1.5 | 3249 | - | 2049 |
| LN-I | - | 180 | - | 1853 | 2459 | 1481 | 457 | 84 | 14.85 | 15.7 | −0.3 | 3048 | - | 1848 |
| VV-I | - | 201 | - | 1840 | 2839 | 1240 | 524 | 546 | 12.21 | 13.4 | 1.7 | 3412 | - | 2212 |
| 2012 | ||||||||||||||
| P | 21 (185) | 206 | 4297 | 2118 | 2544 | 1166 | 376 | 500 | 9.6 | 12.1 | 0.7 | 3207 | 3025 | 2007 |
| LN-II | 23 (205) | 203 | 4286 | 2100 | 2584 | 1279 | 363 | 428 | 10.58 | 12.7 | 0.1 | 3160 | 2955 | 1960 |
| V | 25 (210) | 191 | 3557 | 1873 | 2076 | 1183 | 447 | 207 | 10.49 | 12.9 | 1.2 | 2982 | 2772 | 1782 |
| VV-II | 22 (152) | 226 | 4460 | 2082 | 2771 | 1056 | 312 | 835 | 8.38 | 12 | 1.2 | 3593 | 3430 | 2393 |
| Site | Days | GDD (°C·d) | Temperature Gradient (Tmax−Tmin) |
|---|---|---|---|
| VV | 22 | 152 | 18.1° |
| P | 21 | 185 | 15.5° |
| LN | 23 | 205 | 14.6° |
| V | 25 | 210 | 14.1° |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Sastre, L.F.; Martín-Ramos, P.; Navas-Gracia, L.M.; Hernández-Navarro, S.; Martín-Gil, J. Impact of Climatic Variables on Carbon Content in Sugar Beet Root. Agronomy 2018, 8, 147. https://doi.org/10.3390/agronomy8080147
Sánchez-Sastre LF, Martín-Ramos P, Navas-Gracia LM, Hernández-Navarro S, Martín-Gil J. Impact of Climatic Variables on Carbon Content in Sugar Beet Root. Agronomy. 2018; 8(8):147. https://doi.org/10.3390/agronomy8080147
Chicago/Turabian StyleSánchez-Sastre, Luis F., Pablo Martín-Ramos, Luis M. Navas-Gracia, Salvador Hernández-Navarro, and Jesús Martín-Gil. 2018. "Impact of Climatic Variables on Carbon Content in Sugar Beet Root" Agronomy 8, no. 8: 147. https://doi.org/10.3390/agronomy8080147