Characterization of Root and Shoot Traits in Wheat Cultivars with Putative Differences in Root System Size
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Growth Conditions
2.2. Root Traits: Non-Destructive Measurements
2.3. Destructive Measurements
2.4. Statistical Analysis
3. Results
3.1. Shoot Traits
3.2. Root Traits
3.3. Correlation between Traits
4. Discussion
4.1. Characterising the Root Systems of Cultivars with Contrasting Root System Sizes
4.2. Negative Relationship Between Phenology and Root System Size
4.3. The Relationship Between Root System Size and Shoot Traits
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Palta, J.A.; Chen, X.; Milroy, S.P.; Rebetzke, G.J.; Dreccer, M.F.; Watt, M. Large root systems: Are they useful in adapting wheat to dry environments? Funct. Plant Biol. 2011, 38, 347–354. [Google Scholar] [CrossRef]
- Palta, J.A.; Watt, M. Vigorous crop root systems: Form and function for improving the capture of water and nutrients. In Applied Crop Physiology: Boundaries between Genetic Improvement and Agronomy; VO Sadras, D.C., Ed.; Academic Press: San Diego, CA, USA, 2009; pp. 309–325. [Google Scholar]
- Loes, A.; Gahoonia, T.S. Genetic variation in specific root length in scandinavian wheat and barley accessions. Euphytica 2004, 137, 243–249. [Google Scholar] [CrossRef]
- Botwright Acuña, T.L.; Rebetzke, G.J.; He, X.; Maynol, E.; Wade, L.J. Mapping quantitative trait loci associated with root penetration ability of wheat in contrasting environments. Mol. Breed. 2014, 34, 631–642. [Google Scholar] [CrossRef]
- Hirel, B.; Le Gouis, J.; Ney, B.; Gallais, A. The challenge of improving nitrogen use efficiency in crop plants: Towards a more central role for genetic variability and quantitative genetics within integrated approaches. J. Exp. Bot. 2007, 58, 2369–2387. [Google Scholar] [CrossRef] [PubMed]
- Anbessa, Y.; Juskiw, P. Nitrogen fertilizer rate and cultivar interaction effects on nitrogen recovery, utilization efficiency, and agronomic performance of spring barley. ISRN Agron. 2012, 2012, 531647. [Google Scholar] [CrossRef]
- Wasson, A.P.; Richards, R.A.; Chatrath, R.; Misra, S.C.; Prasad, S.V.; Rebetzke, G.J.; Kirkegaard, J.A.; Christopher, J.; Watt, M. Traits and selection strategies to improve root systems and water uptake in water-limited wheat crops. J. Exp. Bot. 2012, 63, 3485–3498. [Google Scholar] [CrossRef] [PubMed]
- Hamblin, A.; Tennant, D. Root length density and water uptake in cereals and grain legumes: How well are they correlated? Aust. J. Agric. Res. 1987, 38, 513–527. [Google Scholar] [CrossRef]
- Lilley, J.M.; Kirkegaard, J.A. Benefits of increased soil exploration by wheat roots. Field Crop. Res. 2011, 122, 118–130. [Google Scholar] [CrossRef]
- Palta, J.A.; Gregory, P.J. Drought affects the fluxes of carbon to roots and soil in 13c pulse-labelled plants of wheat. Soil Biol. Biochem. 1997, 29, 1395–1403. [Google Scholar] [CrossRef]
- Liao, M.; Fillery, I.R.P.; Palta, J.A. Early vigorous growth is a major factor influencing nitrogen uptake in wheat. Funct. Plant Biol. 2004, 31, 121–129. [Google Scholar] [CrossRef]
- Liao, M.; Palta, J.A.; Fillery, I.R.P. Root characteristics of vigorous wheat improve early nitrogen uptake. Aust. J. Agric. Res. 2006, 57, 1097–1107. [Google Scholar] [CrossRef]
- Palta, J.A.; Yang, J. Crop root system behaviour and yield. Field Crop. Res. 2014, 165, 1–4. [Google Scholar] [CrossRef]
- Waines, J.G.; Ehdaie, B. Domestication and crop physiology: Roots of green-revolution wheat. Ann. Bot. 2007, 100, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Aziz, M.M.; Palta, J.A.; Siddique, K.H.M.; Sadras, V.O. Five decades of selection for yield reduced root length density and increased nitrogen uptake per unit root length in australian wheat varieties. Plant Soil 2016, 413, 181–192. [Google Scholar] [CrossRef]
- Benlloch-Gonzalez, M.; Bochicchio, R.; Berger, J.; Bramley, H.; Palta, J.A. High temperature reduces the positive effect of elevated CO2 on wheat root system growth. Field Crop. Res. 2014, 165, 71–79. [Google Scholar] [CrossRef]
- Nagel, K.A.; Putz, A.; Gilmer, F.; Heinz, K.; Fischbach, A.; Pfeifer, J.; Faget, M.; Blossfeld, S.; Ernst, M.; Dimaki, C.; et al. Growscreen-rhizo is a novel phenotyping robot enabling simultaneous measurements of root and shoot growth for plants grown in soil-filled rhizotrons. Funct. Plant Biol. 2012, 39, 891–904. [Google Scholar] [CrossRef]
- Judd, L.A.; Jackson, B.E.; Fonteno, W.C. Advancements in root growth measurement technologies and observation capabilities for container-grown plants. Plants 2015, 4, 369–392. [Google Scholar] [CrossRef] [PubMed]
- Benlloch-Gonzalez, M.; Berger, J.; Bramley, H.; Rebetzke, G.J.; Palta, J.A. The plasticity of the growth and proliferation of wheat root system under elevated CO2. Plant Soil 2014, 374, 963–976. [Google Scholar] [CrossRef]
- Palta, J.A.; Fillery, I.R.P.; Rebetzke, G.J. Restricted-tillering wheat does not lead to greater investment in roots and early nitrogen uptake. Field Crop. Res. 2007, 104, 52–59. [Google Scholar] [CrossRef]
- Dieffenbach, A.; Gottlein, A.G.; Matzner, E. In-situ soil solution chemistry in an acid forest soil as influenced by growing roots of norway spruce (Picea abies [L.] Karst.). Plant Soil 1997, 192, 57–61. [Google Scholar] [CrossRef]
- Dessureault-Rompré, J.; Nowack, B.; Schulin, R.; Luster, J. Modified micro suction cup/rhizobox approach for the in-situ detection of organic acids in rhizosphere soil solution. Plant Soil 2006, 286, 99–107. [Google Scholar] [CrossRef]
- Pfeifer, J. Elucidation of Root-Soil Interactions of Crops in Space and Time by Establishment and Application of Novel Image Based Non-Invasive Root Phenotyping Methods. Ph.D. Thesis, ETH Zurich, Zürich, Switzerland, 2013. [Google Scholar]
- Adu, M.O.; Chatot, A.; Wiesel, L.; Bennett, M.J.; Broadley, M.R.; White, P.J.; Dupuy, L.X. A scanner system for high-resolution quantification of variation in root growth dynamics of brassica rapa genotypes. J. Exp. Bot. 2014, 65, 2039–2048. [Google Scholar] [CrossRef] [PubMed]
- Isbell, R.F. A Classification System for Australian Soils (Third Approximation); Technical Report 2/1993; CSIRO: Townsville, Australia, 1993. [Google Scholar]
- Flower, K.C.; Cordingley, N.; Ward, P.R.; Weeks, C. Nitrogen, weed management and economics with cover crops in conservation agriculture in a mediterranean climate. Field Crop. Res. 2012, 132, 63–75. [Google Scholar] [CrossRef]
- Ludwig, F.; Milroy, S.P.; Asseng, S. Impacts of recent climate change on wheat production systems in western australia. Clim. Chang. 2008, 92, 495–517. [Google Scholar] [CrossRef]
- Lemerle, D.; Cousens, R.D.; Gill, G.S.; Peltzer, S.J.; Moerkerk, M.; Murphy, C.E.; Collins, D.; Cullis, B.R. Reliability of higher seeding rates of wheat for increased competitiveness with weeds in low rainfall environments. J. Agric. Sci. 2004, 142, 395–409. [Google Scholar] [CrossRef]
- Zadoks, J.C.; Chang, T.T.; Konzak, C.F. A decimal code for the growth stages of cereals. Weed Res. 1974, 14, 415–421. [Google Scholar] [CrossRef]
- Mahfoozi, S.; Limin, A.E.; Ahakpaz, F.; Fowler, D.B. Phenological development and expression of freezing resistance in spring and winter wheat under field conditions in north-west iran. Field Crop. Res. 2006, 97, 182–187. [Google Scholar] [CrossRef]
- Semenov, M.A.; Stratonovitch, P.; Alghabari, F.; Gooding, M.J. Adapting wheat in europe for climate change. J. Cereal Sci. 2014, 59, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Asseng, S.; Foster, I.A.N.; Turner, N.C. The impact of temperature variability on wheat yields. Glob. Chang. Biol. 2011, 17, 997–1012. [Google Scholar] [CrossRef]
- Sadras, V.O.; Monzon, J.P. Modelled wheat phenology captures rising temperature trends: Shortened time to flowering and maturity in australia and argentina. Field Crop. Res. 2006, 99, 136–146. [Google Scholar] [CrossRef]
- Palta, J.A.; Fillery, I.R. Nitrogen accumulation and remobilisation in wheat of 15n -urea applied to a duplex soil at seeding. Aust. J. Exp. Agric. 1993, 33, 233–238. [Google Scholar] [CrossRef]
- Chen, Y.; Ghanem, M.; Siddique, K.H.M. Characterising root trait variability in chickpea (cicer arietinuml.) germplasm. J. Exp. Bot. 2016, 68, 1987–1999. [Google Scholar] [CrossRef] [PubMed]
- Mendiburu, F. Agricolae: Statistical Procedures for Agricultural Research. R package Version 1. 0-9. Available online: http://CRAN.R-project.org/package=agricolae (accessed on 24 October 2017).
- R Development Core Team. R: A Language and Environment for Statistical Computing. Available online: http://www.R-project.org/ (accessed on 27 September 2017).
- Becker, S.R.; Byrne, P.F.; Reid, S.D.; Bauerle, W.L.; McKay, J.K.; Haley, S.D. Root traits contributing to drought tolerance of synthetic hexaploid wheat in a greenhouse study. Euphytica 2015, 207, 213–224. [Google Scholar] [CrossRef]
- Botwright Acuña, T.L.; Wade, L.J. Genotype×environment interactions for root depth of wheat. Field Crop. Res. 2012, 137, 117–125. [Google Scholar] [CrossRef]
- Siddique, K.H.M.; Belford, R.K.; Tennant, D. Root:Shoot ratios of old and modern, tall and semi-dwarf wheats in a mediterranean environment. Plant Soil 1990, 121, 89–98. [Google Scholar] [CrossRef]
- Xie, Q.; Fernando, K.M.; Mayes, S.; Sparkes, D.L. Identifying seedling root architectural traits associated with yield and yield components in wheat. Ann. Bot. 2017, 119, 1115–1129. [Google Scholar] [CrossRef] [PubMed]
- Saradadevi, R.; Bramley, H.; Palta, J.A.; Siddique, K.H.M. Stomatal behaviour under terminal drought affects post-anthesis water use in wheat. Funct. Plant Biol. 2017, 44, 279–289. [Google Scholar] [CrossRef]
- Ehdaie, B.; Merhaut, D.J.; Ahmadian, S.; Hoops, A.C.; Khuong, T.; Layne, A.P.; Waines, J.G. Root system size influences water-nutrient uptake and nitrate leaching potential in wheat. J. Agron. Crop. Sci. 2010, 196, 455–466. [Google Scholar] [CrossRef]
- Watt, M.; Moosavi, S.; Cunningham, S.C.; Kirkegaard, J.A.; Rebetzke, G.J.; Richards, R.A. A rapid, controlled-environment seedling root screen for wheat correlates well with rooting depths at vegetative, but not reproductive, stages at two field sites. Ann. Bot. 2013, 112, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, J.A.; Wingen, L.U.; Griffiths, M.; Pound, M.P.; Gaju, O.; Foulkes, M.J.; Le Gouis, J.; Griffiths, S.; Bennett, M.J.; King, J.; et al. Phenotyping pipeline reveals major seedling root growth qtl in hexaploid wheat. J. Exp. Bot. 2015, 66, 2283–2292. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Du, Y.L.; Wang, J.; Wu, A.; Qiao, S.; Xu, B.; Zhang, S.; Siddique, K.H.M.; Chen, Y. Moderate drought stress affected root growth and grain yield in old, modern and newly released cultivars of winter wheat. Front. Plant Sci. 2017, 8, 672. [Google Scholar] [CrossRef] [PubMed]
- Spitters, C.J.T.; Kramer, T.H. Differences between spring wheat cultivars in early growth. Euphytica 1986, 35, 273–292. [Google Scholar] [CrossRef]
- Brooking, I.R.; Jamieson, P.D. Temperature and photoperiod response of vernalization in near-isogenic lines of wheat. Field Crop. Res. 2002, 79, 21–38. [Google Scholar] [CrossRef]
- McMaster, G.S.; White, J.W.; Hunt, L.A.; Jamieson, P.D.; Dhillon, S.S.; Ortiz-Monasterio, J.I. Simulating the influence of vernalization, photoperiod and optimum temperature on wheat developmental rates. Ann. Bot. 2008, 102, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Porter, J.R.; Gawith, M. Temperatures and the growth and development of wheat: A review. Eur. J. Agron. 2008, 10, 23–36. [Google Scholar] [CrossRef]
- Gregory, P.J.; Palta, J.A.; Batts, G.R. Root systems and root: Mass ratio-carbon allocation under current and projected atmospheric conditions in arable crops. Plant Soil 1997, 187, 221–228. [Google Scholar] [CrossRef]
- Watt, M.; Schneebeli, K.; Dong, P.; Wilson, I.W. The shoot and root growth of brachypodium and its potential as a model for wheat and other cereal crops. Funct. Plant Biol. 2009, 36, 960–969. [Google Scholar] [CrossRef]
- Watt, M.; Kirkegaard, J.A.; Rebetzke, G.J. A wheat genotype developed for rapid leaf growth copes well with the physical and biological constraints of unploughed soil. Funct. Plant Biol. 2005, 32, 695–706. [Google Scholar] [CrossRef]
- Rajala, A.; Peltonen-Sainio, P. Plant growth regulator effects on spring cereal root and shoot growth. Agron. J. 2001, 93, 936–943. [Google Scholar] [CrossRef]
- Pang, J.; Milroy, S.P.; Rebetzke, G.J.; Palta, J.A. The influence of shoot and root size on nitrogen uptake in wheat is affected by nitrate affinity in the roots during early growth. Funct. Plant Biol. 2015, 42, 1179–1189. [Google Scholar] [CrossRef]
- Passioura, J.B. Roots and drought resistance. Agric. Water Manag. 1983, 7, 265–280. [Google Scholar] [CrossRef]
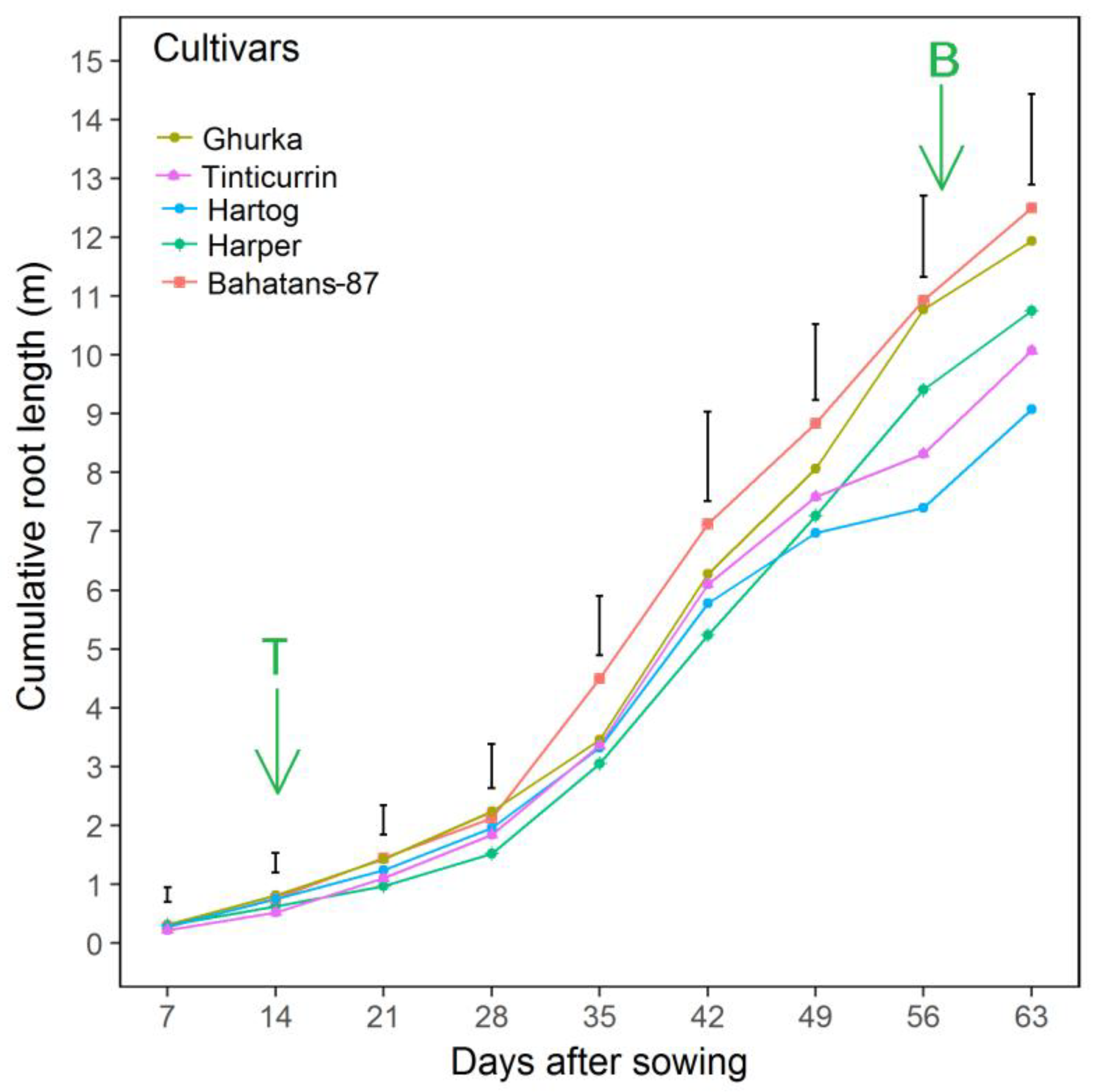

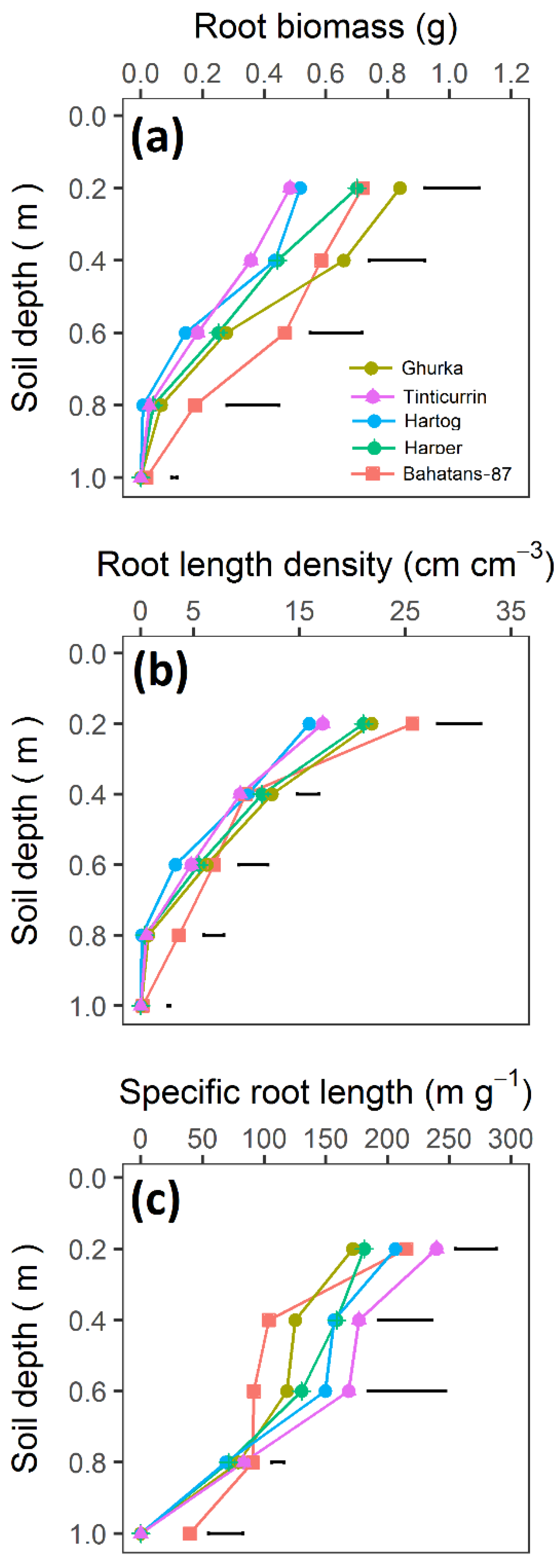
| Cultivars | Country of Origin | Year of Release | Total Root Biomass | Cumulative Root Length |
|---|---|---|---|---|
| Ghurka | Australia | 1916 | Large | Large |
| Tincurrin | Australia | 1978 | Small | Small |
| Hartog | Australia | 1983 | Medium | Medium |
| Harper | Australia | 2010 | Small | Small |
| Bahatans-87 | Algeria | 1924 | Large | Large |
| Cultivars | Time to Tillering (Z2.1) | Time to Booting (Z4.9) | Time to Anthesis (Z6.1) |
|---|---|---|---|
| (DAS) | (DAS) | (DAS) | |
| Ghurka | 16 | 92 a | 104 a |
| Tincurrin | 17 | 59 c | 75 b |
| Hartog | 15 | 57 c | 67 b |
| Harper | 18 | 85 b | 104 a |
| Bahatans 87 | 14 | 95 a | 107 a |
| LSD p < 0.05 | ns | 4.2 | 6.9 |
| Cultivars | LA | SLA | LB | Tillers | Shoot biomass | H |
|---|---|---|---|---|---|---|
| (cm2 plant−1) | (cm2 g−1) | (g plant−1) | (tillers plant−1) | (g plant−1) | (cm) | |
| Ghurka | 555 ab | 277 c | 2.00 ab | 5.3 b | 3.2 b | 44 b |
| Tincurrin | 493 bc | 312 a | 1.58b c | 5.7 b | 4.6 ab | 65 a |
| Hartog | 402 c | 267 cd | 1.51 c | 4.6 b | 5.1 a | 70 a |
| Harper | 491 bc | 255 d | 1.93 abc | 4.8 b | 3.5 b | 43 b |
| Bahatans-87 | 627 a | 295 b | 2.12 a | 8.8 a | 3.3 b | 46 b |
| LSD p < 0.05 | 113 | 14 | 0.42 | 2.1 | 1.4 | 7.1 |
| Cultivars | Root Length | Root Biomass | Specific Root Length | Root Length Density | Shoot Biomass | Root:Shoot Ratio |
|---|---|---|---|---|---|---|
| (m plant−1) | (g plant−1) | (m g−1) | (cm cm−3) | (g plant−1) | ||
| Ghurka | 273 a | 1.94 a | 142 c | 11.37 ab | 3.2 b | 0.62 a |
| Tincurrin | 211 b | 1.05 b | 202 a | 9.75 bc | 4.6 ab | 0.23 c |
| Hartog | 199 b | 1.10 b | 178 b | 9.30 c | 5.1 a | 0.22 c |
| Harper | 230 ab | 1.44 b | 162 bc | 11.64 a | 3.5 b | 0.41 b |
| Bahatans-87 | 278 a | 2.03 a | 139 c | 10.84 abc | 3.3 b | 0.61 a |
| LSD p < 0.05 | 56 | 0.47 | 35 | 1.85 | 1.4 | 0.12 |
| Traits | RL | MRL | RB | SRL | RLD | R:S | LA | SLA | LB | SB | T |
|---|---|---|---|---|---|---|---|---|---|---|---|
| MRL | 0.82 *** | ||||||||||
| RB | 0.87 *** | 0.80 *** | |||||||||
| SRL | −0.50 | −0.51 | −0.84 *** | ||||||||
| RLD | 0.32 | 0.08 | 0.39 | −0.48 | |||||||
| R:S | 0.72 ** | 0.68 ** | 0.92 *** | −0.87 *** | 0.61 ** | ||||||
| LA | 0.86 *** | 0.92 *** | 0.77 *** | −0.43 | 0.12 | 0.62 | |||||
| SLA | −0.03 | 0.13 | −0.07 | 0.21 | −0.22 | −0.05 | 0.28 | ||||
| LB | 0.90 *** | 0.88 **** | 0.83 *** | −0.53 | 0.23 | 0.66 ** | 0.91 *** | −0.15 | |||
| SB | −0.19 | −0.26 | −0.46 | 0.65 ** | −0.74 ** | −0.75 ** | −0.22 | −0.02 | −0.22 | ||
| T | 0.64 | 0.61 | 0.63 | −0.41 | 0.10 | 0.51 | 0.75 ** | 0.39 | 0.59 | −0.09 | |
| H | −0.56 | −0.53 | −0.67 ** | 0.70 ** | −0.83 *** | −0.83 *** | −0.49 | 0.24 | −0.61 | 0.81 *** | −0.29 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Figueroa-Bustos, V.; Palta, J.A.; Chen, Y.; Siddique, K.H.M. Characterization of Root and Shoot Traits in Wheat Cultivars with Putative Differences in Root System Size. Agronomy 2018, 8, 109. https://doi.org/10.3390/agronomy8070109
Figueroa-Bustos V, Palta JA, Chen Y, Siddique KHM. Characterization of Root and Shoot Traits in Wheat Cultivars with Putative Differences in Root System Size. Agronomy. 2018; 8(7):109. https://doi.org/10.3390/agronomy8070109
Chicago/Turabian StyleFigueroa-Bustos, Victoria, Jairo A. Palta, Yinglong Chen, and Kadambot H.M. Siddique. 2018. "Characterization of Root and Shoot Traits in Wheat Cultivars with Putative Differences in Root System Size" Agronomy 8, no. 7: 109. https://doi.org/10.3390/agronomy8070109
APA StyleFigueroa-Bustos, V., Palta, J. A., Chen, Y., & Siddique, K. H. M. (2018). Characterization of Root and Shoot Traits in Wheat Cultivars with Putative Differences in Root System Size. Agronomy, 8(7), 109. https://doi.org/10.3390/agronomy8070109







