Plant Desiccation Tolerance and its Regulation in the Foliage of Resurrection “Flowering-Plant” Species
Abstract
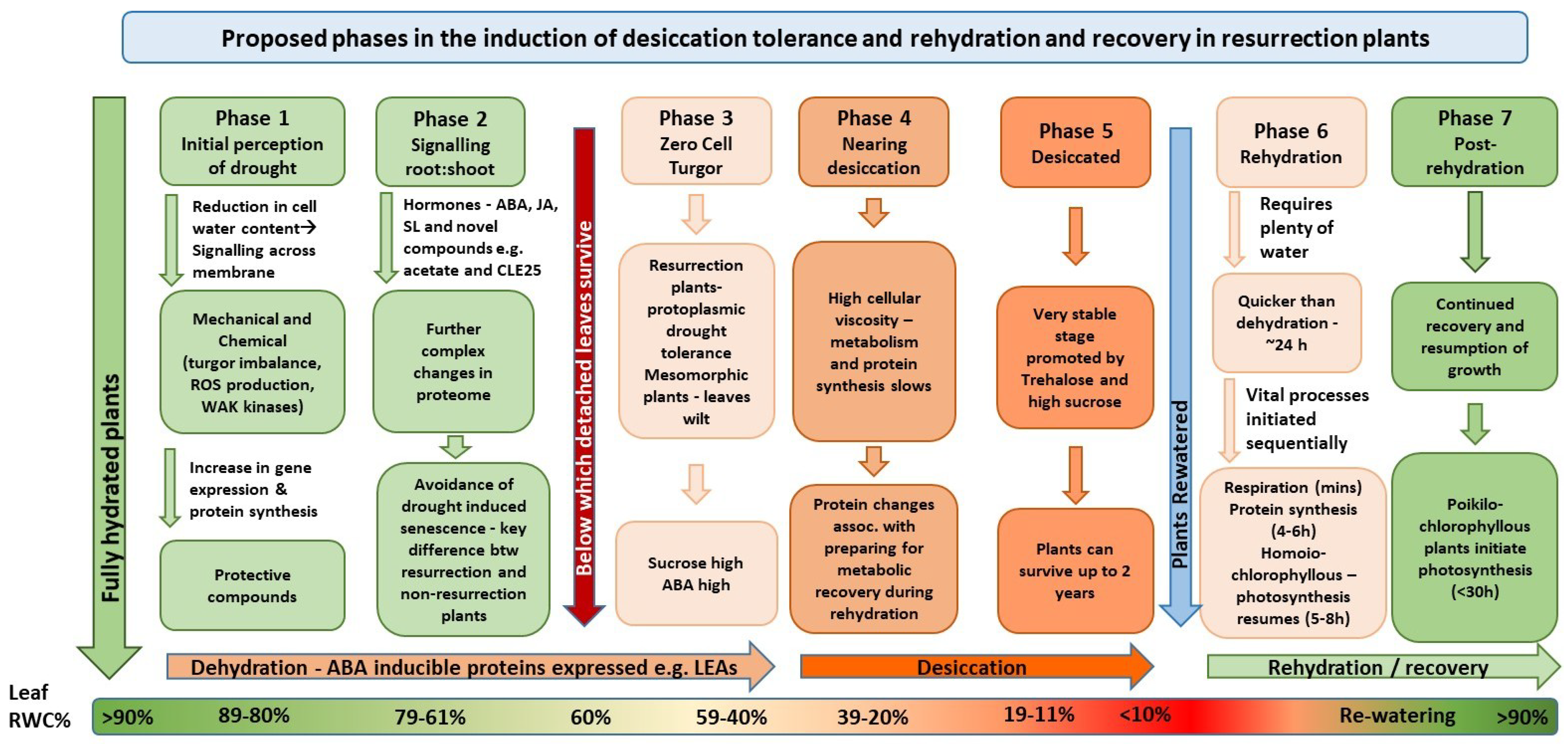
:1. Introduction
2. Single Basic Mechanism of Desiccation Tolerance in Seed, Pollen and Resurrection Foliage
3. Predisposing Factors for Evolution of Angiosperm Resurrection Species
4. Desiccation Tolerance in Plant Taxa That Preceded the Angiosperms
5. Regulation of the Induction of Desiccation Tolerance in Angiosperms
5.1. ABA Induction Pathway for Desiccation Tolerance
5.2. Transcription Factors and the ABA Induction Pathway for Desiccation Tolerance
6. Potential Regulators of Desiccation Tolerance Other Than ABA
6.1. Brassinosteroids (BR), Jasmonates (JA) and Strigolactones (SL)
6.2. Reactive Oxygen Species (ROS)
7. Sucrose and Induction of Desiccation Tolerance
8. The Role of Trehalose-6-Phospate in Desiccation Tolerance
9. Protein-Synthesis in Resurrection Plants under Drought Stress
Processes in Seed and Pollen during Desiccation
10. Identification of Genes Involved in Desiccation Tolerance
11. Direct DNA Manipulation of Desiccation-Related Genes
12. Stress-Induction of Senescence in Mature and Premature Leaves
13. Epigenetic Modifications
14. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Gaff, D.F.; Churchill, D.M. Borya nitida Labill.—An Australian species in the Liliaceae with desiccation-tolerant leaves. Aust. J. Bot. 1976, 24, 209–224. [Google Scholar] [CrossRef]
- Gaff, D.F. Desiccation tolerant vascular plants of Southern Africa. Oecologia 1977, 1, 95–109. [Google Scholar] [CrossRef] [PubMed]
- Gaff, D.F. The Biology of Resurrection Plants; Western Australia Press: Nedlands, Australia, 1981; pp. 114–146. [Google Scholar]
- Gaff, D.F. Desiccation tolerant ‘resurrection’ grasses from Kenya and West Africa. Oecologia 1986, 70, 118–120. [Google Scholar] [CrossRef] [PubMed]
- Gaff, D.F.; Latz, P.K. The occurrence of resurrection plants in the Australian flora. Aust. J. Bot. 1978, 26, 485–492. [Google Scholar] [CrossRef]
- Gaff, D.F.; Bole, P.V. Resurrection grasses in India. Oecologia 1986, 71, 159–160. [Google Scholar] [CrossRef] [PubMed]
- Gaff, D.F.; Ellis, R.P. Southern African grasses with foliage that revives after dehydration. Bothalia Afr. Biodivers. Conserv. 1974, 11, 305–308. [Google Scholar] [CrossRef]
- Iturriaga, G.; Gaff, D.F.; Zentella, R. New desiccation-tolerant plants, including a grass, in the central highlands of Mexico. Aust. J. Bot. 2000, 48, 153–158. [Google Scholar] [CrossRef]
- Gaff, D.F.; Sutaryono, Y.A.; Miszalski, Z. Resurrecting desiccation-tolerant resurrection grasses from dryland areas. Wiad. Bot. 1990, 34, 17–22. [Google Scholar]
- Gaff, D.F.; Oliver, M. The evolution of desiccation tolerance in angiosperm plants: A rare yet common phenomenon. Funct. Plant Biol. 2013, 40, 315–328. [Google Scholar] [CrossRef]
- Farrant, J.; Brandt, W.; Lindsey, G.G. An Overview of Mechanisms of Desiccation Tolerance in Selected Angiosperm Resurrection Plants; Plant Stress: Rondebosch, South Africa, 2007; pp. 72–84. [Google Scholar]
- Angelovici, R.; Galili, G.; Fernie, A.R.; Fait, A. Seed desiccation: A bridge between maturation and germination. Trends Plant Sci. 2010, 15, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Gechev, T.S.; Dinakar, C.; Benina, M.; Toneva, V.; Bartels, D. Molecular mechanisms of desiccation tolerance in resurrection plants. Cell. Mol. Life Sci. 2012, 69, 3175–3186. [Google Scholar] [CrossRef] [PubMed]
- Terrasson, E.; Buitink, J.; Righetti, K.; Ly Vu, B.; Pelletier, S.; Lalanne, D.; Zinsmeister, J.; Leprince, O. An emerging picture of the seed desiccome: Confirmed regulators and newcomers identified using transcriptome comparison. Front. Plant Sci. 2013, 4, 497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farrant, J.M.; Cooper, K.; Hilgart, A.; Abdalla, K.O.; Bentley, J.; Thomson, J.A.; Dace, H.J.W.; Peton, N.; Mundree, S.G.; Rafudeen, M.S. A molecular physiological review of vegetative desiccation tolerance in the resurrection plant Xerophyta viscosa (Baker). Planta 2015, 242, 407–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, A.; Wang, D.; Yu, B.; Yu, X.; Li, W. Maintenance or collapse: Responses of extraplastidic membrane lipid composition to desiccation in the resurrection plant Paraisometrum mileense. PLoS ONE 2014, 9, e103430. [Google Scholar] [CrossRef] [PubMed]
- Franchi, G.G.; Piotto, B.; Nepi, M.; Baskin, C.C.; Baskin, J.M.; Pacini, E. Pollen and seed desiccation tolerance in relation to degree of developmental arrest, dispersal, and survival. J. Exp. Bot. 2011, 62, 5267–5281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eyres, I.; Boschetti, C.; Crisp, A.; Smith, T.P.; Fontaneto, D.; Tunnacliffe, A.; Barraclough, T.G. Horizontal gene transfer in Bdelloid rotifers is ancient, ongoing and more frequent in species from desiccating habitats. BMC Biol. 2015, 13, 90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hilbricht, T.; Varotto, S.; Sgaramella, V.; Bartels, D.; Salamini, F.; Furini, A. Retrotransposons and siRNA have a role in the evolution of desiccation tolerance leading to resurrection of the plant Craterostigma plantagineum. New Phytol. 2008, 179, 877–887. [Google Scholar] [CrossRef] [PubMed]
- Royal Botanic Gardens, Kew. Seed Information Database (SID), version 7.1; Royal Botanic Gardens, Kew: Richmond, UK, 2008. [Google Scholar]
- Holman, R.M.; Brubaker, F. On the Longevity of Pollen; University of California Press: Oakland, CA, USA, 1926. [Google Scholar]
- Visser, T. Germination and Storage of Pollen. Ph.D. Thesis, Wageningen University & Research, Wageningen, The Netherlands, 1955. [Google Scholar]
- Evenari, M.; Shanan, L.; Tadmor, N. The Negev. The Challenge of a Desert; Harvard University Press: Cambridge, MA, USA, 1971. [Google Scholar]
- McFadden, G.I. Origin and evolution of plastids and photosynthesis in eukaryotes. Cold Spring Harb. Perspect. Biol. 2014, 6, a016105. [Google Scholar] [CrossRef] [PubMed]
- Gribaldo, S.; Poole, A.M.; Daubin, V.; Forterre, P.; Brochier-Armanet, C. The origin of eukaryotes and their relationship with the Archaea: Are we at a phylogenomic impasse? Nat. Rev. Microbiol. 2010, 8, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Rochette, N.C.; Brochier-Armanet, C.; Gouy, M. Phylogenomic test of the hypotheses for the evolutionary origin of eukaryotes. Mol. Biol. Evol. 2014, 31, 832–845. [Google Scholar] [CrossRef] [PubMed]
- Anderson, K.L.; Apolinario, E.E.; Sowers, K.R. Desiccation as a long-term survival mechanism for the Archaeon Methanosarcina barkeri. Appl. Environ. Microbiol. 2012, 78, 1473–1479. [Google Scholar] [CrossRef] [PubMed]
- Billi, D.; Potts, M. Life and death of dried prokaryotes. Res. Microbiol. 2002, 153, 7–12. [Google Scholar] [CrossRef]
- Garcia, A.H. Anhydrobiosis in bacteria: From physiology to applications. J. Biosci. 2011, 36, 939–950. [Google Scholar] [CrossRef] [PubMed]
- Stoll, M.; Loveys, B.; Dry, P. Hormonal changes induced by partial rootzone drying of irrigated grapevine. J. Exp. Bot. 2000, 51, 1627–1634. [Google Scholar] [CrossRef] [PubMed]
- Gaff, D.F. Protoplasmic tolerance of extreme stress. In Adaptation of Plants to Water and High Temperature Stress; Turner, N.C., Kramer, P.J., Eds.; John Wiley & Sons: New York, NY, USA, 1980; pp. 207–230. [Google Scholar]
- Hsiao, T.C.; Acevedo, E.; Fereres, E.; Henderson, D.W. Water stress, growth and osmotic adjustment. Philos. Trans. R. Soc. Lond. B 1976, 273, 479–500. [Google Scholar] [CrossRef]
- Sperry, J.S.; Tyree, M.T. Mechanism of water stress-induced xylem embolism. Plant Physiol. 1988, 88, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Christmann, A.; Weiler, E.W.; Steudle, E.; Grill, E. A hydraulic signal in root-to-shoot signalling of water shortage. Plant J. 2007, 52, 167–174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaff, D.F.; Loveys, B.R. Abscisic acid levels in drying plants of a resurrection grass. Trans. Malays. Soc. Plant Physiol. 1993, 3, 286–287. [Google Scholar]
- Blomstedt, C.K.; Griffiths, C.A.; Fredericks, D.P.; Hamill, J.D.; Gaff, D.F.; Neale, A.D. The resurrection plant Sporobolus stapfianus: An unlikely model for engineering enhanced plant biomass? Plant Growth Regul. 2010, 62, 217–232. [Google Scholar] [CrossRef]
- Giarola, V.; Krey, S.; Driesch, B.; Bartels, D. The Craterostigma plantagineum glycine-rich protein CpGRP1 interacts with a cell wall-associated protein kinase 1 (CpWAK1) and accumulates in leaf cell walls during dehydration. New Phytol. 2016, 210, 535–550. [Google Scholar] [CrossRef] [PubMed]
- Davis, F.S.; Merkle, M.G.; Bovey, R.W. Effect of moisture stress on the absorption and transport of herbicides in woody plants. Bot. Gaz. 1968, 129, 183–189. [Google Scholar] [CrossRef]
- Dinant, S.; Lemoine, R. The phloem pathway: New issues and old debates. C. R. Biol. 2010, 333, 307–319. [Google Scholar] [CrossRef] [PubMed]
- Kuang, J.; Gaff, D.; Gianello, R.; Blomstedt, C.; Neale, A.; Hamill, J. Changes in in vivo protein complements in drying leaves of the desiccation-tolerant grass Sporobolus stapfianus and the desiccation-sensitive grass Sporobolus pyramidalis. Funct. Plant Biol. 1995, 22, 1027–1034. [Google Scholar]
- Ismail, A.; Takeda, S.; Nick, P. Life and death under salt stress: Same players, different timing? J. Exp. Bot. 2014, 65, 2963–2979. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Bartels, D. Molecular responses to dehydration and desiccation in desiccation-tolerant angiosperm plants. J. Exp. Bot. 2018, 69, 3211–3222. [Google Scholar] [CrossRef] [PubMed]
- Bastien, C.; Aurélie, E.; Iris, S.; Bernhard, K.; Dorothea, B.; Shaun, P.; Stefan, H. Water deficit induces chlorophyll degradation via the ‘pao/phyllobilin’ pathway in leaves of homoio-(Craterostigma pumilum) and poikilochlorophyllous (Xerophyta viscosa) resurrection plants. Plant Cell Environ. 2014, 37, 2521–2531. [Google Scholar]
- Gaff, D.F.; Loveys, B.R. Abscisic acid content and effects during dehydration of detached leaves of desiccation tolerant plants. J. Exp. Bot. 1984, 35, 1350–1358. [Google Scholar] [CrossRef]
- Griffiths, C.; Gaff, D.; Neale, A. Drying without senescence in resurrection plants. Front. Plant Sci. 2014, 5, 36. [Google Scholar] [CrossRef] [PubMed]
- Ghasempour, H.R.; Anderson, E.M.; Gaff, D.F. Effects of growth substances on the protoplasmic drought tolerance of leaf cells of the resurrection grass Sporobolus stapfianus. Aust. J. Plant Physiol. 2001, 28, 1115–1120. [Google Scholar]
- Wright, S.T.C. Seasonal changes in the levels of free and bound abscisic acid in blackcurrant (Ribes nigrum) buds and beech (Fagus sylvatica) buds. J. Exp. Bot. 1975, 26, 161–174. [Google Scholar] [CrossRef]
- Bano, A.; Hansen, H.; Dörffling, K.; Hahn, H. Changes in the contents of free and conjugated abscisic acid, phaseic acid and cytokinins in xylem sap of drought stressed sunflower plants. Phytochemistry 1994, 37, 345–347. [Google Scholar] [CrossRef]
- Gaff, D.F.; McGregor, G.R. The effect of dehydration and re-hydration on the nitrogen content of various fractions from resurrection plants. Biol. Plant. 1979, 21, 92–99. [Google Scholar] [CrossRef]
- Bartels, D.; Schneider, K.; Terstappen, G.; Piatowski, D.F.S. Molecular cloning of abscisic acid-modulated genes which are induced during desiccation of the resurrection plant Craterostigma plantagineum. Planta 1990, 181, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Neale, A.; Blomstedt, C.; Bronson, P.; Le, T.N.; Guthridge, K.; Evans, J.; Gaff, D.; Hamill, J. The isolation of genes from the resurrection grass Sporobolus stapfianus which are induced during severe drought stress. Plant Cell Environ. 2000, 23, 265–277. [Google Scholar] [CrossRef]
- Huang, J.-C.; Lin, S.-M.; Wang, C.-S. A pollen-specific and desiccation-associated transcript in Lilium longiflorum during development and stress. Plant Cell Physiol. 2000, 41, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.-Y.; Chen, Y.-C.; Jauh, G.Y.; Wang, C.-S. A lily ASR protein involves abscisic acid signaling and confers drought and salt resistance in Arabidopsis. Plant Physiol. 2005, 139, 836–846. [Google Scholar] [CrossRef] [PubMed]
- Meurs, C.; Basra, A.S.; Karssen, C.M.; van Loon, L.C. Role of abscisic acid in the induction of desiccation tolerance in developing seeds of Arabidopsis thaliana. Plant Physiol. 1992, 98, 1484–1493. [Google Scholar] [CrossRef] [PubMed]
- Rushton, D.L.; Tripathi, P.; Rabara, R.C.; Lin, J.; Ringler, P.; Boken, A.K.; Langum, T.J.; Smidt, L.; Boomsma, D.D.; Emme, N.J.; et al. WRKY transcription factors: Key components in abscisic acid signalling. Plant Biotechnol. J. 2012, 10, 2–11. [Google Scholar] [CrossRef] [PubMed]
- Santiago, J.; Dupeux, F.; Betz, K.; Antoni, R.; Gonzalez-Guzman, M.; Rodriguez, L.; Márquez, J.A.; Rodriguez, P.L. Structural insights into PYR/PYL/RCAR ABA receptors and PP2Cs. Plant Sci. 2012, 182, 3–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, F.-Q.; Xin, Q.; Cao, Z.; Liu, Z.-Q.; Du, S.-Y.; Mei, C.; Zhao, C.-X.; Wang, X.-F.; Shang, Y.; Jiang, T.; et al. The magnesium-chelatase h subunit binds abscisic acid and functions in abscisic acid signaling: New evidence in Arabidopsis. Plant Physiol. 2009, 150, 1940–1954. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Hu, J.; Guo, S.; Wang, J.; Cheng, Y.; Dang, X.; Wu, L.; He, Y. Proteome analysis of Physcomitrella patens exposed to progressive dehydration and rehydration. J. Exp. Bot. 2012, 63, 711–726. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.C.D.; Farrant, J.M.; Oliver, M.J.; Ligterink, W.; Buitink, J.; Hilhorst, H.M.W. Key genes involved in desiccation tolerance and dormancy across life forms. Plant Sci. 2016, 251, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Finkelstein, R.; Gampala, S.S.; Lynch, T.J.; Thomas, T.L.; Rock, C.D. Redundant and distinct functions of the ABA response loci ABA-insensitive (ABI) 5 and abre-binding factor (ABF) 3. Plant Mol. Biol. 2005, 59, 253–267. [Google Scholar] [CrossRef] [PubMed]
- Hand, S.C.; Menze, M.A.; Toner, M.; Boswell, L.; Moore, D. LEA proteins during water stress: Not just for plants anymore. Annu. Rev. Physiol. 2011, 73, 115–134. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Kishitani, S.; Ito, Y.; Toriyama, K. Accumulation of raffinose in rice seedlings overexpressing OsWRKY11 in relation to desiccation tolerance. Plant Biotechnol. 2009, 26, 431–434. [Google Scholar] [CrossRef]
- Wang, Z.; Zhu, Y.; Wang, L.; Liu, X.; Liu, Y.; Phillips, J.; Deng, X. A WRKY transcription factor participates in dehydration tolerance in Boea hygrometrica by binding to the W-box elements of the galactinol synthase (BhGOLS1) promoter. Planta 2009, 230, 1155. [Google Scholar] [CrossRef] [PubMed]
- Ghasempour, H.R.; Gaff, D.F.; Williams, R.P.W.; Gianello, R.D. Contents of sugars in leaves of drying desiccation tolerant flowering plants, particularly grasses. Plant Growth Regul. 1998, 24, 185–191. [Google Scholar] [CrossRef]
- Maia, J.; Dekkers, B.J.W.; Provart, N.J.; Ligterink, W.; Hilhorst, H.W.M. The re-establishment of desiccation tolerance in germinated Arabidopsis thaliana seeds and its associated transcriptome. PLoS ONE 2011, 6, e29123. [Google Scholar] [CrossRef] [PubMed]
- Tuteja, N. Abscisic acid and abiotic stress signaling. Plant Signal. Behav. 2007, 2, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Ishitani, M.; Xiong, L.; Stevenson, B.; Zhu, J.-K. Genetic analysis of osmotic and cold stress signal transduction in Arabidopsis: Interactions and convergence of abscisic acid-dependent and abscisic acid-independent pathways. Plant Cell 1997, 9, 1935–1949. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi-Shinozaki, K.; Shinozaki, K. A novel cis-acting element in an Arabidopsis gene is involved in responsiveness to drought, low-temperature, or high-salt stress. Plant Cell 1994, 6, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, K.; Yamaguchi-Shinozaki, K.; Shinozaki, K. The transcriptional regulatory network in the drought response and its crosstalk in abiotic stress responses including drought, cold, and heat. Front. Plant Sci. 2014, 5, 170. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, K.; Kiyosue, T.; Yamaguchi-Shinozaki, K.; Shinozaki, K.A. nuclear gene, erd1, encoding a chloroplast-targeted clp protease regulatory subunit homolog is not only induced by water stress but also developmentally up-regulated during senescence in Arabidopsis thaliana. Plant J. 1997, 12, 851–861. [Google Scholar] [CrossRef] [PubMed]
- Levitt, J. The Hardiness of Plants; Academic Press Inc.: New York, NY, USA, 1956. [Google Scholar]
- Dhawan, S.S.; Sharma, A. Analysis of differentially expressed genes in abiotic stress response and their role in signal transduction pathways. Protoplasma 2014, 251, 81–91. [Google Scholar]
- Islam, S.; Griffiths, C.A.; Blomstedt, C.K.; Le, T.-N.; Gaff, D.F.; Hamill, J.D.; Neale, A.D. Increased biomass, seed yield and stress tolerance is conferred in Arabidopsis by a novel enzyme from the resurrection grass Sporobolus stapfianus that glycosylates the strigolactone analogue GR24. PLoS ONE 2013, 8, e80035. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Rivero, R.M.; Shulaev, V.; Blumwald, E.; Mittler, R. Abiotic and biotic stress combinations. New Phytol. 2014, 203, 32–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anandarajah, K.; Kott, L.; Beversdorf, W.D.; McKersie, B.D. Induction of desiccation tolerance in microspore-derived embryos of Brassica napus L. By thermal stress. Plant Sci. 1991, 77, 119–123. [Google Scholar] [CrossRef]
- Oliver, M.J.; Jain, R.; Balbuena, T.S.; Agrawal, G.; Gasulla, F.; Thelen, J.J. Proteome analysis of leaves of the desiccation-tolerant grass, Sporobolus stapfianus, in response to dehydration. Phytochemistry 2011, 72, 1273–1284. [Google Scholar] [CrossRef] [PubMed]
- Peleg, Z.; Reguera, M.; Tumimbang, E.; Walia, H.; Blumwald, E. Cytokinin-mediated source/sink modifications improve drought tolerance and increase grain yield in rice under water-stress. Plant Biotechnol. J. 2011, 9, 747–758. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walia, H.; Wilson, C.; Wahid, A.; Condamine, P.; Cui, X.; Close, T.J. Expression analysis of barley (Hordeum vulgare L.) during salinity stress. Funct. Integr. Genom. 2006, 6, 143. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Bai, M.-Y.; Chong, K. Brassinosteroid-mediated regulation of agronomic traits in rice. Plant Cell Rep. 2014, 33, 683–696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ha, C.V.; Leyva-González, M.A.; Osakabe, Y.; Tran, U.T.; Nishiyama, R.; Watanabe, Y.; Tanaka, M.; Seki, M.; Yamaguchi, S.; Dong, N.V.; et al. Positive regulatory role of strigolactone in plant responses to drought and salt stress. Proc. Natl. Acad. Sci. USA 2014, 111, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Bu, Q.; Lv, T.; Shen, H.; Luong, P.; Wang, J.; Wang, Z.; Huang, Z.; Xiao, L.; Engineer, C.; Kim, T.H.; et al. Regular of drought tolerance by the F-box protein max2 in Arabidopsis. Plant Physiol. 2014, 164, 424–439. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; He, H.; Vitali, M.; Visentin, I.; Charnikhova, T.; Haider, I.; Schubert, A.; Ruyter-Spira, C.; Bouwmeester, H.J.; Lovisolo, C.; et al. Osmotic stress represses strigolactone biosynthesis in Lotus japonicus roots: Exploring the interaction between strigolactones and ABA under abiotic stress. Planta 2015, 241, 1435–1451. [Google Scholar] [CrossRef] [PubMed]
- Visentin, I.; Vitali, M.; Ferrero, M.; Zhang, Y.; Ruyter-Spira, C.; Novák, O.; Strnad, M.; Lovisolo, C.; Schubert, A.; Cardinale, F. Low levels of strigolactones in roots as a component of the systemic signal of drought stress in tomato. New Phytol. 2016, 212, 954–963. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruiz-Lozano, J.M.; Aroca, R.; Zamarreño, A.M.; Molina, S.; Andreo-Jiméne, B.; Porcel, R.; García-Mina, J.M.; Ruyter-Spira, C.; López-Ráez, J.A. Arbuscular mycorrhizal symbiosis induces strigolactone biosynthesis under drought and improves drought tolerance in lettuce and tomato. Plant Cell Environ. 2016, 39, 441–452. [Google Scholar] [CrossRef] [PubMed]
- El-Maarouf-Bouteau, H.; Bailly, C. Oxidative signaling in seed germination and dormancy. Plant Signal. Behav. 2008, 3, 175–182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.; Twito, S.; Miller, G. New cross talk between ROS, ABA and auxin controlling seed maturation and germination unraveled in APX6 deficient Arabidopsis seeds. Plant Signal. Behav. 2014, 9, e976489. [Google Scholar] [CrossRef] [PubMed]
- Gechev, T.S.; Benina, M.; Obata, T.; Tohge, T.; Sujeeth, N.; Minkov, I.; Hille, J.; Temanni, M.-R.; Marriott, A.S.; Bergström, E.; et al. Molecular mechanisms of desiccation tolerance in the resurrection glacial relic Haberlea rhodopensis. Cell. Mol. Life Sci. 2013, 70, 689–709. [Google Scholar] [CrossRef] [PubMed]
- Moyankova, D.; Mladenov, P.; Berkov, S.; Peshev, D.; Georgieva, D.; Djilianov, D. Metabolic profiling of the resurrection plant Haberlea rhodopensis during desiccation and recovery. Physiol. Plant. 2014, 152, 675–687. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Wang, B.; Phillips, J.; Zhang, Z.-N.; Du, H.; Xu, T.; Huang, L.-C.; Zhang, X.-F.; Xu, G.-H.; Li, W.-L.; et al. Global transcriptome analysis reveals acclimation-primed processes involved in the acquisition of desiccation tolerance in Boea hygrometrica. Plant Cell Physiol. 2015, 56, 1429–1441. [Google Scholar] [CrossRef] [PubMed]
- Beckett, M.; Loreto, F.; Velikova, V.; Brunetti, C.; DiFerdinando, M.; Tattini, M.; Calfapietra, C.; Farrant, J.M. Photosynthetic limitations and volatile and non-volatile isoprenoids in the poikilochlorophyllous resurrection plant Xerophyta humilis during dehydration and rehydration. Plant Cell Environ. 2012, 35, 2061–2074. [Google Scholar] [CrossRef] [PubMed]
- Koussevitzky, S.; Suzuki, N.; Huntington, S.; Armijo, L.; Sha, W.; Cortes, D.; Shulaev, V.; Mittler, R. Ascorbate peroxidase 1 plays a key role in the response of Arabidopsis thaliana to stress combination. J. Biol. Chem. 2008, 283, 34197–34203. [Google Scholar] [CrossRef] [PubMed]
- Prasch, C.M.; Sonnewald, U. Simultaneous application of heat, drought, and virus to Arabidopsis plants reveals significant shifts in signaling networks. Plant Physiol. 2013, 162, 1849–1866. [Google Scholar] [CrossRef] [PubMed]
- Tahir, J.; Dijkwel, P. B-substituting alanine synthases: Roles in cysteine metabolism and abiotic and biotic stress signalling in plants. Funct. Plant Biol. 2016, 43, 307–323. [Google Scholar] [CrossRef]
- Foyer, C.H.; Lelandais, M.; Kunert, K.J. Photooxidative stress in plants. Physiol. Plant. 1994, 92, 696–717. [Google Scholar] [CrossRef]
- Cruz de Carvalho, R.; Bernardes Da Silva, A.; Soares, R.; Almeida, A.M.; Coelho, A.V.; Marques Da Silva, J.; Branquinho, C. Differential proteomics of dehydration and rehydration in bryophytes: Evidence towards a common desiccation tolerance mechanism. Plant Cell Environ. 2014, 37, 1499–1515. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-M.; To, T.K.; Matsui, A.; Tanoi, K.; Kobayashi, N.I.; Matsuda, F.; Habu, Y.; Ogawa, D.; Sakamoto, T.; Matsunaga, S. Acetate-mediated novel survival strategy against drought in plants. Nat. Plants 2017, 3, 17097. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, F.; Suzuki, T.; Osakabe, Y.; Betsuyaku, S.; Kondo, Y.; Dohmae, N.; Fukuda, H.; Yamaguchi-Shinozaki, K.; Shinozaki, K. A small peptide modulates stomatal control via abscisic acid in long-distance signalling. Nature 2018, 556, 235. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M. Studies of Sugars in Relation to Induction of Desiccation Tolerance in Resurrection Grass Sporobolus stapfianus. Ph.D. Thesis, Monash University, Clayton, Australia, 2001. [Google Scholar]
- Bourquin, S.; Bonnemain, J.-L.; Delrot, S. Inhibition of loading of 14C assimilates by p-chloromercuribenzenesulfonic acid: Localization of the apoplastic pathway in Vicia faba. Plant Physiol. 1990, 92, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Qu, H.; Dibley, K.E.; Offler, C.E.; Patrick, J.W. A suite of sucrose transporters expressed in coats of developing legume seeds includes novel pH-independent facilitators. Plant J. 2007, 49, 750–764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chincinska, I.; Gier, K.; Krügel, U.; Liesche, J.; He, H.; Grimm, B.; Harren, F.J.M.; Cristescu, S.M.; Kühn, C. Photoperiodic regulation of the sucrose transporter StSUT4 affects the expression of circadian-regulated genes and ethylene production. Front. Plant Sci. 2013, 4, 26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ibraheem, O.; Botha, C.E.J.; Bradley, G. In silico analysis of cis-acting regulatory elements in 5′ regulatory regions of sucrose transporter gene families in rice (Oryza sativa Japonica) and Arabidopsis thaliana. Comput. Biol. Chem. 2010, 34, 268–283. [Google Scholar] [CrossRef] [PubMed]
- Smeekens, S.; Hellmann, H.A. Sugar sensing and signaling in plants. Front. Plant Sci. 2014, 5, 113. [Google Scholar] [CrossRef] [PubMed]
- Dobrenel, T.; Marchive, C.; Azzopardi, M.; Clément, G.; Moreau, M.; Sormani, R.; Robaglia, C.; Meyer, C. Sugar metabolism and the plant target of rapamycin kinase: A sweet operator? Front. Plant Sci. 2013, 4, 93. [Google Scholar] [CrossRef] [PubMed]
- Yobi, A.; Wone, B.W.; Xu, W.; Alexander, D.C.; Guo, L.; Ryals, J.A.; Oliver, M.J.; Cushman, J.C. Comparative metabolic profiling between desiccation-sensitive and desiccation-tolerant species of Selaginella reveals insights into the resurrection trait. Plant J. 2012, 72, 983–999. [Google Scholar] [CrossRef] [PubMed]
- Phillips, J.R.; Fischer, E.; Baron, M.; Van Den Dries, N.; Facchinelli, F.; Kutzer, M.; Rahmanzadeh, R.; Remus, D.; Bartels, D. Lindernia brevidens: A novel desiccation-tolerant vascular plant, endemic to ancient tropical rainforests. Plant J. 2008, 54, 938–948. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, G.; Gamba, A.; Murelli, C.; Salamini, F.; Bartels, D. Novel carbohydrate metabolism in the resurrection plant Craterostigma plantagineum. Plant J. 1991, 1, 355–359. [Google Scholar] [CrossRef] [PubMed]
- Romero, C.; Bellés, J.M.; Vayá, J.L.; Serrano, R.; Culiáñez-Macià, F.A. Expression of the yeast trehalose-6-phosphate synthase gene in transgenic tobacco plants: Pleiotropic phenotypes include drought tolerance. Planta 1997, 201, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.K.; Kim, J.-K.; Owens, T.G.; Ranwala, A.P.; Choi, Y.D.; Kochian, L.V.; Wu, R.J. Trehalose accumulation in rice plants confers high tolerance levels to different abiotic stresses. Proc. Natl. Acad. Sci. USA 2002, 99, 15898–15903. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grennan, A.K. The role of trehalose biosynthesis in plants. Plant Physiol. 2007, 144, 3–5. [Google Scholar] [CrossRef] [PubMed]
- Wingler, A. The function of trehalose biosynthesis in plants. Phytochemistry 2002, 60, 437–440. [Google Scholar] [CrossRef]
- Muller, B.; Pantin, F.; Génard, M.; Turc, O.; Freixes, S.; Piques, M.; Gibon, Y. Water deficits uncouple growth from photosynthesis, increase C. content, and modify the relationships between C and growth in sink organs. J. Exp. Bot. 2011, 62, 1715–1729. [Google Scholar] [CrossRef] [PubMed]
- Chiou, T.-J.; Bush, D.R. Sucrose is a signal molecule in assimilate partitioning. Proc. Natl. Acad. Sci. USA 1998, 95, 4784–4788. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yadav, U.P.; Ivakov, A.; Feil, R.; Duan, G.Y.; Walther, D.; Giavalisco, P.; Piques, M.; Carillo, P.; Hubberten, H.-M.; Stitt, M.; et al. The sucrose–trehalose 6-phosphate (Tre6P) nexus: Specificity and mechanisms of sucrose signalling by Tre6P. J. Exp. Bot. 2014, 65, 1051–1068. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Primavesi, L.F.; Jhurreea, D.; Andralojc, P.J.; Mitchell, R.A.; Powers, S.J.; Schluepmann, H.; Delatte, T.; Wingler, A.; Paul, M.J. Inhibition of SNF1-related protein kinase1 activity and regulation of metabolic pathways by trehalose-6-phosphate. Plant Physiol. 2009, 149, 1860–1871. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, C.A.; Sagar, R.; Geng, Y.; Primavesi, L.F.; Patel, M.K.; Passarelli, M.K.; Gilmore, I.S.; Steven, R.T.; Bunch, J.; Paul, M.J. Chemical intervention in plant sugar signalling increases yield and resilience. Nature 2016, 540, 574–578. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, C.A.; Paul, M.J.; Foyer, C.H. Metabolite transport and associated sugar signalling systems underpinning source/sink interactions. Biochim. Biophys. Acta (BBA)-Bioenerg. 2016, 1857, 1715–1725. [Google Scholar] [CrossRef] [PubMed]
- Nunes, C.; O’Hara, L.E.; Primavesi, L.F.; Delatte, T.L.; Schluepmann, H.; Somsen, G.W.; Silva, A.B.; Fevereiro, P.S.; Wingler, A.; Paul, M.J. The trehalose 6-phosphate/SnRK1 signaling pathway primes growth recovery following relief of sink limitation. Plant Physiol. 2013, 162, 1720–1732. [Google Scholar] [CrossRef] [PubMed]
- Nuccio, M.L.; Wu, J.; Mowers, R.; Zhou, H.P.; Meghji, M.; Primavesi, L.F.; Paul, M.J.; Chen, X.; Gao, Y.; Haque, E.; et al. Expression of trehalose-6-phosphate phosphatase in maize ears improves yield in well-watered and drought conditions. Nat. Biotechnol. 2015, 33, 862–869. [Google Scholar] [CrossRef] [PubMed]
- Schluepmann, H.; Pellny, T.; van Dijken, A.; Smeekens, S.; Paul, M. Trehalose 6-phosphate is indispensable for carbohydrate utilization and growth in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2003, 100, 6849–6854. [Google Scholar] [CrossRef] [PubMed]
- Lunn, J.E.; Feil, R.; Hendriks, J.H.; Gibon, Y.; Morcuende, R.; Osuna, D.; Scheible, W.R.; Carillo, P.; Hajirezaei, M.R.; Stitt, M. Sugar-induced increases in trehalose 6-phosphate are correlated with redox activation of ADPglucose pyrophosphorylase and higher rates of starch synthesis in Arabidopsis thaliana. Biochem. J. 2006, 397, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Hallam, N.D.; Gaff, D.F. Re-organization of fine structure during rehydration of desiccated leaves of Xerophyta villosa. New Phytol. 1978, 81, 349–355. [Google Scholar] [CrossRef]
- Hallam, N.D.; Gaff, D.F. Regeneration of chloroplast structure in Talbotia elegans: A desiccation-tolerant plant. New Phytol. 1978, 81, 657–662. [Google Scholar] [CrossRef]
- Eastmond, P.J.; van Dijken, A.J.; Spielman, M.; Kerr, A.; Tissier, A.F.; Dickinson, H.G.; Jones, J.D.; Smeekens, S.C.; Graham, I.A. Trehalose-6-phosphate synthase 1, which catalyses the first step in trehalose synthesis, is essential for Arabidopsis embryo maturation. Plant J. 2002, 29, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Gómez, L.D.; Gilday, A.; Feil, R.; Lunn, J.E.; Graham, I.A. AtTPS1-mediated trehalose 6-phosphate synthesis is essential for embryogenic and vegetative growth and responsiveness to aba in germinating seeds and stomatal guard cells. Plant J. 2010, 64, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Avonce, N.; Leyman, B.; Mascorro-Gallardo, J.O.; Van Dijck, P.; Thevelein, J.M.; Iturriaga, G. The Arabidopsis trehalose-6-p synthase AtTPS1 gene is a regulator of glucose, abscisic acid, and stress signaling. Plant Physiol. 2004, 136, 3649–3659. [Google Scholar] [CrossRef] [PubMed]
- Koster, K.L. Glass formation and desiccation tolerance in seeds. Plant Physiol. 1991, 96, 302–304. [Google Scholar] [CrossRef] [PubMed]
- Pilon-Smits, E.A.; Terry, N.; Sears, T.; Kim, H.; Zayed, A.; Hwang, S.; van Dun, K.; Voogd, E.; Verwoerd, T.C.; Krutwagen, R.W. Trehalose-producing transgenic tobacco plants show improved growth performance under drought stress. J. Plant Physiol. 1998, 152, 525–532. [Google Scholar] [CrossRef]
- Magazù, S.; Migliardo, F.; Gonzalez, M.; Mondelli, C.; Parker, S.; Vertessy, B. Molecular mechanisms of survival strategies in extreme conditions. Life 2012, 2, 364. [Google Scholar] [CrossRef] [PubMed]
- Crowe, J.H.; Carpenter, J.F.; Crowe, L.M. The role of vitrification in anhydrobiosis. Ann. Rev. Physiol. 1998, 60, 73–103. [Google Scholar] [CrossRef] [PubMed]
- Wood, A.J.; Oliver, M.J. Translational control in plant stress: The formation of messenger ribonucleoprotein particles (mRNPs) in response to desiccation of Tortula ruralis gametophytes. Plant J. 1999, 18, 359–370. [Google Scholar] [CrossRef]
- Tymms, M.J.; Gaff, D.F.; Hallam, N.D. Protein synthesis in the desiccation tolerant angiosperm Xerophyta villosa during dehydration. J. Exp. Bot. 1982, 33, 332–343. [Google Scholar] [CrossRef]
- Blomstedt, C.K.; Gianello, R.D.; Gaff, D.F.; Hamill, J.D.; Neale, A.D. Differential gene expression in desiccation-tolerant and desiccation-sensitive tissue of the resurrection grass, Sporobolus stapfianus. Funct. Plant Biol. 1998, 25, 937–946. [Google Scholar] [CrossRef]
- Le, T.N.; Blomstedt, C.K.; Kuang, J.; Tenlen, J.; Gaff, D.F.; Hamill, J.D.; Neale, A.D. Desiccation-tolerance specific gene expression in leaf tissue of the resurrection plant Sporobolus stapfianus. Funct. Plant Biol. 2007, 34, 589–600. [Google Scholar] [CrossRef]
- Daniel, V.; Gaff, D.F. Dessication-induced changes in the protein complement of soluble extracts from leaves of resurrection plants and related desiccation-sensitive species. Annu. Bot. 1980, 45, 173–181. [Google Scholar] [CrossRef]
- Gaff, D.F.; Ziegler, H. ATP and ADP contents in leaves of drying and rehydrating desiccation tolerant plants. Oecologia 1989, 78, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Leprince, O.; Buitink, J. Desiccation tolerance: From genomics to the field. Plant Sci. 2010, 179, 554–564. [Google Scholar] [CrossRef] [Green Version]
- Chiu, R.S.; Nahal, H.; Provart, N.J.; Gazzarrini, S. The role of the Arabidopsis FUSCA3 transcription factor during inhibition of seed germination at high temperature. BMC Plant Biol. 2012, 12, 15. [Google Scholar] [CrossRef] [PubMed]
- Xue, G.-P.; Way, H.M.; Richardson, T.; Drenth, J.; Joyce, P.A.; McIntyre, C.L. Overexpression of TaNAC69 leads to enhanced transcript levels of stress up-regulated genes and dehydration tolerance in bread wheat. Mol. Plant 2011, 4, 697–712. [Google Scholar] [CrossRef] [PubMed]
- Zia, A.; Walker, B.J.; Oung, H.M.O.; Charuvi, D.; Jahns, P.; Cousins, A.B.; Farrant, J.M.; Reich, Z.; Kirchhoff, H. Protection of the photosynthetic apparatus against dehydration stress in the resurrection plant Craterostigma pumilum. Plant J. 2016, 87, 664–680. [Google Scholar] [CrossRef] [PubMed]
- Piatkowski, D.; Schneider, K.; Salamini, F.; Bartels, D. Characterization of five abscisic acid-responsive cDNA clones isolated from the desiccation-tolerant plant Craterostigma plantagineum and their relationship to other water-stress genes. Plant Physiol. 1990, 94, 1682–1688. [Google Scholar] [CrossRef] [PubMed]
- Ingram, J.; Bartels, D. The molecular basis of dehydration tolerance in plants. Annu. Rev. Plant Biol. 1996, 47, 377–403. [Google Scholar] [CrossRef] [PubMed]
- Mariaux, J.-B.; Bockel, C.; Salamini, F.; Bartels, D. Desiccation-and abscisic acid-responsive genes encoding major intrinsic proteins (MIPs) from the resurrection plant Craterostigma plantagineum. Plant Mol. Biol. 1998, 38, 1089–1099. [Google Scholar] [CrossRef] [PubMed]
- Bartels, D.; Salamini, F. Desiccation tolerance in the resurrection plant Craterostigma plantagineum. A contribution to the study of drought tolerance at the molecular level. Plant Physiol. 2001, 127, 1346–1353. [Google Scholar] [CrossRef] [PubMed]
- Garwe, D.; Thomson, J.A.; Mundree, S.G. Molecular characterization of XvSAP1, a stress-responsive gene from the resurrection plant Xerophyta viscosa Baker. J. Exp. Bot. 2003, 54, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Garwe, D.; Thomson, J.A.; Mundree, S.G. Xvsap1 from Xerophyta viscosa improves osmotic-, salinity-and high-temperature-stress tolerance in Arabidopsis. Biotechnol. J. 2006, 1, 1137–1146. [Google Scholar] [CrossRef] [PubMed]
- Ndima, T.; Farrant, J.; Thomson, J.; Mundree, S. Molecular characterization of XvT8, a stress-responsive gene from the resurrection plant Xerophyta viscosa Baker. Plant Growth Regul. 2001, 35, 137–145. [Google Scholar] [CrossRef]
- Ramanjulu, S.; Bartels, D. Drought-and desiccation-induced modulation of gene expression in plants. Plant Cell Environ. 2002, 25, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Van den Dries, N.; Facchinelli, F.; Giarola, V.; Phillips, J.R.; Bartels, D. Comparative analysis of LEA-like 11-24 gene expression and regulation in related plant species within the Linderniaceae that differ in desiccation tolerance. New Phytol. 2011, 190, 75–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.; Shang, H.; Liu, Y.; Zheng, M.; Wu, R.; Phillips, J.; Bartels, D.; Deng, X. A role for a cell wall localized glycine-rich protein in dehydration and rehydration of the resurrection plant Boea hygrometrica. Plant Biol. 2009, 11, 837–848. [Google Scholar] [CrossRef] [PubMed]
- Chakrabortee, S.; Boschetti, C.; Walton, L.J.; Sarkar, S.; Rubinsztein, D.C.; Tunnacliffe, A. Hydrophilic protein associated with desiccation tolerance exhibits broad protein stabilization function. Proc. Natl. Acad. Sci. USA 2007, 104, 18073–18078. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Close, T.J. Dehydrins: A commonalty in the response of plants to dehydration and low temperature. Physiol. Plant. 1997, 100, 291–296. [Google Scholar] [CrossRef]
- Dure, L. A repeating 11-mer amino acid motif and plant desiccation. Plant J. 1993, 3, 363–369. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gasulla, F.; vom Dorp, K.; Dombrink, I.; Zähringer, U.; Gisch, N.; Dörmann, P.; Bartels, D. The role of lipid metabolism in the acquisition of desiccation tolerance in Craterostigma plantagineum: A comparative approach. Plant J. 2013, 75, 726–741. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.-C.D.; Artur, M.A.; Maia, J.; Jonkheer, E.; Derks, M.F.; Nijveen, H.; Williams, B.; Mundree, S.G.; Jiménez-Gómez, J.M.; Hesselink, T. A footprint of desiccation tolerance in the genome of Xerophyta viscosa. Nat. Plants 2017, 3, 17038. [Google Scholar] [CrossRef] [PubMed]
- Giarola, V.; Bartels, D. What can we learn from the transcriptome of the resurrection plant Craterostigma plantagineum? Planta 2015, 242, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Giarola, V.; Hou, Q.; Bartels, D. Angiosperm plant desiccation tolerance: Hints from transcriptomics and genome sequencing. Trends Plant Sci. 2017, 22, 705–717. [Google Scholar] [CrossRef] [PubMed]
- VanBuren, R.; Bryant, D.; Edger, P.P.; Tang, H.; Burgess, D.; Challabathula, D.; Spittle, K.; Hall, R.; Gu, J.; Lyons, E. Single-molecule sequencing of the desiccation-tolerant grass Oropetium thomaeum. Nature 2015, 527, 508. [Google Scholar] [CrossRef] [PubMed]
- VanBuren, R.; Wai, C.M.; Zhang, Q.; Song, X.; Edger, P.P.; Bryant, D.; Michael, T.P.; Mockler, T.C.; Bartels, D. Seed desiccation mechanisms co-opted for vegetative desiccation in the resurrection grass Oropetium thomaeum. Plant Cell Environ. 2017, 40, 2292–2306. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, K.; Bi, D.; Zhou, S.; Shao, J. Characterization of the transcriptome and EST-SSR development in Boea clarkeana, a desiccation-tolerant plant endemic to China. PeerJ 2017, 5, e3422. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Yang, G.; Zhang, L.; Yang, X.; Zhao, S.; Ji, Z.; Zhou, Q.; Hu, M.; Wang, Y.; Chen, M.; et al. The resurrection genome of Boea hygrometrica: A blueprint for survival of dehydration. Proc. Natl. Acad. Sci. USA 2015, 112, 5833–5837. [Google Scholar] [CrossRef] [PubMed]
- Farrant, J.M.; Moore, J.P. Programming desiccation-tolerance: From plants to seeds to resurrection plants. Curr. Opin. Plant Biol. 2011, 14, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Frank, W.; Phillips, J.; Salamini, F.; Bartels, D. Two dehydration-inducible transcripts from the resurrection plant Craterostigma plantagineum encode interacting homeodomain-leucine zipper proteins. Plant J. 1998, 15, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Villalobos, M.A.; Bartels, D.; Iturriaga, G. Stress tolerance and glucose insensitive phenotypes in Arabidopsis overexpressing the CpMYB10 transcription factor gene. Plant Physiol. 2004, 135, 309–324. [Google Scholar] [CrossRef] [PubMed]
- Giarola, V.; Jung, N.U.; Singh, A.; Satpathy, P.; Bartels, D. Analysis of pcc13-62 promoters predicts a link between cis-element variations and desiccation tolerance in Linderniaceae. J. Exp. Bot. 2018, 69, 3773–3784. [Google Scholar] [CrossRef] [PubMed]
- Dalton, H.L.; Blomstedt, C.K.; Neale, A.D.; Gleadow, R.; DeBoer, K.D.; Hamill, J.D. Effects of down-regulating ornithine decarboxylase upon putrescine-associated metabolism and growth in Nicotiana tabacum L. J. Exp. Bot. 2016, 67, 3367–3381. [Google Scholar] [CrossRef] [PubMed]
- Furini, A.; Koncz, C.; Salamini, F.; Bartels, D. Agrobacterium-mediated transformation of the desiccation-tolerant plant Craterostigma plantagineum. Plant Cell Rep. 1994, 14, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Smith-Espinoza, C.; Bartels, D.; Phillips, J. Analysis of a LEA gene promoter via agrobacterium-mediated transformation of the desiccation tolerant plant Lindernia brevidens. Plant Cell Rep. 2007, 26, 1681–1688. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Chen, K.; Li, T.; Zhang, Y.; Wang, Y.; Zhao, Q.; Liu, J.; Zhang, H.; Liu, C.; Ran, Y. Efficient DNA-free genome editing of bread wheat using CRISPR/Cas9 ribonucleoprotein complexes. Nat. Commun. 2017, 8, 14261. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Chen, K.; Yan, Y.; Zhang, Y.; Gao, C. Genotyping genome-edited mutations in plants using CRISPR ribonucleoprotein complexes. Plant Biotechnol. J. 2018. [Google Scholar] [CrossRef] [PubMed]
- McCallum, C.M.; Comai, L.; Greene, E.A.; Henikoff, S. Targeted screening for induced mutations. Nat. Biotechnol. 2000, 18, 455–457. [Google Scholar] [CrossRef] [PubMed]
- Till, B.J.; Cooper, J.; Tai, T.H.; Colowit, P.; Greene, E.A.; Henikoff, S.; Comai, L. Discovery of chemically induced mutations in rice by TILLING. BMC Plant Biol. 2007, 7, 19. [Google Scholar] [CrossRef] [PubMed]
- Blomstedt, C.K.; Gleadow, R.M.; O’Donnell, N.; Naur, P.; Jensen, K.; Laursen, T.; Olsen, C.E.; Stuart, P.; Hamill, J.D.; Møller, B.L. A combined biochemical screen and TILLING approach identifies mutations in Sorghum bicolor L. Moench resulting in acyanogenic forage production. Plant Biotechnol. J. 2012, 10, 54–66. [Google Scholar] [CrossRef] [PubMed]
- Blomstedt, C.K.; O’Donnell, N.H.; Bjarnholt, N.; Neale, A.D.; Hamill, J.D.; Møller, B.L.; Gleadow, R.M. Metabolic consequences of knocking out UGT85B1, the gene encoding the glucosyltransferase required for synthesis of dhurrin in Sorghum bicolor (L. Moench). Plant Cell Physiol. 2016, 57, 373–386. [Google Scholar] [CrossRef] [PubMed]
- Iturriaga, G.; Schneider, K.; Salamini, F.; Bartels, D. Expression of desiccation-related proteins from the resurrection plant Craterostigma plantagineum in transgenic tobacco. Plant Mol. Biol. 1992, 20, 555–558. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, Z.; Wang, L.; Wu, R.; Phillips, J.; Deng, X. LEA 4 group genes from the resurrection plant Boea hygrometrica confer dehydration tolerance in transgenic tobacco. Plant Sci. 2009, 176, 90–98. [Google Scholar] [CrossRef]
- Kumar, D.; Singh, P.; Yusuf, M.A.; Upadhyaya, C.P.; Roy, S.D.; Hohn, T.; Sarin, N.B. The Xerophyta viscosa aldose reductase (aldrxv4) confers enhanced drought and salinity tolerance to transgenic tobacco plants by scavenging methylglyoxal and reducing the membrane damage. Mol. Biotechnol. 2013, 54, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Li, Y.J.; Wang, B.; Yu, H.M.; Li, Q.; Hou, B.K. The arabidopsis UGT87A2, a stress-inducible family 1 glycosyltransferase, is involved in the plant adaptation to abiotic stresses. Physiol. Plant. 2017, 159, 416–432. [Google Scholar] [CrossRef] [PubMed]
- Le, T.T.; Williams, B.; Mundree, S.G. An osmotin from the resurrection plant tripogon loliiformis (TlOSM) confers tolerance to multiple abiotic stresses in transgenic rice. Physiol. Plant. 2018, 162, 13–34. [Google Scholar] [CrossRef] [PubMed]
- Blum, A. Effective use of water (EUW) and not water-use efficiency (WUE) is the target of crop yield improvement under drought stress. Field Crops Res. 2009, 112, 119–123. [Google Scholar] [CrossRef]
- Dai, A. Increasing drought under global warming in observations and models. Nat. Clim. Chang. 2013, 3, 52. [Google Scholar] [CrossRef]
- Deikman, J.; Petracek, M.; Heard, J.E. Drought tolerance through biotechnology: Improving translation from the laboratory to farmers’ fields. Curr. Opin. Biotechnol. 2012, 23, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Wellburn, F.A.M.; Wellburn, A.R. Novel chloroplasts and unusual cellular ultrastructure in the “resurrection” plant Myrothamnus flabellifolia Welw. (Myrothamnaceae). Bot. J. Linn. Soc. 1976, 72, 51–54. [Google Scholar] [CrossRef]
- Brewer, P.B.; Koltai, H.; Beveridge, C.A. Diverse roles of strigolactones in plant development. Mol. Plant 2013, 6, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Lim, E.K.; Bowles, D.J. A class of plant glycosyltransferases involved in cellular homeostasis. EMBO J. 2004, 23, 2915–2922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cardoso, C.; Zhang, Y.; Jamil, M.; Hepworth, J.; Charnikhova, T.; Dimkpa, S.O.N.; Meharg, C.; Wright, M.H.; Liu, J.; Meng, X.; et al. Natural variation of rice strigolactone biosynthesis is associated with the deletion of two max1 orthologs. Proc. Natl. Acad. Sci. USA 2014, 111, 2379–2384. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Martínez, J.A.; Poza-Carrión, C.; Cubas, P. Arabidopsis branched1 acts as an integrator of branching signals within axillary buds. Plant Cell 2007, 19, 458–472. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.R.; Chung, K.M.; Park, J.-H.; Oh, S.A.; Ahn, T.; Hong, S.H.; Jang, S.K.; Nam, H.G. Ore9, an F-box protein that regulates leaf senescence in Arabidopsis. Plant Cell 2001, 13, 1779–1790. [Google Scholar] [CrossRef] [PubMed]
- Stirnberg, P.; Furner, I.J.; Ottoline, L.H.M. Max2 participates in an scf complex which acts locally at the node to suppress shoot branching. Plant J. 2007, 50, 80–94. [Google Scholar] [CrossRef] [PubMed]
- Zentgraf, U. Oxidative stress and leaf senescence. In Annual Plant Reviews: Senescence Processes in Plants; Gan, S., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2007; Volume 26. [Google Scholar]
- He, Y.; Fukushige, H.; Hildebrand, D.F.; Gan, S. Evidence supporting a role of jasmonic acid in Arabidopsis leaf senescence. Plant Physiol. 2002, 128, 876–884. [Google Scholar] [CrossRef] [PubMed]
- Lohman, K.N.; Gan, S.; John, M.C.; Amasino, R.M. Molecular analysis of natural leaf senescence in Arabidopsis thaliana. Physiol. Plant. 1994, 92, 322–328. [Google Scholar] [CrossRef]
- McCabe, M.S.; Garratt, L.C.; Schepers, F.; Jordi, W.J.R.M.; Stoopen, G.M.; Davelaar, E.; van Rhijn, J.H.A.; Power, J.B.; Davey, M.R. Effects of p(SAG12)-IPT gene expression on development and senescence in transgenic lettuce. Plant Physiol. 2001, 127, 505–516. [Google Scholar] [CrossRef] [PubMed]
- Weaver, L.M.; Gan, S.; Quirino, B.; Amasino, R.M. A comparison of the expression patterns of several senescence-associated genes in response to stress and hormone treatment. Plant Mol. Biol. 1998, 37, 455–469. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Gan, S. A gene encoding an acyl hydrolase is involved in leaf senescence in Arabidopsis. Plant Cell 2002, 14, 805–815. [Google Scholar] [CrossRef] [PubMed]
- Guerrero, F.D.; Jones, J.T.; Mullet, J.E. Turgor-responsive gene transcription and RNA levels increase rapidly when pea shoots are wilted. Sequence and expression of three inducible genes. Plant Mol. Biol. 1990, 15, 11–26. [Google Scholar] [CrossRef] [PubMed]
- Buchanan-Wollaston, V.; Ainsworth, C. Leaf senescence in Brassica napus: Cloning of senescence related genes by subtractive hybridisation. Plant Mol. Biol. 1997, 33, 821–834. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.M.; Song, H.R.; Han, S.K.; Han, M.; Kim, C.Y.; Park, J.; Lee, Y.H.; Jeon, J.S.; Noh, Y.S.; Noh, B. Hda19 is required for the repression of salicylic acid biosynthesis and salicylic acid-mediated defense responses in Arabidopsis. Plant J. 2012, 71, 135–146. [Google Scholar] [CrossRef] [PubMed]
- Chinnusamy, V.; Zhu, J.-K. Epigenetic regulation of stress responses in plants. Curr. Opin. Plant Biol. 2009, 12, 133–139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.-M.; Sasaki, T.; Ueda, M.; Sako, K.; Seki, M. Chromatin changes in response to drought, salinity, heat, and cold stresses in plants. Front. Plant Sci. 2015, 6, 114. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-M.; To, T.K.; Seki, M. An epigenetic integrator: New insights into genome regulation, environmental stress responses and developmental controls by histone deacetylase 6. Plant Cell Physiol. 2012, 53, 794–800. [Google Scholar] [CrossRef] [PubMed]
- Bej, S.; Basak, J. Abiotic stress induced epigenetic modifications in plants: How much do we know? In Plant Epigenetics; Rajewsky, N., Jurga, S., Barciszewski, J., Eds.; Springer: New York, NY, USA, 2017; pp. 493–512. [Google Scholar]
- Diez, C.M.; Roessler, K.; Gaut, B.S. Epigenetics and plant genome evolution. Curr. Opin. Plant Biol. 2014, 18, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Meyer, P. Epigenetic variation and environmental change. J. Exp. Bot. 2015, 66, 3541–3548. [Google Scholar] [CrossRef] [PubMed]
- Matzke, M.A.; Mosher, R.A. RNA-directed DNA methylation: An epigenetic pathway of increasing complexity. Nat. Rev. Genet. 2014, 15, 394–408. [Google Scholar] [CrossRef] [PubMed]
- Niederhuth, C.E.; Schmitz, R.J. Putting DNA methylation in context: From genomes to gene expression in plants. Biochim. Biophys. Acta 2017, 1860, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Furini, A.; Koncz, C.; Salamini, F.; Bartels, D. High level transcription of a member of a repeated gene family confers dehydration tolerance to callus tissue of Craterostigma plantagineum. EMBO J. 1997, 16, 3599–3608. [Google Scholar] [CrossRef] [PubMed]
- Ferdous, J.; Hussain, S.S.; Shi, B.J. Role of microRNAs in plant drought tolerance. Plant Biotechnol. J. 2015, 13, 293–305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khraiwesh, B.; Zhu, J.-K.; Zhu, J. Role of miRNAs and siRNAs in biotic and abiotic stress responses of plants. Biochim. Biophys. Acta (BBA) Gene Regul. Mech. 2012, 1819, 137–148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith-Espinoza, C.; Phillips, J.; Salamini, F.; Bartels, D. Identification of further Craterostigma plantagineum cdt mutants affected in abscisic acid mediated desiccation tolerance. Mol. Genet. Genom. 2005, 274, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Xu, T.; Shen, C.-Y.; Xu, G.-H.; Chen, S.-X.; Song, L.-Z.; Li, M.-J.; Wang, L.-L.; Zhu, Y.; Lv, W.-T.; et al. Identification of a retroelement from the resurrection plant Boea hygrometrica that confers osmotic and alkaline tolerance in Arabidopsis thaliana. PLoS ONE 2014, 9, e98098. [Google Scholar] [CrossRef] [PubMed]
- Ditzer, A.; Bartels, D. Identification of a dehydration and ABA-responsive promoter regulon and isolation of corresponding DNA binding proteins for the group 4 LEA gene cpc2 from C. plantagineum. Plant Mol. Biol. 2006, 61, 643–663. [Google Scholar] [CrossRef] [PubMed]
- Ryan, D.P.; Owen-Hughes, T. Snf2-family proteins: Chromatin remodellers for any occasion. Curr. Opin. Chem. Biol. 2011, 15, 649–656. [Google Scholar] [CrossRef] [PubMed]
- Strein, C.; Alleaume, A.-M.; Rothbauer, U.; Hentze, M.W.; Castello, A. A versatile assay for RNA-binding proteins in living cells. RNA 2014, 20, 721–731. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verma, J.K.; Gayali, S.; Dass, S.; Kumar, A.; Parveen, S.; Chakraborty, S.; Chakraborty, N. Osalba1, a dehydration-responsive nuclear protein of rice (Oryza sativa L. ssp. Indica), participates in stress adaptation. Phytochemistry 2014, 100, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Kapazoglou, A.; Drosou, V.; Argiriou, A.; Tsaftaris, A.S. The study of a barley epigenetic regulator, HvDME, in seed development and under drought. BMC Plant Biol. 2013, 13, 172. [Google Scholar] [CrossRef] [PubMed]
- Song, C.-P.; Agarwal, M.; Ohta, M.; Guo, Y.; Halfter, U.; Wang, P.; Zhu, J.-K. Role of an arabidopsis ap2/erebp-type transcriptional repressor in abscisic acid and drought stress responses. Plant Cell 2005, 17, 2384–2396. [Google Scholar] [CrossRef] [PubMed]
- To, T.K.; Nakaminami, K.; Kim, J.-M.; Morosawa, T.; Ishida, J.; Tanaka, M.; Yokoyama, S.; Shinozaki, K.; Seki, M. Arabidopsis HDA6 is required for freezing tolerance. Biochem. Biophys. Res. Commun. 2011, 406, 414–419. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.-Y.; Chen, S.-Y. Inducible expression of translation elongation factor 1a gene in rice seedlings in response to environmental stresses. Acta Bot. Sin. 1999, 41, 800–806. [Google Scholar]
- Sani, E.; Herzyk, P.; Perrella, G.; Colot, V.; Amtmann, A. Hyperosmotic priming of Arabidopsis seedlings establishes a long-term somatic memory accompanied by specific changes of the epigenome. Genome Biol. 2013, 14, R59. [Google Scholar] [CrossRef] [PubMed]
- Gupta, D.B.; Rai, Y.; Gayali, S.; Chakraborty, S.; Chakraborty, N. Plant organellar proteomics in response to dehydration: Turning protein repertoire into insights. Front. Plant Sci. 2016, 7, 460. [Google Scholar] [CrossRef] [PubMed]
- Oliver, M.J.; Guo, L.; Alexander, D.C.; Ryals, J.A.; Wone, B.W.M.; Cushman, J.C. A sister group contrast using untargeted global metabolomic analysis delineates the biochemical regulation underlying desiccation tolerance in Sporobolus stapfianus. Plant Cell 2011, 23, 1231–1248. [Google Scholar] [CrossRef] [PubMed]
- Tymms, M.; Gaff, D. Recovery of protein synthesis and RNA synthesis after rewetting of desiccated leaves of the angiosperm Xerophyta villosa. Biochem. Physiol. Pflanz. 1984, 179, 211–217. [Google Scholar] [CrossRef]
| Genus & Species No. of Desiccation Tolerant Species/of Total spp. in Genus | Notes on Tolerance Limits | Genus & Species No. of Desiccation Tolerant Species/of Total spp. in Genus | Notes on Tolerance Limits | ||
|---|---|---|---|---|---|
| Brachyachne 1/10 | Oropetium 3/4 | ||||
| B. patentifolia | 0% RH | >15 cm | O. capense | 0% RH | |
| Eragrostiella 3/5 | O. roxburghianum | 0% RH | |||
| E. bifaria var. bifaria | 15% RH | G | O. thomaeum | 0% RH | |
| E. brachyphylla | 0% RH, | Poa 1/~500 | |||
| E. nardoides | 11% RH | Poa bulbosa | 5% RH | Bs V | |
| Eragrostis 4/350 | Sporobolus 7/160 | ||||
| Er. hispida | 0–5% RH | Bs, >15 cm | S. atrovirens | <4% RWC | |
| Er. invalida | 0% RH | Bs, >30 cm | S. blakei | 0% RH | Bs |
| Er. nindensis | 0–2% RH | G, >15 cm | S. elongatus | <10% RWC | Bs > 30 cm |
| Er. paradoxa | 0% RH | >15 cm | S. festivus | 0% RH | >15 cm |
| Micraira 7/14 | S. lampranthus | 0% RH | Bs | ||
| M. adamsii | 0–2% RH | S. pellucidus | 0–5% RH | >15 cm | |
| M. lazaridis | - | S. stapfianus | 2% RH | >15 cm | |
| M. multinervia | >5% RH | Tripogon 10/30 | |||
| M. spinifera | <7.5% RWC | T. capillatus | 0% RH | >15 cm | |
| M. subulifolia | 5% RH | T. curvatus | 0% RH | ||
| M. tenuis | <8% RWC | T. filiformis | 2% RH | >25 cm | |
| M. viscidula | <7.5% RWC | T. jaquemontii | 0% RH | G | |
| Microchloa 3/4 | T. lisboae | 0% RH | >25 cm | ||
| Mi. caffra | 0–5% RH | >15 cm | T. loliiformis | 0% RH | G |
| Mi. indica | 0% RH | T. major | 0% RH | Bs | |
| Mi. kunthii | 0% RH | T. minimus | 0–2% RH | ||
| T. polyanthus | 2% RH | >15 cm | |||
| T. spicatus | 0% RH | ||||
| DICOTYLEDONS | Tissue Type |
|---|---|
| Gesneriaceae | |
| Conandron ramondioides | Young leaf to 1 cm size |
| Opithandra primuloides | To 2nd youngest leaf (1 cm long) |
| Stylidiaceae | |
| Stylidium sp. | Buds only |
| MONOCOTYLEDONS | |
| Anthericaceae | |
| Borya scirpioides | High in 3 youngest leaves |
| Cyperaceae | |
| Carex physodes | Bs = ~1 cm at base of the leaf lamina, including the leaf meristem |
| Carex pachystilis | Bs |
| Cyperus bellis | Bs |
| Fimbristylis dichotoma | Bs |
| Kyllinga alba Nees | Bs |
| Kyllinga alata | Bs |
| Kyllinga nervosa | Bs |
| Mariscus capensis | Bs |
| Poaceae: see Table 1 |
| Leaf RWC (%) | 94 | 82 | 66 | 61 | 58 | 49 | 48 | 46 |
| Survival % of total leaf area | 6 | 2 | 0 | 48 | 67 | 59 | 85 | 85 |
| RWC (%) | 96 | 90–75 | 75–60 | 45–40 | 30 | 20 | 4 |
| % RNA as Polysomes | ~30 | ~25 | ~45 | ~55 | ~32 | ~1 | 0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blomstedt, C.K.; Griffiths, C.A.; Gaff, D.F.; Hamill, J.D.; Neale, A.D. Plant Desiccation Tolerance and its Regulation in the Foliage of Resurrection “Flowering-Plant” Species. Agronomy 2018, 8, 146. https://doi.org/10.3390/agronomy8080146
Blomstedt CK, Griffiths CA, Gaff DF, Hamill JD, Neale AD. Plant Desiccation Tolerance and its Regulation in the Foliage of Resurrection “Flowering-Plant” Species. Agronomy. 2018; 8(8):146. https://doi.org/10.3390/agronomy8080146
Chicago/Turabian StyleBlomstedt, Cecilia K., Cara A. Griffiths, Donald F. Gaff, John D. Hamill, and Alan D. Neale. 2018. "Plant Desiccation Tolerance and its Regulation in the Foliage of Resurrection “Flowering-Plant” Species" Agronomy 8, no. 8: 146. https://doi.org/10.3390/agronomy8080146





