Spore Density of Arbuscular Mycorrhizal Fungi is Fostered by Six Years of a No-Till System and is Correlated with Environmental Parameters in a Silty Loam Soil
Abstract
:1. Introduction
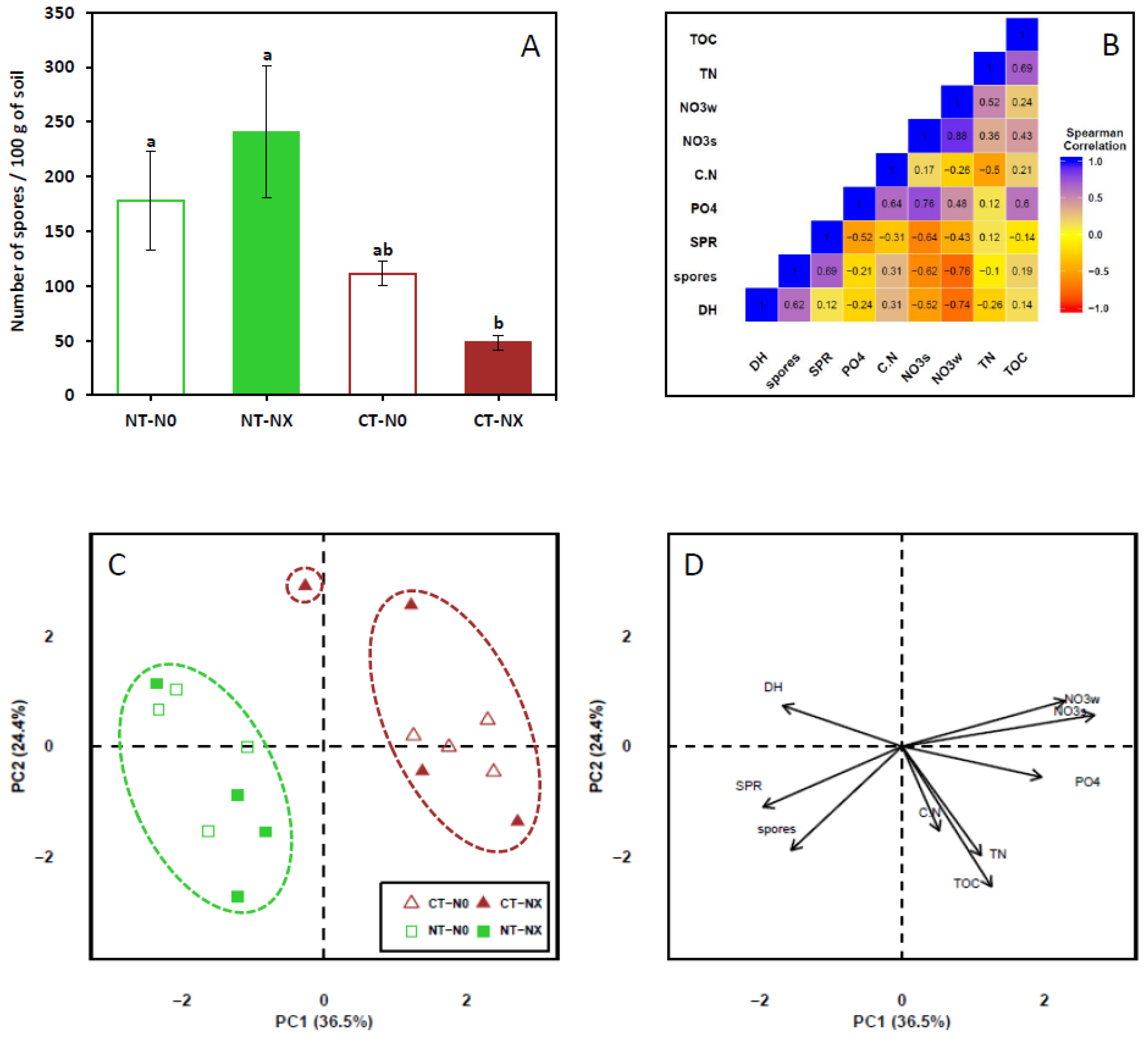
2. Results and Discussion
3. Materials and Methods
3.1. Site Description and Experimental Design
3.2. Sample Collection and Analyzes
3.3. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Selosse, M.A.; Strullu-Derrien, C.; Martin, F.M.; Kamoun, S.; Kenrick, P. Plants, fungi and oomycetes: A 400-million year affair that shapes the biosphere. New Phytol. 2015, 206, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Jansa, J.; Wiemken, A.; Frossard, E. The effects of agricultural practices on arbuscular mycorrhizal fungi. Geol. Soc. 2006, 266, 89–115. [Google Scholar] [CrossRef]
- Brundrett, M.C. Mycorrhizal associations and other means of nutrition of vascular plants: Understanding the global diversity of host plants by resolving conflicting information and developing reliable means of diagnosis. Plant Soil 2009, 320, 37–77. [Google Scholar] [CrossRef]
- Six, J.; Elliott, E.T.; Paustian, K. Soil macroaggregate turnover and microaggregate formation: A mechanism for C sequestration under no-tillage agriculture. Soil Biol. Biochem. 2000, 32, 2099–2103. [Google Scholar] [CrossRef]
- Corkidi, L.; Rowland, D.L.; Johnson, N.C.; Allen, E.B. Nitrogen fertilization alters the functioning of arbuscular mycorrhizas at two semiarid grasslands. Plant Soil 2002, 240, 299–310. [Google Scholar] [CrossRef]
- Brito, I.; Goss, M.J.; de Carvalho, M. Effect of tillage and crop on arbuscular mycorrhiza colonization of winter wheat and triticale under Mediterranean conditions. Soil Use Manag. 2012, 28, 202–208. [Google Scholar] [CrossRef]
- Verzeaux, J.; Roger, D.; Lacoux, J.; Nivelle, E.; Adam, C.; Habbib, H.; Hirel, B.; Dubois, F.; Tetu, T. In winter wheat, no-till increases mycorrhizal colonization thus reducing the need for nitrogen fertilization. Agronomy 2016, 6, 38. [Google Scholar] [CrossRef]
- Schalamuk, S.; Cabello, M. Arbuscular mycorrhizal fungal propagules from tillage and no-tillage systems: Possible effects on Glomeromycota diversity. Mycologia 2010, 102, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Castillo, C.G.; Rubio, R.; Rouanet, J.L.; Borie, F. Early effects of tillage and crop rotation on arbuscular mycorrhizal fungal propagules in an Ultisol. Biol. Fertil. Soils 2006, 43, 83–92. [Google Scholar] [CrossRef]
- Oruru, M.B.; Njeru, E.M. Upscaling Arbuscular Mycorrhizal Symbiosis and Related Agroecosystems Services in Smallholder Farming Systems. BioMed Res. Int. 2016, 2016, 4376240. [Google Scholar] [CrossRef] [PubMed]
- Jansa, J.; Mozafar, A.; Kuhn, G.; Anken, T.; Ruh, R.; Sanders, I.R.; Frossard, E. Soil tillage affects the community structure of mycorrhizal fungi in maize roots. Ecol. Appl. 2003, 13, 1164–1176. [Google Scholar] [CrossRef]
- Berruti, A.; Lumini, E.; Balestrini, R.; Bianciotto, V. Arbuscular mycorrhizal fungi as natural biofertilizers: Let’s benefit from past successes. Front. Microbiol. 2016, 6, 1559. [Google Scholar] [CrossRef] [PubMed]
- Saia, S.; Rappa, V.; Ruisi, P.; Abenavoli, M.R.; Sunseri, F.; Giambalvo, D.; Frenda, A.S.; Martinelli, F. Soil inoculation with symbiotic microorganisms promotes plant growth and nutrient transporter genes expression in durum wheat. Front. Plant. Sci. 2015, 6, 815. [Google Scholar] [CrossRef] [PubMed]
- Janoušková, M.; Krak, K.; Wagg, C.; Štorchová, H.; Caklová, P.; Vosátka, M. Effects of inoculum additions in the presence of a preestablished arbuscular mycorrhizal fungal community. Appl. Environ. Microbiol. 2013, 79, 6507–6515. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Qiao, N.; Xu, X.; Xin, X.; Han, J.Y.; Tian, Y.; Ouyang, H.; Kuzyakov, Y. Nitrogen acquisition by plants and microorganisms in a temperate grassland. Sci. Rep. 2016, 6, 22642. [Google Scholar] [CrossRef] [PubMed]
- Zuber, S.M.; Villamil, M.B. Meta-analysis approach to assess effect of tillage on microbial biomass and enzyme activities. Soil Biol. Biochem. 2016, 97, 176–187. [Google Scholar] [CrossRef]
- Van den Bossche, A.; de Bolle, S.; de Neve, S.; Hofman, G. Effect of tillage intensity on N mineralization of different crop residues in a temperate climate. Soil Tillage Res. 2009, 103, 316–324. [Google Scholar] [CrossRef]
- Pandey, C.B.; Chaudhari, S.K.; Dagar, J.C.; Singh, G.B.; Singh, R.K. Soil N mineralization and microbial biomass carbon affected by different tillage levels in a hot humid tropic. Soil Tillage Res. 2010, 110, 33–41. [Google Scholar] [CrossRef]
- Watts, D.B.; Torbert, H.A.; Feng, Y.; Prior, S.A. Soil microbial community dynamics as influenced by composted dairy manure, soil properties, and landscape position. Soil Sci. 2010, 175, 474–486. [Google Scholar] [CrossRef]
- Nivelle, E.; Verzeaux, J.; Habbib, H.; Kuzyakov, Y.; Decocq, G.; Roger, D.; Lacoux, J.; Duclercq, J.; Spicher, F.; Nava-Saucedo, J.E.; et al. Functional response of soil microbial communities to tillage, cover crops and nitrogen fertilization. Appl. Soil Ecol. 2016, 108, 147–155. [Google Scholar] [CrossRef]
- Das, A.; Lal, R.; Patel, D.P.; Idapuganti, R.G.; Layek, J.; Ngachan, S.V.; Ghosh, P.K.; Bordoloi, J.; Kumar, M. Effects of tillage and biomass on soil quality and productivity of lowland rice cultivation by small scale farmers in North Eastern India. Soil Tillage Res. 2014, 143, 50–58. [Google Scholar] [CrossRef]
- Cid, P.; Carmona, I.; Murillo, J.M. No-tillage permanent bed planting and controlled traffic in a maize-cotton irrigated system under Mediterranean conditions: Effects on soil compaction, crop performance and carbon sequestration. Eur. J. Agron. 2014, 61, 24–34. [Google Scholar] [CrossRef]
- Lipiec, J.; Hatano, R. Quantification of compaction effects on soil physical properties and crop growth. Geoderma 2003, 116, 107–136. [Google Scholar] [CrossRef]
- Bertolino, A.V.F.A.; Fernandes, N.F.; Miranda, J.P.L.; Souza, A.P.; Lopes, M.R.S.; Palmieri, F. Effects of plough pan development on surface hydrology and on soil physical properties in Southeastern Brazilian plateau. J. Hydrol. 2010, 393, 94–104. [Google Scholar] [CrossRef]
- Nawaz, M.F.; Bourrié, G.; Trolard, F. Soil compaction impact and modelling. A review. Agron. Sustain. Dev. 2013, 33, 291–309. [Google Scholar] [CrossRef]
- Kabir, Z. Tillage or no-tillage: Impact on mycorrhizae. Can. J. Plant Sci. 2005, 85, 23–29. [Google Scholar] [CrossRef]
- Hu, J.; Yang, A.; Wang, J.; Zhu, A.; Dai, J.; Wong, M.H.; Lin, X. Arbuscular mycorrhizal fungal species composition, propagule density, and soil alkaline phosphatase activity in response to continuous and alternate no-tillage in Northern China. Catena 2015, 133, 215–220. [Google Scholar] [CrossRef]
- Curaqueo, G.; Barea, J.M.; Acevedo, E.; Rubio, R.; Cornejo, P.; Borie, F. Effects of different tillage system on arbuscular mycorrhizal fungal propagules and physical properties in a Mediterranean agroecosystem in central Chile. Soil Tillage Res. 2011, 113, 11–18. [Google Scholar] [CrossRef]
- Douds, D.D.; Millner, P. Biodiversity of arbuscular mycorrhizal fungi in agroecosystems. Agric. Ecosyst. Environ. 1999, 74, 77–93. [Google Scholar] [CrossRef]
- Kahiluoto, H.; Ketoja, E.; Vestberg, M.; Saarela, I. Promotion of AM utilization through reduced P fertilization 2. Field studies. Plant Soil 2001, 231, 65–79. [Google Scholar] [CrossRef]
- De Varennes, A.; Goss, M.J. The tripartite symbiosis between legumes, rhizobia and indigenous mycorrhizal fungi is more efficient in undisturbed soil. Soil Biol. Biochem. 2007, 39, 2603–2607. [Google Scholar]
- Mortimer, P.E.; Pérez-Fernandez, M.A.; Valentine, A.J. The role of arbuscular mycorrhizal colonization in the carbon and nutrient economy of the tripartite symbiosis with nodulated Phaseolus vulgaris. Soil Biol. Biochem. 2008, 40, 1019–1027. [Google Scholar] [CrossRef]
- Cornejo, P.; Borie, F.; Rubio, R.; Azcon, R. Influence of nitrogen source on the viability, functionality and persistence of Glomus etunicatum fungal propagules in an Andisol. Appl. Soil Ecol. 2006, 35, 423–431. [Google Scholar] [CrossRef]
- Bhadalung, N.N.; Suwanarit, A.; Dell, B.; Nopamornbodi, O.; Thamchaipenet, A.; Rungchuang, J. Effects of long-term NP-fertilization on abundance and diversity of arbuscular mycorrhizal fungi under a maize cropping system. Plant Soil 2005, 270, 371–382. [Google Scholar] [CrossRef]
- Egerton-Warburton, L.M.; Allen, E.B. Shifts in arbuscular mycorrhizal communities along an anthropogenic nitrogen deposition gradient. Ecol. Appl. 2000, 10, 484–496. [Google Scholar] [CrossRef]
- Mbuthia, L.W.; Acosta-Martínez, V.; De Bruyn, J.; Schaeffer, S.; Tyler, D.; Odoi, E.; Mpheshea, M.; Walker, F.; Eash, N. Long term tillage, cover crop, and fertilization effects on microbial community structure, activity: Implications for soil quality. Soil Biol. Biochem. 2015, 89, 24–34. [Google Scholar] [CrossRef]
- Egerton-Warburton, L.M.; Johnson, N.C.; Allen, E.B. Mycorrhizal community dynamics following nitrogen fertilization: A cross-site test in five grasslands. Ecol. Monogr. 2007, 77, 527–544. [Google Scholar] [CrossRef]
- Machet, J.M.; Dubrulle, P.; Louis, P. Azobil1: A computer program for fertiliser N recommendations based on a predictive balance sheet method. In Proceedings of the 1st Congress of the European Society of Agronomy, Paris, France, 5–7 December 1990. [Google Scholar]
- Olsen, S.R.; Cole, C.V.; Watanabe, F.S.; Dean, L.A. Estimation of Available Phosphorus in Soils by Extraction with Sodium Bicarbonate; U.S. Department of Agriculture: Washington, DC, USA, 1954.
- Casida, L.E.; Klein, D.A.; Santoro, T. Soil dehydrogenase activity. Soil Sci. 1964, 98, 371–376. [Google Scholar] [CrossRef]
- McKenney, M.C.; Lindsey, D.L. Improved method for quantifying endomycorrhizal fungi spores from soil. Mycologia 1987, 79, 779–782. [Google Scholar] [CrossRef]
- The R Project for Statistical Computing. Available online: http://www.R-project.org (accessed on 23 January 2017).
- Pohlert, T. PMCMR: Calculate Pairwise Multiple Comparisons of Mean Rank Sums Version 4.1. 2016. Available online: http://cran.r-project.org/ (accessed on 23 January 2017).
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; Wagner, H. Vegan: Community Ecology Package Version 2.3-2. 2015. Available online: http://cran.r-project.org/ (accessed on 23 January 2017).
| Soil Parameter | H (p) | NT-N0 | NT-NX | CT-N0 | CT-NX | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| DH (µg TPF g−1 24 h−1) | 7.35 (0.042) | 12.58 | ± | 1.22 a | 8.35 | ± | 1.6 b | 8.22 | ± | 0.36 b | 7.63 | ± | 0.55 b |
| NO3−s (mg kg−1) | 12.93 (0.005) | 2.91 | ± | 0.06 b | 3.35 | ± | 0.18 b | 11.25 | ± | 1.12 a | 8.49 | ± | 1.21 a |
| NO3−w (mg L−1) | 13.26 (0.004) | 18.13 | ± | 2.04 b | 27.62 | ± | 3.67 b | 44.67 | ± | 2.65 a | 61.76 | ± | 3.81 a |
| PO43− (mg kg−1) | 8.28 (0.040) | 38.08 | ± | 2.21 b | 41.58 | ± | 2.11 ab | 51.37 | ± | 2.38 a | 42.55 | ± | 3.45 ab |
| TN (g kg−1) | NS | 1.18 | ± | 0.04 | 1.21 | ± | 0.06 | 1.18 | ± | 0.02 | 1.25 | ± | 0.07 |
| TOC (g kg−1) | NS | 11.54 | ± | 0.48 | 11.75 | ± | 0.51 | 12.04 | ± | 0.19 | 11.59 | ± | 0.89 |
| C:N ratio | 7.57 (0.048) | 9.74 | ± | 0.12 b | 9.74 | ± | 0.16 b | 10.17 | ± | 0.08 a | 9.29 | ± | 0.31 b |
| SPR (MPa) | 12.11 (0.007) | 0.88 | ± | 0.06 b | 1.265 | ± | 0.08 a | 0.57 | ± | 0.08 c | 0.83 | ± | 0.04 bc |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verzeaux, J.; Nivelle, E.; Roger, D.; Hirel, B.; Dubois, F.; Tetu, T. Spore Density of Arbuscular Mycorrhizal Fungi is Fostered by Six Years of a No-Till System and is Correlated with Environmental Parameters in a Silty Loam Soil. Agronomy 2017, 7, 38. https://doi.org/10.3390/agronomy7020038
Verzeaux J, Nivelle E, Roger D, Hirel B, Dubois F, Tetu T. Spore Density of Arbuscular Mycorrhizal Fungi is Fostered by Six Years of a No-Till System and is Correlated with Environmental Parameters in a Silty Loam Soil. Agronomy. 2017; 7(2):38. https://doi.org/10.3390/agronomy7020038
Chicago/Turabian StyleVerzeaux, Julien, Elodie Nivelle, David Roger, Bertrand Hirel, Frédéric Dubois, and Thierry Tetu. 2017. "Spore Density of Arbuscular Mycorrhizal Fungi is Fostered by Six Years of a No-Till System and is Correlated with Environmental Parameters in a Silty Loam Soil" Agronomy 7, no. 2: 38. https://doi.org/10.3390/agronomy7020038
APA StyleVerzeaux, J., Nivelle, E., Roger, D., Hirel, B., Dubois, F., & Tetu, T. (2017). Spore Density of Arbuscular Mycorrhizal Fungi is Fostered by Six Years of a No-Till System and is Correlated with Environmental Parameters in a Silty Loam Soil. Agronomy, 7(2), 38. https://doi.org/10.3390/agronomy7020038







