Straw Application Strategy to Optimize Nutrient Release in a Southeastern China Rice Cropland
Abstract
1. Introduction
2. Results
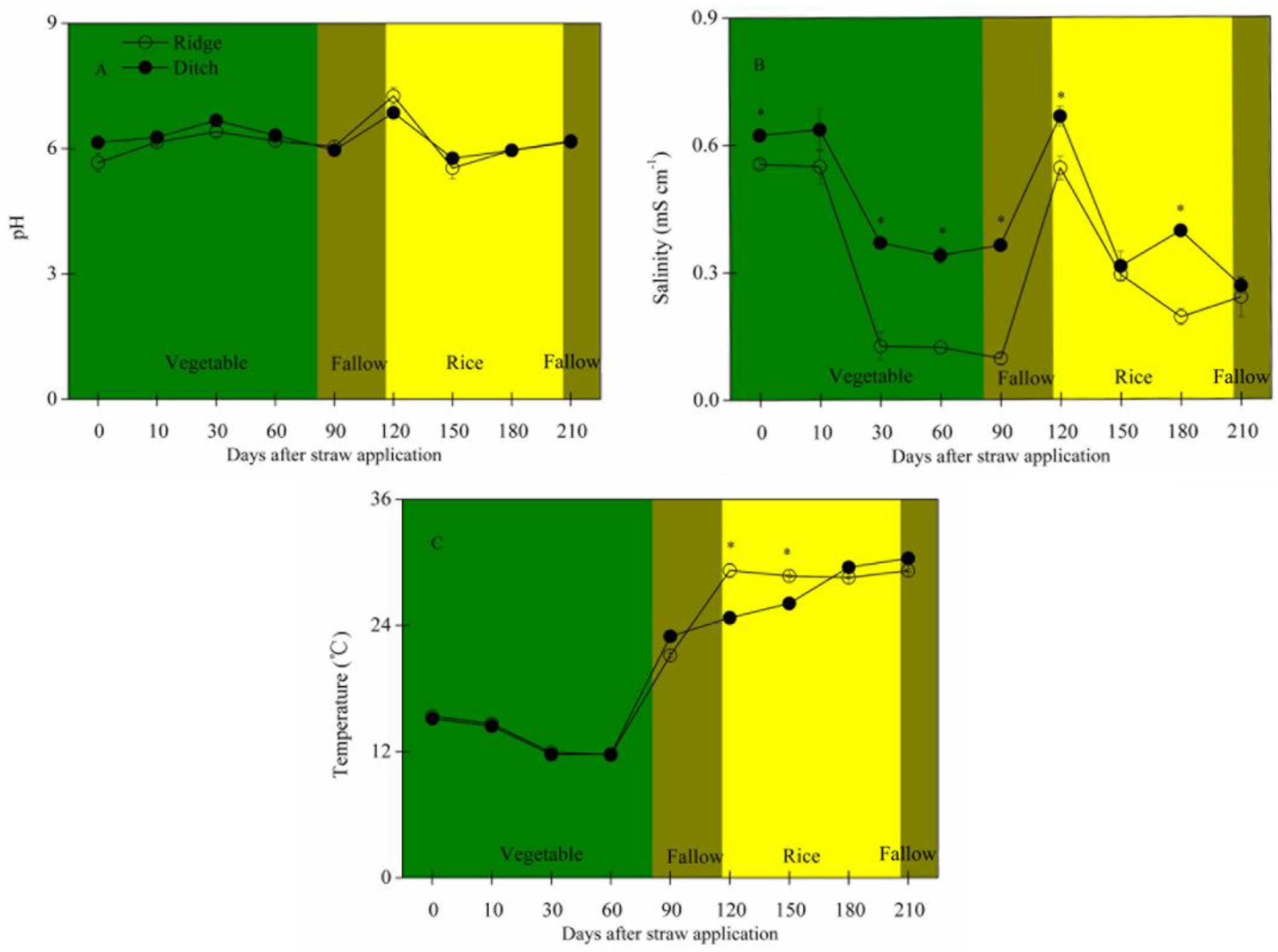
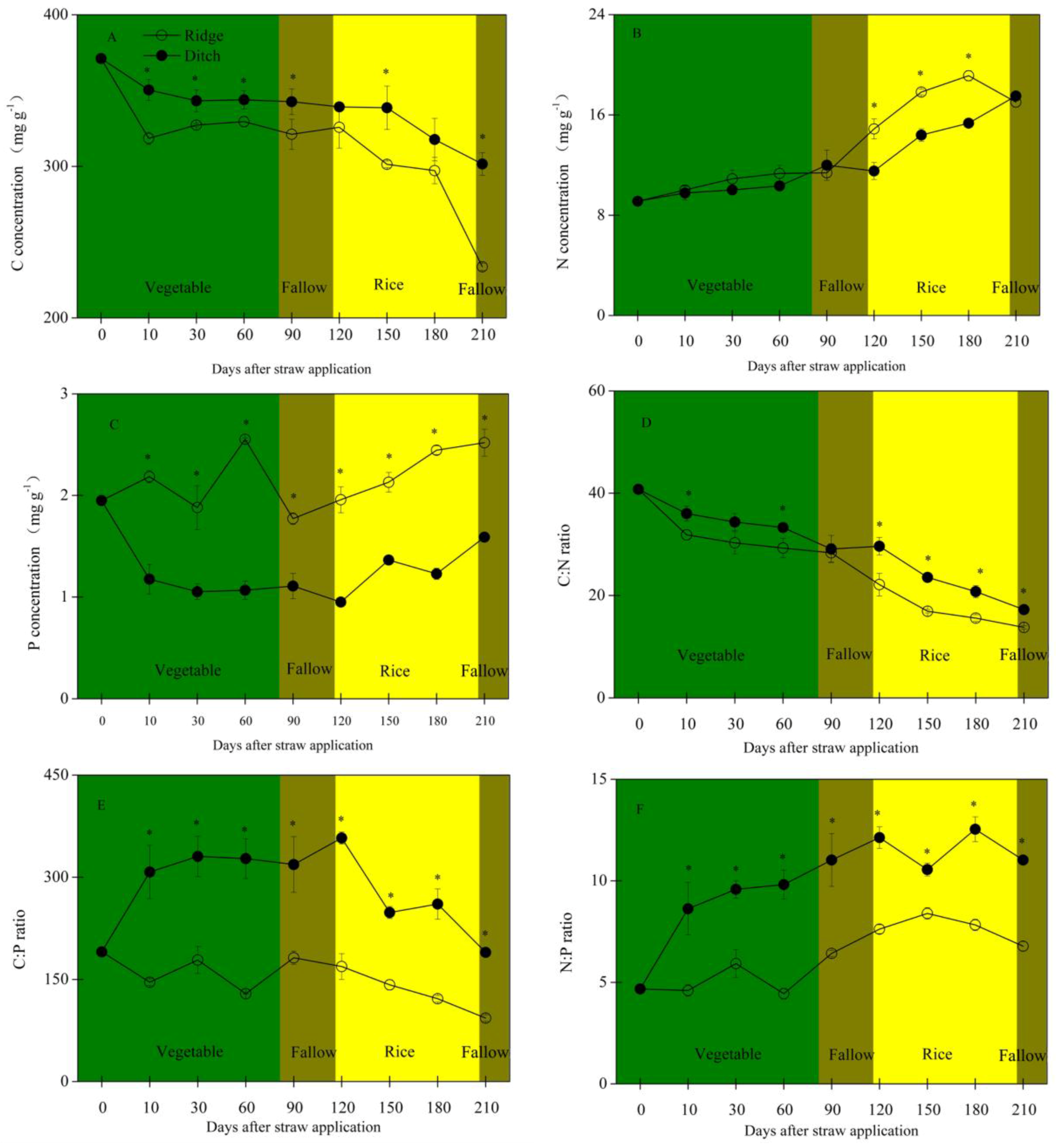
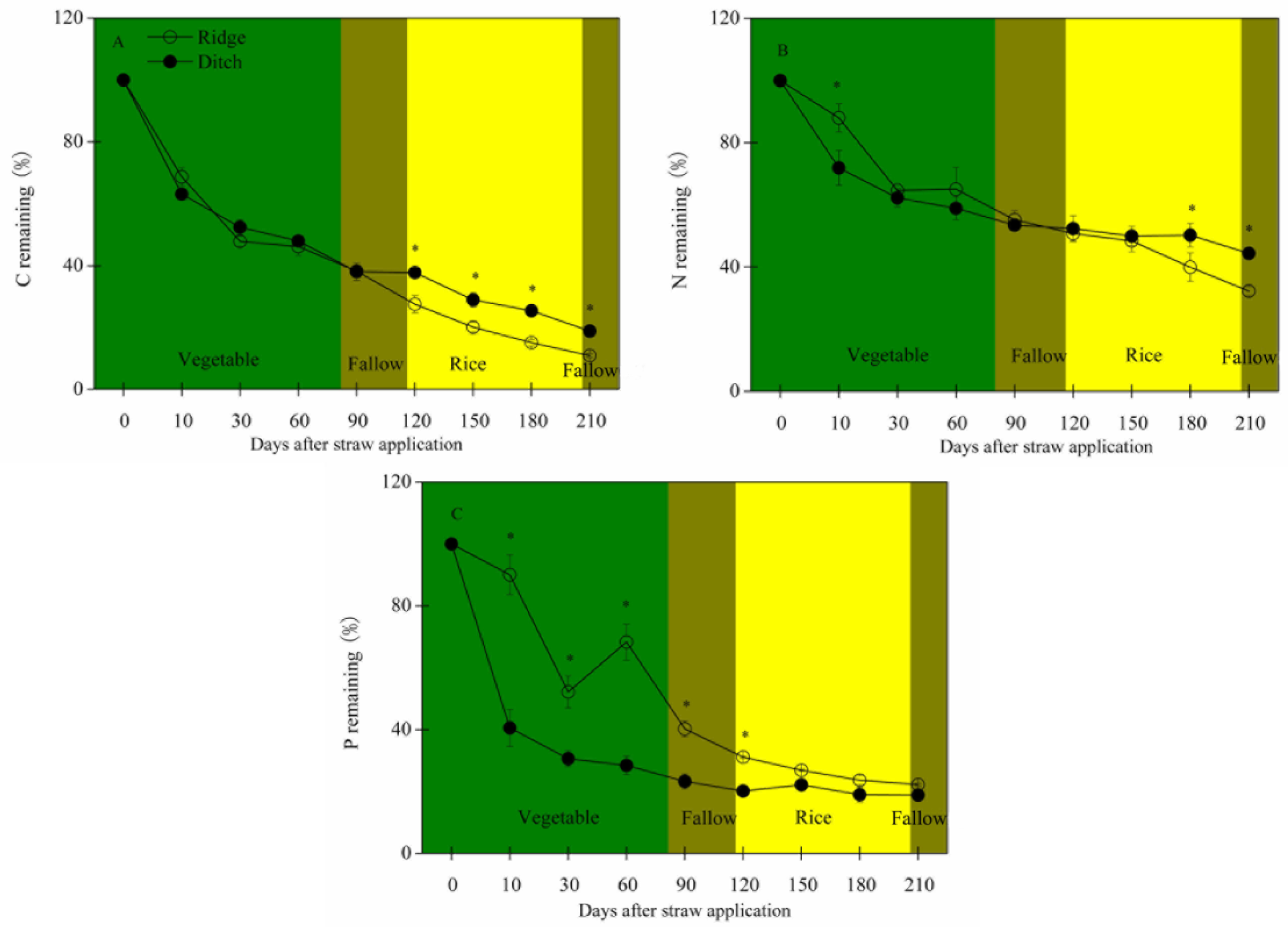
2.1. Straw Mass Remaining, Nutrient Concentration and Stoichiometry, and Soil Traits
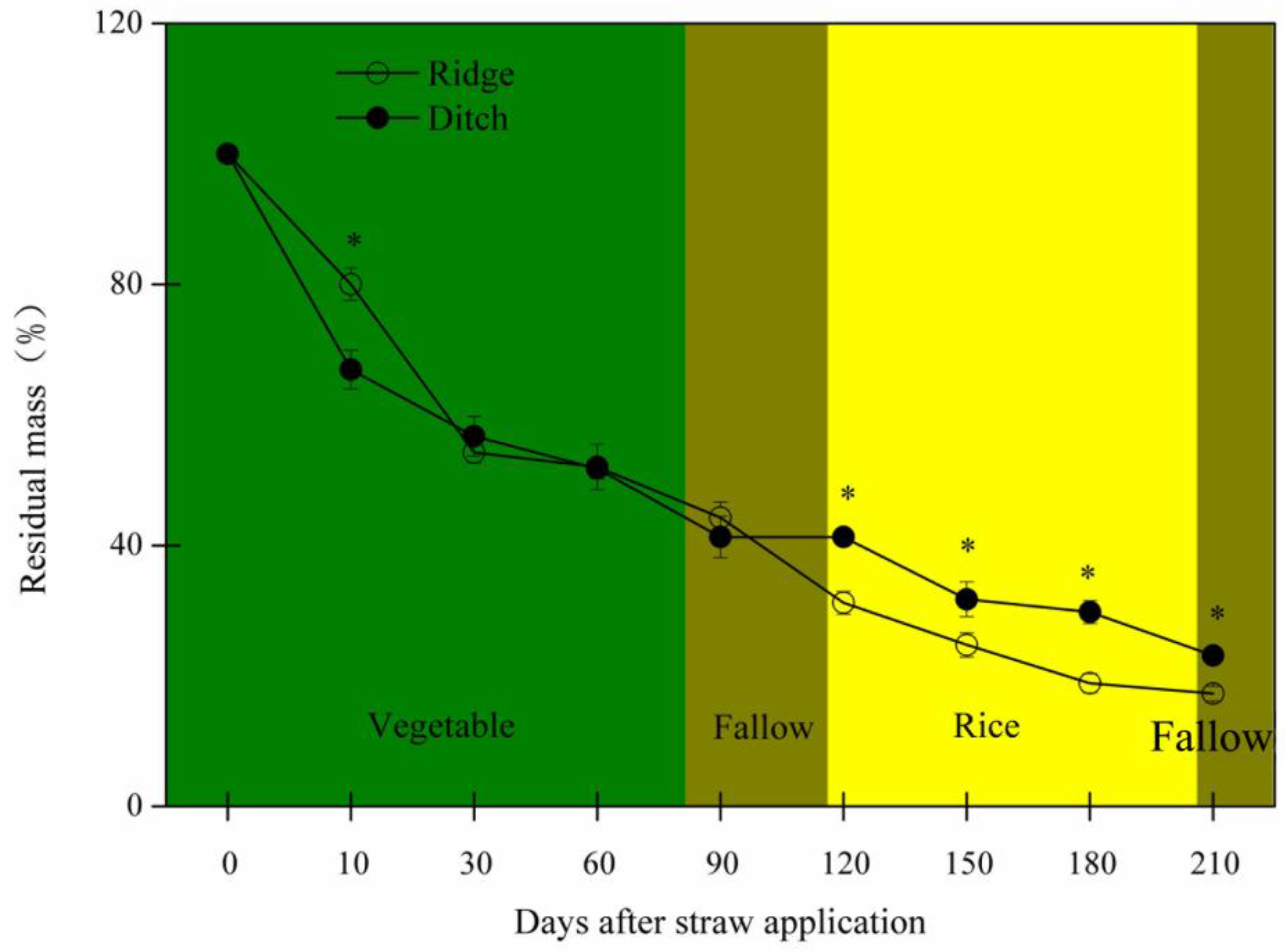
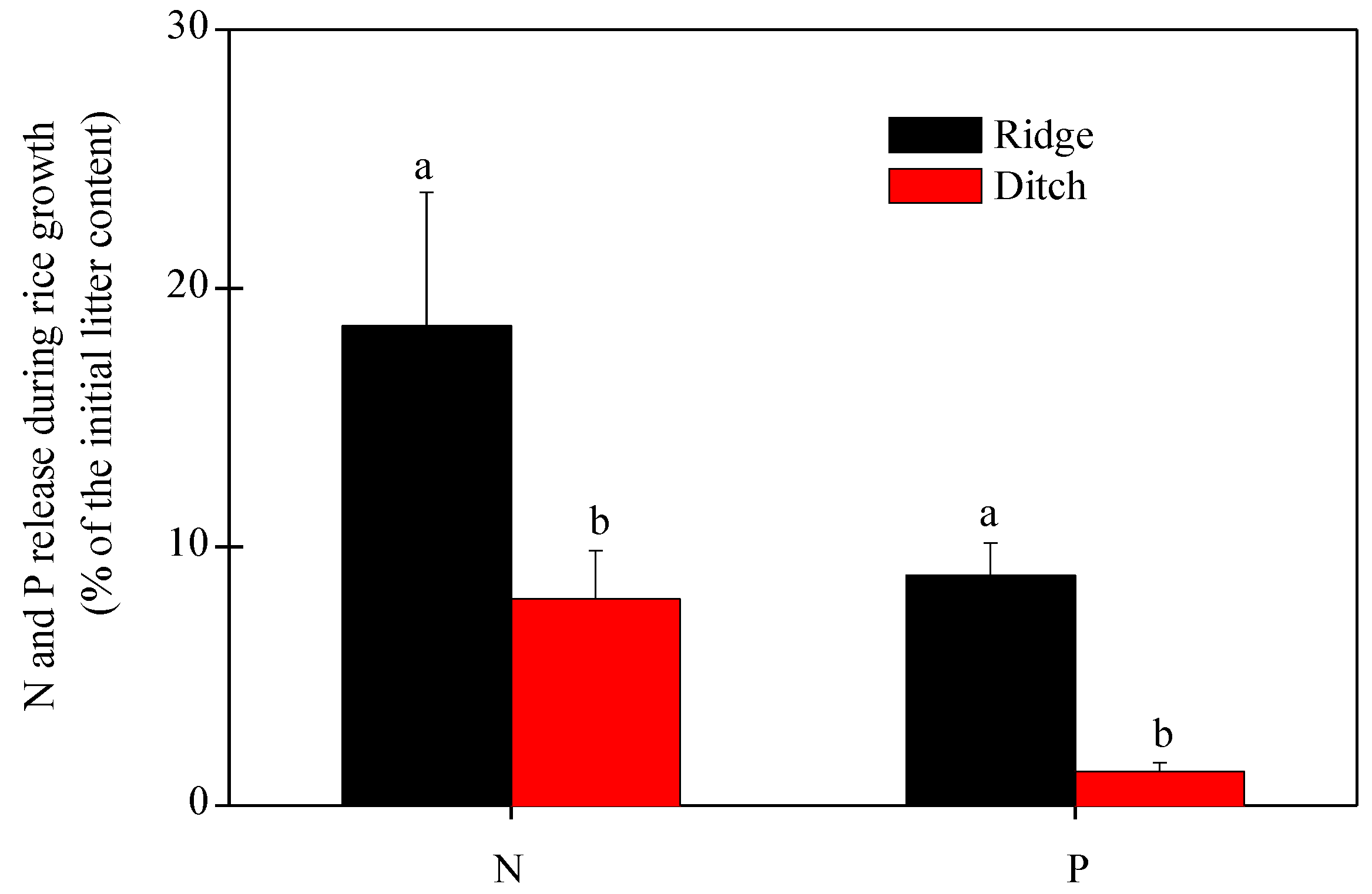
2.2. Residual Straw Mass and Nutrients, Nutrient Release, and Influencing Factors
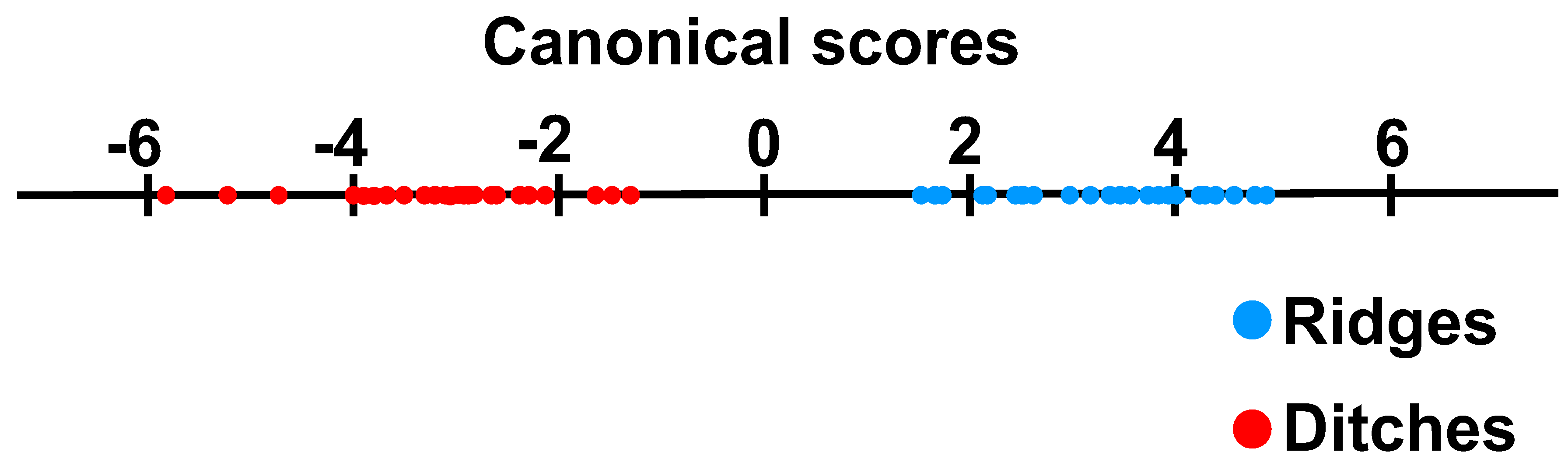
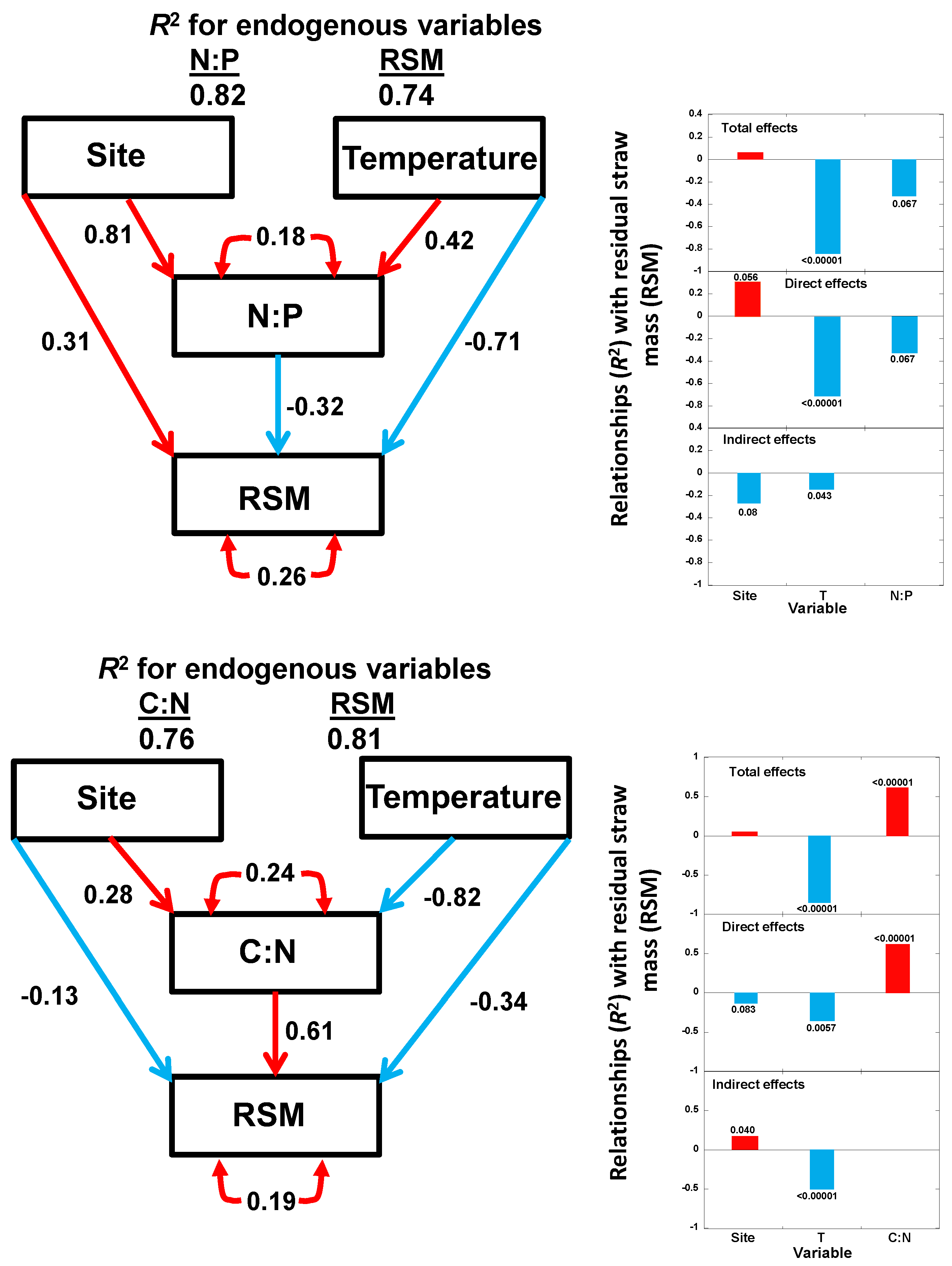
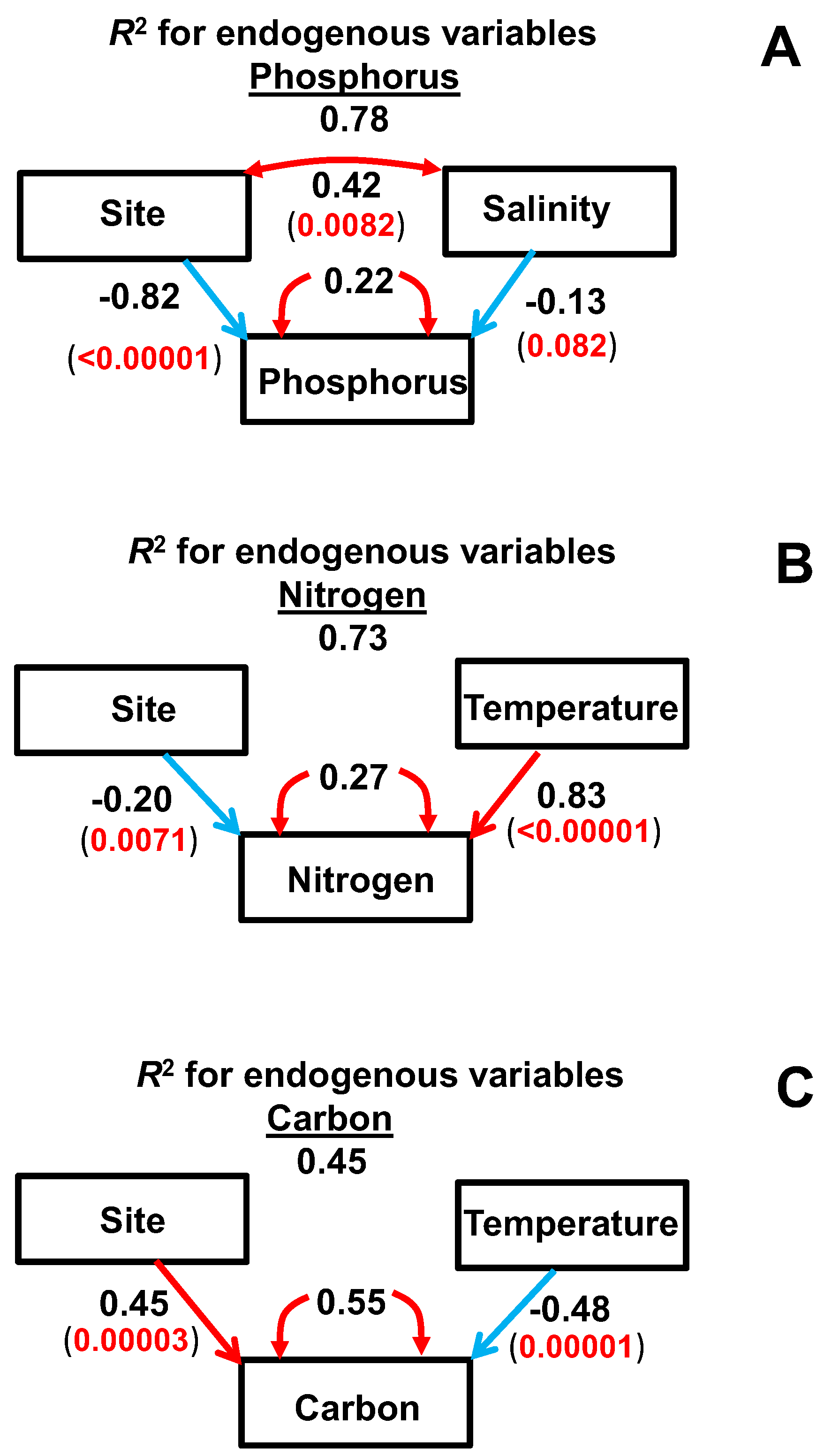
2.3. Multivariate Analyses
2.4. SEM
3. Discussion
3.1. The Impacts of Different Methods of Straw Application on Straw Decomposition
3.2. The Variability of C, N, and P Concentrations and C:N:P Stoichiometry during Straw Decomposition
4. Materials and Methods
4.1. Study Site
4.2. Experimental Design
4.3. Sample Collection and Analysis
4.4. Statistical Analyses
5. Conclusions
- Straw mass decreased faster and C, N, and mainly P concentrations remained higher during the vegetable crop and fallow periods in the ridges than the ditches. C:N, C:P, and N:P ratios of the residual straw were thus lower in the ridges. The straw in the ridges thus had less residual mass and higher N and P concentrations at the beginning of the rice crop period.
- N and P concentrations and contents decreased more in the straw in the ridges than the straw in the ditches during rice growth, so the straw in the ridges provided more N and P than the straw in the ditches.
- Temperature played a key role in the changes of straw C and N concentrations during decomposition, whereas soil salinity had more of an effect on the changes of straw P concentrations.
- The application of straw in the ridges thus allowed a better release of N and P during the rice crop period and produced less residual straw mass than the application of straw in the ditches. Both processes can enhance rice yield in the subtropical rice croplands of China and southeast Asia that use this management system.
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| Variable | Model Type | R2 Model | lme (Variable~Site * Day, Data = Dades, Random = ~1|Day/Plot, Method = “REML”) | ||
|---|---|---|---|---|---|
| Site (Fixed) | Time (Repeat) | Site ^ Time | |||
| Mass remaining (% of initial) | lme | 0.94 | F = 6.86 | F = 59.8 | F = 18.6 |
| p = 0.016 | p = 0.0002 | p = 0.0003 | |||
| Carbon remaining (% of initial) | lme | 0.95 | F = 0.906 | F = 94.8 | F = 16.0 |
| p = 0.35 | p = 0.0001 | p = 0.0006 | |||
| Nitrogen remaining (% of initial) | lme | 0.89 | F = 20.3 | F = 19.3 | F = 27.8 |
| p = 0.0002 | p = 0.0046 | p < 0.0001 | |||
| Phosporus remaining (% of initial) | lme | 0.88 | F = 118 | F = 4.70 | F = 47.9 |
| p < 0.0001 | p = 0.073 | p < 0.0001 | |||
| Carbon concentration in litter | lme | 0.76 | F = 2.43 | F = 5.71 | F = 4.60 |
| p = 0.13 | p = 0.054 | p = 0.043 | |||
| Nitrogen concentration in litter | lme | 0.85 | F = 0.695 | F = 56.4 | F = 2.34 |
| p = 0.41 | p = 0.0003 | p = 0.14 | |||
| Phosphorus concentration in litter | lme | 0.87 | F = 79.7 | F = 2.82 | F = 0.036 |
| p < 0.0001 | p = 0.14 | p = 0.85 | |||
| Litter C:N ratio | lme | 0.88 | F = 7.36 | F = 120 | F = 0.33 |
| p = 0.13 | p < 0.0001 | p = 0.57 | |||
| Litter C:P ratio | lme | 0.85 | F = 87.6 | F = 10.4 | F = 4.06 |
| p < 0.0001 | p = 0.018 | p = 0.056 | |||
| Litter N:P ratio | lme | 0.84 | F = 54.9 | F = 8.06 | F = 0.075 |
| p < 0.0001 | p = 0.030 | p = 0.79 | |||
| Soil pH | lme | 0.83 | F = 1.52 | F = 0.827 | F = 1.27 |
| p = 0.23 | p = 0.40 | p = 0.27 | |||
| Soil salinity | lme | 0.90 | F = 37.7 | F = 0.967 | F = 4.61 |
| p < 0.001 | p = 0.363 | p = 0.043 | |||
| Soil temperature | lme | 0.98 | F = 0.638 | F = 32.6 | F = 0.015 |
| p = 0.43 | p = 0.0012 | p = 0.91 | |||
| Variable | Residual Mass (%) | Soil Temperature (°C) | Soil Salinity (mS cm−1) | Soil pH | ||
|---|---|---|---|---|---|---|
| Residual mass and soil properties | ||||||
| Vegetable crop (both habitats) | 60.29 ± 2.63a | 12.70 ± 0.32a | 0.36 ± 0.05a | 6.33 ± 0.05a | ||
| First fallow period (both habitats) | 42.78 ± 1.90b | 22.07 ± 0.41b | 0.23 ± 0.06ab | 6.00 ± 0.03a | ||
| Rice crop (both habitats) | 29.62 ± 1.77c | 27.81 ± 0.44c | 0.40 ± 0.04ac | 6.22 ± 0.16a | ||
| Second fallow period (both habitats) | 20.22 ± 1.48d | 29.80 ± 0.28d | 0.25 ± 0.02a | 6.17 ± 0.05a | ||
| Crop period | F = 141.319 | F = 2643.159 | F = 61.305 | F = 37.587 | ||
| p < 0.001 | p < 0.001 | p < 0.001 | p < 0.001 | |||
| Ridges | 40.34 ± 4.24 | 21.90 ± 1.57 | 0.27 ± 0.04 | 6.21 ± 0.10 | ||
| Ditches | 42.84 ± 2.97 | 21.46 ± 1.51 | 0.42 ± 0.03 | 6.24 ± 0.07 | ||
| Habitat | F = 3.401 | F = 11.646 | F = 2245.267 | F = 0.159 | ||
| p = 0.139 | p = 0.027 | p < 0.001 | p = 0.711 | |||
| Crop period × habitat | F = 6.809 | F = 48.211 | F = 5.733 | F = 2.418 | ||
| p < 0.001 | p < 0.001 | p < 0.001 | p = 0.045 | |||
| Nutrient concentration | C (mg g−1) | N (mg g−1) | P (mg g−1) | |||
| Vegetable crop (both habitats) | 335.47 ± 3.25a | 10.40 ± 0.21a | 1.65 ± 0.15a | |||
| First fallow period (both habitats) | 331.90 ± 7.58ab | 11.69 ± 0.58a | 1.44 ± 0.16a | |||
| Rice crop (both habitats) | 319.99 ± 5.39b | 15.52 ± 0.62b | 1.68 ± 0.13a | |||
| Second fallow period (both habitats) | 267.66 ± 15.56c | 17.27 ± 0.21c | 2.05 ± 0.22a | |||
| Crop period | F = 18.234 | F = 62.068 | F = 9.660 | |||
| p < 0.001 | p < 0.001 | p < 0.001 | ||||
| Ridges | 306.81 ± 6.55 | 14.06 ± 0.72 | 2.18 ± 0.07 | |||
| Ditches | 334.67 ± 4.18 | 12.61 ± 0.58 | 1.19 ± 0.05 | |||
| Habitat | F = 29.715 | F = 31.669 | F = 623.082 | |||
| p = 0.006 | p = 0.005 | p < 0.001 | ||||
| Crop period × habitat | F = 2.710 | F = 5.286 | F = 3.439 | |||
| p = 0.028 | p = 0.001 | p = 0.009 | ||||
| Nutrient stoichiometry | C:N ratio | C:P ratio | N:P ratio | |||
| Vegetable crop (both habitats) | 32.51 ± 0.77a | 236.57 ± 22.73a | 7.16 ± 0.60a | |||
| First fallow period (both habitats) | 28.70 ± 1.44a | 250.26 ± 35.94a | 8.73 ± 1.18ab | |||
| Rice crop (both habitats) | 21.42 ± 1.20b | 216.54 ± 20.25ab | 9.85 ± 0.51b | |||
| Second fallow period (both habitats) | 15.48 ± 0.83c | 141.53 ± 21.77b | 8.90 ± 0.96ab | |||
| Crop period | F = 44.704 | F = 7.700 | F = 10.184 | |||
| p < 0.001 | p < 0.001 | p < 0.001 | ||||
| Ridges | 23.51 ± 1.50 | 145.18 ± 6.79 | 6.50 ± 0.30 | |||
| Ditches | 27.98 ± 1.40 | 292.60 ± 13.31 | 10.66 ± 0.34 | |||
| Habitat | F = 35.419 | F = 329.071 | F = 127.838 | |||
| p = 0.004 | p < 0.001 | p < 0.001 | ||||
| Crop growth × habitat | F = 0.967 | F = 1.484 | F = 1.369 | |||
| p = 0.474 | p = 0.213 | p = 0.257 | ||||
| Residual nutrients | C (%) | N (%) | P (%) | |||
| Vegetable crop (both habitats) | 54.36 ± 2.21a | 68.46 ± 2.81a | 51.69 ± 5.57a | |||
| First fallow period (both habitats) | 38.13 ± 1.37b | 54.36 ± 1.38b | 31.75 ± 4.11b | |||
| Rice crop (both habitats) | 25.80 ± 1.82c | 48.63 ± 1.59b | 23.82 ± 1.10b | |||
| Second fallow period (both habitats) | 14.86 ± 1.88d | 38.32 ± 2.84c | 20.53 ± 0.82b | |||
| Crop period | F = 178.055 | F = 25.644 | F = 47.704 | |||
| p < 0.001 | p < 0.001 | p < 0.001 | ||||
| Ridges | 34.32 ± 3.87 | 55.55 ± 3.56 | 44.33 ± 4.87 | |||
| Ditches | 39.05 ± 2.94 | 55.44 ± 1.97 | 25.37 ± 1.69 | |||
| Habitat | F = 13.407 | F = 0.002 | F = 94.171 | |||
| p = 0.022 | p = 0.965 | p = 0.001 | ||||
| Crop growth × habitat | F = 4.914 | F = 3.110 | F = 14.328 | |||
| p = 0.001 | p = 0.015 | p < 0.001 | ||||
| Temperature | pH | Salinity | Litter C | Litter N | Litter P | Litter C:N | Litter C:P | Litter N:P | Residual C | Residual N | Residual P | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Residual mass | R = −0.848 | R = 0.199 | R = 0.316 | R = 0.525 | R = −0.876 | R = −0.220 | R = 0.859 | R = 0.284 | R = −0.357 | R = 0.992 | R = 0.925 | R = 0.756 |
| p < 0.001 | p = 0.088 | p = 0.014 | p < 0.001 | p < 0.001 | p = 0.067 | p < 0.001 | p = 0.025 | p = 0.006 | p < 0.001 | p < 0.001 | p < 0.001 | |
| Temperature | R = −0.169 | R = 0.029 | R = −0.491 | R = 0.835 | R = 0.144 | R = −0.827 | R = −0.262 | R = 0.397 | R = −0.857 | R = −0.718 | R = −0.624 | |
| p = 0.125 | p = 0.421 | p < 0.001 | p < 0.001 | p = 0.164 | p < 0.001 | p = 0.036 | p = 0.003 | p < 0.001 | p < 0.001 | p < 0.001 | ||
| pH | R = 0.416 | R = 0.231 | R = −0.314 | R = −0.196 | R = 0.287 | R = 0.241 | R = 0.006 | R = 0.223 | R = 0.147 | R = 0.037 | ||
| p = 0.002 | p = 0.057 | p = 0.015 | p = 0.091 | p = 0.024 | p = 0.049 | p = 0.484 | p = 0.064 | p = 0.159 | p = 0.400 | |||
| Salinity | R = 0.330 | R = −0.251 | R = −0.474 | R = 0.297 | R = 0.500 | R = 0.363 | R = 0.348 | R = 0.311 | R = −0.023 | |||
| p = 0.011 | p = 0.043 | p < 0.001 | p = 0.020 | p < 0.001 | p = 0.006 | p = 0.008 | p = 0.016 | p = 0.439 | ||||
| Litter C | R = −0.628 | R = −0.638 | R = 0.753 | R = 0.708 | R = 0.272 | R = 0.616 | R = 0.472 | R = 0.106 | ||||
| p < 0.001 | p < 0.001 | p < 0.001 | p < 0.001 | p = 0.031 | p < 0.001 | p < 0.001 | p = 0.236 | |||||
| Litter N | R = 0.431 | R = −0.969 | R = −0.508 | R = 0.200 | R = −0.897 | R = −0.688 | R = −0.505 | |||||
| p = 0.001 | p < 0.001 | p < 0.001 | p = 0.086 | p < 0.001 | p < 0.001 | p < 0.001 | ||||||
| Litter P | R = −0.503 | R = −0.952 | R = −0.770 | R = −0.299 | R = −0.123 | R = 0.426 | ||||||
| p < 0.001 | p < 0.001 | p < 0.001 | p = 0.019 | p = 0.203 | p = 0.001 | |||||||
| Litter C:N | R = 0.600 | R = −0.110 | R = 0.901 | R = 0.670 | R = 0.432 | |||||||
| p < 0.001 | p = 0.229 | p < 0.001 | p < 0.001 | p = 0.001 | ||||||||
| Litter C:P | R = 0.712 | R = 0.376 | R = 0.152 | R = −0.358 | ||||||||
| p < 0.001 | p = 0.004 | p = 0.151 | p = 0.006 | |||||||||
| Litter N:P | R = −0.284 | R = −0.334 | R = −0.770 | |||||||||
| p = 0.025 | p = 0.010 | p < 0.001 | ||||||||||
| Residual C | R = 0.918 | R = 0.694 | ||||||||||
| p < 0.001 | p < 0.001 | |||||||||||
| Residual N | R = 0.788 | |||||||||||
| p < 0.001 | ||||||||||||
| Variable | Wilks’ Lambda | p |
|---|---|---|
| Residual C (%) | 0.997 | 0.765 |
| Residual N (%) | 0.969 | 0.298 |
| Residual P (%) | 0.972 | 0.319 |
| C (%) | 0.976 | 0.361 |
| N (%) | 0.990 | 0.550 |
| P (%) | 0.994 | 0.660 |
| C:N | 0.999 | 0.878 |
| C:P | 0.978 | 0.384 |
| N:P | 0.855 | 0.020 |
| pH | 0.802 | 0.006 |
| Salinity | 0.840 | 0.038 |
| Temperature | 0.985 | 0.474 |


References
- Seck, P.A.; Diagne, A.; Mohanty, S.; Wopereis, M.C.S. Crops that feed the world 7: Rice. Food Secur. 2012, 4, 7–24. [Google Scholar] [CrossRef]
- International Rice Research Institute (IRRI). Toward 2000 and Beyond; IRRI: Los Baños, Philippines, 1989. [Google Scholar]
- Frolking, S.; Qiu, J.; Boles, S.; Xiao, X.; Liu, J.; Zhuang, Y.; Li, C.; Qin, X. Combining remote sensing and ground census data to develop new maps of the distribution of rice agriculture in China. Glob. Biogeochem. Cycles 2002, 16, 1091–1101. [Google Scholar] [CrossRef]
- Linquist, B.A.; Adviento-Borbe, M.A.; Pittelkow, C.M.; van Kessel, C.; van Groenigen, K.J. Fertilizer management practices and greenhouse gas emissions from rice systems: A quantitative review and analysis. Field Crops Res. 2012, 135, 10–21. [Google Scholar] [CrossRef]
- Guo, J.H.; Liu, X.J.; Zhang, Y.; Shen, J.L.; Han, W.X.; Zhang, W.F.; Christie, P.; Goulding, K.W.T.; Vitousek, P.M.; Zhang, F.S. Significant acidification in major Chinese croplands. Science 2010, 327, 1008–1010. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wen, M.; Li, X.; Shi, X. Long-term fertilization causes excess supply and loss of phosphorus in purple paddy soil. J. Sci. Food Agric. 2014, 94, 1175–1183. [Google Scholar] [CrossRef] [PubMed]
- Peñuelas, J.; Poulter, B.; Sardans, J.; Ciais, P.; van der Velde, M.; Bopp, L.; Boucher, O.; Godderis, Y.; Llusià, J.; Nardin, E.; et al. Human-induced nitrogen-Phosphorus imbalances alter natural and managed ecosystems across the globe. Nat. Commun. 2013, 4, 2934. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.S.; Pandey, V.C.; Singh, D.P.; Singh, R.P. Influence of pyrite and farmyard manure on population dynamics of soil methanotroph and rice yield in saline rain-fed paddy field. Agric. Ecosyst. Environ. 2010, 139, 74–79. [Google Scholar] [CrossRef]
- Singh, B.; Humphreys, E.; Eberbach, P.L.; Katupitiya, A.; Singh, T.; Kukal, S.S. Growth, yield and water productivity of zero till wheat as affected by rice straw mulch and irrigation schedule. Field Crops Res. 2011, 121, 209–225. [Google Scholar] [CrossRef]
- Wang, J.Y.; Jia, J.X.; Xiong, Z.Q.; Khalil, M.A.K.; Xing, G.X. Water regime–nitrogen fertilizer–straw incorporation interaction: Field study on nitrous oxide emissions from a rice agroecosystem in Nanjing, China. Agric. Ecosyst. Environ. 2011, 141, 437–446. [Google Scholar] [CrossRef]
- Sonnleitner, R.; Lorbeer, E.; Schinner, F. Effect of straw, vegetable oil and whey on physical and microbiological properties of a chernozem. Appl. Soil Ecol. 2003, 22, 195–204. [Google Scholar] [CrossRef]
- Bhattacharyya, P.; Roy, K.S.; Neogi, S.; Adhya, T.K.; Rao, K.S.; Manna, M.C. Effects of rice straw and nitrogen fertilization on greenhouse gas emissions and carbon storage in tropical flooded soil planted with rice. Soil Tillage Res. 2012, 124, 119–130. [Google Scholar] [CrossRef]
- Zhang, Z.S.; Song, X.L.; Lu, X.G.; Xue, Z.S. Ecological stoichiometry of carbon, nitrogen, and phosphorus in estuarine wetland soils: Influences of vegetation coverage, plant communities, geomorphology, and seawalls. J. Soils Sediment. 2013, 13, 1043–1051. [Google Scholar] [CrossRef]
- Pan, T. The Effect of Straw Application on Soil Carbon Pool, Methane and Nitrous Oxide Emissions in the Paddy Fields of Fuzhou. Ph.D. Thesis, Fujian Normal University, Fuzhou, China, 2014. [Google Scholar]
- Sommer, R.; Ryan, J.; Masri, S.; Singh, M.M.; Diekmann, J. Effect of shallow tillage, moldboard plowing, straw management and compost addition on soil organic matter and nitrogen in a dryland barley/wheat-vetch rotation. Soil Tillage Res. 2011, 115, 39–46. [Google Scholar] [CrossRef]
- Yao, S.; Teng, X.; Zhang, B. Effects of rice straw incorporation and tillage depth on soil puddlability and mechanical properties during rice growth period. Soil Tillage Res. 2015, 146, 125–132. [Google Scholar] [CrossRef]
- Qiu, S.J.; Peng, P.Q.; Li, L.; He, P.; Liu, Q.; Wu, J.S.; Christie, P.; Ju, X.T. Effects of applied urea and straw on various nitrogen fractions in two Chinese paddy soils with differing clay mineralogy. Biol. Fertil. Soils 2012, 48, 161–172. [Google Scholar] [CrossRef]
- Zhang, B.; Pang, C.; Qin, J.; Liu, K.; Li, H. Rice straw incorporation in winter with fertilizer-N application improves soil fertility and reduces global warming potential from a double rice paddy field. Biol. Fertil. Soils 2013, 49, 1039–1052. [Google Scholar] [CrossRef]
- Amlin, N.A.; Rood, S.B. Inundation tolerances of riparian willows and cottonwoods. J. Am. Water Resour. Assoc. 2001, 37, 1709–1720. [Google Scholar] [CrossRef]
- Adame, M.F.; Virdi, B.; Lovelock, C.E. Effect of geomorphological setting and rainfall on nutrient exchange in mangroves during tidal inundation. Mar. Freshw. Res. 2010, 61, 1197–1206. [Google Scholar] [CrossRef]
- Kobayashi, T.; Ryder, D.S.; Gordon, G.; Shannon, I.; Ingleton, T.; Carpenter, M.; Jacobs, S.J. Short-term response of nutrients, carbon and planktonic microbial communities to floodplain wetland inundation. Aquat. Ecol. 2009, 43, 843–858. [Google Scholar] [CrossRef]
- Sheng, L.X.; Huang, D.Y.; Xia, H.A.; Xiao, S.Y. The effect of applying economic green manure crops in the paddy field with succession cropping system. Chin. J. Eco-Agric. 2004, 12, 109–111. [Google Scholar]
- Valiela, I.; Teal, J.M.; Allen, S.D.; Van Etten, R.; Goehringer, D.; Volkmann, S. Decomposition in salt marsh ecosystems: The phases and major factors affecting disappearance of above-ground organic matter. J. Exp. Mar. Biol. Ecol. 1985, 89, 29–54. [Google Scholar] [CrossRef]
- Talbot, J.M.; Treseder, K.K. Interactions among lignin, cellulose, and nitrogen drive litter chemistry-decay relationships. Ecology 2012, 93, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Güsewell, S.; Verhoeven, J.T.A. Litter N:P ration indicate whether N or P limits the decomposability of graminoid leaf litter. Plant Soil 2006, 287, 131–143. [Google Scholar] [CrossRef]
- Manzoni, S.; Trofymow, J.A.; Jackson, R.B.; Porporato, A. Stoichiometric controls on carbon, nitrogen, and phosphorus dynamics in decomposing litter. Ecol. Monogr. 2010, 80, 89–106. [Google Scholar] [CrossRef]
- Cleveland, C.C.; Liptzin, D. C:N:P stoichiometry in soil: Is there a “Redfield ratio” for the microbial biomass? Biogeochemistry 2007, 85, 235–252. [Google Scholar] [CrossRef]
- Wang, W.; Lai, D.Y.F.; Li, S.; Kim, P.J.; Zeng, C.; Li, P.; Liang, Y. Steel slag amendment reduces methane emission and increases rice productivity in subtropical paddy fields in China. Wetl. Ecol. Manag. 2014, 22, 683–691. [Google Scholar] [CrossRef]
- Wang, W.; Sardans, J.; Zeng, C.; Zhong, C.; Li, Y.; Peñuelas, J. Responses of soil nutrient concentrations and stoichiometry to different human land uses in a subtropical tidal wetland. Geoderma 2014, 232, 459–470. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Thornton, P.E.; Post, W.M. A global analysis of soil microbial biomass carbon, nitrogen, and phosphorus in terrestrial ecosystems. Glob. Ecol. Biogeogr. 2013, 22, 737–749. [Google Scholar] [CrossRef]
- Tong, C.; Liu, B.G. Litter decomposition and nutrient dynamics in different tidal water submergence environments of estuarine tidal wetland. Geogr. Res. 2009, 28, 118–128. [Google Scholar]
- Richardson, A.E.; Simpson, R.J. Soil microorganisms mediating phosphorus availability update on microbial phosphorus. Plant Physiol. 2011, 156, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Sterner, R.W.; Elser, J.J. Ecological Stoichimetry: The Biology of Elements from Molecules to Biosphere; Princeton University Press: Princeton, NJ, USA, 2002. [Google Scholar]
- Sieger, S.M.; Kristensen, B.K.; Robson, C.A.; Amirsadeghi, S.; Eng, E.W.Y.; Abdel-Mesih, A.; Møller, L.M.; Vanlerberghe, G.C. The role of alternative oxidase in modulating carbon use efficiency and growth during macronutrient stress in tobacco cells. J. Exp. Bot. 2005, 56, 1499–1515. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.Y.; Zhang, Z.C.; Li, X. Influences of salinity on mass and energy dynamics during decomposition of Kandelia candel leaf litter. Chin. J. Plant Ecol. 2010, 34, 1377–1385. [Google Scholar]
- Mu, Z.J.; Huang, A.Y.; Ni, J.P.; Xie, D.T. Linking anual N2O emission in organic soils to mineral nitrogen inputs as estimated by heterotrophic respiration and soil C/N ratio. PLoS ONE 2014, 9, e96572. [Google Scholar] [CrossRef] [PubMed]
- Moore, T.R.; Trofymow, J.A.; Prescott, C.E.; Titus, D.B. Nature and nurture in the dynamics of C, N and P during litter decomposition in Canadian forest. Plant Soil 2011, 339, 163–175. [Google Scholar] [CrossRef]
- Peng, S.; Luo, Y.; Xu, J.; Khan, S.; Jiao, X.; Wang, W. Integrated irrigation and drainage practices to enhance water productivity and reduce pollution in a rice production system. Irrig. Drain. 2012, 61, 285–293. [Google Scholar] [CrossRef]
- Ocio, J.A.; Brookes, P.C.; Jenkinson, D.S. Field incorporation of straw and its effects on soil microbial biomass and soil inorganic N. Soil Biol. Biochem. 1991, 23, 171–176. [Google Scholar] [CrossRef]
- Lu, R.K. Analysis Methods of Soil Science and Agricultural Chemistry; Agricultural Science and Technology Press: Beijing, China, 1999. [Google Scholar]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D.; Core, T.R. Nlme: Linear and Nonlinear Mixed Effects Models. R Package Version 3.1-126. Available online: http://CRAN (accessed on 12 June 2016).
- Barton, K. MuMIn: Multi-Model Inference. Available online: https://cran.r-project.org/package=MuMIn (accessed on 12 June 2017).
- De Maesschalck, R.; Jouan-Rimbaud, D.; Massart, D.L. The Mahalanobis distance. Chemom. Intell. Lab. 2000, 50, 1–18. [Google Scholar] [CrossRef]
- Epskamp, S. semPlot: Unified Visualizations of Structural Equation Models. Struct. Equ. Model. A Multidiscip. J. 2000, 22, 474–483. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, W.; Sardans, J.; Wang, C.; Pan, T.; Zeng, C.; Lai, D.Y.F.; Bartrons, M.; Peñuelas, J. Straw Application Strategy to Optimize Nutrient Release in a Southeastern China Rice Cropland. Agronomy 2017, 7, 84. https://doi.org/10.3390/agronomy7040084
Wang W, Sardans J, Wang C, Pan T, Zeng C, Lai DYF, Bartrons M, Peñuelas J. Straw Application Strategy to Optimize Nutrient Release in a Southeastern China Rice Cropland. Agronomy. 2017; 7(4):84. https://doi.org/10.3390/agronomy7040084
Chicago/Turabian StyleWang, Weiqi, Jordi Sardans, Chun Wang, Ting Pan, Congsheng Zeng, Derrick Y. F. Lai, Mireia Bartrons, and Josep Peñuelas. 2017. "Straw Application Strategy to Optimize Nutrient Release in a Southeastern China Rice Cropland" Agronomy 7, no. 4: 84. https://doi.org/10.3390/agronomy7040084
APA StyleWang, W., Sardans, J., Wang, C., Pan, T., Zeng, C., Lai, D. Y. F., Bartrons, M., & Peñuelas, J. (2017). Straw Application Strategy to Optimize Nutrient Release in a Southeastern China Rice Cropland. Agronomy, 7(4), 84. https://doi.org/10.3390/agronomy7040084







