First Report of Amaranthus hybridus with Multiple Resistance to 2,4-D, Dicamba, and Glyphosate
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material and Growing Conditions
2.2. Dose Response Assays
2.3. Shikimic Acid Accumulation
2.4. Piperonil Butoxide (PBO) Effects on 2,4-D and Dicamba Metabolism
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Netto, A.G.; Nicolai, M.; Carvalho, S.J.P.; Borgato, E.A.; Christoffoleti, P.J. Multiple resistance of Amaranthus palmeri to ALS and EPSPSs inhibiting herbicides in the state of Mato Grosso, Brazil. Planta Daninha 2016, 34, 581–587. [Google Scholar] [CrossRef]
- Faccini, D.; Vitta, J.I. Germination characteristics of Amaranthus quitensis as affected by seed production date and duration of burial. Weed Res. 2005, 45, 371–378. [Google Scholar] [CrossRef]
- Vitta, J.I.; Faccini, D.E.; Nisensohn, L.A. Control of Amaranthus quitensis in soybean crops in Argentina: An alternative to reduce herbicide use. Crop Prot. 2000, 19, 511–513. [Google Scholar] [CrossRef]
- Tuesca, D.; Nisensohn, L. Resistance of Amaranthus quitensis to imazethapyr and clhorimuron-ethyl. Pesqui. Agropecu. Bras. 2001, 36, 601–606. [Google Scholar] [CrossRef]
- Montero Bulacio, N.; Lescano, M.C.; Permingeat, H.; Tuesca, D. Evaluación de la sensibilidad a glifosato en distintos biotipos de Amaranthus hybridus L. (yuyo colorado). Abstr. XVI Congr. Soc. Biol. Rosario 2014, 82. Available online: http://sedici.unlp.edu.ar/bitstream/handle/10915/48707/Documento_completo.pdf-PDFA.pdf?sequence=3 (accessed on 24 April 2015).
- Belgrano, M.J.; Morrone, O.; Zuloaga, F.O.; Instituto de Botánica Darwinion Missouri Botanical Garden. Catálogo de las plantas vasculares del Cono Sur: (Argentina, Sur de Brasil, Chile, Paraguay y Uruguay); Catalog of the vascular plants of the Southern Cone; San Isidro, A., Ed.; Missouri Botanical Garden Press: St. Louis, MO, USA, 2008; ISBN 978-1-930723-76-4. [Google Scholar]
- Heap, I.M. The International Survey of Herbicide Resistant Weeds. Available online: http://www.weedscience.org/ (accessed on 12 June 2017).
- Larran, A.S.; Palmieri, V.E.; Perotti, V.E.; Lieber, L.; Tuesca, D.; Permingeat, H.R. Target-site resistance to acetolactate synthase (ALS)-inhibiting herbicides in Amaranthus palmeri from Argentina. Pest Manag. Sci. 2017, 73, 2578–2584. [Google Scholar] [CrossRef] [PubMed]
- Gaines, T.A.; Shaner, D.L.; Ward, S.M.; Leach, J.E.; Preston, C.; Westra, P. Mechanism of resistance of evolved glyphosate-resistant palmer amaranth (Amaranthus palmeri). J. Agric. Food Chem. 2011, 59, 5886–5889. [Google Scholar] [CrossRef] [PubMed]
- Nandula, V.K.; Wright, A.A.; Bond, J.A.; Ray, J.D.; Eubank, T.W.; Molin, W.T. EPSPS amplification in glyphosate-resistant spiny amaranth (Amaranthus spinosus): A case of gene transfer via interspecific hybridization from glyphosate-resistant Palmer amaranth (Amaranthus palmeri): Glyphosate-resistant spiny amaranth. Pest Manag. Sci. 2014, 70, 1902–1909. [Google Scholar] [CrossRef] [PubMed]
- Pline, W.A.; Wilcut, J.W.; Duke, S.O.; Edmisten, K.L.; Wells, R. Tolerance and accumulation of shikimic acid in response to glyphosate applications in glyphosate-resistant and nonglyphosate-resistant cotton (Gossypium hirsutum L.). J. Agric. Food Chem. 2002, 50, 506–512. [Google Scholar] [CrossRef] [PubMed]
- Arregui, M.C.; Puricelli, E. Mecanismo de Ación de Plaguicidas; Dow Agro Science: Cordoba, Argentina, 2008; ISBN 978-987-05-4191-2. [Google Scholar]
- Torra, J.; Rojano-Delgado, A.M.; Rey-Caballero, J.; Royo-Esnal, A.; Salas, M.L.; De Prado, R. Enhanced 2,4-D metabolism in two resistant papaver rhoeas populations from Spain. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Powles, S. Metabolism-based herbicide resistance and cross-resistance in crop weeds: A threat to herbicide sustainability and global crop production. Plant Physiol. 2014, 166, 1106–1118. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, M.R.A.; Leibhart, L.J.; Reicher, Z.J.; Tranel, P.J.; Nissen, S.J.; Westra, P.; Bernards, M.L.; Kruger, G.R.; Gaines, T.A.; Jugulam, M. Metabolism of 2,4-dichlorophenoxyacetic acid contributes to resistance in a common waterhemp (Amaranthus tuberculatus) population. Pest Manag. Sci. 2017. [Google Scholar] [CrossRef] [PubMed]
- Hess, M.; Barralis, G.; Bleiholder, H.; Buhr, L.; Eggers, T.; Hack, H.; Stauss, R. Use of the extended BBCH scale-general for the descriptions of the growth stages of mono; and dicotyledonous weed species. Weed Res. 1997, 37, 433–441. [Google Scholar] [CrossRef]
- R Foundation for Statistical Computing. A Language and Environment for Statistical; R Development Core Team: Vienna, Austria, 2010. [Google Scholar]
- Ritz, C.; Streibig, J.C. Bioassay analysis using R. J. Stat. Softw. 2005, 12, 1–22. [Google Scholar] [CrossRef]
- Streibig, J.C.; Kudsk, P. Herbicide Bioassays; CRC Press: Cleveland, OH, USA, 1993. [Google Scholar]
- Kniss, A.R.; Vassios, J.D.; Nissen, S.J.; Ritz, C. Nonlinear regression analysis of herbicide absorption studies. Weed Sci. 2011, 59, 601–610. [Google Scholar] [CrossRef]
- Singh, B.; Shaner, D. Rapid determination of glyphosate injury to plants and identification of glyphosate-resistant plants. Weed Technol. 1998, 12, 527–530. [Google Scholar] [CrossRef]
- Burgos, N.R.; Tranel, P.J.; Streibig, J.C.; Davis, V.M.; Shaner, D.; Norsworthy, J.K.; Ritz, C. Review: Confirmation of resistance to herbicides and evaluation of resistance levels. Weed Sci. 2013, 61, 4–20. [Google Scholar] [CrossRef]
- Bernards, M.L.; Crespo, R.J.; Kruger, G.R.; Gaussoin, R.; Tranel, P.J. A waterhemp (Amaranthus tuberculatus) population resistant to 2,4-D. Weed Sci. 2012, 60, 379–384. [Google Scholar] [CrossRef]
- Walsh, M.J.; Powles, S.B.; Beard, B.R.; Parkin, B.T.; Porter, S.A. Multiple-herbicide resistance across four modes of action in wild radish (Raphanus raphanistrum). Weed Sci. 2004, 52, 8–13. [Google Scholar] [CrossRef]
- Watanabe, H.; Ismail, M.Z.; Ho, N.K. Response of 2,4-D resistant biotype of Fimbristylis miliacea (L.) vahl. to 2,4-D dimethylamine and its distribution in the Muda Plain, Peninsular Malaysia. J. Weed Sci. Technol. 1997, 42, 240–249. [Google Scholar] [CrossRef]
- Goggin, D.E.; Cawthray, G.R.; Powles, S.B. 2,4-D resistance in wild radish: reduced herbicide translocation via inhibition of cellular transport. J. Exp. Bot. 2016, 67, 3223–3235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dillon, A.; Varanasi, V.K.; Danilova, T.V.; Koo, D.-H.; Nakka, S.; Peterson, D.E.; Tranel, P.J.; Friebe, B.; Gill, B.S.; Jugulam, M. Physical mapping of amplified copies of the 5-enolpyruvylshikimate-3-phosphate synthase gene in glyphosate-resistant Amaranthus tuberculatus. Plant Physiol. 2017, 173, 1226–1234. [Google Scholar] [CrossRef] [PubMed]
- Hall, L.M.; Holtum, J.A.M.; Powles, S.B. Mechanisms responsible for cross resistance and multiple resistance. In Herbicide Resistance in Plants: Biology and Biochemistry; Powles, S.B., Holtum, J.A.M., Eds.; Lewis Press: Stockport, England, 1994; pp. 243–261. [Google Scholar]
- Hilton, H. Herbicide Tolerant Strains of Weeds; Hawaiian Sugar Planters Association Annual Report: Honolulu, HI, USA, 1957; pp. 69–72. [Google Scholar]
- Switzer, C. The existence of 2,4-D-resistant strains of wild carrot. Proc. Northeast. Weed Control Conf. 1957, 11, 315–318. [Google Scholar]
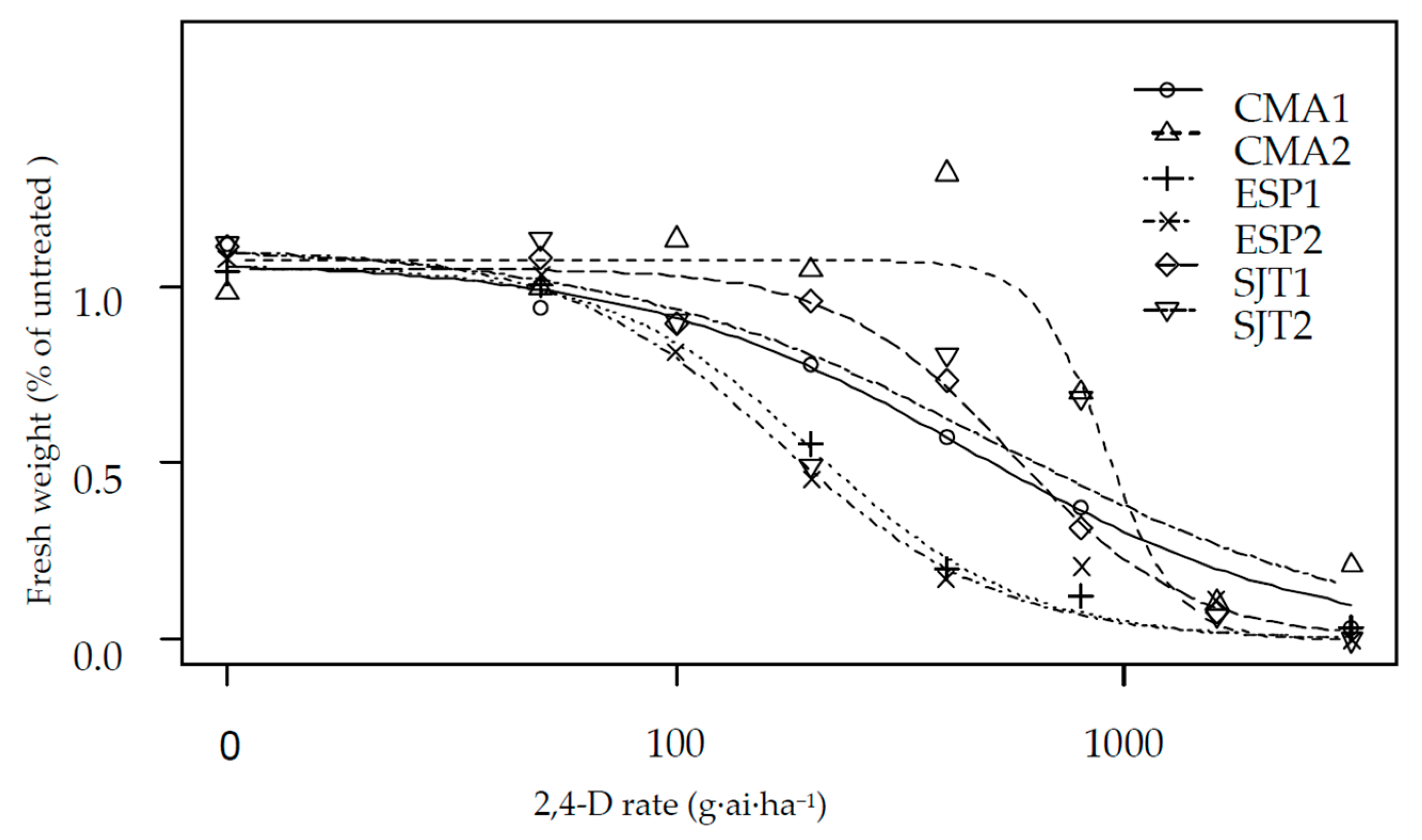
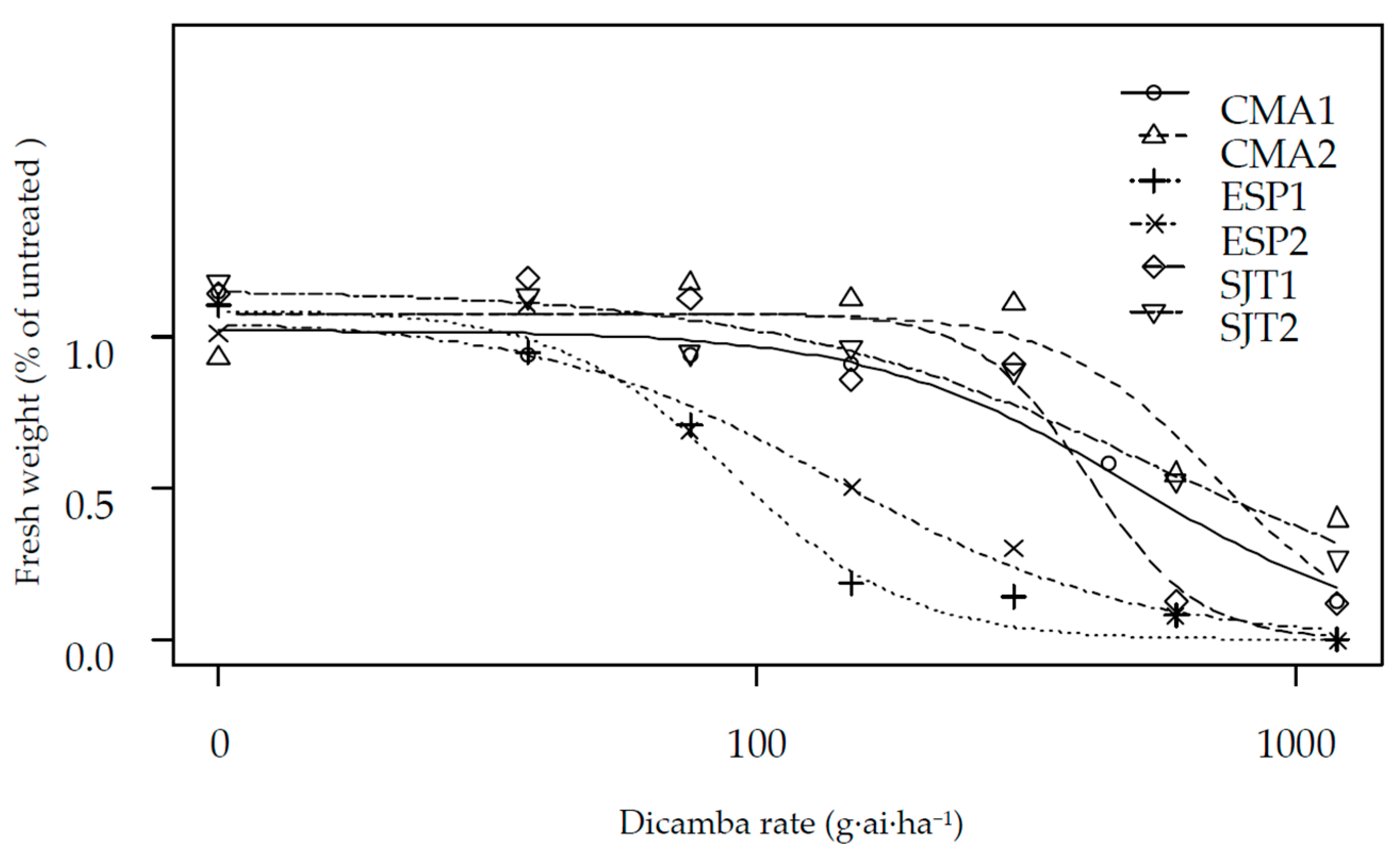
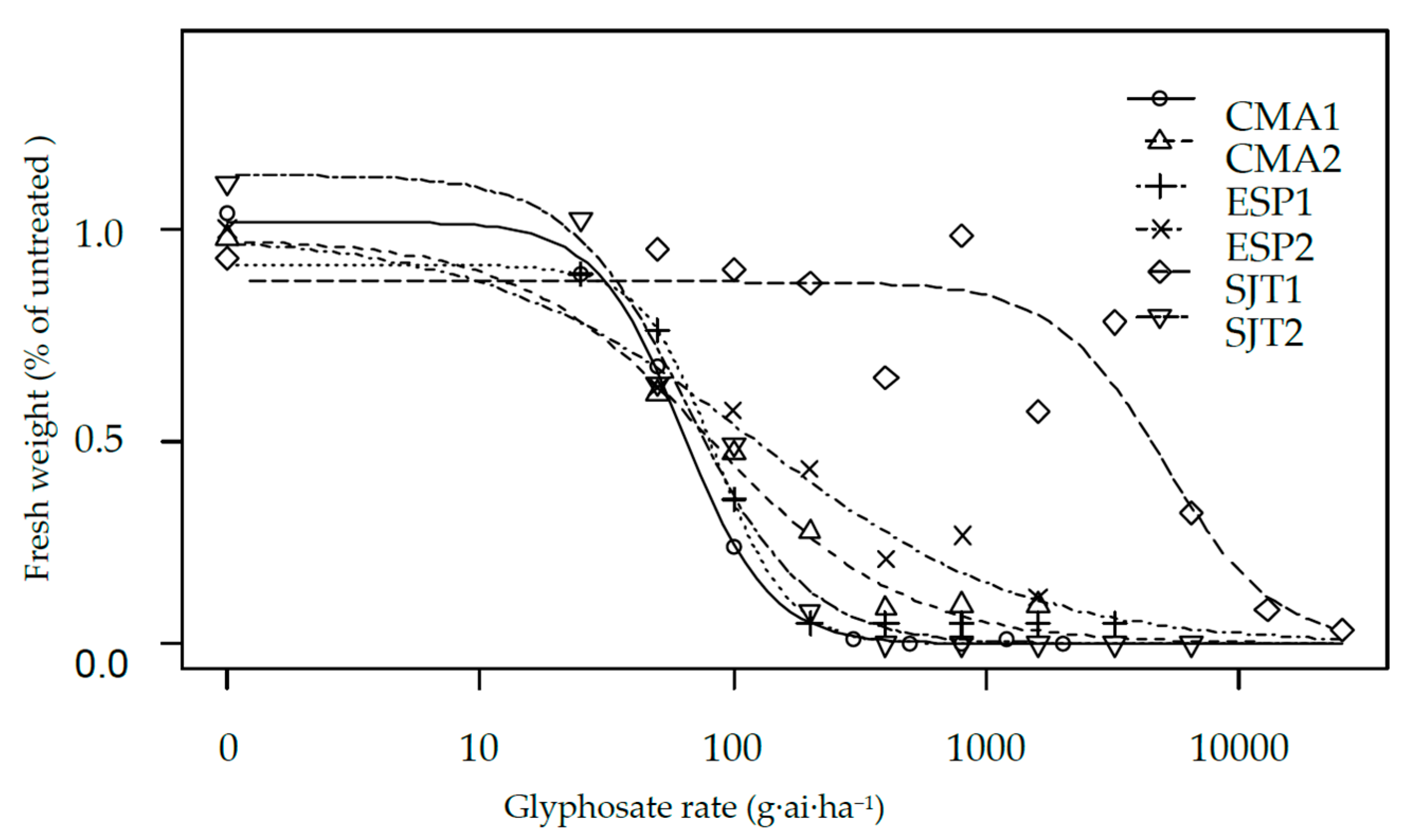
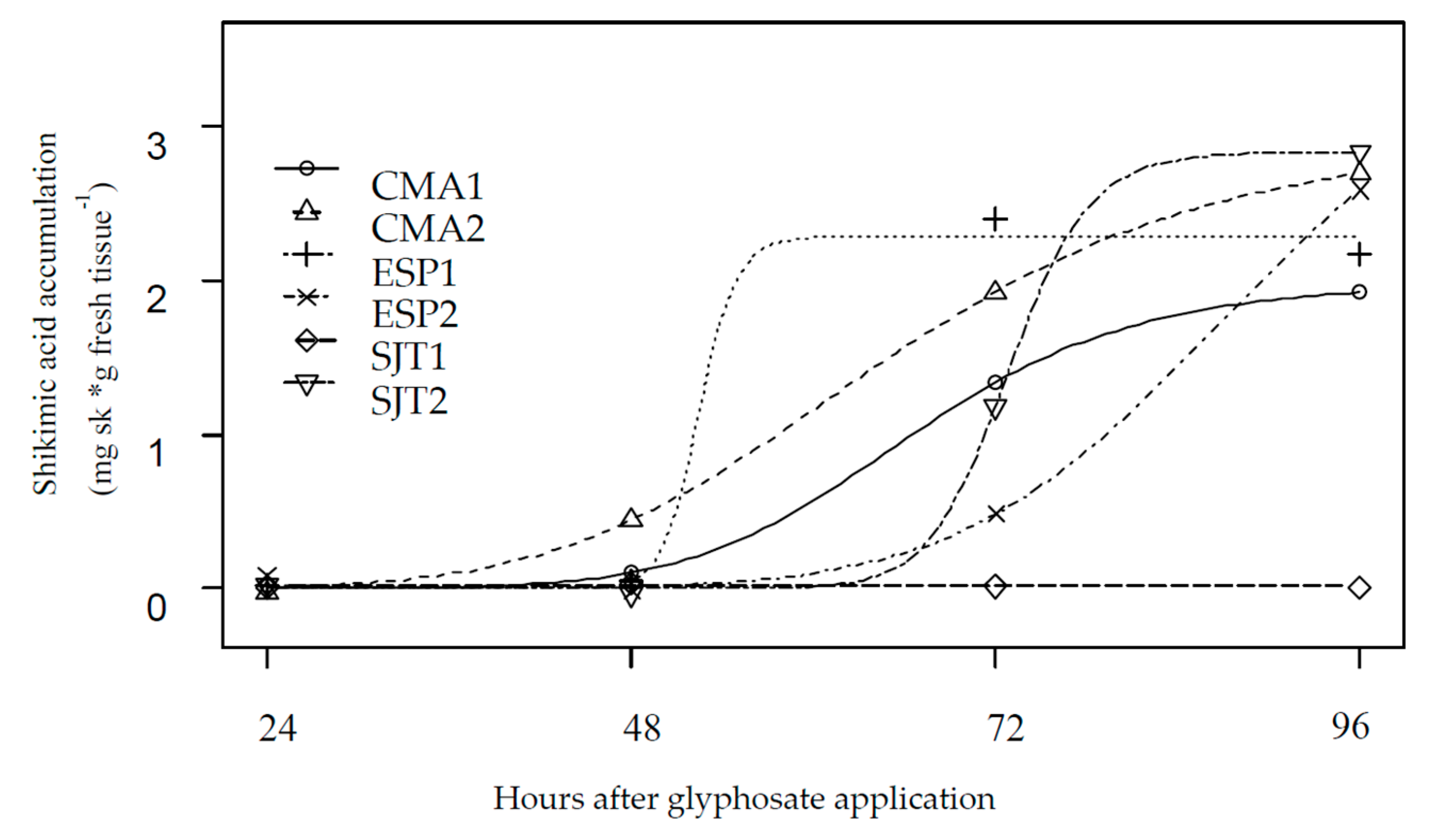
| Population | Herbicide | b | d | GR50 | se | Res. Factor | p-Value | GR80 | se |
|---|---|---|---|---|---|---|---|---|---|
| CMA1 | 2,4-D | 1.161 | 1.066 | 272.78 | 102.86 | 2.64 | 0.001 | 900.30 | 425.07 |
| CMA2 | 2,4-D | 5.563 | 0.075 | 547.03 | 128.11 | 5.31 | 0.012 | 704.33 | 335.80 |
| ESP1 | 2,4-D | 1.849 | 0.148 | 123.13 | 35.45 | 1.20 | 0.591 | 256.19 | 70.46 |
| ESP2 | 2,4-D | 1.686 | 0.114 | 103.10 | 22.41 | - | - | 225.23 | 67.69 |
| SJT1 | 2,4-D | 2.229 | 0.089 | 336.73 | 69.43 | 3.27 | 0.022 | 627.17 | 209.17 |
| SJT2 | 2,4-D | 1.008 | 0.143 | 309.84 | 137.07 | 3.01 | 0.178 | 1224.61 | 472.87 |
| CMA1 | Dicamba | 1.791 | 1.021 | 299.29 | 83.37 | 5.50 | 0.001 | 648.90 | 279.50 |
| CMA2 | Dicamba | 2.971 | 1.077 | 428.28 | 105.77 | 7.87 | 0.001 | 682.90 | 289.50 |
| ESP1 | Dicamba | 2.657 | 1.089 | 54.40 | 8.86 | - | - | 91.72 | 21.35 |
| ESP2 | Dicamba | 1.593 | 1.058 | 83.87 | 24.36 | 1.54 | 0.106 | 200.27 | 77.41 |
| SJT1 | Dicamba | 4.240 | 1.077 | 248.12 | 44.44 | 4.56 | 0.001 | 344.07 | 135.50 |
| SJT2 | Dicamba | 1.192 | 1.157 | 326.36 | 86.78 | 6.00 | 0.001 | 1043.88 | 533.02 |
| CMA1 | Glyphosate | 2.538 | 1.017 | 63.51 | 8.16 | 0.76 | 0.251 | 109.66 | 18.11 |
| CMA2 | Glyphosate | 1.142 | 0.976 | 82.47 | 19.20 | 0.98 | 0.955 | 277.74 | 79.47 |
| ESP1 | Glyphosate | 2.875 | 0.926 | 83.81 | 14.00 | - | - | 135.76 | 32.63 |
| ESP2 | Glyphosate | 0.819 | 0.987 | 119.86 | 70.33 | 1.43 | 0.482 | 652.00 | 336.91 |
| SJT1 | Glyphosate | 1.959 | 0.875 | 5018.2 | 1315.2 | 167.0 | 0.001 | 10,181.8 | 3434.2 |
| SJT2 | Glyphosate | 1.867 | 1.130 | 66.85 | 14.25 | 0.79 | 0.456 | 140.47 | 38.62 |
| Population/Active | 2,4-D | 2,4-D + PBO | Dicamba | Dicamba + PBO |
|---|---|---|---|---|
| CMA1 | 0.79 ± 0.28 | 0.06 ± 0.06 * | 0.68 ± 0.25 | 0.09 ± 0.13 * |
| CMA2 | 0.89 ± 0.33 | 0.06 ± 0.09 * | 0.97 ± 0.25 | 0.03 ± 0.06 * |
| ESP1 | 0.08 ± 0.14 | 0.07 ± 0.15 | 0.57 ± 0.06 | 0.05 ± 0.09 * |
| ESP2 | 0.22 ± 0.08 | 0.07 ± 0.09 * | 0.30 ± 0.15 | 0.10 ± 0.15 * |
| SJT1 | 0.45 ± 0.20 | 0.07 ± 0.06 * | 0.95 ± 0.25 | 0.04 ± 0.04 * |
| SJT2 | 0.81 ± 0.26 | 0.06 ± 0.09 * | 0.78 ± 0.39 | 0.11 ± 0.14 * |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dellaferrera, I.; Cortés, E.; Panigo, E.; De Prado, R.; Christoffoleti, P.; Perreta, M. First Report of Amaranthus hybridus with Multiple Resistance to 2,4-D, Dicamba, and Glyphosate. Agronomy 2018, 8, 140. https://doi.org/10.3390/agronomy8080140
Dellaferrera I, Cortés E, Panigo E, De Prado R, Christoffoleti P, Perreta M. First Report of Amaranthus hybridus with Multiple Resistance to 2,4-D, Dicamba, and Glyphosate. Agronomy. 2018; 8(8):140. https://doi.org/10.3390/agronomy8080140
Chicago/Turabian StyleDellaferrera, Ignacio, Eduardo Cortés, Elisa Panigo, Rafael De Prado, Pedro Christoffoleti, and Mariel Perreta. 2018. "First Report of Amaranthus hybridus with Multiple Resistance to 2,4-D, Dicamba, and Glyphosate" Agronomy 8, no. 8: 140. https://doi.org/10.3390/agronomy8080140
APA StyleDellaferrera, I., Cortés, E., Panigo, E., De Prado, R., Christoffoleti, P., & Perreta, M. (2018). First Report of Amaranthus hybridus with Multiple Resistance to 2,4-D, Dicamba, and Glyphosate. Agronomy, 8(8), 140. https://doi.org/10.3390/agronomy8080140





