Insights into the Positive Effect of Pyraclostrobin on Sugarcane Productivity
Abstract
1. Introduction
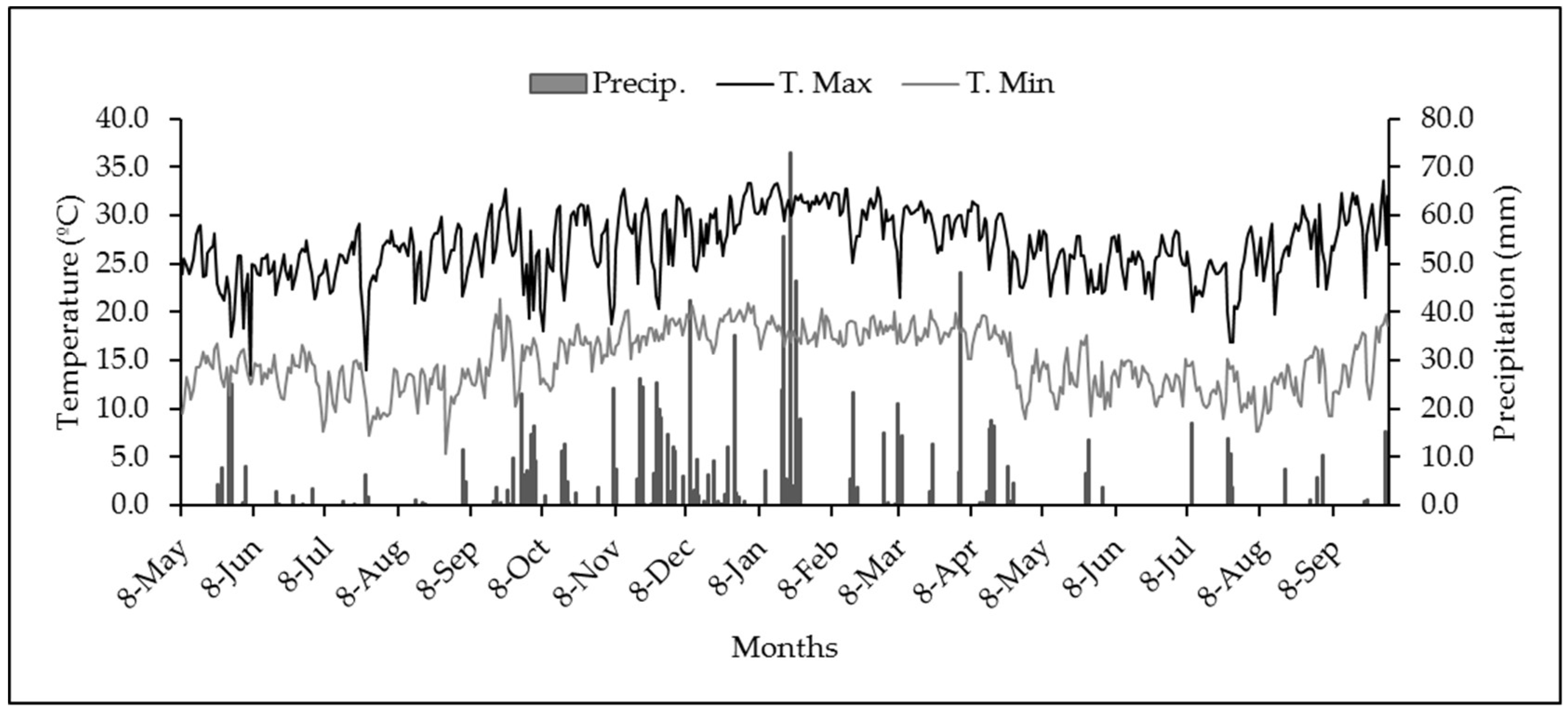
2. Materials and Methods
3. Results and Discussion
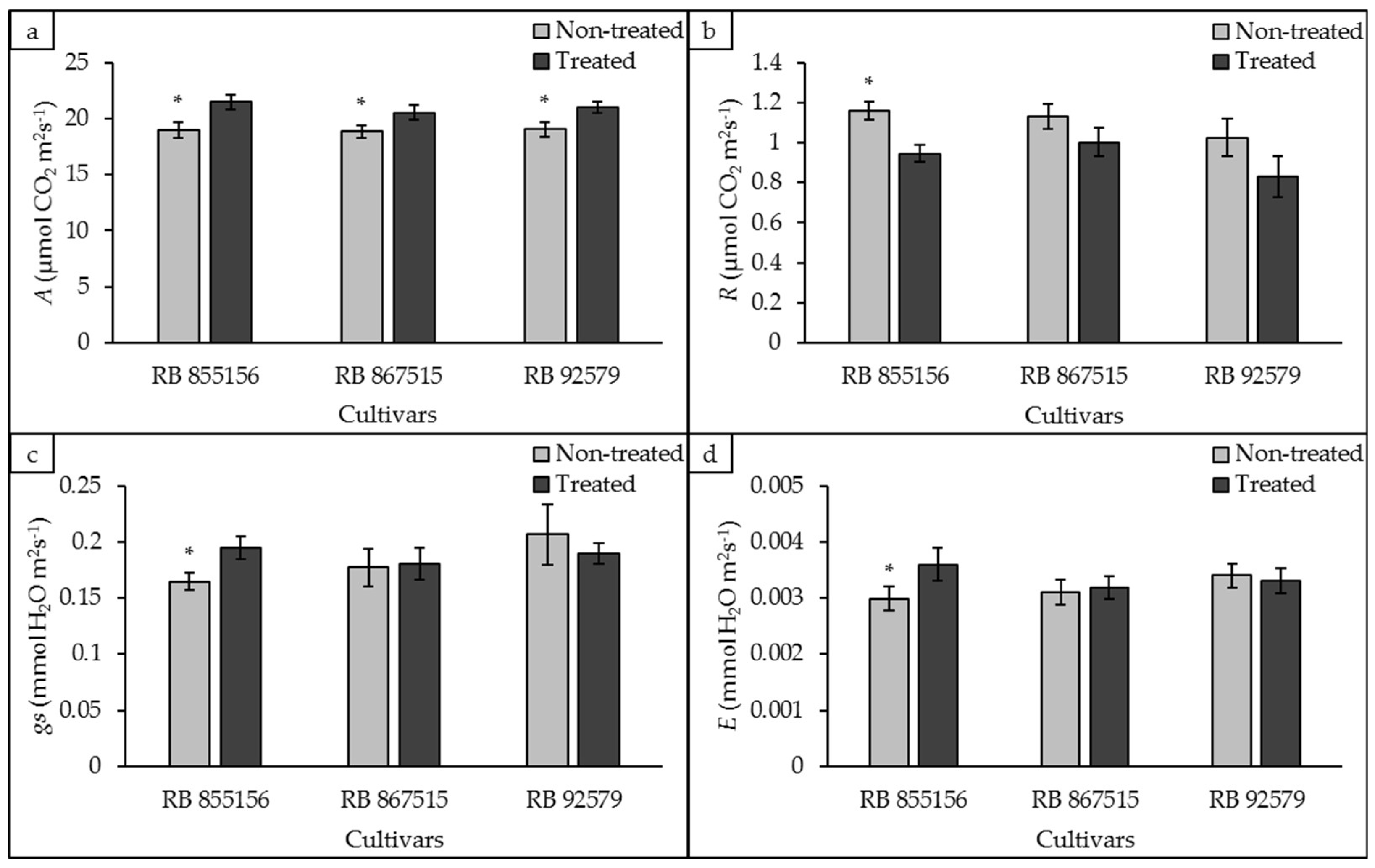
3.1. Gas Exchange
3.2. Chlorophyll Contents
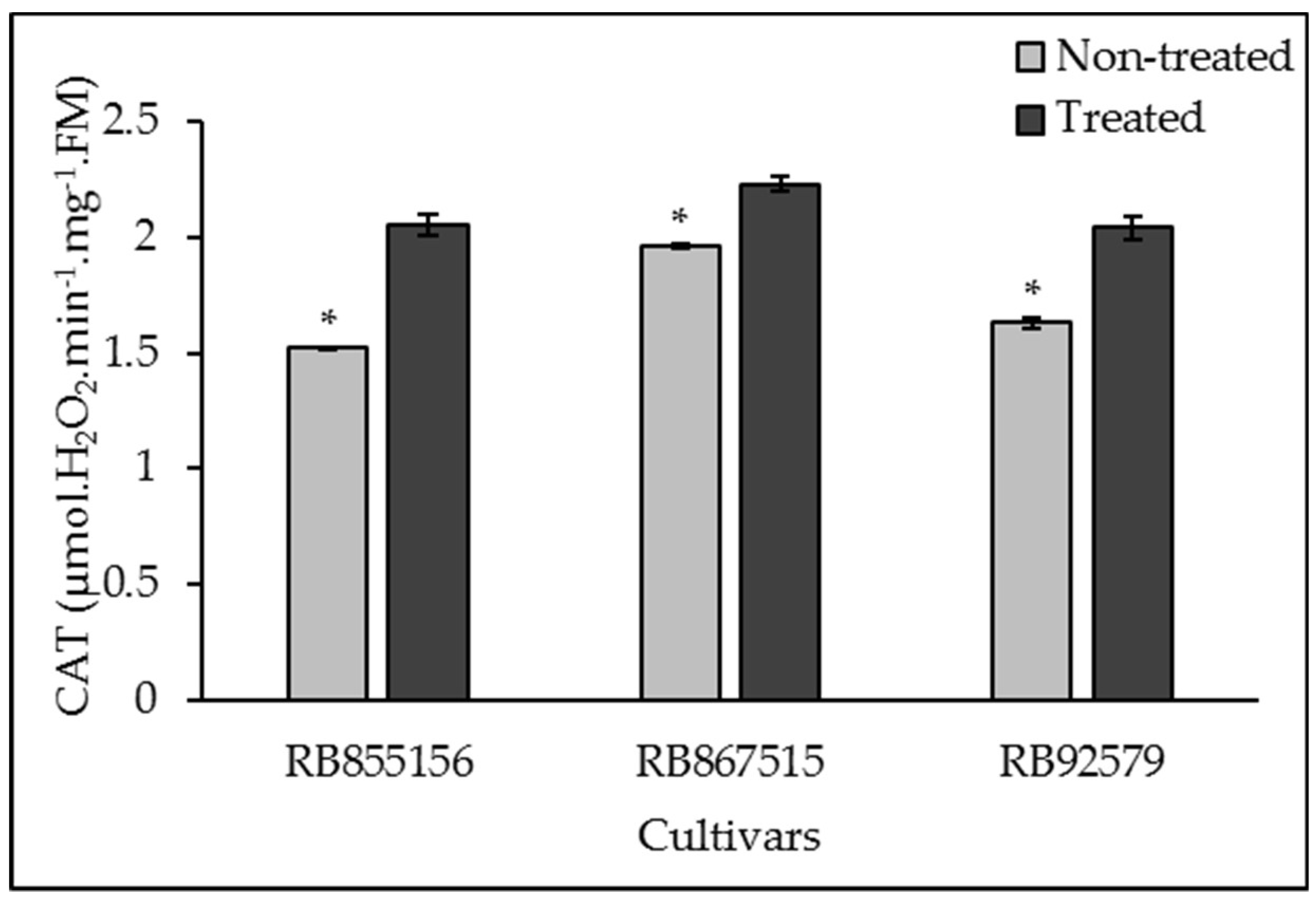
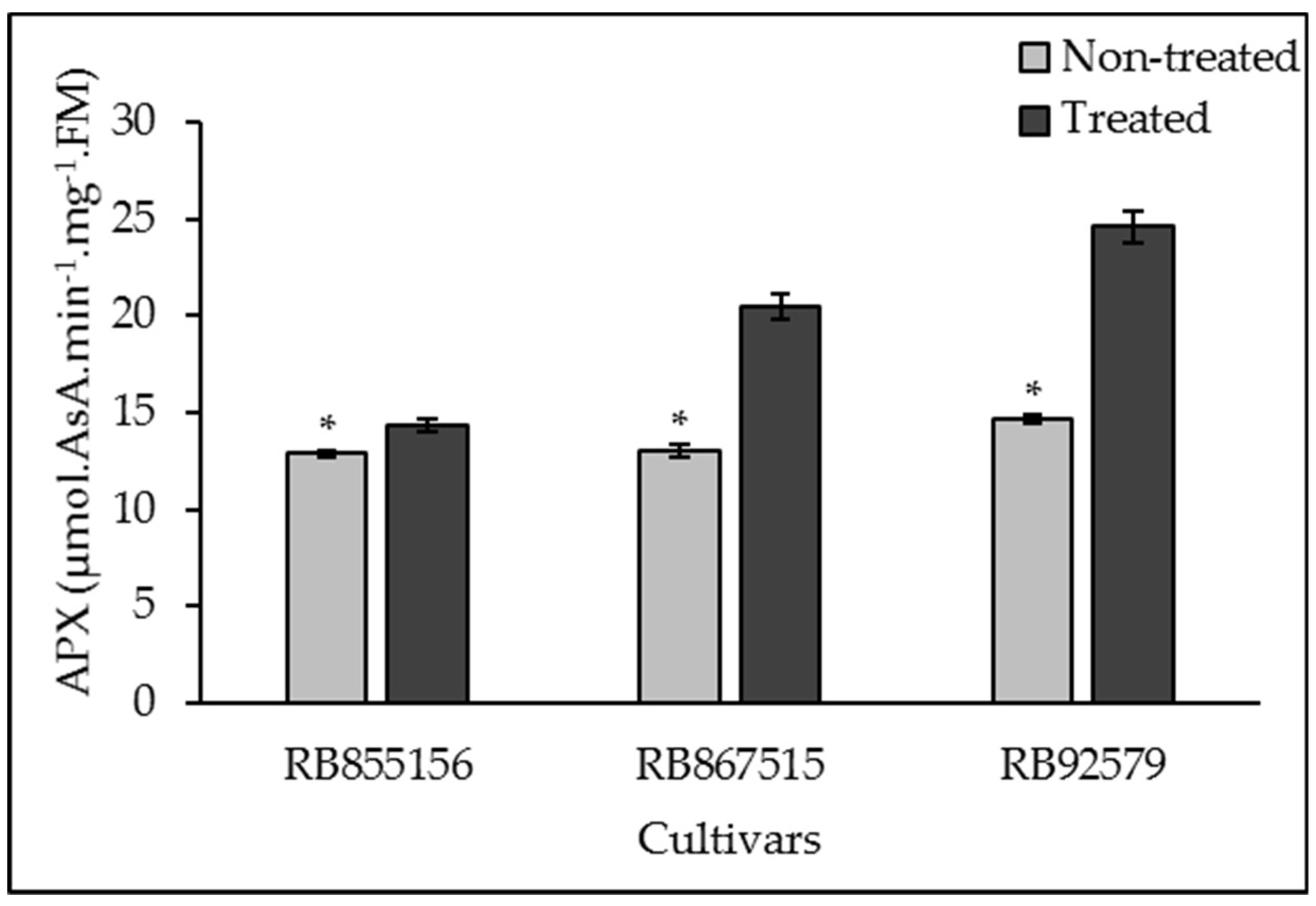
3.3. Antioxidant System
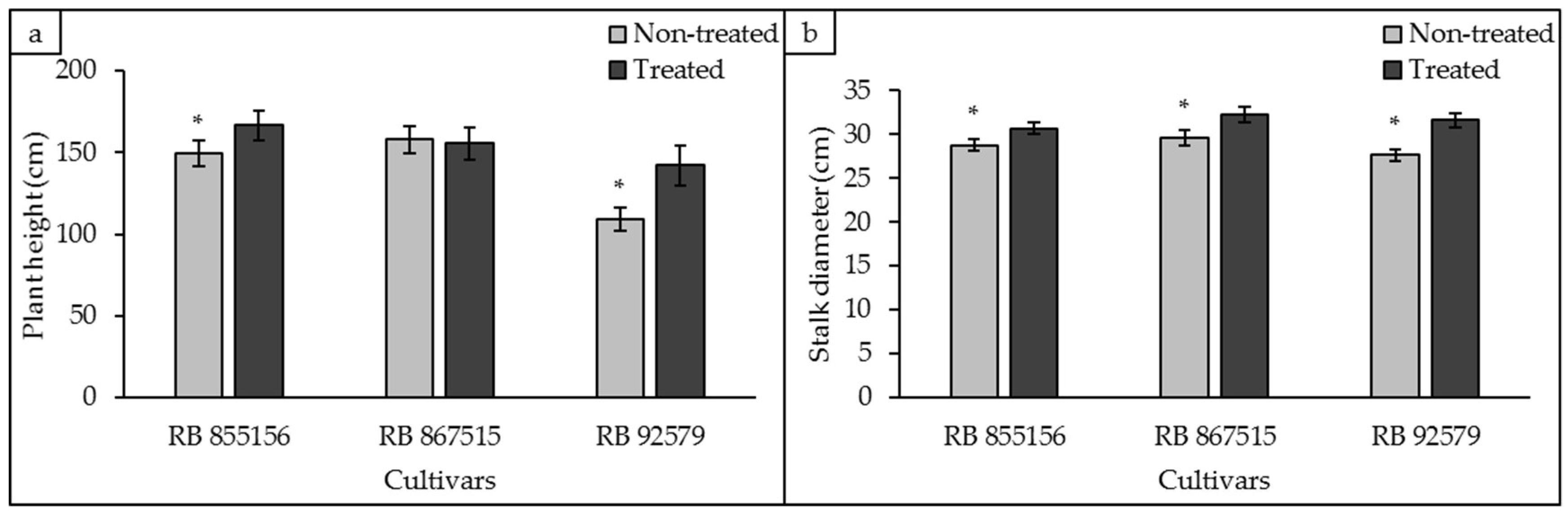
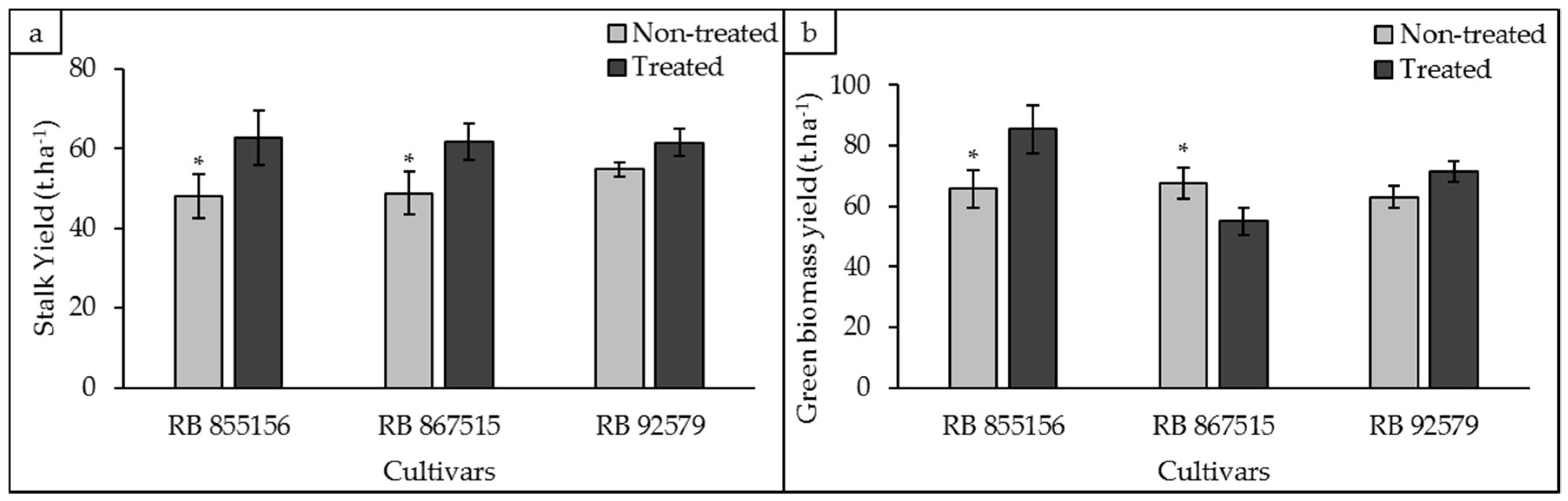
3.4. Biometric Analyses
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Berndes, G.; Hoogwijk, M.; van den Broek, R. The contribution of biomass in the future global energy supply: A review of 17 studies. Biomass Bioenergy 2003, 25, 1–28. [Google Scholar] [CrossRef]
- Haberl, H.; Beringer, T.; Bhattacharya, S.C.; Erb, K.-H.; Hoogwijk, M. The global technical potential of bio-energy in 2050 considering sustainability constraints. Curr. Opin. Environ. Sustain. 2010, 2, 394–403. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, L.A.H.; Lora, E.E.S. Dendroenergia: Fundamentos e Aplicaçôes; Editora Interciência: Rio de Janeiro, Brazil, 2003. [Google Scholar]
- Karp, A.; Shield, I. Bioenergy from plants and the sustainable yield challenge. New Phytol. 2008, 179, 15–32. [Google Scholar] [CrossRef] [PubMed]
- Waclawovsky, A.J.; Sato, P.M.; Lembke, C.G.; Moore, P.H.; Souza, G.M. Sugarcane for bioenergy production: An assessment of yield and regulation of sucrose content. Plant Biotechnol. J. 2010, 8, 263–276. [Google Scholar] [CrossRef] [PubMed]
- Inman-Bamber, N.; Smith, D. Water relations in sugarcane and response to water deficits. Field Crops Res. 2005, 92, 185–202. [Google Scholar] [CrossRef]
- Meschede, D.; Velini, E.D.; Carbonari, C.A.; Silva, J. Alteração fisiológica da cana-de-açúcar pela aplicação de glyphosate e sulfumeturon-methyl. Planta Daninha 2011, 29, 413–419. [Google Scholar] [CrossRef]
- Balba, H. Review of strobilurin fungicide chemicals. J. Environ. Sci. Health 2007, 42, 441–451. [Google Scholar] [CrossRef] [PubMed]
- Venancio, W.S.; Rodrigues, M.A.T.; Begliomini, E.; de Souza, N.L. Physiological effects of strobilurin fungicides on plants. Publicatio UEPG: Ciências Exatas e da Terra, Agrárias e Engenharias 2003, 9. [Google Scholar] [CrossRef]
- Kanungo, M.; Joshi, J. Impact of pyraclostrobin (f-500) on crop plants. Plant Sci. Today 2014, 1, 174–178. [Google Scholar] [CrossRef]
- Bartett, D.W.; Clough, J.M.; Godfrey, C.R.; Godwin, J.R.; Hall, A.A.; Heaney, S.P.; Maund, S.J. Understanding the strobilurin fungicides. Pesticide Outlook 2001, 12, 143–148. [Google Scholar] [CrossRef]
- Sauter, H.; Ammerman, E.; Benoit, R.; Brand, S.; Gold, R.E.; Grammenos, W.; Köhl, H.; Lorenz, G.; Muller, B.; Röhl, F.; et al. Mitochondrial respiration as a target for antifungals: Lessons learned from research on strobilurins. In Antifungal Agents—Discovery and Mode of Action; Dixon, G.K., Copping, L.G., Hollowman, D.W., Eds.; BIOS: Oxford, UK, 1995; pp. 173–191. [Google Scholar]
- Köhle, H.; Grossmann, K.; Jabs, T.; Gerhard, M.; Kaiser, W.; Glaab, J.; Conrath, U.; Seehaus, K.; Herms, S. Physiological effects of the strobilurin fungicide f 500 on plants. In Modern Fungicides and Antifungal Compounds III; Dehne, H.-W., Ed.; AgroConcept: Bonn, Germany, 2002; pp. 61–74. ISBN 3786201447. [Google Scholar]
- Bartlett, D.W.; Clough, J.M.; Godwin, J.R.; Hall, A.A.; Hamer, M.; Parr-Dobrzanski, B. The strobilurin fungicides. Pest Manag. Sci. 2002, 58, 649–662. [Google Scholar] [CrossRef] [PubMed]
- Fagan, E.B.; Dourado Neto, D.; Vivian, R.; Franco, R.B.; Yeda, M.P.; Massignam, L.F.; Oliveira, R.F.D.; Martins, K.V. Efeito da aplicação de piraclostrobina na taxa fotossintética, respiração, atividade da enzima nitrato redutase e produtividade de grãos de soja. Bragantia 2010, 69, 771–777. [Google Scholar] [CrossRef]
- Barbosa, K.A.; Fagan, E.B.; Casaroli, D.; de Carvalho Canedo, S.; Teixeira, W.F. Aplicação de estrobilurina na cultura do milho: Alterações fisiológicas e bromatológicas. Revista do Centro Universitário de Patos de Minas 2011, 2178, 7662. [Google Scholar]
- Nelson, K.A.; Meinhardt, C.G. Foliar boron and pyraclostrobin effects on corn yield. Agron. J. 2011, 103, 1352–1358. [Google Scholar] [CrossRef]
- Costa, R.V.D.; Cota, L.V.; Silva, D.D.D.; Meirelles, W.F.; Lanza, F.E. Viabilidade técnica e econômica da aplicação de estrobilurinas em milho. Trop. Plant Pathol. 2012, 37, 246–254. [Google Scholar] [CrossRef]
- Reboita, M.S.; Rodrigues, M.; Silva, L.F.; Alves, M.A. Aspectos clim ticos do estado de minas gerais (climate aspects in minas gerais state). Revista Brasileira de Climatologia 2015, 17. [Google Scholar] [CrossRef]
- Biemelt, S.; Keetman, U.; Albrecht, G. Re-aeration following hypoxia or anoxia leads to activation of the antioxidative defense system in roots of wheat seedlings. Plant Physiol. 1998, 116, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Giannopolitis, C.N.; Ries, S.K. Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol. 1977, 59, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Mengutay, M.; Ceylan, Y.; Kutman, U.B.; Cakmak, I. Adequate magnesium nutrition mitigates adverse effects of heat stress on maize and wheat. Plant Soil 2013, 368, 57–72. [Google Scholar] [CrossRef]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar]
- Arnon, D.I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. In Methods in Enzymology; Moore, B.S., Ed.; Academic Press: Cambridge, MA, USA, 1987; Volume 148, pp. 350–382. [Google Scholar]
- Grossmann, K.; Retzlaff, G. Bioregulatory effects of the fungicidal strobilurin kresoxim-methyl in wheat (triticum aestivum). Pestic. Sci. 1997, 50, 11–20. [Google Scholar] [CrossRef]
- Kluge, R.A.; Tezotto-Uliana, J.V.; da Silva, P.P. Aspectos fisiológicos e ambientais da fotossíntese. Revista Virtual de Química 2014, 7, 56–73. [Google Scholar]
- Vavasseur, A.; Raghavendra, A.S. Guard cell metabolism and co2 sensing. New Phytol. 2005, 165, 665–682. [Google Scholar] [CrossRef] [PubMed]
- Lima, J.D.; Moraes, W.d.S.; da Silva, M.-G.; Helena, S. Respostas fisiológicas em mudas de banananeira tratadas com estrobilurinas. Semina: Ciências Agrárias 2012, 33, 77–85. [Google Scholar] [CrossRef]
- Logan, B.A.; Adams, W.W.; Demmig-Adams, B. Viewpoint: Avoiding common pitfalls of chlorophyll fluorescence analysis under field conditions. Funct. Plant Biol. 2007, 34, 853–859. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Biochem. 2010, 48, 909–930. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-X.; von Tiedemann, A. Physiological effects of azoxystrobin and epoxiconazole on senescence and the oxidative status of wheat. Pest. Biochem. Physiol. 2001, 71, 1–10. [Google Scholar] [CrossRef]


| Time 1 | Chlorophyll a/b Ratio (Chlorophyll g−1 FM) | Carotenoid Content (µg Carotenoids g−1 FM) | ||
|---|---|---|---|---|
| Non-Treated | Treated | Non-Treated | Treated | |
| 0 | 2.96 aA * | 2.85 cA | 2.81 aB | 4.17 aA |
| 5 | 3.02 aA | 3.26 bcA | 2.52 aA | 2.40 bA |
| 15 | 3.39 aB | 3.96 abA | 3.53 aA | 2.62 bA |
| 25 | 3.38 aB | 4.18 aA | 2.62 aA | 2.27 bA |
| 35 | 3.54 aA | 3.15 bcA | 1.02 bA | 1.65 bA |
| 65 | 3.72 aA | 4.11 aA | 3.68 aA | 2.70 bB |
| Time 1 | Chlorophyll a/b Ratio (µg Chlorophyll g−1 FM) | Carotenoid Content(µg Carotenoids g−1 FM) | ||
|---|---|---|---|---|
| Non-Treated | Treated | Non-Treated | Treated | |
| 0 | 2.94 aA * | 2.54 aA | 3.53 aA | 3.01 abA |
| 5 | 3.17 aA | 2.97 aA | 2.53 bcB | 3.65 aA |
| 15 | 3.72 aA | 3.59 aA | 2.78 abA | 3.67 aA |
| 25 | 3.75 aA | 3.88 aA | 2.49 bcA | 2.39 bcA |
| 35 | 3.03 aA | 2.85 aA | 1.67 cA | 1.42 cA |
| 65 | 3.60 aA | 3.72 aA | 3.66 aA | 2.52 bB |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreira Lopes, A.; Schumacher, P.V.; Terylene Pérez Martínez, A.; Paulino da Costa Netto, A.; Chalfun-Junior, A. Insights into the Positive Effect of Pyraclostrobin on Sugarcane Productivity. Agronomy 2018, 8, 122. https://doi.org/10.3390/agronomy8070122
Moreira Lopes A, Schumacher PV, Terylene Pérez Martínez A, Paulino da Costa Netto A, Chalfun-Junior A. Insights into the Positive Effect of Pyraclostrobin on Sugarcane Productivity. Agronomy. 2018; 8(7):122. https://doi.org/10.3390/agronomy8070122
Chicago/Turabian StyleMoreira Lopes, Amanda, Pedro Vitor Schumacher, Aurora Terylene Pérez Martínez, Antônio Paulino da Costa Netto, and Antonio Chalfun-Junior. 2018. "Insights into the Positive Effect of Pyraclostrobin on Sugarcane Productivity" Agronomy 8, no. 7: 122. https://doi.org/10.3390/agronomy8070122
APA StyleMoreira Lopes, A., Schumacher, P. V., Terylene Pérez Martínez, A., Paulino da Costa Netto, A., & Chalfun-Junior, A. (2018). Insights into the Positive Effect of Pyraclostrobin on Sugarcane Productivity. Agronomy, 8(7), 122. https://doi.org/10.3390/agronomy8070122






